Phenylalanine hydroxylase deficiency in children
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Phenylalanine hydroxylase deficiency in children |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 16 Feb 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Introduction
- - Methods
- - Evidence to recommendations: Considerations
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline.
- - Updating of the guideline
- - References
- - Annexes
- Executive Summary
➡️Introduction
Phenylketonuria (PKU) is an inborn error of metabolism (IEM) caused by inherited deficiency of the enzyme phenylalanine hydroxylase (PAH), which catalyzes the hydroxylation of phenylalanine (Phe) to generate tyrosine (Tyr) [20]. It is considered the most common IEM of amino acid metabolism; estimated worldwide average incidence 1:15000 [21], and of 2638 live births in Egypt [24]. It has been long known that introduction of treatment as early as possible can ameliorate the neurotoxic sequalae of Phe on the developing brain tissue, hence the importance of newborn screening aiming at early diagnosis and management. [22].
➡️Scope
This guideline focuses on diagnosis and management of PAH deficiency in children in Egypt. It is developed to help health care personnel (general practitioners, general pediatricians, family medicine physicians, neonatologists, nutritionists, dieticians, psychologists and nurses aiming at preventing morbidity and neurocognitive disabilities in PKU
Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- The complete European guidelines on phenylketonuria : diagnosis and treatment. Orphanet J Rare Dis. 2017 Oct 12 ;12(1) :162
2- Breast feeding in infants diagnosed with phenylketonuria (PKU) : a scoping review. BMJ Paediatr Open. 2023 Oct ;7(1): e002066
3- Epilepsy and vaccinations : Italian guidelines. Epilepsia, 54(Suppl. 7) :13–22, 2013
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Adaptation for 2 recommendations according to GRADE criteria to be suitable to our Economic implications (Evidence-to-Decision (EtD) table was done)
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. Diagnosis of Phenylketonuria (PAH deficiency)
This guideline emphasizes the best practice on diagnosis of PAH deficiency based on phenylalanine level, type and timing of blood sample and the role of molecular testing in children’s age group from day 1 to 18 years.
B. Management of PAH deficiency
This section includes recommendations and good practice statements on dietary management, follow up and monitoring of PKU children.
C. Prevention of neurocognitive impairments in PAH deficiency
This section includes recommendations and good practice statements on monitoring to prevent neurocognitive impairment of PKU children.
We can summarize the guidelines’ recommendations in the following:
▪️ We recommend that newborn screening should be considered as a national obligation as this has proven to be cost effective, (Intermediate quality evidence, strong recommendation).
▪️ In the differential diagnosis of hyperphenylalaninemia, of any degree, we suggest that BH4 deficiency should be excluded by measuring petrins and dihydropetredine reductase activity in dried blood spot, (low quality evidence, Weak (conditional) recommendation).
▪️ As petrins and dihydropetredine reductase activity in dried blood spot tests are not available in Egypt, careful follow up of patients neurodevelopment after positive newborn screening is highly recommended, (Good practice statement).
▪️ To maintain blood levels in the recommended range, we suggest That patients with PAH deficiency can be classified as either: Not requiring treatment, Requiring diet, BH4 or both, (Very low quality evidence, Weak (conditional) recommendation).
▪️ Patient genotyping should be considered for diagnosing BH4 responsiveness and may help to define the metabolic phenotype and could help in prevention by offering reproductive options, (Very low quality evidence, Weak (conditional) recommendation).
▪️ We suggest that all patients with untreated blood Phe levels >360 µmol/I should be treated. Treatment should start as soon as possible, ideally before 10 days of age, (Very low quality evidence, Weak (conditional) recommendation)
▪️ We suggest that no treatment is recommended when untreated blood levels are <360 µmol/I. But, monitoring of blood Phe (at a lower frequency) until 1 year of age as a minimum is recommended to determine whether levels rise above 360 µmol/l, (Very low quality evidence, Weak (conditional) recommendation).
▪️ We suggest that patients with untreated Phe levels 360-600 µmol/I should be treated until the age of 12 years, (Very low quality evidence, Weak (conditional) recommendation).
▪️ We recommend that, in treated PKU patients up to the age of 12 years, target Phe level should be 120-360 µmol/L, (Intermediate quality evidence, strong recommendation).
▪️ We suggest that, in treated PKU patients aged ≥ 12 years target Phe level should be 120-600 µmol/L, (Very low quality evidence, Weak (conditional) recommendation).
· We recommend that blood Phe levels should be measured to monitor metabolic control, as they are the most clinically relevant biomarkers, (Intermediate quality evidence, strong recommendation).
▪️ We suggest that there is insufficient evidence to support routine evaluation of the Phe fluctuations and the measurement of Phe/tyr ratio in PKU, (Very low quality evidence, Weak (conditional) recommendation).
▪️ We suggest that frequency of blood PHE measurement should be at minimum:
0-1 year Weekly
1-12 years fortnightly
>12 years monthly, (Good practice statement).
▪️ We suggest that all PKU patients should be followed up in a specialized metabolic center, (Very low quality evidence, Weak (conditional) recommendation).
