the Diagnosis and Treatment of Familial Mediterranean Fever during Childhood and Adolescence
| الموقع: | EHC | Egyptian Health Council |
| المقرر الدراسي: | طب الأطفال |
| كتاب: | the Diagnosis and Treatment of Familial Mediterranean Fever during Childhood and Adolescence |
| طبع بواسطة: | Guest user |
| التاريخ: | الأربعاء، 6 مايو 2026، 3:45 AM |
الوصف
"last update: 16 Feb 2025" Download Guideline
جدول المحتويات
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Glossary
- - Introduction
- - Methods
- - Evidence to recommendations: Considerations
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline.
- - Updating of the guideline
- - References
- - Annexes
- Executive Summary
➡️Introduction
Familial Mediterranean fever (FMF) is a monogenic inherited autoinflammatory disorder characterized by self-limited recurrent attacks of fever, serositis, arthritis and erythema. Several factors associated with emotional and physical stress are proposed to trigger FMF attacks.
The febrile attacks are accompanied by a strong acute phase response, and the most severe complication is the development of renal amyloidosis. FMF occurs most commonly among people from the Mediterranean basin (non-Ashkenazi Jews, Arabs, Armenians, Greeks, and Turks) and in other countries, but the epidemiological information remains quite scarce. The emergence, in most patients, occurs before the age of 30 (60% before 10 years and 90% before20 years of age).
FMF diagnosis is mainly clinical, and the genetic testing is indicated to support it . Laboratory tests are not specific, with high ESR, C-reactive protein and serum amyloid A (SAA) protein in the acute phase of this disease, but often, high levels are found even between attacks. SAA levels may be particularly useful in monitoring the effectiveness of treatment.
Typical attacks are defined as recurrent (≥3 of the same type), febrile (rectal temperature of 38 °C or higher), and short (lasting between 12 hours and 3 days). Patients remain asymptomatic between attacks.
FMF is associated with mutations in the Mediterranean fever (MEFV) gene encoding the protein pyrin. MEFV gene is located in the chromosome 16 p13.3 and was first identified in 1997. The gene mutations E148Q, V726A, M680I, M694V and M694I were reported to be the most frequent mutations among Egyptian FMF children.
Colchicine is the mainstay for treatment of FMF. There are two main goals of colchicine therapy in FMF. First, to prevent the clinical FMF attacks and the second is to stop the ongoing subclinical inflammation, hence prevention of the progression to amyloidosis. Initiation of colchicine therapy is recommended as soon as the clinical diagnosis has been made.
➡️Scope
This guideline focuses on diagnosis and management of FMF. In children below 18 year of age to help early and appropriate diagnosis and safe and efficient management by the physicians.
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG recommended using the following source original clinical practice guidelines (CPGs):
1-EULAR recommendations for the management of familial Mediterranean fever 2016
2- Guidelines for the management and treatment of periodic fever syndromes familial Mediterranean fever 2016 (Brazilian)
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. Diagnosis of Familial Mediterranean Fever(FMF)
The guideline covers (Age group) Children and Adolescents (Less than 18 years old)
This guideline emphasis on the clinical criteria for diagnosis
B. Management of Familial Mediterranean Fever
This section includes recommendations and good practice statements on Therapeutic intervention and guarding against complications and drug toxicity.
We can summarize the guidelines’ recommendations for Familial Mediterranean Fever (FMF) in the following:
FMF Diagnosis
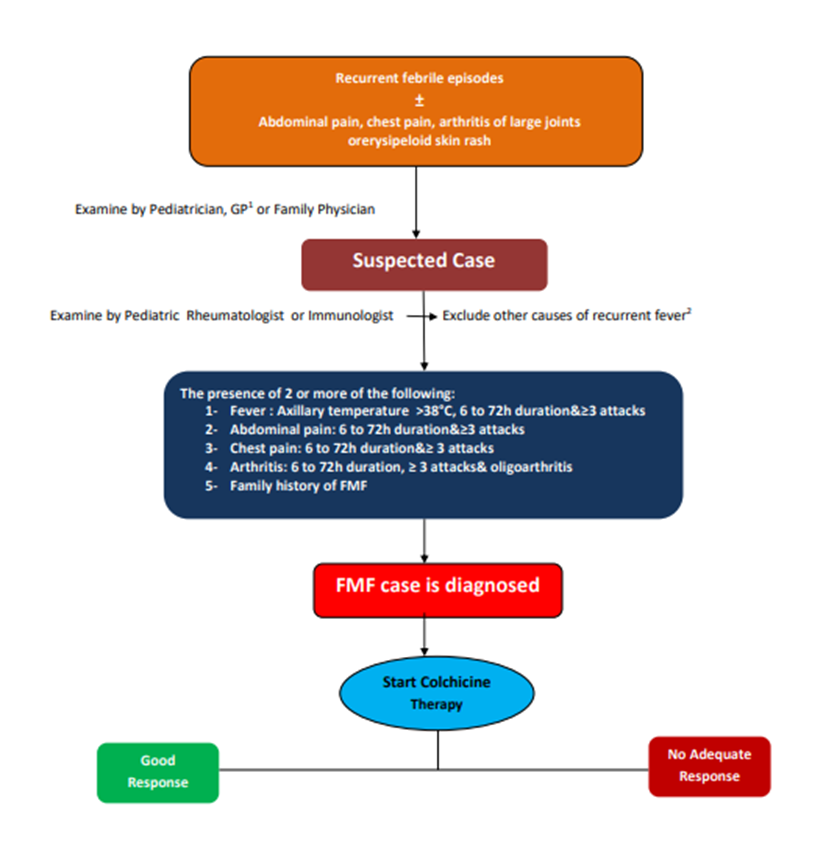
1- FMF should be suspected when there are recurrent febrile episodes associated with abdominal and/or chest pain caused by serositis (peritonitis, pericarditis or pleurisy) and arthritis/synovitis of large joints, accompanied by erysipeloid erythema. QOE Very Low, Strength Conditional
2- The presence of at least 2 of the following 5 criteria after exclusion of other causes can diagnose FMF with high sensitivity: QOE Very Low, Strength GPS
- Fever axillary temperature of >38ᵒC, 6–72 h of duration, ≥3 attacks
