Epithelial ovarian cancer
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Epithelial ovarian cancer |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 9 March 2025" Download Guideline
- Executive Summary
Diagnostic and Staging Work up | |
The standard work-up for patients suspected of having EOC (Epithelial Ovarian Carcinoma) should include detailed history and clinical examination, LFTs, KFTs, serum CA-125, serum CEA and CA 19-9 in case of mucinous carcinoma and endoscopy if either or both are elevated, as well as transabdominal and transvaginal US (should be done by an expert examiner), as well as CT of thorax, abdomen and pelvis. | Strong |
Pathological examination of adequate tumor sample from diagnostic biopsy or surgical specimen should be done. In case of the presence of pleural effusion, cytological assessment should be done. | Strong |
The revised 2017 FIGO staging system for EOC should be used. | Strong |
Management of early EOC (FIGO STAGE I-II) | |
Surgical staging is recommended in presumed early-stage ovarian cancer for classification and recommendation of optimal systemic therapy. | Strong |
The aim of surgery for early EOC is complete resection of the tumour and to undertake adequate staging, which should be performed by midline laparotomy and should include: - Inspection and palpation of the whole abdominal cavity -
Peritoneal washing with cytological
examination | Strong |
Fertility-sparing surgery should be considered in young patients, but always after full discussion with the patient about potential risks. | Strong |
Patients with any stage IA histotype or stage IC1-2 with unilateral ovarian involvement and favorable histology (i.e. low-grade tumors) would be amenable to contralateral ovary and uterus preservation, in combination with the other recommended surgical staging procedures.
| Strong |
Adjuvant chemotherapy in early-stage ovarian cancer is generally recommended for FIGO stage I-IIB (see exceptions below), either paclitaxel-carboplatin or carboplatin alone (six cycles). | Strong |
The benefit of adjuvant chemotherapy is uncertain and can be considered as optional for: - Low grade serous carcinoma (LGSC) stage IB-IC - Clear cell carcinoma (CCC) stage IA-IC1 - Low-grade endometrioid carcinoma (EC) stage IB-IC - Expansile mucinous carcinoma (MC) stage IC - Infiltrative MC stage IA | Conditional |
For patients receiving paclitaxel-carboplatin, a minimum of three cycles are recommended except for high grade serous carcinoma (HGSC) /high-grade endometrioid carcinoma (EC) or any stage IC-II regardless of histotype, for which six cycles should be administered. | Strong |
Adjuvant chemotherapy is not recommended in completely staged patients with LGSC stage IA, low-grade EC stage IA or expansile MC stage IA-IB. | Conditional |
Management of advanced EOC (FIGO STAGE III-IV) | |
Patients with advanced EOC should be evaluated for primary cytoreductive surgery (PCS) by a specialized team, with the aim of achieving complete cytoreduction (absence of all visible residual disease). | Strong |
When complete cytoreductive surgery is feasible, PCS is recommended; otherwise, obtaining adequate biopsy tissue for histology and molecular testing is recommended. | Strong |
PCS should aim to maximal surgical effort and may require intestinal resection, diaphragmatic and peritoneal stripping, splenectomy and removal of bulky para-aortic lymph nodes and, in some cases, extra-abdominal disease. | Strong |
We recommend against systematic lymphadenectomy in patients with macroscopic complete resection and clinically negative nodes as this may lead to unnecessarily increases the rate of post-operative complications and mortality and should not be done. | Strong |
PCS is also recommended in patients with less chemo-sensitive subtypes (e.g. MC or LGSC), even if uncertainty about achieving complete resection exists and a small residual tumour (<1 cm) is likely to remain. | Strong |
When complete cytoreductive surgery is not feasible, neoadjuvant chemotherapy (NACT) for three cycles followed by interval cytoreductive surgery (ICS) and three cycles of paclitaxel-carboplatin are recommended, + staging laparoscopy. | Strong |
Consider the use of bevacizumab in the neoadjuvant setting, before interval cytoreductive surgery (ICS). | Conditional |
When ICS is not possible, and in the absence of overt disease progression, three additional cycles of paclitaxel-carboplatin alone or with bevacizumab are recommended. | Strong |
Paclitaxel (175 mg/m2)-carboplatin (AUC 5-6) every 3 weeks for six cycles is the standard first-line chemotherapy in advanced ovarian cancer. | Strong |
We recommend the schedule of weekly chemotherapy with paclitaxel (60 mg/m2)-carboplatin (AUC 2) as an alternative in frail patients. | Strong |
Bevacizumab may be considered in addition to paclitaxel-carboplatin in high-risk patients, (defined as patients with stage III and macroscopic residual tumour >1 cm or stage IV). | Conditional |
Bevacizumab dose, if given, should be 7.5 mg/kg and the duration of treatment is 12 months. | Strong |
Intraperitoneal chemotherapy and hyperthermic intraperitoneal chemotherapy (HIPEC) are not considered a standard of care in first-line treatment. | Conditional |
