Diagnosis, Management and Prevention of Acute Hemolytic Anemia in Infants, Children and Adolescents
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Diagnosis, Management and Prevention of Acute Hemolytic Anemia in Infants, Children and Adolescents |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:47 AM |
Description
"last update: 18 Feb 2025" Download Guideline
- Executive Summary
➡️Introduction
Acute hemolysis is a common presentation in the Emergency room and General
Pediatrics clinic visits. For most parts of the world, the guideline recommendations are
targeting specific disease entities after initial diagnosis. The purpose of developing the
Egyptian guidelines are to identify strategies and comprehensive actions needed to help
general Pediatricians in the initial approach and management of children presenting with
acute hemolytic episode till reaching a diagnosis and referring the patient to the pediatric
hematologist
➡️Scope
This guideline focuses on diagnosis, prevention and management of the most common causes of acute hemolytic anemia where early interventional treatment can reduce the risk or extent of end-organ failure;” Glucose-6-phosphate-dehydrogenase (G6PD) deficiency, autoimmune hemolytic anemia and hemolytic uremic syndrome” in infants (beyond the neonatal period), children and adolescents
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- Laboratory diagnosis of G6PD deficiency. A British Society for Haematology Guideline.
Br J Haematol. 2020;189(1):24-38 [1].
2- Diagnosis and management of G6PD deficiency. Am Fam Physician. 2005 Oct 1;72(7):1277-82 [2].
3-The diagnosis and management of primary autoimmune haemolytic anaemia.
Br J Haematol. 2017;176(3):395-411 [3].
4- Diagnosis and management of newly diagnosed childhood autoimmune haemolytic anaemia. Recommendations from the Red Cell Study Group of the Paediatric Haemato-Oncology Italian Association. Blood Transfus. 2017;15(3):259-267 [4]
5- 2017 Infectious Diseases Society of America Clinical Practice Guidelines for the Diagnosis and Management of Infectious Diarrhea. Clin Infect Dis. 2017;65(12):1963-1973 [5].
6- Hemolytic uremic syndrome in a developing country: Consensus guidelines.
Pediatr Nephrol. 2019 Aug;34(8):1465-1482 [6].
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
➡️Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following three sub-sections:
A. Diagnosis of acute hemolytic anemia
The guideline covers how to diagnose and differentiate between different causes of acute hemolytic anemia in infants (beyond the neonatal period), children and adolescents.
This section includes recommendations and good practice statements on first level testing in patient presented with AHA, indication and timing for reassessment and when and which further testing are needed.
B. Management of the most pressing causes of AHA namely Glucose-6-phosphate-dehydrogenase (G6PD) deficiency, autoimmune hemolytic anemia and hemolytic uremic syndrome.
This section includes recommendations and good practice statements on first line therapy in AIHA, indication of and precautions during packed red cells transfusion, and follow up plans for patients with G6PD and AIHA
C. Prevention of acute hemolytic anemia due to Glucose-6-phosphate-dehydrogenase (G6PD) deficiency, autoimmune hemolytic anemia and hemolytic uremic syndrome.
This section includes recommendations and good practice statements on which drug to be avoided in patients with G6PD, and indications for empiric antibiotics in children with diarrhea to avoid the development of HUS
We can summarize the guidelines’ recommendations for acute hemolytic anemia in the following:
➡️For diagnosis of Acute Hemolytic Anemia:
▪️ We suggest that First level testing for any patient presented with acute hemolysis includes: * CBC with red cell morphology on peripheral smear, * Reticulocyte count, * Indices of hemolysis (indirect bilirubine, LDH), * Urine analysis, * Direct and indirect antibody test (DAT, Coombs test), * Blood group, [Very Low], Liver transaminases and kidney function (blood urea and serum creatinine). [Low, Conditional]
· We recommend that; If G6PD assay was done during a hemolytic episode of unknown cause, re-assay is warranted to ensure the diagnosis of G6PD deficiency is not missed. [Low, Strong}
▪️ We suggest that a normal G6PD activity during a hemolytic episode should be repeated after 2 months in absence of clinical and laboratory evidence of hemolysis. [Very Low]
▪️ We recommend that G6PD can be diagnosed with a quantitative spectrophotometric analysis or, more commonly, by a rapid fluorescent spot test.[Low, Strong]
▪️ We recommend against relying on screening tests for female patients. Measure G6PD activity by quantitative spectrophotometric assay directly. [Low, Strong]
▪️ We recommend that quantitative assay must be carried out if the screening test is abnormal or borderline. [Low, Strong]
▪️ We recommend that the final G6PD activity should be interpreted in the lights of the reticulocyte count measured on the same sample. [Low, Strong]
▪️ We recommend that samples with very low mean corpuscular hemoglobin (MCH) may give G6PD activity levels above the expected value. [Very Low, Strong]
▪️ We suggest G6PD testing in the following situations; * Favism (hemolysis with fava beans intake), * Hemolysis associated with “oxidant” drugs (Table in implementation tools) or infection, * Abnormal red-cell morphology (Basket cells/ positive Heinz body stain), * Hemoglobinuria, or * Hemolysis with previous history of neonatal pallor/jaundice. [Very Low]
▪️ We recommend that in case of AIHA at a minimum, the DAT should include monospecific anti-IgG and anti-C3d. [Low, Strong]
▪️ We recommend that in case of DAT-negative hemolysis: In patients with unexplained hemolysis and a negative screening DAT, retest with a column agglutination DAT method that includes monospecific anti-IgG, anti-IgA and anti-C3d. [Moderate, Strong]
▪️ We suggest that retest was also negative, consider preparing and investigating a red cell eluate. [Low, Conditional].
