Infertility in she camel
| Site: | EHC | Egyptian Health Council |
| Course: | Theriogenology Guidelines |
| Book: | Infertility in she camel |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 18 May 2025 Download Guideline
Table of contents
- - Acknowledgment
- - Introduction
- - Breeding season of the Middle East and Asian she camel
- - Causes of infertility in she-camel
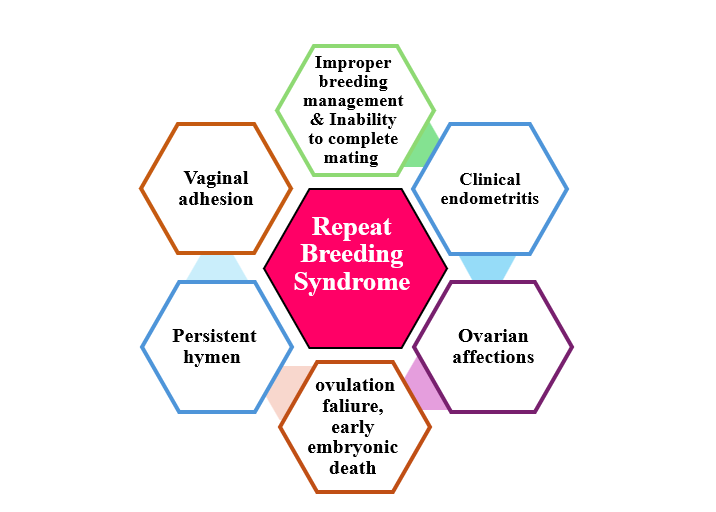
- - A. Repeat Breeding Syndrome
- - B. Early embryonic death (EED syndrome)
- - C. Abnormal behavior (Refuse mating syndrome)
- - D. Congenital abnormalities of the reproductive tract
- - E. Ovarian inactivity
- - REFERENCES
- Acknowledgment
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The Rapporteur of the Committee: Prof. Mohamed Mohamedy Ghanem.
Scientific Group Members:
Prof. Nabil Abd Elgaber, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby. Dr. Mohamed Elsharkawi, Prof. Gamal Sosa, Dr. Naglaa Radwan, Dr. Hend Elsheikh
Authors: Prof.Gamal A.M. sosa, Prof.Ahmed R.Elkhawagah, Dr.Ahmed S.A.M.sosa, Dr.Dina E.M. Rashad
- Introduction
Reproductive capacity plays a vital role in the production and profitability of the camel industry. Common issues in practice include poor reproductive efficiency and infertility. Among domestic farm animals, camelids have the lowest reproductive efficiency, where even slight reductions in fertility can lead to significant consequences (Khanvilkar et al., 2009, Benaissa et al., 2015, Getahun et al., 2021, Mohammed et al., 2025).
Camelids display various anatomical and physiological reproductive differences, such as induced ovulation, overlapping follicular waves, the critical role of the corpus luteum in pregnancy maintenance, a unique intrauterine copulatory mechanism, mating duration, and semen viscosity, which can pose challenges in diagnosing and treating infertility (Belina et al., 2021, Mansour and Karen, 2021, Waqas et al., 2025). Several factors contribute to low reproductive efficiency in female dromedaries, including delayed puberty, a later age at first conception and calving, and extended intervals between births. Furthermore, reproductive performance can be adversely affected by nutritional deficiencies, diseases, hormonal imbalances, and environmental stressors (Kelanemer et al., 2025). It is essential to identify abnormalities in the reproductive tract, particularly in genetically superior animals, as reproductive diseases can significantly diminish fertility.
Arabian she-camels generally reach puberty around 2 years of age. While sexual activity can begin at 2-3 years, she-camels are usually bred starting at approximately 4 years of age. They can continue to breed until they are about 30 years old. At 2 years, she-camels typically weigh around 400 kg, increasing to about 513 kg by 3 years (Beniwal and Chaudhry, 1984, Gherissi et al., 2020, Belina et al., 2021).
She-camels are seasonal polyestrous animals, with maximum breeding occurring during shorter daylight periods, particularly from December to May in regions like Egypt, Saudi Arabia, and the UAE (Table 1). During longer photoperiods, they enter a state of anestrus. The average duration of their estrous cycle varies from 24.2 days in Egypt to about 28 days in Sudan and can range from 11-30 days in Saudi Arabia. Estrous cycles can be less reliable, often accompanied by inconsistent estrus signs (Ashour et al., 2017, El Allali et al., 2017, Al-Bulushi et al., 2019, Ghallab et al., 2024). Ovarian follicles undergo growth in waves approximately every 11-20 days, with active follicles growing to sizes between 10 and 25 mm, and some oversized follicles reaching up to 75 mm under specific conditions (Fig. 1). Ovulation in camels is induced by mating, which has important implications for their breeding management (Skidmore et al., 1996, Anouassi and Tibary, 2013, Ghallab et al., 2024, Mohammed and Alshaibani, 2025).
- Breeding season of the Middle East and Asian she camel
Table 1: Breeding season of the Middle East and Asian she camel (Purohit et al., 2020).
|
Location |
Breeding season |
|
Egypt |
Dec – May |
|
Saudi Arabia |
Dec – March |
|
United Arab Emirates |
Nov - April |
|
Sudan |
March – Aug |
|
Somalia |
May – June, Oct - Dec |
|
Kenya |
Continuous |
|
India |
Nov – March |
|
Pakistan |
Dec – March |
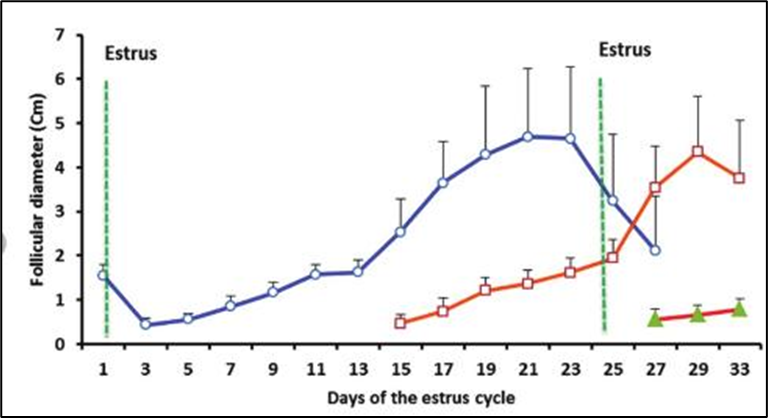
Figure 1: Follicular dynamics during the estrus cycle in camels (Mohamed et al., 2021).
