Prevention of Central Line associated Bloodstream Infections
| Site: | EHC | Egyptian Health Council |
| Course: | Infection Prevention and Control Guidelines |
| Book: | Prevention of Central Line associated Bloodstream Infections |
| Printed by: | Guest user |
| Date: | Sunday, 17 May 2026, 3:40 AM |
Description
"last update: 23 July 2025" Download Guideline
- Executive Summary
Invasive medical devices are a common cause of healthcare associated infections as they provide a route for infectious microorganisms to enter the body. The need for appropriate processes and policies in all health care facilities that ensure proper insertion, use, management, and removal of invasive medical devices is therefore paramount
Aseptic insertion and maintenance of devices is critical in all healthcare settings to reducing risk of infection. Policies involved in the delivery of patients care, are assigned to reduce the risk of infection, during insertion and maintenance of invasive devices. As the procedures are more invasive, the risk of infection is greater.
|
Recommendations |
|
1. Peripheral Catheters and Midline Catheters |
|
In adults, use an upper-extremity site for catheter insertion. Replace a catheter inserted in a lower extremity site to an upper extremity site as soon as possible. (Good Practice Statement) |
|
In pediatric patients, the upper or lower extremities or the scalp (in neonates or young infants) can be used as the catheter insertion site. (Good Practice Statement) |
|
Select catheters on the basis of the intended purpose and duration of use, known infectious and non-infectious complications (e.g., phlebitis and infiltration). (Strong Recommendation) |
|
Avoid the use of steel needles for the administration of fluids and medications that might cause tissue necrosis if extravasation occurs. (Strong Recommendation) |
|
Use a midline catheter or peripherally inserted central catheter (PICC), instead of a short peripheral catheter, when the duration of IV therapy will likely exceed six days. (Good Practice Statement) |
|
Evaluate the catheter insertion site daily by palpation through the dressing to recognze tenderness and by inspection if a transparent dressing is in use. (Good Practice Statement) |
|
Remove peripheral venous catheters if there are signs of phlebitis (warmth, tenderness, erythema or palpable venous cord), infection, or a malfunctioning catheter. (Strong Recommendation) |
|
2. Central Venous Catheters |
|
Weigh the risks and benefits of placing a central venous device at a recommended site to reduce infectious complications against the risk for mechanical complications (e.g., pneumothorax, subclavian artery puncture, subclavian vein stenosis, hemothorax, thrombosis). (Strong Recommendation) |
|
Avoid using the femoral vein for central venous access in adult patients. (Strong Recommendation) |
|
If no risk, use a subclavian site, rather than a jugular or a femoral site, in adult patients to minimize infection risk for non-tunneled CVC placement. (Strong Recommendation) |
|
Avoid the subclavian site in hemodialysis patients and patients with advanced kidney disease, to avoid subclavian vein stenosis. (Strong Recommendation) |
|
Use a fistula or graft in patients with chronic renal failure instead of a CVC for permanent access for dialysis. (Strong Recommendation) |
|
Use ultrasound guidance to place central venous catheters (if this technology is available) to reduce the number of cannulations attempts and mechanical complications. (Strong Recommendation) |
|
Use a CVC with the minimum number of ports or lumens essential for the patient management. (Strong Recommendation) |
|
Promptly remove any intravascular catheter that is no longer essential. (Strong Recommendation) |
|
When adherence to aseptic technique cannot be ensured (i.e., catheters inserted during a medical emergency), replace the catheter as soon as possible, i.e., within 48 hours. (Strong Recommendation) |
|
3. Hand Hygiene, gloves and Aseptic Technique |
|
Perform hand hygiene procedures, either by washing hands with soap and water or with alcohol-based hand rubs (ABHR). Hand hygiene should be performed before and after palpating catheter insertion sites as well as before and after inserting, replacing, accessing, repairing, or dressing an intravascular catheter. Palpation of the insertion site should not be performed after the application of antiseptic, unless aseptic technique is maintained. (Strong Recommendation) |
|
Maintain aseptic technique for the insertion and care of intravascular catheters. (Strong Recommendation) |
|
Wear clean gloves, rather than sterile gloves, for the insertion of peripheral intravascular catheters, if the access site is not touched after the application of skin antiseptics. (Weak Recommendation) |
|
Sterile gloves should be worn for the insertion of arterial, central, and midline catheters. (Strong Recommendation) |
|
Use new sterile gloves before handling the new catheter when guidewire exchanges are performed. (Good Practice Statement) |
|
Wear either clean or sterile gloves when changing the dressing on intravascular catheters. (Weak Recommendation) |
|
Use maximal sterile barrier precautions, including the use of a cap, mask, sterile gown, sterile gloves, and a sterile full body drape, for the insertion of CVCs, PICCs, or guidewire exchange. (Strong Recommendation) |
|
4. Skin Preparation |
|
Prepare clean skin with an antiseptic (70% alcohol, tincture of iodine, or alcoholic chlorhexidine gluconate solution) before peripheral venous catheter insertion. (Strong Recommendation) |
|
Prepare clean skin with a >0.5% chlorhexidine preparation with alcohol before central venous catheter and peripheral arterial catheter insertion and during dressing changes. If there is a contraindication to chlorhexidine, tincture of iodine, an iodophor, or 70% alcohol can be used as alternatives. (Strong Recommendation) |
|
Antiseptics should be allowed to dry according to the manufacturer’s recommendation prior to placing the catheter. (Strong Recommendation) |
|
5. Catheter Site Dressing Regimens |
|
Use either sterile gauze or sterile, transparent, semipermeable dressing to cover the catheter site. (Strong Recommendation) |
|
Replace catheter site dressing if the dressing becomes damp, loosened, or visibly soiled. (Strong Recommendation) |
|
Do not use topical antibiotic ointment or creams on insertion sites, except for dialysis catheters, because of their potential to promote fungal infections and antimicrobial resistance. (Strong Recommendation) |
|
Do not submerge the catheter or catheter site in water. Showering should be permitted if precautions can be taken to reduce the likelihood of introducing organisms into the catheter (e.g., if the catheter and connecting device are protected with an impermeable cover during the shower). (Strong Recommendation) |
|
Replace dressings used on short-term CVC sites every 2 days for gauze dressings. (Good Practice Statement) |
|
Replace dressings used on short-term CVC sites at least every 7 days for transparent dressings, except in those pediatric patients in which the risk for dislodging the catheter may outweigh the benefit of changing the dressing. (Strong Recommendation) |
|
Monitor the catheter sites visually when changing the dressing or by palpation through an intact dressing on a regular basis, depending on the clinical situation of the individual patient. (Strong Recommendation) |
|
Encourage patients to report any changes in their catheter site or any new discomfort to their provider. (Good Practice Statement) |
|
6. Antibiotic/Antiseptic Prophylaxis |
|
Do not administer systemic antimicrobial prophylaxis routinely before insertion or during use of an intravascular catheter to prevent catheter colonization or CRBSI. (Strong Recommendation) |
|
