Retinoblastoma
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatric Oncology Guidelines |
| Book: | Retinoblastoma |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
26 August 2025" Download Guideline
- Executive Summary
This guidance provides a data-supported approach to the diagnosis, treatment and follow up of paediatric patients diagnosed with retinoblastoma.
|
Level Of recommendation |
|
|
1-Work up for newly diagnosed retinoblastoma |
|
|
We recommend bilateral examination under anaesthesia and full ophthalmology assessment for all patients suspected to have retinoblastoma. |
Strong Recommendation |
|
We recommend MRI of brain and orbits with IV contrast prior to therapy in all patients. |
Strong Recommendation |
|
For all tumors stage II and above - according to IRSS- we recommend metastatic work up: Bilateral bone marrow biopsies, bone scan, CSF cytology and whole spine MRI |
Strong recommendation
|
|
Pathology is not recommended for initial confirmation of diagnosis prior to therapy. |
Strong Recommendation |
|
We recommend family counseling for all children with RB. |
Strong recommendation |
|
2- Treatment of Unilateral Intraocular Retinoblastoma |
|
|
Surgery We recommend enucleation for: · IIRC Group E · Failed eye salvage with local treatment and systemic chemotherapy in groups C and D · Unilateral advanced intraocular group D disease with no hope of useful vision. |
Strong Recommendation |
|
Optic nerve resection margin is recommended to be at least 10 mm |
Strong Recommendation |
|
Histopathological assessment and staging of the enucleated eye is recommended for all patients. |
Strong Recommendation |
|
|
|
|
We recommend local treatment for all group A eyes. |
Strong Recommendation |
|
Treatment of Group B |
|
|
We recommend both local treatment and systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) for a total of 6 cycles with 21-28 days interval in between cycles. |
Strong Recommendation |
|
Treatment of Groups C and D |
|
|
We recommend both local treatment and systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) for a total of 6 cycles with 21-28 days interval in between cycles. |
Strong Recommendation |
|
We recommend routine EUA with every 1 cycle and if progressive disease we recommend enucleation and histopathology examination. |
Strong Recommendation |
|
If NO high risk features in pathology, we do not recommend further treatment after enucleation. |
Strong Recommendation |
|
If high risk features are present in pathology, we recommend giving total 6 cycles of systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) with 21-28 days interval in between cycles. |
Strong Recommendation |
|
We recommend starting systemic chemotherapy whithin 35 days of enucleation for high risk features. |
Strong Recommendation |
|
Treatment of Group E |
|
|
We recommend enucleation upfront for all group E eyes |
Strong recommendation |
|
If NO high risk features in pathology, we do not recommend further treatment after enucleation |
Strong recommendation |
|
If high risk features are present in pathology, we recommend giving total 6 cycles of systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) with 21-28 days interval in between cycles. |
Strong Recommendation |
|
We recommend starting systemic chemotherapy whithin 35 days of enucleation for high risk histology. |
Strong Recommendation |
|
3-Treatment of Bilateral Intraocular Retinoblastoma |
|
|
We recommend dictating treatment protocol by the most advanced eye. |
Strong Recommendation
|
|
We recommend upfront enucleation of the most advanced eye followed by histopathology. If the better eye is group A, B or C, we recommend enucleation of group D eyes. If both eyes are group D we recommend chemoreduction and focal therapy for both eyes. |
Strong Recommendation |
|
We recommend chemoreduction for groups B, C and D eyes as well as local therapy similar to unilateral intraocular treatment protocols. |
Strong Recommendation |
|
4- Treatment of Extraocular Retinoblastoma |
|
|
We recommend systemic chemotherapy followed by radiation therapy for IRSS stages II-III orbital and regional lymph node involvement. |
Strong Recommendation |
|
For stage IV metastatic eyes, we recommend systemic chemotherapy, followed by high dose chemotherapy and stem cell rescue. |
Strong Recommendation |
|
Recommended systemic chemotherapy regimen includes 4 cycles of 4 drug regimen (VCR-CDDP-CPM-ETOP) |
Strong Recommendation |
|
5- Surveillance |
|
|
We recommend EUA every 3-4 weeks for patients receiving active anti-tumor treatment till the end of therapy. |
Strong Recommendation |
|
We recommend 1-2 months EUA starting at the end of therapy and continuing for 24 months thereafter for patients who present with unilateral disease and are younger than 2 years of age at presentation or bilateral disease of any age. |
Strong Recommendation |
|
We recommend life long follow up and counseling for bilateral RB patients and patients with positive family history of RB as well as sibling screening. |
Strong Recommendation |
|
For sibling screening we recommend EUA at intervals: • Every 2 weeks since birth till 8 weeks of age, • then monthly till 1 year of age, • then every 3 months till 3 years of age, • then every 6 months till 7 years of age |
Strong recommendation |
|
For unilateral retinoblastoma patients who are older than 2 years of age we recommend extending ophthalmology examination intervals post therapy, reaching 6 monthly by age of 5 and annually thereafter. |
Strong recommendation |
|
We recommend that RB survivors treated with chemotherapy or EBRT , follow up at oncology clinic every 3 months then for longer intervals as clinically warranted. |
Strong Recommendation |
- Acknowledgment
We would like to acknowledge the Guidelines Development Group (GDG) of Paediatric Oncology for adapting this Guideline.
