Screening of prediabetes and Type 2 Diabetes in Pediatrics
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Screening of prediabetes and Type 2 Diabetes in Pediatrics |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 23 March 2025" Download Guideline
- Executive Summary
➡️Introduction
Definition of the disease condition
Diabetes mellitus is a complex metabolic disorder characterized by chronic hyperglycemia resulting from defects in insulin secretion, insulin action, or both. Inadequate insulin secretion and/or diminished tissue responses to insulin in the complex pathways of hormone action result in deficient insulin action on target tissues, which leads to abnormalities of carbohydrate, fat, and protein metabolism. The etiology of diabetes is heterogeneous, however, most cases can be classified into two broad etiopathogenetic categories type 1 diabetes (T1D), which is characterized primarily by deficiency of insulin secretion; or type 2 diabetes (T2D), which results from a combination of resistance to insulin action, as well as an inadequate compensatory insulin secretory response for the degree of insulin resistance, due to progressive non-autoimmune β-cell failure. It was formerly known as adult-onset diabetes or non-insulin dependent diabetes.
➡️Magnitude of the problem worldwide.
While T1D remains the most common form of diabetes in children and adolescents worldwide, T2D is becoming more prevalent and imposes an increasingly important public health burden globally. It is stated that T2D in youth has become widespread in many regions of the world; and its frequency in the pediatric age range is increasing.
In many countries, the incidence of T2DM in adolescents continues to increase. Similarly, the prevalence of prediabetes, defined in adults as a state of high-risk for progression to diabetes, is increasing quickly in some developing countries with the increase of overweight and obesity.
Worldwide incidence and prevalence of T2D in children and adolescents vary substantially among countries, age categories and ethnic groups. Epidemiologic studies have shown variations in the incidence of T2D in children and adolescents ranging from 1 to 51 per 1000
The difference in the incidence and prevalence rates of T2D in children might be attributed to genetics and hormonal factors, environmental influences, different quality of life, and access to health care.
➡️Magnitude of the problem in Egypt with impact on health and economy
In Egypt, T2D is presenting a major public health care burden with a total cost of 25.2 billion EGP equivalent to 3.5 billion USD. According to the IDF, the annual prevalence increment rate of T2D in Egypt is 266%. Moreover, Egypt is thought to jump from the 9th to the 7th rank among the countries with highest prevalence of T2D worldwide by 2030 with a prevalence of 12.4 millions. Data about the prevalence of T2D in children and adolescents are sparse. Studies showed increasing prevalence rates varying from 0.01% in 2002 to 0.3% recently in 2018.
➡️Role of screening and early detection in ameliorating this impact.
T2D is a complex and multifactorial disease, characterized by an association between genetic, epigenetic, and environmental factors. The pathophysiology of T2D in children is more complex than that in adults. Beta-cell dysfunction occurs more rapidly in children than in adults.
Primary prevention approaches aiming at addressing the modifiable risk factors, starting from the intra-uterine period and extending throughout childhood, are of utmost importance in preventing T2DM. Despite the possibility of detecting the genetic susceptibility to T2DM by documenting compromised insulin secretion and insulin sensitivity in otherwise healthy children with a positive family history, it is not cost effective. Since the diagnosis of T2DM in youth is rare before puberty, it is detected more frequently during puberty when physiologic and transient insulin resistance (IR) occurs (insulin sensitivity declines by 25%–30% .
Maternal overweight and obesity were found to be associated with T2DM in offspring, irrespective of various confounding pre-existing or pregnancy-related conditions. Children born to obese or overweight women were found to have a 3.5- and 1.4-fold higher incidence of T2DM respectively, compared to those born to normal-weight women. Hence, one of the earliest points of childhood T2DM prevention is the reduction of maternal overweight and obese status during pregnancy. In addition, studies have documented that offspring of women with gestational diabetes (GDM) are at increased risk of T2DM and obesity, irrespective of the mother’s weight status during pregnancy. Not only overt GDM but also prediabetic conditions during pregnancy have been linked to glucose abnormalities and insulin resistance in the offspring. Protection of the fetus from intrauterine glycemic derangements is, therefore, of paramount importance to prevent prediabetic conditions and T2DM in childhood and adolescence. Other risk factors such as maternal diet, maternal smoking, and paternal obesity should also be considered.
In order to prevent childhood T2DM, one should clearly focus on intrauterine life, but postnatal exposures seem to play an equally important role. Childhood obesity is among the major modifiable risk factors of T2DM in children. Increasing BMI has been reported as a risk factor of glycemic deterioration and progression to T2DM.
The importance of programming in T2DM pathogenesis is growing. Dietary and environmental factors during the first 1000 days from conception to the second birthday have utmost importance in T2DM prevention. Breastfeeding was found to have a strong protective effect against childhood T2DM especially among high-risk populations. Moreover, both high and low birth weight, preterm birth, as well as rapid weight gain during the first months of life have been shown to increase the risk of childhood obesity, insulin resistance and glucose metabolism disturbances, predisposing to T2DM.
In an earlier review in 2007 including reports on acute and long-term comorbidities associated with type 2 diabetes in young people, microvascular complications have been identified at diagnosis, implying long-term, unrecognized hyperglycemia. Children may also present with acute decompensation in diabetic ketoacidosis (DKA) and/or hyperosmolar hyperglycemic state (HHS). This argues for a systematic screening program in children at high risk for type 2 diabetes in order to prevent an acute, life-threatening presentation and to decrease the development of chronic complications. Although not proven in children, it is generally assumed that earlier diagnosis of diabetes will lead to interventions that will improve glycemic control and reduce the related short- and long-term complications.
The optimal approach to screening and diagnosis of prediabetes and diabetes in youth is uncertain. In a recent cross-sectional analysis conducted by Wallace et al including 14,119 youth aged 10 to 19 years in the 1999–2016 NHANES; it was found that HbA1c is a specific and useful non fasting test to identify high-risk youth who could benefit from lifestyle interventions to prevent diabetes and cardiovascular risk in adulthood. Sensitivity and specificity of the screening criteria for detecting any hyperglycemia were low for both HbA1c ≥ 5.7% (sensitivity = 55.5%, specificity = 76.3%) and FPG ≥ 100 mg/dL (sensitivity = 35.8%, specificity = 77.1%). Confirmed undiagnosed diabetes (HbA1c ≥ 6.5% and FPG ≥ 126 mg/dL) was rare, 0.5% of youth. Associations with cardiometabolic risk were consistently stronger and more specific for HbA1c-defined hyperglycemia (specificity = 98.6%; sensitivity = 4.0%) than FPG defined hyperglycemia (specificity = 90.1%; sensitivity = 19.4%).
For better sensitivity, a combination of A1C and fasting or random blood glucose is recommended to screen for type 2 diabetes in children and youth with risk factors; with a 2-hour OGTT to be considered as an initial screening test in children and youth with 3 or more risk factors. A 2-hour OGTT was also recommended in those who show a discrepancy between the A1C and fasting or random blood glucose results on screening.
Recent analyses of data from US adolescents estimate prediabetes to be present in 4–23% of adolescents, depending on criteria used, with other studies finding an 8% risk of progression from prediabetes to T2DM over a 3-year period. These data support the importance of intervention to avoid long-term sequelae, focusing on reducing degree of obesity and insulin resistance
➡️Methods of prevention of progression and possible reversal of the disease condition
Most programs aiming at preventing childhood T2DM are based on obesity prevention, given the etiological connection between increased childhood obesity, metabolic syndrome and T2DM. These programs mostly target dietary interventions like providing healthier school meals, reducing simple sugars, and restricting unhealthy food advertisements aimed at children. Nutritional interventions should be combined with programs targeting increased physical activity and restricting sedentary life and screen time in order to achieve the best long-term outcome.
Regarding adolescents, school-based interventions have proven more effective when the adolescents were addressed directly.
Because of the rarity and the relatively recent emergence of the problem in children and adolescents, there has been limited evidence leading to unique challenges in the diagnosis, management, and monitoring of this vulnerable population. This limited evidence base is further complicated by differences in the characteristics and presentation of the disorder and approaches to treatment in developed and developing countries. Hence we aim to provide guidelines for screening and prevention of childhood T2D in Egypt.
➡️Scope
- This guideline focuses on screening and diagnosis of pre-diabetes and type 2 Diabetes with its comorbidities.
➡️ GUIDELINE OBJECTIVES
1) Screening for early detection of prediabetes allowing the opportunity to prevent or delay progression to type 2 diabetes and its comorbidities.
2) Early detection of type 2 diabetes in high-risk groups, to prevent or delay serious diabetes complications during adolescence and early adulthood.
3) Screening for early diagnosis of comorbidities or complications in adolescents with prediabetes and type 2 diabetes to prevent serious disabilities during adolescence and early adulthood.
➡️ Health/Clinical Question (PIPOH)
- P (patients, target population):
Gender: both genders.
Age group: adolescents and preadolescents in the age group of 10-19 years.
Disease/ condition: prediabetes and type 2 diabetes (T2DM).
Comorbidity: obesity with one or more of the following:
Hypertension, dyslipidemia, NACLD, PCOS, and other organ affection (clinical features suggestive of metabolic syndrome).
Exclusion criteria: other types of diabetes or hyperglycemic states (e.g. type 1 diabetes, MODY, secondary diabetes, and drug-induced diabetes).
- I (interventions and practices considered/ guideline category):
Screening (clinical and laboratory) for prediabetes and type 2 diabetes in adolescents (age group from 10 to 19 years).
Screening for early detection of comorbidities associated with those at risk of prediabetes or type 2 diabetes.
- P (Professionals / intended or target users and clinical specialties):
-Primary health care physicians at Ministry of Health (MOH).
-General pediatricians and school physicians.
-Family medicine physicians.
- O (major outcomes):
Primary outcome:
Early detection and prompt referral of prediabetes and type 2 diabetes in adolescents to pediatric endocrinologist care.
Secondary outcome:
- Prevention of progression of prediabetes to type 2 diabetes.
- Early detection of associated comorbidities of prediabetes and type 2 diabetes.
- H (Healthcare settings):
➡️Type: primary and secondary.
➡️Health care sector: governmental, non-governmental and private sectors.
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- ISPAD guidelines
(2022)
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Adaptation for 2 recommendations according to GRADE criteria to be suitable to our Economic implications (Evidence-to-Decision (EtD) table was done)
- Development of Good Practice Statements
➡️Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following:
➡️Diagnosis of Pre-diabetes and Type 2 Diabetes
The guideline covers Adolescents and preadolescents in the age group of 10-19 This Guideline emphasis on:
2) Early detection of type 2 diabetes in high-risk groups, to prevent or delay serious diabetes complications during adolescence and early adulthood.
