the Management of Epilepsy in Egypt
| Site: | EHC | Egyptian Health Council |
| Course: | Neurology Guidelines |
| Book: | the Management of Epilepsy in Egypt |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 19 May 2025 Download Guideline
- Executive Summary
- To determine whether the patient might have had an epileptic seizure, the patient's and eyewitness's (where possible) detailed histories should be taken . (strong recommendation)
- A neurological examination should be performed since recurrence can be predicted through an abnormal examination after a first seizure. (strong recommendation) .
- In children and young people, a specialist neurologist with good training and expertise in epilepsy should establish the diagnosis of epilepsy (strong recommendation).
- We recommend referral to a more specialized Epilepsy clinic If the epilepsy diagnosis cannot be definitely confirmed. (strong recommendation)
- A normal EEG should not exclude the diagnosis of epilepsy . (strong recommendation) .
- Epilepsy is a clinical diagnosis; however, EEG can be indicated .(conditional recommendation) .
- If there is clinical doubt, EEG should be used to classify epileptic seizures and epilepsy syndromes . (strong recommendation)
- In case of equivocal epilepsy diagnosis or suspected non-epileptic events, refer to tertiary center (strong recommendation)
- When findings of a standard EEG have not supported epilepsy diagnosis or classification, a sleep EEG should be performed . (strong recommendation)
- Increasing recording time to 60 minutes on conventional EEG is a more convenient and cost-effective method of enhancing the EEG diagnostic yield compared to multiple conventional EEGs (strong recommendation) ..
- In the case of patients with a likelihood of a non-convulsive epileptic seizure, as suggested by the clinical history, the finding of epileptiform abnormalities in EEG is specific to assess the risk of seizure recurrence . (strong recommendation) .
- We strong recommend using 21 electrodes should be used (10-20 system) periods when the eyes are closed and open should be included in the recordings (strong recommendation) .
- During EEG Photic stimulation, sleep deprivation, and hyperventilation can be used routinely (unless contraindicated ) (conditional recommendation) .
- In all patients with blackouts, we recommend the 12-lead electrocardiography (ECG) in older age groups with blackouts (strong recommendation) .
- Consider ECG as a routine channel during EEG recording and should be performed as a baseline before starting ASM (conditional recommendation) .
- Magnetic resonance imaging (MRI) epilepsy protocol should follow the ILAE consensus on the"recommendations for using structural MRI in the care of PWE" (strong recommendation)
- We recommend MRI for brain imaging in PWE (before the age of two years and in adults) (strong recommendation).
- Any patient with focal onset on history, EEG, or examination (unless clear evidence of benign focal epilepsy) is indicated for MRI(strong recommendation).
- We recommend Referral for neuropsychological for any PWE who is suffering from learning or occupational challenges and as a part of the pre-surgical evaluation (strong recommendation).
- In adults, appropriate blood tests should be considered (complete blood count (CBC), glucose, calcium, thyroid-stimulating hormone (TSH), plasma electrolytes) to specify possible causes and/or any significant comorbidity(strong recommendation).
- In adolescents and adults, urine toxicology screening should be done if addiction is suspected (strong recommendation).
- We recommend lumbar puncture (LP) for a child <6 months, a patient who fails to return to baseline /meningeal signs, or suspects high intracranial pressure (imaging before LP is mandatory) (strong recommendation).
- In children and young people, other investigations should be considered in special situations (including urine and blood biochemistry) to identify an underlying cause of epilepsy and exclude other diagnoses (strong recommendation)..
- In case of failure of the initial ASM (due to continued seizures or adverse effects), initiation of an additional drug should take place and then escalate up to an adequate or maximum tolerated dose (this drug may be an alternative first‐line or second‐line drug). Afterward, slow tapering off the first drug should be done and caution is needed during the changeover period (strong recommendation).
- In case the second drug is worthless, consider tapering of it or the first drug (conditional recommendation).
- We recommend combination therapy with the newer antiepipetic drugs in patients who do not achieve seizure freedom on monotherapy . (strong recommendation).
- We recommend Drug withdrawal after a minimum of 2-5 years without a seizure based on risk of seizure recurrence. Gradual withdrawal over 2-6 months is recommended.
If seizures recur, the effective well tolerated drug previously used should be restarted using the last effective dose . (strong recommendation).
- In self-limited epilepsy with centrotemporal spikes first line are carbamazepine, oxcarbazepine, valproate. And adjuvant therapy are eslicarpazepine, lacozamide, lamotrigine, levetiracetam, zonisamide (strong recommendation) .
- In self-limited epilepsy with autonomic seizures first line are carbamazepine, sodium valproate. And adjuvant therapy are eslicarpazepine, lacozamide, lamotrigine, levetiracetam, zonisamide (conditional recommendation) .
- In childhood occipital visual epilepsy first line are carbamazepine, lamotrigine, levetiracetam, oxcarbazepine, valproate. And adjuvant therapy are eslicarpazepine, lacozamide, lamotrigine, levetiracetam, zonisamide (strong recommendation) .
- In photosensitive occipital lobe epilepsy first line is avoid factors that provoke seizures, valproate and adjuvant therapy are benzodiazepines, levetiracetam (strong recommendation) .
- In childhood absence epilepsy first line are ethosuximide, valproic acid. And adjuvant therapy are a combination of ethosuximide and valproic acid or lamotrigine (strong recommendation) .
- In epilepsy with myoclonic absence first line are ethosuximide, valproic acid.and adjuvant therapyare a combination of ethosuximide and valproic acid. other options are lamotrigine (strong recommendation) .
- In epilepsy with eyelid myoclonia first line are ethosuximide, valproic acid.and adjuvant therapy are clonazepam, ethosuximide, valproic acid (conditional recommendation) .
- In myoclonic atonic epilepsy first line are valproate in GTCS, ethosuximide in absence seizures, steroids in the periods with multiple atonic seizures, and frequent, prolonged episodes of nonconvulsive status. and adjuvant therapy are lamotrigine, ketogenic diet (conditional recommendation) .
- In lennox-gastaut syndrome first line are lamotrigine, topiramate, valproate.and adjuvant therapy are lamotrigine, topiramate, valproate, rufinamide (strong recommendation) .
- In developmental and/or epileptic encephalopathy with spike-wave activation during sleep first line are standard ASMs, VPA, benzodiazepines, ethosuximide, ACTH or prednisone, high-dose benzodiazepines, intravenous immunoglobulins, epilepsy surgery.other options: acetazolamide, clobazam, lacozamide, lamotrigine, levetiracetam (conditional recommendation) .
- IN Febrile infection-related epilepsy syndrome First line ARE benzodiazepines and barbiturates for treatment of the acute event.And adjuvant therapy is ketogenic diet.other options: IVIG, cannabidiol, anakinra, immunomodulation such as tocilizumab or canakinumab, epilepsy surgery (strong recommendation).
- In hemiconvulsion, hemiplegia epilepsy syndrome first line are Steroids, NMDA receptor blocker as memantine, amantadine.and adjuvant therapy are carbamazepine, phenytoin in cases of persistent seizures.other options: lamotrigine, perampanel, rufinamide, topiramate, valproate, epilepsy surgery (conditional recommendation) .
- In juvenile absence epilepsy (JAE) first line are ethosuximide, lamotrigine, sodium valproate.and adjuvant therapy are a combination of any of the first lines (strong recommendation) .
- In juvenile myoclonic epilepsy (JME) first line aresodium valproate, topiramate.and adjuvant therapy are acetazolamide, lamotrigine, levetiracetam . (strong recommendation). .
- In idiopathic generalized tonic–clonic seizures first line are lamotrigine, sodium valproate. And adjuvant therapy are consider carbamazepine, oxcarbazepine (exacerbating myoclonic and absence seizures) (strong recommendation). .
- In focal aware and impaired awareness seizures first line are carbamazepine, eslicarpazepine acetate, lacozamide, lamotrigine, levetiracetam, oxcarbazepine, sodium valproate.and adjuvant/ add-on: lamotrigine, phenobarbital, phenytoin, topiramate, vigabatrin, zonisamide (strong recommendation).
