Pancreatic Cancer
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Pancreatic Cancer |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 20 January 2025" Download Guideline
- Executive Summary
This guidance provides a data-supported approach to diagnosis, staging, treatment and follow up of patients diagnosed with pancreatic Cancer. This Guideline is intended only for pancreatic adenocarcinoma.
|
Recommendation |
Strength of recommendation |
|
Diagnosis |
|
|
· Labs |
|
|
· Routine labs including LFTs, KFTs, and CBC should be included in the primary diagnosis of pancreatic cancer. |
Good practice statement |
|
· CA 19-9 can be used as a serum marker to measure disease burden and potentially guide treatment decisions. |
Good practice statement. |
|
· Cytology in localized pancreatic lesion, preferably by EUS guidance or biopsy from metastatic site “preferred” should be obtained before initiation of chemotherapy. |
Strong |
|
· Imaging |
|
|
· Multiphasic contrast-enhanced thoracic-abdominal and pelvic CT, including late arterial phase and portal venous phase, should be used as the first-line imaging modality for suspected PC. |
Strong |
|
Strong |
|
|
·Imaging should be carried out in the 4 weeks before starting treatment. |
Strong |
|
· Abdominal MRI may be used when CT cannot be carried out, or inconclusive or for pancreatic cystic lesions. |
Conditional |
|
· We do not recommend PET/CT for diagnosis of primary tumors but may be useful for staging localized tumors and in cases where the presence of distant metastases is uncertain (e.g. Doubtful imaging or high CA 19-9). |
Conditional |
|
· Hepatic MRI is recommended before surgery to confirm the absence of small liver metastases |
Strong |
|
Pathology and immunophenotyping |
|
|
· CA19-9 (or CK19 according to availability), Chromogranin (or synaptophysin according to availability) are recommended for pathologic diagnosis. |
Conditional |
|
Staging and Risk assessment |
|
|
· MDT discussion in expert centers is required to define a recommended treatment strategy for patients with PC. |
Good clinical practice |
|
· Tumors should be staged according to the AJCC staging system |
Strong |
|
· We recommend assessing resectability by anatomical NCCN criteria. |
Strong |
|
· We prefer staging laparoscopy in patients who meet any of the followings: CA19.9 > 150U/ml, low volume ascites, tumor in the body or tail of pancreas, borderline resectable tumor (after neoadjuvant treatment), or tumor > 3 cm in size. |
Conditional |
|
Treatment of resectable PC |
|
|
· We suggest performing frozen section analysis of pancreatic neck transection and of common bile duct transection margins. |
Conditional |
|
· Tumour clearance should be defined for all margins identified by the surgeon |
Good clinical practice |
|
· For adenocarcinomas of the pancreas head and uncinate, a pancreatoduodenectomy (Whipple procedure) should be done. |
Strong |
|
· For patients with tumours in the body or tail, radical anterograde modular pancreatosplenectomy with dissection of the left hemi-circumference of the SMA to the left of the coeliac trunk is recommended. |
Strong |
|
· Standard lymphadenectomy is recommended and should involve the removal of >16 lymph nodes to allow adequate pathological staging of the disease. |
Strong |
|
· The total number of lymph nodes examined and lymph node ratio (number of involved lymph nodes as a proportion of the number of lymph nodes examined) should be reported in the pathological analysis. |
Strong |
|
· Patients undergoing surgery should receive perioperative thromboprophylaxis with either unfractionated heparin or low-molecular-weight heparin (LMWH), unless contraindicated. |
Strong |
|
· If the bilirubin level is >14 mg/l (250 mmol/l), endoscopic drainage is recommended for those planned to receive neoadjuvant treatment or those in whom surgery will be delayed for longer than 2 weeks. |
Strong |
|
· Neoadjuvant therapy is not recommended for resectable PC. |
Conditional |
|
· Following resection of PC, completion of 6 months of adjuvant Chemotherapy is strongly recommended. |
Strong |
|
· Adjuvant mFOLFIRINOX is recommended for patients with resected PC and ECOG PS 0-1. |
Strong |
|
· In patients who are not candidates for mFOLFIRINOX (age >75 years, ECOG PS 2 or contraindication to mFOLFIRINOX), we recommend gemcitabine-capecitabine as an alternative option. |
Strong |
|
· Adjuvant gemcitabine or 5-FU-LV should be limited to frail patients. |