3) Screening for early diagnosis of comorbidities or complications in adolescents with prediabetes and type 2 diabetes to prevent serious disabilities during adolescence and early adulthood.
➡️We can summarize the guidelines’ recommendations for Type 2 Diabetes in the following:
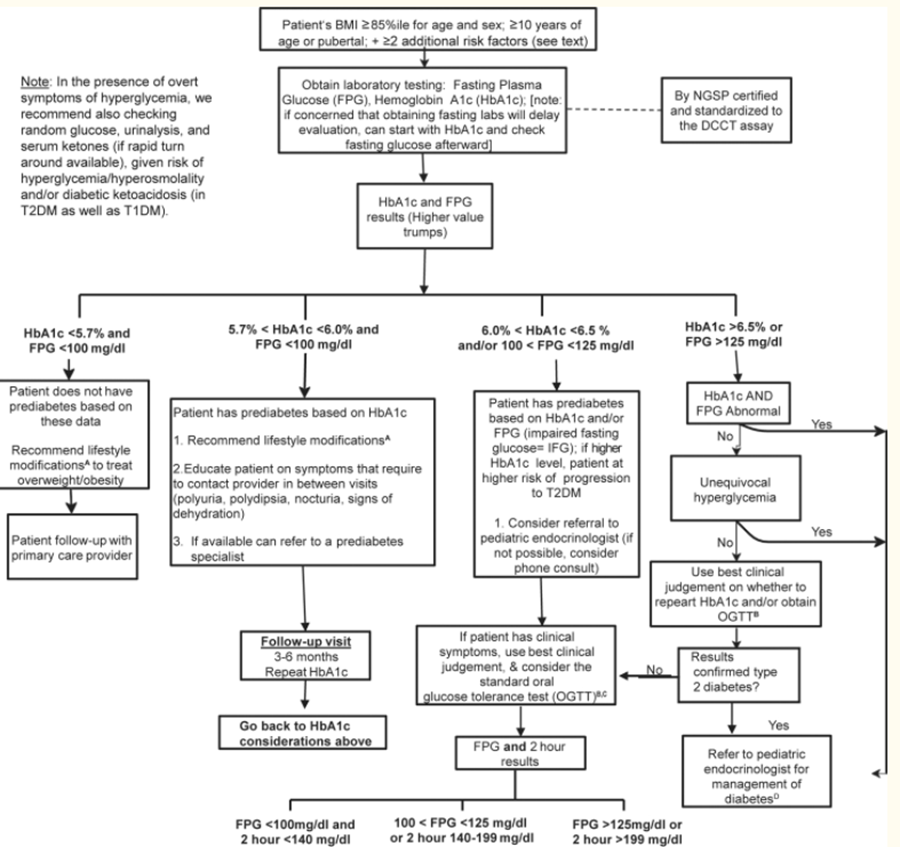
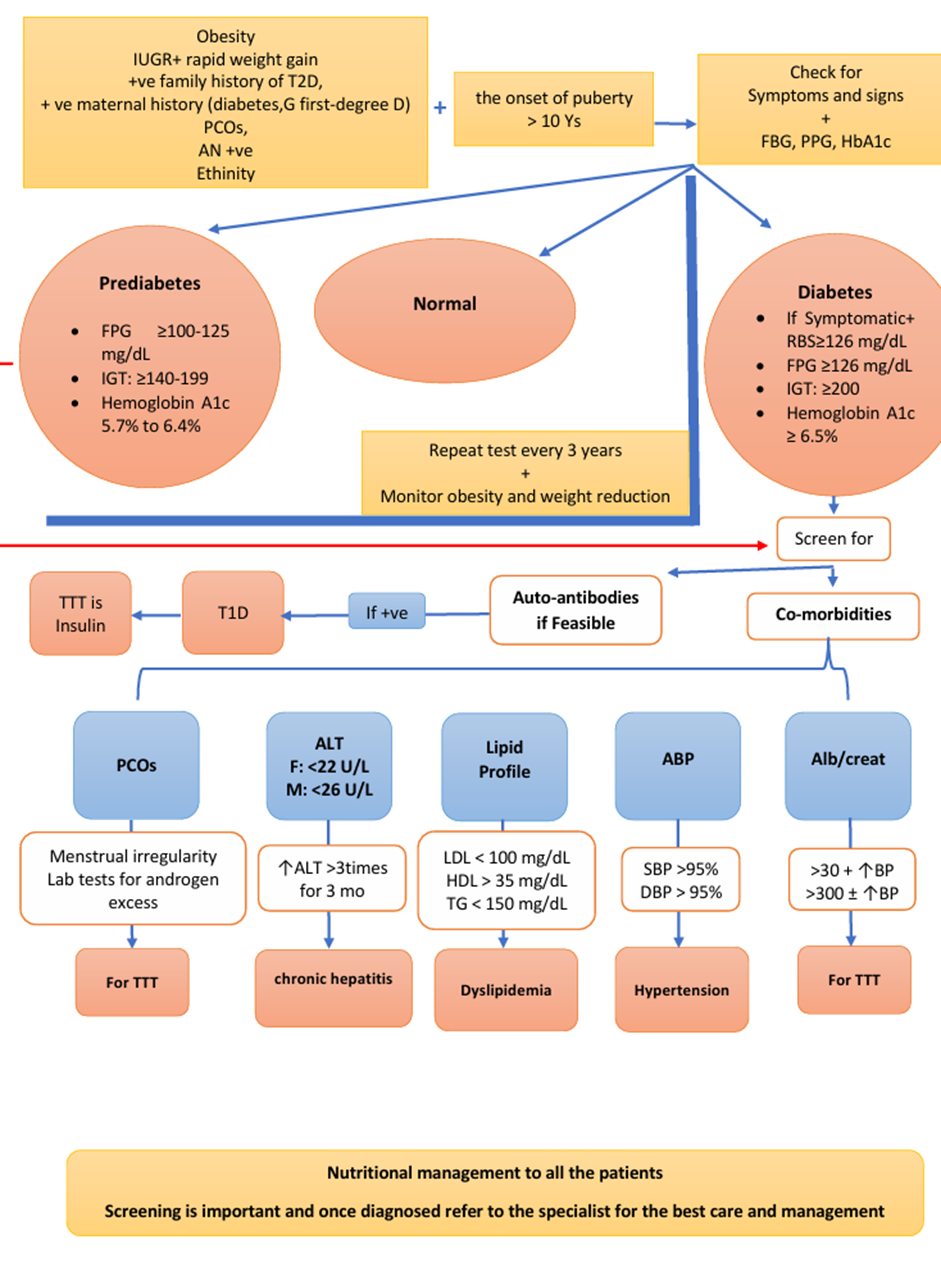
Adolescents at risk that should be screened for prediabetes or type 2 diabetes in primary health care setting or general pediatric department or outpatient clinic?
Risk factors to develop type 2 diabetes (High, Strong) :
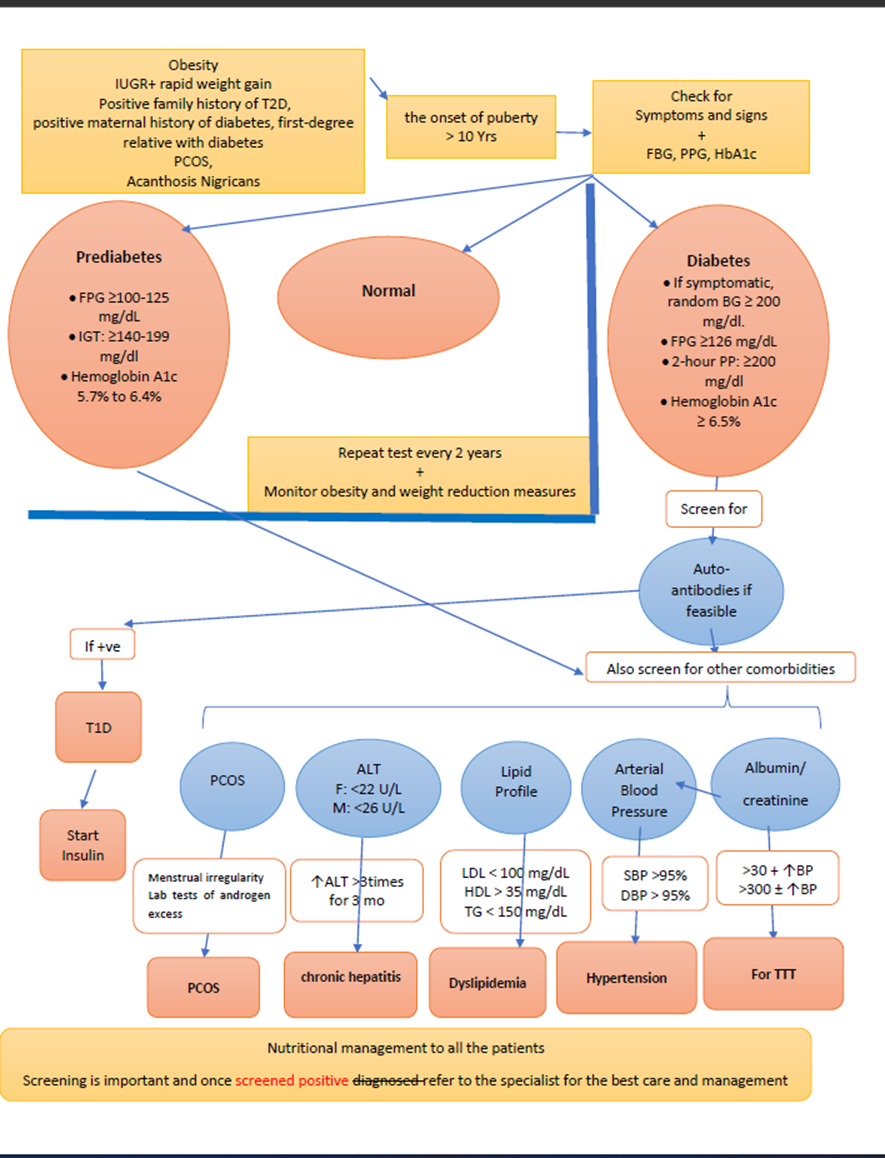
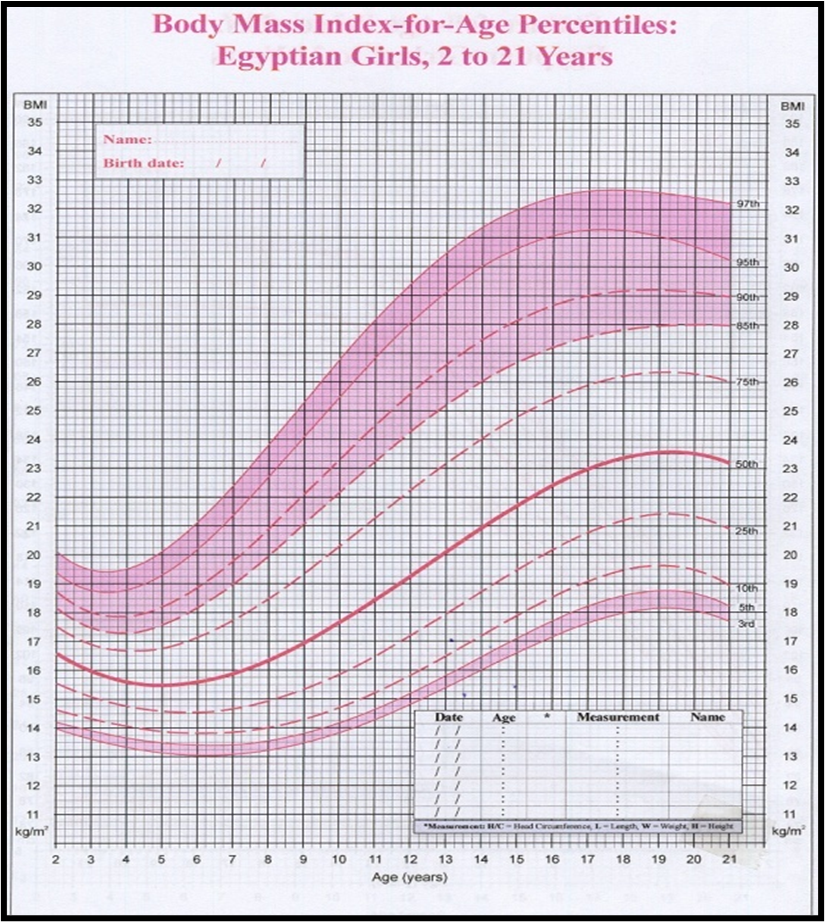
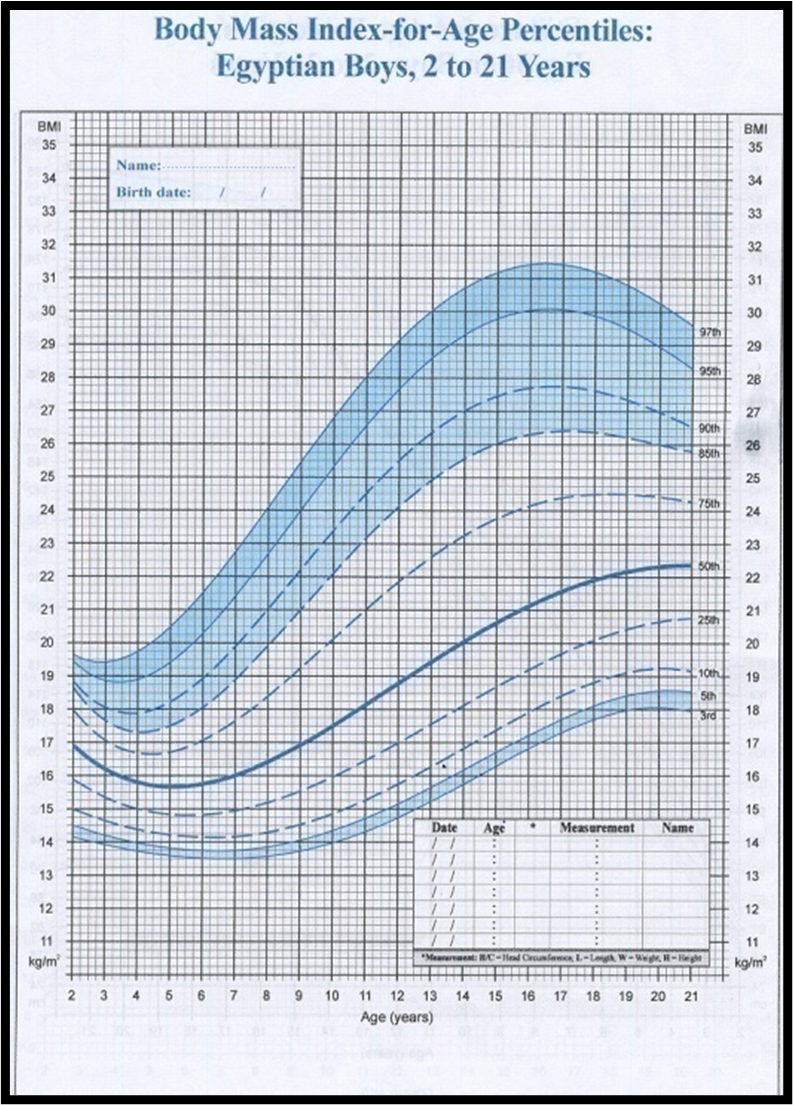
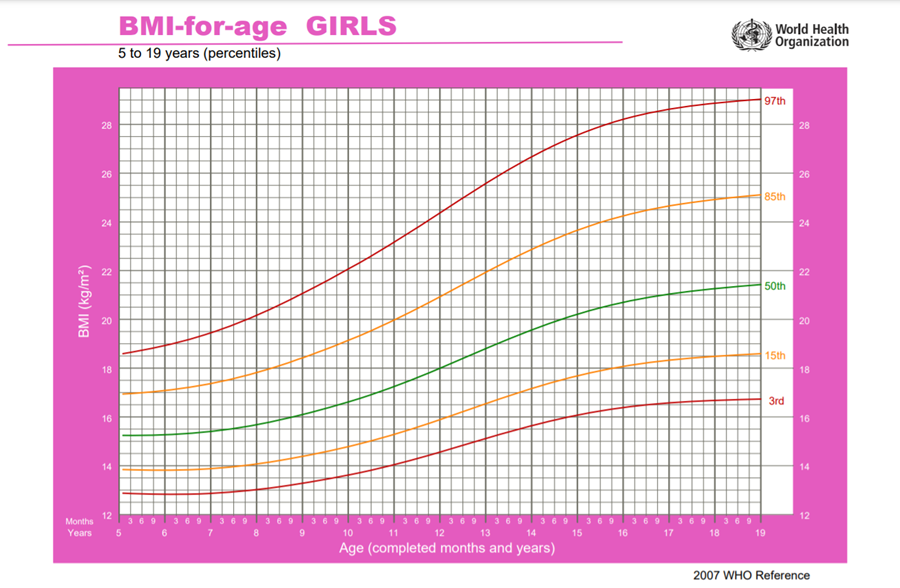
Obesity (BMI > 95th percentile for age and sex), intrauterine growth retardation with rapid infant weight gain, first-degree family history of type 2 diabetes, maternal history of diabetes or gestational diabetes during child's gestation, high-risk ethnicity, polycystic ovary syndrome (PCOS).
Risk-based screening for prediabetes and/or type 2 diabetes should be considered after the onset of puberty or after 10 years of age, whichever occurs earlier in children and adolescents who are overweight (BMI > 85th percentile) or obese (BMI > 95th percentile) and who have one or more additional risk factors for diabetes (Refer to table (1) in implementation tools for details and level of evidence) (High, Strong).
Assessment for dysglycemia in obese at-risk youth should occur in the setting of clinical assessment of other obesity-related comorbidities (metabolic dysfunction-associated steatotic liver disease (MASLD)), dyslipidemia, elevated blood pressure [BP], and polycystic ovary syndrome) that are more prevalent than dysglycaemia (Refer to Q 6) below) (High, Strong).
➡️Tests recommended for screening of prediabetes or type 2 diabetes in primary health care setting or general pediatric department or outpatient clinic:
Fasting plasma glucose, 2-h plasma glucose after 75-g OGTT, and HbA1C can be used to test for prediabetes or diabetes (Intermediate, Strong).
