Acute Gastroenteritis in Infants and Young Children
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Acute Gastroenteritis in Infants and Young Children |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
1 Sep 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Glossary
- - Introduction
- - Purpose and Scope
- - Methods
- - Evidence to recommendations: Considerations
- - Implementation Tools and Considerations
- - research needs
- - Monitoring and evaluating the impact of the guideline.
- - Updating of the guideline
- - References
- - Annexes
- Executive Summary
Acute Gastroenteritis (AGE) is a common pediatric illness. In the Middle East region, AGE persists as the second major cause of pediatric mortality and morbidity following acute lower respiratory tract infections3,4
Acute gastroenteritis (AGE) in children is considered one of the most common causes of visits to health centers, one of the most frequent reasons of hospitalization and the third leading cause of death related to infectious diseases worldwide5,6.
The incidence of diarrhea ranges from 0.5 to 2 episodes per child per year in children <3 years in Europe8. On average, children below 3 years of age in developing countries experience 3 episodes of diarrhea each year7.
Rotavirus (RV)is the leading cause of AGE in infants and young children and the major contributor to hospitalization for diarrhea in countries that have no RV vaccines in their national immunization schedules9,10,11. With the continuing decline in cases of RV-associated AGE, since the implementation of routine childhood vaccination against RV, norovirus (NoV) infection has become the most common cause of medically treated AGE 12,13.
In 2012, a study conducted in two locations in Egypt over 2112 children to determine the causes of acute diarrhea in children younger than 5-years seeking treatment. Bacteria were identified as a sole pathogen in 20%, RV in 14% and Cryptosporidium in 5% of the cases. Adenovirus (AdV), astrovirus (AsV), NoV and G. lamblia were detected as the sole pathogen in 2%, 3%, 9% and 7% of the cases, respectively. E. histolytica was never detected as the sole pathogen14.
The most common enteric infections in the 0-12 months age group were NoV, RV, enterotoxigenic E. coli (ETEC), AsV, Campylobacter spp. and AdV. When identified, Shigella was more commonly identified during the second year after birth (13-24 months). The rates of infection for Cryptosporidium spp. and G. lamblia were similar among children of both age groups14.
High fever (>40 ̊C), overt fecal blood, abdominal pain, and central nervous system involvement each suggests a bacterial pathogen. Vomiting and respiratory symptoms are associated with a viral etiology7.
Many diarrheal deaths are caused by dehydration8. It would be helpful to have a common tool to evaluate dehydration. The Clinical Dehydration Scale (CDS) is easy to use in the assessment of dehydration7.
Dehydration from AGE of any etiology and at any age, except when it is severe, can be safely and effectively treated in over 90% of cases by Oral Rehydration Salts (ORS) solution8.
For prevention of recurrent AGE, exclusive breastfeeding until age six months, and continued breastfeeding with complementary foods until two years of age is considered an important aspect in prevention7.
Contact and standard precautions including (hand hygiene, personal protective equipment, soiled patient-care equipment, environmental control including textiles, laundry and adequate patient placement)8.
This guideline focuses on prevention and management of acute gastroenteritis in infants and young children. The objectives of these Clinical Practice Guidelines are to provide evidence-based guidance on the diagnosis, management and prevention of Acute Gastroenteritis (AGE) specifically adjusted to the customs of the Egyptian community and to integrate it with the already existing health care system through the following:
1- Standardization of clinical practice of acute gastroenteritis in Egypt.
2- Reduction of morbidity and mortality from acute gastroenteritis.
These Clinical Practice Guidelines intended to be used in infants and young children less than 5 years of age presenting with acute gastroenteritis without co-morbidities in all healthcare facilities
Patient Population (P):
· Both genders
· Age: 2months - 5 years
· Disease: Acute gastroenteritis
· Co-morbidities: No
Intervention (I):
· Diagnosis
· Management
· Prevention
Professionals (P):
· Pediatricians, Family Physicians, Emergency Physicians, Primary Health Care Practitioners.
· Nurses.
· Parents and Care-givers.
Outcomes (O):
· Primary outcome: standardization of clinical practice of acute gastroenteritis in Egypt.
