Management of Kawasaki Disease and its Cardiac Sequelae
- Introduction
➡️Background:
The Egyptian experience in the diagnosis and management of Kawasaki disease (KD) has evolved and intensified over the past years, especially with the emergence of atypical, incomplete cases and with the appearance of Multisystem Inflammatory Syndrome in Children (MISC). Meanwhile, a survey conducted by Kawasaki Disease Arab Initiative (Kawarabi) in 13 Arab countries, showed that the quality of medical services received by children with KD in large cities was rated as excellent in 6/13 or good in 7/13 countries compared to fair in 4/13 or poor in 4/13 countries in rural areas and they concluded that KD patients in mid-size cities and rural areas have limited access to standard healthcare in the Arab world (9). Accordingly, the establishment of Egyptian evidence-based guidelines for the management of KD in children is a necessity with consideration of the available facilities and the local limitations.
➡️What is Kawasaki Disease?
KD is an acute, self-limited febrile illness that predominantly affects young children, especially those under 5 years of age, with a median age of onset of 9 to 11 months. Approximately 25% of cases occur in older children, and it rarely affects adults (10). It is more common in males than females by a ratio of 1.5 to 1 (10). Age and gender play an important role as risk factors for complications, as age less than one year or above 9 years, and male gender have universally been identified to be significant risk factors for developing coronary artery aneurysms (11).
➡️Seasonal peaks: -
Evidence of seasonal variations has been demonstrated with a peak incidence in January through March in the Northern hemisphere, compared to a peak in May through June in the Southern hemisphere (12). This seasonality is suggestive of an environmental agent that may play a role in disease causation in different regions and ethnicities. Because the incidence of Kawasaki disease peaks during winter and spring, an infectious agent as a primary trigger may be suggested (13).
➡️Etiology and pathogenesis:
Despite more than 50 years of study, the exact etiology of Kawasaki disease remains unknown. Immunologic response to an exposure in the respiratory system or gastrointestinal (GI) tract or both in a genetically susceptible child is the most accepted theory. The immunologic cascade leads to systemic inflammation in medium-sized arteries and multiple organs in the acute phase (14).
➡️Diagnostic criteria: -
Recommendations for the diagnosis of KD are shown in table A
KD is categorized into two types: Classic KD(CKD) and Incomplete KD (IKD) (11). Typical KD or CKD have clear diagnostic criteria which are shown in figure (1) a and (1) b. Not all features of KD appear at the same time and watchful waiting might be needed to reach a diagnosis.
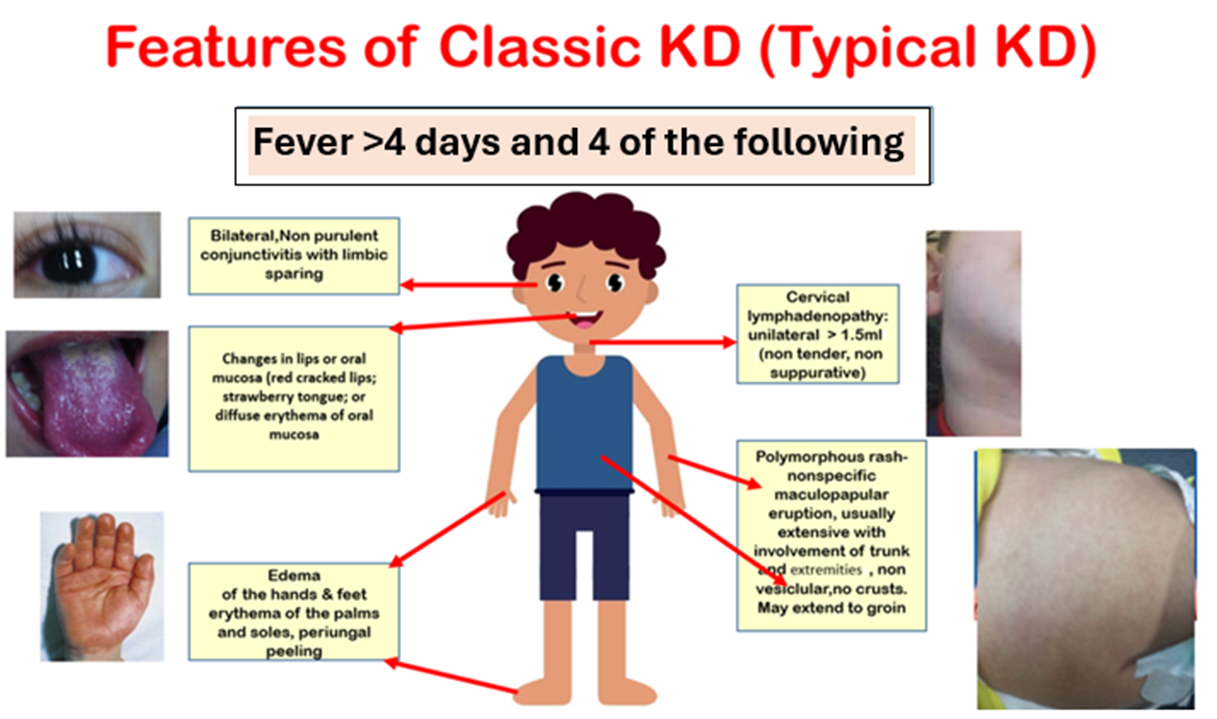
Figure (1a): Features of classic Kawasaki disease (KD).
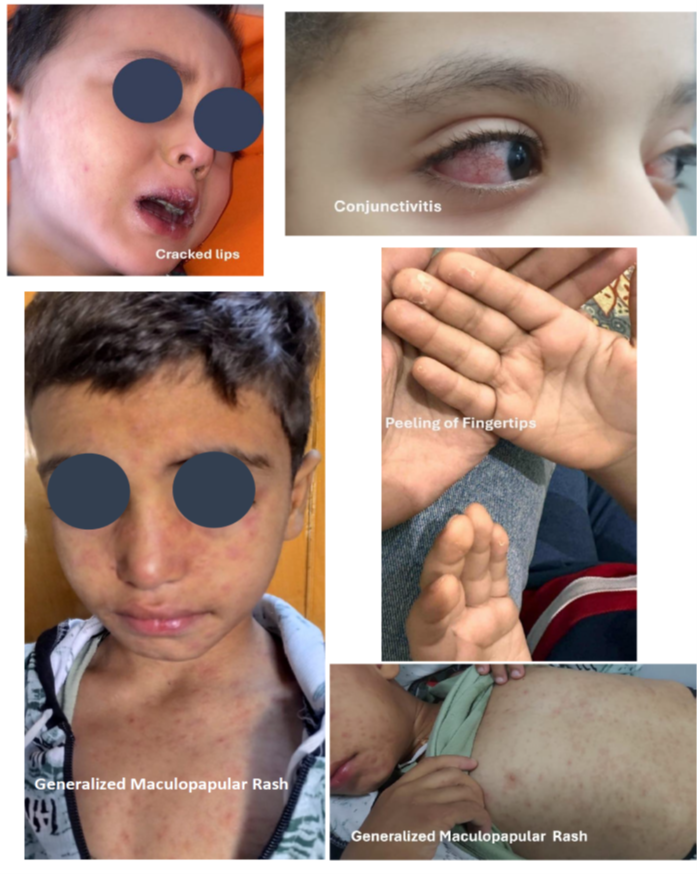
Figure (1b): Features of classic Kawasaki disease (KD).