Recommend using a combination of A1C and fasting or random blood glucose to screen for type 2 diabetes in children and youth with risk factors.
“A 2-hour OGTT may be considered as an initial screening test in children and youth with 3 or more risk factors and should be done in those in whom there is a discrepancy between the A1C and fasting or random blood glucose results” (Good Practice Statement).
➡️Positive screening test result for prediabetes or type 2 diabetes in primary health care setting or general pediatric department?
Criteria for considering diagnosis of prediabetes according to ADA definitions (High, Strong):
• Impaired fasting glycemia (IFG): FPG ≥100-125 mg/dL
• Impaired Glucose tolerance (IGT): Post-challenge plasma glucose is ≥140-199 mg/dL
• Hemoglobin A1c ˂5.7% normal, 5.7% to 6.4% prediabetes.
(Should be laboratory-based, DCCT aligned, NGSP certified methodology).
Criteria for diagnosis of type 2 diabetes (High, Strong):
a) Symptoms of diabetes (polyuria, polydipsia, nocturia, and unexplained weight loss) and a random plasma glucose ≥200 mg/dL OR
• Fasting blood glucose ≥126 mg/dL OR
• 2-hour blood glucose concentration during an oral glucose tolerance test (OGTT) ≥200 mg/dL OR
• Hemoglobin A1c (HbA1c) ≥ 6.5%
(Should be laboratory-based, DCCT aligned, NGSP certified methodology).
Point-of-care measurement of HbA1c is not acceptable for diagnosis.
In the absence of symptoms, testing should be confirmed with a repeat test on a different day (Intermediate, Strong).
Measurement of insulin or C-peptide is NOT recommended as part of routine evaluation.
If tests are normal repeat testing at a minimum of 3-year intervals,
or more frequently if BMI is increasing, the cardiometabolic risk profile is deteriorating, there is a strong family history of T2D, or evidence of pre-diabetes (Low, Conditional).
➡️Referral to pediatric endocrinologist and diabetologist in adolescents with suspected prediabetes or type 2 diabetes in the primary health care setting or general pediatric department:
Referral to pediatric diabetologist/endocrinologist is recommended in children and adolescents suspected to have prediabetes or type 2 diabetes according to the American Diabetes Association (ADA) criteria (High, Strong).
