Management of Multidrug-Resistant Organisms (MDROs) and Outbreak Situations
| Site: | EHC | Egyptian Health Council |
| Course: | Infection Prevention and Control Guidelines |
| Book: | Management of Multidrug-Resistant Organisms (MDROs) and Outbreak Situations |
| Printed by: | Guest user |
| Date: | Sunday, 17 May 2026, 3:46 AM |
Description
"last update: 28 July 2025" Download Guideline
- Executive Summary
Although transmission of MDROs is most frequently documented in acute care facilities, all healthcare settings are affected by the emergence and transmission of antimicrobial-resistant microbes. The severity and extent of disease caused by these pathogens varies by the population(s) affected and by the institution(s) in which they are found.
These Recommendation serve as general guidance for the implementation of strategies and practices to prevent the transmission of MRSA, VRE, and other MDROs. The administration of healthcare organizations and institutions should ensure that appropriate strategies are fully implemented, regularly evaluated for effectiveness, and adjusted such that there is a consistent decrease in the incidence of targeted MDROs. It also guides the initial response following the identification of novel or targeted multidrug-resistant organisms (MDROs) or resistance mechanisms.
|
Recommendations |
|
1. Administrative Measures |
|
Make MDRO prevention and control an organizational patient safety priority (Strong Recommendation) |
|
Provide administrative support, and both financial and human resources, to prevent and control MDRO transmission within the healthcare organization. (Strong Recommendation) |
|
Implement a multidisciplinary process to monitor and improve healthcare personnel (HCP) adherence to recommended practices for Standard and Contact Precautions. (Strong Recommendation) |
|
Implement systems to designate patients known to be colonized or infected with a targeted MDRO and to notify receiving healthcare facilities and personnel prior to transfer of such patients within or between facilities. (Strong Recommendation) |
|
Provide updated feedback at least annually to healthcare providers and administrators on facility and patient-care-unit trends in MDRO infections. Include information on changes in prevalence or incidence of infection, results of assessments for system failures, and action plans to improve adherence to and effectiveness of recommended infection control practices to prevent MDRO transmission. (Strong Recommendation) |
|
2. Education and Training of Healthcare Personnel |
|
Provide education and training on risks and prevention of MDRO transmission during orientation and periodic educational updates for healthcare personnel; include information on organizational experience with MDROs and prevention strategies. (Strong Recommendation) |
|
3. Judicious Use of Antimicrobial Agents |
|
In hospitals and long-term care facilities, ensure that a multidisciplinary process is in place to review antimicrobial utilization, local susceptibility patterns (antibiograms), and antimicrobial agents included in the formulary to foster appropriate antimicrobial use. (Strong Recommendation) |
|
Implement systems (e.g., computerized physician order entry, comment in microbiology susceptibility report, notification from a clinical pharmacist or unit director) to prompt clinicians to use the appropriate antimicrobial agent and regimen for the given clinical situation. (Strong Recommendation) |
|
Provide clinicians with antimicrobial susceptibility reports and analysis of current trends, updated at least annually, to guide antimicrobial prescribing practices. (Strong Recommendation) |
|
In settings that administer antimicrobial agents but have limited electronic communication system infrastructures to implement physician prompts, implement a process for appropriate review of prescribed antimicrobials. Prepare and distribute reports to prescribers that summarize findings and provide suggestions for improving antimicrobial use. (Good Practice Statement) |
|
4. Surveillance |
|
In microbiology laboratories, use standardized laboratory methods and follow published guidance for determining antimicrobial susceptibility of targeted (e.g., MRSA, VRE, MDR-ESBLs) and emerging (e.g., VRSA, MDR- MDROs. (Strong Recommendation) |
|
In all healthcare organizations, establish systems to ensure that clinical microbiology laboratories (in-house and out-sourced) promptly notify infection control staff when a novel resistance pattern for that facility is detected. (Strong Recommendation) |
|
In hospitals and long-term care facilities, develop and implement laboratory protocols for storing isolates of selected MDROs for molecular typing when needed to confirm transmission or delineate the epidemiology of the MDRO within the healthcare setting. (Strong Recommendation) |
|
Monitor trends in the incidence of target MDROs in the facility over time using appropriate statistical methods to determine whether MDRO rates are decreasing and whether additional interventions are needed. (Strong Recommendation) |
|
Specify isolate origin (i.e., location and clinical service) in MDRO monitoring protocols in hospitals and other large multi-unit facilities with high-risk patients. (Strong Recommendation) |
|
Establish a baseline (e.g., incidence) for targeted MDRO isolates by reviewing results of clinical cultures; if more timely or localized information is needed, perform baseline point prevalence studies of colonization in high-risk units. When possible, distinguish colonization from infection in analysis of these data. (Strong Recommendation) |
|
5. Infection Control Precautions to Prevent Transmission of MDROs |
|
Follow Standard Precautions during all patient encounters in all settings in which healthcare is delivered. (Strong Recommendation) |
|
Use masks according to Standard Precautions when performing splash-generating procedures (e.g., wound irrigation, oral suctioning, intubation); when caring for patients with open tracheostomies and the potential for projectile secretions; and in circumstances where there is evidence of transmission from heavily colonized sources (e.g., burn wounds). Masks are not otherwise recommended for prevention of MDRO transmission from patients to healthcare personnel during routine care (e.g., upon room entry) (Strong Recommendation) |
|
Implement Contact Precautions routinely for all patients infected with target MDROs and for patients that have been previously identified as being colonized with target MDROs (e.g., patients transferred from other units or facilities who are known to be colonized). (Strong Recommendation) |
|
Consider the individual patient’s clinical situation and prevalence or incidence of MDRO in the facility when deciding whether to implement or modify Contact Precautions in addition to Standard Precautions for a patient infected or colonized with a target MDRO. (Good Practice Statement) |
|
For relatively healthy residents (e.g., mainly independent) follow Standard Precautions, making sure that gloves and gowns are used for contact with uncontrolled secretions, pressure ulcers, draining wounds, stool incontinence, and ostomy tubes/bags. (Good Practice Statement) |
|
For ill residents (e.g., those totally dependent upon healthcare personnel for healthcare and activities of daily living, ventilator-dependent) and for those residents whose infected secretions or drainage cannot be contained, use Contact Precautions in addition to Standard Precautions. (Good Practice Statement) |
