the Management of Bleeding in Pediatric Patients with Isolated Thrombocytopenia
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | the Management of Bleeding in Pediatric Patients with Isolated Thrombocytopenia |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 17 Feb 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Introduction
- - Methods
- - Evidence to recommendations: Considerations
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline.
- - Updating of the guideline
- - References
- - Annexes
- Executive Summary
➡️Introduction
Pediatric thrombocytopenia is a complex area of medicine covering a wide age range of different patho etiologies. Establishing the cause of thrombocytopenia has obvious clinical repercussions but is sometimes quite challenging.
Compared to adult practice, there is a relative lack of high-quality research to inform evidence-based guidelines for the diagnosis and management of thrombocytopenia in pediatrics.
The aim of this adapted clinical practice guideline (CPG) is to provide evidence-based recommendations for the diagnosis and management of thrombocytopenia in the pediatric age group.
➡️Scope
This guideline focuses on any disease or condition causing bleeding in neonates, infants, children, and adolescents (with focus on thrombocytopenia). It Provides an evidence-based document for the appropriate clues for diagnosis, and management of different causes of thrombocytopenia in pediatrics.
It also identifies neonates, infants, children, and adolescents at high risk of bleeding and methods of prevention of further bleeding episodes in the target population.
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
- American Society of Hematology 2019 guidelines for immune thrombocytopenia. (ASH 2019)1
- The European guideline on management of major bleeding and coagulopathy following trauma: fifth edition. (European 2019)2
- Management of severe perioperative bleeding: guidelines from the European Society of Anaesthesiology. (ESA 2016)3
- Fetal and neonatal alloimmune thrombocytopenia: recommendations for evidence-based practice, an international approach. (BSH, 2019)4
- Guidelines on transfusion for fetuses, neonates and older children. (BSH 2016)5
- Guidelines for the Laboratory Investigations of heritable disorders of platelet function. (BSH 2011)6
- Updated international consensus report on the investigation and management of primary immune thrombocytopenia. (ASH ITP consensus 2019).7
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
➡️Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. Diagnosis of pediatric isolated thrombocytopenia:
The guideline covers recommendations for diagnosis of isolated thrombocytopenia in neonates as well as in infants and children.
This guideline emphasis on clinical manifestations and investigations for different cases of thrombocytopenia.
B. Management of pediatric isolated thrombocytopenia:
This section includes recommendations and good practice statements on how to manage pediatric thrombocytopenia according to different clinical situations and diagnoses.
C. Prevention of pediatric isolated thrombocytopenia:
This section handles the prevention of thrombocytopenia in pediatrics.
We can summarize the guidelines’ recommendations for management of isolated thrombocytopenia in Pediatric Age Groups in the following:
Diagnosis of pediatric isolated thrombocytopenia:
▪️The NeoBAT is a modified WHO bleeding assessment tool to record neonatal bleeding episodes to standardise the clinical recording of bleeding in premature and term neonates in an intensive care setting. GPS.
▪️ A cranial ultrasound should be performed to screen for intracranial hemorrhage (ICH) in all neonates suspected of fetal/neonatal alloimmune thrombocytopenia (FNAIT) within 24 h of delivery. High, Weak (conditional) .
▪️ A neonate with FNAIT should have platelet counts monitored until the platelets are normal in the absence of treatment. High, Weak (conditional).
▪️ Clues for inherited thrombocytopenia diagnosis:
- Thrombocytopenia has been present since early life.
- A positive family history for a similar disorder.
- Characteristic physical features are present.
- Failure to respond to first-line treatment. GPS.
· Mean platelet volume may be used to differentiate ITP from inherited thrombocytopenia. Strong.
▪️ Flow cytometry should be used in the investigation or confirmation of Bernard Soulier syndrome and abnormalities in the collagen (GpVI and GpIa/IIa) and thrombin receptors (PAR-1). High, Weak (conditional).
▪️ The International Society of Hemostasis and thrombosis – Bleeding assessment tool (ISTH-SCC BAT), a validated general bleeding assessment score, could be used for initial screening of bleeding manifestations. GPS.
▪️ The diagnosis of ITP is based principally on the exclusion of other causes of isolated thrombocytopenia using patient history, physical examination, blood count, and evaluation of the peripheral blood film (to exclude other hematological conditions). Strong.
In isolated thrombocytopenia with no abnormal physical findings and no abnormal blood smear, a bone marrow examination is not required in the initial diagnosis, whether or not treatment is recommended. Strong.
▪️ Testing for antinuclear antibodies is not necessary in the evaluation of children and adolescents with newly diagnosed ITP. High, Strong.
▪️ Bone marrow examination is unnecessary in children and adolescents with the typical features of ITP. Strong.
▪️ Bone marrow examination could be appropriate in those relapsing after remission, in patients not responding to initial treatment options, where splenectomy is considered, or if other abnormalities are detected in the blood count (bicytopenia or pancytopenia) or morphology . Weak (conditional).
▪️ Additional evaluation could include:
- DAT is recommended to exclude coexistent autoimmune hemolytic anemia, especially prior to therapy.
- Immunoglobulin levels.
- Lupus and other markers of autoimmune diseases that might require specific treatment (e.g., test for APLAs, ANAs, anti-cardiolipin antibody, lupus anticoagulant, and serum Igs)
- Chronic infections (hepatitis, cytomegalovirus and/or HIV in at-risk populations or when there is no other explanation)
- Complex immunodeficiency diseases
Genetic screening for inherited thrombocytopenia and bone marrow failure syndromes. Weak (conditional).
➡️Treatment of thrombocytopenia:
▪️ In a neonate with FNAIT, platelets should be transfused immediately if life-threatening bleeding is present to maintain platelet counts initially above 100 x 109 /l and then above 50 x 109 /l for at least 7 days. High, Strong.
▪️ If an ICH is suspected clinically, do not delay platelet transfusion while awaiting confirmation by imaging studies. High, Weak (conditional).
▪️ In the absence of life-threatening bleeding in a neonate, such as intracranial or gastrointestinal bleeding, platelets should be transfused to maintain a platelet count above 30 × 109 /l. High, Weak (conditional)
▪️ In the rare circumstance where either HPA unselected or HPA selected platelets are not available; infuse the neonate with IVIG 1 g/kg. High, Weak (conditional).
▪️ For preterm neonates with very severe thrombocytopenia (platelet count below 25 x 109/l) platelet transfusions should be administered in addition to treating the underlying cause of the thrombocytopenia. Intermediate, Weak (conditional).
▪️ In case of maternal ITP:
1- Management of delivery:
- FNAIT should be excluded by parental testing if the neonate presents with severe thrombocytopenia.
- The mode of delivery should be determined by obstetric indications, not by anticipation of the neonatal platelet count.
- Procedures during labor that may be associated with increased hemorrhagic risk to the fetus should be avoided, specifically the use of fetal scalp electrodes, fetal blood sampling, ventouse delivery, and rotational forceps. Weak (conditional).
2) Management after delivery
- Umbilical cord platelet count should be obtained at the time of delivery or as soon as possible.
- Repeat the platelet count as needed depending on platelet levels, trends in the count, and response to treatment (if any). If cord platelet count is <100 x 109/L, repeat the platelet count daily until stable.
- If platelet count is <50 x 109/L at birth, perform a cranial ultrasound.
- In the case of ICH, give IVIg and limited steroids to maintain platelet count > 100 x 109/L for 1 week if possible and > 50 x 109/L for another week.
- If there is symptomatic bleeding or if platelet count is < 30 x 109/L, with or without platelet transfusion, give IVIg.
- If severe thrombocytopenia continues for > 1 week in a breast-fed infant, consider pausing breastfeeding for a few days to see whether platelet count increases. Weak (conditional).
▪️ Indications for hospitalization in ITP:
- Any severe (grade 4) bleeding requires immediate hospital admission and treatment to increase platelet levels until bleeding has decreased.
- Any moderate (grade 3) bleeding requires hospital review and consideration for admission and therapy.
- Patient with ITP with uncertainty about the diagnosis, those with social concerns, those who live far from the hospital, or those for whom follow up cannot be guaranteed, admission to the hospital may be preferable. Weak (conditional).
▪️ Patients not admitted to the hospital should receive education and expedited follow-up with a hematologist (within 24 to 72 hours of the diagnosis or disease relapse). Weak (conditional).
▪️ At diagnosis, children and adolescents with ITP and mild or even moderate bleeding on a pediatric bleeding assessment tool (grade 1-3) may be managed expectantly with supportive advice and a 24-hour contact point, irrespective of platelet count. Weak (conditional).
▪️ In patients with persistent and chronic ITP, observation or watch and wait is less validated because it is based on the expectation of spontaneous future improvement. Weak (conditional).
▪️ In children with newly diagnosed ITP who have non–life-threatening mucosal bleeding and/or diminished HRQoL can start with any of the 1st line therapy:
- Prednisone (2 - 4 mg/kg/day; maximum, 120 mg daily, for 5-7 days) (Time to initial response 4-14 days).
- IVIG in single dose of 0.8 to 1.0 g/kg. (Time to initial response 1-3 days).
- A second dose of IVIg may be administered if there is a suboptimal initial response and/or ongoing bleeding. Weak (conditional).
▪️ Indications of platelets transfusion in thrombocytopenia:
➡️In non-immune thrombocytopenia
- Platelets < 10 x 109/L transfuse Irrespective of signs of hemorrhage.
- Platelets < 20 x 109/L transfuse in:
- Insertion of a non-tunnelled central venous line.
- Platelets < 50 x 109/L transfuse in:
- Moderate hemorrhage (e.g., gastrointestinal bleeding).
- Surgery, unless minor (except at critical sites). Strong.
▪️ Tranexamic acid (TXA) may be useful in certain dental or surgical procedures or a substantial risk for bleeding. GPS.
▪️ In children with ITP lasting ≥3 months who have non-life-threatening mucosal bleeding and/or diminished health-related quality of life and do not respond to first-line treatment, refer to hematologist for second-line therapies presented in the order they should be pursued:
- Thrombopoietin receptor agonist (eltrombopag or romiplostim)
- Rituximab
- Splenectomy (if possible, splenectomy should be delayed as long as possible after diagnosis because of the potential for spontaneous remission in the first year). Weak (conditional).
▪️ For menorrhagia in adolescent girl with thrombocytopenia manage as emergency treatment. Tranexamic acid can be useful and consult gynecologist for hormonal therapy. GPS.
➡️Prevention of thrombocytopenia:
▪️ In patients identified by screening or sisters of patients with FNAIT, the presence and/or concentration of HPA antibodies in subsequent pregnancies may be useful to determine the risk of FNAIT. High, Weak (conditional).
▪️ Consecutive assessments of levels of anti-HPA-1a antibody in HPA-1a-immunised women may be useful in identifying the risk of FNAIT. High, Weak (conditional).
