Diabetic Ketoacidosis
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Diabetic Ketoacidosis |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 26 March 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Funding
- - Glossary
- - Introduction
- - Methods
- - Evidence to recommendations: Considerations
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline.
- - References
- - Annexes
- Executive Summary
➡️Introduction
A practical guideline for the management of diabetic ketoacidosis in children and adolescents has been adapted to fit the Egyptian healthcare system. This process of customizing existing evidence-based clinical practice guidelines for local contexts offers a practical alternative to creating new ones from scratch, potentially enhancing their usefulness while conserving resources. This guideline aims to provide practical guidance for the diagnosis, treatment, and prevention of diabetic ketoacidosis in children and adolescents in Egypt, as well as the adaptation methods employed to create Egypt's first National Guideline for the management of diabetic ketoacidosis in children and adolescents using the Adapted ADAPTE method. The entire adaptation process, encompassing the setup, adaptation, and finalization phases, is thoroughly described. This involved a guideline adaptation group (GAG) and an external review group by experts in clinical content.
The finalized adapted CPG provides pediatricians and healthcare workers in the field of diabetes and endocrinology in Egypt with practical, evidence-based guidance for the management of diabetic ketoacidosis in children and adolescents. This initiative underscores the effectiveness of the Adapted ADAPTE method and emphasizes the significance of collaboration between clinical and methodological experts in adapting national guidelines.
➡️Scope
This guideline focuses on diagnosis and management of DKA in pediatric age group.
➡️Guidelines development and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- The International Society for Pediatric and Adolescent Diabetes (ISPAD) Clinical Practice Consensus Guidelines 2018 and 2022
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Adaptation for 2 recommendations according to GRADE criteria to be suitable to our Economic implications (Evidence-to-Decision (EtD) table was done)
- Development of Good Practice Statements
➡️Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. Diagnosis of DKA
The adapted CPG targets children aged 1 to 18 years presenting with diabetic ketoacidosis and is intended for use by various healthcare providers in the field namely paediatricians, diabetologists or endocrinologists, and intensivists.
B. Management of DKA
This section includes recommendations and good practice statements on management of DKA starting from initial resuscitation therapy, using IV insulin and when to wean off to SC insulin and when an ICU admission is indicated.
C. Prevention of DKA complications such as cerebral oedema and HHS
We can summarize the guidelines’ recommendations for DKA in the following:
We suggest in initial assessment of a patient with DKA to do the following (Very low, Conditional):
In Initial DKA Patient Assessment:
· Obtain vital signs and weight of the patient.
· Note that despite severe dehydration, hypertension occurs in 12% of children with DKA. Such patients require volume replacement despite the hypertension and should be monitored particularly carefully for signs and symptoms of impending cerebral injury.
· Insert two wide bore peripheral cannulas.
· Do Immediate measurement of: blood glucose, blood or urine ketone, venous blood gases, serum electrolytes, blood urea nitrogen and s-creatinine, complete blood count and C-reactive protein (CRP).
· Connect the patient to an ECG monitor and check T waves.
· Assess conscious level: Glasgow coma scale (GCS) assessment (table 4). Examine pupillary size and reflexes
· Obtain appropriate specimens for cultures if there is evidence of infection e.g. fever.
· Obtain history looking for the underlying cause of DKA: In newly diagnosed it is mainly delay in diagnosis. In known diabetics look for missed insulin dose (especially basal insulin) or infection or marked insulin deficiency in children who reached puberty but their basal insulin dose was not adjusted.
➡️Dehydration Severity Assessment in DKA Patient (Very low, Conditional):
· Assess the severity of dehydration (table 1) by:
A- Pulse rate and volume (weak rapid pulse in shock).
B- Capillary refill time (normal capillary refill is ≤ 2 seconds).
C- Skin turgor ('tenting' or inelastic skin) or other signs of dehydration.
D- Patient temperature and temperature of periphery (cold hands and feet indicate poor tissue perfusion and possible shock, hypothermia may also occur in shock).
E- Urine output (ml/hour).
F- Blood pressure. Hypotension is a late sign in shock (blood pressure is maintained for a long time by sympathetic tone, stress hormones and increased osmotic pressure from marked hyperglycemia) (Low, Conditional).
G- Conscious level (reduced in shock and is not alone indicative of brain edema) (Very low, Conditional).
➡️Calculating Anion Gap, Corrected Sodium and Osmolarity (Very low, Conditional):
· Calculate the following in the DKA patient:
o Anion gap = Na – (Cl + HCO3) (Normal = 12 ± 2 mmol/L)
In DKA the anion gap is typically 20-30 mmol/L
an anion gap >35 mmol/L suggests concomitant lactic acidosis (e.g. due to sepsis)
o Corrected sodium = measured Na + 1.6 ([plasma glucose – 100]/100) mg/dL
o Effective osmolality (mOsm/kg) = 2 (plasma Na) + (plasma glucose mg/dl) /18. (Normal range is 275–295 mOsm/kg)
➡️Infection in DKA when to Suspect (High, Strong):
· Suspect infection if the patient has fever, high CRP, or an anion gap more than 35 mmol/l and give antibiotics after obtaining appropriate cultures. Leukocytosis with shift to the left may occur in DKA without presence of infection. Consider Sepsis if acidosis is not improving (lactic acidosis) after revising fluid and insulin infusions.
· We suggest ICU admission in the following conditions:
o Children in severe DKA (pH< 7.1, HCO3- < 5 mEq/L)
o Children at increased risk of cerebral oedema [e.g., <5 years of age, severe acidosis, low pCO2 (<21 mmHg), high blood urea nitrogen (> 20 mg/dl)].
➡️We suggest the following treatment plan (Very Low, Conditional):
· Initial Resuscitation Fluid Therapy:
o For children who are volume depleted but not in shock: Volume expansion (ressuscitation) should begin immediately with 0.9% saline, 10 to 20 ml/kg infused over 20–30 min to restore the peripheral circulation.
➡️The initial fluid bolus SHOULD be subtracted from the calculated fluid deficit.
If tissue perfusion is poor the initial fluid bolus volume should be 20 ml/kg.
o For children with DKA in shock (High, Strong): rapidly restore circulatory volume with 0.9% saline in 20 ml/kg boluses directly infused manually into a large bore cannula as quickly as possible with reassessment of circulatory status / tissue perfusion after each bolus. If the child shock is fluid-responsive, give fluids as needed until circulation is restored guided by patient capillary refill time, pulse rate and volume, central venous pressure, urine output, peripheral temperature, and blood pressure. Rate of fluid infusion does NOT increase the risk of cerebral edema. If the child shock is non fluid -responsive, consult the ICU to assess the need for vasoactive / inotropic drugs.
➡️Boluses given to treat shock SHOULD NOT be subtracted from the calculated fluid deficit.
o Initial resuscitation should take 20-30 minutes. Do not take longer as this may worsen the severity of DKA
o Blood glucose may drop 75-100 mg/dl/hour in this initial rehydration phase.
Type of ressuscitation fluids: Use crystalloid, like normal saline, not colloid or initial volume expansion.
· Subsequent deficit and maintenance fluid (High, Strong):
A- Calculate the total fluid requirement by adding the estimated fluid deficit to the fluid maintenance requirements per 24 hours.
B- Estimating Fluid Deficit: Assume 5% dehydration in mild DKA, 5-7% dehydration (6-10% in infants) in moderate DKA. Assume 7-10% dehydration in severe DKA (˃10-15% in infants).
C- In shocked patients, deficits may exceed 10% body weight.
Use Table (1) for estimating severity of dehydration.
D- Aim to replace the estimated fluid over 24 to 48 hours.
E- ISPAD table (3) provides easy precalculated volumes of replacement and maintenance fluids (provided in this document in implementation tools) can be used when 10% dehydration is assumed and the total fluid replacement will be given over 48 hours. The fluid volume in the table is calculated per 24 hours and per hour based on body weight.
F- For body weights >32 kg, the volumes have been adjusted so as not to exceed twice the maintenance rate of fluid administration.
G-Calculation of fluid infusion rates for obese children should be similar to those of other children. Using ideal body weight for fluid calculations for these children is not necessary. If fluid calculations for obese children exceed those typically used in adult protocols, then adult DKA fluid protocols can be used (e.g., 1 L maximum per bolus and 500 ml/h fluid infusion).
H- I.V. fluids given in another hospital before assessment should be subtracted from the calculations.
I- Replacement of urinary losses should not be routinely done but may only be necessary in some circumstances with severe diuresis, particularly in children with a mixed presentation of DKA and HHS. Careful monitoring of fluid intake and output is essential to ensure positive fluid balance to correct the underlying dehydration (Very Low, Conditional).
Type of subsequent fluid to use (High, Strong):
- Use 0.9% saline to 0.45 saline or a balanced salt solution (Ringer’s lactate) with added potassium chloride for subsequent fluid replacement.
- Introduce glucose to IV fluid to avoid hypoglycemia before resolution of DKA:
Introduce glucose once blood glucose falls below 250-300 mg/dl or the rate of drop of BG exceeds 90 mg/dl/hr and increase glucose concentration as needed to avoid hypoglycemia (Good Practice Statement).
Initially once BG falls below 250-300 mg/dl, or the rate of drop of BG exceeds 90mg/dl/hr, use 250 ml glucose 5% and 250 ml 0.9% saline (which gives 2.5% glucose in 0.45% saline)
If the rate of drop is still rapid or BG reaches 180 mg/dl (usual renal threshold for glucose loss) increase glucose concentration in IV fluids by using 250 ml glucose 10% and 250 ml 0.9% saline (gives 5% glucose in 0.45% saline).
Introduce 10% glucose if the rate of hourly drop of BG exceeds 90 mg/dl/hr or BG reaches 90 mg/dl (e.g. use 200 ml of 25% glucose and 300 ml of 0.9% saline which gives 10% glucose in 0.45% saline).
Increase IV glucose concentration to 12.5% as needed according to drop of BG (made by adding 250 ml glucose 25% and 250 ml of 0.9% saline to give 12.5% glucose in 0.45% saline).
NB. Glucose 10%= 10 gram glucose in 100 ml = 100 mg glucose in 1 ml
-Do NOT reduce the rate of insulin infusion to avoid hypoglycemia (as this will worsen the acidosis and metabolic derangements) but increase the concentration of glucose in IV fluids to avoid hypoglycemia.
Sodium concentration in IV fluids (High, Strong):
- Initial sodium is usually low (due to dilutional effect from osmotic movement of water to extracellular compartment and because of increased sodium free lipid fraction in the blood) and corrected sodium must be calculated.
- Serum sodium trends during DKA treatment largely reflect the balance of sodium and water losses at presentation and sodium concentration in IV fluids. Evidence showed that the drop in corrected sodium concentration during treatment was not associated with cerebral injury.
- Sodium usually rises slowly (by 1.6 mmol/L for each 100 mg/dl decrease in glucose concentration) or remains in normal range with drop in BG.
