Rabbit diseases diagnosis, prevention and control
| Site: | EHC | Egyptian Health Council |
| Course: | Avian and Rabbit Medicine Guidelines |
| Book: | Rabbit diseases diagnosis, prevention and control |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
1 July 2025" Download Guideline
Table of contents
- - Acknowledgement
- - Scope
- - The target audience
- - Aim
- - Abbreviations
- - Guidelines to protect rabbits from diseases
- - Diseases problem and treatment
- - Bacterial Diseases
- - VIRAL Diseases of Rabbit
- - Digestive system problems
- - PROTOZOAL DISEASES
- - Treatment digestive system problems
- - Prevention and control of digestive diseases
- - Listeriosis
- - Skin problems
- - Fungal Dermatitis or Superficial Mycosis
- - Reference
- Acknowledgement
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The rapporteur of the Committee: Prof. Mohamed Mohamedy Ghanem.
Scientific Group Members: Prof. Nabil Yassin, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby, Dr. Mohamed Elsharkawy, Prof. Gamal A. Sosa., Dr. Naglaa Radwan, Dr. Hend El Sheikh
Authors: Prof. Dalia Mansour Hamed, Dr. Essam Elmarakby
- Scope
The guidelines concerned diagnosis, treatment, and prevention of Rabbit diseases. The guidelines also provided landmarks for the evaluation of the severity and the most suitable antibiotics for therapeutic intervention.
- The target audience
The guideline is intended for all veterinarians who are intended to diagnose, treat, and control Rabbit Diseases.
- Aim
Rabbit production is essential to their economies and food security. When proper husbandry and management practices are used, rabbits are relatively sensitive to diseases. A summary of some of the more prevalent diseases affecting rabbit is provided below.
- Abbreviations
Doe: female rabbit
Buck: male rabbit
kits :Rabbits babies
- Guidelines to protect rabbits from diseases
Diseases have a natural occurrence and cannot be entirely eradicated; however, employing effective management practices can help maintain their prevalence at manageable levels.
1. Disease prevention is markedly more effective than treatment.
2. Traits such as high disease resistance, longevity, and productivity can be inherited.
3. Selecting breeding stock based on exceptional performance can yield significant returns on investment of time and resources.
4. Avoid overcrowding by ensuring that housing conditions allow for adequate space and promote proper nutrition.
5. Ensure that the facility offers ample, draft-free ventilation.
6. Structures with solid sides and wire floors can create undesirable updrafts, which should be avoided.
7. Allow your rabbits to access natural sunlight, provided it is not excessively hot. Additionally, maintain cleanliness and dryness in all equipment to reduce the risk of disease outbreaks, and ensure that all tools are in good working condition.
8. Limit unnecessary handling of rabbits, their feed, food and water containers, or any equipment they come into contact with, as clothing and hands of caregivers can transmit pathogens.
9. Isolate any new animals introduced to the herd, regardless of their origin, as well as those that have potentially been exposed to other rabbits, either directly or indirectly through shared equipment and handlers.
10. Isolate rabbits that show symptoms of infectious diseases and attend to healthy animals before addressing those that are ill.
11. Protect your rabbits from stressors, especially nocturnal disturbances.
12. Allow for adequate rest periods during the day, balancing routine care with their need for downtime.
13. If you regularly sell rabbits to a dealer, ensure that your marketable stock is segregated and kept outside the main entry to the rabbitries, facilitating quick and efficient collection, help in preventing disease transmission.
- Diseases problem and treatment
Disease/ condition |
Symptom |
Causative agent |
Treatment/ control |
High mortality |
Hemorrhagic septicemia Respiratory symptom, Bloody nasal discharges |
Pasteurella, Rabbit calicivirus disease |
No effective treatment ,prevention by vaccination with killed vaccine |
Respiratory Symptom,
|
Conjunctivitis, Pneumonia/lung abscess, otitis media, |
Pasteurella multocida, Bordetella bronchiseptica, Staph. aureus, Myxoma virus |
|
Mastitis, Metritis,
|
Inflamed,
swollen, hot,
|
Pasteurella multocida Staph. aureus |
Mastitis could be treated for 3 days
|
Weepy Eye
|
Matted fur at medial corner
|
Inflammation of
|
Neomycin
|
Vent Disease
|
Scabby" inflammation of
|
Usually spread Infection with rabbit s syphilis, (Rabbit spirochetosis) Treponema cuniculi, |
Daily application of penicillin |
Mucoid
|
Jelly-like secretion
|
cecum impact,
|
Reduce stress, place on high |
diarrhea
|
Diarrhea, poor flesh condition
|
Coccidia.E.coli, Salmonella, Enterotoxemia, Clostridium difficile and C. spiroforme |
E.coli or Salmonella or
coccidian: - Enterotoxemia: Tetracycline, Nitrofuran, Metronidazole |
Fur
Mites
|
Loss of fur around face, neck,
|
Mite.
|
chlorhexidine or a povidene-iodine repeat 10 days after |
Ear Mites |
Shaking head, scratching at |
Infestation of |
Saturate inside of ear with oil Ivomec i.m |
Ring worm
|
Loss of hair in circular fashion
with sore in the middle. Most common on feet and legs of young, but can be seen
in adults and on any location of |
Fungus. Contagious to |
Individual animals should be
|
Sore
|
Ulcerated area on the bottom
|
Small foot pad area for |
Use dry solid surface for |
Nervous disorder |
torticollis, opisthotonous and convulsion |
Listeria monocytogenes ,Pasteurella Salmonella |
Tetracycline. |
Abortion |
|
Listeria monocytogenes ,Pasteurellosis, Staph,Salmonella |
Sulpha + trimethoprim at 40mg/kg b.w. Neomycin200-800mg \Liter |
- Bacterial Diseases
1-Pasteurellosis
Etiology
- Pasteurellosis in rabbits is caused by the organism Pasteurella multocida, a bacteria is a gram negative stained short rod, bipolar that includes a large variety of different strains.-Some of these strains are highly virulent and cause serious disease, while others may only induce mild disease or no disease at all.
- Has 5 antigenic group ( A, B, C, D, E) A and D are the most common isolates from rabbit as primary respiratory diseases followed by Staph and Bordetella as secondary cause.
-Different strains also infect different species of animals. The bacteria reside in the upper respiratory airways of clinically normal animals but if the sensitive lining of the respiratory system is damaged by ammonia, dust particles, a virus or other bacteria, Pasteurella invade and initiate an immune response, causing clinical disease.Other strains are involved in enzootic pneumonia and lung abscess.
In chickens, Pasteurella multocida causes fowl cholera.
Pasteurella multocida is the most common bacteria responsible for respiratory infections and also competes with Staphylococcus as the major cause of carcass abscess, reproductive tract infections and mastitis.
- Animal can be symptomless
carrier.
It called overt diseases (stress related).
Culture Media: The organism grows on blood agar. It is aerobic and facultative anaerobic. The colonies are mucoid in appearance.
Predisposing factors :
1- Ammonia is extremely destructive to the cells lining the respiratory system.Therefore, poor ventilation and lack of effective air exchange are the most
important concerns. When combined with hot, humid conditions we have the
potential for high mortalities from snuffles
2-In winter, excessive draught also increases susceptibility to infection. Shed design, ventilation and temperature control have been discussed in a previous section. an even flow of ventilation throughout the shed with maximum air-exchange and minimal draught is the ideal to work towards.
3-Diets that are over 18% in protein when fed to rabbits result in higher production of faecal and urinary ammonia, thus further compounding the problem. As mentioned previously, there is a high carriage of Pasteurella multocida amongst normal rabbits.
4-Although transmission from normal carrier animals to animals completely free of the bacteria is normally very low, animals that develop clinical disease shed bacteria at much higher rates (direct transmission). Thus management factors including no quarantine facilities, poor identification of diseased livestock, cross fostering from diseased does and poor culling policies together with inadequate hygiene to eliminate the bacteria that have been shed will predispose to infection. It is important to note the Pasteurella multocida can remain viable in water nipples and unclean feeders for several days (indirect contact through utensils).
Susceptibility:
- All ages are susceptible.