- Causes of infertility in she-camel
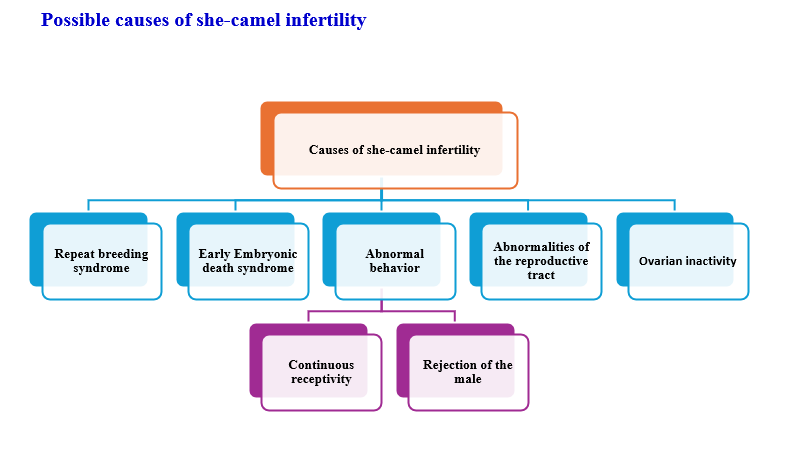
- A. Repeat Breeding Syndrome
It is generally defined as any camel that failed to conceive after at least three regular spaced services, with no clinical abnormalities. It may be caused by different affections
I. Ovarian Affections
It includes ovarian cysts (follicular, luteal cysts, hemorrhagic), ovarian agenesis, ovarian hypoplasia, and Ovarian hydrobursitis.
1.1- Ovarian cysts
Cystic ovarian disease (COD) is a significant ovarian dysfunction resulting from disruptions in the hypothalamic-pituitary-gonadal axis due to either endogenous or exogenous factors. a study by (Hegazy et al., 2001) indicates that the incidence of cystic and inactive ovaries in female camels increases during summer. When a dominant follicle is present without mating, it can lead to one of two outcomes: either regression of the follicle or cystic degeneration, resembling an ovulatory follicle. According to (Dawod and Hamed, 2018), the types of ovarian cysts are categorized as follows:-
1.2- Follicular cysts
It may be unilateral or bilateral either single or multiple cysts, but mostly in a single manner. It is thin-walled, well-vascularized, or slightly opaque and filled with a straw-colored serous fluid. The diameter ranged from 17-40 mm (Fig. 2).
1.3- Luteal cysts
It is thick-walled and becomes partially or totally opaque, tense in consistency, with a grayish-yellow color. The follicular fluids reach 22 ml. The diameter reaches up to 50 mm.
1.4 Hemorrhagic cysts
It is a single, thick wall, unilateral or bilateral, and contains bloody follicular fluid that reaches about 8 ml. The diameter ranged from 10-25 mm.
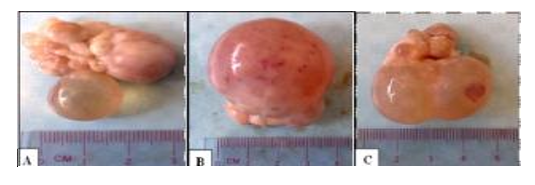
Figure 2. Ovary containing Follicular cyst characterized by a large, thin wall containing serous, clear follicular fluid (A) single small cyst (B) large cyst 40 mm in diameter (C) double cyst (Dawod and Hamed, 2018).
2- Ovarian agenesis
It is the complete absence of ovaries. It may be unilateral or bilateral (Fig. 3). The unilateral ovarian agenesis is characterized by the presence of a large bulging follicle on the right ovary and a small fatty mass on the left one. The bilateral ovarian agenesis is characterized by ending of the fallopian tube in a well-developed bursa that contains a small fatty tissue mass like the pea size while the rest of the genital tract is normal (Dawod and Hamed, 2018).
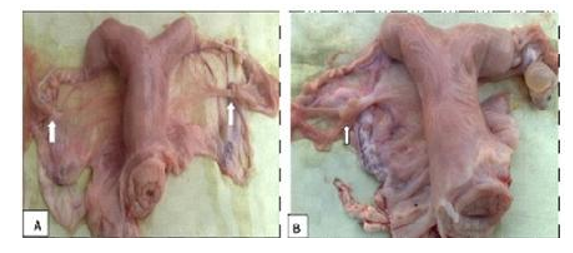
Figure 3. Ovarian agenesis (A) bilateral, (B) left unilateral agenesis with a normal right ovary, and has a large follicle (Dawod and Hamed, 2018).
3- Ovarian hypoplasia
It is characterized by the absence of ovarian follicular activity due to genital and chromosomal abnormalities (73XO, 75XXX, 74XX/74XY, minute chromosome) (Tibary et al., 2011). The ovaries are small, thin, and firm structures without any small follicles, while the rest of the genital tract is normal (Fig. 4) (Dawod and Hamed, 2018).
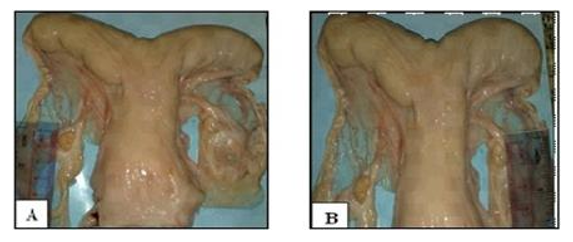
Figure 4. Ovarian hypoplasia (A) left ovary 10 mm (B) right ovary 8 mm in length (Dawod and Hamed, 2018).
Diagnosis:
· The history of multiple matings/continuous receptivity, or persistent rejection of the male.
· Transrectal ultrasonography demonstrates a small uterus and either small follicles that do not progress to a dominant size or an inability to visualize the ovaries entirely.
· Laparoscopic examination to confirm the case.
4- Ovarian hydrobursitis
The camelid's ovary is completely covered with a thin fold of the mesosalpinx, the ovarian bursa. The apex of this bursa forms a large circular orifice within which lie the fimbriae of the oviduct (Fig. 5a–c). In most cases (85%), the ovary was freely movable and could be easily uncovered from the bursa. In about 15% of cases, the bursa appeared as a large sack filled with different amounts of fluid of different physical characters (Fig. 5d–f). The mean diameters of these bursae were 35.18 ± 5.2 × 15.12 cm. On transrectal palpation, the ovarian hydrobursitis (21.8%) was suspected when the genital tract was difficult to retract, and the encapsulated ovary was not palpable (Fig. 5g). on ultrasonographic examination, a large well-demarcated, hypoechogenic sac was found lateral to or beneath the corresponding uterine horn (Fig. 5h, i). In many cases (68.4%) the ovary appeared as a floating structure and was easily identified when it was functionally active. In the other cases (30.6%), the ovary adhered to the bursal wall (Fig. 5j).
