Safe Injection Practices
| Site: | EHC | Egyptian Health Council |
| Course: | Infection Prevention and Control Guidelines |
| Book: | Safe Injection Practices |
| Printed by: | Guest user |
| Date: | Sunday, 17 May 2026, 1:44 AM |
Description
"last update: 6 March 2025" Download Guideline
- Executive Summary
Safe injection practices are every provider's responsibility. A safe injection does not harm the person, expose the provider to risks or result in hazardous waste for the community. Improper use and disposal of syringes, needles, and medications imposes risks on patients and healthcare providers. The following recommendations apply to the use of needles, cannulas that replace needles, and, where applicable, intravenous delivery systems.
|
Recommendations |
|
Prepare medications in a designated clean medication preparation area that is separated from potential sources of contamination, including sinks or other water sources (Strong recommendation) |
|
Use aseptic technique when preparing and administering medications to avoid contamination of sterile injection equipment (Strong recommendation) |
|
Disinfect the access diaphragms of medication vials before inserting a device into the vial (Strong recommendation) |
|
Enter medication containers with a new needle and a new syringe, even when obtaining additional doses for the same patient. (Strong recommendation) |
|
Do not administer medications from a syringe to multiple patients, even if the needle or cannula on the syringe is changed. Needles, cannula and syringes are sterile, single-use items; they should not be reused for another patient nor to access a medication or solution that might be used for a subsequent patient (Strong recommendation) |
|
Use needles and syringes for one patient only (this includes manufactured prefilled syringes and cartridge devices such as insulin pens). (Strong recommendation) |
|
Use fluid infusion and administration sets (i.e., intravenous bags, tubing and connectors) for one patient only and dispose appropriately after use. Consider a syringe or needle/cannula contaminated once it has been used to enter or connect to a patient’s intravenous infusion bag or administration set (Strong recommendation) |
|
Ensure single-dose or single-use vials, ampules, and bags or bottles of parenteral solution are used for one patient only. Do not combine leftover contents for later use (Strong recommendation) |
|
Use single-dose vials for parenteral medications whenever possible (Strong recommendation) |
|
If multidose vials must be used, both the needle or cannula and syringe used to access the multidose vial must be sterile (Strong recommendation) |
|
Dedicate multidose vials to a single patient whenever possible. If multidose vials are used for more than one patient, restrict the medication vials to a centralized medication area and do not bring them into the immediate patient treatment area (e.g., operating room, patient room/cubicle) (Strong recommendation) |
|
Store multidose vials in accordance with the manufacturer’s recommendations; discard if sterility is compromised or questionable (Strong recommendation) |
|
Consider the use of syringes with a sharps injury protection feature (SIP devices), as opposed to syringes without a sharps injury protection feature, by health care workers (HCWs) delivering intramuscular, subcutaneous or intradermal injectable medications to patients (Conditional recommendation) |
|
Consider the use of syringes with a re-use prevention feature (RUP devices), as opposed to devices without, by HCWs delivering intramuscular, subcutaneous or intradermal injectable medications to patients (Conditional recommendation) |
|
Wear a surgical mask when placing a catheter or injecting material into the spinal canal or subdural space (i.e., during myelograms, lumbar puncture and spinal or epidural anesthesia) (Strong recommendation) |
- Recommendations
|
Recommendations |
|
Prepare medications in a designated clean medication preparation area that is separated from potential sources of contamination, including sinks or other water sources (Strong recommendation, High grade evidence) |
|
Use aseptic technique when preparing and administering medications to avoid contamination of sterile injection equipment (Strong recommendation, High grade evidence) |
|
Disinfect the access diaphragms of medication vials before inserting a device into the vial (Strong recommendation, High grade evidence) |
|
Enter medication containers with a new needle and a new syringe, even when obtaining additional doses for the same patient. (Strong recommendation, High grade evidence) |
1.8.1 Remarks
Medication Preparation Area
Facilities should have a designated area in which medications are drawn up. The medication preparation area:
● Should be clean and away from possible sources of contamination such as sinks and other water sources.
● Should not contain any tool or equipment that may have come into contact with blood or body fluids as blood collection tubes, or needle holders.
