Prevention of Catheter associated Urinary Tract Infections
| Site: | EHC | Egyptian Health Council |
| Course: | Infection Prevention and Control Guidelines |
| Book: | Prevention of Catheter associated Urinary Tract Infections |
| Printed by: | Guest user |
| Date: | Sunday, 17 May 2026, 3:37 AM |
Description
"last update: 23 July 2025" Download Guideline
- Executive Summary
Invasive medical devices are a common source of healthcare associated infections (HAIs) and provide a route for infectious agents to enter the body. Pneumonia, urinary tract infections and bloodstream infection account for most of intensive care unit HAIs, and most of these are associated with invasive devices.
Urinary tract infections are the most common type of healthcare-associated infections (HAIs) reported by acute care hospitals. Healthcare-associated UTIs are caused by instrumentation of the urinary tract. Catheter-associated urinary tract infection (CAUTI) has been associated with increased morbidity, mortality, hospital cost, and length of stay. Despite the measurement challenges, CAUTI is among those HAIs targeted for significant improvement, based on evidence showing that more than 50 percent of these infections are preventable.
The following guidelines provide best-practice guidance on strategies for the selection, insertion, maintenance and removal of urinary catheters.
|
Recommendations |
|
1. Appropriate Urinary Catheter Use |
|
Insert catheters only for appropriate indications and leave in place only as long as needed. (Strong Recommendation) |
|
Minimize urinary catheter use and duration of use in all patients, particularly those at higher risk for CAUTI or mortality from catheterization such as women, the elderly, and patients with impaired immunity. (Strong Recommendation) |
|
Avoid use of urinary catheters in patients and nursing home residents for management of incontinence. (Strong Recommendation) |
|
Use urinary catheters in operative patients only as necessary, rather than routinely. (Strong Recommendation) |
|
For operative patients who have an indication for an indwelling catheter, remove the catheter as soon as possible postoperatively, preferably within 24 hours, unless there are appropriate indications for continued use. (Strong Recommendation) |
|
2. Consider using alternatives to indwelling urethral catheterization in selected patients when appropriate. |
|
Consider using external catheters as an alternative to indwelling urethral catheters in cooperative male patients without urinary retention or bladder outlet obstruction. (Good Practice Statement) |
|
Consider alternatives to chronic indwelling catheters, such as intermittent catheterization, in spinal cord injury patients. (Good Practice Statement) |
|
Intermittent catheterization is preferable to indwelling urethral or suprapubic catheters in patients with bladder emptying dysfunction. (Good Practice Statement) |
|
Consider intermittent catheterization in children with myelomeningocele and neurogenic bladder to reduce the risk of urinary tract deterioration. (Good Practice Statement) |
|
3. Proper Techniques for Urinary Catheter Insertion |
|
Perform hand hygiene immediately before and after insertion or any manipulation of the catheter device or site. (Strong Recommendation) |
|
Ensure that only properly trained healthcare worker who know the correct technique of aseptic catheter insertion and maintenance are given this responsibility. (Strong Recommendation) |
|
Insert urinary catheters using aseptic technique and sterile equipment. (Strong Recommendation) |
|
Use sterile gloves, drape, sponges, an appropriate antiseptic or sterile solution for periurethral cleaning, and a single-use packet of lubricant gel for insertion. (Strong Recommendation) |
|
Routine use of antiseptic lubricants is not necessary. (Good Practice Statement) |
|
Properly secure indwelling catheters after insertion to prevent movement and urethral traction. (Strong Recommendation) |
|
Unless otherwise clinically indicated, consider using the smallest bore catheter possible, consistent with good drainage, to minimize bladder neck and urethral trauma. (Good Practice Statement) |
|
4. Proper Techniques for Urinary Catheter Maintenance |
|
Following aseptic insertion of the urinary catheter, maintain a closed drainage system. (Strong Recommendation) |
|
If breaks in aseptic technique, disconnection, or leakage occur, replace the catheter and collecting system using aseptic technique and sterile equipment. (Strong Recommendation) |
|
Consider using urinary catheter systems with preconnected, sealed catheter-tubing junctions. (Good Practice Statement) |
|
Maintain unobstructed urine flow. (Strong Recommendation) |
|
Keep the catheter and collecting tube free from kinking. (Strong Recommendation) |
|
Keep the collecting bag below the level of the bladder at all times. Do not rest the bag on the floor. (Strong Recommendation) |
|
Empty the collecting bag regularly using a separate, clean collecting container for each patient; avoid splashing, and prevent contact of the drainage spigot with the nonsterile collecting container. (Strong Recommendation) |
|
Use Standard Precautions, including the use of gloves, during any manipulation of the catheter or collecting system. (Strong Recommendation) |
|
Changing indwelling catheters or drainage bags at routine, fixed intervals is not recommended. Rather, it is suggested to change catheters and drainage bags based on clinical indications such as infection, obstruction, or when the closed system is compromised. (Good Practice Statement) |