- Abdominal pain 6–72 h of duration ≥3 attacks
- Chest pain 6–72 h duration≥ 3 attacks
- Arthritis 6–72 h duration ≥3 attacks, oligoarthritis
- Family history of FMF*(26)
3- Genetic testing can support the clinical diagnosis but cannot exclude it. QOE Very Low, Strength GPs
4- Laboratory tests are not specific, demonstrating high serum levels of inflammatory proteins in the acute phase of the disease, but also often showing high levels even between attacks. SAA serum levels may be especially useful in monitoring the effectiveness of treatment. QOE Low, Strength Conditional
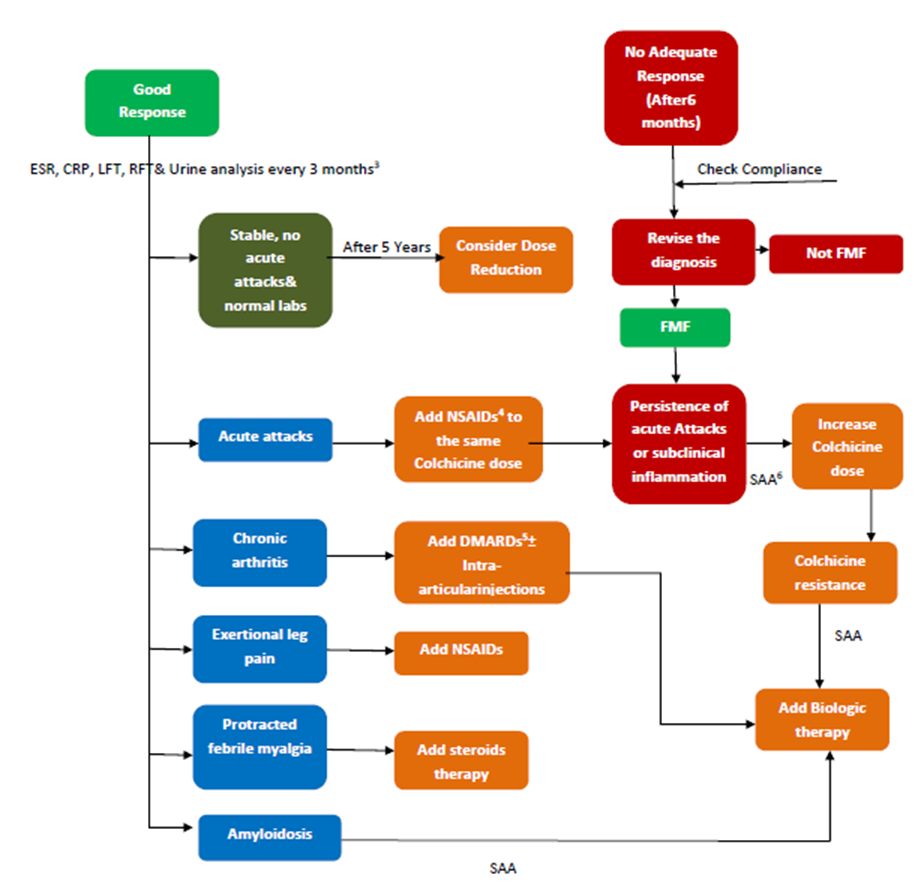
➡️FMF Management
1- Ideally, FMF should be diagnosed and initially treated by a physician with experience in FMF. QOE Very Low, Strength Conditional
2- The ultimate goal of treatment in FMF is to obtain complete control of unprovoked attacks and minimize subclinical inflammation in between attacks. QOE Low, Strength Conditional
3- Treatment with colchicine should be started as soon as a clinical diagnosis is made. QOE High, Strength : Strong.
4- Genetic testing is ideally requested and interpreted by immunology/rheumatology specialist. QOE Very Low, Strength GPS
5- Asymptomatic individuals with homozygous pathogenic mutations, particularly M694V, should be evaluated and followed up by an expert for possible intervention.QQOE Very Low, Strength : GPS
6- Dosing can be in single or in divided doses, depending on tolerance and compliance.QOE Very Low, Strength Conditional
7- The persistence of attacks or subclinical inflammation represents an indication to increase colchicine dose. QOE Low, Strength Conditional
8- FMF treatment needs to be intensified in AA amyloidosis using the maximal tolerated dose of colchicine and supplemented with biologics as required. QOE Low, Strength Conditional.QOE Intermediate, Strength Conditional.
9- Colchicine toxicity is a serious complication that should be given adequate consideration and be prevented. QOE Low, Strength Conditional.
10- Liver enzymes should be monitored regularly in patients with FMF treated with colchicine; if liver enzymes are elevated greater than twofold the upper limit of normal, colchicine shouldbe reduced, and the cause further investigated. QOE Low, Strength Conditional.
11- In patients with decreased renal function, the risk of colchicine toxicity is very high and therefore evidence of toxicity should routinely be sought, and the colchicine dose reduced accordingly. QOE Low, Strength Conditional.
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/- Recommendations
Diagnosis of FMF
|
Health Question |
Recommendation & its Grade |
Level of Evidence |
Source Guidelines |
|
1-When to suspect FMF? |
Recurrent febrile episodes associated with abdominal and/or chest pain caused by serositis (peritonitis, pericarditis or pleurisy) and arthritis/synovitis of large joints, accompanied by erysipeloid erythema. (D) |
D |
Brazilian(25)
|
|
2- How to diagnose FMF? |
The presence of at least 2 of the following 5 criteria after exclusion of other causes can diagnose FMF with high sensitivity: Fever axillary temperature of >38ᵒC, 6–72 h of duration, ≥3 attacks Abdominal pain 6–72 h of duration ≥3 attacks Chest pain 6–72 h duration≥ 3 attacks Arthritis 6–72 h duration ≥3 attacks, oligoarthritis Family history of FMF*(26) |
GPP |
|
|
3- What is the role of genetic study in FMF diagnosis? |
Genetic testing can support the clinical diagnosis but cannot exclude it**(27) |
GPP |
|
|
4- What is the essential laboratory work up for FMF? |
Laboratory tests are not specific, demonstrating high serum levels of inflammatory proteins in the acute phase of the disease, but also often showing high levels even between attacks. SAA serum levels may be especially useful in monitoring the effectiveness of treatment. (C) |
C |
Brazilian |
FMF: Familial mediterranean fever, GPP: good practice point, SAA: Serum amyloid A.
*: Yalçinkaya F, Ozen S, Ozçakar ZB, Aktay N, Çakar N et al. A new set of criteria for the diagnosis of familial Mediterranean fever in childhood. Rheumatology (Oxford) 2009; 48:395–8.
**Giancane G, Haar NMT, Wulffraat N, Vastert SJ, Barron K, Hentgen V, et al. Evidence-based recommendations for genetic diagnosis of familial Mediterranean fever. Ann Rheum Dis. 2015;74(4):635–41.
