THE DIAGNOSIS, TREATMENT, AND PREVENTION OF THROMBOSIS IN PAEDIATRIC AGE GROUPS
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | THE DIAGNOSIS, TREATMENT, AND PREVENTION OF THROMBOSIS IN PAEDIATRIC AGE GROUPS |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 6 March 2025" Download Guideline
- Recommendations
Part I: Diagnosis
|
CPGL Source |
Recommendation |
SoR |
|
|
The clinical presentation of VTE in children depends on several factors, including the patient's age, anatomic location of the thrombosis, affected organ system(s), characteristics of the thrombus (occlusive vs. nonocclusive), and chronicity. ₋ Common symptoms of an acute extremity deep venous thrombosis (DVT) include unilateral limb pain, swelling, and redness of the involved extremity. ₋ In the upper extremity, in specific, DVT extension and occlusion of the superior vena cava (SVC) can result in SVC syndrome characterized by swelling of the neck and face; dilatation of the superficial collateral venous circulation of the arms, neck, and chest; bilateral periorbital edema; and headaches. ₋ In the case of CVC-associated DVTs, the initial presenting symptom is often dysfunction of the CVC. ₋ Chronic DVTs, as in the case of adults, can presents with signs of chronic venous obstruction or post-thrombotic syndrome (PTS) including edema, venous stasis dermatitis, limb pain, and skin ulceration. ₋ In neonates, acute DVT may present with new-onset thrombocytopenia. ₋ Renal vein thrombosis classically presents with thrombocytopenia and hematuria and, if bilateral, can lead to renal insufficiency. In the neonatal period, it can also manifest with a palpable flank mass on examination, while in older children it is often associated with nephrotic syndrome and presents with peripheral and periorbital edema. ₋ Thrombocytopenia can also be one of the presenting symptoms in intracardiac thrombosis and portal vein thrombosis with the latter also resulting in upper gastrointestinal bleeding in children.
|
GPS |
|
|
I- Laboratory investigations |
|
|
BSH 2011 ad 2021 |
Acute venous thrombosis |
|
|
· Laboratory investigations are required to aid the exclusion of systemic disorders in children presenting with a suspected VTE. |
Strong |
|
|
· Haematology investigations (full blood count, clotting screen) and renal function should be undertaken to confirm safe baselines prior to anticoagulation. |
Strong |
|
|
· D-Dimers should not be used to exclude VTE in children. |
Conditional |
|
|
BSH 2011 ad 2021 |
Heritable and acquired thrombophilia |
|
|
· Routine testing for heritable thrombophilia with a first episode of VTE is not indicated. |
Strong |
|
|
· Children presenting with an unprovoked VTE should be tested for the presence of anti-phospholipid antibodies and those with persistently positive results should remain on long-term anticoagulation.
|
Strong |
|
|
BSH 2011 ad 2021 |
Purpura fulminant (PF) |
|
|
· Neonates and children with PF should be tested urgently for protein C and S deficiency. |
Strong |
|
|
· Children with early onset spontaneous thrombotic events should be screened for antithrombin (AT) deficiency. |
Strong
|
|
|
|
II- Radiological |
|
|
BSH 2011 ad 2021 |
CVL and non-CVL related VTE in the upper limb (UL) |
|
|
· Ultrasound (US) is recommended for the initial assessment of the peripheral upper limb, axillary, subclavian and internal jugular veins but may be relatively insensitive for the detection of central intra-thoracic VTE. |
Strong
|
|
|
· Contrast MRV is recommended for assessing the central veins for VTE. |
Strong |
|
|
· Multi-detector computerized tomography (CT) venography (MDCT venography) may be considered for the assessment of the central veins if MRV is unavailable. Magnetic resonance imaging (MRI) should always be preferred to CT due to radiation dose considerations in children. |
Conditional |
|
|
BSH 2011 ad 2021 |
CVL and non-CVL related VTE in the lower limb (LL) |
|
|
· Doppler US is recommended to assess the LL venous system for VTE. |
Strong |
|
|
· If the US is normal and the clinical suspicion of VTE remains high this should be repeated after a week to assess for proximal progression of any calf vein thrombus. |
Strong
|
|
|
· MRV should be considered in children with suspected proximal extension of femoral VTE. |
Conditional |
|
|
· Patients presenting with bilateral lower limb DVT should have their inferior vena cava (IVC) imaged to exclude IVC thrombosis. |
Strong |
|
|
BSH 2011 ad 2021 |
For blocked central venous lines |
|
|
· A chest X-Ray is recommended to visualize the CVL position. |
Conditional |
|
|
· Doppler US, conventional venography or contrast enhanced MRV may be required to exclude large vessel thrombosis. |
Conditional |
|
|
BSH 2011 ad 2021 |
For Diagnosis of cerebral sino-venous thrombosis |
|
|
· Urgent brain MRI including T2* imaging and MRV to detect both intraparenchymal haemorrhage and sinus thrombosis. |
Strong |
|
|
· If urgent MRI is unavailable, a pre- and post-contrast CT scan with CT venography (CTV) should be performed. |
Strong |
|
|
· Children in whome CSVT is suspected on CT could have confirmatory MRI replaced by CTV if MRI/MRV is not available. |
Strong |
|
|
BSH 2022 |
₋ We suggest testing for PNH in patients with thrombosis at unusual sites and abnormal haematological parameters (i.e., cytopenia and abnormal red cell indices) or evidence of haemolysis (i.e., raised lactate dehydrogenase, bilirubin and reticulocyte count). |
Conditional |
|
₋ We recommend testing for myeloproliferative neoplasms (MPN) panel (including JAK2 V617F, JAK2 exon 12, CALR, MPL mutation analysis) in patients with thrombosis at unusual sites and with full blood count abnormalities suggestive of a myeloproliferative neoplasm. |
Strong |
|
|
₋ Screening for antiphospholipid antibodies is recommended following unprovoked VTE because this may alter management including choice of antithrombotic therapy. |
Strong |
|
|
₋ Screening for antiphospholipid antibodies is suggested in patients with VTE provoked by a minor risk factor as this may alter management including choice of antithrombotic therapy. |
Conditional |
|
|
₋ Patients with acute multiple thrombotic events and evidence of organ failure suggestive of CAPS should be tested for antiphospholipid antibodies. |
Strong |
|
|
₋ As antiphospholipid syndrome (APS) is an acquired thrombophilia, screening for antiphospholipid antibodies is not recommended in family members of patients with thrombosis. |
Strong |
|
|
|
Pediatric patients may present with nonspecific (i.e., cough, crackles/ rales, tachycardia, and persistent tachypnea) or mild symptoms in the setting of a PE, especially when the PE is limited to the segmental branches of the pulmonary arteries. Adolescents most frequently present with pleuritic chest pain, dyspnea, cough, and hemoptysis. |
GPS |
|
BSH 2011 |
If available, isotope lung scanning may be considered as the initial imaging investigation, providing the chest X-ray is normal and there is no significant concurrent cardiopulmonary disease. Otherwise, CT pulmonary angiography (CTPA) is recommended as the initial imaging modality for suspected PE. |
Strong |
|
Non-diagnostic isotope lung scanning should be followed by further imaging. |
Strong |
|
|
Patients with a good quality negative CTPA do not require further investigation or treatment. |
Strong |
|
|
Pulmonary magnetic resonance angiography (MRA) should be considered as an alternative to CTPA when iodinated contrast injection or radiation is a significant consideration.
|
Conditonal |
|
|
|
We suggest to assess clues from history and physical examination History clues: Assess for: • Interventional catheterization especially balloon dilatation, valvotomy procedure. • Larger central line catheter size. • Repeated central catheter manipulations. • Longer duration of cannulation. Physical examination: Evaluation of the limb for signs and symptoms of acute peripheral arterial occlusion in the form of: • Non-Palpable pulses. • Difference in blood pressure > 10 mmHg between right and left legs. • Decrease in skin temperature. • Skin discoloration (pallor or cyanosis). • Prolonged capillary refill time. |
GPS |
|
|
Children presenting with sudden onset of the following symptoms are at high risk of stroke and should undergo immediate neurological assessment and consideration of urgent neuroimaging: (i) focal weakness, (ii) visual or speech disturbances, (iii) limb incoordination or ataxia, (iv) altered mental status, (v) headache, (vi) signs of raised intracranial pressure, or (vii) seizures with additional neurological symptoms. |
GPS |
|
In children presenting with neurological symptoms or signs relevant for stroke, the use of adult stroke recognition tools to differentiate childhood stroke from its mimics are not recommended in their current form. |
GPS |
|
|
BSH 2022 |
In patients with stroke, an abnormal full blood count should prompt consideration for testing with an MPN panel and for PNH. |
Conditional |
|
BSH 2022 |
Testing for heritable thrombophilia is not recommended in patients with stroke, regardless of age. |
Strong |
|
BSH 2022 |
Testing for antiphospholipid antibodies should be considered in young patients in the absence of identifiable risk factors of cardiovascular disease because this may alter management including choice of antithrombotic therapy.