Ovarian hydrobursitis is a peculiar disease of the ovarian bursa, characterized by adhesion of the ovarian bursa, fluid accumulation, and encapsulation of the ovary (Fig. 6). It is initially an inflammatory process, and the accumulated bursal fluid is partially originates from the follicular fluid. It is associated with peri-uterine adhesion (vagina/cervix), pyometra, and Paraovarian cysts (Tibary and Anouassi, 2001, Ali et al., 2011). The affected female shows long-standing infertility, refuses to mate, repeats breeding with long heat intervals, early embryonic death, and abortion.The incidence of ovarian hydrobursitis was 6.5% in slaughtered camels and 33.8% in infertile females.
Diagnosis
- Rectal examination reveals a cord-like structure was palpated between the ovarian end of the uterus and the corresponding ovaries.
- Ultrasonography presented variable appearances of the ovarian bursa. The bursal fluid appears as clear anechoic fluid with different sizes, and in a few cases, there are echogenic particles in the bursal fluid. In addition, the ovaries carrying different structures appear as floating masses in the bursal fluid (Fig. 7) (Mansour and Karen, 2021).
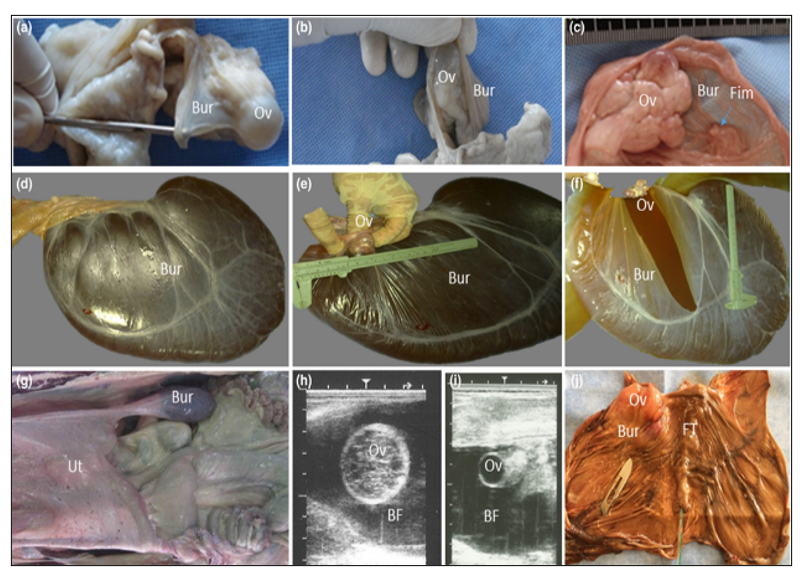
Figure 5. Ovarian bursa in camels: (a, b, c) the ovary is completely covered with a thin fold of the mesosalpinx, and the apex of bursa forms a large circular orifice within which lies the fimbriae of the oviduct (c, arrow). (d–f) A case with ovarian hydrobursitis, the bursa, appeared as a large sack filled with fluid, while the ovary was found floating within the completely closed bursa. (g) On transrectal palpation, the genital tract was hard to withdraw, and the encapsulated ovary was problematic to palpate. (h, i) On ultrasonographic examination, a large, well-demarcated, hypoechogenic sac was found lateral to or beneath the corresponding uterine horn, and the ovary appeared as a floating structure. (j) In some cases, the ovary adhered to the bursal wall. Ov: ovary; Bur: bursa; Fim: fimbria of the oviduct; Ut: uterus; FT: fallopian tube; BF: bursal fluid (Ali and Derar, 2020).
Hormonal and biochemical evaluation of the bursal fluid and its comparison with blood and follicular fluid biochemistry may be useful in clarifying the origin of the bursal fluid.
- Neutrophilia, monocytosis and eosinophilia.
- Bursal fluid had a tendency for high estradiol-17β concentration compared to blood serum. Progesterone and cholesterol concentrations were similar in bursal fluid, follicular fluid and serum.
- Total protein, phosphorus, and magnesium concentrations were greater in the bursal fluid than in serum.
- Histopathology: degeneration and hyperplasia of the lining epithelium, mononuclear cells infiltration, focal aggregation of inflammatory cells, cystic dilatation of multi-acinal structures, tiny hemorrhages, and presence of hemosiderin-laden macrophages.
- Bacterial and protozoal examination may be helpful in explaining the etiopathogenesis of the syndrome.
- Treatment: Surgical ablation in unilaterally affected animals presents potential treatment.
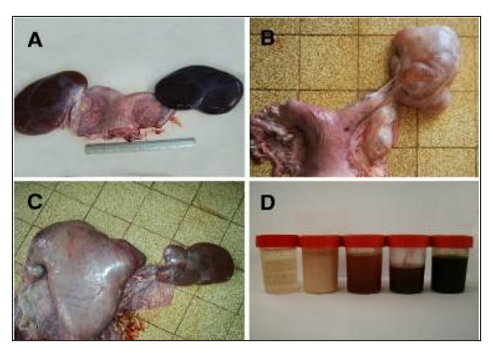
Figure 6. Ovarian hydrobursitis in slaughtered female camels. A, bilateral ovarian hydrobursitis; B, unilateral ovarian hydrobursitis; C, ovarian hydrobursitis in pregnant female; D, different fluids in the bursa (Ali et al., 2011, Benaissa et al., 2014).
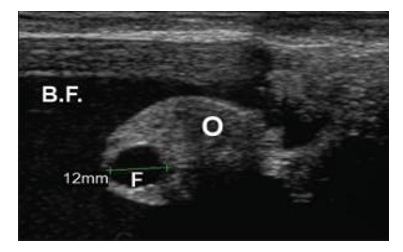
Figure 7. Ultrasound image shows a case of ovarian hydrobursitis. O: ovary; B.F: bursitis fluid; F: Graafian follicle (Mansour and Karen, 2021).
II.Ovulation Failure
It is a common problem in camelids. Anovulatory follicles can become hemorrhagic and, in some cases, luteinize and produce progesterone, resulting in rejection of the male. It occurs due to the lack of luteinizing hormone released after mating (Fig. 8, 9).
Causes:
- Bad breeding time or mating time, in contrast to follicular development.
- The abnormal ovary.
- Males with low potency could not induce the ovulation-inducing factor (OIF) due to the short time of copulation.
- The females with vagina/cervix abnormalities that may inhibit the normal mechanism of induction of ovulation.
- The ovarian follicular activity may be reduced in females that are lactating, obese, heavily parasitized, advanced in age, or have experienced severe
Diagnosis:
- level of progesterone must be kept under check.
- confirmation of ovulation through ultrasonography.
Treatment: Injection of Human chorionic gonadotropin (hCG) or gonadotropin-releasing hormone (GnRH).
