Diagnosis and Treatment of Systemic Juvenile Idiopathic Arthritis (sJIA) in Pediatric Age Groups
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Diagnosis and Treatment of Systemic Juvenile Idiopathic Arthritis (sJIA) in Pediatric Age Groups |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 18 Feb 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Introduction
- - Methods
- - Evidence to recommendations: Considerations
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline.
- - Updating of the guideline
- - References
- - Annexes
- Executive Summary
Disease/Condition: Systemic Juvenile Idiopathic Arthritis (sJIA)
Guideline Objective(s)
1) Establish early diagnosis of sJIA
2) Clarify the role of investigations in disease assessment and follow up
3) Identify treatment modalities for controlling the disease activity as well as prevention and treatment of complications.
▪️Health / Clinical Question (PIPOH)
P: Patient (Target Population):
Children and Adolescents (Less than 18 years old)
I: Interventions and Practices Considered / CPG Category:
Diagnosis, and treatment and prevention of complications
P: Professionals (Intended / Target Users or Stakeholders) :
Pediatric Rheumatologists
General Pediatricians
Family Physicians
General Practioners
O: Major Outcomes Considered:
Primary (Specific) outcome: Early diagnosis of sJIA, proper control of disease activity, and prevention and treatment of complications
Secondary (general) outcome: Prevent delay in diagnosis, reduce inappropriate management and undue complications, improve patient outcome and quality of life, decrease hospitalization, and alleviate burden of drug side effects.
H: Healthcare Settings:
Governmental and private healthcare facilities (hospitals and clinics)
▪️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
- 2021 American College of Rheumatology Guideline for the Treatment of Juvenile Idiopathic Arthritis: Therapeutic Approaches for Oligoarthritis, Temporomandibular Joint Arthritis, and Systemic Juvenile Idiopathic Arthritis. Onel KB, et al. Arthritis Care Res (Hoboken). 2022 Apr;74(4):521-537. doi: 10.1002/acr.24853.1
- Treating juvenile idiopathic arthritis to target: recommendations of an international task force 2018. Ravelli A, et al. Ann Rheum Dis 2018;77:819–828. doi:10.1136/annrheumdis-2018-2130302
- Practice and consensus-based strategies in diagnosing and managing systemic juvenile idiopathic arthritis in Germany 2018 Hinze et al. Pediatric Rheumatology (2018) 16:7. doi:10.1186/s12969-018-0224-23
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
▪️Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. DIAGNOSIS OF JUVENILE IDIOPATHIC ARTHRITIS
B. INVESTIGATION REQUIRED AND POSSIBLE DIFFERENTIAL DIAGNOSIS
C. TREATEMENT RECOMMENDATIONS FOR JIA.
D. LONG TERM MONITORING OF PATIENTS.
We can summarize the guidelines’ recommendations in the following:
▪️ JIA comprises a group of inflammatory disorders that begin before the 18th birthday and persist for at least 6 weeks with other known conditions excluded (GPP)
▪️ Systemic JIA is grouped among JIA disorders: Fever of unknown origin (Excluding infectious, neoplastic, autoimmune, or monogenic autoinflammatory diseases) that is documented to be daily: quotidian fever that rises to ≥ 39°C once a day and returns to ≤ 37 °C between fever peaks for at least 3 consecutive days and reoccurring over a duration of at least 2 weeks plus 2 major criteria OR 1 major criterion and 2 minor criteria.
Major criteria include Evanescent (nonfixed)erythematous rash and Arthritis.
Minor criteria include Generalized lymph node enlargement and/or hepatomegaly and/or splenomegaly, Serositis, Arthralgia lasting 2 weeks or longer (in the absence of arthritis) and Leukocytosis (≥ 15,000/mm3) with neutrophilia. (GPP)
▪️ Once a child is suspected to have sJIA they should be referred to a pediatric rheumatologist ( GPP)
▪️ The demonstration of systemic inflammation, i.e., usually elevated C-reactive protein, erythrocyte sedimentation rate, leukocytes and/or ferritin) is essential for diagnosing sJIA at disease onset (Strong recommendation, High LOE)
▪️ Measurement of specific auto antibodies may be useful to rule out other conditions. (Weak recommendation, very low LOE)
▪️ Sonography and MRI are important modalities to assess joint manifestations, to differentiate from other conditions and to monitor disease activity (Weak recommendation, very low LOE)
▪️ Malignancies are important differential diagnoses for sJIA. If suspected, an extended panel of diagnostic tests, including chest radiography, ultrasound of the abdomen and pelvis, bone marrow aspiration, and, if appropriate, biopsy of lymph nodes or other involved organs should be pursued. An elevated LDH, uric acid and cytopenias represent pertinent findings. (Weak recommendation, very low LOE)
▪️ Infections are important differential diagnoses for sJIA. An adapted search for infections should be pursued as a case of fever of unknown origin (Weak recommendation, very low LOE)
▪️ Hereditary autoinflammatory syndromes are other important differential diagnoses for sJIA. Molecular genetic testing should be pursued if clinical suspicion for a known hereditary autoinflammatory syndrome exists. (Weak recommendation, very low LOE)
▪️ A febrile patient with known or suspected sJIA is classified as having MAS if the following criteria are met: Ferritin > 684 ng/ml and 2 of the following, PLT count ≤ 181x109/liter, AST> 48 U/liter, TGs > 156mg/dl, Fibrinogen ≤ 360 mg/dl. Abnormalities not otherwise explained by other patient condition, such as concomitant ITP, infectious hepatitis, visceral leishmaniasis, or familial hyperlipidemia (GPP)
▪️ High-dose systemic glucocorticoids are an effective and proven treatment for sJIA. (Strong recommendation, High LOE)
▪️ In the case of sJIA with arthritis, high-dose systemic glucocorticoids may be used, either as IV pulse therapy and/or as daily glucocorticoids with subsequent dose reduction. (Strong recommendation, Intermediate LOE)
▪️ Alternatively, IL-1 or IL-6 blockade may be applied, possibly in combination with glucocorticoids and/or methotrexate. (Strong recommendation, High LOE)
▪️ In case of insufficient treatment response, IV glucocorticoid pulse therapy may be repeated, or IL-1 or IL-6 blocking agents may be increased in dose (if feasible). In case of initial glucocorticoid therapy, IL-1 or IL- 6 blockade may be initiated. In case of initial biological monotherapy, glucocorticoids may be added (systemically or locally), the biological agent may be changed, or methotrexate may be added. (Strong recommendation, High LOE)
▪️ In case of a predominant polyarticular arthritis and in case of lack of treatment response despite the utilization of the approved biological agents, second-line agents, e.g., TNF blockers (etanercept or adalimumab) or abatacept may be applied. In addition, the use of methotrexate is reasonable, and intraarticular glucocorticoids may be applied (Strong recommendation, Intermediate LOE)
▪️ Tapering and discontinuing glucocorticoids is strongly recommended after inactive disease has been attained (Weak recommendation, very low LOE)
▪️ Tapering and discontinuing biologic DMARDS is conditionally recommended after inactive disease has been attained (Weak recommendation, very low LOE)
▪️ Glucocorticoids are conditionally recommended as part of the initial treatment of sJIA with MAS.IL-1 and IL-6 inhibitors are conditionally recommended over calcineurin inhibitors alone to achieve inactive disease and resolution of MAS (Weak recommendation, very low LOE)
▪️ Moderate doses of etoposide can be used in refractory cases of MAS. Intravenous immunoglobulins might be considered in refractory cases with variable success (GPP)
▪️ Disease activity should be assessed and documented regularly using a validated composite instrument. (JADAS score) (Weak recommendation, Intermediate LOE)
▪️sJIA with active systemic manifestations will require weekly assessment till resolution; monthly to every 3 months evaluations for patients who have high/moderate disease activity; and less frequent assessments, in states of persistent clinical remission (Weak recommendation, very low LOE)
▪️ The following interim targets are aimed for:
i. Resolution of fever within one week of the start of treatment (Strong recommendation, Intermediate LOE)
ii. Improvement of CRP by at least 50% within one week of the start of treatment (Strong recommendation, Intermediate LOE)
iii. Marked improvement of overall disease activity within four weeks of the start of treatment, i.e., Improvement of the physician global disease activity by at least 50%, reduction of actively inflamed joints (if present) by at least 50% and/or a JADAS10-Score of maximally 5.4 (Strong recommendation, Intermediate LOE)
iv.Clinically inactive disease is aimed for within six to twelve months (Weak recommendation, Intermediate LOE)
▪️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ( ). Link: http://www.guidelines-registry.org/- Recommendations
Recommendations for Diagnosis of sJIA (Quick references guide)
|
Health Question |
Diagnosis Recommendation |
SOR
|
LOE |
Source guidelines |
|
1- What is the general definition of JIA? |
JIA comprises a group of inflammatory disorders that begins before the 18th birthday and persists for at least 6 weeks wit other known conditions excluded. |
|
GPP*(9) |
|
|
2- What are the classification criteria for definite diagnosis of systemic JIA? |
-Systemic JIA is grouped among JIA disorders.
-Fever of unknown origin (Excluding infectious, neoplastic, autoimmune, or monogenic autoinflammatory diseases) that is documented to be daily: quotidian fever that rises to ≥ 39°C once a day and returns to ≤ 37 °C between fever peaks for at least 3 consecutive days and reoccurring over a duration of at least 2 weeks plus 2 major criteria OR 1 major criterion and 2 minor criteria.
