sample collection and sample processing
| Site: | EHC | Egyptian Health Council |
| Course: | Avian and Rabbit Medicine Guidelines |
| Book: | sample collection and sample processing |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:47 AM |
Description
"last update: 25 Sep. 2024" Download Guideline
Table of contents
- - Acknowledgement
- - Abbreviations
- - Scope
- - The target audience
- - Aim
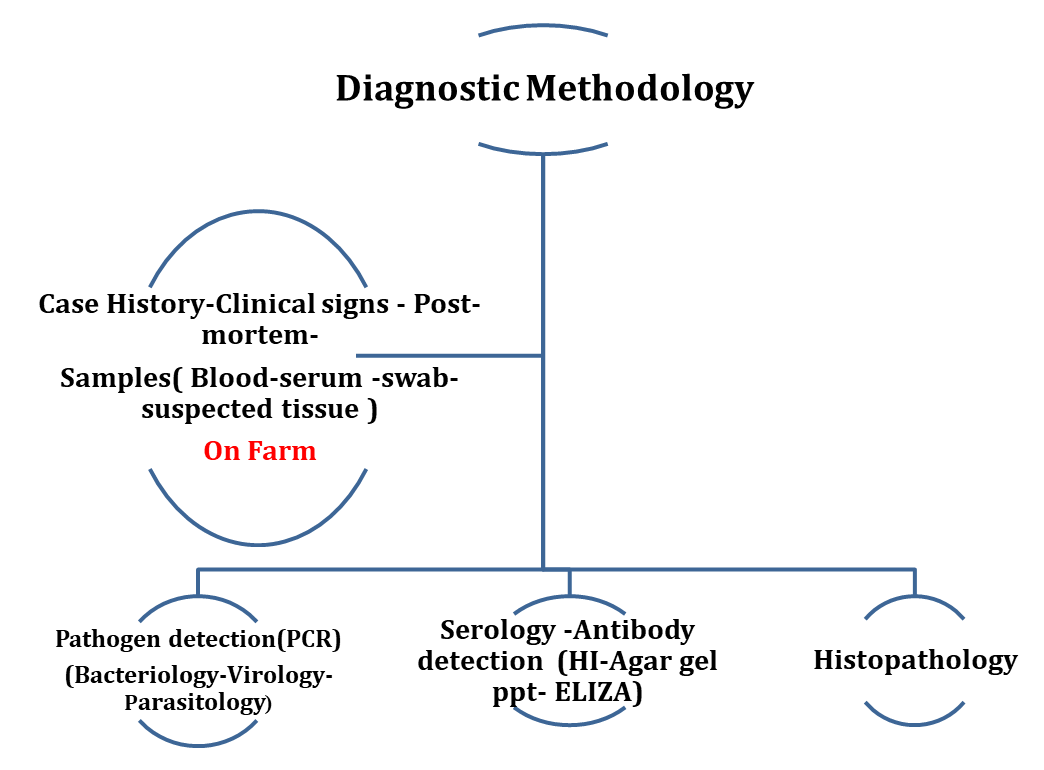
- - Diagnostic Methodology
- - Methods used for collecting blood sample
- - Serum collection and handling
- - Diagnose infectious diseases
- - Ages for Sampling
- - PCR Molecular diagnosis
- - Samples collected in relation to suspected symptom
- - Samples in relation to suspected diagnosis
- - Histopathology as diagnostic tools
- - REFERENCES
- Acknowledgement
We would like to acknowledge the committee of National Egyptian Guidelines for We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The Rapporteur of the Committee: Prof. Mohamed Mohamedy Ghanem.
Scientific Group Members:
Prof. Nabil Abd Elgaber, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby. Dr. Mohamed Elsharkawi, Prof. Gamal Sosa, Dr. Naglaa Radwan, Dr. Hend Elsheikh
Authors: Prof. Dalia Mansour, Dr. Essam Elmarakby.
- Abbreviations
AIV, Avian Influenza Virus, Flu, Influenza, Fowl Plague
ND Newcastle disease virus
ILT Infectious laryngotracheitis
Wet pox Fowl pox
IB Infectious bronchitis
MD Marek's Disease virus
I. Coryza Infectious Coryza
CRD Mycoplasma,
F Cholera Fowl cholera - Pasteurellosis
EDS Egg drop syndrome
- Scope
The guidelines for the evaluation the most suitable sample and diagnostic methodology for accurate diagnosis to prevent and control poultry diseases.