· Secondary outcome: reduction of morbidity and mortality from acute gastroenteritis.
Healthcare Settings (H):
· Primary, secondary and tertiary healthcare facilities.
· Governmental: University, MOH, Ministry of Defense, Ministry of Interior.
· Non-governmental: Private, NGO.
Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- ESPGHAN/ESPID guidelines 2014
2- IDSA guidelines 2017
3-WHO guidelines 2005:
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. Diagnosis of acute gastroenteritis
The guideline covers (Age group) 2months - 5 years
B. Management of acute gastroenteritis.
This section includes recommendations and good practice statements on management of acute gastroenteritis in infants and young children
C. Prevention of acute gastroenteritis.
We can summarize the guidelines’ recommendations for acute gastroenteritis in the following:
· Risk Factors:
Rotavirus is the most severe enteric pathogen of childhood diarrhea. (low, conditional)
The high incidence of dehydration in infants<6 months is related to a higher exposure to rotavirus. (low, conditional)
In developing countries, a young age (<6 months) is related to the severity and persistence of diarrhea. (Intermediate, Strong)
Children attending day care centers have a greater risk of mild and severe diarrheal illness than children at home. (low, Conditional)
Predominant breast-feeding may reduce the risk of AGE in young European infants. (Low, Conditional)
In developing areas early weaning may be associated with earlier onset of severe or prolonged diarrhea. (Low, Conditional)
Fever, severe dehydration, and lethargy, which are more common in rotavirus infection, indicate systematic involvement and are associated with severe diarrhea (low conditional)
In European countries, there is evidence, although weak, of a link between low socioeconomic status and the severity or persistence of diarrhea( low Conditional)
· Diagnosis: Indications for medical Visit
Indications for medical visit: Infants and toddlers with AGE should be referred for medical evaluation if any of the following are present (low, conditional)
Age <2 months
Severe underlying disease (eg. diabetes and renal failure)
Persistent vomiting
High output diarrhea with elevated stool volumes (>8 episodes/day)
Family-reported signs of severe dehydration.
· Dehydration Assessment:
The best measure of dehydration is the percentage loss of body weight. (Very low, Conditional)
Historical points are moderately sensitive as a measure of dehydration. (Low, Conditional)
Classification into subgroups with no or minimal dehydration, mild-moderate dehydration, and severe dehydration is an essential basis for appropriate treatment. (High, Strong)
Parental reports of dehydration symptoms are so low in specificity that they may not be clinically useful; however, parental report of normal urine output decreases the likelihood of dehydration (Very Low, Conditional)
Little is known about the severity of diarrhea and/or vomiting and dehydration in industrialized countries; therefore, recommendations are largely based on data from developing countries. In the latter, infants and young children with frequent high-output diarrhea and vomiting are most at risk (Low, Conditional)
Clinical tests for dehydration are imprecise, generally showing only fair-moderate agreement among examiners. (Low, Conditional)
It would be helpful to have a common tool to evaluate dehydration. The use of the clinical dehydration scale (CDS) is supported by consistent evidence, and it is easy to use in the assessment of dehydration. (Low Conditional)
This scale should be used in combination with other criteria to guide the need of medical interventions in individual cases. (Low Conditional)
High fever (>40oC), overt fecal blood, abdominal pain, and central nervous system involvement each suggests a bacterial pathogen. Vomiting and respiratory symptoms are associated with viral etiology (Low, Conditional)
Laboratory Diagnosis:
Acute gastroenteritis does not generally require a specific diagnostic workup. (Very low Conditional)
Children presenting with AGE do not require routine etiological investigation; however, there may be particular circumstances in which microbiological investigations may be necessary for diagnosis and treatment. (Very low Conditional)
Microbiological investigations may be considered in children with underlying chronic conditions (eg, oncologic diseases, IBDs, etc), in those in extremely severe conditions, or in those with prolonged symptoms in whom specific treatment is considered. (Very low Conditional)
The differentiation of a bacterial from nonbacterial etiology is not likely to change treatment. C-reactive protein (CRP) and procalcitonin measurements are not routinely recommended to identify a bacterial etiology. (Very low conditional)
Based on available data we do not recommend the routine use of fecal markers to distinguish between viral and bacterial AGE in the clinical setting. (Very low, conditional)
Tests of dehydration are imprecise, and, generally, there is only fair-to-moderate agreement with the examiner’s estimate. (low Conditional)
The only laboratory measurement that appears to be useful in decreasing the likelihood of >5% dehydration is serum bicarbonate (normal serum bicarbonate). (Low, Conditional)
Electrolytes should be measured in hospital settings:
In moderately dehydrated children whose history and physical examination findings are inconsistent with a severe diarrheal disease, and in all severely dehydrated children.