▪️ Antenatal IVIG administration to the mother, commencing 1 g/kg/week at 12–16 weeks gestation, increase to 2 g/kg/week at 20 weeks or IVIG 1 g/kg/week at 12–16 weeks with the addition of corticosteroids at 1 mg/kg/day at 20 weeks or IVIG 0.5 g/kg/week at 12–16 weeks for the entire pregnancy or IVIG 2 g/kg/week at 12–16 or IVIG 2 g/kg/week at 12–16 weeks, add corticosteroids 1 mg/kg/day at 20 weeks should be suggested to all women in a subsequent pregnancy with maternal fetal incompatibility who have had a previous fetus or neonate with FNAIT-related ICH. High, Weak (conditional).
▪️ If corticosteroids are used with IVIG, dexamethasone should not be used because of the associated risk of oligohydramnios. High, Weak (conditional).
▪️ In a child with history of bleeding, avoid the following drugs:
- Salicylates.
- NSAID.
- Anticoagulants. GPS.
▪️Consider platelet transfusion to prevent bleeding in severe thrombocytopenia (platelet count < 10 x 109/l) caused by abciximab . Intermediate, Weak (conditional).
▪️Following trauma, severely injured patients should be transported directly to an appropriate trauma facility. The time between injury and bleeding control should be minimized. Local compression is recommended to limit life-threatening bleeding. High, Strong.
▪️ Patients with an obvious bleeding source and those presenting with hemorrhagic shock in extremis and a suspected source of bleeding should undergo an immediate bleeding control procedure. High, Strong.
▪️Use focused assessment with sonography in trauma (FAST) ultrasound for the detection of free fluid in patients with torso trauma. High, Strong.
▪️ Early imaging using contrast-enhanced whole-body CT (WBCT) for the detection and identification of type of injury and potential source of bleeding is recommended. High, Strong.
▪️ Laboratory screening of patients treated or suspected of being treated with anticoagulant agents should be done. High, Strong.
▪️ Platelets should be administered to maintain a platelet count above 50 x 109/L. High, Strong.
▪️ Maintain a platelet count above 100 x 109/L in patients with ongoing bleeding and/or traumatic brain injury. High, Strong.
▪️ Maintain a hemoglobin level of 70 to 90 g/L in patients with ongoing bleeding and/or traumatic brain injury. High, Strong.
▪️ We recommend that TXA be administered to the trauma patient who is bleeding or at risk of significant hemorrhage as soon as possible and within 3 h after injury at a loading dose of 1 g infused over 10 min, followed by IV. infusion of 1 g over 8 h. High, Strong.
▪️ Before surgery or invasive procedures, use a structured patient interview or standardized questionnaire which considers clinical and family bleeding history and detailed information on the patient's medication. High, Weak (conditional).
▪️ Routine use of conventional coagulation screening tests such as activated partial thromboplastin time (aPTT), international normalized ratio (INR) and platelet count is not recommended in elective surgery. High, Weak (conditional).
▪️ In patients with normal platelet counts, preoperative platelet function testing is suggested only in association with a positive bleeding history, decreased platelet function caused by medical conditions or antiplatelet medication. High, Weak (conditional).
▪️Bleeding time is not recommended for preoperative bleeding risk stabilization as it is influenced by many variables. Weak (conditional).
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy) platform, WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: (PREPARE-2022CN791). Link: http://www.guidelines-registry.org/- Recommendations
Part I: Diagnosis of thrombocytopenia:
|
|
CPGL Source |
Recommendation |
Strength of Recommendation |
Quality of Evidence |
|
|
1.1 What is a validated bleeding score for neonates? |
|||||
|
|
The NeoBAT is a modified WHO bleeding assessment tool to record neonatal bleeding episodes to standardise the clinical recording of bleeding in premature and term neonates in an intensive care setting. |
GPS* |
|
||
|
1.2 What is the initial evaluation for neonates presenting with bleeding and thrombocytopenia? |
|||||
|
BSH 2011 |
• An evaluation of patients with abnormal bleeding requires objective clinical assessment of bleeding history, any family history, history of maternal drug intake, history of perinatal sepsis and physical examination for any physical anomalies or organomegaly, and any associated morbidity. • Laboratory investigations of platelet number and function are recommended in any patient where bleeding symptoms are not fully explained by standard clinical laboratory investigations. |
GPS
|
|
||
|
1.3 What is the initial evaluation for fetal/neonatal alloimmune thrombocytopenia (FNAIT)? |
|||||
|
BSH FNAIT 2019 |
• A cranial ultrasound should be performed to screen for intracranial hemorrhage (ICH) in all neonates suspected of fetal/neonatal alloimmune thrombocytopenia (FNAIT) within 24 h of delivery. |
High |
Weak (conditional) |
||
|
ISTH**
|
FNAIT testing should include: • HPA genotyping from the mother, the neonate or, if not available, the father • Alloantibody testing of maternal serum • A crossmatch with paternal platelets |
|
GPS |
||
|
BSH FNAIT 2019 |
• A neonate with FNAIT should have platelet counts monitored until the platelets are normal in the absence of treatment. |
High |
Weak (conditional) |
||
*A novel approach to standardised recording of bleeding in a high-risk neonatal population
Added to the implementation tool 44
** Investigations for fetal and neonatal alloimmune thrombocytopenia: communication from the SSC of the ISTH. Journal of Thrombosis and Hemostasis. 2018
|
1.1 What are the antenatal diagnostic tests for possible FNAIT? |
|||
|
BSH FNAIT 2019 |
Fetal HPA typing, preferably using non-invasive methods, if adequately quality assured, should be performed during pregnancy when the father is unknown, unavailable for testing or heterozygous for the implicated antigen. Balance of harms and benefits: The alternative is amniocentesis, which is associated with risk of fetal demise. |
High |
Strong |
|
1.2 What are the points in history and examination suggestive of inherited thrombocytopenia? |
|||
|
ITP Consensus 2019 |
• Thrombocytopenia has been present since early life • A positive family history for a similar disorder. • Characteristic physical features are present. • Failure to respond to first-line treatment. |
|
GPS |
|
1.3 What are the tests required to exclude inherited thrombocytopenia? |
|||
|
ITP Consensus 2019 |
Mean platelet volume may be used to differentiate ITP from inherited thrombocytopenia; increased mean platelet volume can be suspected on smear if there are many large platelets. |
|
Strong |
|
BSH 2011 |
Flow cytometry should be used in the investigation or confirmation of Bernard Soulier syndrome by moderate to marked reduction in CD42a (GpIX) and CD42b (Gp1bα) |
High |
Weak (conditional) |
|
BSH 2011 |
Flowcytometry may also be used to investigate abnormalities in the collagen (GpVI and GpIa/IIa) and thrombin receptors (PAR-1). |
High |
Strong |
|
1.4 What is the initial evaluation for children and adolescents presenting with bleeding and thrombocytopenia? |
|||
|
ITP consensus 2019 |
A complete history, physical examination, full blood count, and expert analysis of the peripheral blood smear should be performed and carefully evaluated at initial diagnosis to exclude secondary causes of thrombocytopenia. |
|
Weak (conditional) |
|
1.5 What is a validated general bleeding score? |
|||
|
ISTH SSC – BAT* |
International Society of Hemostasis and thrombosis – Bleeding assessment tool (ISTH-SCC BAT)* comprises 14 categories for assessing bleeding symptoms could be used for initial screening of bleeding manifestations |
|
GPS |
* Added to implementation tools
|
1.1 What are the diagnostic criteria for immune thrombocytopenia? |
|||
|
ITP Consensus 2019 |
The diagnosis of ITP is based principally on the exclusion of other causes of isolated thrombocytopenia using patient history, physical examination, blood count, and evaluation of the peripheral blood film (to exclude other hematological conditions). |
|
Strong |
|
ITP consensus 2019 |
In isolated thrombocytopenia with no abnormal physical findings and no abnormal blood smear, a bone marrow examination is not required in the initial diagnosis, whether or not treatment is recommended. |
|
Strong |
|
1.2 What are the additional diagnostic tests required in children and adolescents with ITP? |
|||
|
ITP Consensus 2019 |
Quantitative immunoglobulin (Ig) level testing is indicated to exclude an immune deficiency syndrome or before treatment with IVIg. In children, Ig level testing may be considered at baseline. |
|
Weak (conditional) |
|
ASH 2019 |
Testing for antinuclear antibodies is not necessary in the evaluation of children and adolescents with newly diagnosed ITP. |
High |
Strong |
|
1.3 What are the indications of bone marrow examination in patients with ITP? |
|||
|
ASH 2019 |
Newly diagnosed ITP in children • Bone marrow examination is unnecessary in children and adolescents with the typical features of ITP • Bone marrow examination is not necessary in children who fail IVIG therapy • Bone marrow examination is also not necessary in patients prior to initiation of treatment of corticosteroids. |
High High Intermediate |
Strong Strong Weak (conditional) |
|
ITP consensus 2019 |
• Bone marrow examination could be appropriate in those relapsing after remission, in patients not responding to initial treatment options, where splenectomy is considered, or if other abnormalities are detected in the blood count (bicytopenia or pancytopenia) or morphology • This examination should ideally include an aspirate, biopsy, flow cytometry, and cytogenetics |
Intermediate
|
Weak (conditional) Weak (conditional) |
|
1.4 What are the subsequent investigations in children and adolescents with persistent or chronic ITP? |
|||
|
ITP consensus 2019 |
Additional evaluation could include • A DAT is recommended to exclude coexistent autoimmune hemolytic anemia, especially prior to therapy. • Immunoglobulin levels. • Lupus and other markers of autoimmune diseases that might require specific treatment (e.g., test for APLAs, ANAs, anti-cardiolipin antibody, lupus anticoagulant, and serum Igs) • Chronic infections (hepatitis, cytomegalovirus and/or HIV in at-risk populations or when there is no other explanation) • Complex immunodeficiency diseases • Genetic screening for inherited thrombocytopenia and bone marrow failure syndromes |
|
Weak (conditional) |
|
ITP consensus 2019 |
Bone marrow examination could be appropriate in those relapsing after remission, in patients not responding to initial treatment options, where splenectomy is considered, or if other abnormalities are detected in the blood count or morphology This examination should ideally include an aspirate, biopsy, flow cytometry, and cytogenetics |
Intermediate
|
Weak (conditional) Weak (conditional) |
|
ASH 2019 |
Recommendation against routine testing for H pylori in children with persistent or chronic ITP. |
High |
Strong |
Part II: Treatment of thrombocytopenia:
|
CPGL Source |
Recommendation |
Strength of Recommendation |
Quality of Evidence |
|||
|
2.1 What is the initial treatment of bleeding in a neonate with FNAIT? |
||||||
|
BSH FNAIT 2019 |
Platelets should be transfused immediately if life-threatening bleeding is present. |
High |
Strong |
|||
|
In the presence of life-threatening bleeding such as intracranial or gastrointestinal bleeding, platelets should be transfused to maintain platelet counts initially above 100 x 109 /l and then above 50 x 109 /l for at least 7 days. |
High |
Weak (conditional) |
||||
|
If an ICH is suspected clinically, do not delay platelet transfusion while awaiting confirmation by imaging studies. |
High |
Weak (conditional) |
||||
|
|
In the absence of life-threatening bleeding in a neonate, such as intracranial or gastrointestinal bleeding, platelets should be transfused to maintain a platelet count above 30 × 109 /l. |
High |
Weak (conditional) |
|||
|
|
In the rare circumstance where either HPA unselected or HPA selected platelets are not available; infuse the neonate with IVIG 1 g/kg.