Maintenance with anti-estrogen therapy after first-line platinum-based chemotherapy can be considered in ER positive low grade serous carcinoma (LGSC) or grade I endometrioid carcinoma. | Conditional |
Other recommended regimens (other than paclitaxel/carboplatin regime) such as docetaxel/carboplatin or 5FU/ calcium leucovorin/oxaliplatin or Capecitabin/Oxaliplatin specially in mucinous carcinoma may be used | Conditional |
Management of recurrent EOC. | |
The following should be assessed when selecting treatment for patients with recurrent disease: - Histotype - Number of prior lines of treatment - Exposure and response to prior treatment - TFIp (treatment-free interval from last platinum) - Possibility of achieving a complete secondary surgical cytoreduction - Residual chemotherapy toxicity - The patient’s general condition and preferences | Good Practice Statement
|
Patients with first relapse of ovarian cancer after >6 months of last platinum administration should be evaluated by a team experienced in surgery for ovarian cancer to identify potential candidates for surgical cytoreduction. | Strong |
Patients who have previously responded to platinum without early symptomatic relapses (after >6 months) should be treated with either a platinum-based doublet (paclitaxel or gemcitabine with bevacizumab) or single agent (liposomal doxorubicin). The selection should be based on safety and patient preference. | Strong |
If combination therapy is contraindicated, carboplatin monotherapy remains an option. | Strong |
Treatment is usually recommended for four to six cycles. | Strong |
Bevacizumab should be continued until disease progression (symptomatic) or the next line of treatment is started, as continuation of bevacizumab beyond progression has not been evaluated in the recurrent setting. | Strong |
Platinum rechallenge following treatment with a platinum regimen (monotherapy or combination) should be considered if the tumour is not refractory or resistant. | Strong |
Patients with relapsed EOC for whom platinum is not an option should be defined by: - Proven refractory (progression during platinum) - Expected resistance (early symptomatic progression post-platinum, response to rechallenge unlikely) - Platinum intolerance - Patient choice - QoL issues | Good Practice Statement
|
For patients who are not candidates to receive platinum, integrating palliative care early in the treatment pathway is strongly recommended. | Good Practice Statement
|
Single-agent non-platinum options that are recommended include weekly paclitaxel, a combination of gemcitabine and oral etoposide, navelbine, or metronomic cyclophosphamide. | Strong |
Bevacizumab should be recommended in combination with weekly paclitaxel, or topotecan in patients without contraindications to bevacizumab (e.g. increased risk of intestinal fistulae, history of bowel obstruction or serosal invasion). | Strong |
Hormonal therapy ((e.g. aromatase inhibitors, tamoxifen or luteinising hormone-releasing hormone agonists) is recommended for relapsed LGSC with ER and/or PgR expression. | Strong |
Surveillance | |
Surveillance of ovarian cancer patients can include CA-125 determination, physical examination and CT scan evaluation, first year: every three months, second year: every six months, and annually thereafter. | Good Practice Statement
|
- Recommendations
➡️Diagnostic and Staging Work up
▪️ The standard work-up for patients suspected of having EOC (Epithelial Ovarian Carcinoma) should include detailed history and clinical examination, LFTs, KFTs, serum CA-125, serum CEA and CA 19-9 in case of mucinous carcinoma and endoscopy if either or both are elevated, as well as transabdominal and transvaginal US (should be done by an expert examiner), as well as CT of thorax, abdomen and pelvis.
➡️Strong recommendation, low grade evidence (4,5).
▪️ Pathological examination of adequate tumor sample from diagnostic biopsy
or surgical specimen should be done. In case of the presence of pleural effusion,
cytological assessment should be done.
➡️Strong recommendation, low grade evidence (4).
▪️ The revised 2017 FIGO staging system for EOC should be used.
➡️Strong recommendation, high grade evidence (6).[hk1]
➡️Management of early EOC (FIGO STAGE I-II)
▪️Surgical staging is recommended in presumed early-stage ovarian cancer for classification and recommendation of optimal systemic therapy.
➡️Strong recommendation, moderate grade evidence (6).[hk2]
▪️ The aim of surgery for early EOC is complete resection of the tumour and to undertake adequate staging, which should be performed by midline laparotomy and should include:
- Inspection and palpation of the whole abdominal cavity
- Peritoneal washing with cytological examination
- Biopsies from all visible lesions and all abdominal fields
- Bilateral salpingo-oophorectomy
- Hysterectomy
- Omentectomy
- Appendicectomy in MC
- Systematic pelvic and para-aortic lymphadenectomy
➡️Strong recommendation, low grade evidence (7).[hk3]
▪️ Fertility-sparing surgery should be considered in young patients, but always after full discussion with the patient about potential risks.