- Young developed acute form with high mortality.
- Semi-mature and adult acquire sub-acute and chronic with low morality.
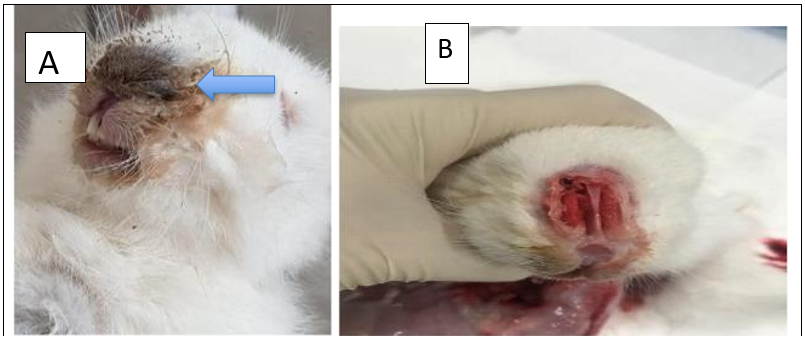
Snuffle
Snuffles is a severe infection of the upper respiratory tract. Young rabbits aswell as lactating does and performing bucks are equally susceptible in death.
The most common clinical signs are:
1- severe nasal discharge, chronic sneezing and wet matted fur on the inside of the fore legs from constantly rubbing the nose.2- sneezing due to snuffles must be differentiated from sneezing due to pellet dust or water droplets entering the nasal cavity. Wet patches on the forelegs and nasal discharge (a mixture of mucus and pus) are the giveaway signs.
3- Infections including conjunctivitis and pneumonia may take place secondary to snuffles.
P.M: Rhinitis, sinusitis, in chronic cases erosion in turbinate bone and atrophy.
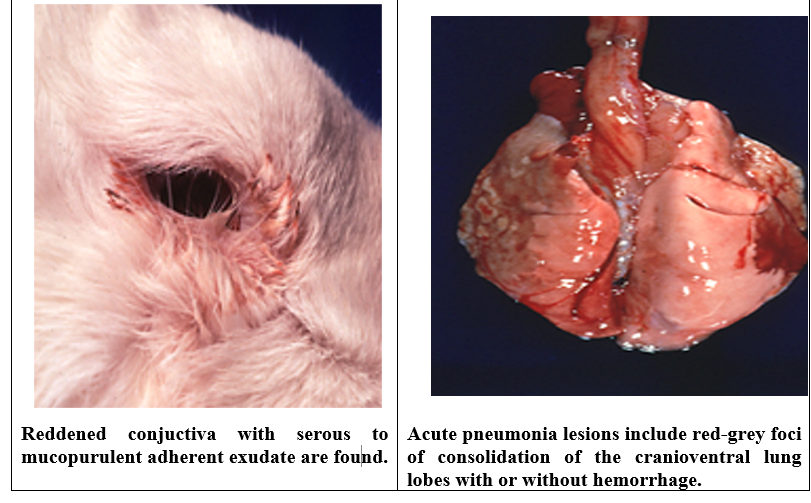
Pneumonia/lung abscess
Tend to be a chronic due to large numbers of organisms in the environment.Infection can either be acute, where sudden death occurs or chronic, usually involving lung
abscesses where rabbits lose weight rapidly and experience deep, difficult respiration (dyspnoea).
D.D: Dyspnoea must be differentiated from laboured breathing due to heat-stress or shock.
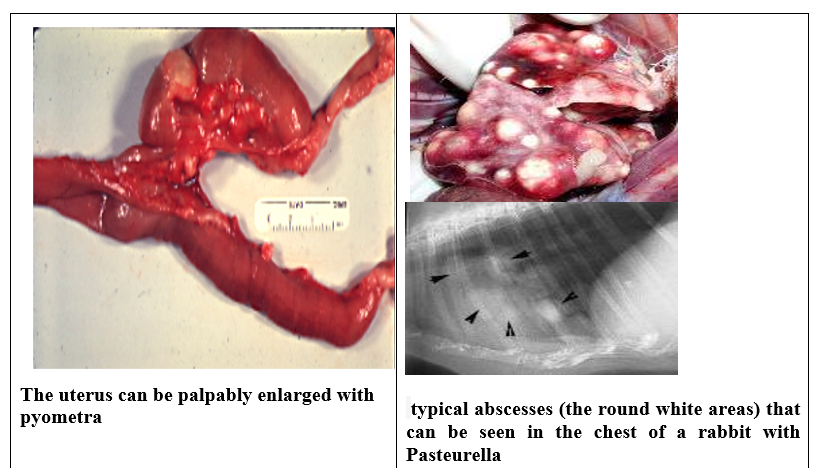
Following infection of the respiratory system, young rabbits usually die within a week. Those that survive fail to thrive and do not reach market weight. Infected animals continually shed bacteria into the environment, resulting in transmission of the disease to other susceptible stock.P.M: Large abscesses can be present in thoracic cavity, hemorrhage on lung fibrin cover , pleural cavity.
Wry-Neck ((Head Tilt) (Otitis Media)
Wry neck is the common name for a
middle ear infection
Pasteurella
can spread from nasal cavity to the ophthalmic bulla via the Eustachian tube.
Presence of inspissated pus in the deeper structure of ear spread along the
nerve leads to neurological signs with rolling and nystagmus.
Rabbits lose their sense of balance and direction and consequently tilt their heads to one side. Wry neck is not particularly serious and whilst some animals may develop brain abscess, the majority go to slaughter at the correct market weight.
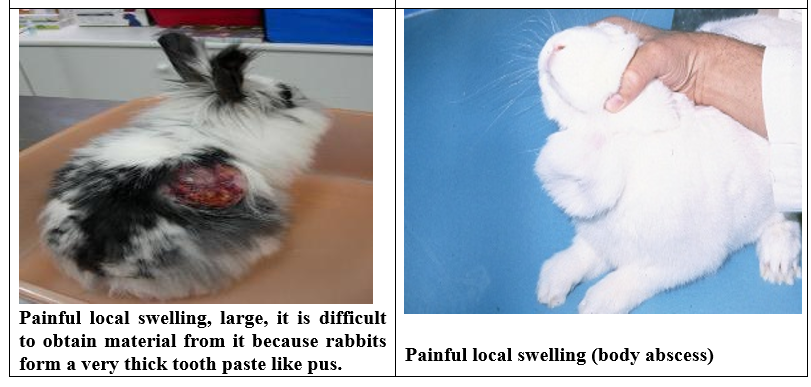
Body abscess
In contract, young growers with severe pasteurella body abscess usually fail to thrive. The abscess continually forms, discharges and regresses, spreading bacteria into the environment. Infection usually results from pre or post weaning injury with the opportunistic bacteria invading wounds caused from fighting. After surgery may cause osteomyelitisRabbits that make slaughter are usually condemned which is worse than the animal dying during the growing period. Abscess along the backbone is a particular problem due to the fact that they are not usually noticed until dressing and result in complete carcass condemnation.
Mastitis/Metritis
Does tend to suffer mastitis or metritis concurrent with snuffles. Lactating does are highly susceptible particularly if nutrition during breeding is inadequate (poor quality feed, too much energy in the ration) they are housed in draughty conditions or suffer heat stress or they are placed under a highly stressful breeding program. Does do not usually die directly, performance drops substantially, kids do not thrive and concurrent snuffles infection results in transmission to neighbouring does who may in turn succumb to infection. Female affected showed vaginal discharge, fail to concept. Bucks showed abscess in tests.Conjunctivitis
Swollen eyes glued shut from exudates congestion of conjunctiva with serous purulent exudates and epiphora (abnormal flow of tears).Haemorrhagic septicemia
In peracute cases no signs animal
die from fever, lung hemorrhage. L.nodes showed sever congestion chest cavity
contain a clear yellowish fluid.
Diagnosis:
1- Clinical signs2- Isolation of causative agent but not relied on (may M.O die during transport, M.O found in deeper tissue, M.O may be normal inhabitant)
3-Serological test
4- PCR
Treatment and prevention
Firstly, it must be stressed that while many antibiotics will successfully destroy theorganism, most of these drugs do not penetrate the nasal cavity of rabbits well and are of little effect in reducing the carrier status of the animals. They will have some advantage in controlling secondary infections such as pneumonia, particularly during hot, humid summers and draughty winters, however their routine use for the control of pasteurellosis is not warranted.