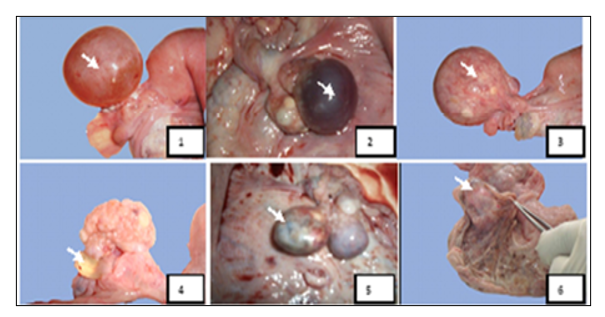
Figure 8: Macroscopic aspects of ovarian affections found in female camels studied at El-Oued slaughterhouse. Key: 1-Follicular cyst, 2-Hemorrhagic cyst, 3-Luteinized follicular cyst, 4-Para-ovarian cyst, 5- Ovarian teratoma, 6- Oophoritis (Gherissi et al., 2019).
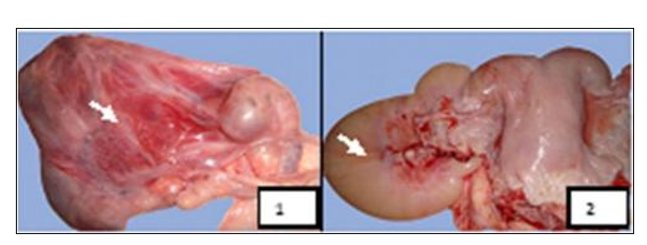
Figure 9: Macroscopic aspects of ovarian bursal affections found in female camels studied at El-Oued Slaughterhouse. Key: 1- Bursitis, 2- Hydrobursitis (Gherissi et al., 2019).
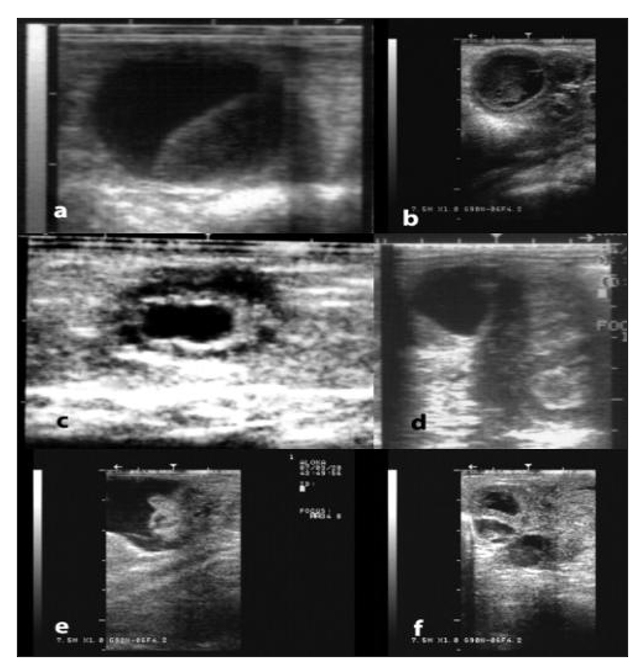
Figure 10: Ultrasonographic appearance of some ovarian and tubal disorders in camelids. a) Large anovulatory follicle, b) Luteinized/hemorrhagic anovulatory follicle, c) Cystic rete ovarii, d) Ovarian teratoma, e) Hydrobursitis, f) Hydrosalpinx (Pearson and Tibary, 2014).
III. Utero-tubal affections
▪️ Hydro-salpinx
The uterine tube is filled with fluids and can be rectally palpated as a tortuous, distended structure near the ovary (Fig. 11, 12). By ultrasonography, the uterus is demonstrated as many hypoechogenic, well-demarcated sacs adjacent to the ovary (Mansour and Karen, 2021).
▪️Clinical endometritis
The uterine inflammation has been described as the most commonly encountered form of infertility in dromedary camels. It can lead to irreversible damage of the salpinx, resulting in a total loss of fertility. It can be classified as acute, sub-acute, or chronic. According to the thickness of the uterine wall and the amount and consistency of the vaginal discharges, endometritis is classified into three degrees (mild degree: uterus of normal consistency with small amount of mucopurulent vaginal discharge; moderate degree: thick-walled uterus with moderate mucopurulent discharge; and severe degree: large-sized uterus with hard consistency and thick or pasty vaginal discharges) (Fig. 13) (Ali et al., 2010).
Causes
- Over-breeding
- Unaddressed postpartum complications
- Improper obstetrical manipulations
- Nutritional deficiencies, such as selenium and copper, may also be linked to an increased incidence of endometritis
Diagnosis
- Observation of vaginal discharges
- Trans-rectal palpation reveals a slight enlargement of the uterine horns
- Vaginal examination: the uterus and/or cervix are enlarged, tough, and have thick wall, with different degrees of vaginal discharges.
- Transrectal ultrasonography: areas of increased echogenicity and thickening of the uterine wall.
- Cytology and culture are necessary in cases of suspected endometritis »»»»» Endometrial biopsy should also be considered in the diagnosis of endometritis and can be a prognostic indicator of future fertility. For camelids, the use of a Turret rectal biopsy punch is recommended. Targeted biopsies can be performed in combination with hysteroscopy. Although a grading system for histological evaluation has been proposed in camelids it is not widely used in a clinical setting. These techniques should be performed when the female has the presence of a dominant follicle and the cervix is open (Table 2).
- Bacteriological culture should include sensitivity testing for the major antibiotics. The most common bacteria isolated from cases of camelid endometritis are Escherichia coli, Streptococcus equi zooepidemicus, β-hemolytic Streptococci, Enterococcus spp., coagulase negative Staphyloccocus spp., Proteus spp, Enterobacter aerogenes, Klebsiella pneumonia, Pseudomonas aeruginosa and Trueperella pyogenes. Venereal transmission should be considered in case of herd infertility or abortion outbreaks. Pseudomonas aeruginosa, Campylobacter fetus fetus, Tritrichomonas foetus, Aspergillus spp., and Mucor spp. have been isolated from infertile camels.
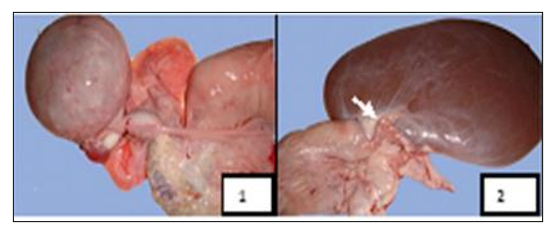
Figure 11: Macroscopic aspects of oviduct affections found in female camels studied at El-Oued slaughterhouse. 1 - Oviduct hypertrophy, 2 - Oviduct torsion (Gherissi et al., 2019).