Use a 2% chlorhexidine wash for daily skin cleansing to reduce CRBSI. (Good Practice Statement) |
|
Use povidone iodine antiseptic ointment or bacitracin/gramicidin/ polymyxin B ointment at the hemodialysis catheter exit site after catheter insertion and at the end of each dialysis session only if this ointment does not interact with the material of the hemodialysis catheter per manufacturer’s recommendation. (Strong Recommendation) |
|
7. Replacement of Peripheral and Midline Catheters |
|
There is no need to replace peripheral catheters more frequently than every 72-96 hours to reduce risk of infection and phlebitis in adults. (Strong Recommendation) |
|
Replace peripheral catheters in children only when clinically indicated. (Strong Recommendation) |
|
Replace midline catheters only when there is a specific indication. (Good Practice Statement) |
|
8. Replacement of CVCs, Including PICCs and Haemodialysis Catheters |
|
Do not routinely replace CVCs, PICCs, hemodialysis catheters, or pulmonary artery catheters to prevent catheter-related infections. (Strong Recommendation) |
|
Do not remove CVCs or PICCs on the basis of fever alone. Use clinical judgment regarding the appropriateness of removing the catheter if infection is evidenced elsewhere or if a noninfectious cause of fever is suspected. (Good Practice Statement) |
|
Do not use guidewire exchanges routinely for non-tunneled catheters to prevent infection. (Strong Recommendation) |
|
Do not use guidewire exchanges to replace a non-tunneled catheter suspected of infection. (Strong Recommendation) |
|
Use a guidewire exchange to replace a malfunctioning non-tunneled catheter if no evidence of infection is present. (Strong Recommendation) |
|
Use new sterile gloves before handling the new catheter when guidewire exchanges are performed. (Good Practice Statement) |
|
9. Umbilical Catheters |
|
Remove and do not replace umbilical artery catheters if any signs of CRBSI, vascular insufficiency in the lower extremities, or thrombosis are present. (Good Practice Statement) |
|
Remove and do not replace umbilical venous catheters if any signs of CRBSI or thrombosis are present. (Good Practice Statement) |
|
Cleanse the umbilical insertion site with an antiseptic before catheter insertion. Avoid tincture of iodine because of the potential effect on the neonatal thyroid. Other iodine-containing products (e.g., povidone iodine) can be used. (Strong Recommendation) |
|
Do not use topical antibiotic ointment or creams on umbilical catheter insertion sites because of the potential to promote fungal infections and antimicrobial resistance. (Strong Recommendation) |
|
Add low-doses of heparin (0.25—1.0 U/ml) to the fluid infused through umbilical arterial catheters (Strong Recommendation) |
|
Remove umbilical catheters as soon as possible when no longer needed or when any sign of vascular insufficiency to the lower extremities is observed. Optimally, umbilical artery catheters should not be left in place >5 days. (Good Practice Statement) |
|
Umbilical venous catheters should be removed as soon as possible when no longer needed but can be used up to 14 days if managed aseptically. (Good Practice Statement) |
|
An umbilical catheter may be replaced if it is malfunctioning, and there is no other indication for catheter removal, and the total duration of catheterization has not exceeded 5 days for an umbilical artery catheter or 14 days for an umbilical vein catheter. (Good Practice Statement) |
|
10. Peripheral Arterial Catheters and Pressure Monitoring Devices for Adult and Paediatric Patients |
|
In adults, use of the radial, brachial or dorsalis pedis sites is preferred over the femoral or axillary sites of insertion to reduce the risk of infection. (Strong Recommendation) |
|
In children, the brachial site should not be used. The radial, dorsalis pedis, and posterior tibial sites are preferred over the femoral or axillary sites of insertion. (Good Practice Statement) |
|
A minimum of a cap, mask, sterile gloves and a small sterile fenestrated drape should be used during peripheral arterial catheter insertion. (Strong Recommendation) |
|
During axillary or femoral artery catheter insertion, maximal sterile barriers precautions should be used. (Good Practice Statement) |
|
Replace arterial catheters only when there is a clinical indication. (Good Practice Statement) |
|
Remove the arterial catheter as soon as it is no longer needed. (Good Practice Statement) |
|
Use disposable, rather than reusable, transducer assemblies when possible. (Strong Recommendation) |
|
Do not routinely replace arterial catheters to prevent catheter-related infections. (Good Practice Statement) |
|
Replace disposable or reusable transducers at 96-hour intervals. Replace other components of the system (including the tubing, continuous-flush device, and flush solution) at the time the transducer is replaced. (Strong Recommendation) |
|
Keep all components of the pressure monitoring system (including calibration devices and flush solution) sterile. (Strong Recommendation) |
|
Minimize the number of manipulations of and entries into the pressure monitoring system. Use a closed flush system (i.e., continuous flush), rather than an open system (i.e., one that requires a syringe and stopcock), to maintain the patency of the pressure monitoring catheters. (Good Practice Statement) |
|
When the pressure monitoring system is accessed through a diaphragm, rather than a stopcock, scrub the diaphragm with an appropriate antiseptic before accessing the system. (Strong Recommendation) |
|
Do not administer dextrose-containing solutions or parenteral nutrition fluids through the pressure monitoring circuit. (Strong Recommendation) |
|
Sterilize reusable transducers according to the manufacturers’ instructions if the use of disposable transducers is not feasible. (Strong Recommendation) |
|
11. Replacement of Administration Sets |
|
In patients not receiving blood, blood products or fat emulsions, replace administration sets that are including secondary sets and add-on devices, no more frequently than at 96-hour intervals, but at least every 7 days. (Strong Recommendation) |
|
Replace tubing used to administer blood, blood products, or fat emulsions (those combined with amino acids and glucose in a 3-in-1 admixture or infused separately) within 24 hours of initiating the infusion. (Strong Recommendation) |
|
Replace tubing used to administer propofol infusions every 6 or 12 hours, when the vial is changed or per the manufacturer’s recommendation. (Strong Recommendation) |
|
12. Needleless Intravascular Catheter Systems |
|
Change needleless connectors no more frequently than every 72 hours or according to manufacturers’ recommendations for the purpose of reducing infection rates. (Good Practice Statement) |
|
Minimize contamination risk by scrubbing the access port with an appropriate antiseptic (chlorhexidine, povidone iodine, an iodophor, or 70% alcohol) and accessing the port only with sterile devices. (Strong Recommendation) |
|
Use a needleless system to access IV tubing. (Weak Recommendation) |
|
When needleless systems are used, a split septum valve may be preferred over some mechanical valves due to increased risk of infection with the mechanical valves. (Good Practice Statement) |
|
13. Performance Improvement |
|
Use collaborative-based performance improvement initiatives in which multifaceted strategies are “bundled” together to improve compliance with evidence-based recommended practices. (Strong Recommendation) |
- Recommendations
|
Recommendations |
|
1. Peripheral Catheters and Midline Catheters |
|
In adults, use an upper-extremity site for catheter insertion. Replace a catheter inserted in a lower extremity site to an upper extremity site as soon as possible. (Good Practice Statement) |
|