|
High |
Weak (conditional) |
|||
|
2.2 What are the indications of platelet transfusion in a neonate with thrombocytopenia? |
||||||
|
BSH 2016
|
For preterm neonates with very severe thrombocytopenia (platelet count below 25 x 109/l) platelet transfusions should be administered in addition to treating the underlying cause of the thrombocytopenia. • If Platelet count < (25 x 109/l) transfuse in Neonates with no bleeding • If Platelet count < (50× 109/l) transfuse in Neonates with bleeding, current coagulopathy, before surgery, or infants with FNAIT if previously affected sibling with ICH. • If Platelet count < (100 x 109/l) transfuse in Neonates with major bleeding or requiring major surgery (e.g., neurosurgery). |
Intermediate |
Weak (conditional) |
|||
|
2.3 What is the management of a neonate of mother with ITP? |
||||||
|
ITP consensus 2019
|
1) Management of delivery • Cordocentesis and fetal scalp blood sampling should be avoided. |
|
Weak (conditional) |
|||
|
• FNAIT should be excluded by parental testing if the neonate presents with severe thrombocytopenia. |
|
Weak (conditional) |
||||
|
• The mode of delivery should be determined by obstetric indications, not by anticipation of the neonatal platelet count. |
|
Strong |
||||
|
• Procedures during labor that may be associated with increased hemorrhagic risk to the fetus should be avoided, specifically the use of fetal scalp electrodes, fetal blood sampling, ventouse delivery, and rotational forceps. |
|
Weak (conditional) |
||||
|
ITP consensus 2019
|
2) Management after delivery • Umbilical cord platelet count should be obtained at the time of delivery or as soon as possible. |
|
Weak (conditional) |
|||
|
• Repeat the platelet count as needed depending on platelet levels, trends in the count, and response to treatment (if any). If cord platelet count is <100 x 109/L, repeat the platelet count daily until stable. |
|
Weak (conditional) |
||||
|
• If platelet count is <50 x 109/L at birth, perform a cranial ultrasound. |
|
Weak (conditional) |
||||
|
• In the case of ICH, give IVIg and limited steroids to maintain platelet count > 100 x 109/L for 1 week if possible and > 50 x 109/L for another week. |
|
Weak (conditional) |
||||
|
• If there is symptomatic bleeding or if platelet count is < 30 x 109/L, with or without platelet transfusion, give IVIg. |
|
Weak (conditional) |
||||
|
• If severe thrombocytopenia continues for > 1 week in a breast-fed infant, consider pausing breastfeeding for a few days to see whether platelet count increases. |
|
Weak (conditional) |
||||
|
2.4 What are the indications of hospitalization in pediatric patients with ITP? |
||||||
|
ITP consensus 2019
|
• Any severe (grade 4) bleeding requires immediate hospital admission and treatment to increase platelet levels until bleeding has decreased. |
|
Weak (conditional) |
|||
|
• Any moderate (grade 3) bleeding requires hospital review and consideration for admission and therapy. |
|
Weak (conditional) |
||||
|
• Worsening bleeding or significant comorbidities |
|
Weak (conditional) |
||||
|
• Risk of ICH (e.g., head trauma or unexplained headaches); patients at higher risk for ICH include those with a history of moderate or severe bleed in the preceding 28 days, recent administration (within 8 hours) of NSAIDs, and another clinically significant coagulopathy (e.g., von Willebrand disease). |
|
Weak (conditional) |
||||
|
• A change in behavior or mood consistent with significant depression or irritability. |
|
Weak (conditional) |
||||
|
• Parents are anxious about bleeding and do not believe that they can control (young child) or restrict (older child) their child’s activity. |
|
Weak (conditional) |
||||
|
• Parents cannot be relied upon to bring the child back readily if there is an emergency (e.g., they live too far away, they cannot afford to return, there are additional social concerns). |
|
Weak (conditional) |
||||
|
• Child has not spontaneously improved and must be overly restricted in activities. |
|
Weak (conditional) |
||||
|
ASH 2019 |
• Patient with ITP with uncertainty about the diagnosis, those with social concerns, those who live far from the hospital, or those for whom follow up cannot be guaranteed, admission to the hospital may be preferable. |
GPS |
|
|||
|
• Patients not admitted to the hospital should receive education and expedited follow-up with a hematologist (within 24 to 72 hours of the diagnosis or disease relapse). |
GPS |
|
||||
|
2.5 What is the initial treatment of pediatric patients with ITP? |
||||||
|
ITP consensus 2019
|
I. Watch-and-wait policy based on clinical classification 1. At diagnosis, children and adolescents with ITP and mild or even moderate bleeding on a pediatric bleeding assessment tool (grade 1-3) may be managed expectantly with supportive advice and a 24-hour contact point, irrespective of platelet count 2. In patients with persistent and chronic ITP, observation or watch and wait is less validated because it is based on the expectation of spontaneous future improvement. |
|
Strong
Weak (conditional) |
|||
|
ASH 2019
|
II. In children with newly diagnosed ITP who have non–life-threatening mucosal bleeding and/or diminished HRQoL can start with any of the 1st line therapy: - Prednisone (2 - 4 mg/kg/day; maximum, 120 mg daily, for 5-7 days) (Time to initial response 4-14 days). For patients receiving corticosteroids, the treating physician should ensure the patient is adequately monitored for potential side effects regardless of the duration or type of corticosteroid selected (see implementation tool) |
GPS
|
Weak (conditional) |
|||
|
ASH 2019
ITP consensus 2019 |
- For patients where corticosteroids are contra-indicated or otherwise not preferred, the intravenous immunoglobulin can be used - IVIG in single dose of 0.8 to 1.0 g/kg. (Time to initial response 1-3 days). - A second dose of IVIg may be administered if there is a suboptimal initial response and/or ongoing bleeding. |
High
|
Strong
Weak (conditional) |
|||
|
2.6 What are the indications of platelet transfusion in pediatric patients with thrombocytopenia? |
||||||
|
BSH 2016 |
In non-immune thrombocytopenia § Platelets < 10 x 109/L transfuse Irrespective of signs of hemorrhage. § Platelets < 20 x 109/L transfuse in: o Insertion of a non-tunnelled central venous line. § Platelets < 50 x 109/L transfuse in: o Moderate hemorrhage (e.g., gastrointestinal bleeding). o Surgery, unless minor (except at critical sites) § Platelets < 75-100 x 109/L transfuse in o Major hemorrhage or significant post-operative bleeding o Surgery at critical sites: central nervous system including eyes |
Intermediate |
Weak (conditional) |
|||
|
In immune thrombocytopenia Patients with immune thrombocytopenia should only be transfused with platelets for life-threatening bleeding. |
Intermediate |
Strong |
||||
|
2.7 What is the treatment of life-threatening bleeding in pediatric patient with thrombocytopenia? |
||||||
|
ITP consensus 2019
|
For children with immune thrombocytopenia 1. Combination therapy including IV corticosteroids, IVIg, with or without platelet transfusion. - Dose: IV methylprednisolone: 30 mg/kg per day, IVIg: 0.8-1.0 g/kg/d. - A second dose of IVIg and IV steroids may be required if a platelet response is not seen within 24 hours of the initial dose. |
|
Weak (conditional) |
|||
|
2. Platelet transfusion. |
|
Weak (conditional) |
||||
|
3. Antifibrinolytics may be given if bleeding continues despite therapy. |
|
Weak (conditional) |
||||
|
4. If there is an ICH, emergency splenectomy and/or neurosurgical control of bleeding should be considered in conjunction with emergency platelet-raising therapy. |
|
Weak (conditional) |
||||
|
5. Thrombopoietin receptor agonist (TPO-Ras) should be considered; they may aid the acute response in patients and prevent a decrease in platelet count if initial response to emergency therapy is lost. |
|
Weak (conditional) |
||||
|
BSH 2016 |
For children with non-immune thrombocytopenia: Platelet transfusion is the main line of treatment. |
Intermediate |
Strong |
|||
|
2.8 What is the adjuvant treatment in pediatric patient with thrombocytopenia? |
||||||
|
ITP Consensus 2019 |
- Tranexamic acid (TXA) may be useful in certain dental or surgical procedures or a substantial risk for bleeding. - Dose: 15-20mg/kg every 8 hours orally and e-aminocaproic acid 1-5 g every 4-6 hours [maximum dose, 24 g/d] |
|
Weak (conditional) |
|||
|
2.9 What is the long-term treatment plan in pediatric patients with thrombocytopenia? |
||||||
|
ASH 2019 |
In children with ITP lasting ≥3 months who have non-life-threatening mucosal bleeding and/or diminished health-related quality of life and do not respond to first-line treatment, refer to hematologist for second-line therapies presented in the order they should be pursued: 1. Thrombopoietin receptor agonist (eltrombopag or romiplostim) 2. Rituximab 3. Splenectomy (if possible, splenectomy should be delayed as long as possible after diagnosis because of the potential for spontaneous remission in the first year) |
|
Weak (conditional) |
|||
|
2.10 what is the treatment of menorrhagia in adolescent girl with thrombocytopenia |
||||||
|
|
Manage as emergency treatment. Tranexamic acid can be useful and consult gynecologist for hormonal therapy |
GPS |
|
|||
Part III: Prevention of thrombocytopenia:
|
CPGL Source |
Recommendation |
Strength of Recommendation |
Quality of Evidence |
||
|
3.1 How to prevent serious bleeding in a fetus/ neonate with FNAIT? |
|||||
|
BSH FNAIT 2019 |
At time of delivery: • If the fetal platelet count is unknown, assisted delivery and invasive procedures on the fetus during delivery should be avoided, including forceps, vacuum-assisted delivery, scalp blood sampling and scalp electrodes • A cord blood sample should be sent for platelet count determination immediately after delivery • HPA-selected platelets should be available at the time of delivery |
High
High High |
Weak (conditional) |
||
|
BSH FNAIT 2019 |
After delivery: • In the absence of life-threatening bleeding in a neonate, such as intracranial or gastrointestinal bleeding, platelets should be transfused to maintain a platelet count above 30 x 109/l. |
High |
Weak (conditional) |
||
|
3.2 How to prevent alloimmunization (maternal & transfusion related)? |
|||||
|
BSH FNAIT 2019 |
In patients identified by screening or sisters of patients with FNAIT, the presence and/or concentration of HPA antibodies in subsequent pregnancies may be useful to determine the risk of FNAIT. |
Intermediate |
Weak (conditional) |
||
|
Consecutive assessments of levels of anti-HPA-1a antibody in HPA-1a-immunised women may be useful in identifying the risk of FNAIT. |
Intermediate |
Weak (conditional) |
|||
|
Antenatal IVIG administration to the mother, commencing 1 g/kg/week at 12–16 weeks gestation, increase to 2 g/kg/week at 20 weeks or IVIG 1 g/kg/week at 12–16 weeks with the addition of corticosteroids at 1 mg/kg/day at 20 weeks or IVIG 0.5 g/kg/week at 12–16 weeks for the entire pregnancy or IVIG 2 g/kg/week at 12–16 or IVIG 2 g/kg/week at 12–16 weeks, add corticosteroids 1 mg/kg/day at 20 weeks should be suggested to all women in a subsequent pregnancy with maternal fetal incompatibility who have had a previous fetus or neonate with FNAIT-related ICH. |