➡️Strong recommendation, low grade evidence (8).
▪️ Patients with any stage IA histotype or stage IC1-2 with unilateral ovarian involvement and favorable histology (i.e. low-grade tumors) would be amenable to contralateral ovary and uterus preservation, in combination with the other recommended surgical staging procedures.
➡️Strong recommendation, low grade evidence (8).
▪️ Adjuvant chemotherapy in early-stage ovarian cancer is generally recommended for FIGO stage I-IIB (see exceptions below), either paclitaxel-carboplatin or carboplatin alone (six cycles).
➡️Strong recommendation, high grade evidence (9-11).
▪️ The benefit of adjuvant chemotherapy is uncertain and can be considered as optional for:
- Low grade serous carcinoma (LGSC) stage IB-IC
- Clear cell carcinoma (CCC) stage IA-IC1
- Low-grade endometrioid carcinoma (EC) stage IB-IC
- Expansile mucinous carcinoma (MC) stage IC
- Infiltrative MC stage IA
➡️Conditional recommendation, low grade evidence (12-14).
▪️ For patients receiving paclitaxel-carboplatin, a minimum of three cycles are recommended except for high grade serous carcinoma (HGSC) /high-grade endometrioid carcinoma (EC) or any stage IC-II regardless of histotype, for which six cycles should be administered
➡️Strong recommendation, high grade evidence (15,16).
▪️ Adjuvant chemotherapy is not recommended in completely staged patients with LGSC stage IA, low-grade EC stage IA or expansile MC stage IA-IB.
Conditional recommendation, moderate grade evidence [hk4] (10,12-14).
➡️Management of advanced EOC (FIGO STAGE III-IV)
▪️ Patients with advanced EOC should be evaluated for primary cytoreductive surgery (PCS) by a specialized team, with the aim of achieving complete cytoreduction (absence of all visible residual disease).
➡️Strong recommendation moderate grade evidence (7,17,18).
▪️ When complete cytoreductive surgery is feasible, PCS is recommended; otherwise, obtaining adequate biopsy tissue for histology and molecular testing is recommended.
➡️Strong recommendation, low grade evidence (19).
▪️PCS should aim to maximal surgical effort and may require intestinal resection, diaphragmatic and peritoneal stripping, splenectomy and removal of bulky para-aortic lymph nodes and, in some cases, extra-abdominal disease.
➡️Strong recommendation, low grade evidence (17,18).
▪️ We recommend against systematic lymphadenectomy in patients with macroscopic complete resection and clinically negative nodes as this may lead to unnecessarily increases the rate of post-operative complications and mortality and should not be done.
➡️Strong recommendation, low grade evidence (20).
▪️ PCS is also recommended in patients with less chemo-sensitive subtypes (e.g. MC or LGSC), even if uncertainty about achieving complete resection exists and a small residual tumour (<1 cm) is likely to remain.
➡️Strong recommendation, low grade evidence (19).
▪️ When complete cytoreductive surgery is not feasible, neoadjuvant chemotherapy (NACT) for three cycles followed by interval cytoreductive surgery (ICS) and three cycles of paclitaxel-carboplatin are recommended, + staging laparoscopy.
➡️ Strong recommendation, high grade evidence (21,22).
▪️ Consider the use of bevacizumab in the neoadjuvant setting, before interval cytoreductive surgery (ICS).
➡️Conditional recommendation, moderate grade evidence (23-27).
▪️ When ICS is not possible, and in the absence of overt disease progression, three additional cycles of paclitaxel-carboplatin alone or with bevacizumab are
recommended.
➡️Strong recommendation, high grade evidence (21,22).
▪️ Paclitaxel (175 mg/m2)-carboplatin (AUC 5-6) every 3 weeks for six cycles is the standard first-line chemotherapy in advanced ovarian cancer.
Strong recommendation, high grade evidence (28,29).
▪️ We recommend the schedule of weekly chemotherapy with paclitaxel (60 mg/m2)-carboplatin (AUC 2) as an alternative in frail patients.
Strong recommendation, high grade evidence (30).
▪️ Bevacizumab may be considered in addition to paclitaxel-carboplatin in high-risk patients, (defined as patients with stage III and macroscopic residual tumour >1 cm or stage IV).
➡️Conditional recommendation, high grade evidence (31).
▪️ Bevacizumab dose, if given, should be 7.5 mg/kg and the duration of treatment is 12 months.
