Recommendations on The Clinical Interventions For The Prevention of The Primary Cesarean Section
| Site: | EHC | Egyptian Health Council |
| Course: | Obstetric and Gynecology Guidelines |
| Book: | Recommendations on The Clinical Interventions For The Prevention of The Primary Cesarean Section |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
4 August 2025" Download Guideline
- Executive Summary
This guideline offers evidence-based recommendations on the clinical interventions for the safe prevention of the primary cesarean section. The recommendations are intended to provide healthcare professionals caring for pregnant women with practical guidance on how to safely avoid the primary cesarean section.
List of Recommendations
|
Strength of Recommendation |
|
|
1. Measures During ANC and Before Labor |
|
|
|
|
|
1.1.1. Discuss the benefits and risks of both caesarean and vaginal birth with women, taking into account their circumstances, concerns, priorities and plans for future pregnancies. (See Appendix I) |
GPS
|
|
1.1.2. Fetal presentation should be assessed and documented beginning at 36 0/7 weeks of gestation to guide the plan of management. |
GPS
|
|
1.2. Breech Presentation & Other Fetal Malpresentation |
|
|
1.2.1. Offer women who have an uncomplicated singleton breech pregnancy after 36+0 weeks, external cephalic version by an experienced obstetrician, unless: ➡️ the woman is in established labor ➡️ there is fetal compromise ➡️ the woman has ruptured membranes or vaginal bleeding ➡️ the woman has any other medical conditions (for example, severe hypertension) that would make external cephalic version inadvisable. |
Conditional
|
|
1.2.2. Before carrying out a caesarean birth for an uncomplicated singleton breech pregnancy, carry out an ultrasound scan to check that the baby is in the breech position. Do this as late as possible before the caesarean birth procedure. |
GPS
|
|
1.3. Suspected Fetal Macrosomia |
|
|
1.3.1 Cesarean delivery to avoid potential birth trauma should be limited to estimated fetal weights of at least 4500 g in women without diabetes and at least 4000 g in women with diabetes. |
GPS
|
|
1.4. Twin Gestations |
|
|
1.4.1. Perinatal outcomes for twin gestations in which first twin is in cephalic presentation are not improved by cesarean delivery. Thus, women with either cephalic/cephalic presenting twins or cephalic/non-cephalic presenting twins should be counseled to attempt vaginal delivery. |
Strong
|
|
1.5. Predicting Caesarean Birth for Cephalopelvic Disproportion |
|
|
1.5.1. Do not use pelvimetry for decision making about mode of birth. |
Strong
|
|
1.5.2. Do not use the following for decision making about mode of birth, as they do not accurately predict cephalopelvic disproportion: ➡️ maternal shoe size ➡️ maternal height ➡️ estimations of fetal size (ultrasound or clinical examination). |
GPS
|
|
1.6. Mother-To-Child Transmission of Hepatitis |
|
|
1.6.1 Hepatitis B virus: Do not offer pregnant women with hepatitis B a planned caesarean birth for this reason alone, as mother-to-baby transmission of hepatitis B can be reduced if the baby receives immunoglobulin and vaccination. |
Strong
|
|
1.6.2 Hepatitis C virus: Do not offer women who are infected with hepatitis C a planned caesarean birth for this reason alone. |
Strong
|
|
1.7. Body Mass Index |
|
|
1.7.1. Do not use a BMI of over 50 kg/m2 alone as an indication for planned caesarean birth. |
GPS
|
|
1.8. Cesarean On Maternal Request |
|
|
1.8.1 If a pregnant woman with no medical indication for a caesarean birth requests a caesarean birth: ➡️ discuss and explore the reasons for the request ➡️ address concerns they have about the birth as pain and pain relief options ➡️ discuss the overall benefits and risks of CS birth compared with vaginal birth (See Appendix I) ➡️ Document the discussion and the decision taken |
GPS
|
|
1.8.2. Don’t do cesarean section on request without a written informed consent from the woman indicating that this is a CS on request in absence of a medical or obstetric indication. |
GPS |
|
1.9. Head Engagement in A Primigravida |
|
|
1.9.1 Non-engagement of the head in a primigravida should not be considered as an only factor requiring cesarean birth. |
GPS |
|
2. Measures During Labor |
|
|
2.1. Induction of Labor |
|
|
2.1.1. Before 41 0/7 weeks of gestation, induction of labor generally should be performed based on maternal and fetal medical indications. Inductions at ≥ 41 0/7 weeks of gestation should be performed to reduce risk of cesarean delivery and risk of perinatal morbidity and mortality. |