- In focal to bilateral tonic-clonic seizure and generalized onset tonic-clonic seizure first line; lamotrigine, sodium valproate.and adjuvant/ add-on: levetiracetam, topiramate (strong recommendation). .
- In generalized non-motor (Absence) seizures first line: ethosuximide, sodium valproate,and adjuvant: clonazepam, lamotrigine, levetiracetam, topiramate, zonisamide (strong recommendation) .
- In myoclonic seizures first line: levetiracetam, sodium valproate, topiramate.and adjuvant are clonazepam, levetiracetam, piracetam (strong recommendation). .
- In tonic & atonic seizures first line: sodium valproateand adjuvant: lamotrigine, topiramate(strong recommendation) .
- In status Epilepticus (SE) the diagnostic evaluation begins in parallel with emergent Initial Therapy :
o For diagnosis, finger stick glucose should be checked, pulse oximeter and cardiac monitoring should be started as soon as possible. Blood and serum laboratory evaluation typically includes a CBC, basic metabolic panel, calcium and magnesium determinations (conditional recommendation) .
o In the case of febrile patients or if there is a suspected subarachnoid haemorrhage or central nervous system infection, LP should be performed, preferably after obtaining the CT scan. Non-contrast CT of the brain is the first imaging study to be considered in the Emergency Department (ED) if MRI is not feasible (conditional recommendation) .
o Emergent prehospital treatment with benzodiazepines is needed and assess any abnormalities (hypoxia, hypoglycemia, or hypotension must be managed accordingly) (strong recommendation) .
o If benzodiazepines are not administered to the patient before ED arrival and are still seizing, IV benzodiazepines should be included in the initial dosing when IV access is immediately available (strong recommendation) .
o When IV access is not available, IM, per rectum (PR), buccal, or intranasal benzodiazepines should be administered together with IV placement (strong recommendation) .
o Unless IV access is immediately available, initiate IM. In adults or children over 40 kg, IM midazolam 10 mg. In children 13–40 kg, the IM midazolam dose is 5 mg, and in children <13 kg, the IM midazolam dose is 0.2 mg/ kg (strong recommendation) .
o Among second-line ASMs, the first-line choice will be either phenytoin or levetiracetam in their standard loading doses. Also lacosamide can be used if the previous ASM failed or is unavailable. It would be better to start the second line simultaneously with the initial benzodiazepine rather than waiting for a response for benzodiazepine (strong recommendation) .
o If seizures have stopped after the initial ASM, and the patient had awakened, a maintenance dose of loading ASMs should be started if indicated and can be given either orally or intravenously (strong recommendation) .
l For treatment of Refractory SE early (within one hour) drug-induced coma with continuous IV infusion of an anaesthetic drug is recommended for RSE with EEG target of burst suppression (strong recommendation) .
l The recommended loading dose of IV midazolam infusion is 0.2 mg/kg at 2 mg/min. Then, repeated boluses every 5 min of 0.2–0.4 mg/kg should be administered until the seizures stop, up to a maximum loading dose of 2 mg/kg. Then, a continuous infusion at 0.05–2 mg/kg/h should be started (strong recommendation) .
- Propofol IV infusions are an alternative at 1–2 mg/kg IV over 3–5 min as a loading dose and then repeated boluses every 3–5 min of the same amount until the seizures stop (strong recommendation).
- The propofol infusion at a rate of 30–200 mcg/kg/min should then be maintained .Pentobarbital at 5 mg/kg as a loading dose, which is followed by a maintenance infusion of 1–3 mg/ kg may be used in children with refractory SE more frequently because of adverse effects with propofol (strong recommendation) .
- For treatment of Super-Refractory SE Thiopental should be initiated in the recommended dose with progressive weaning of the previous anesthetic over the following 24 hours (conditional recommendation).
- If a burst-suppression EEG pattern without epileptic activity has been achieved during 24 hours under thiopental and SE recurs after thiopental weaning, adding phenobarbital should be considered in therapeutic dose for at least 24 hours, after which weaning should again be tried .thiopental anesthesia (epileptiform discharges), then ketamine in the recommended dose should be associated with thiopental (conditional recommendation) .
- In case of persistence or recurrence of SRSE despite the adoption of all previous recommendations and the patient in burst suppression so should be maintained under combined anesthesia with thiopental and ketamine, and all of the following therapeutics should be considered (according to availability and applicability):
A. magnesium sulfate – intravenous bolus (4 g) followed by a continuous infusion (2 - 6 g/h) aiming for plasma levels of 3.5 mmol/L (conditional recommendation).
B. Immunotherapy (conditional recommendation) .
C. Ketogenic diet (conditional recommendation) .
D. Vagus nerve stimulation (conditional recommendation) .
F. Epilepsy surgery (conditional recommendation) .
G. electroconvulsive therapy (conditional recommendation) .
- Patients with focal Drug resistant epilepsy should be referred for evaluation in a specialized/tertiary epilepsy care facility. The expertise of multidisciplinary teams should include psychology, psychiatry, neuroradiology, social work, counseling, clinical nurse specialists, occupational therapy, neurology, neuroanesthesia, neurophysiology, and neurosurgery (conditional recommendation) .
- Once referred to a specialized/tertiary epilepsy facility the patient should undergo :
A. Video-EEG to characterize the epilepsy types and rule out PNES (conditional recommendation) .
B. Imaging (MRI- HARNESS protocol) studies must be scrutinized once epilepsy diagnosis is confirmed to ascertain the presence of an epileptogenic focus and facilitate a possible surgical work-up (conditional recommendation) .
C. In presence of high suspicion of focal seizures; if no epileptogenic lesion is found on MRI, other ancillary tests include:
1. Ictal single-photon emission computed tomography (SPECT) (conditional recommendation) .
2. Positron emission tomography (PET) (strong recommendation) .
3. Functional MRI for eloquent regions of the brain (strong recommendation) .
4. Invasive EEG can be considered in case of unclear localization, or in case a more precise definition of the relationship of the eloquent cortex to the epileptic cortex is needed (strong recommendation) .
l In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For extra-temporal lobe epilepsy related to eloquent areas but not included in the epileptogenic zone; awake craniotomy, mapping techniques, and ECoG guided resection should be applied (conditional recommendation) .
- In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For extra-temporal lobe epilepsy with eloquent areas included in the epileptogenic zone; implementation of procedure such as MST, VNS, or RNS should be applied (strong recommendation).
- In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For unilateral temporal lobe epilepsy with lateral temporal involvement, ATL should be applied (strong recommendation).
- In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For unilateral temporal lobe epilepsy with no lateral temporal involvement, either surgical procedures (ATL or SAH) or minimally invasive procedures (LITT or SRS) should be applied (strong recommendation).
- In the case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For bilateral temporal lobe epilepsy, either VNS or RNS should be applied (conditional recommendation) .
- In the case of hemispheric generalized or multifocal epilepsy, either hemispherectomy (anatomical or functional) or VNS should be applied (conditional recommendation) .
- In the case of generalized poorly localized epilepsy with mostly atonic attacks, palliative management with corpus callosotomy should be applied (conditional recommendation) .
- In case of generalized poorly localized epilepsy with minor atonic attacks, palliative management with VNS or DBS of the anterior nucleus of the thalamus should be applied (strong recommendation) .
- Recommendations
Section I: Definition and classification of epilepsy
To determine whether the patient might have had an epileptic seizure, the patient's and eyewitness's (where possible) detailed histories should be taken . (strong recommendation) (high quality evidence) [1].
A neurological examination should be performed since recurrence can be predicted through an abnormal examination after a first seizure . (strong recommendation) (high quality evidence) [1].
In children and young people, a specialist neurologist with good training and expertise in epilepsy should establish the diagnosis of epilepsy (strong recommendation) (high quality evidence) [2].
We recommend referral to a more specialized Epilepsy clinic If the epilepsy diagnosis cannot be definitely confirmed. (strong recommendation) (high quality evidence) [2].
