Wilms’ tumor
| الموقع: | EHC | Egyptian Health Council |
| المقرر الدراسي: | اورام أطفال |
| كتاب: | Wilms’ tumor |
| طبع بواسطة: | Guest user |
| التاريخ: | الأربعاء، 6 مايو 2026، 3:45 AM |
الوصف
"last update: 12 March 2025" Download Guideline
- Executive Summary
This guidance provides a data-supported approach to the diagnosis, risk stratification, treatment and follow up of paediatric patients diagnosed with Wilms’ tumor.
|
Level Of Recommendations |
|
|
1-Work up for newly diagnosed Wilms’ tumor |
|
|
We recommend complete assessment for signs of associated syndromes including blood pressure measurement and urine analysis. |
Strong Recommendations |
|
Contrast enhanced CT abdomen and pelvis or MRI is recommended (to assess bilaterality, evidence of tumor rupture and evidence of tumor thrombus extension into the renal vein or inferior vena cava, nephrogenic rests) |
Strong Recommendations |
|
CT chest is recommended to assess for metastasis. |
Strong Recommendations |
|
For stage IV WT, we recommend local staging to determine local therapy. |
Strong Recommendations |
|
2-Treatment of unilateral WT with no predisposition to develop bilateral WT |
|
|
We recommend primary nephrectomy with regional LN sampling (5-10 nodes) for all patients, followed by adjuvant treatment according to stage and histology. |
Strong Recommendations |
|
If initially unresectable or resection is contraindicated, we recommend proceeding to chemotherapy without biopsy (either image guided core needle biopsy or open). |
Strong Recommendations |
|
We recommend either image guided core needle biopsies, or open biopsy to confirm WT pathology in the following conditions: · Age < 1year or older than 10 years · Uncertain renal origin · Atypical metastases: bones (any age), central nervous system (any age), isolated pulmonary nodules < 2years. · Elevated LDH > 3-4 folds · Hypercalcaemia and age<4 years. |
Strong recommendations |
|
Management of initially resected WT with no predisposition to develop bilateral WT |
|
|
We recommend the following adjuvant treatment: |
|
|
1. Favorable histology WT: · Regimen EE-4A for LR patients · Regimen DD-4A for SR patients · Regimen M for HR patients. |
Strong Recommendations |
|
2. Focal anaplastic WT: · Stages I-III: Regimen DD4-A · Stage IV: Regimen UH-HR |
Strong Recommendations |
|
3. Diffuse anaplastic WT: · Stage I: Regimen DD4A. · Stages III-IV: Regimen UH-HR |
Strong Recommendations |
|
Management of initially unresectable WT with no predisposition to develop bilateral WT |
|
|
We recommend neoadjuvant treatment, regimen (DD-4A) for initially unresectable tumors. |
Strong Recommendations |
|
We recommend reassessment at week 6 by contrast enhanced CT chest, abdomen and pelvis. |
Strong Recommendations |
|
We recommend total nephrectomy and LN sampling at week 6, if feasible. If not feasible, we recommend continuing (DD-4A) till week 12, followed by reassessment and surgery. |
Strong Recommendations |
|
We recommend the following postoperative adjuvant treatment for initially unresectable WT: |
|
|
· Continuing Regimen DD-4A for SR favorable histology WT, stages I-III focal anaplastic WT and stage I diffuse anaplastic WT. |
Strong Recommendations
|
|
· Switching to Regimen M for HR favorable histology WT. |
|
|
· Switching to Regimen I for blastemal predominant histology. |
|
|
· Switching to Regimen UH-HR for stage IV focal anaplastic WT and stages II-IV diffuse anaplastic WT. |
|
3. Treatment of bilateral WT and unilateral WT with predisposition to develop bilateral WT: |
|
|
We do not recommend upfront nephrectomy either partial or radical.
|
Strong Recommendations |
|
We do not recommend upfront biopsy (either needle or open). If biopsied, a tumor is stage III for determination of chemotherapy regimen, but biopsy alone does not upstage a tumor to stage III for determining whether to give radiation. |
Strong Recommendations |
|
We recommend neoadjuvant treatment (VAD) for 6 weeks. |
Strong Recommendations |
|
We recommend reassessment with contrast enhanced CT chest, abdomen and pelvis at week (6) VAD. |
Strong Recommendations |
|
We recommend bilateral partial nephrectomy (one or both sides) at week 6, if feasible. |
Strong Recommendations |
|
If bilateral partial nephrectomy at week 6 is not feasible, assess for response: · Partial response in both kidneys, we recommend continuing VAD regimen till week 12 then reassess for bilateral partial nephrectomy or total nephrectomy with LN sampling followed by adjuvant treatment based on higher risk histology.
· Less than partial response (<50% reduction of tumor size) in either kidney, we recommend either immediate surgery or bilateral open biopsies followed by adjuvant treatment based on higher risk histology, reimage at week 12 for definitive surgery. |
Strong Recommendations |
|
We recommend the following adjuvant treatment after surgery or biopsy in bilateral WT determined by the highest assigned stage/histology of either kidney: |
Strong Recommendations |
|
EE-4A regimen is recommended for · Stage I - CR with no lesion detectable on imaging after preoperative chemotherapy at week 6, or · Stage I-II favorable histology WT after complete resection or completely necrotic. |
|
|
DD-4A regimen is recommended for · Stage III-IV favorable histology WT, or · Stage III-IV completely necrotic, or · Stage I favorable histology WT with blastemal predominant histology, or · Stage I-III focal anaplastic WT, or · Stage I diffuse anaplastic WT |
|
|
Regimen I is recommended for · Stage II-IV favorable histology WT with blastemal predominant histology. |
|
|
Regimen UH (start week 1) is recommended for · Stage IV focal anaplastic WT, or · Stages II-IV diffuse anaplastic WT |
|
|
4. Management of extrapulmonary metastasis stage IV WT |
|
|
· We recommend upgrading to regimen M in stage IV favorable histology WT and irradiation to all metastatic sites post nephrectomy. |
Strong recommendation |
|
· We do not recommend liver irradiation in the following condition only: Solitary liver metastasis, at presentation (before chemotherapy) completely resected and negative margins with nephrectomy. |
Strong recommendation |
|
· We recommend regimen UH for stage IV anaplastic histology (focal or diffuse) and irradiation to all metastatic sites |
Strong recommendation |
|
5. Radiotherapy (RT): |
|
|
Post nephrectomy (either upfront or delayed), RT should begin close to chemotherapy, preferably by Day 10 (surgery is Day 0), but no later than Day 14. |
Strong Recommendations |
|
Flank RT (10.8 Gy at 1.8 Gy per fraction) is recommended in unilateral WT, bilateral WT and unilateral WT with predisposition to develop WT under the following conditions: · Locally stage III favorable histology WT. · Locally stage I-III focal anaplasia · Locally stage I-II diffuse anaplasia |
Strong Recommendations
|
|
Flank RT (19.8 Gy at 1.8 Gy per fraction) is recommended in unilateral WT, bilateral WT and unilateral WT with predisposition to develop WT under the following conditions: · Locally stage III diffuse anaplastic histology WT. |
Strong Recommendation |
|
For bilateral WT and unilateral WT with predisposition to develop WT, we do not recommend flank RT in these conditions: · Biopsy alone, however reported surgical tumor spill will require RT. · Complete resection with negative surgical margins/nodes. |