|
Strong |
|
|
• Unprovoked thrombosis at an early age • Recurrent episodes of thrombosis or thrombophlebitis • Strong family history of thrombosis or thrombotic events especially at an early age (< 45 years) • Thrombosis at multiple or unusual locations including in cerebral and jugular • Migratory episodes or diffuse form of thrombotic events • Severity out of proportion to any recognized known stimulus for thrombosis • Unexplained neonatal thrombosis or fetal loss • Skin necrosis particularly if on coumarins or warfarin • Unexplained, prolonged, activated partial thromboplastin time • Patients with idiopathic thrombocytopenia, SLE or recurrent thrombosis including deep venous thrombosis, pulmonary embolus or superficial venous thrombosis Thrombosis in arteries with the absence of arterial disease |
GPS |
|
BSH 2011 ad 2021 |
Children with early onset spontaneous thrombotic events should be screened for AT deficiency. |
Strong |
|
BSH 2022 |
Neonates and children with purpura fulminans should be tested urgently for protein C and S deficiency. |
Strong |
|
BSH 2022 |
In neonates with multiple unexplained thrombosis, especially with clinical evidence suggestive of catastrophic antiphospholipid syndrome (CAPS), testing for antiphospholipid antibodies and heritable thrombophilia should be considered |
Conditional |
|
BSH 2022 |
Thrombophilia screening is not routinely recommended for neonatal stroke. |
Conditional |
|
BSH 2011 |
Routine testing for heritable thrombophilia in unselected children presenting with a first episode of VTE is not indicated. |
Strong |
|
BSH 2011 |
Testing for heritable thrombophilia after a first episode of VTE has uncertain predictive value for recurrence. |
Strong |
|
BSH 2011 |
We do not recommend offering routine thrombophilia testing to first-degree relatives of people with a history of VTE. |
Conditional |
|
BSH 2022 |
Genetic testing for variants in genes (e.g., MTHFR, SERPINE1 variants (PAI-1plasma level)) without a clinically significant link to thrombosis is not recommended. |
Conditional |
|
BSH2022 |
Routine testing of coagulation factors to assess the risk of thrombosis is not currently recommended. |
Conditional |
|
BSH 2022 |
The presence of a patent foramen ovale (PFO ) in patients with a stroke is not an indication for thrombophilia testing. |
Conditional |
|
BSH 2022 |
We suggest selective testing of asymptomatic first-degree relatives of probands with protein C, protein S and antithrombin deficiency where this may influence the management and life choices depending on personal circumstances. |
Conditional |
|
BSH 2011 |
Children presenting with an unprovoked VTE should be tested for the presence of anti-phospholipid antibodies and those with persistently positive results should remain on long-term anticoagulation. |
Strong |
|
BSH 2018 |
Assessment of an individual’s VTE risk, based on the factors listed, should be made at diagnosis and at appropriate times during the course of their illness (e.g. re-admission to hospital), particularly in adolescent patients, given their higher baseline risk of thrombosis. |
Conditional |
|
|
Patient-related factors Age >10 years (particularly adolescents) Inherited thrombophilia Personal or family history of VTE Obesity Immobilisation Concurrent infection Disease-related factors Presence of pulmonary/intrathoracic/ pelvic disease Sarcomas (rhabdomyosarcoma, Ewing sarcoma, osteosarcoma) Acute promyelocytic leukaemia Acute lymphoblastic leukaemia Lymphomas Treatment-related factors Major surgery Use of CVLs Induction chemotherapy for ALL: concurrent administration of corticosteroids and asparaginase |
|
|
BSH 2018 |
Routine thrombophilia screening is not recommended outside the context of large, well-conducted prospective studies intended to assess the impact of prothrombotic defects in modern treatment protocols |
Strong |
|
BSH 2022 |
Testing for deficiencies of physiological anticoagulants should be performed only after 3 months of anticoagulation for acute thrombosis. |
Conditional |
Part II: Treatment
|
CPGL Source |
Recommendation |
SoR |
|
ASH 2018 |
• We recommend using anticoagulation rather than no anticoagulation in pediatric patients with symptomatic DVT or PE |
Strong |
|
ASH 2018 |
• We suggest either using anticoagulation or no anticoagulation in pediatric patients with asymptomatic DVT or PE |
Conditional |
|
ASH 2018 |
• We suggest against using thrombolysis followed by anticoagulation; rather, anticoagulation alone should be used in pediatric patients with DVT |
Conditional |
|
ASH 2018 |
• We suggest using either low-molecular-weight heparin or vitamin K antagonists in pediatric patients with symptomatic DVT or PE |
Conditional |
|
BSH 2011 |
• The use of thrombolytic therapy is not indicated for the majority of children with VTE but should be considered in the presence of extensive thrombosis, particularly those involving the pelvic veins, SVC, IVC or intracardiac sites |
Strong |
|
ASH 2018 |
• We suggest against using thrombolysis followed by anticoagulation; rather, anticoagulation alone should be used in pediatric patients with submassive PE |
Conditional |
|
ASH 2018 |
• We suggest using thrombolysis followed by anticoagulation, rather than anticoagulation alone, in pediatric patients with PE with hemodynamic compromise |
Conditional |
|
ASH 2018 |
• We suggest using anticoagulation for <3 months rather than anticoagulation for > 3 months in pediatric patients with provoked DVT or PE |
Conditional |
|
ASH 2018 |
• We suggest using anticoagulation for 6 to 12 months rather than anticoagulation for > 6 to 12 months in pediatric patients with unprovoked DVT or PE |
Conditional |
|
BSH 2011 |
• In recurrent idiopathic VTE and children with antiphospholipid syndrome: duration should be life-long. |
Strong |
|
BSH 2011 |
• Anticoagulation should be initiated with low molecular weight heparin (LWMH) followed by warfarin (INR 2.5) or continuingLMWH. |
Strong |
|
BSH 2011 |
• Unfractionated heparin (UFH) may be used for initial therapy where rapid reversal of anticoagulation may be required. |
Conditional |
|
BSH 2011 |
• Ongoing therapy with LMWH may be preferable in infants under 1 year of Age. |
Conditional |
|
ACCP 2012 |
• We recommend initial treatment with UFH or LMWH for at least 5 days • For ongoing therapy, we recommend LMWH or UFH. For patients in whom clinicians will subsequently prescribe VKAs, we recommend beginning oral therapy as early as day 1 and discontinuing UFH/LMWH on day 6 or later than day 6 if the INR has not exceeded 2.0 compared with no therapy. |
Strong |
|
|
Considerations for initiating DOACs for treatment of pediatric VTE • Stable patient (unlikely to need an urgent procedure) • Tolerating good oral intake or on full nasogastric feeds • Adequate renal and hepatic function • Unlikely to have antiphospholipid antibody syndrome • ≥5 days of parenteral anticoagulation • No drug interactions (consult clinical pharmacy) • Gestational age > 37 weeks and weight > 2.6 kg |
GPS |
|
ASH 2018 |
We suggest against using thrombectomy followed by anticoagulation; rather, anticoagulation alone should be used in pediatric patients with symptomatic DVT or PE. |
Conditional |
|
ASH 2018 |
We suggest against using inferior vena cava (IVC) filter; rather anticoagulation alone should be used in pediatric patients with symptomatic DVT or PE. |
Conditional |
|
ACCP 2012 |
We suggest that therapeutic UFH in children is titrated to achieve a target range of anti-Xa activity of 0.35 to 0.7 units/mL or an activated partial thromboplastin time (aPTT) range that correlates to this anti-Xa range or to a protamine titration range of 0.2 to 0.4 units/mL. |
Conditional |
|
ACCP 2012 |
We suggest, for neonates and children receiving either once- or twice-daily therapeutic LMW that the drug be monitored to a target anti-Xa activity range of 0.5 to 1.0 units/mL in a sample taken 4 to 6 h after subcutaneous injection or 0.5 to 0.8 units/mL in a sample taken 2 to 6 h after subcutaneous injection. |
Conditional |
|
ACCP 2012 |
We suggest, for children receiving vitamin K antagonists (VKAs), that the drug be monitored to a target international normalized ratio (INR) of 2.5 (range, 2.0-3.0), except in the setting of prosthetic cardiac valves where we suggest adherence to the adult recommendations. |
Conditional |
|
ASH 2018 |
We suggest using anticoagulation, rather than no anticoagulation, in pediatric patients with right atrial thrombosis. |
Conditional |
|
ASH 2018 |
We suggest against using thrombolysis or surgical thrombectomy followed by standard anticoagulation; rather, anticoagulation alone should be used in pediatric patients with right atrial thrombosis |
Conditional |
|
ASH 2018 |
We suggest no removal, rather than removal, of a functioning CVAD in pediatric patients with symptomatic CVAD-related thrombosis who continue to require venous access. |
Conditional |
|
ASH 2018 |
We suggest either removal or no removal of a functioning CVAD in pediatric patients who have symptomatic CVAD-related thrombosis with worsening signs or symptoms, despite anticoagulation, and who continue to require venous access. |
Conditional |
|
ASH 2018 |
We recommend removal rather than no removal of a non-functioning or unneeded CVAD in pediatric patients with symptomatic CVAD-related thrombosis. |
Strong |
|
ASH 2018 |
We suggest delayed removal of a CVAD until after initiation of anticoagulation (days), rather than immediate removal in pediatric patients with symptomatic CVAD–related thrombosis who no longer require venous access or in whom the CVAD is nonfunctioning |
Conditional |
|
ASH 2018 |
We recommend using anticoagulation rather than no anticoagulation in pediatric patients with CSVT without hemorrhage. |
Strong |
|
ASH 2018 |
We suggest using anticoagulation rather than no anticoagulation in pediatric patients with CSVT with hemorrhage |
Conditional |
|
ASH 2018 |
We suggest against using thrombolysis followed by standard anticoagulation; rather, anticoagulation alone should be used in pediatric patients with CSVT. |
Conditional |
|
BSH 2011 ad 2021 |
₋ Anticoagulation should be continued for |
|
|
· Three months if there was a clear and treated precipitating factor e.g. infection. |
Strong |
|
|
· Six months if there is no identified precipitant. |
Strong |
|
|
· Anticoagulation may need to be continued for longer in patients where there is an ongoing risk factor (e.g. continuing treatment with asparaginase), in those with recurrent idiopathic CVT and in those with ongoing symptoms or signs attributable to venous hypertension) - duration should be considered on an individual basis. |
Conditional |
|
|
₋ Re-imaging should be undertaken prior to stopping anticoagulation in patients with ongoing symptoms attributable to venous hypertension (e.g. headache, vomiting, papilloedema, visual obscurations, visual field deficit) or with progressive neurological signs. ₋ Re-imaging is not required in patients with stable neurological signs, unless consideration is being given to extending anticoagulant therapy, in which case it may be helpful to establish whether or not recanalization has occurred. |
Conditional |
|
|
ASH 2018 |
• We suggest using anticoagulation rather than no anticoagulation in neonates with RVT. |
Conditional |
|
ASH 2018 |
• We recommend against using thrombolysis, followed by standard anticoagulation; rather, anticoagulation alone should be used in neonates with nonlife-threatening RVT. |
Conditional |
|
ASH 2018 |
• We suggest using thrombolysis followed by standard anticoagulation rather than anticoagulation alone in neonates with life-threatening RVT. |
Conditional |
|
ACCP 2012 |
• For unilateral renal vein thrombosis (RVT) in the absence of renal impairment or extension into the inferior vena cava (IVC), ACCP suggest either (1) supportive care with radiologic monitoring for extension of thrombosis (if extension occurs we suggest anticoagulation) or (2) anticoagulation with UFH/LMWH or LMWH in therapeutic doses rather than no therapy. |
Conditional |
|
ACCP 2012 |
• For unilateral RVT that extends into the IVC, ACCP suggest anticoagulation with UFH/LMWH or LMWH |
Conditional |
|
ACCP 2012 |
• For bilateral RVT with evidence of renal impairment, ACCP suggest anticoagulation with UFH/LMWH or initial thrombolytic therapywith tissue plasminogen activator (tPA) followed by anticoagulation with UFH/LMWH |
Conditional |
|
ACCP 2012 |
• For unilateral renal vein thrombosis (RVT) in the absence of renal impairment or extension into the inferior vena cava, ACCP suggest a total duration of between 6 weeks and 3 months rather than shorter or longer durations of therapy |
Conditional |
|
ACCP 2012 |
• For unilateral RVT that extends into the IVC, ACCP suggest anticoagulation with UFH/LMWH or LMWH for a total duration of between 6 weeks and 3 months |
Conditional |
|
ASH 2018 |
We suggest using anticoagulation rather than no anticoagulation in pediatric patients with PVT with occlusive thrombus, post liver transplant, and idiopathic PVT. |
Conditional |
|
ASH 2018 |
We suggest using no anticoagulation rather than anticoagulation in pediatric patients with PVT with nonocclusive thrombus or portal hypertension |
Conditional |
|
ASH 2018 |
In children who will not be anticoagulated, follow-up monitoring is important, because extension of thrombus or organ dysfunction may require reconsideration of treatment options. |
Conditional |
|
BSH 2018 |
Children with VTE unrelated to CVLs should receive anticoagulation for an initial period of 3 months. |
Strong |
|
BSH 2018 |
Continuation of treatment beyond 3 months should be considered in patients with active cancer or other ongoing risk factors. |
Conditional |
|
BSH 2018 |
Children with cancer who are found to have incidental VTE should be considered for systemic anticoagulation as per protocols for symptomatic disease. However, if the thrombosis is solely CVL-related, then it is reasonable to withhold anticoagulation initially and monitor the patient. |
Conditional |
|
BSH 2018 |
LMWH is the anticoagulant of choice for VTE in children with malignancy |
Conditional |
|
BSH 2018 |
Unfractionated heparin can be also used for the initial treatment of established VTE for patients with cancer when LMWH or direct oral anticoagulants are contraindicated, or not available. |
Conditional |
|
BSH 2018 |
Routine antithrombin supplementation is not recommended during LMWH or unfractionated heparin (UFH) anticoagulation. |
Conditional |
|
BSH 2018 |
Routine measurement of peak anti-Xa activity, with a target range of 0.5–1.0 units/mL, is recommended for children receiving LMWH. |
