Prevention and Management of Ovarian Hyperstimulation Syndrome (OHSS)
| Site: | EHC | Egyptian Health Council |
| Course: | ِAssisted Reproductive Technologies Guidelines |
| Book: | Prevention and Management of Ovarian Hyperstimulation Syndrome (OHSS) |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
9 July 2025" Download Guideline
- Executive Summary
· It is recommended to counsel patients with elevated anti-müllerian hormone levels, polycystic ovary syndrome (PCOS), and anticipated high oocyte yields that they are at increased risk for ovarian hyperstimulation syndrome (OHSS). Interventions to reduce OHSS risk should be focused on this patient population.
(Strong Recommendation).
· It is recommended to dose gonadotropins based on individualized ovarian reserve testing to decrease the risk of OHSS (Strong Recommendation).
· Consider lowering the starting dose of gonadotropins and/or supplementing with oral ovulation-inducing medications (clomiphene citrate and/or letrozole) to decrease the risk of OHSS (Conditional Recommendation).
· Use a GnRH agonist to trigger oocyte maturation as a first-line strategy to reduce the risk of moderate-to-severe OHSS (Strong Recommendation).
· Coasting is generally not recommended as a primary strategy to reduce the risk of moderate-to-severe OHSS. However, when other more effective strategies are not available, coasting in combination with cabergoline and a freeze-only strategy may mitigate the risk (Conditional recommendation).
· Provide adequate luteal support when using a GnRH agonist as a trigger and planning a fresh embryo transfer (Strong Recommendation).
· In patients at risk for moderate-to-severe OHSS, start a dopamine agonist such as cabergoline on the day of the hCG trigger or soon after that and continue for several days (Strong Recommendation).
· Consider a freeze-only cycle and subsequent frozen embryo transfer in patients at risk for OHSS based on a high ovarian response or elevated serum estradiol levels. Multiple high-quality studies have reported a significant reduction in rates of moderate or severe OHSS when this strategy is employed (Strong Recommendation).
· Do not use a lower dose for the human chorionic gonadotropin (hCG)-only trigger as a strategy to reduce the risk of moderate-to-severe OHSS (Conditional Recommendation).
· Do not administer letrozole as an intervention to reduce rates of moderate-to-severe OHSS (Conditional Recommendation).
· Do not administer a luteal GnRH antagonist alone to reduce rates of moderate-to-severe OHSS. Most studies report no reduction in rates of moderate-to-severe OHSS or associated signs or symptoms. Some low-quality evidence suggests modest symptomatic improvement in women with OHSS who received a GnRH antagonist after the hCG trigger (Conditional Recommendation).
· Do not use aspirin as a primary strategy to reduce the incidence of OHSS (Conditional Recommendation).
· Do not administer metformin for the sole purpose of reducing the incidence of OHSS in GnRH antagonist cycles. Most studies do not report a significant reduction in OHSS rates in women with PCOS who were given metformin. However, metformin may be considered for OHSS risk reduction among women with PCOS using a GnRH-agonist protocol (Conditional Recommendation).
· Do not administer medications such as mifepristone, myoinositol, D-chiro-inositol, or glucocorticoids to reduce rates of OHSS as studies have shown these interventions to be ineffective (Conditional Recommendation).
· Do not use volume expanders such as albumin, hydroxyethyl starch, or mannitol in patients at high risk of developing moderate or severe OHSS (Conditional Recommendation).
· Clinicians need to be aware of the symptoms and signs of OHSS, as the diagnosis is based on clinical criteria. (Conditional Recommendation). Table 4
· Women presenting with symptoms suggestive of OHSS should be assessed face-to-face by a clinician. In women presenting with severe abdominal pain or pyrexia, extra care should be taken to rule out other causes of the patient’s symptoms. The input of clinicians experienced in the management of OHSS should be obtained in such cases. (Conditional Recommendation).
· It is recommended that once the diagnosis of OHSS is established, the severity of the disease should be classified as mild, moderate, severe, or critical according to the standardized classification scheme included. (Conditional Recommendation). Table 5
· Outpatient management is recommended for women who have mild or moderate OHSS and only in selected cases with severe OHSS when the physician can ensure that the patient can follow clinical guidelines and ensure that a system is in place to assess the condition every 1 to 2 days. (Conditional Recommendation).
· Women undergoing outpatient management of OHSS should be appropriately counselled and provided with information regarding fluid intake and output monitoring. In addition, they should be provided with contact details to access advice. (Conditional Recommendation)
· Fluid replacement by the oral route, guided by thirst, is the most physiological approach to correcting intravascular dehydration. (Conditional Recommendation)
· Women with moderate OHSS should be evaluated for predisposing risk factors for thrombosis and prescribed either antiembolism stockings or LMWH if indicated. (Conditional Recommendation).
· Women with severe OHSS being managed on an outpatient basis should receive thromboprophylaxis with low molecular weight heparin (LMWH). The duration of treatment should be individualised, taking into account risk factors and whether or not conception occurs. (Conditional Recommendation).
· Women with OHSS being managed on an outpatient basis should be reviewed urgently if they develop symptoms or signs of worsening OHSS. In the absence of these, a review every 2–3 days is likely to be adequate. (Conditional Recommendation).
· Baseline laboratory investigations should be repeated if the severity of OHSS is thought to be worsening. Haematocrit is a useful guide to the degree of intravascular volume depletion. (Conditional Recommendation) Table 6
· Outpatient paracentesis/culdocentesis of ascitic fluid by the abdominal or transvaginal route under ultrasound guidance may be considered for prevention of disease progression in cases of moderate to severe OHSS. (Conditional Recommendation).