● Should be cleaned and disinfected regularly by well trained staff.
● Should have hard, non-porous surface for preparation activities.
● Should contain and have readily accessible necessary supplies such as alcohol-based hand rub, needles and syringes in their sterile packaging, and alcohol wipes.
When drawing a medication
● Use aseptic technique to access parenteral medications:
o Use new sterile syringe and sterile needle.
o Prevent contact between the injection materials and non-sterile environment.
o Perform hand hygiene and wear clean gloves.
o Disinfect the rubber stopper of medication vials and the neck of glass ampules with sterile 70% alcohol before inserting a needle or breaking the ampule.
o Prepare an injection as close as possible to the time of administration to the patient.
o Use a new sterile syringe with new safety needle or a new sterile vial adaptor for each access.
o Avoid touch contamination of the needle or vial adapter prior to penetrating the rubber diaphragm.
o Consider using conventionally manufactured pre-filled syringes if pre-drawn syringes are needed.
o Avoid interrupting healthcare personnel who are drawing up medications.
o Use a quiet area for medication in the medication area.
Table (3): Indications for glove use in health care ( For safe injection practice)
|
Key elements |
Indications |
Precautions |
|
Glove Use
|
Wear non-sterile, well-fitting, single-use gloves: ● When performing venipuncture or venous access injections, because of the potential for blood exposure at the puncture site ● If the health worker's skin is NOT intact (e.g. through eczema, or cracked or dry skin) ● If the patient's skin is NOT intact (e.g. through eczema, burns or skin infections). |
When undertaking injections, DO NOT use gloves: ● For routine intradermal, subcutaneous and intramuscular injections ● If the health worker's skin is intact ● If the patient's skin is intact. |
|
Recommendations |
|
Do not administer medications from a syringe to multiple patients, even if the needle or cannula on the syringe is changed. Needles, cannula and syringes are sterile, single-use items; they should not be reused for another patient nor to access a medication or solution that might be used for a subsequent patient (Strong recommendation, High grade evidence) |
|
Use needles and syringes for one patient only (this includes manufactured prefilled syringes and cartridge devices such as insulin pens). (Strong recommendation, High grade evidence)
|
|
Use fluid infusion and administration sets (i.e., intravenous bags, tubing and connectors) for one patient only and dispose appropriately after use. Consider a syringe or needle/cannula contaminated once it has been used to enter or connect to a patient’s intravenous infusion bag or administration set (Strong recommendation, Moderate grade evidence) |
1.8.2
Rationale (Annex 1. Safe Injection Myths CDC)
●
Spiking a bag, vial, or bottle of sterile fluid with a dispensing
device and leaving that device in place to withdraw medication for multiple
patients increases the risk for microbial contamination.
●
Never store needles and syringes unwrapped because sterility
cannot be ensured. Keep unwrapped syringes in the original package (e.g.,
intradermal syringes).
●
Place only pre-filled flush syringes (e.g., saline, heparin) that
are terminally sterilized by the manufacturer after packaging onto a sterile
field immediately after opening.
●
Never place items sterilized by manufacturers before final
packaging onto a sterile field (e.g., some types of IV tubing and pre-filled
syringes).
●
Never pool left over parenteral medications (vials or IV
solutions) for later administration.
➡️IV
Solutions
● Use an IV solution (e.g., bag, bottle) for only one patient, and then discard.
● Use needleless spiking devices to remove fluid from IV bottles/bags and vials and use for only one patient.
● Never use a container of IV solution (e.g., bag, bottle) to obtain flush solutions for more than one patient.
● Never use infusion supplies, such as needles, syringes, and administration sets, for more than one patient.
● Use needle free systems for all aspects of parenteral medication administration and transfer of solutions between containers.
● Avoid removing closed system transfer devices used for chemotherapy administration once attached. If a second medication needs to be administered, the device should remain on the port, and be flushed before connecting the second medication.
➡️Syringes and Needles
● Remove needle, cannula, syringe and/or accessory items from sterile packaging immediately before use.
● In the clinical setting, avoid using Pharmacy Bulk Packages of sterile unwrapped syringes whenever possible. These bulk packages are primarily intended for use under a Laminar airflow system for medication compounding. If used for a mass immunization clinic or allergy testing, open a new, sterile package and discard any remaining syringes at the conclusion of the activity. Do not save for later use.
