Esophageal and Esophagogastric Junction Cancer
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Esophageal and Esophagogastric Junction Cancer |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:47 AM |
Description
"last update: 19 January 2025" Download Guideline
- Executive Summary
This guidance provides a data-supported approach to the primary prevention, screening, diagnosis, staging, treatment and follow up of patients diagnosed with Esophageal and Esophagogastric Junction Cancer. This guideline intended only for malignant esophageal tumors of epithelial origin, and not for any other non‐epithelial malignant tumors of the esophagus or metastatic malignant esophageal tumors.
|
Recommendation |
Strength of recommendation |
|
1.Primary prevention |
|
|
The following interventions may help to reduce the risk of esophageal cancer: · Treating gastroesophageal reflux disease (GERD) and Barrett's esophagus early · Prevention of injury to the esophagus · Avoidance of tobacco and alcohol · Avoidance of meat, processed food intake, hot beverages. · Diet rich in fruits and vegetables · Avoid obesity
|
Good practice statement
|
|
2.Secondary prevention (Screening) |
|
|
• Screening of esophageal and GEJ tumors in the general population is not cost effective and should not be done.
|
Strong
|
|
3.Diagnosis |
|
|
3A. All patients with new dysphagia, gastrointestinal bleeding, recurrent aspiration or emesis, weight loss and/or loss of appetite should undergo an upper gastrointestinal endoscopy. |
Strong |
|
3B. The location of the tumor relative to the lower incisors and GEJ, the length of the tumor, the extent of circumferential involvement, the presence of Barrett esophagus and the degree of obstruction should be carefully recorded to assist with treatment planning. |
Strong |
|
3C. Multiple biopsies, six to eight, using standard size endoscopy forceps should be performed to provide sufficient material for histologic and molecular interpretation. Larger forceps is recommended during surveillance endoscopy of Barrett esophagus for the detection of dysplasia. |
Strong |
|
3D. Diagnosis should be based on endoscopic biopsies (Chromo-endoscopy if available) with the histological tumor type classified according to the World Health Organization (WHO) criteria. The differentiation between esophageal SCC and AC is of prognostic and therapeutic relevance. |
Strong |
|
3E. Laparoscopy + washings could be done to exclude occult metastatic disease involving peritoneum/diaphragm, especially in locally advanced (T3/T4) adenocarcinoma of the GEJ infiltrating the anatomical cardia. |
Good practice statement
|
|
4.Pathology |
|
|
4A. Histological diagnosis should be reported according to the WHO criteria. |
Good practice statement |
|
4B. Immuno-histochemical staining including HER2 is recommended in poorly differentiated and undifferentiated cancers when differentiation between SCC and AC using morphological characteristics is not possible.
|
Good practice statement |
|
5.Staging and risk assessment |
|
|
5A. Consider Multidisciplinary team meetings (MDTs) for patients with esophageal cancer. MDTs often include surgeons, radiologist, pathologists, medical oncologists, radiation oncologists, gastroenterologists, dietitians, rehabilitation physicians, palliative care specialists and dedicated cancer nurse specialists. |
Conditional |
|
5B. Staging should include a complete clinical examination, Complete blood count (CBC) and comprehensive chemistry profile, endoscopy, chest /abdomen /pelvis CT with oral and IV contrast.
|
Strong |
|
5C. Consider 18F-fluoro-2-deoxy-D-glucose (FDG)-positron emission tomography (PET) in patients who are candidates for esophagectomy.
|
Conditional |
|
5D. Endoscopic ultrasound (EUS) is recommended in early lesions in order to assess tumor depth and lymph node status in patients amenable to upfront surgery or candidates for tri-modality treatment (T3N0, T1-4a and any locoregional N). If not available refer to a specialized center. |
Strong |
|
5E. We recommend bronchoscopy for tumors located at or above the carina in the initial staging, which can help in both surgery and radiotherapy treatments. |
Strong |
|
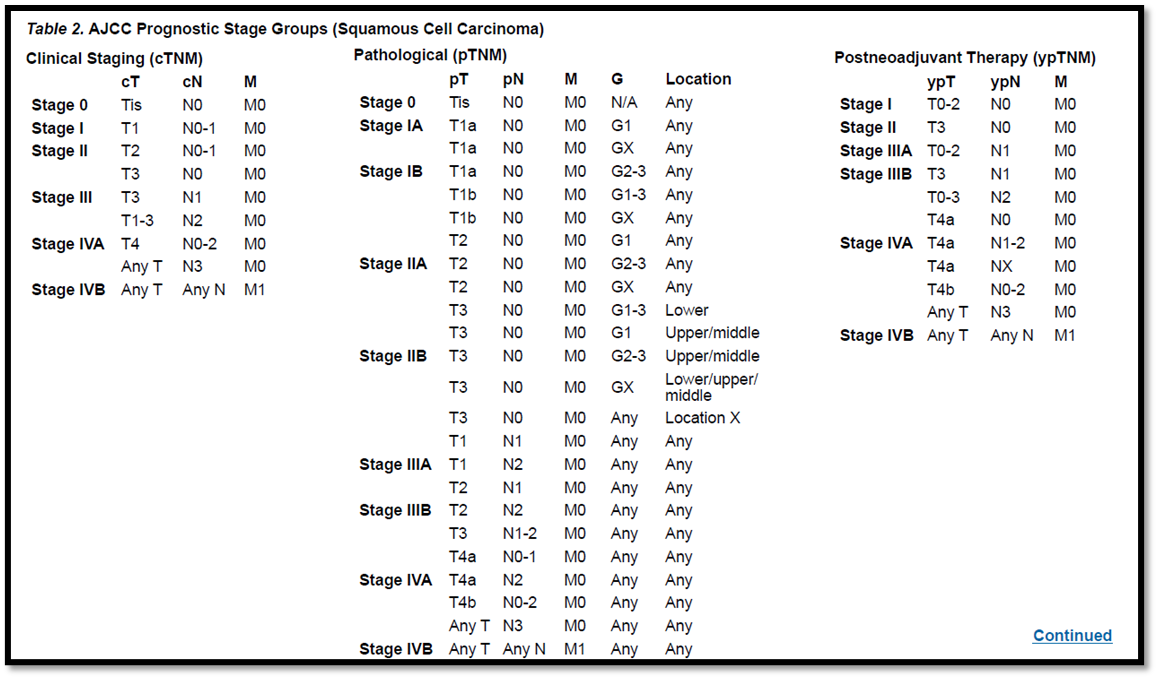
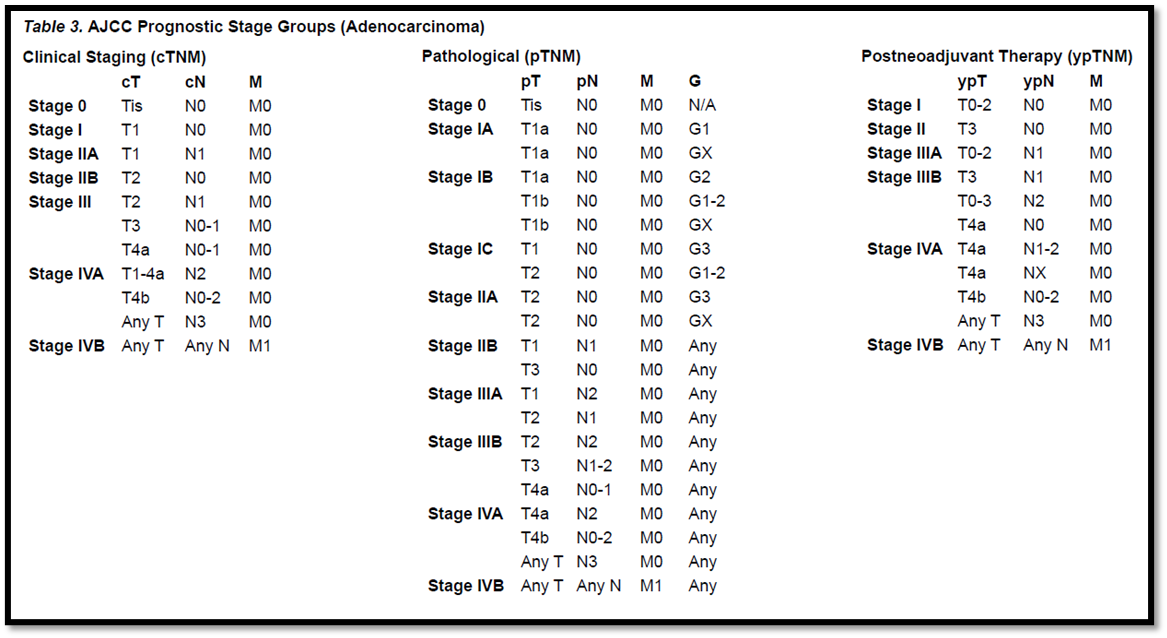
5F. Esophageal cancer should be staged according to the American Joint Committee on Cancer AJCC/UICC TNM (tumor/node/metastases) 8th edition staging system
|
Strong |
|
6.Nutrition |
