Bronchial Asthma Management in Children
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Bronchial Asthma Management in Children |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 18 March 2025" Download Guideline
- Executive Summary
➡️Introduction
Asthma is a chronic inflammatory disease of the airways, characterized by recurrent episodes of wheeze, chest tightness, cough, and shortness of breath and airflow obstruction resulting from edema, bronchospasm, and increased mucus production. Commonly associated with seasonal allergies (allergic rhinitis) and eczema (atopic dermatitis), these three conditions form what is known as the atopic triad. There is a wide range in the frequency and severity of the symptoms, but uncontrolled asthma and acute exacerbations can lead to respiratory failure and death. Around 14% of children worldwide have a diagnosis of asthma, making it the most common chronic respiratory disease of childhood.
The prevalence of asthma among Egyptian children aged 3 - 15 years was estimated to be 8.2%. Poor asthma control is associated with a number of negative effects on children and families. For example, they are more likely to be absent from school, Caregivers also experience missed work days. Some children will experience severe symptoms and life-threatening attacks. Asthma is a multigenetic disease, where both genetic and environmental factors have significant roles in pathogenesis. Effective asthma management involves a holistic approach addressing both pharmacological and non-pharmacological management, as well as education and self-management aspects. Working in partnership with children and families is a key in promoting good outcomes. Education on how to take treatment effectively, trigger avoidance, modifiable risk factors and actions to take during acute attacks via personalized asthma action plans is essential.
➡️Scope
This guideline focuses on diagnosis, treatment and prevention of bronchial asthma in asthmatic children: preschoolers (5 years and younger), children (6 years and older) after treatment of acute asthma exacerbation.
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria the CPGs development group and methodologists recommended using the following guidelines
- SIGN/BTS 158: British guideline on the management of asthma, 2019
- Japanese pediatric guideline for the treatment and management of asthma (JPGL), 2020
- Canadian Thoracic Society 2021 Guideline update: Diagnosis and management of asthma in preschoolers, children, and adults
- European Respiratory Society clinical practice guidelines for the diagnosis of asthma in children aged 5–16 years, 2021
- GINA Report 2024
➡️Recommendations and good practice statements
This version of the guidelines includes recommendations and good practice statements on the following subsections:
▪️ Diagnosis and management of asthma in children 5 years and younger
▪️ Diagnosis and Management of asthma in children 6 years and older
▪️ Prevention of Bronchial asthma
▪️ Difficult to treat asthma
▪️ Asthma resistant to therapy
▪️ Immunotherapy in asthma
We can summarize the guidelines for management of bronchial asthma in the following:
▪️ Clinical assessment to assess the initial probability of asthma is based on proper history taking with observation of clinical improvement in symptoms with inhaled beta-2 agonist, there is no role for x-ray except to exclude other diagnoses
▪️ Lung function testing, do not have a major role in the diagnosis of asthma in children less than 5 years while in children 6 years and older the diagnosis of asthma is based on the history of characteristic symptom patterns and evidence of variable expiratory airflow limitation and reversibility by spirometry or peak expiratory flow if available rather than relying on symptoms alone
▪️ For children aged 5 years and younger the best practice for the initial asthma management based on asthma severity is regular ICS or LTRA as controller treatment
▪️For children aged 6 years and older the best practice for the initial asthma management based on asthma severity is regular ICS, LTRA or ICS-LABA as controller treatment
▪️ Children should be reviewed after initial treatment every 2-3 months to review response and adjust treatment.
▪️ All children with asthma (and/or their parents or caregivers) should be offered self-management education, which should include a written personalized asthma action plan and be supported by regular professional review
▪️The child should be referred for expert assessment if symptom control remains poor and/or flare-ups persist, or if side-effects of treatment are observed or suspected
▪️ For prevention of asthma, breastfeeding should be encouraged for its many benefits, including a potential protective effect in relation to early asthma. Patients with asthma and parents/caregivers of children with asthma should be advised about the dangers of smoking and second-hand tobacco smoke exposure on their children. Also patients with moderate to severe asthma are advised to receive an influenza vaccination every year
▪️ For difficult to treat asthma patients should be systematically evaluated to confirm the diagnosis, check adherence to maintenance therapy, review history and examination for comorbidities and to identify modifiable risk factors and triggers
▪️ Severe asthma is a subset of difficult-to-treat asthma. In children 6 years and older it means asthma that is uncontrolled despite adherence with maximal optimized high-dose ICS-LABA treatment and management of contributory factors, or that worsens when high-dose treatment is decreased. Further assessment and management should be done by a specialist, additional investigations and assessment of asthma phenotype may be required
Allergen immunotherapy may be considered as add-on-therapy for asthmatic children who have clinically significant sensitization to aeroallergens including those with allergic rhinitis.- Recommendations
Table 3. Recommendations |
|
| |||
A. Diagnosis and management of asthma in children 5 years and younger Q1. For children 5years&younger What is the best practice in the diagnostic criteria of asthma? |
|
| |||
N | Health questions | Source Guideline | Recommendations | Quality of evidence | Strength of Recommendation |
1.a | - Is there clinical assessment for probability of asthma in children 5 years and younger |
Canadian thoracic society 2021
Japanese 2020
| Clinical assessment to assess for the initial probability of asthma is based on: • a history of recurrent episodes (3 attacks) of wheeze, cough, breathlessness and chest tightness that vary over time • recorded observation of wheeze heard by a healthcare professional • personal/family history of other atopic conditions (in particular, atopic eczema/dermatitis, allergic rhinitis) • no symptoms/signs to suggest alternative diagnoses.
Symptom improvement can be observed after beta-2 agonist inhalation. | intermediate
intermediate
| Strong
strong
(GPS)
|
1.b | Is assessing clinical improvement after using regular low-dose inhaled corticosteroids (ICS) and as-needed SABA within 2–3 months’ support diagnosis of asthma in children 5 years and younger?
| GINA 2024 | A trial of treatment for at least 2–3 months with as-needed SABA and regular low-dose ICS may provide some guidance about the diagnosis of asthma. Marked clinical improvement during treatment, and deterioration when treatment is stopped, support a diagnosis of asthma. | Very low | Weak (conditional) |
1.c | Is chest X-ray indicated for diagnosis of asthma? | GINA 2024 | Chest X-ray Radiographs are rarely indicated; except, if there is doubt about the diagnosis of asthma help to exclude other diagnoses. |
| GPS |
Q2. For children aged 5 years and younger: What is the best practice for assessing asthma severity? | |||||
2 | How to assess pulmonary function in children 5 years and younger? | GINA 2024 | Due to the inability of most children 5 years and younger to perform reproducible expiratory maneuvers, lung function testing, do not have a major role in the diagnosis of asthma at this age. However, by 5 years of age, many children are capable of performing reproducible spirometry if coached by an experienced technician and with visual incentives |
| GPS |
| Q3- For children aged 5 years and younger: What is the best practice for the initial asthma management based on asthma severity? | ||||
3.a | Which children should be initiated on regular low dose ICS as controller treatment? | GINA 2024 | If symptoms pattern is consistent with asthma, and asthma symptoms are not well controlled, or > 3 exacerbations per year.
| Very low | Weak (conditional) |
|
| GINA 2024 | If symptoms pattern is not consistent with asthma but wheezing episodes requiring SABA occur frequently e.g > 3 per year. Give diagnostic trial for 3 months | Very low | Weak
|
3.b | When to use double low dose ICS as controller therapy in children 5 years and younger | GINA 2024 | Doubling the initial low dose of ICS may be the best option if asthma is not well controlled on low dose ICS after checking inhaler technique and adherence | Low | Weak (conditional) |
3.c | What is the role of LTRA in asthma management? | Japanese 2020 | LTRA can be added to ICS for children whose asthma is not controlled well with ICS alone. | low | Weak (conditional) |
3.d | Can we use LABA in the management of asthma in children 5 years and younger? | GINA 2024 | There are insufficient data about the efficacy and safety of ICS in combination with a long-acting beta2 agonist (LABA) in children <4 years old to recommend their use. |
| GPS |
3.e | How often should a child be reviewed after initial treatment to review response and adjust treatment? | GINA 2024 | -Assessment at every visit (every 2-3 months) should include asthma symptom control and risk factors and side-effects. -If therapy is stepped-down or discontinued, schedule a follow-up visit 3–6 weeks later to check whether symptoms have recurred, as therapy may need to be stepped-up or reinstituted
The child’s height should be measured every year, or more often.