Major criteria: 1- Evanescent (nonfixed)erythematous rash 2-Arthritis.
Minor criteria: 1-Generalized lymph node enlargement and/or hepatomegaly and/or splenomegaly 2-Serositis 3-Arthralgia lasting 2 weeks or longer (in the absence of arthritis) 4-Leukocytosis (≥ 15,000/mm3) with neutrophilia. |
|
GPP*(9) |
|
|
Once a child is suspected to have sJIA they should be referred to a pediatric rheumatologist. |
GPP |
|
|
|
|
4- What is the role of investigations in diagnosis of sJIA? |
- The demonstration of systemic inflammation, i.e., usually elevated C-reactive protein, erythrocyte sedimentation rate, leukocytes and/or ferritin) is essential for diagnosing sJIA at disease onset
-Measurement of specific autoantibodies may be useful in order to rule out other conditions.
-Sonography and MRI are important modalities to assess joint manifestations, to differentiate from other conditions and to monitor disease activity. |
Strong
Weak
Weak
|
High
Very low
Very low |
The German 2018 ³ Statement 2
Statement 3 |
|
5-What is the differential diagnosis of sJIA? |
-Malignancies are important differential diagnoses for sJIA. If suspected, an extended panel of diagnostic tests, including chest radiography, ultrasound of the abdomen and pelvis, bone marrow aspiration, and, if appropriate, biopsy of lymph nodes or other involved organs should be pursued. An elevated LDH, uric acid and cytopenias represent pertinent findings.
-Infections are important differential diagnoses for sJIA. An adapted search for infections should be pursued as a case of fever of unknown origin
-Hereditary autoinflammatory syndromes are other important differential diagnoses for sJIA. Molecular genetic testing should be pursued if clinical suspicion for a known hereditary autoinflammatory syndrome exists. |
Weak
Weak
Weak |
Very low
Very low
Very low |
The German 2018 ³ Statement 3
|
|
6-What are the classification criteria for diagnosis of macrophage activation syndrome (MAS) in patients with sJIA? |
A febrile patient with known or suspected sJIA is classified as having MAS if the following criteria are met: Ferritin > 684 ng/ml and 2 of the following · PLT count ≤ 181x109/liter · AST> 48 U/liter · TGs > 156mg/dl · Fibrinogen ≤ 360 mg/dl Abnormalities not otherwise explained by other patient condition, such as concomitant ITP, infectious hepatitis, visceral leishmaniasis, or familial hyperlipidemia. |
|
GPP**(29) |
|
* Martini A, Ravelli A, Avcin T, Beresford MW, Burgos-Vargas R, Cuttica R, et al. Toward New Classification Criteria for Juvenile Idiopathic Arthritis: First Steps, Pediatric Rheumatology International Trials Organization International Consensus. J Rheumatol 2019; 46(2):190-7. 9
**Ravelli A, Minoia F, Dav S, Horne A,Bovis F, Pistorio A, et al. 2016 Classification criteria for macrophage activation syndrome complicating systemic juvenile idiopathic arthritis. A European League Against Rheumatism/American College of Rheumatology/Paediatric heumatology International Trials Organisation Collaborative Initiative.Arthritis Rheumatol 2016 Mar;68(3):566-76.29
GPP: Good Practice Point (Consensus by the sJIA Guidelines Adaptation Group)
Recommendations For Treatment of sJIA (Quick references guide)
|
Health Questions |
Treatment Recommendations |
GOR |
LOE |
Adapted Guidelines |
|
1-What are the treatment strategies for sJIA without MAS? |
A) Glucocorticoids: High-dose systemic glucocorticoids are an effective and proven treatment for sJIA. B) In the case of sJIA with arthritis, high-dose systemic glucocorticoids may be used, either as IV pulse therapy and/or as daily glucocorticoids with subsequent dose reduction. C) Alternatively, IL-1 or IL-6 blockade may be applied, possibly in combination with glucocorticoids and/or methotrexate. D) In case of insufficient treatment response (see treatment targets in (3C), IV glucocorticoid pulse therapy may be repeated, or IL-1 or IL-6 blocking agents may be increased in dose (if feasible). In case of initial glucocorticoid therapy, IL-1 or IL- 6 blockade may be initiated. In case of initial biological monotherapy, glucocorticoids may be added (systemically or locally), the biological agent may be changed, or methotrexate may be added. E) In case of a predominant polyarticular arthritis and in case of lack of treatment response despite the utilization of the approved biological agents, second-line agents, e.g., TNF blockers (etanercept or adalimumab) or abatacept may be applied. In addition, the use of methotrexate is reasonable and intraarticular glucocorticoids may be applied
F) Tapering and discontinuing glucocorticoids is strongly recommended after inactive disease has been attained
G) Tapering and discontinuing biologic DMARDS is conditionally recommended after inactive disease has been attained |
Strong
Strong
Strong
Strong
Strong
Weak
Weak |
High
Intermediate
High
High
Intermediate
Very low
Very low |
The German 2018³ Statement 5 Statement 7
ACR 2021 ˡ
|
|
2- What are the treatment strategies for sJIA with MAS? |
(A) Glucocorticoids are conditionally recommended as part of initial treatment of sJIA with MAS. (B) IL-1 and IL-6 inhibitors are conditionally recommended over calcineurin inhibitors alone to achieve inactive disease and resolution of MAS
(C) Moderate doses of etoposide can be used in refractory cases of MAS
(D) Intravenous immunoglobulins might be considered in refractory cases with variable success |
Weak
Weak |
Very Low
Very Low
GPP***(46)
GPP**** (47) |
ACR 2021 ˡ
ACR 2021 ˡ
|
|
3- When and how to monitor the patient with sJIA while on treatment?
|
(A) Disease activity should be assessed and documented regularly using a validated composite instrument. (JADAS score (27) (B) sJIA with active systemic manifestations will require weekly assessment till resolution; monthly to every 3 months evaluations for patients who have high/moderate disease activity; and less frequent assessments, in states of persistent clinical remission. (C) The following interim targets are aimed for: i. Resolution of fever within one week of the start of treatment ii. Improvement of CRP by at least 50% within one week of the start of treatment iii. Marked improvement of overall disease activity within four weeks of the start of treatment, i.e., Improvement of the physician global disease activity by at least 50%, reduction of actively inflamed joints (if present) by at least 50% and/or a JADAS10-Score of maximally 5.4 iv.Clinically inactive disease is aimed for within six to twelve months v. Once the treatment target has been achieved, it should be sustained. Ongoing monitoring should occur to ensure maintenance of the target. |
Weak
Weak
Strong
Strong Weak |
Intermediate
Very low
Intermediate
Intermediate
Intermediate |
Treat to Target2018 ²
The German 2018³ Statement 4
Treat to target 2018² |
Tibaldi J, Pistorio A, Aldera E, Puzone L, El Miedany Y, Pal P, et al. Development and initial validation of a composite disease activity score for systemic juvenile idiopathic arthritis. Rheumatology (Oxford). 2020;59(11):3505-14.27
*** Boom V, Anton J, Lahdenne P, Quartier P, Ravelli A, Wulffraat NM, et al. Evidence-based diagnosis and treatment of macrophage activation syndrome in systemic juvenile idiopathic arthritis. Pediatr Rheumatol 13, 55 (2015). 46
**** Ambler WG, Nanda K, Onel KB, Shenoi S. Refractory systemic onset juvenile idiopathic arthritis: current challenges and future perspectives. Ann Med. 2022 Dec;54(1):1839-1850.47
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|
|||
|
Dr Ashraf Abdel Baky |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children's Hospital, Faculty of Medicine, Ain Shams University. Chair of EPG/GAG |
Editor, Clinical expert, GAG member |
|
|||
|
Dr Dalia H. El-Ghoneimy |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Faculty of Medicine, Ain Shams University. |
Clinical expert, GAG member |
|
|||
|
Dr Mohamed Almalky |
Professor of Pediatrics, Faculty of Medicine, Zagazig University. |
Clinical expert, GAG member |
|
|||
|
Dr Shereen Esam Maher |
Professor of Pediatrics, Pediatric Rheumatology, Children's Hospital, Faculty of Medicine, Minia University. |
Clinical expert, GAG member |
|
|||
|
Dr Ali Sobh |
Associate Professor of Pediatrics, Mansoura University Children’s Hospital, Faculty of Medicine, Mansoura University. |
Clinical expert, GAG member |
|
|||
|
Dr Iman Fahmy |
Associate Professor of Pediatics, Pediatric Allergy, Immunology and Rheumatology Unit, Children's Hospital, Faculty of Medicine, Suhag University |
Clinical expert, GAG member |
|
|||
|
Dr Nesrine Radwan
|
Associate Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children's Hospital. Faculty of Medicine, Ain Shams University. |
Clinical expert, GAG member |
|
|||
|
Dr Rasha El-Owaidy |
Associate Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children's Hospital. Faculty of Medicine, Ain Shams University. |
Clinical expert, GAG member |
|
|||
|
Dr Yomna Mohamed Farag |
Associate Professor of Pediatrics, Pediatric Rheumatology Unit, Specialized Children’s Hospital, Faculty of Medicine, Cairo University. |
Clinical expert, GAG member |
|
|||
|
Dr Amira Hattab |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital. Faculty of Medicine, Ain Shams University. |
Clinical expert, GAG member |
|
|||
|
Dr Ghada Abdel Haleem Shousha |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital. Faculty of Medicine, Ain Shams University. |
Clinical expert, GAG member |
|
|||
|
Dr. Sally Gouda |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital. Faculty of Medicine, Ain Shams University. |