Additional symptoms can be evident in KD patients but are not encompassed by the principal criteria (fig 1c and fig 1 d), they include arthritis, gastrointestinal involvement, irritability, lethargy, neurological manifestations, cough, and rhinorrhea. Arthritis, primarily affecting the large joints of the lower extremities (such as the knees, hips, and ankles), can be identified in 7.5% to 25% of patients and is typically temporary and non-deforming (15,16). In exceptional instances, abdominal imaging techniques such as radiographs or computed tomography (CT) scans might reveal indications of pseudo-obstruction, a condition that can sometimes manifest before the appearance of cardinal symptoms (17).
In incomplete KD (IKD), children have some symptoms appearing late or not at all, making it somewhat invisible and overlapping with the clinical symptoms of various pediatric infectious or connective tissue diseases. This increases the likelihood of misdiagnosis and underdiagnosis, leading to residual cardiac complications, and even death (18).
The estimated ratio of male to female patients in children with IKD is 1.5: 1.
Among the mucocutaneous manifestations, nail abnormalities can be observed. Orange-brown transverse chromonychia has also been occasionally described. During the convalescent phase, transverse leukonychia and Beau’s lines are the most common (20).
On the other hand, Atypical KD occurs in patients who, along with the usual clinical features of KD, also have a few unusual clinical manifestations, such as pulmonary involvement and renal impairment (21).
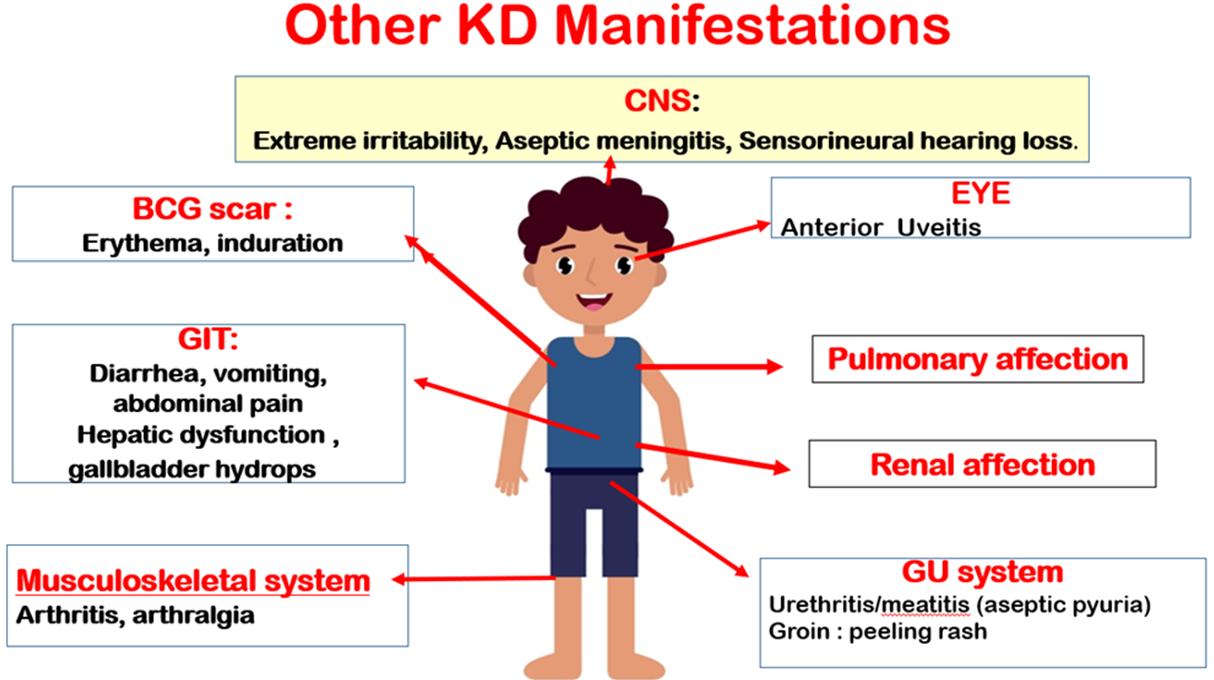
Figure (1)c: Additional clinical features of KD.
CNS: Central Nervous System: Bacillus Calmette Guerin; GIT: Gastrointestinal tract; GU: Genitourinary
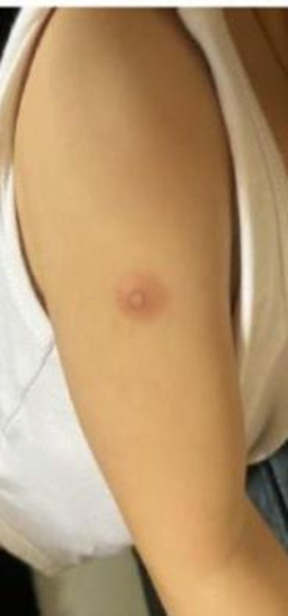
Figure (1)d: Induration of BCG scar in atypical KD patient.
Recommendations for diagnosis of classic, incomplete and atypical Kawasaki disease are shown in Recommendation table A.
➡️Disease Course:
Kawasaki Disease natural clinical course can be divided into three phases:
Acute (1st-2nd Week), Extreme irritability exists, by the end of which CAA can occur
Subacute (3rd-4th week), characterized by defervescence, periungual skin desquamation, thrombocytosis and progress of or appearance of new CAA,
Convalescence phase (5th–8th week), in which all KD signs disappear, inflammatory markers normalize, and transversal nail indentations (Beau’s lines) may appear (22).
➡️Cardiovascular Involvement in Kawasaki Disease:
Coronary artery involvement is the most serious complication in children with KD. Coronary artery abnormalities occur in 25% of untreated patients and 5% of patients treated with intravenous immunoglobulin (IVIG) (7). A study from Egypt reviewed a series of 580 patients ≤ 40 years of age presenting with symptoms of coronary artery ischemia and reported lesions consistent with antecedent KD in 6.7 % of cases (24). Data on exact prevalence of CAA in Egyptian children is not yet determined.
The proximal left anterior descending artery and the proximal right CA are the most frequent locations of CAA, and the posterior descending artery is the least common. (25)
Table 2 Highlights Cardiac affection in KD according to the American Heart ➡️Association (AHA) Guidelines 2017 (7):
|
Positive Echocardiographic findings suggestive of cardiac involvement in acute KD Include: 1. Left anterior descending coronary artery or proximal right coronary artery with a Z-score ≥ 2.5 2. Coronary artery aneurysm formation 3. ≥3 of the following suggestive features may be used to support KD diagnosis if classical clinical features are incomplete: Mitral regurgitation. Pericardial effusion. Decreased left ventricular function. Z-scores in the left anterior descending coronary artery or right coronary artery of 2 to 2.5.
|
Several non-coronary complications have now been identified in this condition, but these are often overlooked. Myocarditis is an integral component of KD and may be more common than coronary artery abnormalities. Myocarditis is universal in almost all patients with KD during the acute phase of disease. Transient left ventricular dysfunction can occur in more than 50% of patients. Pericardial involvement and valvular abnormalities have also been observed in patients with KD (25). Cardiac affection in KD is shown in figure (2).
