Diabetes Care in Hospital Settings
| Site: | EHC | Egyptian Health Council |
| Course: | Diabetes and Endocrinology Guidelines |
| Book: | Diabetes Care in Hospital Settings |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 21 January 2025" Download Guideline
- Executive Summary
This guideline offers evidence-based recommendations on the inpatient management of diabetic patients during hospital stay. The recommendations are intended to provide healthcare professionals with practical guidance on the proper way to manage diabetes and dysglycemia during hospitalization.
- Recommendations
▪️ A1C test should be performed for any patient with random blood glucose sample > 140 mg/dl during hospitalization or for any patient with a known history of diabetes. (strong recommendation, moderate certainty evidence) 3
▪️ Health care facilities should implement validated protocols (written or computerized) and delivered to all departments including: emergency units, ICU units, general wards, obstetrician/delivery units and dialysis suits. (strong recommendation, moderate certainty evidence)4-11
Remarks
It is reasonable to check A1C level in any hospitalized patient with a random blood glucose sample > 140 mg/dl. This is the cutoff point for further screening in hospitalized patients. It is also very important to have validated protocols (written or computerized) and delivered to all departments to ensure high quality management for all the hospitalized patients with dysglycemia.
The National Academy of Medicine advocates for the implementation of computerized provider order entry (CPOE) to mitigate medication-related errors and streamline medication administration processes 10
Summary of evidence
Reviews of randomized controlled trials have revealed notable improvements in glycemic outcomes within hospital settings through the utilization of computerized advice systems. These improvements include increased time spent within the desired glycemic range, reduced average blood glucose levels, and no rise in hypoglycemia rates11. Whenever possible, hospitals should integrate structured order sets to offer computerized guidance for managing glycemic levels effectively. Additionally, there is promising potential in utilizing machine learning algorithms and data from electronic health records (EHRs) to forecast insulin requirements during hospitalization 12.
Evidence supports the preadmission treatment of hyperglycemia in individuals scheduled for elective surgery as an effective strategy for reducing adverse outcomes. 4-12
Rationale for the recommendation
Delivering high-quality hospital care for diabetes necessitates the establishment of standards for care delivery, preferably implemented through structured order sets and quality improvement strategies. However, the consistent implementation of "best practice" protocols, reviews, and guidelines within hospitals remains a challenge. To address this issue, medical centers aiming for optimal inpatient diabetes treatment should develop protocols and structured order sets, incorporating computerized provider order entry (CPOE). Regular audits are recommended to monitor adherence to these protocols, alongside the implementation of educational and training programs to ensure staff remain updated. 3
During initial evaluation, it is essential to specify the type of diabetes, including type 1, type 2, gestational, pancreatogenic, drug-related, or nutrition-related, if known. Given that inpatient treatment and discharge planning are more effective when considering preadmission glycemia, A1C should be assessed for all individuals with diabetes or dysglycemia admitted to the hospital, particularly if no A1C test result is available from the previous three months 4-7. Additionally, admission assessments should include an evaluation of diabetes self-management knowledge and behaviors, with provision for diabetes self-management education, especially if a new treatment plan is being considered. This education should encompass knowledge and skills necessary for post-discharge management, including medication dosing and administration, glucose monitoring, and the recognition and treatment of hypoglycemia 8
To ensure lower incidence of complications during hospitalization, medical centers aiming for optimal inpatient diabetes treatment should develop protocols and structured order sets, incorporating computerized provider order entry (CPOE). Regular audits are recommended to monitor adherence to these protocols, alongside the implementation of educational and training programs to ensure staff remain updated 3.
l When caring of hospitalized patients with existing diabetes or stress induced hyperglycemia, consult with a diabetes specialized team whenever possible. (Conditional recommendation, moderate certainty evidence).13
Remarks
Specialized diabetes team should be responsible for caring with people with diabetes during hospitalization.
Summary of evidence
In a cross-sectional study that compared standard care to cases where diabetes specialists reviewed patient records and provided recommendations remotely through the Electronic Health Record (EHR), both hyperglycemia and hypoglycemia rates saw a reduction of 30-40% 13.
Rationale for the recommendation
The provision of care by adequately
trained specialists or specialized teams could potentially shorten hospital
stays and enhance outcomes related to glycemic control
14-15. Furthermore, the heightened risk of readmission within 30
days post-hospitalization associated with diabetes can be mitigated, resulting
in cost savings, through the delivery of inpatient care by a specialized team
focused on diabetes management 16-17.Diabetes specialized teams
could potentially shorten hospital stays and enhance outcomes related to
glycemic control.13
l Insulin should be initiated and intensified for the treatment of persistent hyperglycemia (blood glucose level ≥ 180 mg/dl on two occasions within 24 hours). (strong recommendation, high certainty evidence)18-22
l Once insulin therapy started, a glycemic goal of 140 – 180 mg/dl is recommended for most critically ill and noncritically ill hospitalized patients. (strong recommendation, high certainty evidence)18-22
l More stringent goals such as 100 – 140 mg/dl may be acceptable in some patients if these goals could be reached without significant hypoglycemia. (conditional recommendation, moderate certainty evidence)18-22
Remarks
Hyperglycemia among hospitalized patients is defined as blood glucose level ≥ 180 mg/dl on two occasions within 24 hours. A glycemic goal of 140 – 180 mg/dl is recommended for most critically ill and noncritically ill hospitalized patients.3
Summary of evidence
In a large clinical study carried out in a surgical intensive care unit (ICU), Van den Berghe et al. revealed that employing an intensive intravenous insulin regimen targeting a glycemic goal of 80-110 mg/dL resulted in a 40% reduction in mortality compared to a conventional approach aiming for a glycemic goal of 180-215 mg/dL in critically ill patients recently subjected to surgery. This investigation offered compelling evidence that actively managing blood glucose levels in hospitalized individuals could yield immediate advantages .18 Nevertheless, a significant multicenter follow-up investigation in critically ill hospitalized individuals, known as the Normoglycemia in Intensive Care Evaluation and Survival Using Glucose Algorithm Regulation (NICE-SUGAR) trial, prompted a reassessment of the optimal glucose-lowering target in critical illness. In this trial, critically ill patients randomly assigned to intensive glycemic control (aiming for 80-110 mg/dL) did not exhibit any substantial treatment advantage compared to a cohort with more moderate glycemic targets (140-180 mg/dL) and experienced slightly but notably higher mortality rates (27.5% vs. 25%). The group subjected to intensive management had markedly elevated rates of hypoglycemia, which likely contributed to the observed adverse outcomes.19
The findings from the NICE-SUGAR trial, corroborated by numerous meta-analyses and a randomized controlled trial, revealed heightened hypoglycemia rates and a rise in mortality associated with more aggressive glycemic control objectives compared to moderate glycemic targets 20-22. Based on these results, insulin should be initiated for the treatment of persistent hyperglycemia > 180 mg/dL. Once therapy is initiated, a glycemic goal of 140-180 mg/dL is recommended for most critically and noncritically ill individuals with hyperglycemia 3. In noncritical care settings, managing hyperglycemia in hospitalized patients typically involves aiming for a glycemic target of 100-180 mg/dL, whether the hyperglycemia is newly diagnosed (e.g., stress hyperglycemia or newly diagnosed diabetes) or is related to diabetes diagnosed prior to admission 3.
