Pharmacological Approach to Type 1 Diabetes in Adults
| Site: | EHC | Egyptian Health Council |
| Course: | Diabetes and Endocrinology Guidelines |
| Book: | Pharmacological Approach to Type 1 Diabetes in Adults |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:47 AM |
- Executive Summary
The prevalence of type 1 diabetes is globally increasing around 2 million with high incidence of acute and chronic complications and the majority of the patients remain uncontrolled despite the presence of many insulin regimens and many new insulin analogues as well as the great improvement of insulin delivery systems.
This guideline provides recommendations for the pharmacological approach to type1 diabetes in adults.
➡️Recommendations
▪️ Treat most adults with type 1 diabetes with multiple daily doses of prandial and basal insulin Strong recommendation ▪️ For most adults with type 1 diabetes, insulin analogs are preferred over injectable human insulins to minimize hypoglycemia risk. Conditional recommendation ▪️ Automated Insulin delivery systems (insulin pens) and insulin analogues could be considered for all adults with type 1 diabetes. Conditional recommendation ▪️ To improve glycemic outcomes and quality of life and minimize hypoglycemia risk, most adults with type 1 diabetes should receive education on how to match mealtime insulin doses to carbohydrate intake and, additionally, to fat and protein intake. They should also be taught how to modify the insulin dose (correction dose) based on concurrent glycemia, glycemic trends (if available), sick-day management, and anticipated physical activity. Strong recommendation ▪️ Insulin treatment plan and insulin-taking behavior should be reevaluated at regular intervals (e.g., every 3–6 months) and adjusted to incorporate specific factors that impact choice of treatment and ensure achievement of individualized glycemic goals. Good practice statement |
- Recommendations
▪️ Treat most adults with type 1 diabetes with multiple daily doses of prandial and basal insulin
Strong recommendation, High-Quality Evidence (systematic review of cross-sectional studies with a consistent reference standard) 3- 5
▪️ For most adults with type 1 diabetes, insulin analogs are preferred over injectable human insulins to 2minimize hypoglycemia risk.
Conditional recommendation, High-Quality Evidence (systematic review of cross-sectional studies with a consistent reference standard) 6 - 10
▪️ Automated Insulin delivery systems (insulin pens) and insulin analogues could be considered for all adults with type 1 diabetes.
Conditional recommendation, High-Quality Evidence (systematic review of cross-sectional studies with a consistent reference standard) 11, 12
▪️ To improve glycemic outcomes and quality of life and minimize hypoglycemia risk, most adults with type 1 diabetes could receive education on how to match mealtime insulin doses to carbohydrate intake and, additionally, to fat and protein intake. They should also be taught how to modify the insulin dose (correction dose) based on concurrent glycemia, glycemic trends (if available), sick-day management, and anticipated physical activity.
Conditional Recommendation, Moderate Quality Evidence (based on observational studies) 13, 14
▪️ Insulin treatment plan and insulin-taking behavior should be reevaluated at regular intervals (e.g., every 3–6 months) and adjusted to incorporate specific factors that impact choice of treatment and ensure achievement of individualized glycemic goals
Conditional Recommendation, Moderate Quality Evidence (based on observational studies) 1, 14, 15
- Acknowledgement
We would like to acknowledge the Diabetes and Endocrinology Scientific Committee for developing these guidelines.
Chair of the GDG: Mohamed Hesham El Hefnawy, National Institute of Diabetes and Endocrinology, Cairo
Members of the Guideline Development Group (GDG):
Mohamed Abdelhady Mohamed Mashahet, Faculty of Medicine, Fayoum University, Fayoum.
Athar Reda Ibrahim, National Institute of Diabetes and Endocrinology, Cairo
Ahmed Mohamed Hamam, Military Hospital, Cairo
Amr Ali Mahfouz, National Institute of Diabetes and Endocrinology, Cairo
Atef Bassyouni, National Institute of Diabetes and Endocrinology, Cairo
Elsayed Abdel Fattah Eid, Faculty of Medicine, Delta University for science and technology, Dakahlia
Fawzy A F Elmessallamy, Faculty of Medicine, Zagazig University, Sharqia
Mai Mohammed Salama, National Hepatology and Tropical Medicine Research Institute, Cairo
Mohamed Abdelmoniem Elmikawy, police hospital, Cairo.
Randa Salam, Faculty of Medicine, Cairo University, Cairo.