Strong
|
|
2.1.2 Cervical ripening methods should be used when labor is induced in women with unfavorable cervix.
|
Strong
|
|
2.2. First Stage of Labor: Latent Phase |
|
|
2.2.1 The use of the following definition of the latent phase of the first stages of labor is recommended for practice: The latent first stage is a period of time characterized by: ➡️ Painful uterine contractions and ➡️ variable changes of the cervix, including some degree of effacement and slower progression of dilatation up to 5 cm for first and subsequent labors. |
Strong
|
|
2.2.2 A prolonged latent phase (eg, >20 hours in nulliparous women and >14 hours in multiparous women) should not be an indication for cesarean delivery. |
Strong
|
|
2.2.3. These durations are not recommended as an indication for intervention when maternal and fetal condition are reassuring. |
Strong
|
|
2.2.4. If slow progress is suspected, assess to identify: ➡️ Developing complications ➡️ Reassuring maternal and fetal condition ➡️ Emotional and physical needs |
Strong
|
|
2.3. First Stage of Labor: Active Phase |
|
|
2.3.1. The use of the following definition of the active phase of the first stages of labor is recommended for practice: ➡️ The active first stage is a period of time characterized by regular painful uterine contractions, and a substantial degree of cervical effacement and more rapid cervical dilatation from 5 cm until full dilatation for first and subsequent labors. |
Strong
|
|
2.3.2 Use a partogram with a 4-hour action line to monitor progress of women in spontaneous labor with an uncomplicated singleton pregnancy at term starting in the active phase, to reduce the likelihood of caesarean birth. |
GPS
|
|
2.3.3. Regarding the Partogram: A minimum cervical dilatation rate of 1 cm/hour throughout active first stage is not recommended for identification of normal labor progression and a slower than 1-cm/hour cervical dilatation rate alone should not be a routine indication for obstetric intervention. |
GPS
|
|
2.3.4. A cervical dilatation of 0.5 cm per hour (2 cm in 4 hours) is considered normal in the active phase. |
GPS
|
|
2.3.5. Protracted labor (slower progress than is usual) is diagnosed when in: · Nulliparous women: a cervical dilatation of < 2 cm in 4 hours is found · Multiparous women: a cervical dilatation of < 2 cm in 4 hours or a slowing in the progress of labor If delay is suspected, check descent and rotation of the baby's head and changes in the strength, duration and frequency of uterine contractions (uterine atony). |
GPS
|
|
2.3.6. For women with intact membranes in whom delay in the established first stage of labor is confirmed: ➡️ consider amniotomy if membranes are intact, ➡️ oxytocin if inertia was diagnosed and ➡️ a repeat vaginal examination 2 hours later. ➡️ If oxytocin is used, ensure that the time between increments of the dose is no more frequent than every 30 minutes and increase oxytocin until there are 4–5 contractions in 10 minutes. ➡️ If cervical dilatation has increased by less than 2 cm after 4 hours of oxytocin, review is required to assess the need for caesarean section. |
GPS
|
|
2.3.7. Labor may not naturally accelerate until a cervical dilatation threshold of 5 cm is reached. Therefore, the use of medical interventions to accelerate labor and birth (such as oxytocin augmentation or caesarean section) before this threshold is not recommended, provided fetal and maternal conditions are reassuring. |
Strong
|
|
2.3.8. Arrest in labor (complete cessation of progress) is diagnosed at cervical dilatation of 6 cm or more with ruptured membranes and no or limited cervical change for 4 hours of adequate contractions. |
Strong
|
|
2.3.9. Cesarean delivery for active-phase arrest in first stage of labor should be reserved for women ≥ 6 cm of dilation with ruptured membranes who fail to progress despite 4 hours of adequate uterine activity |
Strong
|
|
2.4. Fetal Heart Rate Monitoring |
|
|
2.4.1. Intermittent auscultation of the fetal heart rate with either a Doppler ultrasound device or Pinard fetal stethoscope is recommended for healthy pregnant women in labor. |
Strong
|
|
2.4.2. Standardization of protocol of the intermittent auscultation is important for health care planning and medico-legal purposes and, therefore, the WHO adopted the following protocol: · Interval: Auscultate every 15–30 minutes in active first stage of labor, and every 5 minutes in the second stage of labor. · Duration: Each auscultation should last for at least 1 minute; if the FHR is not always in the normal range (i.e. 110–160 bpm), auscultation should be prolonged to cover at least three uterine contractions. · Timing: Auscultate during a uterine contraction and continue for at least 30 seconds after the contraction
|
Strong
|
|
2.5. Second stage of labor |
|
|
2.5.1. The use of the following definition and duration of the second stage of labor is recommended for practice: The second stage is the period of time between full cervical dilatation and birth of the baby, during which the woman has an involuntary urge to bear down, as a result of expulsive uterine contractions. |
STRONG
|
|
2.5.2. The duration of the second stage varies from one woman to another: ➡️ In first labor, birth is usually completed within 2 hours ➡️ In subsequent labors, birth is usually completed within 1 hour. |
GPS
|
|
2.5.3. Before diagnosing arrest of labor in second stage, if maternal and fetal conditions permit, allow for: ➡️ At least 1 h of pushing in multiparous women ➡️ At least 2 h of pushing in nulliparous women Longer durations may be appropriate on individualized basis (e.g., with use of epidural analgesia or with fetal malposition) as long as maternal and fetal conditions are reassuring and progress is being documented. |
Strong
|
|
2.5.4. Delay in active second stage is diagnosed when: ➡️ In nulliparous woman (any of): either insufficient flexion/rotation/descent within 1 hour or the second stage duration is > 2 hours. ➡️ In multiparous woman (any of): either insufficient flexion/rotation/descent within 30 minutes or the second stage duration is > 1 hour. Longer durations may be appropriate where maternal and fetal condition is optimal. |
GPS
|
|
2.5.5. A specific absolute maximum length of second stage (passive plus active) has not been identified. Rather than rigid time limits, base decision-making on continuing assessment of: ➡️ Maternal physical and emotional condition ➡️ Fetal condition ➡️ Progress of labor ➡️ Maternal preferences |
GPS
|
|
2.5.6. Operative vaginal delivery in second stage of labor by experienced and well-trained physicians should be considered safe, acceptable alternative to cesarean delivery. Training in, and ongoing maintenance of, practical skills related to operative vaginal delivery should be encouraged. |
Strong
|
- Recommendations
|
1. Measures During ANC and Before Labor |
|
1.1. Planning of Birth |
|
