Gastric Cancer
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Gastric Cancer |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:54 AM |
Description
"last update: 1 June 2025 Download Guideline
- Executive Summary
This guidance provides a data-supported approach to diagnosis, staging, treatment and follow up of patients diagnosed with gastric cancer. This Guideline is intended only for gastric adenocarcinoma
|
Recommendations |
Strength of recommendations |
|
|
Diagnosis, initial staging and risk assessment |
||
|
Diagnosis, initial staging and risk assessment should include physical examination, full and differential blood count, liver and renal function tests, endoscopy and contrast enhanced CT scan with oral and IV contrast of the thorax, abdomen and pelvis. |
Strong |
|
|
Diagnosis should be made from multiple (5-8) endoscopic biopsies to guarantee an adequate representation of the tumour. |
Strong |
|
|
The histological diagnosis should be reported according to WHO criteria. |
Strong |
|
|
HER2 expression by IHC and/or amplification by in situ hybridisation is a validated predictive biomarker for drug therapy and is recommended in case of adenocarcinoma and metastatic disease. |
Strong |
|
|
Accurate assessment of T and N stage by EUS in potentially operable tumours to determine the proximal and distal extent of tumour is preferred |
Conditional |
|
|
Assessment of nutritional status to detect relevant dietary and nutritional deficiencies in both localised and advanced disease settings is recommended. |
Good practice statement. |
|
|
FDG/PET/CT may be used as problem solving tool only |
Conditional |
|
|
Diagnostic laparoscopy and peritoneal washings for cytology are recommended for patients with resectable gastric cancer who are also candidates for perioperative chemotherapy as patients with cytology positive samples are uncertain candidates for curatively-intended surgical resection. |
Strong |
|
|
Management of local and locoregional disease |
||
|
Multidisciplinary treatment planning before any treatment decision is mandatory. |
Good practice statement
|
|
|
Surgery |
||
|
Endoscopic or surgical resection alone is appropriate for selected very early tumours (stage Tis, IA). |
Strong |
|
|
For stage IB-III gastric cancer, peri-operative therapy and radical gastrectomy is recommended. |
Strong |
|
|
Patients should undergo D2 resection in a high-volume surgical centre. |
Strong |
|
|
En bloc resection of involved structures should be done for T4b tumors. |
Strong |
|
|
Routine splenectomy is not indicated unless the spleen is involved or extensive hilar adenopathy is noted. |
Strong |
|
|
Consider placing feeding tube in selected patients undergoing total gastrectomy (especially if postoperative chemoradiation appears a likely recommendation). |
Good practice statement
|
|
|
Peri-operative chemotherapy |
||
|
Peri-operative (pre- and post-operative) chemotherapy is recommended for patients with stage >IB resectable gastric cancer. |
Strong |
|
|
A triplet chemotherapy regimen including a fluoropyrimidine, a platinum compound and docetaxel should be given in case of good perforance status (ECOG PS 0-1). |
Strong |
|
|
Peri-operative use of FLOT is standard of care for patients who are able to tolerate a triple cytotoxic drug regimen (ECOG PS 0-1). |
Strong |
|
|
For patients unfit for triplet Chemotherapy, a combination of a fluoropyrimidine with cisplatin or oxaliplatin is recommended. |
Strong |
|
|
Adjuvant treatment |
||
|
For patients with stage >IB gastric cancer who have undergone surgery without administration of preoperative chemotherapy, adjuvant chemotherapy is recommended. |
Strong |
|
|
For patients undergoing peri- or post-operative chemotherapy, we recommend against the addition of post-operative RT. |
Strong |
|
|
For patients who have not received preoperative chemotherapy and have not undergone an appropriate D2 lymphadenectomy, adjuvant CRT (see annex 3) can be considered. |
Conditional |
|
|
For patients who have undergone surgery with involved margins (R1), adjuvant RT or CRT (see annex 3) might be considered as an individual recommendation, but is not standard. |
Conditional |
|
|
Management of advanced and metastatic disease |
||
|
First-line systemic therapy |
||
|
First-line chemotherapy with a platinum and fluoropyrimidine is recommended. Oxaliplatin is preferred, especially for older patients. |
Strong |
|
|
Irinotecane 5-FU can be considered an alternative option for patients who do not tolerate platinum compounds. |
Strong |
|
|
Trastuzumabe chemotherapy is recommended in patients with adenocarcinoma HER2-positive tumours. |
Strong |
|
|
Second- and later-line treatment |
||
|
Treatment with trastuzumab is not recommended after first-line therapy in HER2-positive advanced gastric cancer. |
Strong |
|
|