➡️Screen for comorbidities of prediabetes and type 2 diabetes in adolescents at risk in the primary health care setting or general pediatric department or outpatient clinic:
➡️Hypertension:
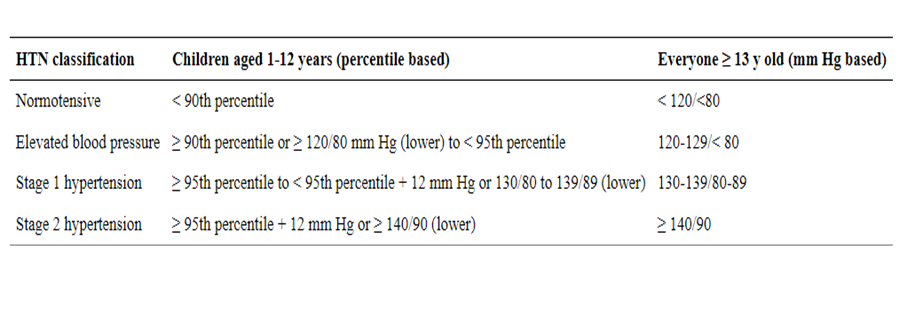
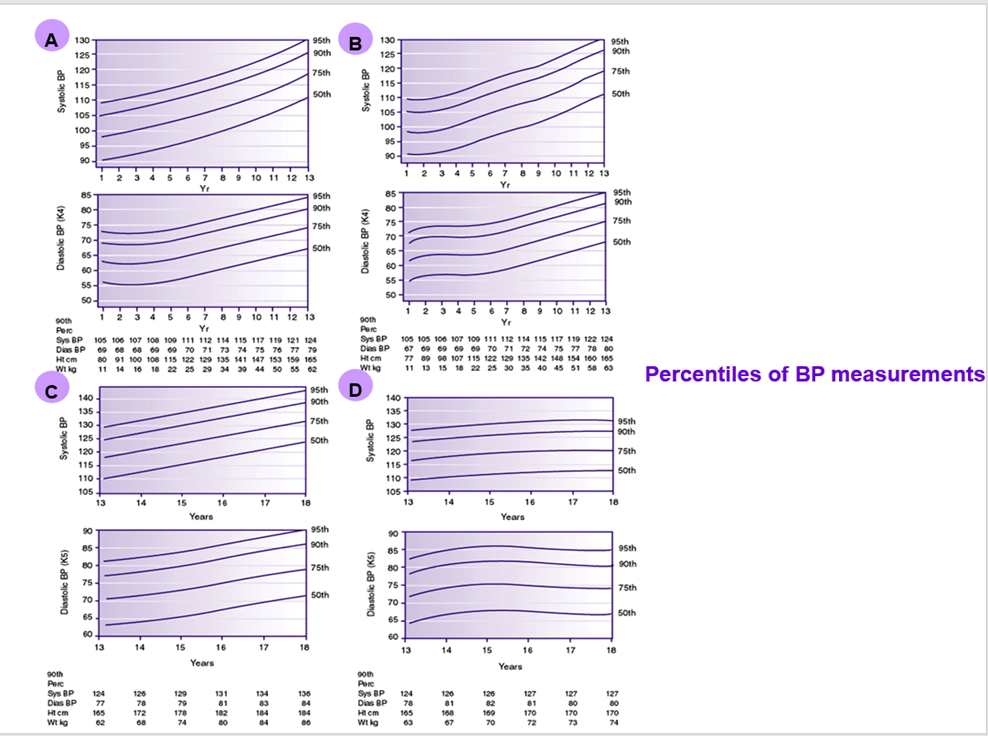
Blood pressure should be monitored at every visit according to standardized techniques specific for children.
Elevated BP should be confirmed on 2 additional separate days. (High, Strong)
Hypertension is defined as an average systolic or diastolic BP > 95th percentile for age, sex, and height, with high normal BP being 90th to <95th percentile.
Initial treatment of elevated blood pressure should consist of weight loss, limitation of dietary salt, and increased physical activity (Very Low, Conditional).
Dyslipidemia:
Lipids should be measured yearly starting at diabetes onset (after optimal glycemic levels are achieved or within 3 months of diagnosis)
Initial screening for dyslipidemia does not require fasting
Goal levels for lipids are:
1. LDL-Cholesterol < 100 mg/dL
2. HDL- Cholesterol > 35 mg/dL
3. Triglycerides < 150 mg/dL (Intermediate, Strong).
Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) :
Evaluation for MASLD by measuring ALT and AST should be done at diagnosis of type 2 diabetes and annually thereafter (High, Strong).
Interpretation of ALT should be based upon sex-specific upper limits of normal in children (22 U/L for girls and 26 U/L for boys) and not individual laboratory upper limits of normal (High, Strong).
➡️If liver enzymes remain >3 times the upper limit of normal after 6 months refer to a pediatric gastroenterologist for consultation to exclude other causes of elevated liver enzymes, imaging and/or liver biopsy (Moderate, Strong)
Polycystic ovary syndrome
Female patients should be asked about menstrual irregularities, and symptoms of hyperandrogenism.
Patients with menstrual irregularities should be tested for hyperandrogenism (Very Low, Conditional).
Diagnosis of PCOS in adolescents should be based on a complete picture that includes clinical signs and symptoms of androgen excess, increased androgen levels, and exclusion of other causes of hyperandrogenemia in the setting of oligomenorrhea (See appendix). If suspected refer to endocrinologist (Intermediate, Strong).
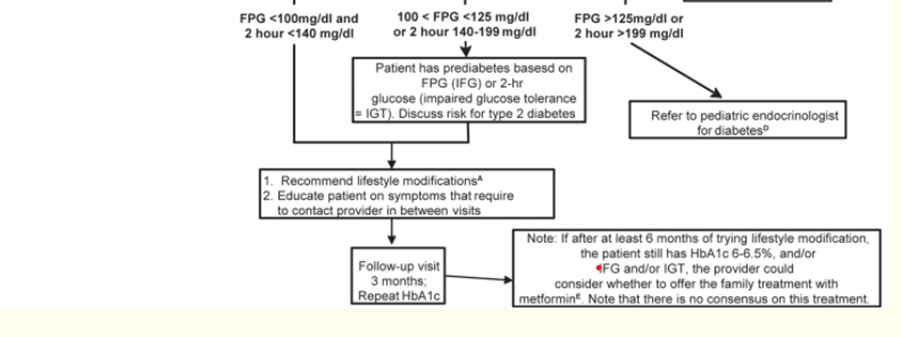
➡️Prevent progression of prediabetes to type 2 diabetes in adolescents in primary health care setting or general pediatric department:
Only lifestyle change, with decreased caloric intake and increased physical activity has been shown to be effective for adolescents with pre-diabetes (High, Strong).
Diet modification should focus on (Intermediate, Strong):
• Eliminating sugar-sweetened soft drinks and juices.
• Reducing the intake of foods made from refined, simple sugars and high fructose corn syrup.
• Limiting use of high-fat and/or high calorie dense foods.
Reducing the use of processed, prepackaged, and convenience foods (Very Low, Conditional):
• Portion control.
• Reducing meals eaten away from home.
• Increasing vegetable intake and limited use of fruit as a substitute for high-calorie and low nutrient foods.
• Changing staple foods from enriched white rice and white flour to brown rice and whole grain items with lower glycemic index to promote gradual and sustainable absorption with meals.
The Mediterranean diet which provides a carbohydrate content < 50% of daily energy and includes abundant use of poly- and mono-unsaturated fat (about 20%), mainly olive oil, was found to reverse dysglycemia (Good Practice Statement).
The use of metformin and/or insulin in youth with prediabetes is not currently recommended (High, Strong).
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((PREPARE-2022CN813)). Link: http://www.guidelines-registry.org/- Recommendations
|
Table 3. Recommendations |
|
|
|||
|
A. Management of Type 2 Diabetes |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A1 |
Who are the adolescents at risk that should be screened for prediabetes or type 2 diabetes in primary health care setting or general pediatric department or outpatient clinic? |
ISPAD 2024 |
Risk factors to develop type 2 diabetes: Consider testing to identify clinical cases of type 2 diabetes in children and adolescents after the onset of puberty or after 10 years of age, whichever occurs earlier, in those who have risk factors for diabetes, which include: Obesity (BMI > 95th percentile for age and sex), intrauterine growth retardation with rapid infant weight gain, first-degree family history of type 2 diabetes, maternal history of diabetes or gestational diabetes during child's gestation, high-risk ethnicity, polycystic ovary syndrome (PCOS). |
High |
Strong |
|
|
|
ISPAD 2024 |
Risk-based screening for prediabetes and/or type 2 diabetes should be considered after the onset of puberty or after 10 years of age, whichever occurs earlier in children and adolescents who are overweight (BMI > 85th percentile) or obese (BMI > 95th percentile) and who have one or more additional risk factors for diabetes (Refer to table (1) in implementation tools for details and level of evidence). |
High |
Strong |
|
|
|
ISPAD 2024 |
Assessment for dysglycemia in obese at-risk youth should occur in the setting of clinical assessment of other obesity-related comorbidities metabolic dysfunction-associated steatotic liver disease (MASLD), dyslipidemia, elevated blood pressure [BP], and polycystic ovary syndrome) that are more prevalent than dysglycaemia (Refer to Q 6) below).
|
High |
Strong |
|
A2 |
What are tests recommended for screening of prediabetes or type 2 diabetes in primary health care setting or general pediatric department or outpatient clinic? |
ISPAD 2022 |
Fasting plasma glucose, 2-h plasma glucose after 75-g OGTT, and A1C can be used to test for prediabetes or diabetes. |
Intermediate
|
Strong |
|
|
|
Diabetes Canada Clinical Practice Guidelines Expert Committee, Panagiotopoulos C, Hadjiyannakis S, Henderson M. Type 2 Diabetes in Children and Adolescents. Can J Diabetes. 2018 Apr;42 Suppl 1:S254. Doi: 10.1016/j.jcjd.2017.10.037.PMID:29650104.
|
Recommend using a combination of A1C and fasting or random blood glucose to screen for type 2 diabetes in children and youth with risk factors. “A 2-hour OGTT may be considered as an initial screening test in children and youth with 3 or more risk factors and should be done in those in whom there is a discrepancy between the A1C and fasting or random blood glucose results”. |
Good Practice Statement |
|
|
A3 |
What is considered a positive screening test result for prediabetes or type 2 diabetes in primary health care setting or general pediatric department? Sub-questions: -What is the cut off value for normal fasting blood glucose in adolescents? -What is the cut off value for normal postprandial glucose in adolescents? -What is the cut off value for normal HbA1c in adolescents? |
ISPAD 2022 |
Criteria for considering diagnosis of prediabetes according to ADA definitions: • Impaired fasting glycemia (IFG): FPG ≥100-125 mg/dL • Impaired Glucose tolerance (IGT): Post-challenge plasma glucose is ≥140-199 mg/dL • Hemoglobin A1c ˂5.7% normal, 5.7% to 6.4% prediabetes. (Should be laboratory-based, DCCT aligned, NGSP certified methodology).
|
High |
Strong |
|
|
|
ISPAD 2022 |
Criteria for diagnosis of type 2 diabetes: a) Symptoms of diabetes (polyuria, polydipsia, nocturia, and unexplained weight loss) and a random plasma glucose ≥200 mg/dL OR · Fasting blood glucose ≥126 mg/dL OR · 2-hour blood glucose concentration during an oral glucose tolerance test (OGTT) ≥200 mg/dL OR · Hemoglobin A1c (HbA1c) ≥6.5% (Should be laboratory-based, DCCT aligned, NGSP certified methodology). Point-of-care measurement of HbA1c is not acceptable for diagnosis.
|
High |
Strong |
|
|
|
ISPAD 2022 |
In the absence of symptoms, testing should be confirmed with a repeat test on a different day.