Yara Muhammad Ahmad Eid, Faculty of Medicine, Ain Shams University, Cairo.
- Abbreviations
AID
Automated
insulin delivery
BGM
Blood glucose monitoring
BMI
Body Mass Index
CGM
Continuous glucose monitoring
CVD
Cardiovascular disease
CSII
Continuous subcutaneous insulin infusion
DPP - inhibitors
Diabetes Prevention Program
DPP - 4
Dipeptidyl peptidase - 4 inhibitors
GLP-1Ra
Glucagon like peptide – 1 receptor agonists
GRADE
Grading of Recommendations Assessment, Development and Evaluation
HbA1c
glycated haemoglobin A1c
ICR
Insulin to carbohydrate ratio
ISF
Insulin Sensitivity Factor
LAA
Long acting analogs
MDI
Multiple daily injections
RAA
Rapid-acting analogs
RCT
Randomized controlled trial
SGLT2i
Sodium glucose co transporter inhibitors
TDD.
Total daily insulin dose
TIR
Time in Range
URAA
Ultra rapid-acting insulin analog.
- Glossary
Cardiovascular diseases (CVDs)
A group of disorders of the heart and blood vessels that include coronary heart disease and cerebrovascular disease and PAD
Dipeptidyl peptidase - 4 inhibitors (DPP - 4 inhibitors or gliptins)
Oral antidiabetic drugs used in treating type 2 diabetes. They suppress the degradation of incretins by blocking the action of the enzyme dipeptidyl - peptidase 4. This stimulates insulin secretion and suppresses glucagon release.
HbA1c
Haemoglobin glycated by non-enzymatic attachment of glucose to haemoglobin. The concentration of HbA1c is the most commonly used measure of chronic glycaemia in clinical trials and diabetes management. It is considered to reflect the integrated mean glucose level over the previous 8–12 weeks.
Metformin
A biguanide oral antidiabetic, agent used in treating type 2 diabetes. It decreases glucose production by the liver
Type 1 diabetes
Diabetes caused by immune destruction of pancreatic beta-cells, resulting in lack of insulin production by the pancreas and need for insulin injections for survival.
Type 2 diabetes
Diabetes characterized by various degrees of disorders of insulin action in the body and insulin secretion by the pancreas. Insulin injections are not needed for survival, but might be needed for controlling blood glucose levels.
Incretins
are a group of metabolic hormones that stimulate a decrease in blood glucose levels.
Basal insulin
Insulin which regulates the body's blood glucose between mealtimes, as well as overnight.
Prandial insulin
insulin covers increases in blood glucose levels following meals
Insulin analogs
synthetically produced variations of insulin that are different in amino acid sequence than that of native human insulin.
- Introduction
Insulin treatment is crucial for individuals with type 1 diabetes due to absent or near-absent β-cell function, which can lead to hyperglycemia, metabolic disturbances, and life-threatening tissue catabolism. The Diabetes Control and Complications Trial (DCCT) found that intensive therapy reduced A1C and improved long-term outcomes, but was associated with a higher rate of severe hypoglycemia. Insulin replacement plans typically consist of basal insulin, mealtime insulin, and correction insulin. Basal insulin analogs have longer duration of action and are associated with less hypoglycemia and weight gain. However, the cost and intensity of treatment may be prohibitive. Regular reassessment of insulin-taking behavior and adjustment of treatment plans are recommended. Individuals with type 1 diabetes require approximately 30-50% of their daily insulin as basal and the remainder as prandial, with total daily insulin requirements ranging from 0.4 to 1.0 units/kg/day. For examples of subcutaneous insulin treatment plans (Regimens) see annexes table 1.
Noninsulin Treatments for Type 1 Diabetes
Injectable and oral glucose-lowering medications have been studied for their efficacy as adjunct to insulin treatment of type 1 diabetes. Pramlintide is based on the naturally occurring β-cell peptide amylin and is approved for use in adults with type 1 diabetes. Clinical trials have demonstrated a modest reduction in A1C (0.3–0.4%) and modest weight loss (∼1 kg) with pramlintide Similar results have been reported for several agents currently approved only for the treatment of type 2 diabetes. The addition of metformin in adults with type 1 diabetes was associated with small reductions in body weight, insulin dose, and lipid levels but did not sustainably improve A1C. The largest clinical trials of glucagon-like peptide 1 receptor agonists (GLP-1 RAs) in type 1 diabetes have been conducted with Liraglutide 1.8 mg daily, and results showed modest A1C reductions (∼0.4%), decreases in weight (∼5 kg), and reductions in insulin doses . Similarly, sodium–glucose co-transporter 2 (SGLT2) inhibitors have been studied in clinical trials in people with type 1 diabetes, and results showed improvements in A1C, reduced body weight, and improved blood pressure . However, SGLT2 inhibitor use in type 1 diabetes was associated with an increased rate of DKA.