· Electroencephalography for epilepsy diagnosis
Normal EEG should not exclude the diagnosis of epilepsy . (strong recommendation) (high quality evidence) [3] .
Epilepsy is a clinical diagnosis; however, EEG can be indicated .(conditional recommendation) (high quality evidence) [1] .
If there is clinical doubt, EEG should be used to classify epileptic seizures and epilepsy syndromes . (strong recommendation) (low quality evidence) [4] .
In case of equivocal epilepsy diagnosis or suspected non-epileptic events, refer to tertiary center (strong recommendation) (low quality evidence) [5] .
When findings of a standard EEG have not supported epilepsy diagnosis or classification, a sleep EEG should be performed . (strong recommendation) (low quality evidence) [6] .
Increasing recording time to 60 minutes on conventional EEG is a more convenient and cost-effective method of enhancing the EEG diagnostic yield compared to multiple conventional EEGs (strong recommendation) (high quality evidence) [7] .
In the case of patients with a likelihood of a non-convulsive epileptic seizure, as suggested by the clinical history, the finding of epileptiform abnormalities in EEG is specific to assess the risk of seizure recurrence . (strong recommendation) (high quality evidence) [8] .
We strong recommend using 21 electrodes and should be used (10-20 system) periods when the eyes are closed and open should be included in the recordings (strong recommendation) (low quality evidence) [9] .
During EEG Photic stimulation, sleep deprivation, and hyperventilation can be used routinely (unless contraindicated ) (conditional recommendation) (low quality evidence) [9] .
· Electrocardiography for epilepsy diagnosis
In all patients with blackouts, we recommend the 12-lead electrocardiography (ECG) in older age groups with blackouts (strong recommendation) (high quality evidence) [10] .
Consider ECG as a routine channel during EEG recording and should be performed as a baseline before starting ASM (conditional recommendation) (low quality evidence) [9] .
· Magnetic resonance imaging for epilepsy diagnosis
Magnetic resonance imaging (MRI) epilepsy protocol should follow the ILAE consensus on the"recommendations for using structural MRI in the care of PWE" (strong recommendation) (moderate quality evidence) [11] .
We recommend MRI for brain imaging in PWE (before the age of two years and in adults)
(strong recommendation) (high quality evidence) [1] . Any patient with focal onset on
history, EEG, or examination (unless clear evidence of benign focal epilepsy) is indicated
for MRI (strong recommendation) (high quality evidence) [1] .
· Neuropsychological assessment
We recommend Referral for neuropsychological for any PWE who is suffering from learning or occupational challenges and as a part of the pre-surgical evaluation (strong recommendation) (high quality evidence) [12] .
· Laboratory studies
In adults, appropriate blood tests should be considered (complete blood count (CBC), glucose, calcium, thyroid-stimulating hormone (TSH), plasma electrolytes) to specify possible causes and/or any significant comorbidity(strong recommendation) (high quality evidence) [1].
In adolescents and adults, urine toxicology screening should be done if addiction is suspected (strong recommendation) (high quality evidence) [1] .
We recommend lumbar puncture (LP) for a child <6 months, a patient who fails to return to baseline /meningeal signs, or suspects high intracranial pressure (imaging before LP is mandatory) (strong recommendation). (high quality evidence) [1] .
In children and young people, other investigations should be considered in special situations (including urine and blood biochemistry) to identify an underlying cause of epilepsy and exclude other diagnoses (strong recommendation). (high quality evidence) [1] .
Section II: Choice of Anti-Seizure Medication (ASM)
1. General Considerations for choosing the ASM:
In case of failure of the initial ASM (due to continued seizures or adverse effects), initiation of an additional drug should take place and then escalate up to an adequate or maximum tolerated dose (this drug may be an alternative first‐line or second‐line drug). Afterward, slow tapering off the first drug should be done and caution is needed during the changeover period (strong recommendation) (high quality evidence) [13] .
In case the second drug is worthless, consider tapering of it or the first drug (conditional recommendation) (low quality evidence) [14] .
We recommend combination therapy with the newer antiepipetic drugs in patients who do not achieve seizure freedom on monotherapy . (strong recommendation) (high quality evidence) [13] .
We recommend Drug withdrawal after a minimum of 2-5 years without a seizure based on risk of seizure recurrence. Gradual withdrawal over 2-6 months is recommended. If seizures recur, the effective well tolerated drug previously used should be restarted using the last effective dose . (strong recommendation) (moderate quality evidence) [15,16] .
2. Choice of ASM According to Different Epileptic Syndromes in Different Age Groups (after excluding treatable metabolic causes):
2.A. Self-limited epilepsy with centrotemporal spikes:
First line: carbamazepine, oxcarbazepine, valproate.
Adjuvant therapy: eslicarpazepine, lacozamide, lamotrigine, levetiracetam, zonisamide.
(strong recommendation) (high quality evidence) [17-21].
2.B. Self-limited epilepsy with autonomic seizures
First line: carbamazepine, sodium valproate.
Adjuvant therapy: eslicarpazepine, lacozamide, lamotrigine, levetiracetam, zonisamide.
(conditional recommendation) (low quality evidence) [22].
2.C. Childhood occipital visual epilepsy
First line: carbamazepine, lamotrigine, levetiracetam, oxcarbazepine, valproate.
Adjuvant therapy: eslicarpazepine, lacozamide, lamotrigine, levetiracetam, zonisamide.
(strong recommendation) (high quality evidence) [23] .
2.D. Photosensitive occipital lobe epilepsy
First line: avoid factors that provoke seizures, valproate.
Adjuvant therapy: benzodiazepines, levetiracetam. (strong recommendation)
(high quality evidence) [23] .
2.E. Childhood absence epilepsy
First line: ethosuximide, valproic acid.
Adjuvant therapy: a combination of ethosuximide and valproic acid or lamotrigine (strong recommendation) (high quality evidence) [24] .
2.F. Epilepsy with myoclonic absence
First line: ethosuximide, valproic acid.
Adjuvant therapy: a combination of ethosuximide and valproic acid.
Other options: lamotrigine (strong recommendation) (high quality evidence) [25].
2.G. Epilepsy with eyelid myoclonia
First line: ethosuximide, valproic acid.
Adjuvant therapy: clonazepam, ethosuximide, valproic acid.
(conditional recommendation) (low quality evidence) [26] .
2.H. Myoclonic atonic epilepsy
First line: valproate in GTCS, ethosuximide in absence seizures, steroids in the periods with multiple atonic seizures, and frequent, prolonged episodes of nonconvulsive status.
Adjuvant therapy: lamotrigine, ketogenic diet.
(conditional recommendation) (low quality evidence) [27].
2.I. Lennox-gas taut syndrome
First line: lamotrigine, topiramate, valproate.
Adjuvant therapy: lamotrigine, topiramate, valproate, rufinamide.
(strong recommendation) (high quality evidence) [28-30].
2.J. Developmental and/or epileptic encephalopathy with spike-wave activation during sleep
First line: standard ASMs, VPA, benzodiazepines, ethosuximide, ACTH or prednisone, high-dose benzodiazepines, intravenous immunoglobulins, epilepsy surgery.
Other options: acetazolamide, clobazam, lacozamide, lamotrigine, levetiracetam.
(conditional recommendation) (low quality evidence) [31] .
2.K, Febrile infection-related epilepsy syndrome
First line: benzodiazepines and barbiturates for treatment of the acute event.
Adjuvant therapy: ketogenic diet.
Other options: IVIG, cannabidiol, anakinra, immunomodulation such as tocilizumab or canakinumab, epilepsy surgery.(strong recommendation) (high quality evidence) [32].
2.L. Hemiconvulsion, hemiplegia epilepsy syndrome
First line: Steroids, NMDA receptor blocker as memantine, amantadine.
Adjuvant therapy: carbamazepine, phenytoin in cases of persistent seizures.
Other options: lamotrigine, perampanel, rufinamide, topiramate, valproate, epilepsy surgery.(conditional recommendation) (low quality evidence) [33].
II.3. Choice of ASM According to Different Epileptic Syndromes in Adolescents:
3.A. Juvenile absence epilepsy (JAE)
First line: ethosuximide, lamotrigine, sodium valproate.