Strong Recommendations |
|
Whole abdomen RT (10.5 Gy at 1.5 Gy per fraction) is recommended in all patients with: · Cytology positive ascites · Preoperative rupture · Diffuse abdominal surgical spillage (reported by surgeon) · Peritoneal seeding, in case of diffuse peritoneal implants (21Gy in 1.5 fractions) |
Strong Recommendations |
|
WLI is not recommended in unilateral favorable histology WT with pulmonary metastasis showing RCR post week 6 |
Strong Recommendations |
|
WLI (at week 7) (12 Gy at 1.5 Gy per fraction or 10.5 Gy at 1.5 Gy per fraction if <12 months) is recommended in patients with: · Unilateral favorable histology WT with pulmonary metastasis showing slow incomplete response at week 6 (with or without surgical excision of residual metastases). · Metastatic bilateral and unilateral WT with predisposition to develop bilateral WT. · Pulmonary metastasis and other extra-thoracic metastases (such as liver, bone, or brain). · LN metastases in the hilum and/or mediastinum, or cytology-positive pleural effusion regardless of response to chemotherapy. |
Strong Recommendations |
|
We recommend dactinomycin and doxorubicin reduction by 50% during or within 6 weeks of completing a course of whole lung or abdominal RT. |
Strong recommendation |
|
|
|
|
We recommend complete evaluation for both local and metastatic sites at relapse with contrast enhanced CT chest, abdomen and pelvis |
Strong Recommendations |
|
We recommend biopsy from site of recurrence to confirm WT relapse. |
Strong recommendation |
|
Standard risk |
|
|
We recommend 2nd line chemotherapy as first treatment using four drugs (combinations of alternating courses of doxorubicin and cyclophosphamide and carboplatin and etoposide) |
Strong Recommendations |
|
We recommend surgical resection with clear resection margins if feasible |
Strong recommendation |
|
We recommend flank RT or whole abdomen irradiation in case of peritoneal extension or ascites. |
Strong recommendation |
|
High and higher risk: |
Strong recommendation |
|
We recommend combination chemotherapy (ICE/CCE) alternating with topotecan/cyclophosphamide up to 10 cycles if feasible |
Strong Recommendations |
|
We recommend surgical resection and consolidation with RT if feasible for local and metastatic sites. |
Strong Recommendations |
|
Autologous bone marrow transplantation is recommended for patients with chemo-sensitive relapse who are not candidates for RT consolidation. |
Strong Recommendations |
|
7- End of treatment evaluation |
|
|
We recommend end of treatment evaluation by contrast enhanced CT chest, abdomen and pelvis to confirm CR before starting follow up. |
Strong Recommendations |
|
8- Surveillance (follow up after end of treatment) |
|
|
We recommend clinical examination together with chest and abdominal imaging every 3 months for 2 years, then every 6 months for 5 years. (Chest x-ray and abdominal US may be used in place of cross-sectional imaging with CT chest and abdomen with IV contrast or MRI). |
Strong recommendation |
|
9- Screening recommendations predisposed patients to develop bilateral WT |
|
|
We recommend renal US every 3 months until 7 years (ie, all of year 6). |
Strong recommendation |
- Recommendations
1-Work up for newly diagnosed Wilms’ tumor
We recommend complete assessment for signs of associated syndromes including blood pressure measurement and urine analysis.
strong recommendation, high quality evidence (retrospective analysis, COG) (11)
Contrast enhanced CT abdomen and pelvis or MRI is recommended (to assess bilaterality, evidence of tumor rupture and evidence of tumor thrombus extension into the renal vein or inferior vena cava, nephrogenic rests).
strong recommendation, high quality evidence (retrospective analysis, COG) (12)
CT chest is recommended to assess for metastasis.
strong recommendation, high quality evidence (randomized trials, COG) (13)
For stage IV WT, we recommend local staging to determine local therapy.
strong recommendation, high quality evidence (COG randomized trial, COG prospective analysis) (14,15)
2-Treatment of unilateral WT with no predisposition to develop bilateral WT
We recommend primary nephrectomy with regional LN sampling (5-10 nodes) for all patients, followed by adjuvant treatment according to stage and histology.
strong recommendation, high quality evidence (systematic review, COG retrospective analysis) (16)
If initially unresectable or resection is contraindicated, we recommend proceeding to chemotherapy without biopsy (either image guided core needle biopsy or open).
strong recommendation, high quality evidence (systematic review, SIOP RTSG) (17)
We recommend either image guided core needle biopsies, or open biopsy to confirm WT pathology in the following conditions
• Age < 1year or older than 10 years
• Uncertain renal origin
• Atypical metastases: bones (any age), central nervous system (any age), isolated pulmonary nodules < 2years.
· Elevated LDH >3-4 folds
• Hypercalcaemia and age<4 years.
strong recommendation, high quality evidence (systematic review, SIOP RTSG). (18)
Management of initially resected WT with no predisposition to develop bilateral WT
We recommend the following adjuvant treatment:
1. Favorable histology WT:
• Regimen EE-4A for LR patients
• Regimen DD-4A for SR patients
• Regimen M for HR patients.
strong recommendation, high quality evidence (COG randomized trials). (19,20,21)
2. Focal anaplastic WT:
• Stages I-III: Regimen DD4-A
• Stage IV: Regimen UH-HR
strong recommendation, high quality evidence (COG report). (22)
3. Diffuse anaplastic WT:
• Stage I: Regimen DD4A.
• Stages II-IV: Regimen UH-HR,
strong recommendation, high quality evidence (COG report, COG prospective analysis). (23,24)
Management of initially unresectable WT with no predisposition to develop bilateral WT
We recommend neoadjuvant treatment, regimen (DD-4A) for initially unresectable tumors.
We recommend reassessment at week 6 by contrast enhanced CT chest, abdomen and pelvis.
We recommend total nephrectomy and LN sampling at week 6, if feasible. If not feasible, we recommend continuing (DD-4A) till week 12, followed by reassessment and surgery.
strong recommendations, high quality evidence (COG retrospective analysis, systematic review, SIOP randomized trial). (25,26,27)
We recommend the following postoperative adjuvant treatment for initially unresectable WT:
• Continuing Regimen DD-4A for SR favorable histology WT, stages I-III focal anaplastic WT and stage I diffuse anaplastic WT.
• Switching to Regimen M for HR favorable histology WT.
strong recommendation, high quality evidence (COG randomized trials). (19,20,21)
• Switching to Regimen I for blastemal predominant histology.
strong recommendation, high quality evidence (SIOP randomized trial). (28)
• Switching to Regimen UH-HR for stage IV focal anaplastic WT and stages II-IV diffuse anaplastic WT.
strong recommendation, high quality evidence (COG report, COG prospective analysis). (23,24)
3. Treatment of bilateral WT and unilateral WT with predisposition to develop bilateral WT:
We do not recommend upfront nephrectomy either partial or radical.
