Sleep apnea syndrome
| Site: | EHC | Egyptian Health Council |
| Course: | Pulmonology Guidelines |
| Book: | Sleep apnea syndrome |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update:
6 August 2025" Download Guideline
- Executive Summary
This guideline offers evidence-based recommendations on the targets of Sleep apnea syndrome. The recommendations are intended to provide healthcare professionals with practical guidance on diagnosis and treatment guidelines of adult obstructive sleep apnea and improving health outcomes for people living with Sleep apnea syndrome.
Recommendations
Diagnosis
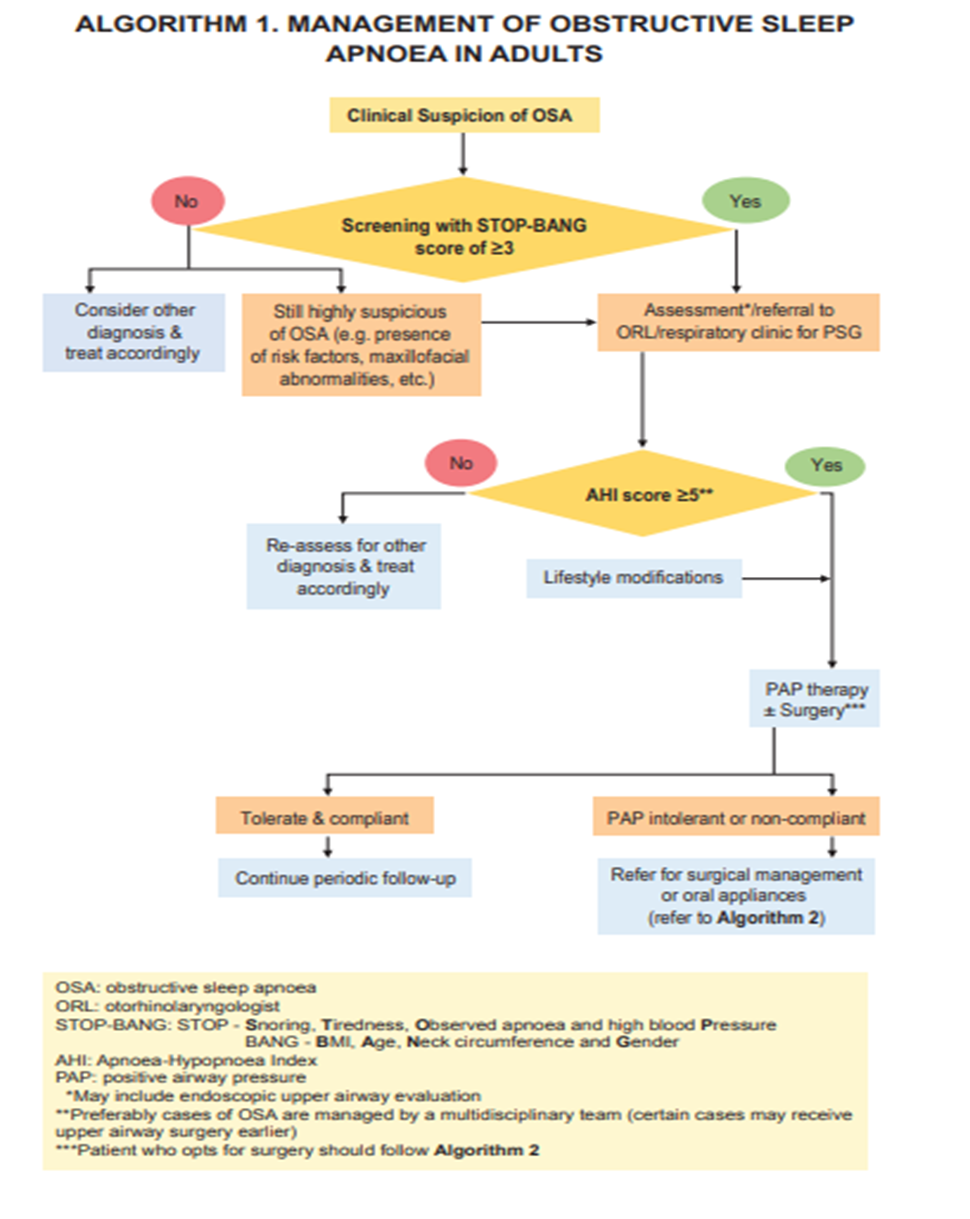
• We recommend either polysomnography or home sleep apnea test be performed to establish diagnosis of obstructive sleep apnea when diagnosis is suspected based on results of comprehensive sleep evaluation which encompasses clinical tools, questionnaires and/or prediction algorithms. (Strong recommendation)
• We recommend either polysomnography or home sleep apnea testing be used to establish diagnosis of obstructive sleep apnea for adult uncomplicated patients who present with signs and symptoms suggesting moderate to severe OSA. (Strong recommendation)
• We recommend that polysomnography be performed to confirm diagnosis of obstructive sleep apnea if home sleep apnea test is negative, inconclusive, or technically inadequate. (Strong recommendation)
• We recommend that polysomnography, rather than home sleep apnea testing, be used for the diagnosis of obstructive sleep apnea in patients with significant cardiorespiratory disease, potential respiratory muscle weakness due to neuromuscular condition, awake hypoventilation or suspicion of sleep related hypoventilation, chronic opioid medication use, history of stroke or severe insomnia. (Strong recommendation)
• We suggest that a second polysomnogram be performed to exclude obstructive sleep apnea when the initial test is negative in patients with clinical suspicion of obstructive sleep apnea. (Conditional recommendation)
• We suggest that, if clinically appropriate, a split-night diagnostic protocol, rather than a full-night diagnostic protocol for polysomnography, be used for the diagnosis of OSA. (Conditional recommendation)
Treatment
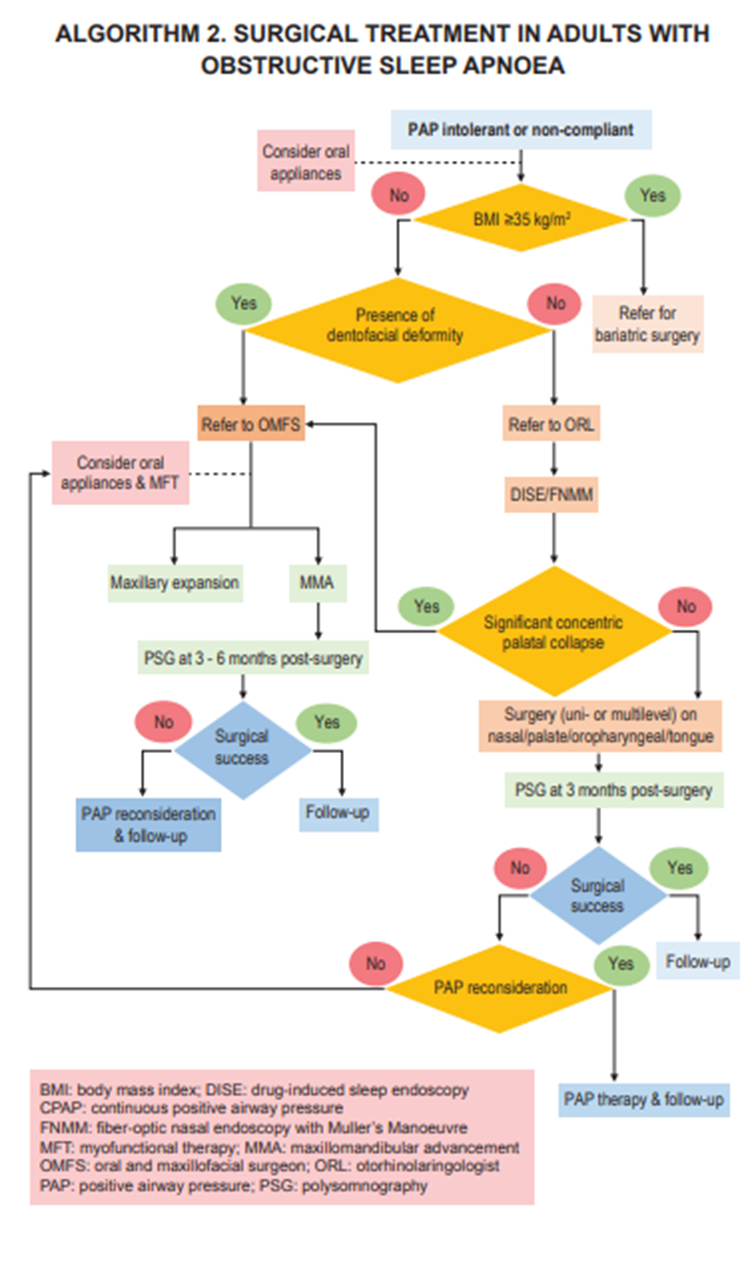
· We suggest that oral appliances to be performed in mild to moderate cases for which CPAP treatment is not indicated, or for cases in which CPAP cannot be used. (Conditional recommendation)
· Weight loss therapy is recommended for obese OSA patients. (Strong recommendation)
· Some OSA patients have apnea alleviated by sleeping in a non-supine position (mainly in a lateral position). We propose that patients with mild cases, along with those who have difficulty with standard treatment such as CPAP treatment, should be instructed regarding their sleeping position, upon confirming that apnea can be reduced in the lateral position. (Conditional recommendation)
· If CPAP or oral appliances cannot be used and there is an indication for otolaryngological surgery, we suggest that it be performed after fully explaining the side effects of the surgery. (Conditional recommendation)
· Oxygen therapy may be performed on patients who cannot use CPAP or oral appliances. (Good Practice Statement)
· If CPAP or oral appliances cannot be used and maxillofacial plastic and reconstructive surgery is indicated, we suggest that it be performed after fully explaining the side effects of the surgery. (Conditional recommendation)
· On the basis of health care professional judgment and patient preference, therapeutic effects are determined by prognosis, reduction of cardiovascular risk factors, effect on complications such as hypertension, quality of life (QOL), effect on subjective symptoms such as drowsiness, and improvement of various indicators in sleep tests. OSA sleepiness improves with proper treatment. (Strong recommendation)
· For OSA patients with insomnia, prioritize treatment of OSA without using hypnotics first. Side effects of hypnotics include an increase in the number of respiratory events and an extension of event time in severe cases. (Conditional recommendation)
· Follow-up PSG or HSAT for routine reassessment of asymptomatic patients with obstructive sleep apnea on PAP therapy, however, follow-up PSG or HSAT can be used to reassess patients with recurrent or persistent symptoms, despite good PAP adherence. (Conditional recommendation)
· Follow-up PSG or HSAT to assess response to treatment with non-PAP interventions. (Strong recommendation)
· Follow-up PSG or HSAT may be used if clinically significant weight gain or loss has occurred since diagnosis of OSA or initiation of treatment. (Conditional recommendation)
· Follow-up PSG may be used for reassessment of sleep-related hypoxemia and/or sleep-related hypoventilation following initiation of treatment for OSA. (Conditional recommendation)
· Follow-up PSG or HSAT may be used in patients being treated for OSA who develop or have a change in cardiovascular disease. (Conditional recommendation)
· Follow-up PSG may be used in patients with unexplained PAP device-generated data. (Conditional recommendation)
- Recommendations
Recommendation
· We recommend either polysomnography or home sleep apnea test be performed to establish diagnosis of obstructive sleep apnea when diagnosis is suspected based on results of comprehensive sleep evaluation which encompasses clinical tools, questionnaires and/or prediction algorithms. (Strong recommendation, Moderate level of evidence)
Remarks
While polysomnography remains the gold standard for the diagnosis of obstructive sleep apnea (OSA), the diagnostic approach of OSA encompasses several steps (1): (i) comprehensive clinical assessment including medical history taking and physical examination of respiratory, cardiovascular, and neurological systems. Sleep history taking should address snoring, witnessed apneas, nocturnal choking or gasping, restlessness, excessive sleepiness. Moreover, special concern should be given to identify OSA-associated conditions such as obesity, hypertension, stroke, and congestive heart failure; (ii) using clinical tools, questionnaires (e.g., Epworth sleepiness scale (2), Berlin Questionnaire (3)) and prediction algorithms (4) which may increase suspicion of OSA through addressing different variables; (iii) polysomnography or home sleep apnea test to confirm diagnosis [1].