Table 2. Classification of endometrial biopsy and the potential effect on fertility (Tibary, 2009).
|
Biopsy Category |
Histopathological Characteristics |
Effect on Fertility |
|
Grade 1A |
-Normal endometrium |
-Normal conception rates |
|
Grade 1B |
Few lymphocytes within the endometrium. |
Low-grade infection or remnants of previous inflammation. Mild surface irritations may indicate a reaction to breeding. May be postpartum or post-abortion |
|
Grade 2A to 2B |
Active and acute, chronic, or chronic active endometritis. Chronic inflammation tends to be more deeply located in the endometrium, compared with active and chronic active inflammation |
Interferes with conception and may cause early embryonic death. |
|
Grade 3A |
Chronic endometritis with glandular fibrosis. |
Interferes with implantation and placentation. May cause early embryonic death |
|
Grade 3B |
Uterine neoplasia |
Pregnancy loss or abortion. |
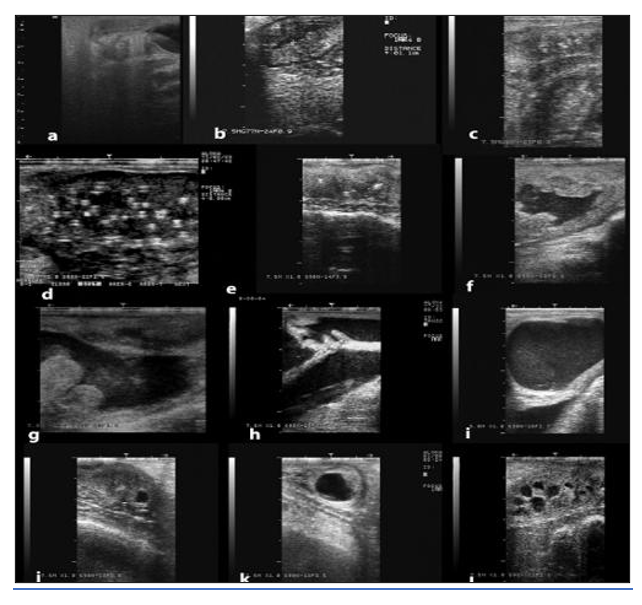
Figure 12: Ultrasonographic appearance of some disorders of the uterus. a) Infantile uterus, b) Endometritis, c) Chronic endometritis, d) Endometrial abscesses, e) Cervical adhesions, f) Pyometra; g) Post-fetotomy pyometra; g) mucometra due vaginal adhesions, note the open cervical rings, i) Mucometra due to vaginal aplasia, j-l) Uterine cysts (Pearson and Tibary, 2014).
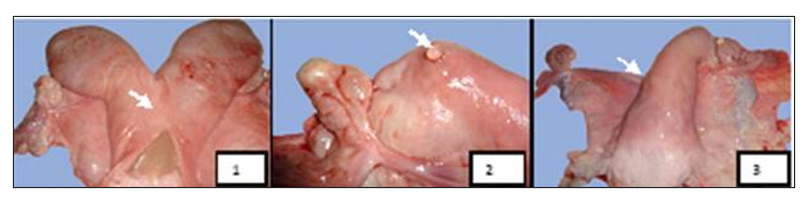
Figure 13: Macroscopic aspects of uterine affections found in female camels studied at El-Oued Slaughterhouse. 1 - Pyometra, 2 - Uterine serosal inclusion cyst, 3 - Uterine agenesis (Gherissi et al., 2019).
IV. Vaginal and cervical adhesions
Vaginal and cervical adhesions are severe long-standing reproductive disorder in dromedaries and consequently result in a high culling rate. The incidence of vaginal and cervical adhesion amounted to about 14.4% and 6.7%, respectively, of the infertility problem. The affected animals are often unable to breed due to obstruction of the genital tract. Moreover, the results of adhesion treatment trials have not been optimistic. Several risk factors are assumed for cervico-vaginal adhesion, such as chronic vaginitis, over-breeding, aggressive mating practice, and injuries during parturition.
Generally, adhesion occurs secondary to surgery, infection, trauma, or radiation. The bacterial infections of the genital tract of the female camel have been shown to be the main causes of reproductive disorders. In camelids, the invasive breeding nature and frequent mating can cause trauma to the cervix and uterus.
Diagnosis
§ Vaginal examination: access to the cranial part of the genital tract was blocked. This adhesion was found at the mid or caudal vagina or the cervix.
§ Trans-rectal ultrasonography: accumulation of hypo- or hyper-echogenic materials in the uterus.
V. Persistent hymen
o It was detected through vaginal examination of some heifers as a membrane cranial to the external opening of the urinary.
o Persistent hymen can be complete and incomplete, leading to accumulation of vaginal fluid or sometimes protruding of Hymen outside the vagina.
o In case of incomplete perforation of the hymen, the female feels pain after mating, because the hymen is very thick, and the opening is small.
o This is treated by giving an incision using a pair of scissors or a scalpel.
- B. Early embryonic death (EED syndrome)
Early embryonic death (EED) refers to the fetal mortality, most probably not later than 50 days of gestation. Animals that refuse the male 2 weeks after mating as if they were pregnant; however, later on (> 30 days) they return to estrus and accept the male.
The loss is considered an early pregnancy loss if it occurs before 50 days of gestation. Usually, there are no generalized or obvious signs of loss, but when pregnancy is beyond or up to 35 days small amounts of fleshy tissue and fluid can be recognized near the manure mass. In several cases, the owner does not even recognize pregnancy and considers the female open because she may show signs of receptivity when there are mating animals or by the presence of any male in the zone. sometimes females do not develop abdominal distention or cushing in late gestation.
Causes of EED
Key factors attributed to EED issues in Camelidae include: -
• Genetic causes
• CL insufficiency
• Unreceptive uterine environment; nevertheless, no factor can be changed to promote embryo sustainability.
Heat stress, chromosomal abnormalities, or genetic factors (such as lethal genes, segregation, mutation, structural abnormality, aneuploidy, e.g., monosomy, trisomy, polyploidy, multiple numbers of haploid chromosomes, polyspermia, etc.), nutritional factors (such as a lack of proteins, vitamins, microminerals, the effect of toxins and nitrate poisoning, etc.), abnormal hormonal situations/endocrine factors (such as low progesterone), energy balance (an important factor in retaining pregnancy), genital infections (which may be specific or nonspecific), venereal diseases (such as Trichomiasis, Brucellosis, Trypanosomiasis, etc.), uterine environment, immunological factors (normally immune suppressors produced by mothers, if not produced, cause fetus rejection), effect of palpation (improper timing of palpation), improper timing of Artificial insemination (late AI aged embryo), twin pregnancy, effect of the male (such as lethal genes, abnormal chromosomes, venereal diseases, etc.), infectious causes, and other proper causes, such as older animals, are among the causes of EED in camels. Moreover, inbreeding of herds is considered one of the basic causes of fetal death. The rate of abortion due to infections varies from 10% to more than 70% in some areas of the world. In llamas and alpacas, the most common reasons for abortion are leptospirosis, chlamydiosis, and toxoplasmosis. Whereas, in camels of the Middle East and Africa, brucellosis and trypanosomiasis are considered major causes of infectious abortion.