· Hospital admission should be considered for women who:
· are unable to achieve satisfactory pain control
· are unable to maintain adequate fluid intake due to nausea
· show signs of worsening OHSS despite outpatient intervention
· are unable to attend for regular outpatient follow-up
· have critical OHSS.
(Conditional
Recommendation).
· Women with severe and critical OHSS should be hospitalized for intravenous hydration and observation. (Conditional Recommendation)
· Intravenous hydration with crystalloid solution should be implemented to prevent hemoconcentration and provide adequate organ perfusion targets. When the use of a crystalloid solution does not allow the maintenance of such an infusion, an alternative colloidal solution should be administered. (Conditional Recommendation).
· Women admitted with OHSS should be assessed at least once daily. More frequent assessment is appropriate for women with critical OHSS and those with complications. (Conditional Recommendation).
· Antiemetics may be used in women with OHSS, avoiding medicines contraindicated in pregnancy. (Conditional Recommendation).
· Women with persistent haemoconcentration despite volume replacement with intravenous colloids may need invasive monitoring, and this should be managed with anaesthetic input. (Conditional Recommendation) (40).
· Features of critical OHSS should prompt consideration of the need for intensive care. Multidisciplinary assistance should be sought for the care of women with critical OHSS and severe OHSS who have persistent haemoconcentration and dehydration. A clinician experienced in the management of OHSS should remain in overall charge of the woman’s care. Intravenous colloid therapy should be considered for women who have large volumes of fluid removed by paracentesis. (Conditional Recommendation)
· In hospitalized patients, pain relief should be conducted with acetaminophen and/or opioid analgesics. (Conditional Recommendation).
· Prophylactic doses of anticoagulants should be considered in women who have severe ovarian hyperstimulation syndrome. The duration of LMWH prophylaxis should be individualized according to patient risk factors and outcome of treatment. (Conditional Recommendation).
· Paracentesis should be performed to alleviate discomfort in hospitalized patients with tense ascites. (Conditional Recommendation)
Indications for paracentesis include the following:
- severe abdominal distension and abdominal pain secondary to ascites
- shortness of breath and respiratory compromise secondary to ascites and increased intra-abdominal pressure
- oliguria despite adequate volume replacement, secondary to increased abdominal pressure causing reduced renal perfusion.
· In addition to the usual symptoms and signs of venous thromboembolism (VTE), thromboembolism should be suspected in women with OHSS who present with unusual neurological symptoms, even if they present several weeks after apparent improvement in OHSS. (Conditional Recommendation)
· Surgery is only indicated in patients with OHSS if there is a coincident problem such as adnexal torsion, ovarian rupture, or ectopic pregnancy, and should be performed by an experienced surgeon. (Conditional Recommendation)
· Non-steroidal anti-inflammatory drugs with antiplatelet properties shouldn't be used for pain relief as they may compromise renal function. (Conditional Recommendation)
· Diuretics should be avoided as they further deplete intravascular volume, but they may have a role in a multidisciplinary setting if oliguria persists despite adequate fluid replacement and drainage of ascites. (Conditional Recommendation)
· There is insufficient evidence to support the use of gonadotrophin-releasing hormone antagonists or dopamine agonists in treating established OHSS. Those drugs should not be used as routine management of OHSS. (Conditional Recommendation).
- Recommendations
I) Prevention of OHSS:
Patient Counselling
· It is recommended to counsel patients with elevated anti-müllerian hormone levels, polycystic ovary syndrome (PCOS), and anticipated high oocyte yields that they are at increased risk for ovarian hyperstimulation syndrome (OHSS). Interventions to reduce OHSS risk should be focused on this patient population.
(Strong Recommendation, high grade evidence) (15).
During Ovarian Stimulation:
GnRH Antagonists
· It is recommended to dose gonadotropins based on individualized ovarian reserve testing to decrease the risk of OHSS (Strong Recommendation, high grade evidence) (16).
Lower Starting Dose
· Consider lowering the starting dose of gonadotropins and/or supplementing with oral ovulation-inducing medications (clomiphene citrate and/or letrozole) to decrease the risk of OHSS (Conditional Recommendation, moderate grade evidence) (18).
GnRH Agonist Trigger
· Use a GnRH agonist to trigger oocyte maturation as a first-line strategy to reduce the risk of moderate-to-severe OHSS (Strong Recommendation, high grade evidence) (19).
Coasting
· Coasting is generally not recommended as a primary strategy to reduce the risk of moderate-to-severe OHSS. However, when other more effective strategies are not available, coasting in combination with cabergoline and a freeze-only strategy may mitigate the risk (Conditional recommendation, low grade evidence) (23).
Luteal Support:
Adequate Luteal Support
· Provide adequate luteal support when using a GnRH agonist as a trigger and planning a fresh embryo transfer (Strong Recommendation, high grade evidence) (20).
Dopamine Agonist:
Cabergoline
· In patients at risk for moderate-to-severe OHSS, start a dopamine agonist such as cabergoline on the day of the hCG trigger or soon after that and continue for several days (Strong Recommendation, high grade evidence) (21).
Freeze-All Strategy:
Freeze-Only Cycle
· Consider a freeze-only cycle and subsequent frozen embryo transfer in patients at risk for OHSS based on a high ovarian response or elevated serum estradiol levels. Multiple high-quality studies have reported a significant reduction in rates of moderate or severe OHSS when this strategy is employed (Strong Recommendation, high grade evidence) (22).