| Very low
Very low | Weak (conditional)
Weak (conditional) |
| For children aged 5 years and younger: when to refer to asthma specialist
| GINA 2024 | The child should be referred for expert assessment if symptom control remains poor and/or flare-ups persist, or if side-effects of treatment are observed or suspected | low | Weak
|
3.f | 4- Is self-management education, including a written personalized asthma action plan should be offered to parents of children 5 years and younger?
| British 2019 | All people with asthma (and/or their parents or carers) should be offered self-management education, which should include a written personalized asthma action plan and be supported by regular professional review. | High | Strong |
|
Table 4. Recommendations |
|
|
|||
|
B. Diagnosis and Management of asthma in children 6 years and older |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
Q4 |
How is asthma diagnosed? |
GINA 2024
ERS 2021 |
The diagnosis of asthma is based on the history of characteristic symptom patterns and evidence of variable expiratory airflow limitation and reversibility by spirometry (appendix) or peak expiratory flow if available rather than relying on symptoms alone.
The task force recommends spirometry as part of the diagnostic work-up of children aged 5–16 years with suspected asthma |
Moderate |
GPS
Strong |
|
Q5 |
What are the characteristic symptom patterns of asthma? |
GINA 2024 |
Wheeze, shortness of breath, cough and/or chest tightness: • Often worse at night or in the early morning. • Vary over time and in intensity. • Triggered by viral infections, exercise, allergen exposure, changes in weather, laughter, or irritants such as car exhaust fumes, smoke or strong smells. |
|
GPS |
|
|
Stepwise approach for management of asthma for children 6 years and older |
||||
|
Q6.a |
In children 6 years and older, what is the criteria to start step 1 to control the symptoms and minimize future risk?
|
GINA 2024 |
Start with step 1 in children with: Symptoms <twice / week As-needed low-dose ICS |
High |
Strong |
|
Q6.b |
What is the preferred controller in step 1? |
GINA 2024 |
Low dose ICS taken whenever SABA taken |
Intermediate |
Strong |
|
Q7.a |
In children 6 years and older, what is the criteria to start step2 to control the symptoms and minimize future risk? |
GINA 2024 |
Start with step 2 in children with: Symptoms ≥twice / week but not daily |
|
GPS |
|
Q7.b |
What is the preferred controller in step 2? |
GINA 2024 |
Daily low dose ICS |
High |
Strong |
|
Q7.c |
What is the other controller options in step 2? |
GINA 2024 |
Daily Leukotrienes receptor antagonist or low dose ICS taken whenever SABA taken |
High
Intermediate |
Strong
Strong |
|
Q8.a |
In children 6 years and older, what is the criteria to start step3 to control the symptoms and minimize future risk? |
GINA 2024 |
Start with step 3 in children with: Symptoms most days or waking from asthma once or more / week |
|
GPS |
|
Q8.b |
What is the preferred controller in step 3? |
GINA 2024 |
Low dose ICS-LABA OR Medium dose ICS or Very low dose ICS-LABA (MART) |
High
Intermediate |
Strong
Strong |
|
Q8.c |
What is the other controller options in step 3? |
GINA 2024 |
Low dose ICS+ LTRA |
|
GPS |
|
Q9.a |
In children 6 years and older, what is the criteria to start step4 to control the symptoms and minimize future risk?
|
GINA 2024 |
Start with step 4 in children with: Symptoms most days or waking from asthma once or more / week + low lung functions if available |
Very low |
Weak
|
|
Q9.b |
What is the preferred controller in step 4? |
GINA 2024 |
Medium dose ICS-LABA OR
Low dose ICS-LABA (MART) OR Refer for expert advice |
Intermediate
Very low |
Strong
Weak (conditional) |
|
Q9.c |
What is the other controller options in step 4? |
GINA 2024 |
Add-on tiotropium
or Add on LTRA |
High |
Strong
GPS |
|
Q10.a |
In children 6 years and older, what is the criteria to start step 5 to control the symptoms and minimize future risk?
|
GINA 2024 |
Start with step 5 in children not controlled with step 4 and refer to expert
|
Very low |
Weak (conditional) |
|
Q10.b |
What is the preferred expert controller in step 5? |
GINA 2024 |
Refer for phenotypic assessment ±higher dose ICS-LABA or Add-on therapy, e.g. anti-IgE,, anti- IL5, Add on long acting muscarinic antagonist (LAMA) (Tiotropium) or anti-IL4R |
High |
Strong
GPS |
|
Q10.c |
What is the other controller options in step 5? |
GINA 2024 |
Consider add on low dose OCS but consider side effects |
|
GPS |
|
|
Difficult to treat asthma |
||||
|
Q11 |
What is difficult to treat asthma? |
GINA 2024 |
It is asthma that is uncontrolled despite prescribing of medium- or high-dose ICS with a second controller (usually a LABA) or with maintenance OCS, or that requires high-dose treatment to maintain good symptom control and reduce the risk of exacerbations. It does not mean a ‘difficult patient’. In many cases, asthma may appear to be difficult to treat because of modifiable factors such as incorrect inhaler technique, poor adherence, smoking or comorbidities, or because the diagnosis is incorrect
|
|
GPS |
|
Q12 |
How to assess difficult to treat asthma?
|
British 2019
British 2019
GINA 2024
GINA 2024 |
Patients with difficult asthma should be systematically evaluated, including:
A. Confirmation of the diagnosis of asthma (refer to Q1 and Q4)
B. Clinician should consider poor adherence to maintenance therapy
C. Review history and examination for comorbidities These include anxiety and depression, obesity, deconditioning, chronic rhinosinusitis, inducible laryngeal obstruction, GERD, obstructive sleep apnea, bronchiectasis, cardiac disease, and kyphosis due to osteoporosis. Investigate according to clinical suspicion.
D. Modifiable risk factors and triggers: identify factors that increase the risk of exacerbations, e.g., environmental tobacco exposure, other environmental exposures (indoor and outdoor) air pollution, molds and noxious chemicals, and medications such as beta-blockers or nonsteroidal anti-inflammatory drugs (NSAIDs).
|
Very low
Low |
Weak (conditional)
Weak
GPS
GPS |
|
|
Asthma resistant to therapy |
||||
|
Q13 |
What is asthma resistant to therapy? |
GINA 2024 |
Severe asthma is a subset of difficult-to-treat asthma. It means asthma that is uncontrolled despite adherence with maximal optimized high-dose ICS-LABA treatment and management of contributory factors, or that worsens when high-dose treatment is decreased |
|
GPS |
|
Q14 |
When to refer patients with asthma resistant to therapy? |
GINA 2024 |
In Children 6 years and older Children with persistent asthma symptoms or exacerbations despite correct inhaler technique and good adherence with Step 4 treatment and in whom other controller options have been considered, should be referred to a specialist with expertise in investigation and management of severe asthma, if available |
Very low |
Weak (conditional) |
|
Q15 |
How to assess asthma resistant to therapy?