|
For MDRO colonized or infected patients without draining wounds, diarrhea, or uncontrolled secretions, establish ranges of permitted ambulation, socialization, and use of common areas based on their risk to other patients and on the ability of the colonized or infected patients to observe proper hand hygiene and other recommended precautions to contain secretions and excretions. (Good Practice Statement) |
|
When single-patient rooms are available, assign priority for these rooms to patients with known or suspected MDRO colonization or infection. Give highest priority to those patients who have conditions that may facilitate transmission, e.g., uncontained secretions or excretions. (Strong Recommendation) |
|
When single-patient rooms are not available, cohort patients with the same MDRO in the same room or patient-care area. (Strong Recommendation) |
|
When cohorting patients with the same MDRO is not possible, place MDRO patients in rooms with patients who are at low risk for acquisition of MDROs and associated adverse outcomes from infection and are likely to have short lengths of stay. (Good Practice Statement) |
|
6. Environmental Measures |
|
Clean and disinfect surfaces and equipment that may be contaminated with pathogens, including those that are in close proximity to the patient (e.g., bed rails, over bed tables) and frequently-touched surfaces in the patient care environment (e.g., door knobs, surfaces in and surrounding toilets in patients’ rooms) on a more frequent schedule compared to that for minimal touch surfaces (e.g., horizontal surfaces in waiting rooms). (Strong Recommendation) |
|
Dedicate noncritical medical items to use on individual patients known to be infected or colonized with MDROs. (Strong Recommendation) |
|
Prioritize room cleaning of patients on Contact Precautions. Focus on cleaning and disinfecting frequently touched surfaces (e.g., bedrails, bedside commodes, bathroom fixtures in the patient’s room, doorknobs) and equipment in the immediate vicinity of the patient. (Strong Recommendation) |
- Recommendation
|
Recommendations |
|
1. Administrative Measures |
|
Make MDRO prevention and control an organizational patient safety priority. (Strong Recommendation, Moderate Grade Evidence ) |
|
Provide administrative support, and both financial and human resources, to prevent and control MDRO transmission within the healthcare organization. (Strong Recommendation, Moderate Grade Evidence ) |
|
Implement a multidisciplinary process to monitor and improve healthcare personnel (HCP) adherence to recommended practices for Standard and Contact Precautions. (Strong Recommendation, Moderate Grade Evidence ) |
|
Implement systems to designate patients known to be colonized or infected with a targeted MDRO and to notify receiving healthcare facilities and personnel prior to transfer of such patients within or between facilities. (Strong Recommendation, Moderate Grade Evidence ) |
|
Provide updated feedback at least annually to healthcare providers and administrators on facility and patient-care-unit trends in MDRO infections. Include information on changes in prevalence or incidence of infection, results of assessments for system failures, and action plans to improve adherence to and effectiveness of recommended infection control practices to prevent MDRO transmission. (Strong Recommendation, Moderate Grade Evidence ) |
|
2. Education and Training of Healthcare Personnel |
|
Provide education and training on risks and prevention of MDRO transmission during orientation and periodic educational updates for healthcare personnel; include information on organizational experience with MDROs and prevention strategies. (Strong Recommendation, Moderate Grade Evidence ) |
|
3. Judicious Use of Antimicrobial Agents |
|
In hospitals and long-term care facilities, ensure that a multidisciplinary process is in place to review antimicrobial utilization, local susceptibility patterns (antibiograms), and antimicrobial agents included in the formulary to foster appropriate antimicrobial use. (Strong Recommendation, Moderate Grade Evidence ) |
|
Implement systems (e.g., computerized physician order entry, comment in microbiology susceptibility report, notification from a clinical pharmacist or unit director) to prompt clinicians to use the appropriate antimicrobial agent and regimen for the given clinical situation. (Strong Recommendation, Moderate Grade Evidence ) |
|
Provide clinicians with antimicrobial susceptibility reports and analysis of current trends, updated at least annually, to guide antimicrobial prescribing practices. (Strong Recommendation, Moderate Grade Evidence ) |
|
In settings that administer antimicrobial agents but have limited electronic communication system infrastructures to implement physician prompts, implement a process for appropriate review of prescribed antimicrobials. Prepare and distribute reports to prescribers that summarize findings and provide suggestions for improving antimicrobial use. (Good Practice Statement) |
|
4. Surveillance |
|
In microbiology laboratories, use standardized laboratory methods and follow published guidance for determining antimicrobial susceptibility of targeted (e.g., MRSA, VRE, MDR-ESBLs) and emerging (e.g., VRSA, MDR-Acinetobacter baumannii) MDROs. (Strong Recommendation, Moderate Grade Evidence ) |
|
In all healthcare organizations, establish systems to ensure that clinical microbiology laboratories (in-house and out-sourced) promptly notify infection control staff when a novel resistance pattern for that facility is detected. (Strong Recommendation, Moderate Grade Evidence ) |
|
In hospitals and long-term care facilities, develop and implement laboratory protocols for storing isolates of selected MDROs for molecular typing when needed to confirm transmission or delineate the epidemiology of the MDRO within the healthcare setting. (Strong Recommendation, Moderate Grade Evidence ) |
|
Monitor trends in the incidence of target MDROs in the facility over time using appropriate statistical methods to determine whether MDRO rates are decreasing and whether additional interventions are needed. (Strong Recommendation, High Grade Evidence ) |
|
Specify isolate origin (i.e., location and clinical service) in MDRO monitoring protocols in hospitals and other large multi-unit facilities with high-risk patients. (Strong Recommendation, Moderate Grade Evidence ) |
|
Establish a baseline (e.g., incidence) for targeted MDRO isolates by reviewing results of clinical cultures; if more timely or localized information is needed, perform baseline point prevalence studies of colonization in high-risk units. When possible, distinguish colonization from infection in analysis of these data. (Strong Recommendation, Moderate Grade Evidence ) |
|
5. Infection Control Precautions to Prevent Transmission of MDROs |
|
Follow Standard Precautions during all patient encounters in all settings in which healthcare is delivered. (Strong Recommendation, Moderate Grade Evidence ) |
|
Use masks according to Standard Precautions when performing splash-generating procedures (e.g., wound irrigation, oral suctioning, intubation); when caring for patients with open tracheostomies and the potential for projectile secretions; and in circumstances where there is evidence of transmission from heavily colonized sources (e.g., burn wounds). Masks are not otherwise recommended for prevention of MDRO transmission from patients to healthcare personnel during routine care (e.g., upon room entry). (Strong Recommendation, Moderate Grade Evidence ) |