Alternative treatments include a taxane, irinotecan, or capecitabine. |
Strong |
|
|
Surgery for metastatic gastric cancer |
||
|
Gastrectomy is not recommended in metastatic gastric cancer unless required for palliation of symptoms. |
Conditional |
|
|
Resection of metastases cannot be recommended in general, but might be considered as an individual approach in highly selected cases with oligometastatic disease and response to chemotherapy. |
Conditional |
|
|
Supportive care and nutrition |
||
|
Care for patients with gastric cancer should include an early palliative care referral and nutritional support. |
Strong |
|
|
Surveillance |
||
|
Regular follow-up is recommended for investigation and treatment of symptoms, psychological support and early detection of recurrence |
Strong
|
|
|
Follow-up should be tailored to the individual patient and stage of disease |
Strong
|
|
|
Dietary support is recommended with attention to vitamin and mineral deficiencies
|
Strong |
|
|
In the advanced disease setting, regular follow-up is recommended to detect symptoms of disease progression before significant clinical deterioration
|
Strong |
|
|
Radiological investigations, specifically CT with oral and IV contrast of the thorax and abdomen, and pelvis should be carried out every 6-12 weeks in patients who are candidates for further cancer specific therapies |
Strong |
|
- Recommendations
Diagnosis, initial staging and risk assessment
· Diagnosis, initial staging and risk assessment should include physical examination, full and differential blood count, liver and renal function tests, endoscopy and contrast enhanced CT scan with oral and IV contrast of the thorax, abdomen and pelvis
Strong recommendation, very low grade evidence. (4)
· Diagnosis should be made from multiple (5-8) endoscopic biopsies to guarantee an adequate representation of the tumour
Strong recommendation, very low grade evidence. (5,6)
· The histological diagnosis should be reported according to WHO criteria
Strong recommendation, very low grade evidence. (7)
· HER2 expression by IHC and/or amplification by in situ hybridisation
is a validated predictive biomarker for drug therapy and is recommended in case of adenocarcinoma and metastatic disease.
Strong recommendation, high grade evidence. (8-10)
· Accurate assessment of T and N stage by EUS in potentially operable tumours to determine the proximal and distal extent of tumour is recommended.
Conditional recommendation, high grade evidence. (11)
· Assessment of nutritional status to detect relevant dietary and nutritional deficiencies in both localised and advanced disease settings is recommended.
Good practice statement.
· FDG/PET/CT is not routinely recommended
Conditional recommendation, low grade evidence. (12)
· Diagnostic laparoscopy and peritoneal washings for cytology are recommended for patients with resectable gastric cancer who are also candidates for perioperative chemotherapy as patients with cytology positive samples are uncertain candidates for curatively-intended surgical resection.
Strong recommendation, low grade evidence. (13)
· The TNM stage should be recorded according to the 8th edition of the AJCC/UICC staging manual
Strong recommendation, very low grade evidence. (14,15)
Management of local and locoregional disease
· Multidisciplinary treatment planning before any treatment decision is mandatory.
Good clinical practice
➡️Resection
· Endoscopic or surgical resection alone is appropriate for selected very early tumours (stage Tis, IA)
Strong recommendation, low grade evidence. (16)
· For stage IB-III gastric cancer, peri-operative therapy and radical gastrectomy is recommended.
Strong recommendation, high grade evidence. (17,18)
· Patients should undergo D2 resection in a high-volume surgical centre
Conditional recommendation, high grade evidence (Figures 1, 2).
• En bloc resection of involved structures should be done for T4b tumors.
Strong recommendation, high grade evidence (19).
• Routine splenectomy is not indicated unless the spleen is involved or extensive hilar
adenopathy is noted.
Strong recommendation, high grade evidence (20).
• Consider placing feeding tube in select patients undergoing total gastrectomy (especially if
postoperative chemoradiation appears a likely recommendation).
Good practice statement
Peri-operative chemotherapy
· Peri-operative (pre- and post-operative) chemotherapy is recommended for patients with stage >IB resectable gastric cancer
Strong recommendation, high grade evidence (21-23)
· A triplet chemotherapy regimen including a fluoropyrimidine, a platinum compound and docetaxel should be given when possible (ECOG PS 0-1).
Strong recommendation, high grade evidence (21-23)
· Peri-operative use of FLOT is standard of care for patients who are able to tolerate a triple cytotoxic drug regimen (ECOG PS 0-1).