▪️ We recommend that the next test to be done before starting treatment; * Immunological markers: ANA, Anti DNA, C3, Antiphospholipid, * Total serum immunoglobulins, * CD markers and * Double negative α/β T cells if hepatosplenomegaly. [High, Strong]
▪️ We suggest that bone marrow aspiration is only recommended in the presence of another associated cytopenia or persistent reticulocytopenia or on suspicion of neoplasia or myelodysplasia. [Very Low]
▪️ We suggest the following initial laboratory panel for patients with suspected post diarrheal hemolytic uremic syndrome; • Complete blood count (hemoglobin <10 g/dl, hematocrit <30%, platelet count <150,000/μl), • Peripheral smear (fragmented red cells - schistocytes ≥ 2%), • Lactate dehydrogenase (LDH > 450 IU/l), • Haptoglobin (undetectable), • Kidney function tests (increased serum creatinine by 50% over baseline level). [Very Low]
For management of Acute Hemolytic Anemia:
▪️ We recommend that if anemia is life threatening, transfuse with ABO, Rh and K matched red cells. [Low, Strong]
▪️ We suggest that transfusion can be avoided in hemodynamically stable patients with regular monitoring until no evidence of hemolysis. [Low, Conditional]
▪️ We suggest to Transfuse if - Hb < 5 g/dl or - Hb is < 7g/dl with cardiac decompensation. GPS
▪️ We suggest to consider the use of a blood warmer for transfusion in patients with cold AIHA (CHAD, mixed AIHA and PCH). [Low, Conditional]
▪️ We suggest the following dose: 3-5 ml/Kg of packed RBCs fully compatible ABO, Rh, K, leucodepleted filtered blood. Transfuse slowly over 4 hours. [Very Low]
▪️ We recommend that the First line therapy of AIHA is prednisolone1-2 mg/kg /day. [Moderate, Strong]
▪️ We suggest that the second line therapy for AIHA should be considered if: - No response to steroids after 3 weeks, - Relapse during or after steroid reduction. And Refer to Hematologist. [Low, Conditional]
▪️ We suggest to consider IV Ig or plasma exchange for severe or life-threatening anemia. [Low, Conditional]
▪️ We recommend for patients with AIHA should receive folic acid supplementation. [Low, Strong]
▪️ We recommend that All patients should receive oral calcium and vitamin D supplements while taking corticosteroids (typically 1200–1500 mg calcium and 800–1000 units’ vitamin D). [High, Strong]
▪️ We recommend follow up by CBC and reticulocyte count every 2-4 weeks when indicated by treating physician. And that G6PD enzyme assay should be tested after complete recovery of the hemolytic attack and patient discharge. [Low, Strong]
➡️For prevention of Acute Hemolytic Anemia:
▪️ We suggest that Patients with G6PD deficiency should avoid exposure to oxidant drugs and ingestion of fava beans. And that patients with suspected G6PD deficiency should avoid exposure to oxidant drugs and ingestion of fava beans till diagnosis. [Low]
▪️ We recommend against the use of antimicrobial therapy in children with infections attributed to certain strain of STEC that produce Shiga toxin 2 (or if the toxin genotype is unknown). [Moderate, Strong]
▪️ We recommend that in immunocompetent children, empiric antimicrobial therapy for bloody diarrhea while waiting for results of investigations is not recommended, except for: * Infants <3 months of age with suspicion of a bacterial etiology. * Ill immunocompetent children with fever, abdominal pain, bloody diarrhea, and bacillary dysentery presumptively due to Shigella. [Low, Strong] * People who have recently travelled internationally with body temperatures ≥38.5°C and/or signs of sepsis. [Low, Conditional]
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/- Recommendations
|
CPGL Source |
Recommendation |
QOE |
SOR |
|
Diagnosis Questions |
|
||
|
|
1.1 What is the initial laboratory panel for infants, children and adolescents presenting with AHA? |
|
|
|
AIEOP 2017 |
First level testing includes • Hemoglobin Level, red cell indices and morphology on peripheral smear • Reticulocyte count • WBC and platelet count • Indices of hemolysis (indirect bilirubin, LDH) • Urine analysis • Direct and indirect antibody test (Coombs test) • Blood group |
Low
|
|
|
BSH 2017 |
• Liver transaminases and kidney function (blood urea and serum creatinine) |
Low |
Conditional |
|
|
1.2 What is the best timing and test to diagnose G6PD deficiency in infants, children and adolescents presenting with AHA? |
|
|
|
BSH 2020
|
• If G6PD assay was done during a hemolytic episode of unknown cause, re-assay is warranted to ensure the diagnosis of G6PD deficiency is not missed. |
Low
|
Strong |
|
|
• A normal G6PD activity during a hemolytic episode should be repeated after 2 months in absence of clinical and laboratory evidence of hemolysis. |
Low* |
|
|
BSH 2020 |
• G6PD can be diagnosed with a quantitative spectrophotometric analysis or, more commonly, by a rapid fluorescent spot test. |
Low |
Strong |
|
BSH 2020 |
• Do not rely on screening tests for female patients. • Measure G6PD activity by quantitative spectrophotometric assay directly. |
Low |
Strong |
|
BSH 2020 |
• Quantitative assay must be carried out if the screening test is abnormal or borderline |
Low |
Strong |
|
BSH 2020 |
• The final G6PD activity should be interpreted in the lights of the reticulocyte count measured on the same sample |
Low |
Strong |
|
BSH 2020 |
• Samples with very low mean corpuscular hemoglobin (MCH) may give G6PD activity levels above the expected value |
Low |
Strong |
|
|
1.3 What are the indications of G6PD testing? |
|
|
|
BSH 2020 |
• Favism (hemolysis with fava beans intake) • Hemolysis associated with “oxidant” drugs (Table in implementation tools) or infection • Red-cell morphology (Basket cells/ positive Heinz body stain) • Hemoglobinuria • Hemolysis with previous history of neonatal pallor/jaundice |
Low |
|
|
|
1.4 What is the best test to diagnose AIHA in infants, children and adolescents presenting with AHA? |
|
|
|
BSH 2017 |
• At a minimum, the DAT should include monospecific anti-IgG and anti-C3d |
Low |
Strong |
|
BSH 2017 |
• Investigation of DAT-negative hemolysis: - In patients with unexplained hemolysis and a negative screening DAT, retest with a column agglutination DAT method that includes monospecific anti-IgG, anti-IgA and anti-C3d - If also negative, consider preparing and investigating a red cell eluate. |
Intermediate
Low |
Strong |
|
|
1.5 What are the additional tests before starting treatment in patients with AIHA? |
|
|
|
BSH 2017
AIOEP 2017 |
• Immunological markers - ANA, Anti DNA, C3, Antiphospholipid - Total serum immunoglobulins - CD markers and double negative α/β T cells if hepatosplenomegaly
• Bone marrow aspiration is only recommended in the presence of another associated cytopenia or persistent reticulocytopenia or on suspicion of neoplasia or myelodysplasia. |
High
Low |
Strong |
|
|
1.6. What is the initial laboratory panel for patients with suspected post diarrheal hemolytic uremic syndrome? |
|
|
|
Bagga et al 2019 |
• Complete blood count (hemoglobin <10 g/dl, hematocrit <30%, platelet count <150,000/μl) • Peripheral smear (fragmented red cells - schistocytes ≥ 2%) • Lactate dehydrogenase (LDH > 450 IU/l) • Haptoglobin (undetectable) • Kidney function tests (increased serum creatinine by 50% over baseline level) |
Low** |
|
|
Management Questions |
|
||
|
|
2.1 What is the emergency treatment for AHA? |
|
|
|
BSH 2017 |
• If anemia is life threatening, transfuse with ABO, Rh and K matched red cells. |
Low |
Strong |
|
|
2.2 When is Packed red cells indicated in AHA? Sub-question: volume/ precautions needed? |
|
|
|
BSH 2017
|
• Transfusion can be avoided in hemodynamically stable patients with regular monitoring until no evidence of hemolysis. • Transfuse if - Hb < 5 g/dl - Hb is < 7g/dl with cardiac decompensation |
Low
GPS |
Conditional |
|
BSH 2017 |
• Consider the use of a blood warmer for transfusion in patients with cold AIHA (CHAD, mixed AIHA and PCH) |
Low |
Conditional |
|
AIEOP 2017 |
• Dose: 3-5 ml/Kg of packed RBCs fully compatible ABO, Rh, K, leucodepleted filtered blood. Transfuse slowly over 4 hours |
Low |
|
|
|
2.3 What
is the medical treatment |
|
|
|
BSH 2017 |
• First line therapy is prednisolone 1-2 mg/kg/day |
Intermediate |
Strong |
|
BSH 2017 |
• Second line therapy should be considered if: - No response to steroids after 3 weeks - Relapse during or after steroid reduction And Refer to Hematologist |