Trigger:
hCG-Only Trigger
· Do not use a lower dose for the human chorionic gonadotropin (hCG)-only trigger as a strategy to reduce the risk of moderate-to-severe OHSS (Conditional Recommendation, low grade evidence) (24).
Adjuvants During Luteal Phase:
Letrozole
· Do not administer letrozole as an intervention to reduce rates of moderate-to-severe OHSS (Conditional Recommendation, moderate grade evidence) (25).
Luteal GnRH Antagonist
· Do not administer a luteal GnRH antagonist alone to reduce rates of moderate-to-severe OHSS. Most studies report no reduction in rates of moderate-to-severe OHSS or associated signs or symptoms. Some low-quality evidence suggests modest symptomatic improvement in women with OHSS who received a GnRH antagonist after the hCG trigger (Conditional Recommendation, low grade evidence) (26).
Aspirin
· Do not use aspirin as a primary strategy to reduce the incidence of OHSS (Conditional Recommendation, low grade evidence) (27).
Metformin
· Do not administer metformin for the sole purpose of reducing the incidence of OHSS in GnRH antagonist cycles. Most studies do not report a significant reduction in OHSS rates in women with PCOS who were given metformin. However, metformin may be considered for OHSS risk reduction among women with PCOS using a GnRH-agonist protocol (Conditional Recommendation, moderate grade evidence) (28).
Ineffective Medications
· Do not administer medications such as mifepristone, myoinositol, D-chiro-inositol, or glucocorticoids to reduce rates of OHSS as studies have shown these interventions to be ineffective (Conditional Recommendation, low grade evidence) (29).
Volume Expanders
· Do not use volume expanders such as albumin, hydroxyethyl starch, or mannitol in patients at high risk of developing moderate or severe OHSS (Conditional Recommendation, low grade evidence) (30).
II) Outpatient management of OHSS:
Assessment of OHSS Symptoms:
· Clinicians need to be aware of the symptoms and signs of OHSS, as the diagnosis is based on clinical criteria. (Conditional Recommendation, low grade evidence) (8). Table 4
· Women presenting with symptoms suggestive of OHSS should be assessed face-to-face by a clinician. In women presenting with severe abdominal pain or pyrexia, extra care should be taken to rule out other causes of the patient’s symptoms. The input of clinicians experienced in the management of OHSS should be obtained in such cases. (Conditional Recommendation, low grade evidence) (8).
Classification of OHSS Severity:
· It is recommended that once the diagnosis of OHSS is established, the severity of the disease should be classified as mild, moderate, severe, or critical according to the standardized classification scheme included. (Conditional Recommendation, low grade evidence) (32). Table 5
Outpatient Management of OHSS:
Outpatient Management
· Outpatient management is recommended for women who have mild or moderate OHSS and only in selected cases with severe OHSS when the physician can ensure that the patient can follow clinical guidelines and ensure that a system is in place to assess the condition every 1 to 2 days. (Conditional Recommendation, low grade evidence) (33).
· Women undergoing outpatient management of OHSS should be appropriately counselled and provided with information regarding fluid intake and output monitoring. In addition, they should be provided with contact details to access advice. (Conditional Recommendation, low grade evidence) (30).
Fluid Replacement in Outpatient Management
· Fluid replacement by the oral route, guided by thirst, is the most physiological approach to correcting intravascular dehydration. (Conditional Recommendation, low grade evidence) (30).
Thromboprophylaxis in outpatient management
· Women with moderate OHSS should be evaluated for predisposing risk factors for thrombosis and prescribed either antiembolism stockings or LMWH if indicated. (Conditional Recommendation, low grade evidence) (34).
· Women with severe OHSS being managed on an outpatient basis should receive thromboprophylaxis with low molecular weight heparin (LMWH). The duration of treatment should be individualised, taking into account risk factors and whether or not conception occurs. (Conditional Recommendation, low grade evidence) (35).
Follow up & Review in Outpatient Management
· Women with OHSS being managed on an outpatient basis should be reviewed urgently if they develop symptoms or signs of worsening OHSS. In the absence of these, a review every 2–3 days is likely to be adequate. (Conditional Recommendation, low grade evidence) (36).
· Baseline laboratory investigations should be repeated if the severity of OHSS is thought to be worsening. Haematocrit is a useful guide to the degree of intravascular volume depletion. (Conditional Recommendation, low grade evidence) (36)Table 6
Paracentesis in Outpatient Management
· Outpatient paracentesis/culdocentesis of ascitic fluid by the abdominal or transvaginal route under ultrasound guidance may be considered for prevention of disease progression in cases of moderate to severe OHSS. (Conditional Recommendation, moderate grade evidence) (8).
III) Inpatient management of OHSS:
Hospital Admission
· Hospital admission should be considered for women who:
· are unable to achieve satisfactory pain control
· are unable to maintain adequate fluid intake due to nausea
· show signs of worsening OHSS despite outpatient intervention
· are unable to attend for regular outpatient follow-up
· have critical OHSS.
(Conditional
Recommendation, low grade evidence) (23).
Intravenous Hydration for Severe OHSS
· Women with severe and critical OHSS should be hospitalized for intravenous hydration and observation. (Conditional Recommendation, low grade evidence) (36).
· Intravenous hydration with crystalloid solution should be implemented to prevent hemoconcentration and provide adequate organ perfusion targets. When the use of a crystalloid solution does not allow the maintenance of such an infusion, an alternative colloidal solution should be administered. (Conditional Recommendation, moderate grade evidence) (37).
