Transfusion of Blood Components in Paediatric Age Groups
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Transfusion of Blood Components in Paediatric Age Groups |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 17 Feb 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Introduction
- - Methods
- - Evidence to recommendations: Considerations
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline.
- - References
- - Annexes
- Executive Summary
➡️Introduction
Pediatric transfusion is a complex area of medicine covering a wide age range from intrauterine life to young adults. The prescriber must balance the risks and benefits of transfusion in each age group and be aware of the indications for special components.
Compared to adult practice, there is a relative lack of high-quality research to inform evidence-based guidelines.
The aim of this adapted clinical practice guideline (CPG) is to provide evidence-based recommendations for the transfusion of blood components in the paediatric age group.
➡️Scope
This guideline focuses on any disease or condition requiring red blood cells, platelets, and/or plasma products transfusion(s) to prevent or control anaemia, bleeding, and thrombotic disorders. It Provides an evidence-based document for the appropriate use of blood components, define indications, dosing, and administration of different blood components.
It also covers safe transfusion practice including pre-transfusion testing, handling of blood components, and close monitoring of transfused patients and includes recommendations in special situations (multi-transfused subjects, massive transfusion, and the critically ill).
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1. Guideline on the investigation and management of acute transfusion reactions Prepared by the BCSH Blood Transfusion Task Force. BSH 20121
2. Guidelines on red cell transfusion in sickle cell disease. BSH 2016a2,3
3. Guidelines for the use of platelet transfusions. BSH 2016b4
4. Recommendations on Red Blood Cell Transfusion in General Critically Ill Children Based on Heamoglobin and/or Physiologic Thresholds from the Paediatric Critical Care Transfusion and Anaemia Expertise Initiative. TAXI 20185
5. Clinical practice guidelines use of blood components in newborn. NNF 2020.6
6. Guidelines on the use of irradiated blood components. BSH 2020a7
7. Guidelines on Transfusion for Fetuses, Neonates and Older Children. BSH 2016 - addendum 2020b8,9
8. British Society of Haematology Guidelines on the spectrum of fresh frozen plasma and cryoprecipitate products: their handling and use in various patient groups in the absence of major bleeding. BSH 2018 – addendum 202010,11
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. Transfusion guidelines in the different age groups:
The guideline covers transfusion guidelines for the different blood products in neonates as well as in infants and children.
This guideline emphasis on indications, dosing, and mode of transfusion.
B. Recommendation on blood product irradiation:
This section includes recommendations and good practice statements on when and how to irradiate blood products according to different clinical situations.
C. Identification and management of transfusion reactions:
This section handles the diagnosis and lines of management in case of blood product reactions.
We can summarize the guidelines’ recommendations for Transfusion of Blood Components in Pediatric Age Groups in the following:
Transfusion in neonates:
▪️ Decision of transfusion is based on the clinical status. For those not requiring cardiopulmonary support or oxygen supply and condition is stable, packed RBCs transfusion is not usually required unless Hb level is below 7g/dl (GPS).
▪️ For non-bleeding neonates, platelet transfusions should not be routinely administered if platelet count is ≥ 25 × 109/l (High LOE, Strong recommendation).
▪️ In neonatal alloimmune thrombocytopenia (FNAIT), maintaining platelet count > 30 x109/l is strongly recommended (High LOE, Weak recommendation).
▪️ FFP may be of benefit in neonates with clinically significant bleeding or prior to invasive procedures with a risk of significant bleeding, and who have an abnormal coagulation profile (Intermediate LOE, weak recommendation).
▪️ FFP should be used for the management of severe hereditary protein S deficiency (Intermediate LOE, Strong recommendation).
▪️FFP transfusion is preferred over cryoprecipitate in the management of disseminated intravascular coagulation (Intermediate LOE, weak recommendation).
▪️ Prophylactic FFP is not recommended in non-bleeding neonates receiving therapeutic hypothermia and having deranged coagulation parameters (High LOE, Strong recommendation).
Transfusion in infants, children, and adolescents
▪️ In children with oncologic diagnoses, those who are critically ill or at risk for critical illness, and hemodynamically stable, a Hb concentration of 7– 8 g/dl is suggested as a threshold for RBC transfusion (Intermediate LOE, weak recommendation).
▪️ A perioperative Hb transfusion threshold of 7 g/dl should be used in stable patients without major co-morbidity or bleeding (High LOE, weak recommendation).
▪️ Patients with chronic anaemia due to red cell aplasia may require an Hb threshold of 8 g/dl (Intermediate LOE, weak recommendation).
▪️ Transfusion is recommended and may be lifesaving in acute sickle complications such as splenic sequestration, hepatic sequestration, aplastic crisis and severe acute chest syndrome (High LOE, strong recommendation). Transfusion is not recommended in uncomplicated painful crises (High LOE, weak recommendation).
▪️ In severe bleeding, maintain the platelet count above 50 ×109 /l (High LOE, weak recommendation).
▪️ In patients with multiple traumas, traumatic brain injury or spontaneous intracerebral hemorrhage, maintain the platelet count above 100 ×109 /l (Intermediate LOE, weak recommendation).
▪️ In patients with bleeding that is not considered severe or life-threatening, consider platelet transfusion if the platelet count is below 30 ×109 /l (Intermediate LOE, weak recommendation).
▪️ In post-transfusion purpura (PTP), intravenous immunoglobulin is the treatment of choice (High LOE, weak recommendation).
▪️ Only use platelet transfusion prior to a procedure or surgery when other treatment has failed and/or the intervention is urgent. Usual threshold counts may be unachievable or unnecessary and individual case review is required (High LOE, weak recommendation),
▪️ In Glanzmann thrombasthenia, consider human leucocyte antigen (HLA)-matched platelets (Intermediate LOE, weak recommendation) where available (GPS) (Intermediate LOE, weak recommendation).
▪️ Use general haemostatic measures to treat bleeding in patients during treatment with aspirin, P2Y12 antagonists or glycoprotein IIa/IIIb inhibitors. If necessary, consider drug cessation and reversal of the effect of co-prescribed anticoagulants (Intermediate LOE, weak recommendation).
▪️ Use TXA to counteract the effect of anti-platelet agents when a risk/benefit assessment would support this (High LOE, strong recommendation).
▪️ Do not use prophylactic platelet transfusions in patients with autoimmune thrombocytopenia (High LOE, weak recommendation).
▪️ Use a ‘no prophylactic platelet transfusion’ strategy for asymptomatic patients with chronic bone marrow failure (Intermediate LOE, Strong recommendation).
▪️ Do not give platelet transfusions routinely prior to minor procedures e.g. bone marrow aspirate or trephine biopsy, peripherally inserted central catheters (PICCs), traction removal of tunneled CVCs, cataract surgery (High LOE, weak recommendation).
▪️ Pathogen-reduced plasma may be used for factor replacement in congenital coagulation factor deficiency If virally inactivated specific clotting factors are not available (High LOE, weak recommendation).
▪️ FFP may be beneficial in children with DIC who have a significant coagulopathy (PT/APTT >1.5 times midpoint of normal range or fibrinogen <1.0 g/l) associated with clinically significant bleeding or prior to invasive procedures (Intermediate LOE, weak recommendation).
▪️Prophylactic FFP should not be administered to non-bleeding children with minor prolongation of the prothrombin time (Intermediate LOE, Strong recommendation)/ activated partial thromboplastin time or decreased fibrinogen including prior to surgery, although it may be considered for surgery to critical sites (Intermediate LOE, weak recommendation).
▪️ FFP should not be used in the management of inherited factor deficiencies other than in a few exceptional circumstances where specific factor concentrates are not available (High LOE, weak recommendation).
▪️Urgent plasma exchange with solvent detergent fresh frozen plasma (SD FFP) is indicated for thrombotic thrombocytopenic purpura (TTP) (High LOE, strong recommendation) and some forms of atypical hemolytic uremic sundrome (HUS) (Intermediate LOE, weak recommendation).
➡️Modification of blood components and related Precautions
▪️ Gamma- or X-irradiation of blood components, is the recommended procedure to prevent TA-GvHD (High LOE, strong recommendation).
▪️ Red cells for neonatal exchange blood transfusion (EBT) should be irradiated (High LOE, weak recommendation).
▪️ Routine irradiation of red cells for transfusion to preterm or term infants (other than for EBT) is not required unless there has been a previous intrauterine transfusion (IUT) (Intermediate LOE, weak recommendation).
▪️ All transfusions of cellular components and fresh plasma from first- or second-degree relatives should be irradiated, even if the patient is immunocompetent. All HLA-selected components should be irradiated even if the patient is immunocompetent (High LOE, strong recommendation).
▪️ All severe congenital T-lymphocyte immunodeficiency syndromes with significant qualitative or quantitative T-lymphocyte deficiency should be considered as indications for irradiation of cellular blood components (High LOE, strong recommendation).
▪️ There is no indication for irradiation of cellular blood components for infants or children with temporary defects of T-lymphocyte function as the result of a viral infection. There is also no indication for irradiation of cellular blood components for adults or children who are HIV-antibody positive or who have acquired immune deficiency syndrome (AIDS) (High LOE, strong recommendation).
▪️ All recipients of allogeneic HSCT should receive irradiated blood components from the time of initiation of conditioning chemo/ radiotherapy (High LOE, strong recommendation).
▪️ All patients undergoing autologous stem cell transplant (ASCT) irrespective of underlying diagnosis or indication for this treatment should receive irradiated cellular blood components from initiation of conditioning chemo/radiotherapy until 3 months post-transplant (High LOE, weak recommendation).
▪️For patients with aplastic anaemia, transfusion of irradiated cellular components is not routinely recommended (High LOE, strong recommendation).
▪️ All adults and children with Hodgkin Lymphoma at any stage of the disease should have irradiated red cells and platelets indefinitely (Intermediate LOE, weak recommendation).
▪️ All patients treated with purine analogue drugs (fludarabine, cladribine, bendamustine and pentostatin) , should receive irradiated blood components indefinitely (Intermediate LOE, weak recommendation).
▪️ Treatment of patients with rituximab is not an indication for use of irradiated cellular blood components (High LOE, strong recommendation).
▪️ Use of irradiated cellular blood components is not indicated for patients undergoing solid organ transplantation (SOT) (High LOE, strong recommendation).
▪️ For patients with recurrent febrile reactions, it is recommended to use a trial of premedication with oral paracetamol given one hour before the reaction is anticipated. Patients who continue to react should have a trial of washed blood components (Intermediate LOE, weak recommendation).
▪️ Provision of CMV safe blood for transfusion in preterm neonates by using CMV seronegative donors or leukoreduction or a combination of both is strongly recommended (High LOE, weak recommendation).
▪️ For intrauterine transfusions use of CMV negative and leuco-depleted packed red blood cell is strongly recommended (High LOE, weak recommendation).
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: (PREPARE-2022CN444). Link: http://www.guidelines-registry.org/- Recommendations
Part I: Transfusion in neonates
|
CPGL Source |
Recommendation |
SoR |
QoE |
|
Pre-transfusion testing |
|||
|
NT.1. What are the samples required for pre-transfusion testing in neonates? |
|||
|
BSH 2020b |
Within the first 4 months, wherever possible, samples from both mother and infant should be obtained for initial ABO and D group determination. The antibody screen should be undertaken on the maternal sample when available. A maternal sample is preferred for antibody testing for the following reasons: · If maternal antibody has bound to fetal cells in vivo, the resulting lower concentration of antibody in neonatal plasma could lead to a false negative antibody screen result. · It is easier to obtain a sufficiently large sample from the mother to allow for screening and antibody identification if required. · Sample collection from the infant exacerbates the anaemia of prematurity. The maternal sample should be collected within 3 days predelivery or collected post-delivery.
|
GPS |
|
|
Red cell transfusion |
|||
|
NR.1. What are the indications and thresholds for transfusion of packed red cells in acute ill neonate without other comorbid conditions? |
|||
|
BSH 2020b |
Studies to date support restrictive transfusion thresholds (2B) and suggested Hb thresholds for top-up transfusions are given in Table 1.