◾ Maternal factors of early embryonic death in she camel
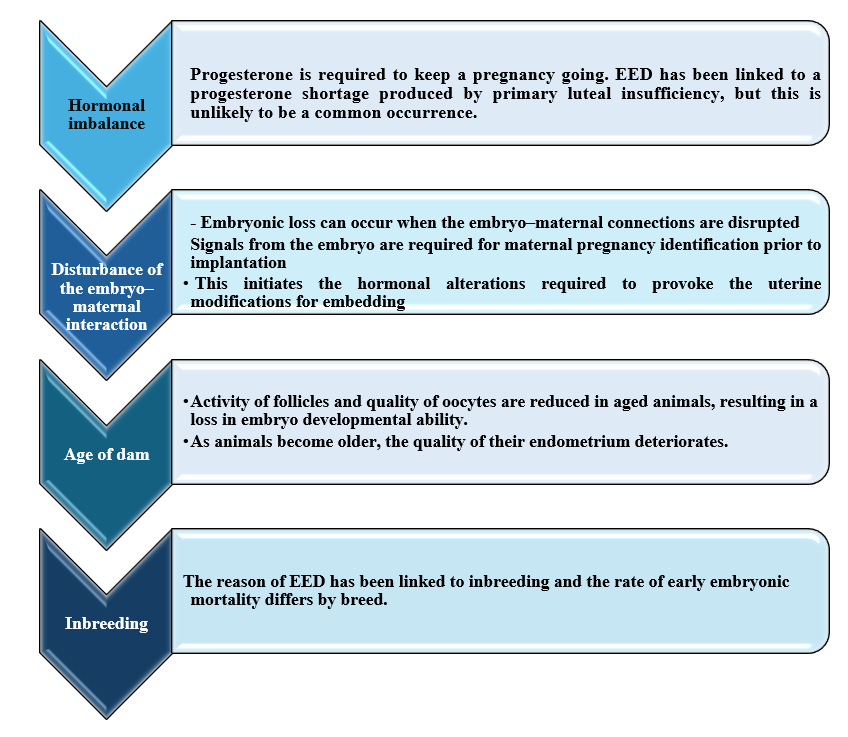
◾ External factors od early embryonic death in she camel
1. High environmental temperature
- Extreme temperature in the course of initial months of pregnancy might also be harmful to the embryo.
- Extreme uterine temperature has been shown to have a direct detrimental effect on the embryo, as well as the pushing of blood away from the uterus to the outside to maintain body temperature, result in a decreased nutrients load.
2. Specific nutritional deficiencies or malnutrition
- The embryo can be harmed by certain nutrient deficits or starvation.
- EED can be caused by a significant deficits of vitamins (vitamin A) or other nutrients (Cu, Zn, and I) that function as metabolic supervisors. Poor nutrition or a serious negative energy balance might impact follicular growth, oocyte quality, and oviduct secretory and motile activity, which is where the fertilization process takes place. Also, nutrition has an impact on the very early phases of conception.
3. Stress: Stress has a negative impact on animal reproductive performance. Stress-causing factors (such as transportation, mechanical trauma, isolation, pain, blood pressure alteration, etc.) impact reproductive function by acting on the hypothalamus (GnRH) or ovarian levels (progesterone).
4. Environmental toxicants, teratogenic compounds, and mycotoxins
- When taken during critical early stages of gestation, toxins in the environment, cancer-causing chemicals, and fungal toxins can have devastating consequences on embryonic life.
- In severe cases, acute endometritis has a direct influence on the embryonic environment following mating or artificial insemination and, in severe cases, is accompanied by the synthesis of luteolytic chemicals such as prostaglandins. Endometrial glands are surrounded by layers of fibrous tissue, resulting in a lack of functional glands, depriving the embryo of protein-rich exocrine secretion.
◾ Noninfectious causes of EED in she camel
- In animals, the primary reason for the failure of a pregnancy in its early stages is chromosomal abnormalities. The chromosomal abnormalities account for around 20% of overall embryonic and fetal loss.
- C. Abnormal behavior (Refuse mating syndrome)
· A female camel that curls her tail dorsally when approached by a rutting male.
· Aggressive behavior and infertility may be associated with ovarian tumors (granulosa theca cell tumors) and unilateral ovariectomy may re-establish ovarian function of the contralateral ovary in some cases.
- D. Congenital abnormalities of the reproductive tract
The most common congenital disorders include:
o Ovarian dysgenesis/hypoplasia
o Segmental aplasia of the uterus
o Uterus didelphys
o Vaginal aplasia
o Persistent hymen
o Vulvar atresia.
Segmental aplasia of the reproductive tract
It may occur at the level of the uterine tube (formation of hydrosalpinx) or the level of the uterine horn (uterus unicornis). The affected females will often present with normal cycles and ovulation but fail to achieve or maintain pregnancy.
Diagnosis: achieved by ultrasonography and confirmed by hysteroscopy or laparoscopy.
Uterus didelphys
It is a rare congenital anomaly of the female reproductive tract characterized by a divided uterine cervix and body (Fig. 14, 15). It occurs due to abnormal development of the paramesonephric (Müllerian) duct (Mahdy and Nasr Eldeen, 2024).
Complete double cervix: It is caused by the persistence of the median walls of the Müllerian ducts along the whole length of the cervix, resulting in two cervices and one uterine body.
Incomplete double cervix: It is caused by the persistence of the median walls of the Müllerian ducts at the posterior part of the cervix, resulting in one cervical canal cranially and two cervical canals caudally.
Double external uterine orifices: It is caused by the persistence of the median walls of the Müllerian ducts at the external uterine orifice, resulting in one cervix with a band of tissue at the external os.