➡️Strong recommendation, high grade evidence (31).
▪️ Intraperitoneal chemotherapy and hyperthermic intraperitoneal chemotherapy (HIPEC) are not considered a standard of care in first-line treatment.
➡️Conditional recommendation, high grade evidence (32,33).
▪️ Maintenance with anti-estrogen therapy after first-line platinum-based chemotherapy can be considered in ER positive low grade serous carcinoma (LGSC) or grade I endometrioid carcinoma.
Conditional recommendation, very low grade evidence (34).
▪️ Other recommended regimens (other than paclitaxel/carboplatin regime) such as docetaxel/carboplatin or 5FU/ calcium leucovorin/oxaliplatin or Capecitabin/Oxaliplatin specially in mucinous carcinoma may be used.
➡️Conditional recommendation, high grade evidence (35,36).
▪️ The following should be assessed when selecting treatment for patients with recurrent disease:
- Histotype
- Number of prior lines of treatment
- Exposure and response to prior treatment
- TFIp (treatment-free interval from last platinum)
- Possibility of achieving a complete secondary surgical
cytoreduction
- Residual chemotherapy toxicity
- The patient’s general condition and preferences
▪️ Patients with first relapse of ovarian cancer after >6 months of last platinum administration should be evaluated by a team experienced in surgery for ovarian cancer to identify potential candidates for surgical cytoreduction.
➡️ Strong recommendation, high grade evidence (37-40).
▪️ Patients who have previously responded to platinum without early symptomatic relapses (after >6 months) should be treated with either a platinum-based doublet (paclitaxel or gemcitabine with bevacizumab) or single agent (liposomal doxorubicin)
The selection should be based on safety and patient preference.
➡️Strong recommendation, high grade evidence (41,42).
▪️If combination therapy is contraindicated, carboplatin monotherapy should be used. Treatment is usually recommended for four to six cycles.
➡️Strong recommendation, high grade evidence (43).
▪️ Bevacizumab should be continued until disease progression (symptomatic) or the next line of treatment is started, as continuation of bevacizumab beyond progression has not been evaluated in the recurrent setting.
➡️Strong recommendation, high grade evidence (44).
▪️ Platinum rechallenge following treatment with a platinum regimen (monotherapy or combination) should be considered if the tumour is not refractory or resistant.
➡️ Strong recommendation, low grade evidence (45).
▪️ Patients with relapsed EOC for whom platinum is not an option should be defined by:
- Proven refractory (progression during platinum)
- Expected resistance (early symptomatic progression
post-platinum, response to rechallenge unlikely)[hk5]
- Platinum intolerance
- Patient choice
- QoL issues
➡️Good Practice Statement
▪️ For patients who are not candidates to receive platinum, integrating palliative care early in the treatment pathway is strongly recommended.
➡️Good Practice Statement
▪️ Single-agent non-platinum options that are recommended include weekly paclitaxel, a combination of gemcitabine and oral etoposide, navelbine, or metronomic cyclophosphamide.
➡️ Strong recommendation, high grade evidence (46-51).
▪️ Bevacizumab should be recommended in combination with weekly paclitaxel, or topotecan in patients without contraindications to bevacizumab (e.g. increased risk of intestinal fistulae, history of bowel obstruction or serosal invasion).
➡️Strong recommendation, high grade evidence (49).
▪️ Hormonal therapy ((e.g. aromatase inhibitors, tamoxifen or luteinising hormone-releasing
hormone agonists) is recommended for relapsed LGSC with ER and/or PgR expression.
➡️ Strong recommendation, high grade evidence (52).
Surveillance of ovarian cancer patients can include CA-125 determination, physical examination and CT scan evaluation, first year: every three months, second year: every six months, and annually thereafter.
➡️Good Practice Statement
➡️ Clinical Indicators
- Visits every 2–4 months for 2 years, then 3–6 months for 3 years, then annually after 5 years
- Physical exam including pelvic exam as clinically indicated
- C/A/P CT, MRI, PET/CT, or PET (skull base to mid-thigh) as clinically indicated
- CBC and chemistry profile as indicated
- CA-125 or other tumor markers if initially elevated
➡️Update of this guideline
This guideline will be updated whenever there is new evidence.
➡️ Research gaps
- Systematic inclusion of cost-benefit analyses in clinical trials with collection of health economic analysis such as incremental cost effectiveness ratio in order to facilitate clinical decision-making.
- Predictive biomarkers: response to specific systemic targeted therapies and immunotherapy.
- Improve models for pre-clinical testing of novel drugs.
- Search for tools to assess quality of life and in clinical trials.
- Dietary supplements, nutritional counselling, physical activity recommendations and psychological support as part of an integrative healthcare approach to care for people with ovarian cancer.