Daily Assessment of Hospitalized OHSS Patients
· Women admitted with OHSS should be assessed at least once daily. More frequent assessment is appropriate for women with critical OHSS and those with complications. (Conditional Recommendation, low grade evidence) (38).
Use of Antiemetics in OHSS
· Antiemetics may be used in women with OHSS, avoiding medicines contraindicated in pregnancy. (Conditional Recommendation, low grade evidence) (39).
Invasive Monitoring for Persistent Haemoconcentration
· Women with persistent haemoconcentration despite volume replacement with intravenous colloids may need invasive monitoring, and this should be managed with anaesthetic input. (Conditional Recommendation, low grade evidence) (40).
Intensive Care Consideration for Critical OHSS
· Features of critical OHSS should prompt consideration of the need for intensive care. Multidisciplinary assistance should be sought for the care of women with critical OHSS and severe OHSS who have persistent haemoconcentration and dehydration. A clinician experienced in the management of OHSS should remain in overall charge of the woman’s care. Intravenous colloid therapy should be considered for women who have large volumes of fluid removed by paracentesis. (Conditional Recommendation, low grade evidence) (40).
Pain Management in Hospitalized OHSS Patients
· In hospitalized patients, pain relief should be conducted with acetaminophen and/or opioid analgesics. (Conditional Recommendation, low grade evidence) (41).
Thromboprophylaxis for Hospitalized OHSS Patients
· Prophylactic doses of anticoagulants should be considered in women who have severe ovarian hyperstimulation syndrome. The duration of LMWH prophylaxis should be individualized according to patient risk factors and outcome of treatment. (Conditional Recommendation, moderate grade evidence) (42).
Paracentesis
· Paracentesis should be performed to alleviate discomfort in hospitalized patients with tense ascites. (Conditional Recommendation, moderate grade evidence) (8)
Indications for paracentesis include the following:
- severe abdominal distension and abdominal pain secondary to ascites
- shortness of breath and respiratory compromise secondary to ascites and increased intra-abdominal pressure
- oliguria despite adequate volume replacement, secondary to increased abdominal pressure causing reduced renal perfusion.
Suspected Thromboembolism with Neurological Symptoms
· In addition to the usual symptoms and signs of venous thromboembolism (VTE), thromboembolism should be suspected in women with OHSS who present with unusual neurological symptoms, even if they present several weeks after apparent improvement in OHSS. (Conditional Recommendation, moderate grade evidence) (43)
Surgery for OHSS
· Surgery is only indicated in patients with OHSS if there is a coincident problem such as adnexal torsion, ovarian rupture, or ectopic pregnancy, and should be performed by an experienced surgeon. (Conditional Recommendation, low grade evidence) (40)
Medication considerations in OHSS:
NSAIDs in Pain Management
· Non-steroidal anti-inflammatory drugs with antiplatelet properties shouldn't be used for pain relief as they may compromise renal function. (Conditional Recommendation, low grade evidence) (40).
Diuretics
· Diuretics should be avoided as they further deplete intravascular volume, but they may have a role in a multidisciplinary setting if oliguria persists despite adequate fluid replacement and drainage of ascites. (Conditional Recommendation, low grade evidence) (40).
GnRH Antagonists or Dopamine Agonists
· There is insufficient evidence to support the use of gonadotrophin-releasing hormone antagonists or dopamine agonists in treating established OHSS. Those drugs should not be used as routine management of OHSS. (Conditional Recommendation, low grade evidence) (21).
• IMPLEMENTATION CONSIDERATIONS
- Patient Information: Fertility clinics should provide verbal and written information concerning OHSS to all women undergoing fertility treatment and notify each patient of her risk of OHSS to which she is exposed. Information should include a 24-hour contact telephone number of a team member who has relevant experience. Special attention for ART patients who have a higher risk of developing OHSS and to patients who are in areas where they do not have easy access to physicians who are knowledgeable in the diagnosis and management of OHSS. (Strength of evidence: LOW; strength of recommendation: conditional) (23).
- OHSS Protocols for Acute and Emergency Units: All acute/emergency units where women with suspected OHSS are likely to present should establish agreed local protocols for the assessment and management of these women and ensure they have access to appropriately skilled clinicians with experience in the management of this condition. (Strength of evidence: LOW; strength of recommendation: conditional) (44).
- Liaison Between Fertility Treatment Centres and Emergency Units: Licensed centres that provide fertility treatment should ensure close liaison and coordination with acute/emergency units where their patients may present. (Strength of evidence: LOW; strength of recommendation: conditional) (8).
- Acknowledgements
We would like to acknowledge the Committee of National Egyptian Guidelines, the Ministry of Health and the OB/GYN Scientific Committee for adapting this guideline.
Chair of the Panel:
Mohamed Yehya Soliman
Scientific Group Members:
Gamal Abo El-Serour, Abdel Maguid Ramzy, Hisham Ali Saleh, Mohamed El Sherbiny, Wafaa Benjamin Basta, Amgad Mounir, Omar Shaaban.
- Abbreviations
ART: Assisted Reproductive Technology
ET: Embryo Transfer
GnRH: Gonadotrophic Releasing Hormone
hCG: human Chorionic Gonadotropin
ICU: Intensive Care Unit
IVF: In Vitro Fertilization
LMWH: Low Molecular Weight Heparin
OHSS: Ovarian Hyperstimulation Syndrome
PCOS: Polycystic Ovarian Syndrome
RCT: Randomized Controlled Trial
- Glossary
ART (assisted reproductive technology): All treatments or procedures that include the handling of human eggs or embryos to help a woman become pregnant. ART includes, but is not limited to, in vitro fertilization (IVF), gamete intrafallopian transfer (GIFT), zygote intrafallopian transfer (ZIFT), tubal embryo transfer, egg and embryo cryopreservation.