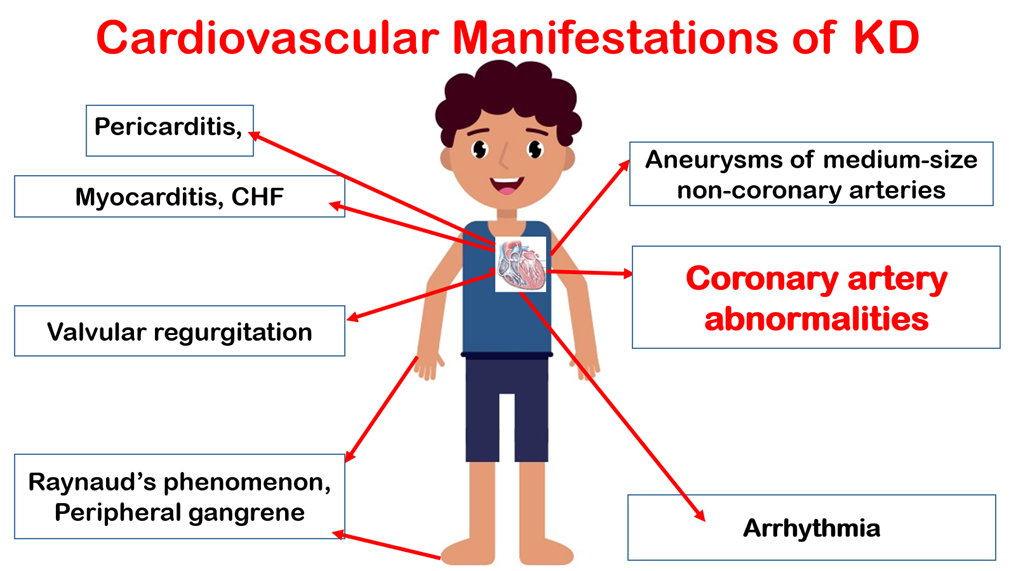
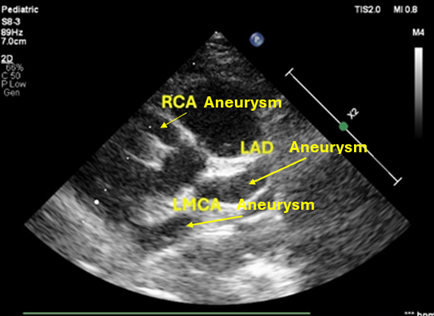
Figure (2): Cardiovascular affection in KD.
CHF: Congestive heart failure, RCA: Right coronary artery, LAD: Left anterior descending, LMCA: Left main coronary artery.
On the other hand, KD shock syndrome (KDSS) is now being recognized and may be difficult to differentiate clinically from toxic shock syndrome (26). In KDSS, KD manifestations are accompanied by multiorgan involvement and reduced organ perfusion due to systolic hypotension. KDSS is a serious condition that can present to the emergency department as an initial feature when typical clinical symptoms of KD have not been detected (27). Patients may have more prominent inflammatory markers and result in shock and hypotension, which requires critical care support at an early stage. Features that enable distinguishing KDSS from the usual KD case presentation include: (1) patients with KDSS are older than the usual patient with KD and have a higher prevalence and severity of GI symptoms; (2) patients with KDSS present with worse biological and inflammatory markers, (3) exhibit a higher resistance rate to IVIG, (4) have a higher rate of CAA, and (5) have greater number of reported cases of ventricular systolic dysfunction and atrioventricular valve regurgitation.
This is different from Toxic Shock syndrome, which should be diagnosed according to the CDC criteria (29)
Kawasaki disease and Multisystem inflammatory syndrome in children: -
Recommendations for differentiation of KD and MISC are shown in Recommendation table A.
MIS-C and KD are considered 2 distinctive diseases triggered by different infectious agents. They may belong to the same umbrella of inflammatory disorders but differ in many aspects of etiology, demography, epidemiology, clinical and laboratory findings, and pathology. Both diseases share many clinical features such as fever, rash, and mucocutaneous involvement, and can affect multiple organ systems. The intensity of the inflammatory response and long-term cardiovascular sequelae diverge between KD and MIS-C. Whereas MIS-C presents a more intense inflammatory syndrome, myocardial dysfunction, and cardiogenic shock. KD vasculitis is associated with pathologic changes in the coronary arteries and long-term cardiovascular sequelae (30,31).
Differential Diagnosis of KD is displayed in Annex Table 4(32-37).
➡️Investigations:
A. Laboratory Investigations
Recommendations on investigations required in KD are provided in Recommendation table B.
Investigations in suspected KD patients are shown in Table2
Table 2: Investigations in suspected KD patients (2)
|
Blood cell count |
|
|
White blood cells |
Increased, especially polymorphonuclear cells. Rare to be decreased. |
|
Red blood cells |
Reduced with normal mean corpuscular volume |
|
Platelets |
Increased till the 2nd or 3rd week, normalized in 4-8 weeks. If decreased suspect Disseminated intravascular coagulopathy |
|
Inflammatory markers |
|
|
Erythrocyte sedimentation rate |
High with slow normalization |
|
C reactive protein* |
High with fast normalization |
|
Liver Function tests |
|
|
Transaminases |
High |
|
Bilirubin |
High |
|
Gamma Glutamyl transferase |
High |
|
Albumin |
Decreased in prolonged severe illness |
|
Other laboratory tests |
|
|
Urine |
>10 white blood cells by High power field |
|
Cerebrospinal fluid |
Aseptic meningitis (mononuclear cells with normal glucose/protein ratio) |
|
Synovial fluid |
Purulent fluid, normal glucose, white blood cells 125000-300000/mm3 |
*Diagnostic positive level used in Egyptian laboratories is > 5mg/L
• CRP: C -reactive protein, ESR: Erythrocyte sedimentation rate, WBCs: White blood cells, HPF: High power field.