1.1.1. Discuss the benefits and risks of both caesarean and vaginal birth with women, taking into account their circumstances, concerns, priorities and plans for future pregnancies. (See Appendix I) [6, GPS, LQE] Rational: Women are part of the decision making process. They should be well informed of the benefits and risks of each route of delivery. |
|
1.1.2. Fetal presentation should be assessed and documented beginning at 36 0/7 weeks of gestation to guide the plan of management. (GPS, VLQE) |
|
1.2. Breech Presentation & Other Fetal Malpresentation |
|
1.2.1. Offer women who have an uncomplicated singleton breech pregnancy after 36+0 weeks, external cephalic version by an experienced obstetrician, unless: ➡️ the woman is in established labor ➡️ there is fetal compromise ➡️ the woman has ruptured membranes or vaginal bleeding ➡️ the woman has any other medical conditions (for example, severe hypertension) that would make external cephalic version inadvisable. [7, 8, 9, Strong, LQE] Conditions: External Cephalic Version minimizes the need of Cesarean Section in breech presentations but needs experience. Only well trained obstetricians have to do it. |
|
1.2.2. Before carrying out a caesarean birth for an uncomplicated singleton breech pregnancy, carry out an ultrasound scan to check that the baby is in the breech position. Do this as late as possible before the caesarean birth procedure. [6, GPS, LQE] |
|
1.3. Suspected Fetal Macrosomia |
|
1.3.1 Cesarean delivery to avoid potential birth trauma should be limited to estimated fetal weights of at least 4500 g in women without diabetes and at least 4000 g in women with diabetes. [GPS, LQE] |
|
1.4. Twin Gestations |
|
1.4.1. Perinatal outcomes for twin gestations in which first twin is in cephalic presentation are not improved by cesarean delivery. Thus, women with either cephalic/cephalic presenting twins or cephalic/non-cephalic presenting twins should be counseled to attempt vaginal delivery. [10, Strong, MQE] |
|
1.5. Predicting Caesarean Birth for Cephalopelvic Disproportion |
|
1.5.1. Do not use pelvimetry for decision making about mode of birth. [11, Strong, LQE] |
|
1.5.2. Do not use the following for decision making about mode of birth, as they do not accurately predict cephalopelvic disproportion: ➡️ maternal shoe size ➡️ maternal height ➡️ estimations of fetal size (ultrasound or clinical examination). [12, 13, GPS, LQE] |
|
1.6. Mother-To-Child Transmission of Hepatitis |
|
1.6.1 Hepatitis B virus: Do not offer pregnant women with hepatitis B a planned caesarean birth for this reason alone, as mother-to-baby transmission of hepatitis B can be reduced if the baby receives immunoglobulin and vaccination. [6, 14, Strong, LQE] Rational: No randomized controlled trials were done to prove that ECS prevents mother-to-child transmission of HBV. According to the CDC, all infants should receive a dose of hepatitis B vaccine at birth regardless of the HBV infection status of the birth parent. This birth dose, together with the HBIG (given within 12 hours of birth), serves as postexposure immunoprophylaxis for infants born to a parent with HBV infection [15]. |
|
1.6.2 Hepatitis C virus: Do not offer women who are infected with hepatitis C a planned caesarean birth for this reason alone. [6, Strong, VLQE] Rational: There's no evidence that a C-section reduces the risk of transmission, and it is not a standard practice to reduce transmission risk for women with HCV alone. |
|
1.7. Body Mass Index |
|
1.7.1. Do not use a BMI of over 50 kg/m2 alone as an indication for planned caesarean birth. [6, 16. GPS, VLQE] Rational: A study found that obese women with BMI > 50 kg/m2 have a 30-50% risk of wound complications, a 20% risk of NICU admission, and a 1% to 2% risk of maternal intensive care unit admission. |
|
1.8. Cesarean On Maternal Request |
|
1.8.1 If a pregnant woman with no medical indication for a caesarean birth requests a caesarean birth: ➡️ discuss and explore the reasons for the request ➡️ address concerns they have about the birth as pain and pain relief options ➡️ discuss the overall benefits and risks of CS birth compared with vaginal birth (See Appendix I) ➡️ Document the discussion and the decision taken [6, GPS, VLQE] |
|
1.8.2. Don’t do cesarean section on request without a written consent from the woman indicating that this is a CS on request in absence of a medical or obstetric indication. [GPS] Rational: The GDG emphasizes the importance of documenting maternal request of C.S. in absence of medical indications and having a signed informed consent from her explaining all the risks associated with the procedure. |
|
1.9. Head Engagement in A Primigravida |
|
1.9.1 Non-engagement of the head in a primigravida should not be considered as an only factor requiring cesarean birth. [GPS] Rational: In a primigravida as well as a multipara, head engagement can occur as late as the second stage of labor. |
|
2. Measures During Labor |
|
2.1. Induction of Labor |
|
2.1.1. Before 41 0/7 weeks of gestation, induction of labor generally should be performed based on maternal and fetal medical indications. Inductions at ≥ 41 0/7 weeks of gestation should be performed to reduce risk of cesarean delivery and risk of perinatal morbidity and mortality. [17,18, Strong, HQE] |
|
2.1.2 Cervical ripening methods should be used when labor is induced in women with unfavorable cervix. [19,20,21, Strong, MQE] Rational: Many studies showed that the use of cervical ripening methods such as misoprostol, prostaglandin E gel, and other methods lead to lower rates of cesarean delivery than induction of labor without cervical ripening. |
|
2.2. First Stage of Labor: Latent Phase |
|
2.2.1 The use of the following definition of the latent phase of the first stages of labor is recommended for practice: The latent first stage is a period of time characterized by: ➡️ Painful uterine contractions and ➡️ variable changes of the cervix, including some degree of effacement and slower progression of dilatation up to 5 cm for first and subsequent labors. [22, Strong, LQE] |
|
2.2.2 A prolonged latent phase (eg, >20 hours in nulliparous women and >14 hours in multiparous women) should not be an indication for cesarean delivery. [23, Strong, MQE] Rational: Most women with a prolonged latent phase ultimately will enter the active phase with expectant management. With few exceptions, the remainder either will cease contracting or, with amniotomy or oxytocin (or both), achieve the active phase [23]. |
|
2.2.3. These durations are not recommended as an indication for intervention when maternal and fetal condition are reassuring. [23, Strong, MQE] |
|
2.2.4. If slow progress is suspected, assess to identify: ➡️ Developing complications ➡️ Reassuring maternal and fetal condition ➡️ Emotional and physical needs [24,25, Strong, HQE] |
|
2.3. First Stage of Labor: Active Phase |