- If measured serum sodium concentration is low and does not rise appropriately with the fall in BG level, increase the sodium content of the fluid (e.g. use 0.675% saline which is 3/4 normal saline, or higher sodium content fluid like 0.9% normal saline).
- In the event that changes in serum sodium concentration are required, the sodium content of intravenous fluids should be adjusted, but not the rate of infusion.
J- Hyperchloremia may occur with large volume fluid administration causing persistence of low serum bicarbonate. This usually resolves spontaneously. To make sure there is no deterioration of patient condition, evaluate other clinical and lab data, and calculate the anion gap or measure blood betahydroxybutyrate ketone level if available to ensure they are decreasing. The chloride load in IV fluid may be reduced by using Ringer’s lactate solution instead of saline.
➡️Correction of acidosis and bicarbonate therapy (Low, Conditional):
DO NOT give bicarbonate as it may cause harm (increases risk of hypokalemia, worsen tissue oxygenation, may cause paradoxical CNS acidosis and significantly increases the risk of development of cerebral edema later). Bicarbonate may be indicated in:
- Severe acidosis (pH< 6.9) with evidence of compromised cardiac contractility. In this case give bicarbonate after initial rapid boluses given rapidly within 30 minutes if the pH remains below 6.9.
- For treatment of life-threatening hyperkalaemia
If bicarbonate is indicated, carefully give 1-2 mmol/kg over 60 minutes.
Major causes of persistent acidosis include insufficient fluid administration, incorrect preparation or administration of IV insulin or associated sepsis.
o Potassium therapy (Very low, Conditional)
A- Assessment of serum potassium:
- If immediate serum potassium measurement is unavailable, an ECG is an alternative, T wave flattening and inversion, prominent U waves indicate hypokalemia while tall peaked T waves indicate hyperkalemia (figure 3).
- Severe hypokalemia (< 2.5 mEq/l) is an independent marker of poor treatment outcome and mortality.
B- Potassium Replacement: Usually there is an average of 5 mEq/ kg (range 3-6 mEq/kg) loss of potassium (lost in urine with polyuria). Potassium shifts out of the cells in the presence of acidosis and with lack of insulin. Hypokalemia may be more severe in malnourished children. Unless the patient is in renal failure with poor urine output, fluids should have added potassium.
- If the child is hypokalemic, start potassium replacement at the time of initial volume expansion and before starting insulin therapy. For children with initial potassium levels <3 mmol/L, defer insulin treatment and give a bolus of potassium (not to exceed 0.5 mmol/Kg/h), along with cardiac monitoring.
- When potassium is infused at the time of initial boluses, only 20 mmol/L potassium can be used if fluid is infused at ≥ 10 ml/kg/hour (e.g. during initial resuscitation) because the maximum allowed rate of potassium infusion is 0.5 mmol/kg/hour.
- The maximum allowed concentration of potassium in a peripheral IV line is 60 mmol/L. Make sure there is no extravasation (potassium is caustic).
- Monitor s.K+ hourly in this case and do cardiac monitoring for any arrythmia.
- If hypokalemia persists despite a maximum rate of potassium replacement, then the rate of insulin infusion can be reduced.
- If s. K+ is 3.5-5 mEq/l (normal range), start potassium chloride at a rate 40 mmol/L fluid at the time of starting insulin after the initial fluid resuscitation.
- Subsequent potassium replacement therapy should be based on serum K+ measurements (do s-K+ 2 hours after starting potassium then every 4 hours in this case).
- If initial s. K+ is above 5.5 mmol/L, wait until urine output is established and s-K+ drops below 5.5 mmol/L to start potassium replacement. Measure potassium hourly to initiate potassium infusion once the serum level drops to normal range.
- Potassium replacement should continue throughout IV fluid therapy.
· Insulin Therapy (Intermediate, Strong) :
A- Timing of starting insulin
- Start I.V. insulin infusion 1 hour AFTER starting fluid replacement therapy, i.e., after the patient has received initial volume expansion. DO NOT take longer time in the initial bolus resuscitation to avoid further deterioration before starting insulin.
- Do not give an IV bolus of insulin at the start of therapy because:
It may precipitate shock by rapidly decreasing osmotic pressure.
It may exacerbate hypokalemia.
B- Insulin Route
- Route of administration: IV (If a child or young person with DKA is using insulin pump therapy, start intravenous insulin therapy and disconnect the pump).
- Infusion tubing should be flushed with the insulin solution before Administration
- Central venous catheters should not be used for insulin administration because the large dead space may cause erratic insulin delivery.
C- Insulin dose
- Insulin therapy should begin with 0.1 U/kg/h (dilute 50 units regular (soluble) insulin in 50mL normal saline, 1 unit=1mL)
- Start at 0.05 unit /kg /hour if the patient shows marked sensitivity to insulin as in:
young children below age of 5 years
some known cases of diabetes who received a dose of insulin prior to presentation in DKA
less severe DKA (pH >7.15)
- The insulin dose may be decreased further provided that metabolic acidosis continues to resolve. (For example, in a child below 5 years and mild DKA, insulin may drop from 0.05 unit/kg/h, to 0.03 unit/kg/h).
- Aim for a decrease in serum glucose of 35-90 mg/dl/hour after insulin is started.
- Increase the rate of insulin infusion if the rate of drop of blood glucose is less than 35 mg/dl/hour.
- The dose of insulin should usually remain at 0.05–0.1 unit/kg/h and should NOT be reduced until resolution of DKA (pH > 7.30, serum bicarbonate >18 mmol/L, closure of anion gap)
- Resolution of DKA takes longer than normalization of blood sugar. So, increase glucose concentration in infused fluid (see fluid section) to be able to maintain insulin infusion without development of hypoglycemia until complete resolution of DKA.
· We suggest the following monitoring schedule (Very low, Conditional):
o Hourly heart rate, respiratory rate, capillary refill time and blood pressure.
o Hourly fluid input and output with measurement of urine output (or more frequently, with the possibility of urinary catheterization when there is impaired consciousness).
o Hourly GCS assessment, neurologic assessment
o Observe for warning signs of cerebral oedema, including headache, irritability, inappropriate slowing of heart rate and rise of blood pressure, repeated vomiting, increased drowsiness, incontinence, specific nerve palsies, change in pupillary size or reaction.
o Hourly capillary blood glucose monitoring
o Do the following laboratory measurements at 2 hours and every 2-4 hours (or hourly in severe cases until stabilization of the patient), venous blood gases, s-sodium, s-potassium, blood urea nitrogen, s-creatinine, s-calcium, magnesium, phosphate (should they be done every 4-6 hours according to need).
Serum may be lipemic, which in extreme cases can interfere with accuracy of electrolyte measurements in some laboratories (eg sodium).
A- Measure body weight each morning
o We suggest the following phosphate therapy in DKA (Very low, Conditional)
o Routine phosphate replacement is not routine unless treatment (e.g. with potassium phosphate) is available but severe hypophosphatemia (< 1 mg/dl) with or without symptoms should be treated immediately.
o Phosphate depletion occurs in DKA due to osmotic losses in urine and shift of intracellular phosphate to extracellular compartment due to acidosis.
o Phosphate level decreases further with treatment (fluid dilution and correction of acidosis causing intracellular movement of phosphate).
o Hypophosphatemia occurs in 50-60% of children during treatment. continuation of intravenous therapy without food consumption beyond 24 hours is a risk factor for clinically significant hypophosphatemia.
o Careful monitoring of serum calcium and magnesium should be done during phosphate replacement to avoid hypocalcemia.
We suggest the following transition to subcutaneous Insulin plan (Very low, Conditional):
B- Transition to subcutaneous therapy and stop intravenous therapy at resolution of DKA WHEN ALL OF THE FOLLOWING occurs:
- ketosis has resolved,
N.B. Absence of ketonuria (ketones in urine) should not be used as an endpoint for determining resolution of DKA. Ketonuria characteristically continues for several hours after serum β- hydroxybutyrate level returns to normal. Note that urine ketone strips measure acetoacetate and acetone while beta-hydroxybutyrate (BOHB) is the main ketone body in tissues. BOHB is eliminated by conversion to acetoacetate which is excreted in urine with DKA resolution.
- pH>7.30, bicarbonate >18 mmol/L and closure of the anion gap.
- Patient is fully conscious.
- Patient can take oral fluids without nausea or vomiting.
o Start subcutaneous insulin before stopping intravenous insulin:
- Shift may be more convenient before a mealtime.
- Give short-acting regular insulin 30 min-1 hour before stopping IV insulin (rapid-acting analogues should be injected 15-30 minutes before stopping IV insulin).
- Timing of intermediate- or long-acting insulin should be determined by the individual patient’s SC insulin regimen. For example, for the patient on a basal-bolus insulin regimen, the first dose of basal insulin may be started in the evening and IV insulin stopped the next morning if DKA has resolved by the morning.
- Do NOT use premixed insulin (to allow more flexibility of dosing insulin rather than a fixed basal to mealtime insulin ratio).
➡️Five General Sick Day Diabetes Management Principles
(ISPAD, 2018) (Good Practice Statement) :
1. More frequent BG and ketone (urine or blood) monitoring
2. DO NOT STOP INSULIN
3. Monitor and maintain hydration with adequate salt and water balance.
4. Treat the underlying precipitating illness
5. Sick day guidelines including insulin adjustment should be taught soon after diagnosis and reviewed at least annually with patients and family members with a goal of minimizing and/or avoiding DKA and similarly minimizing and/or avoiding illness associated hypoglycemia.
➡️We suggest the following management plan for cerebral oedema (CE) (Very low, Conditional):
A- Diagnosis:
o The degree of cerebral edema that develops during DKA correlates with the degree of dehydration and hyperventilation at presentation, but not with initial osmolality or osmotic changes during treatment.
o SUSPECT, who is at high risk?
- younger age, especially below 5 years.
- new onset diabetes or long duration of symptoms.
- severe acidosis.
- high BUN at presentation (>20 mg/dl).
- severe hypocapnia at presentation after adjusting for the degree of acidosis.
- bicarbonate treatment for correction of acidosis.
- In these cases, mannitol or hypertonic saline should be available at the bedside with dose calculated,
o When does CE occur?
Usually within 12 hours after treatment is started but, uncommonly, may occur before the start of treatment or rarely, it can occur within 24-48 hours after start of treatment
o Clinical Diagnosis: CE in DKA is a clinical diagnosis.
One diagnostic criterion, or two major criteria, or one major and two minor criteria (table 5) have a sensitivity of 92%, a specificity of 96% and a false positive rate of only 4% for the early recognition of DKA-related cerebral oedema; early enough to allow for effective treatment.
o When to do cranial imaging?
Start treatment first as with any critically ill patient and do not delay until imaging is done.