Conditional |
|
BSH 2018 |
Coagulopathies should be corrected and fibrinogen levels maintained above 1 g/l in children on anticoagulant therapy. |
Conditional |
|
|
At least 3 months or as long as active cancer or cancer therapy. • For non–catheter-associated DVT or PE recommend indefinite anticoagulation while cancer is active, under treatment, or if risk factors for recurrence persist. • For symptomatic catheter-associated DVT, consider anticoagulation treatment for at least 3 months or as long as the catheter is in place. |
GPS |
|
BSH 2018 |
Therapeutic anticoagulation may be continued while theplatelet count is >50 x 109/l. |
Strong |
|
BSH 2018 |
Outside of these scenarios, an alternative approach of reducing the anticoagulation dose to 50% when the platelet count is between 25 and 50 x109/l and temporarily interrupting anticoagulation when <25 x 109/l could be considered. |
Conditional |
|
BSH 2018 |
For children with normal renal function on LMWH, the last therapeutic dose should be given at least 24 h prior to an invasive procedure and the last prophylactic dose at least 12 h prior to an invasive procedure. LMWH should be restarted no sooner than 4–6 h after a procedure |
Strong |
|
BSH 2018 |
For children on warfarin, the last dose should be given 4– 5 days prior to an invasive procedure. Bridging anticoagulation with LMWH when the International Normalised Ratio (INR) becomes sub-therapeutic will be required for individuals within 1 month of a VTE. This should also be considered for up to 3 months after a VTE in those judged to be at particularly high risk of recurrence |
Conditional |
|
BSH 2018 |
Removal of the CVL is not necessary if it is still required for venous access, in a good position and functioning well |
Conditional |
|
BSH 2018 |
Symptomatic CVL-related VTE should be treated with anticoagulation for a minimum of 3 months |
Strong |
|
BSH 2018 |
Further doses of asparaginase may be administered following an asparaginase-related VTE provided they are covered by prophylactic or therapeutic anticoagulation |
Strong |
|
BSH 2018 |
Such anticoagulation should be continued for at least 3 weeks following each dose of pegylated asparaginase |
Strong |
|
BSH 2018 |
We suggest that that re-exposure to asparaginase is delayed until symptoms of the initial VTE have improved and, in the case of CVT, repeat imaging demonstrates stabilisation or improvement of the thrombosis |
Conditional |
|
|
In the event of VTE recurrence, three options can be considered: (1) increase LMWH by 20–25% or switch to direct oral anticoagulants; (2) for direct oral anticoagulants, switch to LMWH; and (3) for vitamin K antagonist, switch to LMWH or direct oral anticoagulants (guidance, based on evidence of very low quality and an unknown balance between desirable and undesirable effects). |
GPS |
|
ACCP 2012
|
|
GPS |
Part III: Prevention
|
UW Health 2023 |
All hospitalized patients 6 months of age or older should be evaluated for both bleeding and VTE risk within 24 hours of admission, upon transferring level of care, and periodically during hospital stay (every 48-72 hours)
|
Strong |
||||||||
|
|
VTE Risk factors: Acute conditions ₋ Reduced mobility longer than 48 hrs ₋ Central venous access device ₋ Activee infection ₋ Major trauma or burn ₋ Major surgery ₋ Critically ill ₋ Hypoalbuminemia ₋ Blood transfusion withing the previous 48 hours Chronic medical condition ₋ Post-pubertal and/or age 12 years or older ₋ Autoimmune disorders associated with thrombosis ₋ Thrombophilic condition ₋ Active malignancy ₋ Obesity Historical factors ₋ Asparaginase therapy within the previous 30 days ₋ Recentt surgery within the past 30 days ₋ History of thrombosis ₋ Family history of VTE in a 1st degree relative < 40 year old at time of clot. |
GPS |
||||||||
|
|
Risk Factor Stratification definitions: Low VTE risk: No altered mobility and 0-2 risk factors Moderate VTE risk: ₋ No altered mobility and 3-4 risk factors ₋ Altered mobility and 0-2 other risk factors High VTE risk: ₋ No altered mobility and ≥ 5 risk factors ₋ Altered mobility and ≥ 3 other risk factors |
GPS |
||||||||
|
|
Risk assessment
|
|
||||||||
|
UW Health 2023 |
₋ If a patient meets at least one of the criteria of the “Chemical Prophylaxis NOT Recommended” list, avoid chemical prophylaxis as risk outweighs benefit. ₋ If patient meets at least one of the following criteria in the “Consider Avoiding Chemical Prophylaxis” list, consider avoiding chemical prophylaxis. - Consider consulting hematology for recommendations if patient is considered at high risk for VTE, but also with high bleeding risk. - Uncontrolled hypertension is defined as systolic or diastolic blood pressure greater than 95thpercentile for age, height, and gender. |
Strong
Conditional
GPS
GPS |
||||||||
|
UW Health 2023 |
Recommendations for specific factors associated with increased bleeding risk:
|
Strong |
||||||||
|
|
Non-pharmacological (Physical) methods for thromboprophylaxis |
|
||||||||
|
|
- Children should be encouraged to reduce their risk of VTE by maintenance of good hydration, early mobilisation whenever possible. |
GPS |
||||||||
|
BSH 2011 ad 2021 |
- The use of physical methods for VTE risk reduction should be considered in older children and adolescents who are at increased risk of VTE. |
Strong |
||||||||
|
BSH 2011 ad 2021 |
- In suitable patients, physical methods may be helpful when there is a high risk of bleeding or to complement anticoagulant-based prophylaxis when there is a particularly high risk of VTE. |
Strong |
||||||||
|
|
- Mechanical prophylaxis methods may include sequential compression device (SCD), graduated compression stockings (GCS), or consulting physical therapy and/or occupational therapy to assist with movement of the patient . |
GPS |
||||||||
|
UW Health 2023 |
- Contraindications to mechanical prophylaxis: · Extremity has acute fracture, · Extremity has peripheral IV access, · Skin condition affecting extremity (i.e., dermatitis, burn, etc.), · Unable to achieve correct fit due to patient size, · Lower extremity peripheral arterial insufficiency. |
Strong |
||||||||
|
|
Pharmacological thromboprophylaxis (LMWH, VKA) |
|
||||||||
|
BSH 2011 ad 2021 |
- There is no evidence for the use of aspirin for VTE prophylaxis in children. |
Conditional |
||||||||
|
BSH 2011 ad 2021 |
- Children, particularly adolescents, with multiple risk factors for VTE should be considered for thromboprophylaxis with LMWH. |
Conditional |
||||||||
|
|
1. Low molecular weigh heparin (LMWH) |
|
||||||||
|
UW Health 2023 |
· Enoxaparin is the preferred pharmacologic prophylaxis agent for pediatric patient. |
Strong |
||||||||
|
|
· Use of subcutaneous (SQ) injections is preferred due to lack of evidence of intravenous administration in the prophylactic setting. |
GPS |
||||||||
|
UW Health 2023
|
· Avoid enoxaparin or heparin if patient has hypersensitivity to enoxaparin, heparin, pork products, or any component of the formulation. |
Strong |
||||||||
|
|
· Recommended ‘prophylaxis’ doses are usually half the following treatment doses: ₋ Dalteparin: 100 u/kg twice per day or 200 u/kg once per day s.c. ₋ Enoxaparin: 1 mg/kg twice per day or 2 mg/kg once per day s.c. ₋ Tinzaparin: 175 u/kg once per day s.c. · Infants <8 weeks of age and/or <5 kg require 50% larger doses e.gDalteparin 150 u/kg twice per day and Enoxaparin 1.5 mg/ kg twice per day, possibly due to a larger volume of distribution and/or reduced anti thrombin levels. · Target anti-Xa activity taken 4 h following subcutaneous injection. Prophylactic 0.1–0.4 u/ml. |
GPS |
||||||||
|
|
2. Warfarin |
|
||||||||
|
|
₋ Initial loading dose of 0.2 mg/kg p.o. for 2 d. |
GPS |
||||||||
|
₋ Subsequent dose adjustments should be based on the INR result. |
||||||||||
|
|
₋ Reversal of warfarin with Vitamin K: Vitamin K can be given p.o. or i.v., dosing regimens vary but doses of 30 µg/kg or 0.3–5 mg have been reported to be effective (Bolton-Maggs& Brook, 2002). |
GPS |
||||||||
|
|
3. Rivaroxaban |
|
||||||||
|
UW Health 2023
|
· Rivaroxaban, an oral anticoagulant with FDA approval in the pediatric population, may be considered for prophylaxis in patients with high VTE risk if appropriate based on patient characteristics |
Conditional |
||||||||
|
UW Health 2023
|
· Rivaroxaban may be used based on individual considerations. - Only use rivaroxaban in infants weighing at least 2.6 kg whose serum creatinine is less than the 97.5thpercentile and children or adolescents with eGFR > 50 mL/min/1.73 m2. - Patients should receive at least five days of parenteral anticoagulation and at least ten days of enteral feeding prior to starting rivaroxaban for VTE prophylaxis. |
Strong
Strong
Strong |
||||||||
|
|
Recommendations for patients with special risk factors |
|
||||||||
|
ASH 2018 |
Patients with congenital purpura fulminant due to homozygous protein C deficiency The ASH guideline panel suggests using either liver transplantation or no liver transplantation (anticoagulation or protein C replacement) in pediatric patients with congenital purpura fulminans due to homozygous protein C deficiency. |
Conditional |
||||||||
|
BSH 2011 ad 2021 |
Patients with CVL - Thromboprophylaxis for primary prevention of CVL related thrombosis is not recommended. - Consideration may be given to the use of heparin-bonded CVLs, if available, for short-term use. |
Conditional
Conditional |
||||||||
|
BSH 2018 |
Children with cancer - Simple measures should be encouraged: early mobilisation, good hydration and prompt removal of central venous lines (CVLs) when no longer required. - Routine thromboprophylaxis in children with cancer is not recommended, but should be considered in adolescents with multiple risk factors. Patient-related factors Age >10 years(particularly adolescents) Inherited thrombophilia Personal or family history of VTE Obesity Immobilisation Concurrent infection Disease-related factors Presence of pulmonary/intrathoracic/pelvic disease Sarcomas (rhabdomyosarcoma,Ewing sarcoma, osteosarcoma) Acute promyelocytic leukaemia Acute lymphoblastic leukaemia Lymphomas Treatment-related factors Major surgery Use of CVLs Induction chemotherapyfor ALL: concurrentadministration of corticosteroidsand asparaginase |
Conditional
Conditional |
||||||||
|
BSH 2018 |
Children with cancer and CVL - Removal of the CVL is not necessary if it is still required for venous access, in a good position and functioning well |
Conditional |
||||||||
|
BSH 2018 |
- For children requiring a CVL and at high risk of VTE, an internal device (port) should be used in preference to an external tunnelled device (Hickman or Broviac catheter) |
Conditional |
||||||||
|
BSH 2018 |
- Until further paediatric data are available, we suggest using tunnelled lines in preference to peripherally inserted central catheter (PICC) lines in children with cancer, where practicable |
Conditional |
||||||||
|
BSH 2018 |
- There are insufficient data to support a preference for jugular or subclavian vein access for CVL insertion. Femoral vein access in children is associated with a higher risk of thrombosis and should be avoided if possible |
Strong |
||||||||
|
BSH 2018 |
- There is no definitive evidence that deferring CVL placement until the end of acute lymphoblastic leukaemia (ALL) induction therapy reduces the risk of line-associated thrombosis. The decision regarding timing of CVL placement should take into account the physical and psychological consequences of delaying CVL placement in individuals with poor venous access |
Conditional |
||||||||
|
BSH 2018 |
- Symptomatic CVL-related VTE should be treated with anticoagulation for a minimum of 3 months |
Strong |
||||||||
|
BSH 2018 |
- Anticoagulation according to standard VTE protocols is recommended for cerebral venous thrombosis (CVT) in children with cancer and should be given for a minimum of 3 months |
Strong |
||||||||
|
|
A positive thrombophilia evaluation is not a sufficient basis to offer extended anticoagulation following an episode of provoked VTE. |
GPS |
||||||||
|
A negative thrombophilia evaluation is not a sufficient basis to stop anticoagulants following an episode of unprovoked VTE in a patient with low bleeding risk and willingness to continue therapy. |
GPS |
|||||||||
|
Heterozygosity for FVL or PGM does not increase the predicted risk of recurrence after unprovoked VTE to a clinically significant degree. |
GPS |
|||||||||
|
As a family history of VTE confers an excess risk of thrombosis, relatives should be counseled regarding use of prophylaxis in high risk situations. |
GPS |
|||||||||
|
|
Especially in high-risk group for VTE, physicians should be cautious when using some drugs that may alter the balance between different coagulation factors, and some drugs that can increase blood viscosity as hormonal therapy, steroids, intravenous immunoglobulin. |
GPS |
||||||||
|
UW Health 2023 |
1. Complete blood count (CBC): Obtain baseline CBC within 48 hours of initiation of enoxaparin or heparin. |
Strong |
||||||||
|
|
2. International normalized ratio (INR): a target INR of 2.5 is generally accepted as being appropriate for the management of childhood VTE. |
GPS |
||||||||
|
UW Health 2023
|
3. Anti-Xa: - May consider checking anti-Xa level if the patient experiences active bleeding or has evidence of renal dysfunction while receiving enoxaparin. |
Conditional |
||||||||
|
|
- Routine anti-Xa levels are not recommended with the use of enoxaparin, heparin, or rivaroxaban at prophylactic doses. |
GPS |
||||||||
|
|
Good hydration maintains the blood volume and improves viscosity. |
GPS |
||||||||
|
|
Obesity is a risk factor for many diseases including heart diseases and thrombosis, therefore maintaining optimal growth and preventing obesity is recommended. |
GPS |
||||||||
|
|
A well-balanced diet containing a variety nutrient is a way to keep our children healthy. Some food that improves the cholesterol levels, act as antioxidants, and decrease inflammation can prevent blood clot as: ₋ Fruits (grapes, cherries, apples, prunes, pears, citrus) ₋ Whole grains ₋ black or green tea ₋ Nuts ₋ Leeks, onions, and garlic reduce cholesterol production ₋ Legumes and soy products |
GPS |
- Acknowledgment
The Egyptian Pediatric Clinical Practice Guidelines Committee (EPG)would like to present its highest gratitude and acknowledgment to the members of the clinical practice guideline (CPG) adaptation group and the external review group. We acknowledge the collaborative support of the Supreme Council for Egyptian University Hospitals during the adaptation of this evidence-based CPG.
