Fish Samples for Disease Diagnosis
| Site: | EHC | Egyptian Health Council |
| Course: | Aquatic animal medicine Guidelines |
| Book: | Fish Samples for Disease Diagnosis |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 1 May 2024" Download Guideline
- Committee Chair
Committee Chair: Prof. Ahmed M Byomi
The Decision of the Committee: Prof. Dr Mohamed Mohamady Ghanem.
Committee Members: Prof. Nabil yassien; Prof. Ashraf Aldesoky Shamaa; Prof. Amany Abbass; Prof. Dalia Mansour; Dr Essam Sobhy.
Authors: Mohamed Faisal1,2; Amany A. Abbass1 ; Adel A. Shaheen1; Amel M. El Asely1; Eman A. Abd El-Gawad1; Hiam S. Elabd1; Aya F. Matter1; Hadeer A. Youssef1, and Amira M. El-Daim1.
1Department of Aquatic Animal Medicine, Faculty of Veterinary Medicine, Benha University, Egypt.
2College of Veterinary Medicine, Michigan State University, USA.
- Fish Sample Collection
Like in terrestrial animals, the ultimate success of disease control relies primarily on the proper laboratory diagnosis which is a function of the proper choice of samples with the highest likelihood of detecting the pathogen. The integrity of samples upon receipt by the laboratory is of paramount importance to the fidelity of laboratory results. The recommended protocols in this section represent the universal standards accepted by most laboratories and regulatory agencies and should be followed verbatim unless there are other policies or regulations that require omission or addition of other steps at the sampling process. Before you start sampling there are a number of questions that you need to be guided by their answers. These are:
Why do we need to sample farmed or wild fish?
1. To determine cause of morbidity or mortality
2. To determine prevalence and incidence of a disease
3. To determine freedom of disease in a farm, compartment, or a zone
4. For farm inspection or certification purposes
5. To prevent transboundary transmission of pathogen along with live shipments
6. To better understand disease ecology
7. To determine level of Disease impact
8. To determine the epidemiology status of a disease
9. For surveillance purposes:
10. Following an exposure event to sewage or untreated water.
11. To determine the disease status of fry collected from the wild before stocking them with other farmed fish.
What are the general guidelines for sampling?
The following should be considered:
- Representation of all age groups,
- Include all strains of the same species present at the facility,
- In case of testing for freedom of disease, the sample should consist of a
statistically valid number of a specific lot of a susceptible species
- Moribund fish should be included when present.
- If more than one water source is used for fish production, fish from all water sources should be included in the sample.
- Sample collection should be performed under aseptic precautions paying attention not to mix or mislabel samples.
- If the investigation requires RNA isolation, an RNA-stabilizing buffer should be added to the sample (e.g., RNAlater)
- If histopathological examination is ordered, tissues must be placed in a fixative, such as 10% neutral buffered formalin or Davidson’s fixative. Use 10:1 volume of fixative to sample.
- Maximum likelihood of detecting a pathogen during
disease course
When is the maximum likelihood of detecting a pathogen during the disease course?
The end of the incubation time and as soon as clinical disease signs appear because this is a period of the highest pathogen load in infected fish tissues. Samples for serological assays are best taken 14 days from the disease onset (Figure 1). However, for farm inspection, certification, and zone delineation, samples can be taken at any time of the year.
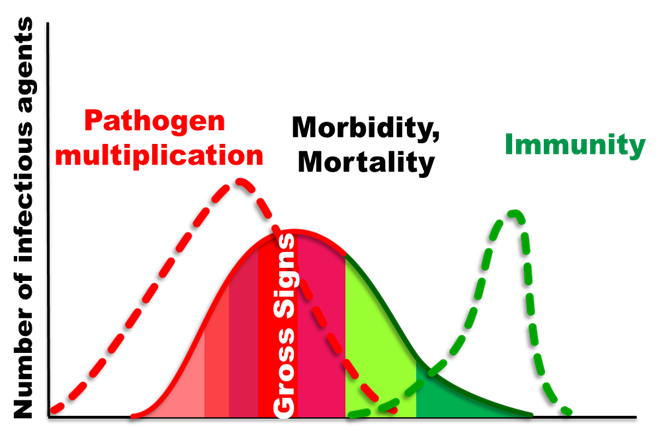
Figure1: A classical infectious disease course.
- Types of Samples and laboratory assay
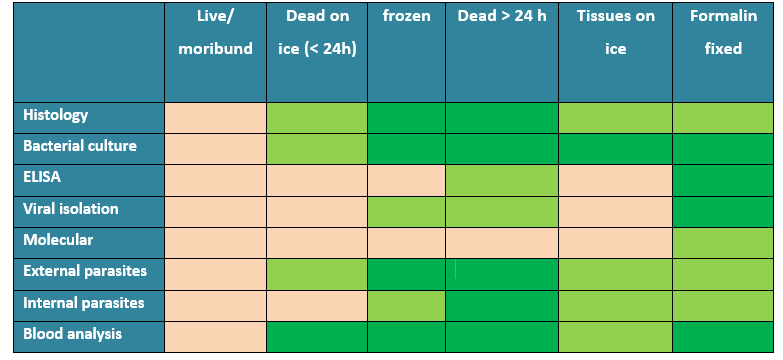
Which sample condition suits which assay?
Table 1 displays all possible sample types and their suitability for laboratory assays.
a) Live fish
The best samples are those collected from live moribund (Table 1).
Advantages:
- You can observe behavior and follow up if necessary.
- No postmortem degradation
- Perfect for blood collection and serology
- No bacterial postmortem invasion
Disadvantages:
- Some fish may not survive the transport.