The primary indications for imaging are focal neurologic deficit (presence of signs of lateralization) for suspicion of:
intracranial hemorrhage which requires emergency neurosurgery
cerebrovascular thrombosis which may require anticoagulation
In both cases the patient will clinically present with focal or severe progressive headache or focal neurologic deficit.
C- Treatment of CE:
o If clinical diagnosis of CE is done, treat immediately. Transfer patient to ICU. Give the most readily available one of the following:
- mannitol 20%, give 0.5–1 g/kg over 10–15 minutes. Effect of mannitol is apparent after 15 minutes and lasts for 2 hours. It can be repeated after 30 minutes if necessary.
- hypertonic sodium chloride (3%), 2.5–5 ml/kg over 10–15 minutes. It can be used if mannitol is not available or in addition to mannitol if there is no response to mannitol after 30 minutes.
- Adjust rate of fluid infusion so as to avoid excessive fluids that might increase cerebral edema while also to maintain a normal blood pressure to avoid cerebral hypoperfusion.
- Elevate the bed head to 30°.
➡️We suggest the following management of HHS (Very low, Conditional):
A- The initial bolus: It should be ≥20 ml/kg of isotonic saline (0.9% NaCl) and additional boluses can be given rapidly if needed to restore peripheral perfusion.
B- Subsequent Fluid Replacement : A fluid deficit of approximately 12% to 15% of body weight should be assumed and urinary losses should be added to the calculated fluids. Use 0.45% to 0.75% NaCl replace the deficit over 24 to 48 h. Isotonic (0.9%) saline should be restarted if perfusion and hemodynamic status appear inadequate as serum osmolality declines.
C- Adjust sodium concentration in fluids to promote a gradual decline in corrected serum sodium concentration and osmolality (A rate of 0.5 mmol/L per hour has been recommended for hypernatremic dehydration).
Mortality has been associated with failure of the corrected serum sodium concentration to decline with treatment.
D- During the initial few hours of rehydration, BG may decline more rapidly. After this phase, if there is a continued rapid fall in BG (>100 mg/dl per hour), add 2.5% or 5% glucose to the rehydration fluid.
E- Potassium should be added to IV fluids just as in the DKA protocol.
F- Bicarbonate is contraindicated.
G- Start insulin once the drop of BG is less than 50 mg/dl/hour with fluids only. Give insulin at a dose of 0.025-0.05 U/Kg/hour. Adjust insulin to achieve a rate of drop of BG of 50-75 mg/dl/hour
H- Treat hypophosphatemia as needed. Replace magnesium in the occasional patient who experiences severe hypomagnesemia and hypocalcemia during therapy. The recommended magnesium dose is 25 to 50 mg/kg per dose for 3 to 4 doses given every 4 to 6 h with a maximum infusion rate of 150 mg/min and 2 g/h.
I- To prevent venous thrombosis, low molecular weight heparin should be considered, especially in children >12 years.
J- Cerebral edema is very rare in HHS and any change in mental status during therapy should be fully investigated.
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/- Recommendations
|
Table 3. Recommendations |
|
|
|||
|
A. Initial Assessment and Calculations |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A1
A2
A3
A4
A5
|
What are the necessary initial steps to be done for a DKA patient before starting treatment?
How to assess the severity of dehydration in a DKA patient clinically?
How to calculate anion gap, corrected sodium and osmolarity?
When to suspect infection in a DKA patient?
When to consider admitting a DKA patient in the ICU? |
ISPAD 2022
ISPAD 2022
ISPAD 2022
ISPAD 2022
ISPAD 2022
ISPAD 2022 |
We suggest in initial assessment of a patient with DKA to do the following: · Obtain vital signs and weight of the patient. Measure height/ length to calculate surface area. Note that despite severe dehydration, hypertension occurs in 12% of children with DKA. Such patients require volume replacement despite the hypertension and should be monitored particularly carefully for signs and symptoms of impending cerebral injury. · Insert two wide bore peripheral cannulas. Do Immediate measurement of : · blood glucose. · blood or urine ketones. · venous blood gases. · serum electrolytes. · blood urea nitrogen and s-creatinine. · complete blood count and C-reactive protein (CRP). · connect the patient to an ECG monitor and check T waves. Assess consciouss level: Glasgow coma scale (GCS) · assessment (table 4 ) Examine pupillary size and reflexes Obtain appropriate specimens for
cultures if there is evidence of infection e.g. fever. Assess the severity of dehydration (table 1) by: · Pulse rate and volume (weak rapid pulse in shock). · Capillary refill time (normal capillary refill is ≤1.5-2 seconds). · Skin turgor ('tenting' or inelastic skin) or other signs of dehydration. · Patient temperature and temperature of periphery (cold hands and feet indicate poor tissue perfusion and possible shock, hypothermia may also occur in shock). · Urine output (ml/hour). · Blood pressure. Hypotension is a late sign in shock (blood pressure is maintained for a long time by sympathetic tone, stress hormones and increased osmotic pressure from marked hyperglycemia). · conscious level (reduced in shock and is not alone indicative of brain edema). Mild DKA assumes 5%, moderate DKA 7% and severe DKA 10% dehydration. Calculate the following: · Anion gap = Na – (Cl + HCO3): - Normal is 12 ± 2 mmol/L - In DKA the anion gap is typically 20-30 mmol/L - an anion gap >35 mmol/L suggests concomitant lactic acidosis (e.g. due to sepsis) Corrected sodium = measured Na + 1.6 ([plasma glucose – 100]/100) mg/dL Effective osmolality (mOsm/kg) = 2 (plasma Na) + (plasma glucose mg/dl) / 18. Normal range is 275–295 mOsm/kg ·Suspect infection if the patient has fever, high CRP, or an anion gap more than 35 mmol/l and give antibiotics after obtaining appropriate cultures. Leucocytosis with shift to the left may occur in DKA without presence of infection. Consider Sepsis if acidosis is not improving (lactic acidosis) after revising fluid and insulin infusions We suggest ICU admission in the following conditions: 1. Children in severe DKA (pH< 7.1, HCO3-< 5 mEq/L) 2. Children at increased risk of cerebral oedema [e.g., <5 years of age, severe acidosis, low pCO2 (<21 mmHg), high blood urea nitrogen (> 20 mg/dl)].
|
Very low
Very low
low
Very low
Very low
Very low High
Very low |
Conditional
conditional
conditional
conditional
conditional
conditional
strong
conditional |
|
Table 4. Recommendations |
|
|
|||
|
A. Fluid Therapy Initial Resuscitation Fluid: |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
In case of volume depleted patient starting fluid expansion before insulin therapy what is the best initial therapy?
|
ISPAD 2022 |
B1. We suggest the following treatment plan
B1. For children who are volume depleted but not in shock, Volume expansion (resuscitation) should begin immediately with 0.9% saline, 10 to 20 ml/kg infused over 20–30 min to restore the peripheral circulation. The initial fluid bolus SHOULD be subtracted from the calculated fluid deficit. · If tissue perfusion is poor the initial fluid bolus volume should be 20 ml/kg. |
Very Low |
Conditional |
|
|
|
|
In the child with DKA in shock, rapidly restore circulatory volume with 0.9% saline in 20 ml/kg boluses directly infused manually into a large bore cannula as quickly as possible with reassessment of circulatory status / tissue perfusion after each bolus. If the child shock is fluid-responsive, give fluids as needed until circulation is restored guided by patient capillary refill time, pulse rate and volume, central venous pressure, urine output, peripheral temperature, and blood pressure. Rate of fluid infusion does NOT increase the risk of cerebral edema. If the child shock is nonfluid -responsive, consult the ICU to assess the need for vasoactive / inotropic drugs. Boluses given to treat shock SHOULD NOT be subtracted from the calculated fluid deficit. |
High |
Strong |
|
B2
|
In a DKA patient what are the available resusciation therapy available and how to calculate the required amount according to each patient? |
ISPAD 2022
|
Initial resuscitation should take 20-30
minutes,
Blood glucose may drop 75-100 mg/dl/hour in this initial rehydration phase. |
Very low
|
Conditional |
|
|
|
ISPAD 2022
|
Type of Resuscitation Fluid Use crystalloid, like normal saline, not colloid for initial volume expansion. |
Very low
|
Conditional |
|
B3 |
How to calculate the fluid deficit in a shocked patient?
|
ISPAD 2022
|
Subsequent Deficit and Maintenance Fluid: · Calculate the total fluid requirement by adding the estimated fluid deficit to the fluid maintenance requirements per 24 hours. · Estimating Fluid Deficit: Assume 5% dehydration in mild DKA, 5-7% dehydration (6-10% in infants) in moderate DKA. Assume 7-10% dehydration in severe DKA (˃10-15% in infants). In shocked patients, deficits may exceed 10% body weight |
High
|
Strong |
|
|
|
ISPAD 2018
|
Use Table 1 for estimating severity of dehydration. · Aim to replace the estimated fluid over 24 to 48 hours. · ISPAD table (3) provides easy precalculated volumes of replacement and maintenance fluids (provided in this document in implementation tools) can be used when 10% dehydration is assumed and the total fluid replacement will be given over 48 hours. The fluid volume in the table is calculated per 24 hours and per hour based on body weight. · For body weights >32 kg, the volumes have been adjusted so as not to exceed twice the maintenance rate of fluid administration. · Calculation of fluid infusion rates for obese children should be similar to those of other children. Using ideal body weight for fluid calculations for these children is not necessary. If fluid calculations for obese children exceed those typically used in adult protocols, then adult DKA fluid protocols can be used (e.g., 1 L maximum per bolus and 500 ml/h fluid infusion). · I.V. fluids given in another hospital before assessment should be subtracted from the calculations. |
High |
Strong |
|
|
|
ISPAD 2022
|
Replacement of urinary losses should not be routinely done but may only be necessary in some circumstances with severe diuresis, particularly in children with a mixed presentation of DKA and HHS. Careful monitoring of fluid intake and output is essential to ensure positive fluid balance to correct the underlying dehydration. |
Very low
|
Conditional |
|
B4
|
When to introduce glucose to the IV fluid to avoid hypoglycemia before DKA resolution?