1-Terramycin is probably the most effective either through the feed or water although it can reduce growth rates by depressing appetite and predispose to diarrhea. The dose rate is 250-500 mg/L drinking water.
2- Cephalaxine used parentally.3- Antibiotics effective against Pasteurella include enrofloxacin trimethoprim sulfa, chloramphenicol, penicillin G, and azithromycin.
4-N.B: pencillin used only parentally as by oral rout leads to diarrhea.
5-Gentamycin effective in local application (eye drop, nasal drop, ointment) to avoid nephrotoxicity.
6-Compination between Enrofloxacine and oxytetracycline will be effective.Culling is by far the most effective policy, operating a high culling rate in the breeding phase of production with plenty of replacements over time yields stock with better resistance and prevents transmission of the organism. Kids from does suffering with pasteurellosis should never be cross-fostered and must be raised independently from
unaffected growers.
Vaccination:
-Dead formalized vaccine at age of 2 month 1cm S/C and boaster after15 days then:
Revaccination every 6
months in non-endemic area
every 2 months in endemic
areas.
The
vaccine includes somatic antigens (3,
12) and capsule types A and D, with
improved cross-immunity observed
among the same serotype strains.
Overall,
effective management of pasteurellosis in rabbits focuses on cautiousantibiotic use, strategic culling, and vaccination programs to enhance herd
immunity
Differential
diagnosis: Other respiratory diseases
Bordetella bronchiseptica is another common inhabitant of the respiratory
tract, but is not normally associated with respiratory disease.
Secondary Staph. aureus infections can also be found.
Myxoma virus causes nasal and ocular discharge, and one form does produce
predominantly respiratory signs, although this is rare
Viral Haemorrhagic Disease produces tracheal and lung haemorrhage and sudden death.
Prevention and control:
-Pasteurellosis must be considered when planning initial shed design.-Ventilation and air exchange must be even through the shed with effective .
- Removal of manure or ammonia
without causing ammonia to rise to cage level.
-Temperature
must be controlled within a 10°C range and no excessive draught should be
apparent.
-Management
techniques including the early diagnosis of diseased livestock and effective
culling policies must be adopted.
- Culling policy should aim at severely affected animals initially and then include mild to sub-clinical carriers as production becomes more successful.
2-Stapylococcosis
Etiology: Staphylococcus aureus.
Most disease causing strains in rabbits are hemolytic, coagulase-positive, mostly type C strains.General: Outbreaks occur sporadically in commercial and laboratory facilities and
manifestation of disease ranges from localized abscessation to acute septicemia.
Transmission:
-Direct contact, aerosol. Carrier animals can harbor the organism in the upper respiratory tract.-Possible entry sites include umbilical vessels and skin abrasions.
Pathogenesis: After inoculation, the organism can spread hematogenously or via local extension resulting in pyoderma, purulent mastitis, internal abscessation, septicemia and purulent bronchopneumonia. Pododermatitis or “sore hock” may occur in association with abscesses or mastitis.
1-Suppurative lesions composed of
thick, purulent exudates can be found in the
subcutaneous , mammary gland, genital tract, conjunctiva and the respiratory
tract.
2-Suppurative emboli can be found in the kidney, lung, brain and heart. With
mastitis, the affected glands have swollen, red areas with indurations of the
overlying skin due to chronic abscessation.
3- In pododermatitis, the swelling and exudates is usually concentrated along
the
ventral hock area.
4- Focal supportive necrotizing lesions with colonies of cocci.
Diagnosis:
Demonstrate Gram positive cocci on Gram-stained sections. Confirm by bacterial
culture, but must determine if the isolate is a pathogenic strain (Beta
hemolytic)
Differential diagnosis: Pasteurellosis,
Tyzzer’s Disease and listeriosis.
Treatment:
Demonstrate Gram positive cocci on Gram-stained sections. Confirm by bacterial
culture, but must determine if the isolate is a pathogenic strain (Betahemolytic)
Differential diagnosis: Pasteurellosis,
Tyzzer’s Disease and listeriosis.
Treatment:
sore hock: Topical ointments with Gentamycin antibiotics, can put flat
surface in cage or put rabbit in solid-bottom cage with soft bedding, but
creates sanitation problems.
3-Bordatellosis
Etiology: Bordatella bronchiseptica
General: The role of this organism in respiratory disease is not firmly established. It can be recovered from the upper and lower respiratory tract of healthy rabbits.In cortisone treated rabbits, suppurative bronchopneumonia has been reproduced
experimentally and the organism has been isolated from natural cases of localized
pneumonic lesions.
Transmission: Direct transmission by aerosol. Bordatella bronchiseptica may impair airway clearance mechanisms and facilitate establishment of Pasteurella multocida.
P.M:
Chronic interstitial pneumonia, chronic bronchiolitis and perivascular and peribronchial infiltrations.Diagnosis:
Bacterial culture. Organisms often present in large numbers.
- VIRAL Diseases of Rabbit
I. Viral Hemorrhagic Disease
Synonyms:
Viral Hemorrhagic Fever,
Viral Necrotizing Hepatitis
"rabbit calicivirus disease" (RCD)?
Why was "rabbit haemorrhagic disease" (RHD) renamed "rabbit calicivirus disease" (RCD)?
History:
The disease was first reported in China in 1984, and has since been reported in Europe, parts of Asia, Mexico and the United States.
In a three-year study at CSIRO
Australian Animal Health Laboratory, rabbits infected with rabbit calicivirus
disease did not die from hemorrhage. "Rabbit Hemorrhagic Disease" is
therefore an inaccurate name for the disease. The disease was renamed Rabbit
Calicivirus. Disease (RCD) because this virus belongs to the virus family
Caliciviridae.
Is rabbit calicivirus disease genetically engineered?
Rabbit calicivirus is not genetically engineered or made in a laboratory. It is a naturally occurring virus.Etiology: A calicivirus has been recovered from infected rabbits. Apparently strains of virus with varying degrees of virulence have been recovered from rabbits from different parts of the world.
Definition: It is a very contagious viral disease can be transmitted via direct and indirect contact and also by insect and fleas .It is caused by host specific calicivirus characterized by high mortality
Transmission:
1-RCD is highly contagious and is spread by contact between susceptible rabbits and an infected rabbit.2-Also by contact with excreta contaminated with the virus, The agent can be spread by direct contact.
3- Biting arthropods and fomites, including handling of infected rabbit meat and byproducts ( Mechanical transmission).
How long does rabbit calicivirus persist in the environment? Under experimental
conditions, the virus can survive up to 25 days at 42 degrees C (can live in tissue such as liver), but for only 2 days at 60 degrees C. It has been detected for 35 days at 20 degrees C. The virus can survive in pH range 3-8.
Pathogenesis: Incubation period: 3-4 Days.
Calicivirus predilection set for hepatocytes that replicate in cytoplasm that
leads to necrosis of liver and spleen
and coagulation of blood intravascular. Thrombi reach vital organ as lung,
kidney and heart internal hemorrhage occur leads to death and impairment in
liver function.
Calicivirus predilection set for hepatocytes that replicate in cytoplasm that leads to necrosis of liver and spleen and coagulation of blood intravascular. Thrombi reach vital organ as lung, kidney and heart internal hemorrhage occur leads to death and impairment in liver function.
Clinical Signs:
Per acute: sudden death may showed severe
hemorrhage.
Acute: There are no specific clinical signs. After about 18 hours of infection with rabbit calicivirus, adult rabbits become progressively quieter; within approximately 30-40 hours of being infected they die quietly with minimal apparent distress.
Some rabbits may appear listless and reluctant to move one day after infection and at this stage, some may have an increased temperature (from 39 degrees C up to 42 degrees C). A few hours prior to death, some rabbits have an increased respiratory rate. The incubation period ranges from 1 to 3 days, at which time one of three forms of the disease may be seen. temperature may be elevated from 40 to 41oC, but rapidly declines prior to death.In subacute disease, clinical disease progresses to include bloody nasal discharge,
opisthotonus and vocalization (nervous signs).Death occurs 2 to 3 hours after the onset of clinical signs. In colony settings, morbidity may reach 90% with 100% mortality.