Surgical Treatment for Type 1 Diabetes
Pancreas and Islet Transplantation
Successful pancreas and islet transplantation can normalize glucose levels and mitigate microvascular complications of type 1 diabetes. However, people receiving these treatments require lifelong immunosuppression to prevent graft rejection and/or recurrence of autoimmune islet destruction. Given the potential adverse effects of immunosuppressive therapy, pancreas transplantation should be reserved for people with type 1 diabetes undergoing simultaneous kidney transplantation, following kidney transplantation, or for those with recurrent ketoacidosis or severe hypoglycemia despite intensive glycemic management.
- SCOPE and Purpose
The scope of this guideline is to provide evidence-based recommendations for pharmacological approach in T1 DM patients. The purpose of developing the guideline is to provide guidance on how to use the different available insulin predations as well as available insulin delivery options and to how to apply different insulin treatment regimens to achieve the glycemic targets with fewer complications
- Target audience
This guideline targets, healthcare professionals (internists, endocrinologists, family medicine physicians) and, researchers, policymakers.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2015 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in a table:
• Developing organisation/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt the American Diabetes Association – Standards of Care in Diabetes – 2024, 2025. 1,2
- Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
• GRADE working group: https://www.gradeworkinggroup.org/
• GRADE online training modules: http://cebgrade.mcmaster.ca/
• GRADE profile software: http://ims.cochrane.org/revman/gradepro
Table 2: Quality and Significance of the four levels of evidence in GRADE:
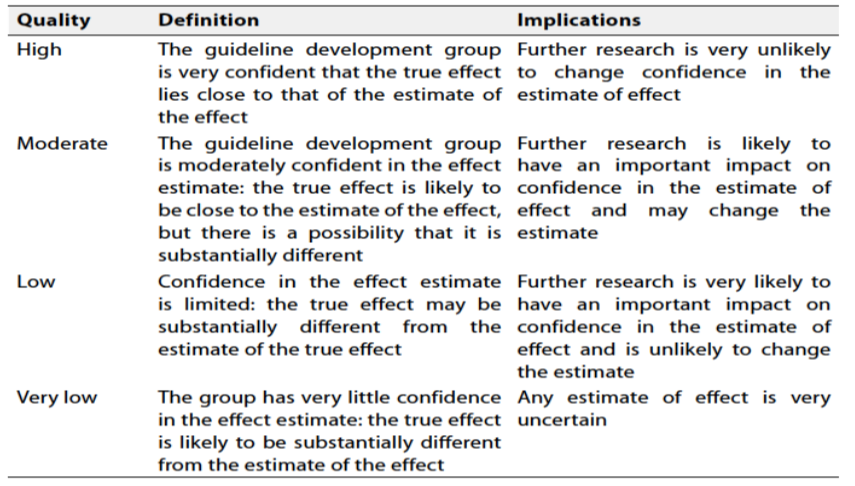
Table 3: Factors that determine How to upgrade or downgrade the quality of evidence
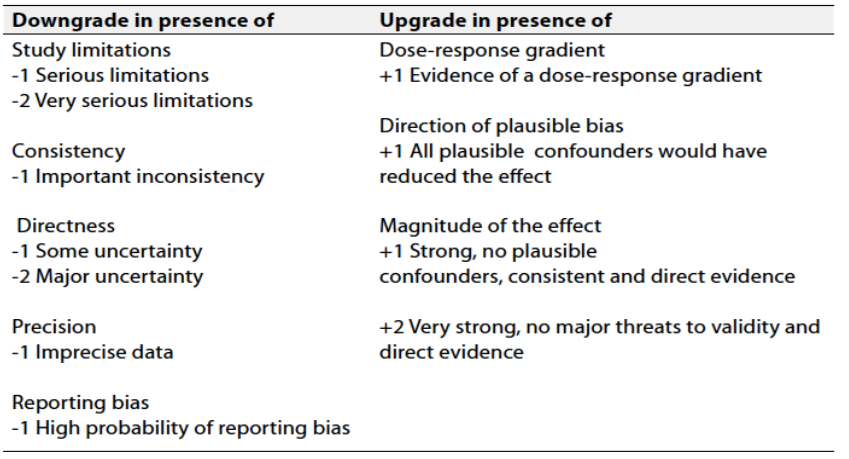
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research needs
During the review of evidence and the development of recommendations, several research gaps were identified regarding Egyptian population. Addressing these will help inform revision of these guidelines.