Low |
Conditional |
|
BSH 2017 |
• Consider IVIg or plasma exchange for severe or life-threatening anemia |
Low |
Conditional |
|
BSH 2017 |
• Patients with AIHA should receive folic acid supplementation |
Low |
Strong |
|
BSH 2017 |
• All patients should receive oral calcium and vitamin D supplements while taking corticosteroids (typically 1200–1500 mg calcium and 800–1000 units vitamin D) |
High |
Strong |
|
|
2.4 For follow up after discharge, what are the laboratory tests needed in patients with G6PD and AIHA? |
|
|
|
BSH 2020 |
• Follow up by CBC and reticulocyte count every 2-4 weeks when indicated by treating physician. • G6PD enzyme assay should be tested after complete recovery of the hemolytic attack and patient discharge. |
Low |
Strong |
|
Prevention Questions |
|
||
|
|
3.1 What are the drugs to be avoided and the dietary modifications required to prevent the occurrence of AHA in patients with G6PD deficiency? |
|
|
|
AFP 2005 |
• Patients with G6PD deficiency should avoid exposure to oxidant drugs and ingestion of fava beans. • Patients with suspected G6PD deficiency should avoid exposure to oxidant drugs and ingestion of fava beans till diagnosis. |
Low |
Strong |
|
|
3.2 For prevention of HUS, when is empiric antibacterial treatment indicated for children with bloody diarrhea? |
|
|
|
IDSA 2017 |
Antimicrobial therapy should be avoided in children with infections attributed to certain strain of STEC that produce Shiga toxin 2 (or if the toxin genotype is unknown) |
Intermediate |
Strong |
|
IDSA 2017 |
In immunocompetent children, empiric antimicrobial therapy for bloody diarrhea while waiting for results of investigations is not recommended, except for: · Infants <3 months of age with suspicion of a bacterial etiology. · Ill immunocompetent children with fever, abdominal pain, bloody diarrhea, and bacillary dysentery presumptively due to Shigella. · People who have recently travelled internationally with body temperatures ≥38.5°C and/or signs of sepsis. |
Low
Low |
Strong
Conditional |
* Expert opinion adopted from: Luzzatto L, Seneca E. G6PD deficiency: a classic example of pharmacogenetics with on-going clinical implications. Br J Haematol. 2014 Feb;164(4):469-80. doi: 10.1111/bjh.12665. PubMed PMID: 24372186; PubMed Central PMCID: PMCPMC4153881.[32]
**Bagga A, Khandelwal P, Mishra K, et al. Hemolytic uremic syndrome in a developing country: Consensus guidelines. Pediatr Nephrol. 2019 Aug;34(8):1465-1482. doi: 10.1007/s00467-019-04233-7. PubMed PMID: 30989342.[33]
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
||||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|
|||
|
Prof. Galila Mokhtar. |
Paediatrics Department, Faculty of Medicine, Ain Shams University, Cairo, Egypt |
Editor, Clinical expert GAG member |
|
|||
|
Prof. Ilham Youssry. |
Professor of Paediatrics and Head of the Paediatric Haematology & BMT unit, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Iman Ragab. |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Laila Sherief. |
Professor of Paediatrics and Paediatrics Haematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Marwa Zakaria. |
Associate Professor of Paediatrics, Zagazig University, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Mervat Hesham. |
Professor of Paediatrics, Paediatric Haematology & Oncology Unit, Zagazig University, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Niveen Salama. |
Associate Professor of Paediatrics, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|
|||
|
Dr. Nouran Yousef. |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Rasha Abdel-Raouf Afifi. |
Professor of Paediatrics and Paediatric Haematology, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|
|||
|
Dr. Sara Makkeyah. |
Associate Professor of Paediatrics, Paediatric Haematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Sherein Abdelhamid Shalaby. |
Professor of Pediatrics, head of Pediatrics department. Helwan University, Egypt |
Clinical expert GAG member |
|
|||
|
Prof. Sonia Adolf. |
Professor of Paediatrics and Paediatric Haematology, National Research Center, Cairo, Egypt |
Clinical expert GAG member |
|
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
||||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|
|||
|
Associate Professor of Pediatrics Ain Shams University, Egypt |
Developing evidence to decision (EtD) frameworks, participating in search and guideline appraisal |
|
||||
|
- Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. - Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. - Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|
||||
|
Dr. Lamis Mohsen Elsholkamy
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|
|||
|
Dr. Ahmed Mahmoud Youssef
|
Fellow of Pediatrics, General Organization for Teaching Hospitals and Institutes (GOTHI). Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|
|||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|
|||
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|
|||
|
External Reviewers Group (ERG) |
||||||
|
External Reviewer(s) for Clinical Content |
||||||
|
||||||
|
Prof. Ahmed Kaddah |
Professor of Pediatrics and Pediatric Hematology, Cairo University, Egypt. |
|||||
|
Prof. Azza Tantawy |
Professor of Pediatrics and Pediatric Hematology/Oncology, Ain Shams University, Egypt. |
|||||
|
Prof. Hoda Hassab |
Professor of Pediatrics and Pediatric Hematology/Oncology, Alexandria University, Egypt |
|||||
|
International Peer Reviewers |
||||||
|
Drew Provan |
Barts and The London School of Medicine and Dentistry, Queen Mary University of London, London, United Kingdom |
|||||
|
External Reviewer(s) for methodology |
||||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
|||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
|||||
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge The British Society for Haematology, The American Family Physicien Society, The Red Cell Study Group of Paediatric Haemato-Oncology Italian Association, The Infectious Diseases Society of America, and The Hemolytic uremic syndrome in a developing country: Consensus guidelines for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️ This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AEA |
Anti-Erythrocyte Autoantibodies |
||
|
AFP |
American Family Physician |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
AHA |
Acute Hemolytic Anemia |
||
|
AHC |
Acute Hemolytic Crisis |
||
|
AIHA |
Autoimmune Hemolytic Anemia |
||
|
AKI |
Acute Kidney Injury |
||
|
AIEOP |
Italian Association of Paediatric Onco-haematology |
||
|
ANA |
Anti Nuclear Antibodies |
||
|
BSH |
British Society of Haematology |
||
|
C3 |
Complement 3 |
||
|
CBC |
Complete Blood Count |
||
|
CHAD |
Cold Hemagglutin Disease |
||
|
CPG |
Clinical Practice Guideline |
||
|
DAT |
Direct antiglobulin test |
||
|
DHS |
Demographic and Health Survey |
||
|
DNA |
Deoxyribo-neucleic Acid |
||
|
EBM |
Evidence Based Medicine |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ER |
Emergency Room |
||
|
ERG |
External Review Group |
||
|
G6PD |
Glucose 6 Phosphate Dehydrogenase |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GIN |
Guideline International Network |
||
|
GL |
Guideline |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
Hb |
Hemoglobin |
||
|
HUS |
Hemolytic uremic syndrome |
||
|
IDSA |
Infectious Diseases Society of America |
||
|
Ig |
Immuno-globulin |
||
|
IV |
Intra-Venous |
||
|
LDH |
Lactate Dehydrogenase |
||
|
MCV |
Mean Corpuscular Volume |
||
|
PCH |
Paroxysmal cold hemoglobinuria |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
QOE |
Quality Of Evidence |
||
|
RBCs |
Red Blood Cells |
||
|
RCT |
Randomized Control Trail |
||
|
RDW |
Red cell Distribution Width |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
||
|
SOR |
Strength Of Recommendation |
||
|
STEC |
Shiga Toxin Producing Escherichia Coli |
||
|
WBC |
White Blood Cell |
||
|
WHO |
World Health Organization |
- Glossary
1. Acceptability;
Is the extent to which the users are likely to adopt a recommendation, based on
internal qualities such as clarity, comprehensiveness, and logical reasoning and on external
factors such as the burden imposed on the process and system of care, patient and
providers’ attitudes and beliefs, and patients’ needs, expectations, and preferences.
