Cervical cancer
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Cervical cancer |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:47 AM |
Description
"last update: 17 March 2025" Download Guideline
- Executive Summary
Diagnostic and Staging Work up | |
Diagnostic and staging work up should include history and physical examination, complete blood count, as well as liver function and renal function studies. | Good practice statement |
We recommend cervical cytology or Papanicolaou (Pap) smears and cervical biopsies for diagnosis. | Strong recommendation |
We recommend cone biopsy (i.e., conization) if the cervical biopsy is inadequate to define invasiveness or if accurate assessment of microinvasive disease is required. | Strong recommendation |
Recommended radiologic imaging includes pelvic MRI, and FDG-PET/CT. | Strong recommendation |
Consider examination under anesthesia (EUA) cystoscopy/proctoscopy for cases having ≥ stage IB. | Good practice statement |
Consider options for fertility sparing or referral to reproductive endocrinology and infertility. | Conditional recommendation |
Staging and risk assessment. | |
Tumor risk assessment should include tumor size, stage, depth of tumor invasion, lymph node status, LVSI and histological subtype. | Strong recommendation |
Management of local/locoregional disease, | |
Surgery | |
Surgical therapy in cervical cancer should be adapted to the stage of disease according to FIGO and TNM classification (Appendix). | Good practice statement |
Surgery should only be considered in patients with earlier stages of cervical cancer (up to FIGO IIA) without risk factors necessitating adjuvant therapy, which results in a multimodal therapy without improvement of survival but increased toxicity. | Strong recommendation |
Microinvasive cervical cancer (stage IA1) without LVSI should be managed with conisation or simple trachelectomy to preserve fertility, and simple hysterectomy is recommended if the patient does not wish to preserve fertility. | Strong recommendation |
In stage IA1 with LVSI, surgical assessment of pelvic lymph nodes is recommended. | Strong recommendation |
In patients with FIGO stage IA2, IB and IIA, radical hysterectomy with bilateral lymph node dissection (with or without SLN) is standard treatment, if the patient does not wish to preserve fertility. | Strong recommendation |
Adjuvant/neoadjuvant treatment | |
Consider NACT with surgery as this may reduce the need for adjuvant RT. | Conditional recommendation |
Intermediate-risk surgicopathologic findings, frequently referred to as Sedlis criteria, are defined by a combination of lymphovascular space involvement, depth of stromal invasion, and tumor size (Table 5, appendix), and they should be treated by whole pelvic RT delivered to a total dose of 4500 to 5040 cGy, in 180 Gy per fraction or 4000 to 4400 Gy in 200 Gy per fraction. | Strong recommendation |
Adjuvant CRT is recommended in high-risk patients (one or more negative prognostic factors such as positive or close surgical margins, positive lymph nodes or microscopic parametrial involvement). For these patients, whole pelvic RT should be delivered to a total dose of 4500 to 5040 cGy, in 180 cGy fractions, with concurrent weekly cisplatin (40mg/m2). | Strong recommendation |
Chemoradiotherapy in locally advanced cervical cancer | |
We recommend CRT for patients with bulky IB2–IVA disease, and the most commonly used regimen is weekly cisplatin 40 mg/m2. | Strong recommendation |
Patients not eligible to cisplatin may receive carboplatin or gemcitabine. | Strong recommendation |
Brachytherapy is needed to obtain a sufficiently high dose to ensure a high rate of local control in advanced cases. | Good practice statement |
Management of advanced/metastatic disease | |
Palliative chemotherapy with the aim of relieving symptoms and improving quality of life is recommended if the patient has a PS< 2 and no formal contraindications. | Strong recommendation |
Cisplatin-based doublets with paclitaxel or topotecan have demonstrated superiority to cisplatin monotherapy in terms of response rate and PFS. | Strong recommendation |
Paclitaxel and cisplatin combined with bevacizumab is recommended as the preferred first-line regimen in metastatic or recurrent cervical cancer based on the balance between efficacy and toxicity profile. | Strong recommendation |
The combination of paclitaxel and carboplatin is recommended as an alternative for patients that are not candidates for cisplatin. | Strong recommendation |
Some patients develop small lung metastases only, which do not rapidly progress and can be managed with stereotactic RT and/or a watchful waiting policy, frequently delaying systemic chemotherapy for a significant period of time. | Conditional recommendation |
Local recurrence of cervical cancer following radical surgery | |
Higher doses of RT can be delivered with brachytherapy and increase the likelihood of local control for patients with small volume central recurrences. | Strong recommendation |
Clinical indicators | |
Follow-up visits with a complete physical examination including a pelvic–rectal exam and a patient history should be conducted by a physician experienced in the surveillance of cancer patients. | Good practice statement |
CT or PET/CT scan should be carried out as clinically indicated. A reasonable follow-up schedule involves follow-up visits every 3–6 months in the first 2 years and every 6–12 months in years 3–5. | Good practice statement |
Patients should return to annual population-based general physical and pelvic examinations after 5 years of recurrence-free follow-up. | Good practice statement |
- Recommendations
➡️Diagnostic and Staging Work up
· Diagnostic and staging work up should include history and physical examination, complete blood count, as well as liver function and renal function studies.