|
2.3.1. The use of the following definition of the active phase of the first stages of labor is recommended for practice: ➡️ The active first stage is a period of time characterized by regular painful uterine contractions, and a substantial degree of cervical effacement and more rapid cervical dilatation from 5 cm until full dilatation for first and subsequent labors. [22, Strong, LQE] |
|
2.3.2 Use a partogram with a 4-hour action line to monitor progress of women in spontaneous labor with an uncomplicated singleton pregnancy at term, once in active phase, to reduce the likelihood of caesarean birth. [26, GPS, VLQE] Rational: Although quality of evidence for clinical benefit of the partogram is low, yet, the partogram provides a pictorial overview of progress, facilitates timely transfer of care, may assist in the detection of prolonged labor, and if an alert line is used in facilities a four hour action line is recommended for triaging women who may require additional care [26, GPS, VLQE]. |
|
2.3.3. Regarding the Partogram: A minimum cervical dilatation rate of 1 cm/hour throughout active first stage is not recommended for identification of normal labor progression and a slower than 1-cm/hour cervical dilatation rate alone should not be a routine indication for obstetric intervention. Rational: There is insufficient evidence to support the use of the alert line as a classifier to detect women at risk of adverse birth outcomes and lead to unnecessary interventions due to the perception that labor progress is pathologically slow. However, women with suspected slow labor progress should be carefully evaluated to exclude developing complications (e.g. cephalopelvic disproportion) [22, GPS, VLQE] |
|
2.3.4. A cervical dilatation of 0.5 cm per hour (2 cm in 4 hours) is considered normal in the active phase. [27, GPS, LQE] |
|
2.3.5. Protracted labor (slower progress than is usual) is diagnosed when in: ➡️ Nulliparous women: a cervical dilatation of < 2 cm in 4 hours is found ➡️ Multiparous women: a cervical dilatation of < 2 cm in 4 hours or a slowing in the progress of labor If delay is suspected, check descent and rotation of the baby's head and changes in the strength, duration and frequency of uterine contractions (uterine atony). [27, GPS, LQE] |
|
2.3.6. For women with intact membranes in whom delay in the established first stage of labor is confirmed: ➡️ consider amniotomy if membranes are intact, ➡️ oxytocin if inertia was diagnosed and ➡️ a repeat vaginal examination 2 hours later. ➡️ If oxytocin is used, ensure that the time between increments of the dose is no more frequent than every 30 minutes and increase oxytocin until there are 4–5 contractions in 10 minutes. ➡️ If cervical dilatation has increased by less than 2 cm after 4 hours of oxytocin, review is required to assess the need for caesarean section [27, GPS, LQE] |
|
2.3.7. Labor may not naturally accelerate until a cervical dilatation threshold of 5 cm is reached. Therefore, the use of medical interventions to accelerate labor and birth (such as oxytocin augmentation or caesarean section) before this threshold is not recommended, provided fetal and maternal conditions are reassuring. [27, Strong, MQE] |
|
2.3.8. Arrest in labor (complete cessation of progress) is diagnosed at cervical dilatation of 6 cm or more with ruptured membranes and no or limited cervical change for 4 hours of adequate contractions. [28,29, Strong, MQE] |
|
2.3.9. Cesarean delivery for active-phase arrest in first stage of labor should be reserved for women ≥ 6 cm of dilation with ruptured membranes who fail to progress despite 4 hours of adequate uterine activity. [28,29, Strong, MQE] |
|
2.4. Fetal Heart Rate Monitoring |
|
2.4.1. Intermittent auscultation of the fetal heart rate with either a Doppler ultrasound device or Pinard fetal stethoscope is recommended for healthy pregnant women in labor. [30,31, Strong, LQE] |
|
2.4.2. Standardization of protocol of the intermittent auscultation is important for health care planning and medico-legal purposes and, therefore, the WHO adopted the following protocol: · Interval: Auscultate every 15–30 minutes in active first stage of labor, and every 5 minutes in the second stage of labor. · Duration: Each auscultation should last for at least 1 minute; if the FHR is not always in the normal range (i.e. 110–160 bpm), auscultation should be prolonged to cover at least three uterine contractions. · Timing: Auscultate during a uterine contraction and continue for at least 30 seconds after the contraction
[30,31, Strong, LQE] |
|
2.5. Second stage of labor |
|
2.5.1. The use of the following definition and duration of the second stage of labor is recommended for practice: The second stage is the period of time between full cervical dilatation and birth of the baby, during which the woman has an involuntary urge to bear down, as a result of expulsive uterine contractions. [22, Strong, LQE] |
|
2.5.2. The duration of the second stage varies from one woman to another: ➡️ In first labor, birth is usually completed within 2 hours ➡️ In subsequent labors, birth is usually completed within 1 hour. [GPS, VLQE] |
|
2.5.3. Before diagnosing arrest of labor in second stage, if maternal and fetal conditions permit, allow for: ➡️ At least 1 h of pushing in multiparous women ➡️ At least 2 h of pushing in nulliparous women Longer durations may be appropriate on individualized basis (e.g., with use of epidural analgesia or with fetal malposition) as long as maternal and fetal conditions are reassuring and progress is being documented. [33, Strong, MQE] |
|
2.5.4. Delay in active second stage is diagnosed when: ➡️ In nulliparous woman (any of): either insufficient flexion/rotation/descent within 1 hour or the second stage duration is > 2 hours. ➡️ In multiparous woman (any of): either insufficient flexion/rotation/descent within 30 minutes or the second stage duration is > 1 hour. Longer durations may be appropriate where maternal and fetal condition is optimal. [GPS, VLQE] |
|
2.5.5. A specific absolute maximum length of second stage (passive plus active) has not been identified. Rather than rigid time limits, base decision-making on continuing assessment of: ➡️ Maternal physical and emotional condition ➡️ Fetal condition ➡️ Progress of labor ➡️ Maternal preferences [34, Strong, MQE] |
|
2.5.6. Operative vaginal delivery in second stage of labor by experienced and well-trained physicians should be considered safe, acceptable alternative to cesarean delivery. Training in, and ongoing maintenance of, practical skills related to operative vaginal delivery should be encouraged. [35,36,37,38,39, STRONG, MQE] |
- Acknowledgements
The Egyptian Health Council would like to acknowledge the Obstetrics & Gynecology Guidelines Development Committee and all the individuals who participated in developing and adapting this guideline.