N-terminal prohormones of brain natriuretic peptide (NT-proBNP) The utility of NT-proBNP as a biological marker in KD is based on the universal myocardial inflammatory component early in the course of the disease. Patients with KD have higher NT-proBNP at the time of diagnosis than other febrile patients, with a pooled sensitivity of 89%, and a specificity of 72%. Moreover, patients with resistance to intravenous immunoglobulin treatment and CAA were found to have higher levels of NT-proBNP, suggesting a prognostic role. Nevertheless, the non-specificity of NT-proBNP to KD limits its use as a stand-alone test. It is worth mentioning that NT-proBNP proved superior to BNP in the evaluation of KD at the onset of the disease, thanks to the longer half-life of the former providing greater sensitivity and specificity (23)
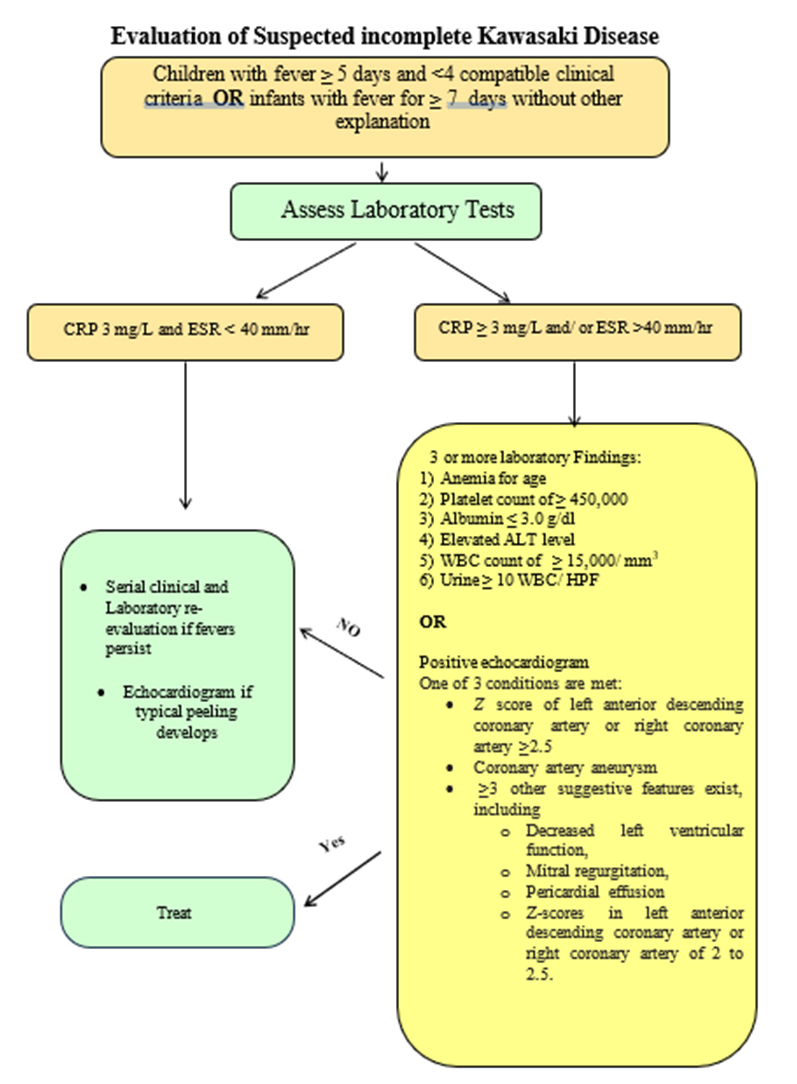
Evaluation of Suspected incomplete KD shown in the following figure
Fig 3: Evaluation of Suspected incomplete Kawasaki Disease (1,36)
B. Echocardiography :
It should be performed in all patients suspected or confirmed to have KD. Recommendations on the protocol of echocardiographic examination in KD are shown in Recommendation table B.
Routine use of CA Z scores has brought a level of standardization to quantification of CA size. It is important for centers to use the same Z score equation for comparisons over time in patients with KD.
Recommended frequency of echocardiographic examination in KD patients is shown in Fig 4.
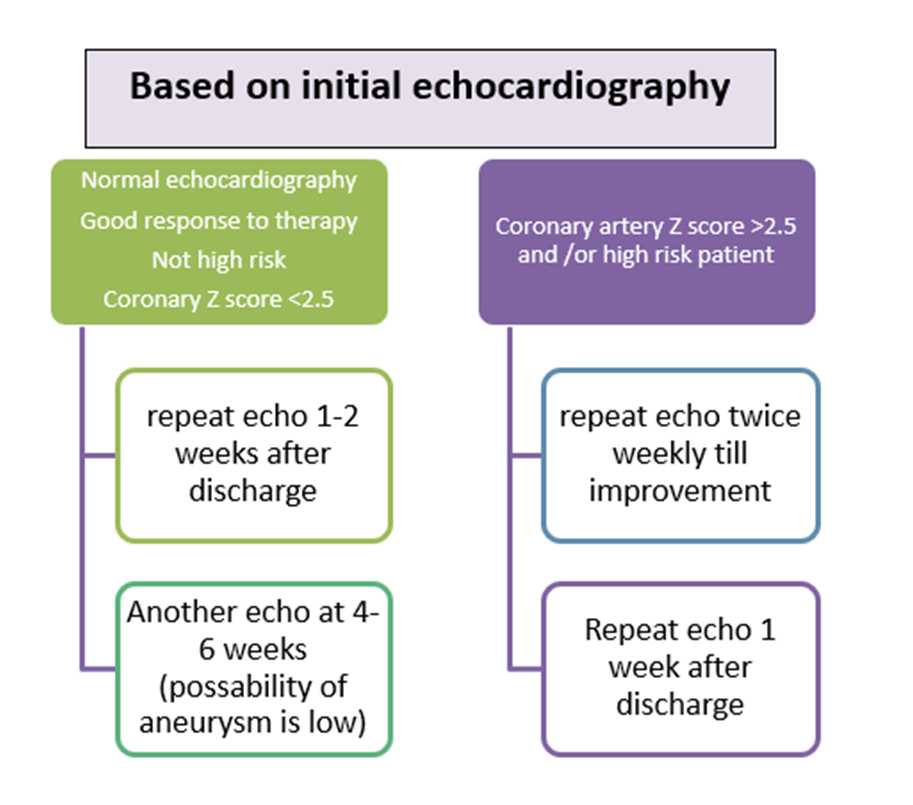
Fig (4): Recommended frequency of echocardiographic examination in KD patients.
Coronary artery measurements should be taken from the inner edge to inner edge of the vessel wall, avoiding the orifices and points of branching which may have normal focal dilatation (23)
The presence of marked perivascular brightness, the absence of the physiological gradual reduction of coronary artery caliber, and mild ectasia, if isolated, are not indicative of KD. These findings could be considered a positive echocardiographic KD sign when are all three simultaneously present (1).
Follow up of KD and CAA: TTE is the primary imaging tool for follow up CAA. Owing to its limited acoustic windows in distal CA, TEE and CT angiography are recommended. Advanced CA imaging using MRI or CTA may be needed for better designations in patients with KD. CTA will delignate aneurysm and stenosis. MRI for Myocardial function analysis and inducible ischemia. Stress echo can be used to follow pts with CCA and ischemia symptoms (36).