|
Unless clinical indications exist (e.g., in patients with bacteriuria upon catheter removal post urologic surgery), do not use systemic antimicrobials routinely to prevent CAUTI in patients requiring either short or long-term catheterization. (Strong Recommendation) |
|
Do not clean the periurethral area with antiseptics to prevent CAUTI while the catheter is in place. Routine hygiene (e.g., cleansing of the meatal surface during daily bathing or showering) is appropriate. (Strong Recommendation) |
|
Unless obstruction is anticipated (e.g., as might occur with bleeding after prostatic or bladder surgery) bladder irrigation is not recommended. (Good Practice Statement) |
|
If obstruction is anticipated, closed continuous irrigation is suggested to prevent obstruction. (Good Practice Statement) |
|
Routine irrigation of the bladder with antimicrobials is not recommended. (Good Practice Statement) |
|
Routine instillation of antiseptic or antimicrobial solutions into urinary drainage bags is not recommended. (Good Practice Statement) |
|
5. Catheter Materials |
|
Silicone might be preferable to other catheter materials to reduce the risk of encrustation in long-term catheterized patients who have frequent obstruction. (Good Practice Statement) |
|
6. Management of Obstruction |
|
If obstruction occurs and it is likely that the catheter material is contributing to obstruction, change the catheter. (Strong Recommendation) |
|
7. Specimen Collection |
|
Obtain urine samples aseptically. (Strong Recommendation) |
|
If a small volume of fresh urine is needed for examination (i.e., urine analysis or culture), aspirate the urine from the needleless sampling port with a sterile syringe/cannula adapter after cleansing the port with a disinfectant. (Strong Recommendation) |
|
Obtain large volumes of urine for special analyses (not culture) aseptically from the drainage bag. (Strong Recommendation) |
- Recommendations
|
Recommendations |
|
1. Appropriate Urinary Catheter Use |
|
Insert catheters only for appropriate indications and leave in place only as long as needed (Strong Recommendation, Moderate Grade Evidence) |
|
Minimize urinary catheter use and duration of use in all patients, particularly those at higher risk for CAUTI or mortality from catheterization such as women, the elderly, and patients with impaired immunity. (Strong Recommendation, Moderate Grade Evidence) |
|
Avoid use of urinary catheters in patients and nursing home residents for management of incontinence. (Strong Recommendation, Moderate Grade Evidence) |
|
Use urinary catheters in operative patients only as necessary, rather than routinely (Strong Recommendation, Moderate Grade Evidence) |
|
For operative patients who have an indication for an indwelling catheter, remove the catheter as soon as possible postoperatively, preferably within 24 hours, unless there are appropriate indications for continued use. (Strong Recommendation, Moderate Grade Evidence) |
|
2. Consider using alternatives to indwelling urethral catheterization in selected patients when appropriate. |
|
Consider using external catheters as an alternative to indwelling urethral catheters in cooperative male patients without urinary retention or bladder outlet obstruction. (Good Practice Statement) |
|
Consider alternatives to chronic indwelling catheters, such as intermittent catheterization, in spinal cord injury patients. (Good Practice Statement) |
|
Intermittent catheterization is preferable to indwelling urethral or suprapubic catheters in patients with bladder emptying dysfunction. (Good Practice Statement) |
|
Consider intermittent catheterization in children with myelomeningocele and neurogenic bladder to reduce the risk of urinary tract deterioration. (Good Practice Statement) |
|
3. Proper Techniques for Urinary Catheter Insertion |
|
Perform hand hygiene immediately before and after insertion or any manipulation of the catheter device or site. (Strong Recommendation, Moderate Grade Evidence) |
|
Ensure that only properly trained healthcare workers who know the correct technique of aseptic catheter insertion and maintenance are given this responsibility. (Strong Recommendation, Moderate Grade Evidence) |
|
Insert urinary catheters using aseptic technique and sterile equipment. (Strong Recommendation, Moderate Grade Evidence) |
|
Use sterile gloves, drape, sponges, an appropriate antiseptic or sterile solution for periurethral cleaning, and a single-use packet of lubricant gel for insertion. (Strong Recommendation, Moderate Grade Evidence) |
|
Routine use of antiseptic lubricants is not necessary. (Good Practice Statement) |
|
Properly secure indwelling catheters after insertion to prevent movement and urethral traction. (Strong Recommendation, Moderate Grade Evidence) |
|
Unless otherwise clinically indicated, consider using the smallest bore catheter possible, consistent with good drainage, to minimize bladder neck and urethral trauma. (Good Practice Statement) |
|
4. Proper Techniques for Urinary Catheter Maintenance |
|
Following aseptic insertion of the urinary catheter, maintain a closed drainage system. (Strong Recommendation, Moderate Grade Evidence) |
|
If breaks in aseptic technique, disconnection, or leakage occur, replace the catheter and collecting system using aseptic technique and sterile equipment. (Strong Recommendation, Moderate Grade Evidence) |