Strong |
|
· Adjuvant CRT is not recommended and should not be given to patients following surgery. |
Strong |
|
Treatment of borderline resectable tumors (BRPC) |
|
|
· Patients with BRPC have a high probability of an R1 resection and should be considered for induction treatment. |
Strong |
|
Strong |
|
|
· Gemcitabine combined with oxaliplatin or capecitabine may be considered, when FOLFIRINOX is not feasible. |
Strong |
|
· CRT with capecitabine may be considered after induction Chemotherapy. |
Conditional |
|
· Following induction therapy, medically fit patients without disease progression and with a decrease in CA 19-9 should undergo surgical exploration, unless contraindicated. |
Strong |
|
Treatment of locally advanced pancreatic cancer (LAPC) |
|
|
Strong |
|
|
· Arterial resection after induction therapy is not recommended but can be considered as a possibility in experienced centers on a case-by-case basis in selected patients according to MDT recommendations. |
Conditional |
|
Treatment of advanced pancreatic cancer |
|
|
First-line treatment |
|
|
· Options to treat patients with metastatic PC should be dependent on PS: o In patients with ECOG PS 0-1 and bilirubin level <1.5 times the ULN, the regimen FOLFIRINOX should be considered. Strong recommendation, high grade evidence (34) o For patients with ECOG PS 2, Karnofsky PS (KPS) >70 and bilirubin level <1.5 times the ULN, gemcitabine-cisplatin can be considered. Strong recommendation, high grade evidence (34). o For patients with ECOG PS 2, KPS <70 and/or bilirubin level >1.5 times the ULN, gemcitabine monotherapy should be considered. Strong recommendation, high grade evidence (34). o For patients with ECOG PS 3-4, symptom-directed and palliative care should be considered Strong recommendation, high grade evidence (34). |
Strong |
|
· The efficacy of treatment should be typically evaluated every 8-12 weeks and should be based on clinical status, CA 19-9 trajectory and imaging. |
Strong |
|
Second-line treatment |
|
|
· After FOLFIRINOX treatment, gemcitabine alone may be offered to patients with ECOG PS 0-1 and a favorable comorbidity profile. |
Conditional |
|
· Oxaliplatin-based second-line treatment (mFOLFOX6 or OFF) may be considered as an alternative in patients with ECOG PS 0-2 if not given previously. |
Conditional |
|
· For patients with ECOG PS 3-4, symptom directed, and palliative care is recommended. |
Strong |
|
· Maintenance therapy with capecitabine (after discussion with patient) may be indicated till disease progression or unacceptable toxicity on a case- by case basis according to MDT recommendations. |
Conditional |
- Recommendations
➡️ Diagnosis
▪️ Labs
▪️ Routine labs including LFTs, KFTs, and CBC should be included in the primary diagnosis of pancreatic cancer.
▪️CA 19-9 can be used as a serum marker to measure disease burden and potentially guide treatment decisions in patients with normal bilirubin.
Good practice statement.
▪️ Cytology in localized pancreatic lesion, preferably by EUS guidance or biopsy from metastatic site “preferred” should be obtained before initiation of chemotherapy.
Strong recommendation, low grade evidence (1).
➡️ Imaging
▪️Multiphasic contrast-enhanced thoracic-abdominal and pelvic CT, including late arterial phase and portal venous phase, should be used as the first-line imaging modality for suspected PC “Pancreatic protocol”.
Strong recommendation, low grade evidence (2).
▪️ We recommend imaging before biliary drainage or stenting in case of jaundice due to an obstructive head PC.
Strong recommendation, very low grade evidence (3).
▪️ Imaging should be carried out in the 4 weeks before starting treatment.
Strong recommendation, low grade evidence (3).
▪️ Abdominal MRI may be used when CT cannot be carried out, or inconclusive or for pancreatic cystic lesions.
Conditional recommendation, very low grade evidence (4).
▪️ We do not recommend PET/CT for diagnosis of primary tumors but may be useful for staging localized tumors and in cases where the presence of distant metastases is uncertain (e.g. Doubtful imaging or high CA 19-9).
Conditional recommendation, low grade evidence (5).
▪️ Hepatic MRI is recommended before surgery to confirm the absence of small liver metastases
Strong recommendation, low grade evidence (4).
➡️Pathology and immunophenotyping
▪️ CA19-9 (or CK19 according to availability), Chromogranin (or synaptophysin according to availability) are recommended for pathologic diagnosis.