- The target audience
The guideline is intended for all veterinarians who are intended to diagnose, treat, and control Poultry Diseases.
- Aim
Design and implement diagnostic methodology and apply control measures to prevent diseases that usually affect poultry farms and should be based on disease prevalence and infectious pressure in the area, the farm conditions, and the correct choice of the type of vaccine.

- Diagnostic Methodology
On-farm, diagnostic activities comprise routine sampling and investigations in line with health control programs. The diagnostic process originally shifted from the traditional veterinarian approach centered on individual birds to the health assessment of entire flocks. Flocks are commonly classified as “healthy” if they perform according to their genetic potential and are considered free from clinical disease.
Clinical examinations are time consuming and labor intensive and may fail to detect diseases; especially subclinical diseases can be challenging to be accurately diagnosed. The manifestation of an infectious disease can vary from subclinical to severe clinical illness, depending on various etiological factors and influences such as the causative agent, host and/or environment altogether complicating diagnosis. Diagnostic procedures continue with post mortem investigations, on farm or in the laboratory, which serve to identify gross pathologic changes in organs and tissues in order to further specify a tentative cause of impaired performance and clinical signs.
Sending diagnostic sample to laboratory:
It is important to provide thorough and relevant flock information on the laboratory submission.
Diagnostic sample submissions include:
• Flock identification and location
• Age of flock
• Date of sample collection
• Vaccination program
• Flock history, including pertinent health or production problems this information is vital to the flock veterinarian and diagnostician to make a meaningful interpretation of serological or diagnostic results and provide recommendations to improve flock health and/or production.
Equipment used for Blood Collection:
-Disposable, sterile 3 or 5 cc syringes are used, depending on the size of the sample to be obtained.
-The size of needle depends on the anatomical site used for blood collection.
-Blood Collection Site Needle Length Needle Gauge Wing vein 0.5–1.0 (1.25–2.54 cm) 20–22 gauge Cardiac puncture 1.5 (3.81 cm) 18–20 gauge.
-Always use disposable needles and replace needles every 5 to 10 birds.
-Dull needles cause tissue trauma and make inaccurate punctures of veins.
- All blood collection equipment must be changed between flocks to eliminate the potential for disease transmission.
- Methods used for collecting blood sample
1.Wing (brachial) vein method using a needle the brachial vein of the wing is an acceptable site for blood collection for birds 4 weeks and older. In younger birds, this vein is too small for efficient blood collection.
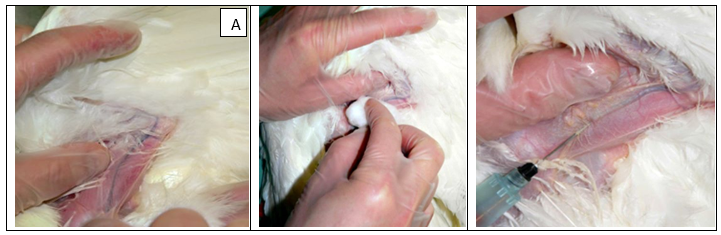
A)Wing vein prepared for puncture must disinfect area around the vein then inserted the syringe correctly.
2. Heart puncture collecting blood directly from the heart can provide rapid blood collection, and allow for collection of larger volumes of blood (4 to 10 mL). Additionally, cleaner blood samples can be collected compared with wing vein method. Cardiac puncture methods should only be practiced by trained personnel. Poor technique in needle placement and repeated attempts to locate the heart can result in fatal hemorrhage; however, this risk is minimized with practice. If fatal hemorrhage is suspected.
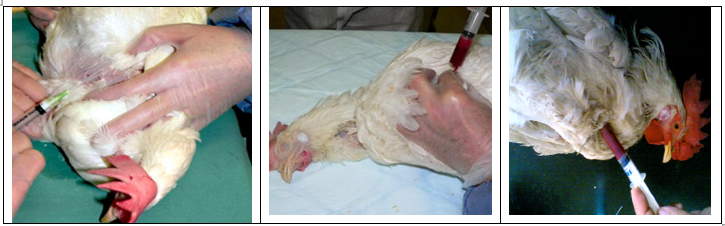
-The route of cardiac puncture was achieved by inserting the needle between the clavicles into the heart.
