Transfusion of Blood Components in Paediatric Age Groups
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing strategies for the transfusion of blood components in paediatric age groups into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:algorithms, pathways, tables, and parents’ educational health guide (in Arabic).
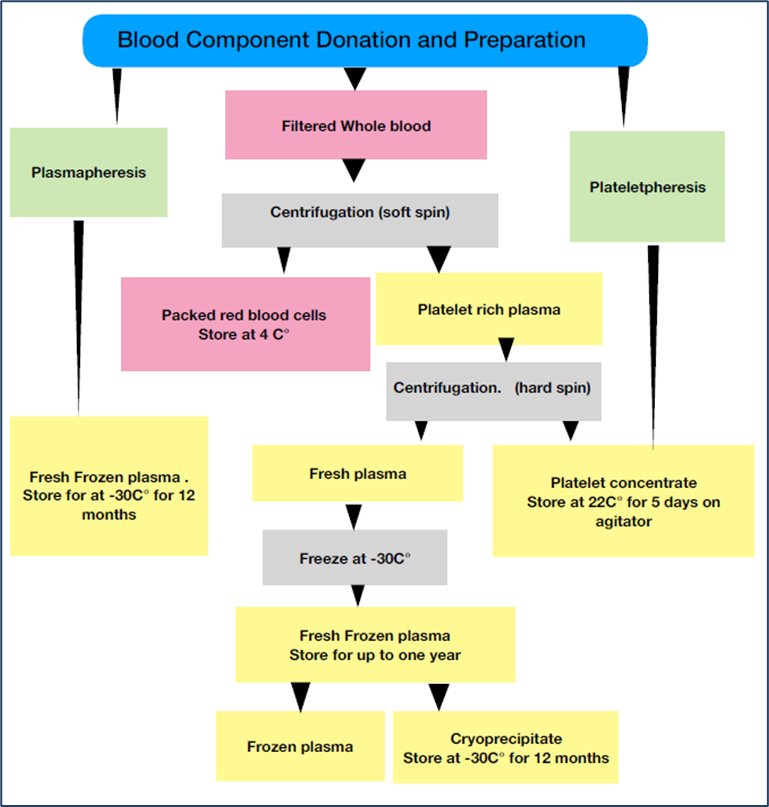
Figure S1: The process by which whole blood is used to produce blood components and plasma derivatives.
Table S1: General transfusion practices


|
|
Cut-off of platelet count |
|
||||
|
|
|
|
|
|
||
|
Less than 20 x103/µl |
|
20 x103 to < 30 x103/µl |
|
30 x103 to 50 x103/µl |
||
|
|
|
|
|
|
||
|
Mandatory transfusion
|
|
Transfusion as clinical situation require (e.g., active bleeding, minor invasive procedure) |
|
Conditional transfusion in the following: - Intracranial hemorrhage - Coagulopathy - Low birth weight < 1000 g - Neonatal sepsis - FNAIT* - Major invasive procedure - Unstable blood pressure |
||
*Washed irradiated platelets resuspended in ABO compatible plasma of maternal origin are used better. The transfused platelet should be lacking the causative antigen.
Figure S2: Indication for platelet transfusion in neonates.
Table S2: Indications of Prophylactic Platelet Transfusion in Pediatrics
|
Indication |
Platelet threshold (x103/µl) |
|
Prior To Procedures or Surgery* |
|
|
· Major surgery or invasive procedure, no active bleeding |
< 50 |
|
· Venous central lines (both tunneled and un-tunneled) ** |
<20 |
|
· Lumbar puncture |
< 40 |
|
· Insertion/removal of epidural catheter |
< 80 |
|
· Major surgery |
<50 |
|
· Neurosurgery or ophthalmic surgery involving the posterior segment of the eye |
<100 |
|
· Percutaneous liver biopsy*** |
<50 |
|
In critically ill child and reversible bone marrow failure |
<10 |
|
In critically ill child and reversible bone marrow failure with additional risk factors for bleeding**** |
<20 |
|
In Stable, non-bleeding child |
<10 |
* Whenever possible use a procedure/equipment associated with the lowest bleeding risk. Apply local measures, such as compression, to reduce the risk of bleeding post-procedure.