|
GINA 2024 |
In Children 6 years and older -Further assessment and management should be done by a specialist, preferably in a multidisciplinary severe asthma clinic if available -Additional investigations may be appropriate for identifying less-common comorbidities and differential diagnoses contributing to symptoms and/or exacerbations and should be based on clinical suspicion -Assess the severe asthma phenotype |
|
GPS |
|
|
Immunotherapy (IT) |
||||
|
Q16 |
What about the role of allergy testing for severe asthma? |
(GINA) 2024 |
• Consider skin prick test (SPT) or specific IgE if not done before
|
|
GPS |
|
Q17 |
What is the role of IT in asthma control? |
(GINA) 2024 |
• Allergen immunotherapy may be considered as add-on-therapy for asthmatic children who have clinically significant sensitization to aeroallergens including those with allergic rhinitis. • When considering SCIT for children with asthma the potenial benefits compared with pharmacological treatment and allergn avoidance must be weighed against the risk of adverse effects and the inconvience and the cost of prolonged coarse of therapy (3-5 years)
|
Very low |
Weak |
|
Q18 |
What is the role of biological therapy in asthma treatment? |
GINA 2024 |
•If available and affordable, consider an add-on Type 2 targeted biologic for patients with severe asthma who have allergic or eosinophilic biomarkers or need maintenance OCS. • Where relevant, test for parasitic infection, and treat if present, before commencing treatment. • Consider whether to start first with anti-IgE, anti-IL5/5Rα, anti-IL4Rα or anti-TSLP |
|
GPS |
|
|
Prevention |
||||
|
Q19 |
What is the best practice to prevent or reduce the severity of asthma attacks? |
||||
|
Q19.a |
Which interventions (avoidance or reduction of exposure to environmental factors) at home, school and outdoor environment can improve asthma control and prevent or reduce the severity of asthma attacks?
|
British 2019
British 2019
|
• Measures to reduce in utero or early life exposure to single aeroallergens, such as house dust mites or pets, or single food allergens, are not recommended for the primary prevention of asthma.
• For children at risk of developing asthma, complex, multifaceted interventions targeting multiple allergens may be considered in families able to meet the costs, demands and inconvenience of such a demanding program.
|
High
High
|
Strong
Strong
|
|
Q19.b |
Is food allergen avoidance for mothers is beneficial for preventing childhood asthma? |
British 2019 |
In the absence of any evidence of benefit and given the potential for adverse effects, maternal food allergen avoidance during pregnancy and lactation is not recommended as a strategy for preventing childhood asthma. |
Intermediate |
Strong |
|
Q19.c |
What is the effect of encouraging breast feeding in infants for asthma prevention? |
British 2019 |
Breastfeeding should be encouraged for its many benefits, including a potential protective effect in relation to early asthma
|
Low |
Weak (conditional) |
|
Q19.d |
Is Avoidance of tobacco smoke and other air pollutants decrease the risk of wheezing in infancy and the risk of persistent asthma. |
British 2019 |
Patients with asthma and parents/carers of children with asthma should be advised about the dangers of smoking and second-hand tobacco smoke exposure on their children including increased wheezing in infancy and increased risk of persistent asthma., and should be offered appropriate support to stop smoking.
|
Intermediate |
Strong |
|
Q19.e.1 |
Is Immunizations have any considerations related to asthma in infants and children |
British 2019 |
Immunizations should be administered independent of any considerations related to asthma. |
Low |
Weak (conditional) |
|
Q19.e.2 |
Is pneumococcal vaccine indicated for asthma? |
GINA 2024 |
Pneumococcal vaccine protects against invasive pneumococcal infection, but asthma alone is not a specific indication for pneumococcal vaccination.
|
|
GPS |
|
Q19.e.3 |
Is influenza vaccine indicated for asthma |
GINA 2024 |
Advise patients with moderate to severe asthma to receive an influenza vaccination every year, or at least when vaccination of the general population is advised |
|
GPS |
➡️Evidence to recommendations: Considerations
The GDG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original sources of appraised Guidelines CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG develops group of good practice statements to improve acceptability and feasibility.
➡️Implementation considerations
To improve Asthma care and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing bronchial asthma diagnosis, treatment, and prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of asthma care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Implementation Tools
Educational materials based on this Adapted CPG for treatment of asthma in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for management of bronchial asthma
2. Arabic Educational materials for nurses and mothers

https://www.magonlinelibrary.com/doi/abs/10.12968/bjsn.2016
1- MDI without spacer
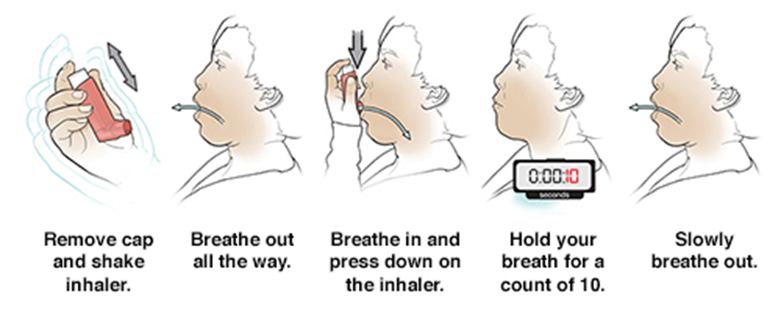
2- MDI with spacer
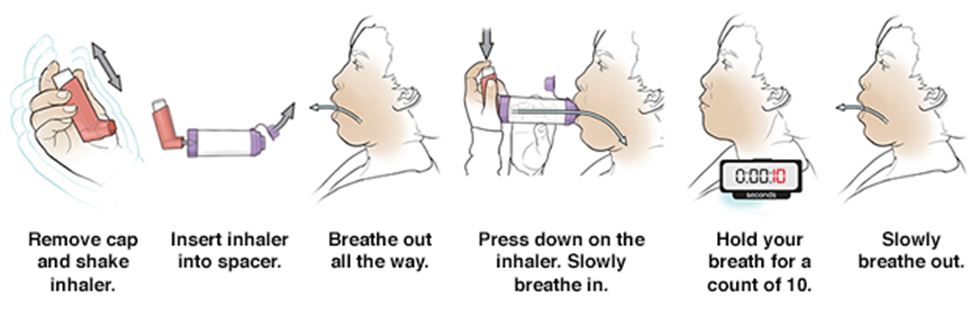
3- How to use a DPI ?