▪️ We suggest that an annual nutritional review is required for any patient who is prescribed in low Phe diet or is self-restricting high protein food. Such a clinical review includes a clinical examination including the anthropometric parameters (weight, height, BMI). It is also recommended that plasma amino acids, plasma homocysteine, and /or methylmalonic acid, hemoglobin, MCV and ferritin are measured. All other micronutrients (vitamins, minerals including calcium, zinc, selenium) or hormones (parathyroid hormones) can be considered if clinically indicated. (low quality evidence, Weak (conditional) recommendation).
▪️ Outpatient clinic visit should be at minimum, given good clinical and metabolic control:
0-1 year: every 2 months
1-18 years: twice per year, (Good practice statement).
▪️ We suggest that health professional support is required throughout life to encourage normal healthy feeding behaviors, with a positive acceptance of a low Phe diet, (Low quality evidence, Weak (conditional) recommendation).
▪️ We suggest that Phe intake should be methodically titrated until Phe is consistently maintained within the Phe target range. If there is stability of control, a challenge with additional Phe should be performed systematically to maximize natural protein permitted, Phe deficiency should be avoided. (Low quality evidence, Weak (conditional) recommendation).
▪️ We suggest that total protein intake should supply the age-related safe level of protein intake (FAQ/WHO/UNU 2007) with an additional 40% from L- amino acid supplements. (Low quality evidence, Weak (conditional) recommendation).
▪️ In PKU, the nutritional intake of energy, macronutrients and micronutrients, should meet the average estimated amounts/ dietary reference values for healthy population. For all age groups, there should be focus on achieving balanced intake of all nutrients, avoiding catabolism or deficiency but preventing excess of any nutrient that may lead to over nutrition or toxicity. Supplementary nutrients (vitamins, minerals and LC-PUFA's) added to Phe -free L-amino acid supplements should be in the amounts that will at least meet normal population dietary reference values. Assessment of dietary intake should be performed in every clinic visit with extra attention directed to patients who are non-adherent, do not have prescribed Phe free L-amino acid supplements (with added micronutrients) or who are at the higher risk of nutritional deficiency, (Low quality evidence, Weak (conditional) recommendation).
▪️ We recommend that fruits and vegetables (except potatoes containing Phe ≤ 75 mg/100 g of food can be safely given without measurements or estimation in a low Phe-diet without loss of Phe control. Some unrestricted fruits and vegetables should be encouraged in the diet in early life to encourage long-term healthy feeding patterns. (Intermediate quality evidence, Strong recommendation).
▪️ We recommend that the artificial sweetener aspartame, particularly from beverages and tables top sweetener’s is best avoided in patients on a low Phe-diet. (Intermediate quality evidence, Strong recommendation).
▪️ The Phe-free protein substitute, in the form of L-amino acids, should be provided in any patient with PKU treated with a low Phe diet consuming less than the FAQ/WHO/UNU 2007 safe levels of natural protein intake. The Phe-free L-amino acid formula should be evenly administered at least 3 times throughout the day. To aid adherence, all patients with PKU should receive a choice of suitable age-appropriate Phe-free L-amino acid supplements. (Intermediate quality evidence, strong recommendation).
▪️ In infants with PKU Breast-Feeding in combination with a Phe-free infant L-amino acid formula should be encouraged, (Low quality evidence, Weak (conditional) recommendation).
▪️ Breast-Feeding
Techniques:
Demand Breast-Feeding with Supplementation: Breast-feed on demand but give a
measured volume of Phe-free infant formula before breast-feeding to reduce
breast milk stimulation and Phe intake.
Alternating Method: Alternate between breast-feeding and Phe-free L-amino acid
infant formula bottle-feeding to achieve acceptable blood Phe control.
Individualized Feeding Plans: Each infant may require a tailored approach based
on their individual blood Phe levels and responses to feeding techniques.
(Low quality evidence, Weak (conditional) recommendation).
▪️ We suggest that in children with PKU, during illness to prevent an excessive rise in blood Phe concentration, it is prudent to encourage the intake of Phe-free L-amino acid supplements and high carbohydrate supplements, (Very low quality evidence, Weak (conditional) recommendation).
▪️ Neurocognitive
evaluations should be performed at 12 and 18 years of age in all patients.
If any of the
stated risk factors applies, perform (additional) neurocognitive tests:
-Non-optimal metabolic control; >50% of the Phe levels are out of target
range over period of 6-12 months (depending on age <12 or 212 years).
-Problems at school or work; not showing progress in
school for 6 months
- Concern of parents / caregivers or family
-Concern of PKU patient
-Concern of metabolic team
Proposed domains of neurocognitive testing:
IQ perception/visuospatial functioning, EF (divided into inhibitory control, working memory and cognitive flexibility), and motor control.
(Good practice statement).
▪️ It is advisable to measure and discuss psychosocial functioning and wellbeing during clinic visits in PKU as this has been demonstrated to improve psychosocial outcomes in patients with other chronic diseases. This should be done in the easiest possible way for each center by interview, paper or electronically, (Intermediate quality evidence, strong recommendation).
▪️ Health related quality of life should be discussed at least annually in the outpatient clinic. It is suggested to consider a questionnaire (preferably PKU specific) to support this at least once during childhood, adolescence and adulthood and during major changes in life, (Very low quality evidence, Weak (conditional) recommendation).
▪️ Adaptive issues (e.g. clinically relevant behavioral problems) should be discussed on an annual basis. Adaptive issues should be screened at 12 and 18 years together with neurocognitive functioning. When adaptive problems occur, a psychology referral should be sought, (Good practice statement).