*Standard definition of preterm is <37 weeks gestational age at birth but table applies to very preterm neonates (<32 weeks). †It is accepted that clinicians may use up to 85 g/dl depending on clinical situation. ‡NIPPV, non-invasive positive pressure ventilation. Table 1 does not include suggested thresholds for moderate to late preterm (≥32 weeks gestational age at birth) or term neonates, as there is little evidence regarding the appropriate thresholds for these groups. Clinicians may consider similar thresholds to those used for preterm babies off oxygen.
|
Intermediate |
Strong |
|
NR.2. What are the indications to transfuse packed red cells in acute ill neonate with neonatal sepsis? |
|||
|
Villeneuve et al 2020 31 |
Decision of transfusion is based on the clinical status. For those not requiring cardiopulmonary support or oxygen supply and condition is stable, transfusion is not usually required unless Hb level is below 7g/dl. |
GPS |
|
|
NR.3. What is the volume required for red cell transfusion in neonates? |
|||
|
BSH 2020b |
Transfusion volumes of 15 ml/kg are generally recommended for non-bleeding neonates. |
Intermediate |
weak |
|
BSH 2020b |
Repeated small volume ‘red cell transfusions (up to 20 mL/kg) are commonly carried out in preterm babies, mainly to replace losses from repeated blood testing exacerbated by reduced red cell production (‘anaemia of prematurity’).
|
Intermediate |
Strong |
|
NR.4. What are the characteristics of red cells used for exchange transfusion in neonates? |
|||
|
BSH 2020b |
- A specific red cell component for neonatal exchange transfusion, usually group O, and should also be compatible with any maternal antibody. - Red cells suitable for neonatal exchange need to be irradiated and ‘fresh’ (before the end of Day 5 following donation, with a 24-h shelf-life post-irradiation in order to reduce the risk of recipient hyperkalaemia. They have a controlled hematocrit 0.5–0.6. They are negative for high-titre anti-A and anti-B antibodies. - Exchange blood transfusion (EBT) should not be undertaken with red cells straight from 4°C storage, and an approved blood-warming device can be used to avoid hypothermia. The use of a blood warmer is only appropriate if the infusion is given at a constant rate (warming is not suited to the intermittent bolus nature of a single vessel EBT where the ‘push-pull’ cycle method is used). Blood warming during EBT should not be uncontrolled, e.g., infusion lines exposed to a radiant heater, because of the risk of red cell hemolysis.
|
GPS |
|
|
Platelets |
|||
|
NP.1. What are the indications and thresholds for platelet transfusion in neonates? |
|||
|
BSH 2020b |
In Preterm neonate - For preterm neonates with very severe thrombocytopenia (Platelet count below 25 × 109/l) platelet transfusions should be administered in addition to treating the underlying cause of the thrombocytopenia |
Intermediate |
Weak |
|
BSH 2020b |
In full-term neonate - For non-bleeding neonates, platelet transfusions should not be routinely administered if platelet count is ≥ 25 × 109/l. |
High |
Strong |
|
BSH 2020b |
- If Platelet count < 25× 109/l transfuse in Neonates with no bleeding - If Platelet count < 50× 109/l transfuse in Neonates with bleeding, current coagulopathy, before surgery, or infants with FNAIT if previously affected sibling with ICH. - If Platelet count < 100× 109/l transfuse in Neonates with major bleeding or requiring major surgery (e.g., neurosurgery) |
Intermediate Intermediate
Intermediate |
Weak Weak
Weak |
|
NNF 2020 |
In antibody mediated thrombocytopenia - In neonatal alloimmune thrombocytopenia (FNAIT), maintaining platelet count > 30 x109/l is strongly recommended. |
High |
Weak |
|
NNF 2020 |
In preterm neonate with PDA - The routine use of platelet transfusion for PDA closure in thrombocytopenic preterm neonates with PDA is not recommended.
|
Intermediate |
weak |
|
NP.2. What is the dose and rate for platelet transfusion in neonates? |
|||
|
BSH 2020b |
Typical transfusion volume: 10–20 ml/kg Transfusion rate: 10–20 ml/kg/h |
GPS |
|
|
NP.3. Is ABO compatibility required for platelet transfusion in neonates? |
|||
|
BSH 2016b |
ABO, Rh matched platelets should be used when available to maximize increments |
Intermediate |
Weak |
|
BSH 2016b |
It is acceptable to use ABO incompatible platelets to reduce wastage. Platelets tested and negative for high titre haemagglutinins and non-group O platelets are associated with a lower risk of haemolysis. |
High |
Strong |
|
Fresh Frozen Plasma (FFP) |
|||
|
NF.1. What are the therapeutic indications of fresh frozen plasma transfusion (FFP) in neonates? |
|||
|
BSH 2020b |
FFP may be of benefit in neonates with clinically significant bleeding (including massive blood loss) or prior to invasive procedures with a risk of significant bleeding, and who have an abnormal coagulation profile, defined as a PT or aPTT significantly above normal gestational and postnatal age-related reference range. (Considering local reference ranges where available). |
Intermediate |
Weak |
|
BSH 2020b |
FFP is appropriate for the early management of severe hereditary protein C deficiency but should not be used in preference to protein C concentrate if this is available. |
Intermediate |
Strong |
|
BSH 2020b |
FFP should be used for the management of severe hereditary protein S deficiency. |
Intermediate |
Strong |
|
BSH 2020b |
Management of DIC, inherited deficiency of clotting factors, vitamin- K deficiency bleeding (prothrombin complex concentrates are preferable to FFP). |
Intermediate |
Weak |
|
BSH 2018 – ad 2020
|
If virally inactivated specific clotting factors are not available, pathogen-reduced plasma may be used for factor replacement in congenital coagulation factor deficiency. |
High |
Weak |
|
NNF 2020 |
FFP transfusion is preferred over cryoprecipitate in the management of disseminated intravascular coagulation. |
Intermediate |
Weak |
|
BSH 2020b |
Where indicated, cryoprecipitate may be used if there is persistent hypofibrinogenemia (<1.0 g/L) despite FFP transfusion, or in conjunction with FFP for very low or rapidly falling fibrinogen. |
Intermediate |
Weak |
|
NF.2. What are the prophylactic indications of fresh frozen plasma transfusion in neonates? |
|||
|
NNF 2020 |
The routine use of prophylactic FFP in preterm neonates is not recommended. |
High |
Weak |
|
NNF 2020 |
Prophylactic FFP is not recommended in non-bleeding neonates receiving therapeutic hypothermia and having deranged coagulation parameters |
High |
Strong |
|
NNF 2020 |
Neonates with deranged coagulation parameters and planned for surgical or invasive procedures should receive FFP. |
High |
Weak |
|
NF.3. What are the indications of fresh frozen plasma transfusion in neonatal emergency? |
|||
|
NNF 2020 |
FFP 15–20 ml/kg given 8–12 hourly may be used as first line therapy to treat acquired neonatal purpura fulminans in association with protein C or protein S deficiency while the underlying cause is being investigated. The underlying cause should be treated, and it may be helpful to monitor PC/PS levels. |
GPS |
|
|
NF.4. What are the contraindications to plasma transfusion in neonates? |
|||
|
NNF 2020 |
There is no evidence to support the routine use of FFP to try to correct abnormalities of the coagulation screen alone in non-bleeding neonates. |
Intermediate |
Weak |
|
NNF 2020 |
Prophylactic FFP is not recommended in non-bleeding neonates receiving therapeutic hypothermia and having deranged coagulation parameters. |
High |
Strong |
|
NNF 2020 |
FFP should not be used in the management of inherited factor deficiencies other than in a few exceptional circumstances where specific factor concentrates are not available. |
High |
Strong |
|
NNF 2020 |
FFP should not be used for simple volume replacement or routinely for prevention of intraventricular hemorrhage. |
Intermediate |
Weak |
|
NNF 2020 |
FFP should not be used for performing a partial exchange transfusion for polycythemia |
GPS |
|
|
NF.5. What is the dose and rate of plasma transfusion in neonates? |
|||
|
BSH 2018 – ad 2020
|
For patients who have abnormal clotting tests and other factors (i.e., personal/family bleeding history, drug history, bleeding risk associated with planned procedure or thrombocytopenia) that indicate a significant bleeding risk during a procedure, then a starting dose of 15 ml/ kg of FFP can be considered. |
High |
Strong |
|
BSH 2018 – ad 2020
|
For the first 15 minutes: 1 mL/kg/hr.; reassess patient, if well tolerated increase rate as per Physician’s order (this is recommended “test dose, slow rate of infusion”). Usual rate: 10 to 20 mL/kg/hr. |
GPS |
|
Part II: Transfusion in infants, children, and adolescents
|
CPGL Source |
Recommendation |
SoR |
QoE |
|
Packed Red Blood Cells (PRBCs) |
|||
|
I. Indications |
|||
|
PR.1. What are the indications and thresholds of red cell transfusion in different paediatric diseases? |
|||
|
BSH 2016b |
In children with cancer • There is insufficient evidence to make recommendations for pre-transfusion Hb thresholds in paediatric haematology/ oncology patients and those undergoing stem cell transplantation.
|
Intermediate |
Weak |
|
|
• In children with oncologic diagnoses who are critically ill or at risk for critical illness, and hemodynamically stable, an Hb concentration of 7– 8 g/dl is suggested as a threshold for RBC transfusion. |
Intermediate |
Weak |
|
BSH 2016b |
In children with pure red cell aplasia • Patients with chronic anaemia due to red cell aplasia may require an Hb threshold of 8 g/dl. |
Intermediate |
Weak |
|
BSH 2016a |
In children with sickle cell disease • Transfusion is recommended and may be lifesaving in acute sickle complications such as splenic sequestration, hepatic sequestration, aplastic crisis and severe acute chest syndrome |
High |
Strong |
|
BSH 2016a |
• Simple transfusion to steady state haemoglobin concentration is indicated for patients with acute exacerbation of anaemia as a result of aplastic crisis or sequestration crisis. • Over-transfusion (to Hb > 8 g/dl) should be avoided in sequestration crises because of the risk of hyperviscosity due to the re-entry of sequestered red cells into the circulation. |
High |
Strong |
|
BSH 2016a |
• There is no evidence that transfusion shortens the duration of a painful crisis. Transfusion is not recommended in uncomplicated painful crises but should be considered if there is a substantial drop in Hb from baseline (e.g., >2 g/dl or to Hb <5 g/dl), haemodynamic compromise or concern about impending critical organ complications. |
High |
Weak |
|
BSH 2016a |
• Transfusion should be considered in the unwell patient with acute multi-organ failure, mesenteric syndrome (1C) and patients with severe sepsis (2C). |
High Intermediate |
Weak
Weak |
|
BSH 2016a |
• Transfusion is recommended in cases of acute chest syndrome with hypoxia. Transfusion may be given by simple or exchange transfusion depending on clinical severity under the guidance of the specialist haemoglobinopathy team. |
High |
Strong |
|
BSH 2016a |
• Adults or children with signs or symptoms suggestive of acute ischaemic stroke should be transfused to sickle haemoglobin (HbS) <30% pending further investigation. Those with confirmed stroke due to sickle cell disease should continue regular transfusions indefinitely. |
High |
Strong |
|
BSH 2016a |
• Transfusion is not recommended to treat steady state anaemia provided that Hb has not fallen over a period of time to symptomatic levels (e.g., with developing chronic kidney disease). |
High |
Weak |
|
TAXI 2018 |
In critically ill children admitted to PICU • In critically ill children or those at risk for critical illness, who are hemodynamically stable and who have an Hb concentration ≥ 7 g/dl, we recommend not administering a RBC transfusion. |
High |
Strong |
|
TAXI 2018 |
• When deciding to transfuse an individual critically ill child, consider not only the hemoglobin (Hb) concentration, but also the overall clinical context (e.g. symptoms, signs, physiological markers, laboratory results) and the risk, benefits, and alternatives to transfusion. |
GPS |
|
|
TAXI 2018 |
• In critically ill children or those at risk for critical illness, we recommend measuring the hemoglobin (Hb) concentration before prescribing each RBC transfusion; knowledge of Hb concentration is not required before RBC transfusion if the patient has life threatening bleeding. |
GPS |
|
|
BSH 2016b |
In the preoperative setting • A perioperative Hb transfusion threshold of 7 g/dl should be used in stable patients without major co-morbidity or bleeding. |
High |
Weak |
|
PR.2. What are the definitions and precautions of transfusion with massive blood loss? |
|||
|
BSH 2016b |
Massive blood loss (MBL) may be defined as either 80 ml/kg in 24 h, 40 ml/kg in 3 h or 2–3 ml/kg/min. In clinical practice, haemodynamic changes compatible with hypovolaemia accompanying evidence or suspicion of serious haemorrhage are the usual triggers. |
GPS |
|
|
BSH 2016b |
Key principles in MBL are: 1 Early recognition of children at risk of MBL using clinical parameters. 2 Education of staff to understand when to activate/trigger the local major haemorrhage protocol. 3 Active resuscitation and control of bleeding. 4 Seek specialist assistance. 5 Rapid provision of O D-negative or group specific red cells. 6 Prescribe all transfused components in ml/kg bodyweight (for children <50 kg) and not as units. 7 Anticipate and treat coagulopathy and thrombocytopenia in trauma with early use of FFP and consideration of platelets and cryoprecipitate in on-going bleeding. 8 Use tranexamic acid in trauma. 9 Avoid hypothermia, hypocalcaemia, acidosis and hyper-kalaemia.