Summary of evidence
A wide variety of studies compared the accuracy of clinical tools, questionnaires or prediction algorithms against polysomnography (PSG) or home sleep apnea test (HSAT), and concluded that clinical tools, questionnaires and prediction algorithms are less accurate than PSG and/or HSAT for the diagnosis of OSA at any threshold of AHI consideration. While sensitivity is high, it is not sufficient to adequately exclude OSA. Specificity tends to be lower, resulting in a higher number of false positives. Such results limit the utility of these clinical or morphometric models and highlight the need of PSG and HSAT to establish diagnosis of OSA. The overall quality of evidence was downgraded to moderate due to inconsistency and imprecision of findings [1].
Rationale for the recommendation
Given the low diagnostic accuracy of clinical tools, questionnaires and prediction algorithms for OSA diagnosis, false negative results aggravate decrements in quality of life (QOL), morbidity, and mortality. PSG is widely accepted as the gold standard test for diagnosis of OSA. Further, PSG has been adopted as reference test for comparison to other diagnostic tests, including HSAT. Besides the diagnosis of OSA, PSG can identify co-existing sleep disorders, including other forms of sleep-disordered breathing. In some cases, and within the appropriate context, the use of HSAT as the initial sleep study may be acceptable.
· We recommend either polysomnography or home sleep apnea testing be used to establish diagnosis of obstructive sleep apnea for adult uncomplicated patients who present with signs and symptoms suggesting moderate to severe OSA. (Strong recommendation, Moderate level of evidence)
Remarks
1. Uncomplicated patients refer to patients who lack specific conditions which may compromise the accuracy of home sleep apnea testing (HSAT) for OSA diagnosis. These conditions include: (i) comorbidities which increased risk of developing non-obstructive sleep-disordered breathing (e.g., central sleep apnea, hypoventilation and sleep related hypoxemia). significant cardiopulmonary disease, potential respiratory muscle weakness due to neuromuscular conditions, history of stroke and chronic opiate medication use; (ii) concern for significant non-respiratory sleep disorder(s) that require evaluation (e.g., disorders of central hypersomnolence, parasomnias, sleep related movement disorders) or interfere with accuracy of HSAT (e.g., severe insomnia); (iii) Environmental or personal factors that preclude the adequate acquisition and interpretation of data from HSAT.
2. The following symptoms and signs suggest diagnosis of moderate to severe OSA: excessive daytime sleepiness occurring on most days, AND the presence of at least two of the following three criteria: (a) habitual loud snoring; (b) witnessed apnea or gasping or choking; or (c) diagnosed hypertension.
In this context (when 1 and 2 are met), either PSG or HSAT is used to establish diagnosis of OSA. Selection between both diagnostic tools can be based on patient preferences and available resources. However, in patients who do not meet these criteria, but in whom there is a concern for OSA based on a comprehensive sleep evaluation, only PSG is recommended.
When HSAT is adopted, monitoring is conducted over at least one night with a minimum of four hours of technically adequate oximetry and flow data. [2]
Summary of evidence
Although here is a potential diagnostic misclassification using HSAT when compared against PSG, however there was not clinically significant difference in treatment outcomes after positive airway pressure (PAP) titration as regard patient-reported sleepiness, QOL, and adherence to continuous positive airway pressure (CPAP) [1].
Rationale for the recommendation
PSG represents the gold standard test for diagnosis of OSA given the superior accuracy when compared to HSAT. However, HSAT may possess potential benefits to patients with suspected OSA. First, HSAT can be performed in the home environment with fewer attached sensors during sleep. Accordingly, HSAT is a convenient alternative for patients who cannot leave home or healthcare settings for testing. Second, HSAT requires less complicated infrastructure, hence, promoting access to diagnostic testing in resource-limited settings. Third, HSAT may be less costly when used appropriately. Finally, HSAT shows to provide adequate clinical outcome when performed with sufficient clinical and technical expertise, using the appropriate devices, for the appropriate patient population, and within the appropriate management pathway (Appendix 1)
Nevertheless, HSAT benefits must be weighed against the potential for harm which could result from HSAT limitations: limited sensitivity when compared to PSG; lack of data to support HSAT use in patient with significant comorbidities or when non-obstructive sleep disordered breathing is suspected. In such conditions, additional diagnostic testing can be required for patients with technically inadequate or inconclusive HSAT findings, or from misdiagnosis and subsequent inappropriate therapy or lack of therapy.
• CPAP treatment is effective for OSA, with CPAP treatment recommended as the first choice for patients with strong clinical symptoms such as daytime sleepiness due to OSA, and for moderate to severe cases. (Strong recommendation) [High level of evidence]
Remarks
CPAP treatment has been reported to improve prognosis and many other related pathologies. A hypotensive effect is seen through CPAP treatment, with a hypotensive effect expected especially in treatment-resistant hypertension.