|
Implement Contact Precautions routinely for all patients infected with target MDROs and for patients that have been previously identified as being colonized with target MDROs (e.g., patients transferred from other units or facilities who are known to be colonized). (Strong Recommendation, Moderate Grade Evidence ) |
|
Consider the individual patient’s clinical situation and prevalence or incidence of MDRO in the facility when deciding whether to implement or modify Contact Precautions in addition to Standard Precautions for a patient infected or colonized with a target MDRO. (Good Practice Statement) |
|
For relatively healthy residents (e.g., mainly independent) follow Standard Precautions, making sure that gloves and gowns are used for contact with uncontrolled secretions, pressure ulcers, draining wounds, stool incontinence, and ostomy tubes/bags. (Good Practice Statement) |
|
For ill residents (e.g., those totally dependent upon healthcare personnel for healthcare and activities of daily living, ventilator-dependent) and for those residents whose infected secretions or drainage cannot be contained, use Contact Precautions in addition to Standard Precautions. (Good Practice Statement) |
|
For MDRO colonized or infected patients without draining wounds, diarrhea, or uncontrolled secretions, establish ranges of permitted ambulation, socialization, and use of common areas based on their risk to other patients and on the ability of the colonized or infected patients to observe proper hand hygiene and other recommended precautions to contain secretions and excretions. (Good Practice Statement) |
|
When single-patient rooms are available, assign priority for these rooms to patients with known or suspected MDRO colonization or infection. Give highest priority to those patients who have conditions that may facilitate transmission, e.g., uncontained secretions or excretions. (Strong Recommendation, Moderate Grade Evidence ) |
|
When single-patient rooms are not available, cohort patients with the same MDRO in the same room or patient-care area. (Strong Recommendation, Moderate Grade Evidence ) |
|
When cohorting patients with the same MDRO is not possible, place MDRO patients in rooms with patients who are at low risk for acquisition of MDROs and associated adverse outcomes from infection and are likely to have short lengths of stay. (Good Practice Statement) |
|
6. Environmental Measures |
|
Clean and disinfect surfaces and equipment that may be contaminated with pathogens, including those that are in close proximity to the patient (e.g., bed rails, over bed tables) and frequently-touched surfaces in the patient care environment (e.g., door knobs, surfaces in and surrounding toilets in patients’ rooms) on a more frequent schedule compared to that for minimal touch surfaces (e.g., horizontal surfaces in waiting rooms). (Strong Recommendation, Moderate Grade Evidence ) |
|
Dedicate noncritical medical items to use on individual patients known to be infected or colonized with MDROs. (Strong Recommendation, Moderate Grade Evidence ) |
|
Prioritize room cleaning of patients on Contact Precautions. Focus on cleaning and disinfecting frequently touched surfaces (e.g., bedrails, bedside commodes, bathroom fixtures in the patient’s room, doorknobs) and equipment in the immediate vicinity of the patient. (Strong Recommendation, Moderate Grade Evidence ) |
Rationale
Multidrug-resistant organisms (MDROs) are an emerging threat to global public health. Inappropriate antibiotic use and lack of infection prevention measures in one facility can affect other facilities because of the interconnected system of health care as patients are transferred among facilities and shared health care providers. MDROs are well recognized by CDC and other public health entities as a concern in health care settings. The potential for rapid spread of MDROs within health care facilities and the difficulties of treating these infections make it critically important for health care personnel to be prepared to prevent and respond to a potential outbreak.
Multi Drug Resistant Organisms are predominantly bacteria resistant to multiple classes of antimicrobial agents.
MDROs include (but are not limited to):
● Methicillin-resistant Staphylococcus aureus (MRSA).
● Vancomycin resistant enterococci (VRE).
● Extended spectrum beta-lactamase producing Enterobacteriaceae (ESBLs).
● Carbapenemase producing Enterobacteriaceae (CPE).
● Other multi drug resistant Gram-negative bacteria (MDRGN).
● Candida auris, a yeast with an emerging concern globally, as it is resistant to Azol group antifungal.
● Administrative measures
Administrative support and involvement are important for the successful control of the target MDROs, authorities in infection control have strongly recommended such support.
Many administrative measures have been associated with a positive impact on prevention of MDROs transmission.
● Education
Educational interventions to encourage a behavior change through improved understanding of the problem MDRO that the facility was trying to control ,enhancing understanding and creating a culture that supported and promoted the desired behavior.
● Judicious Use of Antimicrobial Agents
Effective antimicrobial treatment of infections, use of narrow spectrum agents, treatment of infections and not contaminants, avoiding excessive duration of therapy, and restricting use of broad-spectrum or more potent antimicrobials to treatment of serious infections when the pathogen is not known or when other effective agents are unavailable. Achieving these objectives would likely diminish the selective pressure that favors proliferation of MDROs. Strategies for influencing antimicrobial prescribing patterns within healthcare facilities include education; formulary restriction; prior-approval programs, including pre-approved indications; automatic stop orders. interventions to counteract pharmaceutical influences on prescribing patterns; antimicrobial cycling.
● MDRO Surveillance
Surveillance is a critically important component of any MDRO control program, allowing detection of newly emerging pathogens, monitoring epidemiologic trends, and measuring the effectiveness of interventions.
● Controlling the Spread of MDROs
Take in consideration that both direct patient contact (for example routine patient care) and indirect contact (for example involving environmental contamination) can lead to contamination of the healthcare worker’s hands and clothing.
A 2-level approach is recommended for the prevention and control of MDROs. This involves implementation of:
1. Core strategies for MDRO prevention in all healthcare settings.
2. Organism based or resistance mechanism-based approaches.
1. Core strategies for MDRO prevention in every healthcare setting include
● Standard precautions:
Active surveillance cultures, such as screening for colonization with a target MDRO may fail to identify colonized persons due to a lack of sensitivity of the screening method used, laboratory deficiencies or intermittent patient colonization due to antimicrobial therapy. For this reason, Standard Precautions must be used to limit transmission from potentially colonized patients.
- Hand hygiene is the most important measure to prevent and control the spread of MDROs. Health care personnel, regardless of setting, should perform hand hygiene throughout their shift, especially at key times including:
▶️ Before and after contact with a patient.