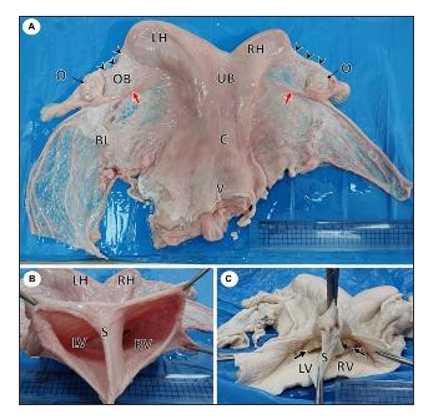
Figure 14. Gross photographs of female reproductive system of a she-camel show: (A) Apparently normal reproductive system consisted of right and left ovaries (O), each ovary was located inside an ovarian bursa (OB), the ovary attached to the uterine horn by the roundligament (red arrow), fallopian tubes (arrowheads), right (RH) and left (LH) horns, uterine body (UB), cervix (C), and vagina (V). Note the broad ligament of the uterus (BL). (B) Per vaginal view shows a completely divided vagina into the left (LV) and right (RV) vagina by a median septum (S). (C) The dorsal wall of the vagina was opened, showing each vagina had its external os (arrow) (Mahdy and Nasr Eldeen, 2024).
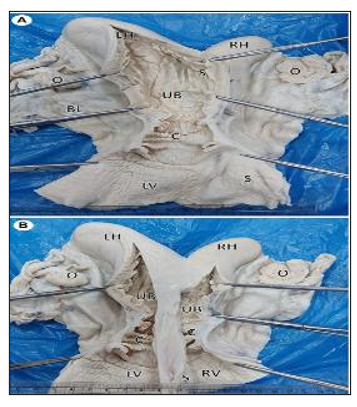
Figure 15. Gross photographs showing a dorsal view of uterus didelphys: (A) An incision was performed in the left horn (LH), body (UB), cervix (C), and vagina (LV). Note the complete septum extending from the fundus to the vagina. (B) Incised right (RH) and left (LH) horns show each horn connected to a separate uterine body that had its own internal and external os (Mahdy and Nasr Eldeen, 2024).
The abnormal condition of the vagina and vulva include: aplasia, persistent hymen, vulvar atresia, and vaginal constriction
Diagnosis
- History of rejecting the male
- Persistent straining or pain during mating or excessive vulvar swelling after mating.
- Transrectal ultrasonography generally demonstrates accumulation of fluid or mucus in the vagina/uterus (hydrometra, mucometra).
- Examination of the external genitalia and vaginal examination.
- Digital palpation is often sufficient to determine an occlusion at the level of the vestibulum.
- Cystitis and vestibular inflammation, or complete adhesion is a long-term result of vaginal inflammation.
- Abnormalities of the vulva are usually seen in small camels, which lead to accumulation of urine in the uterus, a condition known as urometra.
- E. Ovarian inactivity
Ovarian activity in dromedary camel increases during the breeding season in winter and spring months and depends on good food resources, climate conditions, and female body condition.
Causes:
- The adverse body condition.
- Insufficiency of production of gonadotropins from the anterior pituitary.
- failure of the ovaries to respond to gonadotropins.
- improper nutrition.
Clinical signs:
Female camels suffering from ovarian inactivity are usually presented to the clinic with a history of several matings without conception or refuse mating.
Diagnosis
- Trans-rectal palpation: Both ovaries are small, smooth, and firm, with no palpable follicles or corpus luteum, and the uterus is small and atonic.
- Trans-rectal ultrasonography: Absence of any phase of follicular development on the ovaries at two successive examinations with an interval of 7 days (Fig. 16).
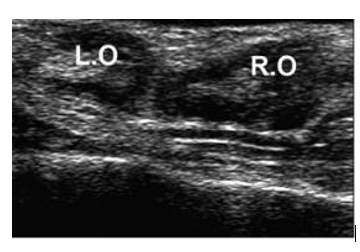
Figure 16. Ultrasound image shows a case of ovarian inactivity. R.O: right ovary; L.O: left ovary (Mansour and Karen, 2021).
Treatment of camel infertility
o For complete vaginal aplasia, females should be separated from males and discarded from the breeding.
o An imperforate hymen is corrected by making an incision through the tissue with a scalpel, however, potential genetic correlation and future breeding should be discussed with the owner.
o Incomplete perforation of the hymen (vestibular narrowing) is fairly common and may be managed successfully with bougienage.
o Vulvar aplasia is also relatively common and surgical repair is controversial.
o Ovarian inactivity: Using multivitamins and mineral supplementation for a week followed by an injection of 1500 IU eCG or using of intravaginal device CIDR for 10 days followed by injection of 1500 IU eCG are reliable protocols to treat the problem of ovarian inactivity in dromedary camels during breeding season (mineral and vitamin premix supplement which contains vitamins A, D, E, B1, B2, B6, B12, C, niacin and pantothenic acid and minerals such as calcium, phosphorus, magnesium, iron, copper, cobalt, iodine, manganese, zinc and selenium was orally administered daily for a week (Day 1-7) (Mansour and Karen, 2021).
o Endometritis:
- Uterine flushing with lactated Ringer’s solution (LRS) or a proprietary equine uterine lavage solution.
- Intrauterine antibiotic infusion, systemic antibiotic administration, or a combination: most antibiotics which can be used in utero can also or alternatively be used systemically and achieve very good concentration in uterine tissue.
- Administration of oxytocin may improve uterine clearance.
- Infusion with 30% DMSO solution or N-acetylcysteine prior to uterine lavage may be considered in chronic cases with thick mucopurulent discharge.
- Biofilm formation is a characteristic of many organisms found in endometritis and the addition of buffered chelating agents (tris-EDTA and Tricide®) may help dissolve the biofilm and improve antimicrobial action.
- A 4% metacresol-sulfonic acid and formaldehyde solution (Lotagen) has provided good results for the treatment of endometritis and metritis in camels.
- Chronic endometritis will lead to the development of degenerative changes or fibrosis. In these cases, the best option in valuable females is embryo transfer.
Success of treatment of endometritis depends on the duration of infection and females should be re-examined after a period of sexual rest.
- REFERENCES
Al-Bulushi, S., Manjunatha, B. M., Bathgate, R., Rickard, J. P. & De Graaf, S. P. (2019). Artificial insemination with fresh, liquid stored and frozen thawed semen in dromedary camels. PLoS One, 14, e0224992.
Ali, A. & Derar, D. R. (2020). Ovary and ovarian bursa in dromedary camels: clinical relevance of the topographical features. Anatomia, Histologia, Embryologia, 49, 325-332.
Ali, A., Al-Sobayil, F. & Al-Hawas, A. (2010). Evaluating the effectiveness of different treatments of uterine infections in female camels (Camelus dromedarius). Theriogenology, 74, 40-44.
Ali, A., Mehana, E., Ahmed, A., El-Tookhy, O., Al-Sobayil, A. & Al-Hawas, A. (2011). Ovarian hydrobursitis in female camels (Camelus dromedarius): clinical findings, histopathology and fertility after unilateral surgical ablation. Theriogenology, 76, 492-499.