Strong recommendation, high grade evidence (21-23)
· For patients unfit for triplet Chemotherapy, a combination of a fluoropyrimidine with cisplatin or oxaliplatin is recommended
Strong recommendation, moderate grade evidence (21-23)
Adjuvant treatment
· For patients with stage >IB gastric cancer who have undergone surgery without administration of preoperative chemotherapy, adjuvant chemotherapy is recommended
Strong recommendation, high grade evidence (24)
· For patients undergoing peri- or post-operative chemotherapy, the addition of post-operative RT has no added benefit and should not be given
Conditional recommendation, high grade evidence (25)
· For patients who have not received preoperative chemotherapy and have not undergone an appropriate D2 lymphadenectomy, adjuvant CRT (see annex 3) can be considered
Conditional recommendation, high grade evidence (26)
· For patients who have undergone surgery with involved margins (R1), adjuvant RT or CRT (see annex 3) might be considered as an individual recommendation, but is not standard
Conditional recommendation, very low grade evidence (27-29)
Management of advanced and metastatic disease
First-line systemic therapy
· First-line chemotherapy with a platinum and fluoropyrimidine is recommended. Oxaliplatin is preferred, especially for older patients
Strong recommendation, high grade evidence (30-36)
· Irinotecane 5-FU can be considered an alternative option for patients who do not tolerate platinum compounds
Strong recommendation, moderate grade evidence (37,38)
· Trastuzumabe chemotherapy is recommended in patients with adenocarcinoma HER2-positive tumours
Strong recommendation, high grade evidence (39)
Second- and later-line treatment
· Treatment with trastuzumab is not recommended after first-line therapy in HER2-positive advanced gastric cancer
Conditional recommendation, high grade evidence (40,41)
· Alternative treatments include a taxane, irinotecan, or capecitabine.
Strong recommendation, high grade evidence (42)
Surgery for metastatic gastric cancer
· Gastrectomy is not recommended in metastatic gastric cancer unless required for palliation of symptoms
Conditional recommendation, high grade evidence (43)
· Resection of metastases cannot be recommended in general, but might be considered as an individual approach in highly selected cases with oligometastatic disease and response to chemotherapy
Conditional recommendation, very low grade evidence (44,45)
Supportive care and nutrition
· Care for patients with gastric cancer should include an early palliative care referral and nutritional support
Strong recommendation, high grade evidence (46-48)
Surveillance
· Regular follow-up is recommended for investigation and treatment of symptoms, psychological support and early detection of recurrence
Strong recommendation, low grade evidence. (49-51)
· Follow-up should be tailored to the individual patient and stage of disease
Strong recommendation, very low grade evidence. (49-51)
· Dietary support is recommended with attention to vitamin and mineral deficiencies
Strong recommendation, very low grade evidence. (49-51)
· In the advanced disease setting, regular follow-up is recommended to detect symptoms of disease progression before significant clinical deterioration
Strong recommendation, very low grade evidence. (49-51)
· Radiological investigations, specifically CT with oral and IV contrast of the thorax and abdomen, and pelvis should be carried out every 6-12 weeks in patients who are candidates for further cancer specific therapies Strong recommendation, very low grade evidence. (49-51)
Clinical indicators for monitoring
- Physical examination at initial diagnosis.
- Full an differential blood count, liver and renal function tests at initial diagnosis.
- Endoscopy and contrast enhanced CT scan with oral and IV contrast of the thorax, abdomen and pelvis at initial diagnosis.
- Multidisciplinary treatment planning before any treatment decision with documentation of the plan in the medical file.
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines, for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines: Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Yousri Wasef & Yousri Rostom.
- Chair of the GIT Cancer Scientific Committee: Prof Yousri Rostom
- The gastric cancer Scientific Group Members: (Alphabitical order): Ahmed EL Kassed, Fouad Aboutaleb, Khaled Abdelkarim, Manal EL Mahdy, Omar S. Omar, and Yousri Wasef.
- Abbreviations
|
CBC |
Complete blood count |
|
CRT |
Combined chemo radiotherapy |
|
Computed Tomography |
|
|
D2 resection |
A standard D2 resection for gastric cancer involves removing not just part or the whole stomach, but also the N1 (groups 1–6) and N2 (groups 7–11) lymph nodes, the greater and lesser omenta and if necessary, the spleen and tail of the pancreas for tumours of the proximal stomach in order to remove groups 10 and 11 lymph nodes (Figs 1 and 2). |
|
ECOG |
Eastern Coperative Oncolgy Group |
|
EHC |
Egyptian Health Council |
|
EUS |
Endoscopic Ultrasound |
|
FLOT |
Fluorouracil, leucovorin, oxaliplatin and docetaxel |
|
GC |
Gastric cancer |
|
IHC |
Immuno-Histochemistry |
|
KFTs |
Kidney function tests |
|
LAGC |
Locally advanced gastric cancer |
|
LFTs |
Liver function tests |
|
MRI |
Magnetic resonance imaging |
|
MDT |
Multidisciplinary team |
|
N |
Node |
|
T |
Tumor |
|
PET/CT |
Positron emission tomography/Computed Tomography |
|
PS |
Performance Status |
|
RT |
Radiotherapy |
|
ULN |
Upper limit of normal |
- Introduction
Less than 1 million (968 784) new cases of gastric cancer were estimated globally in 2022, resulting in 660 175 deaths. These burden estimates will continue to increase due to the
ageing population and increase in risk factors. In Egypt, there was an estimated 3285 new cases of gastric cancer and 2489 deaths occurred because of this disease based on GLOBOCAN 2022.