|
|
6A. All patients with esophageal cancer should be screened regularly for malnutrition by evaluating nutritional intake, weight change and BMI, beginning with diagnosis and repeated depending on the stability of the clinical situation |
Strong |
|
6B. Patients at nutritional risk should be promptly referred for comprehensive nutritional assessment and support clinical nutrition services. |
Good practice statement. |
|
6C. We recommend that during radiotherapy an adequate nutritional intake should be ensured primarily by individualized nutritional counseling and/or with use of ONS, to avoid nutritional deterioration, maintain intake and avoid radiotherapy interruptions. |
Strong |
|
6D. In patients at nutritional risk, we recommend feeding jejunostomy in operable patients and percutaneous gastrostomy tubes for inoperable patients. |
Strong |
|
6E. We recommend that vitamins and minerals be supplied in amounts approximately equal to the recommended daily allowance and discourage the use of high-dose micronutrients in the absence of specific deficiencies. |
Strong |
|
6F. Parenteral nutrition is only recommended if adequate oral/EN is not possible or insufficient e.g. severe mucositis, intractable vomiting, ileus, severe malabsorption, protracted diarrhea or symptomatic gastrointestinal graft versus host disease. |
Strong |
|
6G. For all cancer patients undergoing either curative or palliative surgery we recommend management within an enhanced recovery after surgery (ERAS) program; within this program every patient should be screened for malnutrition and if deemed at risk, given additional nutritional support. |
Strong |
|
7. Early disease (cT1 N0 M0) |
|
|
7A. Multidisciplinary assessment and planning before any treatment is mandatory. |
Good clinical practice |
|
7B. We recommend endoscopic en bloc resection of lesions with intraepithelial high-grade dysplasia and most T1 tumors using either endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD). |
Conditional |
|
7C. Examination of the specimen provides accurate staging and endoscopic resection is considered definitive treatment, unless the deep resection margin is involved or there are significant risk factors for lymph node metastases (e.g. depth of invasion, lymph-vascular invasion, low differentiation grade, ulceration and large tumor size). |
Conditional |
|
7D. Patients with involved deep endoscopic resection margins or significant risk factors for lymph node metastases should be offered further respective surgery with appropriate lymphadenectomy. |
Conditional |
|
8. Locally advanced and resectable disease (cT2-T4 or cN1-3 M0) |
|
|
Squamous cell carcinoma |
|
|
8A. Locally advanced esophageal SCC should be treated with CRT followed by surgery, or definitive CRT with close surveillance and salvage surgery for local tumor persistence or progression (see 10D). |
Strong |
|
8B. For patients not willing to undergo esophageal surgery or who are medically unfit for major surgery, definitive CRT should be preferred as CRT is superior to RT alone. |
Strong |
|
8C. Definitive CRT is recommended for cervically localized tumors where surgery would entail a laryngectomy. |
Good clinical practice |
|
Adenocarcinoma |
|
|
8D. We recommend the use of perioperative chemotherapy or neoadjuvant CRT (see 10D). |
Strong |
|
9. Surgery |
|
|
9A. Esophageal surgery should be carried out in experienced centers only. |
Good clinical practice |
|
9B. We recommend Ivor Lewis procedure, (abdominal and right chest access is used, and reconstruction is carried out with a gastric tube conduit with esophagi-gastric anastomosis in the upper mediastinum) for esophagi-gastric tumors. |
Strong |
|
9C. We recommend McKeown procedure, (abdominal, right chest and cervical access is used with a similar reconstruction to the cervical esophagus) for esophageal tumors. |
Strong |
|
9D. We recommend transhiatal esophagectomy without transthoracic access with a similar reconstruction to the cervical esophagus in frail patients with distal tumors. |
Strong |
|
9E. The Siewert tumor type should be assessed in all patients with adenocarcinoma involving the EGJ. The surgical approach for Siewert type 1 and type 2 should be similar to those described in esophageal cancer. Also, Siewert type III tumors should be considered gastric cancer and surgical approach for these tumors should be similar to those described in gastric cancer. |
Good clinical practice.