** Inserted by experienced staff using ultrasound guidance techniques.
***Consider trans-jugular biopsy if the platelet count is below this level.
****e.g., body temperature > 38°C or undergoing invasive procedure.
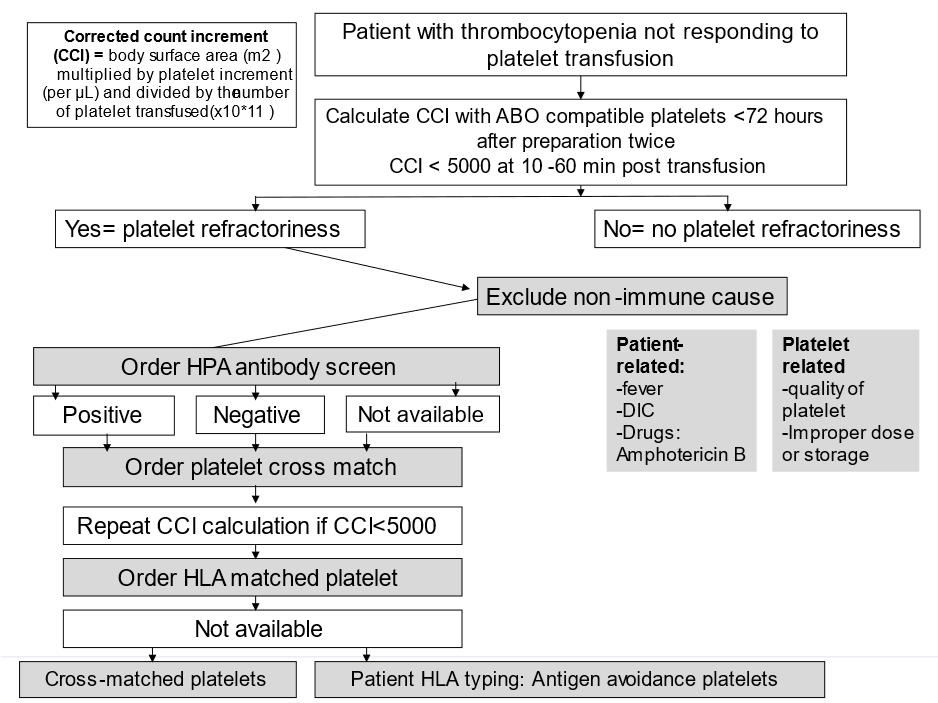
Figure S3: Approach to Platelet Refractoriness
HPA: human platelet antigen, HLA: human leukocyte antigen
Table S3: Indications of fresh frozen plasma transfusion
|
Indications |
Associated conditions |
|
Isolated Coagulation factors deficiency |
When specific therapy (recombinant or viral inactivated product) is not available |
|
Multiple coagulation factor deficiencies |
As DIC, vitamin k deficiency and liver failure If there is active bleeding in addition to treatment of the cause |
|
Anticoagulant |
· Reversal of warfarin (Coumadin)- with active bleeding or require emergency surgery. · Other anticoagulants: before surgery to prevent active bleeding or to treat active bleeding |
|
Microvascular bleeding |
When PT and PTT> 1.5 times normal |
|
Thrombotic thrombo-cytopenic purpura |
Therapeutic plasma exchange |
|
Protein C & protein S deficiency |
Associated with hypercoagulable state and no available concentrates |
Table S4: Indication and dose of cryoprecipitate in various coagulation disorders
|
Indications |
Dose of cryoprecipitate |
|
Fibrinogen Replacement
|
· Cryoprecipitate can be used for fibrinogen replacement in fibrinogen disorders (congenital afibrinogenemia or dysfibrinogenemia) · Dose: 1 unit of cryoprecipitate per 5 kg patient weight will increase fibrinogen by about 100 mg/dL · Number of bags = 0.2 x weight (kg) to provide about 100 mg/dL fibrinogen. |