Cryopreservation: The process of slow freezing or vitrification to preserve biological material (e.g. gametes, zygotes, cleavage-stage embryos, blastocysts or gonadal tissue) at extremely low temperatures.
Embryo transfer: Placement of embryos into a woman’s uterus through the cervix after IVF. In zygote intrafallopian transfer or gamete intrafallopian transfer, zygotes or gametes are placed in a woman’s fallopian tube.
Embryo: The biological organism resulting from the development of the zygote, until eight completed weeks after fertilization, equivalent to 10 weeks of gestational age.
Implantation: the attachment and subsequent penetration by the zona-free blastocyst (usually in the endometrium) that starts 5 to 7 days after fertilization.
Informed consent: All those who are to give their consent should be given an oral explanation, supported by written information in plain language that is provided to them in sufficient time for it to be taken away, read and considered, before the giving of consent to any ART procedure, or any significant step in the procedure that requires consent. This explanation should be given with sensitivity to cultural diversity and accessibility to those with low literacy, disability, and/or those whose first language is not Arabic.
OHSS (Ovarian Hyperstimulation Syndrome): Ovarian hyperstimulation syndrome (OHSS) is an uncommon but serious complication associated with controlled ovarian stimulation during assisted reproductive technology (ART). OHSS is staged (mild, moderate, severe, or critical) by the severity of symptoms and laboratory findings. Ovarian hyperstimulation syndrome is further classified by the timing of onset (early or late).
- INTRODUCTION
Ovarian hyperstimulation syndrome (OHSS) is an uncommon yet serious complication associated with controlled ovarian stimulation during assisted reproductive technology (ART). Historically, moderate-to-severe OHSS has been reported to occur in approximately 1%–5% of in vitro fertilization (IVF) cycles (1-5). However, the true incidence is difficult to delineate due to the absence of a strict, consensus definition. The traditional description of the syndrome generally includes a spectrum of symptoms, such as abdominal distention and discomfort, dyspnea, and findings like ovarian enlargement, ascites, hemoconcentration, hypercoagulability, and electrolyte imbalances (6-9).
OHSS is staged as mild, moderate, severe, or critical, based on the severity of symptoms and laboratory findings (10). Additionally, it is classified by the timing of onset as early or late (11). Table 5
Classification:
- Early-onset OHSS: Occurs after controlled ovarian hyperstimulation and an ovulatory dose of hCG. Symptoms typically begin 4–7 days post-hCG trigger and usually resolve with menses (12).
- Late-onset OHSS: Usually begins at least 9 days post-hCG trigger, in response to rising hCG levels of pregnancy. This type is more severe and significantly prolongs the course of OHSS (13).
Ovarian torsion and ovarian haemorrhage may be considered serious complications of OHSS. Clinicians should be aware of these complications in history taking and examination of cases with OHSS. (31)
Severe OHSS can lead to life-threatening complications, including pleural effusion, acute renal insufficiency, and venous thromboembolism (14).
OHSS association mortality is recorded in the literature as 1:500,000. However, this may be an underestimation of the true related mortality rate as some cases are handled by other specialists. (31)
- Purpose and scope
The purpose of this guideline is to identify quality improvement opportunities in the prevention and management of OHSS and to create explicit and actionable recommendations to implement these opportunities in clinical practice.
Specifically, the goals are to promote the application of preventive interventions that could decrease the incidence of OHSS, especially in high-risk women and to improve early recognition of OHSS symptoms and signs with accurate categorization and prompt management aiming at improving ART health services indicators & avoiding significant physical and psychosocial morbidity that has been associated with OHSS.
This guideline aims to review the literature and provide evidence-based advice to help clinicians prevent, diagnose and manage patients with OHSS.
This guideline provides evidence-based keys to preventing OHSS by identifying the potential risk for the individual patient and planning appropriate strategies to avoid its occurrence during the ART cycle. It also covers the management of established OHSS either by conservative outpatient management in mild to moderate cases or by hospitalization / ICU admission for women with severe or critical OHSS.
- The target audience
The guideline is intended primarily for clinicians who provide assisted conception treatment, but also, for clinicians who look after affected women in emergency and gynaecology departments distinct from the ART treating clinic. Very often, women with OHSS present to clinicians who are not fertility specialists or who do not undertake assisted conception. This guideline should be applied to any setting in which OHSS would be identified, monitored, or managed.
- Methods
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
· Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
· Selecting only national and/or international guidelines
· Specific range of dates for publication (using Guidelines published or updated in 2010 and later)
· Selecting peer-reviewed publications only
· Selecting guidelines written in the English language
· Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
· Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in a table:
• Developing organization/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using the AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigour dimension was retained) This Guideline is adapted mainly from the American Society for Reproductive Medicine (ASRM) Practice Guideline February 2024 “Prevention of moderate and severe ovarian hyperstimulation syndrome”, the RCOG Green-top Guideline No. 5 February 2016 “The Management of Ovarian Hyperstimulation Syndrome” and the Canadian Society of Obstetrics & Gynaecology (SOGC) N 268 November 2017 “Diagnosis and Management of Ovarian Hyperstimulation Syndrome”(5).