- Purpose and scope
These guidelines are developed to improve the quality of care for gastric cancer via providing a uniform standard of care across the country to help in early diagnosis, treatment and follow up for gastric cancer so more optimal treatment options and improved clinical outcomes.
- Target audience
Clinicians who are involved in the care and treatment of patients with gastric cancer, include medical oncologists, radiation oncologists, clinical oncologist, gastroenterologists, surgeons, clinical dietrition interventional radiologists, radiologists, pathologists, and palliative care specialists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The ESMO, NCCN, and NICE guidelines are the main sources used while formulating the national guidelines for gastric cancer (1-3).
- Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group:https://www.gradeworkinggroup.org/
. GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
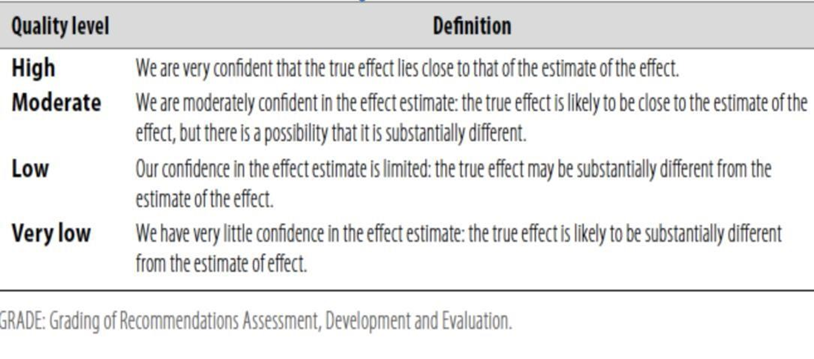
Table 2: Significance of the four levels of evidence

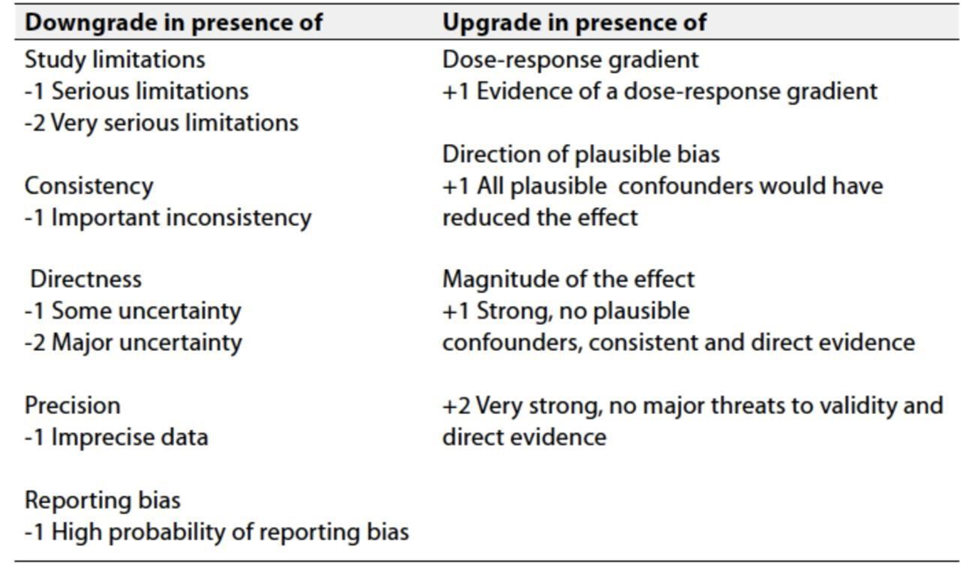
Table 3: Factors that determine How to upgrade or downgrade the quality of evidence
➡️The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research gaps
- Systematic inclusion of cost-benefit analyses in clinical trials with collection of health economic analysis such as incremental cost effectiveness ratio in order to facilitate clinical decision-making.
- Predictive biomarkers: response to specific systemic targeted therapies and immunotherapy.
- Improve models for pre-clinical testing of novel drugs.
- Search for tools to assess quality of life and in clinical trials.
- Dietary supplements, nutritional counselling, physical activity recommendations and psychological support as part of an integrative healthcare approach to care for people with gastric cancer.
- Update of this guideline
- This guideline will be updated whenever there is new evidence.