- Abbreviations
|
ACCP |
American College of Chest Physicians |
|
AIS |
Arterial ischemic stroke |
|
APS |
Antiphospholipid syndrome |
|
ASH |
American Society of Hematolohy |
|
AT |
Antithrombin |
|
BSH |
British Society of Haematology |
|
CPG |
Clinical Practice Guideline |
|
CSVT |
Cerebral sinovenous thrombosis |
|
CT |
Computerized tomography |
|
CTPA |
CT pulmonary angiography |
|
CVAD |
Central venous access device |
|
CVC |
Central venous catheter |
|
CVL |
Cantral venous line |
|
DOACs |
Direct oral anticoagulants |
|
DVT |
Deep venous thrombosis |
|
EPG |
Egyptian Pediatric Clinical Practice Guidelines Committee |
|
FVL |
Factor V Leiden |
|
GDG |
Guidelines Development Group |
|
GPS |
Good practice statement |
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
|
IVC |
Inferior vena cava |
|
JAK2 |
Janase Kinase 2 |
|
LMWH |
Low molecular weight heparin |
|
MRI |
Magnetic resonance imaging |
|
MPN |
Myeloproliferative neoplasms |
|
PC |
Protein C |
|
PE |
Pulmonary embolism |
|
PNH |
Paroxysmal nocturnal hemoglobinuria |
|
PS |
Protein S |
|
PTS |
Post-thrombotic syndrome |
|
PVT |
Portal vein thrombosis |
|
QoE |
Quality of evidence |
|
RVT |
Renal vein thrombosis |
|
SoR |
Strength of recommendation |
|
SVC |
Superior vena cava |
|
tPA |
Tissue plasminogen activator |
|
VKA |
Vitamin K antagonists |
|
VTE |
Venous thromboembolism |
- Executive Summary
➡️Introduction
Pediatric thrombosis is a complex area of medicine covering a wide age range neonatal period to young adults. Compared to adult practice, there is a relative lack of high-quality research to inform evidence-based guidelines.
The aim of this adapted clinical practice guideline (CPG) is to provide evidence-based recommendations for the diagnosis and treatment in the pediatric age group.
➡️Scope
This guideline focuses on the diagnosis and treatment of any venous thrombotic event in pediatric age group. It Provides an evidence-based document for the risk factors , diagnostic tools and manipulation of anticoagulation therapy with defining the indications, dosing, and administration of these drugs and the indications and use of non-pharmacologic therapy.
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
- University of Wisconsin Venous Thromboembolism prophylaxis – Pediatric - Inpatient – Consensus care Guideline. UWHealth 2023
- Thrombophilia testing: A British Society for Hematology guideline. BSH 2022
- Guideline on the investigation, management, and prevention of venous thrombosis in children. BSH 2011 -addendum BSH 2021
- BSH Guideline: management of thrombotic and hemostatic issues in pediatric malignancy. BSH 2018
- American Society of Hematology 2018 Guidelines for management of venous thromboembolism: treatment of pediatric venous thromboembolism. ASH 2018
- Antithrombotic Therapy in Neonates and Children. Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. ACCP 2012
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
➡️Diagnosis :
- The clinical presentation of VTE in children depends on several factors, including the patient's age, anatomic location of the thrombosis, affected organ system(s), characteristics of the thrombus (occlusive vs. nonocclusive), and chronicity. (GPS)
Laboratory diagnosis:
- full blood count, basal coagulation screen,liver and renal function to confirm safe base line.D dimer not required to exclude DVT.(Strong)
- Routine testing for heriditery thrombophilla not required with first episode. (Strong)
- Antiphospholipds antibodies should be screened in unprovoked venous thromboembolisms. (Strong)
- Neonates with purpura fulminant should screened for protein C and S deficiency . Antithrombin III should be screened for early childhood attaks. (Strong)
➡️Radiological diagnosis:
▪️ CVL and non-CVL related VTE in the upper limb (UL): With the use of US, MRV and multidetector CT venography.(Strong)
▪️ CVL and non-CVL related VTE in the lower limb (LL): Doppler US is recommended : where doppler and MRV are recommended(Conditional)
▪️ For blocked central venous lines:Chest X ray, Doppler US and contrast enhanced MRV are recommended. (Conditional)
▪️ For Diagnosis of cerebral sino-venous thrombosis:MRI including T2* imaging. (Strong)
·We recommend chest Xray, CT angiography if clinical symptoms are suggestive of pulmonary embolism. (Strong)
₋ We recommend testing for myeloproliferative neoplasms (MPN) panel (including JAK2 V617F, JAK2 exon 12, CALR, MPL mutation analysis) in patients with thrombosis at unusual sites and with full blood count abnormalities suggestive of a myeloproliferative neoplasm. (Strong)
₋ Screening for antiphospholipid antibodies is recommended following unprovoked VTE because this may alter management including choice of antithrombotic therapy. (Strong)
- We suggest to assess clues from history and physical examination.( GPS)
- Assessment of an individual’s VTE risk, based on the factors:Either patient related or disease related factors. (Conditional)
- Routine thrombophilia screening is not recommended. (Conditional)
-Testing for deficiencies of physiological anticoagulants should be performed only after 3 months of anticoagulation for acute thrombosis. (Conditional)
➡️Treatment
▪️ We recommend using anticoagulation in pediatric patients with symptomatic DVT or PE.(Strong)
▪️ We suggest no anticoagulation in pediatric patients with asymptomatic DVT or PE. (Conditional)
▪️ We suggest to use either low-molecular-weight heparin or vitamin K antagonists in pediatric patients with symptomatic DVT or PE. (Conditional)
▪️ Thrombolytic therapy is not indicated for the majority of children and considered only in the presence of extensive thrombosis and should be followed by anticoagulation in PE with hemodynamic compromise. (Conditional)
▪️ We suggest anticoagulation for < 3 months for patients with provoked DVT or PE. (Conditional)
▪️ We suggest anticoagulation for 6 to 12 months for patients with unprovoked DVT or PE and recurrent idiopathic VTE and children with antiphospholipid syndrome with life-long anticoagulation. (Conditional)
▪️ We recommend anticoagulation should be initiated with low molecular weight heparin (LWMH) followed by warfarin (INR 2.5) or continuing LMWH. (Strong)
▪️ Unfractionated heparin (UFH) may be used for initial therapy where rapid reversal of anticoagulation may be required. (Conditional)
▪️We recommend using anticoagulation in pediatric patients with CSVT without hemorrhage. (Strong)
▪️ We recommend removal of a functioning CVAD in pediatric patients for symptomatic CVAD-related thrombosis with worsening signs or symptoms, despite anticoagulation, and who continue to require venous access. (Strong)
For neonates with RVT:
▪️ In unilateral renal vein thrombosis (RVT) we suggest supportive care with radiologic monitoring for extension of thrombosis and anticoagulation with UFH/LMWH or LMWH in therapeutic doses in a time range of 6 weeks-3 months. (Conditional)
▪️ In bilateral RVT with evidence of renal impairment we suggest anticoagulation with UFH/LMWH or initial thrombolytic therapy with tissue plasminogen activator (tPA) followed by anticoagulation with UFH/LMWH for a time range 6 weeks-3 months. (Conditional)
▪️ For portal vein thrombosis we suggest using anticoagulation in patients with PVT with occlusive thrombus, post liver transplant, and idiopathic PVT. (Conditional)
▪️ Children with VTE unrelated to CVLs should receive anticoagulation for an initial period of 3 months.(Strong)
▪️We suggest continuation of treatment beyond 3 months in patients with active cancer or other ongoing risk factors. (Conditional)
▪️ In children with cancer with incidental VTE we suggest systemic anticoagulation as per protocols for symptomatic disease. (Conditional)
▪️ We suggest LMWH as an anticoagulant of choice for VTE in children with malignancy. Unfractionated heparin can be used initialy when LMWH or direct oral anticoagulants are contraindicated, or not available for at least 3 months or as long as active cancer or cancer therapy. ( Conditional)
▪️ Routine measurement of peak anti-Xa activity, with a target range of 0.5–1.0 units/mL, is suggested for children receiving LMWH.( Conditional)
▪️ For children on warfarin, the last dose should be given 4– 5 days prior to an invasive procedure. Bridging anticoagulation with LMWH when the International Normalised Ratio (INR) becomes sub-therapeutic will be required for individuals within 1 month of a VTE. ( Conditional)
▪️ Symptomatic CVL-related VTE should be treated with anticoagulation for a minimum of 3 months. (Conditional)
▪️ For cancer patients: Doses of asparaginase may be administered following an asparaginase-related VTE covered by prophylactic or therapeutic anticoagulation and continued for 3 weeks following each dose of pegylated asparaginase(Conditional)
➡️Prevention
▪️ Hematology consultation is required if the patient is considered at high risk for VTE. (Strong)
Non-pharmacological (Physical) methods for thromboprophylaxis
▪️ We suggest maintenance of good hydration, early mobilisation whenever possible to reduce the risk of VTE. (Conditional)
▪️ Physical methods may be useful for high risk patients. (Strong)
Pharmacological thromboprophylaxis (LMWH, VKA)
▪️ We recommend not to use of aspirin for VTE prophylaxis in children. (Strong)
▪️ Children, particularly adolescents, with multiple risk factors for VTE should be considered for thromboprophylaxis with LMWH. (Strong)
▪️ Enoxaparin is the preferred pharmacologic prophylaxis agent for pediatric patient. (strong)
Recommendations for patients with special risk factors
▪️ We recommend anticoagulation or protein C replacement in pediatric patients with congenital purpura fulminans due to homozygous protein C deficiency. (Strong)
▪️ We recommend against thromboprophylaxis for primary prevention of CVL related thrombosis. (Strong)
▪️ Routine thromboprophylaxis in children with cancer is not recommended, but should be considered in adolescents with multiple risk factors. (Strong)
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: (PREPARE-2022CN444). Link: http://www.guidelines-registry.org/- Guideline Adaptation Group (GAG)
Name of the Working Group: CPG Adaptation for the Diagnosis, Treatment, and Prevention of Thrombosis in the Pediatric Age Groups.