We do not recommend upfront biopsy (either needle or open). If biopsied, a tumor is stage III for determination of chemotherapy regimen, but biopsy alone does not upstage a tumor to stage III for determining whether to give radiation.
We recommend neoadjuvant treatment (VAD) for 6 weeks.
We recommend reassessment with contrast enhanced CT chest, abdomen and pelvis at week (6) VAD.
We recommend bilateral partial nephrectomy (one or both sides) at week 6, if feasible.
If bilateral partial nephrectomy at week 6 is not feasible, assess for response:
• Partial response in both kidneys, we recommend continuing VAD regimen till week 12 then reassess for bilateral partial nephrectomy or total nephrectomy with LN sampling followed by adjuvant treatment based on higher risk histology.
• Less than partial response (<50% reduction of tumor size) in either kidney, we recommend either immediate surgery or bilateral open biopsies followed by adjuvant treatment based on higher risk histology, reimage at week 12 for definitive surgery.
We recommend the following adjuvant treatment after surgery or biopsy in bilateral WT determined by the highest assigned stage/histology of either kidney:
EE-4A regimen is recommended for
• Stage I - CR with no lesion detectable on imaging after preoperative chemotherapy at week 6, or
• Stage I-II favorable histology WT after complete resection or completely necrotic.
DD-4A regimen is recommended for
• Stage III-IV favorable histology WT, or
• Stage III-IV completely necrotic, or
• Stage I favorable histology WT with blastemal predominant histology, or
• Stage I-III focal anaplastic WT, or
• Stage I diffuse anaplastic WT
Regimen I is recommended for
• Stage II-IV favorable histology WT with blastemal predominant histology.
Regimen UH (start week 1) is recommended for
• Stage IV focal anaplastic WT, or
• Stages II-IV diffuse anaplastic WT
strong recommendation, high quality evidence (COG prospective analysis, COG retrospective analysis). (29,30)
4. Management of extrapulmonary metastasis stage IV WT
We recommend upgrading to regimen M in stage IV favorable histology WT and irradiation to all metastatic sites post nephrectomy.
We do not recommend liver irradiation in the following condition only:
· Solitary liver metastasis, at presentation (before chemotherapy) completely resected and negative margins with nephrectomy.
We recommend regimen UH for stage IV anaplastic histology (focal or diffuse) and irradiation to all metastatic sites
strong recommendation, high quality evidence (COG prospective analysis,). (20,30,31)
5. Radiotherapy:
Timing of RT:
Post nephrectomy (either upfront or delayed), RT should begin close to chemotherapy, preferably by Day 10 (surgery is Day 0), but no later than Day 14.
strong recommendation, high quality evidence (COG reports). (32,33)
Flank RT (10.8 Gy at 1.8 Gy per fraction) is recommended in both unilateral (either with or without predisposing conditions) and bilateral WT, with:
• Locally stage III favorable histology WT.
• Locally stage I-III anaplasia (focal and diffuse).
We do not recommend flank RT in bilateral WT after biopsy alone, however reported surgical tumor spill will require RT.
strong recommendation, high quality evidence (COG reports). (33,34.35)
Whole abdomen RT (10.5 Gy at 1.5 Gy per fraction) is recommended in all patients with:
• Diffuse intraoperative spillage (reported by the surgeon)
• Preoperative rupture
strong recommendation, high quality evidence (COG reports). (37)
WLI is not recommended in unilateral favorable histology WT with pulmonary metastasis showing RCR post week 6 (with or without surgical excision of residual metastases).
strong recommendation, high quality evidence (COG reports). (20)
WLI (at week 7) (12 Gy at 1.5 Gy per fraction or 10.5 Gy at 1.5 Gy per fraction if <12 months) is recommended in patients with:
• Unilateral favorable histology WT with pulmonary metastasis showing slow incomplete response at week 6 (with or without surgical excision of residual metastases).
• Metastatic bilateral and unilateral WT with predisposition to develop bilateral WT.
• Pulmonary metastasis and other extra-thoracic metastases (such as liver, bone, or brain).
• LN metastases in the hilum and/or mediastinum, or cytology-positive pleural effusion regardless of response to chemotherapy.
We recommend dactinomycin and doxorubicin reduction by 50% during or within 6 weeks of completing a course of whole lung or abdominal RT.
strong recommendation, high quality evidence (COG reports). (21,33)
6-Treatment of relapse or refractory disease
We recommend complete evaluation for both local and metastatic sites at relapse with contrast enhanced CT chest, abdomen and pelvis
We recommend biopsy from site of recurrence to confirm WT relapse.
Standard risk
We recommend 2nd line chemotherapy as first treatment using four drugs (combinations of alternating courses of doxorubicin and cyclophosphamide and carboplatin and etoposide)
We recommend surgical resection with clear resection margins if feasible
We recommend flank RT or whole abdomen irradiation in case of peritoneal extension or ascites.
strong recommendation, high quality evidence (COG prospective analysis). (38)
High and higher risk:
We recommend combination chemotherapy (ICE/CCE) alternating with topotecan/cyclophosphamide up to 10 cycles if feasible
We recommend surgical resection and consolidation with RT if feasible for local and metastatic sites.
strong recommendation, high quality evidence (COG prospective analysis, multi-institutional study,). (39,40)
Autologous bone marrow transplantation is recommended for patients with chemo-sensitive relapse who are not candidates for RT consolidation.
strong recommendation, high quality evidence (prospective studies). (41,42,43)
7- End of treatment evaluation
We recommend end of treatment evaluation by contrast enhanced CT chest, abdomen and pelvis to confirm CR before starting follow up.
8- Surveillance (follow up after end of treatment)
We recommend clinical examination together with chest and abdominal imaging every 3 months for 2 years, then every 6 months for 5 years.
(Chest x-ray and abdominal US may be used in place of cross-sectional imaging with chest CT and abdominal CT or MRI).
strong recommendation, high quality evidence (prospective analysis). (44)
9- Screening recommendations predisposed patients to develop bilateral WT
We recommend renal US every 3 months until 7 years (ie, all of year 6).
strong recommendation, high quality evidence (prospective observational study). (45)
➡️Clinical indicators for monitoring:
· Contrast enhanced CT abdomen and pelvis with IV contrast or MRI abdomen.
· CT chest.
· Upfront surgical resection for unilateral WT, otherwise for preoperative chemotherapy
· Upfront biopsy is contraindicated in bilateral WT or unilateral WT with predisposition to develop bilateral WT instead preoperative chemotherapy and management according to response
· Chemotherapy regimens according to histology
· Radiotherapy referral and start ideally day 10 postoperative no more than 14 days.
➡️Update of this guideline
This guideline will be updated whenever there is new evidence.
- Acknowledgment
We would like to acknowledge the Guidelines Development Group (GDG) of Paediatric Oncology for adapting this Guideline.
