Female Reproductive surgery in farm animals
| Site: | EHC | Egyptian Health Council |
| Course: | Theriogenology Guidelines |
| Book: | Female Reproductive surgery in farm animals |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 7 April 2025" Download Guideline
- Acknowledgment
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The Rapporteur of the Committee: Prof. Mohamed Mohamedy Ghanem.
Scientific Group Members:
Prof. Nabil Abd Elgaber, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby. Dr. Mohamed Elsharkawi, Prof. Gamal Sosa, Dr. Naglaa Radwan, Dr. Hend Elsheikh
Authors: Prof. Gamal A.M. Sosa, Prof. Ashraf Aldesoky Shamaa, Prof. Ahmed R.M. H. Elkhawagah, Dr Ahmed S Sosa.
- The female reproductive surgery in farm animals includes
1- Cesarean section
2- Ovariectomy
3- Caslickʾs operation
4- Perineal lacerations
1- Cesarean section
· Cesarean section (CS) involves the fetal extraction from the dam through a surgical incision in the abdominal wall and the uterus [1].
· It is commonly indicated in the cases of dystocia [11] which may be attributed to maternal or fetal causes.
· The maternal causes of dystocia include irreducible uterine torsion, hydropsy, narrow pelvis/pelvic fracture, incomplete cervical dilation, extra-uterine pregnancy, uterine inertia, uterine rupture, urinary bladder carcinoma, irreducible prolapse mass, and bicornual pregnancy. While the fetal causes include fetal abnormalities (hydrocephalus, fetal ascites, anasarca, cleft palate), fetal monsters, fetal mal-disposition, fetal oversize/emphysema, mummified fetus, and macerated fetus [10, 32].
· The cesarean section has several disadvantages including poor dam survival rates and poor fertility due to the increased incidence of peritonitis that leads to uterine adhesions and invariably results in animal death or infertility [1, 9].
· The rate of success of CS is affected by several factors including animal species, fetal viability, asepsis, and surgical technique and approaches [19].
The surgical approaches of cesarean section in different animals:
➡️1- Surgical approaches of cesarean section in cattle
There are different cesarean approaches in cattle [28] including;
1- Standing left paralumbar laparotomy.
2- Standing right paralumbar laparotomy.
3- Recumbent left paralumbar laparotomy.
4- Recumbent right paralumbar laparotomy.
5- Recumbent ventral midline laparotomy.
6- Ventral paramedian laparotomy.
7- Ventrolateral laparotomy.
8- Standing left oblique laparotomy.
The selection of the proper approach determines the anesthetic application technique and depends on the cow's health condition, type of dystocia, the surgeon’s preference, and availability of assistants [28]. The most common technique is the standing left paralumbar celiotomy [1, 28].
For details on methods of anesthesia, refer to Veterinary Anesthesia: - Acknowledgement | EHC | Egyptian Health Council
1- Standing left paralumbar laparotomy
A vertical incision is made in the middle of the left paralumbar fossa, approximately 10 cm ventral to the transverse processes of the lumbar vertebrae and continuing ventrally (Fig. 1) [28].
It has several advantages including its simplicity and easy closure of the abdominal wall, in addition, the rumen prevents small intestines evisceration during the surgery [19]. However, its disadvantage includes the difficulty of lifting the uterus and calf to the paralumbar incision [28].
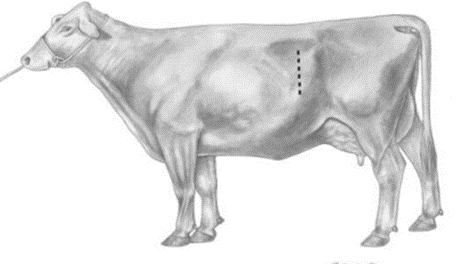
Figure (1). Standing left paralumbar celiotomy.
2- Standing right paralumbar laparotomy
· The surgical incision is made on the right paralumbar fossa in the same way as in the left paralumbar laparotomy.
· It is indicated in cases of a large calf in the right horn, a cow with severe adhesions on the left side due to repeated caesareans, and a cow with hydropsy [20].
· The main disadvantage of this approach is the difficulty of keeping the intestines in the peritoneal cavity with the right paralumbar approach [19].
3- Recumbent left paralumbar laparotomy
· The cow is casted and placed in right lateral recumbency, and the incision is made more ventral than in the standing left paralumbar laparotomy [15].
· This approach has several disadvantages including the more assistance required, the difficulty in exteriorization of the uterus, and the delayed closure of the incision due to increased tension on the muscle layers [28].
4- Recumbent right paralumbar laparotomy
It is similar to that of recumbent left paralumbar celiotomy. It has an additional complication which is the difficulty to keep the intestines in the peritoneal cavity [28].
5- Recumbent ventral midline laparotomy
· The cow is positioned in dorsal recumbency and the incision is performed 5–7 cm caudal to the umbilicus and extended caudally as required (Fig. 2) [28].
· It is indicated in the case of an emphysematous fetus [28]. It has several disadvantages including its limited application in older cows due to the increased size of the udder and the presence of increased ventral vasculature, in addition to the increased tension on the incision [1, 28, 29].
6- Recumbent ventral paramedian laparotomy
· The cow is put in dorsal recumbency and the incision is made 5 cm lateral and parallel to the linea alba and medial to the milk vein (Fig. 2).
· It has an advantage compared to the midline incision in that the abdominal wall closure is more secure [28].
