Foot and Mouth Disease (FMD)
| Site: | EHC | Egyptian Health Council |
| Course: | Large ruminant Medicine and surgery Guidelines |
| Book: | Foot and Mouth Disease (FMD) |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 15 May 2025 Download Guideline
Table of contents
- - Acknowledgement
- - Introduction
- - Etiology and Virus Characteristics
- - Epidemiology
- - Transmission and Pathogenesis
- - Clinical Signs
- - Complications
- - Economic Impact
- - Diagnosis
- - Treatment and Supportive Care
- - Prevention and Control
- - Biosecurity Measures
- - Eradication Strategies
- - FMD in Wildlife and Carrier State
- - Challenges and Future Directions
- - Conclusion
- - References
- Acknowledgement
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The rapporteur of the Committee: Prof. Mohamed Mohamady Ghanem.
Scientific Group Members: Prof. Nabil Yassien, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby, Dr. Mohamed Elsharkawy, Prof. Gamal A. Sosa., Dr. Naglaa Radwan, Dr. Hend El Sheikh
Author Prof. Mohamed Ghanem
- Introduction
Foot and Mouth Disease (FMD) is a highly contagious viral disease affecting cloven-hoofed animals such as cattle, buffalo, pigs, sheep, goats, and various wildlife species. Recognized as one of the most economically devastating diseases for livestock industries worldwide, FMD poses a significant threat not only due to animal health concerns but also due to trade restrictions and massive economic losses. Although mortality is typically low in adult animals, the disease causes severe production losses and high mortality in young animals. Despite decades of global control efforts, FMD remains endemic in many regions, underscoring the persistent challenges in eradicating this transboundary disease.
- Etiology and Virus Characteristics
· FMD is caused by the Foot and Mouth Disease Virus (FMDV), a member of the genus Aphthovirus within the family Picornaviridae. The virus is a non-enveloped, single-stranded RNA virus with high genetic and antigenic variability.
· There are seven serotypes of FMDV: A, O, C, SAT1, SAT2, SAT3, and Asia1. Each serotype contains multiple subtypes, and immunity is serotype-specific, meaning infection or vaccination against one serotype does not confer protection against others.
· The virus is highly labile under certain environmental conditions, especially in cooler temperatures and acidic environments. This contributes to its ability to persist in contaminated environments, animal products, and fomites, making control and eradication difficult.
- Epidemiology
· FMD is endemic in parts of Africa, Asia, the Middle East, and South America. The disease is absent in North America, Western Europe, Australia, and New Zealand due to strict biosecurity measures and effective eradication programs. The epidemiological pattern of FMD varies with geographic region, serotype prevalence, and livestock management practices.
· The World Organisation for Animal Health (WOAH) has established a framework for FMD control known as the Progressive Control Pathway (PCP-FMD), which helps endemic countries improve their control strategies and progress toward eradication.
· Outbreaks often result from illegal animal movement, porous borders, inadequate veterinary infrastructure, and insufficient vaccination coverage. Wildlife reservoirs, particularly African buffalo in sub-Saharan Africa, contribute to the persistence and spread of the virus in certain regions.
- Transmission and Pathogenesis
· FMDV spreads through direct contact with infected animals and indirect contact via contaminated equipment, vehicles, clothing, and feed. Aerosol transmission is also significant, particularly among pigs, which excrete large quantities of virus into the environment. The virus enters the body through the respiratory tract, oral mucosa, or skin abrasions.
· Following entry, the virus replicates in the pharynx and spreads to regional lymph nodes before entering the bloodstream (viremia). This leads to widespread dissemination and the development of characteristic vesicular lesions in the mouth, on the feet, teats, and snout. Infected animals excrete virus in saliva, milk, feces, and semen, often days before clinical signs appear.
- Clinical Signs
The incubation period of FMD typically ranges from 2 to 14 days. Clinical signs vary among species:
Cattle:
- High fever (lasting 2-3 days)
- Blisters (vesicles) on the tongue, lips, gums, teats, and between the hooves
- Excessive salivation (roby) and drooling
- Lameness and reluctance to move
- Reduced milk production
- Loss of appetite and weight loss
|
Ulceration of muzzle in buffalo |
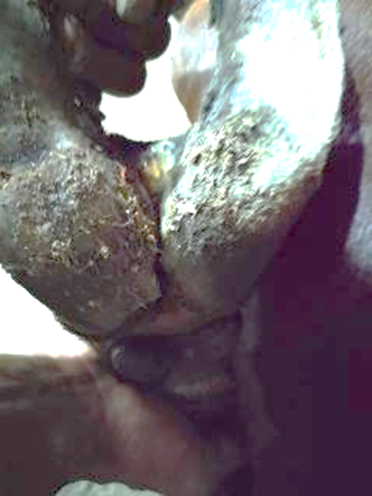
Ulceration of interdigital space |
|
Roby salivation in a cow |
Ulceration of oral mucosa |
|
Massive oral salivation in a buffalo |
Rapid spread of FMD in large ruminant farm |
Sheep and goats: Often subclinical or mild, though vesicles on the feet and oral mucosa may occur.