(25), (26) and (27) are reference numbers
Recommendations For Treatment of FMF
|
Health Question |
Recommendation |
Source (Guidelines) |
Page |
Level of Evidence |
GR |
|
1-Who should start treatment? |
Ideally, FMF should be diagnosed and initially treated by a physician with experience in FMF. |
EULAR(28) |
645 |
5 |
D |
|
2-What are the ultimate goals of treatment of FMF? |
The ultimate goal of treatment in FMF is to obtain complete control of unprovoked attacks and minimize subclinical inflammation in between attacks. |
EULAR |
645 |
4 |
C |
|
3-When to start treatment with colchicine? |
Treatment with colchicine should be started as soon as a clinical diagnosis is made. |
EULAR |
646 |
1b |
A |
|
4-Should asymptomatic Individuals with MEFV gene pathogenic mutations start treatment? |
Genetic testing is ideally requested and interpreted by immunology/rheumatology specialist.** (27)
Asymptomatic individuals with homozygous pathogenic mutations, particularly M694V, should be evaluated and followed up by an expert for possible intervention.**(27) |
|
|
|
GPP
GPP |
|
5-How is colchicine given? |
Dosing can be in single or in divided doses, depending on tolerance and compliance. |
EULAR |
646 |
5 |
D |
|
6-What are the indications for increasing the dose of colchicine? |
The persistence of attacks or subclinical inflammation represents an indication to increase colchicine dose.
|
EULAR |
646 |
3 |
C |
|
7-What is the treatment of amyloidosis? |
FMF treatment needs to be intensified in AA amyloidosis using the maximal tolerated dose of colchicine and supplemented with biologics as required. |
EULAR |
646 |
2b |
C |
|
8-How to monitor toxicity of Colchicine?
|
Colchicine toxicity is a serious complication that should be given adequate consideration and be prevented.
Liver enzymes should be monitored regularly in patients with FMF treated with colchicine; if liver enzymes are elevated greater than twofold the upper limit of normal, colchicine shouldbe reduced, and the cause further investigated.
In patients with decreased renal function, the risk of colchicine toxicity is very high and therefore evidence of toxicity should routinely be sought, and the colchicine dose reduced accordingly. |
EULAR
EULAR
EULAR |
647
647
647 |
4
5
4 |
C
D
C |
|
10-How to treat patients with chronic arthritis? |
Chronic arthritis in a patient with FMF might need additional medications, such as DMARDs, intra-articular steroid injections or biologics. |
EULAR |
645 |
2b |
C |
|
11-How to treat febrile myalgia |
In protracted febrile myalgia, glucocorticoids lead to the resolution of symptoms; NSAID and IL-1-blockade might also be a treatment option. NSAIDs are suggested for the treatment of exertional leg pain. |
EULAR |
645&648 |
2b |
C |
|
12-Should colchicine be stopped during pregnancy or lactation? |
Colchicine should not be discontinued during conception, pregnancy or lactation; current evidence does not justify amniocentesis. |
EULAR |
648 |
3 |
C |
|
13-Should colchicine be stopped before conception in men? |
In general, men do not need to stop colchicine prior to conception; in the rare case of azoospermia or oligospermia proven to be related to colchicine, temporary dose reduction or discontinuation may be needed. |
EULAR |
648 |
3 |
C |
|
14-When to add biologic treatment?
|
Compliant patients not responding to the maximum tolerated dose of colchicine can be considered non-respondent or resistant; alternative biological treatments are indicated in these patients. |
EULAR |
646-647 |
2b |
B |
|
15-For how long should treatment be continued? |
If a patient is stable with no attacks for more than 5 years and no elevated APR, dose reduction could be considered after expert consultation and with continued monitoring. |
EULAR |
648-649 |
5 |
D |
APR: Acute Phase Reactants
DMARDs, disease modifying antirheumatic drugs
EULAR: European League Against Rheumatism
GR: Grade of Recommendations
NSAID, Non-Steroidal Anti-InflammatoryDrugs
SAA: Serum Amyloid A
GPP: Good Practice Point
** Giancane G, Haar NMT, Wulffraat N, Vastert SJ, Barron K, Hentgen V, et al. Evidence-based recommendations for genetic diagnosis of familial Mediterranean fever. Ann Rheum Dis. 2015;74(4):635–41.
(27) and (28) are reference numbers
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
||||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
||||
|
Dr Ashraf Ahmed Galal |
Professor of Pediatrics, Children’s Hospital. Faculty of Medicine, Alexandria University. |
||||
|
Dr Dalia H. El- Ghoneimy |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Faculty of Medicine, Ain Shams University. |
||||
|
Dr Maher Ahmed Abdel Hafez |
Professor of Pediatrics. Head of Pediatric Nephrology and Rheumatology unit. Faculty of Medicine, Tanta University |
||||
|
Dr Mohamed Almalky |
Professor of Pediatrics, Faculty of Medicine, Zagazig University |
||||
|
Dr Sanaa A Mahmoud
|
Clinical Professor of Pediatrics, Allergy and Immunology, Pikeville Osteopathic School of Medicine, KY. Adjunt Faculty, Lincoln Debusk University, Harrogate, TN. Professor of Pediatrics (in sabbatical) Faculty of Medicine, Ain Shams University. |
||||
|
Dr Sheren Esam Maher |
Professor of Pediatrics, Pediatric Rheumatology, Children's Hospital, Faculty of Medicine, Minia University. |
||||
|
Dr Shereen Medhat Reda |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Faculty of Medicine, Ain Shams University. |
||||
|
Dr Zeinab Awad El- Sayed |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology Unit, Children's Hospital, Faculty of Medicine, Ain Shams University. |
||||
|
Dr Ghada El Deriny |
Associate Professor of Pediatrics, Children's Hospital. Faculty of Medicine, Alexandria University. |
||||
|
Dr Rasha El-Owaidy
|
Associate Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Faculty of Medicine, Ain Shams University. |
||||
|
Dr Yomna Mohamed Farag |
Associate Professor of Pediatrics, Pediatric Rheumatology Unit, Specialized Children’s Hospital, Faculty of Medicine, Cairo University. |
||||
|
Dr Ali Sobh |
Lecturer of Pediatrics, Mansoura University Children’s Hospital, Faculty of Medicine, Mansoura University. |
||||
|
Dr Ghada Abdel Haleem Shousha |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital. Faculty of Medicine, Ain Shams University. |
||||
|
Dr Naglaa Samy Mohamed Osman |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology Unit, Children's Hospital, Faculty of Medicine, Assiut University. |
||||
|
Dr Nesrine Radwan
|
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children's Hospital. Faculty of Medicine, Ain Shams University. |
||||
|
Dr Walaa Shoman |
Lecturer of Pediatrics, Children's Hospital. Faculty of Medicine, Alexandria University. |
||||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Dr Tarek Omar |
Prof. of Pediatrics, Alexandria University |
Overseeing the adolopment process of the guidelines. |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto,São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
External ReviewersGroup (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Samia Salah |
Prof. of Pediatrics and Pediatric Rheumatology, Cairo University |
||||
|
Prof Elham Hossny |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Ain Shams University. |
||||
|
Prof Hala Lotfy |
Prof. of Pediatrics and Pediatric Rheumatology, Cairo University |
||||
|
International Peer Reviewers |
|||||
|
1. Prof. Seza Ozen
|
Department of Pediatric Rheumatology, Hacettepe University, Ankara, Turkey |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
▪️ The GDG acknowledge EPG for its help in completing this project.
▪️ We acknowledge The EULAR and Brasilian FMF guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️ This work is not related to any pharmaceutical or industrial company. The members of the GDG and their institutes and universities volunteered their participation and contributions.