· We recommend cervical cytology or Papanicolaou (Pap) smears and cervical biopsies for diagnosis.
➡️Strong recommendation, high grade evidence, (4).
· We recommend cone biopsy (i.e., conization) if the cervical biopsy is inadequate to define invasiveness or if accurate assessment of microinvasive disease is required.
➡️Strong recommendation, high grade evidence, (5).
· Recommended radiologic imaging includes pelvic MRI, and FDG-PET/CT
➡️Strong recommendation, high grade evidence, (6,7).
· Consider examination under anesthesia (EUA) cystoscopy/proctoscopy for cases having ≥ stage IB.
➡️Good practice statement
· Consider options for fertility sparing
➡️ Conditional recommendation, high grade evidence, (8).
➡️Staging and risk assessment
· Tumor risk assessment should include tumor size, stage, depth of tumor invasion, lymph node status, LVSI and histological subtype.
➡️Strong recommendation, high grade evidence, (Table 4, appendix).
➡️Management of local/locoregional disease
➡️Surgery
· Surgical therapy in cervical cancer should be adapted to the stage of disease according to FIGO and TNM classification (Appendix).
➡️Good practice statement
· Surgery should only be considered in patients with earlier stages of cervical cancer (up to FIGO IIA) without risk factors necessitating adjuvant therapy, which results in a multimodal therapy without improvement of survival but increased toxicity.
➡️Strong recommendation, high grade evidence (9).
· Microinvasive cervical cancer (stage IA1) without LVSI should be managed with conisation or simple trachelectomy to preserve fertility, and simple hysterectomy is recommended if the patient does not wish to preserve fertility.
➡️Strong recommendations, high grade evidence (8).
· In stage IA1 with LVSI, surgical assessment of pelvic lymph nodes is recommende
➡️Strong recommendation, moderate grade evidence (10).
· In patients with FIGO stage IA2, IB and IIA, radical hysterectomy with bilateral lymph node dissection (with or without SLN) is standard treatment, if the patient does not wish to preserve fertility.
➡️ Strong recommendation, high grade evidence, (9,11).
Adjuvant/neoadjuvant treatment
· Consider NACT with surgery as this may reduce the need for adjuvant RT.
➡️Conditional recommendation, high grade evidence, (12,13).
· Intermediate-risk surgicopathologic findings, frequently referred to as Sedlis criteria, are defined by a combination of lymphovascular space involvement, depth of stromal invasion, and tumor size (Table 5, appendix), and they should be treated by whole pelvic RT delivered to a total dose of 4500 to 5040 cGy, in 180 Gy per fraction or 4000 to 4400 Gy in 200 Gy per fraction.
➡️Strong recommendation, strong grade evidence, (14-16).
· Adjuvant CRT is recommended in high-risk patients (one or more negative prognostic factors such as positive or close surgical margins, positive lymph nodes or microscopic parametrial involvement). For these patients, whole pelvic RT should be delivered to a total dose of 4500 to 5040 cGy, in 180 cGy fractions, with concurrent weekly cisplatin (40mg/m2).