Appropriate aliquots to be transfused are as follow: - RBCs 20 ml/kg aliquots (maximum four adult units), D-negative or ABO and D-specific (ideally, cross-matched) - Group specific FFP in 20 ml/kg aliquots (maximum four adult units) - Platelets in 15–20 ml/kg aliquots (maximum one adult therapeutic dose) to be considered after every 40ml/kg RBCs - Cryoprecipitate 10 ml/kg (maximum two pools)
Initial immediate transfusion of 20 ml/kg RBCs should be given (up to four adult units). There was no difference between early administration of plasma, platelets and RBCs in a 1:1:1 ratio and in a 1:1:2 ratio32. A ratio of at least 1 FFP:2 RBC is recommended in early resuscitation of major haemorrhage (in major trauma clinicians may consider aiming for a ratio of 1 FFP:1 RBC). Platelets and cryoprecipitate must be considered if active bleeding persists after initial resuscitation. These aliquots should be repeated in recommended ratios as necessary until bleeding is controlled. Ratios should be modified accordingly once laboratory parameters are available. The therapeutic aims should be Hb 8 g/dl, fibrinogen > 1.5 g/l, PT ratio < 1.5, platelet count > 75x109/l. Careful monitoring for adequacy of resuscitation and for circulatory overload is essential. |
GPS |
|
|
II. Volume and rate of PRBCs transfusion |
|||
|
PR.3. What is the best way of ordering volume of PRBCs in children? |
|||
|
|
Prescription of blood components for paediatric transfusion should be in millilitres unless there are local risk-assessed protocols for prescribing in units for older children, and the maximum volume should not be greater than prescribed for adults. |
High |
Weak |
|
PR.4. What is the volume and rate of PRBCs transfusion in in acute ill children? |
|||
|
|
In a non-bleeding infant or child, it is important to take into account the pre-transfusion Hb in relation to the transfusion threshold, and it is recommended that a post-transfusion Hb no more than 2 g/dl above the threshold be aimed for.
|
GPS |
|
|
|
Transfusion rate 5 ml/kg/h (usual maximum rate: 150 ml/h) |
GPS |
|
|
PR.5. What is the transfusion volume for regular transfusion in transfusion-dependent thalassemia in chronic transfusion program? |
|||
|
TIF 2021*33 |
(Desired – actual Hb (g/dl)) x weight (kg) x 3 = ml to be transfused assuming the haematocrit of the unit is 0.58.
|
GPS |
|
|
PR.6. What are the standards for PRBCs transfusion in transfusion-dependent thalassemia? |
|||
|
UK Thalassaemia Society clinical standards34
|
· Blood must be ABO compatible and antigen negative for any clinically significant antibodies the patient is known to have, or to have had previously identified even if not currently detectable. It should be fully matched for all the Rh antigens and K. · Units should be less than 2 weeks old and, in adults, of larger volume where possible. · There should be a clear record of patient’s transfusion requirements outlining volume, frequency and target haemoglobin. · Transfusions will be given on each occasion in a designated age-appropriate area with suitable facilities, experienced regular named nurses and familiar supervising medical team. Pre-arranged transfusions should be started within 30 minutes of the patient’s arrival.
|
GPS |
|
|
BSH 2016b |
Patients with transfusion-dependent anaemia and sickle cell disease should preferably have extended red cell phenotyping or genotyping (D, C, c, E, e, K, Fya, Fyb, Jka, Jkb, M, N, S and s) prior to transfusion and, as a minimum, red cells should be matched for Rh (D, C, c, E, e) and K antigens. |
GPS |
|
|
Bowden 199535 |
GPS |
||
|
Platelets |
|||
|
PP.1. What are the indications for platelet transfusion in infants and children during acute bleeding? |
|||
|
|
In non-immune thrombocytopenia |
|
|
|
BSH 2016b |
• In severe bleeding, maintain the platelet count above 50 ×109 /l. Consider empirical use for the initial management of major haemorrhage. |
High |
Weak |
|
BSH 2016b |
• In patients with multiple trauma, traumatic brain injury or spontaneous intracerebral haemorrhage, maintain the platelet count above 100 ×109 /l |
Intermediate |
Weak |
|
BSH 2016b |
• In patients with bleeding that is not considered severe or life-threatening, consider platelet transfusion if the platelet count is below 30 ×109 /l |
Intermediate |
Weak |
|
|
In immune thrombocytopenia |
|
|
|
BSH 2016b |
• In ITP, consider co-administration of intravenous immunoglobulin in addition to the platelet transfusion |
Intermediate |
Weak |
|
BSH 2016b |
• In post-transfusion purpura (PTP), intravenous immunoglobulin is the treatment of choice |
High |
Weak |
|
BSH 2016b |
• Give therapeutic platelet transfusions (more than one dose) to treat serious bleeding |
High |
Weak |
|
BSH 2016b |
• Only use platelet transfusion prior to a procedure or surgery when other treatment has failed and/or the intervention is urgent. Usual threshold counts may be unachievable or unnecessary and individual case review is required |
High |
Weak |
|
|
In Platelet Function Disorders (Congenital) |
|
|
|
BSH 2016b |
• If pharmaceutical therapies are contraindicated, ineffective or if there is high risk of bleeding, consider transfusion of platelets. In Glanzmann thrombasthenia, consider human leucocyte antigen (HLA)-matched platelets (2C) where available (GPS).
|
Intermediate |
Weak |
|
|
In drug- induced Platelet Function Disorders (Acquired) |
|
|
|
BSH 2016b |
• Do not use platelet transfusion pre-procedure when antiplatelet agents have not been discontinued |
Intermediate |
Weak |
|
BSH 2016b |
• Use general haemostatic measures to treat bleeding in patients during treatment with aspirin, P2Y12 antagonists or glycoprotein IIa/IIIb inhibitors. If necessary, consider drug cessation and reversal of the effect of co-prescribed anticoagulants |
Intermediate |
Weak |
|
BSH 2016b |
• Use TXA to counteract the effect of anti-platelet agents when a risk/benefit assessment would support this |
High |
Strong |
|
BSH 2016b |
• Consider the use of platelet transfusion as an additional measure to those suggested above for critical bleeding |
Intermediate |
Weak |
|
PP.2. What are the indications for prophylactic platelet transfusion? |
|||
|
BSH 2016b |
In critically ill child • Use the platelet count thresholds for reversible bone marrow failure as a general guide for prophylactic platelet transfusion in patients with critical illness in the absence of bleeding or planned procedures. |
Intermediate |
Weak |
|
BSH 2016b |
In autoimmune thrombocytopenia (ITP) • Do not use prophylactic platelet transfusions in patients with autoimmune thrombocytopenia
|
High |
Weak |
|
|
In reversible bone marrow failure (receiving intensive chemotherapy or undergoing allogeneic haematopoietic stem cell transplant (HSCT) |
|
|
|
BSH 2016b |
• Give prophylactic platelet transfusions to patients to maintain a platelet count at or above 10 x 109/l |
High |
Strong |
|
BSH 2016b |
• Use only one adult dose (one unit) routinely for prophylactic platelet transfusions |
High |
Strong |
|
BSH 2016b |
• Consider increasing the threshold for prophylactic platelet transfusion to between 10 and 20 x 109/l /l in patients judged to have additional risk factors for bleeding. Individual review is required. |
Intermediate |
Weak |
|
|
In chronic bone marrow failure (when recovery is not anticipated) |
|
|
|
BSH 2016b |
• Use a ‘no prophylactic platelet transfusion’ strategy for asymptomatic patients with chronic bone marrow failure. |
Intermediate |
Strong |
|
BSH 2016b |
• Give prophylactic platelet transfusions to patients with chronic bone marrow failure receiving intensive treatment. |
High |
Strong |
|
BSH 2016b |
• Manage patients with chronic bleeding of WHO grade 2 or above individually (see implementation tools) , according to the severity of their symptoms and signs. Consider a strategy of prophylaxis (e.g., twice a week). |
Intermediate |
Weak |
|
|
Prior To Procedures or Surgery: |
|
|
|
BSH 2016b |
• Do not give platelet transfusions routinely prior to: - bone marrow aspirate or trephine biopsy - peripherally inserted central catheters (PICCs) - traction removal of tunneled CVCs - cataract surgery |
High Inter. Inter. Inter. |
Weak Weak Weak Weak |
|
BSH 2016b |
• Whenever possible use a procedure/equipment associated with the lowest bleeding risk. Apply local measures, such as compression, to reduce the risk of bleeding post-procedure |
High |
Weak |
|
|
Prior To Procedures or Surgery |
|
|
|
BSH 2016b |
Consider performing the following procedures above the platelet count threshold indicated: |
|
|
|
BSH 2016b |
• venous central lines (both tunneled and un-tunneled), inserted by experienced staff using ultrasound guidance techniques, when the platelet count is >20 ×109 /l |
High |
Weak |
|
BSH 2016b |
• lumbar puncture when the platelet count is ≥40 ×109/l. |
Inter. |
Weak |
|
BSH 2016b |
• insertion/removal of epidural catheter when the platelet count is ≥80 ×109/l. |
Inter. |
Weak |
|
BSH 2016b |
• major surgery – when the platelet count is >50×109/L |
High |
Weak |
|
BSH 2016b |
• neurosurgery or ophthalmic surgery involving the posterior segment of the eye when the platelet count is >100×109 /l |
High |
Weak |
|
BSH 2016b |
• percutaneous liver biopsy when the platelet count is >50 ×109/l. Consider trans-jugular biopsy if the platelet count is below this level |
Inter. |
Weak |
|
PP.3. What is the dose and rate for platelet transfusion? |
|||
|
BSH 2020b |
Typical transfusion volume: • 10–20 ml/kg for children <15 kg, or a single pack for children ≥15 kg • maximum volume 1 pack Transfusion rate: 10–20 ml/kg/h |
GPS |
|
|
PP.4. What are the contraindications of platelet transfusion? |
|||
|
BSH 2016b |
In patients with thrombotic microangiopathies only use platelet transfusions to treat life-threatening bleeding. |
High |
weak |
|
PP.5. How to avoid/ manage the risks of platelets transfusion? |
|||
|
BSH 2016b |
Hospitals should establish a strategy to maximize the transfusion of ABO compatible platelets, especially to patients who require regular platelet support |
Intermediate |
Strong |
|
BSH 2016b |
ABO, Rh matched platelets should be used when available to maximize increments |
Intermediate |
Weak |
|
BSH 2016b |
It is acceptable to use ABO incompatible platelets to reduce wastage. Platelets tested and negative for high titre haemagglutinins and non-group O platelets are associated with a lower risk of haemolysis. Pooled platelets suspended in Platelet Additive Solution (PAS) would also be expected to reduce this risk. |
High |
Strong |
|
BSH 2016b |
RhD negative girls or women of childbearing potential should receive RhD negative platelets. If unavailable, RhD positive platelets can be given with anti-D prophylaxis. |
High |
Strong |
|
BSH 2016b |
For RhD negative boys under 18 years of age, those who already have anti-D antibodies, and transfusion-dependent adults, the platelets of choice are RhD negative. RhD positive platelets should be given if RhD negative platelets are unavailable or to prevent wastage of RhD positive components. Anti-D prophylaxis is not required |
High |
Strong |
|
BSH 2016b |
In patients with a history of allergic transfusion reactions, apart from mild, use platelets suspended in PAS. If reactions continue or are severe, washed platelets (resuspended in 100% PAS) may be required |
High |
Strong |
|
BSH 2016b |
Patients with hypoproliferative thrombocytopenia who are refractory to platelet transfusions and have class I HLA antibodies should receive class I HLA-selected platelet transfusion |
Intermediate |
Weak |
|
BSH 2016b |
Patients with hypoproliferative thrombocytopenia who continue to be refractory to HLA-selected platelet transfusions and have human platelet antigen (HPA) antibodies should receive HPA-selected platelet transfusion |