● Do not prepare medication in one syringe to transfer to another syringe (e.g., Healthcare personal draws up solution into a syringe then transfers the solution to a syringe that has the plunger removed or injects it into the bevel of the syringe).
● Never use a syringe for more than one patient even if the needle has been changed between patients. (Annex 2. One and Only Campaign)
● Use a new sterile syringe and a new sterile needle for each entry into a vial or IV bag.
● Utilize sharps safety devices (needles/syringes) to administer injections whenever possible.
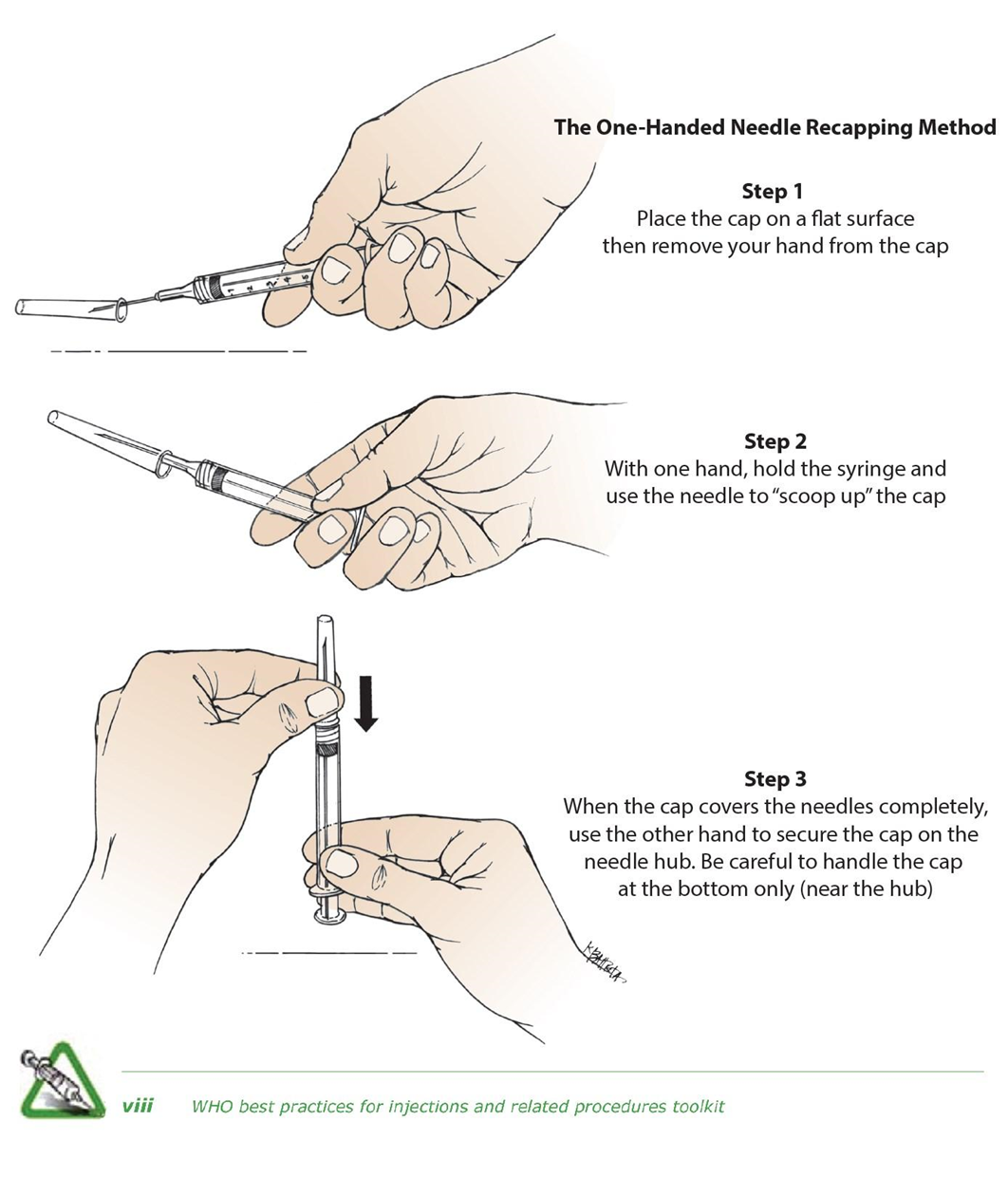
Discard syringes, needles, and cannulas in an approved sharps container/receptacle immediately after use and at the point of use. (Annex 3. One-hand scoop technique)
● Discourage the transporting of medication filled syringes in pockets or clothing.