➡️ Strong recommendation, high grade evidence, (14,17).
Chemoradiotherapy in locally advanced cervical cancer
· We recommend CRT for patients with bulky IB2–IVA disease, and the most commonly used regimen is weekly cisplatin 40 mg/m2;
➡️ Strong recommendation, high grade evidence, (18-23).
· Patients not eligible to cisplatin may receive carboplatin or gemcitabine.
➡️Strong recommendation, very low grade evidence (24,25).
· Brachytherapy is needed to obtain a sufficiently high dose to ensure a high rate of local control in advanced cases.
➡️Good practice statement
Management of advanced/metastatic disease
· Palliative chemotherapy with the aim of relieving symptoms and improving quality of life is recommended if the patient has a PS< 2 and no formal contraindications.
➡️Strong recommendation, high grade evidence, (26,27).
· Cisplatin-based doublets with paclitaxel or topotecan have demonstrated superiority to cisplatin monotherapy and should be used.
➡️Strong recommendation, high grade evidence (26,27).
· Paclitaxel and cisplatin combined with bevacizumab is recommended as the preferred first-line regimen in metastatic or recurrent cervical cancer based on the balance between efficacy and toxicity profile.
➡️ Strong recommendation, high grade evidence, (28-30).
· The combination of paclitaxel and carboplatin is recommended as an alternative for patients that are not candidates for cisplatin.
➡️Strong recommendation, moderate grade evidence, (31).
➡️Strong recommendation, moderate grade evidence, (32).
· Some patients develop small lung metastases only, which do not rapidly progress and can be managed with stereotactic RT and/or a watchful waiting policy, frequently delaying systemic chemotherapy for a significant period of time.
➡️Conditional recommendation, low grade evidence, (33).
Local recurrence of cervical cancer following radical surger
· Higher doses of RT can be delivered with brachytherapy and increase the likelihood of local control for patients with small volume central recurrences.
➡️Strong recommendation, high grade evidence (34).
➡️Clinical indicators
· Follow-up visits with a complete physical examination including a pelvic–rectal exam and a patient history should be conducted by a physician experienced in the surveillance of cancer patients.
➡️Good practice statement
· CT or PET/CT scan should be carried out as clinically indicated. A reasonable follow-up schedule involves follow-up visits every 3–6 months in the first 2 years and every 6–12 months in years 3–5.
➡️Good practice statement
· Patients should return to annual population-based general physical and pelvic examinations after 5 years of recurrence-free follow-up.
➡️Good practice statement
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines, for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines: Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Yousri Wasef & Yousri Rostom.
- Abbreviations
|
ASIR |
Age standardize incidence rate |
|
ASMR |
Age standardize mortality rate |
|
CRT |
Concurrent chemo radiotherapy |
|
EUA |
Examination under anesthesia |
|
FDG-PET/CT |
18-Fluoro-deoxyglucose positron emission tomography |
|
LVSI |
Lympho vascular invasion |
|
MRI |
Magnetic resonance imaging |
|
NACT |
Neoadjuvant chemotherapy |
|
Pap |
Papanicolaou |
|
PS |
Performance status |
|
RT |
Radiotherapy |
|
SLND |
Sentinel lymph node dissection |
- Introduction
In Egypt, there was an estimated 1302 new cases of cervical cancer with an ASIR of 2.8/100 000 normal population and ranks the 13th most common cancer among females. There were 820 (ASMR: 1.8/100 000 normal population) deaths occurred because of this disease based on GLOBOCAN 2022.
- Purpose and scope
These guidelines are developed to improve the quality of care for cervical cancer via providing a uniform standard of care across the country to help in early diagnosis, treatment and follow up for cervical cancer so more optimal treatment options and improved clinical outcomes.
- Target audience
Clinicians who are involved in the care and treatment of patients with cervical cancer, include medical oncologists, radiation oncologists, clinical oncologist, gynecologists, surgeons, radiologists, pathologists, and palliative care specialists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The NCCN, ESMO, NICE guidelines are the main sources used while formulating the national guidelines for cervical cancer.