|
ISPAD 2022
|
Type of subsequent fluid to use: Use 0.9% saline to 0.45 saline or a balanced salt solution (Ringer’s lactate) with added potassium chloride for subsequent fluid replacement. Introducing Glucose to IV Fluid to avoid hypoglycemia before resolution of DKA: • Introduce glucose once blood glucose falls below 250-300 mg/dl or the rate of drop of BG exceeds 90mg/dl/hr and increase glucose concentration as needed to avoid hypoglycemia. • Initially once BG falls below 250-300 mg/dl, or the rate of drop of BG exceeds 90mg/dl/hr, use 250 ml glucose 5% and 250 ml 0.9% saline (which gives 2.5% glucose in 0.45% saline) If the rate of drop is still rapid or BG reaches 180 mg/dl. • (usual renal threshold for glucose loss), increase glucose concentration in IV fluids by using 250 ml glucose 10% and 250 ml 0.9% saline (gives 5% glucose in 0.45% saline). • Introduce 10% glucose if the rate of hourly drop of BG exceeds 90 mg/dl/hr or BG reaches 90 mg/dl. (e.g. use 200 ml of 25% glucose and 300 ml of 0.9% saline which gives 10% glucose in 0.45% saline). • Increase IV glucose concentration to 12.5% as needed according to drop of BG (made by adding 250 ml glucose 25% and 250 ml of 0.9% saline to give 12.5% glucose in 0.45% saline). NB. Glucose 10%= 10 gram glucose in 100 ml = 100 mg glucose in 1 ml |
High
|
Strong |
|
|
|
|
Do NOT reduce the rate of insulin infusion to avoid hypoglycemia (as this will worsen the acidosis and metabolic derangements) but increase the concentration of glucose in IV fluids to avoid hypoglycemia |
Intermediate
|
Strong |
|
B5 |
What is the sodium concentration needed in IV fluid to avoid complications?
|
ISPAD 2022 |
Sodium concentration in IV fluids: Initial sodium is usually low (due to dilutional effect from osmotic movement of water to extracellular compartment and because of increased sodium free lipid fraction in the blood) and corrected sodium must be calculated. · Serum sodium trends during DKA
treatment largely reflect the balance of sodium and water losses at
presentation and sodium concentration in IV fluids. Evidence showed that drop
in corrected sodium concentration during treatment was not associated with
cerebral injury. · Sodium usually rises slowly (by 1.6 mmol/L for each 100 mg/dl decrease in glucose concentration) or remains in normal range with drop in BG. · If measured serum sodium concentration is low and does not rise appropriately with the fall in BG level, increase the sodium content of the fluid (e.g. use 0.675% saline which is 3/4 normal saline, or higher sodium content fluid like 0.9% normal saline). · In the event that changes in serum sodium concentration are required, the sodium content of intravenous fluids should be adjusted, but not the rate of infusion. · Hyperchloremia may occur with large volume fluid administration causing persistence of low serum bicarbonate. This usually resolves spontaneously. To make sure there is no deterioration of patient condition, evaluate other clinical and lab data, and calculate the anion gap or measure blood betahydroxybutyrate ketone level if available to ensure they are decreasing. The chloride load in IV fluid may be reduced by using Ringer’s lactate solution instead of saline. |
High |
Strong |
|
B6 |
When should we use bicarbonate therapy in case of acidosis?
|
ISPAD 2022
|
Acidosis and Bicarbonate therapy: In general, DO NOT give bicarbonate as it may cause harm (increases risk of hypokalemia, worsen tissue oxygenation, may cause paradoxical CNS acidosis and significantly increases the risk of development of cerebral edema later). Bicarbonate may be indicated if: 1. in severe acidosis (pH< 6.9) with evidence of compromised cardiac contractility. In ths case give bicarbonate after initial rapid boluses given rapidly within 30 minutes if the pH remains below 6.9. 2. for treatment of life-threatening hyperkalaemia If bicarbonate is indicated, carefully give 1-2 mmol/kg over 60 minutes. Major causes of persistent acidosis include insufficient fluid administration, incorrect preparation or administration of IV insulin or associated sepsis. |
Very Low |
Conditional |
|
B7 |
How to assess potassium deficit in a DKA patient and how to calculate?
|
ISPAD 2022 |
Potassium Therapy: A-Assessment of serum potassium: If immediate serum potassium measurements unavailable, an ECG is an alternative. • In ECG: T wave flattening and inversion, prominent U waves indicate hypokalemia while tall peaked T waves indicate hyperkalemia (figure 3). • Severe hypokalemia (< 2.5 mEq/l) is an ndependent marker of poor treatment outcome and mortality Potassium Therapy: A-Assessment of serum potassium: If immediate serum potassium measurements unavailable, an ECG is an alternative. • In ECG: T wave flattening and inversion, prominent U waves indicate hypokalemia while tall peaked T waves indicate hyperkalemia (figure 3). • Severe hypokalemia (< 2.5 mEq/l) is an ndependent marker of poor treatment outcome and mortality. B- Potassium Replacement: • Usually there is an average of 5 mEq/ kg (range 3-6 mEq/kg) loss of potassium (lost in urine with polyuria). Potassium shifts out of the cells in the presence of acidosis and with lack of insulin. Hypokalemia maybe more severe in malnourished children. - Unless the patient is in renal failure with poor urine output, fluids should have added potassium. - If initial s.K+ is below 3.5 mmol/L (hypokalemic child), start potassium replacement at the time of initial fluid resuscitation. • If s.K+ is 2.5-3.5 mmol/L,start of insulin treatment may need to be delayed or reduced. • Do not start insulin therapy if the potassium level is at or below 3 mmol/L. • When potassium is infused at the time of initial boluses, only 20 mmol/L potassium can be used if fluid is infused at ≥ 10 ml/kg/hour (e.g. during initial resuscitation) because the maximum allowed rate of potassium infusion is 0.5 mmol/kg/hour. • The maximum allowed concentration of potassium in a peripheral IV line is 60 mmol/L. Make sure there is no extravasation (potassium is a caustic). • Monitor s.K+ hourly in this case and do cardiac monitoring for any arrythmia. • If hypokalemia persists despite a maximum rate of potassium replacement, then the rate of insulin infusion can be reduced. - If s-K+ is 3.5-5 mEq/l (normal range), • start potassium chloride at a rate 40 mmol/L fluid at the time of starting insulin after the initial fluid resuscitation. • Subsequent potassium replacement therapy should be based on serum K+ measurements (do s-K+ 2 hours after starting potassium then every 4 hours in this case). - If initial s-K+ is above 5 mmol/L, wait until urine output is established and s-K+ drops below 5 mmol/L to start potassium replacement. Measure potassium hourly to initiate potassium infusion once the serum level drops to normal range. - Potassium replacement should continue throughout IV fluid therapy. |
Very Low |
Conditional |
|
B8 |
How to initiate and establish insulin therapy?
|
ISPAD 2022
|
Insulin Therapy: A-Timing of starting insulin · Start I.V. insulin infusion 1 hours AFTER starting fluid replacement therapy; i.e., after the patient has received initial volume expansion. DO NOT take longer time in the initial bolus resuscitation to avoid further deterioration before starting insulin. · Do not give an IV bolus of insulin at the start of therapy because: --- It may precipitate shock by rapidly decreasing osmotic pressure. --- It may exacerbate hypokalemia. B-Insulin Route · Route of administration: IV · If a child or young person with DKA is using insulin pump therapy, start intravenous insulin therapy and disconnect the pump. · Infusion tubing should be flushed with the insulin solution before Administration Central venous catheters should not be used for insulin administration because the large dead space may cause erratic insulin delivery C- Insulin dose • Insulin therapy should begin with 0.1 U/kg/h (dilute 50 units regular (soluble) insulin in 50mL normal saline, 1 unit=1mL) • Start at 0.05 unit /kg /hour if The patient shows marked sensitivity to insulin as in: ---young children below age of 5 years, ---some known cases of diabetes who received a dose of insulin prior to presentation in DKA ---less severe DKA (pH >7.15) • The insulin dose may be decreased further provided that metabolic acidosis continues to resolve. (For example, in a child below 5 years and mild DKA, insulin may drop from 0.05 unit/kg/h, to 0.03 unit/kg/h). • Aim for a decrease in serum glucose of 35-90 mg/dl/hour after insulin is started. • Increase the rate of insulin infusion if the rate of drop of blood glucose is less than 35 mg/dl/hour. • The dose of insulin should usually remain at 0.05–0.1 unit/kg/h and should NOT be reduced until resolution of DKA (pH > 7.30, serum bicarbonate >18 mmol/L, closure of anion gap) Resolution of DKA takes longer than normalization of blood sugar. So, increase glucose concentration in infused fluid (see fluid section) to be able to maintain insulin infusion without development of hypoglycemia until complete resolution of DKA. |
Intermediate
|
Strong |
|
B9 |
What are the necessary investigation and clinical findings needed to be done in monitoring a DKA patient?
|
ISPAD 2022 |
· We suggest the following monitoring schedule: · Hourly heart rate, respiratory rate, capillary refill time and blood pressure. · Hourly fluid input and output with measurement of urine output (or more frequently, with the possibility of urinary catheterization when there is impaired consciousness). • Hourly GCS assessment, neurologic assessment Observe for warning signs of cerebral oedema, including headache, irritability, inappropriate slowing of heart rate and rise of blood pressure, repeated vomiting, increased drowsiness, incontinence, specific nerve palsies, change in pupillary size or reaction. • Hourly capillary blood glucose monitoring • do the following laboratory measurements at 2 hours and every 2-4 hours (or hourly in severe cases until stabilization of the patient) o venous blood gases o s-sodium, s-potassium, 2o blood urea nitrogen, s-creatinine o s-calcium, magnesium, phosphate. (should they be done every 4-6 hours according to need) • Measure body weight each morning . Serum may be lipemic, which in extreme cases can interfere with accuracy of electrolyte measurements in some laboratories (eg sodium). |
Very Low |
Conditional |
|
B10 |
Why does phosphate depletion occur and how to manage?
|
ISPAD 2022 |
· We suggest the following phosphate therapy in DKA • Phosphate depletion occurs in DKA due to osmotic losses in urine and shift of intracellular phosphate to extracellular compartment due to acidosis. • Phosphate level decreases further with treatment (fluid dilution and correction of acidosis causing intracellular movement of phosphate). • Hypophosphatemia occurs in 50-60% of children during treatment. continuation of intravenous therapy without food consumption beyond 24 hours is a risk factor for clinically significant hypophosphatemia. • Routine phosphate replacement is not routine unless treatment (e.g. with potassium phosphate) is available but Severe hypophosphatemia (< 1 mg/dl) with or without symptoms should be treated immediately. • Careful monitoring of serum calcium and magnesium should be done during phosphate replacement to avoid hypocalcemia. |
Very low |
Conditional |
|
B11 |
When to transition to SC Insulin?
|
ISPAD 2022 |
We suggest the following transition to subcutaneous Insulin plan: · Transition to subcutaneous therapy and stop intravenous therapy at resolution of DKA which is WHEN ALL OF THE FOLLOWING occurs: 1.ketosis has resolved, N.B. Absence of ketonuria (ketones in urine) should not be used as an endpoint for determining resolution of DKA. Ketonuria characteristically continues for several hours after serum β- hydroxybutyrate level returns to normal. Note that urine ketone strips measure acetoacetate and acetone while beta-hydroxybutyrate (BOHB) is the main ketone body in tissues. BOHB is eliminated by conversion to acetoacetate which is excreted in urine with DKA resolution. 2.