Clinical Chair:Dr. Galila Mokhtar
Methodology Chair: Dr. Ashraf Abdel Baky
Guideline Authorship: The GAG has decided on group authorship and stated the order of authorship.
Registration: PREPARE-2023CN908,Practice guideline REgistration for transPAREncy (PREPARE)http://www.guidelines-registry.org/
Members of the Guideline Adaptation Group (GAG) - Authors
|
Full Name |
Affiliation(s) |
Role/ Clinical specialty |
|
Prof. Galila Mohamed Mokhtar |
Paediatrics Department, PaediatricHaematology/ Oncology & BMT UnitFaculty of Medicine, Ain Shams University, Cairo, Egypt |
Editor, Clinical expert, GAG member |
|
Prof. Amira Adly |
Professor of Paediatrics, PaediatricHaematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|
Prof. Gehan Lotfy |
Professor of Paediatrics, Minia University, Egypt |
Clinical expert GAG member |
|
Prof. Hanafy Hafez |
Professor of Pediatric Oncology, National Cancer Institute, Cairo University, Egypt |
Prof. Hanafy Hafez |
|
Prof. Hoda Hassab |
Professor of Paediatrics and PaediatricsHaematology, Alexandria University, Egypt |
Clinical expert GAG member |
|
Prof. Ilham Youssry |
Professor of Paediatrics and Head of the PaediatricHaematology& BMT unit, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|
Prof. Iman Ragab |
Professor of Paediatrics, PaediatricHaematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|
Prof. Laila Sherief |
Professor of Paediatrics and PaediatricsHaematology& Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|
Prof. Marwa Zakaria |
Associate Professor of Paediatricsand Pediatric Hematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|
Prof. Mervat Hesham |
Professor of Pediatrics and Pediatric Hematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|
Prof. Mervat Mansour |
Professor of Pediatrics and, Pediatric ICU, Ain Shams Universiy, Egypt |
Prof. Mervat Mansour |
|
Dr. Naglaa Shaheen |
Consultant of Pediatric Hematology, Health Insurance Organization |
Clinical expert GAG member |
|
Dr. Nouran Yousef |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
Clinical expert GAG member |
|
Prof. Rasha Abdel-Raouf Afifi |
Professor of Paediatrics and PaediatricHaematology, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|
Dr. Sara Makkeyah |
Associate Professor of Paediatrics, PaediatricHaematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|
Prof. Sonia Adolf |
Professor of Paediatrics and PaediatricHaematology, National Research Center, Cairo, Egypt |
Clinical expert GAG member |
➡️Co-authors: Methodology supervision group
|
Full Name |
Affiliation(s) |
Role/ Clinical specialty |
|
Prof. Ashraf Abdel Baky |
Chairman of EPG Professor of Paediatrics and Pediatric Allergy and Immunology, Ain Shams University, Cairo, Egypt
|
Guideline Methodology expert GAG member |
|
Prof. Tarek Omar |
Vice Chair of EPG Professor of Pediatrics and Pediatric Neurology, Alexandria University, Egypt |
Guideline Methodology expert GAG member |
|
Dr. Yasser S. Amer |
Pediatrician & Guideline Methodologist Research Chair for Evidence-Based Health Care & Knowledge Translation, King Saud University, Riyadh, Saudi Arabia Alexandria Center for Evidence-Based CPG Consultancy Board Member, EPG, Egypt |
Guideline Methodology expert GAG member |
|
Dr. Heba Hussein |
Associate Professor of Oral Medicine, Oral Diagnosis, and Periodontology Department, Faculty of Dentistry, Cairo University, Cairo, Egypt |
Dr. Heba Hussein |
➡️External Review Group
Prof Hanan Ibrahim – Professor of Pediatrics and Pediatric Intensive Care, Ain Shams University, Egypt.
Mervat Mansour-Professor of Pediatrics and Pediatric Intensive Care, Ain Shams University, Egypt.
Prof Khaled Salama - Professor of Paediatric Hematology, Cairo University, Egypt.
Prof Ahmad Darwish –Professor of Pediatric HematologyOncology and Bone Marrow Transplantation, Faculty of Medicine, Mansoura University, Egypt.
Prof. Ivan DFlorez – Pediatrician and Professor, Department of Pediatrics and Clinical Research, University of Antioquia, Medellin, Colombia.
Leonardo Branedao- professor of pediatrics, Institute of Health Policy Management and Evaluation, Toronto University, Toronto, Canada
The Egyptian Pediatric Clinical Practice Guidelines Committee (EPG)
CPG Title: Evidence-Based Clinical Practice Guideline for the Diagnosis, Treatment, and Prevention of Thrombosis in the Pediatric Age Groups
CPG Release Date: 2023/2024
Status: Adapted using ‘Adapted-ADAPTE’ CPG Adaptation Methodology.
Printed Copies: Available upon request from the EPG.
Electronic Sources:Available on the official website of the EPG.
1. University of Wisconsin Venous Thromboembolism prophylaxis – Pediatric - Inpatient – Consensus care Guideline. UWHealth 20231.
2. Thrombophilia testing: A British Society for Haematology guideline. BSH 20222.
3. Guideline on the investigation, management, and prevention of venous thrombosis in children. BSH 2011 -addendum BSH 20213,4 .
4. BSH Guideline: management of thrombotic and hemostatic issues in pediatric malignancy. BSH 20185.
5. American Society of Hematology 2018 Guidelines for management of venous thromboembolism: treatment of pediatric venous thromboembolism. ASH 20186.
6. Antithrombotic Therapy in Neonates and Children. Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. ACCP 20127.
- Methodology
The description of the methodology for the production of this Adapted CPG can be fulfilled byutilizing the sequential process for trans-contextual formal adaptation of CPGs ‘Adapted ADAPTE’ methodf8,9 that was approved by the EPG to be themethod of National CPG production. Details process is reported in the Appendices.
The Egyptian Pediatric Clinical Practice Guidelines Committee (EPG)
Email address: guidelinescommitte@gmail.com
- Introduction
Thrombosis is increasingly recognized in the pediatric age group, especially venous thromboembolism (VTE). The annual incidence of childhood thrombosis was estimated to be 0.07-0.14/ 10,000 children in the 1990s10. However, recent studies showed increasing incidences of childhood thrombotic events due to improved diagnosis, increased survival of children with severe underlying diseases, and increased use of interventions such as central venous lines (CVL). Moreover, the diagnosis of VTE in hospitalized children increased from 34 to 58 cases /10,000 in the United States from year 2001 to 2007. Moreover, the true incidence could have been significantly higher as the majority of VTEs are clinically silent. The increased incidence of thrombosis was observed in all pediatric age categories, nevertheless, neonates, infants (< 2 years old), and adolescents have the greatest risk for VTE11. Egyptian children who have more than one heterozygous mutation even if weakly thrombophilic are prone to develop significant thrombotic events either spontaneously or when exposed to minor risk factors12.
Thrombosis is a serious disease, which leads to mortality and morbidity. The mortality rate is about 2%. Morbidity includes lack of thrombus resolution in 50% of the patients, the development of post-thrombotic syndrome (PTS)11, and recurrent thrombosis in 8.1% with an average follow-up period of 2.86 years 13.
Most of the venous thrombi in children are associated with clinical risk factors such as inheritance of prothrombotic defects, CVL insertion, cardiac disease, surgery, obesity, inflammatory conditions, immobility, medications such as asparaginase and estrogen-containing contraceptives, and cancer patients14. Also, there are some diseases with an increased tendency to thrombosis called acquired thrombophilia diseases as; antiphospholipid syndrome (APS), paroxysmal nocturnal hemoglobinuria (PNH), myeloproliferative neoplasms (MPN) and the presence of a Janase Kinase 2 (JAK2) mutation in the absence of an MPN phenotype2.
The inheritance of prothrombotic defects called inherited thrombophilia diseases which refers to genetic disorders of specific hemostatic proteins that include: the factor V Leiden (FVL) variant (F5 G1691A), the prothrombin gene variant (F2 G20210A), protein C (PC) deficiency, protein S (PS) deficiency, and antithrombin (AT) deficiency. Certainly, the inheritance of any of these prothrombotic genetic defects may lead to spontaneous VTE (unprovoked VTE), VTE with severity out of proportion to the stimulus (provoked VTE), recurrent thrombosis, or VTE at a young age. However, it is still a matter of debate whether it is useful to test for genetic thrombophilia defects in children with a first venous thrombotic event15.
Cancer-associated thrombosis is the second leading cause of death in patients with cancer and its prevalence is increasing. These patients are at high risk of recurrent VTE, io one side, and on the other side, they are at high risk for anticoagulant-related bleeding which is associated with high morbidity and resource use16.
The diagnosis and treatment of pediatric thrombosis are still debatable because children with thrombosis are a heterogeneous group, and it is unlikely that a single approach to testing and/or treatment is optimal or desirable.
The mainstay of treatment of acute pediatric thrombosis is anticoagulation with heparins and/or vitamin K antagonists. The immediate goals of anticoagulation are to prevent the propagation of acute thrombosis, prevent embolization, and prevent secondary VTE. Duration of therapy is therefore predicated on the time-dependent risk of recurrent VTE. Unfortunately, there is limited data by which to accurately define the frequency of VTE recurrence and anticoagulation-associated adverse events in pediatric VTE7. Bleeding is the primary complication of anticoagulant therapy, and is a risk of all anticoagulants, even when maintained within usual therapeutic ranges. Ironically, whereas unfractionated heparin and coumarin, the oldest and most widely used anticoagulants, have specific antidotes for their anticoagulant effect, many of the newer agents currently undergoing clinical evaluation do not have specific antidotes; thus, the best ways to reverse their actions remain to be determined17.
Thus, given that the field of pediatric thrombosis continues to evolve with the scarcity of evidence in the pediatric age group. Therefore, pediatricians dealing with this evolving and serious health problem are in great needof a guideline that may help them for best clinical decisions. Thus, this guideline will answer some of the health questions related to the prevention, diagnosis, and treatment of pediatric thrombosis.
The aim of this adapted clinical practice guideline (CPG) is to provide evidence-based recommendations for the Diagnosis, Treatment, and Prevention of Thrombosis in the Pediatric Age Groups.
These recommendations were adapted from the relevant six CPGs using a formal methodology for CPG adaptation: the Adapted-ADAPTE.
- Scope and Purpose
➡️Disease/Condition:
Thrombosis in different pediatric age groups.