Clinical expert, GAG member |
|
|||
|
Dr Naglaa Samy Mohamed Osman |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology Unit, Children's Hospital, Faculty of Medicine, Assiut University. |
Clinical expert, GAG member |
|
|||
|
Dr Walaa Shoman |
Lecturer of Pediatrics, Children's Hospital. Faculty of Medicine, Alexandria University. |
Clinical expert, GAG member |
|
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
||||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
||||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
||||
|
Dr Tarek Omar |
Prof. of Pediatrics, Alexandria University |
|
||||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
||||
|
Associate Professor of Pediatrics Ain Shams University, Egypt |
Developing evidence to decision (EtD) frameworks, participating in search and guideline appraisal |
|||||
|
- Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. - Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. - Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||||
|
Dr. Lamis Mohsen Elsholkamy
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
Dr. Ahmed Mahmoud Youssef
|
Fellow of Pediatrics, General Organization For Teaching Hospitals and Institutes (GOTHI). Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
External Reviewers Group (ERG) |
||||||
|
External Reviewer(s) for Clinical Content |
||||||
|
||||||
|
Prof. Zeinab Awad El-Sayed |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Ain Shams University, Egypt. |
|||||
|
Prof. Angelo Ravelli |
Professor of Pediatrics, department of Neurosciences, Rehabilitation, Opthalomology, Genetics and Maternal-Infantile Science (DiNOGMI), University of Geno, Italy. |
|||||
|
Dr. Karen Onel |
Chief, Division of Pediatric Rheumatology Hospital for special Surgery. Professor of clinical Pediatrics Weil Cornell Medicine |
|||||
|
Associate Prof. Soad Hashad |
Associate Professor, Pediatric Rheumatology, Tripoli University, Libya. |
|||||
|
Associate Prof. Ghada Al-Deriny |
Associate Prof. Of Pediatrics, Alexandria University, Egypt |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
|||||
▪️ The Egyptian Pediatric Clinical Practice Guideline Committee (EPG) would like to present its highest gratitude and acknowledgment to the members of the clinical practice guideline (CPG) adaptation group (GAG) and the external review group. We acknowledge the collaborative support of the Supreme Council for Egyptian University Hospitals during the adaptation of this evidence-based CPG. We would also like to acknowledge the copyright holder of the source guidelines that were adapted in this work for their generous approval and encouragement. Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️ This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
- Abbreviations
|
AAP |
American Academy of Pediatrics |
|
ACR |
American College of Rheumatology |
|
AOSD |
Adult-onset Still’s disease |
|
CD |
Cluster of Differentiation |
|
CPG |
Clinical Practice Guideline |
|
CPS |
Canadian Pediatric Society |
|
DMARDs |
Disease-modifying antirheumatic drugs |
|
cDMARDs |
Conventional Disease-modifying antirheumatic drugs |
|
EPG |
Egyptian Pediatric Clinical Practice Guidelines Committee |
|
EULAR |
European League against Rheumatism |
|
GAG |
Guideline Adaptation Group |
|
GIN |
Guidelines International Network |
|
GKJR |
German Society for Pediatric Rheumatology |
|
GRADE |
Grading of Recommendations, Assessment, Development and Evaluation |
|
ILAR |
International League of Associations for Rheumatology |
|
IVIG |
Intravenous Immunoglobulin |
|
JADAS |
Juvenile Arthritis Disease Activity Score |
|
JAK |
Janus kinase |
|
MAS |
Macrophage activation syndrome |
|
MOH |
Ministry of Health |
|
MRI |
Magnetic Resonance Imaging |
|
mSMS |
Modified systemic manifestation score |
|
NICE |
National Institute for Health and Care Excellence |
|
NK |
Natural Killer |
|
PET-CT |
Positron Emission Tomography |
|
PRINTO |
Pediatric Rheumatology International Trials Organization |
|
QoE |
Quality of Evidence |
|
SIGN |
The Scottish Intercollegiate Guidelines Network |
|
sJIA |
Systemic Juvenile Idiopathic Arthritis |
|
SoR |
Strength of Recommendations |
- Introduction
Juvenile idiopathic arthritis (JIA) is the most
common chronic rheumatic disease in children (Ravelli and Martini, 2007).
Systemic juvenile idiopathic arthritis (sJIA) is a very distinctive subtype of
JIA with unique clinical manifestations, associated complications, therapeutic
options, and prognosis (Lee and Schneider 2018). It differs from the other
subtypes of JIA in being an autoinflammatory phenotype, presents usually with
fever, rash, lymphadenopathy and marked systemic inflammation (Martini et al.,
2019).
➡️ Epidemiology:
Mean annual incidence and prevalence rates of JIA in
general, and sJIA in particular differ among countries due to combined
immunogenic and environmental factors. In a systematic review conducted by
Thierry and colleagues to evaluate the incidence and prevalence of JIA in
Europe, the incidence estimates varied from 1.6-23/10⁵ and the prevalence
estimates from 3.8-400/10⁵ (Thierry et al., 2014). In Spain, the incidence rate
of JIA was 6.9/10⁵ and the prevalence was 39.7 (36.1-43.7)/10⁵ in children aged
less than 16 years (Modesto et al., 2010). In Egypt, the prevalence rate of JIA
in Sharkia Governate was 3.43/10⁵ in children younger than 16 years and sJIA
constituted 13.6% of the patients (Abou El-Soud et al., 2013).
Systemic JIA can present at any time throughout
childhood and adolescence prior to the age of 16 years, with peak incidence at
2 years There is no gender nor ethnic predilection, with a higher prevalence
rate reported in southeast Asia and Japan (Consolaro et al., 2016). Although it
represents 10 to 20% of all JIA subtypes, sJIA accounts for up to two-thirds of
the mortalities related to JIA (Salah et al., 2009; Martini et al., 2019). sJIA
accounts for 23%-24% of all forms of JIA in Egypt (Hussien et al., 2018), and
in Saudi Arabia, sJIA was the most commonly reported subtype of JIA (Bahabri et
al., 1997). sJIA forms 10% of JIA subtypes in North America and up to 50% in
Asian countries (Fujikawa et al., 1997).
➡️ sJIA immunopathogenesis:
The term sJIA
was preferred, even though arthritis might be absent in some patients.
The Pediatric Rheumatology International Trials Organization (PRINTO)
International Consensus has agreed to keep sJIA among the JIA disorders rather
than the autoinflammatory disorders (Martini et al., 2019).
Similarities between sJIA and adult onset Still’s
disease (AOSD) are both clinical and biological, including the occurrence of
the life-threatening condition known as macrophage activation syndrome (MAS)
and the associated marked activation of interleukin (IL) 1 and IL-6 as a part
of the immunopathogenesis of the diseases. The IL-1 family of cytokines
includes 11 cytokines; of these, IL-1β represents the most potent member and a
therapeutic target in sJIA (Palomo et al., 2015; Toplak et al., 2018).
Therefore, a striking and sustained response to treatment with IL-1β inhibitors
was noticed in sJIA and ASOD (Martini, 2012; Castanda et al., 2016).
Systemic JIA progression often follows a biphasic
course. In the beginning, there is hyperstimulation of the innate immune
response with excessive secretion of IL-1β as a key cytokine. In the second
phase, a dominant adaptive immune response is activated with over production of
IL-17A, explaining the following chronic arthritis which becomes the leading
clinical feature (Kessel et al., 2017). Accordingly, blocking IL-1 is a
rational therapeutic choice in the initial phase of sJIA, but it might need to
be substituted with other biologics when the condition progresses to persistent
polyarthritis (Toplak et al., 2018).
➡️ Diagnosis of sJIA:
Diagnostic criteria of sJIA were defined by the
International League of Associations for Rheumatology (ILAR) (Petty et al.,
2004). Validation of ILAR definition of
sJIA has faced many challenges because it considered the presence of chronic
arthritis a mandatory criterion for the diagnosis, which led to missing up a
sizeable proportion of the patients (Martini et al., 2019; Hinze et al., 2018).
sJIA was recently defined by PRINTO’s new classification criteria for sJIA as
being a fever of unknown origin, after excluding other etiologies such as
infections, malignancies or monogenic autoinflammatory disorders. Fever should
be documented daily in a quotidian pattern (fever that rises to ≥ 39°C once a
day and returns to normal in between) for at least 3 consecutive days and
recurring over a duration of at least 2 weeks. To fulfill the definition, fever
should be accompanied by 2 major criteria or 1 major and 2 minor criteria. The
major ones are (1) presence of arthritis, and (2) evanescent erythematous rash.
Minor criteria include: (1) generalized lymphadenopathy, hepatomegaly, and/or
splenomegaly, (2) serositis, (3) at least 2 weeks of arthralgia, provided that
there is no associated arthritis, and (4) leukocytosis, mainly out of
neutrophilia (Martini et al., 2019). The German consensus has proposed the term
probable sJIA to refer to the patients lacking chronic arthritis (Hinze et al.,
2018).
Disease progression follows one of three courses: a
monophasic course with a single episode of fever, or a polycyclic course, which
is fluctuating between remissions and flares (Singh-Grewal et al., 2006) and
finally the third type of course which is the "chronic persistent".