1. Long-Term Effectiveness: Conduct studies to evaluate the long-term effectiveness of different insulin regimens used to treat TIDM beyond the initial intervention period. Assess the sustainability of behavior changes and diabetes complications risk reduction over extended periods to determine the program's lasting impact.
2. Tailoring Interventions: Investigate the effectiveness of personalized or tailored interventions within T1DM patients. Explore how individual characteristics, cultural factors, socioeconomic status, and health literacy influence regimen outcomes and identify strategies for optimizing intervention customization.
Monitoring and evaluating the impact of the guideline
There are potential indicators that can be used to evaluate the success of implementing a insulin regimens in type 1 DM. These indicators can provide complementary information and a more comprehensive assessment of the program's effectiveness.
1. Hypoglycemia incidence rate reduction: calculate the percentage reduction in the hypglycemia incidence rate using the following formula:
Percentage Reduction = ((Baseline Incidence Rate - Post-Implementation Incidence Rate) / Baseline Incidence Rate) * 100
the same apply to DKA and hospitalization due to dysglycemia
2. Cost-effectiveness: Assess the cost-effectiveness of DIFFERENT REGIMENS by comparing the program's costs to the achieved health outcomes. This indicator considers the financial investment required for the program and the benefits gained in terms of diabetes prevention and reduced healthcare costs.
3. Patient Satisfaction and Feedback: Collect feedback from participants through surveys or interviews to gauge their satisfaction with the REGIMEN , perceived benefits, and suggestions for improvement. Positive patient satisfaction indicates a well-received and impactful program.
- Updating of the guidelines
These guidelines will be updated whenever there is new evidence.
- Reference