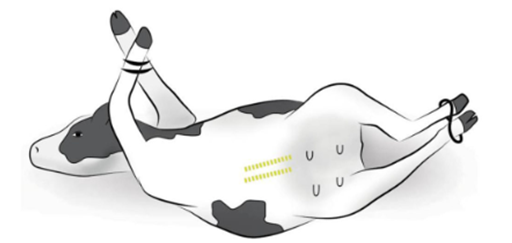
Figure (2). Recumbent ventral midline and paramedian celiotomy
7- Ventrolateral celiotomy
· The cow is positioned in the right lateral recumbency. A curved incision is made 20 cm dorsal to the udder, medial to the stifle fold, and about 50 cm cranio-ventrally (Fig. 3) [28].
· It has several advantages including the avoidance of vascularized musculature of the flank, does not require dorsal recumbency, and allows excellent exteriorization of the uterus. It is suited for older beef and dairy cows [19].
· The disadvantages are the difficult and less secure closure of the incision due to the increased tension, which may be prone to herniation and evisceration [28].
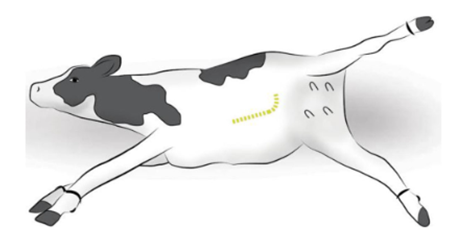
Figure (3) Ventrolateral celiotomy
8- Standing and recumbent left oblique celiotomy
· The incision is made 4-6 cm ventral and cranial to the tuber coxae and is extended cranioventrally toward the caudal rib (Fig. 4) [28].
· It has several advantages including relative accessibility to the apex of the uterine horn, easy exteriorization of the uterus, useful in case of a very heavy calf and the healing of the incision is rapid as the internal abdominal oblique muscle is incised parallel to the muscle fibers [1, 28].
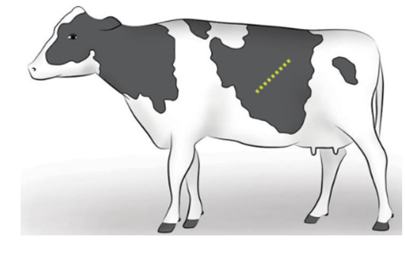
Figure (4) Standing left oblique celiotomy
➡️2- Surgical approaches of cesarean section in small ruminants
CS in the small ruminant can be performed through 2 techniques [6] including:
1. Left paralumbar approach.
2. Ventral midline approach.
The surgical approach chosen must include consideration of the dam and fetal health and the availability of support personnel. The ventral midline approach is preferred when uterine contents may be contaminated [33].
1- Left paralumbar approach.
It is the most common surgical approach in small ruminants [35]. The animal is positioned in the right lateral recumbency. The left flank is clipped and a vertical or slightly oblique 15–25 cm skin incision is made 10 cm below the transverse processes of the lumbar vertebrae centered in the paralumbar fossa [33]. The skin and the cutaneous trunci muscle are incised sharply; the external and internal abdominal oblique muscles are incised with either a scalpel blade or scissors. The uterus incision is made over the greater curvature of the uterus to minimize hemorrhage. The incision can be made directly over a fetal limb or a fetal head. The incision should be long enough to allow for the extraction of the largest portion of the fetus to minimize uterine tears [6].
2- Ventral midline approach.
The animal is positioned in dorsal recumbency and the ventral abdomen from the umbilicus to the cranial udder is surgically clipped. A skin incision is made just cranial to the udder and extended cranially 20–25 cm [6, 33].
➡️3- Surgical approaches of cesarean section in mares
Before cesarean, the mare undergoes a regime of anesthesia as follows; xylazine (0.8 mg/kg bwt, i.v.) followed by diazepam (0.04 mg/kg bwt, i.v.) and ketamine (2.2 mg/kg bwt, i.v.). To counteract the weight of the abdominal contents on the thorax, the animal is kept on halothane or isoflurane and oxygen, with ventilation. Intravenous fluids are administered together with other agents to maintain blood pressure [10]. For details on methods of anesthesia, refer to Veterinary Anesthesia: - Acknowledgement | EHC | Egyptian Health Council
There are several surgical approaches to CS in mare [31]:
1. Ventral midline approach
2. Modified low flank approach
3. Standing flank approaches
1- Ventral midline approach
It is the most frequently used incision in mares [10]. The mare is positioned in dorsal recumbency. A ventral midline incision (25 cm) is made into the abdomen, extending cranially from a point just caudal to the umbilicus.
The uterine horn is exteriorized and large stay sutures are made to stabilize the uterine horn during the procedure; one suture is made near the tip of the uterine horn close to the fetal limb, and one towards the body of the uterus, close to the point of the hocks. The exteriorized horn is isolated using drapes that prevent contamination from uterine contents. An incision is made between the 2 stay sutures through the uterine wall and chorioallantois from the level of the hock to the fetlock. The amniotic membrane is incised and the fetus is pulled from the hindlimbs out of the uterus. The umbilical cord is cut near the foal's body and the ends are clamped. The chorioallantois is separated from the endometrium for approximately 5 cm from its incised edges. Large blood vessels along the uterine incision are ligated. The uterus is sutured in 2 layers (the 1st layer is a simple continuous pattern and the second is a continuous Lembert pattern) using No. 2 polyglycolic acid suture material. The exteriorized uterine horn is rinsed with sterile saline and the contaminated drapes are removed. The abdominal cavity is rinsed with sterile saline and the fluids are removed by suction. The abdomen is closed as follows; Peritoneum and transverse abdominal muscles are sutured together with a double layer of No. 2 polyglycolic acid suture material using a simple interrupted pattern followed by a re-enforcing layer of lock stitch. Internal and external oblique abdominal muscles are sutured together by a lock stitch pattern using a double layer of absorbable suture material (No. 2). The skin incision is closed by an interrupted horizontal mattress pattern with nylon [21].