Summary of evidence
Continuous positive airway pressure (CPAP) is the most effective treatment for adults.[1] Bilevel PAP is also better tolerated by patients who require higher pressure settings (>15 cm H2O). However, despite the high efficacy of CPAP in eliminating respiratory events, its effectiveness is dampened by the decreased use of treatment during sleep and inadequate adherence. Adherence to CPAP among patients with OSA remains a significant challenge, as nearly half of the patients do not adequately adhere to treatment after the first month.[2]
The American Thoracic Society published a recent statement on CPAP adherence tracking systems and the optimal monitoring strategies and outcome measures in adults.[3] Standardizing the CPAP adherence report not only the number of hours used more than 4 hours per night (>70% of nights) but also the amount of mask leak and residual apnea and hypopnea index is important. However, what is the optimal goal in adherence to OSA treatment? Recent studies are looking at the utility of telemedicine adherence interventions, remote monitoring of CPAP, and more interactive features with individual patients and their families have been shown to increase CPAP adherence rates.[4][5][6][7]
Several study results have reported conflicting findings when assessing the effect of CPAP therapy on cardiovascular outcomes in patients with OSA.[8] In a recent randomized control trial, CPAP use for a minimum of 1 year in patients with acute coronary syndrome and OSA without excessive daytime sleepiness did not lower the incidence of cardiovascular events (defined as cardiac-related death or 1 or more of the following outcomes: acute myocardial infarction, non-fatal stroke, hospital admission for heart failure, and new hospitalizations for unstable angina or transient ischaemic attack). The adherence to CPAP therapy was low (2.78 h/night), and follow-up was not long enough, which are significant limitations of this study.[9]
Rationale for the recommendation
In another observational cohort study with long-term follow-up, CPAP use was associated with lower all-cause mortality among patients with severe OSA around years 6 to 7 of follow-up.[10]
In a more recent study, patients with coronary artery disease and OSA without excessive sleepiness who exhibited greater changes in heart rate benefited more from CPAP therapy.[11]
• We suggest that oral appliances to be performed in mild to moderate cases for which CPAP treatment is not indicated, or for cases in which CPAP cannot be used. (Conditional recommendation) [Moderate level of evidence]
QOL can be expected to improve in certain factors by using oral appliances, with some cardiovascular risk factors also improving.
Summary of evidence
For patients unable or unwilling to use CPAP or those unable to access electricity reliably, custom-fitted and titrated oral appliances or mandibular advancement devices (MAD) can bring the lower jaw forward and relieve airway obstruction. This typically works best for candidates with appropriate dentition and mild to moderate sleep apnea. In a randomized clinical trial on 126 patients with moderate-severe OSA, the 24-hour mean arterial pressure was similar between CPAP and MAD after 1-month of therapy. MAD was superior to CPAP for improving quality of life measures.[12] More recently, another randomized clinical trial demonstrated similar long-term improvement for CPAP and MAD in self-reported neurobehavioral outcomes during a 10-year follow-up.[13]
Rationale for the recommendation
The American Academy of Sleep Medicine (AASM) and the American Academy of Dental Sleep Medicine (AADSM) developed guidelines for using MAD in patients with OSA.[14] The AASM/AADSM guidelines recommend the following:
· Oral appliances can be considered rather than no treatment for adult patients with snoring (without OSA) or those with OSA who do not tolerate CPAP therapy or prefer alternate treatment.
· When a sleep physician prescribes oral appliance therapy for an adult patient with obstructive sleep apnea.
· A qualified dentist should use a custom, titratable appliance.
· A follow-up with a qualified dentist after oral appliance therapy is initiated in adult patients with OSA to assess for dental-related side effects.
· A follow-up with sleep testing to confirm treatment efficacy is necessary.
· Weight loss therapy is recommended for obese OSA patients. (Strong recommendation) [Low level of evidence]
· Some OSA patients have apnea alleviated by sleeping in a non-supine position (mainly in a lateral position). We propose that patients with mild cases, along with those who have difficulty with standard treatment such as CPAP treatment, should be instructed regarding their sleeping position, upon confirming that apnea can be reduced in the lateral position. (Conditional recommendation) [Very low level of evidence level]
Remarks
Apnea can be reduced for obese patients by providing them with standard treatment for OSA, including weight loss guidance. Weight loss may improve quality of life and cardiovascular risk factors. The devices used for postural therapy have not been standardized. For patients with mild cases or who have difficulty with standard treatment such as CPAP, instructions are provided regarding their sleeping position, upon confirming that apnea is reduced in the lateral position.
Summary of evidence
The importance of weight loss should be emphasized in patients with OSA who are overweight and obese.[15][16] Although weight loss is recommended and can often decrease the severity of obstructive sleep apnea, it is not usually curative. Patients should be educated on the impact of sleep duration on their health and prioritize getting at least 7 to 8 hours of sleep per night.[17]
OSA that is more prominent in the supine position can be treated with a positioning device to keep a patient on their side, which can be an option.[18][19]
Rationale for the recommendation
Three meta-analyses showed that lifestyle intervention which include diet, exercise or combination of both, improved both AHI and BMI in adults with OSA.
• In the first meta-analysis weight loss through diet and/or physical
activity decreased AHI compared with control (WMD= -6.04/hour,
95% CI -11.18 to -0.90). However, diet alone did not show any significant difference in the outcome.[20]
• The second meta-analysis also supported the finding where intensive lifestyle interventions reduced weight (WMD= -13.76 kg,
95% CI -19.21 to -8.32) and improved both AHI (WMD= -16.09/hour, 95% CI -25.64 to -6.54)) and ODI (WMD= -14.18, 95% CI -24.23 to -4.13). [21]
• In the recent meta-analysis, combination of diet and exercise improved AHI (WMD= -8.09/hour, 95% CI -11.94 to -4.25) and BMI (WMD= -2.41 kg/m2, 95% CI -4.09 to -0.73). Apart from that, diet alone improved both AHI (WMD= -8.61/hour, 95% CI -15.89 to -1.33) and BMI (WMD= -3.97, 95% CI -5.77 to -2.18), while exercise alone only improved AHI (WMD= -8.08/hour, 95% CI -15.78 to -0.42) but not BMI. [22]
• If CPAP or oral appliances cannot be used and there is an indication for otolaryngological surgery, we suggest that it be performed after fully explaining the side effects of the surgery. (Conditional recommendation) [Low level of evidence]
• Oxygen therapy may be performed on patients who cannot use CPAP or oral appliances. (Good Practice Statement) [Low level of evidence]
• If CPAP or oral appliances cannot be used and maxillofacial plastic and reconstructive surgery is indicated, we suggest that it be performed after fully explaining the side effects of the surgery. (Conditional recommendation) [Low level of evidence]
Remarks
Otorhinolaryngological surgery and maxillofacial plastic and reconstructive surgery can be expected to improve QOL in OSA patients under certain circumstances and AHI may improve following surgery, which may improve cardiovascular disorders.
For cases in which CPAP and OA cannot be used, oxygen therapy is possible if desired. That said, in the event a patient with chronic heart failure has been diagnosed, Cheyne-Stokes respiration during sleep is observed, and polysomnography has confirmed an AHI of 20 or higher.
Summary of evidence
Uvulopalatopharyngoplasty (UPPP) surgically removes the uvula and tissue from the soft palate to create more space in the oropharynx.[23] This is sometimes done in conjunction with a tonsillectomy and adenoidectomy. Nevertheless, the long-term efficacy of UPPP is very limited, with less than 50% of patients having a significant increase in the apnea-hypopnea index after the first year.[24]
Maxillomandibular advancement (MMA) requires both the upper and lower jaws to be detached and surgically advanced anteriorly to increase space in the oropharynx.[25] This is best for patients with retrognathia and is less successful in older patients or those with larger neck circumferences. More recently, drug-induced sleep endoscopy has been used for preoperative planning to identify multiple levels of obstruction in these patients and candidacy for surgical treatment such as MMA and hypoglossal nerve stimulator.[26] This allows surgeons to address any nasal, soft palate, and hypopharyngeal obstructions that may be present during a single surgery.[27]
A newer option is the implantable hypoglossal nerve stimulator (HNS), usually implanted unilaterally, although bilateral implantation has been recently reported.[28] This instrument works by stimulating the genioglossus (upper airway dilator muscle) during apneas, resulting in tongue protrusion and relief of the obstruction.[29]
HNS effectively reduces AHI (median AHI score at 12 months decreased by 68%, from 29.3 events per hour to 9.0 events per hour) and improves sleepiness symptoms in those with moderate to severe OSA who are not tolerating CPAP treatment.[30]
Adverse events reported short- and long-term following HNS are not very common. In one study, 134 adverse events were reported from 132 patient reports over 5 years.[31] The most common adverse events reported after HNS are tongue abrasion (11.0%), pain (6.2%), and device malfunction (3% to 6%).[28]
The eligibility criteria for HNS adopted from the original randomized trial include the following characteristics:
· Adults older than 18 years
· Moderate to severe OSA (AHI between 20 to 50 with <25% central or mixed apneas)
· Inability to tolerate CPAP
· No complete concentric collapse at the palate on drug-induced sleep endoscopy [29]
Exclusion criteria for HNS include the following:
Body mass index greater than 32.0 kg/m2
Neuromuscular disease
Hypoglossal-nerve palsy
Severe restrictive or obstructive pulmonary disease
Moderate-to-severe pulmonary arterial hypertension
Severe valvular heart disease
Heart failure, New York Heart Association class III or IV
Recent myocardial infarction or severe cardiac arrhythmias (within the past 6 months)
Persistent uncontrolled hypertension despite medication use
Active psychiatric disease and coexisting nonrespiratory sleep disorders
In extreme cases, OSA can also be treated with a tracheostomy to bypass the oropharyngeal obstruction. This management option is also best addressed at academic or specialty sleep centers that are experienced in treating patients with tracheostomy. Such patients encounter numerous challenges with home care, durable medical equipment, and family/partner education on tracheostomy management. Additionally, many patients with severe OSA requiring tracheostomy have comorbidities.