▪️ Neuroimaging should not be performed in routine care but reserved for PKU patients presenting with unexpected clinical course/and or unexpected neurological deficit, (Very low quality evidence, Weak (conditional) recommendation).
▪️ Patients with PKU should receive the compulsory vaccines and extra vaccines without restriction even if they have convulsions, (Good practice statement).
▪️ We recommend that vaccinations should be performed without contraindication in children with idiopathic or symptomatic epilepsy (Intermediate quality evidence, Strong recommendation).
▪️ We recommend that the risk of epilepsy should not discourage parents from vaccinating their children (Intermediate quality evidence, Strong recommendation).
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/- Recommendations
|
Table 3. Recommendations |
|
|
|||
|
A. Diagnosis of PAH deficiency in children |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A1 |
What is the best clinical practice in diagnosis of asymptomatic neonates and symptomatic children with PAH deficiency? |
The complete European guidelines 2017 |
Newborn screening should be considered as a national obligation as this has proven to be cost effective.
|
Intermediate
|
strong |
|
In the differential diagnosis of hyperphenylalaninemia, of any degree, BH4 deficiency should be excluded by measuring petrins and dihydropetredine reductase activity in dried blood spot.
As these tests are not available in Egypt, careful follow up of patients neurodevelopment after positive newborn screening is highly recommended |
Low |
Weak (conditional)
|
|||
|
Good practice statement |
|||||
|
A2 |
What is the best clinical practice in classification of diagnosed PAH deficiency patients in pediatric age group?
|
The complete European guidelines 2017 |
To maintain blood levels in the recommended range, patients with PAH deficiency can be classified as either: A- Not requiring treatment B- Requiring diet, BH4 or both.
|
Very low |
Weak (conditional) |
|
A3 |
what is the role of molecular testing in management of diagnosed PAH deficiency in pediatric age group? |
The complete European guidelines 2017 |
Patient genotyping should be considered for diagnosing BH4 responsiveness and may help to define the metabolic phenotype and could help in prevention by offering reproductive options.
|
Very low |
Weak (conditional) |
|
A. Management of PAH deficiency in children |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
What is the Phe level and timing at which dietary treatment should be started in asymptomatic neonates and in children with PAH deficiency? (figure (1)
|
The complete European guidelines 2017 |
All patients with untreated blood Phe levels >360 µmol/I should be treated.
Treatment should start as soon as possible, ideally before 10 days of age.
|
Very low |
Weak (conditional)
|
|
No treatment is recommended when untreated blood levels are <360 µmol/I. But, monitoring of blood Phe (at a lower frequency) until 1 year of age as a minimum is recommended to determine whether levels rise above 360 µmol/l. |
Very low |
Weak (conditional)
|
|||
|
Patients with untreated Phe levels 360-600 µmol/I should be treated until the age of 12 years. |
Very low |
Weak (conditional)
|
|||
|
B2 |
What is the target Phe level for optimal management of PAH deficiency in pediatrics age group? Figure (2)
|
The complete European guidelines 2017 |
In treated PKU patients up to the age of 12 years, target Phe level should be 120-360 µmol/L.
|
intermediate |
Strong |
|
In treated PKU patients aged ≥ 12 years target Phe level should be 120-600 µmol/L. |
Very low |
Weak (conditional) |
|||
|
B3 |
What is the best practice in monitoring in PAH deficiency in different pediatric age groups?
|
The complete European guidelines 2017 |
Blood Phe levels should be measured to monitor metabolic control, as they are the most clinically relevant biomarkers
|
intermediate |
Strong |
|
There is insufficient evidence to support routine evaluation of the Phe fluctuations and the measurement of Phe/tyr ratio in PKU
|
Very low |
Weak (conditional) |
|||
|
Frequency of blood PHE measurement should be at minimum: 0-1 year Weekly 1-12 years fortnightly >12 years monthly
|
|
Good practice statement |
|||
|
B4 |
What is the best practice in monitoring growth and development in patients with PAH deficiency in pediatric age group?
|
The complete European guidelines 2017 |
All PKU patients should be followed up in a specialized metabolic center
|
Very low |
Weak (conditional) |
|
An annual nutritional review is required for any patient who is prescribed in low Phe diet or is self-restricting high protein food. Such a clinical review includes a clinical examination including the anthropometric parameters (weight, height, BMI).
It is also recommended that plasma amino acids, plasma homocysteine, and /or methylmalonic acid, hemoglobin, MCV and ferritin are measured.
all other micronutrients (vitamins, minerals including calcium, zinc, selenium) or hormones (parathyroid hormones) can be considered if clinically indicated.
|
low |
Weak (conditional) |
|||
|
|
Outpatient clinic visit should be at minimum, given good clinical and metabolic control: 0-1 year: every 2 months 1-18 years: twice per year
|
|
Good practice statement |
||
|
|
|
Health professional support is required throughout life to encourage normal healthy feeding behaviors, with a positive acceptance of a low Phe diet
|
low |
Weak (conditional) |
|
|
B5 |
What is the Phe, protein, and caloric requirements for children with PAH deficiency? |
The complete European guidelines 2017 |
Phe intake should be methodically titrated until Phe is consistently maintained within the Phe target range. If there is stability of control, a challenge with additional Phe should be performed systematically to maximize natural protein permitted, Phe deficiency should be avoided.
|
low |
Weak (conditional) |
|
Total protein intake should supply the age-related safe level of protein intake (FAQ/WHO/UNU 2007) with an additional 40% from L- amino acid supplements.
|
Low |
Weak (conditional)
|
|||
|
In PKU, the nutritional intake of energy, macronutrients and micronutrients, should meet the average estimated amounts/ dietary reference values for healthy population.