- References
1.https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1453.
2.https://www.esmo.org/guidelines/guidelines-by-topic/esmo-clinical-practice-guidelines.
3.https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/nice-guidelines
4. Allum W, Lordick F, Alsina M, et al. ECCO essential requirements for quality cancer care: oesophageal and gastric cancer. Crit Rev Oncol Hematol. 2018;122:179-193.
5. Gullo I, Grillo F, Molinaro L, et al. Minimum biopsy set for HER2 evaluation in gastric and gastro-esophageal junction cancer. Endosc Int Open. 2015;3(2):E165-E170.
6. Tominaga N, Gotoda T, Hara M, et al. Five biopsy specimens from the proximal part of the tumor reliably determine HER2 protein expression
7. Carneiro F, Fukayama M, Grabsch HI, et al. Gastric adenocarcinoma. In: WHO Classification of Tumours Editorial Board, ed. Digestive System Tumours. 5th ed Lyon, France: International Agency for Research on Cancer; 2019. p. 85-95.
8. Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-esophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376(9742):687-697.
9. Smyth EC, Nilsson M, Grabsch HI, et al. Gastric cancer. Lancet.2020;396(10251):635-648.
10. Haffner I, Schierle K, Raimúndez E, et al. HER2 expression, test deviations, and their impact on survival in metastatic gastric cancer: results from the prospective multicenter VARIANZ study. J Clin Oncol. 2021;39(13):1468-1478.
11. Nie RC, Yuan SQ, Chen XJ, et al. Endoscopic ultrasonography compared with multidetector computed tomography for the preoperative staging of gastric cancer: a meta-analysis. World J Surg Oncol. 2017;15(1):113.
12. Gertsen EC, Brenkman HJF, van Hillegersberg R, et al. 18F-fludeoxyglucose-positron emission tomography/computed tomography and laparoscopy for staging of locally advanced gastric cancer: a multicenter prospective Dutch cohort study (PLASTIC). JAMA Surg. 2021;156(12):e215340.
13. Harmon RL, Sugarbaker PH. Prognostic indicators in peritoneal carcinomatosis from gastrointestinal cancer. Int Semin Surg Oncol. 2005;2(1):3.
14. Stomach cancer. In: Amin MB, Edge S, Greene F, et al., editors. AJCC Cancer Staging Manual. 8th ed. New York, NY: Springer; 2017. p. 203-220.
15, Stomach. In: Brierley JD, Gospodarowicz MK, Wittekind C, editors. UICC TNM Classification of Malignant Tumours. 8th ed. Oxford, UK: Wiley-Blackwell; 2017.
16. Pimentel-Nunes P, Dinis-Ribeiro M, Ponchon T, et al. Endoscopic submucosal dissection: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2015;47(9):829-854.
17. De Manzoni G, Marrelli D, Baiocchi GL, et al. The Italian Research Group for Gastric Cancer (GIRCG) guidelines for gastric cancer staging and treatment: 2015. Gastric Cancer. 2017;20(1):20-30.
18. Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer. 2021;24(1):1-21.
19. Ito H, Clancy TE, Osteen RT, et al. Adenocarcinoma of the gastric cardia: what is
the optimal surgical approach? J Am Coll Surg 2004;199:880-886.
20. Yu W, Choi GS, Chung HY. Randomized clinical trial of splenectomy versus
splenic preservation in patients with proximal gastric cancer.
Br J Surg 2006;93:559-563.
21. Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11-20.
22. Ychou M, Boige V, Pignon JP, et al. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial.J Clin Oncol. 2011;29(13):1715-1721.
23. Al-Batran SE, Homann N, Pauligk C, et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): a randomised, phase 2/3 trial. Lancet.2019;393(10184):1948-1957.
24. Paoletti X, Oba K, Burzykowski T, et al. Benefit of adjuvant chemotherapy for resectable gastric cancer: a meta-analysis. JAMA. 2010;303(17):1729-1737.
25. Cats A, Jansen EPM, van Grieken NCT, et al. Chemotherapy versus chemoradiotherapy after surgery and preoperative chemotherapy for resectable gastric cancer (CRITICS): an international, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19(5):616-628.
26. de Steur WO, van Amelsfoort RM, Hartgrink HH, et al. Adjuvant chemotherapy is superior to chemoradiation after D2 surgery for gastric cancer in the per-protocol analysis of the randomized CRITICS trial. Ann Oncol. 2021;32(3):360-367.
27. Macdonald JS, Smalley SR, Benedetti J, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345(10):725-730.
28. Dikken JL, Jansen EP, Cats A, et al. Impact of the extent of surgery and postoperative chemoradiotherapy on recurrence patterns in gastric cancer. J Clin Oncol. 2010;28(14):2430-2436.