|
Consider using urinary catheter systems with preconnected, sealed catheter-tubing junctions. (Good Practice Statement) |
|
Maintain unobstructed urine flow. (Strong Recommendation, Moderate Grade Evidence) |
|
Keep the catheter and collecting tube free from kinking. (Strong Recommendation, Moderate Grade Evidence) |
|
Keep the collecting bag below the level of the bladder at all times. Do not rest the bag on the floor. (Strong Recommendation, Moderate Grade Evidence) |
|
Empty the collecting bag regularly using a separate, clean collecting container for each patient; avoid splashing, and prevent contact of the drainage spigot with the nonsterile collecting container. (Strong Recommendation, Moderate Grade Evidence) |
|
Use Standard Precautions, including the use of gloves, during any manipulation of the catheter or collecting system. (Strong Recommendation, Moderate Grade Evidence) |
|
Changing indwelling catheters or drainage bags at routine, fixed intervals is not recommended. Rather, it is suggested to change catheters and drainage bags based on clinical indications such as infection, obstruction, or when the closed system is compromised. (Good Practice Statement) |
|
Unless clinical indications exist (e.g., in patients with bacteriuria upon catheter removal post urologic surgery), do not use systemic antimicrobials routinely to prevent CAUTI in patients requiring either short or long-term catheterization. (Strong Recommendation, Moderate Grade Evidence) |
|
Do not clean the periurethral area with antiseptics to prevent CAUTI while the catheter is in place. Routine hygiene (e.g., cleansing of the meatal surface during daily bathing or showering) is appropriate. (Strong Recommendation, Moderate Grade Evidence) |
|
Unless obstruction is anticipated (e.g., as might occur with bleeding after prostatic or bladder surgery) bladder irrigation is not recommended. (Good Practice Statement) |
|
If obstruction is anticipated, closed continuous irrigation is suggested to prevent obstruction. (Good Practice Statement) |
|
Routine irrigation of the bladder with antimicrobials is not recommended. (Good Practice Statement) |
|
Routine instillation of antiseptic or antimicrobial solutions into urinary drainage bags is not recommended. (Good Practice Statement) |
|
5. Catheter Materials |
|
Silicone might be preferable to other catheter materials to reduce the risk of encrustation in long-term catheterized patients who have frequent obstruction. (Good Practice Statement) |
|
6. Management of Obstruction |
|
If obstruction occurs and it is likely that the catheter material is contributing to obstruction, change the catheter. (Strong Recommendation, Moderate Grade Evidence Grade) |
|
7. Specimen Collection |
|
Obtain urine samples aseptically. (Strong Recommendation, Moderate Grade Evidence) |
|
If a small volume of fresh urine is needed for examination (i.e., urine analysis or culture), aspirate the urine from the needleless sampling port with a sterile syringe/cannula adapter after cleansing the port with a disinfectant. (Strong Recommendation, Moderate Grade Evidence) |
|
Obtain large volumes of urine for special analyses (not culture) aseptically from the drainage bag. (Strong Recommendation, Moderate Grade Evidence) |
Rationale
Burden of outcomes associated with CAUTI
1. Urinary tract infections are one of the most common healthcare-associated infections.
2. Urinary catheters remain one of the most common medical devices experienced by adults in emergency departments and hospitals worldwide. Often, these devices are placed and maintained in use without an appropriate clinical indication to justify the risk compared to the benefit. Of patients who have a urinary catheter placed in the hospital, up to half are placed in patients who may not have an appropriate indication for a urinary catheter.
3. The daily risk of development of bacteriuria varies from 3% to 7% when an indwelling urethral catheter remains in situ.
4. The high frequency of catheter use in hospitalized patients means that the cumulative burden of CAUTI is substantial.
5. Infection is only one of several adverse outcomes of urinary catheter use. Noninfectious complications include nonbacterial urethral inflammation, urethral strictures, mechanical trauma, and mobility impairment, and these are described in this document as well.
6. CAUTI has been associated with increased mortality and length of stay, but the association with mortality may be a consequence of confounding by unmeasured clinical variables.
Risk Factors for CAUTI and Reduction Strategies
Risk factors for CAUTI include catheter duration, female anatomy, age related changes of the genitourinary tract, pregnancy, poor nutrition, fecal incontinence, illness severity, paraplegia, cerebrovascular disease, immunocompromised status, comorbid conditions resulting in neurogenic bladder, and equipment required to manage bladder voiding.
Reservoirs of transmission
1. The drainage bag of the bacteriuric patient is a reservoir for organisms that may be transmitted through the hands of healthcare personnel (HCP).
2. The drainage bag can also become contaminated by contact with hands due to inadequate hand hygiene, contact with the patient’s skin or hands, or contact with the floor or vessel used to empty the bag.
3. Outbreaks of infections associated with resistant gram-negative organisms attributable to bacteriuria in catheterized patients have been reported.