●Draw up medication into a syringe as close to administration time as feasible.
●Label all syringes containing medication if not immediately administered.
|
Recommendations |
|
Ensure single-dose or single-use vials, ampules, and bags or bottles of parenteral solution are used for one patient only. Do not combine leftover contents for later use (Strong recommendation, High grade evidence) |
|
Use single-dose vials for parenteral medications whenever possible (Strong recommendation, High grade evidence) |
|
If multidose vials must be used, both the needle or cannula and syringe used to access the multidose vial must be sterile (Strong recommendation, High grade evidence) |
|
Dedicate multidose vials to a single patient whenever possible. If multidose vials are used for more than one patient, restrict the medication vials to a centralized medication area and do not bring them into the immediate patient treatment area (e.g., operating room, patient room/cubicle) (Strong recommendation, High grade evidence) |
|
Store multidose vials in accordance with the manufacturer’s recommendations; discard if sterility is compromised or questionable (Strong recommendation, High grade evidence) |
|
Consider the use of syringes with a sharps injury protection feature (SIP devices), as opposed to syringes without a sharps injury protection feature, by health care workers (HCWs) delivering intramuscular, subcutaneous or intradermal injectable medications to patients (Conditional recommendation, Moderate grade evidence) |
|
Consider the use of syringes with a re-use prevention feature (RUP devices), as opposed to devices without, by HCWs delivering intramuscular, subcutaneous or intradermal injectable medications to patients (Conditional recommendation, very low-quality evidence) |
|
Wear a surgical mask when placing a catheter or injecting material into the spinal canal or subdural space (i.e., during myelograms, lumbar puncture and spinal or epidural anesthesia) (Strong recommendation, Moderate grade evidence) |
1.8.3 Rationale
Outbreak can occur by unsafe injection practices such as syringe reuse and misuse of medications vials. The misuse of vials mainly involves the reuse of single-dose vials. These types of vials should only be used once for a single patient. Single-dose vials typically lack preservatives; therefore, using these vials more than once carries substantial risks for bacterial contamination, growth and infection.
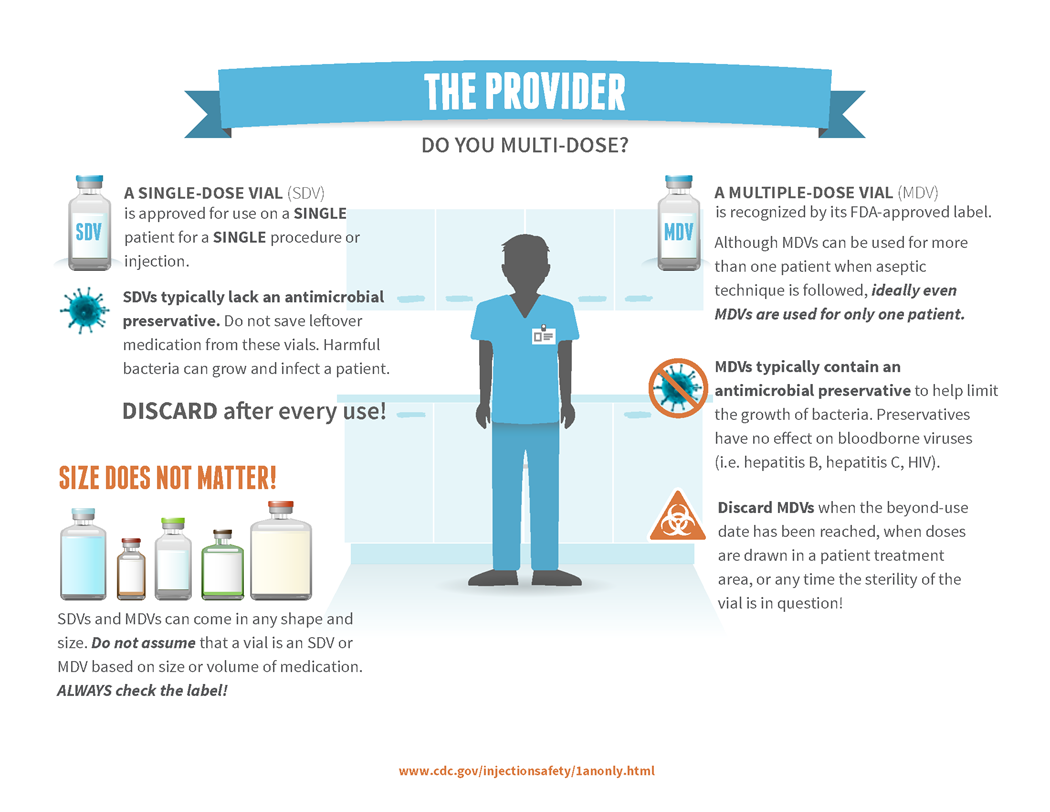
Medication Vials (Annex 4. Single dose vial vs Multi dose vial)
● Always follow the manufacturer’s instructions for storage and use.
● Check the manufacturer’s expiration date on all medication vials prior to use.