In pediatric patients, the upper or lower extremities or the scalp (in neonates or young infants) can be used as the catheter insertion site. (Good Practice Statement) |
|
Select catheters on the basis of the intended purpose and duration of use, known infectious and non-infectious complications (e.g., phlebitis and infiltration). (Strong Recommendation, Moderate Grade Evidence) |
|
Avoid the use of steel needles for the administration of fluids and medications that might cause tissue necrosis if extravasation occurs. (Strong Recommendation, High Grade Evidence) |
|
Use a midline catheter or peripherally inserted central catheter (PICC), instead of a short peripheral catheter, when the duration of IV therapy will likely exceed six days. (Good Practice Statement) |
|
Evaluate the catheter insertion site daily by palpation through the dressing to recognize tenderness and by inspection if a transparent dressing is in use. (Good Practice Statement) |
|
Remove peripheral venous catheters if there are signs of phlebitis (warmth, tenderness, erythema or palpable venous cord), infection, or a malfunctioning catheter. (Strong Recommendation, Moderate Grade Evidence) |
|
2. Central Venous Catheters |
|
Weigh the risks and benefits of placing a central venous device at a recommended site to reduce infectious complications against the risk for mechanical complications (e.g., pneumothorax, subclavian artery puncture, subclavian vein stenosis, hemothorax, thrombosis). (Strong Recommendation, High Grade Evidence) |
|
Avoid using the femoral vein for central venous access in adult patients. (Strong Recommendation, High Grade Evidence) |
|
If no risk, use a subclavian site, rather than a jugular or a femoral site, in adult patients to minimize infection risk for non-tunneled CVC placement. (Strong Recommendation, Moderate Grade Evidence) |
|
Avoid the subclavian site in hemodialysis patients and patients with advanced kidney disease, to avoid subclavian vein stenosis. (Strong Recommendation, High Grade Evidence) |
|
Use a fistula or graft in patients with chronic renal failure instead of a CVC for permanent access for dialysis. (Strong Recommendation, High Grade Evidence) |
|
Use ultrasound guidance to place central venous catheters (if this technology is available) to reduce the number of cannulation attempts and mechanical complications. (Strong Recommendation, Moderate Grade Evidence) |
|
Use a CVC with the minimum number of ports or lumens essential for the patient management. (Strong Recommendation, Moderate Grade Evidence) |
|
Promptly remove any intravascular catheter that is no longer essential. (Strong Recommendation, High Grade Evidence) |
|
When adherence to aseptic technique cannot be ensured (i.e., catheters inserted during a medical emergency), replace the catheter as soon as possible, i.e., within 48 hours. (Strong Recommendation, Moderate Grade Evidence) |
|
3. Hand Hygiene, gloves and Aseptic Technique |
|
Perform hand hygiene procedures, either by washing hands with soap and water or with alcohol-based hand rubs (ABHR). Hand hygiene should be performed before and after palpating catheter insertion sites as well as before and after inserting, replacing, accessing, repairing, or dressing an intravascular catheter. Palpation of the insertion site should not be performed after the application of antiseptic, unless aseptic technique is maintained. (Strong Recommendation, Moderate Grade Evidence) |
|
Maintain aseptic technique for the insertion and care of intravascular catheters. (Strong Recommendation, Moderate Grade Evidence) |
|
Wear clean gloves, rather than sterile gloves, for the insertion of peripheral intravascular catheters, if the access site is not touched after the application of skin antiseptics. (Weak Recommendation, Low Grade Evidence) |
|
Sterile gloves should be worn for the insertion of arterial, central, and midline catheters. (Strong Recommendation, High Grade Evidence) |
|
Use new sterile gloves before handling the new catheter when guidewire exchanges are performed. (Good Practice Statement) |
|
Wear either clean or sterile gloves when changing the dressing on intravascular catheters. (Weak Recommendation, Low Grade Evidence) |
|
Use maximal sterile barrier precautions, including the use of a cap, mask, sterile gown, sterile gloves, and a sterile full body drape, for the insertion of CVCs, PICCs, or guidewire exchange. (Strong Recommendation, Moderate Grade Evidence) |
|
4. Skin Preparation |
|
Prepare clean skin with an antiseptic (70% alcohol, tincture of iodine, or alcoholic chlorhexidine gluconate solution) before peripheral venous catheter insertion. (Strong Recommendation, Moderate Grade Evidence) |
|
Prepare clean skin with a >0.5% chlorhexidine preparation with alcohol before central venous catheter and peripheral arterial catheter insertion and during dressing changes. If there is a contraindication to chlorhexidine, tincture of iodine, an iodophor, or 70% alcohol can be used as alternatives. (Strong Recommendation, High Grade Evidence) |
|
Antiseptics should be allowed to dry according to the manufacturer’s recommendation prior to placing the catheter. (Strong Recommendation, Moderate Grade Evidence) |
|
5. Catheter Site Dressing Regimens |
|
Use either sterile gauze or sterile, transparent, semipermeable dressing to cover the catheter site. (Strong Recommendation, High Grade Evidence) |
|
Replace catheter site dressing if the dressing becomes damp, loosened, or visibly soiled. (Strong Recommendation, Moderate Grade Evidence) |
|
Do not use topical antibiotic ointment or creams on insertion sites, except for dialysis catheters, because of their potential to promote fungal infections and antimicrobial resistance. (Strong Recommendation, Moderate Grade Evidence) |
|
Do not submerge the catheter or catheter site in water. Showering should be permitted if precautions can be taken to reduce the likelihood of introducing organisms into the catheter (e.g., if the catheter and connecting device are protected with an impermeable cover during the shower). (Strong Recommendation, Moderate Grade Evidence) |
|
Replace dressings used on short-term CVC sites every 2 days for gauze dressings. (Good Practice Statement) |
|
Replace dressings used on short-term CVC sites at least every 7 days for transparent dressings, except in those pediatric patients in which the risk for dislodging the catheter may outweigh the benefit of changing the dressing. (Strong Recommendation, Moderate Grade Evidence) |
|
Monitor the catheter sites visually when changing the dressing or by palpation through an intact dressing on a regular basis, depending on the clinical situation of the individual patient. (Strong Recommendation, Moderate Grade Evidence) |
|
Encourage patients to report any changes in their catheter site or any new discomfort to their provider. (Good Practice Statement) |
|
6. Antibiotic/Antiseptic Prophylaxis |
|
Do not administer systemic antimicrobial prophylaxis routinely before insertion or during use of an intravascular catheter to prevent catheter colonization or CRBSI. (Strong Recommendation, Moderate Grade Evidence) |
|
Use a 2% chlorhexidine wash for daily skin cleansing to reduce CRBSI. (Good Practice Statement) |
|
Use povidone iodine antiseptic ointment or bacitracin/gramicidin/ polymyxin B ointment at the hemodialysis catheter exit site after catheter insertion and at the end of each dialysis session only if this ointment does not interact with the material of the hemodialysis catheter per manufacturer’s recommendation. (Strong Recommendation, Moderate Grade Evidence) |
|
7. Replacement of Peripheral and Midline Catheters |
|
There is no need to replace peripheral catheters more frequently than every 72-96 hours to reduce risk of infection and phlebitis in adults. (Strong Recommendation, Moderate Grade Evidence) |