Intermediate |
Weak |
|
BSH 2016b |
Patients with hypoproliferative thrombocytopenia who are not refractory to platelet transfusion may not receive HLA-selected or HPA-selected platelets
|
Intermediate |
Weak |
|
Fresh Frozen Plasma |
|||
|
PF.1. What are the indications for plasma transfusion during acute bleeding in paediatrics? |
|||
|
BSH
2018 – |
Pathogen-reduced plasma may be used for factor replacement in congenital coagulation factor deficiency If virally inactivated specific clotting factors are not available. |
High |
Weak |
|
BSH 2016b |
FFP may be beneficial in children with DIC who have a significant coagulopathy (PT/APTT >1.5 times midpoint of normal range or fibrinogen <1.0 g/l) associated with clinically significant bleeding or prior to invasive procedures |
Intermediate |
Weak |
|
BSH 2016b |
In DIC, cryoprecipitate may be given if the fibrinogen is < 1.0 g/l despite FFP, or in conjunction with FFP for very low or rapidly falling fibrinogen. |
Intermediate |
Weak |
|
BSH 2016b |
Prophylactic FFP should not be administered to non-bleeding children with minor prolongation of the prothrombin time (2B)/ activated partial thromboplastin time including prior to surgery, although it may be considered for surgery to critical sites (2C) |
Intermediate
Intermediate |
Strong
Weak |
|
BSH 2016b |
Prophylactic cryoprecipitate should not be routinely administered to non-bleeding children with decreased fibrinogen including prior to surgery. It may be considered for fibrinogen <1g/l for surgery at risk of significant bleeding or to critical sites. |
High |
Weak |
|
BSH 2016b |
FFP should not be used in the management of inherited factor deficiencies other than in a few exceptional circumstances where specific factor concentrates are not available. |
High |
Weak |
|
BSH 2016b |
Urgent plasma exchange with solvent detergent fresh frozen plasma (SD FFP) is indicated for thrombotic thrombocytopenic purpura (TTP) (1B) and some forms of atypical hemolytic uremic sundrome (HUS) (2C). |
High
Intermediate |
Strong
Weak |
|
PF.2. What is the dose/ frequency for plasma transfusion during acute bleeding in paediatrics? |
|||
|
BSH 2016b |
The recommended therapeutic dose of FFP is 10-15 mL/kg of body weight given 8–12 hourly depending on the clinical situation and laboratory parameters
|
High |
Weak |
|
PF.3. What is the best choice of blood group for plasma transfusion? |
|||
|
BSH
2018 – |
Plasma of donors with identical ABO blood group to the recipient should be used as the first choice. If this is not possible, ABO non-identical but compatible plasma is acceptable if it has ‘low-titre’ anti-A or anti-B activity. |
High |
Strong |
|
BSH
2018 – |
Group O plasma should only be given to group O patients. |
High |
Strong |
|
BSH
2018 – |
Fresh frozen plasma and cryoprecipitate of any RhD group may be transfused. If RhD positive plasma is given to an RhD negative individual, no anti-D prophylaxis is required. |
High |
Strong |
Part III: Modification of blood components and related Precautions
|
MO.1. What are the indications and dose for irradiation of cellular blood components? |
|||
|
BSH 2020a |
- Gamma- or X-irradiation of blood components, by validated systems, is the recommended procedure to prevent TA-GvHD. - The minimum dose achieved in the irradiation volume should be 25 Gy, with no part receiving >50 Gy. |
High
High |
Strong
Strong |
|
BSH 2020a |
In Intrauterine transfusion and neonatal transfusion - Red cells for neonatal exchange blood transfusion (EBT) should be irradiated. |
High |
Weak |
|
BSH 2020a |
- Routine irradiation of red cells for transfusion to preterm or term infants (other than for EBT) is not required unless there has been a previous intrauterine transfusion (IUT), in which case irradiated components should be administered until 6 months after the expected delivery date (40 weeks gestation). |
Intermediate |
Weak |
|
BSH 2020a |
- Routine irradiation of platelet transfusions for preterm or term infants is not required unless there has been a previous IUT, in which case irradiated components should be administered until 6 months after the expected delivery date (40 weeks gestation).
|
Intermediate |
Weak |
|
BSH 2020a |
In first and second-degree relative - All transfusions of cellular components and fresh plasma from first- or second-degree relatives should be irradiated, even if the patient is immunocompetent. All HLA-selected components should be irradiated even if the patient is immunocompetent.
|
High |
Strong |
|
BSH 2020a |
In immunodeficiency - All severe congenital T-lymphocyte immunodeficiency syndromes with significant qualitative or quantitative T-lymphocyte deficiency should be considered as indications for irradiation of cellular blood components. |
High |
Strong
|
|
BSH 2020a |
- Once a diagnosis of severe T-lymphocyte immunodeficiency has been suspected, irradiated components should be given while further diagnostic tests are being undertaken. |
High |
Weak |
|
|
- There is no indication for irradiation of cellular blood components for infants or children with temporary defects of T-lymphocyte function as the result of a viral infection. There is also no indication for irradiation of cellular blood components for adults or children who are HIV-antibody positive or who have acquired immune deficiency syndrome (AIDS). |
High |
Strong |
|
BSH 2020a |
In Haematopoietic stem cell transplant (HSCT) - All recipients of allogeneic HSCT should receive irradiated blood components from the time of initiation of conditioning chemo/ radiotherapy. The recommendation applies for all conditions where HSCT is indicated regardless of the underlying diagnosis. Irradiated components should be continued until all of the following criteria are met: 1. >6 months have elapsed since the transplant date 2. The lymphocyte count is >1x 109/l 3. The patient is free of active chronic GvHD 4. The patient is off all immunosuppression |
High
|
Strong
|
|
|
- If chronic GvHD is present or continued immunosuppressive treatment is required, irradiated blood components should be given indefinitely. |
Intermediate |
Weak |
|
|
- Allogeneic cellular blood components transfused to bone marrow and peripheral blood stem cell donors of all ages within 7 days prior to or during the harvest should also be irradiated. |
Intermediate |
Weak |
|
|
- Patients undergoing bone marrow or peripheral blood stem cell collections for future autologous re-infusion should receive irradiated cellular blood components for 7 days prior to and during the bone marrow/stem cell harvest to prevent the collection of viable allogeneic T lymphocytes, which can potentially withstand cryopreservation. |
High |
Weak |
|
|
- All patients undergoing autologous stem cell transplant (ASCT) irrespective of underlying diagnosis or indication for this treatment should receive irradiated cellular blood components from initiation of conditioning chemo/radiotherapy until 3 months post-transplant (6 months if total body irradiation was used in conditioning) unless conditioning, disease or previous treatment determine indefinite duration, for example previous diagnosis of Hodgkin lymphoma (HL) or previous purine analogue treatment. |
High |
Weak |
|
BSH 2020a
|
In aplastic anaemia - For patients with aplastic anaemia, transfusion of irradiated cellular components is not routinely recommended, except for HLA-selected platelets, transfusion of granulocytes, donations from first- or second- degree relatives, or planned relevant treatment (e.g. ATG, alemtuzumab, HSCT). |
High |
Strong |
|
BSH 2020a
|
In Hodgkin lymphoma (HL) - All adults and children with HL at any stage of the disease should have irradiated red cells and platelets indefinitely. |
Intermediate |
Weak |
|
BSH 2020a
|
Medications - All patients treated with purine analogue drugs (fludarabine, cladribine, bendamustine and pentostatin) should receive irradiated blood components indefinitely. |
Intermediate |
Weak |
|
|
- Patients with haematological diagnosis treated with alemtuzumab should receive irradiated components. |
Intermediate |
Weak |
|
|
- Patients with aplastic anaemia undergoing treatment with ATG or alemtuzumab should receive irradiated blood components. |
Intermediate |
Weak |
|
|
- Patients receiving ATG or other T-lymphocyte-depleting serotherapy for rare types of immune dysfunction conditions should receive irradiated blood components. |
Intermediate |
Weak |
|
|
- Treatment of patients with rituximab is not an indication for use of irradiated cellular blood components unless this is indicated for a different reason (underlying diagnosis, type of component or previous treatment). |
High |
Strong |
|
BSH 2020a |
In solid organ transplantation (SOT) - Use of irradiated cellular blood components is not indicated for patients undergoing solid organ transplantation (SOT) who have received alemtuzumab or ATG as induction therapy or for treatment of graft rejection. |
High |
Strong |
|
MO.2. What are the indications for use of washed PRBCs? |
|||
|
BSH 2012 |
For patients with recurrent febrile reactions, it is recommended to use a trial of premedication with oral paracetamol given one hour before the reaction is anticipated (or non-steroidal anti-inflammatory drugs in patients with predominant chills or rigors - but an assessment of the risks of medication against the severity of reaction should be made in each case). Patients who continue to react should have a trial of washed blood components. |
Intermediate |
Weak |
|
Handbook of transfusion medicine36 |
Patients with recurrent or severe allergic or febrile reactions to red cells, and severely IgA-deficient patients with anti-IgA antibodies for whom red cells from an IgA-deficient donor are not available. |
GPS |
|
|
MO.3. What are the indications to give leukofiltered blood components in neonates? |
|||
|
NNF 2020 |
Provision of CMV safe blood for transfusion in preterm neonates by using CMV seronegative donors or leukoreduction or a combination of both is strongly recommended. |
High |
Weak |
|
NNF 2020 |
For intrauterine transfusions use of CMV negative and leuco-depleted packed red blood cell is strongly recommended. |
High |
Weak |
|
MO.4. What are the characteristics of plasma transfusion and its storage? |
|||
|
BSH 2018 – ad 2020 |
Plasma of donors with identical ABO blood group to the recipient should be used as the first choice. If this is not possible, ABO non-identical but compatible plasma is acceptable if it has ‘low-titre’ anti-A or anti-B activity. |
High |
Strong |
|
BSH 2018 – ad 2020 |
Group O plasma should only be given to group O patients |
High |
Strong |
|
BSH 2018 – ad 2020 |
FFP and cryoprecipitate of any RhD group may be transfused. If RhD positive plasma is given to an RhD negative individual, no anti-D prophylaxis is required. |
High |
Strong |
|
BSH 2018 – ad 2020 |
Once thawed, standard FFP or methylene blue treated FFP (MBFFP) may be stored at 2-4°C in an approved temperature-controlled blood storage refrigerator before administration to the patient, as long as the infusion is completed within 24 hours of thawing. |
Intermediate |
Strong |
|
BSH 2018 – ad 2020 |
Transfusion of FFP should be completed within 4 hours of issue out of a controlled temperature environment. |
Intermediate |
Strong |
|
BSH 2018 – ad 2020 |
Pre-thawed FFP that is out of a controlled temperature environment 2-4°C) can be accepted back into temperature- controlled storage if this occurs on one occasion only of less than 30 min). |
Intermediate |
Strong |
Part IV: Acute Transfusion Reactions
|
Source |
Recommendations |
Quality of evidence |
Level of recommendations |
|
TR.1. How to diagnose acute transfusion reaction (ATR)? |
|||
|
BSH 2012 |