Conditional recommendation, low grade evidence (6).
Staging and Risk assessment
▪️MDT discussion in expert centers is required to define a recommended treatment strategy for patients with PC.
➡️Good clinical practice
▪️ Tumors should be staged according to the AJCC staging system.
Strong recommendation, low grade evidence (7).
▪️ We recommend assessing resectability by anatomical NCCN criteria
Strong recommendation, low grade evidence (8)
▪️ We prefer staging laparoscopy in patients who meet any of the followings:
CA19.9 > 150U/ml, low volume ascites, tumor in the body or tail of pancreas, borderline resectable tumor (after neoadjuvant treatment), or tumor > 3 cm in size.
Conditional recommendation, low grade evidence (9)
➡️Treatment of resectable PC
▪️We suggest performing frozen section analysis of pancreatic neck transection and of common bile duct transection margins.
Conditional recommendation, very low grade evidence (10)
▪️ Tumour clearance should be defined for all margins identified by the surgeon.
▪️ For adenocarcinomas of the pancreas head and uncinate, a pancreatoduodenectomy (Whipple procedure) should be done.
Strong recommendation, very low grade evidence (11)
▪️ For patients with tumours in the body or tail, radical anterograde modular pancreatosplenectomy with dissection of the left hemi-circumference of the SMA to the left of the coeliac trunk is recommended.
Strong recommendation, very low grade evidence (12, 13)
▪️ Standard lymphadenectomy is recommended and should involve the removal of >16 lymph nodes to allow adequate pathological staging of the disease.
Strong recommendation, very low grade evidence (14).
▪️ The total number of lymph nodes examined and lymph node ratio (number of involved lymph nodes as a proportion of the number of lymph nodes examined) should be reported in the pathological analysis.
Strong recommendation, very low grade evidence (14)
▪️ Patients undergoing surgery should receive perioperative thromboprophylaxis with either unfractionated heparin or low-molecular-weight heparin (LMWH), unless contraindicated.
Strong recommendation, high grade evidence (15, 16).
▪️ If the bilirubin level is >14 mg/l (250 mmol/l), endoscopic drainage is recommended for those planned to receive neoadjuvant treatment or those in whom surgery will be delayed for longer than 2 weeks.
Strong recommendation, high grade evidence (17).
▪️ Neoadjuvant therapy is not recommended for resectable PC.
Conditional recommendation, moderate grade evidence (18-20).
▪️ Following resection of PC, completion of 6 months of adjuvant Chemotherapy is strongly recommended.
Strong recommendation, high grade evidence (21-2
▪️ Adjuvant mFOLFIRINOX is recommended for patients with resected PC and ECOG PS 0-1.
Strong recommendation, high grade evidence (23-25)
▪️ In patients who are not candidates for mFOLFIRINOX (age >75 years, ECOG PS 2 or contraindication to mFOLFIRINOX), we recommend gemcitabine-capecitabine as an alternative option.
Strong recommendation, high grade evidence (23-25)
▪️ Adjuvant gemcitabine or 5-FU-LV should be limited to frail patients.
Strong recommendation, high grade evidence (23-25)
▪️ Adjuvant CRT is not recommended and should not be given to patients following surgery (in R0 cases).
Strong recommendation, high grade evidence (26).
➡️Treatment of borderline resectable tumors (BRPC)
▪️ Patients with BRPC have a high probability of an R1 resection and should be considered for induction treatment.
Strong recommendation, high grade evidence (18-20)
▪️ A period of induction chemotherapy (FOLFIRINOX) followed by CRT on a case-by-case basis and subsequent surgery, is recommended according to MDT recommendations
Strong recommendation, low grade evidence (20, 27).
▪️ Gemcitabine combined with oxaliplatin or capecitabine may be considered, when FOLFIRINOX is not feasible.
Strong recommendation, low grade evidence (20,22).
▪️ CRT with capecitabine may be considered after induction Chemotherapy.
Conditional recommendation, low grade evidence (19)
▪️ Following induction therapy, medically fit patients without disease progression and with a decrease in CA 19-9 should undergo surgical exploration, unless contraindicated.
Strong recommendation, strong grade evidence (28).
➡️Treatment of locally advanced pancreatic cancer (LAPC)
▪️ A conversion surgery strategy utilizing the standard of care of up to 6 months of combination Chemotherapy (e.g. FOLFIRINOX) should be chosen.