In all children starting IV therapy, and during therapy, because hyper- or hyponatremia will alter the rate at which IV rehydration fluids will be given. (Very low, Conditional)
There is no indication for endoscopy except in selected circumstances or cases such as differential diagnosis with IBD at its onset. (Very low, conditional)
Home management:
1- ORS:
Reduced osmolarity ORS (50/60 mmol/L Na+) should be used as first-line therapy for the management of children with AGE. (High Strong)
Reduced osmolarity ORS is more effective than full strength ORS as measured by such important clinical outcomes as reduced stool output, reduced vomiting, and reduced need for supplemental IV therapy. (High Strong)
If ORS therapy fails, such children should be given ORS solution by nasogastric (NG) tube or Ringer's Lactate Solution intravenously (IV) (75 ml/kg in four hours), usually in hospital. After confirming that the signs of dehydration have improved, it is usually possible to resume ORS successfully.
ORS therapy should not be given in the following conditions:
• Abdominal distension with paralytic ileus, which may be caused by opiate drugs (e.g. codeine, loperamide) and hypokalaemia.
Glucose malabsorption, indicated by a marked increase in stool output.
2- Nutrition:
Early resumption of feeding after rehydration therapy is recommended. Further studies are, however, needed to determine whether the timing of refeeding affects the duration of diarrhea, total stool output, or weight gain in childhood acute diarrhea. (High Strong)
The routine use of lactose-free feeds is presently not recommended in outpatient setting. (High Strong)
There is insufficient evidence to recommend in favor or against the use of diluted lactose-containing milk. (High Strong)
The bread, rice, apple, toast (BRAT) diet has not been studied and is not recommended. (Very low conditional)
Beverages with a high sugar content should not be used. (low conditional)
3- Pharmacotherapy
Children age> 6 months in developing countries may benefit from the use of zinc in the treatment of AGE; however, in regions where zinc deficiency is rare, no benefit from the use of zinc is expected. (High Strong)
Folic acid is not recommended for the management of children with AGE. (Intermediate Strong)
Active treatment with probiotics, in adjunct to ORS, is effective in reducing the duration and intensity of symptoms of gastroenteritis. Selected probiotics can be used in children with AGE. (High Strong)
The use of the following probiotics should be considered in the management of children with AGE as an adjunct to rehydration therapy:
L rhamnosus GG and S boulardii. (High Strong)
Ondansetron, at the dosages used in the available studies and administered orally or intravenously, may be effective in young children with vomiting related to AGE. Before a final recommendation is made, a clearance on safety in children is, however, needed. (Intermediate Strong)
There is no evidence to support the use of other antiemetics. (Intermediate Strong)
Loperamide is not recommended in the management of AGE in children. (Intermediate Strong)
Smectite can be considered in the management of AGE. (Intermediate Strong)
Smectite plus LGG and LGG alone are equally effective in the treatment of young children with AGE. Combined use of the 2 interventions is not justified. (Intermediate Strong)
Other absorbents (namely, kaolin–pectin and attapulgite-activated charcoal) are not recommended. (Low Conditional)
Racecadotril can be considered in the management of AGE. (Intermediate Strong)
Bismuth subsalicylate is not recommended in the management of children with AGE. (Low Conditional)
New evidence has confirmed that probiotics are effective in reducing the duration of symptoms in children with AGE. (High Strong)
The use of prebiotics in the management of children with AGE is not recommended. (Intermediate Strong)
4- Anti- infective therapy
Anti-infective therapy should not be given to the vast majority of otherwise healthy children with acute gastroenteritis. (Very Low Conditional)
Antibiotic therapy for acute bacterial gastroenteritis is not needed routinely but only for specific pathogens or in defined clinical settings. (Very Low Conditional)
The choice of the antimicrobial agent depends on the local prevalence of the 3 pathogens (Shigella spp, Campylobacter spp, and Salmonella enterica) and the resistance patterns. (Very Low Conditional)