➡️Guideline Objectives
To Provide an evidence-based document for the
1. Identification of neonates, infants, children, and adolescents at high risk of thrombosis.
2. Diagnostic approaches in the target population.
3. Management of thrombosis in the target population.
4. Prevention of thrombosis in the target population
|
Health / Clinical Question (PIPOH)
|
|
|
P: Patient (Target Population):
|
•Gender: Both genders. • Age group: Neonates, Infants, children & adolescents less than 18 years. • Disease/ Condition: thrombosis (arterial/venous, site, disease categories). • Exclusion criteria: thrombotic microangiopathy, disseminated intravascular coagulopathy, thrombosis due to stasis and/ or blood vessel injury. |
|
I: Interventions and Practices Considered / CPG Category:
|
Diagnosis: • Venous thrombosis • Arterial thrombosis Treatment • Venous thrombosis • Arterial thrombosis Prevention |
|
P: Professionals (Intended / Target Users or Stakeholders) :
|
Primary health care physicians at the Ministry of Health, general practitioners, family medicine specialists, pediatricians, hematologists, oncologists, neonatologists, intensive care specialists, surgeons, neurologists, nurses, and medical students. |
|
O: Major Outcomes Considered:
|
• Primary outcome: - Prevent mortality and morbidity from thrombosis. - Prevent sequelae and disabilities • Secondary outcome: - Family counseling - Prevention of recurrence |
|
H: Healthcare Settings:
|
Primary, secondary, and tertiary healthcare services mainly outpatient clinics and emergency rooms in governmental, non-governmental and private sectors in Egypt. |
Guidelines 1-5 used the Grading of Recommendations Assessment Development and Evaluation (GRADE) methodology18.Guideline 6 used an early version of GRADE.
Rating Scheme for the Strength of the Evidence/Recommendations:
➡️GRADE ranking of evidence
|
High |
Further research is very unlikely to change our confidence in the estimate of effect |
|
Moderate |
Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. |
|
Low |
Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. |
|
Very low |
Any estimate of effect is very uncertain. |
GRADE Ratings for Recommendations For or Against Practice
|
Strong |
the panel is confident that the desirable effects of adherence to a recommendation outweigh the unesirable effects |
|
conditional |
the panel concludes that the desirable effects of adherence to a recommendation probably outweigh the undesirable effects, but is not confident. |
➡️Good practice statement refers to
Good practice
statements represent recommendations that the Guidelines Development Group
(GDG) feels are important, although they are not adequate for a formal
certainty of evidence rating according to the Grading of Recommendations
Assessment, Development, and Evaluation (GRADE) working group.
- Implementation strategies or interventions
1. Leadership commitment, engagement, and support.
2. Local clinical and quality champions.
3. Dissemination (printed and electronic).
4. Regular training and education.
5. Regular audit and feedback (along with regular review and update promotes the concept of the ‘living CPGs’.
6. Networking with relevant existing projects.
7. Parents or carers as champions for changes.
➡️Implementation tools
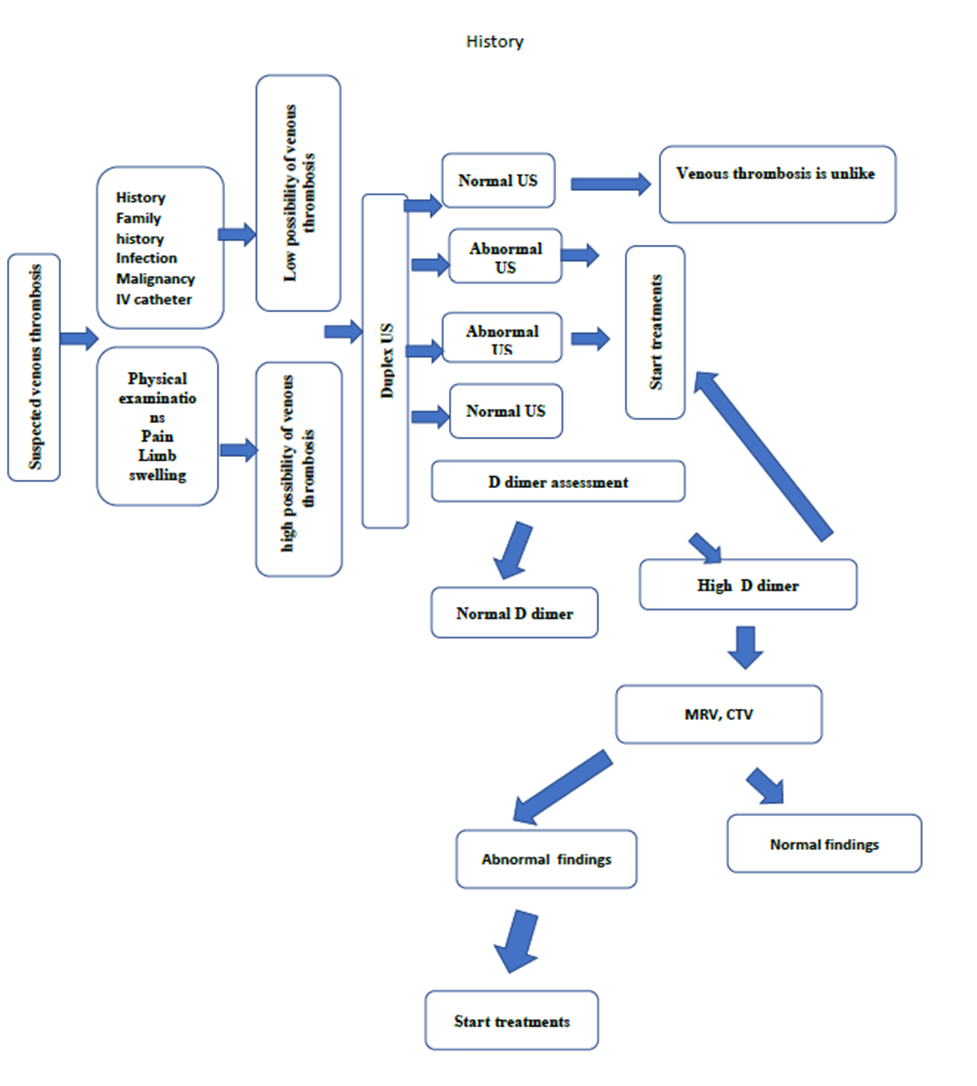
Figure1: Diagnostic
algorithm for Pediatric DVT
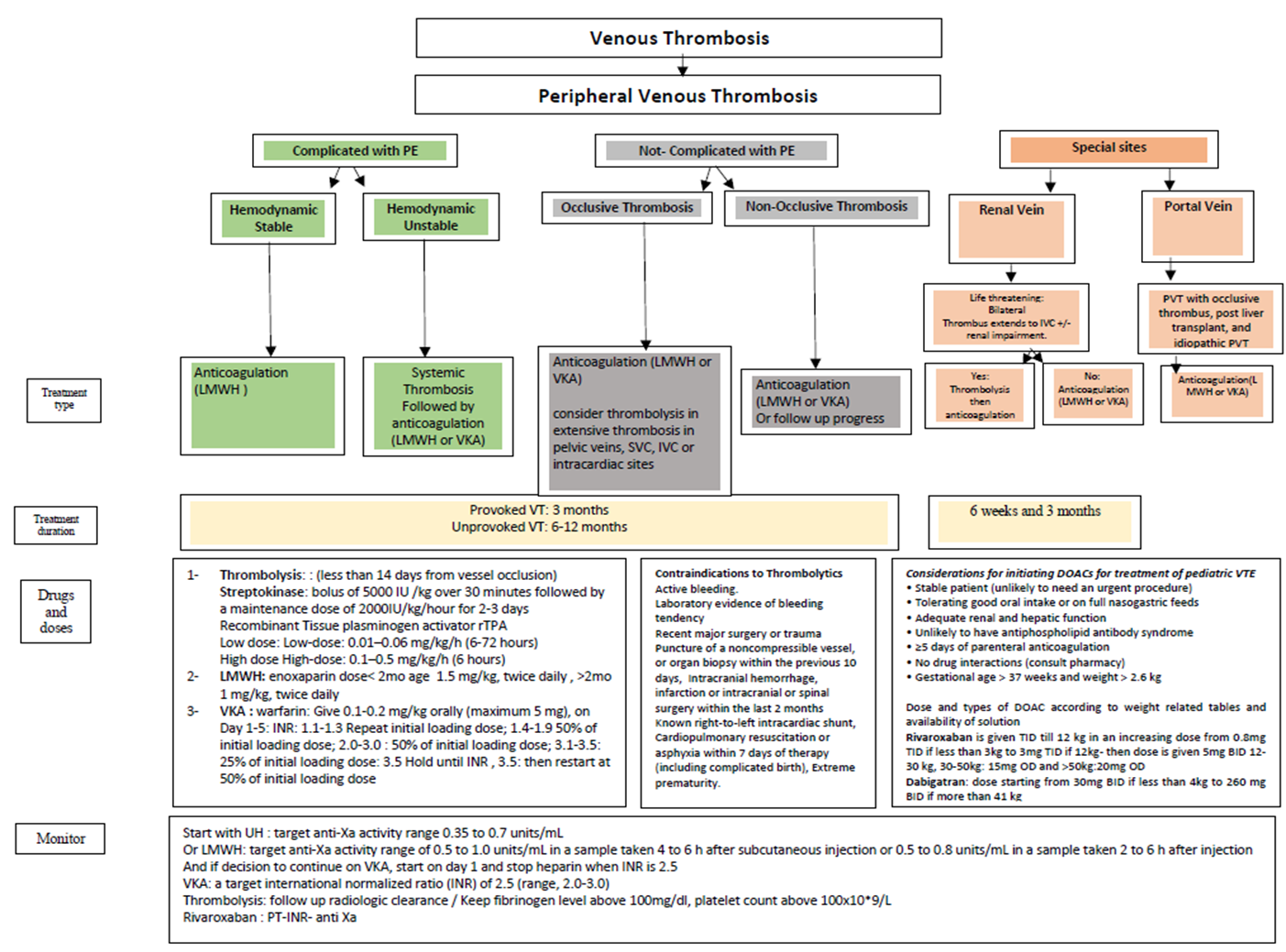
Figure 2: Algorithm
for management of venous thrombosis
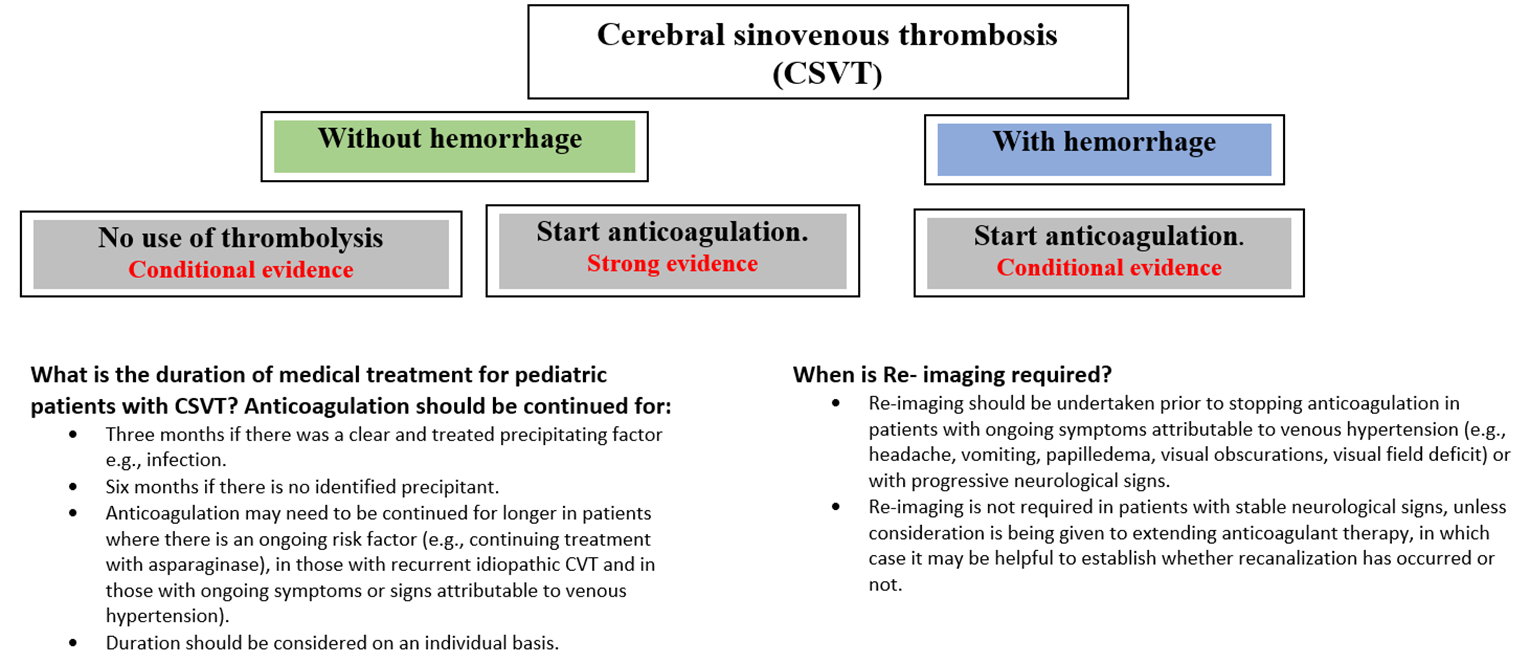
Figure 3: Management plan of cerebral sinovenous thrombosis
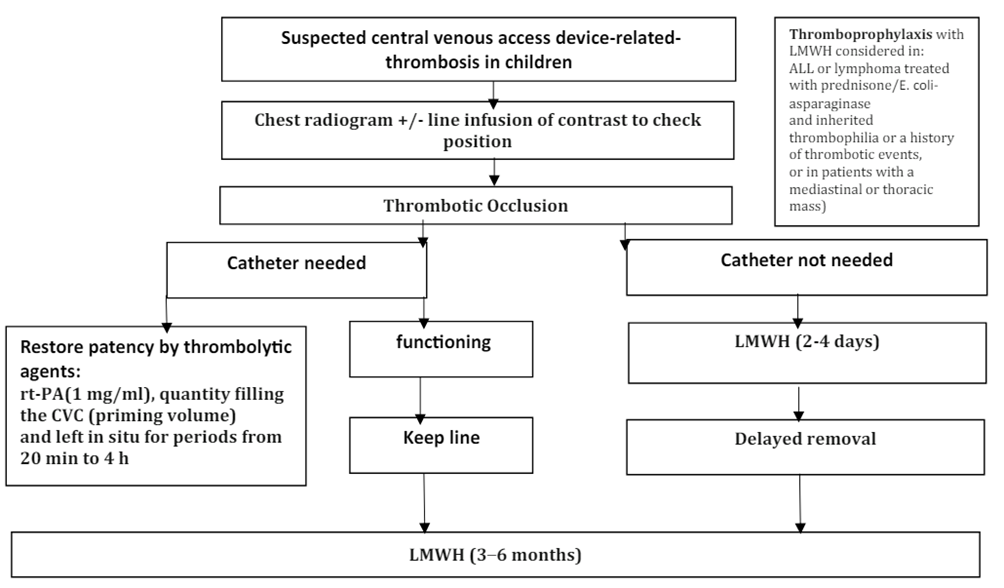
Figure 4: Pathway for management of acute CVAD-associated thrombosis in neonates and children.
|
Drug |
Route |
Dose |
Therapeutic monitoring |
|
UFH |
Continuous IV infusion |
Age < 12 mo: bolus 75 U/kg followed by 28 U/kg/h Age 1 to < 12 y: bolus 75 U/kg followed by 20 U/kg/h Age > 12 y: bolus 80 U/kg followed by 18 U/kg/h |
Target range: aPTT: 1.5-2.5 times control or Anti-Xa level 0.3-0.7 U/mL |
|
Enoxaparin (LMWH) |
Subcutaneous injection |
Age < 2 mo: 1.5-1.7 mg/kg q12 h Age > 2 mo: 1 mg/kg q12 h |
Target range: Anti-Xa 0.5-1.0 U/mL (Sample 3 to 4 h after third dose) |
|
Warfarin |
Oral |
Loading: 0.2 mg/kg x 1 (if INR < 1.3), max 10 mg Maintenance: adjust according to INR (Measure INR daily days 2 to 4) INR 1.1-1.3: repeat loading dose INR 1.4-1.9: 50% of loading dose INR 2.0-3.0: INR 3.1-3.5: 25% of loading dose INR >3.5: Hold until INR < 3.5, reinitiate at 50% of loading dose. |
Target range: INR 2.0 to 3.0 |
|
Rivaroxaban (DOAC) |
Oral |