Noticeably, systemic manifestations of the disease tend to respond earlier and
easier than the articular manifestations, which can become refractory to
multiple therapeutic approaches (Beukelman et al., 2014).
The role of laboratory and radiological
investigations for confirming or excluding the diagnosis of sJIA is debatable.
Given that ILAR PRINTO diagnostic criteria did not include any investigations
except for the leukocytic count, many authors recommend performing other
investigational tests to exclude the differential diagnoses and to properly
detect any associated complications, mainly MAS (Hinze et al., 2018).
Generally, sJIA differs from all other JIA types in the associated elevated
inflammatory markers such as C-reactive protein (CRP), erythrocyte
sedimentation rate (ESR), platelets, and the reduced levels of hemoglobin and
serum albumin (Cimaz et al., 2016). Antinuclear antibodies and rheumatoid
factor are typically negative in sJIA
➡️ Assessment of sJIA activity:
Regular assessment of sJIA systemic and articular
activities is mandatory to determine the disease progression and response to
treatment. The Juvenile Arthritis Disease Activity Score (JADAS) has been
accepted as a tool of JIA activity evaluation, but not specifically for sJIA
(Consolaro et al., 2016). An updated JADAS specific for sJIA (sJADAS) has been
recently released (table 7). The score is composed of 5 main items: 1)
Physician global assessment of the overall disease activity (a scale from 0 to
10), 2) Patient global assessment of the well-being (a scale from 0 to 10), 3)
number of active joints, 4) CRP or ESR levels, and 5) the modified systemic
manifestation score (mSMS), including presence of fever, rash, lymphadenopathy,
hepatomegaly, splenomegaly, serositis, anemia or thrombocytopenia (Tibaldi et
al., 2020).
➡️ Complications of sJIA:
Complications of sJIA include MAS, growth
retardation, damage from severe erosive arthritis, osteoporosis, cardiovascular
events such as pericarditis, pulmonary hypertension and amyloidosis (Woerner et
al, 2015).
Macrophage activation syndrome (MAS): MAS is a
potentially life-threatening condition that was described in association with,
or as a complication of systemic inflammatory disorders such as sJIA (Ravelli
et al., 2016; Çakan et al., 2020). It was reported in 10% of sJIA patients,
either initially or along the course of the disease. However, subclinical MAS
may occur in up to 30-40% of sJIA cases (Behrens et al., 2007). MAS is
characterized by an overwhelming inflammatory reaction due to sustained
dysregulated and dysfunctional immune response involving expansion of T
lymphocytes and macrophages, leading to exaggerated production of
proinflammatory cytokines (Ravelli et al., 2012). MAS is to be suspected when
patients develop high, persistent fever, generalized lymphadenopathy,
hepatosplenomegaly, central nervous system (CNS) dysfunction, and hemorrhagic diathesis.
High levels of ferritin, low fibrinogen and changes in the hematological
parameters towards cytopenia, should raise the suspicion of MAS in any sJIA
patient (Çakan et al., 2020).
Growth retardation: Growth retardation usually
complicates long-standing, refractory sJIA patients. Indeed, impaired growth
might be the result of combined factors such as altered nutritional status,
physical restrictions, emotional impacts, and prolonged use of systemic
glucocorticoids, , in addition to the chronic inflammatory status itself (De
Benedetti et al., 2015).
➡️Differential Diagnosis of sJIA:
Diagnosis of sJIA is basically depending on excluding
the other possible diseases that may mimic the symptoms, most importantly
infections, malignancy or hereditary autoinflammatory diseases (Hinze et al.,
2018). Abdominal sonography, chest imaging and articular sonography as well as
laboratory investigations involving peripheral blood smear, blood cultures, urine
analysis, serum S100 proteins, serum procalcitonin and bone marrow aspirate as
indicated will help to establish specific diagnosis (Hinze et al., 2018).
Furthermore, patients who initially present with MAS need to be differentiated
from the other possible etiologies of primary and secondary hemophagocytic
lymphohistiocytosis (HLH).
➡️ Broad lines of sJIA Treatment:
The American College of Rheumatology (ACR) has
promulgated treatment recommendations for patients with sJIA in 2011 and
updated them in 2013 (Beukelman et al., 2011; Ringold et al., 2013) and lastly
in 2021 (Onel et al 2022). The German consensus has settled their
recommendations based on a treat-to-target approach (Hinze et al., 2018). The
course of the disease might follow a pattern of relapses followed by intervals
of remission, or an unremitting course with persistent arthritis (Martini et
al., 2019). Nevertheless, patients with sJIA do not often respond to treatment
with conventional disease-modifying antirheumatic drugs (cDMARDs). Glucocorticoids
are mandatory to control the disease relapses, and low-doses of glucocorticoids
might be needed to keep the patients in the remission status. However,
prolonged glucocorticoid regimens are associated with significant adverse
effects such as osteoporosis, short stature, cataract, glaucoma, hypertension
and diabetes mellitus type 2. The era of biologics has rescued many patients
from the side effects of prolonged courses of glucocorticoids and from the poor
response in some refractory cases (Correll et al., 2014). Although biologics
targeting IL-1 and IL-6 are the most recommended lines of therapy for sJIA (
Vastert et al., 2014; DeWitt et al., 2012; Onel et al 2022) the long-term
commitment to frequent injections or infusions remains a challenge in young
children (Yokota et al., 2015).
Refractory, steroid-dependent sJIA patients are
treated individually based on the patients’ circumstances and the expert
preferences. In patients refractory to IL-1 and IL-6 blocking agents with
steroid dependency, other treatments can be considered including adding cDMARD
for refractory arthritis and other biological such as abatacept, and the
limited trials of JAK inhibtors and anti-IL17 and anti-IL18 agents. The latter
may be used in severe sJIA through compassionate investigational new drug use.
( Record et al.,2011; Canny et al., 2017; Canna .,2017)
➡️Prognosis:
sJIA accounts for the highest mortality rates among
the other subtypes of JIA, given its associated complications (Huang et al.,
2019).
Early predictors of articular damage and poor
prognosis include young age at the disease onset (<18 months of age), longer
disease duration, prolonged use of glucocorticoids, persistent or recurrent
thrombocytosis, and high inflammatory parameters (Sandborg et al., 2006; Russo et
al., 2013).
Purpose and scope
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: Juvenile Idiopathic Arthritis guidelines, JIA, Systemic Juvenile Idiopathic Arthritis guidelines, sJIA classification, sJIA Guidelines
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric Rheumatology)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) 23-26 by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 3 guidelines:
1.2021 American College of Rheumatology Guideline for the Treatment of Juvenile Idiopathic Arthritis: Therapeutic Approaches for Oligoarthritis, Temporomandibular Joint Arthritis, and Systemic Juvenile Idiopathic Arthritis. Onel KB, et al. Arthritis Care Res (Hoboken). 2022 Apr;74(4):521-537. doi: 10.1002/acr.24853.1
2. Treating juvenile idiopathic arthritis to target: recommendations of an international task force 2018. Ravelli A, et al. Ann Rheum Dis 2018;77:819–828. doi:10.1136/annrheumdis-2018-2130302
3. Practice and consensus-based strategies in diagnosing and managing systemic juvenile idiopathic arthritis in Germany 2018 Hinze et al. Pediatric Rheumatology (2018) 16:7. doi:10.1186/s12969-018-0224-23
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)27-30
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of pediatric Rheumatology.
The main functions of the clinical panel were adolopment of Jeuvenile idiopathic arthritis in pediatrics Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 5 clinical experts who have interest and expertise in as well as eminent international reviewers.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions .
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with the emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is necessary about actual healthcare practice
2- Have large net positive consequences (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing strategies for the transfusion of blood components in pediatric age groups into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on paper or electronically to remind a patient/consumer to perform a particular health-related behavior.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or training courses, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including algorithms, pathways, tables, and parents’ educational health guide (in Arabic).