Rationale for the recommendation
Maxillomandibular advancement (MMA) is one of the surgical
interventions for patients with moderate to severe OSA. In a small RCT comparing MMA and CPAP in 50 patients with severe OSA, there was no significant difference in improvement of AHI and ESS.[31] However, there was no explanation of randomisation or type of analysis done. MMA with or without adjunctive surgical procedures had been shown to be an effective and predictable surgical treatment option for patients with moderate to severe OSA with a surgical success rate of 86% and cure rate ranging from 38 - 50%. [32,33]
A recent meta-analysis of eight clinical trials on moderate to severe
OSA showed that MMA [34]
• achieved surgical success with mean final AHI of 12.4/hour (95%
CI 7.18 to 17.6)
• increased posterior airway volume (PAV) which correlated with
improvements in postoperative AHI (r=0.75, 95% CI 0.65 to 0.85)
In another meta-analysis which examined the impact of MMA in OSA, it was shown that [35]
• upper airway volume increased in the vertical position (MD=8.909 mm3, 95% CI 6.614 to 11.204) and supine position (MD=6.047 mm3, 95% CI 5.539 to 6.565)
• AHI reduced (MD=45.596/hour, 95% CI 50.375 to 40.818)
• RDI reduced (MD=50.474/hour, 95% CI 63.680 to 37.089)
• oxygen saturation increased (MD=8.990%, 95% CI 5.205 to 12.775)
• ESS reduced (MD= -10.491, 95% CI -12.519 to -8.464)
There is no strong evidence to establish the magnitude and direction of maxillary or mandibular movement required to cure OSA and thus this should be tailored to each individual patient based on clinical assessment.
The stability of improvement in airway parameters with MMA has been reported to be sustained significantly over a mean follow-up of 10 months. [36]
The revised Stanford Protocol (2019) state that the following patients can benefit from MMA [37]
• patients with OSA and concurrent dentofacial deformity
those who have a co-existing indication for orthognathic/corrective jaw surgery are indicated for proceeding directly to MMA surgery to concurrently improve speech and mastication function
• patients with moderate to severe OSA without dentofacial
deformity with:
- complete lateral pharyngeal wall collapse on DISE
- low hyoid position and obtuse cervicomental angle
- high inclination of the occlusal plane
Potential complications of MMA have been reported to be low but can be divided into [38]
• intraoperative complications
- Bleeding due to injury to the descending palatine artery during
maxillary down fracture
- Risk of unfavorable fracture patterns due to highly cortical bone
• early postoperative complications
- Airway obstruction which should be acutely recognized; moderate to severe OSA patients must be monitored in the ICU after extubation until the airway is confirmed to be secured
- Due to the large movements, there is increased risk of wound dehiscence and exposure of plate leading to possible infected plates requiring close postoperative monitoring
• late postoperative complications
- malunion of bony segments
- hardware issues relating to chronic sinusitis, acute infection or
persistent pain
- relapse of the OSA symptoms which require revision surgery
• On the basis of health care professional judgment and patient preference, therapeutic effects are determined by prognosis, reduction of cardiovascular risk factors, effect on complications such as hypertension, quality of life (QOL), effect on subjective symptoms such as drowsiness, and improvement of various indicators in sleep tests. OSA sleepiness improves with proper treatment. (Strong recommendation) [Moderate level of evidence]
• For OSA patients with insomnia, prioritize treatment of OSA without using hypnotics first. Side effects of hypnotics include an increase in the number of respiratory events and an extension of event time in severe cases. (Conditional recommendation) [Low level of evidence]
Remarks
It is believed that OSA patients may have reduced QOL mainly due to daytime sleepiness. Because QOL is inherently a comprehensive and multifaceted index that includes sleep quality itself, it is difficult to determine whether QOL is decreased by OSA alone. It can be said that there is consensus on the improvement of subjective sleepiness when OSA is treated with CPAP [39].
Summary of evidence
Many research papers using SF-36 (36-Item Short Form Health Survey), which is considered the gold standard, have been reported as an index of QOL. SF-36 consists of eight subscales, and is roughly divided into physical factors and mental factors. According to a systematic review; that is, report 2 reported in 2017, CPAP therapy was reported to significantly improve QOL compared to the control for physical factors [40,41], while report 1 reported that it improved mental factors significantly [40], although other reports report that there was no significant difference [41]. Similar meta-analyses in the past also state that the QOL, as evaluated by SF-36, was improved by CPAP [42,43, 44]; however, these were mixed with other QOL indicators or deal with only some sub-indexes of SF-36, which is problematic.
Rationale for the recommendation
On the other hand, from the viewpoint of mental factors, many research papers evaluating indicators of anxiety and depression have been reported. Although two meta-analyses both concluded that CPAP can improve anxiety and depression [42,43] it has been reported that the indicators used vary, and that the effect is high when targeting people having a strong tendency to be depressed; therefore, it is not wise to generalize. It should also be noted that these psychological subjective assessments are subject to therapeutic intervention, with contact with medical staff itself tending to have a positive effect.
QOL indexes other than SF-36 include the SAQLI (Sleep Apnea Quality of Life Index) and FOSQ (Functional Outcomes of Sleep Questionnaire), which specialize in sleep quality. According to a meta-analysis that combines these, CPAP therapy significantly improves QOL compared to the control group, a result which is not affected by the presence or absence of subjective sleepiness indicated by the Epworth Sleepiness Scale (ESS) [41].
It is difficult to determine if there is improvement of QOL based on evidence because the conclusion changes depending on what is used as an index. However, improvement of QOL that cannot be explained by sleepiness alone, as an evaluation of sleep, is expected, as well as physical performance during the day regardless of sleep, which may lead to the elimination of adverse mental effects such as anxiety and depressed mood.
We believe that CPAP therapy can be proposed to improve QOL with moderate confidence based on evidence from meta-analyses.
· Follow-up PSG or HSAT for routine reassessment of asymptomatic patients with obstructive sleep apnea on PAP therapy, however, follow-up PSG or HSAT can be used to reassess patients with recurrent or persistent symptoms, despite good PAP adherence (Conditional recommendation) [Moderate level of evidence]
Remarks
There is insufficient evidence to suggest that routine PSG and/or HSAT should be performed in patients who are responding adequately to PAP treatment as determined by symptom control and adherence to therapy. [45,46,47]
If symptoms return despite good adherence to PAP treatment, PSG or HSAT is appropriate. If there is an unexplained change in adherence, or there is clinical suspicion of a separate sleep disorder, then a repeat PSG may be considered, and might include PAP titration. [48] If there is suspicion of another sleep disorder such as narcolepsy, then PSG should precede other testing, such as a multiple sleep latency test.
· Follow-up PSG or HSAT to assess response to treatment with non-PAP interventions (Strong recommendation) [Moderate level of evidence]
Remarks
PAP therapy remains the first-line therapy for most adults with OSA. [48] For patients who are intolerant to PAP or who prefer alternative therapies, several non-PAP treatments for OSA are available, including oral appliance therapy, nasal expiratory positive airway pressure, upper airway surgery, positional therapy, oral pressure therapy, weight loss and hypoglossal nerve stimulation.
Following implementation of non-PAP interventions for OSA, clinical reassessment should include follow-up testing to ensure successful treatment of OSA with the chosen treatment at the appropriate time interval. Follow-up testing is not necessary for positional therapy if it was proven effective on the diagnostic study. As to the type of test, PSG vs HSAT, multiple factors should be considered, including comorbidities (eg, hypoventilation, advanced cardiopulmonary disease), patient comfort, ability to wear/operate the HSAT equipment, cost of testing, and resource utilization.