Effective methods to reduce risk include:
1. Not inserting an IUC unless strict criteria are met (e.g., neurogenic bladder, obstructive uropathy)
2. Using external urinary catheters when appropriate for the patient
3. Limiting the duration of the IUC by using facility-specific removal criteria
4. Following aseptic techniques for insertion and maintenance of IUC
5. Additional approaches to prevent CAUTI:
a. Define and monitor catheter harm in addition to CAUTI, including catheter obstruction, unintended removal, catheter trauma, or reinsertion within 24 hours of removal. Catheter harm includes infectious complications in addition to CAUTI (eg, secondary bacteremia, asymptomatic bacteriuria consequences) and noninfectious catheter complications. Patients with an indwelling urethral catheter are 5 times more likely to experience noninfectious complications (eg, urethral injury, pain, or inadvertent catheter removal) than infectious complications.
b. Establish a system for defining, analyzing, and reporting data on non–catheter-associated UTIs, particularly UTIs associated with devices used as alternatives to indwelling urethral catheters. Non–catheter-associated UTIs are defined as UTIs that occur in hospitalized patients without an indwelling urethral catheter. These include but are not limited to patients who have had no urinary device at all, as well as those with external urinary catheters, urinary stents, or urostomies, or who undergo intermittent catheterization, and thus are not captured by the National Healthcare Safety Network (NHSN) CAUTI definition.
Case Definitions
Surveillance definitions and clinical definitions are intended for different purposes and should not be used interchangeably. Infection preventionists should be able to effectively explain the differences between these three types of definitions.
1. Clinical definitions: These are intended to be used for diagnosis and treatment purposes. Diagnostic criteria for a urinary tract infection vary by care setting and patient population. Clinical diagnosis and treatment for a urinary tract infection should be rendered by a physician.
2. Surveillance definitions: These are intended to define the incidence of a condition and are useful in the measurement and monitoring of performance improvement efforts. NHSN and WHO provides standardized definitions and methodology for performing surveillance for CAUTIs through the Patient Safety Component.
Indwelling urinary devices procedures:
● Bacteria associated with infection in the setting of urinary catheterisation gain access to the urinary tract either through extraluminal contamination - from the health care worker’s hands or from the person’s own colonic or perineal flora.
● This can occur if there is a break in aseptic technique during insertion of the catheter or servicing of the drainage system.
● Intraluminal contamination can occur through reflux of bacteria from a contaminated urine drainage bag
● Education of healthcare workers – healthcare workers performing catheterization should be trained and competent in the technique and familiar with policies and procedures for insertion, maintenance and changing of indwelling urinary devices.
● Aseptic technique protects patients during catheterization by employing infection control measures that minimise, as far as practicably possible, the presence of pathogenic microorganisms.
● While the principles of aseptic technique remain constant for all clinical procedures, the level of practice will change depending upon a standard aseptic technique risk assessment.
Implementation strategies
Preventing CAUTI requires a focus on both technical and socioadaptive (or behavioral) components. Interventions to assist with program implementation and evaluation that have been reported to be associated with improved outcomes are provided. The intervention used a 2-tier system of interventions to determine each hospital’s specific needs relative to their CAUTI rates. Tier 1 describes data gathering needs and a nursing template in the medical record to prompt staff to consider removing catheters at the earliest possible point. This tier alone can create sufficient visibility within the organization to reduce CAUTI rates to the desired level. Tier 2 is a more intensive tier of steps that hospitals can implement for stubborn rates that will not come down, including root-cause analysis.
- Acknowledgements
We would like to acknowledge the Infection Control Guidelines Committee for developing these guidelines.
Head of IPC Guidelines Committee
Professor Ghada Ismail (Professor of Clinical Pathology (Clinical Microbiology), Faculty of Medicine, Ain Shams University, Secretary of Supreme IPC Committee, SCUH, Member of WHO Global Guidelines Groups (GDG) for Infection Prevention)
Secretary of IPC Guidelines Committee
Professor Walaa Abd El-Latif (Professor of Medical Microbiology and Immunology, Faculty of Medicine Ain Shams University, IPC Consultant)
Members of the Committee
▪ Professor Amal Sayed (Deputy Manager of Environmental Affair, Infection Control Director, Cairo University Hospitals)
▪ Professor Amani El-Kholy (Clinical Pathology Department (Microbiology), Faculty of Medicine, Cairo University, Infection Control Consultant)
▪ Dr Asmaa Mohamed Abdelfatah Mohamed (Lecturer, Faculty of Nursing MTI University)
▪ Dr Gehan Mohamed Fahmy (Professor clinical microbiology ASUSH consultant infection control, Board member of IFIC EMERO region)
▪ Professor Hebatallah Gamal Rashed (Clinical Pathology Department (Microbiology), Faculty of Medicine, Assuit University, Infection Control Consultant)
▪ Dr Iman Afifi (Consultant Clinical Pathology (Microbiology) and IPC, Ain Shams University, Director IPC units of Ain Shams internal medicine and Geriatric hospitals
▪ Professor Maha El Touny (Department of internal medicine. Faculty of Medicine, Ain Shams University. Infection Control Consultant)
▪ Brigadier Dr. Mohamed Bakr Al-Attar (Assistant Commander of Military Fever Hospital for Therapeutic Affairs, Tropical Medicine Consultant)
▪ Professor Nagwa Khamis (Emeritus Consultant Clinical Pathology (Microbiology) and IPC, ASU Director IPC Department and CEO Consultant IPC, CCHE-57357)
▪ Professor Nesrine Fathi Hanafi (Professor in Medical Microbiology and Immunology Faculty of Medicine Alexandria University, Head of Infection Prevention and Control, Alexandria University Hospitals)
▪ Dr. Reham Lotfy Abdel Aziz (Environmental Health Director, EEAA, Hazardous Waste Consultant, WMRA, Ministry of Environment)
▪ Professor Sherin ElMasry (Professor of Clinical Pathology, Ain Shams University, Chief Director of IPC ASU, Health Care Quality & Patient Safety Consultant)
- List of Abbreviations
· CAUTI: Catheter associated Urinary Tract Infections
· IPC: Infection Prevention and Control
· CDC: Centers for Disease Control and Prevention
· HAIs: Healthcare Associated Infections
· HCP: Healthcare Personnel
· IUC: Indwelling
urinary catheterization
· NHSN: National Healthcare Safety Network
· UTI: Urinary tract infections
· WHO: World Health Organization
- Glossary
· Catheter: A
hollow flexible tube that is inserted into a body organ.
