ADVANCED/METASTATIC BREAST CANCER
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | ADVANCED/METASTATIC BREAST CANCER |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
23 July 2025" Download Guideline
- Executive Summary
This guidance provides an evidence-based approach to the diagnosis, staging, treatment and follow up of patients diagnosed with advanced breast cancer
|
Strength of the recommendation |
|
|
Diagnosis, pathology and molecular biology |
|
|
At first diagnosis of MBC, a biopsy should be carried out to confirm histology and assess/re-assess tumour biology including ER, PgR, HER2 status & KI 67. |
Strong |
|
Staging and risk assessment |
|
|
The minimum imaging work-up for staging includes computed tomography (CT) of the chest and abdomen, and bone scintigraphy. |
Strong |
|
18F-FDG-PET)/CT may be used instead of CT and bone scans only as problem solving tool. |
Conditional |
|
The interval between imaging and starting treatment should be ≤4 weeks. |
Good practice statement. |
|
Evaluation of response should generally occur every 2-4 months depending on disease dynamics, location, extent of metastasis and type of treatment. |
Good practice statement. |
|
Disease monitoring intervals should not be shortened as there is no evidence of an OS benefit but potential for emotional and financial harm. Less frequent monitoring is acceptable, particularly for indolent disease. |
Good practice statement. |
|
If progression is suspected, additional tests should be carried out in a timely manner irrespective of planned intervals. |
Good practice statement. |
|
Repeat bone scans are a mainstay of evaluation for bone-only/predominant metastases, but image interpretation may be confounded by a possible flare during the first few months of treatment. MRI may be added to define response in specific locations. |
Conditional |
|
Impending fracture risk should be evaluated by CT or X-rays. In the case of suspected cord compression, MRI is the modality of choice. |
Strong |
|
Symptomatic patients should always undergo brain imaging, preferably with MRI. |
Strong |
|
HR-positive, HER2-negative breast cancer First Line. |
|
|
A CDK4/6 inhibitor combined with endocrine therapy (ET) may be used as first-line therapy for patients with ER-positive, HER2-negative MBC. However this depends on the availability, access-ability, patient comorbidity, and budget impact. |
Conditional |
|
Pre- and perimenopausal women should be offered OFS or ovarian ablation in addition to all endocrine-based therapies. |
Strong |
|
Second-line treatment. |
|
|
Selection of second-line therapy (chemotherapy versus further endocrine-based therapy) should be based on disease aggressiveness, extent and organ function, and consideration of the associated toxicity profile. |
Good practice statement. |
|
Everolimus- exemestane is a recommended option. |
Strong |
|
Tamoxifen or fulvestrant can also be combined with everolimus and is recommended. If everolimus is used, stomatitis prophylaxis must be used. |
Strong |
|
At least two lines of endocrine-based therapy are preferred before moving to chemotherapy in the absence of endocrine refractory disease and/or imminent organ failure. |
Strong |
|
In patients with imminent organ failure, chemotherapy is the preferred option. |
Strong |
|
For patients with endocrine-sensitive tumours, continuation of ET with agents not previously received in the metastatic setting is recommended.
|
Strong |
|
Beyond second-line treatment |
|
|
Patients with tumours that are endocrine resistant should be considered for chemotherapy. |
Strong |
|
Sequential single-agent chemotherapy is generally preferred over combination strategies. In patients where a rapid response is needed due to imminent organ failure, combination chemotherapy is preferred. |
Strong |
|
Available drugs for single-agent chemotherapy include anthracyclines, taxanes, capecitabine, vinorelbine, and platinums. |
Strong |
|
HER2-positive breast cancer |
|
|
Standard first-line treatment of HER2-positive MBC should be trastuzumab-docetaxel regardless of HR status. |
Strong |
|
Docetaxel should be given for at least six cycles, if tolerated, followed by maintenance trastuzumab until progression. |
Strong |
|
An alternative taxane (paclitaxel) can be substituted for docetaxel. |
Strong |
|
ET can be added to trastuzumab maintenance after completion of chemotherapy for HER2-positive, HR-positive . |
Strong |
|
If chemotherapy is contraindicated in patients with HER2-positive, HR-negative MBC, HER2-targeted therapy without chemotherapy (e.g. trastuzumab) is recommended. |
Strong |
|
if taxane chemotherapy is contraindicated, a less toxic chemotherapy partner (e.g. capecitabine or vinorelbine) should be considered. |
Strong |
|
In selected cases of HER2-positive, HR-positive MBC where the patient is not suitable for first-line chemotherapy, ET (e.g. an AI) in combination with an HER2-targeted therapy, such as trastuzumab, or lapatinib, can be recommended. |
Strong |
|
The use of single-agent ET without HER2-targeted therapy in HER2-positive, HR-positive MBC is not routinely recommended unless comorbidities (e.g. cardiac disease) preclude the safe use of HER2-directed therapies. |
Conditional |
|
Patients with metastatic recurrence within 12 months of receiving adjuvant trastuzumab should follow the second-line therapy recommendations. |
Strong |
|
In later lines of therapy, lapatinib is an evidence-based therapy option to be used preferably in combinations (e.g. with capecitabine, trastuzumab or ET). |
Conditional |
|
TNBC |
|
|
In cases of imminent organ failure, combination therapy is preferred based on a taxane and/or anthracycline combination. |
|
|
After progression, all chemotherapy recommendations for HER2-negative disease also apply for TNBC, e.g. capecitabine, and vinorelbine. |
|
|
Site-specific management Primary stage IV disease |
|
|
For patients with newly diagnosed stage IV breast cancer and an intact primary tumour, therapeutic decisions should ideally be discussed in a multidisciplinary context.