P.M: A swollen, enlarged spleen; a swollen, friable liver with a lobular pattern
-Tracheal hemorrhages, small focal pulmonary hemorrhages that may join to form large, reddened areas. Occasionally, only two of these organs may be affected.-Myocardial (heart muscle) hemorrhages and renal infarcts(kidney scars) occur more rarely.
-Hematuria, diarrhea ,vaginal discharge or foamy exudates from nose
-Pale carcasses with internal hemorrhage which fail to coagulate.
Diagnosis:
-The disease is tentatively diagnosed based on the rapidly fatal infection and grossnecropsy findings.
-VHD-specific PCR is used to confirm the postmortem diagnosis.
-HA and HI -Eliza
- Frozen carcasses may be Saved for disease confirmation.
Treatment: There is no effective treatment.
Control: -Once the disease is recognized, elimination of all rabbits in the colony has been the only effective way of preventing infection.
- Vet.biosecurity, quarantine and
vaccinate newly introduced rabbit.
Vaccination: The vaccine is a "killed-vaccine", As a standard precaution, the vaccine
should not be used in any rabbit showing any kind of sickness.Vaccines are produced from virus grown in rabbits liver or spleen and the virus inactivated by Beta proprio-Lacton or Formalin.
In slight contaminated area:
Breeder Rabbits are immunized at
about eight to ten weeks of age with annual boosters. The virus is killed by
0.5% sodium hypochlorite.
In highly contaminated area:
Breeder Rabbits obtain primary dose
at 6 weeks and followed by boaster dose after 4 -6 weeks then revaccinated
every 6 months.
In fattening
-Vaccinated once at 4 weeks if
their dams not vaccinated.
-Young
rabbits from vaccinated dams are vaccinated at ten weeks of age, when most of
their maternal antibody has disappeared.
-The
virus is no longer notifiable. Maternal antibodies can be passed to young and
confer temporary immunity.
-Vaccination
of young will then provide permanent immunity. The timing of vaccination is
critical.
- Killed virus vaccines provide protection of native rabbits exposed to the disease,
How does VHD kill rabbits and don’t
kill young rabbit under 8 weeks?
VHD
causes the rapid development of blood clots in major organs such as lungs, heart
and kidneys. These clots block blood vessels and result in death from heart and
respiratory failure in about 30-40 hours. Why do young rabbits under 5 to 8
weeks survive? This is not known, but it is speculated that young kittens have
a relatively immature immune system, and cannot produce enough of the chemicals
that initiate clotting which causes death in older rabbits.

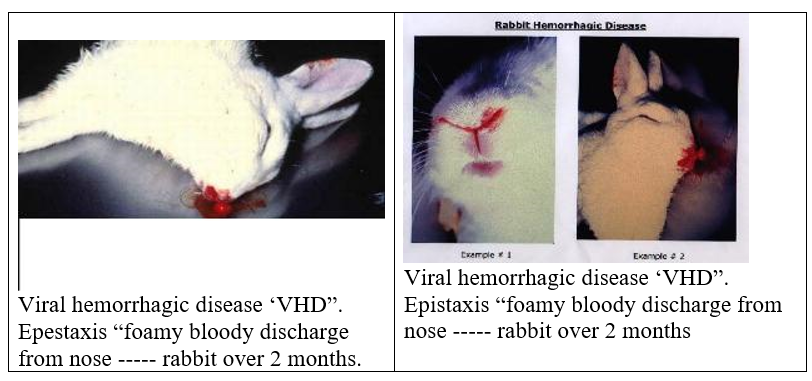
- Digestive system problems
1-Salmonellosis
Etiology
Various
serotypes of Salmonella enteritidis, usually typhimurium or enteritidis.
Gram
negative, non-spore forming bacilli. Grow on enriched media for 24 hours then
transmitted to MacConkey agar (White dew drops colonies) or S-S agar.
Epizootiology:
Morbidity
and mortality can be high. Transmission is via fecal contamination
from
carriers or affected animals. Potentially
zoonotic.
Transmission:
-Direct
by the fecal-oral route.
- Bedding and nest cages, Rodent
excretion that contaminate both food and water. Rabbits housed in unhygienic
conditions.
Clinical:
Usually
nonspecific (General clinical signs), include depression, fever many are found
dead. Some have diarrhea, some abort.
Disease
may spread rapidly, with many cases appearing in a short time.
P.M:
-Rabbits
dying of peracute septicemic disease may have no lesions or only congestion of
the organs, a few petechial hemorrhages.
-Those living a little longer may
have multiple pinpoint focal necrosis on liver, and heart splenomegaly,
lymphadenopathy (mesenteric L.N), and ulceration of Peyer’s patches.
-
In some cases there is fibrinous enteritis.
-Metritis is found in pregnant Does
-Acute
form: Polyserositis, focal hepatic necrosis, splenomegaly, enteritis with
fibrinous exudate and suppurative metritis.
Diagnosis:
Morphology,
culture, serotyping.
D.D:
Systemic diseases Viral (VHD),
Bacterial (pseudotuberculosis, acute pasteurellosis, listeriosis, spirochetosis ,Staphylococcosis.
Protozoal: diseases (Toxaplasmosis)
Control:
Good husbandry. Feed and bedding contaminated by fecal matter of rodents
are a potential source of infection. Human carriers also are a possible.
Treatment and Prevention:
Broad spectrum antibiotics are recommended. However, when administrated orally, most antibiotics do not attain sufficient concentration in the blood stream to be effective against septicemia. Best results obtained with fluoroquinolone antibiotic.
Recommendation:
Treated adults become carriers. Therefore, elimination of carrier is best method for control.
2-Colibacillosis
Etiology: Attaching and effacing (enteropathogenic) strains of Escherichia
coli.
General: E. coli is a major cause of enteritis in commercial rabbitries and
is occasionally a problem in research facilities. The organism is not normally
present or is present in small numbers within the gastrointestinal tract of
suckling and weanling rabbits.
-When a change in intestinal pH occurs, there is a rapid proliferation of the bacteria.
- Factors that promote this growth are intestinal coccidiosis and diets that require a high hydrochloric acid content for digestion.
-The isolated strains are enteropathogenic (cause
intestinal disease, but do not
produce enterotoxins).
- Some strains affect only suckling
rabbits and attach to the full length of the small and large intestine, while
other strains affect weanlings only and attach only to the ileum and large intestine.
Pathogenesis:
- The organism colonizes and attaches to the Peyer’s patches then later colonizes and attaches to enterocytes. Intestinal lesions are most severe at 7-14 days post-inoculation (in experimental infection).
Clinical signs and PM findings
-Morbidity may be sporadic or epizootic and mortality is high.
- Dehydrated carcasses, perineal staining with watery, yellow to brown fecal
material.
-The small intestine is usually grossly normal.
-The cecum and colon may be distended with watery yellow to gray-brown
contents.
-Serosal ecchymoses may be found,
-Edema in the cecal and colonic walls.
- Enlarged mesenteric lymph nodes.
- In the very young (1-14 day old), Watery diarrhea colors the belly and the posterior part of the body with yellow to brown.
- The stomach is full of undigested
clotted milk. The whole nest usually affected at the same time.