2. Adaptation (of guidelines);
Is the systematic approach to considering the use and/or modification of (a) guidelines(s)
produced in one cultural and organizational setting for application in different context?
Adaptation can be used as an alternative to de novo guideline development or for
customizing (an) existing guideline(s) to suit the local context.
3. Adoption (of a guideline);
Is the acceptance of a guideline as a whole after the assessment of its quality, currency, and
content. When health care providers (or other users of recommendations) adopt a
guideline, they feel committed to change their practices in accordance with the
recommendations of the guideline.
4. Applicability;
Is the extent to which the users are able to put a recommendation into practice, based on
internal qualities such as a clearly defined eligible patient population that matches the
population to which the intervention is targeted in the local setting and external factors
such as the availability of the necessary knowledge, skills, provider time, staff, equipment,
and other resources.
Applicability is sometimes taken as a synonym for feasibility:
Feasibility of the acquisition of necessary skills and knowledge
Feasibility of the necessary increase in provider time, staff, equipment, and so on.
5. Culture;
Culture represents the norms and values of a specific group, community, or population.
6. Diffusion;
Is a passive means of transferring knowledge; it is not directed towards a target audience
(e.g. publication of articles in medical journals).
7. Dissemination;
Is more active than diffusion in that it targets a specific audience and involve tailoring the
information for that audience (e.g. of dissemination strategies include targeted mailings,
presentations, and press conferences.
8. Evidence-based principles;
Evidence-Based Medicine (EBM) has been defined as ― the conscientious, explicit, and
judicious use of current best evidence in making decisions about the care of individual
patients. The practice of EBM means integrating individual clinical expertise with the best
available external clinical evidence from systematic research.
9. Evidence tables;
Are summaries of the most salient information from studies identified in the systematic
review. The elements of evidence tables are dependent on the types of information in
studies related to a particular topic but might include information such as the article
reference, the study type (e.g. RCT or Cohort), the number of patients and their
characteristics, and the intervention, comparison arms, outcome measures, and effect sizes.
10. Guideline or Clinical Practice Guideline (CPG);
Systematically developed statements about specific health problems, intended to assist
practitioners and patients in making decisions about appropriate health care.
11. Guideline consistency;
Agreement between the evidence and the recommendations, based on the: Comprehensiveness of the study search and selection process, Coherence between the results of the studies and their interpretation by the guideline authors, and Transparency between interpretation and recommendations.
12. Guideline content;
In the ‘ADAPTE Manual and Resource Toolkit for Guideline Adaptation’ document,
guideline content refers to the recommendations in the source guidelines.
13. Guideline currency;
A CPG may be considered up to date ―when (no) new information on interventions,
outcomes, and performance justifies updating (it).
14. Guideline quality;
By quality of clinical practice guidelines, we mean the confidence that the potential biases
of guideline development addressed adequately and that the recommendations are both
internally and externally valid and are feasible for practice. This process involves taking
into account the benefits, harms and costs of the recommendations, as well as the practical
issues attached to them. Therefore, the assessment (of quality) includes judgments about
the methods used for developing the guidelines, the content of the final recommendations,
and the factors linked to their uptake.
15. Guideline topic;
In the ADAPTE Manual and Resource Toolkit for Guideline Adaptation' document, the
topic refers to the theme of the guideline, as described in the guideline title, for a targeted
population (disease and patients) and intervention. The purpose, the audience, and the
setting intended for the guideline, although not necessarily explicitly stated in the title, are
also part of the topic. A guideline on a given topic may contain more than one health
question.
16. Health question or clinical question or key question;
Is a precisely described health issue (e.g. clinical, professional practice or public health)
relating to the topic of the guideline? Guideline may include one or more questions.
17. Implementation;
Implementation includes methods to promote the uptake of research findings into routine
healthcare in both clinical and policy contexts and hence to improve the quality and
effectiveness of healthcare. It includes the study of influences on healthcare professional
and organizational behavior.
18. Intra-class correlations;
Intra-class correlations provide a measurement of the extent to which two or more raters
agree when rating the same set of things. It is a reliability index and is typically a ratio of
the variance of interest over the sum of the variance of interest plus error.
19. Recommendation;
Any statement that promote or advocate a particular course of action in clinical care.
20. Stakeholder;
A stakeholder is an individual, group and/or organization with a stake in your decision to
implement a guideline. Stakeholders include individuals or groups who will be directly or
indirectly affected by the implementation of a guideline.
21. Source guideline;
In the ADAPTE Manual and Resource Toolkit for Guideline Adaptation document, source
guideline refers to those guidelines selected to undergo assessments of quality, currency,
content, consistency, and acceptability/applicability and upon which an adapted guideline
may be based
- Introduction
Anemia, defined as decreased red blood cell (RBC) mass and or decrease in hemoglobin
level, is a common disease that affects nearly one quarter of the population worldwide. [7,
8] Overall, anemia can be categorized as acute or chronic. [9, 10] The most pressing concerns for the emergency physician are the acute anemias that can cause immediate, life-threatening complications. [10].
The most common causes of acute hemolytic anemia where early interventional treatment can reduce the risk or extent of end-organ failure are Glucose-6-phosphate-dehydrogenase (G6PD) deficiency, autoimmune hemolytic anemia and hemolytic uremic syndrome. [11, 12, 13, 14]
Glucose-6-phosphate-dehydrogenase (G6PD) deficiency; the most common red cell
enzyme deficiency in the world, with over 400 million affected. African, Asian,
Mediterranean, and Middle- Eastern descents are most commonly affected by this disorder.