|
|
10. Chemoradiotherapy |
|
|
10A. The recommended traditional standard regimen for definitive CRT is four cycles of cisplatin 5-FU (or capecitabine) combined with RT to a dose of 50.4 Gy in 28 fractions (or 50 Gy in 25 fractions). |
Strong |
|
10B. Weekly carboplatin - paclitaxel, as used in the CROSS regimen, combined with RT as definitive treatment is also recommended. |
Strong |
|
10C. RT should be delivered using 3D conformal RT, but intensity modulated RT or volumetric arc therapy are preferred if available. |
Strong |
|
10D. We recommend against the use of RT dose >50.4 Gy in the definitive treatment of mid and distal esophageal cancer specially if salvage esophagectomy is considered as a therapeutic strategy. We recommend the use of dose up to 60 Gy in cervical esophageal cancer. |
Strong |
|
11. Preoperative chemotherapy in adenocarcinoma of the esophagus and GEJ |
|
|
11A. In patients with c T2, N0(with high-risk lesions: LVI≥ 3cm, poorly differentiated) or cT1b-cT2N+ or cT3-cT4a, any N who are scheduled to receive surgery as the primary treatment, pre-operative chemotherapy regimens are recommended. |
Strong |
|
11B. FLOT regimen (4 cycles before and after surgery) is the preferred perioperative chemotherapy regimen for patients with good performance status. Cisplatin and 5-fluorouracil (CF) or oxaliplatin-based doublets FOLFOX or CAPOX are also valid options. |
Strong |
|
12. Adjuvant chemotherapy in adenocarcinoma of the esophagus and GEJ (who have not received preoperative chemotherapy) |
|
|
12A. In patients operated without neoadjuvant treatment, postoperative CT is recommended, particularly in case of R1 resection, N+ lesion, or PT3, T4. |
Strong |
|
12B. Postoperative chemotherapy with capecitabine and oxaliplatin is an option in patients with resectable esophageal or GEJ cancers who had not received preoperative therapy. FOLFOX regimen is also a valid option. |
Strong |
|
13. First- line systemic therapy for unresectable, metastatic, recurrent adenocarcinoma of the esophagus and GEJ. |
|
|
13A. Trastuzumab should be added to first-line chemotherapy for patients with advanced HER2 overexpression-positive adenocarcinoma (combination with a fluoropyrimidine and a platinum agent is preferred). |
Strong |
|
13B. The preferred regimens for HER2-negative disease also include a fluoropyrimidine (fluorouracil or capecitabine) combined with either oxaliplatin or cisplatin. |
Strong |
|
13C.We recommend FOLFOX for elderly or frail patients due to lower toxicity. |
Strong |
|
14. Second line and subsequent systemic therapy for unresectable, metastatic, recurrent adenocarcinoma of esophagus and GEJ |
|
|
14A. Single-agent docetaxel, paclitaxel, and irinotecan are preferred options for second-line subsequent therapy. |
Strong |
|
14B. FOLFIRI is a preferred treatment option that can be safely used in the second-line setting if it was not previously used in first-line therapy. |
Strong |
|
15. First line systemic therapy for unresectable, metastatic, recurrent esophageal and GEJ squamous cell carcinoma |
|
|
15A. Standard first-line Chemotherapy for advanced untreated esophageal SCC is a platinum-Fluoropyrimidine doublet chemotherapy. |
Strong |
|
15B. For patients with advanced esophageal SCC, who are unfit for full- dose chemotherapy due to advanced age or frailty, dose-reduced oxaliplatin/capecitabine is an alternative option. |
Strong |
|
16. Second line and subsequent systemic therapy for unresectable, metastatic and recurrent SCC |
|
|
16A.Taxanes (paclitaxel or docetaxel) or irinotecan monotherapies are recommended as further-line treatment options. |
Strong |
- Recommendations
1.Primary preventions
The following intervention may help to reduce the risk of esophageal cancer:
· Treating gastroesophageal reflux disease (GERD) and Barrett's esophagus early
· Prevention of injury to the esophagus
· Avoidance of tobacco and alcohol
· Avoidance of meat, processed food intake, hot beverages.
· Diet rich in fruits and vegetables
· Avoid obesity
2.Secondary prevention (Screening)
• Screening of esophageal and GEJ tumors in the general population is not cost effective and should not be done.
Strong recommendation, high grade evidence (1).
3.Diagnosis
3A. All patients with new dysphagia, gastrointestinal bleeding, recurrent aspiration or emesis,
weight loss and/or loss of appetite should undergo an upper gastrointestinal endoscopy.
Strong recommendation, moderate grade evidence (2).
3B. The location of the tumor relative to the lower incisors and GEJ, the length of the tumor, the
extent of circumferential involvement, the presence of Barrett esophagus and the degree of
obstruction should be carefully recorded to assist with treatment planning.
Strong recommendation, moderate grade evidence (3)
3C. Multiple biopsies, six to eight, using standard size endoscopy forceps should be performed to
provide sufficient material for histologic and molecular interpretation. Larger forceps is
recommended during surveillance endoscopy of Barrett esophagus for the detection of
dysplasia.