2- Modified low flank approach
The mare is positioned in the right lateral recumbency [34]. The incision is made in a cranio-dorsal to the caudo-ventral direction to the depth of the external abdominal oblique muscle. A grid approach through the external and internal abdominal oblique muscles and the transverse abdominal muscle is used to gain entry to the abdomen. The hysterotomy and fetal extraction proceed similarly to that explained above. Closure of the body wall entails suturing each muscle layer separately before closure of the fascia, subcutaneous tissue, and skin [10].
3- Standing flank approaches
A vertical skin incision (15 to 20 cm) is made approximately 5 cm ventral to the lumbar transverse processes, midway between the 18th rib and the tuber coxae in a cranial-caudal direction [27] (Fig. 5). The incision can be extended ventrally if needed. The incision usually is made on the left side unless surgical embryo transfer techniques are being performed. The skin and subcutaneous tissues are sharply incised. The external abdominal oblique is separated along the direction of its fibers. The internal abdominal oblique fibers are separated bluntly. The transversus muscle is sharply incised, and the peritoneum can either be sharply incised or bluntly separated.
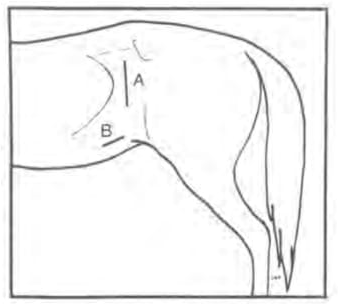
Figure (5). Diagrammatic representation of the left flank in a standing mare. (A): vertical skin incision, (B): oblique skin incision
2- Ovariectomy
Ovariectomy is the surgical removal of the ovary in case of large ovarian tumors and adhesions involving the ovary [13]. It can be performed via a flank or ventral abdominal approach or more recently through standing laparoscopic ovariectomy [3, 25, 26, 30].
1- Surgical approaches of ovariectomy in cattle
There are two different ovariectomy approaches in cattle
1- The left flank approach
A vertical skin incision (30 cm) is made over the left paralumbar fossa extending. All muscular layers were incised with the scalpel. The peritoneum was held with forceps, punctured with the scalpel, and incised with scissors proximally and distally. The uterus and the ovaries are grasped and located 5 cm cranial to the pelvic wall. Lidocaine (2%) is sprayed over the ovarian pedicle and the preformed knot ligates the ovary. After ligation, the ovary can be removed using curved Mayo scissors, and the ovarian pedicle is inspected for hemorrhage. The abdominal is sutured as in the cesarean section [25].
2- Laparoscopic ovariectomy
The cow is restrained in standing stocks and sedated with an I.M. injection of 0.05 mg/kg BW xylazine. The left paralumbar fossa from the transverse processes of the lumbar vertebrae to the level of the stifle and from the second last rib to the level of the tuber coxae was clipped, shaved, and aseptically prepared. Local infiltration with 10 ml of 2% lidocaine is performed in the subcutaneous and muscular tissue of the portal sites. The laparoscopic portal is made at the ventral angle of the paralumbar fossa, approximately 10 cm ventral to a line horizontally drawn through the tuber coxae and at the transition from the middle to the caudal third of the flank (Fig. 6). A vertical skin incision (2cm) is made at this site, and a security obturator is advanced through the musculature and peritoneum until air influx could be heard entering the abdominal cavity. A trocar-cannula unit (20 cm) with an air valve is introduced through the obturator and connected to a carbon dioxide insufflator. The laparoscope is inserted through the trocar. The abdomen is insufflated with carbon dioxide to an intraabdominal pressure of 5–10 mmHg. The two instrument portals, located 20 and 30 cm ventral to the tuber coxae (Fig. 6), are placed under endoscopic guidance. The uterus is identified and the ipsilateral uterine horn is grasped at its greater curvature with grasping forceps (43 cm long with locking handles). The uterine horn is gently pulled cranially, and the ovary within the bursa becomes visible and is grasped with claw forceps (43 cm long with locking handles). The grasping forceps is replaced by an injection cannula through which 20-30 ml 2% lidocaine is injected into different locations inside the mesovarium and mesosalpinx. The injection cannula is replaced with bipolar cauterizing forceps which is placed on the mesovarium 0.5 cm proximal to the ovary. The mesovarium is cauterized until a white discoloration, shrinkage or blister formation can be observed (Fig. 7). The proper ovarian ligament, is then cut with serrated curved scissors (Fig. 8) grasping forceps holding the ovary were pulled to the abdominal wall. The carbon dioxide is allowed to escape through the portals, the peritoneum and external oblique muscle are closed with simple interrupted sutures of five metric polydioxanones, and the skin is closed with simple interrupted sutures of four metric polyamide. The cows should undergo daily clinical and sonographic examinations for the first 5 days post-operatively and then twice a week until 30 days after surgery [3].
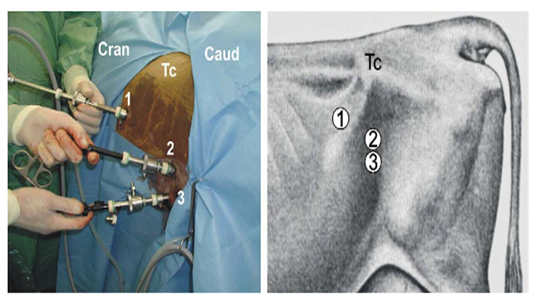
Fig. 6. Portals for laparoscopic ovariectomy via the left flank in a standing cow. TC: tuber coxae, 1: endoscope portal, 2: first instrument portal, 3: second instrument portal [3].