|
Replace peripheral catheters in children only when clinically indicated. (Strong Recommendation, Moderate Grade Evidence) |
|
Replace midline catheters only when there is a specific indication. (Good Practice Statement) |
|
8. Replacement of CVCs, Including PICCs and Hemodialysis Catheters |
|
Do not routinely replace CVCs, PICCs, hemodialysis catheters, or pulmonary artery catheters to prevent catheter-related infections. (Strong Recommendation, Moderate Grade Evidence) |
|
Do not remove CVCs or PICCs on the basis of fever alone. Use clinical judgment regarding the appropriateness of removing the catheter if infection is evidenced elsewhere or if a noninfectious cause of fever is suspected. (Good Practice Statement) |
|
Do not use guidewire exchanges routinely for non-tunneled catheters to prevent infection. (Strong Recommendation, Moderate Grade Evidence) |
|
Do not use guidewire exchanges to replace a non-tunneled catheter suspected of infection. (Strong Recommendation, Moderate Grade Evidence) |
|
Use a guidewire exchange to replace a malfunctioning non-tunneled catheter if no evidence of infection is present. (Strong Recommendation, Moderate Grade Evidence) |
|
Use new sterile gloves before handling the new catheter when guidewire exchanges are performed. (Good Practice Statement) |
|
9. Umbilical Catheters |
|
Remove and do not replace umbilical artery catheters if any signs of CRBSI, vascular insufficiency in the lower extremities, or thrombosis are present. (Good Practice Statement) |
|
Remove and do not replace umbilical venous catheters if any signs of CRBSI or thrombosis are present. (Good Practice Statement) |
|
Cleanse the umbilical insertion site with an antiseptic before catheter insertion. Avoid tincture of iodine because of the potential effect on the neonatal thyroid. Other iodine-containing products (e.g., povidone iodine) can be used. (Strong Recommendation, Moderate Grade Evidence) |
|
Do not use topical antibiotic ointment or creams on umbilical catheter insertion sites because of the potential to promote fungal infections and antimicrobial resistance. (Strong Recommendation, High Grade Evidence) |
|
Add low-doses of heparin (0.25—1.0 U/ml) to the fluid infused through umbilical arterial catheters (Strong Recommendation, Moderate Grade Evidence) |
|
Remove umbilical catheters as soon as possible when no longer needed or when any sign of vascular insufficiency to the lower extremities is observed. Optimally, umbilical artery catheters should not be left in place >5 days. (Good Practice Statement) |
|
Umbilical venous catheters should be removed as soon as possible when no longer needed but can be used up to 14 days if managed aseptically. (Good Practice Statement) |
|
An umbilical catheter may be replaced if it is malfunctioning, and there is no other indication for catheter removal, and the total duration of catheterization has not exceeded 5 days for an umbilical artery catheter or 14 days for an umbilical vein catheter. (Good Practice Statement) |
|
10. Peripheral Arterial Catheters and Pressure Monitoring Devices for Adult and Paediatric Patients |
|
In adults, use of the radial, brachial or dorsalis pedis sites is preferred over the femoral or axillary sites of insertion to reduce the risk of infection. (Strong Recommendation, Moderate Grade Evidence) |
|
In children, the brachial site should not be used. The radial, dorsalis pedis, and posterior tibial sites are preferred over the femoral or axillary sites of insertion. (Good Practice Statement) |
|
A minimum of a cap, mask, sterile gloves and a small sterile fenestrated drape should be used during peripheral arterial catheter insertion. (Strong Recommendation, Moderate Grade Evidence) |
|
During axillary or femoral artery catheter insertion, maximal sterile barriers precautions should be used. (Good Practice Statement) |
|
Replace arterial catheters only when there is a clinical indication. (Good Practice Statement) |
|
Remove the arterial catheter as soon as it is no longer needed. (Good Practice Statement) |
|
Use disposable, rather than reusable, transducer assemblies when possible. (Strong Recommendation, Moderate Grade Evidence) |
|
Do not routinely replace arterial catheters to prevent catheter-related infections. (Good Practice Statement) |
|
Replace disposable or reusable transducers at 96-hour intervals. Replace other components of the system (including the tubing, continuous-flush device, and flush solution) at the time the transducer is replaced. (Strong Recommendation, Moderate Grade Evidence) |
|
Keep all components of the pressure monitoring system (including calibration devices and flush solution) sterile. (Strong Recommendation, High Grade Evidence) |
|
Minimize the number of manipulations of and entries into the pressure monitoring system. Use a closed flush system (i.e., continuous flush), rather than an open system (i.e., one that requires a syringe and stopcock), to maintain the patency of the pressure monitoring catheters. (Good Practice Statement) |
|
When the pressure monitoring system is accessed through a diaphragm, rather than a stopcock, scrub the diaphragm with an appropriate antiseptic before accessing the system. (Strong Recommendation, High Grade Evidence) |
|
Do not administer dextrose-containing solutions or parenteral nutrition fluids through the pressure monitoring circuit. (Strong Recommendation, High Grade Evidence) |
|
Sterilize reusable transducers according to the manufacturers’ instructions if the use of disposable transducers is not feasible. (Strong Recommendation, High Grade Evidence) |
|
11. Replacement of Administration Sets |
|
In patients not receiving blood, blood products or fat emulsions, replace administration sets that are including secondary sets and add-on devices, no more frequently than at 96-hour intervals, but at least every 7 days. (Strong Recommendation, High Grade Evidence) |
|
Replace tubing used to administer blood, blood products, or fat emulsions (those combined with amino acids and glucose in a 3-in-1 admixture or infused separately) within 24 hours of initiating the infusion. (Strong Recommendation, Moderate Grade Evidence) |
|
Replace tubing used to administer propofol infusions every 6 or 12 hours, when the vial is changed or per the manufacturer’s recommendation. (Strong Recommendation, High Grade Evidence) |
|
12. Needleless Intravascular Catheter Systems |
|
Change needleless connectors no more frequently than every 72 hours or according to manufacturers’ recommendations for the purpose of reducing infection rates. (Good Practice Statement) |
|
Minimize contamination risk by scrubbing the access port with an appropriate antiseptic (chlorhexidine, povidone iodine, an iodophor, or 70% alcohol) and accessing the port only with sterile devices. (Strong Recommendation, High Grade Evidence) |
|
Use a needleless system to access IV tubing. (Weak Recommendation, Low Grade Evidence) |
|
When needleless systems are used, a split septum valve may be preferred over some mechanical valves due to increased risk of infection with the mechanical valves. (Good Practice Statement) |
|
13. Performance Improvement |
|
Use collaborative-based performance improvement initiatives in which multifaceted strategies are “bundled” together to improve compliance with evidence-based recommended practices. (Strong Recommendation, Moderate Grade Evidence) |
Rationale
Central line–associated bloodstream infection is associated with significant morbidity and mortality. Implementation of simple evidence-based bundles (hand hygiene, full-barrier precautions during insertion, chlorhexidine to clean the insertion site, avoiding the femoral veins when possible, and removing unnecessary central lines) resulted in a significant, sustained infection reduction.