For all age groups, there should be focus on achieving balanced intake of all nutrients, avoiding catabolism or deficiency but preventing excess of any nutrient that may lead to over nutrition or toxicity.
Supplementary nutrients (vitamins, minerals and LC-PUFA's) added to Phe -free L-amino acid supplements should be in the amounts that will at least meet normal population dietary reference values.
Assessment of dietary intake should be performed in every clinic visit with extra attention directed to patients who are non-adherent, do not have prescribed Phe free L-amino acid supplements (with added micronutrients) or who are at the higher risk of nutritional deficiency
|
Low
|
Weak (conditional)
|
|||
|
|
|
|
Fruits and vegetables (except potatoes containing Phe ≤ 75 mg/100 g of food can be safely given without measurements or estimation in a low Phe-diet without loss of Phe control. Some unrestricted fruits and vegetables should be encouraged in the diet in early life to encourage long-term healthy feeding patterns.
|
Intermediate
|
strong
|
|
The artificial sweetener aspartame, particularly from beverages and tables top sweetener’s is best avoided in patients on a low Phe-diet.
|
Intermediate |
Strong |
|||
|
The Phe-free protein substitute, in the form of L-amino acids, should be provided in any patient with PKU treated with a low Phe diet consuming less than the FAQ/WHO/UNU 2007 safe levels of natural protein intake.
The Phe-free L-amino acid formula should be evenly administered at least 3 times throughout the day
To aid adherence, all patients with PKU should receive a choice of suitable age-appropriate Phe-free L-amino acid supplements.
|
Intermediate |
Strong |
|||
|
B6 |
What is the best practice for introducing breast milk in infants with confirmed PAH deficiency |
The complete European guidelines 2017 |
In infants with PKU Breast-Feeding in
|
low |
Weak (conditional) |
|
Breast feeding in infants diagnosed with phenylketonuria (PKU) 2023 |
Alternating Method: Alternate between breast-feeding and Phe-free L-amino acid infant formula bottle-feeding to achieve acceptable blood Phe control.
|
low |
Weak (conditional) |
||
|
B7 |
Is there specific dietary manipulation needed during management of PKU patients subjected to acute illness?
|
The complete European guidelines 2017 |
In children with PKU, during illness to prevent an excessive rise in blood Phe concentration, it is prudent to encourage the intake of Phe-free L-amino acid supplements and high carbohydrate supplements
|
Very low |
Weak (conditional) |

- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Solaf M Elsayed |
Professor of Medical Genetics, Medical Genetics Department, Ain Shams University, Cairo, Egypt |
Overseeing the medical genetics group. Participating in retrieval of guidelines. Communication with relevant authorities for permission to use the guidelines. Formulating health questions, guideline appraisal, participation in writing and revision and editing the final version of the guidelines |
|||
|
Shaimaa Gad |
Lecturer of Medical Genetics, Medical Genetics Dept. Ain Shams University, Cairo Egypt |
Participating in retrieval of guidelines. Communication with relevant authorities for permission to use the guidelines. Formulating health questions, guideline appraisal, participation in writing the guidelines |
|||
|
Heba Allah Hosny |
Consultant and fellow of Medical and Clinical Genetics, National institute of Neuromotor System, Cairo, Egypt |
Participating in retrieval of guidelines. Communication with relevant authorities for permission to use the guidelines. Formulating health questions, guideline appraisal. participation in writing the guidelines |
|||
|
Rana Mahrous |
Researcher of Medical genetics, National Research Centre, Cairo, Egypt |
Participating in retrieval of guidelines. Communication with relevant authorities for permission to use the guidelines Formulating health questions, guideline appraisal, participation in writing the guidelines |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia. 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia. 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Associate Professor of Pediatrics Ain Shams University, Egypt |
Developing evidence to decision (EtD) frameworks, participating in search and guideline appraisal |
||||
|
- Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. - Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. - Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revising the whole document. |
||||
|
Dr. Lamis Mohsen Elsholkamy
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revising the whole document. |
|||
|
Dr. Ahmed Mahmoud Youssef
|
Fellow of Pediatrics, General Organization for Teaching Hospitals and Institutes (GOTHI). Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revising the whole document. |
|||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revising the whole document. |
|||
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revising the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Eman Marzouk |
Prof. of pediatrics genetics, Alexandria University, Cairo, Egypt |
||||
|
Iman Ehsan Abdel Meguid |
Prof. of pediatrics genetics, Cairo University, Cairo, Egypt |
||||
|
International Peer Reviewers |
|||||
|
Anita MacDonald |
Consultant Dietitian in Inherited Metabolic Disorders at Birmingham Children’s Hospital and an Honorary Professor in Dietetics at Plymouth University, UK. |
||||
|
Júlio César Rocha |
Professor Auxiliar / Assistant Professor Campo Mártires da Pátria, Lisboa | Portugal |
||||
|
Saeedeh Talebi |
Pediatrician, PhD in Nutrition |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
▪️The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge:
- The complete European guidelines on phenylketonuria: diagnosis and treatment. Orphanet J Rare Dis. 2017 Oct 12 ;12(1):162