|
Good practice statement.
|
|
Locoregional treatment of the primary tumour in the absence of symptomatic local disease does not lead to an OS benefit and is not routinely recommended. |
Good practice statement.
|
|
In patients with local symptoms caused by the primary tumour or metastatic disease, the use of local treatment modalities should be evaluated. |
Strong |
|
Surgery of the primary tumour is recommended for patients who may benefit from salvage surgery (e.g. those with bone-only metastases, a good response to initial systemic therapy, HR-positive tumours, HER2-negative tumours, age <55 years and those with OMD). |
Strong |
|
Surgery or RT of the primary tumour should be carefully considered for circumstances in which they provide added value for symptom palliation or prevention of complications. |
Conditional |
|
Oligometastatic disease |
|
|
A multidisciplinary approach is essential to manage patients with bone metastases and prevent skeletal-related events (SREs). |
Good practice statement.
|
|
Patients with OMD should be discussed in a multidisciplinary context to individualise management. |
Good practice statement.
|
|
Multimodality treatment approaches involving locoregional therapy [e.g. high conformal radiotherapy (RT), image-guided ablation,selective internal RT and/or surgery] combined with systemic treatments are recommended, and should be tailored to the disease presentation in the individual patient. |
Strong |
|
Local ablative therapy to all metastatic lesions may be offered on an individual basis after discussion in a multidisciplinary setting. |
Conditional |
|
Bone metastases and bone-modifying agents |
|
|
A multidisciplinary approach is essential to manage patients with bone metastases and prevent skeletal-related events (SREs). |
Strong |
|
An orthopaedic evaluation is advised in the case of significant lesions in the long bones or vertebrae as well as in patients with MSCC to discuss the possible role of surgery. |
Strong |
|
RT is recommended for lesions at moderate risk of fracture and those associated with moderate to severe pain. |
Strong |
|
A single 8-Gy RT fraction is as effective as fractionated schemes in patients with uncomplicated bone metastases. |
Strong |
|
RT should be delivered after surgery for stabilisation or separation surgery for MSCC. |
Strong |
|
Bone-modifying agents (BMAs) are recommended for patients with bone metastases,regardless of symptoms. |
Strong |
|
Before the initiation of BMAs, patients should have a complete dental evaluation and ideally complete any required dental treatment. Calcium and vitamin D supplements should be prescribed. |
Strong |
|
The optimal duration of BMA therapy has not been defined but it is reasonable to interrupt therapy after 2 years for patients in remission. |
Good Practice Statement |
|
The ideal sequence of therapies has not been defined but it seems reasonable to document tumour response with a systemic treatment before suggesting locoregional therapy. |
Conditional |
|
Brain metastases and leptomeningeal metastases |
|
|
Brain metastases should be managed according to the recommendations outlined in the European Association of Neuro-Oncology-ESMO (EANO-ESMO) Clinical Practice Guideline (CPG) for the management of patients with brain metastases from solid tumours. |
Strong |
|
Leptomeningeal metastases should be treated according to the recommendations outlined in the EANO-ESMO CPG for the management of patients with leptomeningeal metastases from solid tumours. |
Strong |
|
Long-term implications and survivorship |
|
|
An interdisciplinary approach is critical, including specialised oncology and/or breast care nurses to proactively screen for and manage treatment-emergent toxicities. |
Good practice statement.
|
|
Patients should be informed about treatment choices and side-effect profiles of recommended systemic treatments. |
Good practice statement.
|
|
All treatments should include formal patient education regarding side-effects of management. |
Strong |
|
All efforts should be done to encourage integrated electronic medical records (EMR) in different hospitals. |
Strong |
|
QoL assessments should be incorporated into the evaluation of treatment efficacy. |
Strong |
|
Dose reduction and delay are effective strategies to manage toxicity in advanced disease. |
Strong |
- Recommendations
Diagnosis, pathology and molecular biology
- At first diagnosis of MBC, a biopsy should be carried out to confirm histology and assess/re-assess tumour biology including ER, PgR, HER2 status & KI 67.
Strong recommendation, high grade evidence (1).
Staging and risk assessment
- The minimum imaging work-up for staging includes computed tomography (CT) of the chest and abdomen, and bone scintigraphy.
Strong recommendation, high grade evidence (2,3).
- 18F-FDG-PET)/CT may be used instead of CT and bone scans only as problem solving tool.
Conditional recommendation, high grade evidence (2,3).
- The interval between imaging and starting treatment should be ≤4 weeks.