Adjuvant therapy: a combination of any of the first lines.(strong recommendation) (high quality evidence) [34].
3.B. Juvenile myoclonic epilepsy (JME)
First line: sodium valproate, topiramate.
Adjuvant therapy: acetazolamide, lamotrigine, levetiracetam .
(strong recommendation) (high quality evidence) [35,36].
3.C.Idiopathic generalized tonic–clonic seizures
First line: lamotrigine, sodium valproate.
Adjuvant therapy: consider carbamazepine, oxcarbazepine (exacerbating myoclonic and absence seizures). (strong recommendation) (high quality evidence)[37].
II.4. Choice of ASM According to Seizure Type:
4.A. Focal aware and impaired awareness seizures
First line: carbamazepine, eslicarpazepine acetate, lacozamide, lamotrigine, levetiracetam, oxcarbazepine, sodium valproate.
Adjuvant/ Add-on: lamotrigine, phenobarbital, phenytoin, topiramate, vigabatrin, zonisamide (strong recommendation) (high quality evidence) [18].
4.B. Focal to bilateral tonic-clonic seizure and generalized onset tonic-clonic seizure
First line; lamotrigine, sodium valproate.
Adjuvant/ Add-on: levetiracetam, topiramate (strong recommendation) (high quality evidence).[38,39].
4.C. Generalized non-motor (Absence) seizures
First line: ethosuximide, sodium valproate
Adjuvant: clonazepam, lamotrigine, levetiracetam, topiramate, zonisamide (strong recommendation) (high quality evidence) [24].
4.D. Myoclonic seizures
First line: levetiracetam, sodium valproate, topiramate.
Adjuvant: clonazepam, levetiracetam, piracetam (strong recommendation) (high quality evidence) [35,36].
$.E. Tonic & atonic seizures
First line: sodium valproate
Adjuvant: lamotrigine, topiramate (strong recommendation) (high quality evidence) [28-30]
Section III: Status Epilepticus (SE)
III.1. Diagnosis and Evaluation of SE:
The diagnostic evaluation begins in parallel with Emergent Initial Therapy.
For diagnosis, finger stick glucose should be checked, pulse oximeter and cardiac monitoring should be started as soon as possible. Blood and serum laboratory evaluation typically includes a CBC, basic metabolic panel, calcium and magnesium determinations (conditional recommendation) (low quality evidence) [40].
In the case of febrile patients or if there is a suspected subarachnoid haemorrhage or central nervous system infection, LP should be performed, preferably after obtaining the CT scan. Non-contrast CT of the brain is the first imaging study to be considered in the Emergency Department (ED) if MRI is not feasible (conditional recommendation) (low quality evidence).[40].
III.2. Emergent treatment of SE:
Emergent prehospital treatment with benzodiazepines is needed while assessing any abnormalities (hypoxia, hypoglycemia, or hypotension must be managed accordingly).
(strong recommendation) (high quality evidence) [41,42].
If benzodiazepines are not administered to the patient before ED arrival and are still seizing, IV benzodiazepines should be included in the initial dosing when IV access is immediately available.
(strong recommendation) (high quality evidence) [43].
When IV access is not available, IM, per rectum (PR), buccal, or intranasal benzodiazepines should be administered together with IV placement (strong recommendation) (high quality evidence)[44].
Unless IV access is immediately available, initiate IM. In adults or children over 40 kg, IM midazolam 10 mg. In children 13–40 kg, the IM midazolam dose is 5 mg, and in children <13 kg, the IM midazolam dose is 0.2 mg/ kg (strong recommendation) (high quality evidence)[44].
Among second-line ASMs, the first-line choice will be either phenytoin or levetiracetam in their standard loading doses. Also lacosamide can be used if the previous ASM failed or is unavailable. It would be better to start the second line simultaneously with the initial benzodiazepine rather than waiting for a response for benzodiazepine.(strong recommendation) (high quality evidence)[45].
If seizures have stopped after the initial ASM, and the patient had awakened, a maintenance dose of loading ASMs should be started if indicated and can be given either orally or intravenously
(strong recommendation) (high quality evidence) [45].
III.3. Treatment of Refractory SE:
Early (within one hour) drug-induced coma with continuous IV infusion of an anaesthetic drug is recommended for RSE with EEG target of burst suppression (strong recommendation) (high quality evidence) [46].
The recommended loading dose of IV midazolam infusion is 0.2 mg/kg at 2 mg/min. Then, repeated boluses every 5 min of 0.2–0.4 mg/kg should be administered until the seizures stop, up to a maximum loading dose of 2 mg/kg. Then, a continuous infusion at 0.05–2 mg/kg/h should be started (strong recommendation) (high quality evidence) [47].
Propofol IV infusions are an alternative at 1–2 mg/kg IV over 3–5 min as a loading dose and then repeated boluses every 3–5 min of the same amount until the seizures stop. The propofol infusion at a rate of 30–200 mcg/kg/min should then be maintained (strong recommendation) (high quality evidence) [48].
Pentobarbital at 5 mg/kg as a loading dose, which is followed by a maintenance infusion of 1–3 mg/ kg may be used in children with refractory SE more frequently because of adverse effects with propofol (strong recommendation) (high quality evidence) [49].
III.4. Treatment of Super-Refractory SE:
Thiopental should be initiated in the recommended dose with progressive weaning of the previous anesthetic over the following 24 hours (conditional recommendation) (low quality evidence) [50].
If a burst-suppression EEG pattern without epileptic activity has been achieved during 24 hours under thiopental and SE recurs after thiopental weaning, adding phenobarbital should be considered in therapeutic dose for at least 24 hours, after which weaning should again be tried (conditional recommendation) (low quality evidence) [51].
If SRSE recurs after weaning of thiopental in association with phenobarbital or if it persists under thiopental anesthesia (epileptiform discharges), then ketamine in the recommended dose should be associated with thiopental (conditional recommendation) (low quality evidence) [52].
In case of persistence or recurrence of SRSE despite the adoption of all previous recommendations; then the patient should be maintained in burst suppression under combined anesthesia with thiopental and ketamine, and all of the following therapeutics should be considered (according to availability and applicability):
· A. magnesium sulfate – intravenous bolus (4 g) followed by a continuous infusion (2 - 6 g/h) aiming for plasma levels of 3.5 mmol/L (conditional recommendation) (low quality evidence) [53].
· B. Immunotherapy (conditional recommendation) (low quality evidence) [54].
· C. Ketogenic diet (conditional recommendation) (low quality evidence) [55].
· D. Vagus nerve stimulation (conditional recommendation) (low quality evidence) [56].
· F. Epilepsy surgery (conditional recommendation) (low quality evidence) [57].
· G. electroconvulsive therapy (conditional recommendation) (low quality evidence) [58].
Section IV: Drug-Resistant Epilepsy (DRE)
IV.1. Characteristics of a specialized/tertiary epilepsy facility
Patients with focal DRE should be referred for evaluation in a specialized/tertiary epilepsy care facility. The expertise of multidisciplinary teams should include psychology, psychiatry, neuroradiology, social work, counseling, clinical nurse specialists, occupational therapy, neurology, neuroanesthesia, neurophysiology, and neurosurgery (conditional recommendation) (low quality evidence) [40].
Once referred to a specialized/tertiary epilepsy facility the patient should undergo
A. Video-EEG to characterize the epilepsy types and rule out PNES [23] (conditional recommendation) (low quality evidence).
B. Imaging (MRI- HARNESS protocol) studies must be scrutinized once epilepsy diagnosis is confirmed to ascertain the presence of an epileptogenic focus and facilitate a possible surgical work-up (conditional recommendation) (low quality evidence) [59][11] .
C. In presence of high suspicion of focal seizures; if no epileptogenic lesion is found on MRI, other ancillary tests include:
1. Ictal single-photon emission computed tomography (SPECT) (conditional recommendation) (low quality evidence) [60].
2. Positron emission tomography (PET). (strong recommendation) (high quality evidence) [61].
3. Functional MRI for eloquent regions of the brain (strong
recommendation) (high quality evidence) [62].