● Inspect vials and discard if sterility is known or suspected to be compromised. Examine vials for particulate matter, discoloration, or turbidity; if present, do not use and discard immediately.
● Read the vial label carefully. Vial size does not indicate whether or not a vial is single- use or multidose.
● Store vials with same-colored labels and/or same medication with different dosages separately.
● Discard any vial that has been placed on a known or visibly contaminated surface or a used procedure tray.
● Following an emergency event, discard all opened or needle-punctured vials.
➡️Single-dose vials
● Single-dose/single-use vials (SDVs) should only be used for a single patient during a single patient encounter.
● Vials must be discarded after this single use. Never return vials after this single use to stock in medication rooms, to medication/drug carts, etc.
● A new needle and a new syringe must be used if a single-dose/single-use vial must be entered more than once during a single procedure for a single patient.
● Never pool, combine, or store the contents of single-dose/single-use vials.
➡️Multi-dose vials
● Only vials that are clearly labeled by the manufacturer as appropriate for multiple uses may be used more than once.
● Whenever possible, use multi-dose vials (MDVs) for only one patient to reduce the risk of contamination.
● Use a new needle and new syringe for every entry into a multi-dose vial.
● Do not leave needles or other objects in vial entry diaphragms between uses. Doing so increases the risk of contamination.
● Using an alcohol pad, disinfect the rubber septum of vials before piercing by scrubbing vigorously.
● Allow the septum to air dry before inserting a needle or other device into the vial.
● If a multi-dose has been opened or accessed (e.g., needle-punctured) the vial should be dated at the time it is opened or accessed and discarded within 28 days unless the manufacturer specifies a different (shorter or longer) date for that opened vial.
●Multi-dose vials should not enter the patient care area and should be stored outside the immediate patient care area and under manufacturer storage recommendations.
➡️Flushing
● Use single-use containers for flush solutions, whenever possible.
● If a multidose vial must be used, use it for only one patient and then discard it.
● Use a new, unused sterile needle and new, unused sterile syringe for each entry into the vial.
➡️Point-Of-Care Testing (e.g., Blood Glucose, Coagulation Studies)
● Lancets
o Use single-use, auto-retracting lancing devices for each patient.
o Dispose of all capillary tubes and sharp devices in a sharps container immediately after use and at the point-of-care.
o Dedicate pen-like lancing devices to one patient and label the device with the patient’s name; do not reprocess for use on other patients.