|
Factor XIII Replacement
|
· 1 unit of cryoprecipitate per 5kg patient weight will provide 10 U/kg of factor XIII. · Number of bags =0.2 x weight (kg). · Factor XIII has a long half-life and can usually be dosed every 3-6 weeks. |
|
Factor VIII Replacement
|
· Dosing depends on patient factor VIII (8) level and requires routine monitoring of factor VIII (8) to determine appropriate dose. · Dosing should be repeated every 8-12 hours but will vary with each patient. · Post-surgery or major trauma replacement may be required for up to 10 days to maintain hemostasis. · Dosing also depends on Plasma Volume (PV) which is a fraction of Total Blood Volume (TBV). · Number of bags = [Desired activity (%) – Current activity (%)] x PV / 80
N.B: PV (mL) = TBV x (1-Hct), TBV (mL) = 70 mL/kg x weight (kg) |
|
Von Willebrand Factor Replacement
|
· Dosing of 1 unit per 10 kg patient weight will usually be enough to control bleeding. · Number of bags = 0.1 x weight (kg) · Repeat dosing may be required every 8-12 hours for up to 3 days followed by once daily dosing. · Follow clinically to adjust dosing and with appropriate lab studies available at your institution. |
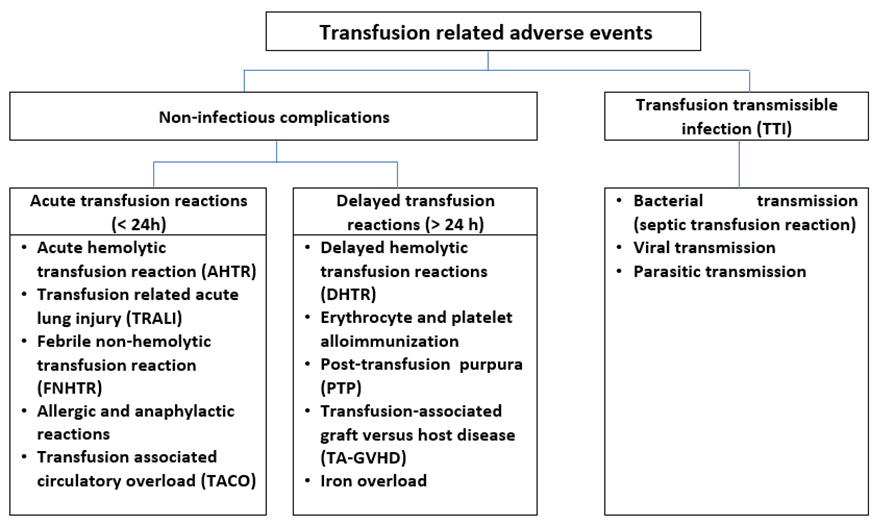
Figure S4: Different types of transfusion related adverse events.
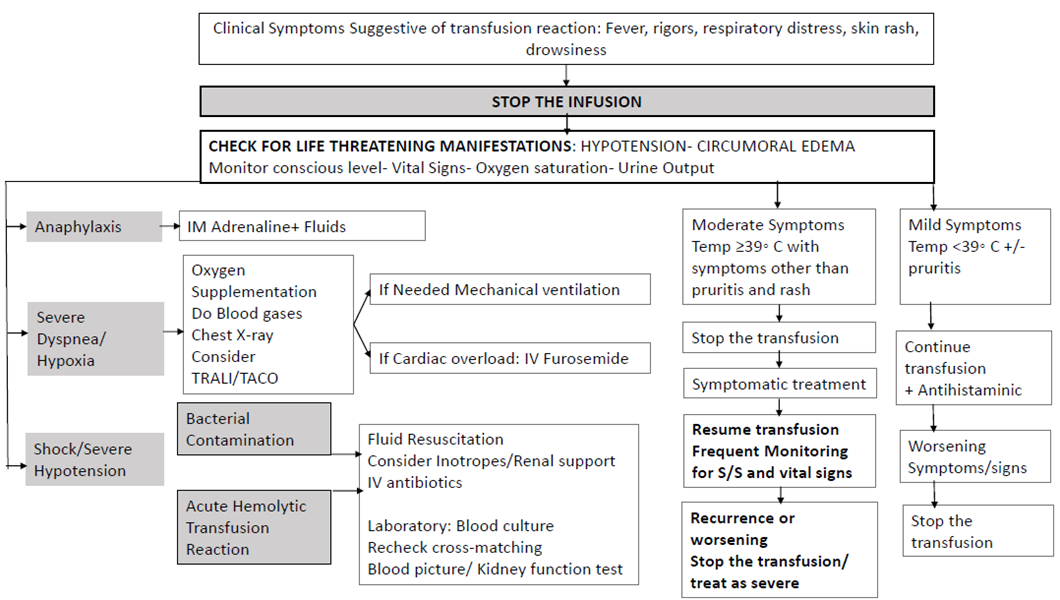
Figure S5: Management of acute transfusion reactions.