29. Ho VKY, Jansen EPM, Wijnhoven BPL, et al. Adjuvant chemoradiotherapy for non-pretreated gastric cancer. Ann Surg Oncol. 2017;24(12):3647-3657.
30. Wagner AD, Syn NL, Moehler M, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2017;8(8):CD004064.
31. Cunningham D, Starling N, Rao S, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med. 2008;358(1):36-46.
32. Al-Batran SE, Hartmann JT, Probst S, et al. Phase III trial in metastatic gastroesophageal adenocarcinoma with fluorouracil, leucovorin plus either oxaliplatin or cisplatin: a study of the Arbeitsgemeinschaft Internistische Onkologie. J Clin Oncol. 2008;26(9):1435-1442.
33. Van Cutsem E, Moiseyenko VM, Tjulandin S, et al. Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol. 2006;24(31):4991-4997.
34. Al-Batran SE, Hartmann JT, Hofheinz R, et al. Biweekly fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) for patients with metastatic adenocarcinoma of the stomach or esophagogastric junction: a phase II trial of the Arbeitsgemeinschaft Internistische Onkologie. Ann Oncol. 2008;19(11):1882-1887.
35. Shah MA, Janjigian YY, Stoller R, et al. Randomized multicenter phase II study of modified docetaxel, cisplatin, and fluorouracil (DCF) versus DCF plus growth factor support in patients with metastatic gastric adenocarcinoma: a study of the US gastric cancer consortium. J Clin Oncol. 2015;33(33):3874-3879.
36. Van Cutsem E, Boni C, Tabernero J, et al. Docetaxel plus oxaliplatin with or without fluorouracil or capecitabine in metastatic or locally recurrent gastric cancer: a randomized phase II study. Ann Oncol. 2015;26(1):149-156.
37. Dank M, Zaluski J, Barone C, et al. Randomized phase III study comparing irinotecan combined with 5-fluorouracil and folinic acid to cisplatin combined with 5-fluorouracil in chemotherapy naive patients with advanced adenocarcinoma of the stomach or esophagogastric junction. Ann Oncol. 2008;19(8):1450-1457.
38. Guimbaud R, Louvet C, Ries P, et al. Prospective, randomized, multicenter, phase III study of fluorouracil, leucovorin, and irinotecan versus epirubicin, cisplatin, and capecitabine in advanced gastric adenocarcinoma: a Frenc intergroup (Fédération Francophone de Cancérologie Digestive, Fédération Nationale des Centres de Lutte Contre le Cancer, and Groupe Coopérateur Multidisciplinaire en Oncologie) study. J Clin Oncol. 2014;32(31):3520-3526.
39. Bang YJ, Van Cutsem E, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376(9742):687-697.
40. Shitara K, Bang YJ, Iwasa S, et al. Trastuzumab deruxtecan in previously treated HER2-positive gastric cancer. N Engl J Med. 2020;382 (25):2419-2430.
41. van Cutsem E, Di Bartolomeo M, Smyth E, et al. Primary analysis of a phase II single-arm trial of trastuzumab deruxtecan (T-DXd) in western patients (Pts) with HER2-positive (HER2þ) unresectable or metastatic gastric or gastroesophageal junction (GEJ) cancer who progressed on or after a trastuzumab-containing regimen. Ann Oncol. 2021;32(suppl 5):S1283-S1346.
42. Wagner AD, Syn NL, Moehler M, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2017;8(8):CD004064.
43. Fujitani K, Yang HK, Mizusawa J, et al. Gastrectomy plus chemotherapy versus chemotherapy alone for advanced gastric cancer with a single non-curable factor (REGATTA): a phase 3, randomized controlled trial. Lancet Oncol. 2016;17(3):309-318.
44. Al-Batran SE, Homann N, Pauligk C, et al. Effect of neoadjuvant chemotherapy followed by surgical resection on survival in patients with limited metastatic gastric or gastroesophageal junction cancer: the AIO-FLOT3 trial. JAMA Oncol. 2017;3(9):1237-1244.
45. Kataoka K, Kinoshita T, Moehler M, et al. Current management of liver metastases from gastric cancer: what is common practice? New challenge of EORTC and JCOG. Gastric Cancer. 2017;20(5): 904-912.
46. Lu Z, Fang Y, Liu C, et al. Early interdisciplinary supportive care in patients with previously untreated metastatic esophagogastric cancer: a phase III randomized controlled trial. J Clin Oncol. 2021;39(7): 748-756.
47. Mansoor W, Roeland EJ, Chaudhry A, et al. Early weight loss as a prognostic factor in patients with advanced gastric cancer: analyses from REGARD, RAINBOW, and RAINFALL phase III studies. Oncologist. 2021;26(9):e1538-e1547.