• Chair of the Committee:
Prof Alaa El-Haddad (Professor of Paediatric Oncology and Former Dean of the National Cancer Institute - Cairo University, Head of the Paediatric Oncology Department and the Bone Marrow Transplant Unit at the Children’s Cancer Hospital Cairo, Egypt).
• The Scientific Committee Members:
Prof Mahmoud Hammad (Committee Rapporteur and Professor of Paediatric Oncology - National Cancer Institute - Cairo University - and Director of the Oncology and Nuclear Medicine Centre at Nasser Institute Hospital).
Prof Iman Sidhom (Professor and Head of Department of Paediatric Oncology - National Cancer Institute - Cairo University).
Prof Emad Ebied (Deputy Director, National Cancer Institute and Professor of Paediatric Oncology - National Cancer Institute - Cairo University).
Dr Ahmed Mustafa (Lecturer of Paediatric Oncology - National Cancer Institute - Cairo University - and Assistant Head of the Specialized Medical Centres Secretariat for Oncology Affairs).
Dr Amal Ahmed Zein (Paediatric Oncology Consultant at Al-Sahel Teaching Hospital- and at Oncology Centre of Nasser Institute Hospital).
Dr Zaki Ahmed Zaki (Consultant and Director, Haematology Unit at Sheikh Zayed Specialized Hospital).
Dr Mahmoud Motaz (Lecturer, South Egypt Oncology Institute, Assiut University, Medical Director and Head of the Paediatric Oncology Department at Shifa Al Orman Oncology Centre).
Dr Esraa Maged (Lecturer of Paediatric Oncology at National Cancer Institute – Cairo University).
Dr Ebtehal Mahmoud Ali (Assistant lecturer of Paediatric Oncology at National Cancer Institute – Cairo University).
Dr Mai Mamdouh (Head of Clinical Pharmacy department, Paediatric Oncology, Dar El Salam Cancer Hospital Harmal).
- Recommendations
1-Work up for newly diagnosed retinoblastoma
We recommend bilateral examination under anaesthesia and full ophthalmology assessment for all patients suspected to have retinoblastoma.
Strong recommendation, high quality level of evidence (Systematic review and meta- analysis) (13)(21-23)
We recommend MRI of brain and orbits with IV contrast prior to therapy in all patients
Strong recommendation, high quality level of evidence (Systematic review and meta- analysis) (13)(21-23)
For all tumors stage II and above - according to IRSS- we recommend metastatic work up: Bilateral bone marrow biopsies, bone scan, CSF cytology and whole spine MRI
Strong recommendation, high quality level of evidence (Systematic review and meta- analysis, Multicenter Registry-Based Study) (24-26)
Pathology is not recommended for initial confirmation of diagnosis prior to therapy.
Strong recommendation, moderate quality level of evidence (Expert consensus review) (27)
We recommend family counseling for all children with RB.
Strong recommendation, moderate quality level of evidence (Expert consensus review)(28)
2- Treatment of Unilateral Intraocular Retinoblastoma
Surgery
We recommend enucleation for:
· IIRC Group E
· Failed eye salvage with local treatment and systemic chemotherapy in groups C and D
· Unilateral advanced intraocular group D disease with no hope of useful vision.
Strong recommendation, high quality level of evidence (Retrospective reviews, SIOP guideline recommendations)(29-31)
Optic nerve resection margin is recommended to be at least 10 mm
Strong recommendation, moderate quality level of evidence (Retrospective review of clinical trials)(32)
Histopathological assessment and staging of the enucleated eye is recommended for all patients
Strong recommendation, high quality level of evidence (Systematic review and meta-analysis)(1)
Treatment of Group A
We recommend local treatment for all group A eyes
Strong recommendation, high quality level of evidence (Systematic review and meta-analysis)(8)(9)
Treatment of Group B
We recommend both local treatment and systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) for a total of 6 cycles with 21-28 days interval in between cycles.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
Treatment of Groups C and D
We recommend both local treatment and systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) for a total of 6 cycles with 21-28 days interval in between cycles.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
We recommend routine EUA with every 1 cycle and if progressive disease we recommend enucleation and histopathology examination.
Strong recommendation, high quality level of evidence (Retrospective reviews, SIOP guideline recommendations)(29-31)
If NO high risk features in pathology, we do not recommend further treatment after enucleation.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
If high risk features are present in pathology, we recommend giving total 6 cycles of systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) with 21-28 days interval in between cycles.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
We recommend starting systemic chemotherapy whithin 35 days of enucleation for high risk features
Treatment of Group E
We recommend upfront enucleation for all group E eyes
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
If NO high risk features in pathology, we do not recommend further treatment after enucleation.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
If high risk features are present in pathology, we recommend giving total 6 cycles of systemic IV three drug chemotherapy (Vincristine-Carboplatin-Etoposide) with 21-28 days interval in between cycles.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
We recommend starting systemic chemotherapy whithin 35 days of enucleation for high risk features
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials)(33)(34)
3-Treatment of Bilateral Intraocular Retinoblastoma
We recommend dictating treatment protocol by the most advanced eye.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials, Systematic review)(34-36)
We recommend upfront enucleation of the most advanced eye followed by histopathology. If the better eye is group A, B or C, we recommend enucleation of group D eyes. If both eyes are group D we recommend chemoreduction and focal therapy for both eyes.