Chair of the GDG:
Abdelhamid Mohamed Attia, MD, FRCOG, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Cairo University
Rapporteur of the GDG:
Alaa Eldin Hamed ElFeky, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Ain Shams University
Members of the GDG:
- Aboubakr Mohamed ElNashar, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Banha University
- Ahmed Ezz El-din Mahran, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Minia University
- Ahmed Fawzy Galal, MD, FRCOG, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Alexandria University
- Ahmed Sekotory Mahmoud, MD, FRCOG, FACOF, Consultant of Obstetrics and Gynecology, Private sector
- Amr Abdel Aziz Nadim, MD, Dean, Faculty of Medicine, 6 October University
- Amr Ahmed AboAlyazid, MD, EFOG, Senior Consultant of Obstetrics and Gynecology, Police Medical Services.
- Amr Essam Abdel Rahman, MD, Senior Consultant of Obstetrics and Gynecology, Ain-Shams University
- Diaa Monier Ajlan, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Tanta University
- Ihab Hassan Abdelfataah, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Galala University
- Magdy Ibrahim Mostafa, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Cairo University
- Mervat Ali Elsersy, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Alexandria University
- Mohamed Mahmoud Fathalla, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Assiut University
- Osama Omar Amer, MD, FRCOG, Major General Dr. of Obstetrics and Gynecology, Armed Forces Hospitals
- Taiseer Maarouf Afifi, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine for girls, AlAzhar University
- Wafaa Benjamin Basta, FRCOG, Obstetrics and Gynecology, Mataria Teaching Hospital, MOHP
GDGs coordinator in the EHC: Dr. Mohamed Mourad Youssef, Consultant Obstetrician and Gynecologist, the General Organization of Teaching Hospitals and Institutes.
- Abbreviations
- ANC: Antenatal Care
- ECS: Elective Cesarean Section
- EHC: Egyptian Health Council
- HBIG: Hepatitis B Immunoglobulins
- HBV: Hepatitis B Virus
- HCV: Hepatitis C Virus
- HQE: High Quality of Evidence
- FHR: Fetal Heart Rate
- GDG: Guidelines Development Group
- GPS: Good Practice Statement
- GRADE: Grading of Recommendations Assessment, Development and Evaluation
- LQE: Low Quality of Evidence
- MQE: Moderate Quality of Evidence
- NICU: Neonatal Intensive Care Unit
- QM: Quality Measure
- QS: Quality Statement
- VLQE: Very Low Quality of Evidence
- Introduction
Cesarean section (CS) rates have increased dramatically worldwide, changing the surgical operation from a lifesaving intervention for high-risk and difficult deliveries to a routine surgical procedure. According to the WHO, the global CS rates tripled from 7% in 1990 to 21.1% in 2021, with projections indicating its rise to 28.5% by 2030 [1].
While medically indicated CS prevents maternal and neonatal mortality and morbidity in complicated deliveries, its overuse, without medical indication, exposes mothers and infants, to unnecessary risks and complications and also strains healthcare systems. In fact, studies have shown a significant increase in the maternal and neonatal mortality and morbidity with higher CS rates due to the short‐ and long‐term effects it poses to the mother, baby, and the subsequent pregnancies risks [2].
Egypt is witnessing a skyrocketing rate of CS. In 2014 Egypt ranked 4th worldwide after Dominican Republic, Brazil and Cyprus with a CS rate of 51.8% [3]. This rate is ever increasing and currently Egypt ranks first with the highest CS rate worldwide reaching 72.2% In 2021 [4] far exceeding the WHO recommended and acceptable CS rate which is around 15% above which there were no proven advantages of decreased maternal and neonatal mortality and morbidity over normal VD [5]. The rapid escalation is particularly pronounced in private health facilities, where rates have reached as high as 81% highlighting the urgent need for targeted interventions to address both medical and non-medical drivers of CS in Egypt.
CS increases maternal risks of hemorrhage (intrapartum and postpartum hemorrhage), infections (genital, UT and surgical site infections), organ injury (UT and GIT injury), TED (DVT and pulmonary embolism), anaesthetic complications and ileus on the short term. On the longer term there is higher incidences of placenta previa, placenta accreta spectrum, uterine rupture, hysterectomies and infertility with the development of chronic conditions like endometriosis, pelvic pain, and surgical adhesions [2].
For the neonatal and child health, CS without labor deprives infants of the physiological adaptations critical for extrauterine transition. The short-term consequences include: higher rates of respiratory distress, NICU admissions, and breastfeeding delays. In the longer-term; there is an altered gut microbiome development which increases the risks for non-communicable diseases like obesity, asthma, allergies, and type 1 diabetes. Childhood obesity rates are 40% higher in CS-born children due to microbiota dysbiosis [2].
For the health system efficiency, reducing CS rates is cost saving and allows better resource allocation both in consumables and in man power as it frees operating rooms and staff for high-need emergencies [2].
Reducing unnecessary CS rates is not merely a clinical concern but a multifaceted public health priority. It requires coordinated action, including the implementation of evidence-based clinical guidelines, public and professional education, and systemic reforms to address financial, personal and institutional incentives that may favor surgical delivery over vaginal birth.
This guideline responds to the WHO call for targeted strategies to reverse CS overuse and is aligned with Egypt’s recent policies of improving women’s and strategies for health system reform
- Scope and Purpose
The objectives of this guideline are:
- To provide guidance for the proper indications of CS.
- To provide tools for auditing the rate of CS on personal, institutional and country basis.
- To optimize outcomes for pregnant women, neonates and children based on their mode of delivery.
- To help reduction of the very high CS rate in Egypt.
- Target Audience
This guideline targets mainly healthcare professionals working as Obstetricians & Gynecologists and healthcare personnel working as auditors, medical administrators, hospital managers, and policy makers who work on the field of maternal and child health.