All patients should be transfused in clinical areas where they can be directly observed, and where staff are trained in the administration of blood components and the management of transfused patients, including the emergency treatment of anaphylaxis. |
High |
Weak |
|
BSH 2012 |
The recognition and immediate management of ATR should be incorporated into local transfusion policies and there should be mandatory transfusion training requirements for all clinical and laboratory staff involved in the transfusion process. |
Intermediate |
Weak |
|
BSH 2012 |
If a patient develops new symptoms or signs during a transfusion, this should be stopped temporarily, but venous access maintained. Identification details should be checked between the patient, their identity band and the compatibility label of the blood component. Perform visual inspection of the component and assess the patient with standard observations. |
High |
Weak |
|
BSH 2012 |
Standard observations during blood components administration include the patient’s pulse rate, blood pressure, temperature and respiratory rate should be monitored and abnormal clinical features, such as fever, rashes or angioedema, frequently assessed. Symptoms and signs of acute transfusion reactions include: · Fever and related inflammatory symptoms or signs, such as chills, rigors, myalgia, nausea or vomiting. · Cutaneous symptoms and signs including urticaria (hives), other skin rashes and pruritus. · Angioedema (localized oedema of the subcutaneous or submucosal tissues), which may be preceded by tingling. · Respiratory symptoms and signs including dyspnoea, stridor, wheeze and hypoxia. · Hypotension. · Pain. · Severe anxiety or ‘feeling of impending doom’. · Bleeding diathesis with acute onset. |
GPS |
|
|
BSH 2012 |
If a patient develops sustained febrile symptoms or signs of moderate severity (temperature ≥39°C OR a rise of ≥2°C from baseline AND/OR systemic symptoms, such as chills, rigors, myalgia, nausea or vomiting), bacterial contamination or a haemolytic reaction should be considered. |
High |
Weak |
|
BSH 2012 |
Patients should be asked to report symptoms that develop within 24 h of completion of the transfusion. |
Intermediate |
Weak |
|
BSH 2012 |
If a patient being transfused for haemorrhage develops hypotension, careful clinical risk assessment is required. If the hypotension is caused by haemorrhage, continuation of the transfusion may be lifesaving. In contrast, if the blood component is considered the most likely cause of hypotension, the transfusion must be stopped or switched to an alternative component and appropriate management and investigation commenced. |
High |
Weak |
|
TR.2. How to treat Acute transfusion reactions? |
|||
|
BSH 2012 |
Initial treatment of ATR is not dependent on classification but should be directed by symptoms and signs. Treatment of severe reactions should not be delayed until the results of investigations are available. |
High |
Weak |
|
BSH 2012 |
For patients with mild reactions, such as pyrexia (temperature of ≥ 38°C AND rise of 1–2°C from baseline), and/or pruritus or rash but WITHOUT other features, the transfusion may be continued with appropriate treatment and direct observation. |
Intermediate |
Strong |
|
BSH 2012 |
Patients with mild isolated febrile reactions may be treated with oral paracetamol (500–1000 mg in adults). Patients with mild allergic reactions may be managed by slowing the transfusion and treatment with an antihistamine. |
Intermediate |
Weak |
|
BSH 2012 |
Anaphylaxis should be treated with intramuscular adrenaline (epinephrine). Patients who are thrombocytopenic or who have deranged coagulation should also receive IM adrenaline if they have an anaphylactic reaction. |
High |
Strong |
|
BSH 2012 |
Shock/severe hypotension associated with wheeze or stridor · For children over 12 years, administer IM adrenaline: 0.5 ml of 1:1000 adrenaline (500 lg) into the anterolateral aspect of the middle third of the thigh. · For children between 6 and 12 years give 0.3 ml of 1:1000 IM adrenaline (300 lg). · For children <6 years give 0.15 ml of 1:1000 IM adrenaline (150 lg). · Adrenaline is repeated, if necessary, at 5-min intervals according to blood pressure, pulse and respiratory function under the direction of appropriately trained clinicians. · Supportive care of anaphylaxis includes: - Rapid fluid challenge of 500–1000 ml crystalloid. - Administration of 10 mg of chlorphenamine IM or by slow intravenous (IV) injection following initial resuscitation. - Administration of 200 mg of hydrocortisone IM or by slow IV injection following initial resuscitation. - If the patient has continuing symptoms of asthma or wheeze, inhaled or intravenous bronchodilator therapy should be considered. |
GPS |
|
|
BSH 2012 |
Shock/severe hypotension without clinical signs of anaphylaxis or fluid overload: - Consider ABO incompatibility or
bacterial contamination. - If bacterial contamination is suspected, take blood cultures from the patient (peripheral vein and through central line, if present) and start broad-spectrum IV antibiotics (the local regimen for patients with neutropenic sepsis would be appropriate). Immediately notify the transfusion laboratory staff and haematologist to arrange culture of the implicated unit/units and contact the blood service so that any other components from the implicated donation can be recalled and quarantined. |
GPS |
|
|
BSH 2012 |
Severe dyspnea without shock. - Consider transfusion-related acute lung injury (TRALI) or transfusion-associated circulatory overload (TACO). Ensure the airway is patent and high-flow oxygen therapy started while urgent expert medical assessment is obtained. Initial investigation should include chest X-ray and oxygen saturation. - Primary treatment of TRALI is ventilatory support |
GPS |
|
|
TR.3. What are the laboratory investigations done for acute transfusion reactions? |
|||
|
BSH 2012 |
In all moderate and severe transfusion reactions, standard investigations, including full blood count, renal and liver function tests and assessment of the urine for haemoglobin should be performed. |
Intermediate |
Weak |
|
BSH 2012 |
If febrile symptoms of moderate severity are sustained, implicated units should be returned to the laboratory for further investigation, the blood service contacted immediately so that associated components from the implicated donation can be withdrawn and the patient sampled for repeat compatibility and culture. |
High |
Weak |
|
BSH 2012 |
Patients who have experienced moderate or severe allergic reactions should have IgA levels measured. Patients with IgA deficiency diagnosed after an ATR should be discussed with an allergist or immunologist regarding future management. |
Intermediate |
Weak |
|
TR.4. What is the subsequent management of recurrent reactions? |
|||
|
BSH 2012 |
For patients with recurrent febrile reactions, it is recommended to give a trial of premedication with oral paracetamol one hour before the reaction is anticipated (or non-steroidal anti-inflammatory drugs in patients with predominant chills or rigors - but an assessment of the risks of medication against the severity of reaction should be made in each case). Patients who continue to react should have a trial of washed blood components. |
Intermediate |
Weak |
|
BSH 2012 |
For recurrent mild allergic reactions, there is no evidence to support routine prophylaxis with antihistamines or steroids. Alternative causes, such as allergy to drugs or latex gloves, should be excluded. |
Intermediate |
Weak |
|
BSH 2012 |
For patients with recurrent moderate or severe allergic reactions, other than those in which the patient is IgA-deficient, options for further transfusion include: • Use of directly monitored transfusion of standard components in a clinical area with resuscitation facilities. Consider antihistamine prophylaxis (although the evidence for efficacy is low, the risks are also low). This may be the only option when further transfusion is urgent and withholding blood is a greater risk. • Transfusion of washed red cells or platelets. • The use of pooled solvent detergent treated FFP when there are recurrent allergic reactions to FFP in patients undergoing plasma exchange. |
Intermediate Intermediate Intermediate |
Weak
Weak
Strong |
|
BSH 2012 |
Patients with confirmed IgA deficiency and a history of reaction to blood should be transfused with components from IgA-deficient donors (first choice) or washed red cells (second choice) if time allows. |
High |
Weak |
|
BSH 2012 |
Life-saving transfusion should not be denied or delayed if these are not immediately available but the facilities and skills to manage severe allergic reactions must be present. |
High |
Weak |
|
TR.5. Do transfusion reactions need to be reported? |
|||
|
BSH 2012 |
All transfusion reactions except mild febrile and/or allergic reactions must be reported to appropriate regulatory and haemovigilance organizations and should also be reviewed within the hospital. |
High |
Weak |
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Galila Mohamed Mokhtar |
Paediatrics Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt |
Editor, Clinical expert, GAG member |
|||
|
Prof. Amira Adly |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Dina Ezzat |
Professor of Paediatrics and Paediatric Haematology, Beni-Suef University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Gehan Lotfy |
Professor of Paediatrics, Minia University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Hoda Hassab |
Professor of Paediatrics and Paediatrics Haematology, Alexandria University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Ilham Youssry |
Professor of Paediatrics and Head of the Paediatric Haematology & BMT unit, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Iman Ragab |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Laila Sherief |
Professor of Paediatrics and Paediatrics Haematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Marwa Zakaria |
Associate Professor of Paediatrics, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Mervat Hesham |
Professor of Paediatrics, Paediatric Haematology & Oncology Unit, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Dr. Naglaa Shaheen |
Consultant of Pediatric Hematology, Health Insurance Organization |
Clinical expert GAG member |
|||
|
Prof. Niveen Salama |
Associate Professor of Paediatrics, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Dr. Nouran Yousef |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Rasha Abdel-Raouf Afifi |
Professor of Paediatrics and Paediatric Haematology, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Rasha El-Ashry |
Professor of Paediatrics, Mansoura University, Egypt |
Clinical expert GAG member |
|||
|
Dr. Sara Makkeyah |
Associate Professor of Paediatrics, Paediatric Haematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Sonia Adolf |
Professor of Paediatrics and Paediatric Haematology, National Research Center, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Associate Professor of Pediatrics Ain Shams University, Egypt |
Developing evidence to decision (EtD) frameworks, participating in search and guideline appraisal |
||||
|
- Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. - Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. - Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
Dr. Lamis Mohsen Elsholkamy
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Ahmed Mahmoud Youssef
|
Fellow of Pediatrics, General Organization For Teaching Hospitals and Institutes (GOTHI). Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof Magdy El-Ekiaby |
Consultant of blood transfusion, director of Shabrawishi Hospital Blood Bank, Vice President of Egyptian Society of Hematology (ESH) |