Strong recommendation, strong grade evidence (29-31).
▪️ Arterial resection after induction therapy is not recommended but can be considered as a possibility in experienced centers on a case-by-case basis in selected patients according to MDT recommendations.
Conditional recommendation, very low grade evidence (32,33).
➡️Treatment of advanced pancreatic cancer
First-line treatment
▪️ Options to treat patients with metastatic PC should be dependent on PS:
o In patients with ECOG PS 0-1 and bilirubin level <1.5 times the ULN, the regimen
FOLFIRINOX should be considered.
Strong recommendation, high grade evidence (34)
o For patients with ECOG PS 2, Karnofsky PS (KPS) >70 and bilirubin level <1.5 times
the ULN, gemcitabine-cisplatin can be considered.
Strong recommendation, high grade evidence (34).
o For patients with ECOG PS 2, KPS <70 and/or bilirubin level >1.5 times the ULN,
gemcitabine monotherapy should be considered.
Strong recommendation, high grade evidence (34).
o For patients with ECOG PS 3-4, symptom-directed and palliative care should be
considered
Strong recommendation, high grade evidence (34).
▪️The efficacy of treatment should be typically evaluated every 8-12 weeks and should be based on clinical status, CA 19-9 trajectory and imaging.
Strong recommendation, high grade evidence (35).
Second-line treatment
▪️ After FOLFIRINOX treatment, gemcitabine alone may be offered to patients with ECOG PS 0-1 and a favorable comorbidity profile.
Conditional recommendation, low grade evidence (36-39).
▪️ Oxaliplatin-based second-line treatment (mFOLFOX6 or OFF) may be considered as an alternative in patients with ECOG PS 0-2 if not given previously.
Conditional recommendation, low grade evidence (36-39).
▪️ For patients with ECOG PS 3-4, symptom directed, and palliative care is recommended.
Strong recommendation, low grade evidence (36-39).
▪️ Maintenance therapy with capecitabine (after discussion with patient) may be indicated till disease progression or unacceptable toxicity on a case- by case basis according to MDT recommendations.
Conditional recommendation, low grade evidence (36-39).
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines, for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines: Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Yousri Wasef & Yousri Rostom.
· Chair of the GIT Cancer Scientific Committee: Prof Yousri Rostom
- The pancreatic cancer Scientific Group Members: (Alphabitical order): Ahmed EL Kassed, Doaa Gennena, Fouad Aboutaleb, Heba Fadlol, Hussein Okasha, Khaled Abdelkarim, Manal EL Mahdy, Mary Gamal, Mohammed Eid, Omar S. Omar, and Yousri Wasef.
- Abbreviations
|
BRPC |
Borderline resectable prostatic cancer |
|
CBC |
Complete blood count |
|
CRT |
Combined chemo radiotherapy |
|
Computed Tomography |
|
|
EHC |
Egyptian Health Council |
|
EUS |
Endoscopic ultrasound |
|
KFTs |
Kidney function tests |
|
LAPC |
Locally advanced pancreatic cancer |
|
LFTs |
Liver function tests |
|
LMWH |
Low-molecular-weight heparin |
|
MRI |
Magnetic resonance imaging |
|
MDT |
Multidisciplinary team |
|
PC |
Pancreatic cancer |
|
PET/CT |
Positron emission tomography/Computed Tomography |
|
PS |
Performance Status |
|
SMA |
Superior mesenteric artery |
|
ULN |
Upper limit of normal |
- Introduction
Pancreatic cancer is a malignancy with high mortality, and the overall prognosis is poor. The
growing trend of pancreatic cancer cases is expected to continue for the next two decades and
beyond. In Egypt, there was an estimated 3349 new cases of pancreatic cancer and 3186 deaths
occurred because of this disease based on GLOBOCAN 2022.
- Purpose and scope
These guidelines are developed to improve the quality of care for pancreatic cancer via providing a uniform standard of care across the country to help in early diagnosis, treatment and follow up for pancreatic cancer so more optimal treatment options and improved clinical outcomes.
- Target audience
Clinicians who are involved in the care and treatment of patients with pancreatic cancer, include medical oncologists, radiation oncologists, clinical oncologist, gastroenterologists, surgeons, clinical dietrition interventional radiologists, radiologists, pathologists, and palliative care specialists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The ESMO, NCCN, and NICE guidelines are the main sources used while formulating the national guidelines for pancreatic cancer (1-3).