4. Invasive EEG can be considered in case of unclear localization, or in case a more precise definition of the relationship of the eloquent cortex to the epileptic cortex is needed (strong recommendation) (high quality evidence) [63].
IV.2. Epilepsy surgery referrals in Egypt:
In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For extra-temporal lobe epilepsy related to eloquent areas but not included in the epileptogenic zone; awake craniotomy, mapping techniques, and ECoG guided resection should be applied (conditional recommendation) (low quality evidence) [64].
In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For extra-temporal lobe epilepsy with eloquent areas included in the epileptogenic zone; implementation of procedure such as MST, VNS, or RNS should be applied (strong recommendation) (high quality evidence).[65]
In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For unilateral temporal lobe epilepsy with lateral temporal involvement, ATL should be applied (strong recommendation) (high quality evidence) [66].
In case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For unilateral temporal lobe epilepsy with no lateral temporal involvement, either surgical procedures (ATL or SAH) or minimally invasive procedures (LITT or SRS) should be applied (strong recommendation) (high quality evidence) [66].
In the case of the localized epileptogenic zone (i.e. for lesional focal epilepsy in concordance with semiology and EEG, or non-lesional focal epilepsy with the localized epileptogenic zone); For bilateral temporal lobe epilepsy, either VNS or RNS should be applied [67, 68] (conditional recommendation) (low quality evidence).
In the case of hemispheric generalized or multifocal epilepsy, either hemispherectomy (anatomical or functional) or VNS should be applied [69] (conditional recommendation) (low quality evidence).
In the case of generalized poorly localized epilepsy with mostly atonic attacks, palliative management with corpus callosotomy should be applied (conditional recommendation) (low quality evidence).[70]
In case of generalized poorly localized epilepsy with minor atonic attacks, palliative management with VNS.[71] or DBS of the anterior nucleus of the thalamus should be applied (strong recommendation) (high quality evidence) [72].
Implementation consideration
It is advised to provide training courses for the physicians at the centers that diagnose and treat epilepsy and in neurophysiology units too .
Clinical indicators for monitoring
For monitoring of the implementation of the guidelines, the files of the patients presenting with the status epilepticus event should include:
- The date of onset of first seizure
- ABCD evaluation
- Treatment of status according to:
First line : Midazolam iv injection or im.
Second line : Phenytoin or levetiracetam.
Third line : midazolam infusion , propofol or phenobarbital in children.
Fourth line : thiopental , ketamine or combined thiopental and ketamine.
- Acknowledgement
We would like to acknowledge the Committee of National Egyptian Guidelines,Ministry of Health and Neurology Scientific Committee for adapting these Guidelines.We would like to acknowledge the epilepsy chapter of Egyptian Society of Neurology, Psychiatry and Neurosurgery(ESNPN)
Chair of the panel: Hassan Hosni
Scientific group members: Azza Abdlnasser, Ahmed Elbassiouny, Mona Ahmed Nada, Magdy Khalaf, Tarek Rageh, Ahmed Attia, Romany Adly, Mohamed Mahmoud Fouad , Ahmed Fawzi Amin , Haitham Elnabawy
- Abbreviations
ACTH: Adrenocorticotropic hormone
ASM: Anti-seizure medications
ATL: Anterior Temporal Lobectomy
CBC: Complete blood count
CT: Computed Tomography
DBS: Deep Brain Stimulation
DP: Delphi panel
DRE: Drug-Resistant Epilepsy
ECG: Electrocardiography
ECoG: Electrocorticography
ED: Emergency Department
EEG: Electroencephalography
EMU: Epilepsy monitoring unit
ESES: Electrical status epilepticus during sleep
EZ: Epileptogenic Zone
GGE: Genetically Generalized Epilepsy
IEEG: Intracranial Electroencephalography
ILAE: International League Against Epilepsy
IM: Intramuscular
IV: Intravenous
JAE: Juvenile absence epilepsy
JME: Juvenile myoclonic epilepsy
LITT: Laser Interstitial Thermal Therapy
LP: Lumbar puncture
MEG: Magnetoencephalography
MRI: Magnetic resonance imaging
MRS: Magnetic Resonance Spectroscopy
MST: Multiple Subpial Transections
NCSE: Non-convulsive status epilepticus
NMDA: N-methyl-D-aspartate
PET: Positron emission tomography
PNES: Psychogenic non-epileptic seizures
PR: Per rectum
PWE: Persons with epilepsy
RNS: Responsive Neurostimulation
RSE: Refractory Status Epilepticus
SAH: Selective Amigdalo-Hippocampectomy
SC: Steering Committee
SD grids: Subdural grids
SE: Status epilepticus
SEC: Specialized Epilepsy Center
SEEG: Stereotactic Electroencephalography
SISCOM: Subtraction Ictal SPECT co-registered with MRI
SPECT: Single-photon emission computed tomography
SRS: Stereotactic Radiosurgery
SRSE: Super-Refractory Status Epilepticus
TSH: Thyroid-stimulating hormone
VNS: Vagal nerve stimulation
WHO: World Health Organization.
- Glossary
· Drug-Resistant Epilepsy: the ILAE definition of DRE as “failure of adequate trials of two or three tolerated, appropriately chosen and used antiepileptic drug schedules (whether as monotherapies or in combination) to achieve sustained seizure freedom”.
· Epilepsy: the ILAE proposed a practical clinical definition for epilepsy include any of the following:
(1) at least two unprovoked (or reflex) seizures occurring more than 24 hours apart,
(2) one unprovoked (or reflex) seizure and a probability of further seizures similar to the general recurrence risk (at least 60%) after two unprovoked seizures, occurring over the next 10 years, or (3) diagnosis of an epilepsy syndrome
· Epileptic seizure: the International League Against Epilepsy (ILAE) defined it as “a transient occurrence of signs and/or symptoms due to abnormal excessive or synchronous neuronal activity in the brain.”
· Epilepsy treatment gap is defined by the International League Against Epilepsy as follows: “The difference between the number of people with active epilepsy and the number whose seizures are being appropriately treated in a given population at a given time expressed as a percentage. This definition includes diagnostic and therapeutic deficits.”
· Refractory Status Epilepticus (RSE): status epilepticus that persists despite administration of at least two appropriately chosen and dosed parenteral antiseizures medications that include benzodiazepine. No specific seizure duration is required.
· Status epilepticus (SE): a neurologic emergency characterized by prolonged seizure with 5 minutes or more of continuous clinical and/or convulsive seizure activity or recurrent seizure activity without recovery of consciousness between seizures with substantial morbidity and mortality rates.
· Super-refractory status epilepticus: SE that persists for at least 24 hours after the start of anesthesia, whether it occurs without interruption despite appropriately selected treatment with anesthesia, or if it recurs while on appropriately selected and dosed anesthetic treatment, or it recurs after withdrawal of anesthesia and requires reintroduction of anesthetic.
- Scope and Purpose
This guideline covers the definition, diagnosis, and management of epilepsy within different age groups in Egypt. Specifically, the goals are to improve diagnostic accuracy, identify DRE patients, and educate clinicians regarding the suitable line of management of epilepsy.
- Introduction
Epilepsy is a chronic brain disorder that is marked by episodes of seizures. The prevalence of epilepsy is around 50 million people worldwide, per the World Health Organization (WHO) figures, and most (almost 80%) reside in low- and middle-income countries. Moreover, the premature mortality risk is almost threefold in persons with epilepsy (PWE) compared to the general public. The prevalence of epilepsy in Egypt was estimated to be 6.98/1000.
Establishing the definitive epilepsy diagnosis and detecting the root cause are challenging tasks, particularly where cultural constraints hinder disease acceptance.
One out of every five patients treated for epilepsy does not really have the disease. Further, the number of available anti-seizure medications (ASMs) has been notable in recent years, making the proper treatment choice more complex. According to the WHO data, proper diagnosis and treatment of epilepsy can provide seizure freedom for 70% of PWE. The remaining 30% presents a major challenge due to resistance and refractoriness to drugs. As in other parts of the world, inadequacies in the care of PWE, such as misdiagnosis, inappropriate medication, and sudden unexpected death, are all well recognized.