pH>7.30, bicarbonate >18 mmol/L and closure of the anion gap. 3. Patient is fully conscious. 4. Patient can take oral fluids without nausea or vomiting. · Shift may be more convenient before a meal time. · Start subcutaneous insulin before stopping intravenous insulin: ---give short-acting regular insulin 30 min-1 hour before stopping IV insulin (rapid-acting analogues should be injected 15-30 minutes before stopping IV insulin). ---timing of intermediate- or long-acting insulin should be determined by the individual patient’s SC insulin regimen. For example, for the patient on a basal-bolus insulin regimen, the first dose of basal insulin may be started in the evening and IV insulin stopped the next morning if DKA has resolved by the morning. Do NOT use premixed insulin (to allow more flexibility of dosing insulin rather than a fixed basal to mealtime insulin ratio). Five General Sick Day Diabetes Management Principles 1. More frequent BG and ketone (urine or blood) monitoring 2. DO NOT STOP INSULIN 3. Monitor and maintain hydration with adequate salt and water balance. 4. Treat the underlying precipitating illness 5. Sick day guidelines including insulin adjustment should be taught soon after diagnosis and reviewed at least annually with patients and family members with a goal of minimizing and/or avoiding DKA and similarly minimizing and/or avoiding illness associated hypoglycemia. |
Very Low
Low
Good Practice Statement |
Conditional
Conditional |
|
B12 |
Cerebral eodema when does it occur , how to diagnose and how treat?
|
ISPAD 2022 |
We suggest the following management plan for cerebral oedema (CE): A-Diagnosis: · SUSPECT, who is at high risk? - younger age, especially below 5 years. - new onset diabetes or long duration of symptoms. - severe acidosis. - high BUN at presentation (>20 mg/dl). - severe hypocapnia at presentation after adjusting for the degree of acidosis. bicarbonate treatment for correction of acidosis. In these cases, mannitol or hypertonic saline should be available at the bedside with dose calculated, · When does CE occur? Usually within 12 hours after treatment is started but, uncommonly, may occur before the start of treatment or rarely, it can occur within 24-48 hours after start of treatment · Clinical Diagnosis: Use criteria in table (4) : CE in DKA is a clinical diagnosis. One diagnostic criterion, or two major criteria, or one major and two minor criteria have a sensitivity of 92%, a specificity of 96% and a false positive rate of only 4% for the early recognition of DKA-related cerebral oedema; early enough to allow for effective treatment. · When to do cranial imaging? Start treatment first as with any critically ill patient and do not delay until imaging is done. The primary indications for imaging are focal neurologic deficit (presence of signs of lateralization) for suspicion of : 1. intracranial hemorrhage which requires emergency neurosurgery 2. cerebrovascular thrombosis which may require anticoagulation In both cases the patient will clinically present with focal or severe progressive headache or focal neurologic deficit. |
Very Low
Low |
Conditional
Conditional |
|
|
|
ISPAD 2018
|
Treatment of CE: A- If clinical diagnosis of CE is done, treat immediately. Transfer patient to ICU. B- Give the most readily available one of the following: · --mannitol 20%, give 0.5–1 g/kg over 10–15 minutes. Effect of mannitol
is apparent after 15 minutes and lasts for 2 hours. It can be repeated after
30 minutes if necessary. --hypertonic sodium chloride (3%), 2.5–5 ml/kg over 10–15 minutes. It can be used if mannitol is not available or in addition to mannitol if there is no response to mannitol after 30 minutes. C-
Adjust rate of fluid infusion so as to avoid excessive fluids that
might increase cerebral edema while also to maintain a normal blood pressure to
avoid cerebral hypoperfusion. Elevate the bed head to 30°. |
Low |
Conditional |
|
B13 |
How to manage a case of HHS?
|
ISPAD 2022 |
We suggest the following management of HHS: Management of HHS: 1- The
initial bolus 2- Subsequent
Fluid replacement: 3- During the initial few hours of rehydration, BG may decline more
rapidly. After this phase, if there is a continued rapid fall in BG (>100
mg/dl per hour), add 2.5% or 5%
glucose to the rehydration fluid. 4- Potassium
should be added to IV fluids just as in
the DKA protocol. Bicarbonate is contraindicated |
Very Low |
Conditional |
|
|
|
|
6- Start insulin once the drop of BG is less than 50 mg/dl/hour with fluids only. Give insulin at a dose of 0.025-0.05 U/Kg/hour. Adjust insulin to achieve a rate of drop of BG of 50-75 mg/dl/hour 7- Treat hypophosphatemia as needed. Replace magnesium in the occasional patient who experiences severe hypomagnesemia and hypocalcemia during therapy. The recommended magnesium dose is 25 to 50 mg/kg per dose for 3 to 4 doses given every 4 to 6 h with a maximum infusion rate of 150 mg/min and 2 g/h. 8- To prevent venous thrombosis, low molecular weight heparin should be considered, especially in children >12 years. Cerebral edema is very rare in HHS and any change in mental status during therapy should be fully investigated |
Low |
Conditional |
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Pediatrics Department, Consultant, Cairo University |
|
||||
|
Ass. Prof. Amal Gaber Mohamed |
Pediatrics Department, Consultant, Al-Azhar University |
|
|||
|
Prof. Amany Kamal El-Hawary |
Pediatrics Department, Consultant, Mansoura University |
|
|||
|
Prof. Amina M. Abdel Wahab |
Pediatrics Department, Consultant, Suez Canal University |
|
|||
|
Prof. Ashraf A. Elsharkawy |
Pediatrics Department, Consultant, Mansoura University |
|
|||
|
Prof. Basma Abd-Elmoez |
Pediatrics Department, Consultant, Minia University |
|
|||
|
Dr. Eman Elshanawany
|
Pediatrics Department, Consultant, Benha University |
|
|||
|
Prof. Ghada Anwar |
Pediatrics Department, Cairo University |
|
|||
|
Prof. Hanaa Abdellateef Mohamad |
Pediatrics Department, Consultant, Assiut University |
|
|||
|
Ass. Prof. Hanan Hassan Aly |
Pediatrics Department, Consultant, Ain Shams University |
|
|||
|
Prof. Hoda Atwa |
Pediatrics Department, Consultant, Suez Canal University |
|
|||
|
Prof. Lubna Fawaz |
Pediatrics Department, Consultant, Cairo University |
|
|||
|
Dr. Mariam Nader Moawad, |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
|||
|
Dr. Marian Fares Nashed |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
|||
|
Ass. Prof. Mona Karem Amin |
Pediatrics Department, Consultant, Suez Canal University |
|
|||
|
Prof. Nora E Badawi |
Pediatrics Department, Consultant, Cairo University |
|
|||
|
Ass. Prof. Nouran Y Salah El-Din |
Department of Pediatrics, Ain Shams University |
|
|||
|
Dr. Ramy Saleh Morsy |
Department of Pediatrics, Consultant, Armed Forces College of Medicine |
|
|||
|
Prof. Randa M. Matter |
Pediatrics Department, Consultant, Ain Shams University |
|
|||
|
Ass. Prof. Remon M. Yousef |
Department of Pediatrics, Consultant, Fayoum University |
|
|||
|
Prof. Sabry M Ghanem |
Department of Pediatrics, Consultant, Al-Azhar University |
|
|||
|
Prof. Safinaz El Habashy |
Department of Pediatrics, Consultant, Ain Shams University |
|
|||
|
Ass. Prof. Shaymaa Elsayed Abdel Meguid |
Department of Pediatrics, Consultant, Alexandria University |
|
|||
|
Prof. Wiam Al Farouk Younis |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
|||
|
Dr. Hamed Khaled Khalifa |
M.B.B.Ch Misr University for Science and Technology. House Officer, Pediatrics Department, Cairo University.
|
|
|||
|
Prof. Tarek Omar |
Pediatrics Department, Faculty of Medicine, Alexandria University, Alexandria, Egypt Alexandria Center for Evidence-Based CPG Consultancy Board, EPG, Egypt |
Professor of Pediatrics, Methodology Supervision Subgroup |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Pediatrics Department, Cairo University/ Egypt |
|||||
|
Prof. Mona Hafez |
Pediatrics Department, Cairo University/ Egypt |
||||
|
Prof. Mona Salem |
Pediatrics Department, Ain Shams University/ Egypt |
||||
|
Prof. Nermin Salah |
Pediatrics Department, Cairo University/ Egypt |
||||
|
Prof.Hesham El Hefnawy |
Prof. of diabetes and endocrinology, former dean of the National Institute of Diabetes and Endocrinology, Cairo, Egypt. Head of National Committee of non-communicable disease |
||||
|
External Reviewer(s) for methodology |
|||||
|
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
|||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge The International Society for Pediatric and Adolescent Diabetes (ISPAD) Clinical Practice Consensus Guidelines 2018 and 2022guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
CPG |
Clinical Practice Guideline |
||
|
DKA |
Diabetic Ketoacidosis |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ERG |
External Review Group |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
ISPAD |
The International Society for Pediatric and Adolescent Diabetes |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
- Funding
▪️This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
- Glossary
Acceptability: Is the extent to which the users are likely to adopt a recommendation It is based on internal qualities such as clarity, comprehensiveness, and logical reasoning and on external factors such as the burden imposed on the process and system of care, patient and providers attitudes and beliefs and patients’ needs expectations and preferences.
Adaptation (of guidelines): It is the systematic approach to considering the use and/or modification of guidelines produced in one cultural and organizational setting for application in different context. Adaptation can be used as an alternative to de novo guidelines development or for customizing existing guidelines to suit the local context.
Adoption (of guidelines): It is the acceptance of guidelines after the assessment of the quality, currency, and content. When health care providers (or other users of recommendations) use the adopted guidelines, they feel committed to change their practices in accordance with the recommendations of the guidelines.
Applicability: It is the extent to which the users can put a recommendation into practice, based on internal qualities such as a clearly defined eligible patient population that matches the population to which the intervention is targeted in the local setting and external factors such as the availability of the necessary knowledge, skills, provider time, staff, equipment, and other resources.
Applicability is sometimes taken as a synonym for feasibility:
• Feasibility of the acquisition of necessary skills and knowledge.
• Feasibility of the necessary increase in provider time, staff, equipment, and so on.
Culture: Culture represents the norms and values of a specific group, community or population.
Diffusion: It is a passive means of transferring knowledge; it is not directed towards a target audience (e.g. publication of articles in medical journals).
Dissemination : It is more active than diffusion in that it targets specific audients and involves tailoring the information for these audients (e.g. dissemination strategies including targeted mailings, presentations and press conferences.
Evidence-based principles: Evidence-Based Medicine (EBM) has been defined as the conscientious, explicit and judicious use of the current best evidence in making decisions about the care of individual patients. The practice of EBM means integrating individual clinical expertise with the best available external clinical evidence from systematic research.