The prevalence of G6PD deficiency among Egyptian neonates is 8.9%. The deficiency is
caused by mutations in G6PD gene, located on the long arm of the X chromosome. A
deficiency of the enzyme causes reduced glutathione, which makes RBCs more
susceptible to oxidant stress and can result in episodic hemolytic anemia. [15,16]
Autoimmune hemolytic anemia; the clinical condition where autoantibodies are
produced and directed against self RBCs. [16, 17] where IgG or IgM antibodies bind to
antigens on the surface of the RBC, leading to decreased survival of the RBC. [17, 18, 19]
Autoimmune hemolytic anemia (AIHA) can be primary (idiopathic) or secondary to a
systemic disease or drugs. A large range of medications can induce AIHA, including methyldopa, penicillin, cephalosporin’s, several nonsteroidal anti-inflammatory
medications, quinidine, and isoniazid [18]. Furthermore, depending on the type and the
thermal sensitivity of the antibody, AIHA can be the warm antibody type, cold antibody
type, or mixed type. [17, 18]. Warm antibody AIHA is caused by IgG antibodies, which bind strongly at room temperature. These antibodies are pan agglutinins – that is, they react with all the red cell antigens. Patients with warm AIHAs can present with severe, life threatening symptoms [17, 18, 20].
The initial treatment of AIHA depends on the severity of hemolysis [12]. As with any
unstable patient, abnormal vital signs should be first addressed. RBC transfusion should be
limited to cases of life-threatening anemia [14, 15, 18]. This is because transfusions can
induce further autoantibody production, and it might be difficult to find an accurate
crosshatch [18]. Corticosteroids slow the rate of hemolysis and considered as a first-line
therapy that should be started as soon as possible [17, 18, 21].
Hemolytic Uremic Syndrome (HUS); a clinical syndrome characterized by progressive
renal failure that is associated with microangiopathic (nonimmune, Coombs-negative)
hemolytic anemia and thrombocytopenia. It is one of the common causes of acute kidney
injury in children and is increasingly recognized in adults. [22]
Typical HUS should be suspected in any child who presents with a history of bloody
diarrhea and new-onset anemia, thrombocytopenia, and acute renal injury. Atypical HUS
should be suspected in patients with similar symptoms but no history of acute diarrheal
illness. [23] The treatment of typical HUS is supportive as well as possible dialysis. [24]
To aid the clinician in AHA management, we present these guidelines that represent and highlight distinct clinical presentations with separate diagnostic and therapeutic pathways
that can be used in clinical practice setting. We also review the parameters present in
diagnostic testing that allow for prognostic insight and present algorithms for both
diagnosis and treatment of the AIHA patient in diverse situations. This is done in the hope
that this work may offer guidance regarding personalized therapy recommendations.
➡️Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of acute hemolytic anemia.
It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to s to assist health care practitioners to apply the best available evidence to clinical decisions about the diagnosis, management, and prevention of acute hemolytic anemia with the following objective in mind: putting a differential diagnosis and approach to the diagnosis and proper management of acute hemolytic anemia in infants, children, and adolescents; identification of infants, children, and adolescents at high risk of developing acute hemolysis; prevention of recurrence of hemolysis in the targeted
population.
This version of the guideline includes recommendations and good practice statements for diagnosis, management, and prevention of Glucose-6-phosphate-dehydrogenase (G6PD) deficiency, autoimmune hemolytic anemia and hemolytic uremic syndrome.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: ‘acute hemolytic anemia’, ‘G6PD deficiency’, ‘autoimmune hemolytic anemia’ ‘hemolytic uremic syndrome’ ‘children’ and adolescents’.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) [25:27] by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 6 guidelines:
1- Laboratory diagnosis of G6PD deficiency. A British Society for Haematology Guideline.
Br J Haematol. 2020;189(1):24-38.
2- Diagnosis and management of G6PD deficiency. Am Fam Physician. 2005 Oct 1;72(7):1277-82.
3- The diagnosis and management of primary autoimmune haemolytic anaemia.
Br J Haematol. 2017;176(3):395-411.
4- Diagnosis and management of newly diagnosed childhood autoimmune haemolytic anaemia. Recommendations from the Red Cell Study Group of the Paediatric Haemato-Oncology Italian Association. Blood Transfus. 2017;15(3):259-267.
5- 2017 Infectious Diseases Society of America Clinical Practice Guidelines for the Diagnosis and Management of Infectious Diarrhea. Clin Infect Dis. 2017;65(12):1963-1973.
6- Hemolytic uremic syndrome in a developing country: Consensus guidelines.
Pediatr Nephrol. 2019 Aug;34(8):1465-1482.
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development) [28:31]
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians’/ healthcare providers’ subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of hematology and bone marrow disorders.
The main functions of the clinical panel were adolopment of Acute Hemolytic Anemia Guidelines, determining the scope of the guideline and guidelines, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians’ subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in AHA; Prof Azza Tantawy, Prof Hoda Hassab, and prof Ahmed Kaddah, as well as eminent international reviewer prof Drew Provan.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group were asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [28]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations.