Measurement of insulin or C-peptide is NOT recommended as part of routine evaluation |
Intermediate
|
Strong |
|
A4 |
When to repeat screening for prediabetes or type 2 diabetes in adolescents if initial tests are normal in those at risk? |
ISPAD 2022 |
If tests are normal:
repeat testing at a minimum of 3-year intervals, or more frequently if BMI is increasing, the cardiometabolic risk profile is deteriorating, there is a strong family history of T2D, or evidence of pre-diabetes |
Low |
Conditional |
|
A5 |
When to consider referral to pediatric endocrinologist and diabetologist in adolescents with suspected prediabetes or type 2 diabetes in the primary health care setting or general pediatric department? |
ISPAD 2022 |
Referral to pediatric diabetologist/endocrinologist is recommended in children and adolescents suspected to have prediabetes or type 2 diabetes according to the American Diabetes Association (ADA) criteria. |
High |
Strong |
|
A6 |
When and how to screen for comorbidities of prediabetes and type 2 diabetes in adolescents at risk in the primary health care setting or general pediatric department or outpatient clinic? Sub-questions: a.When to consider elevated blood pressure? b.What are the normal values for lipids? c.What are the normal values for liver enzymes? d.How to diagnose PCOS? |
ISPAD 2022 |
Hypertension: Blood pressure should be monitored at every visit according to standardized techniques specific for children. Elevated BP should be confirmed on 2 additional separate days.
|
High |
Strong |
|
|
|
ISPAD 2022 |
Hypertension is defined as an average systolic or diastolic BP > 95th percentile for age, sex, and height, with high normal BP being 90th to <95th percentile. Initial treatment of elevated blood pressure should consist of weight loss, limitation of dietary salt, and increased physical activity. |
Very Low |
Conditional |
|
|
|
ISPAD 2022 |
Dyslipidemia: Initial screening for dyslipidemia does not require fasting Goal levels for lipids are: 1. LDL-Cholesterol < 100 mg/dL 2. HDL- Cholesterol > 35 mg/dL 3. Triglycerides < 150 mg/dL |
Intermediate
|
Strong |
|
|
|
ISPAD 2024 |
Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD Evaluation for MAFLD and non-alcoholic Steatohepatitis (NASH) by measuring ALT and AST should be done at diagnosis of type 2 diabetes and annually thereafter |
High |
Strong |
|
|
|
ISPAD 2022 |
Interpretation of ALT should be based upon sex-specific upper limits of normal in children (22 U/L for girls and 26 U/L for boys) and not individual laboratory upper limits of normal.
|
High |
Strong |
|
|
|
ISPAD 2024 |
If liver enzymes remain >3 times the upper limit of normal after 6 months refer to a pediatric gastroenterologist for consultation to exclude other causes of elevated liver enzymes, imaging and/or liver biopsy
|
Intermediate |
Strong |
|
|
|
ISPAD 2022 |
Polycystic ovary syndrome Female patients should be asked about menstrual irregularities, and symptoms of hyperandrogenism. Patients with menstrual irregularities should be tested for hyperandrogenism |
Very low |
Conditional |
|
|
|
ISPAD 2024
|
Diagnosis of PCOS in adolescents should be based on a complete picture that includes clinical signs and symptoms of androgen excess, increased androgen levels, and exclusion of other causes of hyperandrogenemia in the setting of oligomenorrhea (See appendix).
If suspected refer to endocrinologist.
|
Intermediate
|
Strong |
|
A 7 |
1) How to prevent progression of prediabetes to type 2 diabetes in adolescents in primary health care setting or general pediatric department? Sub-questions: a.What are the lifestyle and exercise measures in the primary health care setting or general pediatric department that can help prevent prediabetes development and its progression to type 2 diabetes in adolescent? b.What are the dietary counseling measures in the primary health care setting or general pediatric department that help to prevent prediabetes development and its progression to type 2 diabetes in adolescent? What are the pharmacologic interventions in the primary health care setting or general pediatric department that can help prevent prediabetes development and its progression to type 2 diabetes in children and adolescent? |
ISPAD 2022 |
Only lifestyle change, with decreased caloric intake and increased physical activity has been shown to be effective for adolescents with pre-diabetes. |
High |
Strong |
|
|
|
ISPAD 2022 |
Diet modification should focus on: · Eliminating sugar-sweetened soft drinks and juices. · Reducing the intake of foods made from refined, simple sugars and high fructose corn syrup. · Limiting use of high-fat and/or high calorie dense foods.
|
Intermediate
|
Strong |
|
|
|
|
Reducing the use of processed, prepackaged, and convenience foods. • Portion control. • Reducing meals eaten away from home. • Increasing vegetable intake and limited use of fruit as a substitute for high-calorie and low nutrient foods. • Changing staple foods from enriched white rice and white flour to brown rice and whole grain items with lower glycemic index to promote gradual and sustainable absorption with meals. |
Very Low |
Conditional |
|
|
|
Esposito K, et al. A Journey into a Mediterranean diet and type 2 diabetes: a systematic review with meta-analyses. BMJ Open 2015;5:e008222. doi:10.1136/bmjopen-2015-008222 |
The Mediterranean diet which provides a carbohydrate content < 50% of daily energy and includes abundant use of poly- and mono-unsaturated fat (about 20%), mainly olive oil, was found to reverse dysglycemia. |
Good Practise Statement |
|
|
|
|
ISPAD 2022 |
The use of metformin and/or insulin in youth with prediabetes is not currently recommended. |
High |
Strong |


➡️Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
➡️Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
➡️Steps of implementing Type 2 Diabetes prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
➡️Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physician for Blood pressure and Body mass index screening.
2. Algorithm for Type 2 diabetes screening.
References:
Samuels J, Samuel J. New guidelines for hypertension in children and adolescents. J Clin Hypertens (Greenwich). 2018;20(5):837-839. doi:10.1111/jch.13285
Flynn JT, Kaelber DC, Baker‐Smith CM, et al. Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140:e20171904.
National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents . The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics. 2004;114:555‐576.
Magge SN, Silverstein J, Elder D, Nadeau K, Hannon TS. Evaluation and Treatment of Prediabetes in Youth. J Pediatr. 2020;219:11-22. doi:10.1016/j.jpeds.2019.12.061

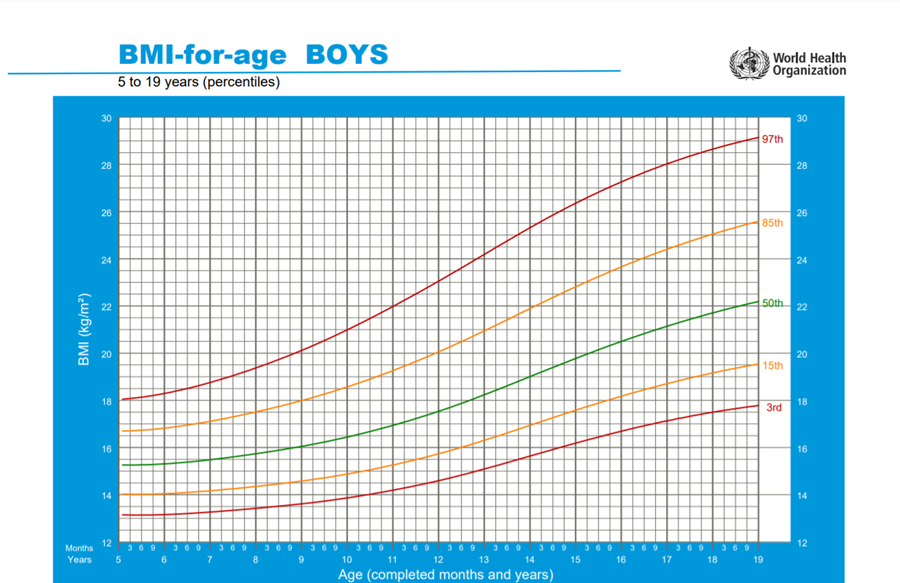
https://www.who.int/tools/growth-reference-data-for-5to19-years/indicators/bmi-for-age

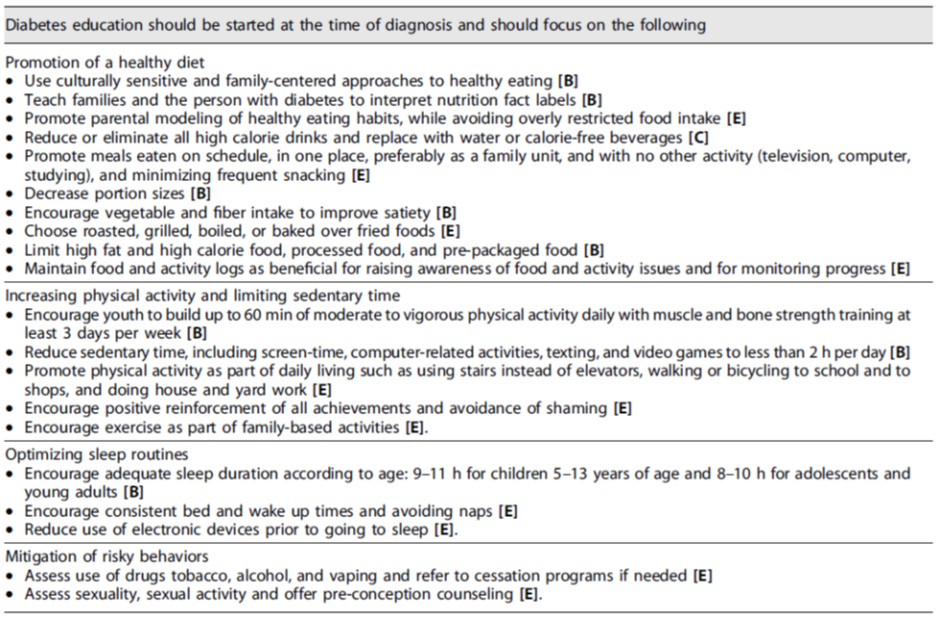
Lifestyle recommendations for youth with T2D
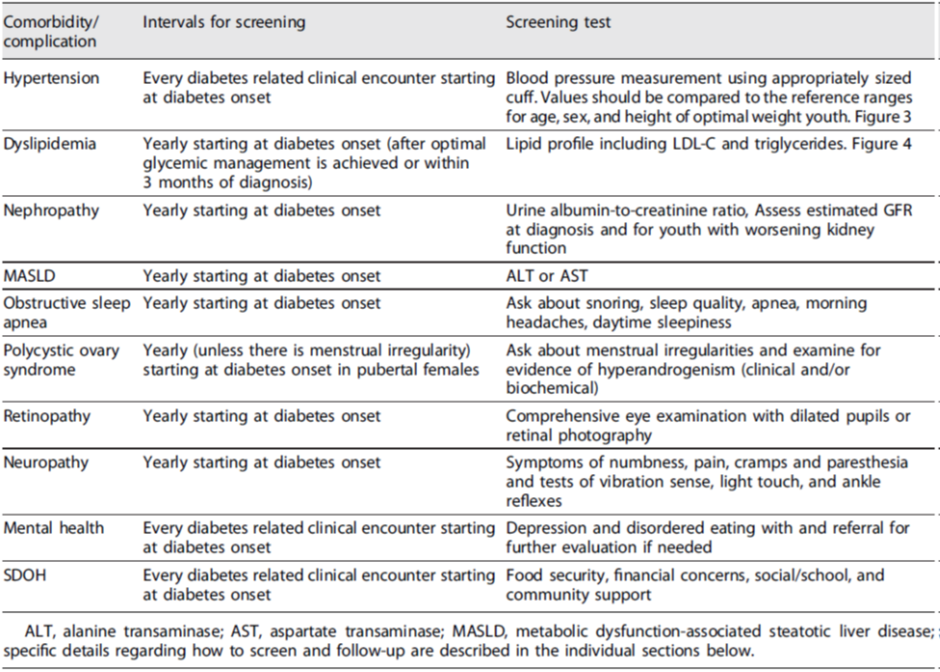
Recommendations for screening of associated comorbidities/complications
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Professor Amany Kamal El Hawary
|
Professor of pediatrics, Director of Pediatric Endocrinology and Diabetes unit, Mansoura University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Amina M. Abdel Wahab |
Professor of pediatrics, Pediatric Endocrinology and Diabetes, Suez Canal University. |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Ashraf A. Elsharkawy |
Professor of pediatrics, Pediatric Endocrinology and Diabetes unit, Mansoura University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Basma Abdel Moez |
Professor of Pediatrics, Head of Pediatric Endocrinology, El Menya University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Hanaa Abdel Latif |
Professor of pediatrics, Head of Pediatric Endocrinology and Diabetes unit, Assiut University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Lubna Fawaz
|
Professor of pediatrics, Former director of the pediatric diabetes, endocrine and metabolism unit, Cairo University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Mona Mamdouh Hassan
|
Professor of pediatrics, Head of the Diabetes, Endocrine and Metabolism Pediatric Unit, Cairo University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Nora El Sayed Badawi |
Professor of Pediatrics, Diabetes, Endocrine and Metabolism Pediatric Unit, Cairo University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Randa M Matter
|