- Complications
· Secondary bacterial infections may complicate recovery
· In young animals, especially calves, sudden death due to myocarditis is common (tiger heart).
· Recovery usually occurs within two weeks, but some animals may become carriers.
· Panting syndrome (inability to tolerate environmental temperature)
· Upper-trichosis (hirsutism): overgrowth of hair in different areas of the body (hyperadrenocorticism).
- Economic Impact
FMD incurs enormous economic costs, including:
- Loss of production (milk, meat).
- Trade restrictions and export bans.
- Costs of control measures and vaccination.
- Cost of hygienic disposal of dead animals.
- Loss of generic resources and market access.
The 2001 FMD outbreak in the United Kingdom is a prime example, resulting in the culling of over 6 million animals and costing the economy over £8 billion. Developing countries need strict biosecurity measures.
- Diagnosis
Accurate diagnosis is crucial for FMD control. Since several vesicular diseases resemble FMD (e.g., vesicular stomatitis), laboratory confirmation is necessary.
Field diagnosis
Field diagnosis may rely on clinical signs and epidemiological context, but confirmation requires laboratory testing to ensure appropriate response and notification to authorities.
Laboratory Methods:
- Virus isolation in cell culture.
- Reverse transcription polymerase chain reaction (RT-PCR) for rapid detection.
- Enzyme-linked immunosorbent assay (ELISA) for antigen and antibody detection.
- Serotyping to identify the specific virus involved.
- Treatment and Supportive Care
· There is no specific antiviral treatment for FMD. Management of affected animals focuses on supportive care, including pain relief, anti-inflammatory drugs (NSAID) and management of secondary infections.
· In endemic regions, affected animals are often allowed to recover naturally, although their productivity may be permanently compromised.
· In non-endemic countries, control policies typically include stamping out (culling infected and in-contact animals), movement restrictions, and disinfection procedures to eliminate the virus.
- Prevention and Control
Vaccination:
Vaccination is the cornerstone of FMD control in endemic areas. Inactivated vaccines are used, and formulations must match circulating strains to be effective. However, vaccines provide only temporary immunity (6 months) and require frequent boosters.
- Monovalent, bivalent, and trivalent vaccines are used depending on the serotypes circulating in the region.
- Emergency vaccination may be deployed during outbreaks in FMD-free zones.
- Biosecurity Measures
- Quarantine and testing of animals before movement.
- Disinfection of transport vehicles, equipment, and facilities.
- Control of animal imports and exports.
- Surveillance and early detection systems.
- Eradication Strategies
- In FMD-free countries, rapid detection, culling, and strict movement controls are enforced.
- In endemic areas, gradual control through mass vaccination, public awareness, and animal health infrastructure and hygiene.
- FMD in Wildlife and Carrier State
Wildlife can serve as reservoirs or spillover hosts. African buffaloes are known long-term carriers of FMDV without showing clinical signs, complicating eradication efforts in Southern Africa.
The carrier state in domestic animals, particularly cattle, is characterized by the persistence of FMDV in the pharyngeal tissues beyond 28 days post-infection.
The significance of carriers in disease transmission remains debated, but their existence necessitates prolonged surveillance post-outbreak.
- Challenges and Future Directions
Several challenges hinder global FMD eradication:
- Antigenic variability and short vaccine duration.
- Inadequate surveillance and reporting.
- Smuggling of animal across the borders.
- Conclusion
Foot and Mouth Disease continues to be a major concern for global animal health and trade. Its highly contagious nature, economic impact, and complex control dynamics make it a priority for national and international animal health organizations. While eradication remains a long-term goal, coordinated surveillance, strategic vaccination, and improved biosecurity are key to managing this disease. Ongoing research, international cooperation, and strong biosecurity measures and managment are essential to control and eventually eliminate FMD from endemic regions.
- References
1. Alexandersen, S., et al. (2003). The Pathogenesis and Diagnosis of Foot-and-Mouth Disease. Journal of Comparative Pathology.
2. FAO and OIE. (2012). Global Foot-and-Mouth Disease Control Strategy.
3. Ghanem, M.M., Abdel-Hamid, O.M. Clinical, haematological and biochemical alterations in heat intolerance (panting) syndrome in Egyptian cattle following natural foot-and-mouth disease (FMD). Trop Anim Health Prod 42, 1167–1173 (2010).
4. Grubman, M. J., & Baxt, B. (2004). Foot-and-Mouth Disease. Clinical Microbiology Reviews.
5. Knight-Jones, T. J. D., & Rushton, J. (2013). The Economic Impacts of Foot and Mouth Disease – What are they, how big are they and where do they occur?. Preventive Veterinary Medicine.
6. OIE (WOAH). (2023). Foot and Mouth Disease. Terrestrial Animal Health Code.