Tilapia Lake Virus disease (TiLVD)
- Diagnosis
o Presumptive diagnostic assays
Common clinical signs
The diseased fish show general clinical signs as anorexia, anemia, emaciation, skin abrasion and discoloration, haemorrhagic patches on the skin, raised scales, exophthalmia, and abdominal distention (Figure 1).

Fig.1: Genral clinical signs of tilapia lake virus infected tilapia showing detached sclaes, skin discoloration and ulceration (red arrow head) [a], skin hemorrahges [b], abdominal distension (black star) [c], and bilateral exophthalmia (blue arrow) [d], corneal opacity [E]. Cited from Jansen, M. D., Dong, H. T., & Mohan, C. V. (2019).
Although TiLV doesn't cause specific signs on fish but the high mortality accompanied with corneal opacity (Eyngor et al., 2014).
Behavioral sign
behavioral changes like gathering in the bottom, sluggish movement, abnormal swimming behavior, and avoidance of schooling before death were also observed during the outbreak (Dong et al., 2017a).
Internal clinical signs
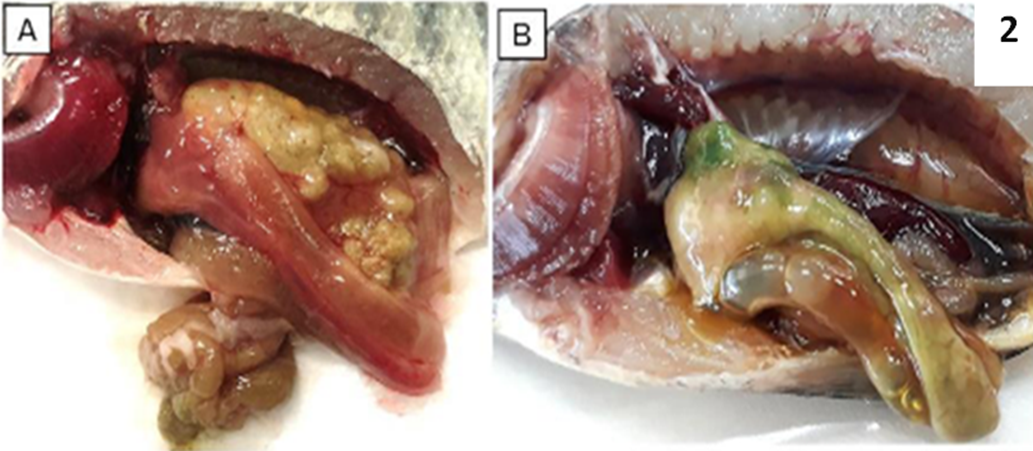
Liver appeared pale and in some cases, the liver tissue turned green or dark color. Also, infected fish show enlarged spleen and gall bladder, accumulation of fluid in the intestine and abdomen cavity (Figure, 2).
Fig 2: PM examination of normal tilapia (A), and TiLV infected tilapia showing pale gills and liver, congested spleen and kidney, enlarged gall bladder and green colored liver (B). cited from Tang, K.F.J., Bondad-Reantaso, M.G., Surachetpong, W., Dong, H.T., Fejzic, N., Wang, Q., Wajsbrot, N. & Hao, B. 2021.Tilapia lake virus disease strategy manual. FAO Fisheries and Aquaculture Circular No. 1220. Rome, FAO.https://doi.org/10.4060/cb7293en
· Lab Diagnosis
Collection of samples
Clinical samples include liver, spleen, kidney, heart, brain, gills, and whole fry. These samples used for histopathology, molecular assays and virus isolation on cell line.
The samples that will be used for:
- Histopathological examination: must be fixed in 10 % buffered formalin.
- Molecular techniques: the sample should be frozen (at -20 °C or -80 °C) and the others are preserved in 95% ethanol (or RNAlater, or equivalent reagent). Indeed, the combination of RNAlater® with deep-freezing at -20 ºC proved to be the most optimal storage conditions for preserving TiLV genomic RNA (Tang et al., 2021).
- Viral isolation: should be aseptically collected and immersed in Hanks’ balanced salt solution (HBSS). Alternatively, the sample can be promptly frozen at -80 °C or preserved on dry ice..
- Non-lethal samples, such as mucus, skin scraping, and blood, can be used as alternatives to killing fish for research purposes. These samples can be used for pond site diagnosis when using portable diagnostic instrument. After the fish have been sampled, they can be safely returned to the ponds.
➡️ Histopathological diagnosis
The liver of tilapia exhibits the presence of syncytial cells, which are characterised by the presence of many nuclei within each cell. These nuclei can range from 3-4 up to 15-20 per cell (Del-Pozo et al., 2017).Other lesions include massive cellular necrosis with pyknotic and karyolytic nuclei are also found in the hepatocytes, together with eosinophilic cytoplasmic inclusion bodies (Senapin et al., 2018). Figure 3.
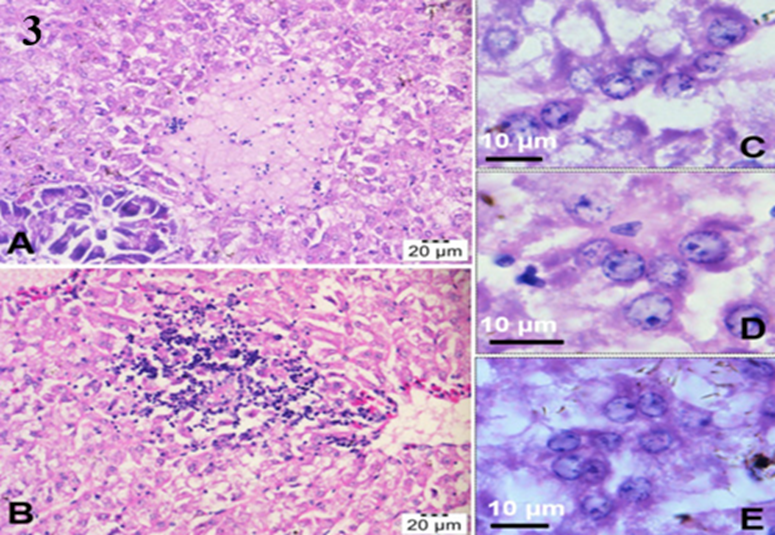
Fig3: H&E stained sections of the fish liver showing focal necrosis of hepatocytes and infiltration of lymphocytic inflammatory cells (A, B). Presence of hepatocytes resembling giant cells which contained multiple nuclei (C–E) was also observed. Senapin et. al (2018).