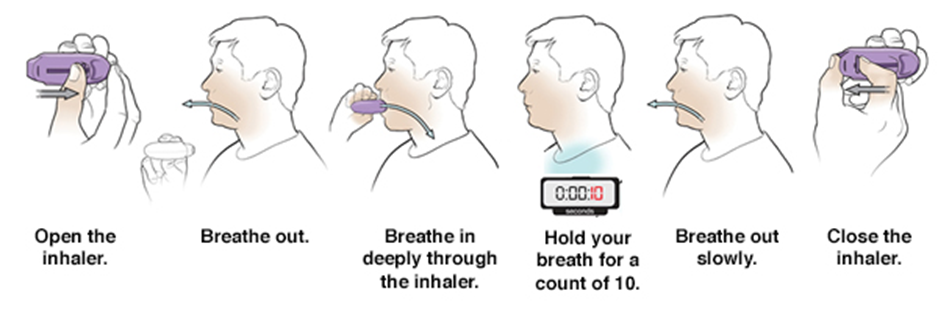
4-Nebulizer
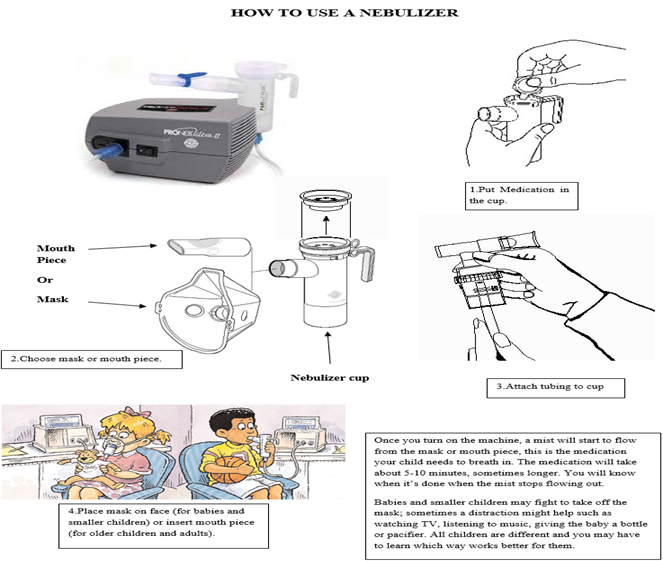
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation/ Development Group (Clinical subgroup) |
||
|
Names |
Affiliations |
Contribution |
|
Prof. Abla Saleh Mostafa |
Professor of Pediatrics, Cairo University |
Clinical expert, GAG member |
|
Dr. Aya Samir Mohamed Saleh |
Lecturer of Pediatrics, Cairo University |
Clinical expert, GAG member |
|
Prof. Dina Hossam-Eldine Hamed |
Professor of Pediatrics, Cairo University |
Clinical expert, GAG member |
|
Prof. Dina Tawfeek Sarhan |
Professor of Pediatrics, Zagazig University |
Clinical expert, GAG member |
|
Prof. Eman Mahmoud Fouda |
Professor of Pediatrics, Ain Shams University |
Editor, Clinical expert, GAG member |
|
Prof. Hala Gouda Elnady |
Professor of Pediatrics, National Research Center |
Clinical expert, GAG member |
|
Prof. Hala Hamdi Shaaban |
Professor of Pediatrics, Cairo University |
Clinical expert, GAG member |
|
Prof. Heba Ahmed Ali |
Ass. Professor of Pediatrics, Ain Shams University |
Clinical expert, GAG member |
|
Prof. Hoda M. Salah El-Din Metwally |
Professor of Pediatrics, Faculty of Medicine Girls Al-Azhar University |
Clinical expert, GAG member |
|
Prof. Magda Hassab Allah Mohamed |
Professor of Pediatrics, Faculty of Medicine Girls Al-Azhar University |
Clinical expert, GAG member |
|
Prof. Mohamed Mahmoud Rashad |
Professor of Pediatrics, Benha University |
Clinical expert, GAG member |
|
Prof. Mona Mohsen Elattar |
Professor of Pediatrics, Cairo University |
Clinical expert, GAG member |
|
Prof. Nesrine Radwan. |
Ass. Professor of Pediatrics, Ain Shams University |
Clinical expert, GAG member |
|
Prof. Shahenaz Mohamoud Hussein |
Professor of Pediatrics, Al-Azhar University |
Clinical expert, GAG member |
|
Prof. Tarek Hamed |
Professor of Pediatrics, Zagazig University |
Editor, Clinical expert, GAG member |
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation/ Development Group (Methodology subgroup) |
||||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
||||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Chairman of EPG |
Overseeing the adolopment process of the guidelines, revision of the final draft, Organizing weekly online meetings of GDG |
||||
|
Dr. Yasser Sami Amer |
Pediatrician and Guideline Methodologist. Alexandria University |
Overseeing the adolopment process of the guidelines |
||||
|
Associate Professor of Pediatrics Ain Shams University, Egypt |
Develop evidence to decision tables of the adaptation, participated in search and appraisal of guidelines |
|||||
|
Dr Lamis Mohsen |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participated in documentation of GDG meetings and in Methodology revision |
||||
|
Dr Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participated in Writing the methodology of adaptation process and revised the whole document. |
||||
|
Dr Nahla Gamaleldin |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participated in search and retrieved guidelines appraisal and revision of the document |
||||
|
Dr Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participated in search and retrieved guidelines appraisal and revision of the document |
||||
|
Guideline Adaptation/ Development Group (External Reviewers subgroup) |
||||||
|
National reviewers |
||||||
|
||||||
|
Prof Zinab Radwan |
Professor of Pediatrics, Cairo University, Egypt |
|||||
|
Prof. Sherif Mostafa Reda |
Professor of Pediatrics, Al-Azhar University, Egypt |
|||||
|
Prof. Dina Shoukri |
Professor of Pediatrics, Zagazig University, Egypt |
|||||
|
International reviewer |
|
|||||
|
||||||
|
Prof. Dr Bulunt Karatag |
Marmara University Faculty of Medicine, Division of Pediatric Pulmonology, Istanbul |
|||||
▪️ We acknowledge the academic members in the GDG for their great role in adaptation, modification, and development of this guideline with addition of their clinical experience.
▪️ The GDG acknowledge EPG for their help in completing this project.
▪️ We acknowledge the original guidelines for their cooperation in providing the permission in adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
- Funding
This work is not related to any pharmaceutical company. The members of
the guideline’s adaptation/development group and their institute and
universities volunteered their participation.
- Abbreviations
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
|
BTS |
British Thoracic Society |
|
CPG |
Clinical Practice Guideline |
|
DHS |
Demographic and Health Survey |
|
EBF |
Exclusive Breast Feeding |
|
EDHS |
Egyptian Demographic and Health Survey |
|
EPGC |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
|
EPG CPG |
EPG Clinical Practice Guideline |
|
ERS |
European Respiratory Society |
|
EtD |
Evidence to Decision |
|
FeNO |
Fractional Exhaled Nitric Oxide |
|
FEV1 |
Forced Expiratory Volume in 1 second |
|
GDG |
Guideline Development Group |
|
GERD |
Gastro-esophageal reflux disease |
|
GINA |
Global Initiative for Asthma |
|
GPS |
Good Practice statement |
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
|
HCP |
Health care Physicians |
|
ICS |
Inhaled Corticosteroids |
|
ICU |
Intensive Care Unit |
|
IgE |
Immunoglobulin E |
|
IL |
Interleukin |
|
IT |
Immune therapy |
|
JPGL |
Japanese Pediatric Guideline |
|
LABA |
Long Acting Beta2 Agonists |
|
LTRA |
Leukotriene Receptor Antagonists |
|
MART |
Maintenance and Reliever Therapy |
|
NSAID |
Non-Steroidal Anti-Inflammatory Drugs |
|
OCS |
Oral Corticosteroids |
|
PICO |
population, intervention, comparison, outcomes |
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
|
RSV |
Respiratory Syncytial Virus |
|
SCIT |
Sub-Cutaneous Immuno-Therapy |
|
SIGN |
Scottish Intercollegiate Guidelines Network |
|
SPT |
Skin Prick Test |
|
SABA |
Short Acting Beta2 Agonists |
|
Th2
|
T-helper2 |
- Glossary
➡️Health professionals
Health professionals’ study, advise on or provide preventive, curative, rehabilitative and promotional health services based on an extensive body of theoretical and factual knowledge in diagnosis and treatment of disease and other health problems. They may conduct research on human disorders and illnesses and ways of treating them and supervise other workers. The knowledge and skills required are usually obtained as the result of study at a higher educational institution in a health-related field for a period of 2–7 years leading to the award of a first degree or higher qualification. Health professionals include doctors, nurses, midwives, pharmacist, paramedical practitioners.
➡️Health workers
Health workers make up the health workforce and are people engaged to deliver health care to individuals and populations as part of the health system. Health workers are divided up into five main categories: health professionals, health associate professionals, personal care workers in health services, health management and support personnel, and other health service providers not elsewhere classified.
➡️Mother/caregiver-infant
This term is used predominantly in relation to infants to highlight the importance of providing services for the mother/caregiver-infant pair together with a holistic approach encompassing all their physical and mental health and nutrition needs and recognizing the interdependence of this unit, especially in the early months of an infant’s life.