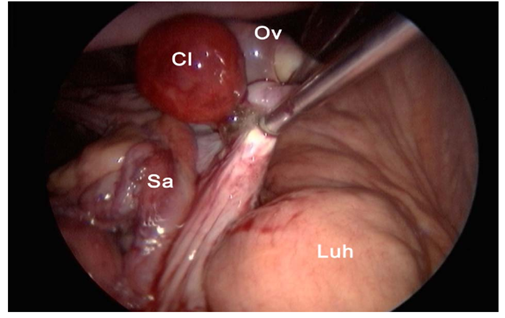
Fig. 7. Cauterization of the mesovarium using cauterization forceps. Cl: corpus luteum, Ov: ovary, Sa: salpinx, Luh: left uterine horn [3].
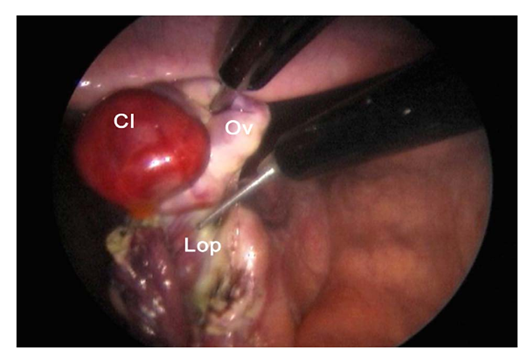
Fig. 8. Transection of the proper ovarian ligament (lop). Cl: corpus luteum, Ov: ovary [3].
2- Surgical approaches of ovariectomy in small Ruminants
There are two different ovariectomy approaches in small ruminants.
1- Ventral midline approach
Ovariectomy is easily performed on the anesthetized animal placed in dorsal recumbency. A small incision (6- 8 cm) is made in the ventral midline just cranial to the udder and continues into the abdominal cavity. The urinary bladder and the uterus are recognized and the uterine horns are exteriorized by gentle traction. For hysterectomy or ovariohysterectomy, the mesometrium is transected after ligation of small blood vessels. The uterus is transected at the level of the body. A circumferential trans-fixation ligature of absorbable suture material is placed close to the cervix. If the remaining portion of the uterine body is large, it should be closed with an inverting suture pattern before replacing it in the abdomen. The vasculature supplying the ovary and horn on one side are ligated and transected. The remaining uterus is closed with an inverting pattern. Some advocate a two-layer uterine closure [33].
2- Laparoscopic approach
Laparoscopy is usually performed on the sedated animal in dorsal recumbency on a cradle that can be tilted. Animals should be fasted for at least 12 hours to reduce rumen fill and the possibility of regurgitation. An area (25 cm x 25 cm) cranial to the mammary gland is prepared by clipping and surgical scrubbing. Three portals are necessary: one each for the laparoscope, a manipulation instrument, and special instruments (insemination gun, suture material). The site of the desired portals is infiltrated with a local anesthetic before introducing a trocar and cannula. For simple technique, the portals are created by making a small skin incision to allow trocar introduction. Visualizing the abdominal viscera requires insufflation with CO2 and elevating the hindquarters to a 40° angle [33]. The rest of the technique is performed as in cattle.
3- Surgical approaches of ovariectomy in mares
There are two different Ovariectomy approaches in mare
1- Flank Approach
The flank ovariectomy approach can be performed in the standing or recumbent position. To perform a standing flank laparotomy, the mare is sedated, and restrained in standing stocks, and the tail is wrapped. The recumbent flank technique requires general anesthesia, resulting in a greater cost and potentially increased risk to the mare. It is generally only used for unilateral ovariectomy, as it offers easy access to only one ovary [5]. The mares are placed in lateral recumbency so that the ovary to be removed is upper-most [16].
The paralumbar fossa is clipped, aseptically prepared, and draped. Regional anesthesia in the form of an inverted L-block or local infiltration of the proposed incision site is required. The incision is started 5 cm ventral to the lumbar transverse processes between the 18th rib and the tuber coxae and is extended ventrally 10 to 15 cm (Fig. 9). Following incision of the skin and subcutaneous tissues, a grid or modified grid approach is used to incise the abdominal musculature. The grid approach involves separating the external abdominal oblique muscle along the direction of its fibers, whereas a modified grid technique involves incising this muscle along the line of the skin incision. The internal abdominal oblique and transversus abdominis muscles are bluntly separated along the direction of their fibers. The peritoneum may be incised or bluntly perforated to allow access to the abdomen, and the ovary is identified and isolated. In standing mares, the mesovarium is anesthetized by topical administration of anesthetic or by direct injection of anesthetic into the mesovarium. The ovary may be transected within the abdomen or after exteriorization. Following removal of the ovary, the mesovarium is observed for hemorrhage and oversewn if required. Closure of the laparotomy incision is routine. Postoperative pain and discomfort may be observed in association with the flank incision. Incisional swelling and discharge may be noted 24 to 72 hours after surgery [16].
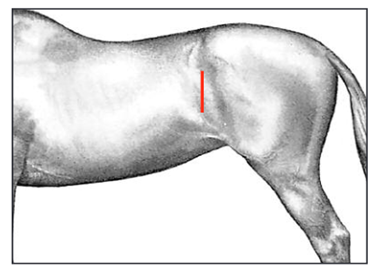
Fig. 9. Position of paralumbar flank incision (in both standing and recumbent horses) for ovariectomy between the 18th rib and tuber coxae [16].