||||
|
Prof Salwa Youssef |
Professor of Haemtaolgy, consultant of Transfusion Medicine, Ain Shams University Egypt, President of the Egyptian society of transfusion medicine. |
||||
|
Prof Seham Ragab |
Professor of Paediatrics and Paediatric Haematolgy and Oncology, Head of Haematology and Oncology Unit, Paeditaric depertment, Menoufia University, Egypt. |
||||
|
International Peer Reviewers |
|||||
|
Yasser Wali |
Pediatric Hematology/Oncology Unit, Child Health Department, Sultan Qaboos University Hospital, Muscat, Oman |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge the British Society of Haematology (BSH), the Pediatric Critical Care Transfusion and Anemia Expertise Initiative (TAXI), and the National Neonatology Forum, India (NNF) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️ This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
|
aPTT |
Activated partial thromboplastin time |
|
ASCT |
Autologous stem cell transplant |
|
ATG |
Antithymocyte globulin |
|
ATR |
Acute transfusion reactions |
|
BCSH |
British Committee for Standards in Haematology |
|
BSH |
British Society for Haematology |
|
CAR-T |
Chimeric antigen receptor T cells |
|
CPG |
Clinical Practice Guideline |
|
DHS |
Demographic and Health Survey |
|
EBT |
Exchange blood transfusion |
|
EPG |
Egyptian Paediatric Clinical Practice Guidelines Committee |
|
EPG CPG |
EPG Clinical Practice Guideline |
|
ERG |
External Review Group |
|
FFP |
Fresh frozen plasma |
|
FNAIT |
Fetal/neonatal alloimmune thrombocytopenia |
|
GAG |
Guideline Adaptation Group |
|
GDG |
Guideline Development Group |
|
GPS |
Good practice statement |
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
|
HDFN |
Haemolytic disease of the fetus and newborn |
|
HSCT |
Haematopoietic stem cell transplant |
|
HL |
Hodgkin lymphoma |
|
Ig |
Immunoglobulin |
|
IHN |
International hemovigilance network |
|
ISBT |
International Society of blood transfusion |
|
ITP |
Immune thrombocytopenia |
|
IUT |
Intrauterine transfusion |
|
PEX |
Plasma exchange |
|
PICO |
population, intervention, comparison, and outcomes |
|
PIPOH |
Patient population, intervention, professionals, outcomes, and healthcare context |
|
PRBCs |
Packed red blood cells |
|
PT |
Prothrombin time |
|
QoE |
Quality of evidence |
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
|
SAG-M |
saline, adenine, glucose and mannitol |
|
SOT |
Solid organ transplantation |
|
SoR |
Strength of recommendation |
|
TTP |
Thrombotic thrombocytopenic purpura |
|
TACO |
Transfusion associated circulatory overload |
|
TRALI |
Transfusion related acute lung injury |
- Introduction
Blood is a vital human tissue and a precious health resource, required to be adequately available, safe, correctly and rationally used and timely administered.12 Blood transfusion is an essential component of modern health-care systems and when used appropriately, it saves lives, improves health conditions and enhances patient outcomes. However, improper or unnecessary use can increase the risk of serious, acute, and delayed adverse complications, and in underprivileged settings it places further strain on already limited health resources.13 In recognition of its critical role in the patient management over a wide variety of medical conditions, blood and blood products are currently incorporated into the World Health Organization (WHO) model list of essential medicines.14
Paediatric transfusion is a complex area of medicine covering a wide age range from intrauterine life to young adults. The prescriber must balance the risks and benefits of transfusion in each age group and be aware of the indications for special components. However, compared to adult practice there is a relative lack of high-quality research to inform evidence-based guidelines.12. The effectiveness of transfusion of whole blood or any of its components depends in large measure on a clear understanding of the anticipated role in supplying a specific deficiency or correcting a pathologic state while realizing the limitations and hazards.15
Children require transfusion of blood components for a vast array of medical conditions, including acute hemorrhage, haematologic and nonhaematologic malignancies, hemoglobinopathy, and allogeneic and autologous stem cell transplant. Evidence-based literature on paediatric transfusion practices is limited, particularly for non-red blood cell products, and many recommendations are extrapolated from studies in adult populations.16 Recognition of these knowledge gaps has led to increasing numbers of clinical trials focusing on children and establishment of paediatric transfusion working groups in recent years.
Red blood cell (RBC) transfusions are used to treat hemorrhage and to improve oxygen delivery to tissues. Transfusion of RBCs should be based on the patient's clinical condition. Indications for transfusion include symptomatic anaemia, acute sickle cell crisis, and acute blood loss of more than 30 percent of blood volume.17
Except during active hemorrhage, holding blood requirements to a minimum frequently serves the patient to greater advantage than maintaining normal hemoglobin levels by frequent and maximal administrations of blood. This restriction is especially pertinent in the refractory anaemias; the aplastic-hypoplastic group. Here multiple transfusions result not alone in hemosiderosis but in a potential suppressive effect on erythropoiesis and hemoglobin synthesis. This restrictive policy, however, does not apply to thalassemia and other hemogobinopathies. It should be emphasized that the hemoglobin concentration at which transfusion is indicated for patients who require single or multiple administrations of blood cannot be arbitrarily fixed but varies with the attendant clinical signs and symptoms and the promise of relief by this form of treatment.18
Fresh frozen plasma (FFP) is indicated for the deficiency of coagulation factors with abnormal coagulation tests in the presence of active bleeding. Fresh frozen plasma is also indicated for a planned surgery or invasive procedure in the presence of abnormal coagulation tests, for the reversal of warfarin in the presence of active bleeding, or planned procedure when vitamin K is inadequate to reverse the warfarin effect, TTP, and congenital or acquired factor deficiency with no alternative therapy. Other situations where the administration of FFP cannot be recommended for or against based on systematic review include FFP transfusion in trauma patients with massive transfusion. Conditions that cause the deficiency of multiple coagulation factors and may require the administration of FFP include liver disease and disseminated intravascular coagulation.19 Platelet transfusion is indicated to prevent hemorrhage in patients with thrombocytopenia. Cryoprecipitate, as a source of FVIII, vWF and FXIII, is used in cases of hypofibrinogenemia, which most often occurs in the setting of massive hemorrhage or consumptive coagulopathy.20
Access to safe and adequate blood supply remains a public health challenge in many countries, particularly in low resource settings (poor economics). In high-income countries the average donation rate is 32.1/1000 population in comparison to 4.6/1000 population in low-income countries, where about 80% of the world’s population lives and has access to only 20% of the world’s safe blood supply.21 While infectious risks are low, non-infectious serious hazards of transfusion, such as transfusion-associated lung injury (TRALI) and transfusion-associated circulatory overload are much more prevalent in critically ill children.22
The aim of this adapted clinical practice guideline (CPG) is to provide evidence-based recommendations for the transfusion of blood components in the paediatric age group. These recommendations were adapted from the relevant CPGs using a formal methodology for CPG adaptation: the Adapted-ADAPTE.
➡️Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of transfusion of blood components in paediatric age groups.
It provides guidance to primary health care providers, pediatricians and specially trained nurses.
This guideline focuses on any disease or condition requiring red blood cells, platelets, and/or plasma products transfusion(s) to prevent or control anaemia, bleeding, and thrombotic disorders. It Provides an evidence-based document for the appropriate use of blood components, define indications, dosing, and administration of different blood components.
It also covers safe transfusion practice including pre-transfusion testing, handling of blood components, and close monitoring of transfused patients and includes recommendations in special situations (multi-transfused subjects, massive transfusion, and the critically ill).
This version of the guideline includes recommendations and good practice statements for the transfusion of blood components in paediatric age groups.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: Packed RBCS, Platelets, fresh frozen plasma, transfusion reaction
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) 23-26 by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria the GDG/ GAG recommended using 8 guidelines:
1. Guideline on the investigation and management of acute transfusion reactions Prepared by the BCSH Blood Transfusion Task Force. BSH 20121
2. Guidelines on red cell transfusion in sickle cell disease. BSH 2016a2,3
3. Guidelines for the use of platelet transfusions. BSH 2016b4
4. Recommendations on Red Blood Cell Transfusion in General Critically Ill Children Based on Heamoglobin and/or Physiologic Thresholds from the Paediatric Critical Care Transfusion and Anaemia Expertise Initiative. TAXI 20185
5. Clinical practice guidelines use of blood components in newborn. NNF 2020.6
6. Guidelines on the use of irradiated blood components. BSH 2020a7
7. Guidelines on Transfusion for Fetuses, Neonates and Older Children. BSH 2016 - addendum 2020b8,9
8. British Society of Haematology Guidelines on the spectrum of fresh frozen plasma and cryoprecipitate products: their handling and use in various patient groups in the absence of major bleeding. BSH 2018 – addendum 2020 10,11
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)27-30
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of pediatric hematology.
The main functions of the clinical panel were adolopment of transfusion of blood components in pediatrics Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as eminent international reviewers.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of
the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13].
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include awell-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing strategies for the transfusion of blood components in paediatric age groups into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:algorithms, pathways, tables, and parents’ educational health guide (in Arabic).
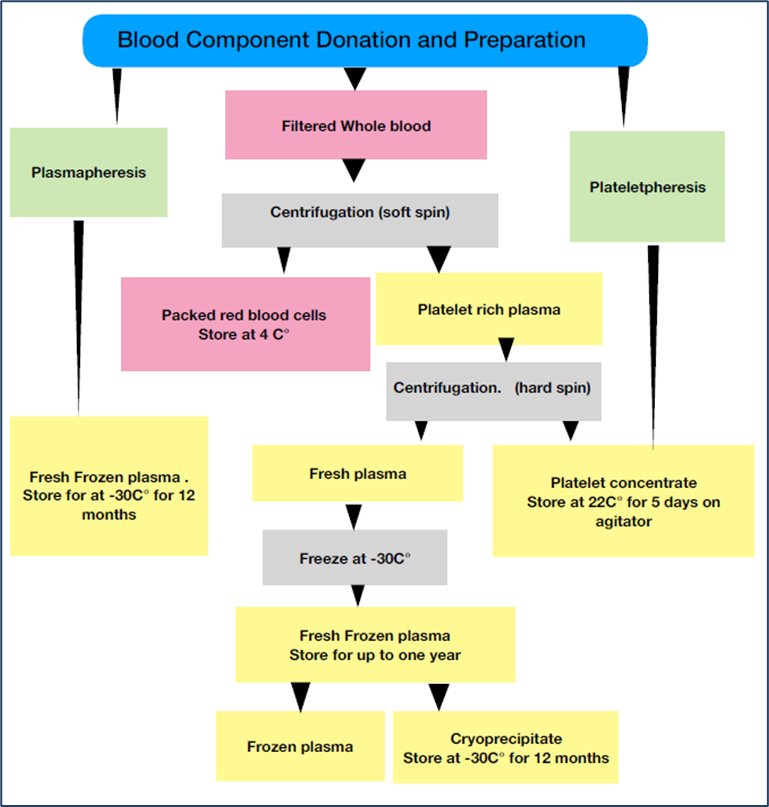
Figure S1: The process by which whole blood is used to produce blood components and plasma derivatives.
Table S1: General transfusion practices


|
|
Cut-off of platelet count |
|
||||
|
|
|
|
|
|
||
|
Less than 20 x103/µl |
|
20 x103 to < 30 x103/µl |
|
30 x103 to 50 x103/µl |
||
|
|
|
|
|
|
||
|
Mandatory transfusion
|
|
Transfusion as clinical situation require (e.g., active bleeding, minor invasive procedure) |
|
Conditional transfusion in the following: - Intracranial hemorrhage - Coagulopathy - Low birth weight < 1000 g - Neonatal sepsis - FNAIT* - Major invasive procedure - Unstable blood pressure |
||
*Washed irradiated platelets resuspended in ABO compatible plasma of maternal origin are used better. The transfused platelet should be lacking the causative antigen.