➡️KD Treatment: -
At presentation, the low-risk KD should be differentiated from high-risk cases (baseline CAA Z score ≥2.5, infants < 12 months or in high-risk category using the Son risk score, in addition to patients presenting with KD shock syndrome) (8) (Fig 5). Patients with standard risk can be treated with IVIg and aspirin. The general approach for treating low risk KD cases is administering a single high dose (2 g/kg) of IVIG intravenously over 10–24 hours within 10 days of disease onset. This should be accompanied by the oral administration of aspirin (30–50 mg/kg), until the patient is afebrile for 48 to 72 hours. In a classic KD, treatment should be initiated without waiting for echocardiography. (23,5) This can quickly and effectively relieve symptoms and decrease the incidence of CALs and CAAs (37).
To reduce the risk of hemolytic anemia in obese patients, IVIG dosing should be based on lean body mass. (38)
Patients with high-risk KD may benefit from intensification of initial therapy with IVIg plus adjunctive anti-inflammatory therapy to reduce the risk of CAA. These therapies include corticosteroids, tumor necrosis factor α inhibitors (eg, Infliximab & Etanercept), interleukin-1 inhibitors (eg, Anakinra), and cyclosporine (8). Proposed treatment regimen for KD is shown in figure 5.
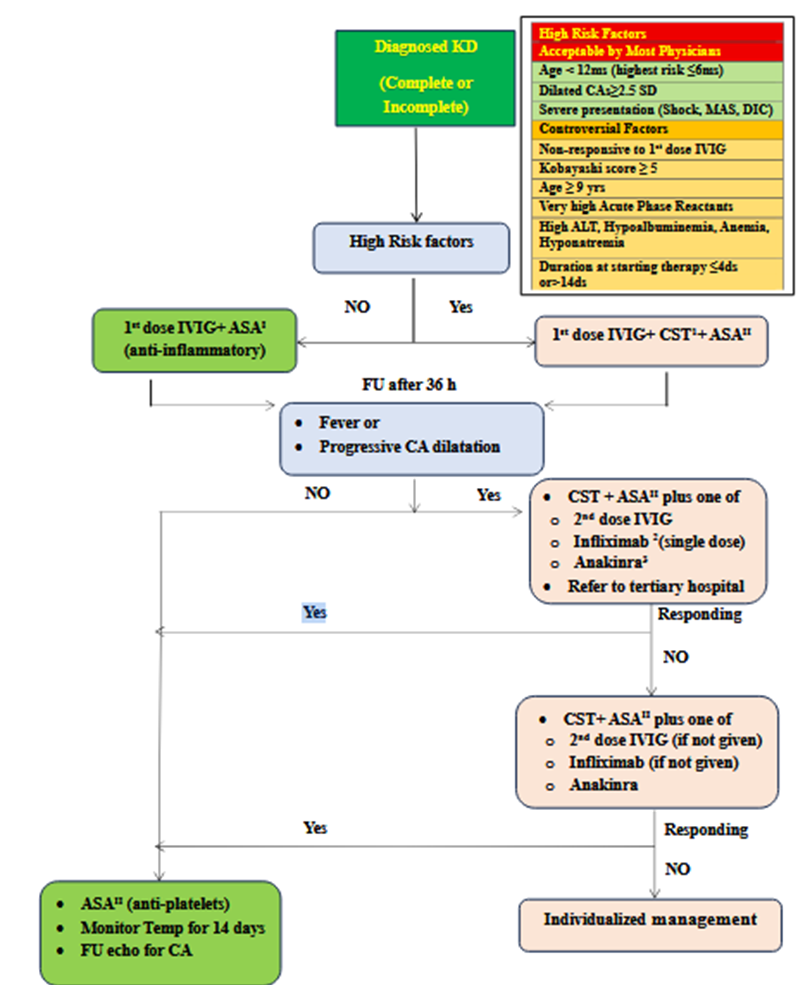
KD: Kawaski Disease, MAS: Macrophage activating Syndrome, DIC: Disseminated Intravascular Coagulation SD: Standard Deviation, ALT: Alanine Transaminase, IVIG: Intravenous Immunoglobulins, ASA: Acetyl Salicylic Acid, CST: Corticosteroid Therapy, CA: Coronary Artery. 1Most practitioners start with standard dose of steroids (2 mg / kg / day IV methylprednisolone) but some practitioners prefer high dose steroids (10-30g / kg / day methylprednisolone for 3-5 days then decrease to standard IV dose ) especially in low income countries in which IVIG treatment could be delayed to 12-24 hours after diagnosis of KD ( or more ) due to shortage of that expensive drug.2 Infliximab is given IV , 10 mg/kg over 2 hours single dose .3 Anakinra ,10 mg/kg/day ,is given IV/SC (preferred IV divided /12 hours).
Figure 5: Proposed algorithm for management of KD(38,39,40).
For corticosteroids therapy as an intensification therapy, the RAISE study suggested the use of IV prednisolone, 2 mg/kg per day divided every 8 h for 5 d (maximum 60 mg/d) while hospitalized; then PO prednisolone 2 mg/kg per d divided every 8 h; slow tapering over 15 d (maximum 30 mg/dose) once CRP normalized. On the other hand, North American studies suggested using Methylprednisolone with the same doses given every 12 hours and to be tapered over 2–4 weeks with the dose cut in half every 5 days. (42,43) Another protocol for non-responders with KD should be managed with a second IVIG cycle and - in case of failure - with 3 pulses of methylprednisolone (30 mg/kg/day), followed by oral prednisone (2 mg/kg/day, then gradually tapered up to the resolution of symptoms and normalization of CRP)(1)
If the child has been ill for more than 14 days and inflammatory symptoms and laboratory changes are no longer present, only low-dose anti- aggregation dose of aspirin (3–5 mg/kg/d) should be administered to prevent thrombosis. Echocardiography should be performed in these patients at the time of presentation and at regular intervals within the next 2 months to follow up on coronary arteries to decide on the duration of aspirin therapy (44). There is increasing evidence that medium- or high-dose aspirin in the acute phase is likely not associated with improved CA outcomes (45,46).
Patients with no coronary dilation should be maintained on LDA (low dose aspirin) for 6-8 weeks with no further interventions. For patients with Z score 5 - <10 and absolute dimension < 8 mm they should be maintained on double antiplatelet ill regression of the aneurysm (23)
Medium-high dose ASA should be replaced in case of concurrent varicella or influenza to avoid the potential development of Reye’s syndrome though low-dose aspirin has not been associated with any documented cases of Reye syndrome and therefore is safe to continue in the event of intercurrent infection. Children may receive dipyridamole (1–5 mg/kg/day divided into 3 doses), ticlopidine (2–7 mg/kg/day divided into 2 doses), or clopidogrel (1 mg/kg/day in a single dose up to a maximum of 75 mg/day in children > 2 years or 0.2 mg/kg of body weight in children < 24 months) in replacement of ASA (47). It is recommended to avoid non-steroidal anti-inflammatory drugs such as ibuprofen in children receiving low-dose aspirin as they can antagonize platelet inhibition sought for with by aspirin (48,49).
|
|
Management recommendations for children with KD are shown in Recommendation table C.