Selection of Catheters and Sites
Central venous catheters (CVCs) or central venous access devices (CVADs), often described by healthcare professionals as “central lines,” refer to a broad category of invasive devices used to administer fluids, medications, blood and blood products, and parenteral nutrition. Unlike the short, temporary catheters inserted into the peripheral vasculature, these central devices access major vessels that are most often located in the neck or adjacent to the heart. There are four general categories of CVADs: non-tunneled (e.g., access via subclavian or internal jugular veins); peripherally inserted central catheters (PICCs) inserted via peripheral veins (such as the cephalic or basilic veins); subcutaneously tunneled; and implanted vascular access ports.
The site at which a catheter is placed influences the subsequent risk for catheter-related infection and phlebitis. The influence of site on the risk for catheter infections is related in part to the risk for thrombophlebitis and density of local skin flora.
The density of skin flora at the catheter insertion site is a major risk factor for CRBSI. No single trial has satisfactorily compared infection rates for catheters placed in jugular, subclavian, and femoral veins. Femoral catheters should also be avoided, when possible, because they are associated with a higher risk for deep venous thrombosis than are internal jugular or subclavian catheters.
Hand Hygiene and Aseptic Technique
Hand hygiene before catheter insertion or maintenance, combined with proper aseptic technique during catheter manipulation, provides protection against infection .
Proper hand hygiene can be achieved through the use of either an alcohol-based product or with soap and water with adequate rinsing. Appropriate aseptic technique does not necessarily require sterile gloves for insertion of peripheral catheters; a new pair of disposable nonsterile gloves can be used in conjunction with a "no-touch" technique for the insertion of peripheral venous catheters. Sterile gloves must be worn for placement of central catheters since a "no-touch" technique is not possible.
Maximal Sterile Barrier Precautions
Maximum sterile barrier precautions are defined as wearing a sterile gown, sterile gloves, and cap and using a full body drape (like the drapes used in the operating room) during the placement of CVC lowered risk of infection
Skin Preparation
Skin preparation with chlorhexidine-containing cutaneous antiseptic regimen , shown lower rates of catheter colonization or CRBSI . Chlorhexidine has become a standard antiseptic for skin preparation for the insertion of both central and peripheral venous catheters, 5% povidone iodine solution in 70% ethanol was associated with a substantial reduction of CVC-related colonization and infection compared with 10% aqueous povidone iodine.
Catheter Site Dressing Regimens
Transparent, semi-permeable polyurethane dressings permit continuous visual inspection of the catheter site and require less frequent changes than do standard gauze and tape dressings. . Transparent dressings can be safely left on peripheral venous catheters for the duration of catheter insertion without increasing the risk for thrombophlebitis .
The choice of dressing can be a matter of preference. If blood is oozing from the catheter insertion site, gauze dressing is preferred.
Patient Cleansing
Daily cleansing of ICU patients with a 2% chlorhexidine impregnated wash cloth may be a simple, effective strategy to decrease the rate of primary BSIs.
Catheter Securement Devices
Catheter stabilization is recognized as an intervention to decrease the risk for phlebitis, catheter migration and dislodgement, and may be advantageous in preventing CRBSIs. Pathogenesis of CRBSI occurs via migration of skin flora through the percutaneous entry site. Sutureless securement devices avoid disruption around the catheter entry site and may decrease the degree of bacterial colonization. Using a sutureless securement device also mitigates the risk of sharps injury to the healthcare provider from inadvertent needlestick injury.
Antimicrobial/Antiseptic Impregnated Catheters and Cuffs
Certain catheters and cuffs that are coated or impregnated with antimicrobial or antiseptic agents can decrease the risk for CRBSI and potentially decrease hospital costs associated with treating CRBSIs, despite the additional acquisition cost of an antimicrobial/antiseptic impregnated .
Antibiotic/Antiseptic Ointments
A variety of topical antibiotic or antiseptic ointments have been utilized in attempts to lower the antimicrobial burden at the catheter insertion site and thus prevent infection.
Nasal carriers of S. aureus are more likely to experience a CRBSI than non-colonized persons.
Antibiotic Lock Prophylaxis, Antimicrobial Catheter Flush and Catheter Lock Prophylaxis
To prevent CRBSI, a wide variety of antibiotic and antiseptic solutions have been used to flush or lock catheter lumens . Catheter lock is a technique by which an antimicrobial solution is used to fill a catheter lumen and then allowed to dwell for a period of time while the catheter is idle. Antibiotics of various concentrations that have been used either alone (when directed at a specific organism) or in combination (to achieve broad empiric coverage) to prophylactically flush or lock central venous catheters include vancomycin, gentamicin, ciprofloxacin, minocycline, amikacin, cefazolin, cefotaxime, and ceftazidime; while antiseptics have included alcohol, taurolidine, trisodium citrate. These agents are usually combined with a compound acting as an anticoagulant, such as heparin or Ethylenediaminetetraacetic acid (EDTA). Although most studies indicate a beneficial effect of the antimicrobial flush or lock solution in terms of prevention of catheter-related infection, this must be balanced by the potential for side effects, toxicity, allergic reactions, or emergence of resistance associated with the antimicrobial agent.
Replacement of Peripheral and Midline Catheters
Scheduled replacement of intravascular catheters has been proposed as a method to prevent phlebitis and catheter-related infections. Because phlebitis and catheter colonization have been associated with an increased risk for catheter-related infection, short peripheral catheter sites commonly are replaced at 72–96-hour intervals to reduce both the risk for infection and patient discomfort associated with phlebitis.
Replacement of CVCs, Including PICCs and Hemodialysis Catheters
Catheter replacement at scheduled time intervals as a method to reduce CRBSI has not lowered rates. Scheduled guidewire exchange of CVCs is another proposed strategy for preventing CRBSI. Catheter replacement over a guidewire has become an accepted technique for replacing a malfunctioning catheter Because of the increased difficulty obtaining vascular access in children, attention should be given to the frequency with which catheters are replaced in these patients.
Umbilical Catheters
Although the umbilical stump becomes heavily colonized soon after birth, umbilical vessel catheterization often is used for vascular access in newborn infants. Umbilical vessels can be cannulated easily and permit both collection of blood samples and measurement of hemodynamic status. The incidences of catheter colonization and BSI are similar for umbilical vein catheters and umbilical artery catheters.
Risk factors for infection differ for umbilical artery and umbilical vein catheters.
Peripheral Arterial Catheters and Pressure Monitoring Devices for Adult and Pediatric Patients
Arterial catheters are usually inserted into the radial or femoral artery and permit continuous blood pressure monitoring and blood gas measurements. The risk of CRBSI for arterial catheters is lower than that associated with non-coated, uncuffed, non-tunneled short-term CVCs. Use of full barrier precautions during arterial cannulation does not appear to reduce the risk of arterial CRBSI .