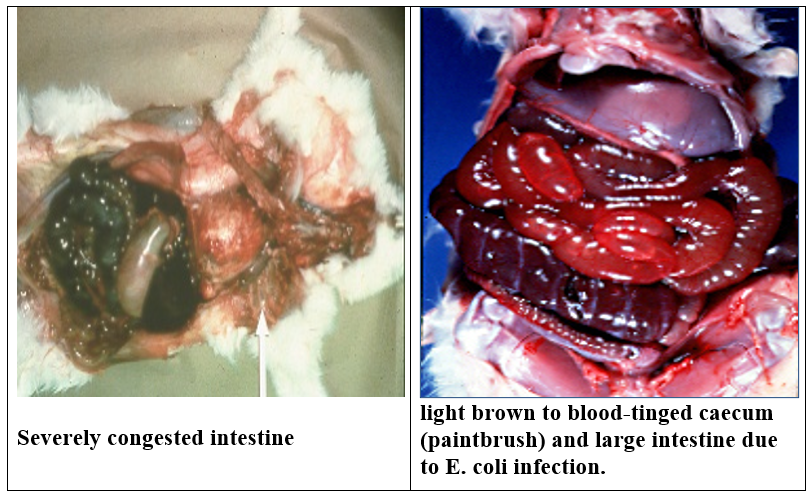
-Changes are most severe and extensive in
weanlings. lesions are limited to caecum and large intestine with watery content,
foul smelling light brown diarrhea tinged with blood.
- longitudinal hemorrhages from outer surface of caecum( paint brush) .
Diagnosis:
1- History and gross and microscopic lesions are suggestive.
2- Morphology, culture on MacConky
give red colonies,
EMB give green metallic chain, serotyping, eliminate other diseases.
And biochemical characterization of the organism.
Differential diagnosis :
Acute coccidiosis, clostridiosis, viral enteridities, Tyzzer’s disease and
mucoid
enteropathy.
Control: 1-Vet. Biosecurity. 2-Antibiotic
sensitivity test ( danofloxacin, Enrofloxacin)
3-Tyzzer’s Disease
Etiology:
“Bacillus”
piliformis, a gram-variable, large pleomorphic filamentous sporeformer of
uncertain classification.
It has not been grown on artificial
medium grow only on tissue culture also it can be cultivated in eggs as the
bacteria is obligatory intracellular.
Epizootiology:
Weanling rabbit 6-12 weeks are susceptible and stress plays a role in infection
Not
often recognized, but possibly widespread; many cases probably is diagnosed.
-Predisposing factors are important and include poor sanitation, stress and
sulfonamide therapy.
Transmission:
- thought to occur via fecal contamination
Infections are thought to be
subclinical parasitism of small numbers of enterocytes unless animals are
stressed or predisposed to disease by other unknown factors.
Morbidity can be sporadic with only a few rabbits affected, Survivors can
become
chronically infected and serve as carriers.
Outbreaks can be severe with 50% or more affected. Mortality is near 100%.
Clinical:
Acute onset of profuse watery diarrhea, depression, dehydration, death in a
day or two.
Chronic: chronic weight loss.
P.M:
Primarily the cecum is affected and sometimes the distal ileum and proximal
colon.
Serosal surfaces are reddened and sometimes petechiated. The wall is edematous.
Contents are brown, flocculent, and watery and the mucosa is rough and granular
with adherent exudate or necrotic debris.
Lesion is necrotizing and multifocal to diffuse.
Multifocal necrotizing hepatitis is common; usually the foci are small (up to
2-3 mm).
A few rabbits may have myocarditis.
Diagnosis:
-Culture the suspected sample in tissue culture Morphology; silver
impregnation (Warthin-Starry) to demonstrate intracellular bacilli
-Serological tests.
-Histopathological detect the intracellular bacilli.
Control:
Closed
colony establish bacilli.
Tetracycline
at half dose for one month 250 ppm in ration or 125mg /liter in drinking water.
4-Clostridiosis (Enterotoxemia)
Etiology :
Clostridium
difficile and C. spiroforme (C.spiroforme appears to be more common).
All
are Gram positive, anaerobic bacilli. C. spiroforme produces a type E iota toxin.
Epizootiology:
-C. difficile is a ubiquitous anaerobe
and may be carried in low numbers by normal rabbits.
C.
difficile causes colitis in rabbits following prolonged therapy with penicillin
and
ampicillin
( as antibiotic destruct normal flora).
-C. spiroforme is reported not to be
part of normal flora, but may colonize if flora upset (e.g., by antibiotic
treatment) At necropsy of diarrheic rabbits, it was isolated from over 50% of
the cases, and of those, 90% of the strains isolated were toxigenic in one
study.
-C. perfringens causes an
enterotoxemia-like condition in young rabbits that results in cecal hemorrhage
and edema.
-High
carbohydrate diet is one of the predisposing factors for enterotoxiaemia and
treatment
with following antibiotic: Lincomycin, penicillin, clindomycin ,erythromycin,
amoxycillin.
Clinical:
Acute sudden death
Peracute form brown watery diarrhea and
death in young or lactating rabbits. Loss of appetite, anorexia collaose, death
may occur within 2days to3 weeks due to loss of electrolyte.
Abdomens
may be enlarged by gas-filled intestines.
Chronic: anorexia, wasting and
intermittent diarrhea over several days.
P.M:
Necrotizing
enterocolitis, often with extensive edema and hemorrhage. thin and dehydrated
carcass; staining of the ventral abdomen, perineum and near legs with watery
green to tarry brown feces; straw colored peritoneal effusion; ecchymoses in
the cecal serosa with very liquid content and gases. Ulcer may be found on
mucosal surface of cecum
Diagnosis:
1-
Lesions, gram-stained fecal smear, anaerobic culture, toxin demonstration.
2- Stain caecal wall scrap u shaped
bacteria or spiral G+ve bacteria
3-Toxins
can be demonstrated by cytotoxicity assay, ELISA, CIE (counter
immunoelectrophoresis), and latex immunoassays.
Latex immunoassays are to be fastest and cheapest, and results agree well with those
of ELISA tests (at least in the case of C.difficile toxin). Both tests offer
positive and negative predictive values approaching or exceeding 90%. This is
better than the cytotoxicity assay, which is not very good at detecting low
concentrations of toxin.
N.B:
We have done cytotoxicity assays in several rabbit diarrhea outbreaks, but have
not yet found an unequivocal case of clostridiosis.
Differential diagnosis: Coccidiosis, Tyzzer’s disease,
and colibacillosis.
Treatment:
Tetracycline,
Nitrofuran, Metronidazole these drugs could be effective.
5-Enteritis Complex
Synonyms: Mucoid Enteropathy (Mucoid Enteritis)
Etiology:
Multifactorial;
one or combinations of bacteria, toxins, dietary irregularity and/or
obstruction
are suggested.
General: A major cause of disease and
mortality in young rabbits. This disease can be reproduced experimentally by
ligating sections of the large intestine. It is a subacute, frequently fatal
disease, characterized by the passage of copious quantities of gelatinous mucus
with feces. Rabbits 7-10 weeks of age are most often affected; however, rabbits
aged between 5-20 weeks may also be affected.
Pathogenesis:
It
is suggested that an alteration in the cecal environment results in the
production of a goblet cell secrete by an undetermined bacterium. The
secreta is absorbed
through
the cecal mucosa and is transported to the colon where it exerts its effect.
Clinical signs:
-Anorexia,
polydipsia and subnormal temperature in addition to the abnormal feces.
- Gastric distention by fluid and
gas; distention of the jejunum by translucent, watery fluid; cecal impaction by
dry contents and gas; distention of the sacculated colon by clear, gelatinous
mucoid exudate.
PM: Striking goblet cell hyperplasia
in the jejunal, ileal and colonic mucosa with minimal or no inflammation. In
the colon, the crypts and lumen are distended with mucus and mucus plugs. There
are minimal to absent lesions in the cecum. Goblet cell hyperplasia of the
gallbladder and mild nephrosis has been described.
Diagnosis:
-Characteristic histologic
appearance. Demonstrate goblet cells with periodic
acid
schiff or Alcian blue stains.
Differential diagnosis: Any infectious or management
problem that results in disruption of normal microbial environment
(coccidiosis, clostridiosis, trichobezars or constipation).
Carbohydrate Overload: low fiber, high starch diets fed
to young animals results in high concentrations of starch in the cecum and
colon which may result in the proliferation of E. coli, Clostridium perfringens, or Clostridium spiroforme.
Bacterial toxins produced during the fermentation process may damage the
mucosal surface and cause movement of water and electrolytes into the lumen,
resulting in diarrhea and dehydration followed by death.
- PROTOZOAL DISEASES
1- Coccidiosis
General: Coccidiosis is a common,
widespread problem in commercial operations
and
research facilities. It is an important economic and complicating disease.