Professor of pediatrics, Pediatric and Adolescence Diabetes Unit, Ain-Shams University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Safinaz El Habashy
|
Professor of pediatrics, Former Head of the Pediatric and Adolescence Diabetes Unit, Ain-Shams University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Wiam Al Farouk Younis
|
Professor of pediatrics, Pediatric Diabetes and Endocrine unit, AFCM |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Assistant Professor Shaymaa Elsayed Abdel Meguid |
Assistant professor of Pediatrics, Pediatric Endocrinology and Diabetology Unit, Faculty of Medicine, Alexandria University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Hanan Hassan Aly
|
Assistant professor of pediatrics, Pediatric and Adolescence Diabetes Unit, Ain-Shams University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Mona Karem Amin
|
Assistant professor of pediatrics, Pediatric Diabetes and Endocrine unit, Suez canal University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Nouran Y Salah El-Din
|
Assistant professor of pediatrics, Pediatric and adolescence diabetes unit, Ain-shams University, AFCM |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Ramy Saleh Morsy
|
Lecturer of pediatrics, Pediatric diabetes and endocrine unit, AFCM |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Remon Magdy |
Assistant professor of pediatrics, Pediatric diabetes and endocrinology unit, Fayoum University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Ghada Mohammad Anwar |
Pediatrics Department /Cairo University |
||||
|
Prof. Mona Hafez |
Pediatrics Department / Cairo University, Egypt
|
||||
|
Prof. Mona Salem |
Pediatrics Department, Ain Shams University/Egypt
|
||||
|
Prof. Nermin Salah |
Pediatrics Department / Cairo University, Egypt
|
||||
|
Prof.Hesham El Hefnawy |
Prof. of diabetes and endocrinology, former dean of the National Institute of Diabetes and Endocrinology, Cairo, Egypt. Head of National Committee of non-communicable disease |
||||
|
International Peer Reviewers |
|||||
|
|
|
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge ISPAD 2022 guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
- Funding
This work is not
related to any pharmaceutical or industrial company. The members of the GDG/
GAG and their institutes and universities volunteered their participation and
contributions.
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
BMI |
Body mass index |
||
|
BP |
Blood Pressure |
||
|
CPG |
Clinical Practice Guideline |
||
|
DHS |
Demographic and Health Survey |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ERG |
External Review Group |
||
|
HCP |
Healthcare Professionals |
||
|
IFG |
Impaired fasting glycemia |
||
|
IGT |
Impaired glucose tolerance |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
MASLD |
metabolic dysfunction-associated steatotic liver disease |
||
|
OGTT |
Oral glucose tolerance test |
||
|
PCOS |
Polycystic Ovary Syndrome |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
RBS |
Random blood sugar |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
||
|
T2D |
Type 2 diabetes |
- Glossary
Admission
Admission, for the purpose of this guideline, refers to a child being registered and entering inpatient care as a patient. This is distinguished from the term “enrolment”, which is used for outpatient care.
- Introduction
➡️Definition of the disease condition
Diabetes mellitus is a complex metabolic disorder characterized by chronic hyperglycemia resulting from defects in insulin secretion, insulin action, or both. Inadequate insulin secretion and/or diminished tissue responses to insulin in the complex pathways of hormone action result in deficient insulin action on target tissues, which leads to abnormalities of carbohydrate, fat, and protein metabolism. The etiology of diabetes is heterogeneous, however, most cases can be classified into two broad etiopathogenetic categories type 1 diabetes (T1D), which is characterized primarily by deficiency of insulin secretion; or type 2 diabetes (T2D), which results from a combination of resistance to insulin action, as well as an inadequate compensatory insulin secretory response for the degree of insulin resistance, due to progressive non-autoimmune β-cell failure (1,2). It was formerly known as adult onset diabetes or non-insulin dependent diabetes (2).
➡️Magnitude of the problem worldwide.
While T1D remains the most common form of diabetes in children and adolescents worldwide, T2D is becoming more prevalent and imposes an increasingly important public health burden globally (3). It is stated that T2D in youth has become widespread in many regions of the world; and its frequency in the pediatric age range is increasing (4,5).
In many countries, the incidence of T2DM in adolescents continues to increase. Similarly, the prevalence of prediabetes, defined in adults as a state of high-risk for progression to diabetes, is increasing quickly in some developing countries with the increase of overweight and obesity (6).
Worldwide incidence and prevalence of T2D in children and adolescents vary substantially among countries, age categories and ethnic groups (3). Epidemiologic studies have shown variations in the incidence of T2D in children and adolescents ranging from 1 to 51 per 1000 (7). The highest reported was in North American Indian adolescents with a prevalence of 50.9 per 1000. The Center for Disease Control and Prevention (CDC) and the National Institutes of Health (NIH) reported increasing prevalence of pediatric T2D in the United States by 35% between 2001 and 2009 with an incidence rate of 5,000 new cases per year (8). Similar trends have been reported in many European countries, although there is a significantly lower incidence rate. Candler et al. (9) showed an incidence of T2D of 0.72/100,000 per year in British children and adolescents (9). Similarly, Schober et al. described the incidence of T2D at 0.29 cases per 100,000 per year between 1999 and 2007 in Austrian children and adolescents (10). In Korea the average incidence rate of T2D in children and adolescents was 0.75/100,000 (11). In China, they reported a T2D prevalence ranging from 2.52/100,000 to 15.64/100,000 (12).
The difference in the incidence and prevalence rates of T2D in children might be attributed to genetics and hormonal factors, environmental influences, different quality of life, and access to health care.
It has been reported that T2D disproportionately involves youth of ethnic and racial minorities, demonstrating substantial differences in insulin secretion and sensitivity that might increase the risk of T2D. It has been postulated that the complexity of psychosocial and cultural environments might influence the onset of T2D in ethnic and racial minorities, where it may be difficult to achieve healthy lifestyle modifications and self-management actions (1).
➡️Magnitude of the problem in Egypt with impact on health and economy
In Egypt, T2D is presenting a major public health care burden with a total cost of 25.2 billion EGP equivalent to 3.5 billion USD (13). According to the IDF, the annual prevalence increment rate of T2D in Egypt is 266%. Moreover, Egypt is thought to jump from the 9th to the 7th rank among the countries with highest prevalence of T2D worldwide by 2030 with a prevalence of 12.4 millions (14). Data about the prevalence of T2D in children and adolescents are sparse. Studies showed increasing prevalence rates varying from 0.01% in 2002 (15) to 0.3% recently in 2018 (16).
➡️Role of screening and early detection in ameliorating this impact.
T2D is a complex and multifactorial disease, characterized by an association between genetic, epigenetic, and environmental factors (17). The pathophysiology of T2D in children is more complex than that in adults. Beta-cell dysfunction occurs more rapidly in children than in adults (18).
Primary prevention approaches aiming at addressing the modifiable risk factors, starting from the intra-uterine period and extending throughout childhood, are of utmost importance in preventing T2DM. Despite the possibility of detecting the genetic susceptibility to T2DM by documenting compromised insulin secretion and insulin sensitivity in otherwise healthy children with a positive family history, it is not cost effective. Since the diagnosis of T2DM in youth is rare before puberty, it is detected more frequently during puberty when physiologic and transient insulin resistance (IR) occurs (insulin sensitivity declines by 25%–30% (19).