-Once a blood sample has been collected into a syringe, the sample should be carefully transferred to a tube to promote clot formation.
-Clotting occurs when all the cells in the blood are drawn together by the coagulation process and separates from the fluid portion of the blood (serum).
• The needle should be removed from the syringe before the blood is pushed into the clotting tube. Forcing the blood back through the needle will rupture red blood cells (hemolysis), resulting in a poor quality sample.
• Slowly inject the blood into the clot tube, allowing it to run down the side of the tube, which encourages clot formation. Blood must be placed in the clot tube before the coagulation process begins.
• Do not disturb the blood tubes while the clotting process is occurring. Tubes should be allowed to stay positioned nearly flat (horizontal) to maximize surface area of the clot as it forms. The amount of serum yielded from clotted blood depends on the surface area of the clot. Tubes held upright in the vertical position have little surface area and produce only a small quantity of serum. Use a test tube holder for keeping tubes in this flat position. If a test tube holder is not available, then a block of wood drilled with appropriate sized holes or a wire rack can be used.
- Serum collection and handling
- After serum has separated from clotted blood, pour serum out of the clot tube into another collection tube, or tease the clot out of the tube with a wooden stick (such as a toothpick), leaving only serum in the tube.
- A clot must be handled gently during the process of separating the serum. A good quality serum sample will appear clear to pale yellow in color.
- Cloudy, slimy, or hemolysis samples should not be sent to the laboratory.
- Sending Serum Samples to the Laboratory Once serum has successfully separated from a clot, it should be kept cool (7°C) and sent immediately to the laboratory.
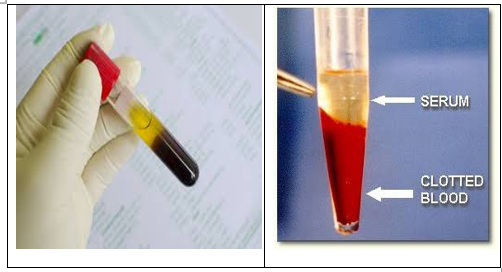
Guidelines for serum Collection:
• Select normal representative birds (10 to 20 sera samples), unless working up a diagnosis.
• Collect 2.0 to 3.0 mL of blood from each bird.
• Samples collected with a needle are cleaner than with a scalpel.
• Do not damage samples by forcing the blood sample back through the needle into the clot?
• Ensure blood runs down the side of the clot tube and position the tubes nearly flat until the clot is formed.
• Leave blood in the clot tube for 10 to 12 hours at 27°C (or for 2h at 4-6 °C).
• Do not shake, roughly handle or freeze the blood while the clot is forming or hemolysis will occur.
• Remove clot gently, or pour off serum.
• Do not mail samples without first removing the clot.
• Keep the serum samples cool and send immediately to the laboratory on wet ice.
- Diagnose infectious diseases
-Starting at the farm with the collection of data on disease history, combined with initial post mortems. Further investigations require a laboratory to proceed with different techniques and disciplines.
Sample selection
Normal, healthy individuals should
provide serum samples for routine serological investigation. Birds must be in good health (Sick or
distressed cull birds) should not be used since their antibody titers are
usually not indicative of the general health status of the flock. Blood
samples, however, taken from birds displaying the clinical symptoms or lesions
of the expected pathogen or syndrome during a possible disease inquiry.
It's crucial to choose birds for caged housing systems from different parts of the
house. The same birds (or cages) should be used for blood collection when a
flock is enrolled in a routine serology program. When compared to drawing blood
from various birds for each test, this will lessen the variation in results.
Ages for routine blood collection in breeder flocks
1. 10 to 12 weeks .
2. At time of transfer (grow to lay farm) .
3. Every 10 to 12 weeks during egg production .
Ages for routine blood collection in commercial layer flocks :
1. One time prior to transfer (grow to lay farm) .
2. Every 10 to 12 weeks during egg production .
Pathogen detection from suspected material for definite diagnosis :
-Routine samples include whole blood, serum, formalin-fixed tissue and swabs: (tracheal, oropharyngeal, cloacal, organs and joints).