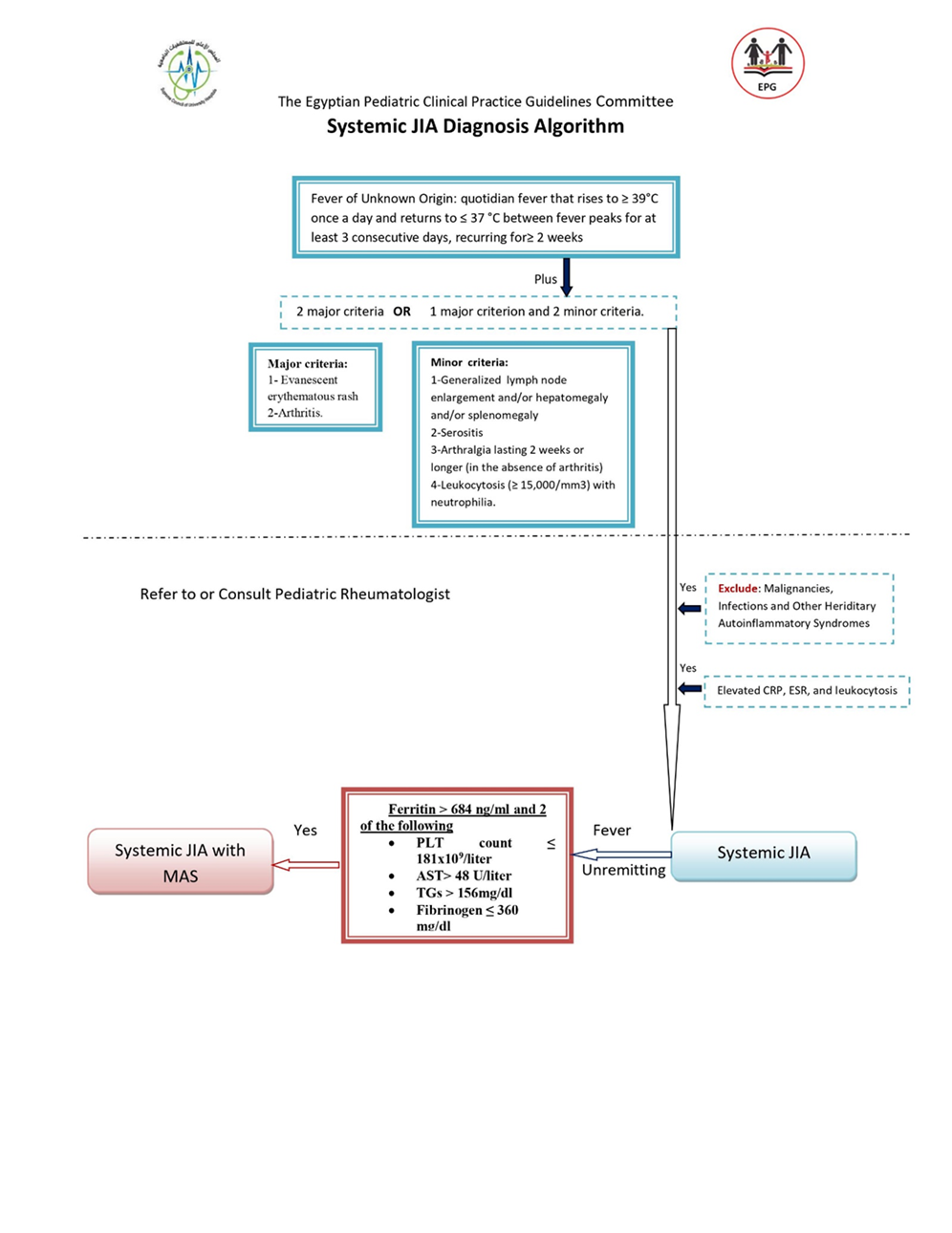
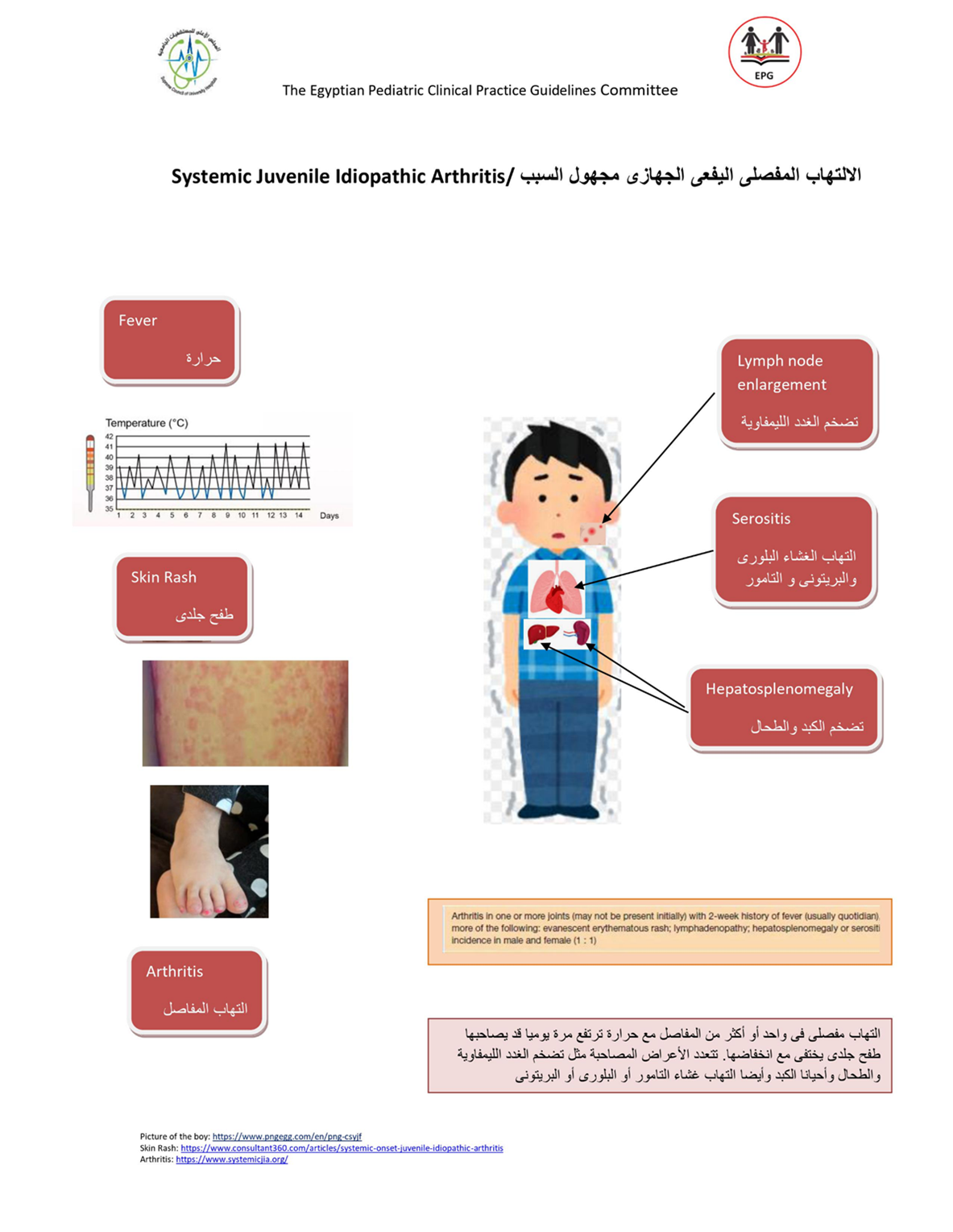
Table (7) : sJADAS10 composite score for assessment of sJIA activity
|
|
sJADAS 10 |
|
Physician global assessment |
0-10 VAS |
|
Parent/patient global assessment |
0-10 VAS |
|
Active joints count |
Simple 0-10 joints a |
|
Acute phase reactants (range) |
Normalized ESR b or CRP (0-10) c |
|
Modified systemic manifestations score (range) |
0-10 |
|
Total score range |
0-50 |
a : Up to 10 joints, irrespective of their type, censored at 10. b: According to the formula: (value in mm/h – 20)/10, where values 120 mm/h are converted to 120. c :According to the formula (value in mg/l – 10)/10, where values 110 mg/l are converted to 110. sJADAS 10: systemic Juvenile Arthritis Disease Activity Score in 10 joints (Adapted from Tibaldi et al., 2020)
d: Modified systemic manifestations score:
|
Clinical or laboratory feature |
Points |
|
Fever * 37.5–38⁰C >38–39⁰C >39–40⁰C >40⁰C |
1 2 3 4 |
|
Evanescent erythematous rash |
1 |
|
Generalized lymphadenopathy (enlargement of >3 lymph node stations) |
1 |
|
Hepatomegaly and/or splenomegaly |
1 |
|
Serositis (pleuritis, pericarditis or peritonitis) |
1 |
|
Anaemia (haemoglobin <9 gm/dl) |
1 |
|
Platelet count >600 109 /L or ferritin >500 ng/ml |
1 |
*:Fever is defined as the maximum temperature either in the past 24 h, 3 days or week. In the assessment of fever, the possible pharmacologic suppression of temperature by paracetamol, NSAIDs or glucocorticoids should be taken into
account. 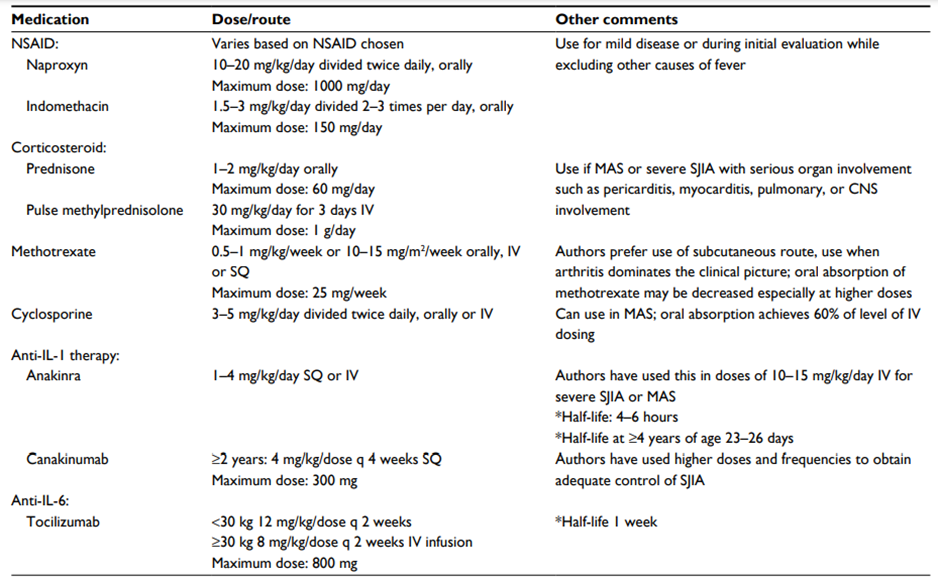
Table (8) Therapeutic agents commonly used for sJIA treatment
Abbreviations: NSAID, nonsteroidal anti-inflammatory drug; SQ, subcutaneous; IV, intravenous; CNS, central nervous system; MAS, macrophage activation syndrome; q, every; sJIA, systemic juvenile idiopathic arthritis. Shenoi S, Wallace CA, 2016 (46)
➡️Parent and carer health educational guide (in Arabic)
الالتهاب المفصلي اليفعي الجهازي مجهول السبب
الالتهاب المفصلي اليفعي الجهازي مجهول السببsJIA يعد نوع مميز من أنواع الالتهاب المفصلي اليفعي مجهول السبب JIA,يتميزبوجود بعض الأعراض الأكلينيكية التى تميزه عن باقي الأنواع وكذلك المضاعفات وطرق التشخيص والخيارات العلاجية الخاصه به , و يمثل هذا النوع حوالي 10 ٪ إلى 20 ٪ من مجمل حالات JIA ، مع معدلات إصابة تتراوح من 0.4 إلى 0.8 طفل لكل 100.000 طفل.