➡️Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group:https://www.gradeworkinggroup.org/
. GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
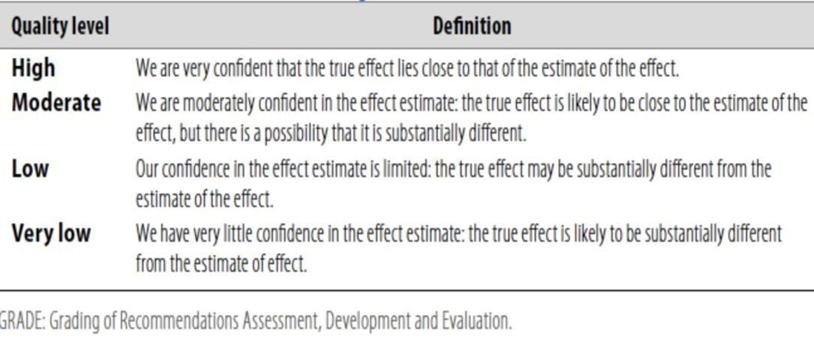
Table 2: Significance of the four levels of evidence

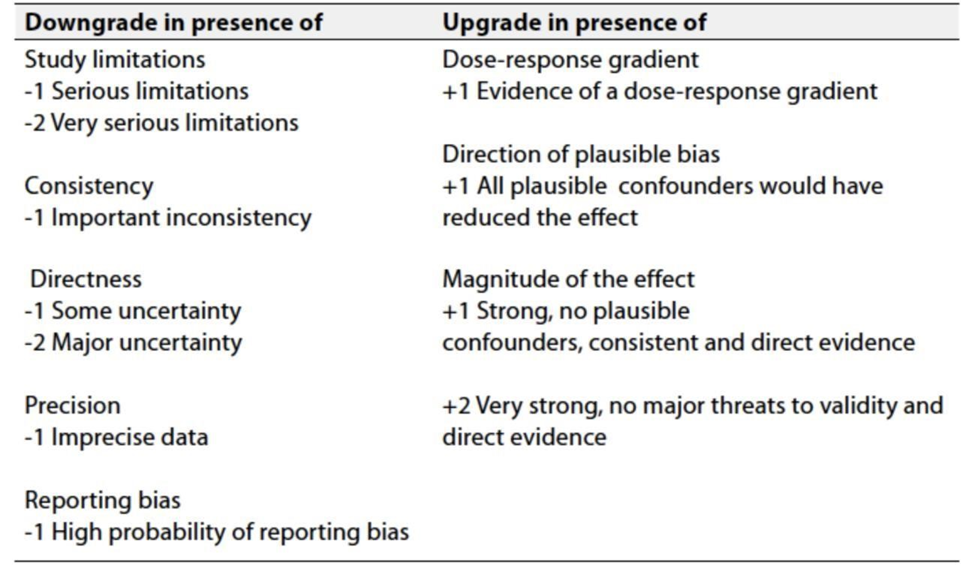
Table 3: Factors that determine How to upgrade or downgrade the quality of evidence
➡️ The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Clinical indicators for monitoring
For patients newly diagnosed with pancreatic cancer:
- laboratory evaluation (CBC, LFT, and KFT)
- imaging
- tissue biopsy for pathological confirmation &IHC
- Research gaps
▪️ Systematic inclusion of cost-benefit analyses in clinical trials with collection of health economic analysis such as incremental cost effectiveness ratio in order to facilitate clinical decision-making.
▪️ Predictive biomarkers: response to specific systemic targeted therapies and immunotherapy.
▪️ Improve models for pre-clinical testing of novel drugs.
▪️ Search for tools to assess quality of life and in clinical trials.
▪️ Dietary supplements, nutritional counselling, physical activity recommendations and psychological support as part of an integrative healthcare approach to care for people with pancreatic cancer.
- Update of this guideline
▪️ This guideline will be updated whenever there is new evidence.
- References
1. Tsuchida H, Fujii T, Mizuma M, et al. Prognostic importance of peritoneal washing cytology in patients with otherwise resectable pancreatic ductal adenocarcinoma who underwent pancreatectomy: a nationwide, cancer registry-based study from the Japan Pancreas Society. Surgery. 2019;166(6):997-1003.
2. Fukukura Y, Kumagae Y, Fujisaki Y, et al. Adding delayed phase images to dual-phase contrast-enhanced CT increases sensitivity for small pancreatic ductal adenocarcinoma. AJR Am J Roentgenol 2021;217(4):888-897.