- Methodology
A comprehensive search for guidelines was done to identify the most relevant ones to consider for adaptation. The inclusion/exclusion criteria that were followed in the search and retrieval of guidelines to be adapted are:
We select guidelines only if they are:
- Evidence-based guidelines
- National and/or international guidelines
- Guidelines published from 2014 to 2025
- Peer reviewed publications
- Guidelines written in English language
We Exclude guidelines that are:
- Written by a single author not on behalf of an organization as guideline to be valid and comprehensive ideally requires multidisciplinary input.
- Published without references as the panel needs to know whether a thorough literature review was conducted and whether the current evidence was used in the preparation of the recommendations.
The following characteristics of the retrieved guidelines were summarized in a table:
- Developing organization/authors
- Date of publication, posting, and release
- Country/language of publication
- Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained).
Guidelines used in the adoption and adaptation process:
1. Queensland Clinical Guidelines. Normal birth Guideline No. MN22.25-V5-R27 Queensland Health. 2022. Available from: http://www.health.qld.gov.au/qcg
2. National Institute for Health and Care Excellence (NICE). (2021). Caesarean birth. (NICE guideline, NG192). Published 31 March 2021. Last updated 10 June 2025.
3. WHO recommendations: intrapartum care for a positive childbirth experience. Geneva: World Health Organization; 2018. Licence: CC BY-NC-SA 3.0 IGO.
4. Aaron B. Caughey, Alison G. Cahill, Jeanne-Marie Guise, Dwight J. Rouse. Safe prevention of the primary cesarean delivery. American Journal of Obstetrics & Gynecology, Volume 210, Issue 3, 2014. Pages 179 – 193
Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because they represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
- GRADE working group: https://www.gradeworkinggroup.org/
- GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1: Quality and Significance of the four levels of evidence in GRADE:
|
Quality |
Definition |
Implications |
|
High |
The guideline development group is very confident that the true effect lies close to that of the estimate of the effect |
Further research is very unlikely to change confidence in the estimate of effect |
|
Moderate |
The guideline development group is moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different |
Further research is likely to have an important impact on confidence in the estimate of effect and may change the estimate |
|
Low |
Confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the true effect |
Further research is very likely to have an important impact on confidence in the estimate of effect and is unlikely to change the estimate |
|
Very low |
The group has very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of the effect |
Any estimate of effect is very uncertain |
Table 2; Factors that determine How to upgrade or downgrade the quality of evidence
|
Downgrade in presence of |
Upgrade in presence of |
|
Study limitations - 1 Serious limitations - 2 Very serious limitations |
Dose-response gradient + 1 Evidence of a dose-response gradient |
|
Consistency - 1 Important inconsistency |
Direction of plausible bias + 1 All plausible confounders would have reduced the effect |
|
Directness - 1 Some uncertainty - 2 Major uncertainty |
Magnitude of the effect + 1 Strong, no plausible confounders, consistent and direct evidence + 2 Very strong, no major threats to validity and direct evidence |
|
Precision - 1 Imprecise data |
|
|
Reporting bias - 1 High probability of reporting bias |
|
The strength of recommendations
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations: The GDG found that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted.
Conditional recommendations: This means that the GDG found that there is:
- Greater uncertainty about the strength of evidence, or
- The recommendation may account for a greater variety in patient values and preferences, or
- The resource use makes the intervention suitable for some, but not for other locations.
Conditional recommendations are still the best available evidence to date and it can be adopted if it meets the conditions mentioned with it.
Good Practice Statement: Statements based on opinion of respected authorities, e.g. the RCOG, ACOG, and the guidelines development group.
- Research Needs
➡️ Individuals, organizations, and governing bodies should work to ensure that research is conducted to provide better knowledge base to guide decisions regarding cesarean delivery and to encourage policy changes that safely lower rate of primary cesarean delivery.
➡️ Does auditing cesarean section indications in grand rounds reduces cesarean section rates.
- Implications For Practice
Reducing unnecessary CS rates is not merely a clinical concern but a multifaceted public health priority. It requires coordinated action, including the implementation of evidence-based clinical guidelines, public and professional education, and systemic reforms to address financial and institutional incentives that may favor surgical delivery over vaginal birth.
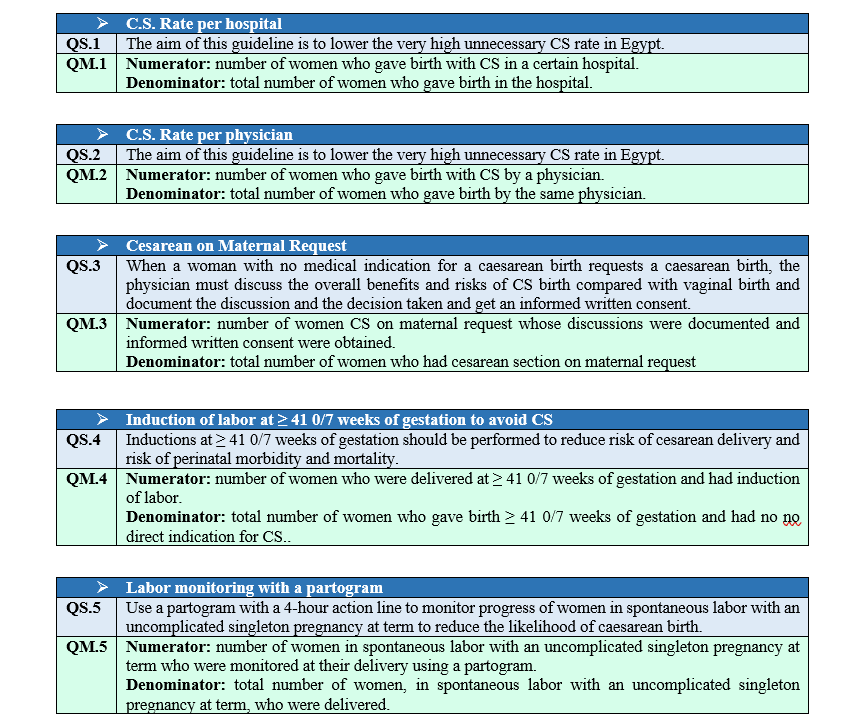
- Clinical Quality Standards for Monitoring
Updating of the guidelines
This guideline will be updated whenever there is new evidence.