l For hospitalized individuals with diabetes who are consuming meals, it's recommended to conduct point-of-care (POC) blood glucose monitoring before meals. For those who are not eating, glucose monitoring every 4-6 hours is advised. However, when using intravenous insulin therapy, more frequent POC blood glucose monitoring, ranging from every 30 minutes to every 2 hours, is necessary to ensure safe implementation. (Good clinical practice, low certainty evidence)3, 23
l Basal insulin or a basal plus bolus correction insulin is the preferred treatment for noncritically ill hospitalized patients with poor oral intake or who are fasting. (strong recommendation, high certainty evidence )(29-35)
l An insulin regimen consisting of basal, prandial and correction components is the preferred treatment of for noncritically ill hospitalized patients with adequate nutritional intake. (strong recommendation, high certainty evidence)(24-30)
l The sole use of sliding scale insulin regimen in the inpatient settings is strongly discouraged. (strong recommendation, high certainty evidence )(24-30)
Remarks
In most cases, insulin is the preferred treatment for hyperglycemia in hospitalized individuals. Continuous intravenous insulin infusion stands out as the most effective approach for attaining specific glycemic targets and averting hypoglycemia in critical care settings. Beyond critical care units, scheduled subcutaneous insulin regimens are recommended for managing hyperglycemia in individuals with diabetes. For noncritically ill hospitalized patients with insufficient or limited oral intake, basal insulin or a basal plus bolus correction regimen is favored. Meanwhile, for most noncritically ill hospitalized individuals with diabetes who have adequate nutritional intake, an insulin regimen incorporating basal, prandial, and correction components is preferred. (31-35)
Summary of evidence
A randomized controlled trial has shown that basal plus bolus treatment improved glycemic outcomes and reduced hospital complications compared with a correction or supplemental insulin without basal insulin (formerly known as sliding scale) for people with type 2 diabetes admitted for general surgery.36
A prospective randomized inpatient study of 70/30 intermediate-acting (NPH)/ regular insulin mixture versus basal-bolus therapy showed comparable glycemic outcomes but significantly increased hypoglycemia in the group receiving insulin mixture.37Therefore, insulin mixtures such as 75/ 25, 70/30, or 50/50 insulins are not routinely recommended for in-hospital use. Data on the use of glargine U-300 and degludec U-100 or U-200 in the inpatient and perioperative settings are limited. A few studies have shown that they demonstrated similar efficacy and safety compared with glargine U-100.(38-42) At this time, there is no available evidence for weekly insulin use in hospital or surgical settings.3
· Top of Form
l For patients with type 2 diabetes who are admitted with heart failure (after recovery from the acute illness), it is recommended to initiate or continue SGLT 2 inhibitors during hospitalization and upon discharge, if there are no contraindications to this group. (Strong recommendation, high certainty evidence).36-41
Remarks
Individuals with type 2 diabetes hospitalized due to heart failure are advised to consider initiating or continuing the use of a sodium-glucose cotransporter 2 (SGLT2) inhibitor during hospitalization and upon discharge, provided there are no contraindications and after recovery from the acute illness. However, SGLT2 inhibitors should be avoided in cases of severe illness, ketonemia or ketonuria, and during prolonged fasting or surgical procedures.3
Proactive adjustment of diuretic dosing is advised during hospitalization and/or discharge, particularly in collaboration with a cardiology/heart failure consult team. Additionally, the FDA cautions that SGLT2 inhibitors should be stopped 3 days before scheduled surgeries (4 days in the case of ertugliflozin). 42
l In adult patients who are hospitalized for noncritical illness and experience hyperglycemia while receiving glucocorticoids (GCs) glycemic management should be pursued with either neutral protamine Hagedorn (NPH)-based insulin or basal bolus insulin (BBI) regimens (conditional recommendation, low certainty evidence) 43,44
Remarks
An NPH-based regimen may consist of NPH (with or without prandial insulin) given in divided doses depending on the timing, pharmacokinetics, and frequency of the specific GC being administered. NPH insulin may be added to BBI if the patient is already ont this regimen.45
Management of patients with GC-associated hyperglycemia requires ongoing BG monitoring with adjustment of insulin dosing. All therapies require safeguards to avoid hypoglycemia when doses of GCs are tapered or abruptly discontinued.45
The feasibility of implementing complex insulin regimens may be difficult for nursing personnel, placing additional burdens that have potential to affect patient safety. However, from a patient perspective, a once-daily morning NPH regimen may be easier to learn than multiple daily injections, particularly for patients who will be discharged home on GC therapy .45
patient perspective, a once-daily morning NPH regimen may be easier to learn than multiple daily injections, particularly for patients who will be discharged home on GC therapy.46
Summary of evidence
The systematic review identified 6 RCTs and 1 non-RCT to address this question (1. Much variability occurred among the studies regarding the insulin regimens used in both the NPH-based groups and the comparator groups. 47
NPH showed a lower BG (blood glucose ) in 4 RCTs while 2 RCTs found no difference and one observational study . No differences occurred in rates of hypoglycemia measured as the number of patients with an event or the number of events per patient in the 5 other studies that reported these data (all with very low certainty of evidence). In 2 RCTs, no differences occurred in hospital length of stay (low certainty evidence).48
Comments
These recommendations has been based on low-certainty evidence demonstrating similar glycemic outcomes for mean BG, hyperglycemia, hypoglycemia, and hospital LOS with NPH and BBI-based regimens for patients with GC-associated hyperglycemia in the hospital. Therefore it has been suggested that either NPH- or BBI-based regimens for glycemic management of GC-associated hyperglycemia. Neither regimen demonstrated cost, feasibility, acceptability, or equity advantages.49
Barriers to addressing GC-associated hyperglycemia may be bridged by establishing protocols and guidelines that outline best practices for achieving and maintaining glycemic control, such as administration of NPH at the same time as intermediate-acting GCs, such as prednisone or methylprednisolone. 49
l In select adult patients with mild hyperglycemia and T2D hospitalized for a noncritical illness, we suggest using either DPP4i with correction insulin or scheduled insulin therapy. (Conditional recommendation, low certainty evidence) 47
Remarks
Select patients include those with T2D that is moderately well-managed as reflected by a recent HbA1c < 7.5% (9.4 mmol/L), BG < 180 mg/dL (10 mmol/L), and, if on insulin therapy before hospitalization, to have a total daily insulin dose < 0.6 units/kg/day; this recommendation applies both to patients taking the DPP4i before admission and those who are not.34