يمكن أن تظهرأعراضsJIA في أي وقت خلال الطفولة والمراهقة ، مع ذروة الإصابة عند عمر سنتين,يصاب به الأولاد والبنات على حد سواء, يحدث عند الأطفال من جميع الخلفيات العرقية ، مع معدل انتشار أعلى في الجنوب الشرقي واليابان.
▪️معايير التصنيف الالتهاب المفاصل البفعي مجهول السبب وفقا لرابطة الجمعيات الدولية للروماتيزم:
يتطلب تشخيص sJIA وجود التهابات في واحد من المفاصل أو أكثر، مع أو قد يسبقها ظهور حمى مستمرة لمدة أسبوعين على الأقل ، تحدث بشكل يومي (على الأقل3 أيام يومية) ، ويرافقها واحد أو أكثر من هذه الاعراض:الطفح الجلدي أحمر غير ثابت في مكانه , تضخم العقد اللمفاوية المنتشرة في الجسم, تضخم الكبد أو الطحال, التهاب في الاغشية المصلية. ولتشخيص الحالة انها sJIA فيجب استبعاد وجود :
- مرض الصدفية أواي تاريخ مرضي للاصابه بها لدى المريض أو أقاربه من الدرجة الأولى.
- ان تكون التهابات المفاصل في طفل ذكر عمره اكتر من ست سنوات ولديه HLA-B27ايجابي.
- مرض التيبس الفقاري المناعي ، التهاب المفاصل المرتبط بالتهاب ألأوتار ERA ، الالتهاب المفصلي العجزي الحرقفي المصاحب لالتهابات القولون المناعية ، التهاب القزحي الأمامي الحاد ، أو تاريخ أحد هذه الأمراض لدى المريض او أحد أقاربه من الدرجة الأولى.
- تحليل عامل الروماتويدي RF موجب مرتين يفصل بينهما ثلاثة أشهر علي الاقل.
▪️الاعراض الاكلينيكية:
§ حمى يومية ترتفع فيها الحرارة الي39-40 درجة مئوية مرة أو مرتين في اليوم ثم تنخفض سريعا إلى الطبيعي.
§ طفح جلدي بقعي أو لطاخي متحرك من مكان لمكان (غير ثابت)، غير مسبب للحكة، يظهر في الجذع والرقبة والجزء من الأطراف القريب من الجذع ,ومن المميز له ظهوره مع نوبات الحمى وتلاشيه عند زوال الحمى.
§ التهابات المفاصل تكون في أربع مفاصل أو أقل أو فد تكون متعددة الي أكثر من خمس مفاصل و المفاصل الأكثر التهابا (المعصمين, الركبتين, والكاحلين). ظهور التهاب المفاصل عادة يكون في خلال الأشهر الثلاثة الاولي من ظهور المرض، إلا أنه قد يتأخر في الظهور لأسابيع اوأشهرأو نادرًا الي سنوات.
§ تضخم العقد اللمفاوية المنتشرة في الجسم ويكون التضخم واضح مما يثير الشكوك من انه ورم خبيث.
§ تضخم الكبد والطحال.
§ التهاب في الأغشية المصلية والذي يكون عادة بدون أعراض ويمكن اكتشافه فقط بمخطط صدى القلب أو تصوير شعاعي للصدر.
▪️التشخيص
لا توجد تحاليل محددة لـتشخيص مرضsJIA)). التحاليل المعملية تعد داعمة للتشخيص وتعكس الطبيعة الألتهابية للمرض ولاستبعاد الحالات التي تسبب أعراض مماثلة.صورة الدم قد تشير الي وجود زيادة في عدد كريات الدم البيضاء والصفائح الدموية وانيميا فقر الدم,ويلاحظ وجود ارتفاع في معاملات الألتهاب مثل معدل ترسيب كرات الدم الحمراء (ESR) ، بروتين سي التفاعلي (CRP) ونسبة الفيرتين بالدم. اما الأجسام المضادة الذاتية مثل الأجسام المضادة الذاتية المضادة للنواة (ANA) والعامل الروماتويدي (RF) فتكون دائمًا سلبيًة ولا تفيد في التشخيص(لوحظ وجود ANA إيجابي في حوالي 4٪ إلى 6٪ من مرضى sJIA)).قد نحتاج الي نضح وخزعة نخاع العظام لاستبعاد الأورام الخبيثة في الدم.
الفحص الإشعاعي قد يبين تورم الأنسجة الرخوة وهشاشة في العظام.فقدان الغضاريف وتاكل العظام عادا ما يظهر في غضون عامين من ظهور المرض.
▪️المضاعفات
متلازمة تنشيط البلاعم MAS هي أحد مضاعفات التي تهدد حياةالأطفال المصابين بـ sJIA.حيث يعاني المريض من حمى شديدة غير مستجيبة وتدهور للحالة العامة للمريض, مع تضخم بالكبد والطحال، وخلل عصبي، ومظاهر نزفية, وفشل متعدد في وظائف أعضاء الجسم.تظهر التحاليل المعملية انخفاض بكريات الدم , وارتفاع في وظائف الكبد ونازعة هيدروجين اللاكتات والدهون الثلاثية والفيرتين بالدم ، مع انخفاض مستويات الفيبرينوجين وانخفاض نسبة سرعة الترسيب ESR بعد أن كانت مرتفعة سابقا ، مع استمرار ارتفاع البروتين التفاعلي CRP. قد نحتاج الي نضح وخزعة نخاع العظام للتأكد من تشخيص MAS.
التهاب المفاصل التآكلي المبكر من المضاعفات المتكررة في حالات المصابين بsJIA وتؤثر عادة علي الركبتين والكاحلين والرسغين. كذلك هشاشة العظام وتأخر النمو من المضاعفات المحتملة الي جانب المضاعفات الناتجة عن تناول الأدوية مثل عقار الكورتيزون لفترات طويلة والادوية المثبطة لجهازللمناعة والتى تزيد نسبة الإصابة بالعدوي والأورام الخبيثة.
المرضي المصابين ب sJIA لديهم احتمال أكبر لحدوث عجز وظيفي متوسط و شديد مقارنة بالأنواع الاخرى من JIA و قد يعزى هذا إلى عدد المفاصل واجهزة الجسم المتأثرة بالاصابة.
▪️العلاج
الهدف من العلاج هو السيطرة على الألتهاب النشط من أجل تخفيف الأعراض والوقاية من المضاعفات المرتبطة بالمرض.نظرًا لطبيعة مرض sJIA وتأثيره علي العديد من أجهزة الجسم، يجب أن تركز بروتوكولات العلاج على الطفل ككل وليس على جانب واحد فقط من المرض.يشمل العلاج الناجح كلا من التدخل الدوائي وغير الدوائي بما في ذلك التغذية العلاجية والعلاج الطبيعي والدعم النفسي والأجتماعي للمريض.
في البداية ، تستخدم مضادات الألتهاب غير الستيرويدية والكورتيزون للسيطرة على المرض, ولكن بسبب الآثار الجانبية غير المرغوب فيها والمرتبطة باستخدام هذه الأدوية فتهدف البروتوكولات الحديثة في العلاج إلى التقليل التدريجي أو التوقف التام عن استخدام هذه الأدوية بعد ان يتم إجراء التشخيص الدقيق للمرض.
تم استخدام العلاج البيولوجى مثل مثبطات TNF بشكل متكرر، ولكنها أظهرت فعالية منخفضة مقارنة بنتائجها في المرضى المصابين بالأنواع الأخرى من .JIAاما العلاجات المضادة لـ IL-1 و IL-6 اثبتت فعالية كبيرة في مرضي sJIA وكلما كان العلاج بهذه العقاقير مبكرا كلما اعطي فرصة افضل لتحسين النتايج.
الحالات المصابة ب MASتحتاج لجرعات كبيرة من الكورتيزون عن طريق الوريد اولا (ميثيل بريدنيزولون لمدة ثلاثة أيام متتالية) متبوعًا بالكورتيزون عن طريق الفم و يستخدم عقارالسيكلوسبورين وهو مثبط الكالسينيورين كعلاج ثانوي وكذلك العلاجات البيولوجية وخاصة مضادIL-1((Anakinraالذي يستخدم بنجاح في علاج كل من MAS و sJIA النشط ويعد مفيدًا في تحقيق التخفيض السريع لجرعة الكورتيزون.