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines, for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines: Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Yousri Wasef & Yousri Rostom.
- Abbreviations
|
ASIR |
Age Standardized Incidence Rate |
|
AUC |
Area under the curve |
|
CCC |
Clear cell carcinoma |
|
CT |
Computed tomography |
|
EC |
Endometrioid carcinoma |
|
EHC |
Egyptian Health Council |
|
EOC |
Epithelial Ovarian Carcinoma |
|
ESMO |
European Society of Medical Oncology |
|
HGSC |
High grade serous carcinoma |
|
HIPEC |
Hyperthermic intraperitoneal chemotherapy |
|
ICS |
Interval cytoreductive surgery |
|
KFTs, |
Kidney function tests |
|
LFTs |
Liver function tests |
|
LGSC |
Low grade serous carcinoma |
|
MC |
Mucinous carcinoma |
|
NACT |
Neoadjuvant chemotherapy |
|
NCCN |
National Comprehensive Cancer Network |
|
NICE |
National Institute of Health and Care Excellence |
|
PCS |
Primary cytoreductive surgery |
|
TFIp |
Treatment-free interval from last platinum |
|
US |
Ultrasound |
|
WHO |
World Health Organization |
- Introduction
In Egypt, there was an estimated 3070 new cases of ovarian cancer with an ASIR of 6.4/100 000 normal population and ranks the 4th most common cancer among females (4%). There were 1944 deaths because of this disease based on GLOBOCAN 2022.
- Purpose and scope
These guidelines are developed to improve the quality of care for ovarian cancer via providing a uniform standard of care across the country to help in early diagnosis, treatment and follow up for epithelial ovarian cancer so more optimal treatment options and improved clinical outcomes.
- Target audience
Clinicians who are involved in the care and treatment of patients with Ovarian cancer, include medical oncologists, radiation oncologists, clinical oncologist, gynecologists, onco-surgeons, radiologists, pathologists, and palliative care specialists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The NCCN (National Comprehensive Cancer Network), ESMO (European Society of Medical Oncology), NICE (National Institute of Health and Care Excellence) guidelines are the main sources used while formulating the national guidelines for ovarian cancer (1-3).
- Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group: https://www.gradeworkinggroup.org/
. GRADE online training modules:http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
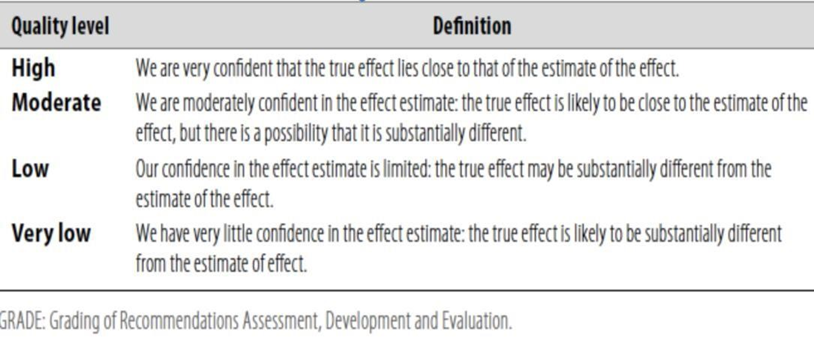
Table 2: Significance of the four levels of evidence

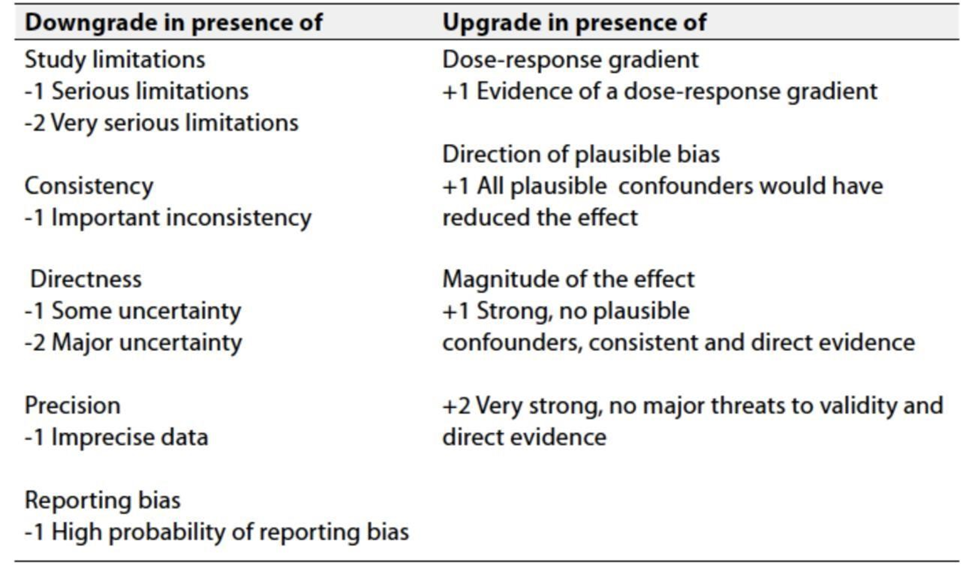
Table 3: Factors that determine How to upgrade or downgrade the quality of evidence
➡️ The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- References
1.https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1453.