- References
1- Ana Pilar Betran, Jiangfeng Ye, Ann-Beth Moller, João Paulo Souza, Jun Zhang - Trends and projections of caesarean section rates: global and regional estimates: BMJ Global Health 2021;6:e005671.
2- Angolile CM, Max BL, Mushemba J, Mashauri HL. Global increased cesarean section rates and public health implications: A call to action. Health Sci Rep. 2023 May 18;6(5):e1274. doi: 10.1002/hsr2.1274. PMID: 37216058; PMCID: PMC10196217.
3- Ministry of Health and Population [Egypt], El-Zanaty and Associates [Egypt], and ICF International. 2015. Egypt Demographic and Health Survey 2014. Cairo, Egypt and Rockville, Maryland, USA: Ministry of Health and Population and ICF International.
4- Central Agency for Public Mobilization and Statistics (CAPMAS). Egypt Family Health Survey 2021. Cairo-CAPMAS-December 2022
5- WHO . WHO statement on caesarean section rates; 2015. Accessed at 20/7/2025 https://www.who.int/publications/i/item/WHO-RHR-15.02
6- National Institute for Health and Care Excellence (NICE). (2021). Caesarean birth. (NICE guideline, NG192). Published 31 March 2021. Last updated 10 June 2025.
7- Clock C, Kurtzman J, White J, Chung JH. Cesarean risk after successful external cephalic version: a matched, retrospective analysis. J Perinatol 2009;29:96-100.
8- American College of Obstetricians and Gynecologists. Mode of term singleton breech delivery: ACOG committee opinion no. 340. Obstet Gynecol 2006;108:235-7.
9- Yoshida M, Matsuda H, Kawakami Y, et al. Effectiveness of epidural anesthesia for external cephalic version (ECV). J Perinatol 2010;30: 580-3.
10- Barrett JF, Hannah ME, Hutton EK, et al. A randomized trial of planned cesarean or vaginal delivery for twin pregnancy: twin birth study collaborative group. N Engl J Med 2013;369: 1295-305.
11- Pattinson RC, Cuthbert A, Vannevel V. Pelvimetry for fetal cephalic presentations at or near term for deciding on mode of delivery. Cochrane Database Syst Rev. 2017 Mar 30;3(3):CD000161. doi: 10.1002/14651858.CD000161.pub2. PMID: 28358979; PMCID: PMC6464150.
12- Gorman RE, Noble A, Andrews CM. The relationship between shoe size and mode of delivery. Midwifery Today 1997;70–1.
13- 112. Hanzal E, Kainz C, Hoffmann G, Deutinger J. An analysis of the prediction of cephalopelvic disproportion. Arch Gynecol Obstet 1993;253:161–6.
14- Hu, Y., Chen, J., Wen, J. et al. Effect of elective cesarean section on the risk of mother-to-child transmission of hepatitis B virus. BMC Pregnancy Childbirth 13, 119 (2013). https://doi.org/10.1186/1471-2393-13-119
15- The center of disease control (CDC). Hepatitis B Perinatal Vaccine Information. Accessed on 25/7/2025 https://www.cdc.gov/hepatitis-b/hcp/perinatal-provider-overview/vaccine-administration.html
16- Smid MC, Dotters-Katz SK, Silver RM, Kuller JA. Body Mass Index 50 kg/m2 and Beyond: Perioperative Care of Pregnant Women With Superobesity Undergoing Cesarean Delivery. Obstet Gynecol Surv. 2017 Aug;72(8):500-510. doi: 10.1097/OGX.0000000000000469. PMID: 28817167.
17- Aaron B. Caughey, Alison G. Cahill, Jeanne-Marie Guise, Dwight J. Rouse. Safe prevention of the primary cesarean delivery. American Journal of Obstetrics & Gynecology, Volume 210, Issue 3, 2014. Pages 179 – 193
18- Gulmezoglu AM, Crowther CA, Middleton P, Heatley E. Induction of labor for improving birth outcomes for women at or beyond term. Cochrane Database Syst Rev 2012;CD004945.
19- Jozwiak M, Bloemenkamp KW, Kelly Mol BW, Irion O, Boulvain M. Mechanical methods for induction of labor. Cochrane Database Syst Rev 2012;3:CD001233.
20- Caughey AB, Nicholson JM, Cheng YW, Lyell DJ, Washington AE. Induction of labor and cesarean delivery by gestational age. Am J Obstet Gynecol 2006;195:700-5.
21- Osmundson S, Ou-Yang RJ, Grobman WA. Elective induction compared with expectant management in nulliparous women with an unfavorable cervix. Obstet Gynecol 2011;117:583-7.
22- WHO recommendations: intrapartum care for a positive childbirth experience. Geneva: World Health Organization; 2018. Licence: CC BY-NC-SA 3.0 IGO.
23- Friedman EA, Sachtleben MR. Amniotomy and the course of labor. Obstet Gynecol 1963;22:755-70.
24- Queensland Clinical Guidelines. Normal birth Guideline No. MN22.25-V5-R27 Queensland Health. 2022. Available from: http://www.health.qld.gov.au/qcg
25- Abalos E, Oladapo OT, Chamillard M, et al. Duration of spontaneous labour in 'low-risk' women with 'normal' perinatal outcomes: A systematic review. Eur J Obstet Gynecol Reprod Biol. 2018 Apr;223:123-132.
26- Dalal AR, Purandare AC. The Partograph in Childbirth: An Absolute Essentiality or a Mere Exercise? J Obstet Gynaecol India. 2018 Feb;68(1):3-14. doi: 10.1007/s13224-017-1051-y.
27- National Institute for Health and Care Excellence (NICE). (2017). Intrapartum care for healthy women and babies. (NICE guideline, NG190).
28- Rouse DJ, Owen J, Hauth JC. Active-phase labor arrest: oxytocin augmentation for at least 4 hours. Obstet Gynecol 1999;93:323-8.