-For specific investigations, PCR, rt-PCR, whole blood or isolates from any type of swab.
Virology
Poultry is the host of numerous viruses. PCR-based viral infection diagnosis has emerged as the preferred technique due to its quick nucleic acid detection. However, isolation is frequently required to deal with functional elements of the disease or to get antigen for the creation of vaccines. Specific application of PCR is a DIVA (differentiating infected from vaccinated birds) PCR, a method used for screening a vaccinated flock for infections with a filed/virulent strain, The initial isolation of a virus depends mainly on adaptation and propagation in embryonated specific pathogen-free (SPF) chicken eggs and cells obtained from those embryos or hatched SPF birds.
Bacteriology
A certain trend to introduce molecular techniques (PCR) that complement or substitute classical bacteriology. The clinical picture and post mortem findings cannot be attributed to a specific bacteria or fungi. Therefore, one of the main goals of bacteriological and fungal examination in diagnostics is the isolation of the pathogen. In addition to disease diagnosis, culturing of bacteria is also obligatory for some surveillance programs, such as that of Salmonella, which are based on the isolation of live bacteria
Parasitology
Diagnoses of poultry parasites depends particularly on macroscopically and microscopically examination.
Serology
In contrast to the isolation or detection of infectious agents, especially viruses, serological testing is fast and easy to perform with minimal laboratory requirements. Primarily, it focuses on the presence, absence or level of specific antibodies in the serum. In practice, antibodies can be detected in egg, tears, saliva, and mucus secretion.
Many of the classical serological techniques (e.g., the agar gel precipitation test, plate agglutination tests (RSA), and hemagglutination inhibition tests (HI)) have been employed for decades; standardized reference sera and antigens for many poultry pathogens are commercially available worldwide. Considering the high flexibility, quick turnaround and the large sample size to be processed at a time, ELISA assays have become routine procedures for flock health monitoring and for poultry diagnostics alike, with commercial systems available for the majority of poultry pathogens.
Serology importance:
The study of serum antibody levels, also known as titers. The immune system develops antibodies that circulate in the blood after a bird is exposed to an antigen, whether by vaccination or exposure to a wild-strain pathogen. Antibodies are found in the serum portion of blood (the liquid portion after the clot develops). Serum is free of all blood cells and clotting factors. The flock’s serum antibody titers are used to monitor efficacy of vaccination programs, evaluate field challenges or diagnose disease. The value of this information depends on the quality of the serum samples received by the laboratory. Poor quality samples lead to erroneous and misleading results. Selection of birds for blood collection, techniques used to collect blood, and handling of blood samples and serum all influence laboratory results.
NB: Selection of Birds For routine serological monitoring, serum samples should be collected from normal, healthy birds. Do not use cull birds that are sick or appear distressed, as their antibody titers are not typically representative of the overall flock health status. During a potential disease investigation, however, blood samples should be collected from birds that are exhibiting the clinical signs or lesions of the suspected pathogen or syndrome.
Number of Samples Twenty good quality serum samples should be collected for routine flock profiling and for disease investigation; however, a minimum of 10 samples may be sufficient to estimate flock antibody titers.
- Ages for Sampling
- Routine monitoring for antibody titer in laying house, the first blood collection should be 10 to 12 weeks of age. By this age, a pullet flock has an opportunity to respond to early live vaccinations and maternal antibodies are absent.
-Antibody titers from this age group can be used to assess the overall immune status of a young flock and priming effect of live vaccines used in vaccination programs. This early serology assessment can screen for potential disease challenge in the grow house.
-Another important time for antibody titer evaluation is immediately prior to transfer of the pullet flock to the laying house. This is a good time to check the pullet’s immune response against (Mycoplasma gallisepticum (MG), Mycoplasma synoviae (MS), Newcastle disease (NDV), infectious bronchitis (IB), avian encephalomyelitis (AE), and avian influenza (AI).
Routine monitoring for antibody titer in breeders house, transfer is also an ideal time to assess adequate sero-conversion for chicken anemia virus (CAV) and avian encephalomyelitis (AE). Collecting serum before transfer establishes a baseline titer level for a flock moved to a multi-age complex.