3. Zins M, Matos C, Cassinotto C. Pancreatic adenocarcinoma staging in the era of preoperative chemotherapy and radiation therapy. Radiology. 2018;287(2):374-390.
4. Hong SB, Choi SH, Kim KW, et al. Meta-analysis of MRI for the diagnosis of liver metastasis in patients with pancreatic adenocarcinoma. J Magn Reson Imaging. 2019;51(6):1737-1744.
5. Wang L, Dong P, Wang WG, et al. Positron emission tomography modalities prevent futile radical resection of pancreatic cancer: a meta-analysis. Int J Surg. 2017;46:119-125.
6. Janick Selves, Elodie Long-Mira, Marie-Christine Mathieu, Philippe Rochaix, Marius Ilié, Immunohistochemistry for Diagnosis of Metastatic Carcinomas of Unknown Primary Site, Cancers (Basel). 2018 Apr 5;10(4):108.
7. AJCC Cancer Staging Manual, Eighth Edition (2017) published by Springer International Publishing.
8. Al-Hawary MM, Francis IR, Chari ST, et al. Pancreatic ductal adenocarcinoma radiology reporting template: consensus statement of the Society of Abdominal Radiology and the American Pancreatic Association. Radiology 2014;270:248-260.
9. Andreas Karachristos, Nikolaos Scarmeas, John P Hoffman, CA 19-9 levels predict results of staging laparoscopy in pancreatic cancer, J Gastrointest Surg., 2005 Dec;9(9):1286-92.
10. Ghabi E, He J., Positive pancreatic neck margins-a telltale sign of complex biology. Hepatobiliary Surg Nutr. 2022 Apr;11(2):302-304.
11. Delpero JR, Bachellier P, Regenet N, et al. Pancreaticoduodenectomy for pancreatic ductal adenocarcinoma: a French multicentre prospective evaluation of resection margins in 150 evaluable specimens. HPB (Oxford). 2014;16(1):20-33.
12. Delpero JR, Boher JM, Sauvanet A, et al. Pancreatic adenocarcinoma with venous involvement: is up-front synchronous portal-superior mesenteric vein resection still justified? A survey of the association française de chirurgie. Ann Surg Oncol. 2015;22(6):1874-1883.
13. Mitchem JB, Hamilton N, Gao F, et al. Long-term results of resection of adenocarcinoma of the body and tail of the pancreas using radical antegrade modular pancreatosplenectomy procedure. J Am Coll Surg. 2012;214(1):46-52.
14. Tol JA, Gouma DJ, Bassi C, et al. Definition of a standard Lymphadenectomy in surgery for pancreatic ductal adenocarcinoma: a consensus statement by the International Study Group on Pancreatic Surgery (ISGPS). Surgery. 2014;156(3):591-600.
15. Farge D, Bournet B, Conroy T, et al. Primary thromboprophylaxis in pancreatic cancer patients: why clinical practice guidelines should be implemented. Cancers. 2020;12(3):618.
16. Schrag D, Uno H, Rosovsky R, et al. Direct oral anticoagulants vs low-molecular-weight heparin and recurrent VTE in patients with cancer:a randomized clinical trial. JAMA. 2023;329(22):1924-1933.
17. van der Gaag NA, Rauws EAJ, van Eijck CHJ, et al. Preoperative biliary drainage for cancer of the head of the pancreas. N Engl J Med. 2010;362(2):129-137.
18. Versteijne E, Vogel JA, Besselink MG, et al. Meta-analysis comparing upfront surgery with neoadjuvant treatment in patients with resectable or borderline resectable pancreatic cancer. Br J Surg.2018;105(8):946-958.
19. Cloyd JM, Heh V, Pawlik TM, et al. Neoadjuvant therapy for resectable and borderline resectable pancreatic cancer: a meta-analysis of randomized controlled trials. J Clin Med. 2020;9(4):1129.
20. Ghanem I, Lora D, Herradón N, et al. Neoadjuvant chemotherapy with or without radiotherapy versus upfront surgery for resectable pancreatic adenocarcinoma: a meta-analysis of randomized clinical trials. ESMO Open. 2022;7(3):100485.