- Breast feeding in infants diagnosed with phenylketonuria (PKU): A scoping review. BMJ Paediatr Open. 2023 Oct ;7(1): e002066
- Epilepsy and vaccinations: Italian guidelines Epilepsia, 54(Suppl. 7):13–22, 2013
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️ This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
CPG |
Clinical Practice Guideline |
||
|
DHS |
Demographic and Health Survey |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ERG |
External Review Group |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
IEM |
Inborn errors of Metabolism |
||
|
LNAAs |
large neutral amino acids |
||
|
|
|
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
||
|
PAH |
Phenylalanine Hydroxylase |
||
|
Phe |
Phenylalanine |
||
|
PKU |
Phenylketonuria |
||
|
Tyr |
Tyrosine |
||
|
QoL |
Quality of life |
- Introduction
Phenylketonuria (PKU) is a rare inherited metabolic disorder caused by a deficiency of the enzyme phenylalanine hydroxylase (PAH), responsible for breaking down phenylalanine to Tyrosine (Tyr). Being a rare disease, the estimated global incidence is approximately 1 in 10,000 to 15,000 live births. However, the prevalence can vary significantly by geographic region and ethnic background [23]. The incidence among Egyptians was estimated to be 1: 2638 live births, with higher rates in Lower Egypt and South Sinai [24]. This higher rate is attributed to the high rate of consanguineous marriages in the country, that reaches 35.3% on average, especially among first cousins (86%) [25].
The PAH gene is located on chromosome 12, and over 1,000 pathogenic variants have been identified in this gene eventually leading to a reduction or complete absence of functional PAH enzyme [26]. High Phe concentrations may disrupt brain development, myelination, and neurotransmitter synthesis. The transport of large neutral amino acids (LNAAs) within the brain tissue is hindered by increased Phe concentration and this explains the deficiency of dopamine and serotonin in the brain tissue of PKU patients [27].
Because of widespread ոеwborn screening programs, overt clinical manifestations of РKU are rare. Neonates are asymptomatic until the initiation of feeds containing Phe (e.g., breast milk or standard infant formula). The serious manifestations of PKU include irreversible intellectual disability, developmental delays, seizures, behavioral abnormalities, microcephaly, and eczematous rash, decreased skin pigmentation and in some cases gait abnormalities [27].
The cornerstone in the diagnosis of PKU, is the direct estimation of Phe level in blood. The most useful laboratory method for screening for PKU is tandem mass spectrometry. In addition to being a high through output method, it can measure additional amino acids including tyrosine and acylcarnitine ester. A high concentration of Phe together with elevated Phe/tyrosine ratio suggests the diagnosis of PKU [28]. Enzyme assay is not performed to confirm the diagnosis, because PΑH activity is expressed only in the liver [27].
Once the biochemical diagnosis is established, treatment should be initiated as soon as possible to avoid irreversible brain damage. Lifetime dietary restriction of Phe has been the mainstay of treatment of PKU children [27]. The diet consists of Phe-free protein substitute in addition to normal (intact) protein calculated in reference to the age and body weight of each child and adjusted according to Phe tolerance. The intact protein includes breast milk, standard infant formula in neonates or vegetarian diet in older children [29].
Rocha and Macdonald, 2016 stated that the goal of dietary management is to reduce the blood phenylalanine concentration into the treatment range of 120–360 μmol/L (2–6 mg/dL) and to provide sufficient macronutrients and micronutrients essential for normal growth [29].
Sapropterin, a biologically active synthetic form of ΒH4, was approved by the US Food and Drug Administration (FDA) in December 2007 [30]. Ѕарrοptеriո may be used as an adjunct to dietary restriction in only ΡΚU patients who are responsive to ѕарrорtеriո [31].
The prognosis of PKU patients vary significantly depending on the timing of initiation of therapy and effectiveness of treatment. Early diagnosis and strict adherence to the phenylalanine-restricted diet from a young age can greatly improve outcomes. With appropriate management, individuals with PKU can often achieve normal or near-normal intellectual and cognitive development, as well as a good quality of life.
➡️Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement guidance on diagnosis and management of PAH deficiency in children. It provides guidance and recommendations to primary health care providers, general practitioners, general pediatricians, family medicine physicians, neonatologists, nutritionists, psychologists, and nurses aiming at preventing morbidity and neurocognitive disabilities in PAH in children.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for searching are:
Phenylketonuria, PKU, Phenylalanine hydroxylase, PAH, nutritional management, PKU treatment.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 3 guidelines:
Guideline (1): The complete European guidelines on phenylketonuria: diagnosis and treatment. Orphanet J Rare Dis. 2017 Oct 12;12(1):162
Guideline (2): Breast feeding in infants diagnosed with phenylketonuria (PKU): a scoping review. BMJ Paediatr Open. 2023 Oct;7(1): e002066
Guideline (3): Epilepsy and vaccinations: Italian guidelines. Epilepsia, 54(Suppl. 7):13–22, 2013
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Adaptation for two recommendations according to GRADE criteria to be suitable to our Economic implications (Evidence to Decision (EtD) table was done)
- Development of Good Practice Statement.