46. Dai Y, Li C, Xie Y, et al. Interventions for dysphagia in oesophageal cancer. Cochrane Database Syst Rev. 2014;2014(10):CD005048.
48. Bergquist H, Wenger U, Johnsson E, et al. Stent insertion or endoluminal brachytherapy as palliation of patients with advanced cancer of the esophagus and gastroesophageal junction. Results of a randomized, controlled clinical trial. Dis Esophagus. 2005;18(3):131-139.
49. Baiocchi GL, D’Ugo D, Coit D, et al. Follow-up after gastrectomy for cancer: the Charter Scaligero Consensus Conference. Gastric Cancer. 2016;19(1):15-20.
50. Hu Y, Kim HI, Hyung WJ, et al. Vitamin B(12) deficiency after gastrectomy for gastric cancer: an analysis of clinical patterns and risk factors. Ann Surg. 2013;258(6):970-975.
51. Baek KH, Jeon HM, Lee SS, et al. Short-term changes in bone and mineral metabolism following gastrectomy in gastric cancer patients. Bone. 2008;42(1):61-67.
52. Smalley SR, Benedetti JK, Haller DG, et al. Updated analysis of SWOG-directed Intergroup study 0116: a phase III trial of adjuvant radiochemotherapy versus observation after curative gastric cancer resection. J Clin Oncol 2012;30:2327-2333.
53. ee HS, Choi Y, Hur WJ, et al. Pilot study of postoperative adjuvant chemoradiation for advanced gastric cancer: adjuvant 5-FU/cisplatin and chemoradiation with capecitabine. World J Gastroenterol 2006;12:603-607.