Rationale for the recommendation
Oral appliances require fitting by a dental sleep specialist, and serial advancement of the mandible to promote airway opening. [49,50] Follow-up PSG or HSAT should be performed following patient acclimatization, reviewed by a trained sleep medicine specialist and necessary adjustments should be made to the device to ensure adequate control of OSA. [51]
· Follow-up PSG or HSAT may be used if clinically significant weight gain or loss has occurred Since diagnosis of OSA or initiation of treatment (Conditional recommendation) [Moderate level of evidence]
· Remark
Auto-adjusting PAP, which is considered to be therapeutically equivalent to fixed CPAP in uncomplicated OSA, [45] may obviate the need for repeat testing in certain circumstances of weight change.
· Follow-up PSG may be used for reassessment of sleep-related hypoxemia and/or sleep-related hypoventilation following initiation of treatment for OSA (Conditional recommendation) [Moderate level of evidence]
Remarks
Physiologic mechanisms resulting in hypoxemia while awake or asleep include residence at high-altitude (or other causes of low fraction of inspired oxygen, FiO2), ventilation-perfusion mismatch, pulmonary diffusion abnormality, shunting and hypoventilation.[52]
The most commonly associated conditions found in the sleep center include ventilation-perfusion mismatch (resulting in hypoxemia with/or without hypercapnia) and hypoventilation (resulting in hypercapnia with/or without hypoxemia). Some patients with OSA, particularly those with underlying cardiopulmonary disease, may have hypoxemia or hypoventilation that persists or develops following initial implementation of treatment such as PAP therapy.
· Follow-up PSG or HSAT may be used in patients being treated for OSA who develop or have a change in cardiovascular disease. (Conditional recommendation) [Moderate level of evidence]
· Remarks
Various cardiovascular (CV) diseases are strongly linked to OSA, with increased risk of conditions such as systemic hypertension, heart failure (HF), cardiac arrhythmias, stroke and sudden death. [53]
Rational of Evidence
Many patients are referred for evaluation and treatment of OSA to potentially improve CV outcomes. For initial diagnosis of suspected sleep apnea, current guidelines strongly recommend attended PSG over HSAT for patients with HF and/or history of stroke, since such patients are at risk for nonobstructive sleep-disordered breathing (central sleep apnea, sleep-related hypoxemia). For CV conditions other than HF and stroke, where the risk of nonobstructive sleep apnea is less, either PSG or HSAT is recommended. [54]
· Follow-up PSG may be used in patients with unexplained PAP device-generated data. (Conditional recommendation) [Moderate level of evidence]
Remarks
Monitoring automated event detection (AED) has become common practice in the management of patients with OSA treated with PAP devices. Event data can be downloaded by smartcard or displayed on the internet for viewing by patients, durable medical equipment companies, and practitioners. [55,56]
Rationale of Evidence
Some studies have shown a correlation between PAP-scored AHI and manual-scored AHI. [57,58] An AED obtained from a PAP device may be considered in the decision to perform follow-up PSG or HSAT, though it is an important caveat that such data are derived from proprietary software, and their validity has been questioned. [46,48]
Rationale for the recommendation
The task force review did not identify any articles measuring outcomes from the use of AED to determine when to perform follow-up PSG or HSAT. In addition to allowing for follow-up of treatment adherence and effectiveness, PAP device software downloads may reveal reasons for suboptimal therapy (such as interface leak). [59] Once minimum adherence has been confirmed and interface issues have been optimized, follow-up PSG may be indicated based on the AED when PAP device software residual AHI remains elevated, particularly in the face of sleep symptoms. It is likely that many clinicians will empirically adjust machine pressure settings based upon the AED, which can obviate the need for testing in some cases. The threshold for repeat testing is a matter of debate and is contextual. For example, a mildly elevated residual AHI (5 to 15) in a patient with re-emergence of sleep apnea symptoms warrants either empiric intervention or repeat testing. However, an elevated residual AHI in the absence of signs or symptoms of under-treated OSA is of unknown significance and may not represent an obvious indication for follow-up testing.
There was no direct evidence found from the literature reviewed to inform best practice on follow-up sleep apnea testing. Indirect evidence was derived from a handful of studies, such as those that incorporated follow-up sleep apnea testing as part of a protocolized clinical trial which also measured outcomes of interest relevant to our clinical guidance statements.
- Acknowledgement
We would like to acknowledge Sleep apnea syndrome Scientific Committee for adapting these guidelines.
Chairman of the GDG: Safy Zahid Kaddah, Professor of Pulmonology, chairman of SBD unit, kasr Al-Ainy Faculty of Medicine Cairo university, Cairo.
Members of the Guideline Development Group (GDG):
Hesham Mohamed Fahmy El-Samadony, Consultant of Respiratory Medicine, Al Abbasia Chest Hospital, MOH, lecturer of Respiratory Medicine, school of Medicine, Newgiza University, Cairo.
Iman Galal, Professor of Pulmonology, chairman of SBD unit Faculty of Medicine, Ain Shams University, Cairo.
Lucy Soliman, Professor of Pulmonology, chairman of SBD unit Faculty of Medicine, Mansoura University, Dakahlia.
Maha Youseff, Professor of Pulmonology, chairman of SBD unit Faculty of Medicine, Menofia University, Menofia.
Neveen ElFayoumy, Professor of Clinical Neurophysiology, Faculty of Medicine Cairo university, Cairo.
Ramiz Reda, Professor of Neurology, Faculty of Medicine, Ain Shams University, Cairo.
- Abbreviations
AADSM
American Academy of Dental Sleep Medicine
AASM
American Academy of Sleep Medicine
AED
Automated event detection
AHI
Apnea hypopnea index
BMI
Body Mass index
CPAP
Continuous positive airway pressure
CSAS
Central sleep apnea syndrome
CSB
Cheyne-Stokes Breathing
DISE
Drug induced sleep endoscopy
EEG
Electroencephalogram
ESS
Epworth Sleepiness Scale
HSAT
Home sleep apnea test
MAD
Mandibular advancement devices
NPPV
Non-Invasive Positive Pressure Ventilation
OCST
Out-of-center sleep testing
ODI
Oxygen desaturation index
OSA
Obstructive sleep apnea
OSAHS
Obstructive sleep apnea-hypopnea syndrome
OSAS
Obstructive sleep apnea syndrome
PSG
Polysomnography
QOL
Quality of life
RDI
Respiratory Disturbance index
REI
Respiratory event index
SAS
Sleep apnea syndrome
SDB
Sleep disordered breathing
SRBD
Sleep-related breathing disorders- Glossary
Hypopnea
Can be defined based on 1 of 2 criteria:
A reduction in airflow of at least 30% for more than 10 seconds associated with at least 4% oxygen desaturation (Acceptable AASM criteria) or a reduction in airflow of at least 30% for more than 10 seconds associated with at least 3% oxygen desaturation or an arousal from sleep on EEG (Recommended AASM criteria).
Apnea
Scoring apnea requires both of the following criteria to be met:
A. Drop in the peak signal excursion by ≥90% of pre-event baseline flow
B. Duration of the drop in flow is ≥10 seconds
Obstructive apnea
Occurs if there is increased effort throughout the entire apnea
Central apnea
Occurs if there is no effort throughout the entire apnea
Mixed apnea
Occurs if there is no effort in the first part and there is an effort in the second part of the apnea.
- Introduction
Obstructive sleep apnea (OSA) is characterized by episodes of a complete airway collapse or a partial collapse with an associated decrease in oxygen saturation or arousal from sleep. Other symptoms include loud, disruptive snoring, witnessed apneas during sleep, and excessive daytime sleepiness. This disturbance results in fragmented, nonrestorative sleep. OSA has significant implications for cardiovascular health, mental illness, quality of life, and driving safety.
Through scientific exploration of the cause, risk factors, pathophysiology, evaluation and treatment of obstructive sleep apnea, healthcare professionals gain insights into the latest advancements in diagnostic techniques, such as polysomnography and home sleep apnea testing, and evidence-based treatment modalities, including continuous positive airway pressure therapy, oral appliances, and surgical interventions. Interdisciplinary collaboration enhance competence in managing OSA, fostering effective communication and teamwork among sleep specialists, pulmonologists, otolaryngologists, dentists, and other relevant professionals. This collaborative approach ensures comprehensive care delivery and improves patient outcomes in obstructive sleep apnea management.
- Scope and Purpose
The objectives of this guidelines are
- To provide guidance for the proper management of obstructive sleep apnea patients.
- To provide clinical guidance statements on indications for follow-up PSG and HSAT in adult patients with OSA. These guidance statements apply to the use of PSG and HSATs after a diagnosis of OSA has been established and decisions about treatment implementation have been made. The guidance statements do not apply to the initiation of treatment (eg, initial titration of PAP devices).
- To provide guidance on various methods for assessment to be used by health professionals to improve patient outcomes in obstructive sleep apnea management for better symptom control, reducing side effects, and increasing self-efficacy and QOL.