➡️Transmission electron microscope
Diagnosis of TiLV can be confirmed by Transmission electron micrographs (TEM), which reveals the presence of spherical virus particles in the cytoplasmic area (Dong et al., 2017b) Figure 4.
NB. Samples which are used for (TEM) must be fixed in 2 % glutaraldehyde and stored at 4 °C
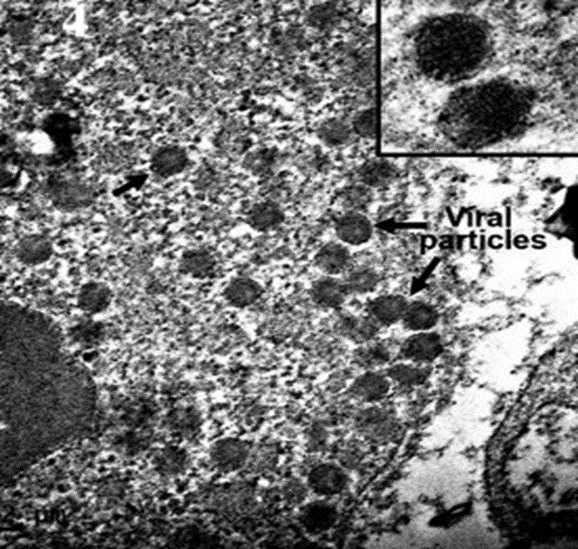
Fig 4: TEM ultrastrcutre of TiLV infected liver, with intracytoplasmic virions observed at higher magnification showing round-shaped particles with diameter around 100 nm, Dong et al. (2017b).
Molecular Techniques
- RT-PCR
Several techniques of RT-PCR for accurate and sensitive detection of TiLV in samples even in a symptomtic or apparent healthy fish. Single RT-PCR, nested RT-PCR, SYBR green-based qPCR, probed-based qPCR, semi-nested RT-PCR, in situ hybridization (ISH) using DIG-labelled probe. The scientists have developed commercial kits based on RT-PCR and RT-qPCR.
- RT-LAMP
This approach enables the identification of TiLV at the pond site using inexpensive equipment such as a water bath.
- Cell culture
TiLV can be cultured on many cell lines including E-11 (derived from Channa striata, a species of snakehead fish), CFF (from Pristolepis fasciatus, a species of Malayan leaffish), OmB (from Oreochromis mossambicus), OnlB (from Nile tilapia brain), OnlL (from Nile tilapia liver), or CAMB (from hybrid snakehead brain). Following the introduction of the virus into the cells, the cells should be placed in an incubator at a temperature range of 25 to 27 ºC for a period of 4 to 12 days. During this time, the cells should be monitored daily for any cytopathic effects (CPE). Infected cells exhibit CPE such as shrinkage, aggregation, rounding up, and detachment from the culture surface (Figure, 5) Tattiyapong et al., (2017)
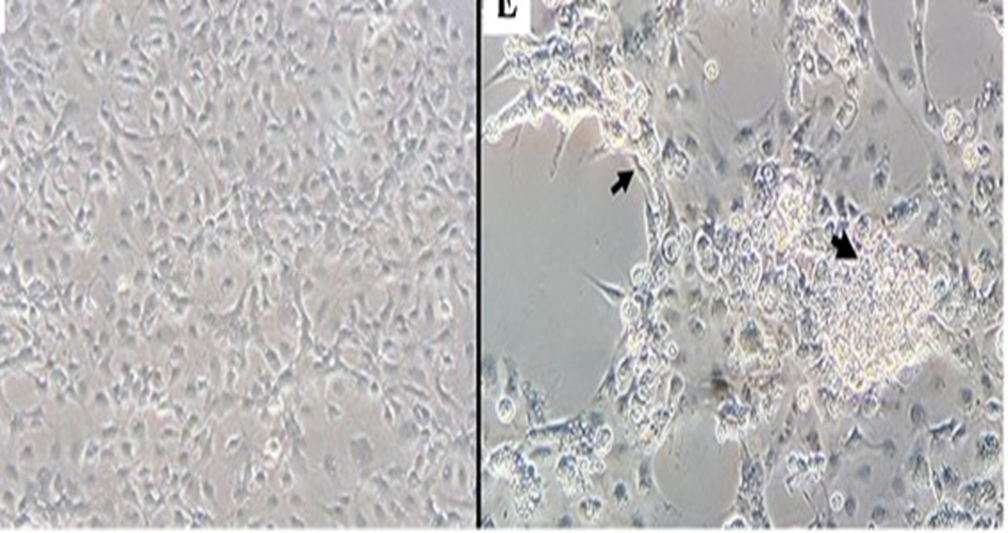
Fig 5: (A) E-11cells inoculated with tissue homogenate prepared from normal tilapia brain; (B) E-11 cells inoculated with tissue homogenate prepared from TiLV-positive brain, CPE with cell shrinkage and syncytial formation (black arrow), four days post-inoculation. Tattiyapong, et al, (2017).