· Catheter-associated Urinary Tract Infection (CAUTI): The presence of symptoms or signs attributable to
microorganisms that have invaded the urinary tract, where the patient has, or
has recently had a urinary catheter.
· Closed continuous bladder irrigation: The infusion
of sterile fluid into the bladder, usually using a closed triple lumen
catheter. One lumen is used to drain urine, another is used to inflate the
catheter balloon and the third is used to infuse the sterile irrigation fluid.
· Closed drainage system: The indwelling catheter is attached to a urine drainage
bag. The drainage bag can be attached aseptically at insertion, or the catheter
and drainage bag are supplied as one unit from the manufacturer
(pre-connected).
· Indwelling Urinary Catheterization (IUC): The insertion of a catheter into the bladder, that
remains in situ to allow continuous drainage of urine. Short-term: Catheter
remains in situ for ≤ 28 days. Long-term: Catheter remains in situ for > 28
days.
· Intermittent catheterization: The periodic insertion of a catheter into the bladder
and its immediate removal when the bladder is drained.
· Suprapubic catheterization: Catheter inserted into the bladder via the abdominal
wall.
- Introduction
A urinary tract infection (UTI) is an infection that involves any of the organs or structures of the urinary tract, (e.g., kidneys, ureters, bladder and urethra). Virtually all healthcare-associated UTIs are caused by instrumentation of the urinary tract. In addition, bacteriuria commonly leads to unnecessary antimicrobial use, and urinary drainage systems are often reservoirs for multidrug-resistant bacteria and a source of transmission to other patients.
The use of indwelling urinary catheters can result in the development of CAUTI. Each day that an indwelling urinary catheterization (IUC) remains in place, the risk of acquiring a CAUTI increases.
The core components of a urinary catheter bundle typically include:
1. Appropriate Catheter Use:
● Avoid unnecessary catheterization: Catheters should only be inserted when there is a clear medical indication.
● Consider alternatives: Explore options like intermittent catheterization, external catheters (condom catheters), or other methods for managing urinary incontinence or retention when appropriate.
● Prompt removal: The catheter should be removed as soon as it is no longer needed. Regular assessment of the continued need for the catheter is crucial.
2. Proper Catheter Insertion Technique:
● Hand hygiene: Perform thorough hand hygiene immediately before and after any manipulation of the catheter insertion site or device.
● Aseptic technique: Use sterile gloves, drapes, and equipment during catheter insertion.
● Skin preparation: Clean the periurethral area with an appropriate antiseptic solution before insertion.
● Appropriate catheter size: Use the smallest catheter size consistent with adequate drainage.
● Lubrication: Apply sterile lubricant gel to the catheter before insertion.
● Securement: Properly secure the catheter after insertion to prevent movement and urethral traction.
3. Maintaining the Catheter:
● Closed drainage system: Maintain a sterile, continuously closed drainage system. Avoid disconnecting the catheter and drainage tubing unless absolutely necessary.
● Unobstructed urine flow: Ensure the catheter and drainage tubing are free from kinks and that the collection bag is positioned below the level of the bladder.
● Regular emptying of the drainage bag: Empty the collection bag regularly using a clean container for each patient, avoiding contact between the drainage spigot and the container.
● Perineal care: Perform routine perineal hygiene.
● Avoid routine catheter changes: Do not change indwelling catheters or drainage bags at fixed intervals unless clinically indicated.
4. Daily Review and Prompt Removal:
● Assess the necessity of the catheter daily: Review the indication for the urinary catheter each day and document this assessment.
● Remove the catheter as soon as it is no longer indicated: Implement protocols or reminders to facilitate timely removal.
● Education and training: Ensure that healthcare personnel (HCP) involved in catheter insertion and maintenance are properly trained and competent in performing these procedures using aseptic techniques.
● Documentation: Document the indication for catheter insertion, the date and time of insertion, the type and size of the catheter, and the plan for removal.
● Surveillance: Implement surveillance programs to monitor CAUTI rates and provide feedback to healthcare teams to improve practices.