- Target Audience
This guideline targets; healthcare professionals, policy makers, national Sleep related breathing disorders programme managers, as well as non-governmental organizations (NGOs) and other stakeholders to afford the most appropriate tools for individuals with OSA.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2015 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in a table:
• Developing organisation/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained).
The GDG decided to adapt the American Academy of Sleep Medicine Clinical Practice Guideline (2024) [1].
Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available
on the following sites:
• GRADE working group: https://www.gradeworkinggroup.org/
• GRADE online training modules:http://cebgrade.mcmaster.ca/
Table 1 Quality and Significance of the four levels of evidence in GRADE:
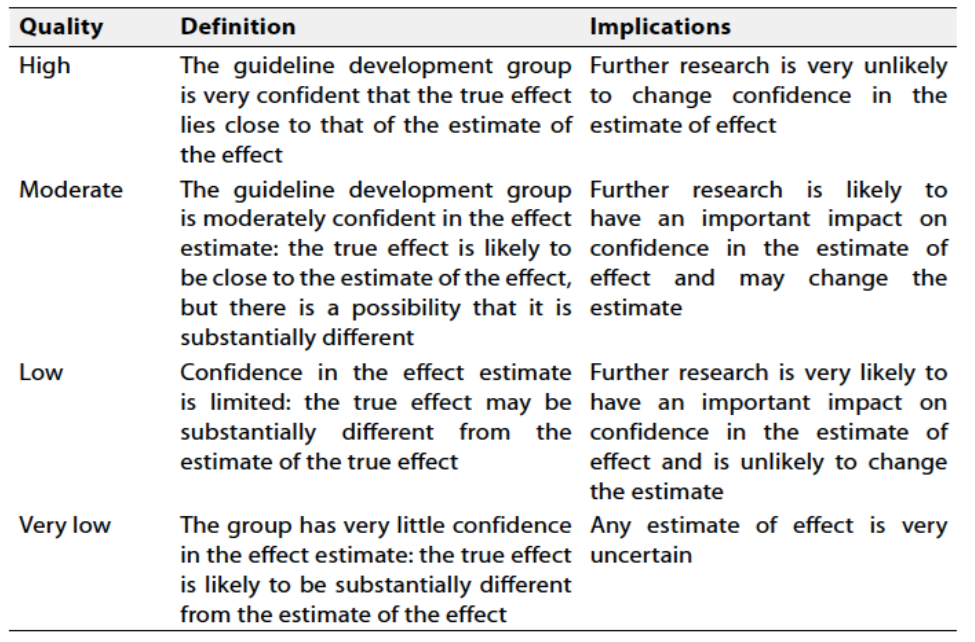
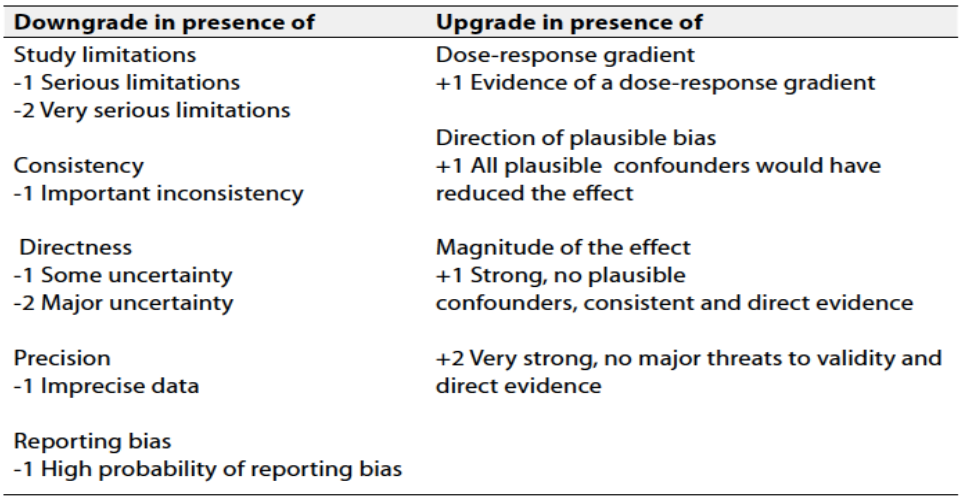
Table 2 Factors that determine How to upgrade or downgrade the quality of evidence
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Implementation considerations
Several barriers may hinder the effective implementation and scale-up of the recommendations in this guideline. These factors may be related to the behaviours of patients (or families), the behavior of healthcare professionals, the organization of care, health service delivery or financial arrangements.
Obstacles to effective implementation include:
➡️ Patient engagement
➡️ Collaboration; person centered, team-based collaboration between clinician, sleep specialists, pulmonologists, otolaryngologists, dentists, and others involved in care delivery
➡️ Management changes: information, guidance and support delivered easily and consistently can help assess sustained management changes.
- Research gaps
Pharmacological Interventions:
1- Study the efficacy of medications in treating OSA. The epilepsy drug sulthiame has shown promise in reducing symptoms of sleep apnea by inhibiting carbonic anhydrase, which helps regulate breathing patterns during sleep.
Weight Management and Metabolic Treatments:
2- Study the efficacy of patients treated with tirzepatide in substantial reductions in apneic events and improvements in related health parameters.
Weight loss has been identified as a critical factor in managing OSA. Medications like tirzepatide, recently approved by the FDA under the brand name Zepbound, have been effective in inducing significant weight loss, thereby reducing the severity of sleep apnea.
Technological Innovations:
Advancements in technology have led to the development of more comfortable and effective treatment options for sleep breathing disorders. Improvements in CPAP machine design, better-fitting masks, and supportive technologies have enhanced patient compliance and treatment efficacy. Additionally, telemedicine and remote monitoring are becoming integral in managing sleep disorders, allowing for real-time data collection and personalized treatment adjustments.
3- Study Personalized Medicine in treatment of obstructive sleep apnea patients: Tailoring treatments based on individual patient profiles, including genetic, anatomical, and lifestyle factors, to improve outcomes.
4- Evaluation of Non-Invasive such as oral appliances and positional therapies in treatment of obstructive sleep apnea patients to increase patient adherence.
5- Integrating multidisciplinary approaches that address comorbid conditions associated with sleep breathing disorders, enhancing overall patient health. Comprehensive Disease Management study.
6- Studies designed to enhance HSAT specificity to reliably rule out OSA among patients receiving treatment.
6- Comparison of clinician global assessment, HSAT and PSG as reliable tools to evaluate changes in symptoms and/or outcomes in patients receiving therapy.
7- Establish reliability and utility of HSAT in patients with cardiopulmonary or neuromuscular disease.
8- Determine the optimal timing of follow-up sleep apnea testing after surgical modifications of the upper airway for OSA.
9- Identify the ideal timing for follow-up testing in patients with OSA who have experienced a change in weight, both by medical/dietary means, and following bariatric surgery.
10- Explore the utility of pulse oximetry as a tool for follow-up of patients with OSA, including those with cardiopulmonary disease.
11- Comparative studies of outcomes between empiric adjustments of OSA therapy based upon clinical assessment or PAP machine AED with interventions based upon follow-up testing.
12- Collaboration with partners in industry to broadly validate and standardize algorithms and adopt common language and terminology for events on PAP downloads.
13- Increase understanding of whether treatment-emergent central sleep apnea encountered on machine AED or on follow-up testing is significant and warrants intervention.
- Monitoring and evaluating the impact of the guideline
Assessment of effectiveness of adult obstructive sleep apnea syndrome services:
· Assessment of sleep apnea symptoms including snoring and excessive daytime sleepiness assessed by ESS.
· Measurement of BMI.
· Polysomnography results including AHI, RDI, snoring index, arrythmia index and oxygen saturation.
· Recommended type of management including CPAP titration.
Updating of the guidelines:
These guidelines will be updated whenever there is new evidence.