- Abbreviations
|
CPGs |
Clinical Practice Guidelines |
|
Adolopment |
Adoption-Adaptation-Development |
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
|
DMARDs |
Disease-Modifying Anti-Rheumatic Drugs |
|
EBCPG |
Evidence Based Clinical Practice Guidelines |
|
EPG |
Egyptian Pediatric Clinical Practice Guidelines Committee |
|
EULAR |
European League Against Rheumatism |
|
FMF |
Familial Mediterranean Fever |
|
FMFGAG |
Familial Mediterranean Fever Guidelines Adaptation Group |
|
GP |
General Practitioner |
|
GPS |
Good Practice Statement |
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
|
IL-1 |
Interleukin-1 |
|
JIA |
Juvenile Idiopathic Arthritis |
|
MEFV |
Mediterranean Fever Gene |
|
PFAPA |
(Periodic Fever, Aphthous Stomatitis, Pharyngitis, Adenitis) |
|
SAA |
Serum Amyloid A Protein |
|
TNF |
Tumor Necrosis Factor |
- Glossary
Amyloidosis:
the inflammatory episodes in persons with FMF lead to the excess production of amyloid A protein in the acute phase and reactant serum amyloid A with subsequent deposition in the kidneys. However, only patients with specific MEFV haplotypes develop amyloidosis.
- Introduction
Familial Mediterranean fever (FMF) is a monogenic inherited autoinflammatory disorder characterized by self-limited recurrent attacks of fever, serositis, arthritis and erythema. Several factors associated with emotional and physical stress are proposed to trigger FMF attacks (1,2).
The febrile attacks are accompanied by a strong acute phase response, and the most severe complication is the development of renal amyloidosis. FMF occurs most commonly among people from the Mediterranean basin (non-Ashkenazi Jews, Arabs, Armenians, Greeks, and Turks) and in other countries, but the epidemiological information remains quite scarce. The emergence, in most patients, occurs before the age of 30 (60% before 10 years and 90% before20 years of age) (3,4)
FMF diagnosis is mainly clinical, and the genetic testing is indicated to support it (5). Laboratory tests are not specific, with high ESR, C-reactive protein and serum amyloid A (SAA) protein in the acute phase of this disease, but often, high levels are found even between attacks. SAA levels may be particularly useful in monitoring the effectiveness of treatment. (6)
Typical attacks are defined as recurrent (≥3 of the same type), febrile (rectal temperature of 38 °C or higher), and short (lasting between 12 hours and 3 days). Patients remain asymptomatic between attacks. Incomplete attacks are defined as painful and recurrent attacks that differ from typical attacks in one or two features, as follows (5):
1. The temperature is normal or lower than 38 °C.
2. The attacks are longer or shorter than specified (but not shorter than 6 hr or longer than a week).
3. No signs of peritonitis are recorded during the abdominal attacks.
4. The abdominal attacks are localized.
5. The arthritis involves joints other than hip, knee and ankle.
- 9 -Genetic diagnosis:
FMF is associated with mutations in the Mediterranean fever (MEFV) gene encoding the protein pyrin. MEFV gene is located in the chromosome 16 p13.3 and was first identified in 1997. Approximately one-third of the patients have either a single or no gene mutation (7).
Previous studies on FMF patients and animal models suggest that MEFV mutations lead to dysregulation of the inflammasome, a complex intracellular multiprotein structure, ending in gain of pyrin function with increased IL-1b secretion by monocytes and a prolonged inflammatory response (8).
The gene mutations E148Q, V726A, M680I, M694V and M694I were reported to be the most frequent mutations among Egyptian FMF children(9).Early onset and severe phenotypes were commonly associated with M694V (10).
The inheritance of FMF, unlike other monogenic SAIDs, is not an ordinary autosomal recessive disorder. Presence of clinical phenotype among heterozygous patients of FMF was reported in about one quarter of clinically diagnosed patients. Thus, FMF was suggested to be a dominant condition with low penetrance (11).
Comorbidities and complications
Many inflammatory and autoimmune diseases have been seen in association with FMF whether related to the activated innate immune system and high pro-inflammatory state or incidentally discovered. Yildiz et al. studied a large cohort of pediatric FMF patients and observed that nearly a fifth of them had comorbid diseases and needed additional medications. The most common was juvenile idiopathic arthritis, henoch-schonlein purpura, uveitis, inflammatory bowel diseases, polyarteritis nodosa, and PFAPA (12).
Arthritis is one of the common clinical features of FMF, and concomitant presence of FMF and juvenile idiopathic arthritis or ankylosing spondylitis has been described. Mutations of MEFV gene might be one of the genetic determinants of JIA especially systemic onset type with elevated IL-1 cytokines (13).
- 10 -Amyloidosis, the most serious complication of FMF, is the deposition of an insoluble serum protein called serum amyloid A, which is produced by the liver, and is considered one of acute phase reactants. The persistence of subclinical inflammation and delay of diagnosis are important risk factors for the development of amyloidosis in FMF (14).It usually involves the kidneys with early proteinuria and later development of renal impairment. It was observed in up to 10.5% of FMF patients. Amyloidosis could be also seen involving liver, intestine, or heart (15).
Increased awareness of the disease with good control of the inflammation, strict follow up, and judicious use of colchicine and biologics could greatly prevent the development of amyloidosis. (13).
➡️Treatment
Colchicine is the mainstay for treatment of FMF. The exact mechanism of action by which colchicine prevents the attacks of FMF and suppresses the inflammation is not well understood. Colchicine prevents activation of neutrophils, it binds to β-tubulin making β- tubulin-colchicine complexes; this way inhibits assembly of microtubules and mitotic spindle formation; moreover, its mode of action includes modulation of chemokines, prostanoids production, and inhibition of neutrophil and endothelial cell adhesion molecules(16). It is thought that colchicine has an effect on the transcription and the expression of the genes involved in neutrophils migration and activation. This latter effect is delayed and doesn’t happen immediately after administration of colchicine, a matter that may explain why colchicine doesn’t have an immediate effect in the acute attacks of FMF (17).
There are two main goals of colchicine therapy in FMF. First, to prevent the clinical FMF attacks and the second is to stop the ongoing subclinical inflammation, hence prevention of the progression to amyloidosis. Initiation of colchicine therapy is recommended as soon as the clinical diagnosis has been made. Individuals who have positive genotype (one or more MEFV mutation) however do not express clinical disease and do not have elevated acute phase reactants, are not recommended to start treatment.
- 11 -Special attention and close follow up should be given to individuals with homozygous
M694V/M694V as they have a higher risk to develop amyloidosis, so they should start treatment as soon as they express the clinical manifestations associated with elevation of the acute phase reactants (18).
The usual initial dose is ≤0.5 mg/day (≤0.6 mg/day if tablets contain 0.6 mg) for children <5 years of age, 0.5–1.0 mg/day (1.2 mg/day if tablets contain 0.6 mg) for children 5–10 years of age, 1.0–1.5 mg/day (1.8 mg/day in case tablets contain 0.6 mg) in children >10 years of age. The maximum dose in children is 2mg/day. All patients should be monitored for disease control as evidenced by minimizing the number of clinical attacks and normalization of the acute phase reactants and SAA in between the attacks. The dose of colchicine should be adjusted according to the degree of disease control. Colchicine is a lifelong therapy especially in poorly controlled cases and in cases with high risk for amyloidosis. Some experts currently suggest that if the patient remained attack free with normal acute phase reactants for more than five years, dose reduction can be considered with close monitoring and after expert consultation (18).