Anouassi, A. & Tibary, A. (2013). Development of a large commercial camel embryo transfer program: 20 years of scientific research. Animal Reproduction Science, 136, 211-221.
Ashour, A., Zeidan, A., Badr, M., Amer, A., Fatma, R., El-Aziz, A. & Al-Akhras, A. (2017). Influence of the different ages during breeding and non-breeding seasons on ovarian activity of the dromedary she-camels. Egyptian Journal of Applied Science, 32, 73-90.
Belina, D., EshetU, A., Alemu, S., Shasho, B., Mohammed, T., Mohammed, A., Mummed, B. & Regassa, D. (2021). Reproductive diseases and disorders of female camels: an assessment and pathological and bacteriological study in Eastern Ethiopia. Veterinary Medicine International, 2021, 6641361.
Belina, D., Eshetu, A., Alemu, S., Shasho, B., Mohammed, T., Mohammed, A., Mummed, B. & Regassa, D. (2021). Reproductive diseases and disorders of female camels: an assessment and pathological and bacteriological study in Eastern Ethiopia. Veterinary Medicine International, 2021, 6641361.
Benaissa, M. H., Faye, B. & Kaidi, R. (2014). Ovarian hydrobursitis in slaughtered female camels (Camelus dromedarius) in Southeast Algeria. Emirates Journal of Food and Agriculture.
BeNAISSA, M. H., Faye, B. & Kaidi, R. (2015). Reproductive abnormalities in female camel (Camelus dromedarius) in Algeria: relationship with age, season, breed and body condition score. Journal of Camel Practice and Research, 22, 67-73.
Beniwal, B. & Chaudhry, A. (1984). Age at first calving in Bikaneri camel. Indian Journal of Animal Sciences, 54, 598-599.
Dawod, A. & Hamed, T. (2018). Ovarian reproductive affections in she-camels. Journal of Veterinary Science & Technology, 9, 562.
El Allali, K., El Bousmaki, N., Ainani, H. & Simonneaux, V. (2017). Effect of the camelid’s seminal plasma ovulation-inducing factor/β-NGF: a kisspeptin target hypothesis. Frontiers in Veterinary Science, 4, 99.
Getahun, A. M., Hunderra, G. C., Gebrezihar, T. G., Boru, B. G., Desta, N. T. & Ayana, T. D. (2021). Comparative study on lesions of reproductive disorders of cows and female dromedary camels slaughtered at Addis Ababa, Adama and Akaki abattoirs with bacterial isolation and characterization. BMC Veterinary Research, 17, 1-15.
Ghallab, R., Hassan, M. A., Askar, A. R., Rashad, A. & El-Shereif, A. A. (2024). A comparative study of follicular dynamics, hormonal profiles, ovarian measurements, and endometrial thickness between well-fed nulliparous and multiparous dromedary she-camels during the breeding season. Journal of Advanced Veterinary Research, 14, 754-759.
Gherissi, D. E., Bouzebda, Z., Afri, F. B., Lamraoui, R. & Chacha, F. (2019). Cross-sectional survey of congenital and acquired genital disorders in sahraoui female camels (Camelus dromedarius) at El Oued abattoir, south-east Algeria. Animal Research International, 16, 3450–3462-3450–3462.
Gherissi, D. E., Monaco, D., Bouzebda, Z., Bouzebda, F. A., Gaouar, S. B. S. & Ciani, E. (2020). Camel herds’ reproductive performance in Algeria: Objectives and thresholds in extreme arid conditions. Journal of the Saudi Society of Agricultural Sciences, 19, 482-491.
Hegazy, A., Al-Eknah, M., Kotb, M., Hemeida, N. & Serur, B. (2001). Pathological and microbiological studies on the reproductive tract of the female camel. Journal of the Egyptian Veterinary Medical Association, 4, 9-25.
Kelanemer, R., Adel, D., Medrouh, B., Belala, R., Sellali, S., Saidi, A., Kalem, A., Rahmoune, Y., Dellal, N. & Fettata, S. (2025). She-camel (Camelus Dromedarius) reproductive status and biochemical blood parameters assay in extensive breeding in southern Algeria. Livestock Science, 292, 105646.
Khanvilkar, A., Samant, S. & Ambore, B. (2009). Reproduction in camel. Veterinary World, 2, 72.
Mahdy, M. A. & Nasr Eldeen, M. S. (2024). True uterus didelphys in she-camel: a case report and review of literature. Frontiers in Veterinary Science, 11, 1419234.
Mansour, N. & Karen, A. (2021). Ovarian inactivity in female dromedary camels. Emirates Journal of Food and Agriculture, 33, 171-177.
Mohamed, R. H., El-Maaty, A. M. A., Mohamed, R. S., Wehrend, A., Ali, F. & Hussein, H. A. (2021). Investigations on the vaginal temperature, cycle stages, and steroid hormone concentrations during the breeding season in camels (Camelus dromedarius). Veterinary world, 14, 1102.
Mohammed, A. & Alshaibani, N. (2025). The Potential Impacts of Assisted Reproductive Techniques in Camel Development and Future Prospects: A Review. Indian Journal of Animal Research, 59.
Mohammed, A., Almuyidi, A., Almarri, H., Alkhalifah, H., Alhmad, A., Alali, H., Alhuwaish, O. & Alkhawaher, M. (2025). Unique Characteristics of Camel Body Systems: Adaptation to Harsh Conditions, Productive and Reproductive Performances: A Review. Indian Journal of Animal Research, 1-10.
Pearson, L. & Tibary, A. (2014). Reproductive failure in female camelids. Clinical Theriogenology, 6, 555-570.
Purohit, G., Vyas, S., Sarswat, C. S., Kumar, A. & Dholpuria, S. (2020). Interesting features of female dromedary (Camelus dromedarius) reproduction. Indian Journal of Animal Reproduction, 41, 2.
Skidmore, J., Billah, M. & Allen, W. (1996). The ovarian follicular wave pattern and induction of ovulation in the mated and non-mated one-humped camel (Camelus dromedarius). Journal of Reproduction and Fertility, 106, 185-192.
Tibary, A. & Anouassi, A. (2001). Retrospective study on an unusual form of ovario-bursal pathology in the camel (Camelus dromedarius). Theriogenology, 56, 415-424.
Tibary, A. (2009). Uterine diseases in camelids. 346-348.
Tibary, A., Picha, Y. & Pearson, L. (2011). Congenital and possibly hereditary causes of infertility in camelids.
Waqas, M.-S., AnOUASSI, A. & Tibary, A. (2025). Manipulation of ovarian activity in camelids. Clinical Theriogenology, 17, 36-48.