Patients who develop persistently elevated BG [eg, >180 mg/dL (10 mmol/L)] on DPP4i therapy should be managed with scheduled insulin therapy; this recommendation does not apply to patients with T1D or other forms of insulin-dependent diabetes.34
Summary of evidence
Based on a metanalysis of 3 RCTs performed in individuals with established T2D prior to hospitalization, DPP4i dosed once daily compared with BBI therapy may provide no benefit on glycemic management . In select patients, there may be a reduced insulin requirement and lower frequency of hypoglycemic events [RR 0.27 (95% CI 0.09 to 0.84); low certainty evidence]. 47
The incidence of hypoglycemia was reduced with use of DPP4i in several trials; however, patients with impaired renal function and those considered to be at higher risk of hypoglycemia and hyperglycemia were excluded from enrollment. DPP4is are approved for use and considered safe in patients with any degree of kidney disease (note that dose adjustment for renal dysfunction is required for select DPP4is; eg, sitagliptin and alogliptin). Therefore, while patients with advanced kidney disease may benefit from reduced hypoglycemia, this remains unknown.1, 50
Of importance, a meta-analysis excluded those studies in which the intervention was a combination of DPP4i and scheduled insulin. However, all RCTs except 1 comparing DPP4i to BBI allowed the use of correction insulin for intermittent hyperglycemia. Finally, all RCTs included criteria for conversion to scheduled insulin therapy in the case of persistent hyperglycemia.1, 50
l For adult patients with diabetes undergoing elective surgical procedures, we suggest targeting preoperative hemoglobin A1c (HbA1c) levels < 8% (63.9 mmol/mol) and blood glucose (BG) concentrations 100 to 180 mg/dL (5.6 to10 mmol/L). (Conditional recommendation very low certainty evidence)(47,51, 52-62)
l For adult patients with diabetes undergoing elective surgical procedures, when targeting hemoglobin A1c (HbA1c) to < 8% (63.9 mmol/mol) is not feasible, we suggest targeting preoperative blood glucose (BG) concentrations 100 to 180 mg/dL (5.6 to 10 mmol/L). (Conditional recommendation, very low certainty evidence)(63-76)
Summary of evidence
A systematic review identified 44 observational (non-RCT) studies to address this question 47. A minority of patients with T1D were included, a single study recruited only patients with T1D 51, and many studies did not specify the type of diabetes. The majority of studies were performed in patients undergoing cardiac and orthopedic surgery, but other surgeries were included. Studies also used different cutoff values for HbA1c and BG concentrations and different strategies and interventions for peri- and intraoperative glucose control, which are likely to have affected outcomes. A meta-analysis of 11 non-RCTs that measured hospital length of stay comparing patients with a preoperative HbA1c < 7% vs ≥ 7% (<53 mmol/mol vs ≥53 mmol/mol) reported a shorter length of stay [MD −0.45 days (95% CI –0.89 to 0.00); very low level of certainty].(52-62)
In 10 non-RCTs, postoperative infections were less frequent in patients with a preoperative HbA1c < 7% vs ≥ 7% [<53 mmol/mol vs ≥53 mmol/mol; odds ratio 0.54 (95% CI 0.40 to 0.73); very low level of certainty] (52, 53, 58, 63-69). Similar findings were observed in 2 non-RCTs that compared patients with a preoperative HbA1c < 8% vs ≥ 8% [<63.9 mmol/mol vs ≥63.9 mmol/mol; odds ratio 0.83 (95% CI 0.15 to 4.63); very low level of certainty] (70,71). One study reported a HbA1c ≥ 7.8% (61.7 mmol/mol) as the threshold above which a significantly higher rate of wound complications occurred (70). The incidence of postoperative infections may also be reduced in patients with better glycemic measures in studies using different HbA1c cutoff levels (<6.5% vs ≥6.5%, <7.5% vs ≥7.5%, and < 8% vs ≥8%; <48.6 mmol/mol vs ≥48.6 mmol/mol, <58.5 mmol/mol vs ≥58.5 mmol/mol, and < 63.9 mmol/mol vs ≥63.9 mmol/mol, respectively) (72-76).
l In adult patients hospitalized for noncritical illness who are receiving enteral nutrition with diabetes-specific and nonspecific formulations, use neutral protamine Hagedorn (NPH)-based or basal bolus regimens. (conditional recommendation , low certainty evidence)(77-81)
Summary of evidence
The systematic review identified 2 systematic reviews, 1 RCT, and 3 non-RCTs that address this question. Studies found little to no difference in mean daily blood glucose between basal or basal bolus insulin vs NPH-based regimens with correctional (sliding scale) insulin. One observational (non- RCT) study found that the average hospital length of stay may be reduced by 1.57 days with NPH regimens compared to basal bolus (95% CI −1.71 to 4.85), and another found that the number of hypoglycemic events may result in an increase of 41% with basal-bolus compared to 70/30-biphasic insulin [IRR 2.92 (95% CI 0.70 to 12.20)], but these results are uncertain. No studies reported outcomes related to nurse time and effort. (77-81)
l Metformin administration should be withheld on the day of surgery. Other oral glucose-lowering agents should be withheld on the morning of the surgery or procedure, and instead, administer half of the NPH dose or 75-80% doses of long-acting analog insulin. (Good clinical practice )3
l SGLT2 inhibitors should be discontinued 3–4 days before surgery. (Good clinical practice )3
l Reducing basal insulin by 25% the evening before surgery, compared to usual dosing, is more likely to help achieve perioperative blood glucose goals with a reduced risk of hypoglycemia. (Good clinical practice, low certainty evidence )3
l In individuals undergoing noncardiac general surgery, utilizing basal insulin in combination with premeal short or rapid-acting insulin (basal-bolus) coverage has been linked to enhanced glycemic outcomes and reduced rates of perioperative complications compared to relying solely on reactive, correction-only short- or rapid-acting insulin coverage without basal insulin dosing.(Good clinical practice, low certainty evidence )3
l Treatment plans should be reviewed and changed as necessary to prevent hypoglycemia and recurrent hypoglycemia when a blood glucose value of <70 mg/dL (<3.9 mmol/L) is documented. (Conditional recommendation, low certainty evidence)3
Remarks
All health care professionals should be aware of hypoglycemia management protocol
Rationale for the recommendation
Numerous instances of inpatient hypoglycemia can be prevented. Therefore, it's essential for each hospital or hospital system to adopt and implement a hypoglycemia prevention and management protocol.3
l A structured discharge plan discharge plan should be tailored to the individual with diabetes. (strong recommendation, moderate certainty evidence )82
Remarks
Implementing a structured discharge plan customized to each individual can potentially decrease the length of hospital stay and reduce readmission rates while enhancing satisfaction with the hospital experience. Key strategies include providing diabetes self-management education before discharge, reconciling diabetes medications with a focus on access, and scheduling virtual and/or face-to-face follow-up visits post-discharge. It's crucial to initiate discharge planning upon admission and to continually update it as the individual's needs evolve 82.