Commonly used medications in treatment are shown in table (3).
|
Table (3): Commonly Used Medications in initial treatment of standard risk KD patients |
|||
|
Drug |
Dose |
Adverse reactions/precautions |
Pharmaceutical form |
|
IVIG |
Single dose ( infusion): 2 g/kg (IV) |
· Possible anaphylactic reaction, especially in children with IgA deficiency (preparation with the least amount of IgA should be selected) · Hemolytic anemia · Aseptic meningitis · Fever, chills, headache, myalgia, nausea, vomiting. Associated with a high infusion rate at beginning of treatment, in 5---15%: During or up to 1---2 days after infusion. · Infusion rate: a test dose is usually done in the first 30-60 min of infusion then the dose is completed. Total Duration 12 hours in patients with Normal Cardiac Function. In Patients with impaired cardiac function usually Total duration is 18-24 hours. |
Vials: 1 g, 2.5 g, 5 g, 10 g
|
|
ASA |
Anti-inflammatory: 30-50 mg/kg /day divided in 3-4 doses (Oral) |
· Gastrointestinal changes and bleeding · Hypoprothrombinemia. · Rhinitis. · Paroxysmal bronchospasm. · Hypersensitivity. · Mild chronic salicylate intoxication, which is characterized by tinnitus and hearing loss. Discontinue treatment if these symptoms appear. Given after meal or with large glass of milk |
Tablets: 75 . 81 . 100, 300, 320 mg Maximum Dose: 4000 mg |
|
|
Anti-platelets: 3-5 mg/kg/day in 1 dose (Oral) |
Tablets: 75. 81. 100 mg Maximum Dose : 150 mg |
|
|
Corticosteroids |
Methylprednisolone: (used in Egypt) · 10-30 mg /kg/day IV for 3-5 days, followed by methylprednisolone, prednisolone or prednisone 2 mg/kg/day IV or Oral with gradual taper based on patient evolution · 2 mg/kg/day IV until fever resolves and CRP levels decrease, with gradual taper based on patient evolution Prednisolone: · 2 mg/kg/day IV until fever resolves and CRP levels decrease, with gradual taper based on patient evolution (not present in Egypt)
|
· High blood pressure. · Fluid retention. · Peptic ulcer. · Infection. · Hirsutism. · Hypokalemia. · Alkalosis. · Weaknesses. · Myopathy with muscle atrophy. · Acne. · Cataract. · Raised intracranial pressure. · Osteoporosis. · Cushing syndrome. · Adrenal suppression. · Glucose intolerance. · Amenorrhoea. · Delayed growth |
Methylprednisolone: Vials: 500, 1000 mg. Maximum Dose: 1000 mg
Prednisolone: Suspension, 1mg/mL, 5 mg/mL Tablets: 5, 20 mg. Maximum Dose : 80 mg
|
IVIG resistance: -
Recommendations for the identification of risk factors of IVIG resistance as well as management are shown in Recommendation table C.
Patients with persistent or recurrent fever ≥36 hours after the completion of the initial IVIg infusion are defined as IVIg resistant (50). Fifteen to 20% of individuals with KD seem to be IVIG resistant and are at greater risk of developing CAA formation (15%) than those who respond to IVIG (5%). Various IVIG resistance (refractory) prediction scores, such as the Kobayashi score (table 4) have been devised for predicting IVIG resistance if total score exceeds 4. Son risk score also can be used to determine risk score. It comprises age <6 months, Asian race, CA Z score >2 on initial echocardiogram, and C-reactive protein >13 mg/dL (each with 1 point assigned, except for 2 points assigned to CA). A risk score ≥3 points is strongly predictive of CAA by 8 weeks after acute illness (23). These scoring systems are thought to contribute to the continuous reduction of CAL occurrence (50). Current evidence supports the use of infliximab as rescue therapy in IVIG- and methylprednisolone-refractory patients with KD. IL-1 blockade with anakinra is highly promising in treating the most dramatically severe multi-refractory patients with KD, with potential benefits also on the cardiovascular complications (51). Table 5 shows the additional anti-inflammatory therapies in high-risk KD patients or those resistant to initial IVIG treatment
|
|
Cut off |
Score |
|
· Na |
≤133 mmol/L |
2 |
|
· AST |
≥100 IU/L |
2 |
|
· Day of starting treatment (or diagnosis) |
Day 4 of illness or earlier |
2 |
|
· Neutrophils |
≥80% |
2 |
|
· CRP |
≥10 mg/dL |
1 |
|
· Platelets |
<300,000 / μL |
1 |
|
· Age (months) |
≤12 months |
1 |
Table (4) Kobayashi Scoring System for prediction of IVIG resistance (50)
AST: Aspartate aminotransferase, CRP: C reactive protein. Kobayashi score ≥5 points has a sensitivity of 76% and a specificity of 80% in Japanese population
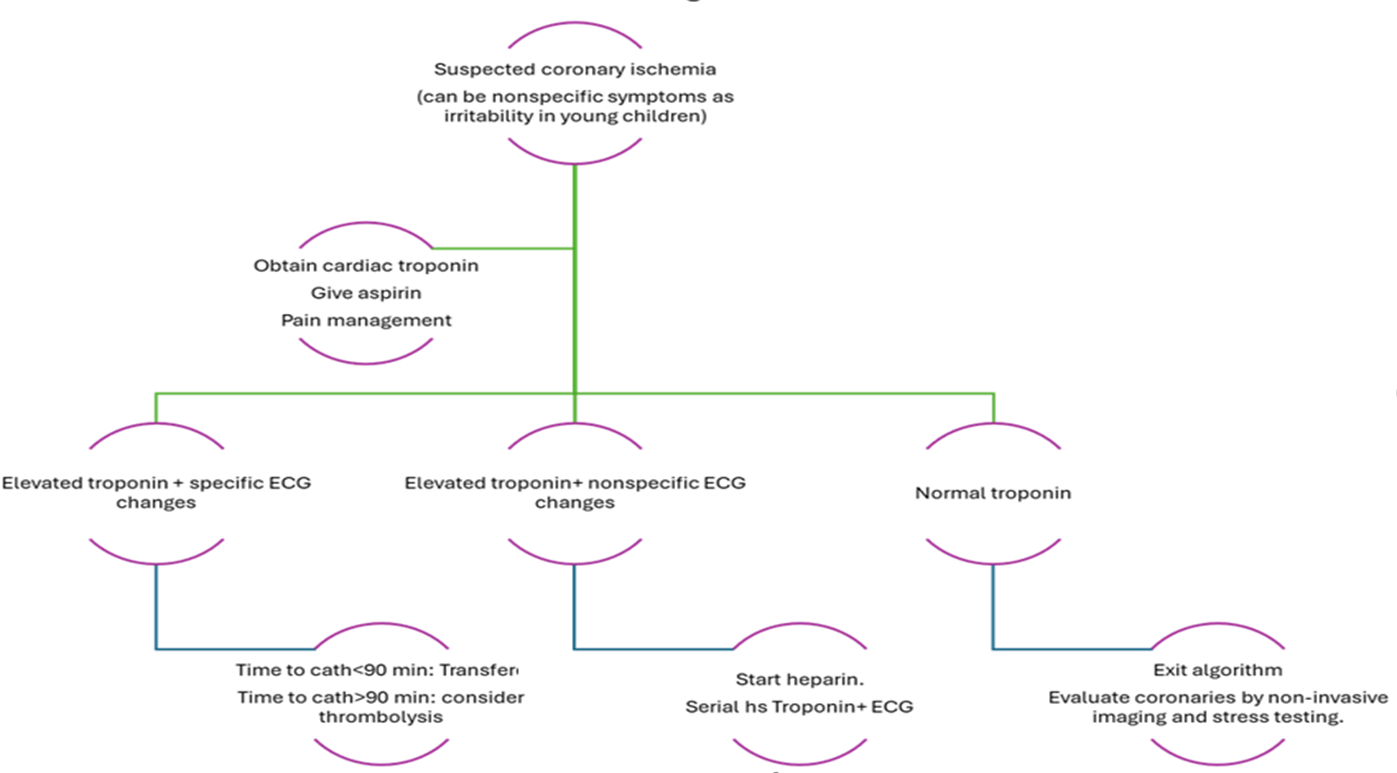
Table (5): Additional anti-inflammatory therapies in high-risk KD patients or those resistant to initial IVIG treatment *(51,52,53)
|
Drug name |
Mechanism of action |
Indication |
Dose |
Precautions |
Pharmaceutical form |
|
Anti-TNF alpha |
|||||
|
|
|||||
|
Infliximab
|
Chimeric murine/human IgG1 monoclonal antibody to TNF-alpha |
in KD patients with persistent inflammation despite IVIG, aspirin and corticosteroid treatment, after consultation with a specialist unit. |
IV, 10 mg/kg given over 2 h |
Exclusion of tuberculosis and infectious hepatitis before use Hold for suspected bacterial infection, fungal infection, varicella, or measles. |
100 mg of lyophilized infliximab in a 20 mL vial for IV use |
|
Etanercept |
Soluble receptor that binds TNFα and TNFβ |
SC, 0.8 mg/kg weekly ×3 doses |