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups: the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of medical genetics and pediatric genetics/metabolic specialists.
The main functions of the clinical panel were adolopment of PAH deficiency Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing the strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinician’s subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 2 clinical national experts who have interest and expertise in as well as eminent international reviewers.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interest:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations (both ETD and changing strength of recommendation were not done in this guideline).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequity, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is necessary about actual healthcare practice
2- Have large net positive consequences (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations, however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops a group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing PAH diagnosis, treatment, and prevention of neurocognitive impairment strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts (figure 1,2).
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️Reminders: the provision of information verbally, on paper or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️Educational meetings: lectures, workshops or training courses, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physicians for diagnosis and algorithm for management of acute malnutrition
3. Arabic Educational materials for nurses and mothers
- Limitations and suggestions for further research needs
Future research recommendations for the management of PKU in children in the Egyptian context could include:
▪️ Documents for detailed available food allowed with Phe content.
▪️ An educational supplementary material to schools, teachers, and nurses
▪️ A guideline for management of PKU in adults and in pregnanacy
▪️Adding the new therapies once available in Egypt
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective management strategies for PAH defeciency in children.
➡️Challenges
▪️ The small number of specialized physicians in relation to the large number of newly diagnosed PKU patients each year.
▪️ The maldistribution of specialized metabolic clinics being focused on the capital and large universities
▪️ The lack of specialized nurses and health workers in metabolic specialty
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for PKU in children:
1. Adherence to PKU Guidelines
▪️ Numerator: Number of children with PAH deficiency who received treatment as per guideline recommendations.
▪️ Denominator: Total number of children diagnosed with PAH deficiency.
▪️ Data Source: Metabolic outpatients/ clinic patient records.
2. Neurocognitive outcome
▪️ Numerator: Total number of children with PAH deficiency with average IQ and neurocognitive functions.
▪️ Denominator: Total number of children diagnosed with PAH deficeinc.
▪️ Data Source: Metabolic outpatients/ clinic patient records..
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization and the success of the treatment in preventing further complications (neurocognitive outcome).
- Updating of the guideline
The EPG genetics GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
2. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
3. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
4. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
5. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
6. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
7. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
8. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
9. Williams RA, Mamotte CD, Burnett JR. Phenylketonuria: an inborn error of phenylalanine metabolism. Clin Biochem Rev. 2008 Feb;29(1):31-41.
10. Shoraka HR, Haghdoost AA, Baneshi MR, Bagherinezhad Z, Zolala F. Global prevalence of classic phenylketonuria based on Neonatal Screening Program Data: systematic review and meta-analysis. Clin Exp Pediatr. 2020 Feb;63(2):34-43.
11. Al Hafid N, Christodoulou J. Phenylketonuria: a review of current and future treatments. Transl Pediatr. 2015 Oct;4(4):304-17.
12. Hillert A, Anikster Y, Belanger-Quintana A, Burlina A, Burton BK, Carducci C, Chiesa AE, Christodoulou J,ĐorđevićM, Desviat LR, Eliyahu A, Evers RAF, Fajkusova L, Feillet F, Bonfim-Freitas PE, Giżewska M, Gundorova P, Karall D, Kneller K, Kutsev SI, Leuzzi V, Levy HL, Lichter-Konecki U, Muntau AC, Namour F, Oltarzewski M, Paras A, Perez B, Polak E, Polyakov AV, Porta F, Rohrbach M, Scholl-Bürgi S, Spécola N, StojiljkovićM, Shen N, Santana-da Silva LC, Skouma A, van Spronsen F, Stoppioni V, Thöny B, Trefz FK, Vockley J, Yu Y, Zschocke J, Hoffmann GF, Garbade SF, Blau N. The Genetic Landscape and Epidemiology of Phenylketonuria. Am J Hum Genet. 2020;107(2):234. Epub 2020 Jul 14.
13. Abdelaziz AE, Fahim M, Samy S, Abu Elsood H, Afifi S, Khafagy H, Abd El Fattah M, Elgunidy N, Kandeel A. Incidence and Risk Factors of Congenital Hypothyroidism and Phenylketonuria in Egypt: Results from Egypt National Newborn Screening Program, 2018-2021. TEPHINET.
14. Shawky RM., El-Awady MY., Elsayed SM., Hamadan GE. Consanguineous matings among Egyptian population. Egyptian Journal of Medical Human Genetics. 2011 ; 12 (2) : 157-163.
15. Chen, T., Xu, W., Wu, D. et al. Mutational and phenotypic spectrum of phenylalanine hydroxylase deficiency in Zhejiang Province, China. Sci Rep 2018 ; 8, 17137.
16. van Spronsen FJ, Blau N, Harding C, Burlina A, Longo N, Bosch AM. Phenylketonuria.
a. Nat Rev Dis Primers. 2021 ;7(1):36. Epub 2021 May 20.
17. Lowe TB, DeLuca J, Arnold GL. Similarities and differences in key diagnosis, treatment, and management approaches for PAH deficiency in the United States and Europe. Orphanet J Rare Dis. 2020 Sep 25;15(1):266.