Strong recommendation, moderate grade evidence (4)
3D. Diagnosis should be based on endoscopic biopsies (Chromo-endoscopy if available) with the
histological tumor type classified according to the World Health Organization (WHO) criteria.
The differentiation between esophageal SCC and AC is of prognostic and therapeutic relevance.
Strong recommendation, high grade evidence (5)
3E. Laparoscopy + washings could be done to exclude occult metastatic disease involving
peritoneum/diaphragm, especially in locally advanced (T3/T4) adenocarcinoma of the GEJ
infiltrating the anatomical cardia.
Good practice statement
4.Pathology
4A. Histological diagnosis should be reported according to the WHO criteria.
4B. Immuno-histochemical staining including HER2 is recommended in poorly differentiated and
undifferentiated cancers when differentiation between SCC and AC using morphological
characteristics is not possible.
Good practice statement.
5.Staging and risk assessment
5A. Consider Multidisciplinary team meetings (MDTs) for patients with esophageal cancer.
MDTs often include surgeons, radiologist, pathologists, medical oncologists, radiation
oncologists, gastroenterologists, dietitians, rehabilitation physicians, palliative care specialists
and dedicated cancer nurse specialists.
Conditional recommendation, moderate grade evidence (6).
5B. Staging should include a complete clinical examination, Complete blood count (CBC) and
comprehensive chemistry profile, endoscopy, chest /abdomen /pelvis CT with oral and IV
contrast.
Strong recommendation, high grade evidence (7).
5C. Consider 18F-fluoro-2-deoxy-D-glucose (FDG)-positron emission tomography (PET)
in patients who are candidates for esophagectomy.
Conditional recommendation, moderate grade evidence (8).
5D. Endoscopic ultrasound (EUS) is recommended in early lesions in order to assess tumor depth and
lymph node status in patients amenable to upfront surgery or candidates for tri-modality
treatment (T3N0, T1-4a and any locoregional N). If not available refer to a specialized center.
Strong recommendation, high grade evidence (9).
5E. We recommend bronchoscopy for tumors located at or above the carina in the initial staging,
which can help in both surgery and radiotherapy treatments.
Strong recommendation, moderate grade evidence (10).
5F. Esophageal cancer should be staged according to the American Joint Committee on Cancer
AJCC/UICC TNM (tumor/node/metastases) 8th edition staging system
Strong recommendation, high grade evidence (11).
6.Nutrition
6A. All patients with esophageal cancer should be screened regularly for malnutrition by evaluating
nutritional intake, weight change and BMI, beginning with diagnosis and repeated depending on the
stability of the clinical situation.
Strong recommendation, moderate grade evidence (12)
6B. Patients at nutritional risk should be promptly referred for comprehensive nutritional assessment and
support clinical nutrition services.
Good practice statement.
6C. We recommend that during radiotherapy an adequate nutritional intake should be ensured primarily
by individualized nutritional counseling and/or with use of ONS, to avoid nutritional deterioration,
maintain intake and avoid radiotherapy interruptions.
Strong recommendation, high grade evidence (13-14).
6D. In patients at nutritional risk, we recommend feeding jejunostomy in operable patients and
percutaneous gastrostomy tubes for inoperable patients..
Strong recommendation, moderate grade evidence (15-16).
6E. We recommend that vitamins and minerals be supplied in amounts approximately equal to the
recommended daily allowance and discourage the use of high-dose micronutrients in the absence
of specific deficiencies.
Strong recommendation, high grade evidence (17-18).
6F. Parentral nutrition is only recommended if adequate oral/EN is not possible or insufficient e.g.
severe mucositis, intractable vomiting, ileus, severe malabsorption, protracted diarrhea or
symptomatic gastrointestinal graft versus host disease.
Strong recommendation, moderate grade evidence (19-20).
6G. For all cancer patients undergoing either curative or palliative surgery we recommend
management within an enhanced recovery after surgery (ERAS) program; within this program
every patient should be screened for malnutrition and if deemed at risk, given additional
nutritional support
Strong recommendation, high Grade evidence (21).
7. Early disease (cT1 N0 M0)
7A. Multidisciplinary assessment and planning before any treatment is mandatory.
Good clinical practice
7B. We recommend endoscopic en bloc resection of lesions with intraepithelial high-grade dysplasia and most T1 tumors using either endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD).
Conditional recommendation, low grade evidence (22,23).
7C. Examination of the specimen provides accurate staging and endoscopic resection is considered definitive treatment, unless the deep resection margin is involved or there are significant risk factors for lymph node metastases (e.g. depth of invasion, lymph-vascular invasion, low differentiation grade, ulceration and large tumor size).
Conditional recommendation, Low quality evidence (22-25).
7D. Patients with involved deep endoscopic resection margins or significant risk factors for lymph node metastases should be offered further respective surgery with appropriate lymphadenectomy.
Conditional recommendation low grade evidence (25).
8. Locally advanced and resectable disease (cT2-T4 or cN1-3 M0)
Squamous cell carcinoma
8A. Locally advanced esophageal SCC should be treated with CRT (see 10D) followed by surgery, or definitive CRT with close surveillance and salvage surgery for local tumor persistence or progression.
Strong recommendation, high grade evidence (26,27).
8B. For patients not willing to undergo esophageal surgery or who are medically unfit for major surgery, definitive CRT should be preferred as CRT is superior to RT alone.
Strong recommendation, high grade evidence (28)
8C. Definitive CRT is recommended for cervically localized tumors where surgery would entail a laryngectomy
Adenocarcinoma
8D. We recommend the use of perioperative chemotherapy or neoadjuvant CRT (see 10D).
Strong recommendation, high grade evidence (29)
9. Surgery
9A. Esophageal surgery should be carried out in experienced centers only.
9B. We recommend Ivor Lewis procedure, (abdominal and right chest access is used, and reconstruction is carried out with a gastric tube conduit with esophagi-gastric anastomosis in the upper mediastinum) for esophagi-gastric tumors.
Strong recommendation, low grade evidence (30).