- Titer response from inactivated (killed) vaccines will peak at 3 to 5 weeks post-vaccination. When monitoring flocks during the egg production period, a 10 to 12 week interval is sufficient to monitor changes in antibody titer levels.
- Blood Sample With proper collection and handling technique, 2.0 to 3.0 milliliters (mL or cc) of whole blood will yield 1.0 to 1.5 mL of serum. This volume of serum is sufficient for routine for the following testing:
|
Serology test |
Disease |
|
ELISA testing |
- Newcastle disease (NDv) - Infectious bronchitis (IB) - infectious bursal disease (IBD or Gumboro), |
|
Agar gel immunodiffusion (AGID), |
- AE - AI |
|
Plate agglutination testing. |
- MG, MS - Pullorum-typhoid |
|
HI |
- Avian influenza - Newcastle disease virus - Infectious bronchitis |
NB: Sufficient serum should be kept frozen in reserve, in case additional testing is required in the future.
- PCR Molecular diagnosis
-The advent of molecular diagnostics such as PCR and rt-PCR has provided new tools for rapidly and accurately diagnosing poultry diseases. It is now possible to sequence the genome of many pathogens. Sequencing allows comparing isolates to better understand the disease epidemiology.
Samples: Tissues, swabs and body fluids can be submitted for molecular diagnostics.
-Samples for PCR, virus isolation, bacterial isolation or other tests can be obtained from swabbing the oral, trachea, cloaca, affected joints, and organs.
- Collection of Swabs Cotton tipped swabs are an effective, non-invasive method for sampling for Mycoplasmas, bacteria, and many viruses (examples: infectious bronchitis, avian influenza, infectious laryngotracheitis, Newcastle).
- Samples collected in relation to suspected symptom
|
Disease of Concern |
Tissues should be collected |
|
Gumboro (IBD) |
Bursa of Fabricius, Thymus ) |
|
Infectious Laryngotracheitis |
( Trachea • Larynx • Conjunctiva) |
|
AI |
Trachea • Larynx • Conjunctiva |
|
ND |
( Trachea • Larynx • Conjunctiva) |
|
Marek’s Disease |
( Sciatic Nerve ,Brain , Eye, Tumors ) |
|
Coccidia |
Enteritis, ceca |
|
REO |
-Portions of the gastrointestinal tract affected. -focal duodenal necrosis: |
|
Adeno |
Liver tissue, spleen |
- Samples in relation to suspected diagnosis
- When submitting samples to a diagnostic laboratory, it is important to provide thorough and relevant flock information on the laboratory submission form.
-Send the swabs to the diagnostic laboratory for immediate analysis.
-Do not freeze samples.
|
Pathogen |
Sample Pooling for PCR |
Media Used |
Site of the samples |
|
Avian Influenza |
pool of 10 swabs |
in 5.5mL media pool 5 swabs in 3mL media BHI (Brain heart infusion) |
-Tracheal -oropharyngeal |
|
Newcastle Disease |
pool of 5 swabs |
in 5.5mL media BHI |
- Tracheal -oropharyngeal |
|
Mycoplasma gallisepticum/synoviae |
pool of 5 swabs |
or BHI media |
-Dry Tracheal -oropharyngeal |
|
Bacteria and/or Viruses only pool tissues from a single bird |
pool by organ system (respiratory, enteric, reproductive) |
media provided in the culture tube |
-Affected organs |
- Histopathology as diagnostic tools
- refers to the evaluation of cells and tissues using a microscope.
-As a follow-up to the post-mortem exam, histology can be a valuable tool in assessing flock health. Some poultry diseases can only be diagnosed by histopathology.
- For example, the clinical presentation of infectious laryngotracheitis virus or wet pox within a flock can be virtually identical, but the diseases cause distinctly different and characteristic histopathologic changes that allow a definitive diagnosis.
-Successful use of histopathology practice requires the availability of appropriately selected and preserved samples.
-Sample Collection Collect specimens for histopathology as soon as possible after death to avoid deterioration of tissues.
-Fresh tissue samples from birds humanely euthanized immediately prior to postmortem examination provide the best quality slides.
-If mortality must be used for tissue collection, they should be determined to be fresh as possible, and not decomposed.