2- Laparoscopic Techniques
Mares are sedated using either xylazine hydrochloride or detomidine hydrochloride in combination with butorphanol tartrate. For bilateral ovariectomy, both paralumbar fossae are prepared for aseptic surgery and draped [16]. The paralumbar fossa is desensitized using regional anesthesia or by direct infiltration of the proposed laparoscope and instrument portal sites [17]. The abdominal cavity can be insufflated with carbon dioxide the trocar–cannula [4]. A 15 mm skin incision is made at the dorsal border of the internal abdominal oblique muscle, and the sharp laparoscopic trocar–cannula is introduced into the abdominal cavity perpendicular to the paralumbar fossa (Fig. 10). The trocar is replaced by the laparoscope, and the ovary is identified. The first portal is made 4 to 8 cm ventral to the laparoscope portal, and a second portal is made 4 to 8 cm ventral to the first (Fig. 11). The mesovarium is infiltrated with a local anesthetic using a long spinal needle. Hemostasis of the mesovarium can be achieved using suture ligatures. The ovary is transected using laparoscopic scissors distal to the site of ligation or coagulation. After removing the ovary, the abdomen is deflated through a laparoscopic cannula. The superficial abdominal fascia and skin at the portals are closed separately [16].
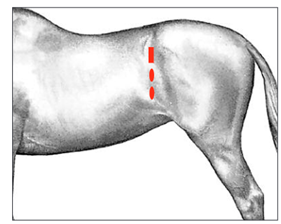
Fig. 10. Position of the laparoscope (rectangle) and instrument portals (ovals) for laparoscopic ovariectomy in the standing horse. The laparoscope portal is positioned between the 18th rib and the tuber coxae, just dorsal to the crus of the internal abdominal oblique muscle. Instrument portals are created 4 to 8 cm ventral to the laparoscope portal [16].
3- Caslickʾs operation
Caslick’s operation is indicated when malformation or inadequate closure of the vulva (Fig. 11) is present. The compromised vulval seal is associated with pneumovagina, or wind sucking, which can predispose ascending contamination and infection. Contamination of the caudal reproductive tract can initially cause vaginitis, which may extend to induce acute endometritis and, in late gestation, will predispose placentitis [14, 22]. The inadequate closure of the vulva is attributed to several causes including trauma during foaling, weight loss, or congenital deviation from the ideal conformation, ageing, and poor body condition score [8, 18]. Moreover, poor vulval conformation is predisposed in mares with a flat-topped croup and a tail setting level with the sacroiliac joint [8, 23].
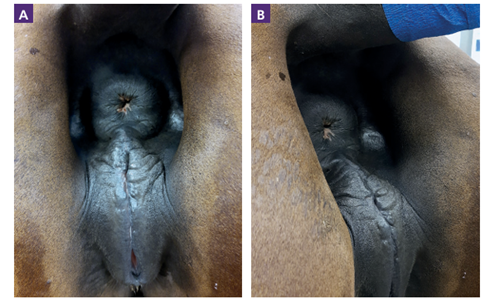
Figure 11. Vulval malconformation in a mare. A cranial sloping of the dorsal commissure of the vulva due to a sunken anus [2]. B: before correction, A: after correction.
The vulval malconformation can be diagnosed and assessed by the Caslick’s index [23]. It is the only way to objectively quantify the degree of abnormality and correlates positively with the likelihood of developing pneumovagina [7, 8, 23]. It is calculated by multiplying the effective length of the vulva in centimeters by the angle of declination of the vulva in degrees [7, 23]. A value of >150 is considered indicative of the need for a Caslick’s vulvoplasty [8, 24]. The need for a Caslick’s operation is suggested by a positive wind sucker test (parting the vulval labia and listening to the sound of air rushing into the vagina), which indicates an in competent vestibulovaginal fold, or the detection of air in the vagina and uterus during per rectum ultrasound examination [8].
Procedures of Caslick’s operation:
Caslick’s operation should be performed on the standing, restrained mare under local anesthesia. Adequate restraint can be achieved by placing the mare in examination stocks, using a twitch and/or by positioning the back-end of the mare just inside a stable door (Bradecamp, 2011). The mare’s rectum should be emptied, with the tail bandaged and held or tied to one side. The perineal region should then be gently cleaned with disinfectant soap such as povidone iodine soap, dried with paper towels or cotton, and prepared with a minimally-irritant antiseptic solution (0.5% chlorhexidine or 30% alcohol). About 15–20ml of local anesthetic (2% lidocaine) should be injected subcutaneously using a 20-gauge or smaller needle along the mucocutaneous junction of both vulval labia, starting at the most ventral aspect of the proposed vulvoplasty and progressing to the dorsal commissure of the vulva (2), (Fig. 12).
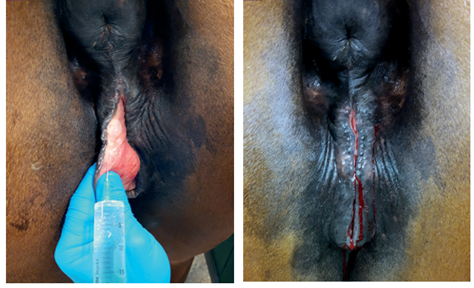
Figure 12. Subcutaneous injection of 2% Lidocaine along the mucocutaneous junction of both labia [2].