Evidence tables: They are summaries of the most salient information from studies identified in the systematic review. The elements of evidence tables are dependent on the types of information in studies related to a particular topic but might include information such as the article reference, the study type (e.g. RCT or Cohort), the number of patients and their characteristics and the intervention, comparison arms, outcome measures and effect sizes.
Guidelines or Clinical Practice Guidelines (CPG): Systematically developed statements about specific health problems, intended to assist practitioners and patients in making decisions about appropriate health care.
Guidelines consistency: Agreement between the evidence and the recommendations, based on:
• Comprehensiveness of the study search and selection process.
• Coherence between the results of the studies and their interpretation by the guidelines authors.
• Transparency between interpretation and recommendations.
Guidelines content: In the ADAPTE Manual and Resource Toolkit for Guidelines Adaptation document, guidelines content refers to the recommendations in the source guidelines.
Guidelines currency: A CPG may be considered up to date when no new information on interventions, outcomes and performance justifies updating it.
Guidelines quality: By quality of clinical practice guidelines, we mean the confidence that the potential biases of guidelines development addressed adequately and that the recommendations are both internally and externally valid and are feasible for practice. This process involves considering the benefits, harms and costs of the recommendations as well as the practical issues attached to them. Therefore, the assessment of quality includes judgments about the methods used for developing the guidelines, the content of the final recommendations, and the factors linked to their uptake.
Guidelines topic: In the ADAPTE Manual and Resource Toolkit for Guidelines Adaptation document, the topic refers to the theme of the guidelines, as described in the guidelines title, for a targeted population (disease and patients) and intervention. The purpose, the audience, and the setting intended for the guidelines, although not necessarily explicitly stated in the title, are also part of the topic. A guideline on a given topic may contain more than one health question.
Health question or clinical question or key question: It is a precisely described health issue (e.g. clinical, professional practice or public health) relating to the topic of the guidelines? Guidelines may include one or more questions.
Implementation: Implementation includes methods to promote the uptake of research findings into routine healthcare in both clinical and policy contexts and hence to improve the quality and effectiveness of healthcare. It includes the study of influences on healthcare professional and organizational behaviour.
Intra-class correlations: Intra-class correlations provide a measurement of the extent to which two or more raters agree when rating the same set of things. It is a reliability index and is typically a ratio of the variance of interest over the sum of the variance of interest plus error.
Recommendation: Recommendation is any statements that promote or advocate a particular course of action in clinical care.
Stakeholder: A stakeholder is an individual, group and/or organization with a stake in your decision to implement a guideline. Stakeholders include individuals or groups who will be directly or indirectly affected by the implementation of a guidelines.
Source guidelines: In the ADAPTE Manual and Resource Toolkit for Guidelines Adaptation document, source guidelines refer to those guidelines selected to undergo assessment of quality, currency, content, consistency, and acceptability/applicability and upon which an adapted guidelines may be based.
- Introduction
➡️Diagnosis of Diabetic Ketoacidosis (DKA): (ISPAD 2022)
▪️ Clinical manifestations of DKA:
o Dehydration
o Tachypnea, rapid deep, sighing (Kussmaul’s) respiration
o Nausea, vomiting without diarrhea, and abdominal pain that may mimic an acute abdominal condition
o Confusion, drowsiness
Not all children or caregivers give history of classic symptoms of diabetes (polyuria, polydipsia) at the time of diagnosis of DKA, and other symptoms are non-specific (eg, weight loss, fatigue, vomiting, abdominal pain). Therefore, fingerstick blood glucose measurements should be considered for all children presenting with rapid breathing or with vomiting and abdominal pain without diarrhea.
▪️ The biochemical criteria for the diagnosis of DKA are:
o Hyperglycemia : blood glucose ≈200 mg/dl
o Venous pH < 7.3 or serum bicarbonate < 18 mmol/L (C)
o Ketonemia (blood ß-hydroxybuyrate ≥3 mmol/L if available, a sensitive indicator of DKA) (C) or Moderate or large ketonuria. Urinary ketones must be read 15 seconds after stick is dipped.
▪️ The Severity of DKA is categorized by the degree of acidosis: (ISPAD 2022)
o Mild: venous pH < 7.3 or serum bicarbonate <18 mmol/L
o Moderate: pH < 7.2 or serum bicarbonate <10 mmol/L
o Severe: pH < 7.1 or serum bicarbonate <5
mmol/L
▪️Hyperglycemic Hyperosmolar State (HHS): (ISPAD , 2022)
- Plasma glucose concentration above 600 mg/dl
- Venous pH > 7.25; arterial pH > 7.30 (arterial sample is not necessary)
- Serum bicarbonate >15 mmol/L
- Small ketonuria, absent to mild ketonemia
- Effective serum osmolality >320 mOsm/kg
- Obtundation, combativeness, or seizures (in approximately 50%)
Formerly called Hyperosmolar non-ketotic coma, it is
characterized by extremely elevated serum glucose concentrations and
hyperosmolality without significant ketosis or acidosis. HHS manifests with
gradually increasing polyuria and polydipsia that may go unrecognized resulting
in profound dehydration and electrolyte losses at the time of presentation. HHS
may occur in children with type 2 diabetes, type 1 diabetes, cystic fibrosis,
and in infants, especially those with neonatal diabetes. Medications such as
corticosteroids and atypical antipsychotics can precipitateHHS.
DKA and HHS may overlap and particular care in the assessment is needed to
diagnose such a condition so that modification in management can be done to
address the associated biochemical disturbances.
Hyperosmolar Hyperglycaemic State (HHS) requires different treatment. Differences in treatment strategy between HHS and DKA include the volume of fluid administered, the timing of insulin administration, and monitoring of the decline in corrected serum sodium concentration.
➡️Causes of DKA (severe insulin deficiency and increased level of counter-regulatory hormones):
▪️ In newly diagnosed patients, DKA is frequently the consequence of a delay in diagnosis (E)
▪️ In children with established diabetes most cases are due to insulin omission (especially basal component) or interruption of insulin delivery in children using insulin pumps. A minority of DKA cases in these children are caused by infection (mostly avoidable if sick day rules are followed).
▪️ Simply eating high carbohydrate diet does NOT cause DKA.
➡️Therapy of DKA
The child with DKA should receive care in a unit that has:
▪️ Experienced nursing (with one-to-one nursing) and medical staff trained in pediatric DKA management who are available to perform meticulous monitoring until DKA has resolved.
▪️ Clinical practice guidelines. Staff should have access to clinical practice guidelines in written or electronic format.
▪️ Access to a laboratory that can provide frequent and timely Lab results.
▪️ Whenever possible, a
specialist/consultant pediatrician with expertise in the management of DKA
should direct patient management. (E)
▪️ Goals of therapy:
- To correct dehydration
- To correct acidosis (caused mainly by volume depletion followed by insulin deficiency and increase in ketone bodies, free amino acids and free fatty acids in blood. Lactic acidosis due to tissue hypoperfusion may also contribute to the acidosis)
- To reverse ketosis
- To gradually restore hyperosmolality and blood glucose concentration to near normal
- To monitor for acute complications
- To identify and treat any precipitating event.
Estimation of the degree of dehydration in DKA is imprecise (dehydration in DKA is hyperosmolar dehydration) and may vary among examiners.
- Mild: pH < 7.3 or serum bicarbonate <18 mmol/L.
- Moderate: pH < 7.2 or serum bicarbonate <10 mmol/L.
- Severe: pH < 7.1 or serum bicarbonate <5 mmol/L.
Laboratory measures have been found to be better predictors of dehydration severity than clinical signs. These include:
o Higher serum urea nitrogen (>20 mg/dl)
o Lower pH (<7.1)
- Wide anion gap
- ≥10% dehydration is suggested by the presence of weak or impalpable peripheral pulses, hypotension or oliguria.
Mild DKA assumes 5%, moderate DKA 7% and severe DKA 10% dehydration.
Hypertension occurs in 12% of children with DKA at presentation and develops during treatment in an additional 16%. It should not be considered sign for cerebral injuy in the absence of other signs.
➡️Causes of Morbidity and Mortality of DKA (Complications): (ISPAD 2022)
• Mortality is mainly due to cerebral injury.
• It is infrequent to have permanent severe neurological sequelae resulting from DKA related brain injuries.
• Renal tubular damage (RTD) and acute kidney injury (AKI) are more common in children with severe acidosis and volume depletion (AKI Stage 1, 2, or 3 is defined by serum creatinine 1.5, 2, or 3 times estimated baseline creatinine). They are managed by restoration of fluid volume and correction of acidosis.
➡️Other complications include:
• Hypokalemia * (potassium may decrease rapidly during treatment, predisposing to cardiac arrhythmias. Severe hypokalemia below 2.5 meq/l is an independent marker of mortality). Potassium replacement is required regardless of the serum potassium concentration, except if renal failure is present).
• Hypoglycemia
• Hypocalcemia, hypomagnesemia
•Severe hypophosphatemia *
• Hyperchloremic acidosis
• Hypochloremic alkalosis
• Other central nervous system complications including cerebral venous sinus thrombosis, basilar artery thrombosis, intracranial hemorrhage, cerebral infarction
• Deep venous thrombosis *
• Pulmonary embolism *
• Rhinocerebral or pulmonary mucormycosis
• Aspiration pneumonia*
• Pulmonary edema *
• Adult respiratory distress syndrome (ARDS)
• Prolonged QTc
• Pneumothorax, pneumomediastinum and subcutaneous emphysema
• Rhabdomyolysis *
• Ischemic bowel necrosis
• Renal failure*
• Acute pancreatitis *
*= These are more frequent in HHS.
The aim of this adapted clinical practice guideline (CPG) is to provide evidence-based recommendations for the management of diabetic ketoacidosis in children and adolescents.
These recommendations were adapted from the relevant International Society for Pediatric and Adolescent Diabetes (ISPAD) Clinical Practice Consensus Guidelines 2018 and 2022 CPG(s) using a formal methodology for CPG adaptation: the Adapted-ADAPTE.
➡️Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of DKA in age group 1 to 18 years old. It provides guidance to primary health care providers, pediatricians and specially trained nurses. The guidelines aimed to for use by various healthcare providers in the field namely paediatricians, diabetologists or endocrinologists, and intensivists
This version of the guideline includes recommendations and good practice statements for:
· Diagnosis and management of DKA.
• Prevention of Cerebral edema and HHS.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: diabetic ketoacidosis, pediatrics
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 2 guidelines:
1- The International Society for Pediatric and Adolescent Diabetes (ISPAD) Clinical Practice Consensus Guidelines 2018 and 2022
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement.
➡️Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of endocrinologist
The main functions of the clinical panel were adolopment of DKA diagnosis and management Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinician’s subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as eminent international reviewers.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of
the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations.
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
➡️GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
➡️Steps of implementing DKA diagnosis, treatment, and prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for management of DKA.