Figure S2: Indication for platelet transfusion in neonates.
Table S2: Indications of Prophylactic Platelet Transfusion in Pediatrics
|
Indication |
Platelet threshold (x103/µl) |
|
Prior To Procedures or Surgery* |
|
|
· Major surgery or invasive procedure, no active bleeding |
< 50 |
|
· Venous central lines (both tunneled and un-tunneled) ** |
<20 |
|
· Lumbar puncture |
< 40 |
|
· Insertion/removal of epidural catheter |
< 80 |
|
· Major surgery |
<50 |
|
· Neurosurgery or ophthalmic surgery involving the posterior segment of the eye |
<100 |
|
· Percutaneous liver biopsy*** |
<50 |
|
In critically ill child and reversible bone marrow failure |
<10 |
|
In critically ill child and reversible bone marrow failure with additional risk factors for bleeding**** |
<20 |
|
In Stable, non-bleeding child |
<10 |
* Whenever possible use a procedure/equipment associated with the lowest bleeding risk. Apply local measures, such as compression, to reduce the risk of bleeding post-procedure.
** Inserted by experienced staff using ultrasound guidance techniques.
***Consider trans-jugular biopsy if the platelet count is below this level.
****e.g., body temperature > 38°C or undergoing invasive procedure.
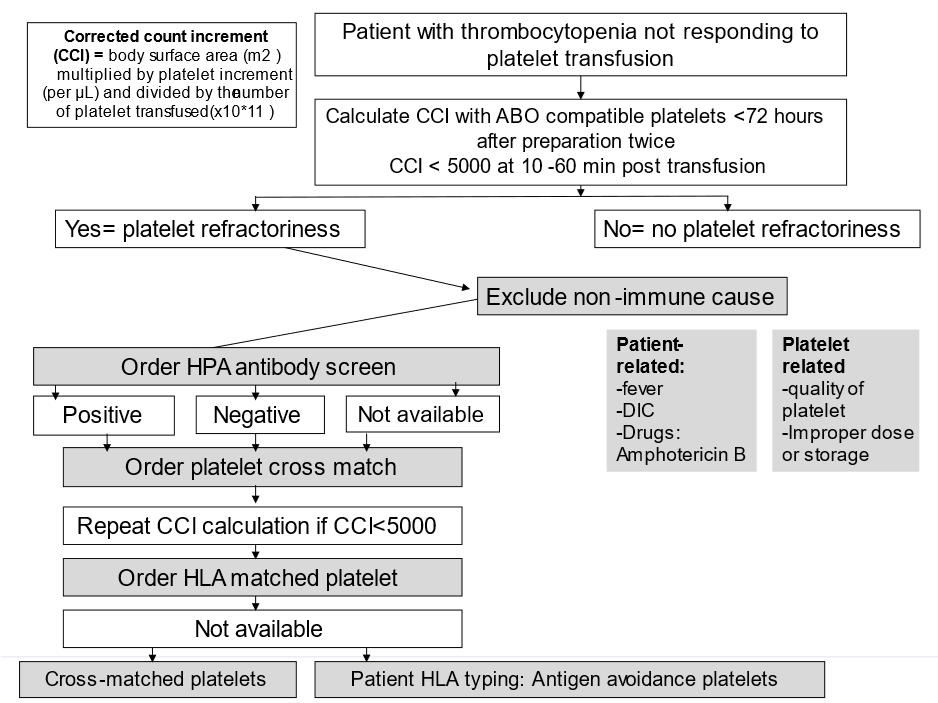
Figure S3: Approach to Platelet Refractoriness
HPA: human platelet antigen, HLA: human leukocyte antigen
Table S3: Indications of fresh frozen plasma transfusion
|
Indications |
Associated conditions |
|
Isolated Coagulation factors deficiency |
When specific therapy (recombinant or viral inactivated product) is not available |
|
Multiple coagulation factor deficiencies |
As DIC, vitamin k deficiency and liver failure If there is active bleeding in addition to treatment of the cause |
|
Anticoagulant |
· Reversal of warfarin (Coumadin)- with active bleeding or require emergency surgery. · Other anticoagulants: before surgery to prevent active bleeding or to treat active bleeding |
|
Microvascular bleeding |
When PT and PTT> 1.5 times normal |
|
Thrombotic thrombo-cytopenic purpura |
Therapeutic plasma exchange |
|
Protein C & protein S deficiency |
Associated with hypercoagulable state and no available concentrates |
Table S4: Indication and dose of cryoprecipitate in various coagulation disorders
|
Indications |
Dose of cryoprecipitate |
|
Fibrinogen Replacement
|
· Cryoprecipitate can be used for fibrinogen replacement in fibrinogen disorders (congenital afibrinogenemia or dysfibrinogenemia) · Dose: 1 unit of cryoprecipitate per 5 kg patient weight will increase fibrinogen by about 100 mg/dL · Number of bags = 0.2 x weight (kg) to provide about 100 mg/dL fibrinogen. |
|
Factor XIII Replacement
|
· 1 unit of cryoprecipitate per 5kg patient weight will provide 10 U/kg of factor XIII. · Number of bags =0.2 x weight (kg). · Factor XIII has a long half-life and can usually be dosed every 3-6 weeks. |
|
Factor VIII Replacement
|
· Dosing depends on patient factor VIII (8) level and requires routine monitoring of factor VIII (8) to determine appropriate dose. · Dosing should be repeated every 8-12 hours but will vary with each patient. · Post-surgery or major trauma replacement may be required for up to 10 days to maintain hemostasis. · Dosing also depends on Plasma Volume (PV) which is a fraction of Total Blood Volume (TBV). · Number of bags = [Desired activity (%) – Current activity (%)] x PV / 80
N.B: PV (mL) = TBV x (1-Hct), TBV (mL) = 70 mL/kg x weight (kg) |
|
Von Willebrand Factor Replacement
|
· Dosing of 1 unit per 10 kg patient weight will usually be enough to control bleeding. · Number of bags = 0.1 x weight (kg) · Repeat dosing may be required every 8-12 hours for up to 3 days followed by once daily dosing. · Follow clinically to adjust dosing and with appropriate lab studies available at your institution. |
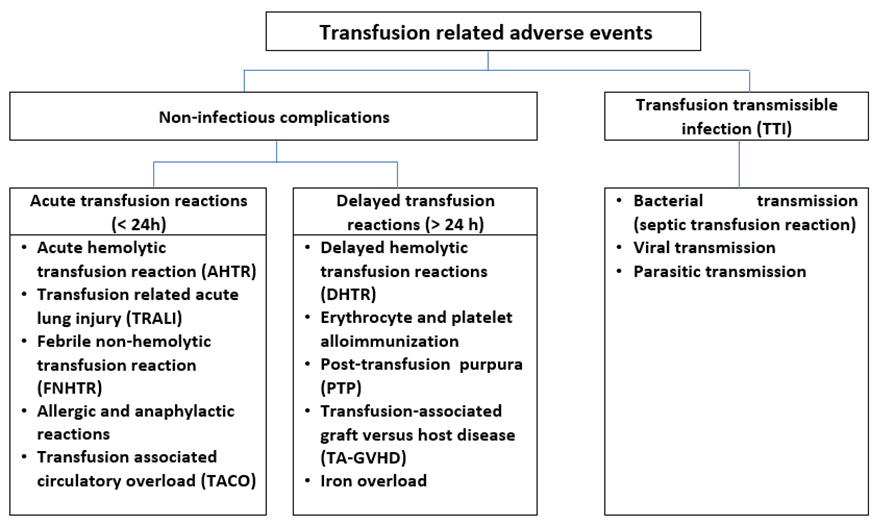
Figure S4: Different types of transfusion related adverse events.
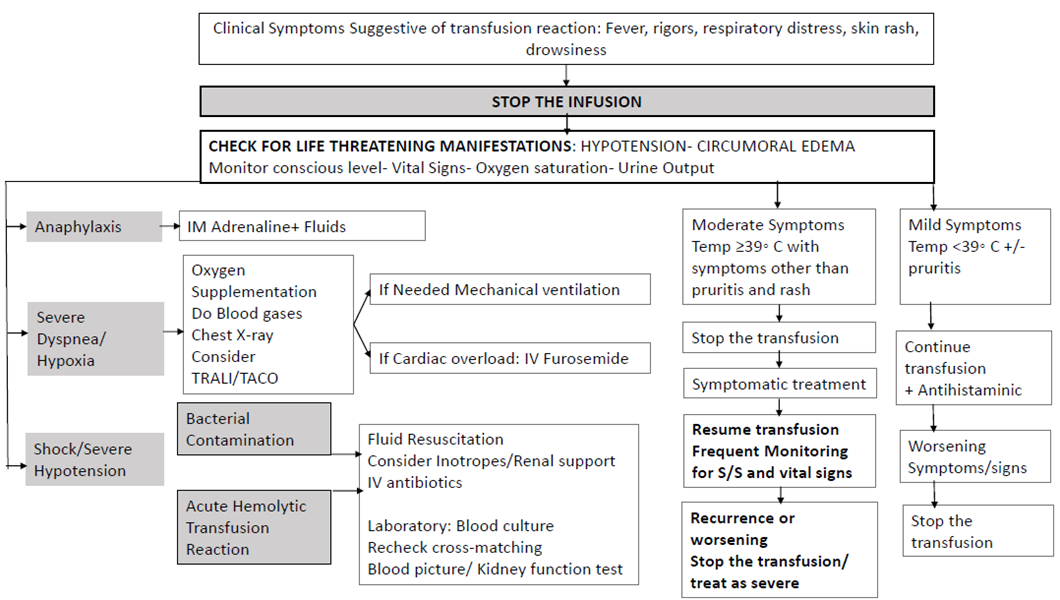
Figure S5: Management of acute transfusion reactions.
- Limitations and suggestions for further research needs
Future research recommendations for the management of transfusion of blood components in paediatric age groups in the Egyptian context could include:
▪️ Implementation of hemovigilance system at different institutional levels.
▪️ Blood components modification for specific populations.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for Blood products transfusion in children.
Challenges
▪️ Implementation of international guidelines in resource limited settings.
▪️ The optimum use of the available resources to fulfill the requirements of Egyptian population.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are two performance measures or indicators for implementing this adapted CPG for Blood products transfusion in children:
1. Adherence to Blood products transfusion Guidelines
▪️ Numerator: Number of children with transfusion who received treatment as per guideline recommendations.
▪️ Denominator: Total number of children who received transfusion.
▪️ Data Source: Hospital or clinic patient records.
2. Adherence to blood products transfusion reaction guidelines
▪️ Numerator: Number of children readmitted with symptoms of transfusion reaction within a certain period (e.g., 30 days) and managed according to the guidelines.
▪️ Denominator: Total number of children admitted with transfusion reactions.
▪️Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines.