In children with watery diarrhea, antibiotic therapy is not recommended unless the patient has recently traveled or may have been exposed to cholera. ( Very Low Conditional)
Bloody diarrhea with low or no fever is typical of STEC (enterohemorrhagic E coli), but can be mild shigellosis or salmonellosis. Antibiotics are not recommended unless epidemiology suggests shigellosis. (Very Low Conditional)
Parenteral rather than oral antibiotic therapy is recommended for:
• Patients unable to take oral medications (vomiting, stupor, etc)
• Patients with underlying immune deficiency who have AGE with fever
• Severe toxemia, suspected or confirmed bacteremia
Neonates and young infants (< 3 months) with fever. Sepsis workup and antibiotics should be considered according to local protocols. (Very Low Conditional)
Antiparasitic treatment is generally not needed in otherwise healthy children; however, it may be considered if symptoms are severe. (Low conditional)
Severe cases of giardiasis can be treated with metronidazole, nitazoxanide, albendazole, or tinidazole. (Low conditional)
Cryptosporidiasis should be treated mainly in immunocompromised children with nitazoxanide. (Low conditional)
Amebic colitis should be treated with metronidazole. (Low conditional)
Specific antiviral treatment is usually not indicated in AGE. (Very Low Conditional)
Severe cytomegalovirus colitis, especially in an immunocompromised child, should be treated with ganciclovir. (Low conditional)
Hospital management:
The recommendations for hospital admission are based on consensus and include any of the following conditions:
• Shock
• Severe dehydration (>9% of body weight)
• Neurological abnormalities (lethargy, seizures, etc)
• Intractable or bilious vomiting
• Failure of oral rehydration
• Suspected surgical condition
Conditions for a safe follow-up and home management are not met. (Very low Conditional)
Contact precautions are advised in addition to standard precautions (hand hygiene, personal protective equipment, soiled patient-care equipment, environmental control including textiles, laundry and adequate patient placement). (Very low Conditional)
Prompt discharge from hospital should be considered in children admitted for AGE when the following conditions are fulfilled:
• Sufficient rehydration is achieved as indicated by weight gain and/or clinical status
• IV fluids are no longer required
• Oral intake equals or exceeds losses
Medical follow-up is available via telephone or office visit. (Very low Conditional)
2- Rehydration therapy (enteral and parenteral):
When oral rehydration is not feasible, enteral rehydration by the nasogastric (NG) route is the preferred method of rehydration, and should be proposed before IV rehydration. (High Strong)
Enteral rehydration is associated with significantly fewer major adverse events and a shorter hospital stay than IV rehydration and is successful in most children. (High Strong)
The rapid (40–50 mL/kg within 3–6 hours) and standard (24 hours) NG rehydration regimens are equally effective and may be recommended. (Intermediate Strong)
IV fluids are required in the following cases:
• Shock
• Dehydration with altered level of consciousness or severe acidosis
• Worsening of dehydration or lack of improvement despite oral or enteral rehydration therapy
• Persistent vomiting despite appropriate fluid administration orally or via an NG tube
Severe abdominal distension and ileus. (Very low,
Conditional)
Children presenting with shock secondary to AGE should receive rapid IV infusion of isotonic crystalloid solution (0.9% saline or lactated Ringer’s solution) with a 20-mL/kg bolus. (Very Low Conditional)
If the blood pressure has not improved after the first bolus, a second (or even a third) bolus of 20 mL/kg should be administered >10 to 15 minutes and other possible causes of shock should be considered. (Very Low Conditional)
Children with severe dehydration requiring IV fluids may receive rapid rehydration with 20mL/kg/h of 0.9% saline solution for 2 to 4 hours. (Intermediate Strong)
In IV-rehydrated children, a dextrose-containing solution may be used for maintenance. ( Low Conditional)