29- Rhoades JS, Cahill AG. Defining and Managing Normal and Abnormal First Stage of Labor. Obstet Gynecol Clin North Am. 2017 Dec;44(4):535-545. doi: 10.1016/j.ogc.2017.07.001. PMID: 29078937.
30- Downe S, Finlayson K, Oladapo OT, Bonet M, Gülmezoglu AM. What matters to women during childbirth: A systematic qualitative review. PloS one 2018;13(4):e0194906
31- Martis R, Emilia O, Nurdiati DS, Brown J.. Intermittent auscultation (IA) of fetal heart rate in labour for fetal well-being. Cochrane Database Syst Rev. 2017
32- Lee L, Dy J, Azzam H. Management of Spontaneous Labour at Term in Healthy Women. J Obstet Gynaecol Can. 2016 Sep;38(9):843-865. doi: 10.1016/j.jogc.2016.04.093. Epub 2016 Jun 25. PMID: 27670710.
33- Spong CY, Berghella V, Wenstrom KD, Mercer BM, Saade GR. Preventing the first cesarean delivery: summary of a joint Eunice Kennedy Shriver National Institute of Child Health and Human Development, Society for MaternaleFetal Medicine, and American College of Obstetricians and Gynecologists workshop. Obstet Gynecol 2012;120:1181-93.
34- American College of Obstetricians and Gynecologists. Committee opinion. Approaches to limit intervention during labor and birth. No. 766. Obstetrics & Gynecology 2017 (reaffirmed 2021);133(2):e164.
35- Towner D, Castro MA, Eby-Wilkens E, Gilbert WM. Effect of mode of delivery in nulliparous women on neonatal intracranial injury. N Engl J Med 1999;341:1709-14.
36- O’Mahony F, Hofmeyr GJ, Menon V. Choice of instruments for assisted vaginal delivery. Cochrane Database Syst Rev 2010;11:CD005455.
37- American College of Obstetricians and Gynecologists. Operative vaginal delivery: ACOG practice bulletin 17. Washington, DC: ACOG; 2000.
38- Powell J, Gilo N, Foote M, Gil K, Lavin JP. Vacuum and forceps training in residency: experience and self-reported competency. J Perinatol 2007;27:343-6.
39- Shaffer BL, Caughey AB. Forceps delivery: potential benefits and a call for continued training. J Perinatol 2007;27:327-8.
- Appendices
Appendix I: Benefits And Risks of Vaginal and Caesarean Birth
|
Outcomes |
Risk with vaginal birth |
Risk with caesarean birth |
|
Outcomes for women that may be more likely with caesarean birth |
||
|
Peripartum Hysterectomy |
100 / 100,000 would be expected to have a peripartum hysterectomy |
200 / 100,000 would be expected to have a Peripartum hysterectomy |
|
Maternal Death |
4 / 100,000 would be expected to die |
25 / 100,000 would be expected to die |
|
Placenta Accreta In Any Future Pregnancy |
30 / 100,000 would be expected to have a placenta accreta in a future pregnancy |
100 / 100,000 would be expected to have a placenta accrete in a future pregnancy |
|
Uterine Rupture In Any Future Pregnancy |
7 / 100,000 would be expected to have a uterine rupture in a future pregnancy |
200 / 100,000 would be expected to have a uterine rupture in a future pregnancy |
|
Outcomes for babies that may be more likely with caesarean birth |
||
|
Neonatal Mortality |
30 / 100,000 would be expected to die |
85 / 100,000 would be expected to die |
|
Asthma |
1,500 / 100,000 children would be expected to have asthma |
1,809 / 100,000 children would be expected to have asthma |
|
Outcomes for women that may be less likely with caesarean birth |
||
|
Urinary incontinence occurring more than 1 year after birth compared to unassisted and assisted vaginal birth |
48,700 / 100,000 women would be expected to have urinary incontinence (unassisted vaginal birth) 19,800 / 100,000 women would be expected to have urinary incontinence (assisted vaginal birth) |
19,600 / 100,000 women would be expected to have urinary incontinence 7,300 / 100,000 women would be expected to have urinary incontinence |
|
Faecal incontinence occurring more than 1 year after birth; compared to assisted vaginal birth |
15,100 / 100,000 women would be expected to have faecal incontinence after assisted vaginal birth |
7,800 / 100,000 women would be expected to have faecal incontinence |
|
Vaginal tear |
560 / 100,000 women would be expected to have a vaginal tear |
0 / 100,000 women would be expected to have a vaginal tear |
|
Perineal/abdominal pain during birth and 3 days after birth |
Median pain scores of 7.3 (during birth) and 5.2 (3 days after birth), 1 is no pain, 10 is most severe pain |
Median pain scores of 1.0 (during birth) and 4.5 (3 days after birth) |
|
Outcomes for women and babies that are likely to be similar for caesarean or vaginal birth |
|
|
Outcomes for women |
Outcomes for babies/children |
|
• thromboembolic disease • major obstetric haemorrhage • postnatal depression • faecal incontinence (occurring more than 1 year after birth; compared to unassisted vaginal birth) |
• admission to neonatal unit • infection • persistent verbal delay • infant mortality (up to 1 year) |
|
Outcomes for women and babies that have conflicting or limited evidence about the risk with caesarean or vaginal birth |
|
|
Outcomes for women |
Outcomes for babies/children |
|
• ITU admission • stillbirth in a subsequent pregnancy. |
• respiratory morbidity • cerebral palsy • obesity (childhood) • autism spectrum condition • type 1 diabetes. |
Source: Adopted from: National Institute for Health and Care Excellence (NICE). (2021). Caesarean birth. (NICE guideline, NG192). Published 31 March 2021. Last updated 10 June 2025.
Disclaimer
This guideline must only be used by licensed personnel who have the proper experience in the field of the guideline and should never be relied upon as a substitute for proper assessment and management with respect to the particular circumstances of each case and the needs of each patient. The applicability of guideline content and suitability to a particular case is the responsibility of the user not the guideline producers.