Good practice statement.
- Evaluation of response should generally occur every 2-4 months depending on disease dynamics, location, extent of metastasis and type of treatment.
Good practice statement.
- Disease monitoring intervals should not be shortened as there is no evidence of an OS benefit but potential for emotional and financial harm. Less frequent monitoring is acceptable, particularly for indolent disease.
Good practice statement.
- If progression is suspected, additional tests should be carried out in a timely manner irrespective of planned intervals.
Good practice statement.
- Repeat bone scans are a mainstay of evaluation for bone-only/predominant metastases, but image interpretation may be confounded by a possible flare during the first few months of treatment. MRI may be added to define response in specific locations.
Conditional recommendation, low grade evidence (3).
- Impending fracture risk should be evaluated by CT or X-rays. In the case of suspected cord compression, MRI is the modality of choice.
Strong recommendation, high grade evidence (4).
- Symptomatic patients should always undergo brain imaging, preferably with MRI.
Strong recommendation, high grade evidence (5).
HR-positive, HER2-negative breast cancer
- A CDK4/6 inhibitor combined with endocrine therapy (ET) may be used as first-line therapy for patients with ER-positive, HER2-negative MBC. However this depends on the availability, access-ability, patient comorbidity, and budget impact.
Conditional recommendation, high grade evidence (6-8).
- Pre- and perimenopausal women should be offered OFS or ovarian ablation in addition to all endocrine-based therapies.
Strong recommendation, high grade evidence (9).
Second-line treatment
- Selection of second-line therapy (chemotherapy versus further endocrine-based therapy) should be based on disease aggressiveness, extent and organ function, and consideration of the associated toxicity profile.
Good practice statement.
- Everolimus- exemestane is a recommended option
Strong recommendation, high grade evidence(10-13).
- Tamoxifen or fulvestrant can also be combined with everolimus and is recommended. If everolimus is used, stomatitis prophylaxis must be used.
Strong recommendation, high grade evidence (10-13).
- At least two lines of endocrine-based therapy are preferred before moving to chemotherapy in the absence of endocrine refractory disease and/or imminent organ failure.
Strong recommendation, very low grade evidence (14).
- In patients with imminent organ failure, chemotherapy is the preferred option.
Strong recommendation, low grade evidence(14).
- For patients with endocrine-sensitive tumours, continuation of ET with agents not previously received in the metastatic setting is recommended.
Strong recommendation, low grade evidence (14).
Beyond second-line treatment
- Patients with tumours that are endocrine resistant should be considered for chemotherapy.
Strong recommendation, very low grade evidence (14).
- Sequential single-agent chemotherapy is generally preferred over combination strategies. In patients where a rapid response is needed due to imminent organ failure, combination chemotherapy is preferred.
Strong recommendation, high grade evidence (14).
- Available drugs for single-agent chemotherapy include anthracyclines, taxanes, capecitabine, vinorelbine,and platinums .
Strong recommendation, high grade evidence(14).
HER2-positive breast cancer
- Standard first-line treatment of HER2-positive MBC should be trastuzumab-docetaxel regardless of HR status.
Strong recommendation, high grade evidence (15,16).
- Docetaxel should be given for at least six cycles, if tolerated, followed by maintenance trastuzumab until progression.
Strong recommendation, high grade evidence(15,16).
- An alternative taxane (paclitaxel) can be substituted for docetaxel
Strong recommendation, high grade evidence(15,16).
- ET can be added to trastuzumab maintenance after completion of chemotherapy for HER2-positive, HR-positive .
Strong recommendation, high grade evidence (17).
- If chemotherapy is contraindicated in patients with HER2-positive, HR-negative MBC, HER2-targeted therapy without chemotherapy (e.g. trastuzumab) is recmmended.
Strong recommendation, low grade evidence (18).
- if taxane chemotherapy is contraindicated, a less toxic chemotherapy partner (e.g. capecitabine or vinorelbine) should be considered.
Strong recommendation, low grade evidence.
- In selected cases of HER2-positive, HR-positive MBC where the patient is not suitable for first-line chemotherapy, ET (e.g. an AI) in combination with an HER2-targeted therapy, such as trastuzumab, or lapatinib, is recommended.
Strong recommendation, high grade evidence (19,20).
- The use of single-agent ET without HER2-targeted therapy in HER2-positive, HR-positive MBC is not routinely recommended unless comorbidities (e.g. cardiac disease) preclude the safe use of HER2-directed therapies.
Conditional recommendation, low grade evidence (21).
- Patients with metastatic recurrence within 12 months of receiving adjuvant trastuzumab should follow the second-line therapy recommendations.
Strong recommendation, high grade evidence (22).
- In later lines of therapy, lapatinib is an evidence-based therapy option to be used preferably in combinations (e.g. with capecitabine, trastuzumab or ET).
Conditional recommendation, low grade evidence(23).
TNBC
- In cases of imminent organ failure, combination therapy is preferred based on a taxane and/or anthracycline combination.
Conditional recommendation, low grade evidence (14).