A solution containing not <0.45% saline (at least 77 mmol/L [Na+]) is recommended during the first 24 hours of IV rehydration therapy to prevent hyponatremia Low Conditional)
After the child starts to urinate and if serum electrolyte values are known, add 20 mmol/L of K+ chloride. (Very Low Conditional)
Rapid rehydration with 20mL/kg/h for 2 to 4 hours followed by oral rehydration or continuous infusion of dextrose solution is adequate for initial rehydration of most patients requiring hospital assistance. (Intermediate Strong)
More rapid IV rehydration may be associated with electrolyte abnormalities and is associated with long time to hospital discharge, and therefore is not recommended. (Intermediate Strong)
Isotonic (0.9%) saline solution effectively reduces the risk of hyponatremia and is recommended for initial rehydration in most cases. In the rare but extremely severe cases of shock, Ringer’s lactate solution is recommended. (Low Conditional)
Glucose may be added to saline solution once fluid volume has been restored in the subsequent phase of IV rehydration (‘‘maintenance’’). (Low Conditional)
Management of electrolyte imbalance:
Oral or NG rehydration with hypo osmolar ORS is an effective and safe treatment and has fewer adverse effects than IV rehydration. (Low Conditional)
If the child is hypernatremia and needs IV rehydration:
• Use an isotonic solution (0.9% saline) for fluid deficit replacement and maintenance.
• Replace the fluid deficit slowly, typically for 48 hours, with the aim of reducing it to <0.5mmol/L/h.
Monitor plasma sodium frequently. (Low Conditional)
ORS solution is safe and effective therapy for nearly all children with hyponatremia. An exception is children with oedema, for whom ORS solution provides too much sodium.
Hypokalemia is worsened when base (bicarbonate or lactate) is given to treat acidosis without simultaneously providing potassium.
Hypokalemia can be prevented, and the potassium deficit corrected, by using ORS solution for rehydration therapy and by giving foods rich in potassium during diarrhea and after it has stopped.
Prevention:
1- Strategies:
Hand hygiene should be performed after using the toilet, changing diapers, before and after preparing food, before eating, after handling garbage or soiled laundry items, and after touching animals or their feces or environments, especially in public settings such as petting zoos. Intermediate (moderate) Strong)
Infection control measures including use of gloves and gowns, hand hygiene with soap and water, or alcohol-based sanitizers should be followed in the care of people with diarrhea. (High Strong)
The selection of a hand hygiene product should be based upon a known or suspected pathogen and the environment in which the organism may be transmitted. (low Strong)
Appropriate food safety practices are recommended to avoid cross-contamination of other foods or cooking surfaces and utensils during grocery shopping, food preparation, and storage; ensure that foods containing meats and eggs are cooked and maintained at proper temperatures. (moderate Strong)
Healthcare providers should direct educational efforts toward all people with diarrhea, but particularly to people with primary and secondary immune deficiencies, pregnant women, parents of young children, and the elderly as they have increased risk of complications from diarrheal disease. (low Strong)
Ill people with diarrhea should avoid swimming, water-related activities, and sexual contact with other people when symptomatic while adhering to meticulous hand hygiene. (low Strong)
2-Vaccination
Rotavirus vaccine should be administered to all infants without a known contraindication. (high Strong)
Two typhoid vaccines (oral and injectable) are licensed in the United States but are not recommended routinely. Typhoid vaccination is recommended as an adjunct to hand hygiene and the avoidance of high-risk foods and beverages, for travelers to areas where there is moderate to high risk for exposure to Salmonella enterica subspecies enterica serovar Typhi, people with intimate exposure (eg, household contact) to a documented Salmonella enterica subspecies enterica serovar Typhi chronic carrier, and microbiologists and other laboratory personnel routinely exposed to cultures of Salmonella enterica subspecies enterica serovar Typhi. Booster doses are recommended for people who remain at risk. (High Strong).