High |
Weak (conditional) |
|||
|
If corticosteroids are used with IVIG, dexamethasone should not be used because of the associated risk of oligohydramnios. |
High |
Weak (conditional) |
|||
|
3.3 What are the drugs to be avoided in a child with history of bleeding? |
|||||
|
|
• Salicylates • NSAID • Anticoagulants |
|
GPS |
||
|
3.4 How to prevent bleeding in a child receiving antiplatelet medications? |
|||||
|
BSH 2016 |
Consider platelet transfusion to prevent bleeding in severe thrombocytopenia (platelet count < 10 x 109/l) caused by abciximab |
Intermediate |
Weak (conditional) |
||
|
3.5 How to prevent further bleeding in a child following trauma? |
|||||
|
Euro Trauma 2019 |
Severely injured patients should be transported directly to an appropriate trauma facility. |
High |
Strong |
||
|
The time between injury and bleeding control should be minimized. |
High |
Strong |
|||
|
Local compression is recommended to limit life-threatening bleeding. |
High |
Strong |
|||
|
Use adjunct tourniquet to stop life-threatening bleeding from open extremity injuries in the pre-surgical setting. |
High |
Strong |
|||
|
Use adjunct pelvic binder to limit life-threatening bleeding in the presence of a suspected pelvic fracture in the pre-surgical setting. |
High |
Strong |
|||
|
Patients with an obvious bleeding source and those presenting with hemorrhagic shock in extremis and a suspected source of bleeding should undergo an immediate bleeding control procedure. |
High |
Weak (conditional) |
|||
|
Use focused assessment with sonography in trauma (FAST) ultrasound for the detection of free fluid in patients with torso trauma. |
High |
Weak (conditional) |
|||
|
Early imaging using contrast-enhanced whole-body CT (WBCT) for the detection and identification of type of injury and potential source of bleeding is recommended. |
High |
Strong |
|||
|
Laboratory screening of patients treated or suspected of being treated with anticoagulant agents should be done. |
High |
Weak (conditional) |
|||
|
Platelets should be administered to maintain a platelet count above 50 x 109/L. |
High |
Weak (conditional) |
|||
|
Maintain a platelet count above 100 x 109/L in patients with ongoing bleeding and/or traumatic brain injury. |
Intermediate |
Weak (conditional) |
|||
|
Transfuse at an initial dose of four to eight single platelet units or one aphaeresis pack. |
Intermediate |
Weak (conditional) |
|||
|
Maintain a hemoglobin level of 70 to 90 g/L in patients with ongoing bleeding and/or traumatic brain injury. |
High |
Weak (conditional) |
|||
|
|
We recommend that TXA be administered to the trauma patient who is bleeding or at risk of significant hemorrhage as soon as possible and within 3 h after injury at a loading dose of 1 g infused over 10 min, followed by IV. infusion of 1 g over 8 h. |
High |
Strong |
||
|
|
We recommend that protocols for the management of bleeding patients consider administration of the first dose of TXA en route to the hospital. |
High |
Weak (conditional) |
||
|
3.6 How to assess the risk of bleeding in children during preoperative evaluation? |
|||||
|
ESA 2016 |
Before surgery or invasive procedures, use a structured patient interview or standardized questionnaire which considers clinical and family bleeding history and detailed information on the patient's medication. |
High |
Weak (conditional) |
||
|
Routine use of conventional coagulation screening tests such as activated partial thromboplastin time (aPTT), international normalized ratio (INR) and platelet count is not recommended in elective surgery. We recommend the use of standardized questionnaires on bleeding and drug history as preferable to the routine use of conventional coagulation screening tests such as activated partial thromboplastin time (aPTT), international normalized ratio (INR) and platelet count in elective surgery. |
High |
Weak (conditional) |
|||
|
In patients with normal platelet counts, preoperative platelet function testing is suggested only in association with a positive bleeding history, decreased platelet function caused by medical conditions or antiplatelet medication. |
Intermediate |
Strong |
|||
|
Bleeding time is not recommended for preoperative bleeding risk stabilization as it is influenced by many variables. |
|
Weak (conditional) |
|||
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Galila Mohamed Mokhtar |
Paediatrics Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt |
Editor, Clinical expert, GAG member |
|||
|
Prof. Amira Adly |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Dina Ezzat |
Professor of Paediatrics and Paediatric Haematology, Beni-Suef University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Gehan Lotfy |
Professor of Paediatrics, Minia University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Hoda Hassab |
Professor of Paediatrics and Paediatrics Haematology, Alexandria University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Ilham Youssry |
Professor of Paediatrics and Head of the Paediatric Haematology & BMT unit, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Iman Ragab |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Laila Sherief |
Professor of Paediatrics and Paediatrics Haematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Marwa Zakaria |
Associate Professor of Paediatrics, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Mervat Hesham |
Professor of Paediatrics, Paediatric Haematology & Oncology Unit, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Niveen Salama |
Associate Professor of Paediatrics, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Dr. Nouran Yousef |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Rasha Abdel-Raouf Afifi |
Professor of Paediatrics and Paediatric Haematology, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Rasha El-Ashry |
Professor of Paediatrics, Mansoura University, Egypt |
Clinical expert GAG member |
|||
|
Dr. Sara Makkeyah |
Associate Professor of Paediatrics, Paediatric Haematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Sonia Adolf |
Professor of Paediatrics and Paediatric Haematology, National Research Center, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Eman Abdel-Raouf |
Professor of Pediatrics and Pediatric Hematology, Cairo University, Egypt. |
||||
|
Prof. Mervat Atfy |
Professor of Pediatrics and Pediatric Hematology, Zagazig University, Egypt. |
||||
|
Prof. Mohamed Abdel-Mohsen |
Professor of Pediatrics and Pediatric Hematology/Oncology, Ain Shams University, Egypt. |
||||
|
International Peer Reviewers |
|||||
|
Prof. James Bussel |
Professor pf Pediatric Hematology/Oncology, Pediatrics Department, Well Cornell Medical College, Newyork, USA |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge the British Society of Haematology (BSH), the American Society of Hematology (ASH), and the European Society of Anaesthesiology (ESA) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
|
ANA |
Anti-nuclear antibody |
|
APLA |
Anti-phospholipid antibody |
|
aPTT |
activated partial thromboplastin time |
|
ASH |
American Society of Hematology |
|
BSH |
British Society of Haematology |
|
BSH |
British Society for Haematology |
|
CPG |
Clinical Practice Guideline |
|
DAT |
Direct Antiglobulin test |
|
EPG |
Egyptian Paediatric Clinical Practice Guidelines Committee |
|
EPG CPG |
EPG Clinical Practice Guideline |
|
ERG |
External Review Group |
|
FNAIT |
Fetal / neonatal alloimmune thrombocytopenia |
|
FAST |
Focused assessment with sonography in trauma |
|
GAG |
Guideline Adaptation Group |
|
GDG |
Guideline Development Group |
|
Gp |
Glycoprotein |
|
GPS |
Good practice statement |
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
|
HPA |
Human platelet antibody |
|
HRQoL |
Health related quality of life |
|
ICH |
Intracranial hemorrhage |
|
Igs |
Immunoglobulins |
|
INR |
International normalized ratio |
|
ISTH |
International society of Thrombosis and Hemostasis |
|
ISTH-SSC |
|
|
ITP |
Immune thrombocytopenia |
|
ITP IWG |
ITP international working group |
|
IVIg |
Intravenous immunoglobulin |
|
HDFN |
Haemolytic disease of the fetus and newborn |
|
HSCT |
Haematopoietic stem cell transplant |
|
HL |
Hodgkin lymphoma |
|
Ig |
Immunoglobulin |
|
IHN |
International hemovigilance network |
|
Non-IT |
Non-immune thrombocytopenia |
|
NSAIDs |
Non-steroidal anti-inflammatory drugs |
|
PAR-1 |
protease-activated receptor-1 |
|
PICO |
population, intervention, comparison, and outcomes |
|
PIPOH |
Patient population, intervention, professionals, outcomes, and healthcare context |
|
QoE |
Quality of evidence |
|
RBC |
Red blood cell |
|
RCT |
Randomized-controlled trials |
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
|
SoR |
Strength of recommendation |
|
WBC |
White blood cells |
|
WBCT |
Whole-body CT |
- Introduction
Thrombocytopenia was defined as a platelet count of less than 150 000 per microliter and can be acquired or inherited.8 However, platelet counts between 100 and 150 × 109/L do not necessarily indicate disease if they have been stable,9 and a cutoff value of 100 ×109/L may be more appropriate to identify a pathologic condition.10 Isolated thrombocytopenia is defined as a low platelet count in the absence of abnormalities of RBC and WBC lineages and no signs or symptoms of systemic illness. The most prevalent etiologies are ITP and drug-induced ITP (DITP).11
Establishing the cause of thrombocytopenia has obvious clinical repercussions but is sometimes quite challenging. We aimed to describe the different etiologies, clinical presentation, clues for diagnosis and management of primary immune thrombocytopenic purpura (ITP) and non-immune thrombocytopenia (non-IT).
An individual with newly diagnosed thrombocytopenia is more likely to have an acquired disorder rather than an inherited genetic mutation. The myriad causes of acquired thrombocytopenia include autoimmune diseases, increased platelet consumption, splenomegaly, marrow suppression, and bone marrow failure. Two important clinical characteristics for recognizing inherited thrombocytopenia syndromes are the age of presentation and the chronicity/duration of symptoms. In the event of severe thrombocytopenia or profound platelet dysfunction, thrombocytopenia is usually recognized during the perinatal period. Milder disorders are noted sporadically at times of hemostatic stress (onset of menses).12
Depending on the patient’s age of onset, the causes of thrombocytopenia will vary. In newborn, a prevalence of thrombocytopenia reaching 22–35% of all admissions neonatal intensive care units has been reported, 13, 14 particularly in very low birth weight preterm neonates in which a prevalence of 70–80% has been reported. 15, 16 A recent meta-analysis reported a prevalence of 24 % across 21 studies.