The ventral extent of the Caslick’s operation should continue to about 2–3cm below the level of the ischial tuberosities, and in the case of a cranially sloping vulva, to below the level where the vulva orifice assumes a vertical orientation. At least 3cm of the vulva must be left open to allow unconstrained urination. A thin strip of mucosa is removed from the mucocutaneous junction of the labial rim using tissue scissors, to expose a strip of submucosa around 8–10mm wide. To allow removal of the tissue, thumb forceps can be used to grasp a ribbon of mucosa [12]. Care should be taken that the dorsal commissure is not omitted, by creating a wound with an inverted U-shape. The labia are sutured together to oppose the freshly created wounds using either a simple continuous, interrupted or continuous interlocking suture pattern (Bradecamp, 2011). It is advisable to use a suture material of approximate size 0 (4 metric) with a curved, round bodied, sharp needle. Suture material can be either absorbable (Vicryl), or non-absorbable (Ethilon). The suture should pass through the middle to deep part of the incised tissue to ensure adequate healing and minimize the risk of fistula formation (Fig. 13). In addition, the sutures should exit the skin approximately 1cm away from the labial rim to prevent them tearing through. The individual stitches should be placed 0.5–1cm apart to avoid gaps and fistula formation (Fig. 14). For a regular Caslick’s procedure, postoperative topical or systemic antibiotics are not indicated [12]. Aftercare includes avoiding or minimizing rectal palpation and manipulation of the surgical field for 10–14 days. Remove the sutures after 7–14 days if non-absorbable suture material is used. To prevent tearing of the vulva at foaling, a Caslick’s operation should be reopened shortly before parturition (less than 14 days before the estimated date of birth) [12]. Opening requires sufficient local anesthesia and involves a straight cut in the direction of the scar between the opposed labia, using either tissue scissors or a scalpel. Postpartum, a Caslick’s operation can be repeated 1–2 days after parturition [12], or after the foal heat, with assurance that the mare has no fluid in the uterus and is no longer at risk of postpartum metritis.
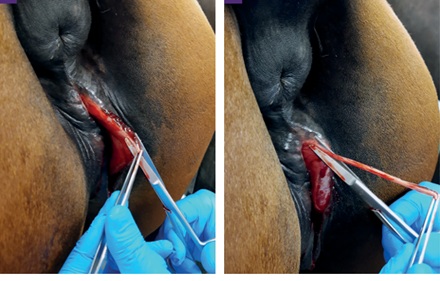
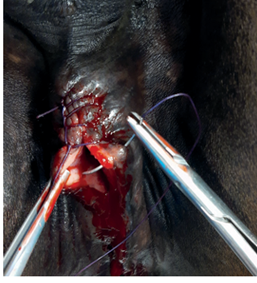
Figure 13. A thin strip of mucosa is removed from the mucocutaneous junction, creating an approximately 8–10mm wide strip of exposed submucosa. The labia are sutured using a simple interrupted, continuous or continuous interlocking suture pattern and exit approximately 1cm from the labial rim (c)
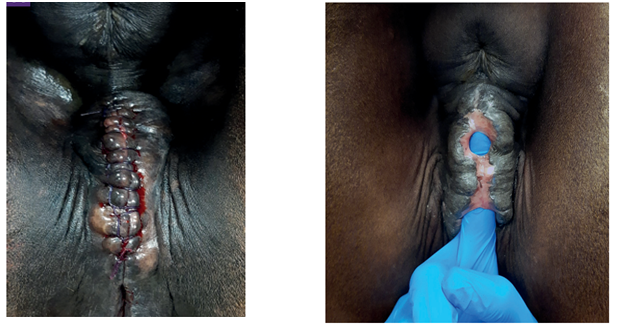
Figure 14. Caslick’s procedure using an interlocking suture technique. Adequate closure can be checked by inserting a finger and applying gentle pressure to the suture from the vaginal aspect.
4- Perineal lacerations
Perineal (rectovaginal) lacerations occur in different species but are more common in mares [36]. Most lacerations arise at the time of foaling due to excessive manipulation during assisted delivery for extraction of the oversized or malpositioned fetus [37]. Depending on their extent, the perineal lacerations have been classified into 3 grades: first, second, and third-grade lacerations as follows [38]:
First-grade laceration: involves the skin of the vulval dorsal commissure and mucosa of the vestibule.
Second-grade lacerations: compromise the closure of the labia and predispose to pneumovagina. Extend through the perineal body, and involve the mucosa and submucosa of the vestibule and the constrictor vulvae muscle. If the perineal body is not reconstructed, the mare will develop a sunken perineum and be predisposed to pneumovagina and urovagina.
Third-grade lacerations: occur penetration of the rectum, perineal body, and anal sphincter, resulting in a complete loss of barrier between the rectum and vestibule (Fig. 15) resulting in a common passage for urination and defecation [38]. The fecal contamination of the reproductive tract results in vaginitis and endometritis. In addition, the loss of the constrictor vulva muscle or a functional rectal sphincter leads to a "wind sucker" during exercise that causes unattractive noise and leads to pneumovagina and further bacterial dissemination to the reproductive tract.
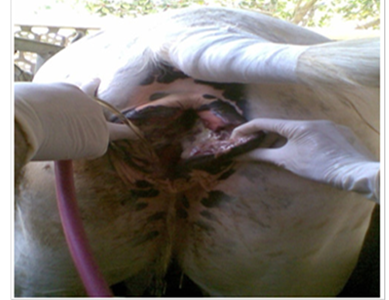
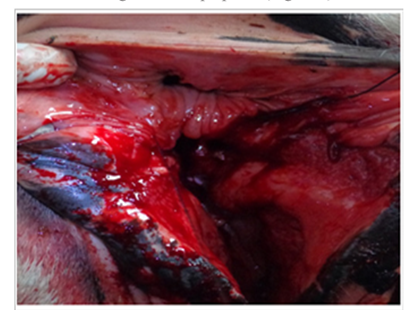
Figure 15. 3rd grade of perineal lacerations with separate shelf for rectum & vagina [38].
Treatment
First-grade injuries do not require surgical intervention. If needed, a Caslickʾs procedure can be performed [37].
Second and third-grade injuries require a Caslickʾs procedure and reconstruction of the perineal body.
In third-grade injuries, the management of the laceration includes 2 parts: immediate treatment and delayed surgical repair [39]. The initial therapy includes daily wound care and cleaning of the contaminated tissues with diluted povidone-iodine solution for ten days to allow subsidization of the perineal swelling. The animal is administered tetanus toxoid along with antibiotics, anti-inflammatory, and anti-histaminic [38].