In general, colchicine has a good safety profile and is well tolerated with minimal side effects. The main side effects include GIT disturbances, lactose intolerance, and elevation of the liver enzymes, alopecia, neutropenia and peripheral neuropathy. Dividing the dose of colchicine and dietary modifications markedly decrease the side effects. In case of elevated liver enzymes, transient reduction or stoppage of the colchicine will eventually be helpful then the usual dose can be resumed. Colchicine has been theoretically incriminated to affect the spermatogenesis as it acts by suppressing the microtubules and arrest of mitosis, however, recent and old studies showed no effect of colchicine on spermatogenesis nor any teratogenic effect and if there is any fertility problem in patients with FMF it is better to be attributed to the disease itself and the amyloidosis of testis or ovaries. For these reasons, it is now recommended not to stop colchicine neither before nor during pregnancy (18).
There is no standard definition for refractory or resistant FMF however, the recent guidelines stated that FMF can be considered resistant to treatment if:(18)
- 12 -1- The patient continued to have ≥ one attack per month despite the maximally tolerated continuous colchicine dose for ≥ 6 months.
2- Persistent subclinical inflammation that is a risk factor for amyloidosis.
3- If the patient developed renal amyloidosis.
In case of uncontrolled disease, the second line drugs, the biological agents, should be added. Anti-interleukin I (anti-IL-1) biological drugs are recommended in the patients with FMF. Several types of anti-IL-1 are available namely; recombinant homologue of IL-1 receptors (anakinra)(20),fully human immunoglobulin G1 monoclonal antibody against IL- 1 (canakinumab)(21), and the third one is Rilonacept, dimeric FC fusion protein capturing IL-1.
Despite the marked efficacy of these biologic drugs in the treatment of FMF and their ability to reverse proteinuria in cases with renal amyloidosis; their efficacy in prevention of amyloidosis is not yet proven and colchicine should continue during biological drug treatment to prevent amyloidosis. Of note, the three anti-IL-1 agents are not interchangeable, and the patient may respond to one drug and not to the other one. This role applies to FMF and other diseases treated by biologics (18).
Treatment of amyloidosis includes measures to support failing organ function, including blood pressure control and dialysis for patients with renal disease (18). The majority of patients with FMF and amyloidosis will eventually require renal replacement therapy (22). Recent experience of renal transplantation in selected patients has been encouraging with long-term graft and patient survival matching that of the age-matched general transplant population (20).
Management of the acute attacks include continuation of the colchicine therapy on the same dose and adding nonsteroidal anti-inflammatory drugs to alleviate the pain. There is no evidence to support increasing the dose of colchicine during the acute attack. Other drugs may include glucocorticoids in severe attacks and in the syndrome of protracted febrile myalgia, disease modifying anti-rheumatic drugs (DMARDs) in chronic arthritis accompanying the FMF. Tumor necrosis factor receptor antagonists (anti-TNF) may also be used in chronic arthritis (23,24).
➡️Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of Familial Mediterranean Fever (FMF).
It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to
1) Provide accurate diagnosis of FMF to avoid over and under diagnosis.
2) Define the role of investigations in diagnosis and optimizing management and prevention of complications
3) Identify the best practice management of FMF patients.
This version of the guideline includes recommendations and good practice statements for diagnosis and treatment of Familial Mediterranean Fever.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: FMF, Familial Mediterranean, Guidelines, Pediatric, and Management.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria the GDG/ GAG recommended using 2 guidelines:
1- EULAR recommendations for the management of familial Mediterranean fever 2016
2- Guidelines for the management and treatment of periodic fever syndromes familial Mediterranean fever 2016 (Brazilian)
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of pediatric immunology and rheumatology.
The main functions of the clinical panel were adolopment of FMF Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulatingevidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 2 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE IIassessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as an eminent international reviewer.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of
the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetingswere also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of eachmeeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendationformulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interestand confidentiality undertaking forms. All guideline members and methodologists were also asked to fillin and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s)was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations (we did not need to do this).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
Developing good practice statements:
The GDG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to helpwith their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability,feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing FMF diagnosis and treatment, strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, andall Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️Gantt chart has been designedto manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
Evidence to Decision Tables: (if any)
Guideline Implementation Tools
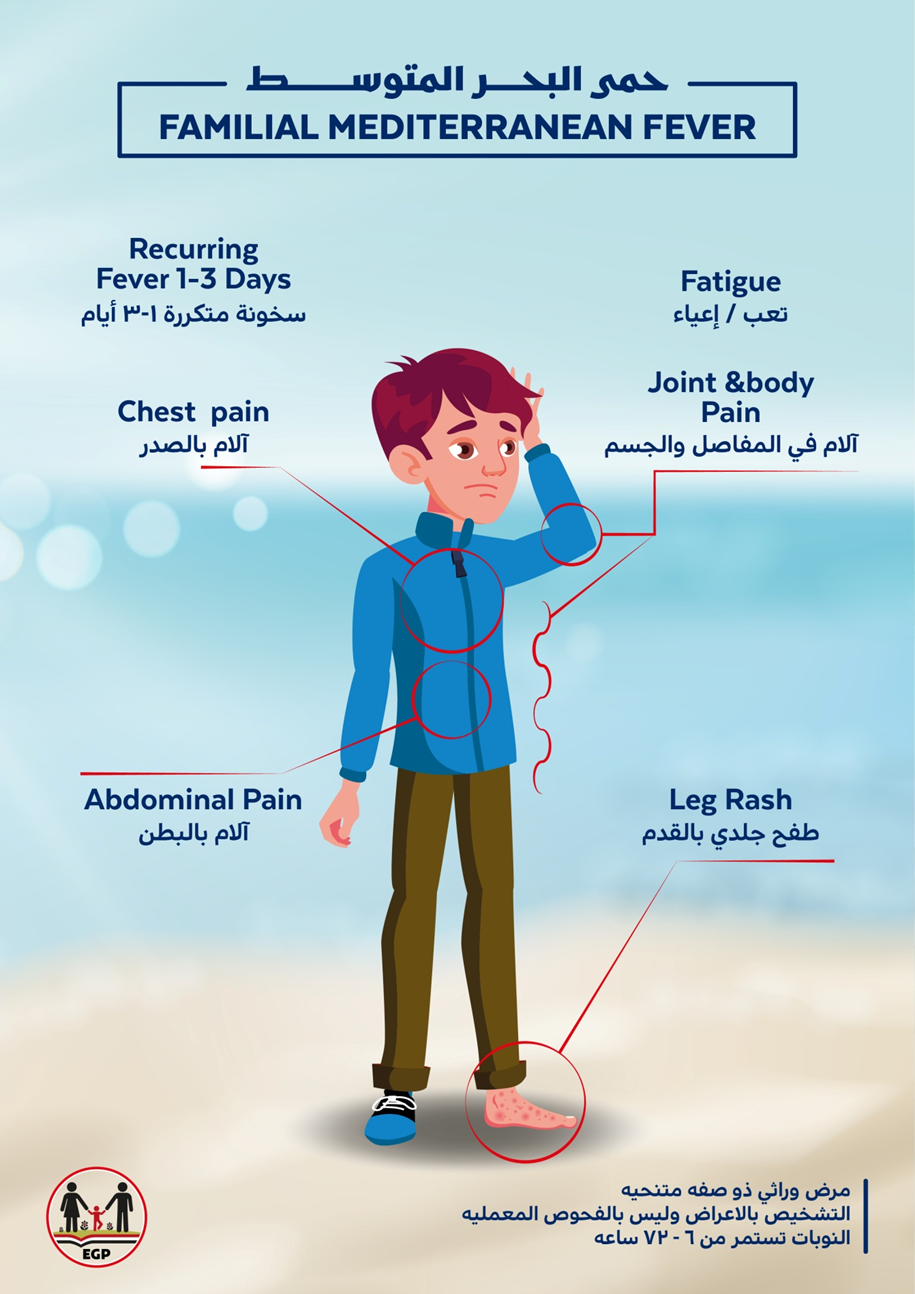
Educational materials; based on this Adapted CPG for diagnosis and treatment of FMF in children; have been made available in several forms including:
1. Manual for physician for diagnosis and treatment algorithms for management of FMF
3. Arabic Educational materials for nurses and mothers
Algorithm of
Diagnosis and Management of Familial Mediterranean Fever in Children

- Limitations and suggestions for further research needs
Future research recommendations for the diagnosis and management of FMF in children in the Egyptian context could include:
▪️ Establishing a registry for cases of FMF
▪️ Assesment of the need for FMF biologic therapy in Egyptian children
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for FMF in children.