- Scope and Purpose
Scope of a urinary catheter bundle guideline typically encompasses all healthcare settings where urinary catheters are used, including:
● Acute care hospitals
● Long-term care facilities
● Outpatient clinics
● Home healthcare
The purpose of a urinary catheter bundle guideline is to:
● Reduce the incidence of Catheter-Associated Urinary Tract Infections: CAUTIs are a significant cause of healthcare-associated infections, leading to increased patient morbidity, mortality, length of stay, and healthcare costs.
● Standardize best practices: The guidelines provide a clear, evidence-based framework for urinary catheter management. This ensures consistent and high-quality care across different settings and providers.
● Improve patient outcomes: By preventing CAUTIs, the guidelines contribute to safer and more effective patient care.
● Provide guidance for appropriate catheter use: Emphasizing the importance of avoiding unnecessary catheterization and promoting the use of alternatives when appropriate.
● Ensure proper insertion and maintenance techniques: Detailing the steps for aseptic insertion and ongoing care to minimize the risk of infection.
● Promote timely catheter removal: Encouraging regular assessment of the continued need for the catheter and its prompt removal when no longer indicated.
● Educate healthcare professionals: Raising awareness about the risks associated with urinary catheters and providing the knowledge and skills necessary for their safe management.
● Support quality improvement initiatives: Providing a benchmark for auditing practices and measuring the effectiveness of interventions aimed at reducing CAUTIs.
- Target Audience
● Infection Prevention and Control personnel
● All Healthcare Workers: Including Clinicians, Nurses, Head Nurses
● HAIs Surveillance Officers
● The Treating Physician
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
● Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
● Selecting only national and/or international guidelines
● Specific range of dates for publication (using Guidelines published or updated in 2013 and later)
● Selecting peer reviewed publications only
● Selecting guidelines written in English language
● Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
● Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
The following characteristics of the retrieved guidelines were summarized in:
● Developing organization/authors
● Date of publication, posting, and release
● Country/language of publication
● Date of posting and/or release
● Dates of the search used by the source guideline developers.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The committee decided to adapt from:
● National Health and Medical Research Council (2019) Australian Guidelines for the Prevention and Control of Infection in Healthcare. Canberra: Commonwealth of Australia.
● Center of Disease Control (2024). Guideline for Prevention of Catheter-Associated Urinary Tract Infections (2009) update March 25, 2024
Evidence assessment
According to the World Health Organization (WHO) Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
● GRADE working group: http://www.gradeworkingroup.org
● GRADE online training modules: http://cebgrade.mcmaster.ca/
● GRADE profile software: http://ims.cochrane.org/revman/gradepro
Table (1) Quality and Significance of the four levels of evidence in GRADE
|
Quality |
Definition |
Implications |
|
High |
The guideline development group is very confident that the true effect lies close to that of the estimate of the effect |
Further research is very unlikely to change confidence in the estimate effect |
|
Moderate |
The guideline development group is moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibly that it is substantially different |
Further research is likely to have an important impact on confidence in the estimate of the effect and may change the estimate |
|
Low |
Confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the true effect |
Further research is very likely to have an important on confidence in the estimate of effect and is unlikely to change the estimate |
|
Very low |
The group has very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of the effect |
Any estimate of the effect is very uncertain |
Table (2) Factors that determine How to upgrade or downgrade the quality of evidence.
|
Downgrade in presence of |
Upgrade in presence of |
|
Study limitations. 1- Serious limitations 2- Very serious limitations |
Dose- response gradient. +1 Evidence of a dose-response gradient |
|
Consistency 1- Important inconsistency |
Direction of plausible bias + All plausible confounders would have reduced the effect |
|
Directness 1- Some uncertainty 2- Major uncertainty |
Magnitude of the effect +1 Strong, no plausible Confounder, consistent and direct evidence |
|
Precision 1- Imprecise data |
+2 very strong, no major threats to validity and direct evidence |
|
Reporting bias 1. High probability of reporting bias |
|
The strength of the recommendations
The strength of a recommendation communicates the importance of adherence to the recommendation.
● Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
● Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations?
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Indicators for Monitoring
To ensure the appropriate prevention of catheter associated urinary tract infection in hospitals and reduce the risk of CAUTI, specific indicators should be monitored regularly. These are some indicators which can provide measurable data to assess compliance, identify areas for improvement, and guide interventions. Here are some key indicators that can be included in hospital guidelines for prevention of CAUTI.
a) Process Measures:
1. Compliance with educational program:
Calculate percent of personnel who have proper training:
● Numerator: number of personnel who insert urinary catheters and who have proper training
● Denominator: number of personnel who insert urinary catheters
● Standardization factor: 100 (i.e., multiply by 100 so that measure is expressed as a percentage)
2. Compliance with documentation of catheter insertion and removal dates:
Conduct random audits of selected units and calculate compliance rate:
● Numerator: number of patients on unit with catheters with proper documentation of insertion and removal dates
● Denominator: number of patients on the unit with a catheter in place at some point during admission
● Standardization factor: 100 (i.e., multiply by 100 so that measure is expressed as a percentage)
3. Compliance with documentation of indication at insertion
Percentage of patients with IUC insertion records in which an indication was documented at or prior to the time of insertion
● Numerator: Patients with IUC insertion records in which an indication was documented at or prior to the time of insertion
● Denominator: number of patients on the unit with inserted catheter
● Standardization factor: 100 (i.e., multiply by 100 so that measure is expressed as a percentage)
b) Outcome Measures:
1. Rates of CAUTI (Number of CAUTI per 1000 catheter-days)
Measurement of rates allows an individual facility to gauge the longitudinal impact of implementation of prevention strategies:
● Numerator: number of CAUTIs in each location monitored
● Denominator: total number of urinary catheter-days for all patients that have an indwelling urinary catheter in each location monitored
● Standardization factor: Multiply by 1000 so that the measure is expressed as cases per 1000 catheter-days.