9C. We recommend McKeown procedure, (abdominal, right chest and cervical access is used with a similar reconstruction to the cervical esophagus) for esophageal tumors.
Strong recommendation, low grade evidence (30).
9D. We recommend transhiatal esophagectomy without transthoracic access with a similar reconstruction to the cervical esophagus in frail patients with distal tumors.
Strong recommendation, low grade evidence (30).
9E. The Siewert tumor type should be assessed in all patients with adenocarcinoma involving the EGJ.
The surgical approach for Siewert type 1 and type 2 should be similar to those described in esophageal cancer. Also, Siewert type III tumors should be considered gastric cancer and surgical approach for these tumors should be similar to those described in gastric cancer.
Good clinical practice.
10. Chemoradiotherapy
10A. The recommended traditional standard regimen for definitive CRT is four cycles of cisplatin5-FU (or capecitabine) combined with RT to a dose of 50.4 Gy in 28 fractions (or 50 Gy in 25 fractions).
Strong recommendation, high grade evidence (31)
10B. Weekly carboplatin - paclitaxel, as used in the CROSS regimen, combined with RT as definitive treatment is also recommended
Strong recommendation, high grade evidence (32)
10C. RT should be delivered using 3D conformal RT, but intensity modulated RT or volumetric arc therapy are preferred if available.
Strong recommendation, moderate grade evidence (33)
10D. We recommend against the use of RT dose >50.4 Gy in the definitive treatment of mid and distal
esophageal cancer specially if salvage esophagectomy is considered as a therapeutic strategy.
We recommend the use of dose up to 60 Gy in cervical esophageal cancer.
Strong recommendation, high grade evidence (34,35).
11. Preoperative chemotherapy in adenocarcinoma of the esophagus and GEJ
11A. In patients with c T2, N0(with high-risk lesions: LVI≥ 3cm, poorly differentiated) or
cT1b-cT2N+ or cT3-cT4a, any N who are scheduled to receive surgery as the primary
treatment, pre-operative chemotherapy regimens are recommended.
Strong recommendation, high grade evidence (36).
11B. FLOT regimen (4 cycles before and after surgery) is the preferred perioperative
chemotherapy regimen for patients with good performance status. Cisplatin and 5-fluorouracil (CF) or oxaliplatin-based doublets FOLFOX or CAPOX are also valid options.
Strong recommendation, high grade evidence (37,38)
12. Adjuvant chemotherapy in adenocarcinoma of the esophagus and GEJ (who have not
received preoperative chemotherapy)
12A. In patients operated without neoadjuvant treatment, postoperative CT is recommended,
particularly in case of R1 resection, N+ lesion, or PT3, T4.
Strong recommendation, high grade evidence (39).
12B. Postoperative chemotherapy with capecitabine and oxaliplatin is an option in patients with
resectable esophageal or GEJ cancers who had not received preoperative therapy. FOLFOX regimen is also a valid option.
Strong recommendation, high grade evidence (40).
13. First- line systemic therapy for unresectable, metastatic, recurrent adenocarcinoma of the esophagus and GEJ.
13A. Trastuzumab should be added to first-line chemotherapy for patients with advanced
HER2 overexpression-positive adenocarcinoma (combination with a fluoropyrimidine
and a platinum agent is preferred).
Strong recommendation, high grade evidence (41).
13B. The preferred regimens for HER2-negative disease also include a fluoropyrimidine
(Fluorouracil or capecitabine) combined with either oxaliplatin or cisplatin
Strong recommendation, high grade evidence (42).
13C.We recommend FOLFOX for elderly or frail patients due to lower toxicity.
Strong recommendation, high grade evidence (42,43).
14. Second line and subsequent systemic therapy for unresectable, metastatic, recurrent adenocarcinoma of esophagus and GEJ
14A. Single-agent docetaxel, paclitaxel, and irinotecan are preferred options for second-line
subsequent therapy
Strong recommendation, high grade evidence (43,44).
14B. FOLFIRI is a preferred treatment option that can be safely used in the second-line setting
if it was not previously used in first-line therapy.
Strong recommendation, moderate grade evidence (45).
15. First line systemic therapy for unresectable, metastatic, recurrent esophageal and GEJ squamous cell carcinoma
15A. Standard first-line Chemotherapy for advanced untreated esophageal SCC is a
platinum-Fluoropyrimidine doublet chemotherapy.
Strong recommendation, moderate grade evidence (46).
15B. For patients with advanced esophageal SCC, who are unfit for full- dose
chemotherapy due to advanced age or frailty, dose-reduced oxaliplatin/capecitabine is
an alternative option.
Strong recommendation, high grade evidence (47).
16. Second line and subsequent systemic therapy for unresectable, metastatic and
recurrent SCC
Taxanes (paclitaxel or docetaxel) or irinotecan monotherapies are recommended as
further-line treatment options
Strong recommendation, moderate grade evidence (48,49).
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines, for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines: Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Yousri Wasef & Yousri Rostom.
· Chair of the GIT Cancer Scientific Committee: Prof Yousri Rostom
- The Esophageal and Esophagogastric Junction Cancer Scientific Group Members: (Alphabitical order): Ahmed EL Kassed, Doaa Gennena, Fouad Aboutaleb, Heba Fadlol, Hussein Okasha, Khaled Abdelkarim, Manal EL Mahdy, Mary Gamal, Mohammed Eid, Omar S. Omar, and Yousri Wasef.
- Abbreviations
AC adenocarcinoma
BE Barret’s esophagus
BMI body mass index
C clinical
CHT chemotherapy
CT Computed tomography
EMR Endoscopic Mucosal Resection
EN enteral nutrition
ERAS Enhanced recovery after surgery
ESD Endoscopic submucosal dissection
ESGE European Society of Gastrointestinal Endoscopy
EUS Endoscopic Ultrasound
FNA/FNB Fine needle aspiration/biopsy
GEJ Esophagogastric Junction
GERD Gastroesophageal reflux disease
GI gastrointestinal
MRI Magnetic Resonance imaging
ONS oral nutritional supplements
PEG percutaneous endoscopic gastrostomies
PET/CT Positron Emission tomography
PN parenteral nutrition
RHT radiotherapy
SCC. squamous cell carcinoma
- Introduction
Esophageal cancer is a familiar malignancy with high incidence and mortality, and the overall prognosis is poor. esophageal cancer has become an urgent global health challenge and the growing trend of esophageal cancer cases is expected to continue for the next two decades and beyond. In Egypt, there were an estimated 1543 new cases of esophageal cancer and 1510 deaths occurred because of this disease based on GLOBOCAN 2022.