In preterm neonates, early-onset thrombocytopenia (<72 h) is usually secondary to antenatal causes, has a characteristic pattern and resolves without complications or the need for treatment. By contrast, late-onset thrombocytopenia in preterm neonates (>72 h) is nearly always due to post-natal acquired bacterial infection and/or necrotizing enterocolitis, which rapidly leads to severe thrombocytopenia (platelet count < 50 x 109/l). Thrombocytopenia is much less common in term neonates and the most important cause is neonatal alloimmune thrombocytopenia (NAIT).17
Immune thrombocytopenia (ITP) is relatively common in childhood, with an annual incidence of 1.9–6.4 per 100,000 children.18 Diagnosis is made when isolated thrombocytopenia occurs in the absence of identifiable and specific precipitants.19 Guidelines have recommended a minimum evaluative process to look for secondary causes of thrombocytopenia before the diagnosis of ITP.20 Differential diagnosis includes infectious, immune, hematologic, endocrine, and neoplastic causes of thrombocytopenia. 21-25
Diagnosis of ITP in children is essentially one of exclusion. To differentiate it from other conditions, medical history should include type and severity of bleeding, systemic symptoms, history of respiratory infections, recent live viral vaccine, medications, presence of bone pain, and family history of bleeding disorders. Clinical examination should include observation for any dysmorphic features, especially skeletal anomalies, and the presence or absence of hepatosplenomegaly and/ or lymphadenopathy.26
Khalifa et al. reported in a large Egyptian study a 30% rate of chronic ITP, matching the international reports.27 There was no gender preference in most acute ITP studies; however, chronic ITP was more frequent in females in Egypt.28 A retrospective report from Lebanon indicated a much lower chronicity rate of 10%.29
Secondary ITP and non-IT are rare and sometimes difficult to recognize. Moreover, other manifestations of the underlying disease may emerge only during the follow-up period.30 Red flags that raise the suspicion of secondary ITP and other non-immune causes of thrombocytopenia include positive family history, older age (adolescence), chronic course, platelet size either above or below the normal range, moderate (instead of severe) thrombocytopenia at first presentation, nonresponse to first-line treatments, and new symptoms or laboratory abnormalities during the disease course.31
Patients with thrombocytopenia typically experience mucocutaneous bleeding. The presence of joint or extensive soft tissue bleeding suggests coagulation abnormalities. Ischemic limb of skin necrosis should raise suspicion of heparin-induced thrombocytopenia.32
There is no single hematologic or biochemical test that is conclusive for a given mechanism of thrombocytopenia.33 If a decision is made to observe the child, even in typical cases, a CBC and blood smear should be repeated periodically to exclude the evolution of a serious bone marrow or other hematologic disorder until the diagnosis is clear or recovery has occurred.7
From registry data, 0% to 4% of children with newly diagnosed ITP have severe bleeding requiring immediate intervention34 with incidence of ICH from; 0% to 1%.35 Risk factors for ICH include low platelet count, non-steroidal anti-inflammatory drugs (NSAIDs) and arteriovenous malformation. Consensus favors consideration of multiple factors when deciding to treat or not, including bleeding symptoms, platelet count, recent trauma, existence of headache, recent medication use, psychosocial and lifestyle issues.7
In children, the symptoms of ITP are of greater impact in treatment decisions than platelet count. The ITP IWG recommends a treatment goal of a safe platelet count in the absence of bleeding, not a normal platelet count.10 Most children do not require therapy “Expectant watch-and-wait policy”.7
The most recent updated consensus report on the investigations and management of primary ITP stated the following: 1) Treatment goals should be individualized to the patient and the phase of the disease, 2) Treatment should prevent severe bleeding episodes, 3) treatment should maintain a target platelet level of 20-30 x 109 at least for symptomatic patients (because risk of major bleeding increases below this level), 4) treatment should be with minimal toxicity, 4) treatment should optimize health related quality of life (HRQoL).7
Although there are numerous publications on the diagnosis and treatment of ITP, knowledge gaps remain, and up-to-date expert opinion and experience are key elements for management of cases.
The aim of this adapted clinical practice guideline (CPG) is to provide evidence-based recommendations for the diagnosis and managment of thrombocytopenia in the paediatric age group. These recommendations were adapted from the relevant CPGs using a formal methodology for CPG adaptation: the Adapted-ADAPTE.
➡️Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of bleeding in pediatric patients with isolated thrombocytopenia.
It provides guidance to primary health care physicians at Ministry of Health (MOH), general practitioners, family medicine specialists and pediatricians.
This guideline focuses on the diagnosis, treatment and prevention of thrombocytopenia in pediatrics. It provides an evidence-based document for how to prevent mortality and morbidity from bleeding, prevent sequelae and disabilities, and prevent further episodes.
This version of the guideline includes recommendations and good practice statements for the diagnosis, management and prevention of bleeding in the pediatric age group with special focus on thrombocytopenia.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search were: childhood thrombocytopenia, pediatric thrombocytopenia, immune thrombocytopenic purpura, idiopathic thrombocytopenic purpura, autoimmune thrombocytopenic purpura, isolated thrombocytopenia, neonatal thrombocytopenia, and Inherited thrombocytopenia.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2011 and later or the last 10 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) 36-39 by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria the GDG/ GAG recommended using 7 guidelines:
- 1. American Society of Hematology 2019 guidelines for immune thrombocytopenia. (ASH 2019)1
- The European guideline on management of major bleeding and coagulopathy following trauma: fifth edition. (European 2019)2
- Management of severe perioperative bleeding: guidelines from the European Society of Anaesthesiology. (ESA 2016)3
- Fetal and neonatal alloimmune thrombocytopenia: recommendations for evidence-based practice, an international approach. (BSH, 2019)4
- Guidelines on transfusion for fetuses, neonates and older children. (BSH 2016)5
- Guidelines for the Laboratory Investigations of heritable disorders of platelet function. (BSH 2011)6
- Updated international consensus report on the investigation and management of primary immune thrombocytopenia. (ASH ITP consensus 2019).7
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)40-43
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of pediatric hematology.
The main functions of the clinical panel were adolopment of thrombocytopenia in pediatrics Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were two guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as eminent international reviewers.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of
the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions. 36-39
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include awell-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing strategies for the diagnosis, prevention and management of thrombocytopenia in pediatric age group into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including: algorithms, pathways, tables, and parents’ educational health guide (in Arabic).
Table S1: Modified WHO Bleeding assessment score
Table S2. Causes of neonatal thrombocytopenia 45
|
|
Ill-appearing, Premature |
Well-appearing, mature |
||
|
|
Early Onset |
Late Onset |
Early Onset |
Late Onset |
|
Common |
Sepsis TORCH infection Birth asphyxia DIC NEC |
Sepsis Thrombosis DIC NEC Drug-induced |
Placental insufficiency Autoimmune Alloimmune (FNAIT) Occult infection |
Occult infection NEC |
|
Rare |
Chromosomal disorders • Trisomy 13 • Trisomy 18 • Trisomy 21 • Turner syndrome |
Inborn errors of metabolism Fanconi anemia |
Inherited syndromes • Bernard-Soulier • Wiskott-Aldrich • Thrombocytopenia absent radii • Others Vascular tumors • Kasabach-Merritt |
Inborn errors of metabolism Fanconi anemia |
Sillers L, Van Slambrouck C, Lapping-Carr G. Neonatal Thrombocytopenia: Etiology and Diagnosis. Pediatr Ann. 2015 Jul;44(7):e175-80. doi: 10.3928/00904481-20150710-11. PubMed PMID: 26171707; PubMed Central PMCID: PMCPMC6107300.
Table S3. Suggested thresholds of platelet count for neonatal platelet transfusion 46
|
Platelet count (× 109/l) |
Indication for platelet transfusion |
|
<25 |
Neonates with no bleeding (including neonates with NAIT if no bleeding and no family history of ICH) |
|
<50 |
Neonates with bleeding, current coagulopathy, before surgery, or infants with NAIT if previously affected sibling with ICH |
|
<100 |
Neonates with major bleeding or requiring major surgery (e.g., neurosurgery) |
NAIT, neonatal alloimmune thrombocytopenia; ICH, intracranial hemorrhage.
New HV, Berryman J, Bolton-Maggs PH, Cantwell C, Chalmers EA, Davies T, Gottstein R, Kelleher A, Kumar S, Morley SL, Stanworth SJ; British Committee for Standards in Haematology. Guidelines on transfusion for fetuses, neonates and older children. Br J Haematol. 2016 Dec;175(5):784-828. doi: 10.1111/bjh.14233. Epub 2016 Nov 11. PMID: 27861734.
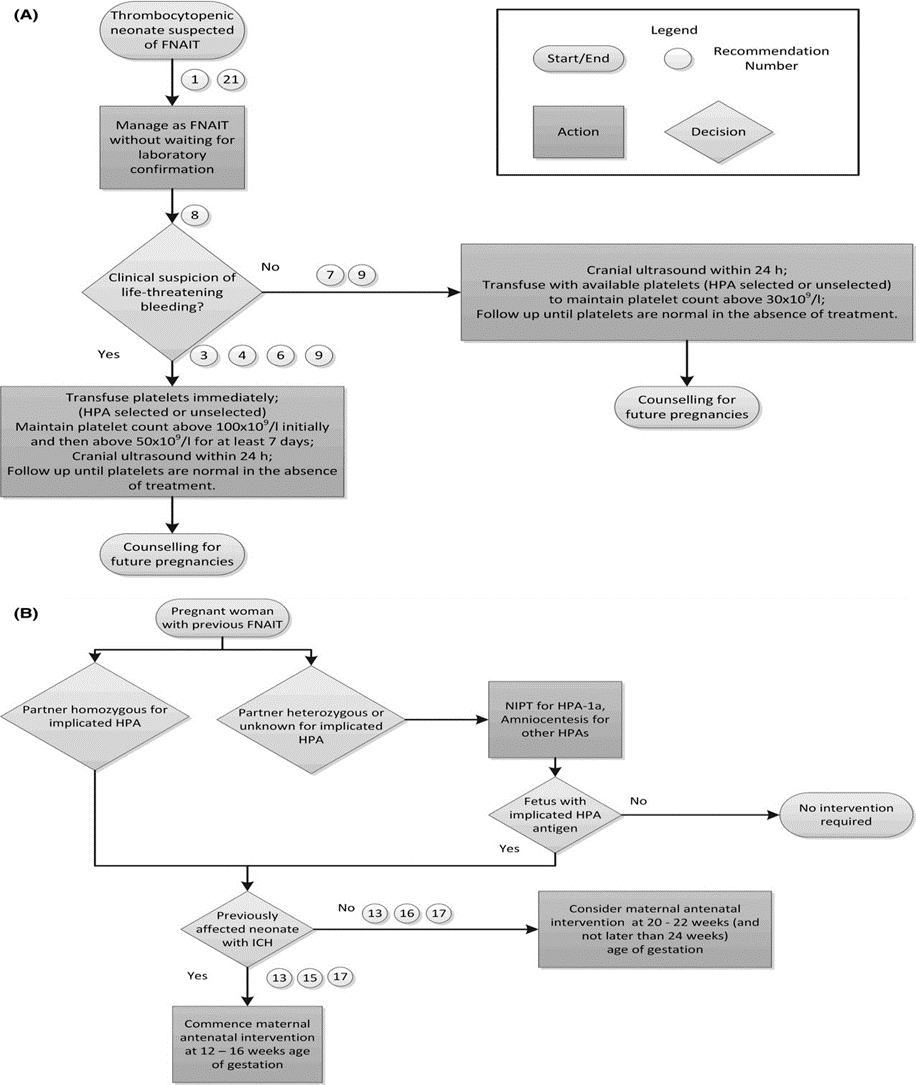
Figure S1: (A) Postnatal Algorithm for FNAIT. (B) Antenatal Algorithm for FNAIT 47
FNAIT, fetal and neonatal alloimmune thrombocytopenia; HLA, human leucocyte antigen; ICH, intracranial hemorrhage; NIPT, non-invasive prenatal testing.