Injection solution vials 25 mg and 50 mg for SC use |
||
|
Anti-IL1 |
|||||
|
Anakinra |
Interleukin-1 receptor antagonist |
In children with a refractory KD, Kawasaki disease shock syndrome, macrophage activation syndrome, persistent fever and laboratory abnormalities, worsening of coronary aneurysms, coronary aneurysms and increased proBNP levels and in patients with features overlapping with MIS-c |
IV/SC, 10 mg/kg per d (IV divided q12 h preferred to SC) while hospitalized; wean once ready for discharge (5 mg/kg per d for 1 d, then stop). |
Exclusion of tuberculosis and infectious hepatitis before use · Serum lipid monitoring after 2–3 months of therapy · Monitor for liver functions baseline and regularly |
Injection solution 100 mg/0.67ml for subcutaneous and intravenous use |
|
Plasma exchange |
|||||
|
|
Mechanical removal of inflammatory cytokines |
a high-risk procedure, should be reserved for extreme cases of refractory KD in whom all reasonable medical therapies have failed, especially if there are complications like severe infections or KD shock syndrome |
Displacing solution set at 5% albumin; 1–1.5× the patient’s circulating plasma volume is exchanged. Usually given for 3 continuous days (upper limit: 6 days) |
|
|
|
Other disease modifying antirheumatic drugs |
|||||
|
Cyclosporine |
Inhibitor of calcineurin – NFAT (nuclear factor of activated T cells) pathway |
May be considered in patients with refractory KD in whom a second IVIG infusion, infliximab, or a course of steroids has failed. In patients with MAS in combination with corticosteroids, IVIG and IL-1 antagonists |
PO, 5 mg/kg per d divided every 12 h; check 2 h level after 3rd dose (goal of 300–600 ng/mL); start to taper (by 10% every 3 d) once patient afebrile, clinically improving, and CRP ≤1.0 mg/dL or 10 d of therapy, whichever is longer |
If using liquid form, use glass dropper; may be mixed with milk, apple juice, or orange juice. Monitor blood pressure. Initial and regular blood counts; AST, ALT, BUN, creatinine, uric acid Reduce dose if creatinine increases by 30% Mg supplementation must be given while on cyclosporine to prevent hypomagnesemia. Should not be administered with statins as both are metabolized by cyt-P450 |
Oral solution (100 mg/ml) Oral capsules 25 mg, 50 mg Solution for intravenous (50 mg/ml) |
|
Cyclophosphamide |
Alkylating agent blocks DNA replication |
Might be used in refractory-KD. Should only be considered in severe refractory cases because of potential adverse reactions |
IV, 10 mg/kg per d in 1 or 2 doses |
Monitor blood counts, AST, ALT, BUN, creatinine. Adjust dosing or discontinue cyclophosphamide if WBC <1500/mm3, platelets <100,000/mm3, or hematuria Administer IV form with Mesna, good hydration and frequent bladder evacuation |
Injection powder solution 1gm/ 2 ml, 200 mg/ml
|
*Plasma exchange and biologic disease modifying anti-rheumatic drugs (DMARDs) like infliximab, anakinra cannot be recommended, except on an individual basis after consultation with a specialist unit.
*Non-biologic DMARDs such as cyclosporin, cyclophosphamide and methotrexate might be used in refractory-KD. They should only be considered in severe refractory cases because of potential adverse reactions, after consultation with a specialist unit.
IgG: immunoglobulin G, TNF: Tumor necrosis factor, SC: Subcutaneous, AST: Aspartate aminotransferase, ALT: Alanine aminotransferase, BUN: Blood urea nitrogen, MAS: Macrophage activation syndrome.
KD and Macrophage activation syndrome (MAS):
The recommendation of the identification and management of MAS in patients with KD is shown in Recommendation table C. Formal diagnostic criteria for MAS in the setting of KD have not been developed. However, drawing on experience with other secondary Hemophagocytic syndrome presentations, MAS may be suspected in KD patients with persistent fever, splenomegaly, elevated ferritin levels, and thrombocytopenia. Inadequate treatment of either KD or MAS could result in severe consequences. These include large coronary aneurysms or coronary artery stenosis, leading to death via cardiac infarct or coronary rupture in KD, or death due to multiorgan dysfunction in MAS. Thus, to ensure appropriate therapy, each disease entity should be considered separately with appropriate targeted therapy. KD should be treated with IVIG as the first-line therapy, and MAS should also be treated with appropriate agents for targeting cytokine storms or underlying triggers. Anakinra and glucocorticoids are preferred for treatment in these patients over a primary HLH-directed treatment protocol with cytotoxic agents. (51)
Thromboprophylaxis
KD patients with small or medium-size aneurysms receive antiplatelet therapy, following the belief that thrombus development is initiated by platelet activation and adhesion. In large aneurysms, however, humoral clotting factors are involved secondary to flow stasis; therefore, anticoagulant therapy is deemed necessary. Both Warfarin and LMWH have been shown to have similar efficiency in preventing thrombosis in KD patients with large aneurysms. They both have similar safety profiles with no substantial differences in the cumulative incidence of major bleeding complications (52).