➡️ Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group: https://www.gradeworkinggroup.org/
. GRADE online training modules:http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
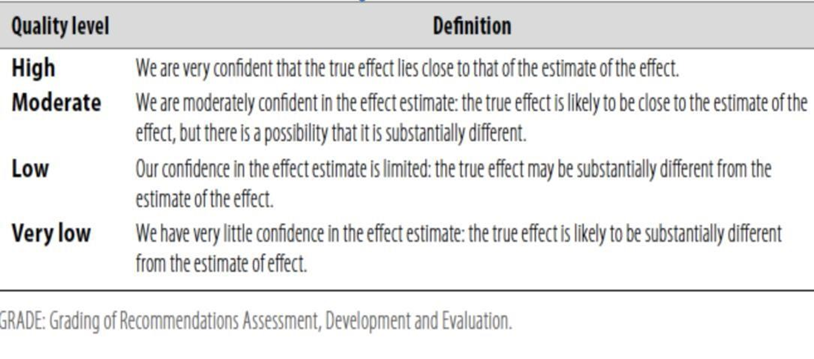
Table 2: Significance of the four levels of evidence

Table 3: Factors that determine How to upgrade or downgrade the quality of evidence
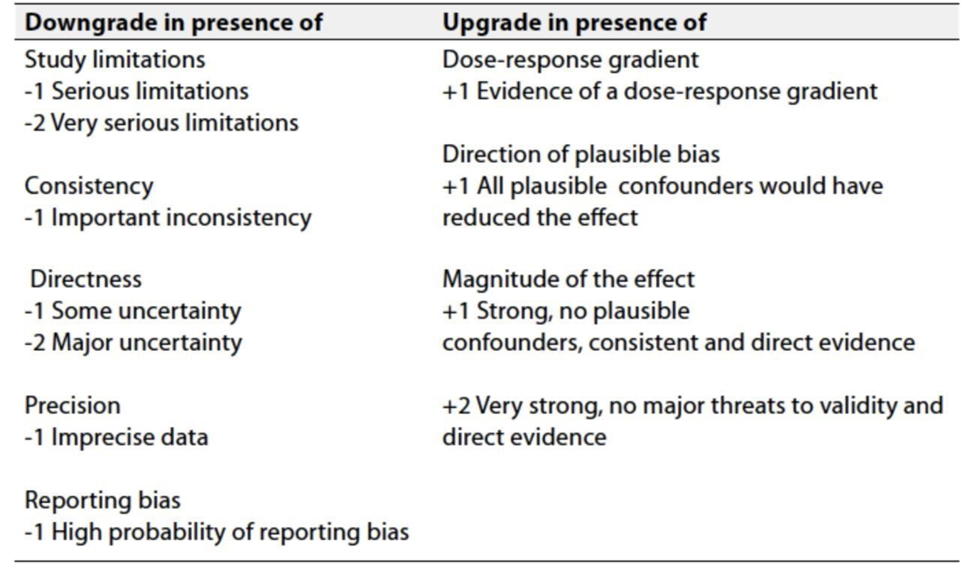
➡️ The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
➡️Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
➡️Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Update of this guideline
This guideline will be updated whenever there is new evidence.
- Research gaps
▪️ Systematic inclusion of cost-benefit analyses in clinical trials with collection of health economic analysis such as incremental cost effectiveness ratio in order to facilitate clinical decision-making.
▪️ Predictive biomarkers: response to specific systemic targeted therapies and immunotherapy.
▪️ Improve models for pre-clinical testing of novel drugs.
▪️ Search for tools to assess quality of life and in clinical trials.
▪️ Dietary supplements, nutritional counselling, physical activity recommendations and psychological support as part of an integrative healthcare approach to care for people with ovarian cancer.
- References
1. https://www.nccn.org/guidelines/category_1
3. https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/nice-guidelines
4. Ronco G, Dillner J, Elfstro¨m KM et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: follow-up of four European randomised controlled trials. Lancet 2014; 383: 524–532.