2.https://www.esmo.org/guidelines/guidelines-by-topic/esmo-clinical-practice-guidelines-gynaecological-cancers
3.https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/nice-guidelines
4. Timmerman D, Planchamp F, Bourne T, et al. ESGO/ISUOG/IOTA/ESGE Consensus Statement on pre-operative diagnosis of ovarian tumors. Int J Gynecol Cancer. 2021;31(7):961-982.
5. Charkhchi P, Cybulski C, Gronwald J, et al. CA125 and ovarian cancer:a comprehensive review. Cancers. 2020;12(12):3730.
6. Ceska Gynekol, Current FIGO staging classification for cancer of ovary, fallopian tube and peritoneum. 2017 Summer;82(3):230-236.
7. du Bois A, Reuss A, Pujade-Lauraine E, et al. Role of surgical outcomeas prognostic factor in advanced epithelial ovarian cancer: a combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials. Cancer. 2009;115(6):1234-1244.
8. Fruscio R, Corso S, Ceppi L, et al. Conservative management of early stage epithelial ovarian cancer: results of a large retrospective series. Ann Oncol. 2013;24(1):138-144.
9. Trimbos JB, Parmar M, Vergote I, et al. International Collaborative Ovarian Neoplasm trial 1 and Adjuvant ChemoTherapy In Ovarian Neoplasm trial: two parallel randomized phase III trials of adjuvant chemotherapy in patients with early-stage ovarian carcinoma. J Natl Cancer Inst. 2003;95(2):105-112.
10. Collinson F, Qian W, Fossati R, et al. Optimal treatment of early-stage ovarian cancer. Ann Oncol. 2014;25(6):1165-1171.
11. Lawrie TA, Winter-Roach BA, Heus P, et al. Adjuvant (post-surgery) chemotherapy for early stage epithelial ovarian cancer. Cochrane Database Syst Rev. 2015;2015(12):CD004706.
12.Oseledchyk A, Leitao MM Jr, Konner J, et al. Adjuvant chemotherapy in patients with stage I endometrioid or clear cell ovarian cancer in the platinum era: a Surveillance, Epidemiology, and End Results Cohort Study, 2000-2013. Ann Oncol. 2017;28(12):2985-2993.
13. Mizuno M, Kajiyama H, Shibata K, et al. Adjuvant chemotherapy for stage I ovarian clear cell carcinoma: is it necessary for stage IA? Int J Gynecol Cancer. 2012;22(7):1143-1149.
14. Gouy S, Saidani M, Maulard A, et al. Staging surgery in early-stage ovarian mucinous tumors according to expansile and infiltrative types. Gynecol Oncol Rep. 2017;22:21-25.
15 Bell J, Brady MF, Young RC, et al. Randomized phase III trial of three versus six cycles of adjuvant carboplatin and paclitaxel in early stage epithelial ovarian carcinoma: a Gynecologic Oncology Group study. Gynecol Oncol. 2006;102(3):432-439.
16. Chan JK, Tian C, Fleming GF, et al. The potential benefit of 6 vs. 3 cycles of chemotherapy in subsets of women with early-stage highrisk epithelial ovarian cancer: an exploratory analysis of a Gynecologic Oncology Group study. Gynecol Oncol. 2010;116(3):301-306.
17.Ataseven B, Chiva LM, Harter P, et al. FIGO stage IV epithelial ovarian, fallopian tube and peritoneal cancer revisited. Gynecol Oncol. 2016;142(3):597-607.
18. Fotopoulou C, Concin N, Planchamp F, et al. Quality indicators for advanced ovarian cancer surgery from the European Society of Gynaecological Oncology (ESGO): 2020 update. Int J Gynecol Cancer.2020;30(4):436-440.
19. Colombo N, Sessa C, du Bois A, et al. ESMOeESGO consensus conference recommendations on ovarian cancer: pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Ann Oncol. 2019;30(5):672-705.
20. Harter P, Sehouli J, Lorusso D, et al. A randomized trial of lymphadenectomy in patients with advanced ovarian neoplasms. N Engl J Med. 2019;380(9):822-832.