• Chair of the Committee:
Prof Alaa El-Haddad (Professor of Paediatric Oncology and Former Dean of the National Cancer Institute - Cairo University, Head of the Paediatric Oncology Department and the Bone Marrow Transplant Unit at the Children’s Cancer Hospital Cairo, Egypt).
• The Scientific Committee Members:
Prof Mahmoud Hammad (Committee Rapporteur and Professor of Paediatric Oncology - National Cancer Institute - Cairo University - and Director of the Oncology and Nuclear Medicine Centre at Nasser Institute Hospital).
Prof Iman Sidhom (Professor and Head of Department of Paediatric Oncology - National Cancer Institute - Cairo University).
Prof Emad Ebied (Deputy Director, National Cancer Institute and Professor of Paediatric Oncology - National Cancer Institute - Cairo University).
Prof Youssef Madney (Professor of Paediatric Oncology - National Cancer Institute - Cairo University - and Consultant of Paediatric Oncology at Dar Al Salam Cancer Hospital Harmal).
Dr Ahmed Mustafa (Lecturer of Paediatric Oncology - National Cancer Institute - Cairo University - and Assistant Head of the Specialized Medical Centres Secretariat for Oncology Affairs).
Dr Zaki Ahmed Zaki (Consultant and Director, Haematology Unit at Sheikh Zayed Specialized Hospital).
Dr Amal Ahmed Zein (Paediatric Oncology Consultant at Al-Sahel Teaching Hospital- and at Oncology Centre of Nasser Institute Hospital).
Prof Shady Fadel (Assistant Professor, Faculty of Medicine, Alexandria University - Director and Head, Paediatric Oncology Department, Borg El Arab Hospital).
Dr Mahmoud Motaz (Lecturer, South Egypt Oncology Institute, Assiut University, Medical Director and Head of the Paediatric Oncology Department at Shifa Al Orman Oncology Centre).
Dr Esraa Maged (Lecturer of Paediatric Oncology at National Cancer Institute – Cairo University).
Dr Ebtehal Mahmoud Ali (Assistant lecturer of Paediatric Oncology at National Cancer Institute – Cairo University).
Dr Mai Mamdouh (Head of Clinical Pharmacy department, Paediatric Oncology, Dar El Salam Cancer Hospital Harmal).
- Abbreviations
CCE (cyclophosphamide-carboplatin-etoposide)
CR (complete remission)
CT (computed tomography)
COG (Children’s Oncology Group)
EFS (event-free survival)
HR (high risk)
ICE (ifosfamide- carboplatin-etoposide)
LR (low risk)
Lymph node (LN)
MRI (magnetic resonance imaging)
NCCN (National Comprehensive Cancer Network)
RCR (rapid complete response)
RT (radiation therapy)
SIR (slow incomplete response)
SR (standard risk)
US (ultrasound)
WLI (whole lung irradiation)
WT (Wilms’ tumor)
- Glossary
Children’s Oncology Group (COG) histologic classification (1)
Favorable histology Wilms’ tumor
- No evidence of anaplasia
Un-favorable histology Wilms’ tumor
a. Focal anaplastic Wilms’ tumor
- Anaplasia confined to one or more discrete sites within the primary tumor with no extrarenal involvement
- No nuclear unrest outside anaplastic foci
b. Diffuse anaplastic Wilms’ tumor
- Non-localized anaplasia
- Anaplasia in invasive sites or extrarenal deposits
- Localized anaplasia with severe nuclear unrest
- Anaplasia in a random biopsy specimen
- Anaplasia involving the edge of one or more sections.
Blastemal predominant histology (post chemotherapy) (2)
▪️ Presence of residual undifferentiated blastemal cells of over 66% in a tumour with more than 33% of cells viable after preoperative chemotherapy.
Risk group definition for patients with favorable histology WT (3)
Low risk:
▪️ Stages I-II.
Standard risk:
▪️ Stage III.
▪️ Stage IV (pulmonary metastasis only) and RCR post week 6.
High risk:
▪️ Stage IV (pulmonary metastasis only) with SIR post week 6.
▪️ Stage IV (extra pulmonary metastasis).
Response criteria for stage IV favorable histology with pulmonary metastasis: (4)
Rapid complete responder (RCR): complete resolution of pulmonary metastases after 6 weeks of pre-nephrectomy chemotherapy with vincristine, dactinomycin, and doxorubicin.
Slow incomplete responder (SIR): incomplete resolution of pulmonary metastases after 6 weeks of pre-nephrectomy chemotherapy with vincristine, dactinomycin, and doxorubicin.
Risk stratification at relapse definitions: (5)
- Standard risk: patients with initial stage I−II low-risk or intermediate-risk tumours, who received only vincristine and/or actinomycin D (no radiotherapy) in their first-line treatment.
- High risk: Patients without initial diffuse anaplasia or blastemal-type histology, who have already received doxorubicin in their initial treatment.
- Very high risk: Patients with recurrent anaplastic or blastemal-predominant tumor.
- Introduction
Wilms’ tumor, also known as nephroblastoma, is the most common primary renal tumor in children. WT accounts for more than 90% of primary renal tumors in patients younger than 20 years and for 5% of all childhood cancers. Most children (75%) present with WT between 1 and 5 years of age, most commonly at 3 years. (6)
Additionally, bilateral tumors or multifocal tumors in a single kidney can occur in approximately 5% to 13% of patients and 10% of patients, respectively, and tend to be more prevalent in individuals with genetic predisposition syndromes.(7) For unilateral tumors, the median age at diagnosis is 35 months for males and 42 months for females, while the median age at diagnosis is 23 months for males and 28.5 months for females for bilateral tumors. (8)
Five-year survival is more than 90% for children with all stages of favorable histology WT who receive appropriate treatment. However, survival remains poor for children with higher stage diffuse anaplastic WT. (9,10)
- Scope and purpose
This guideline was developed aiming to enhance the quality of care for paediatric Wilms’ tumor (WT) patients by establishing a consistent standard of care nationwide. They focus on aiding in the early diagnosis, treatment, and follow-up of Wims tumor to achieve better clinical outcomes.
- Target audience
Clinicians who are involved in the care and treatment of patients with Wilms’ tumor (WT), including paediatric oncologists, surgeons, radiologists, pathologists, and palliative care specialists.
- Methodology
➡️ A comprehensive search for guidelines was undertaken to identify the most
relevant guidelines to consider for adaptation.
➡️ Inclusion/exclusion criteria followed in the search and retrieval of
guidelines to be adapted:
- Selecting only evidence-based guidelines (guideline must include a
report on systematic literature searches and explicit links between
individual Recommendations and their supporting evidence).
- Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or
updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an
organization in order to be valid and comprehensive, a guideline
ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs
to know whether a thorough literature review was conducted and
whether current evidence was used in the preparation of the
Recommendations.