- After progression, all chemotherapy recommendations for HER2-negative disease also apply for TNBC, e.g. capecitabine, and vinorelbine.
Strong recommendation, low grade evidence (14).
Site-specific management
Primary stage IV disease
- For patients with newly diagnosed stage IV breast cancer and an intact primary tumour, therapeutic decisions should ideally be discussed in a multidisciplinary context.
Good practice statement.
- Locoregional treatment of the primary tumour in the absence of symptomatic local disease does not lead to an OS benefit and is not routinely recommended.
Good practice statement.
- In patients with local symptoms caused by the primary tumour or metastatic disease, the use of local treatment modalities should be evaluated.
Strong recommendation, high grade evidence(24).
- Surgery of the primary tumour is recommended for patients who may benefit from salvage surgery (e.g. those with bone-only metastases, a good response to initial systemic therapy, HR-positive tumours, HER2-negative tumours, age <55 years and those with OMD).
Strong recommendation, high grade evidence(25).
- Surgery or RT of the primary tumour should be carefully considered for circumstances in which they provide added value for symptom palliation or prevention of complications.
Conditional recommendation, very low grade evidence(25).
Oligometastatic disease
- A multidisciplinary approach is essential to manage patients with bone metastases and prevent skeletal-related events (SREs).
Good practice statement.
- Patients with OMD should be discussed in a multidisciplinary context to individualise management.
Good practice statement.
- Multimodality treatment approaches involving locoregional therapy [e.g. high conformal radiotherapy (RT), image-guided ablation,selective internal RT and/or surgery] combined with systemic treatments are recommended, and should be tailored to the disease presentation in the individual patient.
Strong recommendation, very low grade evidence (26).
- Local ablative therapy to all metastatic lesions may be offered on an individual basis after discussion in a multidisciplinary setting.
Conditional recommendation, moderate grade evidence (26).
Bone metastases and bone-modifying agents
- A multidisciplinary approach is essential to manage patients with bone metastases and prevent skeletal-related events (SREs).
Strong recommendation, very low grade evidence (26).
- An orthopaedic evaluation is advised in the case of significant lesions in the long bones or vertebrae as well as in patients with MSCC to discuss the possible role of surgery.
Strong recommendation, very low grade evidence (26).
- RT is recommended for lesions at moderate risk of fracture and those associated with moderate to severe pain.
Strong recommendation, very low grade evidence (26).
- A single 8-Gy RT fraction is as effective as fractionated schemes in patients with uncomplicated bone metastases.
Strong recommendation, high grade evidence (26).
- RT should be delivered after surgery for stabilisation or separation surgery for MSCC.
Strong recommendation, low grade evidence(26).
- Bone-modifying agents (BMAs) are recommended for patients with bone metastases,regardless of symptoms.
Strong recommendation, high grade evidence (26).
- Before the initiation of BMAs, patients should have a complete dental evaluation and ideally complete any required dental treatment. Calcium and vitamin D supplements should be prescribed.
Strong recommendation, low grade evidence (26).
- The optimal duration of BMA therapy has not been defined but it is reasonable to interrupt therapy after 2 years for patients in remission.
- The ideal sequence of therapies has not been defined but it seems reasonable to document tumour response with a systemic treatment before suggesting locoregional therapy.
Conditional recommendation, very low grade evidence(26).
Brain metastases and leptomeningeal metastases
- Brain metastases should be managed according to the recommendations outlined in the European Association of Neuro-Oncology-ESMO (EANO-ESMO) Clinical Practice Guideline (CPG) for the management of patients with brain metastases from solid tumours.
Strong recommendation, high grade evidence (27).
- Leptomeningeal metastases should be treated according to the recommendations outlined in the EANO-ESMO CPG for the management of patients with leptomeningeal metastases from solid tumours.
Strong recommendation, high grade evidence (27).
Long-term implications and survivorship
- An interdisciplinary approach is critical, including specialised oncology and/or breast care nurses to proactively screen for and manage treatment-emergent toxicities.
Good practice statement.
- Patients should be informed about treatment choices and side-effect profiles of recommended systemic treatments.
Good practice statement.
- All treatments should include formal patient education regarding side-effects of management.
Strong recommendation, high grade evidence (28).
- All efforts should be done to encourage integrated electronic medical records (EMR) in different hospitals
Strong recommendation, high grade evidence(28).
- QoL assessments should be incorporated into the evaluation of treatment efficacy.
Strong recommendation, high grade evidence(28).
- Dose reduction and delay are effective strategies to manage toxicity in advanced disease.
Strong recommendation, high grade evidence(28).
- Acknowledgement
· We would like to acknowledge the Oncology Committee of Egyptian health council (EHC) Guidelines and Breast Cancer Scientific Committee, for adapting this Guidelines.
· Chair of the Committee of Egyptian health council Guidelines: Prof Hussein Khaled.
· The Oncology Committee Members: Ebtesam Saad Eldin, Ehab Khalil, Emad Hamada, Fouad Abuotaleb, Hesham Elghazaly, Hesham Tawfik, Khaled Abdelkarim, Lobna ezz Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Samir Shehata, Yousri Wasef & Yousri Rostom.