Replacement of Administration Sets
Recent studies suggest that administration sets may be used safely for up to 7 days if used in conjunction with antiseptic catheters When a fluid that enhances microbial growth is infused (e.g., fat emulsions and blood products), more frequent changes of administration sets are indicated as these products have been identified as independent risk factors for CRBSI.
Performance Improvement
Clinical decision makers, healthcare payers, and patient safety advocates emphasize the importance of translating research findings into everyday practice. Rigorous evaluations of CRBSI preventive practices using study designs with high internal validity and including study populations that optimize external validity remain necessary.
Once practices have been determined to be effective and economically efficient, the next step is to implement these evidence-based practices, so they become part of routine clinical care.
- Acknowledgements
We would like to acknowledge the Infection Control Guidelines Committee for developing these guidelines.
Head of IPC Guidelines Committee
Professor Ghada Ismail (Professor of Clinical Pathology (Clinical Microbiology), Faculty of Medicine, Ain Shams University, Secretary of Supreme IPC Committee, SCUH, Member of WHO Global Guidelines Groups (GDG) for Infection Prevention)
Secretary of IPC Guidelines Committee
Professor Walaa Abd El-Latif (Professor of Medical Microbiology and Immunology, Faculty of Medicine Ain Shams University, IPC Consultant)
Members of the Committee
▪ Professor Amal Sayed (Deputy Manager of Environmental Affair, Infection Control Director, Cairo University Hospitals)
▪ Professor Amani El-Kholy (Clinical Pathology Department (Microbiology), Faculty of Medicine, Cairo University, Infection Control Consultant)
▪ Dr Asmaa Mohamed Abdelfatah Mohamed (Lecturer, Faculty of Nursing MTI University)
▪ Dr Gehan Mohamed Fahmy (Professor clinical microbiology ASUSH consultant infection control, Board member of IFIC EMERO region)
▪ Professor Hebatallah Gamal Rashed (Clinical Pathology Department (Microbiology), Faculty of Medicine, Assuit University, Infection Control Consultant)
▪ Dr Iman Afifi (Consultant Clinical Pathology (Microbiology) and IPC, Ain Shams University, Director IPC units of Ain Shams internal medicine and Geriatric hospitals
▪ Professor Maha El Touny (Department of internal medicine. Faculty of Medicine, Ain Shams University. Infection Control Consultant)
▪ Brigadier Dr. Mohamed Bakr Al-Attar (Assistant Commander of Military Fever Hospital for Therapeutic Affairs, Tropical Medicine Consultant)
▪ Professor Nagwa Khamis (Emeritus Consultant Clinical Pathology (Microbiology) and IPC, ASU Director IPC Department and CEO Consultant IPC, CCHE-57357)
▪ Professor Nesrine Fathi Hanafi (Professor in Medical Microbiology and Immunology Faculty of Medicine Alexandria University, Head of Infection Prevention and Control, Alexandria University Hospitals)
▪ Dr. Reham Lotfy Abdel Aziz (Environmental Health Director, EEAA, Hazardous Waste Consultant, WMRA, Ministry of Environment)
▪ Professor Sherin ElMasry (Professor of Clinical Pathology, Ain Shams University, Chief Director of IPC ASU, Health Care Quality & Patient Safety Consultant)
▪ Dr Shimaa El-Garf: Clinical Pathology Specialist, Coordinator of HAI Surveillance and Audit Electronic System for University Hospitals, RLEUH- SCUH
- List of Abbreviations
· ABHR: Alcohol-based Hand Rubs
· APIC: Association for Professionals in Infection Control and
Epidemiology
· BSI: Blood Stream Infection
· CDC: Centers for Disease Control and Prevention
· CLABSI: Center-Line Associated Blood Stream Infection
· CRBSI: Catheter-related Blood Stream Infection
· CVAD: Central Venous Access Devices
· CVC: Central Venous Catheter
· DUR: Device Utilization Ratio
· EDTA: Ethylenediaminetetraacetic acid
· HAIs: Healthcare Associated Infections
· ICU: Intensive Care Units
· IPC: Infection Prevention and Control
· IV: Intravenous
· NHSN: National Healthcare Safety Network
· PICC: Peripherally Inserted Central Catheter
· WHO: World Health Organization
- Glossary
1.1.1
Access: The
performance of any of the following activities during the current inpatient
admission:
o
Line placement
o
Use of (entering the line with a needle or needleless device) any
central line for:
o Infusion
o Withdrawal of blood
o Use for hemodynamic monitoring
1.1.2 Catheter-related blood stream infection (CRBSI): Clinical definition of BSI used for diagnosis and
treatment of infection that requires definitive laboratory evidence that the
central venous catheter is the source of an individual patient’s BSI.
1.1.3 Central line: An
intravascular access device or catheter (now commonly referred to as a central
venous access device), which terminates at or close to the heart or in one of
the great vessels. The line may be used for infusion or hemodynamic monitoring.
A central line may be inserted centrally or peripherally in the patient; it
terminates in one of the great vessels or in or near the heart.
1.1.4 Central Line associated bloodstream infection (CLABSI): refers to surveillance definitions applied to
populations at risk. Therefore, the CDC’s National Healthcare Safety Network
(NHSN) definition is “a CLABSI is a primary BSI in a patient that had a central
line for more than two consecutive calendar days (on or after CL Day 3),
following the first access of the central line before the development of the
BSI and is not related to an infection at another site. However, because some
BSIs are secondary to other sources other than the central line (e.g.,
pancreatitis, mucositis) that may not be easily recognized, the surveillance
definition may overestimate the true incidence of CRBSI.
1.1.5 Extravasation:
Inadvertent leakage of a vesicant solution into surrounding tissue.
1.1.6 Infiltration:
Inadvertent leakage of a non-vesicant solution into surrounding tissue
1.1.7 Infusion: The
administration of any solution through the lumen of a catheter into a blood
vessel. Infusions include continuous infusion (for example, nutritional fluids
or medications), intermittent infusion (for example, IV flush), IV
antimicrobial administration, and blood transfusion or hemodialysis treatment.
1.1.8 Phlebitis: An
inflammation of the wall of a vein, characterized by pain, erythema, swelling
and palpable thrombosis of the cannulated vein.
- Introduction
A central line bundle is a collection of evidence-based strategies that, when implemented together, significantly reduce the risk of Central Line-Associated Bloodstream Infections (CLABSIs). CLABSIs are serious healthcare-associated infections that can lead to increased morbidity, mortality, prolonged hospital stays, and higher healthcare costs.
The introduction of central line bundle guidelines aims to:
● Reduce the incidence of CLABSIs: By providing a structured approach to central line insertion and maintenance, the bundle minimizes the opportunity for pathogens to enter the bloodstream.