➡️Updating of the guideline
The EPG Haematology GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Tinegate H, Birchall J, Gray A, et al. Guideline on the investigation and management of acute transfusion reactions. Prepared by the BCSH Blood Transfusion Task Force. British journal of haematology. Oct 2012;159(2):143-53. doi:10.1111/bjh.12017
2. Davis BA, Allard S, Qureshi A, et al. Guidelines on red cell transfusion in sickle cell disease. Part I: principles and laboratory aspects. 2017;176(2):179-191. doi:https://doi.org/10.1111/bjh.14346
3. Davis BA, Allard S, Qureshi A, et al. Guidelines on red cell transfusion in sickle cell disease Part II: indications for transfusion. 2017;176(2):192-209. doi:https://doi.org/10.1111/bjh.14383
4. Estcourt LJ, Birchall J, Allard S, et al. Guidelines for the use of platelet transfusions. British journal of haematology. Feb 2017;176(3):365-394. doi:10.1111/bjh.14423
5. Doctor A, Cholette JM, Remy KE, et al. Recommendations on RBC Transfusion in General Critically Ill Children Based on Hemoglobin and/or Physiologic Thresholds From the Pediatric Critical Care Transfusion and Anemia Expertise Initiative. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies. Sep 2018;19(9S Suppl 1):S98-s113. doi:10.1097/pcc.0000000000001590
6. Pandita A, Kumar A, Gupta G, Taligasalam V, Tewari VV. Use of Blood Components in Newborns. 2020;34(4):199-217. doi:10.1177/0973217921989856
7. Foukaneli T, Kerr P, Bolton-Maggs PHB, et al. Guidelines on the use of irradiated blood components. 2020;191(5):704-724. doi:https://doi.org/10.1111/bjh.17015
8. New HV, Berryman J, Bolton-Maggs PH, et al. Guidelines on transfusion for fetuses, neonates and older children. British journal of haematology. Dec 2016;175(5):784-828. doi:10.1111/bjh.14233
9. New HV, Stanworth SJ, Gottstein R, et al. British Society for Haematology Guidelines on transfusion for fetuses, neonates and older children (Br J Haematol. 2016;175:784–828). Addendum August 2020. 2020;191(5):725-727. doi:https://doi.org/10.1111/bjh.17109
10. Green L, Bolton-Maggs P, Beattie C, et al. British Society of Haematology Guidelines on the spectrum of fresh frozen plasma and cryoprecipitate products: their handling and use in various patient groups in the absence of major bleeding. 2018;181(1):54-67. doi:https://doi.org/10.1111/bjh.15167
11. Green L, Bolton-Maggs P, Beattie C, et al. British Society of Haematology Guidelines on the spectrum of fresh frozen plasma and cryoprecipitate products: their handling and use in various patient groups in the absence of major bleeding (Br J Haematol. 2018;181:54-67). Addendum August 2020. British journal of haematology. Dec 2020;191(5):728-729. doi:10.1111/bjh.17104
12. Valentine SL, Bembea MM, Muszynski JA, et al. Consensus Recommendations for RBC Transfusion Practice in Critically Ill Children From the Pediatric Critical Care Transfusion and Anemia Expertise Initiative. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies. Sep 2018;19(9):884-898. doi:10.1097/pcc.0000000000001613
13. Al-Saqladi AWM, TA A. A Study of Blood Transfusion in Pediatric Patients at a Teaching Hospital, Aden, Yemen. International Journal of Clinical Transfusion Medicine. 2021;9:1-9. doi:https://doi.org/10.2147/IJCTM.S293720
14. WHO. World Health Organization Model List of Essential Medicines. Vol. 22nd List. 2021.
15. Liumbruno GM, Bennardello F, Lattanzio A, Piccoli P, Rossetti G. Recommendations for the transfusion management of patients in the peri-operative period. II. The intra-operative period. Blood transfusion = Trasfusione del sangue. Apr 2011;9(2):189-217. doi:10.2450/2011.0075-10
16. Mo YD, Delaney M. Transfusion in Pediatric Patients: Review of Evidence-Based Guidelines. Clinics in laboratory medicine. Mar 2021;41(1):1-14. doi:10.1016/j.cll.2020.10.001
17. Sharma S, Sharma P, Tyler LN. Transfusion of blood and blood products: indications and complications. American family physician. Mar 15 2011;83(6):719-24.
18. Tinmouth AT, McIntyre LA, Fowler RA. Blood conservation strategies to reduce the need for red blood cell transfusion in critically ill patients. CMAJ : Canadian Medical Association journal = journal de l'Association medicale canadienne. Jan 1 2008;178(1):49-57. doi:10.1503/cmaj.071298
19. Khawar H, Kelley W, Stevens JB, Guzman N. Fresh Frozen Plasma (FFP). StatPearls. StatPearls Publishing
Copyright © 2022, StatPearls Publishing LLC.; 2022.
20. Nair PM, Rendo MJ, Reddoch-Cardenas KM, Burris JK, Meledeo MA, Cap AP. Recent advances in use of fresh frozen plasma, cryoprecipitate, immunoglobulins, and clotting factors for transfusion support in patients with hematologic disease. Seminars in hematology. Apr 2020;57(2):73-82. doi:10.1053/j.seminhematol.2020.07.006
21. Custer B, Zou S, Glynn SA, et al. Addressing gaps in international blood availability and transfusion safety in low- and middle-income countries: a NHLBI workshop. Transfusion. May 2018;58(5):1307-1317. doi:10.1111/trf.14598
22. Benson AB, Moss M, Silliman CC. Transfusion-related acute lung injury (TRALI): a clinical review with emphasis on the critically ill. British journal of haematology. Nov 2009;147(4):431-43. doi:10.1111/j.1365-2141.2009.07840.x
23. Brouwers MC, Kerkvliet K, Spithoff K. The AGREE Reporting Checklist: a tool to improve reporting of clinical practice guidelines. Bmj. Mar 8 2016;352:i1152. doi:10.1136/bmj.i1152
24. Brouwers MC, Kho ME, Browman GP, et al. AGREE II: advancing guideline development, reporting and evaluation in health care. J Clin Epidemiol. Dec 2010;63(12):1308-11. doi:10.1016/j.jclinepi.2010.07.001
25. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist. Ann Intern Med. May 2022;175(5):710-719. doi:10.7326/m21-4352
26. AGREE Tools. The AGREE Research Trust. 2024. www.agreetrust.org
27. Abdel Baky A, Omar TEI, Amer YS, the Egyptian Pediatric Clinical Practice Guidelines C. Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bulletin of the National Research Centre. 2023/06/13 2023;47(1):88. doi:10.1186/s42269-023-01059-0
28. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. East Mediterr Health J. Jul 31 2023;29(7):540-553. doi:10.26719/emhj.23.070
29. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. Journal of evaluation in clinical practice. Dec 2015;21(6):1095-106. doi:10.1111/jep.12479
30. Klugar M, Lotfi T, Darzi AJ, et al. GRADE guidance 39: using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. J Clin Epidemiol. Aug 6 2024;174:111494. doi:10.1016/j.jclinepi.2024.111494
31. Villeneuve A, Arsenault V, Lacroix J, Tucci M. Neonatal red blood cell transfusion. 2021;116(4):366-378. doi:https://doi.org/10.1111/vox.13036
32. Holcomb JB, Tilley BC, Baraniuk S, et al. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: the PROPPR randomized clinical trial. Jama. Feb 3 2015;313(5):471-82. doi:10.1001/jama.2015.12
33. Cappellini MD., Farmakis D., Porter J., A T. Guidelines for the Management of Transfusion Dependent Thalassaemia (4th edition – Version 2.0). 4th ed. Thalassaemia International Federation; 2021.
34. Yardumian A, Telfer P, Shah F, et al. Standards for the Clinical Care of Children and Adults with Thalassaemia in the UK. 3rd ed. 2016.
35. Bowden RA, Slichter SJ, Sayers M, et al. A comparison of filtered leukocyte-reduced and cytomegalovirus (CMV) seronegative blood products for the prevention of transfusion-associated CMV infection after marrow transplant. Blood. Nov 1 1995;86(9):3598-603.
36. Norfolk DR, McClelland DBL, United Kingdom Blood Transfusion Service. Handbook of transfusion medicine. Fifth edition. ed. Stationery Office; 2013:xiv, 170 pages.- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Galila Mohamed Mokhtar |
Paediatrics Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Amira Adly |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Dina Ezzat |
Professor of Paediatrics and Paediatric Haematology, Beni-Suef University, Egypt |
None |
Not Applicable |
|
Prof. Gehan Lotfy |
Professor of Paediatrics, Minia University, Egypt |
None |
Not Applicable |
|
Prof. Hoda Hassab |
Professor of Paediatrics and Paediatrics Haematology, Alexandria University, Egypt |
None |
Not Applicable |
|
Prof. Ilham Youssry |
Professor of Paediatrics and Head of the Paediatric Haematology & BMT unit, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Iman Ragab |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Laila Sherief |
Professor of Paediatrics and Paediatrics Haematology & Oncology, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Marwa Zakaria |
Associate Professor of Paediatrics, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Mervat Hesham |
Professor of Paediatrics, Paediatric Haematology & Oncology Unit, Zagazig University, Egypt |
None |
Not Applicable |
|
Dr. Naglaa Shaheen |
Consultant of Pediatric Hematology, Health Insurance Organization |
None |
Not Applicable |
|
Prof. Niveen Salama |
Associate Professor of Paediatrics, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Nouran Yousef |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Rasha Abdel-Raouf Afifi |
Professor of Paediatrics and Paediatric Haematology, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Rasha El-Ashry |
Professor of Paediatrics, Mansoura University, Egypt |
None |
Not Applicable |
|
Dr. Sara Makkeyah |
Associate Professor of Paediatrics, Paediatric Haematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Sonia Adolf |
Professor of Paediatrics and Paediatric Haematology, National Research Center, Cairo, Egypt |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanies Sulieman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Prof Magdy El-Ekiaby |
Consultant of blood transfusion, director of Shabrawishi Hospital Blood Bank, Vice President of Egyptian Society of Hematology (ESH) |
None |
Not Applicable |
|
Prof Salwa Youssef |
Professor of Haemtaolgy, consultant of Transfusion Medicine, Ain Shams University Egypt, President of the Egyptian society of transfusion medicine. |
None |
Not Applicable |
|
Prof Seham Ragab |
Professor of Paediatrics and Paediatric Haematolgy and Oncology, Head of Haematology and Oncology Unit, Paeditaric depertment, Menoufia University, Egypt. |
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
None |
Not Applicable |
|
International Peer Reviewers |
|
|
|
|
Yasser Wali |
Pediatric Hematology/Oncology Unit, Child Health Department, Sultan Qaboos University Hospital, Muscat, Oman |
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the blood products transfusion in children " on PubMed are: Packed RBCs, platelet transfusion, Fresh frozen plasma transfusion, transfusion reaction.
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for transfusion of blood components in pediatric age groups
|
CPGs AGREE II DOMAINS |
CPG1 |
CPG2 |
CPG3 |
CPG4 |
CPG5 |
CPG6 |
CPG7 |
CPG8 |
|
D1: Scope & Purpose |
88.8 |
77.8 |
90.7 |
83.3 |
88.9 |
94.4 |
87.7 |
83.3 |
|
D2: Stakeholder Involvement |
100 |
72.2 |
70.4 |
77.8 |
66.7 |
77.8 |
78.7 |
94.4 |
|
D3: Rigour of Development |
79.2 |
83.3 |
87.5 |
83.3 |
77 |
100 |
89.1 |
93.7 |
|
D4: Clarity & Presentation |
94.4 |
88.9 |
98.2 |
83.3 |
83.3 |
100 |
87.7 |
94.4 |
|
D5: Applicability |
87.5 |
62.5 |
83.3 |
66.7 |
76 |
75 |
81.6 |
87.5 |
|
D6: Editorial Independence |
100 |
100 |
100 |
83.3 |
83.3 |
100 |
81.6 |
91.7 |
|
OA 1 |
78.5 |
75.4 |
89.1 |
79.7 |
79.2 |
91.2 |
85.4 |
90.8 |
|
OA 2 (Recommend for use) |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
This table uses the AGREE II Domain Score Color codes (< 40% red; > 41 – 70% yellow; > 71 % green) |
||||||||
Annex Table 3. Annex Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist
|
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Practice guideline REgistration for transPAREncy (PREPARE) Guideline Protocol Registration Receipt