· The Breast Cancer Scientific Committee Members: Mohamed Gamil, Lobna Ezz Elarab, Hesham Elghazaly, Ehab Khalil, Rasha Fahmi, Rasha Shaltout, Omar Sherif Omar.
- Abbreviations
AI Aromatase inhibitor
AJCC American joint committee on cancer
Ax Axilla
BC Breast cancer
BCT Breast conserving therapy
BCS Breast conserving surgery
Bx Biopsy
CEM Contrast-enhanced mammography
CT Chemotherapy
ECOG The Eastern Cooperative Oncology Group
EMR Electronic medical records
ESMO European society for medical oncology
ET Endocrine therapy
ER Estrogen receptors
FH Family history
G Gauge
H&E Hematoxylin and eosin
HER2 Human epidermal growth factor receptor2
HR Hormone receptors
IBC Inflammatory BC
IBTR Ipsilateral breast tumor recurrence
IHC Immunohistochemistry
IKWG International Ki67 in Breast Cancer Working Group
IM Internal mammary
ISH In-situ hybridization
LABC Locally advanced breast cancer
LN Lymph nodes
MDT Multi-disciplinary team
Mets Metastasis
MRI Magnetic resonance imaging
MRM Modified radical mastectomy.
NACT Neoadjuvant chemotherapy
NCCN National comprehensive cancer network
NMBC Non metastatic breast cancer
NSM Nipple-sparing mastectomy
OFS Ovarian function suppression
pCR Pathological complete response
PgR Progesterone receptor
PO Per oral
PST Primary systemic therapy
QoL Quality of Life
RT Radiotherapy
SC Supraclavicular
SLNB Sentinel LN biopsy
SNM Sono-mammography
SSM Skin-sparing mastectomy
TNBC Triple negative breast cancer
TCH Taxotere carboplatin Herceptin
U/S Ultrasound
WLE Wide local excision.
- Glossary
Menopausal status (Defined by NCCN)
Postmenopausal status:
· Permanent cessation of menses includes a profound and permanent decrease in ovarian estrogen synthesis.
Premenopausal status:
· It is the ongoing process of menses and normal ovarian estrogen synthesis.
Perimenopausal status:
· It is the transition status between the pre- and post-menopausal status with irregularities in menses and estrogen levels.
Definitions of molecular subtypes of breast cancer according to: (The ESMO Clinical Practice Guidelines, 2023):
◾ Luminal A: (ER-positive, HER2-negative, Ki67 low, PgR high)
◾ Luminal B:
(HER2-negative subtype): (ER-positive, HER2-negative, and either, Ki67 high or PgR low)
(HER2-positive subtype): (ER-positive, HER2-positive, any Ki67, any PgR)
◾ HER2 overexpression (nonluminal): (HER2-positive, ER and PgR absent)
◾ Triple-negative: (ER and PgR absent, HER2-negative)
Cancers with 1%–100% ER IHC staining are considered ER-positive.
Cancers with 1%–10% ER IHC staining are considered ER-low-positive.
HER2 positive patients (IHC+++ or ISH positive for gene amplification (HER2/CEP17 ratio ≥2.0 AND average HER2 copy number ≥ 4.0 signals/cell))
Ki67 index of 5% or less, or 30% or more, can be used to estimate prognosis for T1-2, N0-1 patients as per the International Ki67 in Breast Cancer Working Group (IKWG)
Definition of High-risk patients:
-HER2-positive disease
-TNBC
- ≥ cT2 or ≥ cN1
-Large primary tumor relative to breast size
- Introduction
Breast cancer is the most common cancer in females and the second most common in the Egyptian population with more than 26 thousand newly diagnosed cases(1). Moreover, it is also the second cause of cancer death in Egypt after hepatocellular carcinoma with estimated mortality rate around 10% in 2022. Approximately 46,000 incident cases are forecasted in 2050.
- Purpose and scope
These guidelines will help to improve the quality of care for advanced breast cancer patients via providing a uniform standard of care across the country to help in early diagnosis and treatment for breast cancer, with less aggressive treatment options and improved clinical outcomes. These guidelines cover primary diagnosis, staging, treatment and follow-up of advanced breast cancer patients.
- Target audience
Clinicians who are involved in the care and treatment of patients with breast cancer, including medical oncologists, radiation oncologists, clinical oncologists , surgeons, interventional radiologists, radiologists and pathologists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The NCCN, ESMO, NICE guidelines are the main sources used while formulating the national guidelines for breast cancer.
- Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group: https://www.gradeworkinggroup.org/
. GRADE online training modules:http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
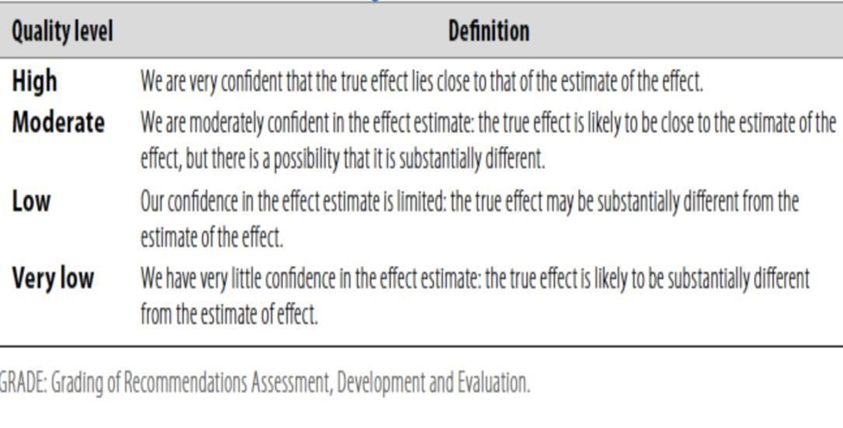
Table 2: Significance of the four levels of evidence

Table 3: Factors that determine how to upgrade or downgrade the quality of evidence.
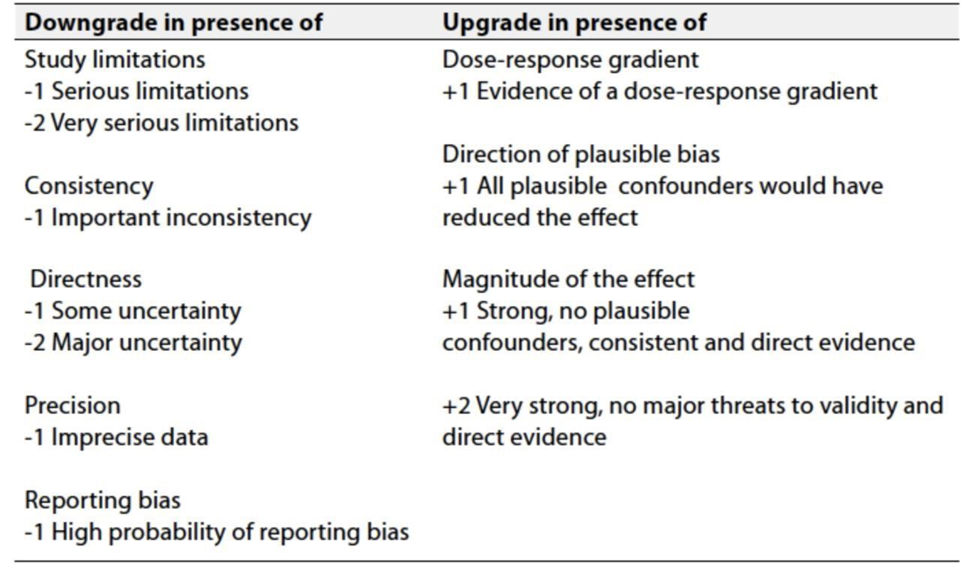
➡️ The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research Gaps
• Head-to-Head comparison for adjuvant Olaparib versus Capecitabine in TNBC received neoadjuvant treatment with residual disease.
• Pembrolizumab neoadjuvant exclusively versus neoadjuvant and adjuvant regarding survival benefit, toxicity including financial toxicity and quality of life.
• This guideline will be updated whenever there is new evidence.
• Randomized trials to determine the risks and benefits of brain screening in TNBC needed.
- Update of this guidline
This guideline will
be updated whenever there is new evidence.
- Annexes
Annex 1.
American Joint Committee on Cancer (AJCC) TNM Staging System For Breast Cancer
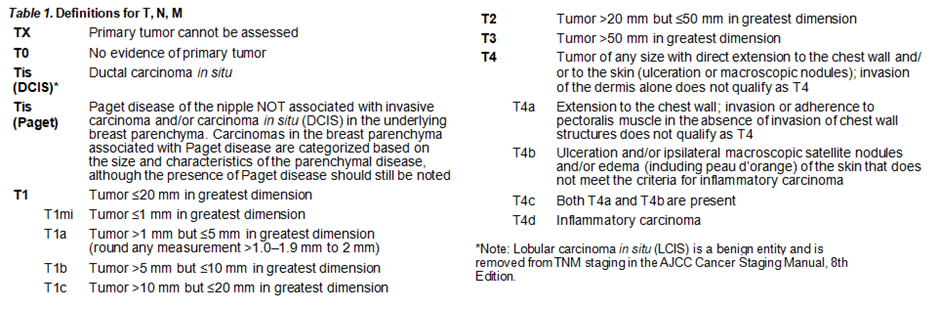
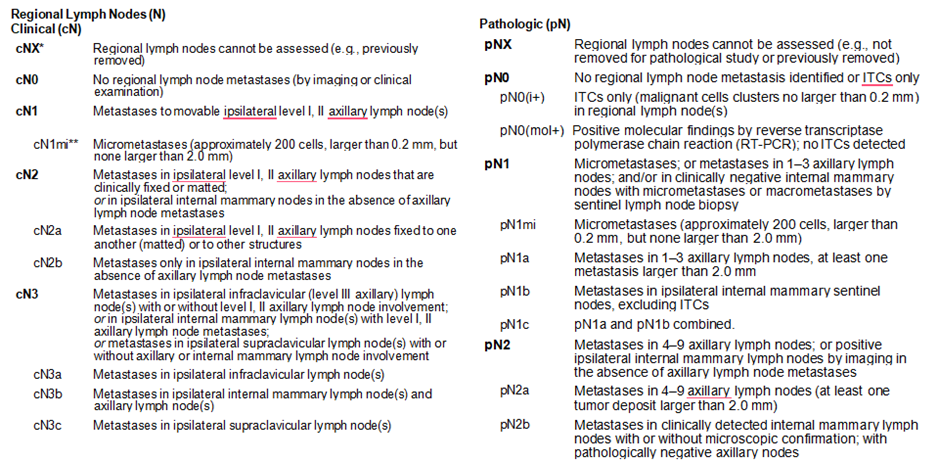


The AJCC Cancer Staging Manual, Eighth Edition (2017) published by Springer International Publishing
Annex 2.