18. Rocha JC, MacDonald A. Dietary intervention in the management of phenylketonuria: current perspectives. Pediatric Health Med Ther. 2016 ; (1)7:155-163.
19. U.S. Food and Drug Administration Center for Drug Evaluation and Research. Kuvan. www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseaction=Search.DrugDetails (Accessed on January 07, 2008).
20. Douglas TD, Jinnah HA, Bernhard D, Singh RH. The effects of sapropterin on urinary monoamine metabolites in phenylketonuria. Mol Genet Metab. 2013 ;109(3):243. Epub 2013 May 1.
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Solaf M Elsayed |
Professor of Medical Genetics, Medical Genetics Dept, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Shaimaa Gad |
Lecturer of Medical Genetics,Medical Genetics Dept., Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Heba Allah Hosny |
Medical Genetics, national institute of Neuromotor System, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Rana Mahrous |
Researcher of Medical genetics, National Research Centre, Cairo, Egypt |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanis Sulieman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
|
|
|
|
|
International Peer Reviewers |
|
|
|
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for "Guideline for PAH deficiency in children. " on PubMed are: ……Phenylketonuria, phenylalanine hydroxylase deficiency, PKU treatment, PKU management .
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for The complete European guidelines on phenylketonuria: diagnosis and treatment.
|
AGREE II/ CPGs |
………… |
|
Domain 1 (Scope) |
%93 |
|
Domain 2 (Stakeholder) |
%92 |
|
Domain 3 (Rigour) |
%94 |
|
Domain 4 (Clarity) |
%96 |
|
Domain 5 (Applicability) |
%70 |
|
Domain 6 (Independence) |
%100 |
|
Overall assessment. |
%100 |
|
Recommend for use (Overall assessment .) |
YES |
Results of the AGREE II assessment of the three source guidelines for Epilepsy and vaccinations: Italian guidelines.
|
AGREE II/ CPGs |
………… |
|
Domain 1 (Scope) |
%61 |
|
Domain 2 (Stakeholder) |
%37 |
|
Domain 3 (Rigour) |
%65 |
|
Domain 4 (Clarity) |
%89 |
|
Domain 5 (Applicability) |
%22 |
|
Domain 6 (Independence) |
%86 |
|
Overall assessment. |
%83 |
|
Recommendation for use (Overall assessment .) |
YES, with modifications |
PRISMA-ScR-scoping review-Checklist, Breast feeding in infants diagnosed with phenylketonuria (PKU) a scoping review.
|
SECTION |
ITEM |
PRISMA-ScR CHECKLIST ITEM |
REPORTED ON PAGE # |
|
TITLE |
|||
|
Title |
1 |
Identify the report as a scoping review. |
P1 |
|
ABSTRACT |
|||
|
Structured summary |
2 |
Provide a structured summary that includes (as applicable): background, objectives, eligibility criteria, sources of evidence, charting methods, results, and conclusions that relate to the review questions and objectives. |
P1 |
|
INTRODUCTION |
|||
|
Rationale |
3 |
Describe the rationale for the review in the context of what is already known. Explain why the review questions/objectives lend themselves to a scoping review approach. |
P1 |
|
Objectives |
4 |
Provide an explicit statement of the questions and objectives being addressed with reference to their key elements (e.g., population or participants, concepts, and context) or other relevant key elements used to conceptualize the review questions and/or objectives. |
P1 |
|
METHODS |
|||
|
Protocol and registration |
5 |
Indicate whether a review protocol exists; state if and where it can be accessed (e.g., a Web address); and if available, provide registration information, including the registration number. |
Methods P2 |
|
Eligibility criteria |
6 |
Specify characteristics of the sources of evidence used as eligibility criteria (e.g., years considered, language, and publication status), and provide a rationale. |
Methods P2 |
|
Information sources* |
7 |
Describe all information sources in the search (e.g., databases with dates of coverage and contact with authors to identify additional sources), as well as the date the most recent search was executed. |
Methods P2 till September 22 |
|
Search |
8 |
Present the full electronic search strategy for at least 1 database, including any limits used, such that it could be repeated. |
Methods P2 |
|
Selection of sources of evidence† |
9 |
State the process for selecting sources of evidence (i.e., screening and eligibility) included in the scoping review. |
Methods P2 |
|
Data charting process‡ |
10 |
Describe the methods of charting data from the included sources of evidence (e.g., calibrated forms or forms that have been tested by the team before their use, and whether data charting was done independently or in duplicate) and any processes for obtaining and confirming data from investigators. |
Methods P2 |
|
Data items |
11 |
List and define all variables for which data were sought and any assumptions and simplifications made. |
P4 |
|
Critical appraisal of individual sources of evidence§ |
12 |
If done, provide a rationale for conducting a critical appraisal of included sources of evidence; describe the methods used and how this information was used in any data synthesis (if appropriate). |
Not available |
|
Synthesis of results |
13 |
Describe the methods of handling and summarizing the data that were charted. |
Results P3 |
|
RESULTS |
|||
|
Selection of sources of evidence |
14 |
Give numbers of sources of evidence screened, assessed for eligibility, and included in the review, with reasons for exclusions at each stage, ideally using a flow diagram. |
Methods P4 |
|
Characteristics of sources of evidence |
15 |
For each source of evidence, present characteristics for which data were charted and provide the citations. |
Not available |
|
Critical appraisal within sources of evidence |
16 |
If done, present data on critical appraisal of included sources of evidence (see item 12). |
Not available |
|
Results of individual sources of evidence |
17 |
For each included source of evidence, present the relevant data that were charted that relate to the review questions and objectives. |
P4 |
|
Synthesis of results |
18 |
Summarize and/or present the charting results as they relate to the review questions and objectives. |
Results P5 |
|
DISCUSSION |
|||
|
Summary of evidence |
19 |
Summarize the main results (including an overview of concepts, themes, and types of evidence available), link to the review questions and objectives, and consider the relevance to key groups. |
P5 |
|
Limitations |
20 |
Discuss the limitations of the scoping review process. |
Limitation of the study P5 |
|
Conclusions |
21 |
Provide a general interpretation of the results with respect to the review questions and objectives, as well as potential implications and/or next steps. |
Discussion P6 |
|
FUNDING |
|||
|
Funding |
22 |
Describe sources of funding for the included sources of evidence, as well as sources of funding for the scoping review. Describe the role of the funders of the scoping review. |
Funding for the scoping review P7 |
JBI = Joanna Briggs Institute; PRISMA-ScR = Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews.