Lieberman L, Greinacher A, Murphy MF, Bussel J, Bakchoul T, Corke S, Kjaer M, Kjeldsen-Kragh J, Bertrand G, Oepkes D, Baker JM, Hume H, Massey E, Kaplan C, Arnold DM, Baidya S, Ryan G, Savoia H, Landry D, Shehata N; International Collaboration for Transfusion Medicine Guidelines (ICTMG). Fetal and neonatal alloimmune thrombocytopenia: recommendations for evidence-based practice, an international approach. Br J Haematol. 2019 May;185(3):549-562. doi: 10.1111/bjh.15813. Epub 2019 Mar 3. PMID: 30828796.
Table S4. ISTH-SSC Bleeding Assessment Tool 48
|
|
SCORE |
||||
|
SYMPTOMS (up to the time of diagnosis) |
0§ |
1§ |
2 |
3 |
4 |
|
Epistaxis |
No/trivial |
- > 5/year |
Consultation only* |
Packing or cauterization or antifibrinolytic |
Blood transfusion or replacement therapy (use of hemostatic blood components and rFVIIa) or desmopressin |
|
Cutaneous |
No/trivial |
For bruises 5 or more (> 1cm) in exposed areas |
Consultation only* |
Extensive |
Spontaneous hematoma requiring blood transfusion |
|
Bleeding from minor wounds |
No/trivial |
- > 5/year |
Consultation only* |
Surgical hemostasis |
Blood transfusion, replacement therapy, or desmopressin |
|
Oral cavity |
No/trivial |
Present |
Consultation only* |
Surgical hemostasis or antifibrinolytic |
Blood transfusion, replacement therapy or desmopressin |
|
GI bleeding |
No/trivial |
Present (not associated with ulcer, portal hypertension, hemorrhoids, angiodysplasia) |
Consultation only* |
Surgical hemostasis, antifibrinolytic |
Blood transfusion, replacement therapy or desmopressin |
|
Hematuria |
No/trivial |
Present (macroscopic) |
Consultation only* |
Surgical hemostasis, iron therapy |
Blood transfusion, replacement therapy or desmopressin |
|
Tooth extraction |
No/trivial or none done |
Reported in ≤25% of all procedures, no intervention** |
Reported in ≥25% of all procedures, no intervention** |
Resuturing or packing |
Blood transfusion, replacement therapy or desmopressin |
|
Surgery |
No/trivial or none done |
Reported in ≤25% of all procedures, no intervention** |
Reported in ≥25% of all procedures, no intervention** |
Surgical hemostasis or antifibrinolytic |
Blood transfusion, replacement therapy or desmopressin |
|
Menorrhagia |
No/trivial |
Consultation only* |
- Time off work/school > 2/year |
- Requiring combined treatment with
antifibrinolytics and hormonal therapy |
- Acute menorrhagia requiring hospital admission and
emergency treatment |
|
Post-partum hemorrhage |
No/trivial or no deliveries |
Consultation only* |
- Iron therapy |
- Requiring blood transfusion, replacement therapy,
desmopressin |
- Any procedure requiring critical care or surgical intervention (e.g. hysterectomy, internal iliac artery legation, uterine artery embolization, uterine brace sutures) |
|
Muscle hematomas |
Never |
Post trauma, no therapy |
Spontaneous, no therapy |
Spontaneous or traumatic, requiring desmopressin or replacement therapy |
Spontaneous or traumatic, requiring surgical intervention or blood transfusion |
|
Hemarthrosis |
Never |
Post trauma, no therapy |
Spontaneous, no therapy |
Spontaneous or traumatic, requiring desmopressin or replacement therapy |
Spontaneous or traumatic, requiring surgical intervention or blood transfusion |
|
CNS bleeding |
Never |
|
|
Subdural, any intervention |
Intracerebral, any intervention |
|
Other bleedings^ |
No/trivial |
Present |
Consultation only* |
Surgical hemostasis, antifibrinolytics |
Blood transfusion or replacement therapy or desmopressin |
In addition to the guidance offered by the table, it is mandatory to refer to the text for more detailed instructions.
§ Distinction between 0 and 1 is of critical importance. Score 1 means that the symptom is judged as present in the patient’s history by the interviewer but does not qualify for a score 2 or more
* Consultation only: the patient sought medical evaluation and was either referred to a specialist or offered detailed laboratory investigation.
** Example: 1 extraction/surgery resulting in bleeding (100%): the score to be assigned is 2; 2 extractions/surgeries, 1 resulting in bleeding (50%): the score to be assigned is 2; 3 extractions/surgeries, 1 resulting in bleeding (33%): the score to be assigned is 2; 4 extractions/surgeries, 1 resulting in bleeding (25%): the score to be assigned is 1.
# If already available at the time of collection.
^ Include: umbilical stump bleeding, cephalohematoma, cheek hematoma caused by sucking during breast/bottle feeding, conjunctival hemorrhage or excessive bleeding following circumcision or venipuncture. Their presence in infancy requires detailed investigation independently from the overall score
Normal range is <4 in adult males, <6 in adult females and <3 in children (3).
Rodeghiero F, Tosetto A, Abshire T, Arnold DM, Coller B, James P, Neunert C, Lillicrap D; ISTH/SSC joint VWF and Perinatal/Pediatric Hemostasis Subcommittees Working Group. ISTH/SSC bleeding assessment tool: a standardized questionnaire and a proposal for a new bleeding score for inherited bleeding disorders. J Thromb Haemost. 2010 Sep;8(9):2063-5. doi: 10.1111/j.1538-7836.2010.03975.x. PMID: 20626619.
Table S5. Differential diagnosis of thrombocytopenia 49
|
Previously diagnosed or possible high risk for conditions that may be associated with immune thrombocytopenia (e.g., infections [HIV, HCV, HBV]), autoimmune/immunodeficiency disorders (CVID, systemic lupus erythematosus, or APS), and malignancy (eg, lymphoproliferative disorders) |
|
Liver disease (including cirrhosis or portal hypertension) |
|
Splenomegaly |
|
Drugs (prescription or nonprescription), including heparin, alemtuzumab, PD-1 inhibitors, abciximab, valproate, alcohol abuse, consumption of quinine (tonic water), exposure to environmental toxins, or chemotherapy |
|
Bone marrow diseases, including myelodysplastic syndromes, leukemias, other malignancies, metastatic disease, myelofibrosis, aplastic anemia, megaloblastic anemia, myelophthisis, and Gaucher disease |
|
Recent transfusions (rare possibility of posttransfusion purpura) and recent vaccinations |
|
Inherited thrombocytopenia: TAR syndrome, radioulnar synostosis, congenital amegakaryocytic thrombocytopenia, Wiskott-Aldrich syndrome, MYH9-related disease, Bernard-Soulier syndrome, type IIB VWD, or platelet-type VWD |
|
Other thrombocytopenic disorders (DIC, TTP, HUS, Evans syndrome) |
APS, antiphospholipid syndrome; CVID, common variable immunodeficiency; DIC, disseminated intravascular coagulation; HUS, hemolytic-uremic syndrome; MYH9, myosin heavy chain 9; PD-1, programmed cell death protein 1; TAR, thrombocytopenia-absent radius; TTP, thrombotic thrombocytopenic purpura; VWD, von Willebrand disease.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896.
Table S6 Bleeding assessment tool in pediatric patient with primary immune thrombocytopenia 49
|
Grade |
Bleeding |
Management approach |
|
Grade 1 (minor) |
Minor bleeding, few petechiae (≤100 total) and/or ≤5 small bruises (≤3 cm in diameter), no mucosal bleeding |
Consent for observation |
|
Grade 2 (mild) |
Mild bleeding, many petechiae (>100 total) and/or >5 large bruises (>3 cm in diameter), no mucosal bleeding |
Consent for observation |
|
Grade 3 (moderate) |
Moderate bleeding, overt mucosal bleeding, troublesome lifestyle |
Intervention to reach grade 1 or 2 |
|
Grade 4 (severe) |
Severe bleeding, mucosal bleeding leading to decrease in Hb > 2 g/dL or suspected internal hemorrhage |
Intervention |
This bleeding scale is based on the one used in the previous consensus report,1 updated based on the authors’ opinion. Hb, hemoglobin.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896
Table S7 Recommendations for the diagnosis of ITP in children and adults 49
|
Basic evaluation in all patients |
Tests of potential utility in the management of an ITP patient |
Tests of unproven or uncertain benefit* |
|
Patient history |
Glycoprotein-specific antibody (can be used in difficult cases, has poor sensitivity, and is not a primary diagnostic test) |
TPO level |
|
Family history |
Anti-phospholipid antibodies (including anti-cardiolipin and lupus anticoagulant) if there are clinical features of antiphospholipid syndrome |
Reticulated platelets/immature platelet fraction |
|
Physical examination |
Anti-thyroid antibodies and thyroid function |
|
|
CBC and reticulocyte count |
Pregnancy test in women of childbearing potential |
Bleeding time |
|
Peripheral blood film |
Antinuclear antibodies |
Serum complement |
|
Quantitative Ig level measurement† |
Viral PCR for EBV, CMV, and parvovirus |
|
|
Blood group (Rh) |
Bone marrow examination (in selected patients; refer to text) |
|
|
HIV‡ |
Direct antiglobulin test |
|
|
HCV‡ |
H pylori‡ |
|
|
HBV |
|
|
CMV, cytomegalovirus; EBV, Epstein-Barr virus; PCR, polymerase chain reaction; PTT, partial thromboplastin time; Rh, rhesus; TPO, thrombopoietin.