- Purpose and scope
These guidelines are developed to improve the quality of care for Esophageal and GJ cancer via providing a uniform standard of care across the country to help in primary prevention, screening, early diagnosis, treatment and follow up for esophageal and GEJ cancer so more optimal treatment options and improved clinical outcomes.
- Target audience
Clinicians who are involved in the care and treatment of patients with esophageal and GEJ cancer, including medical oncologists, radiation oncologists, clinical oncologist, gastroenterologists, surgeons, clinical dietrition interventional radiologists, radiologists, pathologists, and palliative care specialists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained) The NCCN, ESMO, NICE guidelines are the main sources used while formulating the national guidelines for bladder cancer (1-3).
Ø Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group: https://www.gradeworkinggroup.org/
. GRADE online training modules: http://cebgrade.mcmaster.ca/
. GRADE profile software: http://ims.cochrane.org/revman/gradepro
Table 1: Quality of evidence in GRADE
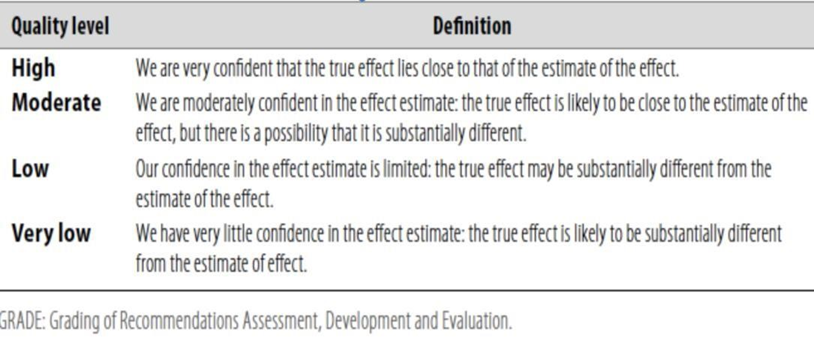
Table 2: Significance of the four levels of evidence

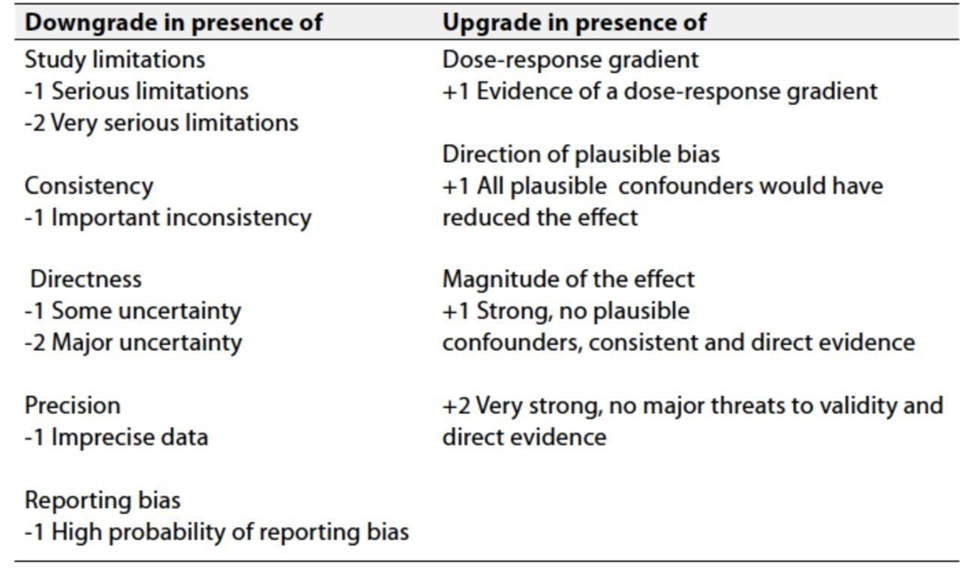
Table 3: Factors that determine How to upgrade or downgrade the quality of evidence
Ø The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Clinical indicators for monitoring
For patients newly diagnosed with esophageal & EGJ cancer:
- laboratory evaluation (CBC, LFT, and KFT)
- imaging
- endoscopic biopsy for pathological confirmation &IHC
- Research gaps
§ Systematic inclusion of cost-benefit analyses in clinical trial with collection of health economic analysis such as incremental cost effectiveness ratio in order to facilitate clinical decision-making.
§ Predictive biomarkers: response to specific systemic targeted therapies and immunotherapy.
§ Improve models for pre-clinical testing of novel drugs.
§ Search for tools to assess quality of life and in clinical trials.
§ Dietary supplements, nutritional counselling, physical activity recommendations and psychological support as part of an integrative healthcare approach to care for people with esophageal cancer.
- Update of this guideline
§ This guideline will be updated whenever there is new evidence.
- References
1. Liam Zakko, Lori Lutzke, Kenneth K Wang, Screening and Preventive Strategies in Esophagogastric Cancer: Surg Oncol Clin N Am. 2017 Apr;26(2):163–178.
2. Rustgi AK, El-Serag HB. Esophageal carcinoma. N Engl J Med. 2014;371(26):2499-2509.
3. Weusten Bas LAM et al. Diagnosis and management Barrett esophagus: European . Society of Gastrointestinal Endoscopy (ESGE) Guideline Endoscopy 2023; 55: 1124–1146
4. Graham DY Schwartz JT, Cain GD, Gyorkey F. Prospective evaluation of biopsy number in the diagnosis of esophageal and gastric carcinoma. Gastroenterology 1982; 82:228-231.