Spine instability neoplastic score
(SINS)
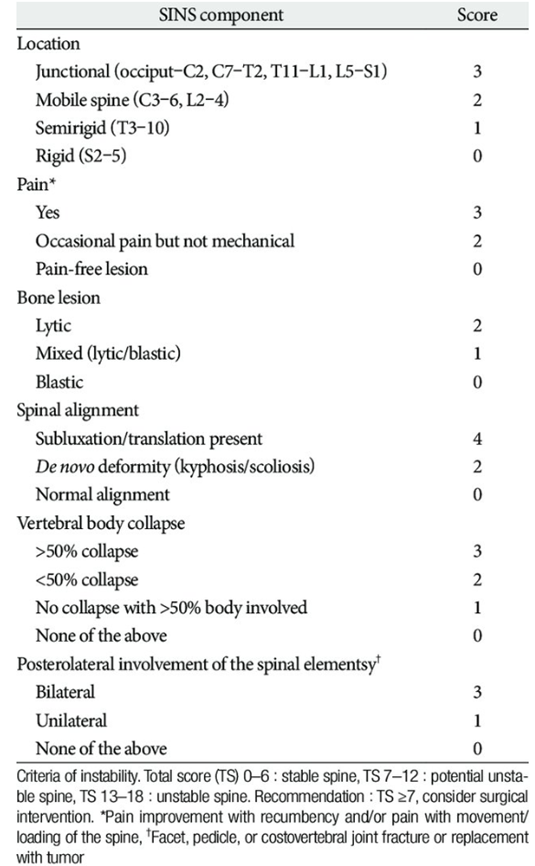
Ho-Young Park et.al., J Korean Neurosurg Soc 57 (2) : 100-107, 2015
- References
1. Gennari A, André F, Barrios CH, et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Ann Oncol. 2021;32(12):1475-1495
2. Cardoso F, Paluch-Shimon S, Senkus E, et al. 5th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 5). Ann Oncol. 2020;31(12):1623-1649.
3. Lee CI, Gold LS, Nelson HD, et al. Comparative effectiveness of imaging modalities to determine metastatic breast cancer treatment response. Breast. 2015;24(1):3-11.
4. Fourney DR, Frangou EM, Ryken TC, et al. Spinal instability neoplastic score: an analysis of reliability and validity from the spine oncology study group. J Clin Oncol. 2011;29(22):3072-3077.
5. Komorowski AS, Warner E, MacKay HJ, et al. Incidence of brain me- tastases in nonmetastatic and metastatic breast cancer: is there a role for screening? Clin Breast Cancer. 2020;20(1):e54-e64.
6. Cristofanilli M, Rugo HS, Im SA, Slamon DJ, Harbeck
N, Bondarenko I, Masuda N et al.
Overall Survival with Palbociclib and Fulvestrant in Women with HR+/HER2- ABC:
Updated Exploratory Analyses of PALOMA-3, a Double-blind, Phase III
Randomized Study.Clin Cancer Res. 2022 Aug
15;28(16):3433-3442. doi: 10.1158/1078-0432.CCR-22-0305.
7. Goetz MP, Toi M, Huober J, Sohn J, Trédan O, Park IH et al. Abemaciclib plus a nonsteroidal aromatase inhibitor as initial therapy for HR+, HER2- advanced breast cancer: final overall survival results of MONARCH 3., Ann Oncol. 2024 Aug;35(8):718-727.
8. Ribociclib plus Endocrine Therapy in Early Breast Cancer.Slamon D, Lipatov O, Nowecki Z, McAndrew N, Kukielka-Budny B, Stroyakovskiy et al. N Engl J Med. 2024 Mar 21;390(12):1080-1091.
9. Klijn JG, Beex LV, Mauriac L, et al. Combined treatment with buserelin and tamoxifen in premenopausal metastatic breast cancer: a ran- domized study. J Natl Cancer Inst. 2000;92(11):903-911.
10. Baselga J, Campone M, Piccart M, et al. Everolimus in post- menopausal hormone-receptor-positive advanced breast cancer. N Engl J Med. 2012;366(6):520-529.