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
➡️Levels of evidence and grades of recommendation:
The panel used the original grading system of the reference guidelines after their permission. Code A represents a high quality of evidence, B represents a moderate quality of evidence, C represents a low quality of evidence, and D represents a very low quality of evidence, while number 1 stands for a strong level of recommendations and number 2 stands for a weak
level of recommendations
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing AHA diagnosis, management, and prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of health care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, management and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behavior.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of AHA in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for management of acute hemolytic anemia
3. Arabic Educational materials for nurses and mothers
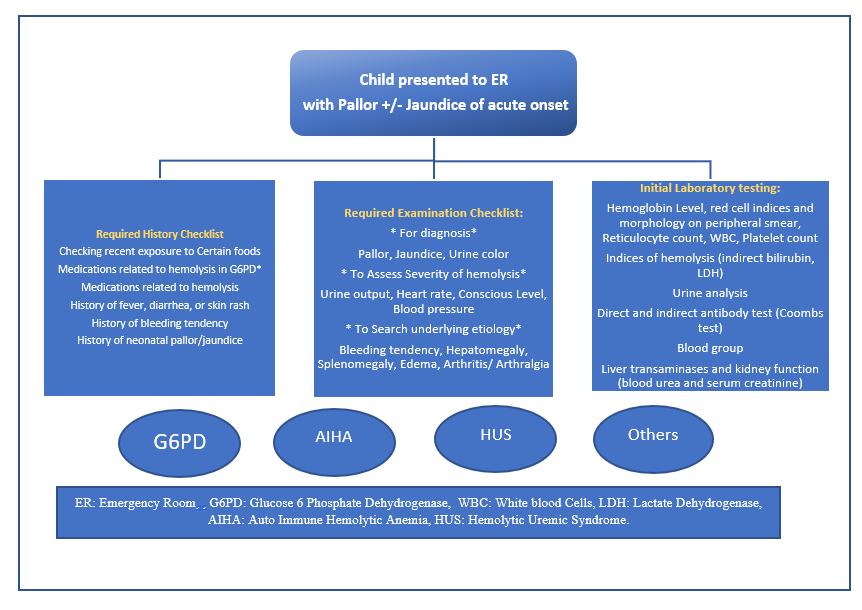
Figure 1. Diagnostic Approach to the child Presenting to the ER with acute Pallor +/- Jaundice
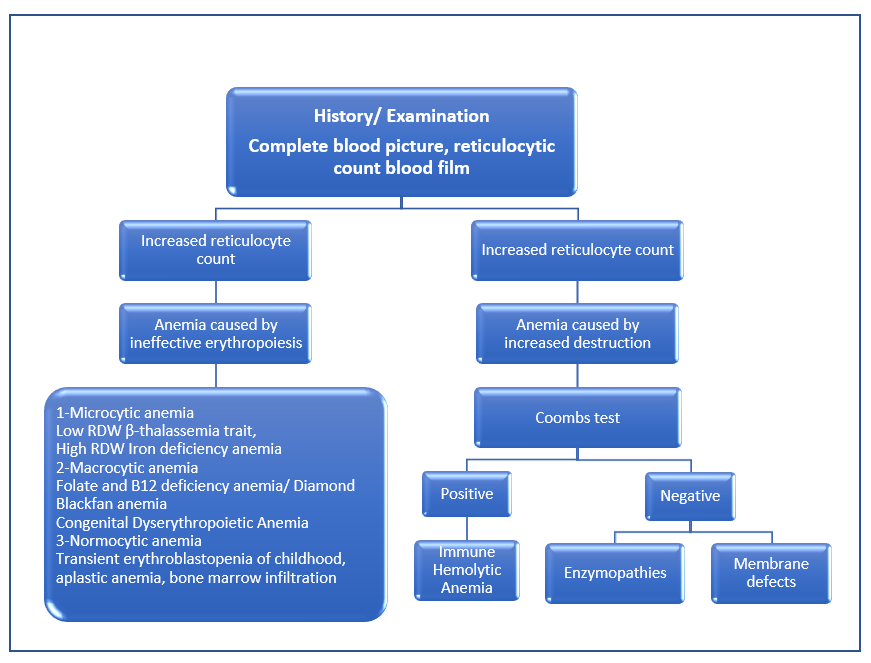
Figure 2. Diagnostic Approach to Child with Acute Pallor +/- Jaundice According to Reticulocyte count
Table 3: Health Questions used to develop this Adapted Clinical Practice Guideline:
|
Diagnosis |
History and clinical examination: (see figure 1 & 2) Laboratory diagnosis: 1.What is the initial laboratory panel for infants, children and adolescents presenting with AHA? 2.What is the best timing and test to diagnose G6PD deficiency in infants, children and adolescents presenting with AHA? 3.What are the indications of G6PD testing? 4.What is the best test to diagnose AIHA in infants, children and adolescents presenting with AHA? 5.What are the additional immunological tests before starting treatment in patients with AIHA? 6.What is the initial laboratory panel for patients with suspected post diarrheal hemolytic uremic syndrome? |
|
Management |
Emergency management: 1.What is the emergency treatment for infants, children and adolescents presenting with AHA? 2.When is Packed red cells indicated? Sub-question: volume/ precautions needed? Medical Pharmacological treatment: 3.What is the medical treatment in infants, children, and adolescents with AIHA? What are the criteria of adequate /good response to treatment in infants, children, and adolescents with AIHA? Monitoring and Follow-up 4.For follow up after discharge, what are the laboratory tests needed in patients with G6PD and AIHA? |
|
Prevention |
1.What are the drugs to be avoided and the dietary modifications required to prevent the occurrence of AHA in patients with G6PD deficiency? 2.For prevention of HUS, when is empiric antibacterial treatment indicated for children with bloody diarrhea? |
Table 4. Differential Diagnosis of AIHA19:
|
Congenital forms |
Spherocytosis and other defects of the erythrocyte membrane proteins; erythrocyte enzyme deficiency; - dyserythropoietic anemia; - haemoglobinopathies; Wilson's disease. |
|
Hemolytic anemia from mechanical causes |
Synthetic heart valves; march hemoglobinuria; cardiopulmonary bypass. |
|
Hemolytic anemia due to vascular injury |
Microangiopathic anemia; thrombotic thrombocytopenic purpura; hemolytic-uremic syndrome; disseminated intravascular coagulation; arterio-venous malformations. |
|
Hemolytic anemia due to thermal damage |
Extensive burns. |
|
Hemolytic anemia from chemical causes |
Chemicals: solvents; methyl chloride; lead; arsenic and hydrogen; snake venom. |
|
Hemolytic anemia due to infectious agents |
Bacteria (Mycoplasma pneumoniae, Clostridium welchii); viruses (cytomegalovirus, herpes virus); protozoa (Plasmodium spp.). |
Table 5: Common drugs that patients with G6PD deficiency should avoid or use with caution23, 24:
|
Generic name |
Risk level |
Generic name |
Risk level |
|
Acetaminophen |
Low |
Moxifloxacin |
High |
|
Acetylsalicylic acid |
Variable* |
Naphthalen |
High |
|
Ascorbic acid (Vitamin C) |
High with high dose |
Nitric oxide |
High |
|
Chloramphenicol |
High |
Nitrofurantoin |
High |
|
Ciprofloxacin |
High |
Nitroglycerin |
High |
|
Colchicine |
Low |
Phenazopyridine (pyridium) |
High |
|
Dapsone |
High |
Primaquine |
High** |
|
Diphenhydramine |
Low |
Probenecid |
High |
|
Glimepiride |
High |
Rasburicase |
High*** |
|
Glipizide |
High |
Sodium nitroprusside |
High |
|
Glyburide |
Use with caution |
Streptomycin |
Low |
|
Hydroxychloroquine |
High |
Sulfacetamide |
High |
|
Isoniazid |
Low |
Sulfamethoxazole |
High**** |
|
Levofloxacin |
High |
Trimethoprim |
Low**** |
|
Methylene blue |
High |
Vitamin K |
Low to high† |
* Risk level low to none, use with caution
** Reduce dose with medical therapy if required
*** Prescribing modification required
**** Also, when in combination with the other drug
† High with vitamin K3 (menadione), low with vitamin K1 (phytomenadione)
- Limitations and suggestions for further research needs
The lake of availability of national guidelines that address acute hemolytic anemia management in recourse limited country is the main limitation to our work.
Future research recommendations for acute hemolytic anemia in children in the Egyptian context could include:
▪️ The impact of time lapses between presentation to the ER and starting transfusion on the morbidity and mortality of patients with acute hemolytic crisis.
▪️ The incident of AIHA in Egypt on the post COVID era.
Other recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for acute hemolytic anemia in children.