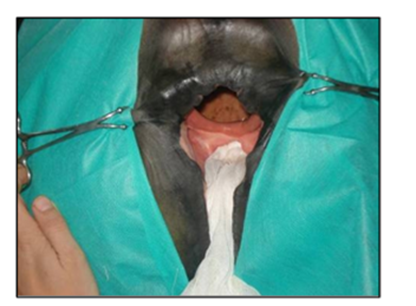
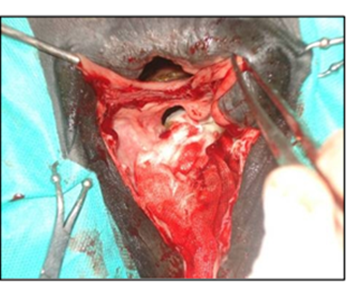
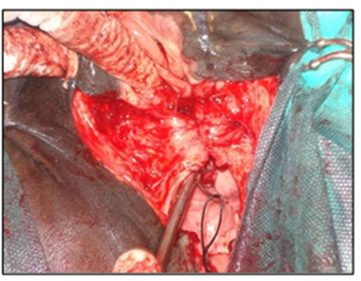
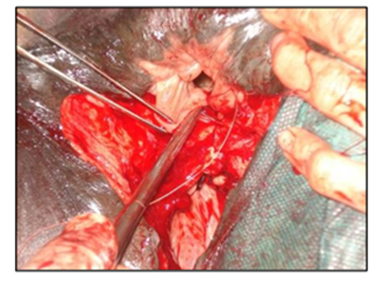
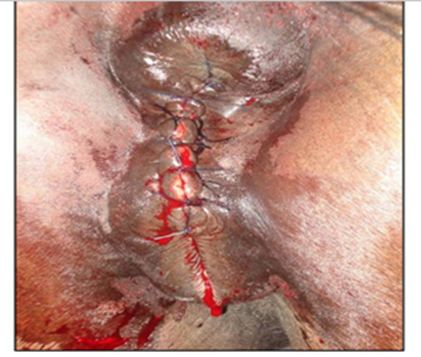
The surgical repair of the perineal body is applied after 10-15 days until the perineal swelling is completely subsided. The mare is controlled in a standing position and sedated with xylazine (5-10ml, slow i.v. route) together with epidural anesthesia with the injection of 5-10ml lignocaine Hcl in between the 1st & 2nd intra-coccygeal space. The perineal area is prepared for the surgery with povidone-iodine and the anus, perineum, and vulvar lips are retracted with towel clamps (Fig. 16). A combination of sharp and blunt dissection is used to separate the tissue into rectal (two-thirds of the thickness) and vestibular shelves (one-third of the thickness). The dissection is continued cranially for 3 to 5 cm and laterally until the tissue shelves can be opposed on the midline without tension (Fig. 17). The septum between the rectum and vagina was sutured by a six-bite pattern using a nonabsorbable suture material (No. 2) (Fig. 18). The rectal mucosa is sutured with simple interrupted suturing using 0 vicryl (Fig. 19). The skin of the perineal body is sutured with non-absorbable suture material (No. 2) in a horizontal mattress pattern (Fig. 20) [37, 38].
|
|
|
|
|
|
|
Figure 20. The skin of the perineal body was closed with non-absorbable suture material (No. 2) in a horizontal mattress pattern [38].
|
|
- References
[1]. Adugna S.A., Kitessa J.D., Feyissa C.T., Adem S.A., 2022. Review on a cesarean section in the cow: Its incision approaches, relative advantage, and disadvantages. Vet Med Sci., 8: 1626–1631.
[2]. Arnold L. and Stout T. 2022. Indications for and how to perform Caslick’s operation in the mare. Equine 6 (1): 6-10.
[3]. Bleul U, Hollenstein K, Kähn W. (2005). Laparoscopic ovariectomy in standing cows. Anim Reprod Sci 90: 193–200.
[4]. Boure L, Marcoux M, Laverty S (1997). Paralumbar fossa laparoscopic ovariectomy in horses with use of endoloop ligatures. Vet Surg26:478–483,
[5]. Colbern GT: Ovariectomy, in McKinnon AO, Voss JL (eds): Equine Reproduction. Philadelphia, Lea & Febiger, 1993, pp 450–455
[6]. Cowley J., Stockler J., Maxwell H. 2023. A review of small ruminant Cesarean section: case selection, surgical techniques, care of the neonates, and postoperative care of the dam. Clinical Theriogenology 15, 9620.
[7]. Crabtree JR, Pycock JF. (2020). Examination of mares and fillies for breeding purposes. UK Vet Equine.4: 77–83
[8]. Dascanio JJ. (2011). External reproductive anatomy. In: McKinnon A, Squires E, Vaala W, Varner D (eds). Equine Reproduction, 2nd ed. Wiley-Blackwell: Chichester; 1577–1581.
[9]. Dhindsa, S.S., Dhaliwal, G.S., Singh, H., Ghuman, S.P.S., 2019. A retrospective study on consequences of cesarean section in bovine. International Journal of Current Microbiology and Applied Sciences, 8(1), 480–484.
[10]. Embertson R.M. (2002). Indications and surgical techniques for cesarean section in the mare. Equine Vet. Educ., 5: 60-64.
[11]. Fesseha, H., Negash, G., Gebrekidan, B., 2020. Caesarean operation in cow due to prolonged pregnancy. Veterinary Medicine—Open Journal, 5(1), 9–13.
[12]. Hendrickson DA. (2013). Equine urogenital surgery. Hendrickson DA, Baird AN, Turner AS (eds). In: Turner and McIlwraith’s Techniques in Large Animal Surgery. 4th edn. Wiley-Blackwell, 156–159.