21. Oettle H, Neuhaus P, Hochhaus A, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA. 2013;310(14):1473-1481.
22. Neoptolemos JP, Stocken DD, Bassi C, et al. Adjuvant chemotherapy with fluorouracil plus folinic acid vs gemcitabine following pancreatic cancer resection: a randomized controlled trial. JAMA. 2010;304(10):1073-1081.
23. Valle JW, Palmer D, Jackson R, et al. Optimal duration and timing of adjuvant chemotherapy after definitive surgery for ductal adenocarcinoma of the pancreas: ongoing lessons from the ESPAC-3 study. J Clin Oncol. 2014;32(6):504-512.
24. Conroy T, Hammel P, Hebbar M, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379(25):2395-2406.
25. Conroy T, Castan F, Lopez A, et al. Five-year outcomes of FOLFIRINOX vs gemcitabine as adjuvant therapy for pancreatic cancer: a randomized clinical trial. JAMA Oncol. 2022;8(11):1571-1578.
26. Seufferlein T, Hammel P, Delpero JR, et al. Optimizing the management of locally advanced pancreatic cancer with a focus on induction chemotherapy: expert opinion based on a review of current evidence.Cancer Treatment Rev. 2019;77:1-10.
27. Katz MHG, Shi Q, Meyers J, et al. Efficacy of preoperative mFOLFIRINOX vs mFOLFIRINOX plus hypofractionated radiotherapy for borderline resectable adenocarcinoma of the pancreas: the A021501 phase 2 randomized clinical trial. JAMA Oncol. 2022;8(9): 1263-1270.
28. Ye C, Sadula A, Ren S, et al. The prognostic value of CA19-9 response after neoadjuvant therapy in patients with pancreatic cancer: a systematic review and pooled analysis. Cancer Chemother Pharmacol.2020;86(6):731-740.
29. Seufferlein T, Hammel P, Delpero JR, et al. Optimizing the management of locally advanced pancreatic cancer with a focus on induction chemotherapy: expert opinion based on a review of current evidence. Cancer Treatment Rev. 2019;77:1-10.
30. Suker M, Beumer BR, Sadot E, et al. FOLFIRINOX for locally advanced pancreatic cancer: a systematic review and patient-level meta-analysis. Lancet Oncol. 2016;17(6):801-810.
31. Van Laethem J-L. Induction chemotherapy in borderline (non-)resectable pancreatic cancer: a unique window of opportunity for understanding pancreatic cancer. Eur J Cancer. 2019;106:34-36.
32. Loos M, Kester T, Klaiber U, et al. Arterial resection in pancreatic cancer surgery: effective after a learning curve. Ann Surg. 2022;275(4):759-768.
33. Tee MC, Krajewski AC, Groeschl RT, et al. Indications and perioperative outcomes for pancreatectomy with arterial resection. J Am Coll Surg. 2018;227(2):255-269.
34. Lambert A, Schwarz L, Borbath I, et al. An update on treatment options for pancreatic adenocarcinoma. Ther Adv Med Oncol. 2019;11:175883591987556.
35. Halle-Smith JM, Hall L, Daamen LA, et al. Clinical benefit of surveillance after resection of pancreatic ductal adenocarcinoma: a systematic review and meta-analysis. Eur J Surg Oncol. 2021;47(9):2248-2255.
36. Oettle H, Riess H, Stieler JM, et al. Second-line oxaliplatin, folinic acid, and fluorouracil versus folinic acid and fluorouracil alone for gemcitabine-refractory pancreatic cancer: outcomes from the CONKO-003 trial. J Clin Oncol. 2014;32(23):2423-2429.
37. Gill S, Ko Y-J, Cripps C, et al. PANCREOX: a randomized phase III study of fluorouracil/leucovorin with or without oxaliplatin for second-line advanced pancreatic cancer in patients who have received gemcitabine-based chemotherapy. J ClinOncol. 2016;34(32):3914-3920.
38. Ohkawa S, Okusaka T, Isayama H, et al. Randomised phase II trial of S- 1 plus oxaliplatin vs S-1 in patients with gemcitabine-refractory pancreatic cancer. Br J Cancer. 2015;112(9):1428-1434.
39. de la Fouchardiere C, Malka D, Chabaud S, et al. LBA60 Evaluation of gemcitabine and paclitaxel versus gemcitabine alone after FOLFIRINOX failure or intolerance in metastatic pancreatic ductal adenocarcinoma: results of the randomized phase III PRODIGE 65-UCGI 36-GEMPAX UNICANCER study. Ann Oncol. 2022;33:S1426.