5. Yoneda JY, Braganca JF, Sarian LO et al. Surgical treatment of microinvasive cervical cancer: analysis of pathologic features with implications on radicality. Int J Gynecol Cancer 2015; 25: 694–698.
6. Wagenaar HC, Trimos JB, Postema S et al. Tumor diameter and volume assessed by magnetic resonance imaging in the prediction of outcome for invasive cervical cancer. Gynecol Oncol 2001; 82: 474–482.
7. Patel CN, Nazir SA, Khan Z et al. 18F-FDG PET/CT of cervical carcin- oma. AJR Am J Roentgenol 2011; 196: 1225–1233.
8. Dittrich R, Lotz L, Hackl J et al. Fertilit€atserhalt bei Krebserkrankungen. Frauenarzt 2014; 55: 240–246.
9. Plante M, Kwon JS, Ferguson S, Samouëlian V, Ferron G, Maulard A, de Kroon C, Van Driel W, Tidy J, Williamson K, Mahner S, Kommoss S, Goffin F, Tamussino K, Eyjólfsdóttir B, Kim JW, Gleeson N, Brotto L, Tu D, Shepherd LE; CX.5 SHAPE investigators; CX.5 SHAPE Investigators, Simple versus Radical Hysterectomy in Women with Low-Risk Cervical Cancer, N Engl J Med. 2024 Feb 29;390(9):819-829
10. Diab Y. Sentinel lymph nodes mapping in cervical cancer a comprehensive review. Int J Gynecol Cancer 2017; 27: 154–158.
11. Kim SM, Choi HS, Byun JS. Overall 5-year survival rate and prognostic factors in patients with stage IB and IIA cervical cancer treated by radical hysterectomy and pelvic lymph node dissection. Int J Gynecol Cancer 2000; 10: 305–312.
12. Neoadjuvant Chemotherapy for Locally Advanced Cervical Cancer Meta-analysis Collaboration. Neoadjuvant chemotherapy for locally advanced cervical cancer: a systematic review and meta-analysis of individual patient data from 21 randomised trials. Eur J Cancer 2003; 39: 2470–2486.
13. Rydzewska L, Tierney J, Vale CL, Symonds PR. Neoadjuvant chemotherapy plus surgery versus surgery for cervical cancer. Cochrane Database Syst Rev 2012; 12: CD007406.
14. CORRIGENDUM, Cervical cancer: ESMO Clinical Practice Guidelines for C. Marth, F. Landoni, S. Mahner, M. McCormack, A. Gonzalez-Martin & N. Colombo, on behalf of the ESMO Guidelines Committee, diagnosis, treatment and follow-up, Annals of Oncology 29 (Supplement 4): iv262, 2018.
15. Sedlis A, Bundy BN, Rotman MZ, Lentz SS, Muderspach LI, Zaino RJ. A randomized trial of pelvic radiation therapy versus no further therapy in selected patients with stage IB carcinoma of the cervix after radical hysterectomy and pelvic lymphadenectomy: A Gynecologic Oncology Group Study. Gynecol Oncol. 1999;73:177-183.
16. Rotman M, Sedlis A, Piedmonte MR, et al. A phase Ill randomized trial of postoperative pelvic irradiation in Stage IB cervical carcinoma with poor prognostic features: follow-up of a gynecologic oncology group study. Int J Radiat Oncol Biol Phys. 2006;65:169-176.
17. Peters W.A., III, Liu P.Y., Barrett R.J., II, Stock R.J., Monk B.J., Berek J.S., Souhami L., Grigsby P., Gordon W., Jr., Alberts D.S. Concurrent Chemotherapy and Pelvic Radiation Therapy Compared with Pelvic Radiation Therapy Alone as Adjuvant Therapy After Radical Surgery in High-Risk Early-Stage Cancer of the Cervix. Clin Oncol. 2000;18:1606–1613.
18. Whitney CW, Sause W, Bundy BN et al. Randomized comparison of fluorouracil plus
cisplatin versus hydroxyurea as an adjunct to radiation therapy in stage IIB–IVA carcinoma of the cervix with negative paraaortic lymph nodes: a Gynecologic Oncology Group and Southwest Oncology Group study. J Clin Oncol 1999; 17: 1339–1348.