Strong recommendation, high quality level of evidence (Prospective clinical trial, systematic review of COG trials, Systematic review)(34-36)
We recommend chemoreduction for groups B, C and D eyes as well as local therapy similar to unilateral intraocular treatment protocols.
Strong recommendation, high quality level of evidence (Prospective clinical trial)(31)
4- Treatment of Extraocular Retinoblastoma
We recommend systemic chemotherapy followed by radiation therapy for stages II-III orbital and regional lymph node involvement.
Strong recommendation, moderate quality level of evidence (COG prospective clinical trial)(37)
For stage IV metastatic eyes, we recommend systemic chemotherapy, followed by high dose chemotherapy and stem cell rescue.
Strong recommendation, moderate quality level of evidence (COG prospective clinical trial)(37)
Recommended systemic chemotherapy regimen includes 4 cycles of 4 drug regimen (VCR-CDDP-CPM-ETOP) with 3 weeks between cycles.
Strong recommendation, moderate quality level of evidence (COG prospective clinical trial)(37)
5- Surveillance
We recommend EUA every 3-4 weeks for patients receiving active anti-tumor treatment till the end of therapy.
Strong recommendation, high quality level of evidence (systematic reviews, SEER based analysis)(38-40)
We recommend 1-2 months EUA starting at the end of therapy and continuing for 24 months thereafter for patients who present with unilateral disease and are younger than 2 years of age at presentation or bilateral disease of any age.
Strong recommendation, high quality level of evidence (systematic reviews, SEER based analysis)(38-40)
We recommend life long follow up and counseling for bilateral RB patients and patients with positive family history of RB as well as sibling screening
Strong recommendation, high quality level of evidence (systematic reviews, SEER based analysis)(38-40)
For sibling screening we recommend EUA at intervals:
• Every 2 weeks since birth till 8 weeks of age,
• then monthly till 1 year of age,
• then every 3 months till 3 years of age,
then every 6 months till 7 years of age
Strong recommendation, high quality level of evidence (systematic reviews, SEER based analysis)(38-40)
For unilateral retinoblastoma patients who are older than 2 years of age we recommend extending ophthalmology examination intervals post therapy, reaching 6 monthly by age of 5 and annually thereafter.
We recommend that RB survivors treated with chemotherapy or EBRT , follow up at oncology clinic every 3 months then for longer intervals as clinically warranted.
Strong recommendation, high quality level of evidence (systematic reviews, SEER based analysis)(38-41)
- Abbreviations
BM (bone marrow)
CDDP (cisplatin)
CNS (central nervous system)
COG (Children’s Oncology Group)
CPM (cyclophosphamide)
CSF (cerebrospinal fluid)
CT (computed tomography)
EBRT (external beam radiation therapy)
ETOP (etoposide)
EUA (examination under anaesthesia)
FAB (French American British)
HICs (high-income countries)
IHC (immunohistochemistry)
IIRC (International Intraocular Retinoblastoma Classification)
IRSS (International Retinoblastoma Staging System)
IT (intrathecal)
IV (intravenous)
LMICs (low- and middle-income countries)
LN (lymph node)
MRI (magnetic resonance imaging)
NCCN (National Comprehensive Cancer Network)
RB (retinoblastoma)
RT (radiation therapy)
SEER (Surveillance, Epidemiology and End Results)
VCR (vincristine)
- Glossary
Intraocular
Retinoblastoma: localized to
the eye. It may be confined to the retina or may extend to involve other
structures such as the choroid, ciliary body, anterior chamber, and optic nerve
head. Intraocular retinoblastoma, however, does not extend beyond the eye into
the tissues around the eye or to other parts of the body. (1)(2)
Extraocular
Retinoblastoma: extends beyond the eye. It may be confined
to the tissues around the eye (orbital retinoblastoma), it may have spread to
the central nervous system, or it may have spread systemically to the bone
marrow or lymph nodes (metastatic retinoblastoma). (3)
Enucleation: The surgical procedure that involves removal of the entire globe and its intraocular contents, with preservation of all other periorbital and orbital structures. (4)
Exenteration: surgical procedure involving removal of the entire globe and its surrounding structures including muscles, fat, nerves, and eyelids (extent determined by disease being treated). (4)
Local treatment: Treatment that is administered by the ophthalmologist directly to the tumor including: (5-12)
1) Cryotherapy through a cryoprobe that uses extreme cold to destroy tumor tissue on the peripheral retina.
2) Laser therapy traditional photocoagulation (argon laser), in which the laser was applied around the tumor to target the tumor vasculature.
3) Brachytherapy (plaque radiation therapy) which can provide an effective means for local control.