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/
- Recommendations
|
Table 3. Recommendations |
|
|
|||
|
A. What are the risk factors that can influence the clinical outcome of children with severe AGE? |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A1 |
ESPGHAN/ESPID 2014 |
Rotavirus is the most severe enteric pathogen of childhood diarrhea. |
Low |
conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
The high incidence of dehydration in infants<6 months is related to a higher exposure to rotavirus. |
Low |
conditional |
|
|
|
ESPGHAN/ESPID 2014 |
In developing countries, a young age (<6 months) is related to the severity and persistence of diarrhea. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Children attending day care centers have a greater risk of mild and severe diarrheal illness than children at home. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Predominant breast-feeding may reduce the risk of AGE in young European infants. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
In developing areas early weaning may be associated with earlier onset of severe or prolonged diarrhea. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Fever, severe dehydration, and lethargy, which are more common in rotavirus infection, indicate systematic involvement and are associated with severe diarrhea. |
low |
conditional |
|
|
|
ESPGHAN/ESPID 2014 |
In European countries, there is evidence, although weak, of a link between low socioeconomic status and the severity or persistence of diarrhea. |
low |
Conditional |
|
|
|
||||
|
A. Diagnosis |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
Indications for medical visit: |
ESPGHAN/ESPID 2014 |
Infants and toddlers with AGE should be referred for medical evaluation if any of the following are present: · Age <2 months · Severe underlying disease (eg. diabetes and renal failure) · Persistent vomiting · High output diarrhea with elevated stool volumes (>8 episodes/day) · Family-reported signs of severe dehydration. |
Low
Very low
Low
Low
low |
Conditiona1
Conditional
Conditional
Conditional
conditional |
|
|
|
||||
|
Diagnosis: Assessment of Dehydration |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
ESPGHAN/ESPID 2014 |
The best measure of dehydration is the percentage loss of body weight. |
Very low |
Conditional |
||
|
|
|
ESPGHAN/ESPID 2014 |
Historical points are moderately sensitive as a measure of dehydration. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Classification into subgroups with no or minimal dehydration, mild-moderate dehydration, and severe dehydration is an essential basis for appropriate treatment. |
High |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Parental reports of dehydration symptoms are so low in specificity that they may not be clinically useful; however, parental report of normal urine output decreases the likelihood of dehydration. |
Very Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Little is known about the severity of diarrhea and/or vomiting and dehydration in industrialized countries; therefore, recommendations are largely based on data from developing countries. In the latter, infants and young children with frequent high-output diarrhea and vomiting are most at risk. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Clinical tests for dehydration are imprecise, generally showing only fair-moderate agreement among examiners. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
It would be helpful to have a common tool to evaluate dehydration. The use of the clinical dehydration scale (CDS) is supported by consistent evidence, and it is easy to use in the assessment of dehydration. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
This scale should be used in combination with other criteria to guide the need of medical interventions in individual cases. |
Low |
Conditional |
|
Table 6: |
|
|
|||
|
Diagnosis Clinical features suggestive of bacterial versus viral etiology of diarrhea: |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
ESPGHAN/ESPID 2014 |
High fever (>40oC), overt fecal blood, abdominal pain, and central nervous system involvement each suggests a bacterial pathogen. Vomiting and respiratory symptoms are associated with viral etiology.