▶️ Before an aseptic task, such as inserting an IV or preparing injectable medications.
▶️ Immediately after touching blood, body fluids, non-intact skin, mucous membranes, or contaminated items (even when gloves are worn during contact).
▶️ Immediately after removing personal protective equipment (PPE).
▶️ When moving from contaminated body sites to clean body sites while providing patient care.
▶️ After touching objects and medical equipment in the immediate patient-care vicinity.
▶️ Before eating.
▶️ After using the restroom.
▶️ After coughing or sneezing into a tissue as part of respiratory hygiene.
Hand hygiene supplies should be readily available and easily accessible. Facilities should educate staff on the importance of hand hygiene, as well as when hand hygiene with alcohol-based hand sanitizer versus soap and water should be done. Patients who are colonized or infected with an MDRO should also be educated and encouraged to perform hand hygiene
routinely.
- Proper use of personal protective equipment: single use gloves and isolation gown are required for direct patient contact as well as environmental contact are anticipated
- Environmental hygiene
● Contact precautions:
Contact Precautions are intended to prevent transmission of transmissible organisms, including MDRO whose spread is not interrupted by Standard Precautions alone and have the potential to contaminate the environment. Contact Precautions, in addition to Standard Precautions, should be routinely implemented in all acute healthcare facilities for any patient known to be infected with or colonized with an MDRO.
Contact precautions in this context are generally not appropriate in healthcare settings other than acute hospital in-patient settings.
Patient placement
● Place the colonized or infected patient with MDROs in a single room.
● If single room is not available, cohorting patients with the same MDRO.
● However, do not cohort patients with the same MDRO species if they have a different resistance mechanism or different resistance patterns (different susceptibility to multiple antimicrobial agents).
● CPE positive patients should not be grouped together unless they are known to carry the same type of CPE.
● Priority of isolation when demand for single rooms exceeds availability should take in account risk assessment guided by the IPC and clinical teams, taking into account the clinical needs of the patient, the background epidemiological picture and the risk category of the patient. The highest priority for isolation should be given to those patients who have conditions which may facilitate transmission of an MDRO, i.e. those with uncontained excretions or secretions such as:
o Diarrhea
o Draining wounds
o Incontinence of urine or faeces
o Copious respiratory secretions
o Prioritize patients requiring airborne precautions.
● Factors that should be considered in determining isolation practices include:
o Healthcare facility type: Hospital versus long-term care facility
o Ward type: Non-acute, acute, critical care or other high-risk unit such as haematology, oncology or transplant ward, burns unit, neonatal intensive care unit (NICU)
o Facilities available for patient isolation: single rooms, en-suite toilet facilities, availability of dedicated commodes
o The nature of colonisation of the affected patient (whether the patient is likely to be a heavy disperser of the MDRO via uncontrolled secretions or excretions)
o Resistance pattern, virulence and potential transmissibility of the particular MDRO
● In long-term residential care facilities isolation is generally not appropriate. Standard precautions, including hand hygiene and appropriate use of PPE, for individual residents and environmental contact is preferred.
Patient equipment
● Disposable or patient dedicated equipment is preferred (for example electronic thermometers).
Environmental cleaning
● In acute hospital areas there is evidence that poor environmental hygiene is associated with acquisition of MDRO.
● Regular, cleaning and disinfection of all patients surrounding surfaces specially frequently touched surfaces are important.
Environmental monitoring
● In acute hospital environmental sampling to detect possible environmental reservoirs of MDRO should be performed in addition to visual inspection to identify persistent environmental reservoirs of infection in the context of an outbreak of infection.
● Outbreaks should be managed promptly by an Outbreak Control Team, which has the authority and competence to investigate the outbreak. Laboratory based surveillance system to record the presence of MDROs infections can assist in the timely reporting and notification of cases or outbreaks.
2. Organism based or resistance mechanism-based approaches.
When core strategies fail to control spread of MDRO in a hospital /unit further measure to control transmission should be considered.
Further measures may include:
a. Targeted active surveillance cultures (ASC) for the MDRO to identify people who are colonized and act as reservoirs of MDRO in the health care environment.
b. When ASC are obtained as part of an intensified MDRO control program, implement Contact Precautions until the surveillance culture is reported negative for the target MDRO.
c. Implement contact precautions if surveillance testing is positive for MDRO.
d. Decolonisation - interventions for MRSA may be:
● Topical - whole body washes and topically applied antimicrobial agents.
● Systemic - orally administered antimicrobial agents.
● Combinations of systemic and topical therapy.
e. Surveillance used to monitor the effect of interventions implemented to control spread of MDROs.
f. Surveillance results should be shared with concerned health care workers and hospital management.
Decolonization
Decolonization involves administration of treatment to patients colonized with a specific MDRO to eradicate carriage of that organism. Most healthcare facilities limit the use of decolonization to those patients colonized with MRSA, where evidence exists for this intervention. Although attempts have been made to develop regimens for the decolonization of patients with other MDRO, such as VRE, few have been successful. Currently there are no recommended regimens available for the routine decolonization of patients harboring MDRO other than MRSA
Antimicrobial stewardship
The mise use of antimicrobial is considered the primary driver for development of MDRO. There is a correlation between the levels of antimicrobial resistance and the total quantity of antimicrobials used in a community. For patients, previous antimicrobial therapy is a risk factor for colonization and infection with MDROs.
Preparing to Implement an MDRO Prevention Plan
Step 1: Determine the focus MDROs
Although most prevention strategies are anticipated to reduce spread of all MDROs, health departments should select focus MDROs to inform selection of MDRO Prevention Plan activities, and process and outcome measures. Health departments should include multiple focus MDROs, aiming to include as many targeted MDROs as their resources allow.
If resources initially allow for only one focus MDRO, jurisdictions should consider phased inclusion of additional focus MDROs in subsequent years. Health departments may wish to evaluate, on a yearly basis, whether additional focus MDROs should be added, based on the epidemiology of MDROs within their region and in surrounding regions. Focus MDROs may differ across areas in a single public health jurisdiction to reflect differences in local epidemiology.
Step 2: Risk stratify the healthcare facilities within a jurisdiction
Certain healthcare facility characteristics (e.g., length of patient stay, acuity of care provided, admission and discharge patterns, and IPC program and practices) predict a healthcare facility’s likely role in regional MDRO spread.