- American Diabetes Association Professional Practice Committee; 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2024. Diabetes Care 1 January 2024; 47 (Supplement_1): S158–S178. https://doi.org/10.2337/dc24-S009.
- American Diabetes Association Professional Practice Committee; 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2025. Diabetes Care 1 January 2025; 48 (Supplement_1): S181–S206. https://doi.org/10.2337/dc25-S009
- Lachin JM, Bebu I; DCCT/EDIC Research Group. The beneficial effects of earlier versus later implementation of intensive therapy in type 1 diabetes. Diabetes Care 2021;44:2225–2230
- Russell-Jones D, Bode BW, De Block C, et al. Fast-acting insulin aspart improves glycemic control in basal-bolus treatment for type 1 diabetes: results of a 26-week multicenter, active-controlled, treatto-target, randomized, parallel-group trial (onset 1). Diabetes Care 2017;40:943–950
- Holt RIG, DeVries JH, Hess-Fischl A, et al. The management of type 1 diabetes in adults. a consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2021;44:2589–2625
- DeWitt DE, Hirsch IB. Outpatient insulin therapy in type 1 and type 2 diabetes mellitus: scientific review. JAMA 2003;289:2254–2264
- Tricco AC, Ashoor HM, Antony J, et al. Safety, effectiveness, and cost effectiveness of long acting versus intermediate acting insulin for patients with type 1 diabetes: systematic review and network meta-analysis. BMJ 2014;349:g5459
- Bartley PC, Bogoev M, Larsen J, Philotheou A. Long-term efficacy and safety of insulin detemir compared to neutral protamine Hagedorn insulin in patients with Type 1 diabetes using a treat-totarget basal-bolus regimen with insulin aspart at meals: a 2-year, randomized, controlled trial. Diabet Med 2008;25:442–449
- Lane W, Bailey TS, Gerety G, et al.; Group Information; SWITCH 1. Effect of insulin degludec vs insulin glargine U100 on hypoglycemia in patients with type 1 diabetes: the SWITCH 1 randomized clinical trial. JAMA 2017;318:33–44