It is recommended that an outpatient follow-up visit with the primary care clinician, endocrinologist, or diabetes care and education specialist within one month of discharge. If there have been changes to glycemic medications or if glucose management is not optimal at discharge, an earlier appointment (within 1-2 weeks) is preferred. In some cases, frequent contact may be necessary to prevent hyperglycemia and hypoglycemia.A discharge algorithm for adjusting glycemic medication, which takes into account admission A1C levels, diabetes medications before admission, and insulin usage during hospitalization, has been found to be useful in guiding treatment decisions and significantly improving A1C levels after discharge 5.
Rationale for the recommendation
Implementing a structured discharge plan tailored to each individual has the potential to reduce the length of hospital stay and lower readmission rates, all while enhancing satisfaction with the hospital experience.
- Acknowledgement
We would like to acknowledge the Diabetes and Endocrinology Scientific Committee for developing these guidelines.
Chair of the GDG: Mohamed Hesham El Hefnawy, National Institute of Diabetes and Endocrinology, Cairo
Members of the Guideline Development Group (GDG):
Yara Muhammad Ahmad Eid, Faculty of Medicine, Ain Shams University, Cairo.
Athar Reda Ibrahim, National Institute of Diabetes and Endocrinology, Cairo
Ahmed Mohamed Hamam, Military Hospital, Cairo
Amr Ali Mahfouz, National Institute of Diabetes and Endocrinology, Cairo
Atef Bassyouni, National Institute of Diabetes and Endocrinology, Cairo
Elsayed Abdel Fattah Eid, Faculty of Medicine, Delta University for science and technology, Dakahlia
Fawzy A F Elmessallamy, Faculty of Medicine, Zagazig University, Sharqia
Mai Mohammed Salama, National Hepatology and Tropical Medicine Research Institute, Cairo
Mohamed Abdelhady Mohamed Mashahet, Faculty of Medicine, Fayoum University, Fayoum.
Mohamed Abdelmoniem Elmikawy, police hospital, Cairo.
Randa Salam, Faculty of Medicine, Cairo University, Cairo.
The Egyptian Diabetes Guidelines Development Group gratefully acknowledges the contributions made for developing these guidelines by:
Bahaa Mohamed Sharaf Eldin, MSc Diabetes, University of South Wales, England (Egyptian Ministry of Health)
- Abbreviations
BGM
Blood Glucose Monitoring
DKA
Diabetic ketoacidosis
DDP4i
Dipeptidyl peptidase 4 inhibitors
GLP1
Glucagon like peptide 1
GRADE
Grading of Recommendations Assessment, Development and Evaluation
HbA1c
Glycated hemoglobin A1C
HHS
Hyperglycemic hyperosmolar state
POC
Point Of Care
RCT
Randomized controlled trial
SGLT 2
Sodium Glucose Co-transporter 2
SSI
Sliding Scale Insulin
- Glossary
HbA1c
Glycated haemoglobin by non-enzymatic attachment of glucose to haemoglobin. The concentration of HbA1c is the most commonly used measure of chronic glycaemia in clinical trials and diabetes management. It is considered to reflect the integrated mean glucose level over the previous 8–12 weeks.
POC
Point Of Care blood glucose monitoring
SSI
Use of correction or supplemental insulin without basal insulin
- Recommendations
|
▪️ A1C test should be performed for any patient with random blood glucose sample > 140 mg/dl during hospitalization or for any patient with a known history of diabetes. (strong recommendation) ▪️ Health care facilities should implement validated protocols for diabetes inpatient management (written or computerized) and delivered to all departments including: emergency units, ICU units, general wards, obstetrician/delivery units and dialysis suits. (strong recommendation) ▪️ When caring of hospitalized patients with existing diabetes or stress induced hyperglycemia, consult with a diabetes specialized team whenever possible. (conditional recommendation) ▪️ Insulin should be initiated and intensified for the treatment of persistent hyperglycemia (blood glucose level ≥ 180 mg/dl on two occasions within 24 hours). (strong recommendation) ▪️ Once insulin therapy started, a glycemic goal of 140 – 180 mg/dl is recommended for most critically ill and noncritically ill hospitalized patients. (strong recommendation) ▪️ More stringent goals such as 110-140 mg/dl may be acceptable in some patients if these goals could be reached without significant hypoglycemia. (conditional recommendation) ▪️ For hospitalized individuals with diabetes who are consuming meals, it's recommended to conduct point-of-care (POC) blood glucose monitoring before meals. For those who are not eating, glucose monitoring every 4-6 hours is advised. However, when using intravenous insulin therapy, more frequent POC blood glucose monitoring, ranging from every 30 minutes to every 2 hours, is necessary to ensure safe implementation. (Good clinical practice ) ▪️ Basal insulin or a basal plus bolus correction insulin is the preferred treatment for noncritically ill hospitalized patients with poor oral intake or who are fasting. (strong recommendation) ▪️ An insulin regimen consisting of basal, prandial and correction components is the preferred treatment for noncritically ill hospitalized patients with adequate nutritional intake. (strong recommendation) ▪️ The sole use of sliding scale insulin regimen in the inpatient settings is strongly discouraged. (strong recommendation) ▪️ For patients with type 2 diabetes who are admitted with heart failure (after recovery from the acute illness), it is recommended to initiate or continue SGLT 2 inhibitors during hospitalization and upon discharge, if there are no contraindications to this group. (strong recommendation) ▪️ In adult patients who are hospitalized for noncritical illness and experience hyperglycemia while receiving glucocorticoids (GCs) glycemic management should be pursued with either neutral protamine Hagedorn (NPH)-based insulin or basal bolus insulin (BBI) regimens (conditional recommendation) ▪️ In select adult patients with mild hyperglycemia and type 2 diabetes (T2D) hospitalized for a noncritical illness, we suggest using either dipeptidyl peptidase-4 inhibitor (DPP4i) with correction insulin or scheduled insulin therapy. (conditional recommendation) ▪️ For adult patients with diabetes undergoing elective surgical procedures, we suggest targeting preoperative hemoglobin A1c (HbA1c) levels < 8% (63.9 mmol/mol) and blood glucose (BG) concentrations 100 to 180 mg/dL (5.6 to10 mmol/L). (conditional recommendation) ▪️ For adult patients with diabetes undergoing elective surgical procedures, when targeting hemoglobin A1c (HbA1c) to < 8% (63.9 mmol/mol) is not feasible, we suggest targeting preoperative blood glucose (BG) concentrations 100 to 180 mg/dL (5.6 to 10 mmol/L). (conditional recommendation) ▪️ In adult patients