*These tests have no proven role in the differential diagnosis of ITP from other thrombocytopenias and do not guide patient management.
†Quantitative Ig level measurement should be considered in children with ITP and is recommended in children with persistent or chronic ITP as part of the reassessment evaluation.
‡Recommended by the majority of the panel for adult patients in the appropriate geographic setting.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896.
Table S8: Therapies for the treatment of ITP 49
|
Clinical situation |
Therapy option |
|
Initial treatment of newly diagnosed ITP |
Corticosteroids |
|
Dexamethasone |
|
|
Methylprednisolone |
|
|
Prednis(ol)one |
|
|
IVIg |
|
|
Anti-D (licensed and available for ITP in only a few countries) |
|
|
Subsequent treatment |
Medical therapies |
|
Medical therapies with robust evidence |
|
|
Rituximab |
|
|
TPO-RAs: eltrombopag, avatrombopag, romiplostim |
|
|
Fostamatinib |
|
|
Medical therapies with less robust evidence |
|
|
Azathioprine |
|
|
Cyclophosphamide |
|
|
Cyclosporine A |
|
|
Danazol |
|
|
Dapsone |
|
|
Mycophenolate mofetil |
|
|
TPO-RA switch |
|
|
Vinca alkaloids |
|
|
Surgical therapies |
|
|
Splenectomy |
|
|
Treatment of patients failing multiple therapies |
Accessory splenectomy |
|
Alemtuzumab |
|
|
Combination of initial and subsequent therapies |
|
|
Combination chemotherapy |
|
|
Clinical trials |
|
|
HSCT |
|
|
Splenectomy, if not already performed |
|
|
Supportive care |
Treatment options for ITP are listed in alphabetical order and do not imply a preferred treatment option.
HSCT, hematopoietic stem cell transplantation; TPO-RA, TPO receptor agonist.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896
Table S9. Suggested thresholds of platelet count for platelet transfusion in children with non-immune thrombocytopenia 50
|
Platelet count (× 109/l) |
Clinical situation to trigger platelet transfusion |
|
<10 |
Irrespective of signs of hemorrhage (excluding ITP, TTP/HUS, HIT) |
|
<20 |
Severe mucositis Sepsis Laboratory evidence of DIC in the absence of bleedinga Anticoagulant therapy Risk of bleeding due to a local tumor infiltration Insertion of a non-tunnelled central venous line |
|
<40 |
Prior to lumbar punctureb |
|
<50 |
Moderate hemorrhage (e.g., gastrointestinal bleeding) including bleeding in association with DIC Surgery, unless minor (except at critical sites)
|
|
<75–100 |
Major hemorrhage or significant post-operative bleeding (e.g., post cardiac surgery) Surgery at critical sites: central nervous system including eyes |
· ALL, acute lymphoblastic leukemia; DIC, disseminated intravascular coagulation; HIT, heparin-induced thrombocytopenia; HUS, hemolytic uremic syndrome; ITP, immune thrombocytopenia; LP, lumbar puncture; TTP, thrombotic thrombocytopenic purpura.
· a Note: routine screening by standard coagulation tests not advocated without clinical indication; for laboratory evidence of DIC see Section 5.6.4.
· b It is accepted that prior to lumbar puncture some clinicians will transfuse platelets at higher counts (e.g., 50 × 109/l) in clinically unstable children, non-ALL patients, or for the first LP in newly-diagnosed ALL patients to avoid hemorrhage and cerebrospinal fluid contamination with blasts, or at lower counts (≤20 × 109/l) in stable patients with ALL, depending on the clinical situation. These practices emphasize the importance of considering the clinical setting and patient factors.
New HV, Berryman J,
Bolton-Maggs PH, Cantwell C, Chalmers EA, Davies T, Gottstein R, Kelleher A,
Kumar S, Morley SL, Stanworth SJ; British Committee for Standards in
Haematology. Guidelines on transfusion for fetuses, neonates and older
children. Br J Haematol. 2016 Dec;175(5):784-828. doi: 10.1111/bjh.14233. Epub
2016 Nov 11. PMID: 27861734.


- Limitations and suggestions for further research needs
Future research recommendations for the diagnosis, prevention and management of thrombocytopenia in pediatric age groups in the Egyptian context could include:
▪️ Implementation of hemovigilance system at different institutional levels.
▪️ Screening programs for specific populations.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention, diagnosis and management strategies for Thrombocytopenia and Bleeding in children.
Challenges
▪️ Implementation of international guidelines in resource limited settings.
▪️ The optimum use of the available resources to fulfill the requirements of Egyptian population.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are two performance measures or indicators for implementing this adapted CPG for Management of isolated thrombocytopenia in pediatrics:
Adherence to Management of isolated thrombocytopenia Guidelines
▪️ Numerator: Number of children with isolated thrombocytopenia who received management as per guideline recommendations.
▪️Denominator: Total number of children who had isolated thrombocytopenia.
▪️ Data Source: Hospital or clinic patient records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines.
- Updating of the guideline
The EPG Haematology GAG
has decided to conduct the next review of this adapted CPG for updates after five
years. This should be carried out in 2027 after checking for updates in the
source CPGs, consultation of expert opinion on the changes needed for updating
according to the newest evidence and recommendations published in this area and
the clinical audit and feedback from implementation efforts in the aforementioned
local healthcare settings except if any breakthrough evidence- based
recommendations are published before that date. The process will be guided by
the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that
is freely provided by the AGREE Enterprise and by the Reporting Items for
Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines
RIGHT-Ad@pt Checklist.
- References
1. Neunert
C, Terrell DR, Arnold DM, et al. American Society of Hematology 2019 guidelines
for immune thrombocytopenia. Blood Adv. 2019 Dec 10;3(23):3829-3866. doi:
10.1182/bloodadvances.2019000966. PubMed PMID: 31794604; PubMed Central PMCID:
PMCPMC6963252.
2. Spahn DR,
Bouillon B, Cerny V, et al. The European guideline on management of major
bleeding and coagulopathy following trauma: fifth edition. Crit Care. 2019 Mar
27;23(1):98. doi: 10.1186/s13054-019-2347-3. PubMed PMID: 30917843; PubMed
Central PMCID: PMCPMC6436241.
3. Kozek-Langenecker
SA, Ahmed AB, Afshari A, et al. Management of severe perioperative bleeding:
guidelines from the European Society of Anaesthesiology: First update 2016. Eur
J Anaesthesiol. 2017 Jun;34(6):332-395. doi: 10.1097/EJA.0000000000000630.
PubMed PMID: 28459785.
4. Lieberman L,
Greinacher A, Murphy MF, et al. Fetal and neonatal alloimmune thrombocytopenia:
recommendations for evidence-based practice, an international approach. Br J
Haematol. 2019 May;185(3):549-562. doi: 10.1111/bjh.15813. PubMed PMID:
30828796.
5. New HV,
Berryman J, Bolton-Maggs PH, et al. Guidelines on transfusion for fetuses,
neonates and older children. Br J Haematol. 2016 Dec;175(5):784-828. doi:
10.1111/bjh.14233. PubMed PMID: 27861734.
6. Harrison P,
Mackie I, Mumford A, et al. Guidelines for the laboratory investigation of
heritable disorders of platelet function. Br J Haematol. 2011 Oct;155(1):30-44.
doi: 10.1111/j.1365-2141.2011.08793.x. PubMed PMID: 21790527.
7. Provan D,
Arnold DM, Bussel JB, et al. Updated international consensus report on the
investigation and management of primary immune thrombocytopenia. Blood Adv.
2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PubMed
PMID: 31770441; PubMed Central PMCID: PMCPMC6880896.
8. Johnson B,
Fletcher SJ, Morgan NV. Inherited thrombocytopenia: novel insights into
megakaryocyte maturation, proplatelet formation and platelet lifespan.
Platelets. 2016 Sep;27(6):519-25. doi: 10.3109/09537104.2016.1148806. PubMed
PMID: 27025194; PubMed Central PMCID: PMCPMC5000870.
9. Stasi R,
Amadori S, Osborn J, et al. Long-term outcome of otherwise healthy individuals
with incidentally discovered borderline thrombocytopenia. PLoS Med. 2006
Mar;3(3):e24. doi: 10.1371/journal.pmed.0030024. PubMed PMID: 16401142; PubMed
Central PMCID: PMCPMC1326262.
10. Rodeghiero F,
Stasi R, Gernsheimer T, et al. Standardization of terminology, definitions and
outcome criteria in immune thrombocytopenic purpura of adults and children:
report from an international working group. Blood. 2009 Mar 12;113(11):2386-93.
doi: 10.1182/blood-2008-07-162503. PubMed PMID: 19005182.
11. Stasi R. How to
approach thrombocytopenia. Hematology Am Soc Hematol Educ Program.
2012;2012:191-7. doi: 10.1182/asheducation-2012.1.191. PubMed PMID: 23233580.
12. Drachman JG.
Inherited thrombocytopenia: when a low platelet count does not mean ITP. Blood.
2004 Jan 15;103(2):390-8. doi: 10.1182/blood-2003-05-1742. PubMed PMID:
14504084.
13. Roberts I,
Stanworth S, Murray NA. Thrombocytopenia in the neonate. Blood Rev. 2008
Jul;22(4):173-86. doi: 10.1016/j.blre.2008.03.004. PubMed PMID: 18433954.
14. Sola-Visner M,
Saxonhouse MA, Brown RE. Neonatal thrombocytopenia: what we do and don't know.
Early Hum Dev. 2008 Aug;84(8):499-506. doi: 10.1016/j.earlhumdev.2008.06.004.
PubMed PMID: 18684573.
15. Ferrer-Marin F,
Chavda C, Lampa M, et al. Effects of in vitro adult platelet transfusions on
neonatal hemostasis. Journal of thrombosis and haemostasis : JTH.
2011;9(5):1020-1028. doi: 10.1111/j.1538-7836.2011.04233.x. PubMed PMID:
21320282; eng.
16. Mehta P, Vasa
R, Neumann L, et al. Thrombocytopenia in the high-risk infant. J Pediatr. 1980
Nov;97(5):791-4. doi: 10.1016/s0022-3476(80)80272-1. PubMed PMID: 7431175.
17. Chakravorty S,
Roberts I. How I manage neonatal thrombocytopenia. Br J Haematol. 2012
Jan;156(2):155-62. doi: 10.1111/j.1365-2141.2011.08892.x. PubMed PMID:
21950766; eng.
18. Terrell DR,
Beebe LA, Vesely SK, et al. The incidence of immune thrombocytopenic purpura in
children and adults: A critical review of published reports. Am J Hematol. 2010
Mar;85(3):174-80. doi: 10.1002/ajh.21616. PubMed PMID: 20131303.