We sincerely hope this can be changed, which is now changing with the emergence of this nationwide consensus. With all the efforts that this initiative bears, it represents one of a series of efforts devoted to creating a significant impact on the health of Egyptians in recent years.
We believe implementing these structured recommendations will substantially improve the quality of care and treatment outcomes for PWE across all age groups (children and adults) in Egypt.
- The target audience
The guideline is intended for all clinicians who are likely to diagnose and manage PWE, and it applies to any setting in which epilepsy would be diagnosed or managed.
- Methods
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2015 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author not on behalf of an organization in order to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
All Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The Guideline Development Group has decided to adapt the current guidelines guided by most recent guidelines and standard of practice reported from
NICE guideline 2022 and updated in 2025
https://www.nice.org.uk/guidance/ng217 was used for adaptation
Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the GRC secretariat and on the following sites:
■ GRADE working group: https://www.gradeworkinggroup.org/
■ GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
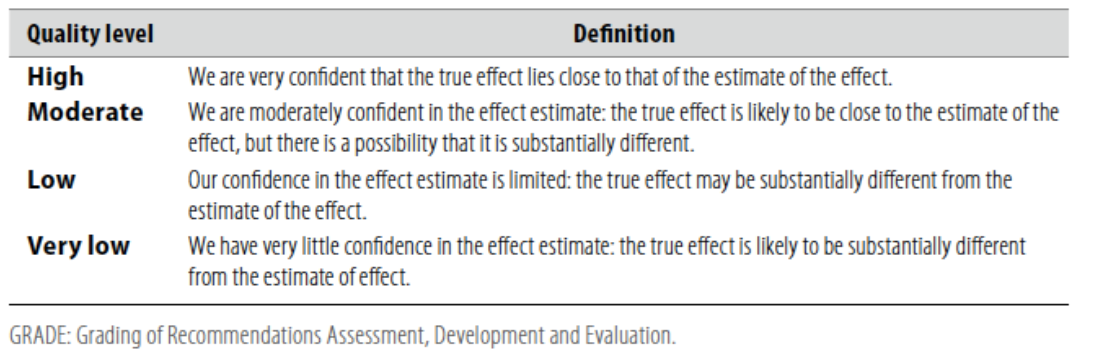
■ GRADE profile software: http://ims.cochrane.org/revman/gradepro
Table 2: Significance of the four level of evidence
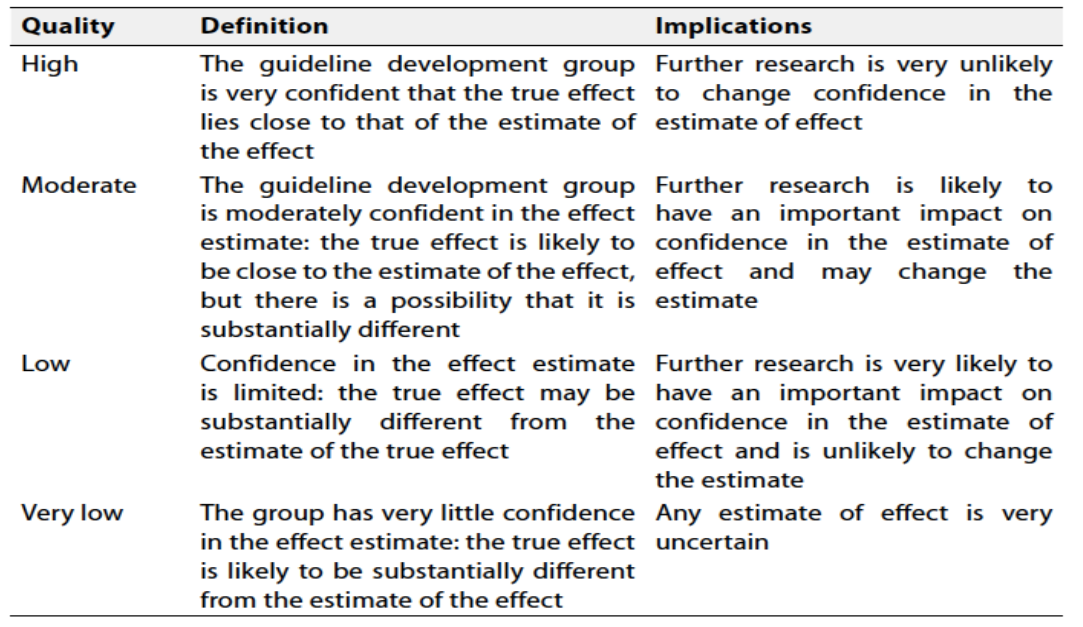
Table 3: Factors that determine How to upgrade or downgrade the quality of Evidence
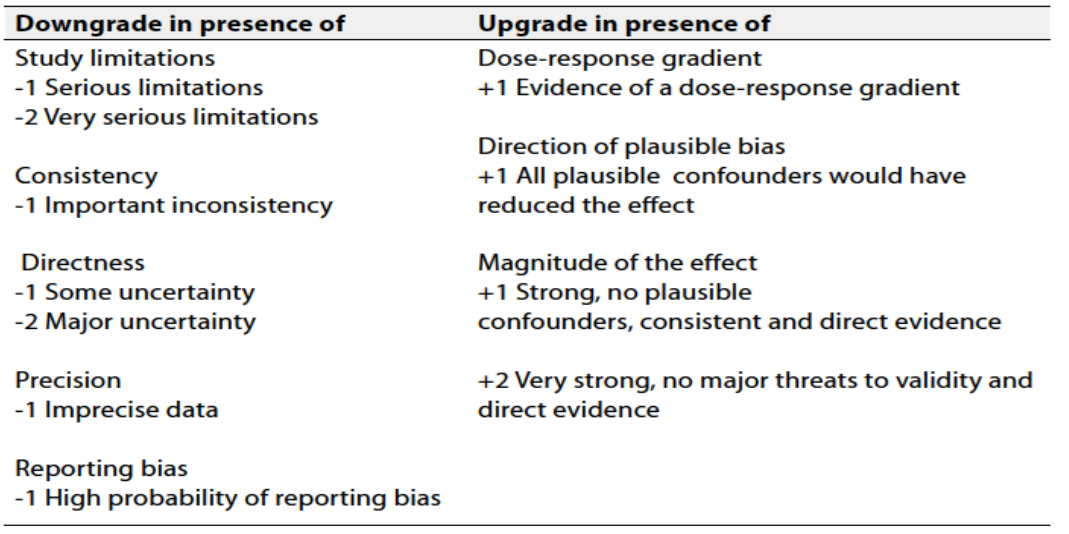
The strength of the recommendation: The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Updating of the guidelines
To keep these recommendations up to date and ensure its validity it will be periodically updated. This will be done whenever strong new evidence is available and necessitates updating.
- Research needs
1. Public awareness about various non-pharmacological lines of treatment in DRE patients, particularly epilepsy surgery.
2. Develop prognostic indicators to identify the best candidates for epilepsy surgery.
3. Cost-effectiveness studies of various non-pharmacological lines of treatment in DRE patients.
- Annexes
Valproic acid (VPA): ia as a highly prevalent medication with multifaceted therapeutic applications in various neurological and psychiatric disorders. The therapeutic uses of VPA include epilepsy treatment across different seizure types, The mechanism of action for VPA involves the enhancement of inhibitory neurotransmission and the modulation of voltage-gated ion channels, If taken during pregnancy, sodium valproate can cause problems for a baby's development, including birth defects and lifelong learning difficulties. Sodium valproate is not recommended if there's a chance that you could become pregnant. [73].
Phenytoin: The FDA approved phenytoin in 1939 for the treatment of epilepsy) generalized tonic-clonic seizures, complex partial seizures, status epilepticus(
it is a hydantoin derivative, a first-generation anti-convulsant drug that is effective in the treatment of generalized tonic-clonic seizures, complex partial seizures, and status epilepticus without significantly impairing neurological function.