11. Cook MM, Al Rabadi L, Kaempf AJ, et al. Everolimus plus exemestane treatment in patients with metastatic hormone receptor-positive breast cancer previously treated with CDK4/6 inhibitor therapy. Oncologist. 2021;26(2):101-106.
12. Jerusalem G, de Boer RH, Hurvitz S, et al. Everolimus plus exemestane vs everolimus or capecitabine monotherapy for estrogen receptor- positive, HER2-negative advanced breast cancer: the BOLERO-6 ran- domized clinical trial. JAMA Oncol. 2018;4(10):1367-1374.
13. Piccart M, Hortobagyi GN, Campone M, et al. Everolimus plus exemestane for hormone-receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: overall survival results from BOLERO-2. Ann Oncol. 2014;25(12):2357-2362.
14. Im SA, Gennari A ,Park YH, Kim JH et al., Pan-Asian adapted ESMO Clinical Practice Guidelines for the diagnosis, staging and treatment of patients with metastatic breast cancer, Ann. Oncol, 8,(3): 1-24, 2023
15. Kaufman B, Mackey JR, Clemens MR, et al. Trastuzumab plus anas- trozole versus anastrozole alone for the treatment of post- menopausal women with human epidermal growth factor receptor 2- positive, hormone receptor-positive metastatic breast cancer: results from the randomized phase III TAnDEM study. J Clin Oncol. 2009;27(33):5529-5537.
16. Yuan Z, Huang J-J, Hua X, et al. Trastuzumab plus endocrine therapy or chemotherapy as first-line treatment for metastatic breast cancer with hormone receptor-positive and HER2-positive: the SYSUCC-002 randomized clinical trial. J Clin Oncol. 2021;39(suppl 15):1003.
17. Rimawi M, Ferrero JM, de la Haba-Rodriguez J, et al. First-line tras- tuzumab plus an aromatase inhibitor, with or without pertuzumab, in human epidermal growth factor receptor 2-positive and hormone receptor-positive metastatic or locally advanced breast cancer (PERTAIN): a randomized, open-label phase II trial. J Clin Oncol. 2018;36(28):2826-2835.
18. Cortés J, Fumoleau P, Bianchi GV, et al. Pertuzumab monotherapy after trastuzumab-based treatment and subsequent reintroduction of trastuzumab: activity and tolerability in patients with advanced hu- man epidermal growth factor receptor 2-positive breast cancer. J Clin Oncol. 2012;30(14):1594-1600
19. Johnston SRD, Hegg R, Im SA, et al. Phase III, randomized study of dual human epidermal growth factor receptor 2 (HER2) blockade with lapatinib plus trastuzumab in combination with an aromatase inhib- itor in postmenopausal women with HER2-positive, hormone receptor-positive metastatic breast cancer: ALTERNATIVE. J Clin Oncol. 2018;36(8):741-748.
20. Johnston S, Pippen J Jr, Pivot X, et al. Lapatinib combined with letrozole versus letrozole and placebo as first-line therapy for post- menopausal hormone receptor-positive metastatic breast cancer. J Clin Oncol. 2009;27(33):5538-5546.
22. Bartsch R, Cameron D, Ciruelos E, Criscitiello C, Curigliano G, Duhoux FP, Foukakis T, Gligorov J, Harbeck N, LeVasseur N, Okines A, Penault-Llorca F, Müller V.Expert recommendations on treatment sequencing and challenging clinical scenarios in human epidermal growth factor receptor 2-positive (HER2-positive) metastatic breast cancer.Cancer Treat Rev. 2025 Jan;132:102853.
23. ASCO Educational Book: 2022,https://ascopubs.org/doi/pdf/10.1200/EDBK_351222#:~:text=In%20later%20lines%20of%20therapy,in%20the%20late%2Dline%20scenario.
24. Ryckman JM, Thomas TV, Wang M, Wu X, Siva S, Spratt DE, Slotman B, Pal S, Chapin BF, Fitzal F, Soran A, Bex A, Louie AV, Lehrer EJ, Zaorsky NG., Local Treatment of the Primary Tumor for Patients With Metastatic Cancer (PRIME-TX): A Meta-Analysis.
Int J Radiat Oncol Biol Phys. 2022 Dec 1;114(5):919-935.
25. Demirors B, Goktepe B, Medeck H, Ozbas S, Soran A, et al., The Role of Primary Surgery in De Novo Metastatic Breast Carcinoma.Eur J Breast Health. 2023 Apr 1;19(2):110-114.
26. Gennari A, André F, Barrios C H et al., ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer, 32,12:1475-1495.
27. Le Rhun E, Guckenberger M, Smits M, et al. EANO-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up of patients with brain metastasis from solid tumours. Ann Oncol. 2021;32(11): 1332-1347.
28. John Sieh Dumbuya,Bashir Ahmad, Cizheng Zeng, Xiuling Chen & Jun Lu, Assessing the effectiveness of measurement scales in evaluating the health-related quality of life in rare disease patients after treatment: a systematic review Health and Quality of Life Outcomes, 22,108 (2024).