- References
1. American Academy of Sleep Medicine Guidelines 2024. Hptts://aasm,org. Clinical Resources. Practice standards.
2. Xanthopoulos MS, Kim JY, Blechner M, Chang MY, Menello MK, Brown C, Matthews E, Weaver TE, Shults J, Marcus CL. Self-Efficacy and Short-Term Adherence to Continuous Positive Airway Pressure Treatment in Children. Sleep. 2017 Jul 01;40(7) [PubMed]
3. Schwab RJ, Badr SM, Epstein LJ, Gay PC, Gozal D, Kohler M, Lévy P, Malhotra A, Phillips BA, Rosen IM, Strohl KP, Strollo PJ, Weaver EM, Weaver TE., ATS Subcommittee on CPAP Adherence Tracking Systems. An official American Thoracic Society statement: continuous positive airway pressure adherence tracking systems. The optimal monitoring strategies and outcome measures in adults. Am J Respir Crit Care Med. 2013 Sep 01;188(5):613-20. [PMC free article] [PubMed]
4. Fox N, Hirsch-Allen AJ, Goodfellow E, Wenner J, Fleetham J, Ryan CF, Kwiatkowska M, Ayas NT. The impact of a telemedicine monitoring system on positive airway pressure adherence in patients with obstructive sleep apnea: a randomized controlled trial. Sleep. 2012 Apr 01;35(4):477-81. [PMC free article] [PubMed]
5. Bakker JP, Weaver TE, Parthasarathy S, Aloia MS. Adherence to CPAP: What Should We Be Aiming For, and How Can We Get There? Chest. 2019 Jun;155(6):1272-1287. [PubMed]
6. Khan NNS, Todem D, Bottu S, Badr MS, Olomu A. Impact of patient and family engagement in improving continuous positive airway pressure adherence in patients with obstructive sleep apnea: a randomized controlled trial. J Clin Sleep Med. 2022 Jan 01;18(1):181-191. [PMC free article] [PubMed]
7. Thong BKS, Loh GXY, Lim JJ, Lee CJL, Ting SN, Li HP, Li QY. Telehealth Technology Application in Enhancing Continuous Positive Airway Pressure Adherence in Obstructive Sleep Apnea Patients: A Review of Current Evidence. Front Med (Lausanne). 2022;9:877765. [PMC free article] [PubMed]
8. Sánchez-de-la-Torre M, Sánchez-de-la-Torre A, Bertran S, Abad J, Duran-Cantolla J, Cabriada V, Mediano O, Masdeu MJ, Alonso ML, Masa JF, Barceló A, de la Peña M, Mayos M, Coloma R, Montserrat JM, Chiner E, Perelló S, Rubinós G, Mínguez O, Pascual L, Cortijo A, Martínez D, Aldomà A, Dalmases M, McEvoy RD, Barbé F., Spanish Sleep Network. Effect of obstructive sleep apnoea and its treatment with continuous positive airway pressure on the prevalence of cardiovascular events in patients with acute coronary syndrome (ISAACC study): a randomised controlled trial. Lancet Respir Med. 2020 Apr;8(4):359-367. [PubMed]
9. Yasir M, Pervaiz A, Sankari A. Cardiovascular Outcomes in Sleep-Disordered Breathing: Are We Under-estimating? Front Neurol. 2022;13:801167. [PMC free article] [PubMed]
10. Lisan Q, Van Sloten T, Marques Vidal P, Haba Rubio J, Heinzer R, Empana JP. Association of Positive Airway Pressure Prescription With Mortality in Patients With Obesity and Severe Obstructive Sleep Apnea: The Sleep Heart Health Study. JAMA Otolaryngol Head Neck Surg. 2019 Jun 01;145(6):509-515. [PMC free article] [PubMed]
11. Azarbarzin A, Zinchuk A, Wellman A, Labarca G, Vena D, Gell L, Messineo L, White DP, Gottlieb DJ, Redline S, Peker Y, Sands SA. Cardiovascular Benefit of Continuous Positive Airway Pressure in Adults with Coronary Artery Disease and Obstructive Sleep Apnea without Excessive Sleepiness. Am J Respir Crit Care Med. 2022 Sep 15;206(6):767-774. [PMC free article] [PubMed]
12. Phillips CL, Grunstein RR, Darendeliler MA, Mihailidou AS, Srinivasan VK, Yee BJ, Marks GB, Cistulli PA. Health outcomes of continuous positive airway pressure versus oral appliance treatment for obstructive sleep apnea: a randomized controlled trial. Am J Respir Crit Care Med. 2013 Apr 15;187(8):879-87. [PubMed] Phillips CL, Grunstein RR, Darendeliler MA, Mihailidou AS, Srinivasan VK, Yee BJ, Marks GB, Cistulli PA. Health outcomes of continuous positive airway pressure versus oral appliance treatment for obstructive sleep apnea: a randomized controlled trial. Am J Respir Crit Care Med. 2013 Apr 15;187(8):879-87. [PubMed]
13. Uniken Venema JAM, Doff MHJ, Joffe-Sokolova D, Wijkstra PJ, van der Hoeven JH, Stegenga B, Hoekema A. Long-term obstructive sleep apnea therapy: a 10-year follow-up of mandibular advancement device and continuous positive airway pressure. J Clin Sleep Med. 2020 Mar 15;16(3):353-359. [PMC free article] [PubMed]
14. Ramar K, Dort LC, Katz SG, Lettieri CJ, Harrod CG, Thomas SM, Chervin RD. Clinical Practice Guideline for the Treatment of Obstructive Sleep Apnea and Snoring with Oral Appliance Therapy: An Update for 2015. J Clin Sleep Med. 2015 Jul 15;11(7):773-827. [PMC free article] [PubMed] Ng SSS, Tam WWS, Lee RWW, Chan TO, Yiu K, Yuen BTY, Wong KT, Woo J, Ma RCW, Chan KKP, Ko FWS, Cistulli PA, Hui DS. Effect of Weight Loss and Continuous Positive Airway Pressure on Obstructive Sleep Apnea and Metabolic Profile Stratified by Craniofacial Phenotype: A Randomized Clinical Trial. Am J Respir Crit Care Med. 2022 Mar 15;205(6):711-720. [PubMed]
15. Wong AM, Barnes HN, Joosten SA, Landry SA, Dabscheck E, Mansfield DR, Dharmage SC, Senaratna CV, Edwards BA, Hamilton GS. The effect of surgical weight loss on obstructive sleep apnoea: A systematic review and meta-analysis. Sleep Med Rev. 2018 Dec;42:85-99. [PubMed]
16. Al Lawati NM, Patel SR, Ayas NT. Epidemiology, risk factors, and consequences of obstructive sleep apnea and short sleep duration. Prog Cardiovasc Dis. 2009 Jan-Feb;51(4):285-93. [PubMed]
17. Cerritelli L, Caranti A, Migliorelli A, Bianchi G, Stringa LM, Bonsembiante A, Cammaroto G, Pelucchi S, Vicini C. Sleep position and obstructive sleep apnea (OSA): Do we know how we sleep? A new explorative sleeping questionnaire. Sleep Breath. 2022 Dec;26(4):1973-1981. [PubMed]
18. Jo JH, Kim SH, Jang JH, Park JW, Chung JW. Comparison of polysomnographic and cephalometric parameters based on positional and rapid eye movement sleep dependency in obstructive sleep apnea. Sci Rep. 2022 Jun 14;12(1):9828. [PMC free article] [PubMed]
19. Ravesloot MJ, de Vries N. Reliable calculation of the efficacy of non-surgical and surgical treatment of obstructive sleep apnea revisited. Sleep. 2011 Jan 01;34(1):105-10. [PMC free article] [PubMed]
20. Araghi MH, Chen YF, Jagielski A, et al. Effectiveness of lifestyle interventions on obstructive sleep apnea (OSA): systematic review and meta-analysis. Sleep. 2013;36(10):1553-1562, 1562a-1562e.
21. Mitchell LJ, Davidson ZE, Bonham M, et al. Weight loss from lifestyle interventions and severity of sleep apnoea: a systematic review and meta-analysis. Sleep Medicine. 2014;15(10):1173-1183.
22. Edwards BA, Bristow C, O’Driscoll DM, et al. Assessing the impact of diet, exercise and the combination of the two as a treatment for OSA: A systematic review and meta-analysis. Respirology. 2019;24(8):740-751.