19. Neunert C, Lim
W, Crowther M, et al. The American Society of Hematology 2011 evidence-based
practice guideline for immune thrombocytopenia. Blood. 2011 Apr
21;117(16):4190-207. doi: 10.1182/blood-2010-08-302984. PubMed PMID: 21325604.
20. Provan D, Stasi
R, Newland AC, et al. International consensus report on the investigation and
management of primary immune thrombocytopenia. Blood. 2010 Jan 14;115(2):168-86.
doi: 10.1182/blood-2009-06-225565. PubMed PMID: 19846889.
21. Elmas Dal S,
Ersoy Y, Ali Erkurt M, et al. An uncommon case of acute brucellosis presenting
with severe thrombocytopenia. Intern Med. 2012;51(23):3291-3. doi:
10.2169/internalmedicine.51.7365. PubMed PMID: 23207127.
22. Kumar S, Nair
S, Rajam L. Case series of pediatric systemic lupus erythematosus from Kerala:
comparison with other Indian series. Int J Rheum Dis. 2010 Oct;13(4):391-5.
doi: 10.1111/j.1756-185X.2010.01536.x. PubMed PMID: 21199476.
23. Hatta K,
Kunishima S, Suganuma H, et al. A family having type 2B von Willebrand disease
with a novel VWF p.R1308S mutation: Detection of characteristic platelet
aggregates on peripheral blood smears as the key aspect of diagnosis. Thrombosis
research. 2015 Oct;136(4):813-7. doi: 10.1016/j.thromres.2015.08.004. PubMed
PMID: 26278967; eng.
24. Lee AC, Li CH,
Wong LM. Childhood thrombocytopenia associated with Graves disease is distinct
from idiopathic thrombocytopenic purpura. Pediatr Hematol Oncol. 2003
Jan-Feb;20(1):39-42. PubMed PMID: 12687752.
25. Olaniyi JA. An
Acute Leukaemia Masquerading as Immune Thrombocytopaenic Purpura (ITP)? A Case
Report. Clin Med Case Rep. 2009;2:31-34. doi: 10.4137/ccrep.s2394. PubMed PMID:
24179370; eng.
26. British
Committee for Standards in Haematology General Haematology Task F. Guidelines
for the investigation and management of idiopathic thrombocytopenic purpura in
adults, children and in pregnancy. Br J Haematol. 2003 Feb;120(4):574-96. doi:
10.1046/j.1365-2141.2003.04131.x. PubMed PMID: 12588344.
27. Khalifa AS,
Tolba KA, el-Alfy MS, et al. Idiopathic thrombocytopenic purpura in Egyptian
children. Acta Haematol. 1993;90(3):125-9. doi: 10.1159/000204392. PubMed PMID:
8291370.
28. ElAlfy M, Farid
S, Abdel Maksoud A. Predictors of chronic idiopathic thrombocytopenic purpura.
Pediatr Blood Cancer. 2010 Jul 1;54(7):959-62. doi: 10.1002/pbc.22481. PubMed
PMID: 20405514.
29. Bryant N, Watts
R. Thrombocytopenic syndromes masquerading as childhood immune thrombocytopenic
purpura. Clin Pediatr (Phila). 2011 Mar;50(3):225-30. doi:
10.1177/0009922810385676. PubMed PMID: 21098529.
30. Aboud N, Depré
F, Salama A. Is Autoimmune Thrombocytopenia Itself the Primary Disease in the
Presence of Second Diseases Data from a Long-Term Observation. Transfusion
Medicine and Hemotherapy. 2017;44(1):23-28. doi: 10.1159/000449038.
31. Rotz SJ, Ware
RE, Kumar A. Diagnosis and management of chronic and refractory immune
cytopenias in children, adolescents, and young adults. Pediatr Blood Cancer.
2018 Oct;65(10):e27260. doi: 10.1002/pbc.27260. PubMed PMID: 29856527.
32. Pons I,
Monteagudo M, Lucchetti G, et al. Correlation between immature platelet
fraction and reticulated platelets. Usefulness in the etiology diagnosis of
thrombocytopenia. European journal of haematology. 2010 Aug;85(2):158-63. doi:
10.1111/j.1600-0609.2010.01468.x. PubMed PMID: 20477866; eng.
33. Monteagudo M,
Amengual MJ, Munoz L, et al. Reticulated platelets as a screening test to
identify thrombocytopenia aetiology. QJM. 2008 Jul;101(7):549-55. doi:
10.1093/qjmed/hcn047. PubMed PMID: 18400777.
34. Grimaldi-Bensouda
L, Nordon C, Leblanc T, et al. Childhood immune thrombocytopenia: A nationwide
cohort study on condition management and outcomes. Pediatr Blood Cancer. 2017
Jul;64(7). doi: 10.1002/pbc.26389. PubMed PMID: 27905681.
35. Heitink-Polle
KMJ, Uiterwaal C, Porcelijn L, et al. Intravenous immunoglobulin vs observation
in childhood immune thrombocytopenia: a randomized controlled trial. Blood.
2018 Aug 30;132(9):883-891. doi: 10.1182/blood-2018-02-830844. PubMed PMID:
29945954.
36. Venkatesh V,
Curley A, Khan R, et al. A novel approach to standardised recording of bleeding
in a high risk neonatal population. 2013;98(3):F260-F263. doi:
10.1136/archdischild-2012-302443 %J Archives of Disease in Childhood - Fetal
and Neonatal Edition.
37. Sillers L, Van
Slambrouck C, Lapping-Carr G. Neonatal Thrombocytopenia: Etiology and
Diagnosis. Pediatr Ann. 2015 Jul;44(7):e175-80. doi: 10.3928/00904481-20150710-11.
PubMed PMID: 26171707; PubMed Central PMCID: PMCPMC6107300.
38. Rodeghiero F,
Tosetto A, Abshire T, et al. ISTH/SSC bleeding assessment tool: a standardized
questionnaire and a proposal for a new bleeding score for inherited bleeding
disorders. J Thromb Haemost. 2010 Sep;8(9):2063-5. doi:
10.1111/j.1538-7836.2010.03975.x. PubMed PMID: 20626619.
References
of materials used in creating this EBCPG
1- ADAPTE
Resource Toolkit versions 2.0 (2009) Available from:
www.g-i-n.net/document-store/adapte-resource-toolkit-guideline-adaptation-version-2
(Version 2.0 downloaded free without registration).
2- Amer YS,
Elzalabany MM, Omar TI, Ibrahim AG and Dowidar NL. The ‘Adapted ADAPTE’: an
approach to improve utilization of the ADAPTE guideline adaptation resource
toolkit in the Alexandria Center for Evidence-Based Clinical Practice
Guidelines. Journal of Evaluation in Clinical Practice 2015; 21: 1095 – 106.
3- AGREE (II)
Instrument (if used) available from thewww.agreecollaboration.org/instrument/
(downloaded free).
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Galila Mohamed Mokhtar |
Paediatrics Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Amira Adly |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Dina Ezzat |
Professor of Paediatrics and Paediatric Haematology, Beni-Suef University, Egypt |
None |
Not Applicable |
|
Prof. Gehan Lotfy |
Professor of Paediatrics, Minia University, Egypt |
None |
Not Applicable |
|
Prof. Hoda Hassab |
Professor of Paediatrics and Paediatrics Haematology, Alexandria University, Egypt |
None |
Not Applicable |
|
Prof. Ilham Youssry |
Professor of Paediatrics and Head of the Paediatric Haematology & BMT unit, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Iman Ragab |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Laila Sherief |
Professor of Paediatrics and Paediatrics Haematology & Oncology, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Marwa Zakaria |
Associate Professor of Paediatrics, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Mervat Hesham |
Professor of Paediatrics, Paediatric Haematology & Oncology Unit, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Niveen Salama |
Associate Professor of Paediatrics, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Nouran Yousef |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Rasha Abdel-Raouf Afifi |
Professor of Paediatrics and Paediatric Haematology, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Rasha El-Ashry |
Professor of Paediatrics, Mansoura University, Egypt |
None |
Not Applicable |
|
Dr. Sara Makkeyah |
Associate Professor of Paediatrics, Paediatric Haematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Sonia Adolf |
Professor of Paediatrics and Paediatric Haematology, National Research Center, Cairo, Egypt |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
External Review Group |
|||
|
Prof. Eman Abdel-Raouf |
Professor of Pediatrics and Pediatric Hematology, Cairo University, Egypt. |
None |
Not Applicable |
|
Prof. Mervat Atfy |
Professor of Pediatrics and Pediatric Hematology, Zagazig University, Egypt. |
None |
Not Applicable |
|
Prof. Mohamed Abdel-Mohsen |
Professor of Pediatrics and Pediatric Hematology/Oncology, Ain Shams University, Egypt. |
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
None |
Not Applicable |
|
International Peer Reviewers |
|
|
|
|
Prof. James Bussel |
Professor pf Pediatric, Hematology/Oncology, Pediatrics Department, Well Cornell Medical College, Newyork, USA |
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the management of pediatric patients with isolated thrombocytopenia" on PubMed are: childhood thrombocytopenia, pediatric thrombocytopenia, immune thrombocytopenic purpura, idiopathic thrombocytopenic purpura, autoimmune thrombocytopenic purpura and isolated thrombocytopenia, neonatal thrombocytopenia, Inherited thrombocytopenia.
Annex Table 2. Results of the AGREE II assessment of the seven source guidelines for bleeding and thrombocytopenia in pediatric age groups
|
CPGs AGREE II DOMAINS |
CPG1 |
CPG2 |
CPG3 |
CPG4 |
CPG5 |
CPG6 |
CPG7 |
|
D1: Scope & Purpose |
95.7 |
92.9 |
81.4 |
85.7 |
85.7 |
75.7 |
94.3 |
|
D2: Stakeholder Involvement |
100 |
82.9 |
81.4 |
75.7 |
81.4 |
75.7 |
92.9 |
|
D3: Rigour of Development |
100 |
90 |
85.7 |
87.1 |
87.1 |
94.3 |
97.1 |
|
D4: Clarity & Presentation |
95.7 |
85.7 |
100 |
85.7 |
85.7 |
100 |
100 |
|
D5: Applicability |
89.3 |
75 |
85.7 |
66.7 |
66.7 |
89.3 |
67.9 |
|
D6: Editorial Independence |
92.9 |
61.4 |
85.7 |
78.6 |
78.6 |
92.9 |
85.7 |
|
OA 1 |
95.7 |
73.6 |
74.3 |
85.5 |
87.7 |
87.7 |
94 |
|
OA 2 (Recommend for use) |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
This table uses the AGREE II Domain Score Color codes (< 40% red; > 41 – 70% yellow; > 71 % green) |
|||||||
Appendix Table 3. The RIGHT-Ad@pt checklist
|
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Practice guideline REgistration for transPAREncy (PREPARE) Guideline Protocol Registration Receipt