➡️All retrieved Guidelines were screened and appraised using AGREE II
instrument (www.agreetrust.org) by at least two members. the panel decided
a cut-off points or rank the guidelines (any guideline scoring above 50% on
the rigour dimension was retained)
The NCCN guidelines are the main source used while formulating the national guidelines for Burkitt lymphoma (NHL).
➡️ Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading
of Recommendations, Assessment, Development and Evaluation) approach
to assess the quality of a body of evidence, develop and report
Recommendations. GRADE methods are used by WHO because these
represent internationally agreed standards for making transparent
Recommendations. Detailed information on GRADE is available through the
on the following sites:
. GRADE working group:https://www.gradeworkinggroup.org/
. GRADE online training modules: http://cebgrade.mcmaster.ca/
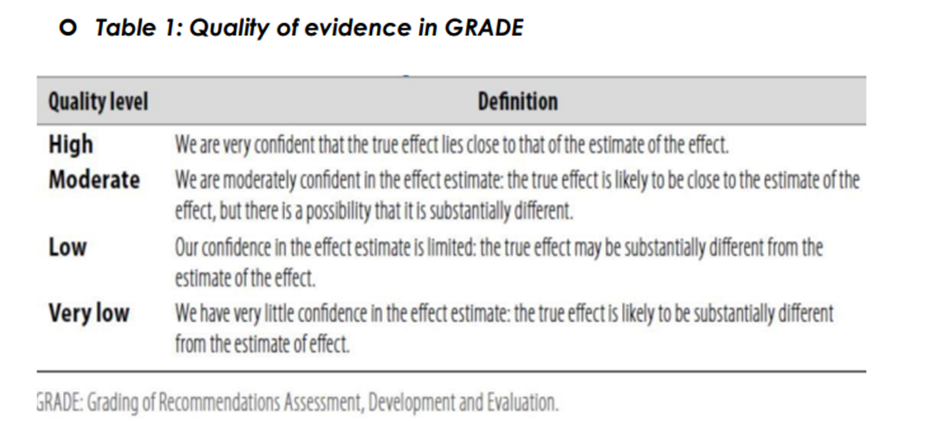
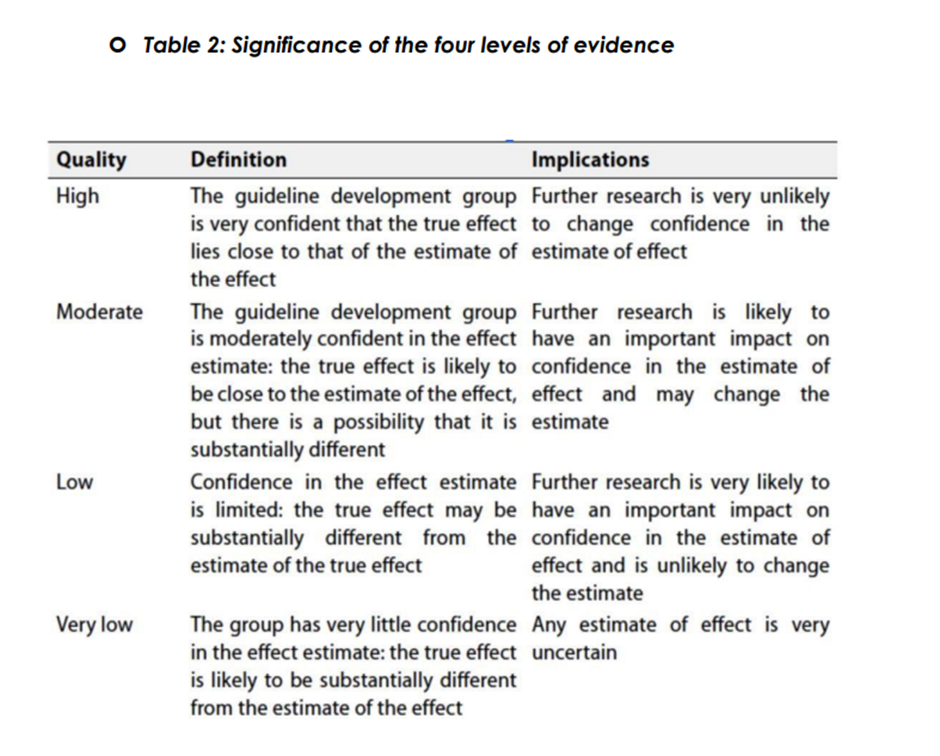
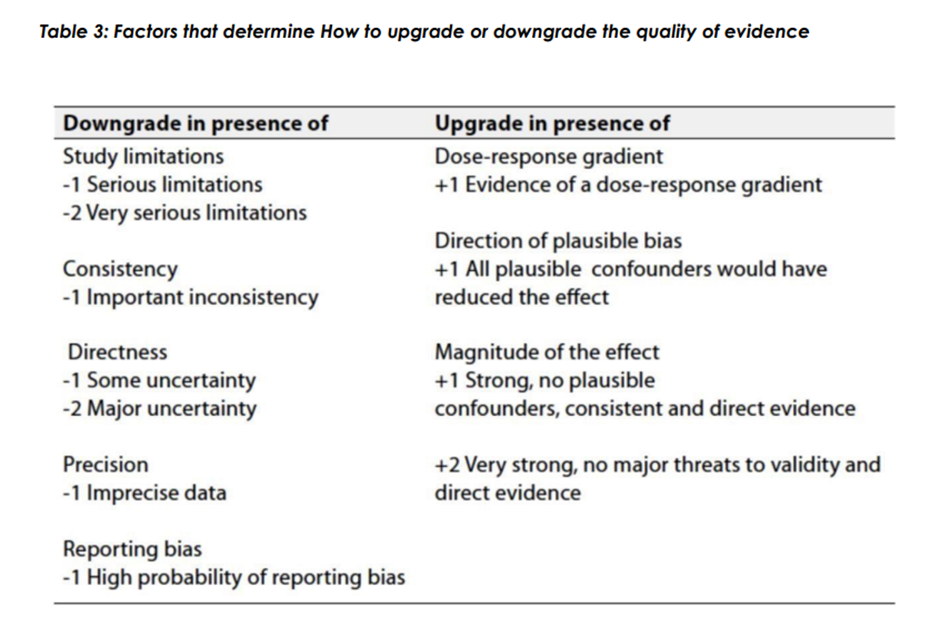
➡️ Table 1: Quality of evidence in GRADE
➡️The strength of Recommendations
The strength of Recommendations communicates the importance of adherence to the Recommendations:
➡️Strong Recommendations
With strong Recommendations, the guideline communicates the message that
the desirable effects of adherence to the Recommendations outweigh the
undesirable effects. This means that in most situations the Recommendations
can be adopted as policy.
➡️Conditional Recommendations
These are made when there is greater uncertainty about the four factors
above or if local adaptation must account for a greater variety in values and
preferences, or when resource use makes the intervention suitable for some,
but not for other locations. This means that there is a need for substantial
debate and involvement of stakeholders before this Recommendations can be
adopted as policy.
➡️When not to make Recommendations.
When there is lack of evidence on the effectiveness of an intervention, it may
be appropriate not to make a Recommendations.