Coccidia may act as a co-pathogen in other infections. As with the other causes
of enteritis, changes in management practices such as feeding or experimental
procedures can predispose to infection and disease.
There are two forms of coccidiosis in rabbits - an intestinal form, and a hepatic form. Eimeria species commonly cause enteric disease in large groups of rabbits, especially in young animals.
Disease Forms/Subtypes
Hepatic
caused by:
Eimeria stiedae.
-Subclinical
disease is common.
-Acute mortality is associated with large infective oocyst dose.
-When clinical disease is present, the signs are variable.
Intestinal form (Intestinal Coccidiosis)
- Subclinical disease is common in adult rabbits. Pathogenicity varies with Species of Eimeria. All intestinal species of Eimeria appear to be pathogenic in young rabbits.
- In adult rabbits, E. coecicola, E. irresidua, and E. magna are highly pathogenic; E. piriformis and E. media are moderately pathogenic; and E. perforans is mildly pathogenic.
Although rabbits are cecotrophic, it is generally accepted that cecotropes do not contain infectious oocyst.
Transmission:
Fecal-oral.
After passage in the feces, the oocysts require one or more days to sporulate.
Pathogenesis:
-After
the sporulated oocysts are ingested, sporozoites are released which invade
enterocytes
and multiply via schizogony.
- One or more sexual cycles
(depending on the species) takes place, then gametogony occurs and oocysts are
formed and passed in the feces.
-
Most Eimeria species in rabbits affect one or more segments of small
intestine;
a few affect cecum or colon also.
- Parasitized enterocytes are lost
resulting in superficially necrotizing enteritis. Severity of epithelial
destruction and degree of inflammatory response vary considerably.
Clinical signs
Intestinal Coccidiosis
Varies from none to profuse watery, even bloody, diarrhea and death, depending
on susceptibility of host (young or
not previously exposed are more susceptible),
species
of causative organism, and inoculating dose. Mild or no signs are more
common.
The cecum and colon contain dark green to brown, watery, foul smelling
material. The mucosa is congested and edematous.
Pathology: Location of the lesions is
dependent on the species involved. Destruction of
enterocytes,
villous atrophy, marked heterophilic infiltration and presence of gametocytes
and oocytes.
Hepatic coccidiosis
Etiology: Eimeria stiedae.
-Infected
rabbits showed poor weight gains, clinical disease and even death in affected
colonies.
-Weanlings are most often affected; older rabbits develop immunity.
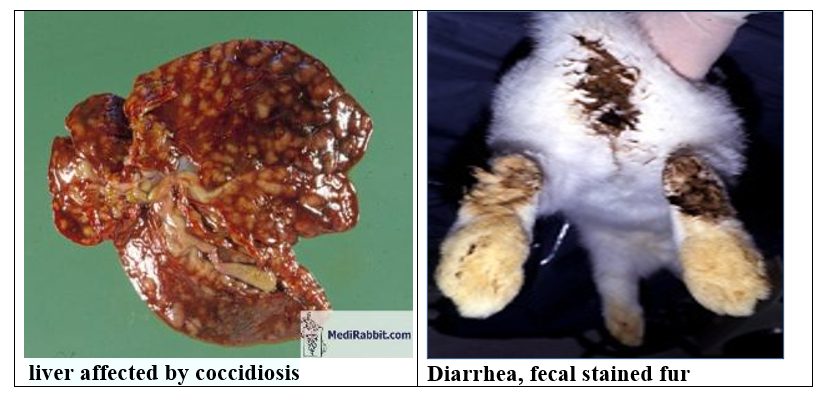
-The liver is enlarged owing to papillary hyperplasia of the bile duct epithelium (and gallbladder occasionally) with different developmental stages of coccidia within bile ducts.
-Acute cases may show numerous miliary hepatic abscesses.
-Chronic cases develop a fibrotic response around affected ducts.
-Other organs are not infected.
Life-cycle of Eimeria stiedae.
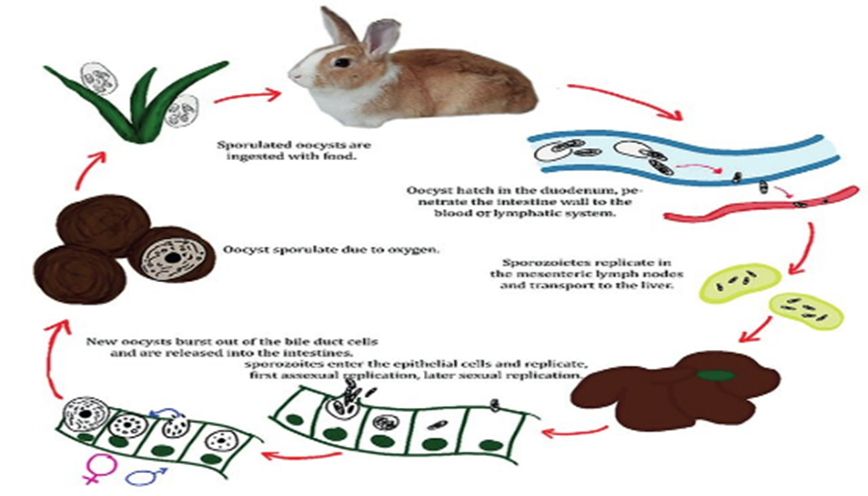
Transmission:
After
ingestion of sporulated oocysts, sporozoites penetrate intestinal epithelial
cells then are transported to the liver where they invade epithelial cells
lining bile ducts and undergo schizogony. After gametogony, oocysts are
released into bile ducts, pass to the intestinal tract via the bile and are
then passed into the feces.
Clinical
signs:
-
None to anorexia, debilitation, constipation or diarrhea. May also see
hepatomegaly, pendulous abdomen, icterus and death. Elevated liver enzymes and serum bilirubin on clinical
pathology.
-
Hepatomegaly with multifocal, raised, yellow to pearl grey, circumscribed,
0.5-2 um foci which contain an inspissated dark green to tan material. In the
liver; these are bile ducts chronically inflamed and dilated with bile and
exudate.
- Microscopic:
chronic proliferative cholangitis and cholecystitis, with numerous schizonts,
microgametes, macrogametes, and developing oocysts in the epithelial cells;
also, oocysts in the lumen.
D.D:
Enteric
Diseases: (enterotoxemia, Tyzzer’s disease, coccidiosis, and mucoid
enteropathy, E.coli , salmonella)
- Treatment digestive system problems
Treatment
Diarrhea due to, E.coli or Salmonella or coccidian:
- Sulpha + trimethoprim at 40mg/kg b.w.
Neomycin200-800mg \Liter.
- Gram negative bacteria in intestinal infection: Enrofloxacin (10% oral solution )100mg\ Liter.
- Treatment Diarrhea due to Enterotoxaemia, Tyzzer’s disease
-Mebendazole (10%) 1 g twice \day for 2kg body weight, Metrindazole
40mg/kgb.w or tetracycline or Nitrofurane .
- Cholestramine 0.5g/kgb.w. as prophylactic
against enterotoxaemia.
- Chronic diarrhea:
Dexamithazone 1-3mg/kg.b.w. IM or IV.
- Enteritis:
Iopermide hydrochloride (0.1mg/kgb.w for 3 days .3
time
per day then once for 2 days + drinking water substituted by Ringer lactate
0.1% glucose intrapertonial in badly affected rabbits.
- In Rabbit showed dehydration sterile
solution containing amino
acids, electrolytes, glucose and
vit. B complex can give intraperitoneally once a day.
- Treating constipation and impaction: liquid
profain 1-2ml/kg b.w.
- Toxic antibiotic:
- Lincomycin and clindamycin are very toxic reach 100%
mortality
-
Penicillin produce mortality 0-100%,
-Ampicillin minimize toxicity with macroloides, spiramycin, tylosin, erythromycin, oleandomycin.
NB:
Vancomycin used as antidote to antibiotic toxicity but It is too expensive.
Anticoccidial drugs work best in the early stages of the disease:
- Sulfonamides have the advantage that they are often also effective against secondary bacterial pathogens.