- Annexes
NCCN resectability Criteria
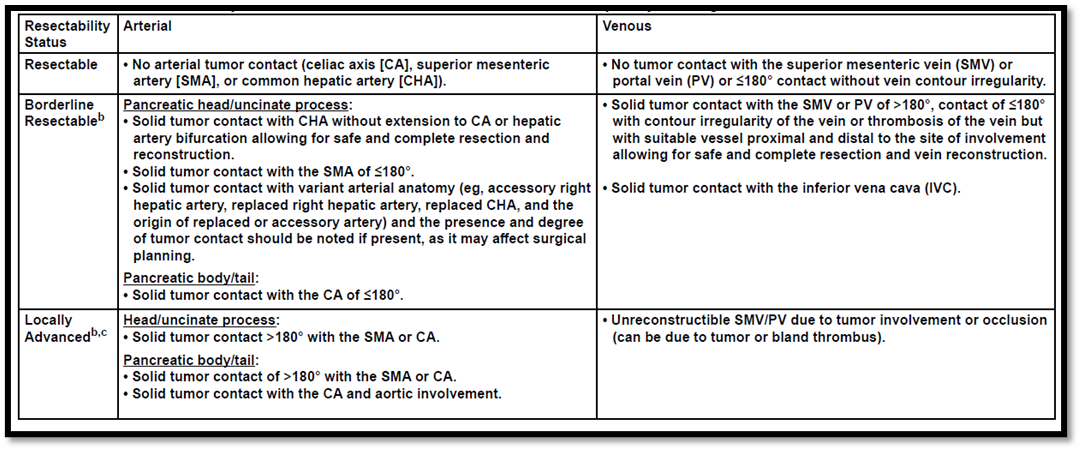
Al-Hawary MM, Francis IR, Chari ST, et al. Pancreatic ductal adenocarcinoma radiology reporting template: consensus statement of the Society of Abdominal Radiology and the American Pancreatic Association. Radiology 2014;270:248-260.
American Joint Committee on Cancer (AJCC) TNM Staging of Pancreatic Cancer (8th ed., 2017)
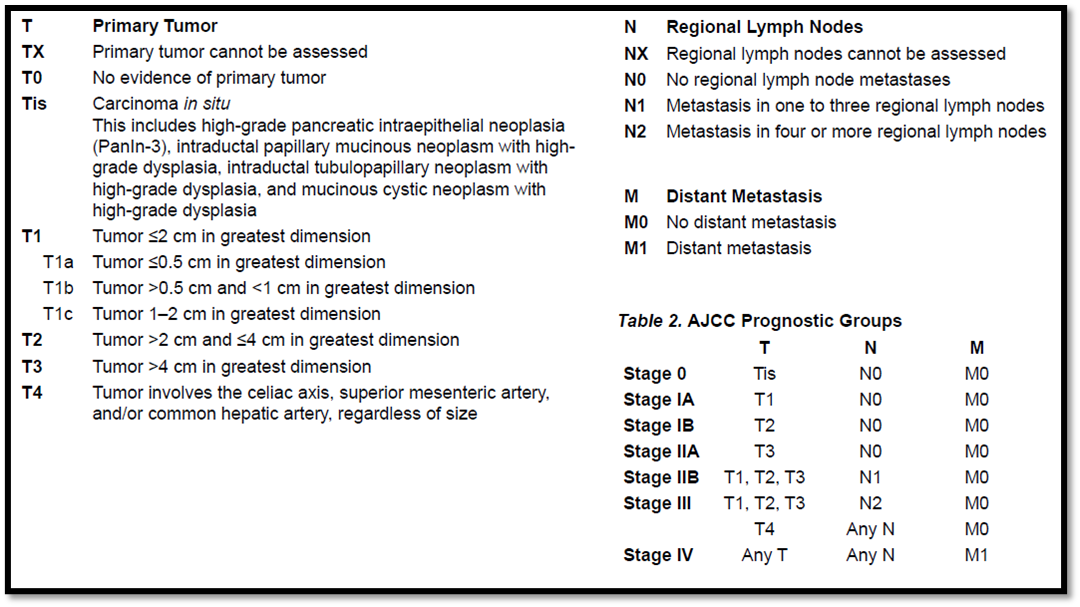
AJCC Cancer Staging Manual, Eighth Edition (2017) published by Springer International Publishing.