hospitalized for noncritical illness who are receiving enteral nutrition with diabetes-specific and nonspecific formulations, we suggest using neutral protamine Hagedorn (NPH)-based or basal bolus regimens. (conditional recommendation) ▪️ Metformin administration should be withheld on the day of surgery. Other oral glucose-lowering agents should be withheld on the morning of the surgery or procedure, and instead, administer half of the NPH dose or 75-80% doses of long-acting analog insulin. (Good clinical practice ) ▪️ SGLT2 inhibitors should be discontinued 3–4 days before surgery. (Good clinical practice) ▪️ Reducing basal insulin by 25% the evening before surgery, compared to usual dosing, is more likely to help achieve perioperative blood glucose goals with a reduced risk of hypoglycemia. (Good clinical practice ) ▪️ In individuals undergoing noncardiac general surgery, utilizing basal insulin in combination with premeal short or rapid-acting insulin (basal-bolus) coverage has been linked to enhanced glycemic outcomes and reduced rates of perioperative complications compared to corrective rapid/short acting insulin dosing. (Good clinical practice ) ▪️ Treatment plans should be reviewed and changed as necessary to prevent hypoglycemia and recurrent hypoglycemia when a blood glucose value of <70 mg/dL (<3.9 mmol/L) is documented. (Conditional recommendation) ▪️ A structured discharge plan tailored to the individual may reduce the length of hospital stay and readmission rates and increase satisfaction with the hospital experience. (strong recommendation) |
- Introduction
Hyperglycemia, hypoglycemia and blood glucose variability are frequently observed in hospitalized individuals, regardless of whether they have diabetes or not. This condition is prevalent among both medical and surgical patients and is associated with adverse health outcomes. Patients with dysglycemia are very prone to different complications such as pneumonia, acute renal failure, sepsis, urinary tract infections, prolonged hospital stays, and post-admission disability. These complications contribute to increased morbidity and mortality rates. The pathophysiology of hyperglycemia in hospitalized patients involves several mechanisms. During stressful situations like hospitalization, there is an excessive secretion of counter-regulatory hormones such as glucagon, growth hormone, epinephrine, and cortisol, which elevate blood glucose levels by promoting hepatic glucose production. Additionally, the overproduction of pro-inflammatory cytokines disrupts carbohydrate metabolism and reduces the peripheral utilization of glucose in skeletal muscles and adipose tissue, leading to hyperglycemia. High-quality hospital care for diabetes requires standards for care delivery, which are best implemented using structured order sets and quality improvement strategies for process improvement. Unfortunately, "best practice" protocols, reviews, and guidelines are inconsistently implemented within hospitals. We will focus in this guideline on the value of established protocols in every health care facility and the benefits of implementing these protocols1.
Adult patients with diabetes account for 25% of noncritically ill hospitalized patients . Another 12% to 25% of hospitalized patients experience hyperglycemia, defined as blood glucose (BG) > 140 mg/dL 2.
Diabetes management in the inpatient setting is facilitated by identification and treatment of hyperglycemia prior to elective procedures, a dedicated inpatient diabetes management service applying validated standards of care, and a proactive transition plan for outpatient diabetes care with timely prearranged follow-up appointments. These steps can improve outcomes, shorten hospital stays, and reduce the need for readmission and emergency department visits 2.
This guidelines document is intended to provide guidance regarding inpatient hospital management of hyperglycemia for critically ill and non-critically ill patients. The guidelines cover the patient pathway within hospital including required laboratory assessment, diagnostic blood sugar cut-off values and glycemic goals recommended for critically ill and non-critical ill inpatients. The guidelines provide clear recommendations for insulin and non-insulin therapies and measures to prevent and monitor hypoglycemia.
As up to 20% of individuals undergoing general surgery have diabetes, and 23–60% have prediabetes or undiagnosed diabetes, the guidelines provide a suggested approach for perioperative management for these patients regarding pharmacological therapy dose adjustment and monitoring 2. Finally Discharge procedure and transition of patients to outpatient clinic care is detailed within this guidance
- Scope and purpose
The objectives of this guidelines is
- To provide guidance for the proper management of dysglycemia during hospital stay.
- To provide guidance on the validated protocols needed for proper management of the proper way to implement these protocols.
- Target audience
This guideline targets; healthcare professionals, policy makers, national diabetes program managers, as well as non-governmental organizations (NGO) and other stakeholders to afford the most appropriate tool for individuals with diabetes.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2015 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in a table:
• Developing organization/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt the American Diabetes Association – Standards of Care in Diabetes – 2024 and The Endocrine society guidelines for inpatient management of hyperglycemia 2022. 2, 3
- Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of
Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
• GRADE working group: https://www.gradeworkinggroup.org/
• GRADE online training modules: http://cebgrade.mcmaster.ca/
• GRADE profile software: http://ims.cochrane.org/revman/gradepro
Table 1 Quality and Significance of the four levels of evidence in GRADE:

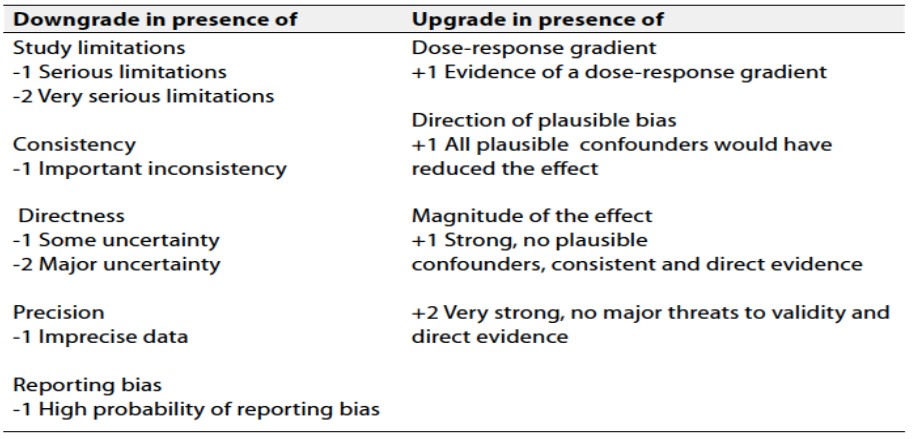
Table 2 Factors that determine How to upgrade or downgrade the quality of evidence
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation- Research needs
Clinical trials to evaluate the use of other insulin regimens during hospitalization such as basal plus insulin regimen. Also further research is needed to evaluate the efficacy and safety of newer agents such as SGLT 2 inhibitors and glucagon like peptide receptor agonists.
- Updating of the guidelines
These guidelines will be updated whenever there is new evidence.
- References
1. Umpierrez, G. E., & Pasquel, F. J. (2017). Management of inpatient hyperglycemia and diabetes in older adults. Diabetes care, 40(4), 509-517.