Phenytoin works by blockade of voltage-dependent membrane sodium channels responsible for increasing the action potential. This action obstructs the positive feedback that sustains high-frequency repetitive firing, thus preventing the spread of the seizure focal point.
Hypersensitivity to phenytoin or other hydantoins is a contraindication for using phenytoin. Pregnancy is another absolute contraindication for phenytoin use.
Hanson et al. reported a prevalence of fetal hydantoin syndrome (FHS) of 11% in pregnant women receiving treatment for epilepsy with phenytoin, with an additional 30% of the in utero-exposed children expressing some of the syndrome’s features such as epicanthic folds, hypertelorism, broad flat nasal bridges, an upturned nasal tip, wide prominent lips and, also, distal digital hypoplasia, intrauterine growth retardation, and diminished mental capacity. [74]
Carbamazapine: is FDA indicated for epilepsy, trigeminal neuralgia, and acute manic and mixed episodes in bipolar I disorder. Indications for epilepsy are specifically for partial seizures with complex symptomatology (psychomotor, temporal lobe), generalized tonic seizures (grand mal), and mixed seizure patterns. Carbamazepine is not indicated for absence seizures
It modulates voltage-gated sodium channels (VGSC), causing inhibition of action potentials and decreased synaptic transmission. Similar to other anti-convulsions, carbamazepine is suggested to bind to the alpha subunit of VGSC, specifically at a binding pocket formed by the external pore loop and the pore-lining part of domain IV. Researchers proposed that carbamazepine keeps sodium channels in inactivated states, leading to fewer channels to open and thus inhibits the generation of action potentials. Carbamazepine also binds to other voltage-gated ion channels, such as voltage-gated calcium channels
it is contraindicated in patients with bone marrow depression and hypersensitivity to this drug or tricyclic compounds such as amitriptyline. Before administration, monoamine oxidase inhibitors should be discontinued for a minimum of 14 days. Usage carbamazepine and nefazodone together may result in insufficient plasma concentration of nefazodone. Carbamazepine is contraindicated in use with nefazodone. [75]
Oxcarbazepine :is a 10-keto derivative of carbamazepine, which came to the market in 2000. However, the minor structural differences between oxcarbazepine and carbamazepine have led to significant differences in the induction of metabolic pathways and the metabolism of the two medications
Oxcarbazepine is a first-line choice for treating focal-onset epilepsy in several countries, including the USA. The oral suspension is a popular dosage form among clinicians, oxcarbazepine might be efficacious in decreasing seizure frequency for drug-resistant epilepsy when used as an add-on drug. Oxcarbazepine can cause ataxia, somnolence, and hyponatremia
Avoid abrupt withdrawal of oxcarbazepine. Prescribers should exercise caution in pediatric, elderly, pregnant, and renal impairment patients who have had a hypersensitivity reaction to carbamazepine. It is also contraindicated in treatment-naive patients with the HLA-B*1502 allele. [76]
Eslicarbazepine acetate: is a new anti-epileptic drug belonging to the dibenzazepine carboxamide family that is currently approved as adjunctive therapy and monotherapy for partial-onset (focal) seizures. The drug enhances slow inactivation of voltage-gated sodium channels and subsequently reduces the activity of rapidly firing neurons. Eslicarbazepine acetate has few, but some, drug–drug interactions. It is a weak enzyme inducer and it inhibits cytochrome P450 2C19, but it affects a smaller assortment of enzymes than carbamazepine. Clinical studies using eslicarbazepine acetate as adjunctive treatment or monotherapy have demonstrated its efficacy in patients with refractory or newly diagnosed focal seizures. The drug is generally well tolerated, and the most common side effects include dizziness, headache, and diplopia. One of the greatest strengths of eslicarbazepine acetate is its ability to be administered only once per day. Eslicarbazepine acetate has many advantages over older anti-epileptic drugs, and it should be strongly considered when treating patients with partial-onset epilepsy. [77]
Levetiracetam :is a novel antiepileptic drug used to treat partial, myoclonic, and tonic-clonic seizures. In 2000, the FDA approved the use of the oral formulation as adjunctive therapy for the treatment of focal seizures, myoclonic seizures, and primary generalized seizures. In addition, the FDA approved intravenous levetiracetam (LEV) in 2006 for use in patients older than 15 years as adjunctive anticonvulsant therapy when the oral formulation is not tolerated. In Europe, it is approved for treating partial seizures as a single agent and as an add-on treatment for partial seizures, tonic-clonic seizures, and myoclonic seizures.
It is chemically unrelated to other antiepileptic drugs. However, its favorable safety profile, distinct mechanism of action, and fewer drug interactions make it an attractive therapeutic choice for treating seizures.
Most common adverse effects are neurobehavioral, like sedation, fatigue, mood swings, headache, agitation, irritability, aggression, depression, memory loss, confusion, paresthesia, a decline in cognition, and increased suicide risk. Most of the time, the side effects are mild. [78].
Brivaracetam: is a third-generation antiepileptic racetam derivative and a 4-n-propyl analogue of levetiracetam. While the exact mechanism of action of brivaracetam is unknown, its anticonvulsant effects are believed to be due to its highly selective affinity for synaptic vesicle protein 2A (SV2A) in the brain. The SV2A glycoprotein is a protein-coding gene implicated in synaptic signal transduction. It is believed to play a role in the regulation of neurotransmission by stimulating vesicle fusion and maintaining a reserve of secretory vesicles. Studies in SV2A-deficient animals have demonstrated an increased propensity for seizures. Brivaracetam’s actions as an SV2A ligand lend it broad-spectrum activity against partial onset seizure.
Brivaracetam approved as adjunctive therapy for the treatment of POS in adults and adolescents 16 years of age and older. This medication has shown promise in patients with epilepsy whose seizures persist despite adequate treatment. Brivaracetam was well tolerated throughout all three phase 3 trials, which led to its FDA approval. The drug offers improved seizure control with minimal safety risks for patients with epilepsy. [79]
Topiramate: is approved as monotherapy for epilepsy for individuals aged 2 years and older with primary generalized onset tonic-clonic or partial-onset seizures. Additionally, it is sanctioned for adjunctive therapy in adults and pediatric populations from ages 2 to 16 years with primary generalized onset tonic-clonic seizures, partial-onset seizures, and for those aged 2 years or older with seizures associated with Lennox-Gastaut syndrome.
Topiramate blocks voltage-gated sodium channels, likely leading to sustained depolarization control during seizures. Topiramate reduces membrane depolarization via AMPA/Kainate receptors and enhances GABA-A receptor activity, strengthening inhibitory effects. Additionally, topiramate acts as a weak carbonic anhydrase inhibitor; acidosis in the brain partially protects against seizures by downregulating NMDA receptor activity.
The most common adverse effects in epilepsy trials included central nervous system involvement (paresthesia, fatigue, cognitive problems, dizziness, somnolence, psychomotor slowing, memory/concentration difficulties, nervousness, confusion), endocrine/metabolism issues (weight loss, anorexia), respiratory problems (infection), and miscellaneous effects (fever, flushing). In trials involving patients with migraine, the most common adverse effects are paresthesia and dysgeusia.
Severe adverse drug reactions include:
Acute myopia and secondary angle-closure glaucoma
Oligohidrosis and hyperthermia (uncommon, reversible with cessation of the drug). [80]
Lamotrigine: is an anti-seizure/anti-epilepsy drug first approved for use in the USA in 1994. It is considered a first-line treatment for primary generalized tonic-clonic seizures (including simple and complex partial seizures and focal-onset tonic-clonic seizures) and Lennox-Gestault syndrome
The mechanism of action for lamotrigine is not entirely understood. It is a triazine, and research has shown that lamotrigine selectively binds and inhibits voltage-gated sodium channels, stabilizing p resynaptic neuronal membranes and inhibiting presynaptic glutamate and aspartate releas
Lamotrigine follows first-order kinetics with a half-life of 29 hours
Lamotrigine can cause serious rashes requiring hospitalization and discontinuation of this medication. Rash severity varies but includes a risk for Stevens-Johnson syndromeLamotrigine is present in breast milk and is detectable in the blood of breastfed infants. Symptoms of lamotrigine in infants include poor feeding, drowsiness, rash, and apnea. These symptoms can improve with the discontinuation of lamotrigine. If the benefits outweigh the risks in treating epilepsy during lactation/breastfeeding, clinicians should consider monitoring infant lamotrigine levels. [81]
Lacosamide: is an antiepileptic drug used to treat seizures. As a chiral functionalized amino acid, it works by blocking slowly inactivating components of voltage-gated sodium currents. Lacosamide exhibits a stereoselective mode of interaction with sodium channels
In the US and Europe, lacosamide is indicated for the treatment of partial-onset seizures in children and adults. In Canada, it is reserved for use in adults.