According to weight 2.6 to < 3 kg:0.8 mg per dose TDS 3 to < 4 kg: 0.9 mg per dose TDS 4 to < 5 kg: 1.4 mg per dose TDS 5 to < 7 kg: 1.6 mg per dose TDS 7 to < 8 kg: 1.8 mg per dose TDS 8 to < 9 kg: 2.4 mg per dose TDS 9 to < 10 kg: 2.8 mg per dose TDS 10 to < 12 kg: 3 mg per dose TDS 12 to < 30 kg: 5 mg per dose BD 30 to < 50 kg: 15 mg OD ≥ 50 kg: 20 mg OD |
|
Table 2: Common anticoagulant therapies for use in pediatric VTE
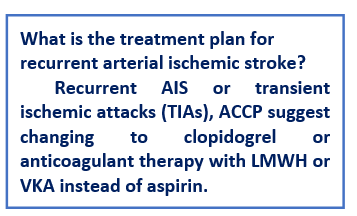
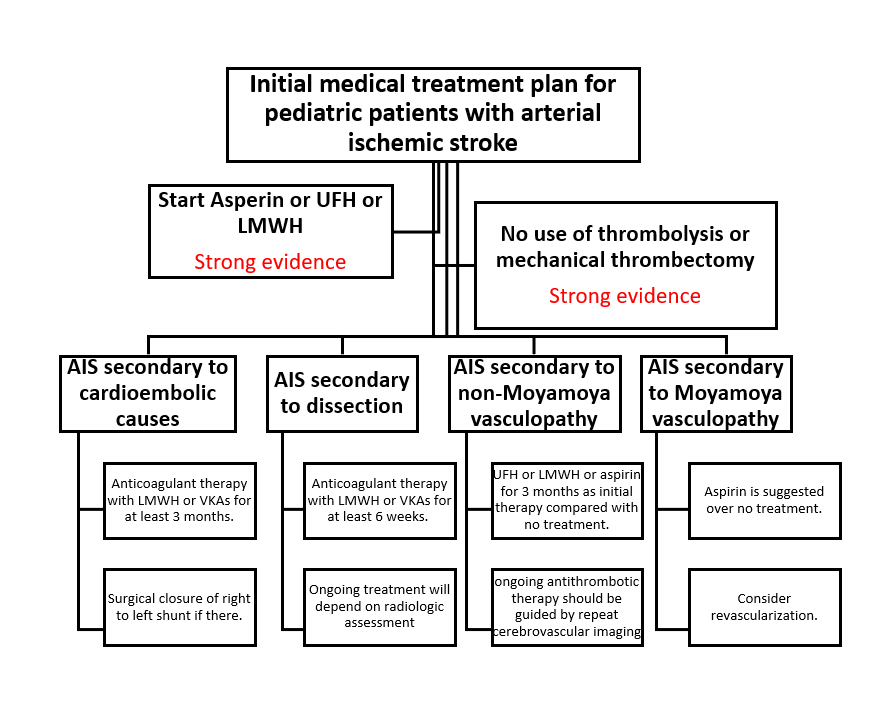

Figure 5: Management
plan of arterial ischemic stroke



- Plan for Scheduled Review and Update
The CPG Adaptation Group decided to review this adapted CPG for updates after fiveyears from its publication date (2023/2024) which should be in (2028) after checking for updates in the source guidelines, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the Egyptian healthcare setting. The updating process will be guided by the CheckUp (Checklist for the Reporting of Updated Guidelines) Tool.
1. EPG
2. Supreme Council
3. The funding bodies are official governmental organizations that have provided non-financial logistic and expert clinical and methodological support and have not influenced the formulation of the final evidence-based recommendations. Moreover, this guideline adaptation project was not related to any pharmaceutical, industrial, or any other commercial entity.
All of the GAG members have no conflicts of interest to declare. None of them were associated with or involved in any of the source CPGs that were appraised. Moreover, the competing interests are assessed as part of the AGREE II assessment (Domain 6 Editorial independence).
Future research recommendations for the management of pediatric venous thrombosis in different age groups in the Egyptian context could include:
▪️ Implementation of hemovigilance system at different institutional levels.
▪️ Anticoagulation therapy for specific populations.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for venous thromboembolismin children.
➡️Challenges
▪️ Implementation of international guidelines in resource-limited settings.
▪️ The optimum use of the available resources to fulfill the requirements of the Egyptian population.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Updating the guideline
The EPG Haematology GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area, and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence-based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Cheng Y. Venous Thromboembolism Prophylaxis – Pediatric – Inpatient
Consensus Care Guideline. University of Wisconsin Hospitals and Clinics Authority; 20 April 2023 2023.
2.Arachchillage DJ, Mackillop L, Chandratheva A, Motawani J, MacCallum P, Laffan M. Thrombophilia testing: A British Society for Haematology guideline. Br J Haematol. 2022;198(3):443-458.
3. Biss TT, Chalmers EA. Addendum to British Society for Haematology guideline on the investigation, management and prevention of venous thrombosis in children (Br. J. Haematol. 2011; 154: 196-207). Br J Haematol. 2021;194(6):996-998.
4. Chalmers E, Ganesen V, Liesner R, et al. Guideline on the investigation, management and prevention of venous thrombosis in children. Br J Haematol. 2011;154(2):196-207.
5. Sibson KR, Biss TT, Furness CL, et al. BSH Guideline: management of thrombotic and haemostatic issues in paediatric malignancy. Br J Haematol. 2018;180(4):511-525.
6. Monagle P, Cuello CA, Augustine C, et al. American Society of Hematology 2018 Guidelines for management of venous thromboembolism: treatment of pediatric venous thromboembolism. Blood Adv. 2018;2(22):3292-3316.
7. Monagle P, Chan AKC, Goldenberg NA, et al. Antithrombotic therapy in neonates and children: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e737S-e801S.
8. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. Journal of evaluation in clinical practice. 2015;21(6):1095-1106.
9. Fervers B, Burgers JS, Voellinger R, et al. Guideline adaptation: an approach to enhance efficiency in guideline development and improve utilisation. BMJ Qual Saf. 2011;20(3):228-236.
10. van Ommen CH, Heijboer H, Buller HR, Hirasing RA, Heijmans HS, Peters M. Venous thromboembolism in childhood: a prospective two-year registry in The Netherlands. J Pediatr. 2001;139(5):676-681.
11. Goldenberg NA, Donadini MP, Kahn SR, et al. Post-thrombotic syndrome in children: a systematic review of frequency of occurrence, validity of outcome measures, and prognostic factors. Haematologica. 2010;95(11):1952-1959.
12. Abdelwahab M, Abdelraouf R, Adel F, Zagloul N. PB2444 INHERITED THROMBOPHILIA IN EGYPTIAN CHILDREN: A 13 YEAR SINGLE CENTER STUDY. HemaSphere. 2019;3(S1).