● Standardize best practices: The guidelines outline specific, evidence-based interventions that should be consistently applied by all healthcare professionals involved in central line care. This reduces variability in practice and promotes a high standard of care.
● Improve patient safety: Preventing CLABSIs directly contributes to safer patient care, reducing the likelihood of severe complications such as sepsis and death.
● Provide a framework for education and training: The bundle serves as a basis for educating healthcare personnel on the proper techniques for central line insertion, maintenance, and early removal.
● Facilitate quality improvement efforts: The guidelines provide measurable elements that can be monitored to assess compliance and the effectiveness of implemented strategies, driving continuous improvement in central line care.
A typical central line insertion bundle includes the following key components:
1. Hand Hygiene: Performing thorough hand hygiene immediately before catheter insertion.
2. Maximal Barrier Precautions: Utilizing sterile gloves, gown, mask, cap, and a full-body sterile drape during the insertion procedure.
3. Skin Antisepsis: Cleaning the insertion site with an appropriate antiseptic solution, such as chlorhexidine with alcohol.
4. Optimal Site Selection: Avoiding the femoral vein in adult patients whenever possible due to a higher risk of infection. The subclavian vein is often preferred.
5. Daily Review of Line Necessity: Assessing the ongoing need for the central line each day and promptly removing it when no longer indicated.
In addition to the insertion bundle, maintenance bundles address the care of the central line after placement and typically include:
● Hand hygiene before accessing the line.
● Scrubbing the access port with an appropriate antiseptic before each use.
● Proper dressing changes at recommended intervals using aseptic technique.
● Replacing administration sets and tubing at recommended intervals.
- Scope and Purpose
The scope of a central line bundle guideline typically encompasses all healthcare settings where central venous catheters are inserted and maintained, including:
● Intensive Care Units (ICUs)
● Medical-Surgical Units
● Emergency Departments
● Operating Rooms
● Outpatient Infusion Centers
● Hemodialysis Units
The purpose of a central line bundle guideline is to:
● Significantly reduce the incidence of Central Line-Associated Bloodstream Infections: This is the primary goal, as CLABSIs are a major cause of preventable morbidity and mortality in hospitalized patients.
● Establish and promote evidence-based best practices: The guidelines synthesize the most current research and recommendations into a practical framework for clinical practice.
● Standardize care processes: By outlining specific steps for insertion and maintenance, the bundle aims to ensure consistent and high-quality care across different providers and settings.
● Improve patient safety and outcomes: Reducing CLABSIs directly translates to fewer complications, shorter hospital stays, decreased healthcare costs, and improved patient survival.
● Provide a clear and actionable framework for healthcare professionals: The bundle offers practical guidance that can be readily implemented in daily practice.
● Serve as a foundation for education and training: The guidelines provide essential information for educating new and experienced healthcare professionals on the proper management of central lines.
● Facilitate performance measurement and quality improvement initiatives: The elements of the bundle can be used as metrics to monitor adherence to best practices and to drive continuous improvement in central line care.
● Foster a culture of safety: By emphasizing the importance of meticulous technique and consistent adherence to protocols, the bundle contributes to a healthcare environment where infection prevention is a top priority.
- Target Audience
● Infection
Prevention and Control Team
● All
Healthcare Workers: Including Clinicians, Nurses, Head Nurses
● Healthcare
associated infections (HAIs) Surveillance Officers
● The
Treating Physician
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
● Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
● Selecting only national and/or international guidelines
● Specific range of dates for publication (using Guidelines published or updated in 2013 and later)
● Selecting peer reviewed publications only
● Selecting guidelines written in English language
● Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
● Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
The following characteristics of the retrieved guidelines were summarized in:
● Developing organization/authors
● Date of publication, posting, and release
● Country/language of publication
● Date of posting and/or release
● Dates of the search used by the source guideline developers.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The committee decided to adapt from:
● Centers of Disease Control (2024). Guidelines for the Prevention of Intravascular Catheter-Related Infections (2011) update February 28, 2024
● World Health Organization (2024). Guidelines for the prevention of bloodstream infections and other infections associated with the use of intravascular catheters, Part I: peripheral catheters
● National Health and Medical Research Council (2019) Australian Guidelines for the Prevention and Control of Infection in Healthcare. Canberra: Commonwealth of Australia.
Evidence assessment
According to the World Health Organization (WHO) Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
● GRADE working group: https://www.gradeworkinggroup.org/
● GRADE online training modules: http://cebgrade.mcmaster.ca/
Table (1) Quality and Significance of the four levels of evidence in GRADE
|
Quality |
Definition |
Implications |
|
High |
The guideline development group is very confident that the true effect lies close to that of the estimate of the effect |
Further research is very unlikely to change confidence in the estimate effect |
|
Moderate |
The guideline development group is moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibly that it is substantially different |
Further research is likely to have an important impact on confidence in the estimate of the effect and may change the estimate |
|
Low |
Confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the true effect |
Further research is very likely to have an important on confidence in the estimate of effect and is unlikely to change the estimate |
|
Very low |
The group has very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of the effect |
Any estimate of the effect is very uncertain |
Table (2) Factors that determine How to upgrade or downgrade the quality of evidence.
|
Downgrade in presence of |
Upgrade in presence of |
|
Study limitations. 1- Serious limitations 2- Very serious limitations |
Dose- response gradient. +1 Evidence of a dose-response gradient |
|
Consistency 1- Important inconsistency |
Direction of plausible bias + All plausible confounders would have reduced the effect |
|
Directness 1- Some uncertainty 2- Major uncertainty |
Magnitude of the effect +1 Strong, no plausible Confounder, consistent and direct evidence |
|
Precision 1- Imprecise data |
+2 very strong, no major threats to validity and direct evidence |
|
Reporting bias 1. High probability of reporting bias |
|
The strength of the recommendations
The strength of a recommendation communicates the importance of adherence to the recommendation.
● Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
● Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations?
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Indicators for Monitoring
To ensure the appropriate prevention of catheter associated blood stream infection in hospitals and reduce the risk of CLABSI, specific indicators should be monitored regularly. These are some indicators which can provide measurable data to assess compliance, identify areas for improvement, and guide interventions. Here are some key indicators that can be included in hospital guidelines for prevention of CLABSI.
a) CLABSI Prevention Process Measures
1. Documentation of daily assessment regarding patient’s need for continuing CVC access
Calculate the % of patients who received daily assessment for continuing need for CVC access
● Numerator: Number of CVC insertions with documentation of daily assessment
● Denominator: number of patients with CVC
● Standardization factor: 100 (i.e., multiply by 100 so that measure is expressed as a percentage)
b) Outcome Measures:
1. Rates of CLABSI (Number of CLABSI per 1000 device-days)
Measurement of rates allows an individual facility to gauge the longitudinal impact of implementation of prevention strategies:
● Numerator: Number of central line-associated bloodstream infections (BSIs) identified for the patient care unit(s) under surveillance
● Denominator: Number of central line-days for the patient care units under surveillance
● Standardization factor: Multiply by 1000 so that the measure is expressed as cases per 1000 device-days.