4) Transthermal thermotherapy which is application of infrared light directly to the tumor.
5) Intravitreal chemotherapy intravitreal injection of melphalan or topotecan to control vitreous seeds.
6) Intra-arterial chemotherapy Direct delivery of chemotherapy melphalan into the eye via cannulation of the ophthalmic artery.
The International Intraocular Retinoblastoma Classification Grouping System (13-15)
Group A
· Small intraretinal tumors away from the foveola and disc.
· All tumors are 3 mm or smaller in greatest dimension, confined to the retina and All tumors are located further than 3 mm from the foveola and 1.5 mm from the optic disc.
Group B
· All other tumours confined to the retina not in Group A.
· Tumour-associated subretinal fluid less than 3 mm from the tumour with no subretinal seeding.
· Tumour located closer than 3 mm to the optic nerve or fovea.
Group C
· Discrete local disease with minimal subretinal or vitreous seeding.
· Subretinal fluid, present or past, without seeding involving up to one-fourth of the retina
· Local fine vitreous seeding may be present close to the discrete tumor.
· Local subretinal seeding less than 3 mm (2 DD) from the tumor
Group D
· Diffuse disease with significant vitreous or subretinal seeding.
· Tumour(may be massive or diffuse.
· Subretinal fluid present or past without seeding, involving up to total retinal detachment.
· Diffuse or massive vitreous disease may include greasy seeds or avascular tumour masses.
· Diffuse subretinal seeding may include subretinal plaques or tumour nodules.
Group E
· Presence of any one or more of the following poor prognosis features:
1- Tumour touching the lens.
2- Tumour anterior to anterior vitreous face involving ciliary body or anterior segment.
3- Diffuse infiltrating retinoblastoma.
4- Neovascular glaucoma.
5- Opaque media from haemorrhage.
6- Tumour necrosis with aseptic orbital cellulites.
7- Phthisis bulbi.
Unilateral advanced intraocular group D disease: Large tumors filling the vitreous for which there is little or no likelihood of restoring vision, extension of tumor to the anterior chamber, or diffuse extensive intraocular distribution of RB seeds whether subretinal or in vitreous. (16)(17)
Useful vision: The retention of functional visual acuity, sufficient for age-appropriate visual tasks. It varies with age, tumour location, and treatment goals. (18)
- Introduction
Retinoblastoma is the most common primary intraocular malignancy in children, accounting for approximately 2–4% of all pediatric cancers. It typically manifests at a median age of two years. Significant advances in multidisciplinary care combined with implementation of novel therapeutic modalities have improved survival rates in HICs, with survival exceeding 95%. However, these outcomes remain difficult to replicate in LMICs, where delayed diagnosis and limited access to specialized care, persist as major barriers to effective management and timely treatment. The principal therapeutic objective remains the preservation of life, followed by ocular salvage and visual function retention whenever feasible.(19)(20)
- Scope and purpose
This guideline was developed aiming to enhance the quality of care for retinoblastoma patients by establishing a consistent standard of care nationwide. They focus on aiding in the early diagnosis, screening, proper referral, treatment in a multidisciplinary approach, and follow-up of retinoblastoma to achieve better clinical outcomes.
- Target audience
Clinicians who are involved in the care and treatment of patients with retinoblastoma, including paediatric oncologists, ophthalmologists, radiologists, radiotherapy experts, pathologists, social workers, geneticists and palliative care specialists.
- Methodology
◾ A comprehensive search for guidelines was undertaken to identify the most
relevant guidelines to consider for adaptation.
◾ inclusion/exclusion criteria followed in the search and retrieval of
guidelines to be adapted:
- Selecting only evidence-based guidelines (guideline must include a
report on systematic literature searches and explicit links between
individual recommendations and their supporting evidence).
- Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or
updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an
organization in order to be valid and comprehensive, a guideline
ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs
to know whether a thorough literature review was conducted and
whether current evidence was used in the preparation of the
recommendations.
◾ All retrieved Guidelines were screened and appraised using AGREE II
instrument (www.agreetrust.org) by at least two members. the panel decided
a cut-off points or rank the guidelines (any guideline scoring above 50% on
the rigour dimension was retained)
The COG and NCCN guidelines are the main sources used while formulating the national guidelines for retinoblastoma.