19. Rose PG, Bundy BN, Watkins EB et al. Concurrent cisplatin-based radiotherapy and
chemotherapy for locally advanced cervical cancer. N Engl J Med 1999; 340: 1144–1153.
20. Morris M, Eifel PJ, Lu J et al. Pelvic radiation with concurrent chemotherapy compared with
pelvic and para-aortic radiation for high-risk cervical cancer. N Engl J Med 1999; 340: 1137–
1143.
21.Keys HM, Bundy BN, Stehman FB et al. Cisplatin, radiation, and adjuvant hysterectomy
compared with radiation and adjuvant hysterectomy for bulky stage IB cervical carcinoma. N
Engl J Med 1999; 340: 1154–1161.
22.Peters WA, 3rd, Liu PY, Barrett RJ, 2nd et al. Concurrent chemotherapy and pelvic radiation
therapy compared with pelvic radiation therapy alone as adjuvant therapy after radical surgery
in high-risk early-stage cancer of the cervix. J Clin Oncol 2000; 18: 1606–1613.
23.Chemoradiotherapy for Cervical Cancer Meta-Analysis Collaboration. Reducing uncertainties
about the effects of chemoradiotherapy for cervical cancer: a systematic review and meta-
analysis of individual patient data from 18 randomized trials. J Clin Oncol 2008; 26: 5802–
5812.
24. Eun Ji Nam, Maria Lee, Ga Won Yim, Jae Hoon Kim, Sunghoon Kim, Sang Wun Kim, Jae
Wook Kim, Young Tae Kim, Comparison of Carboplatin‐ and Cisplatin‐Based Concurrent
Chemoradiotherapy in Locally Advanced Cervical Cancer Patients With Morbidity Risks
The Oncologist, Volume 18, Issue 7, July 2013, Pages 843–849,
25. Cetina, Lucely Rivera, Lesbia, Candelaria, Myrna de la Garza, Jaim, Dueñas-González,
Alfonso, Chemoradiation with gemcitabine for cervical cancer in patients with renal failure
Anti-Cancer Drugs 15(8):p 761-766, September 2004.
26.Moore DH, Blessing JA, McQuellon RP et al. Phase III study of cisplatin with or without
paclitaxel in stage IVB, recurrent, or persistent squamous cell carcinoma of the cervix: a
Gynecologic Oncology Group Study.J Clin Oncol 2004; 22: 3113–3119.
27.Long HJ 3rd, Bundy BN, Grendys EC Jr et al. Randomized phase III trial of cisplatin with or
without topotecan in carcinoma of the uterine cervix: a Gynecologic Oncology Group Study. J
Clin Oncol 2005; 23:4626–4633.
28.Monk BJ, Sill MW, Burger RA et al. Phase II trial of bevacizumab in the treatment of
persistent or recurrent squamous cell carcinoma of the cervix: a Gynecologic Oncology Group
study. J Clin Oncol 2009; 27: 1069–1074.
29.Tewari KS, Sill MW, Long HJ 3rd et al. Improved survival with bevacizumab in advanced
cervical cancer. N Engl J Med 2014; 370: 734–743.
30.Kitagawa R, Katsumata N, Shibata T et al. Paclitaxel plus carboplatin versus paclitaxel plus
cisplatin in metastatic or recurrent cervical cancer: the openlabel randomized phase III trial
JCOG0505. J Clin Oncol 2015; 33: 2129–2135
31. Andres Redondo, Nicoletta Colombo, Mary McCormack, Lydia Dreosti, et al., Primary results
from CECILIA, a global single-arm phase II study evaluating bevacizumab, carboplatin and
paclitaxel for advanced cervical cancer, Gynecol Oncol., 2020 Oct;159(1):142-149.
32. https://interactiveguidelines.esmo.org/esmo-web-
app/toc/index.php?subjectAreaID=7&loadPdf=1
33.Tomoki Kimura, Toshiki Fujiwara, Tsubasa Kameoka, Yoshinori Adachi, Shinji Kariya,
Stereotactic body radiation therapy for metastatic lung metastases, Jpn J Radiol. 2022 Sep
13;40(10):995–1005.