2. Rate of bloodstream infections secondary to CAUTI (Number of bloodstream infections secondary to CAUTI per 1000 catheter-days):
Numerator: number of episodes of bloodstream infections secondary to CAUTI
Denominator: total number of urinary catheter-days for all patients that have an indwelling urinary catheter in each location monitored
Standardization factor: Multiply by 1000 so that the measure is expressed as cases per 1000 catheter-days
3. Device Utilization Ratio (DUR): The catheter utilization ratio is the number of urinary catheter-days per number of patient-days in a given period. This is a measure of the total patient-days in which a urinary catheter was used and can be used as a marker for risk of infection.
● Numerator: Number of Catheter Days for a location
● Denominator: Number of Patient Days for a location
According to the approved CDC criteria,
*Device days are the total number of days of exposure to urinary catheter for all the patients during the selected time period.
*Patient days are the total number of days patients are in the ICU during the selected time period
1.10 Plan to Update this National Clinical Guideline
This guideline will be reviewed and updated when new evidence emerges that is likely to influence the recommendations.
- Annex
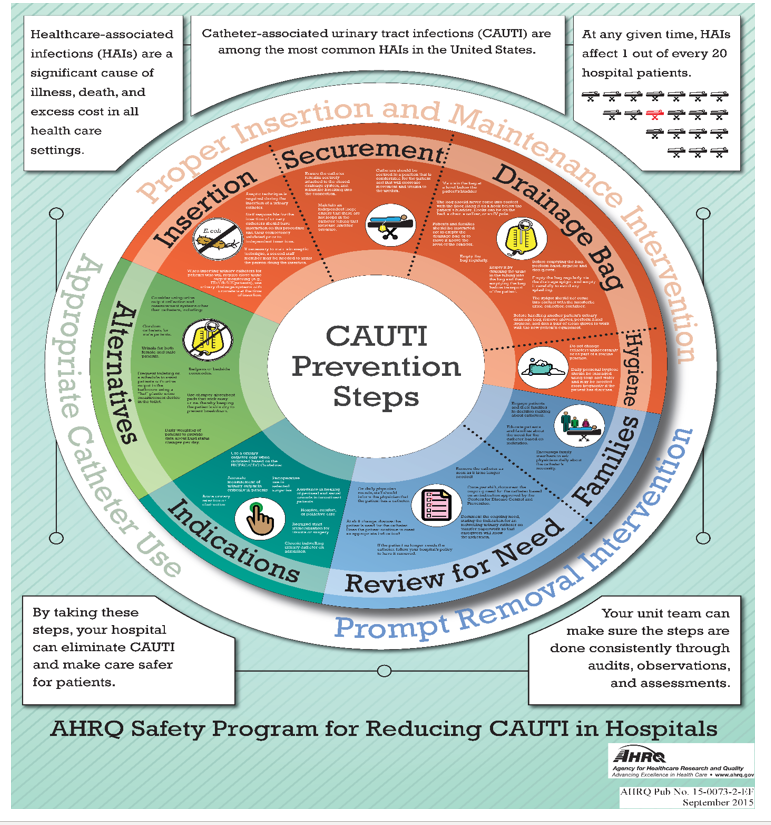
Annex 1. Poster on CAUTI
Prevention from AHRQ Safety Program for Reducing CAUTI in Hospitals
Annex 2. CAUTI Tiers. AHRQ Safety Program for ICUs: Preventing CLABSI and CAUTI
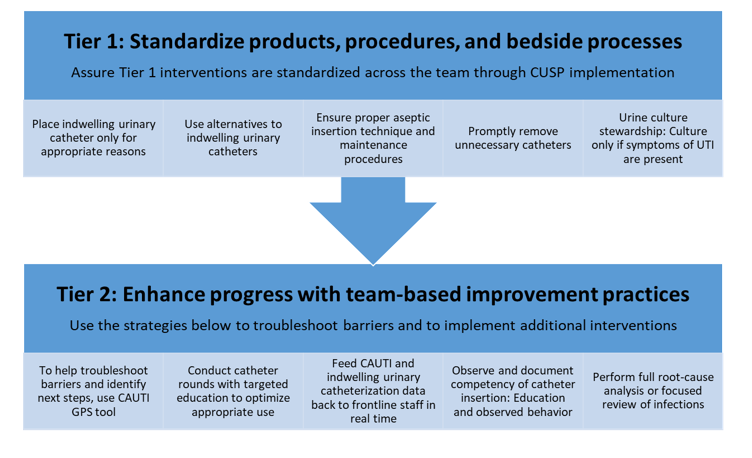
Abbreviations: CAUTI = catheter-associated urinary tract infection; CUSP: Comprehensive Unit-based Safety Program, GPS = Guide to Patient Safety; UTI = urinary tract infection
These materials were expanded, enhanced, and adapted from tiered interventions implemented in cohorts 1 and 2 of AHRQ Safety Program for ICUs: Preventing CLABSI and CAUTI from materials developed for CAUTI prevention by faculty and staff at the Department of Veterans Affairs and the University of Michigan.