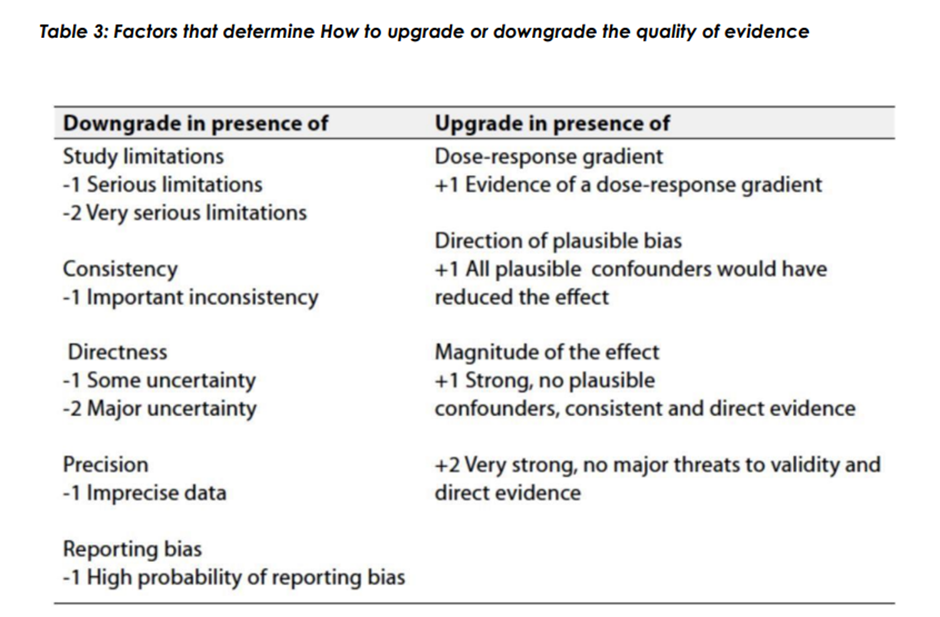
◾ Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading
of Recommendations, Assessment, Development and Evaluation) approach
to assess the quality of a body of evidence, develop and report
recommendations. GRADE methods are used by WHO because these
represent internationally agreed standards for making transparent
recommendations. Detailed information on GRADE is available through the
on the following sites:
. GRADE working group: https://www.gradeworkinggroup.org/
. GRADE online training modules: http://cebgrade.mcmaster.ca/
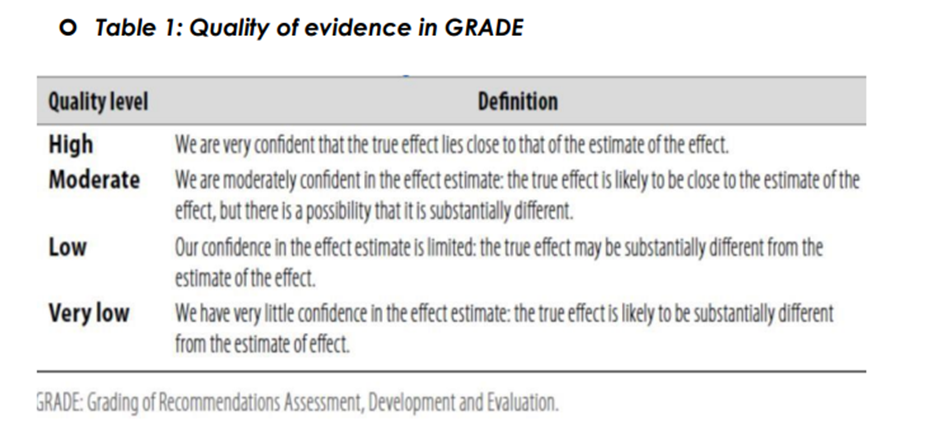
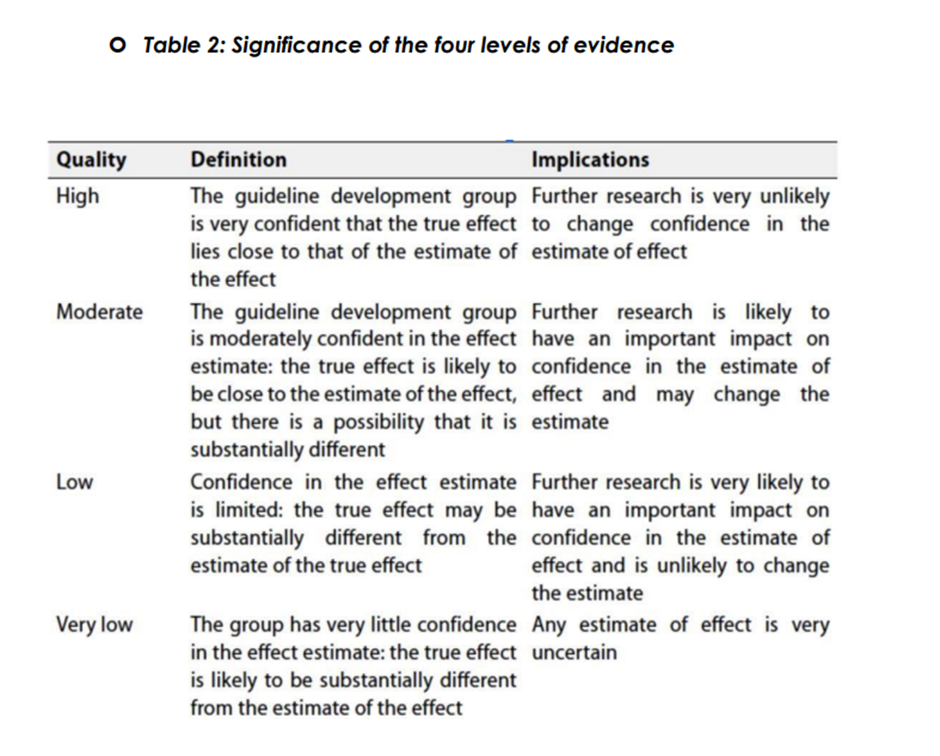
◾ Table 1: Quality of evidence in GRADE
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations
With strong recommendations, the guideline communicates the message that
the desirable effects of adherence to the recommendation outweigh the
undesirable effects. This means that in most situations the recommendation
can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors
above or if local adaptation must account for a greater variety in values and
preferences, or when resource use makes the intervention suitable for some,
but not for other locations. This means that there is a need for substantial
debate and involvement of stakeholders before this recommendation can be
adopted as policy.