➡️Referral
Referral, for the purpose of this guideline, refers predominantly to a child being referred to inpatient care from outpatient care. Asthmatic child might however also get referred to expert in asthma management for follow-up.
- Introduction
Asthma is a chronic inflammatory disease of the airways, characterized by recurrent episodes of wheeze, chest tightness, cough, and shortness of breath and airflow obstruction resulting from edema, bronchospasm, and increased mucus production. Commonly associated with seasonal allergies (allergic rhinitis) and eczema (atopic dermatitis), these three conditions form what is known as the atopic triad (1). There is a wide range in the frequency and severity of the symptoms, but uncontrolled asthma and acute exacerbations can lead to respiratory failure and death (2).
Poor asthma control has a significant impact on patients, their families, the healthcare system, and the community as a whole in terms of poor quality of life, frequent emergency department visits, hospitalizations, and deaths (3-5).
Asthma is a devastating global disease that affects people of all ages, with prevalence ranging from 1% to 21% in adults and up to 20% of children aged 6–7 years experiencing severe wheezing bouts within a year (6). The global prevalence of asthma ranges from 1% to 18% in different studies, with national prevalence estimates ranging from 1.7% to 53% in different countries and at different ages (7). The prevalence of asthma has increased dramatically in the last 20 years, indicating that environmental factors (allergens, infections, lifestyle, and food) play a significant role in the development of asthma (8). Bronchial asthma prevalence varies significantly by region, with industrialized countries having a higher frequency than developing countries (9).
Asthma is a leading cause of hospitalization in Egypt, and it is estimated that one out of every four children with asthma is unable to attend school on a regular basis due to poor asthma management (10).
The prevalence was 8.2 percent among Egyptian children aged 3 to 15 years. It was reported that asthma prevalence was 4.8% in Egyptian infants and children aged less than 4 years, from five governorates. Studies from Egypt reported that prevalence of asthma is 9.4% in 11– 15-year-old school in Cairo and 8.2% was reported in another study of children with age of 3–15 years (11).
➡️Pathophysiology of asthma:
Asthma is a chronic inflammatory airway disease that results in a narrow airway lumen. The airway narrowing is caused by smooth muscle contraction, bronchospasm, and increased mucus secretion as well as bronchial wall thickening due to edema, smooth muscle hypertrophy, and subepithelial fibrosis. The pathophysiological mechanisms that underlie these changes are diverse and heterogeneous. They are driven by a variety of cell types including immune cells; mainly T-helper cells (Th2, Th17, Th1), B-cells, mast cells, eosinophils, dendritic cells, and neutrophils; as well as structural bronchial cells such as epithelial cells, myofibroblasts, and smooth muscle cells (12). The mechanisms of asthma include the following: (figure 1) (13)
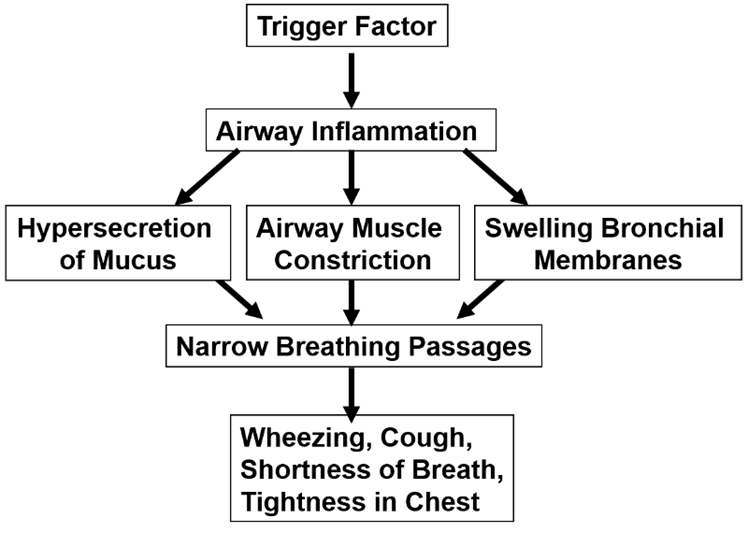
Figure 1: pathophysiology of asthma (Liu et al., 2021) (13).
Asthma Patterns in Childhood, Based on Natural History
1- Transient non atopic wheezing:
It is characterized by recurrent cough/wheeze that is primarily triggered by common respiratory viral infections and is most common in early preschool years. Usually resolves during the preschool and early school years, without increased risk for asthma in later life (14).
2- Persistent atopy-associated Asthma:
It starts in the early preschool years and is linked to atopy : either: Clinical (e.g., atopic dermatitis in childhood, allergic rhinitis, food allergy), or Biologic (e.g. elevated serum immunoglobulin E, increased blood eosinophils). Highest risk for persistence into later childhood and adulthood. Those who begin before the age of three have diminished airflow by the time they reach school age (15).
➡️Asthma with deteriorating lung function:
It is associated with hyperinflation in childhood and male gender. Children experience progressive airflow limitation (16).
➡️Asthma triggers:
Asthma attacks commonly occur following exposure to one or several triggers. Viral respiratory infections remain the leading cause, but there are a number of other known triggers, including aeroallergens, second hand smoke exposure, or changes in ambient air temperature or humidity. Identification and documentation of specific asthma triggers should be part of routine care. Education on trigger recognition and avoidance is essential (17)
➡️Risk factors of asthma:
There are a number of risk factors that should be explored in the history of children who present with features of asthma. In symptomatic children, a personal or family history of atopic features, including asthma, eczema or rhinitis, supports a diagnosis of asthma. Education on modifiable risk factors, for example, exposure to secondhand smoke or air pollution and obesity, should be delivered routinely during consultations and asthma reviews. A range of social determinants that are linked to poverty impact on outcomes and the health of children with asthma (18).
➡️Clinical Presentation of asthma:
Children with asthma typically present with a symptom triad of wheeze, shortness of breath and cough. However, ‘asthma’ is an umbrella term used to describe this collection of symptoms and, when present, should prompt practitioners to ask, ‘What type of asthma is this?’ There are a number of asthma subtypes that present and respond to treatment differently. Identification of the features of asthma and modifiable or treatable traits should only be the start of the diagnostic journey (19). Asthma symptoms are normally intermittent in nature and may not be present at the time of clinical review, making the diagnosis challenging in some cases (20). Additionally, disease phenotypes are not fixed and may evolve over time, necessitating ongoing review of symptoms and treatment. Wheeze is a key feature of asthma and, if not present, a diagnosis of asthma in a child is unlikely (21).
The prevalence of ‘preschool wheeze’ is an additional challenge when diagnosing asthma in young children. In the first few years of life, many children will experience wheeze, but not all will go on to develop true asthma. The diagnosis of asthma should therefore be reviewed routinely to identify true asthma and alter treatment where necessary (22). Favorable response to an appropriate trial of asthma treatment is an important confirmatory piece of diagnostic evidence. Clinical examination may be normal in children and adolescents with asthma if they present during asymptomatic periods. During acute attacks, use of accessory muscles of respiration and widespread wheeze may be present (23). Chest hyperinflation may be identified in acute and chronic disease settings.
Many of children with asthma develop symptoms before five years of age, but the disease is frequently misdiagnosed or not suspected particularly in infants and toddlers. Evaluating the presence of asthma symptoms is an important first step in establishing a proper diagnosis. (Figure 2) (24).