- Annexes
Contraindications to Primary Resection (16)
· High risk of renal failure for those with germline WT1 mutations (Denys-Drash, WAGR).
· Unacceptable anesthesia risk due to:
a. Disease burden
b. Massive pulmonary disease or tumor embolus
c. Very large abdominal tumors causing pulmonary compromise
· Surgeon judgment: Operation would lead to significant morbidity/ mortality, tumor spill, or residual tumor
· Solitary kidney
· IVC tumor thrombus above the level of the hepatic veins is an absolute contraindication; extension of thrombus to the retrohepatic cava is a relative contraindication
· Bilateral tumors or unilateral disease in patients with a predisposing condition
Staging of Wilms’ tumor according to COG staging system (46)
Stage I:
· Tumor limited to kidney, completely resected.
· The renal capsule is intact.
· The tumor was not ruptured or biopsied prior to removal.
· The vessels of the renal sinus are not involved.
· There is no evidence of tumor at or beyond the margins of resection.
Stage II:
· The tumor is completely resected and there is no evidence of tumor at or beyond the margins of resection.
· The tumor extends beyond kidney, as is evidenced by any one of the following criteria:
-There is regional extension of the tumor (i.e. penetration of the renal capsule, or extensive invasion of the soft tissue of the renal sinus, as discussed below).
-Blood vessels within the nephrectomy specimen outside the renal parenchyma, including those of the renal sinus, contain tumor.
Stage III:
· Residual non-hematogenous tumor present following surgery and confined to abdomen. Any one of the following may occur:
· Lymph nodes within the abdomen or pelvis are involved by tumor. (Lymph node involvement in the thorax, or other extraabdominal sites is a criterion for Stage IV.)
· The tumor has penetrated through the peritoneal surface.
· Tumor implants are found on the peritoneal surface. • Gross or microscopic tumor remains postoperatively (eg, tumor cells are found at the margin of surgical resection on microscopic examination).
· The tumor is not completely resectable because of local infiltration into vital structures.
· Tumor spillage occurring either before or during surgery.
· The tumor was biopsied (whether tru-cut, open or fine needle aspiration) before removal.
· Tumor is removed in greater than one piece (eg, tumor cells are found in a separately excised adrenal gland; a tumor thrombus within the renal vein is removed separately from the nephrectomy specimen).
Note: Extension of the primary tumor within vena cava into thoracic vena cava and heart is considered Stage III, rather than Stage IV even though outside the abdomen.
Stage IV:
· Hematogenous metastases (lung, liver, bone, brain, etc), or lymph node metastases outside the abdominopelvic region are present. (The presence of tumor within the adrenal gland is not interpreted as metastasis and staging depends on all other staging parameters present).
Stage V:
· Bilateral renal involvement by tumor is present at diagnosis. An attempt should be made to stage each side according to the above criteria based on the extent of disease.
Predisposing factors to develop bilateral WT: (47)
1. Genetic disorders:
· Beckwith-Wiedemann Syndrome.
· WAGR Syndrome (Wilms tumor, aniridia, genitourinary abnormalities, mental retardation).
· Simpson-Golabi-Behmel-Syndrome.
· Denys-Drash Syndrome.
· Frasier Syndrome.
· Perlman Syndrome.
2. Associated genitourinary anomalies.
3. Contralateral nephrogenic rests in children <12 months.
4. Diffuse hyperplastic perilobar nephroblastomatosis.
- References
1. Green, D. M., D’Angio, G. J., Beckwith, J. B., Breslow, N. E., Grundy, P. E., Ritchey, M. L., & Thomas, P. R. M. (1996). Wilms tumor. CA: A Cancer Journal for Clinicians, 46(1), 46–63. https://doi.org/10.3322/CANJCLIN.46.1.46
2. Vujanić, G. M., Sandstedt, B., Harms, D., Leuschner, I., Kelsey, A., & dE Kraker, J. (2002). Revised International Society of Paediatric Oncology (SIOP) working classification of renal tumors of childhood. Medical and Pediatric Oncology, 38(2), 79–82. https://doi.org/10.1002/MPO.1276
3. Dome, J. S., Perlman, E. J., & Graf, N. (2014). Risk stratification for wilms tumor: current approach and future directions. American Society of Clinical Oncology Educational Book. American Society of Clinical Oncology. Annual Meeting, 34, 215–223. https://doi.org/10.14694/EDBOOK_AM.2014.34.215
4. Verschuur, A., Van Tinteren, H., Graf, N., Bergeron, C., Sandstedt, B., & De Kraker, J. (2012). Treatment of pulmonary metastases in children with stage IV nephroblastoma with risk-based use of pulmonary radiotherapy. Journal of Clinical Oncology : Official Journal of the American Society of Clinical Oncology, 30(28), 3533–3539. https://doi.org/10.1200/JCO.2011.35.8747
5. Van Den Heuvel-Eibrink, M. M., Hol, J. A., Pritchard-Jones, K., Tinteren, H. Van, Furtwängler, R., Verschuur, A. C., Vujanic, G. M., Leuschner, I., Brok, J., Rübe, C., Smets, A. M., Janssens, G. O., Godzinski, J., Ramírez-Villar, G. L., Camargo, B. De, Segers, H., Collini, P., Gessler, M., Bergeron, C., … Graf, N. (2017). Rationale for the treatment of Wilms tumour in the UMBRELLA SIOP–RTSG 2016 protocol. Nature Reviews Urology 2017 14:12, 14(12), 743–752. https://doi.org/10.1038/nrurol.2017.163
6. Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2018, based on November 2020 SEER data submission, posted to the SEER web site, April 2021. Bethesda, MD: National Cancer Institute. Available at: https://seer.cancer.gov/csr/1975_2018/. 2. Breslow N, Olshan A, Beckwith JB, Green DM. Epidemiology of Wilms tumor. Med Pediatr Oncol 1993;21:172-181. Available at: https://www.ncbi.nlm.nih.gov/pubmed/7680412.
7. Charlton J, Irtan S, Bergeron C, Pritchard-Jones K. Bilateral Wilms tumour: a review of clinical and molecular features. Expert Rev Mol Med 2017;19:e8. Available at: https://www.ncbi.nlm.nih.gov/pubmed/28716159.
8. Steliarova-Foucher E, Colombet M, Ries LAG, et al. International incidence of childhood cancer, 2001-10: a population-based registry study. Lancet Oncol 2017;18:719-731. Available at: https://www.ncbi.nlm.nih.gov/pubmed/28410997.
9. Gratias EJ, Dome JS, Jennings LJ, et al. Association of Chromosome 1q Gain With Inferior Survival in Favorable-Histology Wilms Tumor: A Report From the Children's Oncology Group. J Clin Oncol 2016;34:3189- 3194. Available at: https://www.ncbi.nlm.nih.gov/pubmed/27400937.
10. Irtan S, Ehrlich PF, Pritchard-Jones K. Wilms tumor: "State-of-the-art" update, 2016. Semin Pediatr Surg 2016;25:250-256. Available at: https://www.ncbi.nlm.nih.gov/pubmed/27955727.