- Limitations and suggestions for further research needs
▪️Resource
barriers
Treatment
guidelines are hard to implement in daily practice due to a lack of resources
of patients particularly the cost of biologic therapy
▪️System barriers
Treatment
guidelines are not approved by official bodies e.g. MOH
▪️Attitudinal
barriers
Physicians’ autonomy in practice
using their usual approach.
Disagreement of Senior Pediatrician
with the guidelines recommendations.
Treatment guidelines may limit
flexibility and individual approach.
Patient barriers
Patients do not want doctors to conform to treatment guidelines (expensive medication, for fear of side effects)
In addition, Lack of relevant national data or JIA registry disable our estimates for diagnostic and management difficulties. We have no multicenter studies from which reasonable conclusion could be derived and included in the recommendations.
There is no financial funding from any source for our EPG project till now which of course limit our ability for research, developing evidence-based recommendations and doing an effective implementation plan.
Some of the relevant international guidelines did not grant permission for adaptation or did not respond. Others may ask for a paid adaptation permission which is unaffordable.
- Monitoring and evaluating the impact of the guideline.
The following are two performance measures or indicators for implementing this adapted CPG for Jeuvenile idiopathic arthritis in children:
1. Adherence to JIA Guidelines
▪️Numerator: Number of children who received treatment as per guideline recommendations.
▪️ Denominator: Total number of children who received treatement.
▪️ Data Source: Hospital or clinic patient records.
2. Adherence to JIA guidelines
▪️ Numerator: Number of children readmitted with symptoms of JIA within a certain period (e.g., 30 days) and managed according to the guidelines.
▪️ Denominator: Total number of children admitted with JIA.
▪️ Data Source: Hospital readmission records.
These key performance indicators are designed to measure effectiveness and adherence to the guidelines.
- Updating of the guideline
The EPG Rheumatology GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2028 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Onel KB, Horton DB, Lovell DJ, Shenoi S, Cuello CA, Angeles‐Han ST, et al. 2021 American College of Rheumatology guideline for the treatment of juvenile idiopathic arthritis:Therapeutic approaches for Oligoarthritis, Temporomandibular joint arthritis, and Systemic juvenile idiopathic arthritis. Arthritis Care Res (Hoboken). 2022 Apr;74(4):553-69. doi: 10.1002/acr.24853
2. Ravelli A, Consolaro A, Horneff G, et al.Treating juvenile idiopathic arthritis to target: recommendations of an international task force Annals of the Rheumatic Diseases 2018;77:819-828.doi:10.1136/annrheumdis-2018-213030.
3. Hinze et al. Practice and consensus-based strategies in diagnosing and managing systemic juvenile idiopathic arthritis in Germany 2018. Pediatric Rheumatology (2018) 16:7. DOI 10.1186/s12969-018-0224-2
4. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. J EvalClinPract. 2015 Dec;21(6):1095-106. doi: 10.1111/jep.12479
5. Fervers B, Burgers JS, Voellinger R, Brouwers M, Browman GP, Graham ID, Harrison MB, Latreille J, Mlika-Cabane N, Paquet L, Zitzelsberger L. Guideline adaptation: an approach to enhance efficiency in guideline development and improve utilisation. BMJ quality & safety. 2011 Mar 1;20(3):228-36.
6. AGREE (II) Instrument available from the www.agreecollaboration.org/instrument/ (downloaded free).
7. Ravelli A, Martini A. Juvenile idiopathic arthritis. Lancet 2007; 369: 767-79.
8. Lee JJY, Schneider R. Systemic Juvenile Idiopathic Arthritis. Pediatr Clin North Am. 2018;65(4):691-709.
9. Martini A, Ravelli A, Avcin T, Beresford MW, Burgos-Vargas R, Cuttica R, et al. Toward New Classification Criteria for Juvenile Idiopathic Arthritis: First Steps, Pediatric Rheumatology International Trials Organization International Consensus. J Rheumatol 2019; 46(2):190-7.
10. Thierry S, Fautrel B, Lemelle I, Guillemin F. Prevalence and incidence of juvenile idiopathic arthritis: a systematic review. Joint Bone Spine. 2014;81(2):112–7
11. Modesto C, Antón J, Rodriguez B, Bou R, Arnal C, Ros J, et al. Incidence and prevalence of juvenile idiopathic arthritis in Catalonia (Spain). Scandinavian Journal of Rheumatology 2010: 39; 472–9.
12. Abou El-Soud A, El-Najjar A, El-Shahawy E, Amar H, Hassan T, Abd-Allaha S, Ragab H. Prevalence of juvenile idiopathic arthritis in Sharkia Governorate, Egypt: epidemiological study. Rheumatol Int 2013; 33:2315–22
13. Consolaro A, Ravelli A. Defining criteria for disease activity states in juvenile idiopathic arthritis. Rheumatology 2016;55(4):595–6.
14. Salah S, Hamshary A, Lotfy H, Abdel Rahman H. Juvenile Idiopathic Arthritis, the Egyptian Experience. Journal of Medical Sciences 2009;9: 98- 102.
15. Hussein ZM, Wagdy R, Shawki M, Zohni S, Shehawy I. The pattern of juvenile idiopathic arthritis; a retrospective Egyptian study. Egypt J Pediatr Allergy Immunol 2018; 16(1):7-14.
16. Bahabri S, Al-Sewairi W, Al-Mazyad A, Karrar A, Al-Ballaa S, El-Ramahai K, et al. Juvenile rheumatoid arthritis: The Saudi experience. Ann Saudi Med 1997; 4:413-8.
17. Fujikawa S, Okuni M. Clinical analysis of 570 cases with juvenile rheumatoid arthritis: results of a nationwide retrospective survey in Japan. Acta PaediatrJpn. 1997;39(2):5–249.
18. Palomo J, Dietrich D, Martin P, Palmer G, Gabay C. The interleukin (IL)-1 cytokine family – balance between agonists and antagonists in inflammatory diseases. Cytokine 2015;76(1):25–37.
19. Toplak N, Blazina S, Avcin T. The role of IL-1 inhibition in systemic juvenile idiopathic arthritis: current status and future perspectives. Drug Des DevelTher 2018; 12:1633-43.
20. Martini A. Systemic juvenile idiopathic arthritis. Autoimmun Rev 2012;12:56–9.
21. Castaneda S, Blanco R, Gonzalez-Gay MA. Adult-onset Still’s disease: advances in the treatment. Best Prac Res Clin Rheumatol. 2016;30(2):222-38.
22. Kessel C, Lippitz K, Weinhage T, Hinze C, Wittkowski H, Holzinger D, et al. Proinflammatory cytokine environmentas can drive interleukin-17 overexpression by γ/δ T cells in systemic juvenile idiopathic arthritis. ArthritsRheumatol. 2017;69(7):1480-94.
23. Petty RE, Southwood TR, Manners P, Baum J, Glass DN, Goldenberg J, et al, International League of Associations for Rheumatology. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J Rheumatol. 2004;31:390–2
24. Singh-Grewal D, Schneider R, Bayer N, Feldman BM. Predictors of disease course and remission in systemic juvenile idiopathic arthritis: significance of early clinical and laboratory features. Arthritis Rheum 2006;54(5):1595–601.
25. Beukelman T. Treatment advances in systemic juvenile idiopathic arthritis. F1000Prime Rep 2014;6:21.
26. Cimaz R. Systemic onset juvenile idiopathic arthritis. Autoimmun Rev 2016;15(9):931–4.
27. Tibaldi J, Pistorio A, Aldera E, Puzone L, El Miedany Y, Pal P, et al. Development and initial validation of a composite disease activity score for systemic juvenile idiopathic arthritis. Rheumatology (Oxford). 2020;59(11):3505-14.
28. Woerner A, von Scheven-Gête A, Cimaz R, Hofer M. Complications of systemic juvenile idiopathic arthritis: risk factors and management recommendations. Expert Rev Clin Immunol. 2015 May;11(5):575-88. doi: 10.1586/1744666X.2015.1032257.
29. Ravelli A, Minoia F, Davi S, Horne AC, Bovis F, Pistorio A, et al. 2016 Classification criteria for macrophage activation syndrome complicating systemic juvenile idiopathic arthritis. A European League Against Rheumatism/American College of Rheumatology/ Pediatric Rheumatology International Trials Organization Collaborative Initiative. Arthritis Rheumatol 2016. DOI 10.1002/ART.39332
30. Çakan M, Karadag SG, Tanatar A, Ayaz NA. The frequency of macrophage activation syndrome and disease course in systemic juvenile idiopathic arthritis. Mod Rheumatol2020; 30(5):900-4.
31. Behrens EM, Beukelman T, Paessler M, Cron RQ. Occult macrophage activation syndrome in patients with systemic juvenile idiopathic arthritis. J Rheumatol. 2007;34(5):1133–8
32. Ravelli A, Grom AA, Behrens EM, Cron RQ. Macrophage activation syndrome as a part of systemic juvenile idiopathic arthritis: diagnosis, genetics, pathophysiology and treatment. Genes Immun 2012;13(4):289–98.
33. De Benedetti F, Brunner H, Ruperto N, Schneider R, Xavier R, Allen R,et al. Catch-up growth during tocilizumab therapy for systemic juvenile idiopathic arthritis: results from a phase III trial. Arthritis Rheumatol. 2015;67(3):840–848.