- Overnight mailing is not available in remote areas.
- Difficult with larger fish
- High Cost
- Water can leak.
- Fish can develop diseases while in transport.
b) Freshly dead whole fish specimens placed immediately on ice.
This is the second choice for submission of samples for diagnostic purposes.
Advantages:
- Acceptable alternative for fish not expected to survive transport.
- Works well for viral isolation, ELISA, and molecular assays
- Less weight for shipping
- Does not require access to compressed oxygen.
Disadvantages:
- Overnight mailing may not be available in remote areas.
- Not ideal for proper bacteriology testing
- Not ideal for histopathology
- Not ideal for serological assays
- Not ideal for detection of external parasites
- Cannot observe behavior.
c) Frozen fish
Freshly dead fish specimens that are immediately frozen for diagnostic purposes
Advantages:
- Can be held until able to ship.
- Can be used for viral isolation, molecular assays, and some antigen capture assays.
Disadvantages:
- Not ideal for proper bacteriology testing
- Cannot be used for histopathology.
- Cannot collect blood or serum.
- Not ideal for detection of external parasites
- Cannot observe behavior.
- Requires access to dry ice for shipment.
Table 1: Types of samples and suitability for each laboratory assay.
- Number of fish to be sampled
What is the optimal number of fish to be sampled?
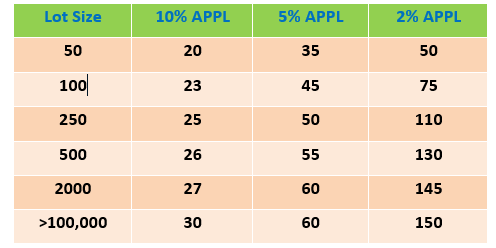
In the case of collecting samples for diagnosis a disease that is associated with clinical sign, mortality, or reduced growth, collect no less than ten fish preferably showing gross disease signs. In the case of inspection, certification, or zone delineation, number of fish to be collected from each lot must be in accordance with a plan that provides 95% confidence that at least one infected fish will be collected if the minimum assumed pathogen prevalence level (APPL) of infection equals or exceeds 5%,however, the level of confidence may vary from one country to the other, therefore, the veterinarian in charge of collecting samples should be aware of regulation, policies, and the nature of pathogen in question. For example, to determine freedom of virus in a lot requires a 2% level of confidence. Table (2) displays the number of fish needed to sample from each lot to achieve the desired level of confidence of laboratory results.
Table 2: Sample number based on an
assumed pathogen prevalence level (APPL) in the population of 10%, 5 %, or 2%.
Does fish age determine which organ to collect?
Yes, it does. In the case of fry and yolk sac fry the entire fish is collected after the removal of the yolk sac. For fish measuring 4-6 cm, the entire viscera and kidneys are collected in addition to the brain. For fish over 6 cm, the kidneys, spleen, heart, and brain are collected in addition to other tissues appropriate for the specific pathogen being tested for (e.g., skin and fins in case of herpesviruses or ovarian fluid in case of flavobacteriois).
- Fish Packaging and Shipping
How to package and ship live fish?
- Use sturdy plastic bags!
- Label each bag.
- Fill with one-third water, fish, and two-thirds compressed oxygen (Figure 2 and 3)
- Twist and fold top of bag
- Seal tightly with strong rubber band
- Double bag, sealed separately in same manner.
- Label each bag with the necessary information (Figure 4)
- Surround each bag with ice, as appropriate for species and weather.
- Contain fish samples and ice within another sealed plastic bag.
- Place in strong insulated cooler
- Fill the remaining empty space with packing material.
- Include submission forms and case history)
- Place in a cardboard box, if possible, for shipping
Precautions:
- Keep fish at low densities in bags.
- Do not feed fish for 24 hours prior to bagging.
- Pack fish immediately prior to transport
- Make sure there is enough gas in bag (twice as much O2 as water)
- Add an extra bag if fish have spines.
- Add extra ice during warm weather.
- Make sure bags are sealed tightly.
- Make sure ice is bagged to prevent leaking as it melts.
- Make sure the receiving laboratory knows the date and time of the expected shipment.
- Use tracking numbers immediately to locate fish if they do not arrive as expected.
How to package and ship freshly dead fish on ice?
- Use strong airtight plastic bags.
- Label each bag.
- Collected fish are placed in bags immediately following humane euthanasia.
- No water or air in bag
- ~5 fish per bag, dependent on size
- Surround each bag with ice.
- Contain samples & ice within another sealed plastic bag.
- Place in strong insulated cooler
- Include submission forms and case history.
- Place cooler in a cardboard box, if possible, for shipping
How to package and ship frozen fish?
- Freeze euthanized fish immediately after collection.
- Keep samples frozen until testing.
- Label each bag.
- Use strong airtight plastic bags.
- No water or air in bag
- Five fish per bag, dependent on size
- Seal bags tightly
- One layer of samples per bag
- Surround each bag with dry ice.
- Contain fish & dry ice within another sealed plastic bag.
- Place dry ice on top of fish, and newspaper or packing peanuts used to fill the air space above.
- Place in strong insulated cooler
- Include submission forms and case history.
- Place cooler in a cardboard box, if possible, for shipping
What information should be on the label on the outside of the box containing the samples?
- Date
- Location/source
- Species/lot ID
- Sample type
- Specimen number
- Type of preservative
- Submitted by
- Purpose of investigation
- Needed tests.
Photograph courtesy of Dr Mohamed Faisal, Michigan State University.