➡️Challenges
▪️ Training primary care doctor to spot patients with acute hemolysis and know how to approach such cases, recognizing patients that need urgent hospitalization or intensive care management and avoidance of any delay that may results in grave consequences.
▪️ Emphasis on avoiding empiric antibiotics description for patients with diarrhea.
▪️ Implementation of international guidelines in resource limited settings.
▪️ The optimum use of the available resources to fulfill the requirements of Egyptian population.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks
➡️Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for acute hemolytic anemia in children:
1. Adherence to acute hemolytic anemia Guidelines
▪️ Numerator: Number of children with acute hemolytic anemia who received treatment as per guideline recommendations.
▪️ Denominator: Total number of children diagnosed with acute hemolytic anemia.
▪️ Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
▪️ Numerator: Total number of hospital stay days for children with acute hemolytic anemia.
▪️ Denominator: Total number of children admitted with acute hemolytic anemia.
▪️ Data Source: Hospital admission and discharge records.
3. Rate of Readmission
▪️ Numerator: Number of children readmitted with symptoms of acute hemolytic anemia within a certain period (e.g., 30 days) after discharge.
▪️ Denominator: Total number of children initially admitted with acute hemolytic anemia.
▪️ Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG …GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Roper D, Layton M, Rees D, Lambert C, Vulliamy T, De la Salle B, D’Souza C, British Society for Haematology. Laboratory diagnosis of G6PD defciency. A British Society for Haematology Guideline. Br J Haematol. 2020. https://doi.org/10.1111/bjh.16366. PubMed PMID : 31991476.
2. Frank JE (2005) Diagnosis and management of G6PD defciency. Am Fam Physician 72(7) :1277–1282 PubMed PMID : 16225031.
3. Hill QA, Stamps R, Massey E, Grainger JD, Provan D, Hill A, British Society for Haematology (2017) The diagnosis and management of primary autoimmune haemolytic anaemia. Br J Haematol 176(3) :395–411. https://doi.org/10.1111/bjh.14478. PubMed PMID : 28005293.
4. Ladogana S, Maruzzi M, Samperi P, Perrotta S, Del Vecchio GC, Notaran‑gelo LD et al (2017) Diagnosis and management of newly diagnosed childhood autoimmune haemolytic anaemia. Recommendations from the Red Cell Study Group of the Paediatric Haemato-Oncology Italian Association. Blood Transfusion 15(3) :259. https://doi.org/10.2450/2016.0072-16. PubMed. PMID : 28151390 ; PubMed Central PMCID PMCPMC5448833
5. Shane AL, Mody RK, Crump JA, Tarr PI, Steiner TS, Kotlof K et al (2017) 2017 Infectious Diseases Society of America clinical practice guidelines for the diagnosis and management of infectious diarrhea. Clin Infect Dis 65(12) : e45-80. https://doi.org/10.1093/cid/cix959. PubMedPMID:291945. 9 ; PubMedCentralPMCID : PMCPMC5848254
6. Bagga A, Khandelwal P, Mishra K, Thergaonkar R, Vasudevan A, Sharma J et al (2019) Hemolytic uremic syndrome in a developing country : consensus guidelines. Pediatr Nephrol 34 :1465–1482. https://doi.org/10.1007/s00467-019-04233-7
7. Adamson JW, Longo DL. Anemia and polycythemia. In : Kasper DL, Harrison TR, editors. Harrison's principles of internal medicine. New York : McGraw-Hill, Medical Pub. Division ; 2005
8. World Health O. Haemoglobin concentrations for the diagnosis of anaemia and assessment of severity. Geneva : World Health Organization ; 2011.
9. Dhaliwal G, Cornett PA, Tierney LM, Jr. Hemolytic anemia. Am Fam Physician. 2004 Jun 1 ;69(11) :2599-606. PubMed PMID : 15202694.
10. Janz T, Hamilton G, editors. CHAPTER 121 Anemia, Polycythemia, and White Blood Cell Disorders2013.
11. Barcellini W, Fattizzo B. Clinical Applications of Hemolytic Markers in the Differental Diagnosis and Management of Hemolytic Anemia. Dis Markers. 2015 ;2015 :635670. doi : 10.1155/2015/635670. PubMed PMID : 26819490 ; PubMed Central PMCID : PMCPMC4706896.
12. Bain BJ. Diagnosis from the blood smear. N Engl J Med. 2005 Aug 4 ;353(5) :498-507. doi : 10.1056/NEJMra043442. PubMed PMID : 16079373.
13. George JN, Charania RS. Evaluation of patients with microangiopathic hemolytic anemia and thrombocytopenia. Semin Thromb Hemost. 2013 Mar ;39(2) :153-60. doi: 10.1055/s-0032-1333538. PubMed PMID : 23390027.
14. Baker KR, Moake J. Fragmentation Hemolytic Anemia. 2016. In : Williams hematology [Internet]. New York, N.Y. : McGraw-Hill Education LLC, 9th. [1 - 30 -online resource]. Available from: https://yale.idm.oclc.org/login?URL=https://accessmedicine.mhmedical.com/book. aspx ? bookID=1581.
15. Cappellini MD, Fiorelli G. Glucose-6-phosphate dehydrogenase deficiency. Lancet. 2008 Jan 5 ;371(9606) :64-74. doi : 10.1016/S0140-6736(08)60073-2. PubMed PMID : 18177777.
16. Kasemy ZA, Bahbah WA, El Hefnawy SM, et al. Prevalence of and mothers’ knowledge, attitude and practice towards glucose-6-phosphate dehydrogenase deficiency among neonates with jaundice : a cross-sectional study BMJ Open 2020 ;10 : e034079. doi : 10.1136/bmjopen-2019-034079.
17. Gehrs BC, Friedberg RC. Autoimmune hemolytic anemia. Am J Hematol. 2002 Apr ;69(4) :258-71. doi : 10.1002/ajh.10062. PubMed PMID : 11921020.
18. Packman CH. Hemolytic Anemia Resulting from Immune Injury. 2016. In : Williams hematology [Internet]. New York, N.Y. : McGraw-Hill Education LLC, 9th. [1 online resource]. Available from: https://yale.idm.oclc.org/login?URL=https://accessmedicine.mhmedical.com/book. aspx?bookID=1581.
19. Bunn HF, Rosse W. Hemolytic anemias and acute blood loss. In : Kasper DL, Harrison TR, Fauci AS, et al., editors. Harrison's Principles of Internal Medicine : McGraw-Hill, Medical Pub. Division ; 2005.
20. Packman CH. Hemolytic anemia due to warm autoantibodies. Blood reviews. 2008 Jan ;22(1) :17-31. doi : 10.1016/j.blre.2007.08.001. PubMed PMID : 17904259 ; eng.
21. Petz LD. Emergency Transfusion Guidelines for Autoimmune Hemolytic Anemia. Laboratory Medicine. 2005 ;36(1) :45-48. doi : 10.1309/NE3BH8U3K6N1149V %J Laboratory Medicine.