It is also used as an adjunctive therapy in the treatment of primary generalized tonic-clonic seizures in patients four years of age and older.
The extended-release capsules of lacosamide are indicated for the treatment of partial-onset seizures in adults and in pediatric patients weighing at least 50 kg.
lacosamide selectively enhances the slow inactivation of voltage-gated sodium channels, shifting the slow inactivation curve to more hyperpolarized potentials and augmenting the maximal fraction of channels in the slow inactivated state. This results in the stabilization of hyperexcitable neuronal membranes and inhibition of repetitive neuronal firing. Lacosamide does not affect the fast component of voltage-gated sodium currents, unlike traditional sodium channel blockers.The most common side effects are Double vision,Dizziness, Headache,Nausea ,Sleepiness and the most serious are Suicidal Thoughts and Actions,Heart Rhythm Changes and severe allergic reactions . [82]
Perampanil: was FDA approved in October 2012 as an adjunctive agent for the treatment of POS with or without secondary generalization in patients with epilepsy at least 12 years of age. In June 2015, the agency approved a second indication for primary generalized tonic-clonic (PGTC) seizures in patients with epilepsy who are at least 12 years of age.
is a novel non-competitive selective antagonist at the postsynaptic ionotropic alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) glutamate receptor.
the most common adverse effects seen in patients taking perampanel were dizziness, fatigue, headache, somnolence, irritability, vertigo, vomiting, weight gain, contusion, nausea,abdom inal pain, and anxiety
This medication is classified as pregnancy category C, indicating it may cause fetal harm. [83]
Rufinamide: is a tri-azole derivative drug structurally unrelated to any other anti-seizure drug (ASD).[1] It was developed first in 2004 and later was granted an orphan drug status in October 2004, and was first marketed in Europe in January 2007. It was approved by the US FDA on November 14, 2008, for the adjunctive treatment of seizures associated with Lennox Gastaut syndrome (LGS).
it stabilizes the inactive state of the sodium channel, limiting the sustained bursts of high-frequency action potentials and preventing sodium channels from returning to an activated state, therefore decreasing the neuronal hyperexcitability and action potential propagation. In this way, it modulates the activity of the sodium channel, prolonging its inactivity.
The major contraindication to the use of rufinamide is familial short QT syndrome. As discussed above, rufinamide was associated with significant ventricular arrhythmia, including cardiac death in patients with a history of short QT syndrome. [84]
Vigabatrin :was first formulated in 1974 for the treatment of seizures. Five years later, clinical trials on the drug started in Europe, followed by the US in 1980. This testing led to the approval of vigabatrin in the UK's market in 1989, which was then prescribed widely for infantile spasms and refractory complex partial seizures. However, despite the increased incidence of peripheral vision loss in patients on vigabatrin, officials raised concerns about its safety in 1997 despite its efficacy. Finally, in 2009, after a series of studies, the FDA approved vigabatrin for the treatment of infantile spasms as a single drug and refractory complex partial seizures as an additional drug to other anti-epileptic drugs. Given the potential risks of visual loss, the approval comes with a supplemental "Risk Evaluation and Mitigation Strategy”.
Vigabatrin: is an irreversible inhibitor of gamma-amino-butyric acid transaminase (GABA-T), an enzyme that degrades GABA. It is structurally the same as GABA with an extra vinyl group. Given this fact, it acts as a substrate for GABA-T, freeing GABA in the synaptic cleft. The concentration of GABA, a neuro-inhibitory transmitter, increases in the brain, terminating seizure activity. Besides inhibiting GABA-T, vigabatrin prevents neuronal uptake of GABA and stimulates its release into the synapse. Some studies show that vigabatrin enhances the action of the inhibitory neurotransmitter glutamine, which researchers believe adds to its anticonvulsant effect.
Significant side effects include visual disturbances ,Peripheral visual field defect (VFD) concentrically occurs in both eyes as early as 9 months and 11 months in adults and children, respectively, after treatment onset. On average, visual field defects are mostly detected 5 to 6 years after treatment with vigabatrin. Patients tend to turn their heads and move their eyes in a particular direction to compensate for the visual loss. In contrast to peripheral vision, central vision remains mostly unaffected. Because of potential toxicity, the FDA made it compulsory to conduct a baseline ophthalmologic examination before starting vigabatrin treatment in any patient. For patients older than 9 years of age, perimetry testing serves to detect any VFD. On the other hand, electroretinography should be done twice for younger patients to confirm VFD diagnosis,and MRI changes including hyperintensities in the basal ganglia, thalami, and brainstem on diffusion-weighted and T2/FLAIR sequences. Such findings are insignificant and disappear on vigabatrin cessation. [85]
- References
53 . Fa, Z., et al., Magnesium Sulfate for non-Eclamptic Status Epilepticus. Seizure, 2015. 32.
68. Khankhanian, P., et al., Combined VNS-RNS Neuromodulation for Epilepsy. 2022. 39(2): p. e5-e9.
73. S. Chateauvieux, F. Morceau, and M. Diederich, “Valproic acid,” in Encyclopedia of Toxicology, Fourth Edition: Volume 1-9, vol. 9, Treasure Island (FL), 2023, pp. V9-705-V9-713. doi: 10.1016/B978-0-12-824315-2.00578-9.
74. M. Gupta and J. Tripp, “Phenytoin.,” Treasure Island (FL), 2024.
75. J. S. Maan, T. vi H. Duong, and A. Saadabadi, “Carbamazepine.,” Treasure Island (FL), 2024.
76. C. V Preuss, G. Randhawa, T. J. P. Wy, and A. Saadabadi, “Oxcarbazepine.,” Treasure Island (FL), 2024.
77. G. L. Galiana, A. C. Gauthier, and R. H. Mattson, “Eslicarbazepine Acetate: A New Improvement on a Classic Drug Family for the Treatment of Partial-Onset Seizures.,” Drugs R. D., vol. 17, no. 3, pp. 329–339, Sep. 2017, doi: 10.1007/s40268-017-0197-5.
78. A. Kumar, K. Maini, and R. Kadian, “Levetiracetam.,” Treasure Island (FL), 2024.
79. F. Khaleghi and E. C. 2nd Nemec, “Brivaracetam (Briviact): A Novel Adjunctive Therapy for Partial-Onset Seizures.,” P T, vol. 42, no. 2, pp. 92–96, Feb. 2017.
80, K. A. Fariba and A. Saadabadi, “Topiramate.,” Treasure Island (FL), 2024.
81. N. T. Betchel, K. A. Fariba, and A. Saadabadi, “Lamotrigine.,” Treasure Island (FL), 2024.
82. P. Doty, G. D. Rudd, T. Stoehr, and D. Thomas, “Lacosamide.,” Neurother. J. Am. Soc. Exp. Neurother., vol. 4, no. 1, pp. 145–148, Jan. 2007, doi: 10.1016/j.nurt.2006.10.002.
83. J. Greenwood and J. Valdes, “Perampanel (Fycompa): A Review of Clinical Efficacy and Safety in Epilepsy.,” P T, vol. 41, no. 11, pp. 683–698, Nov. 2016.
84. M. J. Humayun and R. Wadhwa, “Rufinamide.,” Treasure Island (FL), 2024.
85. R. Singh and R. P. Carson, “Vigabatrin.,” Treasure Island (FL), 2024.