11. Coppes MJ, Arnold M, Beckwith JB, et al: Factors affecting the risk of contralateral Wilms tumor development: a report from the National Wilms Tumor Study Group. Cancer 85:1616-25, 1999
12. Servaes S, Khanna G, Naranjo A, et al. Comparison of diagnostic performance of CT and MRI for abdominal staging of pediatric renal tumors: a report from the Children's Oncology Group. Pediatr Radiol 2015;45:166-172. Available at: https://www.ncbi.nlm.nih.gov/pubmed/25135711.
13. Grundy, P. E., Green, D. M., Dirks, A. C., Berendt, A. E., Breslow, N. E., Anderson, J. R., & Dome, J. S. (2012). Clinical significance of pulmonary nodules detected by CT and Not CXR in patients treated for favorable histology Wilms tumor on national Wilms tumor studies-4 and -5: A report from the Children’s Oncology Group. Pediatric Blood & Cancer, 59(4), 631–635. https://doi.org/10.1002/PBC.24123
14. Shamberger, R. C., Guthrie, K. A., Ritchey, M. L., Haase, G. M., Takashima, J., Beckwith, J. B., D’Angio, G. J., Green, D. M., & Breslow, N. E. (1999). Surgery-related factors and local recurrence of Wilms tumor in National Wilms Tumor Study 4. Annals of Surgery, 229(2), 292–297. https://doi.org/10.1097/00000658-199902000-00019
15. Gow KW, Roberts IF, Jamieson DH, et al. Local staging of Wilms' tumor--computerized tomography correlation with histological findings. J Pediatr Surg 2000;35:677-679. Available at: https://www.ncbi.nlm.nih.gov/pubmed/10813321.
16. Kieran K, Ehrlich PF. Current surgical standards of care in Wilms tumor. Urol Oncol 2016;34:13-23. Available at: https://www.ncbi.nlm.nih.gov/pubmed/26122713. Kieran K, Anderson JR, Dome JS, et al.: Lymph node involvement in Wilms tumor: results from National Wilms Tumor Studies 4 and 5. J Pediatr Surg 47 (4): 700-6, 2012. [PUBMED Abstract]
17. Vujanić, G. M., Gessler, M., Ooms, A. H. A. G., Collini, P., Coulomb-l’Hermine, A., D’Hooghe, E., de Krijger, R. R., Perotti, D., Pritchard-Jones, K., Vokuhl, C., van den Heuvel-Eibrink, M. M., & Graf, N. (2018). The UMBRELLA SIOP-RTSG 2016 Wilms tumour pathology and molecular biology protocol. Nature Reviews. Urology, 15(11), 693–701. https://doi.org/10.1038/S41585-018-0100-3
18. Jackson, T. J., Brisse, H. J., Pritchard-Jones, K., Nakata, K., Morosi, C., Oue, T., Irtan, S., Vujanic, G., van den Heuvel-Eibrink, M. M., Graf, N., & Chowdhury, T. (2022). How we approach paediatric renal tumour core needle biopsy in the setting of preoperative chemotherapy: A Review from the SIOP Renal Tumour Study Group. Pediatric Blood & Cancer, 69(9). https://doi.org/10.1002/PBC.29702
19. D'Angio GJ, Breslow N, Beckwith JB, et al: Treatment of Wilms' tumor. Results of the Third National Wilms' Tumor Study. Cancer 64:349-60, 1989
20. Dix, D. B., Seibel, N. L., Chi, Y. Y., Khanna, G., Gratias, E., Anderson, J. R., Mullen, E. A., Geller, J. I., Kalapurakal, J. A., Paulino, A. C., Perlman, E. J., Ehrlich, P. F., Malogolowkin, M., Gastier-Foster, J. M., Wagner, E., Grundy, P. E., Fernandez, C. V., & Dome, J. S. (2018). Treatment of stage IV favorable histology wilms tumor with lung metastases: A report from the children’s oncology group AREN0533 study. Journal of Clinical Oncology, 36(16), 1564–1570. https://doi.org/10.1200/JCO.2017.77.1931
21. Dix, D. B., Gratias, E. J., Seibel, N., Anderson, J. R., Mullen, E. A., Geller, J. I., Khanna, G., Kalapurakal, J. A., Perlman, E. J., Ehrlich, P. F., Malogolowkin, M. H., Grundy, P. E., & Dome, J. (2014). Treatment of stage IV favorable histology Wilms tumor with incomplete lung metastasis response after chemotherapy: A report from Children’s Oncology Group study AREN0533. Journal of Clinical Oncology, 32(15_suppl), 10001–10001. https://doi.org/10.1200/JCO.2014.32.15_SUPPL.10001
22. Armstrong AE, Daw NC, Renfro LA, et al. Treatment of focal anaplastic Wilms tumor (FAWT): A report from the Children’s Oncology Group (COG) AREN0321 and AREN03B2 studies. J Clin Oncol 2023;41:10005-10005. Available at: https://ascopubs.org/doi/abs/10.1200/JCO.2023.41.16_suppl.10005.
23. Daw N, Chi YY, Kim Y, et al. Treatment of stage I anaplastic Wilms’tumor: a report from the Children’s Oncology Group AREN0321 study.Eur J Cancer. 2019;118:58-66.12.
24. Dome JS, Cotton CA, Perlman EJ, et al: Treatment of anaplastic histology Wilms’ tumor: Results from the fifth National Wilms’ Tumor Study. J Clin Oncol 24:2352-2358, 2006
25. Graf, N., Tournade, M. F., & De Kraker, J. (2000). THE ROLE OF PREOPERATIVE CHEMOTHERAPY IN THE MANAGEMENT OF WILMS’ TUMOR: The SIOP Studies. Urologic Clinics of North America, 27(3), 443–454. https://doi.org/10.1016/S0094-0143(05)70092-6
26. Rutigliano DN, Kayton ML, Steinherz P, et al.: The use of preoperative chemotherapy in Wilms' tumor with contained retroperitoneal rupture. J Pediatr Surg 42 (9): 1595-9, 2007. [PUBMED Abstract]
27. Tournade MF, Com-Nougue C, de Kraker J, et al. Optimal duration of preoperative therapy in unilateral and nonmetastatic Wilms' tumor in children older than 6 months: results of the Ninth International Society of Pediatric Oncology Wilms' Tumor Trial and Study. J Clin Oncol 2001;19:488-500. Available at: https://www.ncbi.nlm.nih.gov/pubmed/11208843.