● Testing devices (e.g., blood glucose meters)
o Whenever possible, blood glucose meters should be assigned to an individual person and not be shared.
o If blood glucose meters must be used on more than one patient, ensure they are labelled by the manufacturer for multiple patient use and include adequate instructions for disinfection of the meter between patients.
o Clean and disinfect multiple patient use meters (specifically for bloodborne pathogens and other infectious agents) after each patient use, using manufacturer recommendations. If the manufacturer does not provide instructions for cleaning and disinfection, then the testing meter should not be used for more than one patient.
o Avoid handling test strip containers with soiled gloves to avoid contamination. If a new test strip is needed, discard soiled gloves and perform hand hygiene before obtaining a new test strip.
o Clean visible blood and dirt from meters before disinfecting.
o Use an Environmental Protection Agency (EPA) / Egyptian Ministry of Health and Population (MohP) approved disinfectant and follow manufacturer’s contact time when disinfecting meters between patients.
● Use a mask to contain respiratory droplets when preparing and injecting solution into an intracapsular space (joint), the spine and during lumbar puncture.
● Disinfect catheter hubs, needleless connectors, and injection ports before accessing. Use either an antiseptic containing port protector cap or vigorously apply mechanical friction with chlorhexidine/alcohol, sterile 70% isopropyl alcohol, or other approved disinfectant swab.
➡️Injection Safety Breaches
Some types of infection control breaches, including, but not limited to, medication injection practices pose a risk of blood borne pathogen transmission. Infection control breaches are divided into two categories based on the qualitative risk assessment:
● Category A errors correspond to gross mistakes in infection control practices, typically with identifiable risk. The risk assessment is based on documented bloodborne pathogen transmission in association with similar practices in the past, or the observed or very high likelihood of blood exposure as a result of the breach. Examples of Category A errors include: 1) reuse of needles or syringes between patients; and 2) reuse of contaminated syringes to access multi-dose medication vials or intravenous fluid bags.
● Category B errors correspond to breaches of infection control where the likelihood of blood exposure resulting from the breach is uncertain but thought to be less than would occur with a Category A breach.
1.9 Indicators for Monitoring
1.9.1 Facility-level injection safety
● KPI Definition:
Percentage of healthcare facilities where all therapeutic injections are given
with new, disposable, single-use injection equipment.
● What to measure:
This indicator assesses the implementation of policies to ensure that all
health facilities practice injection safety.
● Rationale:
This indicator is relevant in countries that experience outbreaks of healthcare
injection-associated HIV infection, that have a history of unsafe injection
practices (more than 0.25 unsafe injections/per person/per year) and/or that
have a prevalence of hepatitis C infection >2%.
● Numerator:
Number of sampled healthcare facilities where all therapeutic injections are
given with new, disposable, single-use injection equipment
● Denominator:
Number of facilities sampled
● Method of measurement: For the numerator and denominator. Health facility
survey
● Disaggregation:
Type of site.
1.10 Plan to Update this National Clinical Guideline
1.10.1 This guideline
will be reviewed and updated at least every three years, or when new evidence
emerges that is likely to influence the recommendations.
- Acknowledgements
We would like to acknowledge the Infection Control Guidelines Committee for developing these guidelines.
Head of IPC Guidelines Committee
Professor Ghada Ismail (Professor of Clinical Pathology (Clinical Microbiology), Faculty of Medicine, Ain Shams University, Secretary of Supreme IPC Committee, SCUH, Member of WHO Global Guidelines Groups (GDG) for Infection Prevention)
Secretary of IPC Guidelines Committee
Professor Walaa Abd El-Latif (Professor of Medical Microbiology and Immunology, Faculty of Medicine Ain Shams University, IPC Consultant)
Members of the Committee
▪ Professor Amal Sayed (Deputy Manager of Environmental Affair, Infection Control Director, Cairo University Hospitals)
▪ Professor Amani El-Kholy (Clinical Pathology Department (Microbiology), Faculty of Medicine, Cairo University, Infection Control Consultant)
▪ Dr Asmaa Mohamed Abdelfatah Mohamed (Lecturer, Faculty of Nursing MTI University)