[13]. Hofmeyr CFB. (1987). The female genitalia. In: Hofmeyr CFB, ed. Ruminant Urogenital Surgery. Ames, Iowa: Iowa State Univer Pr, 1987:122–147.
[14]. Inoue Y, Sekiguchi M. (2017). Vestibuloplasty for persistent pneumovagina in mares. J Equine Vet Sci. 48: 9–14.
[15]. Jos, B.M.N., 2020. Surgical management of dystocia in a cross-bred holstein friesian heifer by caesarean section—A case report. International. Journal of Current Microbiology and Applied Sciences, 9(7), 3928–3931.
[16]. Loesch, D.A. and Rodgerson, DH. (2003). Surgical Approaches to Ovariectomy in Mares. Compendium 25(11):862-871.
[17]. Marien T, De Clercq T, Adriaenssen F, Segers L (2000). Standing laparoscopic ovariectomy in mares using monopolar electrocoagulation and extracorporeal Tayside slipping knot technique. Equine Vet Educ 12(4):202–206
[18]. McCue PM. (2008). The problem mare: management philosophy, diagnostic procedures, and therapeutic options. J Equine Vet Sci. 28:619–626.
[19]. Newman, K.D. & Anderson, D.E., 2005. Cesarean section in cows. Veterinary Clinics of North America—Food Animal Practice, 21(1), 73–100.
[20]. Newman, K.D., 2008. Bovine cesarean section in the field. Veterinary Clinics of North America—Food Animal Practice, 24(2), 273–293.
[21]. Ninu, A.R., Saxena, A.C., Sivanarayanan, T.B., Remya, V., Binsila, B.K., Maiti, S.K., Zama, M.M.S. (20215). Caesarean in mare by Marcenac incision under local anesthesia. IJVR, 16 (1): 117-119
[22]. Papa FO, Melo CM, Monteiro GA et al. (2014). Equine perineal and vulvar conformation correction using a modification of Pouret’s Technique. J Equine Vet Sci. 34: 459–464.
[23]. Pascoe RR. (1979). Observations on the length and angle of declination of the vulva and its relation to fertility in the mare. J Reprod Fertil. 27:299–305.
[24]. Pascoe RR. (2007). Vulvar conformation. In: Samper JC, Pycock JF, McKinnon AO (eds). Current therapy in equine reproduction. Saunders Elsevier: St. Louis (MO). 140–145
[25]. Peiró, J.R., Nogueira, G.M., Nogueira, G.P., Perri, S.H.V., Cardoso, D. (2009). Ovariectomy by left flank approach in prepubertal Nelore (Bos indicus) heifers. The Canadian Journal of Veterinary Research. 73: 237 - 240
[26]. Rodgerson DH, Belknap JK, Wilson DA. (2001). Laparoscopic ovariectomy using sequential electrocoagulation and sharp transection of the equine mesovarium. Vet Surg 30:572–579.
[27]. Ross M.W. (1991). Standing abdominal surgery. Specific indications and preoperative considerations. Veterinary Clinics of North America: Equine Practice, 7 (3): 739-749.
[28]. Schultz, L.G., Tyler, J.W., Moll, H.D., Constantinescu, G.M., 2008. Surgical approaches for cesarean section in cattle. Canadian Veterinary Journal, 49(6), 565–568.
[29]. Sexton, J. W. (1954). Ventral midline cesarian section in the cow. Journal of Iowa State University Veterinarian, 16(2), 81–82
[30]. Shoemaker RW, Read EK, Duke T. (2004). In situ coagulation and transection of the ovarian pedicle an alternative to laparoscopic ovariectomy in juvenile horses. Can J Vet Res 68:27–32.
[31]. Taylor, T.S., Verner, D.D., Martin, M.T., Blanchard, T.L., Scrutchfield, W.L. and Elmore, R.G. (1989) Management of dystocia in mares: uterine torsion and cesarean section. Comp. cont. Educ. pract. Vet. 11, 1265-1273.
[32]. Thangamani, A., Srinivas, M., Prasad, B.C., 2018. Obstetrical operation and epidural analgesia in domestic animals: An overview. Journal of Veterinary and Technology, 7(2), 10–15
[33]. Tibary A. and Metre D.V. (2004). Surgery of the Sheep and Goat Reproductive System and Urinary Tract. In: Farm Animal Surgery, Susan L. Fubini, Norm G. Ducharme (eds), W.B. Saunders, Pages 527-547
[34]. Vandeplassche, M., Bouters, R., Spincemaille, J. and Bonte, P. (1977). Cesarean section in the mare. Proc. Am. Ass. equine Practnrs. 23, 75-80.
[35]. Vivrette S: Cesarean section in goats. In: Morrow DA: editor. Current Therapy in Theriogenology. 2nd edition, Philadelphia, PA; Saunders: 1986:592–593.
[36]. Belknap, J.K. and Nickels, F.A. (1992). A one-stage repair of third-degree perineal lacerations and rectovestibular fistulae in 17 mares. Vet. Surg. Assoc. 21(5): 378-381.
[37]. Ghamsari, S.M., Malekshahi Nejad, M.M. Moradi, o. (2008). Evaluation of Modified Surgical Technique in Repair of Third-Grade Perineal Lacerations in Mare IJVS, 3(3), 71-75.
[38]. Singh, P. and Saharan, S. (2017). Surgical management of perineal laceration in mares. MOJ Surg., 4(4):86‒87.
[39]. Woodie, B. (2006). the vulva, vestibule, vagina and cervix. In: Auer, Stick, ed. Equine surgery. 2nd ed. Philadelphia: WB Saunders Co, 845-849.