34. Beukelman T, Patkar NM, Saag KG, Tolleson-Rinehart S, Cron RQ, DeWitt EM, et al. 2011 American College of Rheumatology recommendations for the treatment of juvenile idiopathic arthritis: initiation and safety monitoring of therapeutic agents for the treatment of arthritis and systemic features. Arthritis Care Res. 2011;63(4):465–82.
35. Ringold S, Weiss PF, Beukelman T, EM DW, Ilowite NT, Kimura Y, et al. 2013 update of the 2011 American College of Rheumatology recommendations for the treatment of juvenile idiopathic arthritis: recommendations for the medical therapy of children with systemic juvenile idiopathic arthritis and tuberculosis screening among children receiving biologic medications. Arthritis Rheum. 2013;65(10):2499–512.
36. Correll CK, Binstadt BA. Advances in the pathogenesis and treatment of systemic juvenile idiopathic arthritis. Pediatr Res 2014;75(1–2):176–83.
37. Vastert SJ, de Jager W, Noordman BJ, Holzinger D, Kuis W, Prakken BJ, et al. Effectiveness of first-line treatment with recombinant interleukin-1 receptor antagonist in steroid-naive patients with new-onset systemic juvenile idiopathic arthritis: results of a prospective cohort study. Arthritis Rheumatol. 2014;66(4):1034–43.
38. DeWitt EM, Kimura Y, Beukelman T, Nigrovic PA, Onel K, Prahalad S, et al. Consensus treatment plans for new-onset systemic juvenile idiopathic arthritis. Arthritis Care Res. 2012;64(7):1001–10.
39. Yokota S, Itoh Y, Morio T, Sumitomo N, Daimaru K, Minota S. Macrophage activation syndrome in patients with systemic juvenile idiopathic arthritis under treatment with Tocilizumab. J Rheumatol. 2015;42(4):712-22.
40. Record JL, Beukelman T, Cron RQ. Combination therapy of abatacept and anakinra in children with refractory systemic juvenile idiopathic arthritis: a retrospective case series. J Rheumatol. 2011;38(1):180–1.
41. Canny S, Mellins E. New frontiers in the treatment of systemic juvenile idiopathic arthritis. F1000Res 2017;6:971.
42. Canna SW, Girard C, Malle L, de Jesus A, Romberg N, KelsenJ,et al. Life threatening NLRC4-associated hyperinflammation successfully treated with IL-18 inhibition. J Allergy Clin Immunol 2017;139(5):1698–701.
43. Huang Z, Lee PY, Yao X, Zheng S, Li T. Tofacitinib treatment of refractory systemic juvenile idiopathic arthritis. Pediatrics 2019; 143(5):e20182845.
44. Sandborg C, Holmes TH, Lee T, Biederman K, Bloch DA, Emery H, et al. Candidate early predictors for progression to joint damage in systemic juvenile idiopathic arthritis. J Rheumatol 2006;33(11):2322–9.
45. Russo RA, Katsicas MM. Patients with very early onset systemic juvenile idiopathic arthritis exhibit more inflammatory features and a worse outcome. J Rheumatol 2013;40(3):329–34.
46. Boom, V., Anton, J., Lahdenne, P. et al. Evidence-based diagnosis and treatment of macrophage activation syndrome in systemic juvenile idiopathic arthritis. Pediatr Rheumatol 13, 55 (2015). https://doi.org/10.1186/s12969-015-0055-3
47. Ambler WG, Nanda K, Onel KB, Shenoi S. Refractory systemic onset juvenile idiopathic arthritis: current challenges and future perspectives. Ann Med. 2022 Dec;54(1):1839-1850.
48. Shenoi S, Wallace CA. Diagnosis and Treatment of Systemic Juvenile Idiopathic Arthritis. J Pediatr. 2016 Oct;177:19-26. doi: 10.1016/j.jpeds.2016.06.056. Epub 2016 Aug 4. PMID: 27499217.
49. WHO. World Health Organization Model List of Essential Medicines. Vol. 22nd List. 2021.
50. Brouwers MC, Kerkvliet K, Spithoff K. The AGREE Reporting Checklist: a tool to improve reporting of clinical practice guidelines. Bmj. Mar 8 2016;352:i1152. doi:10.1136/bmj.i1152
51. Brouwers MC, Kho ME, Browman GP, et al. AGREE II: advancing guideline development, reporting and evaluation in health care. J Clin Epidemiol. Dec 2010;63(12):1308-11. doi:10.1016/j.jclinepi.2010.07.001
52. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist. Ann Intern Med. May 2022;175(5):710-719. doi:10.7326/m21-4352
53. AGREE Tools. The AGREE Research Trust. 2024. www.agreetrust.org
54. Abdel Baky A, Omar TEI, Amer YS, the Egyptian Pediatric Clinical Practice Guidelines C. Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bulletin of the National Research Centre. 2023/06/13 2023;47(1):88. doi:10.1186/s42269-023-01059-0
55. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. East Mediterr Health J. Jul 31 2023;29(7):540-553. doi:10.26719/emhj.23.070
56. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. Journal of evaluation in clinical practice. Dec 2015;21(6):1095-106. doi:10.1111/jep.12479
57. Klugar M, Lotfi T, Darzi AJ, et al. GRADE guidance 39: using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. J Clin Epidemiol. Aug 6 2024;174:111494. doi:10.1016/j.jclinepi.2024.111494
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Dr Ashraf Abdel Baky |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children's Hospital, Faculty of Medicine, Ain Shams University. Chair of EPG/GAG |
None |
Not Applicable |
|
Dr Dalia H. El-Ghoneimy |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Faculty of Medicine, Ain Shams University. |
None |
Not Applicable |
|
Dr Mohamed Almalky |
Professor of Pediatrics, Faculty of Medicine, Zagazig University. |
None |
Not Applicable |
|
Dr Shereen Esam Maher |
Professor of Pediatrics, Pediatric Rheumatology, Children's Hospital, Faculty of Medicine, Minia University. |
None |
Not Applicable |
|
Dr Ali Sobh |
Associate Professor of Pediatrics, Mansoura University Children’s Hospital, Faculty of Medicine, Mansoura University. |
None |
Not Applicable |
|
Dr Iman Fahmy |
Associate Professor of Pediatics, Pediatric Allergy, Immunology and Rheumatology Unit, Children's Hospital, Faculty of Medicine, Suhag University |
None |
Not Applicable |
|
Dr Nesrine Radwan
|
Associate Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children's Hospital. Faculty of Medicine, Ain Shams University. |
None |
Not Applicable |
|
Dr Rasha El-Owaidy |
Associate Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children's Hospital. Faculty of Medicine, Ain Shams University. |
None |
Not Applicable |
|
Dr Yomna Mohamed Farag |
Associate Professor of Pediatrics, Pediatric Rheumatology Unit, Specialized Children’s Hospital, Faculty of Medicine, Cairo University. |
None |
Not Applicable |
|
Dr Amira Hattab |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital. Faculty of Medicine, Ain Shams University. |
None |
Not Applicable |
|
Dr Ghada Abdel Haleem Shousha |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital. Faculty of Medicine, Ain Shams University. |
None |
Not Applicable |
|
Dr. Sally Gouda |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital. Faculty of Medicine, Ain Shams University. |
None |
Not Applicable |
|
Dr Naglaa Samy Mohamed Osman |
Lecturer of Pediatrics, Pediatric Allergy, Immunology and Rheumatology Unit, Children's Hospital, Faculty of Medicine, Assiut University. |
None |
Not Applicable |
|
Dr Walaa Shoman |
Lecturer of Pediatrics, Children's Hospital. Faculty of Medicine, Alexandria University. |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanies Soliman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Prof. Zeinab Awad El-Sayed |
Professor of Pediatrics, Pediatric Allergy, Immunology and Rheumatology unit, Children’s Hospital, Ain Shams University, Egypt. |
None |
Not Applicable |
|
Prof. Angelo Ravelli |
Professor of Pediatrics, department of Neurosciences, Rehabilitation, Opthalomology, Genetics and Maternal-Infantile Science (DiNOGMI), University of Geno, Italy. |
None |
Not Applicable |
|
Dr. Karen Onel |
Chief, Division of Pediatric Rheumatology Hospital for special Surgery. Professor of clinical Pediatrics Weil Cornell Medicine |
None |
Not Applicable |
|
Associate Prof. Soad Hashad |
Associate Professor, Pediatric Rheumatology, Tripoli University, Libya. |
None |
Not Applicable |
|
Associate Prof. Ghada Al-Deriny |
Associate Prof. Of Pediatrics, Alexandria University, Egypt |
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
None |
Not Applicable |
|
International Peer Reviewers |
|
|
|
|
Yasser Wali |
Pediatric Hematology/Oncology Unit, Child Health Department, Sultan Qaboos University Hospital, Muscat, Oman |
None |
Not Applicable |
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for "Guideline for the Juvenile idiopathic arthritis in children " on PubMed are: Juvenile Idiopathic Arthritis guidelines, JIA, Systemic Juvenile Idiopathic Arthritis guidelines, sJIA classification, sJIA Guidelines
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for transfusion of blood components in pediatric age groups

Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Practice guideline REgistration for transPAREncy (PREPARE) Guideline Protocol Registration Receipt