Step 3: Decide where to begin MDRO Prevention Plan implementationPrevention activities will be most impactful when they are implemented in all influential and highly connected facilities. Operationally, most health departments will implement prevention plans in phases, generally by piloting implementation in a subset of facilities and then expanding as experience and capacity grow. Selection of facilities for initial implementation should take into consideration local capacity, current epidemiology of focus MDROs, number and type of facilities in the jurisdiction, and health equity.
Step 4: Evaluate jurisdictional clinical laboratory surveillance
MDRO surveillance from both clinical cultures and colonization screening should guide prevention planning and be used to monitor the impact of prevention activities. Based on this evaluation, health departments should work strategically with clinical laboratories to improve detection and reporting of the focus MDROs and resistance mechanisms from both clinical cultures and surveillance testing to best meet their jurisdictional goals.
Including clinical cultures in MDRO detection efforts can augment colonization screening strategies used for early detection and increases opportunities to identify individuals with a targeted MDRO for implementation of appropriate infection control measures.
Step 5: Define process and outcome measures
Health departments should define an overall prevention goal and process and outcome measures for the MDRO Prevention Plan prior to implementation. Progress should be shared regularly (e.g., at least annually) with partners.
Relationship between Prevention and Response Activities
MDRO prevention strategies that is described in Public Health Strategies to prevent the Spread of Novel and Targeted MDROs should be considered in all epidemic stages. These is an ongoing intervention across a group of facilities or geographic regions that are implemented based on the local epidemiology and healthcare facility characteristics. These are intended for pre-endemic stages of spread and are implemented.
Ideally, response activities should be layered on existing prevention interventions. Combining these strategies has the potential to be more effective than either strategy implemented in isolation.
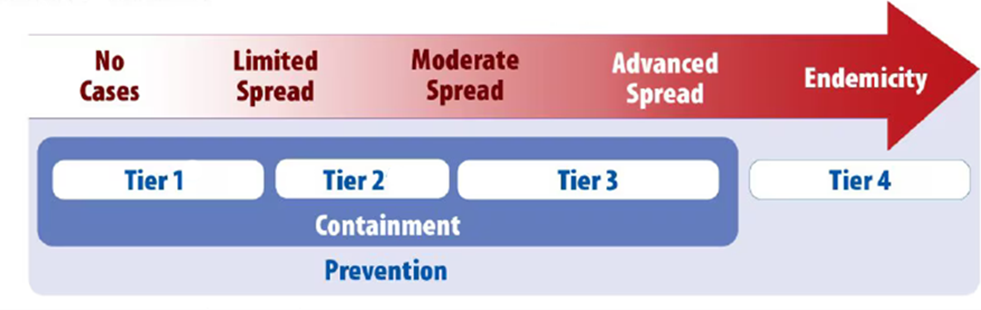
The Relationship between epidemic stages, response tiers, containment response, and prevention activities for novel or targeted MDROs.
● Tier 1 organisms: Includes organisms or resistance mechanisms that have never (or very rarely) been identified in a country.
● Tier 2 organisms: Information is available. or comparable settings about how transmission of these organisms occurs and the groups primarily at risk.
● Tier 3 organisms: Information is available about how transmission of these organisms occurs and the groups primarily at risk. These are MDROs targeted by the facility or region for their clinical significance and potential to spread rapidly (e.g., to other regions where they are less common). Tier 3 MDROs have been identified more frequently across a region than Tier 2 MDRO
● Endemic (Tier 4) organisms: Organisms are endemic in a region. These are MDROs that have been targeted by public health for their clinical significance and potential to spread rapidly.
Table (3): Summary of response Recommendation for MDRO containment by Tier
|
Epidemic stages |
No cases identified limited spread |
Limited to moderate spread |
Moderate to advanced spread |
Epidemic |
|
Containment Tier |
Tier 1 |
Tier 2 |
Tier 3 |
Tier 4 |
|
Tier definition |
Organisms or resistance mechanisms never or very rarely identified in country |
Mechanisms and organisms not regularly found in a region. Pan-not susceptible organisms with the potential for wider spread in a region |
Mechanisms and organisms regularly (i.e frequently) found in a region but not endemic.
|
Mechanisms and organisms that are endemic |
|
Response Elements |
Prioritize prevention; containment principles generally don’t apply |
|||
|
Healthcare Investigation |
||||
|
Review the patient’s healthcare exposures prior to and after the positive culture |
ALWAYS Typical review period: 30 days prior to culture collection to present |
ALWAYS Typical review period: 30 days prior to culture collection to present |
ALWAYS Typical review period: Current admission and sometimes immediately prior admission |
|
|
Contact Investigations |
||||
|
Screening of healthcare contacts (i.e, residents, and patients) |
ALWAYS |
ALWAYS |
USUALLY, |
|
|
Household Contact Screening |
USUALLY, |
RARELY |
RARELY |
|
|
Healthcare Personnel Screening |
USUALLY, |
RARELY |
RARELY |
|
|
Additional Actions if Transmission identified in healthcare |
||||
|
Recurring response-driven point prevalence survey |
ALWAYS |
ALWAYS |
RARELY |
|
|
Evaluate potential spread to healthcare facilities that regularly with the index healthcare facility |
USUALLY, |
USUALLY, |
RARELY |
|
|
Clinical Laboratory Surveillance |
||||
|
Retrospective lab surveillance |
ALWAYS |
ALWAYS |
RARELY |
|
|
Prospective lab surveillance |
ALWAYS |
ALWAYS |
ALWAYS |
|
|
Environmental Cultures |
|
|||
|
Environmental Sampling |
SOMETIMES |
RARELY |
RARELY |
|
|
Infection Control Measures |
|
|||
|
Notify healthcare providers; promptly implement appropriate transmission-based precautions |
ALWAYS |
ALWAYS |
ALWAYS |
|
|
Infection Control Assessment with observations of practice |
ALWAYS |
ALWAYS |
SOMETIMES |
|
|
Clear communication of patient status with transferring facilities |
ALWAYS |
ALWAYS |
ALWAYS |
|
- Acknowledgements
We would like to acknowledge the Infection Control Guidelines Committee for developing these guidelines.