When not to make recommendations.
When there is lack of evidence on the effectiveness of an intervention, it may
be appropriate not to make a recommendation.
- Clinical indicators for monitoring
• Time of initiation of chemotherapy • Initial MRI brain and orbit • Pathology of the enucleated eye • EUA visits initially and between treatment • Initial grouping of intraocular retinoblastoma- Update of this guideline
This guideline will be updated whenever there is new evidence.
- Annexes
The international Retinoblastoma Staging System(1-3)(13) Stage 0: • Eye has not been enucleated and no dissemination of disease. Stage I: • Eye enucleated, completely resected histologically. Stage II: • Eye enucleated, microscopic residual tumor. Stage III: • Regional extension III A: Overt orbital disease III B: Preauricular or cervical lymph node extension Stage IV: • Metastatic disease. IVa: Hematogenous metastasis (without CNS involvement) IVa1: Single lesion IVa2: Multiple lesions IVb: CNS extension (with or without any other site of regional or metastatic disease) IVb1: Prechiasmatic lesion IVb2: CNS mass IVb3: Leptomeningeal and CSF disease High risk pathology features (1-3) (13) • The main high-risk feature is extension of the tumour in the optic nerve to the margin of the surgical resection. • Post laminar optic nerve invasion; • Involvement of anterior segment; • Massive invasion of choroid > 3mm; • Invasion of sclera; • Extra scleral tumour extension. • Any degree of concomitant choroid and optic nerve involvement.- References
1) Abramson, D. H., & Shields, C. L. (2023). Re: Tomar et al.: American Joint Committee on Cancer Ophthalmic Oncology Task Force. High-risk pathologic features based on presenting findings in advanced intraocular retinoblastoma: A multicenter, international data-sharing American Joint Committee on Cancer study (Ophthalmology, 2022; 129:923–932). Ophthalmology, 130(2), e87–e88. https://doi.org/10.1016/j.ophtha.2022.10.024
2) Tomar, A. S., Finger, P. T., Gallie, B. L., Wilson, M. W., Chantada, G. L., Zhao, J., ... & Mallipatna, A. (2022). Metastatic death based on presenting features and treatment for advanced intraocular retinoblastoma: A multicenter registry-based study. Ophthalmology, 130(3), 262–274. https://doi.org/10.1016/j.ophtha.2022.10.018
3) Vempuluru, V. S., Maniar, A., & Kaliki, S. (2024). Global retinoblastoma studies: A review. Clinical & Experimental Ophthalmology, 52(3), 334–354. https://doi.org/10.1111/ceo.14357
4) Honavar, S. G., & Rao, R. (2019). Enucleation and exenteration. In S. Chaugule, S. Honavar, & P. Finger (Eds.), Surgical Ophthalmic Oncology (pp. 203–221). Springer. https://doi.org/10.1007/978-3-030-18757-6_12
5) Shields, C. L., Shields, J. A., Cater, J., De Potter, P., Toth, K., & Meehan, K. (2001). Plaque radiotherapy for retinoblastoma: Long-term tumor control and treatment complications in 208 tumors. Ophthalmology, 108(11), 2116–2121. https://doi.org/10.1016/S0161-6420(01)00812-4
6) Merchant, T. E., Gould, C. J., Wilson, M. W., Kun, L. E., Pratt, C. B., & Haik, B. G. (2004). Episcleral plaque brachytherapy for retinoblastoma. Pediatric Blood & Cancer, 43(2), 134–139. https://doi.org/10.1002/pbc.20044
7) Shields, C. L., Mashayekhi, A., Sun, H., Thangappan, A., & Shields, J. A. (2006). Iodine 125 plaque radiotherapy as salvage treatment for retinoblastoma recurrence after chemoreduction in 84 tumors. Ophthalmology, 113(11), 2087–2092. https://doi.org/10.1016/j.ophtha.2006.06.024
8) Francis, J. H., Levin, A. M., Zabor, E. C., Gobin, Y. P., Dunkel, I. J., & Abramson, D. H. (2018). Ten-year experience with ophthalmic artery chemosurgery: Ocular and recurrence-free survival. PLoS ONE, 13(5), e0197081. https://doi.org/10.1371/journal.pone.0197081
9) Yousef, Y. A., Soliman, S. E., Astudillo, P. P. P., Gallie, B. L., & Dimaras, H. (2016). Intra-arterial chemotherapy for retinoblastoma: A systematic review. JAMA Ophthalmology, 134(5), 584–591. https://doi.org/10.1001/jamaophthalmol.2016.0403