Maternal overweight and obesity were found to be associated with T2DM in offspring, irrespective of various confounding pre-existing or pregnancy-related conditions (20). Children born to obese or overweight women were found to have a 3.5- and 1.4-fold higher incidence of T2DM respectively, compared to those born to normal-weight women (21). Hence, one of the earliest points of childhood T2DM prevention is the reduction of maternal overweight and obese status during pregnancy. In addition, studies have documented that offspring of women with gestational diabetes (GDM) are at increased risk of T2DM and obesity, irrespective of the mother’s weight status during pregnancy (22). Not only overt GDM but also prediabetic conditions during pregnancy have been linked to glucose abnormalities and insulin resistance in the offspring (23). Protection of the fetus from intrauterine glycemic derangements is, therefore, of paramount importance to prevent prediabetic conditions and T2DM in childhood and adolescence. Other risk factors such as maternal diet, maternal smoking, and paternal obesity should also be considered (24-26).
In order to prevent childhood T2DM, one should clearly focus on intrauterine life, but postnatal exposures seem to play an equally important role. Childhood obesity is among the major modifiable risk factors of T2DM in children. Increasing BMI has been reported as a risk factor of glycemic deterioration and progression to T2DM (27).
The importance of programming in T2DM pathogenesis is growing. Dietary and environmental factors during the first 1000 days from conception to the second birthday have utmost importance in T2DM prevention (28). Breastfeeding was found to have a strong protective effect against childhood T2DM especially among high risk populations (29, 30). Moreover, both high and low birth weight, preterm birth, as well as rapid weight gain during the first months of life have been shown to increase the risk of childhood obesity, insulin resistance and glucose metabolism disturbances, predisposing to T2DM (31-33).
In an earlier review in 2007 (34) including reports on acute and long-term comorbidities associated with type 2 diabetes in young people, microvascular complications have been identified at diagnosis, implying long-term, unrecognized hyperglycemia. Children may also present with acute decompensation in diabetic ketoacidosis (DKA) and/or hyperosmolar hyperglycemic state (HHS). This argues for a systematic screening program in children at high risk for type 2 diabetes in order to prevent an acute, life-threatening presentation and to decrease the development of chronic complications. Although not proven in children, it is generally assumed that earlier diagnosis of diabetes will lead to interventions that will improve glycemic control and reduce the related short- and long-term complications (34).
The optimal approach to screening and diagnosis of prediabetes and diabetes in youth is uncertain (35). In a recent cross-sectional analysis conducted by Wallace et al (35) including 14,119 youth aged 10 to 19 years in the 1999–2016 NHANES; it was found that HbA1c is a specific and useful non fasting test to identify high-risk youth who could benefit from lifestyle interventions to prevent diabetes and cardiovascular risk in adulthood. Sensitivity and specificity of the screening criteria for detecting any hyperglycemia were low for both HbA1c ≥ 5.7% (sensitivity = 55.5%, specificity = 76.3%) and FPG ≥ 100 mg/dL (sensitivity = 35.8%, specificity = 77.1%). Confirmed undiagnosed diabetes (HbA1c ≥ 6.5% and FPG ≥ 126 mg/dL) was rare, 0.5% of youth. Associations with cardiometabolic risk were consistently stronger and more specific for HbA1c-defined hyperglycemia (specificity = 98.6%; sensitivity = 4.0%) than FPG defined hyperglycemia (specificity = 90.1%; sensitivity = 19.4%).
For better sensitivity, a combination of A1C and fasting or random blood glucose is recommended to screen for type 2 diabetes in children and youth with risk factors; with a 2-hour OGTT to be considered as an initial screening test in children and youth with 3 or more risk factors. A 2-hour OGTT was also recommended in those who show a discrepancy between the A1C and fasting or random blood glucose results on screening (36).
Recent analyses of data from US adolescents estimate prediabetes to be present in 4–23% of adolescents, depending on criteria used, with other studies finding an 8% risk of progression from prediabetes to T2DM over a 3-year period. These data support the importance of intervention to avoid long-term sequelae, focusing on reducing degree of obesity and insulin resistance (37).
➡️Methods of prevention of progression and possible reversal of the disease condition
Healthy diet and physical activity are important in preventing childhood obesity. This is especially important in the preschool period. Rapid weight gain in preschool children was found to be associated with a high risk of overweight or obesity in adolescence, increasing the risk of childhood T2DM (38). Most programs aiming at preventing childhood T2DM are based on obesity prevention, given the etiological connection between increased childhood obesity, metabolic syndrome and T2DM. These programs mostly target dietary interventions like providing healthier school meals, reducing simple sugars, and restricting unhealthy food advertisements aimed at children. Nutritional interventions should be combined with programs targeting increased physical activity and restricting sedentary life and screen time in order to achieve the best long-term outcome (39).
Regarding adolescents, school-based interventions have proven more effective when the adolescents were addressed directly (40).
Because of the rarity and the relatively recent emergence of the problem in children and adolescents, there has been limited evidence leading to unique challenges in the diagnosis, management, and monitoring of this vulnerable population. This limited evidence base is further complicated by differences in the characteristics and presentation of the disorder and approaches to treatment in developed and developing countries. Hence we aim to provide guidelines for screening and prevention of childhood T2D in Egypt.
- Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the screening of prediabetes and type 2 diabetes in children and adolescents. It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to guide the HCPs on:
1) Screening for early detection of prediabetes allowing the opportunity to prevent or delay progression to type 2 diabetes and its comorbidities.
2) Early detection of type 2 diabetes in high-risk groups, to prevent or delay serious diabetes complications during adolescence and early adulthood.
3) Screening for early diagnosis of comorbidities or complications in adolescents with prediabetes and type 2 diabetes to prevent serious disabilities during adolescence and early adulthood.
This version of the guideline includes recommendations and good practice statements for screening of prediabetes and type 2 diabetes in children and adolescents.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: prediabetes, type 2 diabetes, children, adolescents, OGTT.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria the GDG/ GAG recommended using 2 guidelines:
1- ISPAD Guidelines (2024)
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
➡️Contributors to the guideline development process:
➡️Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of Type 2 Diabetes.
The main functions of the clinical panel were adolopment of ISPAD 2024 Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinician’s subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as eminent international reviewers in Type 2 diabetes in children.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of
the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations (although this was not done in this guideline).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
➡️GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2.Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include awell-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Limitations and suggestions for further research needs
Future research recommendations for the screening of Type 2 Diabetes in children in the Egyptian context could include:
· The accuracy of the HBA1c, OGTT, Fasting blood glucose in large scale screening.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention strategies for Type 2 Diabetes in children.
➡️Challenges
· Low specificity and sensitivity of HbA1c
· Cost of the laboratory procedures
· Lack of patient’s compliance to follow up
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for Type 2 Diabetes. in children:
1. Adherence to screening program of T2D:
· Numerator: Number of children diagnosed with prediabetes and T2D
· Denominator: Total number of children diagnosed with T2D
· Data Source: Hospital or clinic patient records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG Type 2 Diabetes GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
- American Diabetes Association. Classification and Diagnosis of Diabetes: <i>Standards of Medical Care in Diabetes-2018. Diabetes Care. 2018 Jan; 41(Suppl 1):S13-S27.
- Weber D.R. and Jospe N. Diabetes Mellitus. Classification of Diabetes Mellitus. In: Nelson textbook of pediatrics (21st ed.) Kleigman R. and St Geme J.(editors), 2021: p.11814-11822.
- Zeitler, P, Arslanian, S, Fu, J, et al (2018). ISPAD Clinical Practice Consensus Guidelines 2018: Type 2 diabetes mellitus in youth. Pediatr Diabetes. 19(27): 28– 46.
- 3- Shah, A.S., Barrientos-Pérez, M., Chang, N., Fu, J.F., Hannon, T.S., Kelsey, M., Peña, A.S., Pinhas-Hamiel, O., Urakami, T., Wicklow, B. and Wong, J., 2024. ISPAD Clinical Practice Consensus Guidelines 2024: Type 2 diabetes in children and adolescents. Hormone Research in Paediatrics, pp.1-41.
- Mayer-Davis EJ, Lawrence JM, Dabelea D, et al. Incidence trends of type 1 and type 2 diabetes among youths, 2002-2012. N Engl J Med. 2017;376(15):1419-1429.
- Ingelfinger JR, Jarcho JA. Increase in the incidence of diabetes and its implications. N Engl J Med. 2017;376(15):1473-1474 REF 3
- Magge SN, Silverstein J, Elder D, Nadeau K and Hannon TS. Evaluation and treatment of prediabetes in youth. The Journal of Pediarics. 2020. https://www.jpeds.com/article/S0022-3476(19)31740-8/fulltext
- Pulgaron ER, Delamater AM. Obesity and type 2 diabetes in children: epidemiology and treatment. Curr Diab Rep. 2014 Aug; 14(8):508.
- Lawrence JM, Imperatore G, Dabelea D, Mayer-Davis EJ, Linder B, Saydah S, et al. Trends in incidence of type 1 diabetes among non-Hispanic white youth in the U.S., 2002–2009. Diabetes 2014;63:3938-45. 9.
- Candler TP, Mahmoud O, Lynn RM, Majbar AA, Barrett TG, Shield JP. Continuing rise of type 2 diabetes incidence in children and young people in the UK. Diabet Med 2018;35:737-44.
- Schober E, Waldhoer T, Rami B, Hofer S. Austrian Diabetes Incidence Study Group: incidence and time trend of type 1 and type 2 diabetes in Austrian children 1999-2007. J Pediatr 2009;155:190-3.
- Lee JH, Kim YM, Kwak MJ, Kim SY, Kim HJ, Cheon CK. Incidence trends and associated factors of diabetes mellitus in Korean children and adolescents: a retrospective cohort study in Busan and Gyeongnam. Ann Pediatr Endocrinol Metab 2015;20:206-12.
- Ye Q, Fu JF. Paediatric type 2 diabetes in China-Pandemic, progression, and potential solutions. Pediatr Diabetes 2018;19:27-35.