2. Korytkowski MT, Muniyappa R, Antinori-Lent K, Donihi AC, Drincic AT, Hirsch IB, Luger A, McDonnell ME, Murad MH, Nielsen C, Pegg C, Rushakoff RJ, Santesso N, Umpierrez GE. Management of Hyperglycemia in Hospitalized Adult Patients in Non-Critical Care Settings: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2022 Jul 14;107(8):2101-2128. doi: 10.1210/clinem/dgac278. PMID: 35690958; PMCID: PMC9653018.
3. American Diabetes Association Professional Practice Committee; 16. Diabetes Care in the Hospital: Standards of Care in Diabetes—2024. Diabetes Care 1 January 2024; 47 (Supplement_1): S295–S306. https://doi.org/10.2337/dc24-S016
4. Garg R, Schuman B, Bader A, et al. Effect of preoperative diabetes management on glycemic control and clinical outcomes after elective surgery. Ann Surg 2018;267:858–862
5. van den Boom W, Schroeder RA, Manning MW, Setji TL, Fiestan GO, Dunson DB. Effect of A1C and glucose on postoperative mortality in noncardiac and cardiac surgeries. Diabetes Care 2018;41:782–788
6. Setji T, Hopkins TJ, Jimenez M, et al. Rationalization, development, and implementation
7. of a preoperative diabetes optimization program designed to improve perioperative outcomes and reduce cost. Diabetes Spectr 2017;30:217–223
8. Okabayashi T, Shima Y, Sumiyoshi T, et al. Intensive versus intermediate glucose control in
9. surgical intensive care unit patients. Diabetes Care 2014;37:1516–1524
10. Institute of Medicine. Preventing Medication Errors. Aspden P, Wolcott J, Bootman JL, Cronenwett LR, Eds. Washington, DC, National Academies Press, 2007
11. Sly B, Russell AW, Sullivan C. Digital interventions to improve safety and quality of inpatient diabetes management: a systematic review. Int JMed Inform 2022;157:104596
12. Nguyen, M., Jankovic, I., Kalesinskas, L., Baiocchi, M., & Chen, J. H. (2021). Machine learning for initial insulin estimation in hospitalized patients. Journal of the American Medical Informatics Association, 28(10), 2212-2219.
13. Rushakoff, R. J., Sullivan, M. M., MacMaster, H. W., Shah, A. D., Rajkomar, A., Glidden, D. V., & Kohn, M. A. (2017). Association between a virtual glucose management service and glycemic control in hospitalized adult patients: an observational study. Annals of Internal Medicine, 166(9), 621-627.
14. Akiboye, F., Sihre, H. K., Al Mulhem, M., Rayman, G., Nirantharakumar, K., & Adderley, N. J. (2021). Impact of diabetes specialist nurses on inpatient care: A systematic review. Diabetic Medicine, 38(9), e14573.
15. Wang, Y. J., Seggelke, S., Hawkins, R. M., Gibbs, J., Lindsay, M., Hazlett, I., ... & Draznin, B. (2016). Impact of glucose management team on outcomes of hospitalization in patients with type 2 diabetes admitted to the medical service. Endocrine Practice, 22(12), 1401-1405.
16. Bansal, V., Mottalib, A., Pawar, T. K., Abbasakoor, N., Chuang, E., Chaudhry, A., ... & Hamdy, O. (2018). Inpatient diabetes management by specialized diabetes team versus primary service team in non-critical care units: impact on 30-day readmission rate and hospital cost. BMJ Open Diabetes Research and Care, 6(1), e000460.
17. Ostling, S., Wyckoff, J., Ciarkowski, S. L., Pai, C. W., Choe, H. M., Bahl, V., & Gianchandani, R. (2017). The relationship between diabetes mellitus and 30-day readmission rates. Clinical diabetes and endocrinology, 3, 1-8.
18. Van den Berghe, G., Wouters, P., Weekers, F., Verwaest, C., Bruyninckx, F., Schetz, M., ... & Bouillon, R. (2001). Intensive insulin therapy in critically ill patients. New England journal of medicine, 345(19), 1359-1367.
19. Finfer S, Chittock DR, Su SY, et al.; NICESUGAR Study Investigators. Intensive versus conventional glucose control in critically ill patients. N Engl J Med 2009;360:1283–1297
20. Kansagara D, Fu R, Freeman M, Wolf F, Helfand M. Intensive insulin therapy in hospitalized patients: a systematic review. Ann Intern Med 2011;154:268–282
21. Sathya B, Davis R, Taveira T,Whitlatch H,Wu WC. Intensity of peri-operative glycemic control and postoperative outcomes in patients with diabetes: a meta-analysis. Diabetes Res Clin Pract 2013;102:8–15
22. Umpierrez G, Cardona S, Pasquel F, et al. Randomized controlled trial of intensive versus conservative glucose control in patients undergoing coronary artery bypass graft surgery: GLUCO-CABG trial. Diabetes Care 2015;38:1665–1672.