10) Shields, C. L., Kaliki, S., Al-Dahmash, S. A., Rojanaporn, D., Bianciotto, C., & Shields, J. A. (2013). Management of advanced retinoblastoma with intravenous chemotherapy then intra-arterial chemotherapy as alternative to enucleation. Retina, 33(10), 2103–2109. https://doi.org/10.1097/IAE.0b013e31829d1c75
11) Francis, J. H., Abramson, D. H., Gaillard, M. C., Marr, B. P., Beck-Popovic, M., Munier, F. L., ... & Gobin, Y. P. (2015). The classification of vitreous seeds in retinoblastoma and response to intravitreal melphalan. Ophthalmology, 122(6), 1173–1179. https://doi.org/10.1016/j.ophtha.2015.02.001
12) Shields, C. L., Manjandavida, F. P., Arepalli, S., Kaliki, S., & Shields, J. A. (2014). Intravitreal melphalan for persistent or recurrent retinoblastoma vitreous seeds: Preliminary results. JAMA Ophthalmology, 132(3), 319–325. https://doi.org/10.1001/jamaophthalmol.2013.8001
13) Kaliki, S., Shields, C. L., Rojanaporn, D., Al-Dahmash, S. A., Nagaiah, G., & Shields, J. A. (2013). High-risk retinoblastoma based on International Classification of Retinoblastoma: Analysis of 519 enucleated eyes. Ophthalmology, 120(5), 997–1003. https://doi.org/10.1016/j.ophtha.2012.10.032
14) Chantada, G. L., Doz, F., Orjuela, M., Qaddoumi, I., Sitorus, R. S., Kepak, T., ... & Rodriguez-Galindo, C. (2008). World disparities in risk definition and management of retinoblastoma: A report from the International Retinoblastoma Staging Working Group. Pediatric Blood & Cancer, 50(3), 692–694. https://doi.org/10.1002/pbc.21427
15) Murphree, L. M. (2005). Intraocular retinoblastoma: The case for a new group classification. Ophthalmology Clinics of North America, 18(1), 41–53. https://doi.org/10.1016/j.ohc.2004.10.002
16) Munier, F. L. (2014). Classification and management of seeds in retinoblastoma. Ophthalmic Genetics, 35(4), 193–207. https://doi.org/10.3109/13816810.2014.973045
17) Shields, C. L., Honavar, S. G., Shields, J. A., Demirci, H., Meadows, A. T., & Naduvilath, T. J. (2002). Factors predictive of recurrence of retinal tumors, vitreous seeds, and subretinal seeds following chemoreduction for retinoblastoma. Archives of Ophthalmology, 120(4), 460–464. https://doi.org/10.1001/archopht.120.4.460
18) Warda, O., Naeem, Z., Roelofs, K. A., Sagoo, M. S., & Reddy, M. A. (2023). Retinoblastoma and vision. Eye, 37(5), 797–808. https://doi.org/10.1038/s41433-021-01845-y
19) Dimaras, H., Kimani, K., Dimba, E. A. O., Gronsdahl, P., White, A., Chan, H. S. L., & Gallie, B. L. (2015). Retinoblastoma. The Lancet, 386(9997), 803–812. https://doi.org/10.1016/S0140-6736(14)60345-0
20) Fabian, I. D., Stacey, A. W., Chowdhury, T., Duncan, C., Taktak, A., Scheimberg, I., ... & Reddy, M. A. (2019). Global retinoblastoma presentation and analysis by national income level. JAMA Oncology, 5(6), 865–873. https://doi.org/10.1001/jamaoncol.2019.0266
21) Khurana, A., Eisenhut, C. A., Wan, W., et al. (2013). Comparison of the diagnostic value of MR imaging and ophthalmoscopy for the staging of retinoblastoma. European Radiology, 23(5), 1271–1280. https://doi.org/10.1007/s00330-012-2707-8
22) de Graaf, P., Göricke, S., Rodjan, F., Galluzzi, P., Maeder, P., Castelijns, J. A., ... & Moll, A. C. (2012). Guidelines for imaging retinoblastoma: Imaging principles and MRI standardization. Pediatric Radiology, 42(1), 2–14. https://doi.org/10.1007/s00247-011-2202-2
23) Cho, S. J., Kim, J. H., Baik, S. H., Sunwoo, L., Bae, Y. J., & Choi, B. S. (2020). Diagnostic performance of MRI of post-laminar optic nerve invasion detection in retinoblastoma: A systematic review and meta-analysis. Neuroradiology, 63(4), 499–509.
24) Chantada, G. L., Sampor, C., Bosaleh, A., Solernou, V., Fandiño, A., & de Dávila, M. T. (2013). Comparison of staging systems for extraocular retinoblastoma: Analysis of 533 patients. JAMA Ophthalmology, 131(9), 1127–1134. https://doi.org/10.1001/jamaophthalmol.2013.260
25) Tomar, A. S., Finger, P. T., Gallie, B., et al. (2022). Metastatic death based on presenting features and treatment for advanced intraocular retinoblastoma: A multicenter registry-based study. Ophthalmology, 129(8), 933–945.