NB: Do not collect samples from birds that have been previously frozen. The freeze and thaw processes can disrupt cellular features, leading to poor quality slides. An individual sample should be no larger than 1 cm3 (1x1x1 cm) to allow for adequate penetration of the tissue with fixative.
- Larger pieces of tissue will decompose in the center before adequate penetration by the fixative (formalin).
-Sampling for Specific Diseases When there is concern for a particular disease based on regional risk, a suspicious result on surveillance testing, or clinical signs in the flock, specific tissues should be collected.
-Samples preservation for histopathology:
- Submerged in a solution of 10% neutral buffered formalin for preservation.
-The volume of formalin solution in a single container should be at least 10 times the volume of all tissues.
-Samples must be fully immersed in the solution to be adequately saturated by fixative to prevent deterioration.
- Lung tissue and other air-containing tissues may be wrapped gently in absorbent cotton to aide immersion.
-Gently open the lumen of trachea and intestine samples to release trapped air.
-Sample submission, when submitting samples to a diagnostic laboratory, it is important to provide thorough and relevant flock information on the laboratory submission form.
-Critical information that should accompany all diagnostic sample submissions beside, Special shipping regulations may apply for formalin filled containers, locally appropriate biohazard labelling on all transport containers. This information is vital to the flock veterinarian and diagnostician to make a meaningful interpretation of diagnostic results and provide recommendations to improve flock health and/or production.
-Sample processing, after arrival at the diagnostic laboratory, the formalin preserved tissues are embedded into a paraffin block, and then sectioned with a microtome into thin slices. Tissue slices of this thickness (4 micron) are thin enough to be examined by the pathologist under a light microscope. These slices are fixed on glass slides and stained. Various stains can be used to highlight different cell types, or other aspects of the tissue. The most frequently used stain for disease diagnosis is hematoxylin and eosin (H&E) stain.
- REFERENCES
1. Astill, J.; Dara, R.A.; Fraser, E.D.G.; Sharif, S. Detecting and predicting emerging disease in poultry with the implementation of new technologies and big data: A focus on avian influenza virus. Front. Vet. Sci. 2018, 5, 263. [Google Scholar] [CrossRef]
2. Bermudez, Alex J. and Bruce Stewart-Brown. Chapter 1: Principles of Disease Prevention: Diagnosis and Control, “Disease Prevention and Diagnosis”. Diseases of Poultry. 13th edition. Ames: Wiley-Blackwell, 2013. 2. USDA-APHIS. United States Veterinary Permit for Importation and Transportation of Controlled Materials and Organisms and Vectors. U.S. Department of Agriculture. 2016.
3. Collett, S.R.; Smith, J.A.; Boulianne, M.; Owen, R.L.; Gingerich, E.; Singer, R.S.; Johnson, T.J.; Hofacre, C.L.; Berghaus, R.D.; Stewart-Brown, B. Principles of Disease Prevention, Diagnosis, and Control. In Diseases of Poultry; Swayne, D.E., Boulianne, M., Logue, C.M., McDougald, L.R., Nair, V., Suarez, D.L., Wit, S., Grimes, T., Johnson, D., Kromm, M., et al., Eds.; Wiley: Hoboken, NJ, USA, 2020; pp. 1–78. ISBN 9781119371168. [Google Scholar]
4. Grafl, B.; Aigner, F.; Liebhart, D.; Marek, A.; Prokofieva, I.; Bachmeier, J.; Hess, M. Vertical transmission and clinical signs in broiler breeders and broilers experiencing adenoviral gizzard erosion. Avian Pathol. 2012, 41, 599–604. [Google Scholar] [CrossRef]
5. OIE World Organisation for Animal Health. Manual of Diagnostic Tests and Vaccines for Terrestrial Animals Chapter 3.3.4: Chapter 3.3.4 Avian Influenza (Including Infection with High Pathogencity Avian Infleunza Viruses); OIE World Organisation for Animal Health: Paris, France, 2021. [Google Scholar]
6. Skinner, J.T.; Bauer, S.; Young, V.; Pauling, G.; Wilson, J. An economic analysis of the impact of subclinical (mild) necrotic enteritis in broiler chickens. Avian Dis. 2010, 54, 1237–1240. [Google Scholar] [CrossRef].
7. Lab Animal volume 42, pages359–361 (2013).