Head of IPC Guidelines Committee
Professor Ghada Ismail (Professor of Clinical Pathology (Clinical Microbiology), Faculty of Medicine, Ain Shams University, Secretary of Supreme IPC Committee, SCUH, Member of WHO Global Guidelines Groups (GDG) for Infection Prevention)
Secretary of IPC Guidelines Committee
Professor Walaa Abd El-Latif (Professor of Medical Microbiology and Immunology, Faculty of Medicine Ain Shams University, IPC Consultant)
Members of the Committee
▪ Professor Amal Sayed (Deputy Manager of Environmental Affair, Infection Control Director, Cairo University Hospitals)
▪ Professor Amani El-Kholy (Clinical Pathology Department (Microbiology), Faculty of Medicine, Cairo University, Infection Control Consultant)
▪ Dr Asmaa Mohamed Abdelfatah Mohamed (Lecturer, Faculty of Nursing MTI University)
▪ Dr Gehan Mohamed Fahmy (Professor clinical microbiology ASUSH consultant infection control, Board member of IFIC EMERO region)
▪ Professor Hebatallah Gamal Rashed (Clinical Pathology Department (Microbiology), Faculty of Medicine, Assuit University, Infection Control Consultant)
▪ Dr Iman Afifi (Consultant Clinical Pathology (Microbiology) and IPC, Ain Shams University, Director IPC units of Ain Shams internal medicine and Geriatric hospitals
▪ Professor Maha El Touny (Department of internal medicine. Faculty of Medicine, Ain Shams University. Infection Control Consultant)
▪ Brigadier Dr. Mohamed Bakr Al-Attar (Assistant Commander of Military Fever Hospital for Therapeutic Affairs, Tropical Medicine Consultant)
▪ Professor Nagwa Khamis (Emeritus Consultant Clinical Pathology (Microbiology) and IPC, ASU Director IPC Department and CEO Consultant IPC, CCHE-57357)
▪ Professor Nesrine Fathi Hanafi (Professor in Medical Microbiology and Immunology Faculty of Medicine Alexandria University, Head of Infection Prevention and Control, Alexandria University Hospitals)
▪ Dr. Reham Lotfy Abdel Aziz (Environmental Health Director, EEAA, Hazardous Waste Consultant, WMRA, Ministry of Environment)
▪ Professor Sherin ElMasry (Professor of Clinical Pathology, Ain Shams University, Chief Director of IPC ASU, Health Care Quality & Patient Safety Consultant)
▪ Dr Shimaa El-Garf (Coordinator): Clinical Pathology Specialist, Coordinator of HAI Surveillance and Audit Electronic System for University Hospitals, RLEUH- SCUH
- List of Abbreviations
1.1.1 ASC: Active Surveillance
Culture
1.1.2 C. difficile: Clostridioides
difficile
1.1.3 CDC: Centers for Disease
Control and Prevention
1.1.4 CPE: Carbapenemase
producing Enterobacteriaceae
1.1.5 ESBL: Extended
spectrum beta-lactamase producing Enterobacteriaceae
1.1.6 HAIs: Healthcare Associated
Infections
1.1.7 GNB: Gram Negative Bacilli
1.1.8 HCP: Healthcare
Personnel
1.1.9 IPC: Infection prevention
and control
1.1.10 HCP: Healthcare Personnel
1.1.11 KPIs: Key Performance
Indicators.
1.1.12 MDR-ESBLs: Multi-drug resistance-
extended-spectrum β-lactamases
1.1.13 MDRGN: Multi
Drug Resistant Gram-negative
1.1.14 MDROs: Multidrug-Resistant
Organisms
1.1.15 MRSA: Methicillin-resistant
Staphylococcus aureus
1.1.16 NICU: Neonatal Intensive Care Unit
1.1.17 PPE: Personal Protective
Equipment
1.1.18 VRE: Vancomycin Resistant
Enterococci.
1.1.19 VRSA: Vancomycin Resistant
Staphylococcus Aureus
1.1.20 WHO: World Health
Organization
- Glossary
● Cohorting: In the context of this guideline, this term applies to the practice of grouping patients infected or colonized with the same infectious agent together to confine their care to one area and prevent contact with susceptible patients (cohorting patients). During outbreaks, healthcare personnel may be assigned to a cohort of patients to further limit opportunities for transmission (cohorting staff).
● Contact Precautions: Contact Precautions are a set of practices used to prevent transmission of infectious agents that are spread by direct or indirect contact with the patient or the patient's environment. Contact Precautions also apply where the presence of excessive wound drainage, fecal incontinence, or other discharges from the body suggest an increased transmission risk. A single patient room is preferred for patients who require Contact Precautions. When a single patient room is not available, consultation with infection control is helpful to assess the various risks associated with other patient placement options (e.g., cohorting, keeping the patient with an existing roommate). In multi-patient rooms, ≥1 meter spatial separation of between beds is advised to reduce the opportunities for inadvertent sharing of items between the infected/colonized patient and other patients. Healthcare personnel caring for patients on Contact Precautions wear a gown and gloves for all interactions that may involve contact with the patient or potentially contaminated areas in the patient's environment. Donning of gown and gloves upon room entry, doffing before exiting the patient room and performance of hand hygiene immediately upon exiting are done to contain pathogens.
● Endemic: The constant presence of a disease or infectious agent within a given geographic area or population group, without importation from outside; may also refer to the usual or expected frequency of the disease within such area or population group.
● Epidemic: Epidemic refers to an increase, often sudden, in the number of cases of a disease above what is normally expected in a specific geographical area. Epidemics occur when an agent and susceptible hosts are present in adequate numbers, and the agent can be effectively conveyed from a source to the susceptible hosts. More specifically, an epidemic may result from:
− A recent increase in the amount or virulence of the agent,
− The recent introduction of the agent into a setting where it has not been before,
− An enhanced mode of transmission so that more susceptible persons are exposed,
− A change in the susceptibility of the host response to the agent, and/or
− Factors that increase host exposure or involve introduction through new portals of entry.
● Hand hygiene: A general term that applies to any one of the following:
− Handwashing with plain (non-antimicrobial) soap and water);
− Antiseptic hand rub (waterless antiseptic product, most often alcohol-based, rubbed on all surfaces of hands); or
− Surgical hand antisepsis (antiseptic hand wash or antiseptic hand rub performed preoperatively by surgical personnel to eliminate transient hand flora and reduce resident hand flora).
● Healthcare-associated infection (HAI): An infection that develops in a patient who is cared for in any setting where healthcare is delivered (e.g., acute care hospital, chronic care facility, ambulatory clinic, dialysis center, surgy center, long term home care) and is related to receiving health care (i.e., was not incubating or present at the time healthcare was provided). In ambulatory and home settings, HAI would apply to any infection that is associated with a medical or surgical intervention performed in those settings.