2. Device Utilization Ratio (DUR): The catheter utilization ratio is the number of central line-days per number of patient-days in a given period. This is a measure of the total patient-days in which a central line was used and can be used as a marker for risk of infection.
● Numerator: total of central line-days for the location(s) under surveillance for the specified time period.
● Denominator: the total number of days that patients are in the location during the selected time period.
According to the approved CDC criteria,
*Device days are the total number of days of exposure to urinary catheter for all the patients during the selected time period.
*Patient days are the total number of days patients are in the ICU during the selected time period
1.1 Plan to Update this National Clinical Guideline
This guideline will be reviewed and updated when new evidence emerges that is likely to influence the recommendations.
- Annex
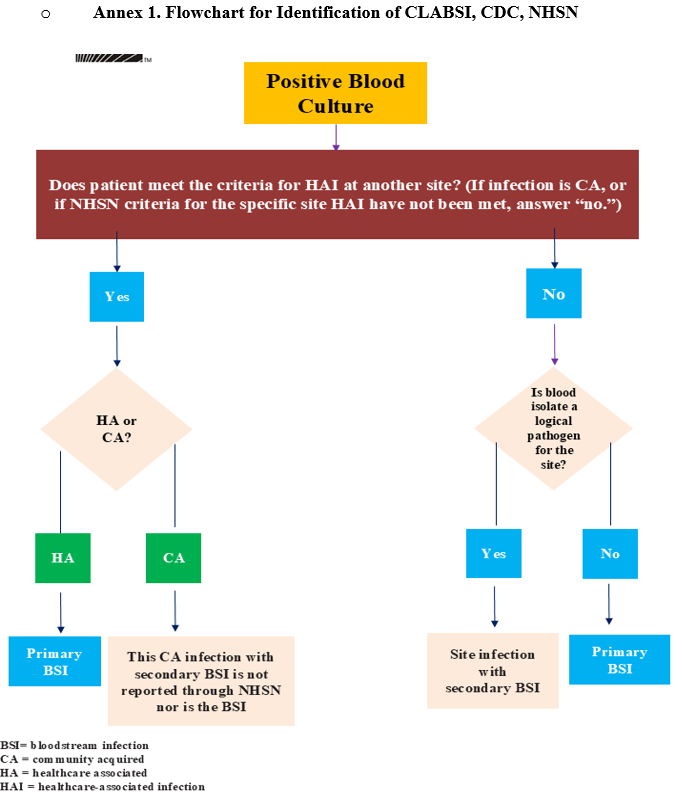
Annex 2. Differentiating CLABSI and CRBSI (APIC 2014)
|
Criteria |
CLABSI |
CRBSI |
|
Purpose of definition |
Surveillance |
Clinical diagnosis |
|
Device removal required |
Usually no |
Usually yes |
|
Cultures |
Qualitative blood cultures |
Blood cultures with differential time to positivity |
|
Catheter tip culture recommended |
No |
Yes |
|
Major advantage |
Convenience, lower cost, readily available in most laboratories |
High sensitivity; better positive predictive value |
|
Major disadvantage |
Often unable to distinguish a primary and secondary BSI; may overstate the true incidence of primary CLABSI |
More complex, less convenient, expensive |
Annex 3. Many IV devices are available to
deliver medications, fluids and nutrition into patients' bodies, and monitor
vital signs. Each carries its own benefits and risks for different patients.
Credit: University of Michigan
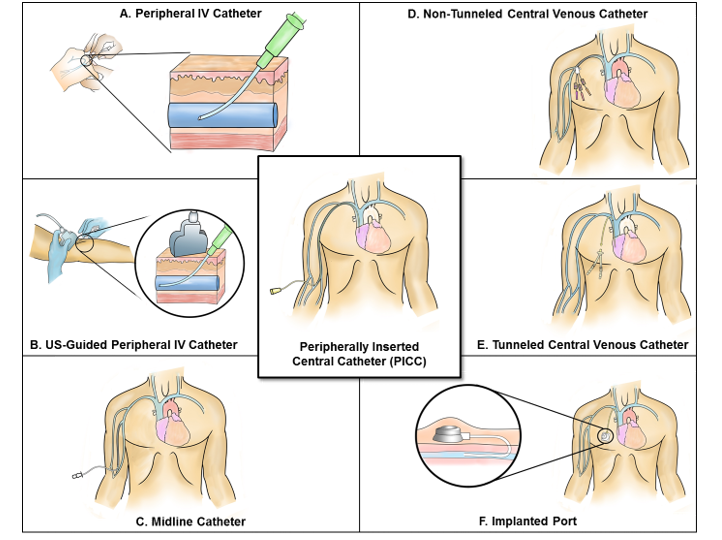
Examples of central lines: Tunnelled and non-tunnelled central venous catheters, implanted ports, pulmonary artery catheters, dialysis or haemofiltration catheters in a great vessel and peripherally inserted central catheters. An introducer is considered a central line if the tip is situated in a great vessel.
Examples of devices that are not considered to be central lines for the purpose of CLABSI surveillance: Arterial catheters, arteriovenous fistulas, arteriovenous grafts, atrial catheters (also known as transthoracic intra-cardiac catheters, those catheters inserted directly into the right or left atrium via the heart wall), extracorporeal membrane oxygenation (ECMO), femoral arterial catheters, intra-aortic balloon pump (IABP) devices, haemodialysis reliable outflow (HeRO) dialysis catheters, peripheral intravenous cannulae or midline catheters, and ventricular assist devices (VAD). Pacemaker wires and other solid or non‑lumen devices inserted into central blood vessels, or the heart are not considered central lines, because fluids are not infused, pushed, or withdrawn through such devices.
- References
1- Centers for Disease Control and Prevention (CDC). Patient Safety Component Manual. National Healthcare Safety Network (NHSN); 2025.
2- Centers for Disease Control and Prevention (CDC). Guidelines for the Prevention of Intravascular Catheter-Related Infections (2011). Updated February 28, 2024.
3- World Health Organization (WHO). Guidelines for the Prevention of Bloodstream Infections and Other Infections Associated with the Use of Intravascular Catheters, Part I: Peripheral Catheters. Geneva: WHO; 2024.
4- Buetti N, Marschall J, Drees M, Fakih MG, Hadaway L, Mermel LA, et al. Strategies to prevent central line-associated bloodstream infections in acute-care hospitals: 2022 update. Infect Control Hosp Epidemiol. 2022.
5- National Health and Medical Research Council (NHMRC). Australian Guidelines for the Prevention and Control of Infection in Healthcare. Canberra: Commonwealth of Australia; 2019.
6- Association for Professionals in Infection Control and Epidemiology (APIC). Guide to Preventing Central Line-Associated Bloodstream Infections. 2015.
7- Chopra V, Flanders SA, Saint S, Woller SC, O'Grady NP, Safdar N, et al. The Michigan Appropriateness Guide for Intravenous Catheters (MAGIC): Results from a multispecialty panel using the RAND/UCLA appropriateness method. Ann Intern Med. 2015.