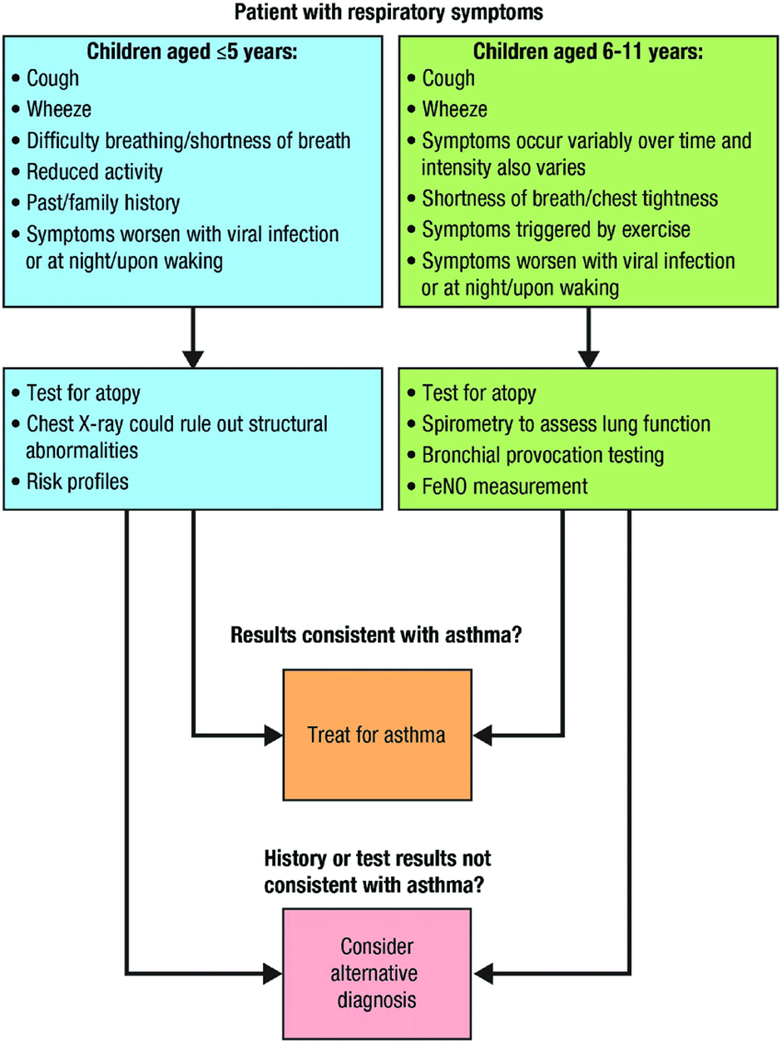
Figure 2: Diagnosis of asthma in children (Devani et al., 2022)(25)
➡️Asthma comorbidities:
Asthma is often associated with various comorbidities. The most frequently reported asthma comorbid conditions include:
I. Allergic rhinitis (26).
II. Sinusitis (27)
III. Gastroesophageal reflux disease (28).
IV. Allergic conjunctivitis (29).
V. Obesity (30).
VI. Obstructive sleep apnea (31).
VII. Eczema (31).
VIII. Food allergy (28).
➡️Differential diagnosis of asthma:
Table 1: Age-Related Differential Diagnosis for Wheezing (Kaplan et al., 2019) (32).

➡️Assessment of asthma control:
Treatment should be designed to ensure the asthma is well controlled, this means:
▪️ The patient doesn’t have symptoms on more than 2 days a week.
▪️ The patient doesn’t need his reliever medication on more than 2 days a week.
▪️ Asthma doesn’t limit what the patient can do.
▪️ The patient doesn’t have symptoms at night or on wakening up.
▪️ Achieving good asthma control and control quality of life (33).
Assessment of level of asthma symptom control (34)

➡️Difficult-to-treat asthma:
It is asthma that is un controlled despite prescribing of medium or high-dose ICS with a second controller or with maintenance OCS, or that require high-dose treatment to maintain good symptoms control and reduce the risk of exacerbations. It doesn’t mean a ‘difficult patient’. In many cases, asthma may appear to be difficult because of modifiable factors such as incorrect inhaler technique, poor adherence, exposure to smoking or other irritants, presence of comorbidities, or because the diagnosis is in correct. (35)
➡️Severe asthma:
It is a subset of difficult-to-treat asthma. It means asthma that is un controlled despite adherence with maximal optimized high-dose controller treatment and management of contributory factors, or that worsen when high-dose treatment is decreased.
Asthma is not defined as severe if it markedly improves when contributory factors are addressed. (35)
➡️Prognosis of asthma:
Recurrent coughing and wheezing occur in 35% of preschool-age children. Of these, approximately one-third continue to have persistent asthma into later childhood, and approximately two-thirds improve on their own through their teen years (36).
Asthma severity by the ages of 7-10 year is predictive of asthma persistence in adulthood. Children with moderate to severe asthma and with lower lung function measures are likely to have persistent asthma as adults. Children with milder asthma and normal lung function are likely to improve over time, with some becoming periodically asthmatic (disease-free for months to years); however, complete remission for 5 years in childhood is uncommon (37).
➡️Asthma predictive index
3 or more wheezing episodes plus at least 1 major criteria or 2 minor criteria.
I. Major criteria
• Parental history of asthma
• Physician-diagnosed atopic dermatitis
• Inhalant allergen sensitization
II. Minor criteria
• Physician-diagnosed allergic rhinitis
• Wheezing without colds
• Blood eosinophil count ≥4%
• Food allergen sensitization
The role of these tools is to help identify children at
greater risk of developing persistent asthma symptoms not as criteria for the diagnosis of asthma in
young children(38).
➡️Prevention of asthma:
A hygiene hypothesis purports that naturally occurring microbial exposures in early life might drive early immune development away from allergic sensitization, persistent airways inflammation, and remodeling. If these natural microbial exposures truly have an asthma-protective effect, without significant adverse health consequences, then these findings may foster new strategies for asthma prevention (39).
Several non-pharmacotherapeutic measures with numerous positive health attributes—avoidance of environmental tobacco smoke (beginning prenatally), exclusive breastfeeding (>4 months), an active lifestyle, and a healthy diet—might reduce the likelihood of asthma development (40).
The vaccinations are safe and effective in asthma, may help prevent asthma development, and pneumococcal and annual influenza vaccination in particular should be offered to asthmatics. Vaccines against Respiratory Syncytial Virus (RSV), which are the main triggers for asthma exacerbation and have been linked to asthma inception, should help reduce asthma morbidity and mortality (41).
➡️Purpose & Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of bronchial asthma in children. It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to improve early case detection and referral, case management of bronchial asthma. As a sequence, there will be an improvement in quality of life and medical condition for asthmatic children.
This version of the guideline includes recommendations and good practice statements for children suffering from bronchial asthma.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Using keywords: Management, Asthma, ICS, LABA, children 5 years and below, and children 6 years and above.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2019 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria the GDG & methodologists recommended using 6 guidelines:
1. SIGN/BTS 158: British guideline on the management of asthma, 2019
2. Japanese pediatric guideline for the treatment and management of asthma (JPGL), 2020
3. Canadian Thoracic Society 2021 Guideline update: Diagnosis and management of asthma in preschoolers, children, and adults
4. European Respiratory Society clinical practice guidelines for the diagnosis of asthma in children aged 5–16 years, 2021
5. GINA Report 2024
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
➡️Contributors to the guideline development process:
➡️Guideline Development Group (GDG):
The GDG for the guideline on prevention and management of Bronchial Asthma included experts with a range of technical skills and diverse perspectives in the field of pulmonology.
The main functions of the GDG were adolopment of selected guidelines for Asthma (2019&2024) determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
The protocol of this CPG adaptation Project was published in the official journal of the Guidelines International Network. (42-43)
➡️Guideline Methodologists:
There were guideline methodologists with expertise in guidelines development, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach & also, provided AGREE II assessment of the source guidelines in conjunction with CDG.(44-45)
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical experts who have interest and expertise in the prevention and management of Asthma in children. They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development Group meetings:
GDG meetings were organized virtually twice weekly. Due to the extensive scope of
the guideline, EPG Chair was responsible for the timetable and objectives of each meeting. GDG meetings were also attended by members of the methodologists and systematic. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) table was done while considering changing strength of recommendations according to availability of some resources in the recommendations (we did this for only 2 recommendations).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
➡️GRADE EtD criteria and considerations that link to the strength of recommendations:
Criteria Considerations:
Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost- effectiveness are more likely to support a strong recommendation.
Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements (GPS):
The GDG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
- Limitations and suggestions for further research needs
Future research recommendations for the management of Asthma in children in the Egyptian context could include:
▪️ More epidemiological research.
▪️ Studies on parents attitude, behavior and knowledge about asthma management
▪️ Efficacy studies on the management protocols
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for ASTHMA in children.
➡️Challenges
▪️ Socially and economically unprivileged population
▪️ Lack of enough trained healthcare workers to deal with these problems
▪️ Limited resources
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for Asthma in children:
1. Adherence to asthma Guidelines
▪️ Numerator: Number of children with asthma who received treatment as per guideline recommendations.
▪️ Denominator: Total number of children diagnosed with asthma
▪️ Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
▪️ Numerator: Total number of hospital stay days for children with severe asthma
▪️ Denominator: Total number of children admitted with asthma
▪️ Data Source: Hospital admission and discharge records.
3. Rate of Readmission
▪️ Numerator: Number of children readmitted with symptoms of asthma within a certain period (e.g., 30 days) after discharge.
▪️ Denominator: Total number of children initially admitted with asthma.
▪️ Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG WGAG has decided to conduct the next review of this adapted CPG for updates after three years. This should be carried out in 2027 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the fore mentioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The updating will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) that is one of the AGREE Tools
- References
- Khan, M. S. A., Khan, A. A. & Latafat, T. Developments in the Understanding of Bronchial Asthma and Contribution of Greeco-Arab Physicians. Journal of Integrated Community Health 2020; (ISSN 2319-9113), 9, 22-7.
- Kuruvilla, M. E., Lee, F. E.-H. & Lee, G. B. Understanding asthma phenotypes, endotypes, and mechanisms of disease. Clinical reviews in allergy & immunology 2019; 56, 219-33.
- Mohamed Hussain S, Ayesha Farhana S, Mohammed Alnasser S. Time trends and regional variation in prevalence of asthma and associated factors in Saudi Arabia: A systematic review and meta-analysis. Biomed Res Int 2018; 2018:1-9.
- Alatawi A, Alanazi M. Barriers of asthma care among asthmatic children in Saudi Arabia: Maternal perspectives. Open J Pediatr 2020; 10:302.
- Taminskiene V, Alasevicius T, Valiulis A, Vaitkaitiene E, Stukas R, Hadjipanayis A, et al. Quality of life of the family of children with asthma is not related to asthma severity. Eur J Pediatr 2019; 178:369-76
- Dharmage, S. C., Perret, J. L. & Custovic, A. Epidemiology of asthma in children and adults. Frontiers in pediatrics 2019; 7, 246.
- Agarwal, R., Dhooria, S., Aggarwal, A. N., Maturu, V. N., Sehgal, I. S., Muthu, V., et al. Guidelines for diagnosis and management of bronchial asthma: Joint ICS/NCCP (I) recommendations. Lung India 2015; 32, S3-S42.
- Asher, M. I., García-Marcos, L., Pearce, N. E. & Strachan, D. P. 2020. Trends in worldwide asthma prevalence. European Respiratory Journal 2020; 56 (6):2002094.
- Ibrahim, N. M., Almarzouqi, F. I., Al Melaih, F. A., Farouk, H., Alsayed, M. & AlJassim, F. M. Prevalence of asthma and allergies among children in the United Arab Emirates: A cross-sectional study. World Allergy Organization Journal 2021; 14, 100588.
- Mohammed, M., ABDELAKHER, A. R., SHOKRY, D. M. & SAFAA, A. E. Prevalence of bronchial asthma among school aged children in Elmaraghah Center in Sohag Governorate. The Medical Journal of Cairo University 2020; 88, 1097-101
- Ghonem, M. G. A. Prevalence of Bronchial Asthma among Primary School Children. The Egyptian Journal of Hospital Medicine 2022; 88, 3256-61.
- Pavord ID, Beasley R, Agusti A, Anderson GP, Bel E, Brusselle G, et al. After asthma: Redefining airways diseases. Lancet 2018; 391:350-400
- Liu, E. G., Yin, X., Swaminathan, A. & Eisenbarth, S. C. Antigen-presenting cells in food tolerance and allergy. Frontiers in immunology 2021; 11, 616020.
- Divaret‐Chauveau, A., Mauny, F., Hose, A., Depner, M., Dalphin, M. L., Kaulek, V., et al. Trajectories of cough without a cold in early childhood and associations with atopic diseases. Clinical & Experimental Allergy 2023; 53, 429-42.
- Laubhahn, K. & Schaub, B. From preschool wheezing to asthma: Immunological determinants. Pediatric Allergy and Immunology 2023; 34, e14038.
- Gaillard, E. A., Kuehni, C. E., Turner, S., Goutaki, M., Holden, K. A., de Jong, C. C., et al. European Respiratory Society clinical practice guidelines for the diagnosis of asthma in children aged 5–16 years. European respiratory journal 2021; 58.
- Gautier C, Charpin D. Environmental triggers and avoidance in the management of asthma. J Asthma Allergy 2017; 10:47–56.
- Keet CA, Matsui EC, McCormack MC, et al. Urban residence, neighborhood poverty, race/ethnicity, and asthma morbidity among children on Medicaid. J Allergy Clin Immunol 2017; 140:822–7.
- Bush A, Pavord ID. The Lancet Asthma Commission: treating children in primary care. Prescriber 2018; 29:28–32.
- McCormack MC, Enright PL. Making the diagnosis of asthma. Respir Care 2008; 53:583–90.
- Kuruvilla ME, Lee FE-H, Lee GB. Understanding asthma phenotypes, Endotypes, and mechanisms of disease. Clin Rev Allergy Immunol 2019; 56:219–33.
- Fainardi V, Santoro A, Caffarelli C. Preschool wheezing: trajectories and long-term treatment. Front Pediatr 2020; 8:240.
- Townshend J, Hails S, McKean M. Diagnosis of asthma in children. BMJ 2007; 335:198–202
- Liu, Z., Fu, Z., Dai, J. H. & Niu, C. [Clinical features of children with bronchial asthma complicated by pulmonary fungal infection and risk factors for pulmonary fungal infection]. Zhongguo Dang Dai Er Ke Za Zhi 2019; 21, 431-5.
- Devani, P., Lo, D. K. H. & Gaillard, E. A. Practical approaches to the diagnosis of asthma in school-age children. Expert Review of Respiratory Medicine 2022; 16, 973-81.
- Goniotakis, I., Perikleous, E., Fouzas, S., Steiropoulos, P., & Paraskakis, E. A Clinical Approach of Allergic Rhinitis in Children. Children 2023; 10(9), 1571.
- Laidlaw, T. M., Mullol, J., Woessner, K. M., Amin, N., & Mannent, L. P. Chronic rhinosinusitis with nasal polyps and asthma. The Journal of Allergy and Clinical Immunology: In Practice 2021; 9(3), 1133-1141.
- Ullmann, N., Mirra, V., Di Marco, A., Pavone, M., Porcaro, F., Negro, V., ... & Cutrera, R. Asthma: differential diagnosis and comorbidities. Frontiers in pediatrics 2018; 6, 276.
- Sanchez-Hernandez, M. C., DORDAL, T., Navarro, A. M., Dávila-García, M. I., Fernández-Parra, B., Colás, C., & Izquierdo-Dominguez, A. Severity and duration of allergic conjunctivitis: are they associated with severity and duration of allergic rhinitis and asthma? Eur Ann Allergy Clin Immunol 2022 Nov;54(6):277-283.