➡️Challenges
▪️ Increase awareness among pediatrician about FMF diagnosis
▪️ The expenses of biologic therapy in colchicines resistant cases
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for FMF in children:
1. Adherence to FMF Guidelines
▪️ Numerator: Number of children with FMF who received treatment as per guideline recommendations.
▪️ Denominator: Total number of children diagnosed with FMF
▪️ Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
▪️ Numerator: Total number of hospital stay days for children with FMF acute attacks.
▪️ Denominator: Total number of children admitted with acute attacks of FMF.
▪️ Data Source: Hospital admission and discharge records.
3. Rate of Readmission
▪️ Numerator: Number of children readmitted with symptoms of FMF attacks within a certain period (e.g., 30 days) after discharge.
▪️ Denominator: Total number of children initially admitted with acute attack of FMF
▪️ Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG …GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Ozen S, Bilginer Y. A clinical guide to autoinflammatory diseases: familial Mediterranean fever and next-of-kin. Nat Rev Rheumatol2014;10:135-47.
2. Yenokyan G, Armenian HK. Triggers for attacks in familial Mediterranean fever: application of the case-crossover design. Am J Epidemiol 2012;175:1054-61.
3. Fujikura K.Global epidemiology of Familial Mediterranean fever mutations using population exome sequences. Molecular Genetics & Genomic Medicine. 2015; 3(4): 272–282.
4. Berkun Y, Eisenstein E, Ben-Chetrit E. FMF – clinical features, new treatments and the role of genetic modifiers: a critical digest of the 2010–2012 literature. Clin Exp Rheumatol. 2012;30 Suppl.72:S90–5.
5. Alghamdi M. Familial Mediterranean fever, review of the literature. Clin Rheumatol. 2017; 36: 1707–13.
6. Çakan M, Karadağ SG, Tanatar A, Sönmez HE, and Ayaz NA. The Value of Serum Amyloid A Levels in Familial Mediterranean Fever to Identify Occult Inflammation During Asymptomatic Periods. J Clin Rheumatol. 2021 Jan 1;27(1):1-4. doi: 10.1097/RHU.0000000000001134.
7. Ozdogan H, Ugurlu S. Familial Mediterranean Fever.Presse Med. 2019 Feb;48(1 Pt 2):e61-e76. doi: 10.1016/j.lpm.2018.08.014.
8. Chae, J. J., Y. H. Cho, G. S. Lee, J. Cheng, P. P. Liu, L. Feigenbaum, et al. 2011. Gain-of-functionpyrinmutationsinduce NLRP3 protein-independent interleukin-1β activation and severe autoinflammation in mice. Immunity 34: 755– 768.
9. Hala S. Talaat, Maha F. Sheba, Rehab H. Mohammed, Mohamed Ali Gomaa, Nihal El Rifaei. Genotype Mutations in EgyptianChildrenwithFamilialMediterranean Fever: ClinicalProfile, and Response to Colchicine. Mediterr J Rheumatol. 2020 Jun; 31(2): 206–213.
10. Ozturk C, Halicioglu O, Coker I, Gulez N, Sutçuoglu S, Karaca N, et al. Association of clinical and genetical features in FMF with focus on MEFV strip assay sensitivity in 452 children from western Anatolia, Turkey. Clin Rheumatol. 2012; 31:493–501.
11. Marek-Yagel D, Berkun Y, Padeh S, Abu A, Reznik-Wolf H, Livneh A. Clinical disease among patients heterozygous for familial Mediterranean fever. Arthritis Rheum. 2009; 60(6):1862–6.
12. Yildiz M, Adrovic A, Tasdemir E, Baba-Zada K, Aydin M et al. Evaluation of co-existing diseases in children with familial Mediterranean fever. Rheumatol Int. 2020; 40(1): 57-64.
13. Balcı-Peynircioğlu B, Kaya-Akça Ü, Arıcı Z S, Avcı E, Akkaya-Ulum Z Y et al. Comorbidities in familial Mediterranean fever: analysis of 2000 genetically confirmed patients. Rheumatology, 2020; 59(6):1372-80.
14. Hashkes P J. 50 Years Ago in The Journal of Pediatrics: Amyloidosis in Childhood. J Pediatr, 2019; 205: 54
15. Touitou I, Sarkisian T, Medlej-Hashim M, Tunca M, Livneh A et al. International Study Group for PhenotypeGenotype Correlation in Familial Mediterranean Fever. Country as the primary risk factor for renal amyloidosis in familial Mediterranean fever. Arthritis Rheum, 2007; 56(5): 1706-12.)
16. Cerquaglia C, Diaco M, Nucera G, La Regina M, Montalto M, Manna R. Pharmacological and clinical basis of treatment of familial Mediterranean fever (FMF) with colchicine or analogues: an update. Curr Drug Targets Inflamm Allergy 2005;4(1):117-24.
17. Ben-Chetrit E, Bergmann S, Sood R. Mechanism of the anti-inflammatory effect of colchicine in rheumatic diseases: a possible new outlook through microarray analysis. Rheumatology (Oxford) 2006;45(3):274-82.