▪ Dr Gehan Mohamed Fahmy (Professor clinical microbiology ASUSH consultant infection control, Board member of IFIC EMERO region)
▪ Professor Hebatallah Gamal Rashed (Clinical Pathology Department (Microbiology), Faculty of Medicine, Assuit University, Infection Control Consultant)
▪ Dr Iman Afifi (Consultant Clinical Pathology (Microbiology) and IPC, Ain Shams University, Director IPC units of Ain Shams internal medicine and Geriatric hospitals
▪ Professor Maha El Touny (Department of internal medicine. Faculty of Medicine, Ain Shams University. Infection Control Consultant)
▪ Professor Nagwa Khamis (Emeritus Consultant Clinical Pathology (Microbiology) and IPC, ASU Director IPC Department and CEO Consultant IPC, CCHE-57357)
▪ Professor Nesrine Fathi Hanafi (Professor in Medical Microbiology and Immunology Faculty of Medicine Alexandria University, Head of Infection Prevention and Control, Alexandria University Hospitals)
▪ Dr. Reham Lotfy Abdel Aziz (Environmental Health Director, EEAA, Hazardous Waste Consultant, WMRA, Ministry of Environment)
▪ Professor Sherin ElMasry (Professor of Clinical Pathology, Ain Shams University, Chief Director of IPC ASU, Health Care Quality & Patient Safety Consultant)
▪ Dr Shimaa El-Garf (Coordinator): Clinical Pathology Specialist, Coordinator of HAI Surveillance and Audit Electronic System for University Hospitals, RLEUH- SCUH
- Abbreviations
1.1.1
CDC: Centers for Disease Control and Prevention
1.1.2
EPA: Environmental Protection Agency
1.1.3
FDA: Food and Agriculture Agency
1.1.4
HCW: Healthcare worker
1.1.5
HIV: Human Immunodeficiency Virus
1.1.6
IV: Intravenous
1.1.7
MDV: Multidose Vial
1.1.8
MoHP: Ministry of Health and Population
1.1.9
NSPs: Needle and syringe programs
1.1.10
RUP: Re-use prevention feature
1.1.11
SDV: Single Dose Vials
1.1.12
SIP: Sharp’s injury protection
1.1.13
SIS: Supervised injection sites
1.1.14
WHO: World Health Organization
- Glossary
1.1.1 Multi-dose vial: It is a bottle of liquid medication that contains more
than one dose of medication and is recognized by its FDA-approved label.
Healthcare providers use multi-dose vials to deliver a variety of medications
and vaccines. If handled safely, they can be accessed for more than one
patient. Healthcare providers should always use a new needle and syringe when
accessing the contents of a multi-dose vial. Reuse of needles or syringes can
contaminate medicine with germs, which can spread to others.
1.1.2 Single-dose vial: It is a bottle of liquid medication healthcare
providers give to a patient using a needle and syringe. A single-dose vial is
approved for use on a single patient for a single procedure or injection.
- Introduction
1.1.1
Safe injection practices refer to a set of guidelines and
techniques designed to reduce the health risks associated with injecting drugs.
The goal is to prevent the spread of bloodborne pathogens, while minimizing
harm to the individual using the drugs.
1.1.2
Safe injection involves using sterile syringes, disinfecting the
injection site, and safely disposing of used equipment to avoid harm to the
individual and the broader community.
1.1.3
Harm reduction strategies, such as needle and syringe programs
(NSPs) and supervised injection sites (SIS), play a crucial role in encouraging
safer behaviors among people who inject drugs, while also connecting them with
health services and support for substance use treatment.
1.1.4
By adopting safe injection practices and expanding harm reduction
services, public health systems can reduce the negative impacts of injection
drug use, improve individual health outcomes, and lessen the overall burden on
healthcare systems.
- Scope and Purpose
➡️
Scope
The scope of injection safety guidelines focuses on ensuring
safe injection practices, in various healthcare settings and for all persons
involved, including patients, healthcare workers, and the wider community.
Covers all aspects of safe injection practices, including preparation,
administration, and disposal of medications.
➡️Purpose
- Prevent the Transmission of
Infectious Diseases: By adhering to safe injection techniques, healthcare providers
and individuals can significantly reduce the risk of transmitting blood-borne
pathogens, such as HIV and hepatitis B and C.
- Minimize Complications Associated
with Injections: Safe injection practices help prevent complications like
abscesses, infections, and tissue damage, which can occur if injections are not
administered correctly.
- Promote Public Health: Safe injection practices
contribute to the overall health and well-being of a community by reducing the
burden of infectious diseases and improving healthcare outcomes.