Frequent blood work is required for routine monitoring of warfarin to ensure maintenance within a therapeutic window, and dosing can be challenging due to warfarin’s possible interactions with food. Certain foods that are rich in vitamin K may affect the therapeutic effects of warfarin, while other foods, such as grapefruit juice, interact with the metabolism of warfarin via the cytochrome P450 pathway (53). Furthermore, warfarin has many known drug interactions. Antimicrobials such as trimethoprim-sulfamethoxazole and erythromycin have been known to enhance the effects of warfarin, whereas rifampicin has been reported to reduce its effects. In addition, genetic polymorphisms have been found to be associated with warfarin metabolism, which further complicates the issues with dosing (54).
LMWH has added advantages over warfarin because of its somewhat less intensive monitoring requirements and faster achievement of monitoring levels within the therapeutic window, although potential negative effects on bone health associated with long-term treatment and discomfort associated with the injections make it less than ideal as a solution. It is a particularly useful option for younger patients in whom frequent blood work is less feasible (55)
Warfarin is advantageous compared with LMWH because of its ability to counter the anticoagulation effect promptly by means of vitamin K administration. The anti–factor Xa activity of LMWH is only partially reversible with protamine.
In the last decade, direct-acting oral anticoagulants, namely, direct thrombin inhibitors and anti–factor Xa agents, and novel antiplatelet agents have been introduced in adults. (29) These agents have been known to have certain advantages over classic anticoagulants, such as minor or absent food and drug interactions. In addition, they require limited or no monitoring and are delivered via the oral route, which may be a better option for patients who are averse to daily injections. Although their nonreversible nature has long been a limitation of direct oral anticoagulants from a safety point of view, antidote therapies for those agents are emerging which will allow reversal in case of severe bleeding events. However, these agents have not been adequately studied in children. Once there is more safety and efficacy data, regulatory approval, and availability of pediatric preparations, direct-acting oral anticoagulants will likely replace warfarin, not only for KD patients, but also for many other pediatric patients (56).
Recommendations table D shows the Management of coronary involvement in children with KD.
Coronary thrombosis and Myocardial Infarction in relation to KD:
KD patients with CAA are at highest risk of myocardial infarction in the first 2-3 months after illness. The presentation of these patients is different than adults. Patients with giant CAA have a lifelong risk of ischemia (58,59,3). The algorithm for management is shown in figure 6 .
Figure (6): Management of possible coronary thrombosis in children with KD
Long-term management (after convalescence) of KD patients with coronary lesions
Recommendations table E shows the long-term management of KD
Medical therapy for myocardial protection such as beta- blockers (carvedilol, metoprolol, or bisoprolol) decrease the risk of myocardial infarction and death by reducing myocardial oxygen demand. Angiotensin converting enzyme (ACE) inhibitors or Angiotensin receptor blockers (ARBs) also protect against myocardial infarction and death. Statins in addition to their cholesterol-lowering action have other pleiotropic effects in inflammation, endothelial dysfunction, oxidative stress, platelet aggregation, coagulation, and fibrinolysis, which make them useful in the management of KD. Huang, et al. reported a beneficial effect of short-term (3 months) statin treatment (simvastatin, 10 mg/day as a single dose at bedtime) in KD patients complicated with CAL. Chronic vascular inflammation is also significantly improved, as well as endothelial dysfunction, with no adverse effects. (61) However, long-term and randomized control trials are needed before further conclusions can be made.
The use of an ACE inhibitor in combination with a beta-blocker during the acute phase of Kawasaki disease (KD) in normotensive infants has been proposed in some observational studies; however, this approach has not been formally recommended by current guidelines. While there is a theoretical rationale based on the role of angiotensin II in promoting endothelial proliferation and vascular remodeling, which may contribute to coronary artery aneurysm (CAA) progression, the clinical evidence supporting such an intervention in the acute phase of KD is limited and of low certainty. Therefore, the panel judged this strategy to be speculative and not supported by sufficient high-quality data to inform a clinical recommendation. Further research is needed to assess the potential benefits and risks of this approach.
Following up of patients depends largely on the degree of coronary affection:
Patients with no coronary dilation should be maintained on LDA (low dose aspirin) for 6 weeks with no further interventions. If dilatation only was present at the acute stage, it should be reassessed at regular intervals till 1 year post convalescence.
For patients with small coronary artery aneurysms (Z score 2.5–4.9), regular follow-up with echocardiography is recommended. Aspirin may continue until normalization of coronary dimensions. The routine use of stress testing or coronary CT angiography (CTA) in this group is not currently supported by strong evidence and may be reserved for selected cases based on clinical risk factors or symptoms. Further studies are needed to determine the optimal imaging interval in this subgroup.
Patients with Medium and Large Coronary Aneurysms (Z score ≥5):
For medium aneurysms (Z 5–<10, diameter <8 mm): Dual antiplatelet therapy (e.g., aspirin + clopidogrel) should be considered until aneurysm regression with baseline ischemia assessment (stress echo or coronary CTA) at ~1-year post-illness to be repeated every 2–5 years, based on symptoms or clinical risk. Beta-blockers and stains may be considered for possible endothelial protection and ischemia risk reduction (62).
For Large aneurysms (Z ≥10 or diameter ≥8 mm): should consider combined antiplatelet and anticoagulation therapy in addition to beta blockers. Contact or high-impact sports should be restricted due to bleeding and ischemic risk. In addition to regular advanced imaging and close specialist follow-up (63).
Transition to adult care:
Patients with KD and coronary artery lesions, either persistent or those that have been remodeled and have decreased to a normal internal luminal dimension, should have long‐term follow‐up, with their care transitioned to an adult cardiologist once they reach adulthood, generally between the ages of 18 and 21 years.
The transition from pediatric to adult care for these patients needs to be a process involving a deliberate and coordinated series to ensure uninterrupted care. Transition should involve 6 cores:
1.The policy: preparing an office transition guide for families, including discussion of the practice's approach to an adult model of care around privacy and consent.
2. Tracking: ensuring that patient is offered a transition readiness/self‐care skill assessment conducted periodically.
3. Transition readiness. The pediatric clinician should have a standardized way of assessing the youth's self‐care skills.
4. Planning should include creating a transition plan jointly developed with youth and families that includes preparing decision-making support, assembling a medical summary and emergency care plan shared with youth and families, identifying an adult cardiologist and primary care clinician, and preparing transfer information for and communication with the new clinician.
5. Transfer of care, which requires engagement during this vulnerable transition time of both the adult clinician and the pediatric subspecialist to help the young adult schedule a follow‐up appointment with the adult health care clinician within 3 to 6 months of leaving the pediatric clinician.
6. The sixth core element is transition completion, during which the pediatric clinician confirms that the young adult has been seen by the adult clinician and facilitates obtaining patient feedback about the HCT process.
The 6 core elements is a process that evolves over several years and includes working with the adolescent's developmental stages to achieve maturity, education, and self‐management. More details on transition can be checked on the scientific statement from AHA 2021 (64).