- Trimethoprim/sulfamethoxazole 30 mg/kg q 24 h PO (mean given orally every 24 hours);
- sulfadimethoxine 50 mg/kg first dose, then 25 mg/kg q 24 h PO
- Toltrazuril (25 mg/kg daily for 2 days PO, then repeat after 5 days) is a highly effective anticoccidial treatment.
Provide supportive care for dehydration:
- Correct dehydration over 12 to 24 hours. can be given subcutaneously, intravenously.
- In collapsed rabbits, track the response by monitoring blood pressure.
-Provide nutritional support by syringe feeding or via a nasogastric tube.
-Prokinetic drugs help normalize gut motility (e.g., metoclopramide 0.5-1 mg/kg q 6-8 h PO, SC; cisapride 0.5-1 mg/kg q 6-8 h PO)
-H2 histamine-receptor antagonists (e.g., ranitidine 2-5 mg/kg q 12 h PO) may stimulate gut motility but may also reduce the risk of gastric ulceration.
N.B:- Drug calculation could be estimated through a software program - https://morfz.com/rx/drugcalc.html
- Prevention and control of digestive diseases
-Frequent cleaning of rabbit accommodation, feeding, and water containers
-Efficient removal of feces daily (before oocysts become infective)
-Change bedding litter frequently, and keep it dry.
-Control vermin and flies that could serve as mechanical vectors.
-Avoid fecal contamination of food.
-Keep rabbits younger than 4 months old out of contaminated areas.
-Where there is a problem in spite of good husbandry, prophylactic anticoccidials may be needed.
-Because coccidiosis primarily affects young rabbits just after weaning (5- to 6-week-old animals), disease prevention must be initiated before weaning.
- Listeriosis
Etiology:
Listeria
monocytogenes. Gram positive, motile, non-spore-forming coccobacillus
General:
Occurs sporadically as an epizootic in rabbits.
Clinical signs
Pathogenesis:
This organism has tropism for the uterus
and placenta.
-Characterized
by depression, anorexia and fever, abortions in pregnant and sudden death in
does in late gestation.
- Infected
newborn kits may develop systemic disease, may have stunted growth and/or
develop
meningoencephalitis.
-Nervous
disorder as torticollis, opisthotonous and convulsion
P.M: Macro
Stillbirths,
dead fetus in uterus, Multiple necrotic foci in the liver and spleen;
lymphadenopathy;
ecchymoses; acute metritis; hydrothorax; ascites; and anasarca straw colored
fluid in peritoneal cavity, visceral congestion.
Micro:
Coagulative necrosis with polymorphonuclear cell infiltration.
Transmission:
Ingestion
(contaminated feed and water are possible sources) and transplacental
infection.
Diagnosis: Bacterial culture.
D.D: Pasterullosis, Salmonella
Treatment: 1-Vet.Biosecurity 2-Tetracycline.
- Skin problems
1-Rabbit Syphilis
Spirochetosis, Treponematosis
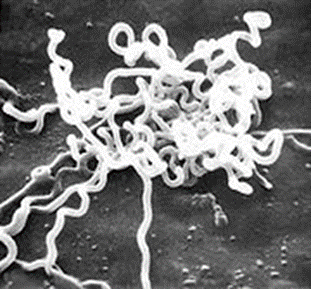
Etiology:
Treponema
cuniculi. Characteristic Gram-negative, 5-20 um, helical rods with tight or
irregular spirals
General: The disease is also referred to as
rabbit syphilis and vent disease and
is
common
in wild hares. This disease occurs occasionally in conventional facilities and
asymptomatic rabbits may be serologically positive.
Transmission: Zoonotic disease (from human to
rabbit not from rabbit to human)
Venereal,
although transmission through extragenital contact can occur. The organism can
penetrate intact mucous membranes. Susceptibility is age and breed dependent.
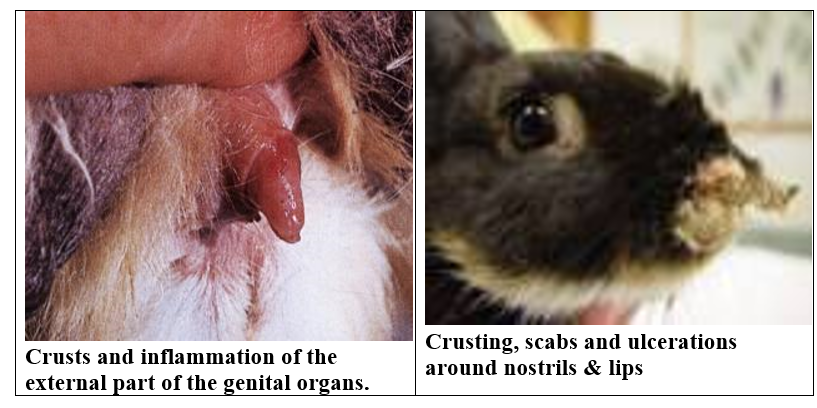
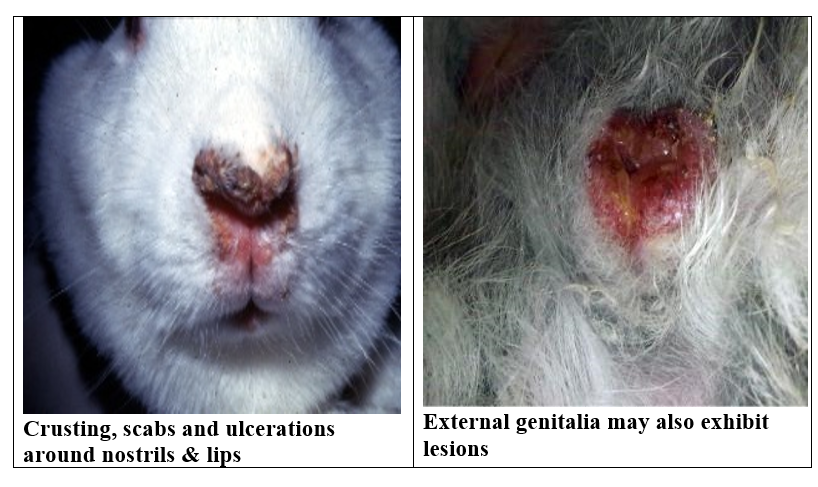
Clinical signs:
Edema
and erythema at the mucocutaneous junctions of the vulva, prepuce, anal region,
muzzle and periorbital area. Lesions are often crusty. Popliteal and inguinal
lymph nodes may be enlarged. Desire for mating is lost.
P.M:
Lesion
is confined to the epithelium and superficial dermis.
Epidermal
hyperplasia, epidermal cell necrosis, erosions and ulceration and infiltration
by predominantly plasma cells and lymphocytes with fewer heterophils.
Diagnosis:
-Scrapings
with wet mount preparation and examination under dark-field microscopy or in
silver-stained tissue sections.
-Serological
( Reagin antibody and fluorescent treponemal antigen test) .
Differential diagnosis:
Pasteurella sp. infections of the
external genitalia and lesions due to trauma, Listeriosis, Fleas,
Pseudotuberculosis(yersiniosis)
Treatment:
Penicillin
40,000 IU/kg q7days for rabbit (syphilis) may lead to diarrhea., so
tetracycline or arsenic preparation injected.
2-Ectoparasites (Mange)
Mange (“ear canker.”)
A non-burrowing fur mite that is
non-pathogenic even in heavy infestations The life cycle is about 5 weeks.. It
is not zoonotic.
Psoroptes cuniculi Rabbit ear mite.
-Non-burrowing mite that causes intense irritation – head shaking, scratching of the ears, hyperaemia - and the production of exudate leading to thick crust formation filling the auditory canal. Mites irritate the lining of the ear and cause serum and thick brown crusts to accumulate, creating an “ear canker.”
Lesions can spread to the face and neck, the ear drum can perforate leading to a purulent otitis media (secondary bacterial infection) and meningitis, reach the CNS, and result in torticollis.

N.B:
-The brown crumbly exudate should never be removed in a conscious rabbit, because this is very painful.
- The crusts will slowly slough off as the mites die and the tissue underneath heals. -The incidence is much lower when rabbits are housed in wire cages instead of solid cages.