26) Bakhshi, S., Meel, R., Kashyap, S., & Sharma, S. (2011). Bone marrow aspirations and lumbar punctures in retinoblastoma at diagnosis. Journal of Pediatric Hematology/Oncology, 33(5), e182–e185.
27) Munier, F. L., Beck-Popovic, M., Chantada, G. L., Cobrinik, D., Kivelä, T. T., Lohmann, D., ... & Stathopoulos, C. (2019). Conservative management of retinoblastoma: Challenging orthodoxy without compromising the state of metastatic grace. Progress in Retinal and Eye Research, 73, 100764. https://doi.org/10.1016/j.preteyeres.2019.05.005
28) Gombos, D. S., & Skalet, A. H. (2024a). Screening children at risk for retinoblastoma. In Clinical Ophthalmic Oncology (pp. 289–291). Springer. https://doi.org/10.1007/978-3-031-77177-4_30
29) Berry, J. L., Kogachi, K., Aziz, H. A., et al. (2017). Risk of metastasis and orbital recurrence in advanced retinoblastoma eyes treated with systemic chemoreduction versus primary enucleation. Pediatric Blood & Cancer, 64(4), e26270. https://doi.org/10.1002/pbc.26270
30) Chantada, G., Luna-Fineman, S., Sitorus, R. S., Kruger, M., Israels, T., Leal-Leal, C., et al. (2013). SIOP-PODC recommendations for graduated-intensity treatment of retinoblastoma in developing countries. Pediatric Blood & Cancer, 60(5), 719–727. https://doi.org/10.1002/pbc.24456
31) Shields, C. L., & Shields, J. A. (2010). Retinoblastoma management: Advances in enucleation, intravenous chemoreduction, and intra-arterial chemotherapy. Current Opinion in Ophthalmology, 21(3), 203–212. https://doi.org/10.1097/ICU.0b013e328338676a
32) Chantada, G. L., Guitter, M. R., Fandiño, A. C., Raslawski, E. C., de Davila, M. T. G., Vaiani, E., & Scopinaro, M. J. (2008). Treatment results in patients with retinoblastoma and invasion to the cut end of the optic nerve. Pediatric Blood & Cancer, 52(2), 218–222. https://doi.org/10.1002/pbc.21735
33) Aerts, I., Sastre-Garau, X., Savignoni, A., et al. (2013). Results of a multicenter prospective study on the postoperative treatment of unilateral retinoblastoma after primary enucleation. Journal of Clinical Oncology, 31(11), 1458–1463. https://doi.org/10.1200/JCO.2012.42.3962
34) Shah, R., & Gombos, D. S. (2024). Children’s Oncology Group (COG) trials for retinoblastoma. In J. L. Berry, B. E. Damato, & A. D. Singh (Eds.), Clinical Ophthalmic Oncology. Springer. https://doi.org/10.1007/978-3-031-77177-4_31
35) Rodriguez-Galindo, C., Orbach, D. B., & VanderVeen, D. (2015). Retinoblastoma. Pediatric Clinics of North America, 62(1), 201–223. https://doi.org/10.1016/j.pcl.2014.09.014
36) Ancona-Lezama, D., Dalvin, L. A., & Shields, C. L. (2020). Modern treatment of retinoblastoma: A 2020 review. Indian Journal of Ophthalmology, 68(11), 2356–2365. https://doi.org/10.4103/ijo.IJO_721_20
37) Dunkel, I. J., Piao, J., Chantada, G. L., Banerjee, A., Abouelnaga, S., Buchsbaum, J. C., ... & Chintagumpala, M. M. (2022). Intensive multimodality therapy for extraocular retinoblastoma: A children’s oncology group trial (ARET0321). Journal of Clinical Oncology, 40(33), 3839–3847. https://doi.org/10.1200/jco.21.02337
38) Skalet, A. H., Gombos, D. S., Gallie, B. L., Kim, J. W., Shields, C. L., Marr, B. P., Plon, S. E., & Chévez-Barrios, P. (2018). Screening children at risk for retinoblastoma: Consensus report from the American Association of Ophthalmic Oncologists and Pathologists. Ophthalmology, 125(3), 453–458.https://doi.org/10.1016/j.ophtha.2017.09.001
39) Reddy, M. A., Butt, M., Hinds, A. M., Duncan, C., Price, E. A., Sagoo, M. S., & Onadim, Z. (2021). Prognostic Information for Known Genetic Carriers of RB1 Pathogenic Variants (Germline and Mosaic). Ophthalmology. Retina, 5(4), 381–387. https://doi.org/10.1016/j.oret.2020.08.010
40) Shinohara, E. T., DeWees, T., & Perkins, S. M. (2014). Subsequent malignancies and their effect on survival in patients with retinoblastoma. Pediatric Blood & Cancer, 61(1), 116–119. https://doi.org/10.1002/pbc.24714
41) Temming, P., Arendt, M., Viehmann, A., et al. (2017). Incidence of second cancers after radiotherapy and systemic chemotherapy in heritable retinoblastoma survivors: A report from the German reference center. Pediatric Blood & Cancer, 64(1), 71–80. https://doi.org/10.1002/pbc.26193