21 Vergote I, Tropé CG, Amant F, et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. N Engl J Med. 2010;363(10):943-953.
22. Kehoe S, Hook J, Nankivell M, et al. Primary chemotherapy versus primary surgery for newly diagnosed advanced ovarian cancer (CHORUS): an open-label, randomised, controlled, non-inferiority trial. Lancet. 2015;386(9990):249-257.
23 Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. 2011;365(26):2473-2483.
24. Perren TJ, Swart AM, Pfisterer J, et al. A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med. 2011;365(26):2484-2496. 22. Pfisterer J, Joly F, Kristensen G, et al. Optimal treatment duration of bevacizumab as front-line therapy for advanced ovarian cancer: AGOOVAR
25.BOOST/GINECO OV118/ENGOT Ov-15 open-label randomized phase III trial. J Clin Oncol. 2023;41(4):893-902.
26. Rouzier R, Gouy S, Selle F, et al. Efficacy and safety of bevacizumabcontaining neoadjuvant therapy followed by interval debulking surgery in advanced ovarian cancer: results from the ANTHALYA trial. Eur J Cancer. 2017;70:133-142.
27. Garcia Garcia Y, de Juan Ferré A, Mendiola C, et al. Efficacy and safety results from GEICO 1205, a randomized phase II trial of neoadjuvant chemotherapy with or without bevacizumab for advanced epithelial ovarian cancer. Int J Gynecol Cancer. 2019;29(6):1050-1056.
28 Stuart GCE, Kitchener H, Bacon M, et al. 2010 Gynecologic Cancer InterGroup (GCIG) consensus statement on clinical trials in ovarian cancer: report from the Fourth Ovarian Cancer Consensus Conference. Int J Gynecol Cancer. 2011;21(4):750-755.
29. BookmanMA.The addition of new drugs to standard therapy in the first line treatment of ovarian cancer. Ann Oncol. 2010;21:vii211-vii217.
30 Pignata S, Scambia G, Katsaros D, et al. Carboplatin plus paclitaxel once a week versus every 3 weeks in patients with advanced ovarian cancer (MITO-7): a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2014;15(4):396-405.
31 Oza AM, Cook AD, Pfisterer J, et al. Standard chemotherapy with or without bevacizumab for women with newly diagnosed ovarian cancer (ICON7): overall survival results of a phase 3 randomised trial.Lancet Oncol. 2015;16(8):928-936.
32 van Driel WJ, Koole SN, Sikorska K, et al. Hyperthermic intraperitoneal chemotherapy in ovarian cancer. N Engl J Med. 2018;378(3):230-240.
33. Lim MC, Chang S-J, Yoo HJ, et al. Randomized trial of hyperthermic intraperitoneal chemotherapy (HIPEC) in women with primary advanced peritoneal, ovarian, and tubal cancer. J Clin Oncol.2017;35(suppl 15):5520.
34 Gershenson DM, Bodurka DC, Coleman RL, et al. Hormonal maintenance therapy for women with low-grade serous cancer of the ovary or peritoneum. J Clin Oncol. 2017;35(10):1103-1111.
36.Ledermann JA, Luvero D, Shafer A, et al. Gynecologic Cancer InterGroup (GCIG) consensus review for mucinous ovarian carcinoma. Int J Gynecol Cancer 2014;24:S14-19.
37. Harter P, Sehouli J, Reuss A, et al. Prospective validation study of a predictive score for operability of recurrent ovarian cancer: the Multicenter Intergroup Study DESKTOP II. A project of the AGO Kommission OVAR, AGO Study Group, NOGGO, AGO-Austria, and MITO. Int J Gynecol Cancer. 2011;21(2):289-295.
38. Harter P, Sehouli J, Vergote I, et al. Randomized trial of cytoreductive surgery for relapsed ovarian cancer. N Engl J Med. 2021;385(23): 2123-2131.
39. Shi T, Zhu J, Feng Y, et al. Secondary cytoreduction followed by chemotherapy versus chemotherapy alone in platinum-sensitive relapsed ovarian cancer (SOC-1): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2021;22(4):439-449.
40.Coleman RL, Brady MF, Herzog TJ, et al. Bevacizumab and paclitaxel carboplatin chemotherapy and secondary cytoreduction in recurrent, platinum-sensitive ovarian cancer (NRG Oncology/Gynecologic Oncology Group study GOG-0213): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2017;18(6):779-791.
41. Pfisterer J, Plante M, Vergote I, et al. Gemcitabine plus carboplatin compared with carboplatin in patients with platinum-sensitive recurrent ovarian cancer: an intergroup trial of the AGO-OVAR, the NCIC CTG, and the EORTC GCG. J Clin Oncol. 2006;24(29):4699-4707.