5. Nagtegaal ID, Odze RD, Klimstra D, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76(2):182-188.
6. Mats Lindblad, Christine Jestin, Jan Johansson, David Edholm, Gustav Linder, Multidisciplinary team meetings improve survival in patients with esophageal cancer, Diseases of the Esophagus, 2024;,doae061.
7. van Vliet EP, Heijenbrok-Kal MH, Hunink MG et al. Staging investiga- tions for oesophageal cancer: ameta-analysis. Br J Cancer 2008; 98(3):547–557.
8. Flamen P, Lerut A, Van Cutsem E, De Wever W, Bormans G, Hiele M, De Leyn P, Van Raemdonck D,et al.Utility of positron emission tomography for the staging of patients with potentially operable esophageal carcinoma. J Clin Oncol. 2000 Sep 15;18(18):3202-10. doi: 10.1200/JCO.2000.18.18.3202. PMID: 10986052.
9. Li X, Wang Y, Kong M, Lin J. Systematic review and meta-analysis of endoscopic ultrasonography in staging diagnosis of esophageal cancer after neoadjuvant radiotherapy and chemotherapy. J Gastrointest Oncol. 2022 Aug;13(4):1525-1540. doi: 10.21037/jgo-22-437. PMID: 36092358; PMCID:PMC9459206.
10. Riedel M, Stein HJ, and Mounyam L, et al (2000) Bronchoscopy in the preoperative staging of esophageal cancer below the tracheal bifurcation: a prospective study Eur Respir J 16(1) 134–139
11. Rice TW, Ishwaran H, Blackstone EH, et al. Recommendations for clinical staging (cTNM) of cancer of the esophagus and esophagogastric junction for the 8th edition AJCC/UICC staging manuals. Dis Esophagus.2016;29(8):913-919.
12. Pan H, Cai S, Ji J, Jiang Z, Liang H, Lin F, et al. The impact of nutritional status, nutritional risk, and nutritional treatment on clinical outcome of 2248 hospitalized cancer patients: a multi-center, prospective cohort study in Chinese teaching hospitals. Nutr Cancer 2013;65:62e70.
13. Ravasco P, Monteiro-Grillo I, Vidal PM, Camilo ME. Dietary counseling improves patient outcomes: a prospective, randomized, controlled trial in colorectal cancer patients undergoing radiotherapy. J Clin Oncol 2005;23: 1431e8.
14. Langius JA, Zandbergen MC, Eerenstein SE, van Tulder MW, Leemans CR, Kramer MH, et al. Effect of nutrition therapy on nutritional status, quality of life and mortality in patients with head and neck cancer receiving (chemo) radiotherapy: a systematic review. Clin Nutr 2013;32:671e8.
15. Odelli C, Burgess D, Bateman L, Hughes A, Ackland S, Gillies J, et al. Nutrition support improves patient outcomes, treatment tolerance and admission characteristics in oesophageal cancer. Clin Oncol R Coll Radiol 2005;17: 639e45.
16.Marcy PY, Magne N, Bensadoun RJ, Bleuse A, Falewee MN, Viot M, et al. Systematic percutaneous fluoroscopic gastrostomy for concomitant radio chemotherapy of advanced head and neck cancer: optimization of therapy. Support Care Cancer 2000;8:410e3.
17.Rock CL, Doyle C, Demark-Wahnefried W, Meyerhardt J, Courneya KS, Schwartz AL, et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J Clin 2012; 62:243e74
18.Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C. Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: systematic review and meta-analysis. JAMA 2007;297:842e57
19.Bozzetti F, Santarpia L, Pironi L, Thul P, Klek S, Gavazzi C, et al. The prognosis of incurable cachectic cancer patients on home parenteral nutrition: a multicenter observational study with prospective follow-up of 414 patients. Ann Oncol 2014;25:487e93.
20.Scolapio JS, Ukleja A, Burnes JU, Kelly DG. Outcome of patients with radiation enteritis treated with home parenteral nutrition. Am J Gastroenterol 2002;97:662e6.
21.Mortensen K, Nilsson M, Slim K, Schafer M, Mariette C, Braga M, et al., Enhanced Recovery After Surgery (ERAS®) Group. Consensus guidelines for enhanced recovery after gastrectomy: enhanced recovery after surgery (ERAS®) society recommendations. Br J Surg 2014;101:1209e29
22. di Pietro M, Canto MI, Fitzgerald RC. Endoscopic management of early adenocarcinoma and squamous cell carcinoma of the esophagus: screening, diagnosis, and therapy. Gastroenterology. 2018;154(2):421-436.
23. Weusten B, Bisschops R, Coron E, et al. Endoscopic management of Barrett’s esophagus: European Society of Gastrointestinal Endoscopy (ESGE) Position Statement. Endoscopy. 2017;49(2):191-198.
24.. Pimentel-Nunes P, Dinis-Ribeiro M, Ponchon T, et al. Endoscopic submucosal dissection: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2015;47(9):829-854.
25. Yamashina T, Ishihara R, Nagai K, et al. Long-term outcome and metastatic risk after endoscopic resection of superficial esophageal squamous cell carcinoma. Am J Gastroenterol. 2013;108(4):544-551.
26. van Hagen P, Hulshof MCCM, van Lanschot JJB, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366(22):2074-2084.
27.Vincent J, Mariette C, Pezet D, et al. Early surgery for failure after chemoradiation in operable thoracic oesophageal cancer. Analysis of the non-randomised patients in FFCD 9102 phase III trial: chemoradiation followed by surgery versus chemoradiation alone. Eur J Cancer. 2015;51(13):1683-1693.
28. Jiaxin Li, Yibin Jia, Yufeng Cheng, Jianbo Wang, Chemoradiotherapy vs radiotherapy for nonoperative early stage esophageal cancer: A seer data analysis,Cancer Med. 2020 May 22;9(14):5025–5034.
29. Xiaozheng Kang, Jianjun Qin, Ruixiang Zhang, et al, 2021 NCC/CATS/CSTCVS/STM expert consensus on perioperative immunotherapy for esophageal cancer,AOE, Vol 4 (September 25, 2021)
30. Hulscher JBF, van Sandick JW, de Boer AGEM, et al. Extended transthoracic resection comparedwith limited transhiatal resection for adenocarcinoma of the esophagus. N Engl JMed. 2002;347(21):1662-1669.