* Where sources of evidence (see second footnote) are compiled from, such as bibliographic databases, social media platforms, and Web sites.
† A more inclusive/heterogeneous term used to account for the different types of evidence or data sources (e.g., quantitative and/or qualitative research, expert opinion, and policy documents) that may be eligible in a scoping review as opposed to only studies. This is not to be confused with information sources (see first footnote).
‡ The frameworks by Arksey and O’Malley (6) and Levac and colleagues (7) and the JBI guidance (4, 5) refer to the process of data extraction in a scoping review as data charting.
§ The process of systematically examining research evidence to assess its validity, results, and relevance before using it to inform a decision. This term is used for items 12 and 19 instead of "risk of bias" (which is more applicable to systematic reviews of interventions) to include and acknowledge the various sources of evidence that may be used in a scoping review (e.g., quantitative and/or qualitative research, expert opinion, and policy document).
From: Tricco AC, Lillie E, Zarin W, O'Brien KK, Colquhoun H, Levac D, et al. PRISMA Extension for Scoping Reviews (PRISMAScR): Checklist and Explanation. Ann Intern Med. 2018;169:467–473. doi: 10.7326/M18-0850.
Annex Table 3. Annex Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
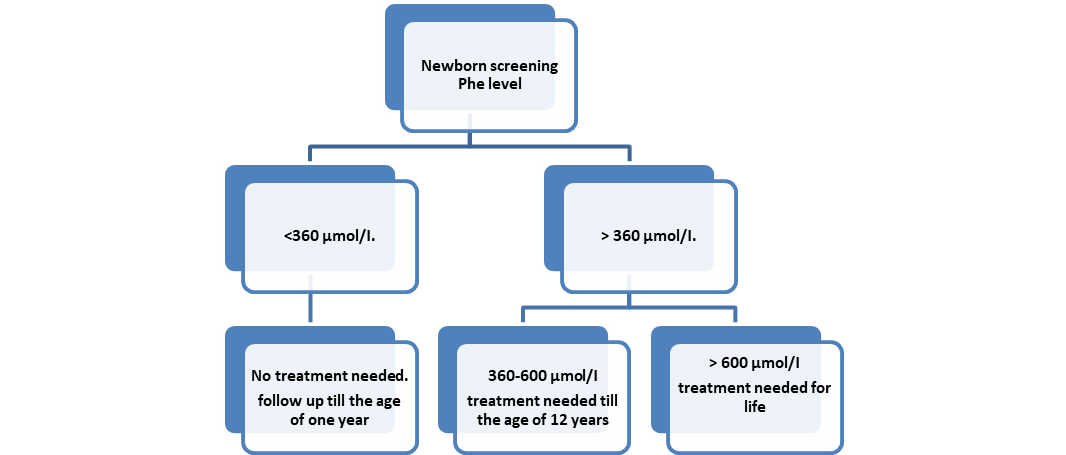
Figure (1): Treatment of PAH deficiency according to Phe level
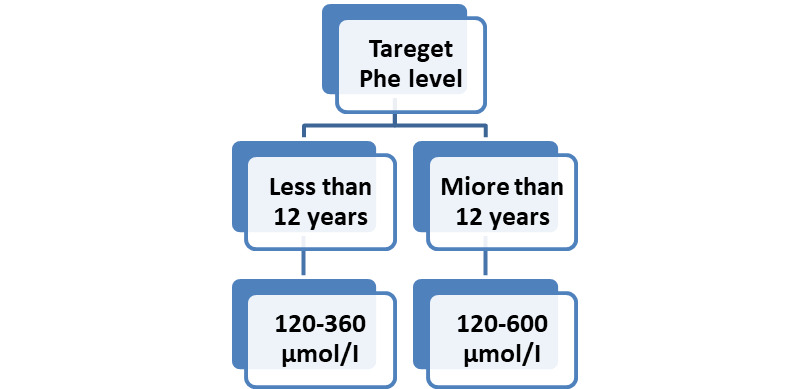
Figure (2): Target Phe levels for optimal management in PAH deficiency