Transmission:
The mite is readily transmitted by direct contact.
Diagnosis:
-Mites can be visualized on otoscopic examination.
- Microscopic examination of aural debris.
Diagnosis
- The examined areas of the body are ears, head, neck, thorax, abdomen, and extremities.
In ear mange, the lesion score is as follows:
0 for apparently normal ears
1 for lesions inside the ear
2 for lesions on the bottom third of the ear
3 for lesions extending to the two thirds of the ea
and 4 for lesions with a greater extension than two thirds of the ear.
- Serological tests such as enzyme-linked immunosorbent assay are used to detect the antibodies against Sarcoptic scabiei
Different strategies to control mange in rabbits
Control of rabbit mange:
-Hygienic measures
-Using acaricides:
A) Systemic ( organophosphorous compounds, synthetic pyrethroids).
b) local (Mcrolactones, benzyl- benzoatessulpher- based compounds).
-Using vitamin (A,D and E)
Treatment:
-Should be aggressive with supportive therapy given for
toxic shock, and ivermectin will kill any larvae that emerge from unremoved
eggs.
- Bath with chlorhexidine or a povidene-iodine cleanser.
Give fliuds s/c or, i.v. or i.p. plus ivermectin i.m. 200mcg/kg to kill those maggots out of reach.
- Injectable ivermectin treatment regimens effective against both fur and ear mites have been reported, with the dosage of ivermectin 200–400 mcg/kg, SC, two or three treatments 10–21 days apart.
-Mites may also be treated with selamectin (20 mg topically every 7 days has been effective).
- Fungal Dermatitis or Superficial Mycosis

Def: Fungal dermatitis, also known as superficial mycosis, refers to skin infections caused by fungi that primarily affect the outermost layers of the skin, hair, and nails. These infections are common and often contagious, especially in warm, humid environments.
The causing agents are two pathogenic fungi:
- Trichophyton mentagrophytes = ringworm
May be carried asymptomatically in the coat of the rabbit.
- Microsporum canis
Occasionally rabbits are infected through contact with cats or dogs; this fungus can carry one or more zoonotic diseases (diseases that can be transmitted from animal to human).
Ringworm is the main cause of mycosis. It will infect not only the epidermis, but also the annex structures such as hair follicles and hair shafts. In some cases, cultures of infected tissue have revealed the presence of Microsporum sp.
Clinical signs:
Typically, lesions start around the head and spread to the legs and feet, more specifically to the toenail beds. The wound is raised, circumscribed and erythematous. It shows dry crusts with little or no pruritus and patchy alopecia. The tissue under the crusts usually shows inflammation and the hair follicles show abscessation, as the secondary result of a bacterial invasion.
Diagnosis:
1. Fungal or Dermatophyte Cultures: Grow the organisms on specialized fungal or dermatophyte media to identify the causative fungi.
2. Skin Scrapings Examination: Mount skin scrapings in 10% potassium hydroxide (KOH) to visualize and identify spores under the microscope.
3. Fluorescence under UV Light: Fluorescent examination offers limited assistance. *Trichophyton mentagrophytes* generally does not fluoresce, whereas *Microsporum canis* may fluoresce depending on the strain—some strains glow, others do not.
4. Special Stains: Techniques such as Gomori- methenamine silver stain, Gridley fungus stain, and periodic acid-Schiff (PAS) reaction help detect and identify spores in tissue samples.
Treatment: The hair around the lesion should be clipped, and disposed off safely. The best treatment for fungal dermatitis is oral administration of griseofulvin (25-50 mg/kg PO q24h or divided q12h). (Wear gloves while administering this drug). The treatment should continue two weeks after the disappearance of the clinical signs. Topical or systemic treatments are also possible: Antifungal cream or lotion; or spray; Miconazole cream.
The environment of the rabbit should be cleaned carefully: vacuum cleaning, boiling of towels and cleaning of surfaces with 1:10 bleach water.
D.D: The diagnosis should be distinguished from other causes of crusty hair loss on the scalp and ears, such as genetic hairlessness, trauma, depilatory hair loss, or fur pulling.
- Reference
- Bortolotti A, Castelli D, Bonati M.
1989. Hematology and serum chemistry values of adult, pregnant and newborn New
Zealand rabbits (Oryctolagus cuniculus). Lab Anim Sci 39:437-439.
- Broderson
JR. 1989. A retrospective review of lesions associated with the use of
Freund's
adjuvant. Lab Anim Sci 39:400-405.
- Brown
RL et al. 1969. Ultrastructural pathology of an acute fatal enteritis of
captive cottontail rabbits. Search for an etiologic agent. Am J Pathol
57:93-107.
- Burgess
EC, Windberg LA. 1989. Borrelia sp. infection in coyotes, black-tailed jack rabbits
and desert cottontails in southern Texas. J Wildl Dis 25:47-51.
- Fallon
MT et al. 1988. Diagnostic exercise: eye lesions in a rabbit. Lab Anim Sci
38:612-613
-Fox, R.R et al. 1971. Hereditary
cortical cysts in the rabbit. J Heredity 62:105-109.
- Garibaldi
BA, Goad ME. 1988. Hypercalcemia with secondary nephrolithiasis in a rabbit.
Lab Anim Sci 38:331-333.
- Garibaldi
BA, Goad ME. 1988. Lipid keratopathy in the Watanabe (WHHL) rabbit. Vet Pathol
25:173-174.
- Gillett
NA et al. 1983. Medical and surgical management of gastric obstruction from a
hair ball in the rabbit. J Am Vet Med Assoc 183:1176-1178.
- Green
PW et al. 1984. Spontaneous degenerative spinal disease in the laboratory
rabbit.
J Orthop Res 2:161-168.
- Green
RA, Baker DC. 1987. Coagulation defects of aflatoxin intoxicated rabbits. Vet
Pathol 24:62-70.
- Hanglow,
A.C et al. 1986. Synovitis associated with serum IgM rheumatoid factor arising
spontaneously in 'Old English' rabbits. Ann Rheum Dis 45:331-338.
- Hill
JE et al. 1988. Spontaneous storage-like disease in a rabbit. Vet Pathol
25:91-92.
-Hinton
M. 1981. Kidney disease in the rabbit: A histological survey. Lab Anim
15:263-265.
- Hofmann
JR Jr, Hixson CJ. 1986. Amyloid A protein deposits in a rabbit with pyometra. J
Am Vet Med Assoc 189:1155-1186.
- Lelkes L. 1987. A review of
rabbit enteric diseases: A new perspective. J Appl Rabbit Res 10:55-61.
- Leary
SL et al. 1984. Experimental and naturally-occurring gastric foreign bodies in
laboratory rabbits. Lab Anim Sci 34:58-61.
- Lee
KJ et al. 1978. Hydronephrosis caused by urinary lithiasis in a New Zealand
white rabbit (Oryctolagus cuniculus). Vet Pathol. 15:676-678.
- Machii
K et al. 1989. Infection of rabbits with Sendai virus. Lab Anim Sci 39:334-337.
- Weisbroth SH, Flatt RE, Kraus AL
(eds). The Biology of the Laboratory Rabbit.
Academic
Press, New York, 1974.
- https://lbah.com/rabbit/pasteurella-rabbit/
- https://www.raising-rabbits.com/rabbit-syphilis.html
- https://www.medirabbit.com/EN/Skin_diseases/Parasitic/Mange/Sarcoptes.htm
- https://www.researchgate.net/publication/360584494_Ultrasonographic_diagnosis_of_hepatic_coccidiosis_in_rabbits/
- https://link.springer.com/article/10.1007/s00580-020-03115-x/figures/1
- https://www.medirabbit.com/EN/Skin_diseases/Bacterial/Syph_gen.html
- https://www.fao.org/4/t0756e/T0756E09.htm
https://www.medirabbit.com/EN/GI_diseases/Protozoal_diseases/Cocc_en.htm.
- https://atlas-parazitov.uvlf.sk/?p=917
- https://www.medirabbit.com/EN/Skin_diseases/Fungal/Fungal_en.htm.
- https://morfz.com/rx/drugcalc.html