31. Herskovic A, Martz K, al-Sarraf M, et al. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326(24):1593-1598.
32. van Hagen P, Hulshof MC, van Lanschot JJ, et al., CROSS Group. Preoperative chemoradiotherapy for esophageal or junctional cancer.N Engl J Med. 2012 May 31;366(22):2074-84
33. Minsky BD, Pajak TF, Ginsberg RJ, et al. INT 0123 (Radiation Therapy Oncology Group 94-05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol. 2002;20(5):1167-1174.
34. Hulshof M, Geijsen ED, Rozema T, et al. Randomized study on dose escalation in definitive chemoradiation for patients with locally advanced esophageal cancer (ARTDECOStudy). J ClinOncol. 2021;39(25):2816-2824.
35. de Vos-Geelen J, Hoebers FJP, Geurts SME, et al. A national study to assess outcomes of definitive chemoradiation regimens in proximal esophageal cancer. Acta Oncol. 2020;59(8):895-903.
36.Csontos A, Fazekas A, Szakó L, Farkas N, Papp C, Ferenczi S, Bellyei S, Hegyi P, Papp A. Effects of neoadjuvant chemotherapy vs chemoradiotherapy in the treatment of esophageal adenocarcinoma: A systematic review and meta-analysis.World J Gastroenterol 2024; 30(11): 1621-1635.
37.Al-Batran SE, Homann N, Pauligk C, et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): a randomised, phase 2/3 trial. Lancet 2019;393:1948-195
38.Conroy T, Galais MP, Raoul JL, et al. Definitive chemoradiotherapy with FOLFOX versus fluorouracil and cisplatin in patients with oesophageal cancer (PRODIGE5/ACCORD17): final results of a randomised, phase 2/3 trial. Lancet Oncol. 2014;15(3):305-314.
39.Ychou M, Boige V, Pignon JP, Conroy T, Bouché O, Lebreton G, Ducourtieux M, Bedenne L, Fabre JM, Saint-Aubert B, Genève J,Lasser P, Rougier P. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial. J Clin Oncol. 2011 May 1;29(13):1715-21.
40. Paolet X, Oba K, Burzykowski T, et al; GASTRIC (Global Advanced/ Adjuvant Stomach Tumor Research International Collaboration) Group. Benefit of adjuvant chemotherapy for resectable gastric cancer: a meta-analysis. JAMA. 2010;303:1729-1737.
41.Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, et al; ToGA Trial Investigators. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer(ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010 Aug 28;376(9742):687-97.
42.Al-Batran SE, Hartmann JT, Probst S, Schmalenberg H, Hollerbach S, et al; Arbeitsgemeinschaft Internistische Onkologie. Phase III trial in metastatic gastroesophageal adenocarcinoma with fluorouracil, leucovorin plus either oxaliplatin or cisplatin: a study of the Arbeitsgemeinschaft Internistische Onkologie. J Clin Oncol. 2008 Mar 20;26(9):1435-42.
43.Montagnani F, Turrisi G, Marinozzi C, Aliberti C, Fiorentini G. Effectiveness and safety of oxaliplatin compared to cisplatin for advanced,unresectable gastric cancer: a systematic review and meta-analysis. Gastric Cancer. 2011 Mar;14(1):50-5.
43.Ford HE, Marshall A, Bridgewater JA, Janowitz T, Coxon FY, Wadsley J, Mansoor W, et al; COUGAR-02 Investigators. Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): an open-label, phase 3 randomised controlled trial. Lancet Oncol. 2014 Jan;15(1):78-86.
44.Hironaka S, Ueda S, Yasui H, Nishina T, Tsuda M, Tsumura ,et al. Randomized, open-label, phase III study comparing irinotecan with paclitaxel in patients with advanced gastric cancer without severe peritoneal metastasis after failure of prior combination chemotherapy using fluoropyrimidine plus platinum: WJOG 4007 trial. J Clin Oncol. 2013 Dec 10;31(35):4438-44.
45.Sym SJ, Hong J, Park J, Cho EK, Lee JH, et al. A randomized phase II study of biweekly irinotecan monotherapy or a combination of irinotecan plus 5-fluorouracil/leucovorin (mFOLFIRI) in patients with metastatic gastric adenocarcinoma refractory to or progressive after first-line chemotherapy. Cancer Chemother Pharmacol. 2013 Feb;71(2):481-8.
46.Hayashi K, Ando N, Watanabe H, Ide H, Nagai K, Aoyama N, et al. Phase II evaluation of protracted infusion of cisplatin and 5-fluorouracil in advanced squamous cell carcinoma of the esophagus: a Japan Esophageal Oncology Group (JEOG) Trial (JCOG9407). Jpn J Clin Oncol. 2001 Sep;31(9):419-23.
47.Hall PS, Swinson D, Cairns DA, Waters JS, Petty R, Allmark C, et al; GO2 Trial Investigators. Efficacy of Reduced-Intensity Chemotherapy With Oxaliplatin and Capecitabine on Quality of Life and Cancer Control Among Older and Frail Patients With Advanced Gastroesophageal Cancer: The GO2 Phase 3 Randomized Clinical Trial. JAMA Oncol. 2021 Jun 1;7(6):869-877.
48.Kato K, Tahara M, Hironaka S, Muro K, Takiuchi H, Hamamoto Y, Imamoto H, Amano N, Seriu T. A phase II study of paclitaxel by weekly 1-h infusion for advanced or recurrent esophageal cancer in patients who had previously received platinum-based chemotherapy. Cancer Chemother Pharmacol. 2011 Jun;67(6):1265-72.
49.Burkart C, Bokemeyer C, Klump B, Pereira P, Teichmann R, Hartmann JT. A phase II trial of weekly irinotecan in cisplatin-refractory esophageal cancer. Anticancer Res. 2007 Jul-Aug;27(4C):2845-8.
- Annexes
AJCC Cancer Staging Manual, Eighth Edition (2017)