13. Monagle P, Adams M, Mahoney M, et al. Outcome of pediatric thromboembolic disease: a report from the Canadian Childhood Thrombophilia Registry. Pediatr Res. 2000;47(6):763-766.
14. Branchford BR, Betensky M, Goldenberg NA. Pediatric issues in thrombosis and hemostasis: The how and why of venous thromboembolism risk stratification in hospitalized children. Thromb Res. 2018;172:190-193.
15. Hoppe C, Matsunaga A. Pediatric thrombosis. Pediatr Clin North Am. 2002;49(6):1257-1283.
16. Weitz JI, Haas S, Ageno W, et al. Cancer associated thrombosis in everyday practice: perspectives from GARFIELD-VTE. J Thromb Thrombolysis. 2020;50(2):267-277.
17. Ng HJ, Crowther MA. New anti-thrombotic agents: emphasis on hemorrhagic complications and their management. Semin Hematol. 2006;43(1 Suppl 1):S77-83.
18. Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336(7650):924-926.
19. Parasuraman S, Goldhaber SZ. Venous thromboembolism in children. Circulation. 2006;113(2):e12-16.
20. Witmer C, Raffini L. Treatment of venous thromboembolism in pediatric patients. Blood. 2020;135(5):335-343.
21. Kearon C, Kahn SR, Agnelli G, Goldhaber S, Raskob GE, Comerota AJ. Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133(6 Suppl):454S-545S.
22. Young G, Yee DL, O'Brien SH, Khanna R, Barbour A, Nugent DJ. FondaKIDS: a prospective pharmacokinetic and safety study of fondaparinux in children between 1 and 18 years of age. Pediatr Blood Cancer. 2011;57(6):1049-1054.
- Appendices
➡️CPG databases and libraries:
1. Guidelines International Network Library and Registry (GIN).https://g-i-n.net/international-guidelines-library/
2. World Health Organization (WHO) Guidelines
3. National Institute of Clinical and Health Excellence (NICE) UK. http://www.nice.org.uk/guidance/
4. scottish Intercollegiate Guidelines Network (SIGN) UK. http://www.sign.ac.uk/guidelines/
5. ECRI Guidelines Trust (USA). https://guidelines.ecri.org/
6. DynaMed (Summaries: Guidelines and Resources)
➡️Bibliographic databases:
1. PubMed/ MEDLINE https://pubmed.ncbi.nlm.nih.gov/
➡️Specialized professional societies:
1. American Society of Hematology (ASH) https://www.hematology.org/
2. American College of Chest Physicians (ACCP) https://www.chestnet.org/
3. British Society for Haematology (BSH) https://b-s-h.org.uk/
4. Children’sOncologyGroup (COG)
5. International Society on Thrombosis and Haemostasis (ISTH)https://www.isth.org/
6. NationalComprehensiveCancer Network (NCCN)https://www.nccn.org/
Appendix 2. Search strategy
Keywords:Pediatric Thrombosis – Thrombophilia – Hypercoagulability – venous thrombosis– arterial thrombosis - Stroke – Pulmonary embolism - Thromboprophylaxis - Thrombectomy
PubMed/ MEDLINE https://pubmed.ncbi.nlm.nih.gov/
Appendix 3. CPG Databases
CPG Databases:
www./epg.edu.eg
Appendix 4. Adaptation Process
Methodology
Phase One – Set-Up
A preliminary rapid search showed a large number of published CPGs addressing this topic.
The project lead was assigned by the EPG who nominated the clinical chair (Ashraf Abdel Baky), and worked closely with the methodology chair (Tarek Omar), to define the initial scope of the CPG. A group of clinical experts was defined from the network of the EPG to be the members of the GAG (full list provided in Table 1). The methodology followed was according to the ‘Adapted-ADAPTE’ method.
Detailed composition of the GAG including clinical specialties and roles.
The topic of “Thrombosis in the Pediatric Age Groups” was identified by the EPG as one of the main high-priority topics for pediatric practice in Egypt.
The methodology chair conducted capacity-building sessions for the GAG members on the CPG adaptation methodology, a systematic review of CPGs, and evaluation of CPGs at the outset of the project in addition to hands-on technical support throughout the process.
All members of the GAG declared their conflicts of interest and agreed on the terms of references for CPG projects proposed by the EPG.
A timeline for the CPG adaptation project was outlined by the EPG and agreed upon by all GAG members.
Refer to the Scope and Purpose section. The PIPOH Model was used to identify the health questions that guided the search and screening process.
A comprehensive review of evidence-based CPGs for pediatric venous thrombosiswas conducted.
# Included: 6
# Excluded: 9
The GAG relied on the inclusion and exclusion (eligibility) criteria for screening and filtration.
➡️Inclusion / Exclusion CPGs Selection (eligibility) Criteria
1. Methods of Development:Evidence-Based CPGs: (Detailed Methodology of Development Documented; link Recommendations with Evidence; link to Systematic Reviews) rather than Consensus-based CPGs (Expert opinion)
2. Author(s) Organization (CPG development group) from CPGs Database (Producer or Finder) and Specialized Society (clinical specialty) rather than single authors.
3. Country: International or national CPGs.
4. Date of Publication: Range of year of publications (2024-2025) Last 5 years
5. Language: English and Arabic CPGs only
6. Status: Only source CPG (de novo developed) rather than adapted CPGs
The Appraisal of Guidelines Research & Evaluation (AGREE) II Instrument was used to assess the quality of the five eligible Source CPGs using the online platform (MY AGREE PLUS) by four independent appraisers from the GAG. Disagreements in the ratings of the AGREE II items were resolved by discussions.
Appendix Table 1. AGREE II assessment results and the standardized domain scores for the XX included CPGs
|
CPGs AGREE II DOMAINS |
CPG1
|
CPG2
|
CPG3 |
CPG4 |
CPG5 |
CPG6 |
|
D1: Scope & Purpose |
94.44 |
77.78 |
72.2 |
61 |
91.65 |
83.33 |
|
D2: Stakeholder Involvement |
50 |
77.78 |
83.3 |
72.2 |
91.65 |
72.22 |
|
D3:Rigour of Development |
68.75 |
87.75 |
93.75 |
68.8 |
86.45 |
85.42 |
|
D4: Clarity & Presentation |
94.44 |
83.33 |
100 |
77.8 |
88.9 |
88.88 |
|
D5: Applicability |
50 |
62.5 |
83.3 |
58.3 |
83.35 |
83.33 |
|
D6: Editorial Independence |
60 |
91.67 |
100 |
883.3 |
91.65 |
83.33 |
|
OA 1 |
68.12 |
80.12 |
89.1 |
71.7 |
88.1 |
84.06 |
|
OA 2 (Recommend for use) |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
This table uses the AGREE II Domain Score Color codes (< 40% red; > 41 – 70% yellow; > 71 % green) |
||||||
1. CPG1: University of Wisconsin Venous Thromboembolism prophylaxis – Pediatric - Inpatient – Consensus care Guideline. UWHealth 2023
2. CPG2: Thrombophilia testing: A British Society for Haematology guideline. BSH 2022
3. CPG3: Guideline on the investigation, management, and prevention of venous thrombosis in children. BSH 2011 -addendum BSH 2021
4. CPG4: BSH Guideline: management of thrombotic and hemostatic issues in pediatric malignancy. BSH 2018
5. CPG5: American Society of Hematology 2018 Guidelines for the management of venous thromboembolism: treatment of pediatric venous thromboembolism. ASH 2018
6. CPG6: Antithrombotic Therapy in Neonates and Children. Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. ACCP 2012
Based on the results of the AGREE II assessment, the GAG decided to adapt/ adopt all of the recommendations from the six CPGs.
Appendix Table 3. Currency survey of the CPG developer
|
1. Are you aware of any new evidence relevant to this CPG statement? Yes/ No |
|
2. Is there any new evidence to invalidate any of the recommendations comprising the CPG? Yes/ No |
|
3. Are there any plans to update the CPG in the near future? Yes/ No |
|
4. When the CPG was last updated? 5. What is the citation for the latest version? See Reference www.epg.edu.eg |
Steps 13-15 were not conducted as the GAG relied on the results of the AGREE II assessment to reach the required assessments of CPG content, consistency, acceptability, and applicability.
Steps 16 and 17: the AGREE II assessments were discussed among the members of the GAG. Therefore, the pediatric venous thrombosis CPG adaptation group decided to select the six CPG and adapt all of its recommendations and implementation tools.
The first draft of the adapted CPG was prepared and sent for external review via email with the related clinical review and methodology review forms.
Details of the members of the external review group.
The adapted CPG was approved by the EPG.
16. Step 21. Consulting with the source CPG developer(s)


1. Step 22. Acknowledging the source documents
The source CPG(s), relevant articles, and websites have been acknowledged in the adapted CPG document.
The GAG decided on a plan for reviewing and updating the adapted CPG using the CheckUp Tool that is freely provided by the AGREE Enterprise.
This document represents the finalized adapted CPG full document. Table 9 presents the full map of the CPG adaptation process including the steps that were followed and the steps that were not followed with the relevant reasons.
Appendix Table 4. Decision Support for EPGAdaptation Working Group for CPG for ‘Management of pediatric venous thrombosis. Chairperson: Dr. Ashraf Abdelbaky Salama
|
PHASE |
MODULE |
STEP |
TOOL |
DECISION |
REASON (if not utilized) |
||||
|
Utilized |
Not utilized |
||||||||
|
ONE: SET-UP |
1.1. Preparation |
1 |
1 |
√ |
|
|
|||
|
2 |
√ |
|
|
||||||
|
2 |
|
√ |
|
|
|||||
|
3 |
|
√ |
|
|
|||||
|
4 |
|
√ |
|
|
|||||
|
5 |
3 |
√ |
|
|
|||||
|
4 |
√ |
|
|
||||||
|
1 |
√ |
|
|
||||||
|
6 |
5 |
√ |
|
|
|||||
|
|
|||||||||
|
TWO: ADAPTATION |
2.1. Scope and Purpose |
7 |
6 |
√ |
|
|
|||
|
2.2. Search and Screen |
8 |
2 |
√ |
|
|
||||
|
7 |
√ |
|
|
||||||
|
9 |
8 |
√ |
|
|
|||||
|
10 |
9 |
|
√ |
Decided to rely on inclusion/ exclusion criteria (filters) & PIPOH compatibility |
|||||
|
10 |
√ |
||||||||
|
2.3. Assessment |
11 |
9 |
√ |
|
|
||||
|
10 |
√ |
|
|
||||||
|
12 |
11 |
√ |
|
|
|||||
|
13 |
12 |
|
√ |
Decided to select NICU (Amer et al,2015) of the CPG |
|||||
|
14 |
13 |
|
√ |
Decided to rely on D3 Scores of AGREE II |
|||||
|
14 |
|
√ |
|||||||
|
15 |
15 |
|
√ |
Decided to rely on D5, D2 Scores of AGREE II |
|||||
|
2.4. Decision and Selection |
16 |
Table |
√ |
|
|
||||
|
17 |
Decision making and selection |
√ |
|
|
|||||
|
|
2.5. Customization |
18 |
16 |
√ |
|
|
|||
|
|
|||||||||
|
THREE: FINALIZATION |
3.1. External Review and Acknowledgment Module |
19 |
17 |
√ |
|
|
|||
|
20 |
|
√ |
|
|
|||||
|
21 |
|
√ |
|
|
|||||
|
22 |
|
√ |
|
|
|||||
|
3.2. Aftercare Planning |
23 |
18 |
√ |
|
|
||||
|
3.3. Final Production |
24 |
|
√ |
|
|
||||