|
Low |
Conditional |
||
|
Table 7. Recommendations |
|
|
|||
|
Laboratory Diagnosis |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
ESPGHAN/ESPID 2014 |
Acute gastroenteritis does not generally require a specific diagnostic workup. |
Very low |
Conditional |
||
|
|
|
ESPGHAN/ESPID 2014 |
Children presenting with AGE do not require routine etiological investigation; however, there may be particular circumstances in which microbiological investigations may be necessary for diagnosis and treatment. |
Very low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Microbiological investigations may be considered in children with underlying chronic conditions (eg, oncologic diseases, IBDs, etc), in those in extremely severe conditions, or in those with prolonged symptoms in whom specific treatment is considered. |
Very low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
The differentiation of a bacterial from nonbacterial etiology is not likely to change treatment. C-reactive protein (CRP) and procalcitonin measurements are not routinely recommended to identify a bacterial etiology. |
Very low |
conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Based on available data we do not recommend the routine use of fecal markers to distinguish between viral and bacterial AGE in the clinical setting. |
Very low |
conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Tests of dehydration are imprecise, and, generally, there is only fair-to-moderate agreement with the examiner’s estimate. |
low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
The only laboratory measurement that appears to be useful in decreasing the likelihood of >5% dehydration is serum bicarbonate (normal serum bicarbonate). |
low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Electrolytes should be measured in hospital settings: · In moderately dehydrated children whose history and physical examination findings are inconsistent with a severe diarrheal disease, and in all severely dehydrated children. In all children starting IV therapy, and during therapy, because hyper- or hyponatremia will alter the rate at which IV rehydration fluids will be given. |
Very low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
There is no indication for endoscopy except in selected circumstances or cases such as differential diagnosis with IBD at its onset. |
Very low |
conditional |
|
|
|
|
|
|
|
|
Table 8:Home Management |
|
|
|||
|
A- ORS |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
Home management ORS |
ESPGHAN/ESPID 2014 |
Reduced osmolarity ORS (50/60 mmol/L Na+) should be used as first-line therapy for the management of children with AGE. |
High |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
Reduced osmolarity ORS is more effective than full strength ORS as measured by such important clinical outcomes as reduced stool output, reduced vomiting, and reduced need for supplemental IV therapy. |
High |
Strong |
|
|
|
WHO 2005 |
If ORS therapy fails, such children should be given ORS solution by nasogastric (NG) tube or Ringer's Lactate Solution intravenously (IV) (75 ml/kg in four hours), usually in hospital. After confirming that the signs of dehydration have improved, it is usually possible to resume ORS successfully. |
Good Practice Statement |
|
|
|
|
WHO 2005 |
ORS therapy should not be given in the following conditions: · Abdominal distension with paralytic ileus, which may be caused by opiate drugs (e.g. codeine, loperamide) and hypokalaemia. Glucose malabsorption, indicated by a marked increase in stool output. |
Good Practice Statement |
|
|
|
|
||||
|
A. Nutrition |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
ESPGHAN/ESPID 2014 |
Early resumption of feeding after rehydration therapy is recommended. Further studies are, however, needed to determine whether the timing of refeeding affects the duration of diarrhea, total stool output, or weight gain in childhood acute diarrhea. |
High |
Strong |
||
|
|
|
ESPGHAN/ESPID 2014 |
The routine use of lactose-free feeds is presently not recommended in outpatient setting. |
High |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
There is insufficient evidence to recommend in favor or against the use of diluted lactose-containing milk. |
High |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
The bread, rice, apple, toast (BRAT) diet has not been studied and is not recommended. |
Very low |
conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Beverages with a high sugar content should not be used. |
low |
conditional |
|
Pharmacology Therapy: |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A1 |
ESPGHAN/ESPID 2014 |
Children age> 6 months in developing countries may benefit from the use of zinc in the treatment of AGE; however, in regions where zinc deficiency is rare, no benefit from the use of zinc is expected. |
High |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
Folic acid is not recommended for the management of children with AGE. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Active treatment with probiotics, in adjunct to ORS, is effective in reducing the duration and intensity of symptoms of gastroenteritis. Selected probiotics can be used in children with AGE. |
High |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
The use of the following probiotics should be considered in the management of children with AGE as an adjunct to rehydration therapy: L rhamnosus GG and S boulardii. |
High |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Ondansetron, at the dosages used in the available studies and administered orally or intravenously, may be effective in young children with vomiting related to AGE. Before a final recommendation is made, a clearance on safety in children is, however, needed. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