3. Arabic Educational materials for nurses and mothers
- Limitations and suggestions for further research needs
Future research recommendations for the management of DKA in children in the Egyptian context could include:
· Future researches should be directed to efficacy of the fluid protocol and the impact of application of the guidelines on the outcome of DKA
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for DKA. in children.
➡️Challenges
· Presence of trained staff (physicians and nurses) skilled to manage DKA cases
· availability of well-equipped ICU
· Access to a laboratory that can provide frequent and timely Lab results.
· distribution of the guidelines to medical care units.
· Lack of trained nurses dealing with emergency cases (unexperienced with IV canulation, fluids preparation)
· Absence of laboratory providing frequent timely results
· unavailability of some preparations like: phosphate to correct hypophosphatemia, half normal saline to be used in hyperosmolarity, and mannitol or hypertonic saline.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for DKA in children:
1.Adherence to DKA Guidelines
· Numerator: Number of children with DKA who received treatment as per guideline recommendations.
· Denominator: Total number of children diagnosed with diabetes
· Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
· Numerator: Total number of hospital stay days for children with DKA
· Denominator: Total number of children admitted with type 1 diabetes
· Data Source: Hospital admission and discharge records.
3. Rate of Readmission
· Numerator: Number of children readmitted with symptoms of DKA within a certain period (e.g., 30 days) after discharge.
· Denominator: Total number of children initially admitted with diabetes
· Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
➡️Updating of the guideline
The EPG Endocrin GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Craig, M. E., Jefferies, C., Dabelea, D., Balde, N., Seth, A., & Donaghue, K. C. (2018). ISPAD Clinical Practice Consensus Guidelines 2018: Definition, epidemiology, and classification of diabetes in children and adolescents. Pediatric Diabetes, 19(Suppl. 27), 7-19. https://doi.org/10.1111/pedi.12773
2. Donaghue, K. C., Wadwa, R. P., Dimeglio, L. A., Wong, J. C., Nadeau, K., & Chiarelli, F. (2022). ISPAD Clinical Practice Consensus Guidelines 2022: Microvascular and macrovascular complications in children and adolescents. Pediatric Diabetes, 23(Suppl. 27), 45-57. https://doi.org/10.1111/pedi.13285
3. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
4. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
5. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
6. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
7. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
8. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
9. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
10. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|
|||||||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|||||
|
Interest identified |
Management plan & decision |
|
||||||
|
Prof. Mona Mamdouh Hassan |
Pediatrics Department, Consultant, Cairo University |
|
||||||
|
Ass. Prof. Amal Gaber Mohamed |
Pediatrics Department, Consultant, Al-Azhar University |
|
||||||
|
Prof. Amany Kamal El-Hawary |
Pediatrics Department, Consultant, Mansoura University |
|
||||||
|
Prof. Amina M. Abdel Wahab |
Pediatrics Department, Consultant, Suez Canal University |
|
||||||
|
Prof. Ashraf A. Elsharkawy |
Pediatrics Department, Consultant, Mansoura University |
|
||||||
|
Prof. Basma Abd-Elmoez |
Pediatrics Department, Consultant, Minia University |
|
||||||
|
Dr. Eman Elshanawany
|
Pediatrics Department, Consultant, Benha University |
|
||||||
|
Prof. Ghada Anwar |
Pediatrics Department, Cairo University |
|
||||||
|
Prof. Hanaa Abdellateef Mohamad |
Pediatrics Department, Consultant, Assiut University |
|
||||||
|
Ass. Prof. Hanan Hassan Aly |
Pediatrics Department, Consultant, Ain Shams University |
|
||||||
|
Prof. Hoda Atwa |
Pediatrics Department, Consultant, Suez Canal University |
|
||||||
|
Prof. Lubna Fawaz |
Pediatrics Department, Consultant, Cairo University |
|
||||||
|
Dr. Mariam Nader Moawad, |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
||||||
|
Dr. Marian Fares Nashed |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
||||||
|
Ass. Prof. Mona Karem Amin |
Pediatrics Department, Consultant, Suez Canal University |
|
||||||
|
Prof. Nora E Badawi |
Pediatrics Department, Consultant, Cairo University |
|
||||||
|
Ass. Prof. Nouran Y Salah El-Din |
Department of Pediatrics, Ain Shams University |
|
||||||
|
Dr. Ramy Saleh Morsy |
Department of Pediatrics, Consultant, Armed Forces College of Medicine |
|
||||||
|
Prof. Randa M. Matter |
Pediatrics Department, Consultant, Ain Shams University |
|
||||||
|
Ass. Prof. Remon M. Yousef |
Department of Pediatrics, Consultant, Fayoum University |
|
||||||
|
Prof. Sabry M Ghanem |
Department of Pediatrics, Consultant, Al-Azhar University |
|
||||||
|
Prof. Safinaz El Habashy |
Department of Pediatrics, Consultant, Ain Shams University |
|
||||||
|
Ass. Prof. Shaymaa Elsayed Abdel Meguid |
Department of Pediatrics, Consultant, Alexandria University |
|
||||||
|
Prof. Wiam Al Farouk Younis |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
||||||
|
Dr. Hamed Khaled Khalifa |
M.B.B.Ch Misr University for Science and Technology. House Officer, Pediatrics Department, Cairo University.
|
|
||||||
|
Prof. Tarek Omar |
Pediatrics Department, Faculty of Medicine, Alexandria University, Alexandria, Egypt Alexandria Center for Evidence-Based CPG Consultancy Board, EPG, Egypt |
Professor of Pediatrics, Methodology Supervision Subgroup |
||||||
|
|
|
None |
Not Applicable |
|
||||
|
Guideline Adaptation Group (Methodology Subgroup) |
|
|||||||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
||||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
||||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
||||
|
External Review Group |
|
|||||||
|
Prof. Ghada Anwar |
Pediatrics Department, Cairo University/ Egypt |
|
||||||
|
Prof. Mona Hafez |
Pediatrics Department, Cairo University/ Egypt |
|
||||||
|
Prof. Mona Salem |
Pediatrics Department, Ain Shams University/ Egypt |
|
||||||
|
Prof. Nermin Salah |
Pediatrics Department, Cairo University/ Egypt |
|
||||||
|
Hesham El Hefnawy |
Prof. of diabetes and endocrinology, former dean of the National Institute of Diabetes and Endocrinology, Cairo, Egypt. Head of National Committee of non-communicable disease |
|
||||||
|
External Reviewer for methodology |
|
|||||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
|
|||||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
|
|||||||
|
International Peer Reviewers |
|
|
|
|||||
|
|
|
None |
Not Applicable |
|
||||
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for "Guideline for the prevention and management of DKA’’" on PubMed are: DKA, pediatrics, diagnosis, management
|
Pediatrics Department, Cairo University/ Egypt |
|
|
Prof. Mona Hafez |
Pediatrics Department, Cairo University/ Egypt |
|
Prof. Mona Salem |
Pediatrics Department, Ain Shams University/ Egypt |
|
Prof. Nermin Salah |
Pediatrics Department, Cairo University/ Egypt |
|
Hesham El Hefnawy |
Prof. of diabetes and endocrinology, former dean of the National Institute of Diabetes and Endocrinology, Cairo, Egypt. Head of National Committee of non-communicable disease |
Appendix 1. List of the websites and databases we searched:
CPG databases and libraries:
1. Guidelines International Network (GIN) International Guidelines Library.
https://g-i-n.net/international-guidelines-library/
2. ECRI Guidelines Trust (USA). https://guidelines.ecri.org/
3. National Institute of Clinical and Health Excellence (NICE) UK. http://www.nice.org.uk/guidance/
4. Scottish Intercollegiate Guidelines Network (SIGN) UK. http://www.sign.ac.uk/guidelines/
5. EBSCO DynaMed (USA) https://www.dynamed.com/ (subscription required)
Bibliographic databases:
1. PubMed/ MEDLINE https://pubmed.ncbi.nlm.nih.gov/
2. Embase https://www.embase.com/landing?status=grey (subscription required)
3. CINAHL https://www.ebsco.com/products/research-databases/cinahl-complete
Specialized professional societies:
1. American Academy of Pediatrics (AAP) https://www.aap.org/
2. Canadian Paediatric Society (CPS) https://www.cps.ca/
Royal College of Paediatrics and Child Health (RCPCH) https://www.rcpch.ac.uk/
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for DKA management
|
CPGs AGREE II DOMAINS |
CPG1 |
CPG2 |
CPG3 |
CPG4 |
CPG5 |
|
D1: Scope & Purpose |
60% |
53% |
61% |
99% |
67% |
|
D2: Stakeholder Involvement |
38% |
43% |
36% |
94% |
69% |
|
D3: Rigour of Development |
15% |
9% |
26% |
99% |
60% |
|
D4: Clarity & Presentation |
65% |
68% |
78% |
100% |
94% |
|
D5: Applicability |
9% |
7% |
20% |
96% |
74% |
|
D6: Editorial Independence |
56% |
56% |
4% |
90% |
71% |
|
OA 1 |
33% |
38% |
38% |
100% |
75% |
|
OA 2 (Recommend for use) |
No (4) |
No (4) |
No (4) |
Yes (4) |
Yes (3) Yes with modif. (1) |
|
This table uses the AGREE II Domain Score Color codes (< 40% red; > 41 – 70% yellow; > 71 % green)
|
|||||
Annex Table 3. Annex
Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Implementation tools:
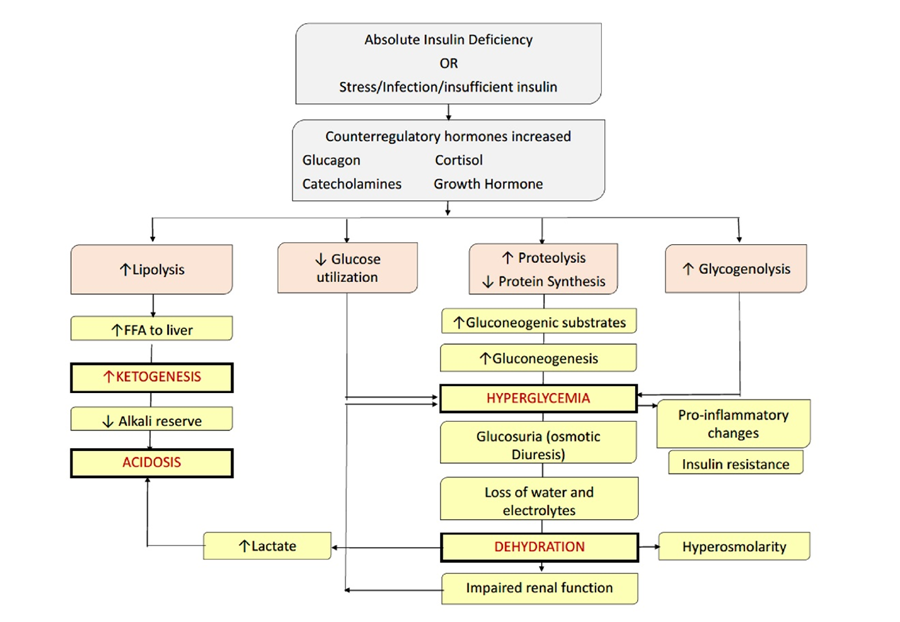
Figure (1): Pathophysiology of diabetic ketoacidosis.