- Ensure Patient Safety: By following safe injection
guidelines, healthcare providers can protect their patients from harm and build
trust in the healthcare system.
- Target Audience
1.1.1
All health workers (Primarily
aimed at workers who give injections or draw blood and at those who handle
medical waste).
1.1.2
Health facility
administrators.
1.1.3
Those responsible for
infection-control policy and practice.
1.1.4
Those responsible for
procurement of injection equipment and other health-care supplies.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
● Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
● Selecting only national and/or international guidelines
● Specific range of dates for publication (using Guidelines published or updated in 2013 and later)
● Selecting peer reviewed publications only
● Selecting guidelines written in English language
● Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
● Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
The following characteristics of the retrieved guidelines were summarized in:
● Developing organization/authors
● Date of publication, posting, and release
● Country/language of publication
● Date of posting and/or release
● Dates of the search used by the source guideline developers.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The committee decided to adapt from:
1. CDC Preventing Unsafe Injection Practices March 26, 2024
2. CDC Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings (2007) updated November 27, 2023
3. WHO guideline on the use of safety-engineered syringes for intramuscular, intradermal and subcutaneous injections in health care settings 2016
➡️Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
● GRADE working group:https://www.gradeworkinggroup.org/
● GRADE online training modules: http://cebgrade.mcmaster.ca/
Table (1) Quality and Significance of the four levels of evidence in GRADE
|
Quality |
Definition |
Implications |
|
High |
The guideline development group is very confident that the true effect lies close to that of the estimate of the effect |
Further research is very unlikely to change confidence in the estimate effect |
|
Moderate |
The guideline development group is moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibly that it is substantially different |
Further research is likely to have an important impact on confidence in the estimate of the effect and may change the estimate |
|
Low |
Confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the true effect |
Further research is very likely to have an important on confidence in the estimate of effect and is unlikely to change the estimate |
|
Very low |
The group has very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of the effect |
Any estimate of the effect is very uncertain |
Table (2) Factors that determine How to upgrade or downgrade the quality of evidence.
|
Downgrade in presence of |
Upgrade in presence of |
|
Study limitations. 1- Serious limitations 2- Very serious limitations |
Dose- response gradient. +1 Evidence of a dose-response gradient |
|
Consistency 1- Important inconsistency |
Direction of plausible bias + All plausible confounders would have reduced the effect |
|
Directness 1- Some uncertainty 2- Major uncertainty |
Magnitude of the effect +1 Strong, no plausible Confounder, consistent and direct evidence |
|
Precision 1- Imprecise data |
+2 very strong, no major threats to validity and direct evidence |
|
Reporting bias 1- High probability of reporting bias |
|
➡️The strength of the recommendations
The strength of a recommendation communicates the importance of adherence to the recommendation.
● Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
● Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations?
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Annexes
Annex 1. Safe Injection Myths CDC

Annex 2. One and Only Campaign

Annex 3. One-hand scoop technique
Annex 4. Single dose vial vs Multi dose vial
- References
1. CDC Preventing Unsafe Injection Practices March 26, 2024
2. CDC Guideline for Isolation Precautions: Preventing Transmission of Infectious Agents in Healthcare Settings (2007)updated November 27, 2023
3. Harm Reduction International. (2020). The Global State of Harm Reduction 2020. London: HRI Press.
4. WHO Strategic Information Guidelines, 2020 available at https://indicatorregistry.unaids.org/indicator/facility-level-injection-safety
5. Association for Professionals in Infection Control and Epidemiology (APIC), Safe Injection, Infusion, and Medication Vial Practices in Health Care, 2016
6. WHO guideline on the use of safety-engineered syringes for intramuscular, intradermal and subcutaneous injections in health care settings 2016
7. World Health Organization. (2012). Guide to Developing and Implementing Needle and Syringe Programmes. Geneva: WHO Press.
8. Marshall, B. D., Milloy, M. J., Wood, E., Montaner, J. S., & Kerr, T. (2011). Reduction in overdose mortality after the opening of North America’s first medically supervised safer injecting facility: a retrospective population-based study. The Lancet, 377(9775), 1429-1437.