- Home PD, Bergenstal RM, Bolli GB, et al. New insulin glargine 300 units/mL versus glargine 100 units/mL in people with type 1 diabetes: a randomized, phase 3a, open-label clinical trial (EDITION 4). Diabetes Care 2015;38:2217–2225
- Yeh HC, Brown TT, Maruthur N, et al. Comparative effectiveness and safety of methods of insulin delivery and glucose monitoring for diabetes mellitus: a systematic review and metaanalysis. Ann Intern Med 2012;157:336–347
- Speight J, Choudhary P, Wilmot EG, et al. Impact of glycaemic technologies on quality of life and related outcomes in adults with type 1 diabetes: a narrative review. Diabet Med 2023; 40:e14944
- Hopkins D, Lawrence I, Mansell P, et al. Improved biomedical and psychological outcomes 1 year after structured education in flexible insulin therapy for people with type 1 diabetes: the U.K. DAFNE experience. Diabetes Care 2012;35:1638– 1642
- Speight J, Amiel SA, Bradley C, et al. Longterm biomedical and psychosocial outcomes following DAFNE structured education to promote intensive insulin therapy in adults with sub-optimally controlled Type 1 diabetes. Diabetes Res Clin Pract 2010;89:22–29
- Kahn, P.A., Liu, S., McCoy, R., Gabbay, R.A. and Lipska, K., 2021. Glucagon use by US adults with type 1 and type 2 diabetes. Journal of Diabetes and its Complications, 35(5), p.107882.
- Annexes
Table (1): Examples of subcutaneous insulin treatment plans (Regimens)
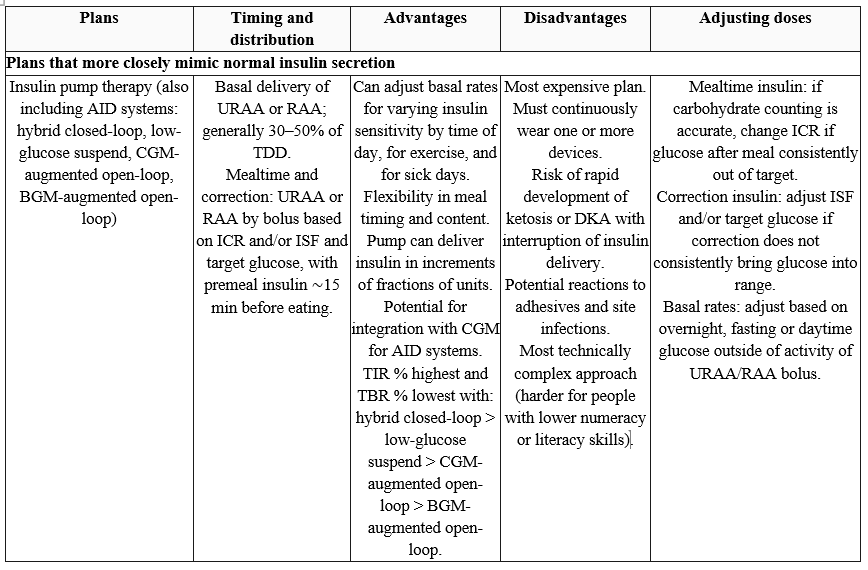
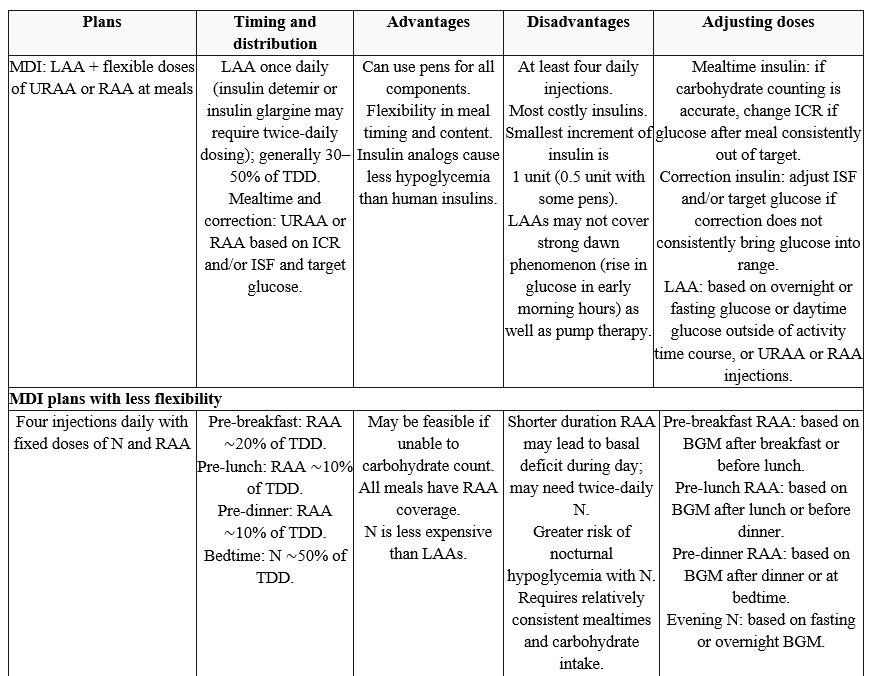
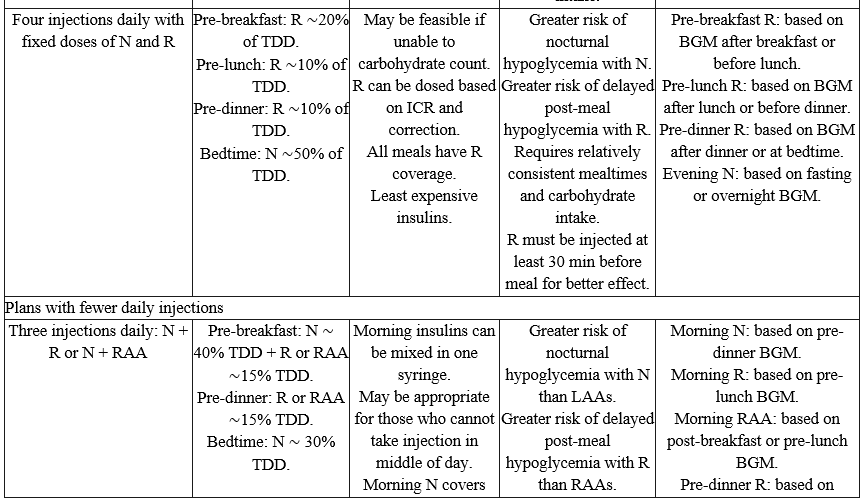
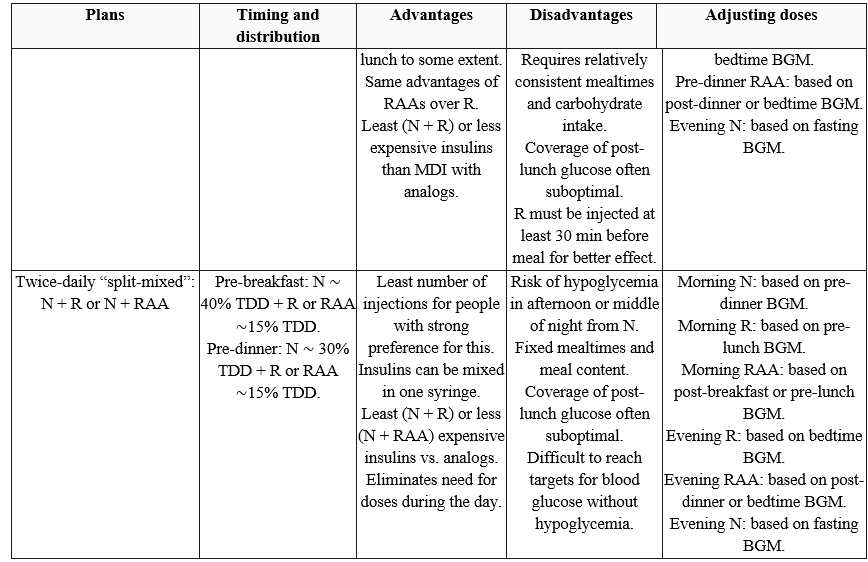
AID, automated insulin delivery; BGM, blood glucose monitoring; CGM, continuous glucose monitoring; ICR, insulin-to-carbohydrate ratio; ISF, insulin sensitivity factor; LAA, long-acting analog; MDI, multiple daily injections; N, NPH insulin; R, short-acting (regular) insulin; RAA, rapid-acting analog; TBR, time below range; TDD, total daily insulin dose; TIR, time in range; URAA, ultra-rapid-acting analog. Adapted from Holt et al. 5