23. Zhang R, Isakow W, Kollef MH, Scott MG. Performance of a modern glucose meter in ICU
and general hospital inpatients: 3 years of real-world paired meter and central laboratory results. Crit Care Med 2017;45:1509–1514
24. Umpierrez GE, Smiley D, Jacobs S, et al. Randomized study of basal-bolus insulin therapy in the inpatient management of patients with type 2 diabetes undergoing general surgery (RABBIT 2 surgery). Diabetes Care 2011;34:256–261
25. Colunga-Lozano LE, Gonzalez Torres FJ, Delgado-Figueroa N, et al. Sliding scale insulin for non-critically ill hospitalised adults with diabetes mellitus. Cochrane Database Syst Rev 2018;11: CD011296
26. Migdal AL, Fortin-Leung C, Pasquel F,Wang H, Peng L, Umpierrez GE. Inpatient glycemic control with sliding scale insulin in noncritical patients with type 2 diabetes: who can slide? J Hosp Med 2021;16:462–468
27. Bellido V, Suarez L, Rodriguez MG, et al. Comparison of basal-bolus and premixed insulin regimens in hospitalized patients with type 2 diabetes. Diabetes Care 2015;38:2211–2216
28. Galindo RJ, Pasquel FJ, Vellanki P, et al. Degludec hospital trial: a randomized controlled trial comparing insulin degludec U100 and glargine U100 for the inpatient management of patients with type 2 diabetes. Diabetes Obes Metab 2022; 24:42–49
29. Pasquel FJ, Lansang MC, Khowaja A, et al. A randomized controlled trial comparing glargine U300 and glargine U100 for the inpatient management of medicine and surgery patients with type 2 diabetes: glargine U300 hospital trial. Diabetes Care 2020;43:1242–1248
30. Perez A, Carrasco-S_anchez FJ, Gonz_alez C, et al. Efficacy and safety of insulin glargine 300 U/mL (Gla-300) during hospitalization and therapy intensification at discharge in patients with insufficiently controlled type 2 diabetes: results of the phase IV COBALTA trial. BMJ Open Diabetes Res Care 2020;8:e001518
31. Brown KE, Hertig JB. Determining current insulin pen use practices and errors in the inpatient setting. Jt Comm J Qual Patient Saf 2016;42:568–575, AP1–AP7
32. Horne J, Bond R, Sarangarm P. Comparison of inpatient glycemic control with insulin vials versus insulin pens in general medicine patients. Hosp Pharm2015;50:514–521
33. Veronesi G, Poerio CS, Braus A, et al. Determinants of nurse satisfaction using insulin pen devices with safety needles: an exploratory factor analysis. Clin Diabetes Endocrinol 2015;1:15
34. Najmi U, Haque WZ, Ansari U, et al. Inpatient insulin pen implementation, waste, and potential cost savings: a community hospital experience. J Diabetes Sci Technol 2021;15:741–747
35. Bueno E, Benitez A, Rufinelli JV, et al. Basal bolus regimen with insulin analogues versus human insulin in medical patients with type 2 diabetes: a randomized controlled trial in Latin America. Endocr Pract 2015;21:807–813
36. Galindo RJ, Dhatariya K, Gomez-Peralta F, Umpierrez GE. Safety and efficacy of inpatient diabetes management with non-insulin agents an overview of international practices. Curr Diab Rep 2022;22:237–246
37. Pasquel FJ, Lansang MC, Dhatariya K, Umpierrez GE. Management of diabetes and hyperglycaemia in the hospital. Lancet Diabetes Endocrinol 2021;9:174–188
38. Umpierrez GE, Gianchandani R, Smiley D, et al. Safety and efficacy of sitagliptin therapy for the inpatient management of general medicine and surgery patients with type 2 diabetes: a pilot, randomized, controlled studyDiabetes Care 2013;36:3430–3435
39. Fushimi N, Shibuya T, Yoshida Y, Ito S, Hachiya H, Mori A. Dulaglutide-combined basal plus correction insulin therapy contributes to ideal glycemic control in non-critical hospitalized patients. J Diabetes Investig 2020;11:125–131
40. Fayfman M, Galindo RJ, Rubin DJ, et al. A randomized controlled trial on the safety and efficacy of exenatide therapy for the inpatient management of general medicine and surgery patients with type 2 diabetes. Diabetes Care 2019;42:450–456
41. Perez-Belmonte LM, Osuna-S_anchez J, Mill_an-G_omez M, et al. Glycaemic efficacy and safety of linagliptin for the management of noncardiac surgery patients with type 2 diabetes in a real-world setting: Lina-Surg study. Ann Med 2019;51:252–261
42. U.S. Food and Drug Administration. FDA revises labels of SGLT2 inhibitors for diabetes to include warnings about too much acid in the blood and serious urinary tract infections. Accessed 21 August 2023. Available from https:// www.fda.gov/drugs/drug-safety-and-availability/ fda-revises-labels-sglt2-inhibitors-diabetes-include -warnings-about-too-much-acid-blood-and-serious
43. Seisa MO, Saadi S, Nayfeh T, et al. A systematic review supporting the endocrine society clinical practice guideline for the management of hyperglycemia in adults hospitalized for non-critical illness or undergoing elective surgical procedures. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgac277.
44. Ruiz de Adana MS, Colomo N, Maldonado-Araque C, et al. Randomized clinical trial of the efficacy and safety of insulin glargine vs. NPH insulin as basal insulin for the treatment of glucocorticoid induced hyperglycemia using continuous glucose monitoring in hospitalized patients with type 2 diabetes and respiratory disease. Diabetes Res Clin Pract. 2015;110(2):158-165.
45. Delfs N, Struja T, Gafner S, et al. Outcomes of hospitalized patients with glucocorticoid-induced hyperglycemia—a retrospective analysis. J Clin Med. 2020;9(12):4079.
46. Radhakutty A, Burt MG. Management of endocrine disease: critical review of the evidence underlying management of glucocorticoid induced hyperglycaemia. Eur J Endocrinol. 2018;179(4):R207 -R218.
47. Seisa MO, Saadi S, Nayfeh T, et al. A systematic review supporting the endocrine society clinical practice guideline for the management of hyperglycemia in adults hospitalized for non-critical illness or undergoing elective surgical procedures. J Clin Endocrinol Metab. https://doi.org/10.1210/clinem/dgac277
48. Ruiz de Adana MS, Colomo N, Maldonado-Araque C, et al. Randomized clinical trial of the efficacy and safety of insulin glargine vs. NPH insulin as basal insulin for the treatment of glucocorticoid induced hyperglycemia using continuous glucose monitoring in hospitalized patients with type 2 diabetes and respiratory disease. Diabetes Res Clin Pract. 2015;110(2):158-165.
49. Gerards MC, Tervaert EC, Hoekstra JB, Vriesendorp TM, Gerdes VE. Physician’s attitudes towards diagnosing and treating glucocorticoid induced hyperglycaemia: sliding scale regimen is still widely used despite guidelines. Diabetes Res Clin Pract. 2015;109(2):246-252.
50. Umpierrez GE, Gianchandani R, Smiley D, et al. Safety and efficacy of sitagliptin therapy for the inpatient management of general medicine and surgery patients with type 2 diabetes: a pilot, randomized, controlled study. Diabetes Care. 2013;36(11):3430-3435.
51. Nyström T, Holzmann MJ, Eliasson B, Kuhl J, Sartipy U. Glycemic control in type 1 diabetes and long-term risk of cardiovascular events or death after coronary artery bypass grafting. J Am Coll Cardiol. 2015;66(5):535-543
52. Halkos ME, Lattouf OM, Puskas JD, et al. Elevated preoperative hemoglobin A1c level is associated with reduced long-term survival after coronary artery bypass surgery. Ann Thorac Surg. 2008;86(5):1431-1437.
53. Ramadan M, Abdelgawad A, Elshemy A, et al. Impact of elevated glycosylated hemoglobin on hospital outcome and 1 year survival of primary isolated coronary artery bypass grafting patients. Egyptian Heart J. 2018;70(2):113-118.
54. Festejo Villamiel KM, Yao C, Sioson M. Enhanced recovery after surgery (ERAS) outcomes in patients with prior diagnosis of diabetes. J ASEAN Fed Endocr Soc. 2019;34(1):73-79.
55. Lenguerrand E, Beswick AD, Whitehouse MR, Wylde V, Blom AW. Outcomes following hip and knee replacement in diabetic versus nondiabetic patients and well versus poorly controlled diabetic patients: a prospective cohort study. Acta Orthop. 2018;89(4):399-405.