22. Siegler R, Oakes R. Hemolytic uremic syndrome ; pathogenesis, treatment, and outcome. Curr Opin Pediatr. 2005 Apr ;17(2) :200-4. doi : 10.1097/01.mop.0000152997. 66070.e9. PubMed PMID : 15800412.
23. Tarr PI, Gordon CA, Chandler WL. Shiga-toxin-producing Escherichia coli and haemolytic uraemic syndrome. Lancet. 2005 Mar 19-25 ;365(9464) :1073-86. doi : 10.1016/S0140-6736(05)71144-2. PubMed PMID : 15781103.
24. Scheiring J, Andreoli SP, Zimmerhackl LB. Treatment and outcome of Shiga toxin-associated hemolytic uremic syndrome (HUS). Pediatr Nephrol. 2008 Oct ;23(10) :1749-60. doi : 10.1007/s00467-008-0935-6. PubMed PMID : 18704506 ; PubMed Central PMCID : PMCPMC6901419.
25. Brouwers MC, Kho ME, Browman GP, et al. AGREE II: advancing guideline development, reporting and evaluation in health care. J Clin Epidemiol. Dec 2010;63(12):1308-11. doi:10.1016/j.jclinepi.2010.07.001
26. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist. Ann Intern Med. May 2022;175(5):710-719. doi:10.7326/m21-4352
27. AGREE Tools. The AGREE Research Trust. 2024. www.agreetrust.org
28. Abdel Baky A, Omar TEI, Amer YS, the Egyptian Pediatric Clinical Practice Guidelines C. Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bulletin of the National Research Centre. 2023/06/13 2023;47(1):88. doi:10.1186/s42269-023-01059-0
29. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. East Mediterr Health J. Jul 31 2023;29(7):540-553. doi:10.26719/emhj.23.070
30. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. Journal of evaluation in clinical practice. Dec 2015;21(6):1095-106. doi:10.1111/jep.12479
31. Klugar M, Lotfi T, Darzi AJ, et al. GRADE guidance 39: using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. J Clin Epidemiol. Aug 6 2024;174:111494. doi:10.1016/j.jclinepi.2024.111494
32. Luzzatto L, Seneca E. G6PD deficiency : a classic example of pharmacogenetics with on-going clinical implications. Br J Haematol. 2014 Feb ;164(4) :469-80. doi : 10.1111/bjh.12665. PubMed PMID : 24372186 ; PubMed Central PMCID : PMCPMC4153881.
33. Bagga A, Khandelwal P, Mishra K, et al. Hemolytic uremic syndrome in a developing country : Consensus guidelines. Pediatr Nephrol. 2019 Aug ;34(8) :1465-1482. doi : 10.1007/s00467-019-04233-7. PubMed PMID : 30989342.
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Galila Mokhtar
|
Professor of Pediatrics, Pediatric Hematology and Oncology unit, Ain Shams University, Cairo, Egypt. |
None |
Not Applicable |
|
Prof. Ilham Youssry |
Professor of Pediatrics and Head of the Pediatric Hematology and BMT unit, Cairo University, Cairo, Egypt. |
None |
Not Applicable |
|
Prof. Iman Ragab |
Professor of Pediatrics, Pediatric Hematology and Oncology Unit, Ain Shams University, Cairo, Egypt. |
None |
Not Applicable |
|
Prof. Laila M Sherief |
Professor of Pediatrics and pediatrics Hematology and Oncology, Zagazig University, Egypt. |
None |
Not Applicable |
|
Prof. Marwa Zakaria |
Associate Professor of Pediatrics, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Mervat Abdallah Hesham |
Professor of Pediatrics, Pediatric Hematology and Oncology Unit, Zagazig University, Egypt |
|
|
|
Prof. Niveen Salama |
Professor of Pediatrics, Cairo University, Cairo, Egypt. |
None |
Not Applicable |
|
Dr. Nouran Yousef Salah |
Lecturer of Pediatrics, Pediatric Diabetes, Endocrinology and Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt. |
None |
Not Applicable |
|
Prof. Rasha Abdel-Raouf |
Professor of Pediatrics and Pediatric Hematology, Cairo University, AFCM, Cairo, Egypt. |
None |
Not Applicable |
|
Dr. Sara Makkeyah |
Lecturer of Pediatrics, Pediatric Hematology and Oncology Unit, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Sherein Abdelhamid Shalaby |
Professor of Pediatrics, head of Pediatrics department. Helwan University, Egypt |
None |
Not Applicable |
|
Prof. Sonia Adolf Habib |
Professor of Pediatrics and Pediatric Hematology, National Research Center, Cairo, Egypt |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics, Ain Shams University, Egypt. Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanis Sulieman |
Associate Professor of Pediatrics, Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Prof. Ahmed Kaddah |
Professor of Pediatrics and Pediatric hematology, Cairo University, Egypt. |
None |
Not Applicable |
|
Prof. Azza Tantawy |
Professor of Pediatrics and Pediatric Hematology/Oncology, Ain Shams University, Egypt. |
None |
Not Applicable |
|
Prof. Hoda Hassab |
Professor of Pediatrics and Pediatric Hematology/Oncology, Alexandria University, Egypt |
None |
Not Applicable |
|
International Peer Reviewers |
|||
|
Drew Provan |
Barts and The London School of Medicine and Dentistry, Queen Mary University of London, London, United Kingdom |
None |
Not Applicable |
|
External Reviewer for methodology |
|
|
|
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
None |
Not Applicable |
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
None |
Not Applicable |
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the prevention and management of Acute hemolytic anemia" on PubMed are: Guideline adaptation, Acute hemolytic anemia, Autoimmune hemolytic anemia, Glucose-6-phosphat dehydrogenase deficiency, Hemolytic uremic syndrome.
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for acute hemolytic anemia
|
CPGs AGREE II DOMAINS |
CPG1 |
CPG2 |
CPG3 |
CPG4 |
CPG5 |
CPG6 |
|
D1: Scope & Purpose% |
63.63 |
85.71 |
85.7 |
76.19 |
95.32 |
87.5 |
|
D2: Stakeholder Involvement% |
61.9 |
61.9 |
82.14 |
80.95 |
85.71 |
78.57 |
|
D3: Rigour of Development% |
86.75 |
87.5 |
71.8 |
85.71 |
85.71 |
85.71 |
|
D4: Clarity & Presentation% |
61.9 |
100 |
88.09 |
76.19 |
95.32 |
85.71 |
|
D5: Applicability% |
88.73 |
78.57 |
64.28 |
52.85 |
92.85 |
61.9 |
|
D6: Editorial Independence% |
78.57 |
85.71 |
78.57 |
92.85 |
92.85 |
100 |
|
OA 1% |
73.58 |
83.23 |
78.57 |
77.45 |
91.29 |
83.23 |
|
OA 2 (Recommend for use) |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
This table uses the AGREE II Domain Score Color codes (< 40% red; > 41 – 70% yellow; > 71 % green) |
||||||
Appendix
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|