42.Parmar MK, Ledermann JA, Colombo N, et al. Paclitaxel plus platinum-based chemotherapy versus conventional platinum-based chemotherapy in women with relapsed ovarian cancer: the ICON4/ AGO-OVAR-2.2 trial. Lancet. 2003;361(9375):2099-2106.
43 Raja FA, Counsell N, Colombo N, et al. Platinum versus platinum combination chemotherapy in platinum-sensitive recurrent ovarian cancer: a meta-analysis using individual patient data. Ann Oncol.2013;24(12):3028-3034.
44.Pignata S, Lorusso D, Joly F, et al. Carboplatin-based doublet plus bevacizumab beyond progression versus carboplatin-based doublet alone in patients with platinum-sensitive ovarian cancer: a randomised, phase 3 trial. Lancet Oncol. 2021;22(2):267-276.
45 González-Martín A, Harter P, Leary A, Lorusso D, Miller RE, Pothuri B, Ray-Coquard I, Tan DSP, Bellet E, Oaknin A, Ledermann JA, Newly diagnosed and relapsed epithelial ovarian cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023 Oct;34(10):833-848.
46. Spiliopoulou P, Hinsley S, McNeish IA, et al. Metronomic oral cyclophosphamide in relapsed ovarian cancer. Int J Gynecol Cancer. 2021;31(7):1037-1044.
47. Markman M, Blessing J, Rubin SC, et al. Phase II trial of weekly paclitaxel (80 mg/m2) in platinum and paclitaxel-resistant ovarian and primary peritoneal cancers: a Gynecologic Oncology Group study. Gynecol Oncol 2006;101:436-440.
48. Ferrandina G, Ludovisi M, Lorusso D, et al. Phase III trial of gemcitabine compared with pegylated liposomal doxorubicin in progressive or recurrent ovarian cancer. J Clin Oncol 2008;26:890-896.
49. Pujade-Lauraine E, Hilpert F, Weber B, et al. Bevacizumab combined with chemotherapy for platinum-resistant recurrent ovarian cancer: The AURELIA open-label randomized phase III trial. J Clin Oncol 2014;32:1302-1308.
50. Rose PG, Blessing JA, Mayer AR, Homesley HD. Prolonged oral etoposide as second-line therapy for platinum-resistant and platinum-sensitive ovarian carcinoma: a Gynecologic Oncology Group study. J Clin Oncol 1998;16:405-410.
51. Rothenberg ML, Liu PY, Wilczynski S, et al. Phase II trial of vinorelbine for relapsed ovarian cancer: a Southwest Oncology Group study. Gynecol Oncol 2004;95:506-512.
52. Gershenson DM, Bodurka DC, Coleman RL, et al. Hormonal maintenance therapy for women with low-grade serous cancer of the ovary or peritoneum. J Clin Oncol. 2017;35(10):1103-1111.
- Annex.
Table 1. FIGO staging system for EOC
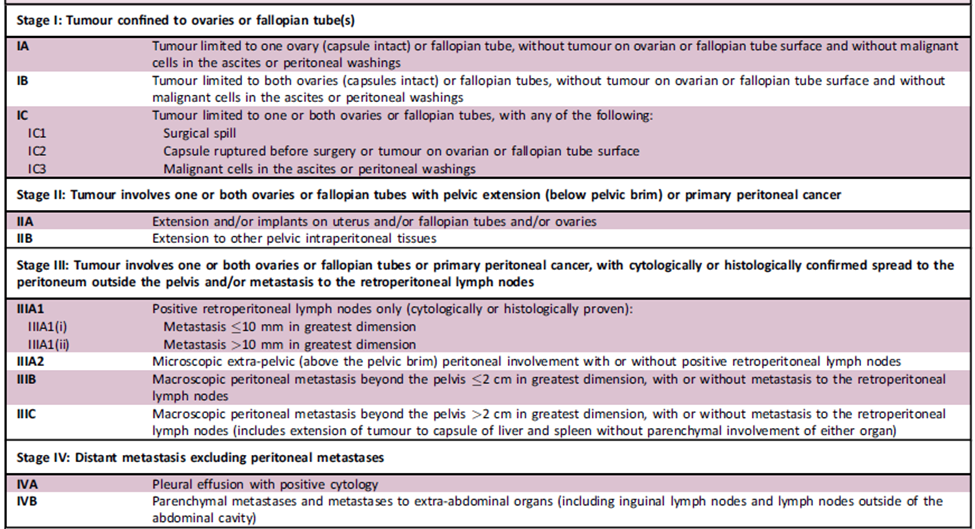
EOC, epithelial ovarian cancer; FIGO, International Federation of Gynecology and Obstetrics.