Copyright, 2006 American Diabetes Association. From Diabetes Care,
Vol. 29, 2006:1150–9. ISPAD 2018
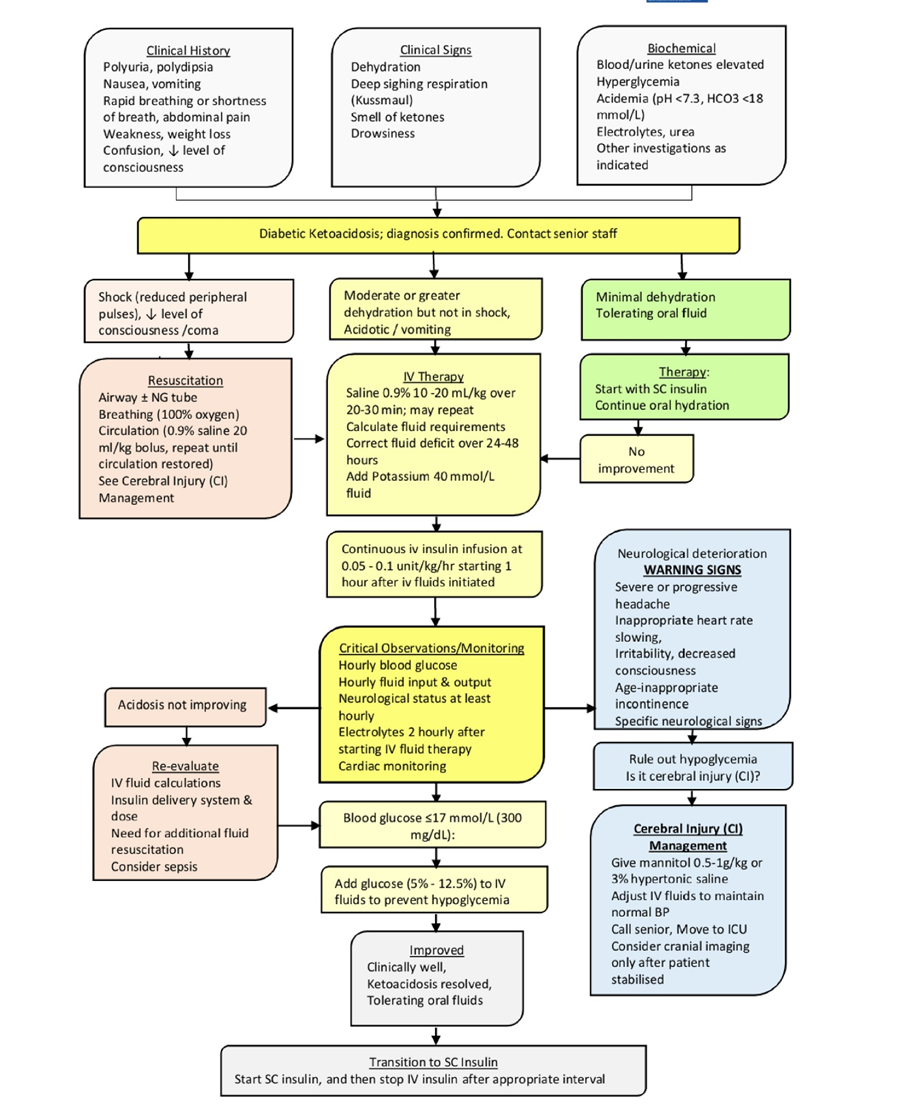
Figure (2): Algorithm for the management of DKA
Pinhas-Hamiel O, Sperling M. Diabetic ketoacidosis. In: Hochberg Z, ed. Practical Algorithms in Pediatric Endocrinology. 3rd, revised edition ed. Basel: Karger; 2017:112-113
· Figure (3): ECG findings in hypo- and hyperkalemia.
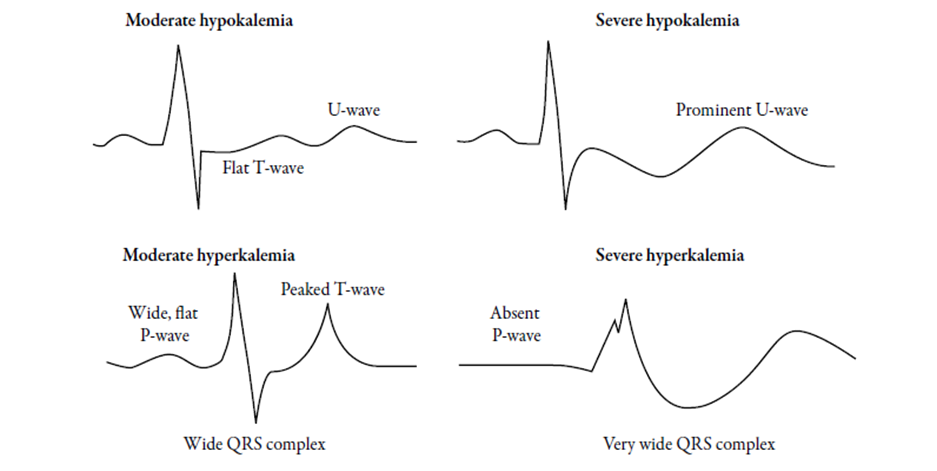
Table (2): Losses of fluids and electrolytes in diabetic ketoacidosis and maintenance requirements in normal children.
(ISPAD clinical practice consensus guidelines 2018, Pediatric Diabetes October 2018; 19 (Suppl.27): 155-177).

Table (3): Glasgow Coma Scale
Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale Lancet 1974: 2: 81–4

Table (4): An alternative example of fluid volumes for the subsequent phase of rehydration. Diabetic Ketoacidosis and Hyperglycemic Hyperosmolar State: A Consensus Statement from the International Society for Pediatric and Adolescent Diabetes. Pediatric Diabetes 2018; 19 (Suppl 27): 155
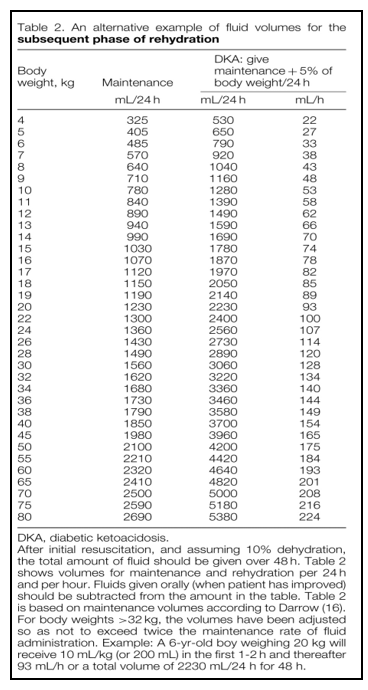
Table (5): Determining Clinical cerebral edema risk.
Muir AB, Quisling RG, Yang MC, Rosenbloom AL. Cerebral Edema in Childhood Diabetic Ketoacidosis: Natural history, radiographic findings, and early identification. Diabetes Care. 2004; 27 (7):1541-1546.
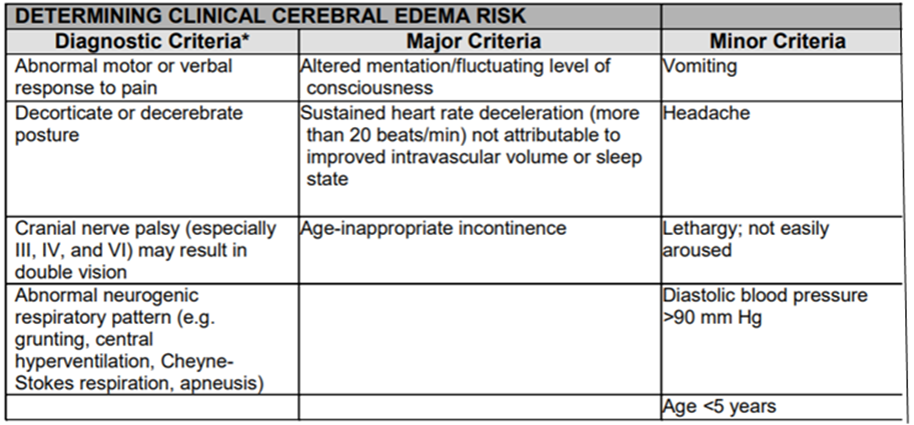
*One diagnostic criterion, or two major criteria, or one major and two minor criteria have a sensitivity of 92%, a specificity of 96% and a false positive rate of only 4% for the early recognition of DKA-related cerebral edema; early enough to allow for effective treatment.
Figure (4): Agree II Score for ISPAD Clinical Consensus Guidelines 20181

Follow up sheet for DKA ,DEMPU1 Table (6)
Instructions in Arabic for Family and Patient Education
From DEMPU Diabetes Education Booklet, Cairo University
كتيب التقيف لسكر الأطفال و الشباب بوحدة السكر و الغدد الصماء و الميتابوليزم، مستشفى الأطفال- جامعة القاهرة، اعداد د سحر أبو الليل، مراجعة أ.د.نرمين صلاح.
رقم ايداع2008/ 19133
: ص 28-30





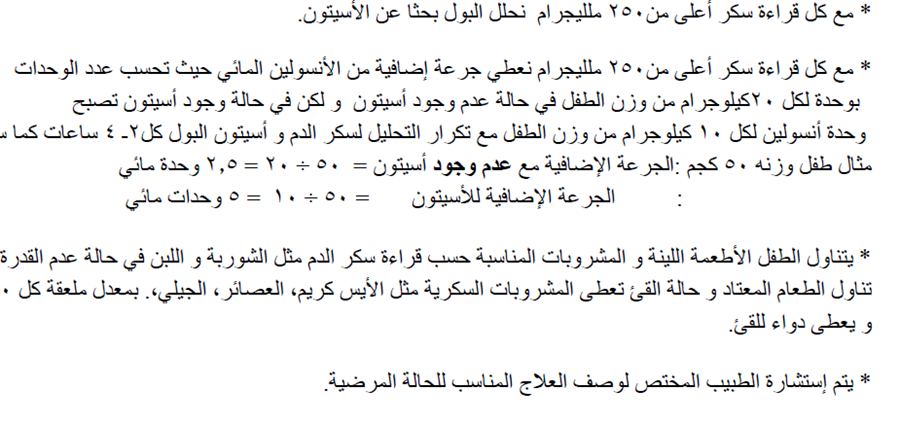
Annex Table 4. The RIGHT-Ad@pt checklist
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☒ Yes ☐ No ☐ Unclear |
61 |
|
|