34.Friedlander M, Grogan M; U.S. Preventative Services Task Force. Guidelines for the treatment
of recurrent and metastatic cervical cancer.Oncologist 2002; 7: 342–347.
- Annexes

Table 5. Intermediate Risk-factors for Cervical Cancer (2).
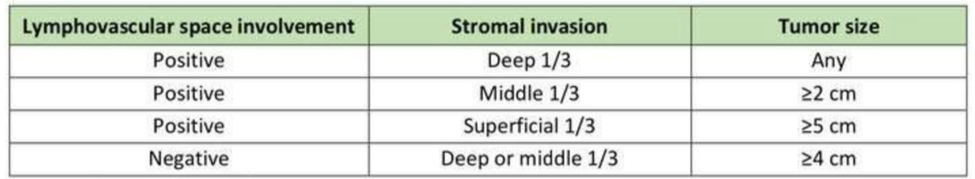
|
The staging of cervical tumours is by the Fe´deration Internationale de Gyne´cologie et d’Obste´trique (FIGO) and TNM classification (Union for International Cancer Control), (2). |
||
|
TNM clinical classification |
||
|
TNM categories |
FIGO stages |
Definition |
|
T – Primary Tumour |
|
|
|
TX |
|
Primary tumour cannot be assessed |
|
T0 |
|
No evidence of primary tumour |
|
Tis |
|
Carcinoma in situ (preinvasive carcinoma) |
|
T1 |
I |
Tumour confined to the cervix |
|
T1a |
IA |
Invasive carcinoma diagnosed only by microscopy. Stromal invasion with a maximal depth of |
|
|
|
5.0 mm measured from the base of the epithelium and a horizontal spread of 7.0 mm or less |
|
IA1 |
Measured stromal invasion 3.0 mm or less in depth and 7.0 mm or less in horizontal spread |
|
|
IA2 |
Measured stromal invasion more than 3.0 mm and not more than 5.0 mm with a horizontal |
|
|
|
|
spread of 7.0 mm or less |
|
T1b |
IB |
Clinically visible lesion confined to the cervix or microscopic lesion greater than T1a/IA2 |
|
IB1 |
Clinically visible lesion 4.0 cm or less in greatest dimension |
|
|
IB2 |
Clinically visible lesion more than 4.0 cm in greatest dimension |
|
|
II |
Tumour invades beyond uterus but not to pelvic wall or to lower third of vagina |
|
|
IIA |
Tumour without parametrial invasion |
|
|
T2a1 |
IIA1 |
Clinically visible lesion 4.0 cm or less in greatest dimension |
|
IIA2 |
Clinically visible lesion more than 4.0 cm in greatest dimension |
|
|
T2b |
IIB |
Tumour with parametrial invasion |
|
T3 |
III |
Tumour involves lower third of vagina, or extends to pelvic wall, or causes hydronephrosis or |
|
|
|
non-functioning kidney |
|
T3a |
IIIA |
Tumour involves lower third of vagina |
|
T3b |
IIIB |
Tumour extends to pelvic wall, or causes hydronephrosis or non-functioning kidney |
|
T4 |
IVA |
Tumour invades mucosa of the bladder or rectum, or extends beyond true pelvise |
|
N – Regional Lymph Nodes |
|
|
|
NX |
|
Regional lymph nodes cannot be assessed |
|
N0 |
|
No regional lymph node metastasis |
|
N1 |
|
Regional lymph node metastasis |
|
M – Distant Metastasis |
|
|
|
M0 |
|
No distant metastasis |
|
M1 |
|
Distant metastasis (includes inguinal lymph nodes and intraperitoneal disease). It excludes |
|
|
|
metastasis to vagina, pelvic serosa, and adnexa |
|
James D, Brierley JD, Gospodarowicz MK et al. (eds). TNM Classification of Malignant Tumours, 8th edition. Oxford, UK: John Wiley & Sons, Inc. 2016.
|
||