18. Ozen S, Demirkaya E, Erer B, Livneh A, Ben-Chetrit E, Giancane G, et al. EULAR recommendations for the management of familial Mediterranean fever. Ann Rheum Dis 2016;75(4):644-51.
19. Haimov-Kochman R, Ben-Chetrit E. The effect of colchicine treatment on sperm production and function: a review. Hum Reprod 1998;13(2):360-2.
20. Erdem E, Karatas A, Kaya C, et al Renal transplantation in patients with familial Mediterranean fever. Clin Rheumatol 2012;31:1183–6.
21. Dinarello CA, Simon A, Van derMeer JW. Treating inflammation by blocking interleukin-1 in a broad spectrum of diseases. Nat Rev Drug Discov2012;11(8):633-52.
22. Keven K, Sengul S, Kutlay S, Ekmekci Y, AnadoletEal Long-term outcome of renal transplantation in patients with familial Mediterranean fever amyloidosis: a single-center experience. Transplant Proc 2004; 36:2632–4.
23. Amital H, Ben-Chetrit E. Therapeutic approaches to familial Mediterranean fever. What do we know and where are we going to? Clin Exp Rheumatol 2004;22(Suppl 34):S4–7.
24. Meinzer U, Quartier P, Alexandra JF, Hentgen V, Retornaet F al. Interleukin-1 targeting drugs in familial Mediterranean fever: a case series and a review of the literature. Semin Arthritis Rheum 2011;41:265–71.
25. TerreriM, Bernardo W, Len C, Artur C, Ribeiro C et al. Guidelines for the management and treatment of periodic fever syndromes familial Mediterranean fever. rev bras reumatol. 2016;5 6(1):37–43
26. Yalçinkaya F, Ozen S, Ozçakar ZB, Aktay N, Çakar N et al. A new set of criteria for the diagnosis of familial Mediterranean fever in childhood. Rheumatology (Oxford) 2009; 48:395–8.
27. Giancane, G., N. M. Ter Haar, N. Wulffraat, S. J. Vastert, K. Barron, V. Hentgen, et al. 2015. Evidence-basedrecommendations for geneticdiagnosisoffamilialMediterraneanfever. Ann. Rheum. Dis.74:635–41.
28. Ozen S, Demirkaya E, Erer B, Livneh A, Ben-Chetrit E, Giancane G, et al. EULAR recommendations for the management of familial Mediterranean fever. Ann Rheum Dis. 2016; 75(4):644–51.
29. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. J Eval Clin Pract. 2015 Dec;21(6):1095-106
30. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
31. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/.
32. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352(Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Tarek Omar |
Professor of Pediatrics Alexandria University, Egypt |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
External Review Group |
|||
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
|
|
|
|
|
International Peer Reviewers |
|
|
|
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for "Guideline for the prevention and management of Familial Mediterranean Fever" on PubMed are:
Familial Mediterranean Fever, FMF.
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for FMF
|
Guidelines |
Domain 1 Score % |
Domain 2 |
Domain3 |
Domain4 |
Domain5 |
Domain6 |
Decision |
||||||
|
EULAR |
82% |
90% |
84% |
93% |
79% |
96% |
INCLUDED |
||||||
|
BRASILIAN |
74% |
57% |
56% |
81% |
43% |
72% |
INCLUDED |
||||||
|
FRENCH |
63% |
54% |
42% |
54% |
19% |
56% |
NOT INCLUDED |
||||||

Annex Table 3. Annex Nurses and Parents Educational Guide in Arabic
برنامج تعليمى لمرضى حمى البحر الأبيض المتوسط
المقدمة
حمى البحر الأبيض المتوسط (FMF)هو مرض وراثي يتميز بنوبات متكررة من الحرارة وألم في البطن أو الصدر والتهاب المفاصل وطفح وتقرحات في الغشاء المخاطي للفم. وعادة تحدث هجمات المرض نتيجة عدة عوامل مرتبطة بالإجهاد العاطفي والجسدي. وأما عن سبب التسمية فلأنه (FMF) أكثر شيوعا بين الناس من حوض البحر الأبيض المتوسط (مثل اليهود غير الأشكناز، الأرمن، العرب، اليونان، والأتراك) وأيضا في بلدان أخرى، ولكن المعلومات عن معدلات انتشارها لا تزال محدودة جدا. ويظهر في معظم المرضى قبل سن 30 (60٪ قبل 10 سنوات و90٪ قبل 20 سنة من العمر).
وتشخيص مرض حمى البحر الأبيض المتوسط هو أساسا تشخيص اكلينيكي ويمكن عمل الاختبار الجيني لدعم التشخيص الاكلينيكي والتحاليل المعملية ليس لها دور تشخيصي لهذا المرض ولكن قد يكون هناك ارتفاع في سرعة الترسيب، رد الفعل البروتينيCوالاميلويد بروتين (SAA)أثناء النوبات الحادة من هذا المرض، و قد تكون هناك مستويات عالية حتى بين النوبات وقد تكون مستويات SAA مفيدة في مراقبة فعالية العلاج.
وتعرف النوبات النموذجية المكتملة بأنها متكررة (≥3 من نفس النوع)، ومصحوبة بحرارة (درجة حرارة المستقيم 38 درجة مئوية أو أعلى)، وقصيرة (تتراوح بين 6 ساعات و3 أيام). ولا توجد اي أعراض بين النوبات.
وتعرف النوبات غير المكتملة بأنها نوبات مؤلمة ومتكررة تختلف عن الهجمات النموذجية في واحدة أو اثنتين، على النحو التالي:
درجة الحرارة طبيعية أو أقل من 38 درجة مئوية.1
النوبات أطول أو أقصر من المحدد (ولكن ليس أقصر من 6 ساعات أو أكثر من أسبوع).2
لا يتم تسجيل أي علامات التهاب بريتوني حاد خلال نوبات الم البطن.3
نوبات ألم البطن مركزة في أماكن محددة ولا تشمل البطن كله..4
التهاب المفاصل يؤثر على مفاصل أخرى غير الورك والركبة والكاحل.5
التشخيص الجيني
وترتبط حمى البحر الأبيض المتوسط مع طفرات في جين MEFVالذي يقع في الكروموسوم 16 p13.3 وتم التعرف عليه لأول مرة في عام 1997. وما يقرب من ثلث المرضى لديهم إما طفرة جينية واحدة أو لا توجد أي طفرات. وهذه الطفرة تؤدي إلى التهابات مستمرة وتم تحديد مئة من الطفرات ولكن الطفرات الخمسة الأساسية هي: E148Q و M680I و M694I و M694V و V726A. واشد الحالات واسرعها ظهورا مرتبطة عادة بالطفرةM694V.
وعلى عكس الأنماط الجينية أحادية المنشأ الأخرى مرض حمى البحر الأبيض المتوسط ليس اضطرابا متنحيا تلقائيا عاديا فقد تم رصد أنماط غير متجانسة جينيا (heterozygous) في حوالي ربع المرضى الذين تم تشخيصهم اكلينيكيا.