- Evidence assessment
According to the WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence and develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
■ GRADE working group:https://www.gradeworkinggroup.org/
■ GRADE online training modules: http://cebgrade.mcmaster.ca/
■ GRADE profile software: http://ims.cochrane.org/revman/gradepro
For further details, Tables are included in the Annexes
o Table 1 Quality of Evidence in GRADE
o Table 2 Significance of the four levels of evidence
o Table 3 Factors that determine How to upgrade or downgrade the quality of evidence
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
- Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
- Conditional recommendations
These are made when there is greater uncertainty about the four factors above, if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
- When not to make recommendations
When there is a lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Good practice statement (GPPs)
GPPs are not an alternative to evidence-based recommendations; any evidence relating to a key question excludes the possibility of using a GPP to make a recommendation. GPPs might be used to emphasize the importance of patient participation in decision-making about specific procedures or advise caution where there is a perceived risk of harm but no available direct evidence of such harm
- RESEARCH GAPS
There is a need to compare outpatient versus inpatient management of severe OHSS regarding safety, efficacy, patient acceptability and health economic assessment.- CLINICAL INDICATORS FOR MONITORING
1. Proportion of women undergoing stimulated assisted reproduction treatment who receive comprehensive verbal and written information on OHSS symptoms and 24-hour emergency contact details.
2. Reporting severe and critical cases of OHSS admitted to hospital .
3. Notification by Emergency units to licensed fertility centres of any patient seen with a suspected diagnosis of OHSS .
4. Daily clinical review of hospitalized women with severe OHSS, including weight and abdominal girth measurements, and monitoring of fluid intake and output .
5. Prescription of low-molecular-weight heparin (LMWH) for all women with severe or critical OHSS, unless contraindicated, regardless of hospital admission status .cs
- UPDATE OF THE GUIDELINE
This guideline will be updated whenever there is new evidence
- Annexes
Table 1 Quality of Evidence in GRADE
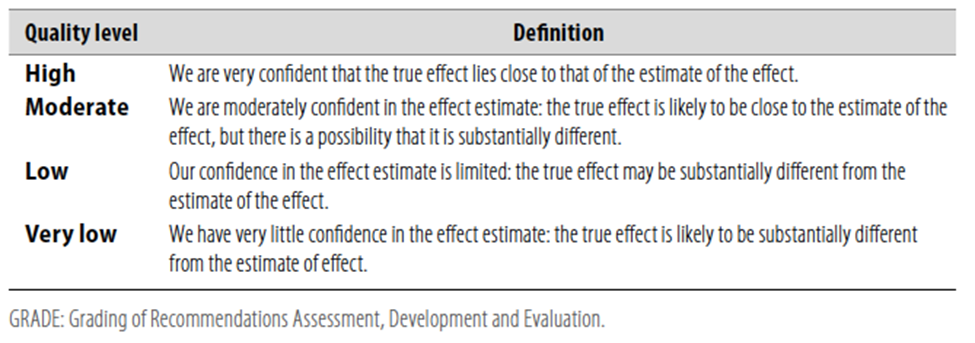
Table 2 Significance of the four levels of evidence
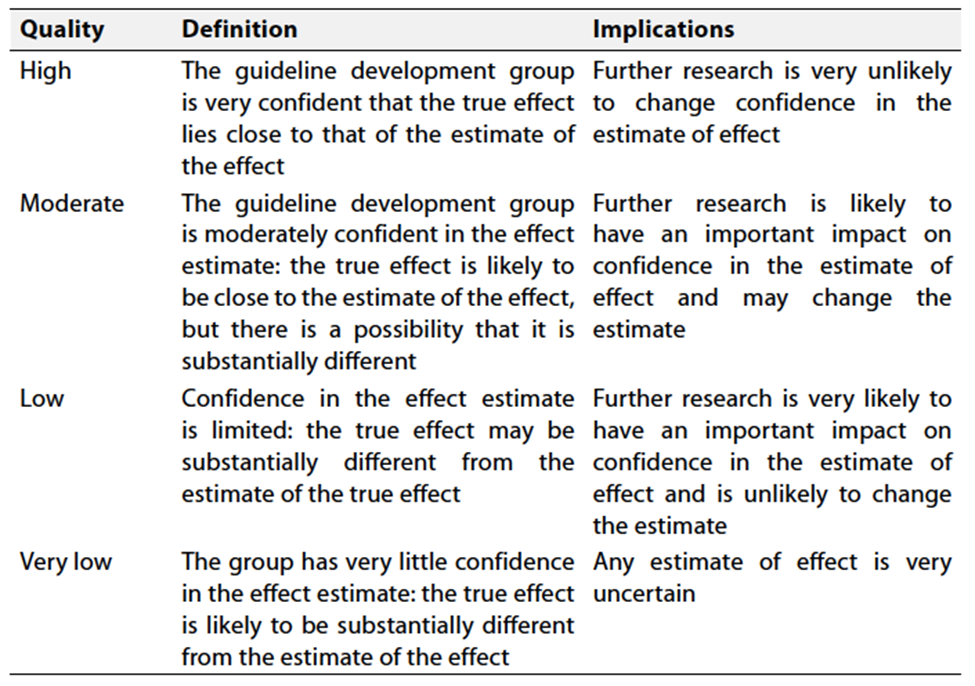
Table 3 Factors that determine How to upgrade or
downgrade the quality of evidence
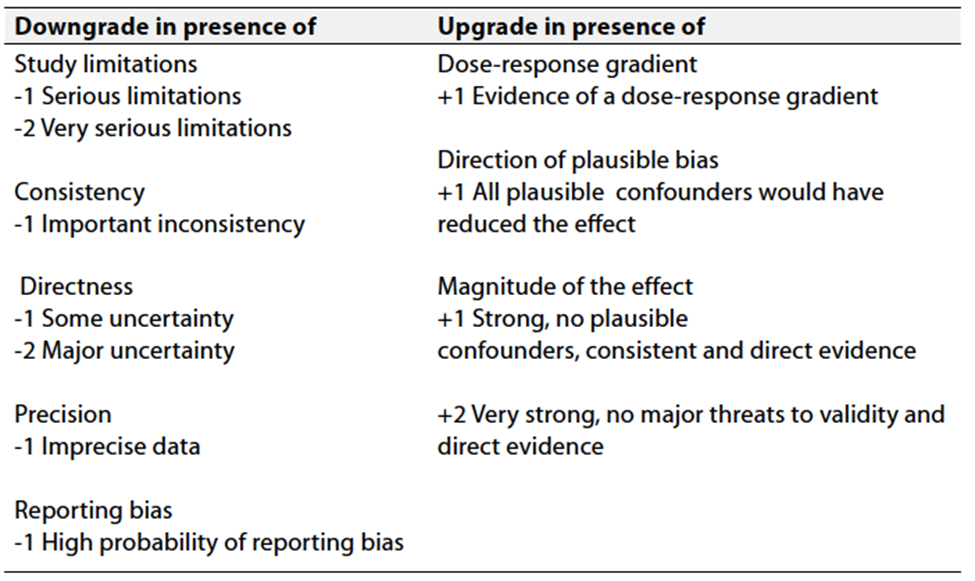
Table 4 Relevant history from a woman suspected to be suffering from OHSS adopted from Green-top Guideline No. 5, February 2016 – The Management of Ovarian Hyperstimulation Syndrome.
|
History |
|
Time of onset of symptoms relative to trigger Medication used for trigger (hCG or GnRH agonist) Number of follicles on final monitoring scan Number of eggs collected Were embryos replaced and how many? Polycystic ovary syndrome diagnosis? |
|
Symptoms |