- Lin, N. Y., & Guilbert, T. W. Management of Severe Asthma in Children. Current Treatment Options in Pediatrics 2018; 4, 438-455.
- Wang, R., Mihaicuta, S., Tiotiu, A., Corlateanu, A., Ioan, I. C., & Bikov, A. Asthma and obstructive sleep apnoea in adults and children–an up-to-date review. Sleep Medicine Reviews 2022; 61, 101564.
- Kaplan, A., Hardjojo, A., Yu, S., & Price, D. Asthma across age: insights from primary care. Frontiers in pediatrics 2019; 7, 162.
- Beasley, R., Braithwaite, I., Semprini, A., Kearns, C., Weatherall, M., & Pavord, I. D. Optimal asthma control: time for a new target. American journal of respiratory and critical care medicine 2020; 201(12), 1480-1487
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, 2022, (GINA, 2022). Available at: www.ginasthma.org/reports
35. Global Initiative for Asthma. Difficult-to –treat & severe asthma in adolescent and adult patients, v5.0,2024. Available from: www.ginasthma.org/reports
- Trivedi, M., & Denton, E. Asthma in children and adults—what are the differences and what can they tell us about asthma? Frontiers in pediatrics 2019; 7, 256.
- Izadi, N., Baraghoshi, D., Curran-Everett, D., Zeiger, R. S., Szefler, S. J., & Covar, R. A. Factors associated with persistence of severe asthma from late adolescence to early adulthood. American Journal of Respiratory and Critical Care Medicine 2021; 204(7), 776-787.
- Chang T S.,, Lemanske R F., Guilbert T W, Gern J E., , Coen M H.,, Evans M D., Gangnon R E.,. Page D, and Jackson J. Evaluation of the Modified Asthma Predictive Index in High-Risk Preschool Children J Allergy Clin Immunol Pract. 2013 March 1; 1(2): . doi:10.1016/j.jaip.2012.10.008.
- Krusche, J., Basse, S., & Schaub, B. 2020, February. Role of early life immune regulation in asthma development. In Seminars in immunopathology 2020 Feb;42(1):29-42.
- Abreo, A., Gebretsadik, T., Stone, C. A., & Hartert, T. V. The impact of modifiable risk factor reduction on childhood asthma development. Clinical and translational medicine 2018; 7(1), 1-12.
- Edwards, M. R., Walton, R. P., Jackson, D. J., Feleszko, W., Skevaki, C., Jartti, T., … Khaitov, M. R. The potential of anti-infectives and immunomodulators as therapies for asthma and asthma exacerbations. Allergy: European Journal of Allergy and Clinical Immunology 2018; 73(1), 50-63
- Abdel Baky A, Youssef A, Elsholkamy L, Saber M, Gamal N, Soliman N, et al. A protocol for adapting a clinical practice guideline for the treatment of paediatric asthma for the Egyptian Pediatric Clinical Practice Guidelines Committee. Clin Pub Health Guidelines. 2024; 1:e12011. https://doi.org/10.1002/gin2.12011
- Abdel Baky, A., Omar, T.E.I., Amer, Y.S. et al. Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent 47, 88 (2023). https://doi.org/10.1186/s42269-023-01059-0
- Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. J Eval Clin Pract. 2015 Dec;21(6):1095-106. doi: 10.1111/jep.12479. Epub 2015 Dec 14.
- Klugar M, Lotfi T, Darzi AJ, et al. GRADE guidance 39: using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. J Clin Epidemiol. 2024 Oct;174:111494. doi: 10.1016/j.jclinepi.2024.111494.
- Annexes
Annex Table 1.
Conflict of Interest
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Abla Saled Mostafa |
Professor of Pediatrics, Cairo University |
Non |
Not Applicable |
|
Dr. Aya Samir Mohamed Saleh |
Lecturer of Pediatrics Cairo University, Egypt |
Non |
Not Applicable |
|
Prof. Dina Hossam-Eldine Hamed |
Professor of Pediatrics Cairo University, Egypt |
Non |
Not Applicable |
|
Prof. Dina Tawfeek Sarhan |
Professor of Pediatrics, Zagazig University |
Non |
Not Applicable |
|
Prof. Eman Mahmoud Fouda |
Professor of Pediatrics, Ain Shams University |
Non |
Not Applicable |
|
Prof. Hala Gouda Elnady |
Professor of pediatrics, National Research center |
Non |
Not Applicable |
|
Prof. Hala Hamdy Shaaban |
Professor of Pediatrics, Cairo University |
Non |
Not Applicable |
|
Ass. Prof. Heba Ali |
Ass. Prof. of pediatrics Ain Shams University |
Non |
Non Applicable |
|
Prof. Hoda M. Salah El-Din Metwally |
Professor of Pediatrics, AL-Azhar University |
Non |
Not Applicable |
|
Prof. Magda Hassab Allah Mohamed |
Professor of Pediatrics, AL-Azhar University |
Non |
Not Applicable |
|
Prof. Mohamed Mahmoud Rashad |
Professor of Pediatrics, Benha University |
Non |
Not Applicable |
|
Prof. Mona Mohsen Ellattan |
Professor of Pediatrics, Cairo University |
Non |
Not Applicable |
|
Prof. Mostafa Al-Saeed |
Professor of Pediatrics, Assuit University |
Non |
Not Applicable |
|
Prof. Nesrine Radwan |
Professor of Pediatrics, Ain Shams University |
Non |
Not Applicable |
|
Shahenaz Mahmoud Hussein |
Professor of Pediatrics, AL-Azhar University |
Non |
Not Applicable |
|
Tarek Hamed |
Professor of Pediatrics, Zagazig University |
Non |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics, MTI, AFCM/ Ain Shams University, Egypt Chair of EPG |
Non |
Not Applicable |
|
Dr. Yasser Sami Amer |
Pediatrician and Guideline Methodologist. Alexandria University |
Non |
Not Applicable |
|
Dr. Nanis Sulieman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
Non |
Not Applicable |
|
Dr Lamis Mohsen |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Non |
Not Applicable |
|
Dr Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Non |
Not Applicable |
|
Dr Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Non |
Not Applicable |
|
Dr Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Non |
Not Applicable |
|
External Review Group |
|||
|
Prof Ahmad ab del raziq |
Professor of Pediatrics, Tanta University, Egypt |
Non |
Not Applicable |
|
Prof. |
Professor of Pediatrics, Assiut University, Egypt |
Non |
Not Applicable |
|
External Reviewer for methodology |
|||
|
Prof. Yasser Samy |
Chair, Adaptation working Group, Guidelines International Network, Scotland. Pediatrics Department and Clinical Practice Guidelines Unit, King Saud University, Saudi Arabia. |
Non |
Not Applicable |
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for " Guideline for the prevention and management of Asthma management in children " on PubMed are: asthma, ICS, LABA / children 5 years and younger, children 6 years and older, difficult to treat asthma, immunotherapy / prevention, / management.
Annex Table 2. Results of the AGREE II assessment of the five source guidelines for asthma.
|
AGREE II/ CPGs |
ERS |
GINA |
JPGL |
BTS/SIGN |
CTS |
|
Domain 1 (Scope) |
53% |
83% |
81% |
63% |
89% |
|
Domain 2 (Stakeholder) |
53% |
80% |
80% |
81% |
85% |
|
Domain 3 (Rigor) |
49% |
85% |
76% |
73% |
84% |
|
Domain 4 (Clarity) |
54% |
85% |
83% |
80% |
89% |
|
Domain 5 (Applicability) |
45% |
78% |
65% |
68% |
89% |
|
Domain 6 (Independence) |
46% |
69% |
89% |
78% |
78% |
|
Overall assessment 1 |
54% |
83% |
78% |
78% |
94% |
|
Recommend for use (Overall assessment 2) |
YES-2 |
YES-0 |
YES-1 |
YES-1 |
YES-0 |