- S Assaad-Khalil,IN Elebrashy,Y Afify,B Abdelmordy,W Zakaria,R Aboushady,S Zanaty,E Basiouny,A Ibrahim,R Sallam,I Anan. PDB20 The Financial Burden of Diabetes Mellitus Type 1 And Type 2 In Egypt. Value in Health. 2017
- IDF Diabetes Atlas: Global estimates of the prevalence of diabetes for 2011 and 2030. Whiting, David R. et al. Diabetes Research and Clinical Practice, Volume 94, Issue 3, 311 – 321
- Salem M, El Samahy M, Mounir E, Tash F, Zaki M, Farid S (2002). Prevalence of obesity in Egyptian school children and its link to type 2 diabetes mellitus. Egyptian journal of pediatrics. 19(3): 397-416.
- Hafez M, Musa N, Mansour M, Hamdy H (2018). Screening for T2D in High Risk Egyptian Children and Adolescents Using Strip HbA1c and OGTT. Hormone Research in Paediatrics Volume 82 Supplement1.
- Todd JN, Srinivasan S, Pollin TI. Advances in the genetics of youth-onset type 2 diabetes. Curr Diab Rep 2018;18:57
- Arslanian S, Bacha F, Grey M, Marcus MD, White NH, Zeitler P. Evaluation and management of youth-onset type 2 diabetes: a position statement by the American Diabetes Association. Diabetes Care 2018;41:2648-68. 22.
- Castorani V, Polidori N, Giannini C, Blasetti A, Chiarelli F (2020). Insulin resistance and type 2 diabetes in children. Annals of Pediatric Endocrinology & Metabolism. 25. 217-226. 10.6065/apem.2040090.045.
- Lahti-Pulkkinen M, Bhattacharya S, Wild SH, Lindsay RS, Räikkönen K, Norman JE, Reynolds RM. Consequences of being overweight or obese during pregnancy on diabetes in the offspring: a record linkage study in Aberdeen, Scotland. Diabetologia. 2019;62:1412-1419.
- Chernausek SD, Arslanian S, Caprio S, Copeland KC, El ghormli L, Kelsey MM, Koontz MB, Orsi CM, Wilfley D. Relationship Between Parental Diabetes and Presentation of Metabolic and Glycemic Function in Youth With Type 2 Diabetes: Baseline Findings From the TODAY Trial. Diabetes Care. 2016 Jan; 39(1):110-7.
- Dabelea D, Mayer-Davis EJ, Lamichhane AP, D'Agostino RB Jr, Liese AD, Vehik KS, Narayan KM, Zeitler P, Hamman RF. Association of intrauterine exposure to maternal diabetes and obesity with type 2 diabetes in youth: the SEARCH Case-Control Study. Diabetes Care. 2008;31:1422-1426.
- Scholtens DM, Kuang A, Lowe LP, Hamilton J, Lawrence JM, Lebenthal Y, Brickman WJ, Clayton P, Ma RC, McCance D, Tam WH, Catalano PM, Linder B, Dyer AR, Lowe WL Jr, Metzger BE; HAPO Follow-up Study Cooperative Research Group; HAPO Follow-Up Study Cooperative Research Group. Hyperglycemia and Adverse Pregnancy Outcome Follow-up Study (HAPO FUS): Maternal Glycemia and Childhood Glucose Metabolism. Diabetes Care. 2019;42:381-392.
- Chen LW, Aris IM, Bernard JY, Tint MT, Chia A, Colega M, Gluckman PD, Shek LP, Saw SM, Chong YS, Yap F, Godfrey KM, van Dam RM, Chong MF, Lee YS. Associations of Maternal Dietary Patterns during Pregnancy with Offspring Adiposity from Birth Until 54 Months of Age Nutrients. 2016;9.
- Ino T. Maternal smoking during pregnancy and offspring obesity: meta-analysis. Pediatr Int. 2010;52:94-99.
- Sharp GC, Lawlor DA. Paternal impact on the life course development of obesity and type 2 diabetes in the offspring. Diabetologia. 2019;62:1802-
- Serbis A, Giapros V, Kotanidou EP, Galli-Tsinopoulou A, Siomou E. Diagnosis, treatment and prevention of type 2 diabetes mellitus in children and adolescents. World J Diabetes 2021; 12(4): 344-365 [PMID: 33889284 DOI: 10.4239/wjd.v12.i4.344]
- Schwarzenberg SJ, Georgieff MK; COMMITTEE ON NUTRITION. Advocacy for Improving Nutrition in the First 1000 Days to Support Childhood Development and Adult Health. Pediatrics. 2018;141.
- Horta BL, de Lima NP. Breastfeeding and Type 2 Diabetes: Systematic Review and Meta-Analysis. Curr Diab Rep. 2019;19:1.
- Vandyousefi S, Goran MI, Gunderson EP, Khazaee E, Landry MJ, Ghaddar R, Asigbee FM, Davis JN. Association of breastfeeding and gestational diabetes mellitus with the prevalence of prediabetes and the metabolic syndrome in offspring of Hispanic mothers. Pediatr Obes. 2019;14:e12515.
- Wang G, Divall S, Radovick S, Paige D, Ning Y, Chen Z, Ji Y, Hong X, Walker SO, Caruso D, Pearson C, Wang MC, Zuckerman B, Cheng TL, Wang X. Preterm birth and random plasma insulin levels at birth and in early childhood. JAMA. 2014;311:587-596.
- Taveras EM, Rifas-Shiman SL, Belfort MB, Kleinman KP, Oken E, Gillman MW. Weight status in the first 6 mo of life and obesity at 3 years of age. Pediatrics. 2009;123:1177-1183.
- Fabricius-Bjerre S, Jensen RB, Færch K, Larsen T, Mølgaard C, Michaelsen KF, Vaag A, Greisen G. Impact of birth weight and early infant weight gain on insulin resistance and associated cardiovascular risk factors in adolescence. PLoS One. 2011;6:e20595.
- Pinhas-Hamiel O, Zeitler P. Acute and chronic complications of type 2 diabetes mellitus in children and adolescents. Lancet 2007;369:1823–31.
- Wallace AS, Wang D, Shin J, et al. Screening and Diagnosis of Prediabetes and Diabetes in US Children and Adolescents. Pediatrics. 2020;146(3):e20200265
- Panagiotopoulos C, Hadjiyannakis S, Henderson M. Diabetes Canada 2018 Clinical Practice Guidelines for the Prevention and Management of Diabetes in Canada: Type 2 Diabetes in Children and Adolescents. Can J Diabetes 2018;42(Suppl 1):S88-S103.
- Esquivel Zuniga R, DeBoer MD. Prediabetes in Adolescents: Prevalence, Management and Diabetes Prevention Strategies. Diabetes Metab Syndr Obes. 2021;14:4609-4619. Published 2021 Nov 25. doi:10.2147/DMSO.S284401
- Geserick M, Vogel M, Gausche R, Lipek T, Spielau U, Keller E, Pfäffle R, Kiess W, Körner A. Acceleration of BMI in Early Childhood and Risk of Sustained Obesity. N Engl J Med. 2018;379:1303-1312.
- Sobol-Goldberg S, Rabinowitz J, Gross R. School-based obesity prevention programs: a meta-analysis of randomized controlled trials. Obesity (Silver Spring). 2013;21:2422-2428.
- Haynos AF, O'Donohue WT. Universal childhood and adolescent obesity prevention programs: review and critical analysis. Clin Psychol Rev. 2012;32:383-399.
42. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
43. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
44. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
45. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
46. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
47. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
48. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
49. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Professor Amany Kamal El Hawary
|
Professor of pediatrics, Director of Pediatric Endocrinology and Diabetes unit, Mansoura University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Amina M. Abdel Wahab |
Professor of pediatrics, Pediatric Endocrinology and Diabetes, Suez Canal University. |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Ashraf A. Elsharkawy |
Professor of pediatrics, Pediatric Endocrinology and Diabetes unit, Mansoura University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Basma Abdel Moez |
Professor of Pediatrics, Head of Pediatric Endocrinology, El Menya University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Hanaa Abdel Latif |
Professor of pediatrics, Head of Pediatric Endocrinology and Diabetes unit, Assiut University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Lubna Fawaz
|
Professor of pediatrics, Former director of the pediatric diabetes, endocrine and metabolism unit, Cairo University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Mona Mamdouh Hassan
|
Professor of pediatrics, Head of the Diabetes, Endocrine and Metabolism Pediatric Unit, Cairo University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Nora El Sayed Badawi |
Professor of Pediatrics, Diabetes, Endocrine and Metabolism Pediatric Unit, Cairo University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Randa M Matter
|
Professor of pediatrics, Pediatric and Adolescence Diabetes Unit, Ain-Shams University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Safinaz El Habashy
|
Professor of pediatrics, Former Head of the Pediatric and Adolescence Diabetes Unit, Ain-Shams University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Professor Wiam Al Farouk Younis
|
Professor of pediatrics, Pediatric Diabetes and Endocrine unit, AFCM |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Assistant Professor Shaymaa Elsayed Abdel Meguid |
Assistant professor of Pediatrics, Pediatric Endocrinology and Diabetology Unit, Faculty of Medicine, Alexandria University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Hanan Hassan Aly
|
Assistant professor of pediatrics, Pediatric and Adolescence Diabetes Unit, Ain-Shams University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Mona Karem Amin
|
Assistant professor of pediatrics, Pediatric Diabetes and Endocrine unit, Suez canal University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Nouran Y Salah El-Din
|
Assistant professor of pediatrics, Pediatric and adolescence diabetes unit, Ain-shams University, AFCM |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Ramy Saleh Morsy
|
Lecturer of pediatrics, Pediatric diabetes and endocrine unit, AFCM |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
Dr. Remon Magdy |
Assistant professor of pediatrics, Pediatric diabetes and endocrinology unit, Fayoum University |
Author contributed in formulating the following recommendations, PIPOH, implementation tools, health question, reviewing the literature, discussion and adaptation |
|||
|
|
|
|
|||
|
|
|
|
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Ghada Mohammad Anwar |
Prof. of pediatric diabetes and endocrinology/ Cairo University |
||||
|
|
|
||||
|
|
|
||||
|
International Peer Reviewers |
|||||
|
|
|
||||
|
|
|
||||
|
|
|
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the prevention of. " on PubMed are: screening, T2 D, prediabetes, children, adolescents. OGTT
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for ISPAD 2022
|
AGREE II/ CPGs |
ISPAD 2022 |
|
Domain 1 (Scope) |
% 89 |
|
Domain 2 (Stakeholder) |
% 75 |
|
Domain 3 (Rigour) |
% 59 |
|
Domain 4 (Clarity) |
% 97 |
|
Domain 5 (Applicability) |
% 94 |
|
Domain 6 (Independence) |
% 73 |
|
Overall assessment . |
% |
|
Recommend for use (Overall assessment .) |
YES |
➡️Annex Table 3.
Annex Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|