● Healthcare personnel (HCP): All paid and unpaid persons who work in a healthcare setting, also known as healthcare workers (e.g. any person who has professional or technical training in a healthcare-related field and provides patient care in a healthcare setting or any person who provides services that support the delivery of healthcare such as dietary, housekeeping, engineering, maintenance personnel).
● MDRO: For epidemiologic purposes, MDROs are defined as microorganisms, predominantly bacteria, that are resistant to one or more classes of antimicrobial agents. Although the names of certain MDROs describe resistance to only one agent (e.g., MRSA, VRE), these pathogens are frequently resistant to most available antimicrobial agents. These highly resistant organisms deserve special attention in healthcare facilities. In addition to MRSA and VRE, certain GNB, including those producing extended spectrum beta-lactamases (ESBLs) and others that are resistant to multiple classes of antimicrobial agents, are of particular concern.
● Outbreak: Outbreak carries the same definition of epidemic but is often used for a more limited geographic area. An outbreak is the occurrence of more cases of disease than expected in a given area or among a specific group of people over a particular period of time. Usually, the cases are presumed to have a common cause or to be related to one another in some way.
● Personal protective equipment (PPE): Specialized clothing or equipment worn by an employee for protection against a hazard. General work clothes (e.g., uniforms, pants, shirts) not intended to function as protection against a hazard are not considered to be PPE.
- Introduction
Multidrug-resistant organisms are bacteria, fungi, and other microbes that have developed resistance to multiple. This makes them difficult to treat, leading to increased morbidity, mortality, and healthcare costs. Outbreaks of MDROs in healthcare settings pose a significant threat to patient safety.
Key Management Strategies
1. Infection Prevention and Control (IPC) Measures:
a. Standard Precautions: These are the foundation of infection prevention and apply to all patients, regardless of their diagnosis. They include hand hygiene, proper use of personal protective equipment (PPE), safe injection practices, and respiratory hygiene.
b. Transmission-Based Precautions: These are additional measures implemented for patients with known or suspected infections that can be transmitted through contact, droplets, or airborne routes. They include contact precautions for organisms spread by direct or indirect contact, droplet precautions for organisms spread by respiratory droplets, and airborne precautions for organisms spread by small airborne particles.
2. Antimicrobial Stewardship:
a. Judicious Antibiotic Use: This involves using antibiotics only when necessary, selecting the most appropriate antibiotic for the infection, and using the correct dose and duration of therapy.
b. Antimicrobial Stewardship Programs: These programs involve a team of healthcare professionals who work together to optimize antibiotic use and minimize the development of antibiotic resistance.
3. Surveillance and Outbreak Management:
a. Active Surveillance: This involves routine screening of patients for MDROs, even in the absence of symptoms. This helps to identify carriers and prevent the spread of infection.
b. Outbreak Investigation: When an outbreak of an MDRO occurs, a thorough investigation is conducted to identify the source of the outbreak, determine the extent of transmission, and implement control measures.
4. Environmental Cleaning and Disinfection:
a. Thorough Cleaning: Regular and thorough cleaning of patient rooms, equipment, and other surfaces is essential to prevent the spread of MDROs.
b. Disinfection: Using appropriate disinfectants to kill MDROs on surfaces.
5. Education and Training:
a. Healthcare Workers: All healthcare workers need to be educated about the risks of MDROs and the importance of infection control measures.
b. Patients and Families: Patients and their families should also be educated about the importance of hand hygiene and other infection prevention measures.
Managing MDROs and outbreaks requires a multi-faceted approach that involves a strong commitment to infection prevention and control, judicious antibiotic use, and ongoing surveillance and outbreak management. By implementing these strategies, healthcare facilities can reduce the burden of MDROs and improve patient safety. Specific management strategies will vary depending on the type of MDRO, the healthcare setting, and the specific outbreak situation.- Scope and Purpose
The scope of managing MDROs and outbreak situations generally includes:
● Healthcare Settings: Hospitals, clinics, long-term care facilities, ambulatory care centers, and any other setting where healthcare is provided.
● MDROs: Specific types of MDROs may vary, but commonly include bacteria like MRSA, VRE, and C. difficile, as well as fungi such as Candida auris and other resistant organisms.
● Outbreak Situations: Any instance where the number of cases of an MDRO exceeds what would be expected in a particular setting.
The purpose of managing MDROs and outbreak situations is to:
● Prevent the spread of infection: Protect patients, healthcare workers, and visitors from MDROs.
● Maintain a safe environment: Create a safe and hygienic environment for healthcare delivery.
● Improve patient outcomes: Reduce the morbidity and mortality associated with MDRO infections.
● Comply with infection control guidelines: Adhere to established infection control standards and regulations.
● Optimize resource utilization: Minimize the impact of MDROs on healthcare resources.
By effectively managing MDROs and outbreak situations, healthcare facilities can significantly reduce the risk of healthcare-associated infections (HAIs) and improve the overall quality of patient care.
- Target Audience
● Infection Prevention and Control Team
● All Healthcare Workers Including Clinicians, Nurses, Head Nurses, Environmental Workers, Technicians (Lab – Radiology Department….)
● Department Managers
● Medical Supply Manager
● Isolated Patients
● Visitors
● HAIs Surveillance Officers
● The Treating Physician
● Microbiology Laboratory Manager
● Clinical Pharmacists
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
● Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual Recommendation and their supporting evidence)
● Selecting only national and/or international guidelines
● Specific range of dates for publication (using Guidelines published or updated in 2013 and later)
● Selecting peer reviewed publications only
● Selecting guidelines written in English language
● Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
● Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the Recommendation.