54. Minsky BD, Pajak TF, Ginsberg RJ, et al. INT 0123 (Radiation Therapy Oncology Group 94-05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol 2002;20:1167-1174.
55. Minsky BD, Pajak TF, Ginsberg RJ, et al. INT 0123 (Radiation Therapy Oncology Group 94-05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol 2002;20:1167-1174.
- Annexes
➡️(Annex 1), Figure 1 Lymph node groups 1–6 (N1)
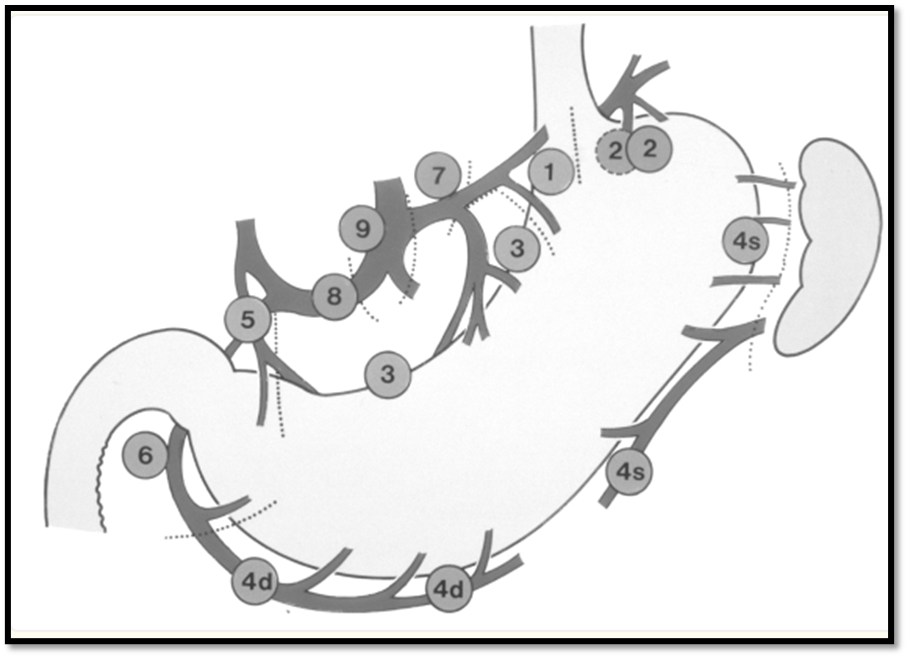
Chul-Hyo Jeon et al. BMC Cancer,23, 1192 (2023)
➡️Figure 2. Lymph node groups 7–11 (N2)
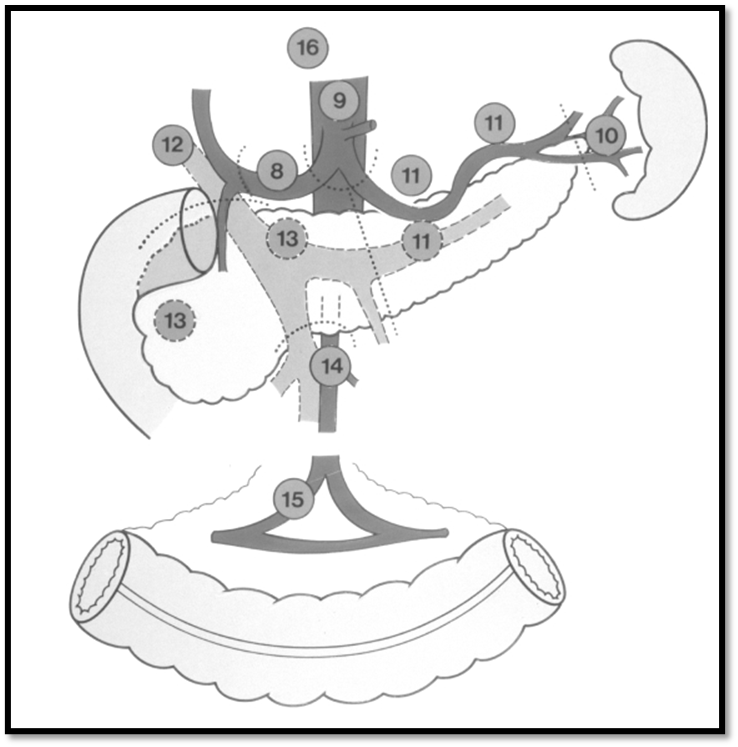
Chul-Hyo Jeon et al. BMC Cancer,23, 1192 (2023)
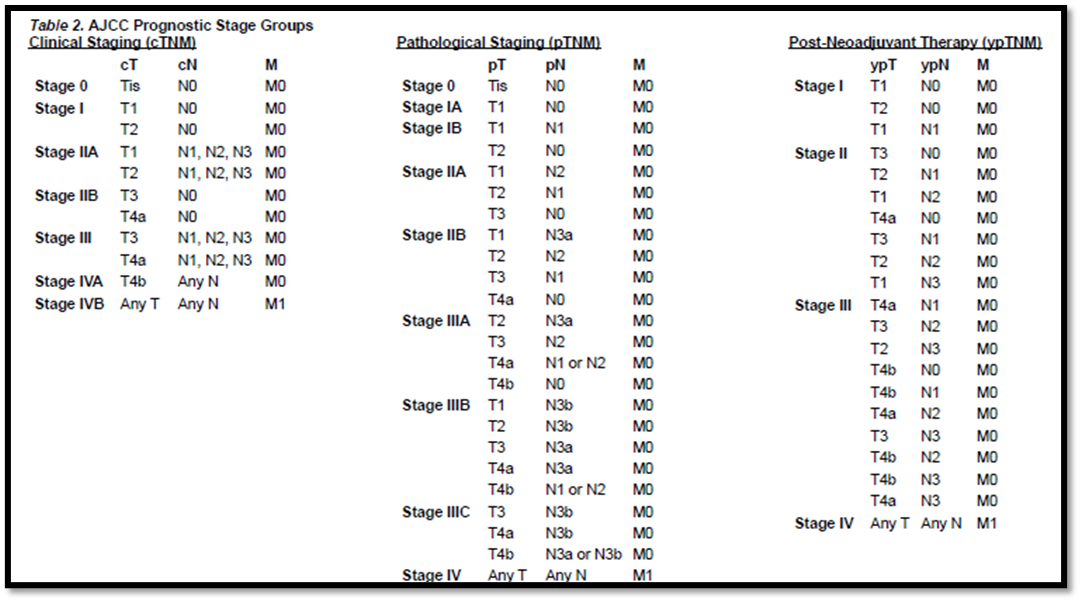
➡️(Annex 2) American Joint Committee on Cancer (AJCC)
TNM Staging Classification for Carcinoma of the Stomach (8th ed., 2017)

➡️(Annex 3) POSTOPERATIVE CRT
▪️ If Fluorouracil is used then: 2 cycles before and 4 cycles after CRT.
▪️ With radiation Fluorouracil 200–250 mg/m2 IV continuous infusion over 24 hours daily on Days 1–5 Weekly for 5 weeks
▪️ If Capecitabine is used then: 1 cycle before and 2 cycles after CRT. With radiation Capecitabine 625–825 mg/m2 PO BID on Days 1–5 Weekly for 5 weeks
▪️ CT simulation and conformal treatment planning should be used with either three-dimensional conformal radiation therapy (3D-CRT) or intensity-modulated radiation therapy (IMRT)
▪️ RT Dose: 45–50.4 Gy (1.8 Gy/day) (total 25–28 fractions)
▪️ Higher doses may be used for positive surgical margins in selected cases as a boost to that area.