28.Van Den Heuvel-Eibrink, M. M., Van Tinteren, H., Bergeron, C., Coulomb-L’Hermine, A., De Camargo, B., Leuschner, I., Sandstedt, B., Acha, T., Godzinski, J., Oldenburger, F., Gooskens, S. L., De Kraker, J., Vujanic, G. M., Pritchard-Jones, K., & Graf, N. (2015). Outcome of localised blastemal-type Wilms tumour patients treated according to intensified treatment in the SIOP WT 2001 protocol, a report of the SIOP Renal Tumour Study Group (SIOP-RTSG). European Journal of Cancer, 51(4), 498–506. https://doi.org/10.1016/J.EJCA.2014.12.011
29. Ehrlich, P., Chi, Y. Y., Chintagumpala, M. M., Hoffer, F. A., Perlman, E. J., Kalapurakal, J. A., Warwick, A., Shamberger, R. C., Khanna, G., Hamilton, T. E., Gow, K. W., Paulino, A. C., Gratias, E. J., Mullen, E. A., Geller, J. I., Grundy, P. E., Fernandez, C. V., Ritchey, M. L., & Dome, J. S. (2017). Results of the First Prospective Multi-institutional Treatment Study in Children With Bilateral Wilms Tumor (AREN0534): A Report From the Children’s Oncology Group. Annals of Surgery, 266(3), 470–478. https://doi.org/10.1097/SLA.0000000000002356
30. Shamberger RC, Haase GM, Argani P, et al. Bilateral Wilms' tumors with progressive or nonresponsive disease. J Pediatr Surg 2006;41:652- 657; discussion 652-657. Available at: https://www.ncbi.nlm.nih.gov/pubmed/16567171.
31. Benedetti DJ, Varela CR, Renfro LA, et al.: Treatment of children with favorable histology Wilms tumor with extrapulmonary metastases: A report from the COG studies AREN0533 and AREN03B2 and NWTSG study NWTS-5. Cancer 130 (6): 947-961, 2024. [PUBMED Abstract]
32. Stokes CL, Stokes WA, Kalapurakal JA, et al. Timing of radiation therapy in pediatric Wilms tumor: a report from the National Cancer Database. Int J Radiat Oncol Biol Phys 2018;101:453-461.
33. Kalapurakal JA, Li SM, Breslow NE, et al; National Wilms' Tumor Study Group. Influence of radiation therapy delay on abdominal tumor recurrence in patients with favorable histology Wilms' tumor treated on NWTS-3 and NWTS-4: a report from the National Wilms' Tumor Study Group. Int J Radiat Oncol Biol Phys 2003;57:495- 499
34. Thomas PR, Tefft M, Compaan PJ, Norkool P, Breslow NE, D’Angio GJ.Results of two radiation therapy randomizations in the third NationalWilms’ Tumor Study. Cancer. 1991;68(8):1703-1707.
35. Daw NC, Chi YY, Kalapurakal JA, et al. Activity of vincristine andirinotecan in diffuse anaplastic Wilms tumor and therapy outcomes of stage II to IV disease: results of the Children’s Oncology GroupAREN0321 Study. J Clin Oncol. 2020;38(14):1558-1568.
36. Ehrlich PF, Chi YY, Chintagumpala MM, et al. Results of treatment forpatients with multicentric or bilaterally predisposed unilateral Wilmstumor (AREN0534): a report from the Children’s Oncology Group.Cancer. 2020;126(15):3516-3525.
37. Shamberger RC, Guthrie KA, Ritchey ML, et al. Surgery related factors and local recurrence of Wilms tumor in National Wilms Tumor Study 4. Ann Surg 2000;229:292– 297.
38. DM Green, CA Cotton, M Malogolowkin, etal: Treatment of Wilms tumor relapsing after initial treatment with vincristine and actinomycin D: A report from the National Wilms Tumor Study Group Pediatr Blood Cancer 48: 493– 499,2007
39. DM Green, CA Cotton, M Malogolowkin, etal: Treatment of Wilms tumor relapsing after initial treatment with vincristine and actinomycin D: A report from the National Wilms Tumor Study Group Pediatr Blood Cancer 48: 493– 499,2007
40. Metzger, M. L., Stewart, C. F., Freeman, B. B., Billups, C. A., Hoffer, F. A., Wu, J., Coppes, M. J., Grant, R., Chintagumpala, M., Mullen, E. A., Alvarado, C., Daw, N. C., & Dome, J. S. (2007). Topotecan is active against Wilms’ tumor: results of a multi-institutional phase II study. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology, 25(21), 3130–3136. https://doi.org/10.1200/JCO.2007.10.9298
41. B Kremens, B Gruhn, T Klingebiel, etal: High-dose chemotherapy with autologous stem cell rescue in children with nephroblastoma Bone Marrow Transplant 30: 893– 898,2002
42. AD Campbell, SL Cohn, M Reynolds, etal: Treatment of relapsed Wilms' tumor with high-dose therapy and autologous hematopoietic stem-cell rescue: The experience at Children's Memorial Hospital J Clin Oncol 22: 2885– 2890,2004
43. F Spreafico, G Bisogno, P Collini, etal: Treatment of high-risk relapsed Wilms tumor with dose-intensive chemotherapy, marrow-ablative chemotherapy, and autologous hematopoietic stem cell support: Experience by the Italian Association of Pediatric Hematology and Oncology Pediatr Blood Cancer 51: 23– 28,2008
44. Brok J, Lopez-Yurda M, Tinteren HV, et al. Relapse of Wilms’ tumour and detection methods: a retrospective analysis of the 2001 Renal Tumour Study Group–International Society of Paediatric Oncology Wilms’ tumour protocol database. Lancet Oncol 2018; 19: 1072–81.
45. Jedrzejewski, G., Wozniak, M. M., Pawelec, A., Matera, A., Kunach, M., Madej, T., Wieczorek, A. P., & Nowakowska, K. (2016). Ultrasound screening for neoplasms in children up to 6 years old. Medicine (United States), 95(42). https://doi.org/10.1097/MD.0000000000005124
46. Beckwith, J. B. (1998). National Wilms Tumor Study: An update for pathologists. Pediatric and Developmental Pathology, 1(1), 79–84. https://doi.org/10.1007/S100249900010
47. Liu, E. K., & Suson, K. D. (2020). Syndromic Wilms tumor: a review of predisposing conditions, surveillance and treatment. Translational Andrology and Urology, 9(5), 2370. https://doi.org/10.21037/TAU.2020.03.27
2) Bhatnagar, S. (2009). Management of Wilms’’ tumor: NWTS vs SIOP. Journal of Indian Association of Pediatric Surgeons, 14(1), 6. https://doi.org/10.4103/0971-9261.54811
3) Gratias EJ, Dome JS, Jennings LJ, et al. Association of chromosome 1q gain with inferior survival in favorable-histology Wilms’ tumor: a report from the Children’s Oncology Group. J Clin Oncol 2016;34:3189-3194.
4) Grundy PE, Breslow NE, Li S, et al. Loss of heterozygosity for chromosomes 1p and 16q is an adverse prognostic factor in favorable-histology Wilms’ tumor: a report from the National Wilms’ Tumor Study Group. J Clin Oncol 2005;23:7312- 7321.
5) Adapted from Wilms’ Tumor and Other Childhood Kidney Tumors Treatment (PDQ®)–Health Professional Version. National Cancer Institute. Accessed February 2, 2023. Available at: https://www.cancer.gov/types/kidney/hp/Wilms’-treatment-pdq.