23. Maniaci A, Di Luca M, Lechien JR, Iannella G, Grillo C, Grillo CM, Merlino F, Calvo-Henriquez C, De Vito A, Magliulo G, Pace A, Vicini C, Cocuzza S, Bannò V, Pollicina I, Stilo G, Bianchi A, La Mantia I. Lateral pharyngoplasty vs. traditional uvulopalatopharyngoplasty for patients with OSA: systematic review and meta-analysis. Sleep Breath. 2022 Dec;26(4):1539-1550. [PubMed]
24. He M, Yin G, Zhan S, Xu J, Cao X, Li J, Ye J. Long-term Efficacy of Uvulopalatopharyngoplasty among Adult Patients with Obstructive Sleep Apnea: A Systematic Review and Meta-analysis. Otolaryngol Head Neck Surg. 2019 Sep;161(3):401-411. [PubMed]
25. Martin MJ, Khanna A, Srinivasan D, Sovani MP. Patient-reported outcome measures following maxillomandibular advancement surgery in patients with obstructive sleep apnoea syndrome. Br J Oral Maxillofac Surg. 2022 Sep;60(7):963-968. [PubMed]
26. Zhou N, Ho JTF, de Vries N, Bosschieter PFN, Ravesloot MJL, de Lange J. Evaluation of drug-induced sleep endoscopy as a tool for selecting patients with obstructive sleep apnea for maxillomandibular advancement. J Clin Sleep Med. 2022 Apr 01;18(4):1073-1081. [PMC free article] [PubMed]
27. Huang Z, Bosschieter PFN, Aarab G, van Selms MKA, Vanhommerig JW, Hilgevoord AAJ, Lobbezoo F, de Vries N. Predicting upper airway collapse sites found in drug-induced sleep endoscopy from clinical data and snoring sounds in patients with obstructive sleep apnea: a prospective clinical study. J Clin Sleep Med. 2022 Sep 01;18(9):2119-2131. [PMC free article] [PubMed]
28. Lewis R, Pételle B, Campbell MC, MacKay S, Palme C, Raux G, Sommer JU, Maurer JT. Implantation of the nyxoah bilateral hypoglossal nerve stimulator for obstructive sleep apnea. Laryngoscope Investig Otolaryngol. 2019 Dec;4(6):703-707. [PMC free article] [PubMed]
29. Kompelli AR, Ni JS, Nguyen SA, Lentsch EJ, Neskey DM, Meyer TA. The outcomes of hypoglossal nerve stimulation in the management of OSA: A systematic review and meta-analysis. World J Otorhinolaryngol Head Neck Surg. 2019 Mar;5(1):41-48. [PMC free article] [PubMed]
30. Strollo PJ, Soose RJ, Maurer JT, de Vries N, Cornelius J, Froymovich O, Hanson RD, Padhya TA, Steward DL, Gillespie MB, Woodson BT, Van de Heyning PH, Goetting MG, Vanderveken OM, Feldman N, Knaack L, Strohl KP., STAR Trial Group. Upper-airway stimulation for obstructive sleep apnea. N Engl J Med. 2014 Jan 09;370(2):139-49. [PubMed]
31. Vicini C, Dallan I, Campanini A, et al. Surgery vs ventilation in adult severe obstructive sleep apnea syndrome. American Journal Otolaryngology.2010;31(1):14-20.
32. Zaghi S, Holty JE, Certal V, et al. Maxillomandibular Advancement for Treatment of Obstructive Sleep Apnea: A Meta-analysis. JAMA Otolaryngol Head Neck Surg. 2016;142(1):58-66.
33. Holty JE, Guilleminault C. Maxillomandibular advancement for the treatment of obstructive sleep apnea: a systematic review and meta-analysis. Sleep Medicine Reviews. 2010;14(5):287-297.
34. Giralt-Hernando M, Valls-Ontañón A, Guijarro-Martínez R, et al. Impact of surgical maxillomandibular advancement upon pharyngeal airway volume and the apnoea-hypopnoea index in the treatment of obstructive sleep apnoea: systematic review and meta-analysis. BMJ Open Respir Res. 2019;6(1):e000402.
35. Rojo-Sanchis C, Almerich-Silla JM, Paredes-Gallardo V, et al. Impact of Bimaxillary Advancement Surgery on the Upper Airway and on Obstructive Sleep Apnea Syndrome: a Meta-Analysis. Scientific Reports. 2018;8(1):5756.
36. AlSaty G, Xiang J, Burns M, et al. Follow-up observation of patients with obstructive sleep apnea treated by maxillomandibular advancement. American Journal of Orthodontics and Dentofacial Orthopedics. 2020;158(4):527-534.
37. Liu SY, Awad M, Riley R, et al. The Role of the Revised Stanford Protocol in Today’s Precision Medicine. Sleep Medicine Clinics. 2019;14(1):99-107.
38. Camacho M, Liu SY, Certal V, et al. Large maxillomandibular advancements forobstructive sleep apnea: An operative technique evolved over 30 years. Journal of Cranio-Maxillo-Facial Surgery. 2015;43(7):1113-1118.
39. D.J. Bratton, T. Gaisl, C. Schlatzer, M. Kohler. Comparison of the effects of continuous positive airway pressure and mandibular advancement devices on sleepiness in patients with obstructive sleep apnoea: a network meta-analysis. Lancet Respir Med, 3 (2015), pp. 869-878
40. E. Kuhn, E.I. Schwarz, D.J. Bratton, V.A. Rossi, M. Kohler. Effects of CPAP and mandibular advancement devices on health-related quality of life in OSA: a systematic review and meta-analysis. Chest, 151 (2017), pp. 786-794
41. D.E. Jonas, H.R. Amick, C. Feltner, R.P. Weber, M. Arvanitis, A. Stine, et al. Screening for obstructive sleep apnea in adults: evidence report and systematic review for the US preventive services task force. J Am Med Assoc, 317 (2017), pp. 415-433
42. M.A. Gupta, F.C. Simpson, D.C. Lyons. The effect of treating obstructive sleep apnea with positive airway pressure on depression and other subjective symptoms: a systematic review and meta-analysis. Sleep Med Rev, 28 (2016), pp. 55-68
43. M. Povitz, C.E. Bolo, S.J. Heitman, W.H. Tsai, J. Wang, M.T. James. Effect of treatment of obstructive sleep apnea on depressive symptoms: systematic review and meta-analysis. PLoS Med, 11 (2014), p. e1001762
44. J. Jing, T. Huang, W. Cui, H. Shen. Effect on quality of life of continuous positive airway pressure in patients with obstructive sleep apnea syndrome: a meta-analysis. Lung, 186 (2008), pp. 131-144
45. Patil SP, Ayappa IA, Caples SM, Kimoff RJ, Patel SR, Harrod CG. Treatment of adult obstructive sleep apnea with positive airway pressure: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2019;15(2):335–343.
46. Morgenthaler TI, Aurora RN, Brown T, et al.. Practice parameters for the use of autotitrating continuous positive airway pressure devices for titrating pressures and treating adult patients with obstructive sleep apnea syndrome: an update for 2007. An American Academy of Sleep Medicine report. Sleep. 2008;31(1):141–147.
47. Sawyer AM, Gooneratne NS, Marcus CL, Ofer D, Richards KC, Weaver TE. A systematic review of CPAP adherence across age groups: clinical and empiric insights for developing CPAP adherence interventions. Sleep Med Rev. 2011;15(6):343–356.
48. Epstein LJ, Kristo D, Strollo PJ, et al.. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med. 2009;5(3):263–276
49. Marklund M. Long-term efficacy of an oral appliance in early treated patients with obstructive sleep apnea. Sleep Breath. 2016;20(2):689–694
50. Palotie T, Riekki S, Mäkitie A, Bachour A, Arte S, Bäck L. The effect of mandible advancement splints in mild, moderate, and severe obstructive sleep apnea-the need for sleep registrations during follow up. Eur J Orthod. 2017;39(5):497–501.
51. Takaesu Y, Tsuiki S, Kobayashi M, Komada Y, Nakayama H, Inoue Y. Mandibular advancement device as a comparable treatment to nasal continuous positive airway pressure for positional obstructive sleep apnea. J Clin Sleep Med. 2016;12(8):1113–1119.
52. Berry RB, Chediak A, Brown LK, et al.. Best clinical practices for the sleep center adjustment of noninvasive positive pressure ventilation (NPPV) in stable chronic alveolar hypoventilation syndromes. J Clin Sleep Med. 2010;6(5):491–50
53. Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, Hla KM. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177(9):10061014
54. Kapur VK, Auckley DH, Chowdhuri S, et al.. Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: an American Academy of Sleep Medicine clinical practice guideline. J Clin Sleep Med. 2017;13(3):479–504
55. Gagnadoux F, Pevernagie D, Jennum P, et al.. Validation of the system one RemStar auto a-flex for obstructive sleep apnea treatment and detection of residual apnea-hypopnea index: a European randomized trial. J Clin Sleep Med. 2017;13(2):283–290.
56. Ueno K, Kasai T, Brewer G, et al.. Evaluation of the apnea-hypopnea index determined by the S8 auto-CPAP, a continuous positive airway pressure device, in patients with obstructive sleep apnea-hypopnea syndrome. J Clin Sleep Med. 2010;6(2):146–151
57. Berry RB, Kushida CA, Kryger MH, Soto-Calderon H, Staley B, Kuna ST. Respiratory event detection by a positive airway pressure device. Sleep. 2012;35(3):361–367
58. Stepnowsky C, Zamora T, Barker R, Liu L, Sarmiento K. Accuracy of positive airway pressure device-measured apneas and hypopneas: role in treatment follow up. Sleep Disord. 2013;2013:314589
59. Rühle KH, Domanski U, Franke KJ, Nilius G. [Studies of leakage measurements of automatic CPAP-devices]. Pneumologie. 2007;61(4):213–218.
- Appendix
Appendix 1
Appendix 2