|
Abdominal bloating Abdominal discomfort/pain, need for analgesia Nausea and vomiting Breathlessness, inability to lie flat or talk in full sentences Reduced urine output Leg swelling Vulval swelling Associated comorbidities such as thrombosis |
Table 5. Proposed RCOG classification of severity of OHSS adopted from Green-top Guideline No. 5, February 2016 - The Management of Ovarian Hyperstimulation Syndrome,
|
Category Features |
|
Mild OHSS Abdominal bloating Mild abdominal pain Ovarian size usually < 8 cma
|
|
Moderate OHSS Moderate abdominal pain Nausea ± vomiting Ultrasound evidence of ascites Ovarian size usually 8–12 cma |
|
Severe OHSS Clinical ascites (± hydrothorax) Oliguria (< 300 ml/day or < 30 ml/hour) Haematocrit > 0.45 Hyponatraemia (sodium < 135 mmol/l) Hypo-osmolality (osmolality < 282 mOsm/kg) Hyperkalaemia (potassium > 5 mmol/l) Hypoproteinaemia (serum albumin < 35 g/l) Ovarian size usually > 12 cma |
|
Critical OHSS Tense ascites/large hydrothorax Haematocrit > 0.55 White cell count > 25 000/ml Oliguria/anuria Thromboembolism Acute respiratory distress syndrome |
|
|
Ovarian size may not correlate with the severity of OHSS in cases of assisted reproduction because of the effect of follicular aspiration. Women demonstrating any feature of severe or critical OHSS should be classified in that category.
Table 6. Examination and investigation of women with suspected OHSS adopted from Green-top Guideline No. 5, February 2016 - The Management of Ovarian Hyperstimulation Syndrome,
|
Examination |
|
General: assess for dehydration, oedema (pedal, vulval and sacral); record heart rate, respiratory rate, blood pressure, body weight Abdominal: assess for ascites, palpable mass, peritonism; measure girth Respiratory: assess for pleural effusion, pneumonia, pulmonary oedema |
|
Investigations |
|
Full blood count Haematocrit (haemoconcentration) C-reactive protein (severity) Urea and electrolytes (hyponatraemia and hyperkalaemia) Serum osmolality (hypo-osmolality) Liver function tests (elevated enzymes and reduced albumin) Coagulation profile (elevated fibrinogen and reduced antithrombin) hCG (to determine outcome of treatment cycle) if appropriate Ultrasound scan: ovarian size, pelvic and abdominal free fluid. Consider ovarian Doppler if torsion suspected |
|
Other tests that may be indicated |
|
Arterial blood gases D-dimers Electrocardiogram (ECG)/echocardiogram Chest X-ray Computerised tomography pulmonary angiogram (CTPA) or ventilation/perfusion (V/Q) scan |
- References
1. Golan A, et al. Ovarian hyperstimulation syndrome: an update. Fertil Steril. 1989.
- Delvigne A, Rozenberg S. Epidemiology and prevention of ovarian hyperstimulation syndrome (OHSS): a review. Hum Reprod Update. 2002.
- Mathur R, et al. Review of the evidence base strategies to prevent ovarian hyperstimulation syndrome. Hum Fertil (Camb). 2007.
- Rizk B, Aboulghar M. Modern management of ovarian hyperstimulation syndrome. Hum Reprod. 1991.
- Soares SR, et al. High ovarian response to controlled ovarian stimulation: how to manage and prevent ovarian hyperstimulation syndrome. Reprod Biomed Online. 2008.