The following characteristics of the retrieved guidelines were summarized in:
● Developing organization/authors
● Date of publication, posting, and release
● Country/language of publication
● Date of posting and/or release
● Dates of the search used by the source guideline developers.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The committee decided to adapt from:
1. Management of Multidrug-Resistant Organisms in Healthcare Settings (2006); Update MARCH 18, 2024 available at https://www.cdc.gov/infection-control/hcp/mdro-management/summary-Recommendation.html
2. MDRO Prevention Strategies, CDC, February 11, 2025
Evidence assessment
According to World Health Organization (WHO) Handbook for Guidelines, we used the GRADE (Grading of Recommendation, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report Recommendation. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent Recommendation. Detailed GRADE information is available on the following sites:
● GRADE working group: https://www.gradeworkinggroup.org/
● GRADE online training modules: http://cebgrade.mcmaster.ca/
Table (1) Quality and Significance of the four levels of evidence in GRADE
|
Quality |
Definition |
Implications |
|
High |
The guideline development group is very confident that the true effect lies close to that of the estimate of the effect |
Further research is very unlikely to change confidence in the estimate effect |
|
Moderate |
The guideline development group is moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibly that it is substantially different |
Further research is likely to have an important impact on confidence in the estimate of the effect and may change the estimate |
|
Low |
Confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the true effect |
Further research is very likely to have an important on confidence in the estimate of effect and is unlikely to change the estimate |
|
Very low |
The group has very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of the effect |
Any estimate of the effect is very uncertain |
Table (2) Factors that determine How to upgrade or downgrade the quality of evidence.
|
Downgrade in presence of |
Upgrade in presence of |
|
Study limitations. 1- Serious limitations 2- Very serious limitations |
Dose- response gradient. +1 Evidence of a dose-response gradient |
|
Consistency 1- Important inconsistency |
Direction of plausible bias + All plausible confounders would have reduced the effect |
|
Directness 1- Some uncertainty 2- Major uncertainty |
Magnitude of the effect +1 Strong, no plausible Confounder, consistent and direct evidence |
|
Precision 1- Imprecise data |
+2 very strong, no major threats to validity and direct evidence |
|
Reporting bias 1. High probability of reporting bias |
|
The strength of the Recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
● Strong Recommendation
With strong Recommendation, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
● Conditional Recommendation
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make Recommendation?
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Indicators for Monitoring
To ensure the effective management of MDROs practices in hospitals and reduce the risk of HAIs, specific indicators should be monitored regularly. These are some indicators which can provide measurable data to assess compliance, identify areas for improvement, and guide interventions. Here are some key indicators that can be included in hospital guidelines for management of MDROs:
● Surveillance and Detection of MDRO Colonization
− Definition: Percentage of patients screened for MDRO on admission
− Formula: (Number of patients screened for MDRO on admission ÷ Total number of patients admitted) × 100
● Antimicrobial Stewardship for MDRO
− Definition: Percentage adherence to antimicrobial stewardship protocols for MDRO cases.
− Formula: (Number of MDRO cases treated in accordance with stewardship guidelines ÷ Total number of MDRO cases) × 100
● Education and Training of HCW
− Definition: Percentage of healthcare workers trained in MDRO management
− Formula: (Number of healthcare workers trained ÷ Total number of healthcare workers) × 100
● Patient and Visitor Involvement
− Definition: Percentage of patients and families educated on MDRO prevention measures
− Formula: (Number of patients educated ÷ Total number of MDRO-positive patients) × 100
● Outcome Measures of MDRO
− Definition: Reduction in healthcare-associated infection rates
− Formula:
o ((Baseline MDRO infection rate − Current MDRO infection rate) ÷ Baseline MDRO infection rate) × 100
o The rate of healthcare- associated MDRO/ 1000 patient days. Example: The rate of HAI- MRSA/ 1000 patient day
1.1 Plan to Update this National Clinical Guideline
This guideline will be reviewed and updated when new evidence emerges that is likely to influence the Recommendation. If no new reports or information are identified for a particular recommendation, the recommendation will be revalidated. The focus will be on Recommendation supported by very-low- or low certainty and conditional grade evidence and where new Recommendation or a change in the published Recommendation may be needed.
- Annex
Annex1. Relationship between epidemic stages, response tiers, containment response, and prevention activities for novel or targeted MDROs. (CDC, 2022)
- References
- Center for Disease Control and Prevention (CDC). Management of Multidrug-Resistant Organisms in Healthcare Settings (2006) Update March 18, 2024.
- MDRO Prevention Strategies [Internet]. Atlanta GA: Center for Disease Control and Prevention; 2024 Feb 11. Available from: https://www.cdc.gov/infectioncontrol/hcp/mdro-management/index.html
- Center for Disease Control and Prevention (CDC). Interim Guidance for a Public Health Response to Contain Novel or Targeted Multidrug-resistant Organisms (MDROs) Atlanta, GA: Center for Disease Control and Prevention (CDC); January 2019.
- Siegel JD, Rhinehart E, Jackson M, Chiarello L. Management of multidrug-resistant organisms in healthcare settings, 2006. Atlanta, GA: Center for Disease Control and Prevention (CDC); 2006.
- Chan M. Ten Years in Public Health, 2007–2017: A Global Health Guardian: Climate Change, Air Pollution and Antimicrobial Resistance. Geneva: World Health Organization (WHO); 2017.
- Palmore TN, Henderson DK. Managing transmission of carbapenem-resistant Enterobacteriaceae in healthcare settings: A view from the trenches. Clin Infect Dis. 2013;57(11):1593–9.
- Trick WE, Lin MY, Cheng-Leidig R, et al. Electronic public health registry of extensively drug-resistant organisms, Illinois, USA. Emerg Infect Dis. 2015;21(10):1725–32.
- Center for Disease Control and Prevention (CDC). Containing Unusual Resistance. CDC Vital Signs. Atlanta, GA: April 2018.
- Tacconelli E, Cataldo MA, Dancer SJ, et al. ESCMID guidelines for the management of the infection control measures to reduce transmission of multidrug-resistant Gram-negative bacteria in hospitalized patients. Clin Microbiol Infect. 2014;20: Suppl 1:1–55.
- Kourtis AP, Hatfield K, Baggs J, et al. Vital signs: Epidemiology and recent trends in methicillin-resistant and in methicillin-susceptible Staphylococcus aureus bloodstream infections - United States. MMWR Morb Mortal Wkly Rep. 2019;68(9):214–9.
- Yokoe DS, Anderson DJ, Berenholtz SM, et al. A compendium of strategies to prevent healthcare-associated infections in acute care hospitals: 2014 updates. Infect Control Hosp Epidemiol. 2014;35(8):967–77.
- Calfee DP, Salgado CD, Milstone AM, et al. Strategies to prevent methicillin-resistant Staphylococcus aureus transmission and infection in acute care hospitals: 2014 update. Infect Control Hosp Epidemiol. 2014;35(7):772–96.