الأعراض والمضاعفات
لقد تم رصد العديد من الأمراض الالتهابية والمناعة الذاتية في مرضي حمى البحر الأبيض المتوسط. وقد تمت دراسة مجموعة كبيرة من المرضى الأطفال ولوحظ أن ما يقرب من خمسهم يعانون من أمراض إضافية وكان أكثرها شيوعا التهاب المفاصل مجهول السبب، فرفرية هينوش شونلين ، التهاب العنبية (مرض بهجت)، وأمراض الأمعاء الالتهابية، التهاب القولون الدوديلكن أخطر مضاعفات مرض حمى البحر الأبيض المتوسط هو الداء النشواني الذي هو ترسب لبروتين غير قابل للذوبان يسمى الأميلويد A وهو أحد متفاعلات الالتهاب الحادة الذي ينتجه الكبد مع أي التهاب حاد في الجسم. وتأخر التشخيص أوعدم العلاج لمرضي حمي البحر الأبيض المتوسط مع استمرار الالتهاب الإكلينيكي يؤدي الي حدوث الداء النشواني وترسب الأميلويد A في الكلى مما يؤدي الي فشل كلوي وقد يحدث الداء النشواني أيضا في الكبد أو الأمعاء أو القلب لكن زيادة الوعي بالمرض مع السيطرة الجيدة على الالتهاب، والمتابعة الدقيقة مع الاستخدام الرشيد للكولشيسين أو حتى العلاج البيولوجي يمكن أن تمنع إلى حد كبير تطور الداء النشواني.
العلاج
الكولشيسين هو العلاج الأساسي لحمي البحر الأبيض المتوسط ويعمل علي منع نوبات المرض وتقليل الالتهابات بين النوبات وبالتالي تقليل المضاعفات بالداء النشواني. ويجب بدء العلاج فور التشخيص الأكلينيكي وجرعة الكولشيسين تختلف باختلاف العمر والوزن. والعلاج يجب ان يستمر طوال العمر كما يجب متابعة تأثير العلاج عن طريق الاعراض الاكلينيكية ومدي السيطرة عليها وكذلك عن طريق مستوي بروتين الاميلويد (أ) ومستوي متفاعلات الالتهابات الحادة في الدم. كما يجب متابعة المريض خوفا من حدوث مضاعفات من العلاج نفسه فبالرغم من ان الكولشيسين عقار آمن الا ان بعض الأعراض الجانبية قد تحدث مع الأستعمال مثل اضطرابات الجهاز الهضمي وارتفاع مستوي انزيمات الكبد وتساقط الشعر وانخفاض عدد كرات الدم البيضاء وتقليل الجرعة او تقسيمها في هذه الحالات يقلل بشكل كبير من هذه الأعراض الجانبية. وفي النوبات الحادة للمرض لا يتم زيادة جرعة الكولشيسين ولكن يمكن اضافة بعض الادوية المسكنة مثل الايبوبروفين او حتي بعض الكورتيزون لتقليل الألم والألتهابات ولا يتأثر الحمل أو الأنجاب سواء في الذكور أوالأناث بعقار الكولشيسين ولذا لا يوقف قبل او اثناء الحمل او بعد الولادة واثناء الرضاعة الطبيعية.
وقد لا يستجيب بعض المرضي للعلاج بالكولشيسين وهؤلاء نطلق عليهم مرضي مقاومون للكولشيسين ولكن قبل ان نضع المريض في هذا التصنيف يجب التاكد اولا ان الجرعات وصلت الي حدها الأقصي للعمر وان المريض منتظم علي العلاج وانه :
1- تحدث الهجمات اكثر من مره في الشهر الواحد بعد 6 اشهر من العلاج
2- هناك ارتفاع في مستويات متفاعلات الالتهابات الحادة في الدم بين النوبات الحادة.
3- هناك حدوث للداء النشواني في الكلي
وفي حالة حدوث ذلك يعتبر المريض مقاوما او غير مستجيب للعلاج بالكولشيسين وفي هذه الحالة يجب استشارة الطبيب المعالج فورا.
أسئلة شائعة :
1- كيف يتم تشخيص الحالة ؟
بعد تكرار حدوث نوبات من الحرارة وألم البطن التى قد يصاحبها أيضا ألم بالصدر و المفاصل وطفح جلدى أكثر من 3 مرات وكل مرة يستمر من 6-72 ساعة عندها يجب استشارة طبيب متخصص للتأكد من صحة التشخيص.
2- ما دور التحليل الجينى في التشخيص ؟
التشخيص يعتمد على تقييم الأعراض الاكلينيكية بواسطة طبيب متخصص ولا يعتمد على التحليل الجينى أو الاختبارات المعملية الأخرى.
3- ما فائدة عمل التحليل الجينى والفحوصات المعملية الأخرى ؟
التحليل الجينى قد ينبئ عن مدى شدة المرض واستجابته للعلاج.
أما الفحوصات المعملية الأخرى مثل سرعة الترسيب، رد الفعل البروتين- Cوالاميلويد بروتين فلها دور فى متابعة نشاط المرض ومدى استجابته للعلاج
4- ماهوالعلاج ؟
العلاج هو عقار الكولشيسين مدى الحياة.
5- ماذا لو لم يتم السيطرة على نشاط المرض بالكولشيسين ؟
بعد الوصول الى أعلى جرعة مسموح بها طبقا لعمر المريض وبعد فترة زمنية كافية للحكم على تأثير العقار (بحد أقصى 6 شهور) هنا يمكن اضافة العلاج البيولوجى حسب توصية الطبيب المتخصص.
6- ما هو علاج النوبات الحادة ؟
الاستمرار على نفس الجرعة من عقار الكولشيسين مع اعطاء أدوية مضادة للالتهاب حسب ما يوصى به الطبيب المتخصص.
7- كيف يتم متابعة المريض ؟
المتابعة تكون شهرية فى أول 3-6 شهور بعد تشخيص المرض والبدء بالعلاج(حسب استجابة المريض للعلاج) ثم تكون كل 3 شهور بعد ذلك لمتابعة الأعراض والفحوصات المعملية لمعرفة مدى استجابتها للعلاج ومعرفة أى تأثيرات جانبية للعلاج والتعامل معها.
8- ما هى تأثيرات المرض على المدى الطويل ؟
لا يوجد تأثيرات مرضية على المدى الطويل طالما كان نشاط المرض تحت السيطرة طول الوقت و يتم التعامل مع ما يستجد فى حينه بواسطة طبيب متخصص.
9- هل هناك علاج وقائى ؟
ليس هناك علاج وقائى ولكن اذا اكتشفنا أن أحد أفراد المريض يحمل نسختين من أحد الجينات المرتبطة بالنشاط المرضى العنيف مثل M694V يتم عمل متابعة لاحتمال ظهور أعراض في بدء العلاج.
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|