- Aboulghar MA, Mansour RT. Ovarian hyperstimulation syndrome: classifications and critical analysis of preventive measures. Hum Reprod Update. 2003.
- Practice Committee of the American Society for Reproductive Medicine. Ovarian hyperstimulation syndrome. Fertil Steril. 2016.
- RCOG Green-top Guideline No. 5. The management of ovarian hyperstimulation syndrome. 2016.
- Lee TH, et al. Risk factors for ovarian hyperstimulation syndrome in Korean women: implications for individualized prevention. J Korean Med Sci. 2010.
- Papanikolaou EG, et al. An ovarian prediction index to prevent ovarian hyperstimulation syndrome. Reprod Biomed Online. 2006.
- Tarlatzis BC, et al. Ovulation induction and ovarian hyperstimulation syndrome. Baillieres Clin Obstet Gynaecol. 2000.
- D'Angelo A, Amso NN. OHSS: pathophysiology and prediction. Hum Fertil (Camb). 2002.
- Nastri CO, et al. Low dose human chorionic gonadotropin in lieu of luteinizing hormone activity supplementation in IVF cycles: a systematic review and meta-analysis. Hum Reprod Update. 2012.
- Mathur R, et al. Pregnancy following severe ovarian hyperstimulation syndrome resulting in successful twin live birth: case report. Hum Reprod. 1997.
- The Rotterdam ESHRE/ASRM-sponsored PCOS consensus workshop group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum Reprod. 2004.
- Al-Inany HG, et al. Gonadotrophin-releasing hormone antagonists for assisted reproductive technology. Cochrane Database Syst Rev. 2016.
- Oudshoorn SC, et al. Individualized versus standard follicle-stimulating hormone dosing in women starting assisted reproductive technology: an open-label, multicentre, randomized, controlled, non-inferiority trial. Lancet. 2017.
- Tarlatzis BC, et al. Comparison of clomiphene citrate and letrozole for induction of ovulation in women with polycystic ovary syndrome. Fertil Steril. 2007.
- Humaidan P, et al. GnRH agonist trigger: efficacy and safety. Reprod Biomed Online. 2014.
- Rienzi L, et al. Luteal support in IVF cycles: the role of progesterone supplementation. Hum Reprod Update. 2011.
- Broer SL, et al. The use of dopamine agonists in the prevention of OHSS: a systematic review. Reprod Biomed Online. 2012.
- Gook DA, et al. The role of the freeze-all strategy in preventing OHSS: a systematic review. Fertil Steril. 2016.
- Shapiro BS, et al. Coasting strategies and their efficacy in reducing the risk of OHSS. J Assist Reprod Genet. 2006.
- Cakmak H, et al. The use of lower-dose hCG trigger in IVF cycles: effects on OHSS risk. Fertil Steril. 2012.
- Aydin N, et al. Letrozole in the prevention of OHSS: evidence from clinical studies. Hum Reprod. 2008.
- Melica G, et al. Efficacy of luteal GnRH antagonists in reducing OHSS risk. J Assist Reprod Genet. 2008.
- Koster MPH, et al. Aspirin as a preventive measure for OHSS: a systematic review. Fertil Steril. 2015.
- Biliński J, et al. Metformin and OHSS: an updated review. J Assist Reprod Genet. 2010.
- Strowitzki T, et al. Effectiveness of mifepristone and other adjunctive medications in preventing OHSS: a meta-analysis. Hum Reprod Update. 2005.
- Schweder M, et al. The use of volume expanders in OHSS prevention: a systematic review. Reprod Biomed Online. 2013.
- Serour G. Clinical manifestations of ovarian hyperstimulation syndrome. Ovarian hyperstimulation syndrome book. Jan Gerris et al., Chapter 4 25:40. 2006
- National Institute for Health and Care Excellence (NICE) Clinical Guideline [CG156]: Fertility problems: assessment and treatment, 2013.
- The Practice Committee of the American Society for Reproductive Medicine, "Ovarian hyperstimulation syndrome," Fertility and Sterility, 2016.
- Prevention of Venous Thromboembolism: British Committee for Standards in Haematology Guidelines, 2010.
- RCOG Green-top Guideline No. 37a, "Reducing the Risk of Venous Thromboembolism during Pregnancy and the Puerperium," 2015.
- Critical Care Obstetrics and Gynecology, "Acute Care and Management of Ovarian Hyperstimulation Syndrome," 2019.
- Society for Assisted Reproductive Technology (SART) and ASRM, "Management of Ovarian Hyperstimulation Syndrome," 2014.
- ACOG Practice Bulletin No. 131, "Management of Ovarian Hyperstimulation Syndrome," 2015.
- ACOG Practice Bulletin, "Nausea and vomiting of pregnancy," 2018.
- Ferraretti AP, et al. "Management of OHSS and guidelines for prevention: International consensus," Fertility and Sterility, 2011.
- The American Society of Anesthesiologists (ASA) Guidelines, 2018.
- Royal College of Obstetricians and Gynaecologists (RCOG) Green-top Guideline No. 37a, 2015.
- Lockwood CJ, et al. "Venous thromboembolism in pregnancy." New England Journal of Medicine, 2017.
- American College of Obstetricians and Gynecologists (ACOG) Practice Bulletin No. 200: "Management of Ovarian Hyperstimulation Syndrome," 2018.
- ASRM Practice Committee, "Ovarian Hyperstimulation Syndrome: Review and Guideline," Fertility and Sterility, 2016.