56. Knapik P, Cieśla D, Filipiak K, Knapik M, Zembala M. Prevalence and clinical significance of elevated preoperative glycosylated hemoglobin in diabetic patients scheduled for coronary artery surgery. Eur J Cardiothorac Surg. 2011;39(4):484-489.
57. Arslan U, Memetoğlu ME, Kutlu R, et al. Preoperative HbA1c level in prediction of short-term morbidity and mortality outcomes following coronary artery bypass grafting surgery. Russian Open Med J. 2015;4(2):e0204.
58. Kim HJ, Shim JK, Youn YN, Song JW, Lee H, Kwak YL. Influence of preoperative hemoglobin A1c on early outcomes in patients with diabetes mellitus undergoing off-pump coronary artery bypass surgery. J Thorac Cardiovasc Surg. 2020;159(2):568-576.
59. Abu Tailakh M, Ishay SY, Awesat J, Poupko L, Sahar G, Novack V. Hemoglobin A1c in patients with diabetes predict long-term mortality following coronary artery surgery. J Clin Med.2021;10(12):2739.
60. Ali F, Khilji S, Tahir S. Impact of elevated glycosylated haemoglobin on outcomes following CABG in patients with diabetes mellitus. Pakistan J Med Health Sci. 2020;14(4):1167-1169.
61. Özmen R, Tunçay A, Yılmaz H, Sarı G, Taşdemir HK. The importance of HbA1c and left ventricular ejection fraction in predicting the development of postoperative mortality and complications in coronary artery bypass graft surgery. Braz J Cardiovasc Surg. Published online July 7, 2021; doi:10.21470/1678-9741-2020-0542
62. Shoghli M, Jain R, Boroumand M, et al. Association of preoperative hemoglobin A1c with in-hospital mortality following valvular heart surgery. Braz J Cardiovasc Surg. 2020;35(5):654-659.
63. Godshaw BM, Ojard CA, Adams TM, Chimento GF, Mohammed A, Waddell BS. Preoperative glycemic control predicts perioperative serum glucose levels in patients undergoing total joint arthroplasty. J Arthroplasty. 2018;33(7s):S76-S80.
64. Strahan S, Harvey RM, Campbell-Lloyd A, Beller E, Mundy J, Shah P. Diabetic control and coronary artery bypass: effect on short-term outcomes. Asian Cardiovasc Thorac Ann. 2013;21(3):281-287.
65. O’Sullivan CJ, Hynes N, Mahendran B, et al. Haemoglobin A1c (HbA1c) in non-diabetic and diabetic vascular patients: is HbA1c an independent risk factor and predictor of adverse outcome? Eur J Vasc Endovasc Surg. 2006;32(2):188-197.
66. Dronge AS, Perkal MF, Kancir S, Concato J, Aslan M, Rosenthal RA. Long-term glycemic control and postoperative infectious complications. Arch Surg. 2006;141(4):375-380.
67. Lamloum SM, Mobasher LA, Karar AH, et al. Relationship between postoperative infectious complications and glycemic control for diabetic patients in an orthopedic hospital in Kuwait. Med Princ Pract. 2009;18(6):447-452.
68. Iorio R, Williams KM, Marcantonio AJ, Specht LM, Tilzey JF, Healy WL. Diabetes mellitus, hemoglobin A1c, and the incidence of total joint arthroplasty infection. J Arthroplasty. 2012;27(5):726- 729.e721.
69. Zhuang T, Shapiro LM, Fogel N, Richard MJ, Gardner MJ, Kamal RN. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg. 2021;46(8):675-684.e610.
70. Cunningham DJ, Baumgartner RE, Federer AE, Richard MJ, Mithani SK. Elevated preoperative hemoglobin A1c associated with increased wound complications in diabetic patients undergoing primary, open carpal tunnel release. Plast Reconstr Surg. 2019;144(4):632e-638e.
71. Colibaseanu DT, Osagiede O, McCoy RG, et al. Proactive protocolbased management of hyperglycemia and diabetes in colorectal surgery patients. Endocr Pract. 2018;24(12):1073-1085.
72. Sato H, Carvalho G, Sato T, Lattermann R, Matsukawa T, Schricker T. The association of preoperative glycemic control, intraoperative insulin sensitivity, and outcomes after cardiac surgery. J Clin Endocrinol Metab. 2010;95(9):4338-4344.
73. Matsuura K, Imamaki M, Ishida A, Shimura H, Niitsuma Y, Miyazaki M. Off-pump coronary artery bypass grafting for poorly controlled diabetic patients. Ann Cardiothorac Surg. 2009;15(1):18-22.
74. Peng W, Liang Y, Lu T, et al. Multivariate analysis of incision infection after posterior lumbar surgery in diabetic patients: a single center retrospective analysis. Medicine. 2019;98(23):e15935.
75. Hwang JS, Kim SJ, Bamne AB, Na YG, Kim TK. Do glycemic markers predict occurrence of complications after total knee arthroplasty in patients with diabetes? Clin Orthop Relat Res. 2015;473(5):1726-1731.
76. Han HS, Kang SB. Relations between long-term glycemic control and postoperative wound and infectious complications after total knee arthroplasty in type 2 diabetics. Clin Orthop Surg. 2013;5(2):118-123.
77. Laesser CI, Cumming P, Reber E, Stanga Z, Muka T, Bally L. Management of glucose control in noncritically ill, hospitalized patients receiving parenteral and/or enteral nutrition: a systematic review. J Clin Med. 2019;8(7):935.
78. Vercoza Viana M, Vercoza Viana L, Tavares AL, de Azevedo MJ. Insulin regimens to treat hyperglycemia in hospitalized patients on nutritional support: systematic review and meta-analyses. Ann Nutr Metab. 2017;71(3-4):183-194.
79. Hijaze D, Szalat A. Retrospective evaluation of glycemic control with basal-bolus or neutral protamine Hagedorn insulin regimens in patients receiving continuous enteral nutrition therapy in medicine wards. Nutr Clin Pract. 2017;32(4):557-562.
80.Hsia E, Seggelke SA, Gibbs J, Rasouli N, Draznin B. Comparison of 70/30 biphasic insulin with glargine/lispro regimen in noncritically ill diabetic patients on continuous enteral nutrition therapy. Nutr Clin Pract. 2011;26(6):714-717.
81. Korytkowski MT, Salata RJ, Koerbel GL, et al. Insulin therapy and glycemic control in hospitalized patients with diabetes during enteral nutrition therapy: a randomized controlled clinical trial. Diabetes Care. 2009;32(4):594-596
82. Gregory, N. S., Seley, J. J., Dargar, S. K., Galla, N., Gerber, L. M., & Lee, J. I. (2018). Strategies to prevent readmission in high-risk patients with diabetes: the importance of an interdisciplinary approach. Current diabetes reports, 18, 1-7.