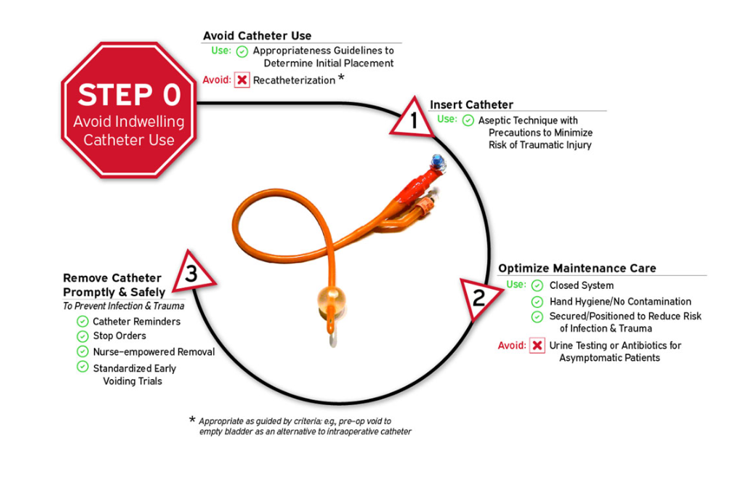
Annex 3. Disrupting the life cycle of the indwelling urethral catheter to reduce catheter-associated infection and trauma. from AHRQ Safety Program for Reducing CAUTI in Hospital
Annex 4. CAUTI insertion bundle
|
Verification Of Need Prior To Insertion. |
Insert Urinary Catheter Using Aseptic Technique. |
Maintain Urinary Catheter Based on Recommended Guidelines. |
|
● Urinary retention/Obstruction ● Severely ill/Immobility ● Lack bladder control ● Patient request/End of life ● Perioperative – selected surgical procedures ● Assisting with pressure ulcer healing for incontinent patients. |
● Hand hygiene ● Catheter insertion kit with sterile gloves, ● drape, cleaning supplies, sterile lubricant, sterile urinary catheter attached ● to a drainage bag. |
● Secure catheter to prevent irritation of the urethra ● Maintain an unobstructed flow, maintain the drainage bag below the level of the bladder and off the floor ● Perform hand hygiene before and after each patient contact ● Provide individual labelled collection container at the bedside ● Review urinary catheter necessity daily, remove catheter promptly when not needed. |
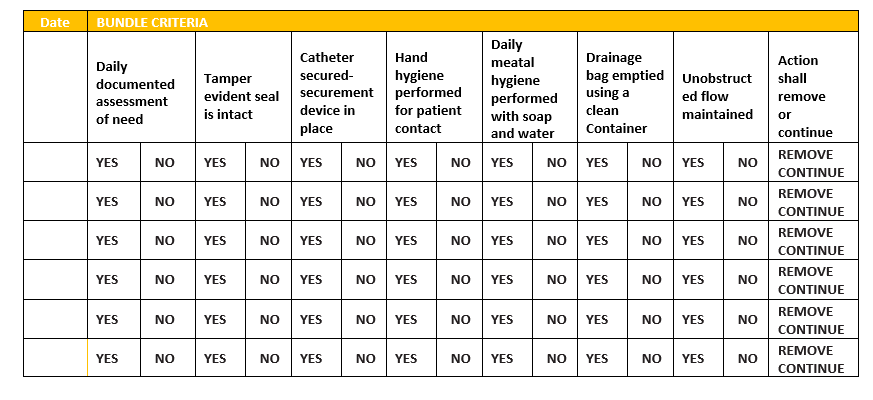
Annex 5. CAUTI Maintenance bundle
- References
1- Centers for Disease Control and Prevention (CDC). Patient Safety Component Manual. National Healthcare Safety Network (NHSN); 2025.
2- Association for Professionals in Infection Control and Epidemiology (APIC). Guide to Preventing Catheter-Associated Urinary Tract Infections. 2025.
3- Centers for Disease Control and Prevention (CDC). Guideline for Prevention of Catheter-Associated Urinary Tract Infections (2009). Updated March 25, 2024.
4- Patel PK, Greene MT, Fakih MG, Saint S, Meddings J. Strategies to prevent catheter-associated urinary tract infections in acute-care hospitals: 2022 update. Infect Control Hosp Epidemiol. 2023;44(8):1209–31. doi:10.1017/ice.2023.137.
5- National Health and Medical Research Council (NHMRC). Australian Guidelines for the Prevention and Control of Infection in Healthcare. Canberra: Commonwealth of Australia; 2019.