There is no evidence to support the use of other antiemetics. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Loperamide is not recommended in the management of AGE in children. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Smectite can be considered in the management of AGE. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Smectite plus LGG and LGG alone are equally effective in the treatment of young children with AGE. Combined use of the 2 interventions is not justified. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Other absorbents (namely, kaolin–pectin and attapulgite-activated charcoal) are not recommended. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
Racecadotril can be considered in the management of AGE. |
Intermediate |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
Bismuth subsalicylate is not recommended in the management of children with AGE. |
Low |
Conditional |
|
|
|
ESPGHAN/ESPID 2014 |
New evidence has confirmed that probiotics are effective in reducing the duration of symptoms in children with AGE. |
High |
Strong |
|
|
|
ESPGHAN/ESPID 2014 |
The use of prebiotics in the management of children with AGE is not recommended. |
Intermediate |
Strong |
|
Home management |
|
|
||||
|
Anti- Infective Therapy |
|
|
||||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
|
ESPGHAN/ESPID 2014 |
Anti-infective therapy should not be given to the vast majority of otherwise healthy children with acute gastroenteritis. |
Very Low |
Conditional |
|||
|
|
|
ESPGHAN/ESPID 2014 |
Antibiotic therapy for acute bacterial gastroenteritis is not needed routinely but only for specific pathogens or in defined clinical settings. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
The choice of the antimicrobial agent depends on the local prevalence of the 3 pathogens (Shigella spp, Campylobacter spp, and Salmonella enterica) and the resistance patterns. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
In children with watery diarrhea, antibiotic therapy is not recommended unless the patient has recently traveled or may have been exposed to cholera. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Bloody diarrhea with low or no fever is typical of STEC (enterohemorrhagic E coli), but can be mild shigellosis or salmonellosis. Antibiotics are not recommended unless epidemiology suggests shigellosis. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Parenteral rather than oral antibiotic therapy is recommended for: · Patients unable to take oral medications (vomiting, stupor, etc) · Patients with underlying immune deficiency who have AGE with fever · Severe toxemia, suspected or confirmed bacteremia Neonates and young infants (< 3 months) with fever. Sepsis workup and antibiotics should be considered according to local protocols. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Antiparasitic treatment is generally not needed in otherwise healthy children; however, it may be considered if symptoms are severe. |
Low |
conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Severe cases of giardiasis can be treated with metronidazole, nitazoxanide, albendazole, or tinidazole. |
Low |
conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Cryptosporidiasis should be treated mainly in immunocompromised children with nitazoxanide. |
Low |
conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Amebic colitis should be treated with metronidazole. |
Low |
conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Specific antiviral treatment is usually not indicated in AGE. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Severe cytomegalovirus colitis, especially in an immunocompromised child, should be treated with ganciclovir. |
Low |
conditional |
|
|
Hospital Management |
|
|
||||
|
Hospitalization |
|
|
||||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
|
ESPGHAN/ESPID 2014 |
The recommendations for hospital admission are based on consensus and include any of the following conditions: · Shock · Severe dehydration (>9% of body weight) · Neurological abnormalities (lethargy, seizures, etc) · Intractable or bilious vomiting · Failure of oral rehydration · Suspected surgical condition Conditions for a safe follow-up and home management are not met. |
Very low |
Conditional |
|||
|
|
|
ESPGHAN/ESPID 2014 |
Contact precautions are advised in addition to standard precautions (hand hygiene, personal protective equipment, soiled patient-care equipment, environmental control including textiles, laundry and adequate patient placement). |
Very low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Prompt discharge from hospital should be considered in children admitted for AGE when the following conditions are fulfilled: · Sufficient rehydration is achieved as indicated by weight gain and/or clinical status · IV fluids are no longer required · Oral intake equals or exceeds losses Medical follow-up is available via telephone or office visit. |
Very low |
Conditional |
|
|
2- Rehydration therapy (enteral and parenteral): |
||||||
|
|
|
ESPGHAN/ESPID 2014 |
When oral rehydration is not feasible, enteral rehydration by the nasogastric (NG) route is the preferred method of rehydration, and should be proposed before IV rehydration. |
High |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
Enteral rehydration is associated with significantly fewer major adverse events and a shorter hospital stay than IV rehydration and is successful in most children. |
High |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
The rapid (40–50 mL/kg within 3–6 hours) and standard (24 hours) NG rehydration regimens are equally effective and may be recommended. |
Intermediate |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
IV fluids are required in the following cases: · Shock · Dehydration with altered level of consciousness or severe acidosis · Worsening of dehydration or lack of improvement despite oral or enteral rehydration therapy · Persistent vomiting despite appropriate fluid administration orally or via an NG tube Severe abdominal distension and ileus. |
Very low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Children presenting with shock secondary to AGE should receive rapid IV infusion of isotonic crystalloid solution (0.9% saline or lactated Ringer’s solution) with a 20-mL/kg bolus. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
If the blood pressure has not improved after the first bolus, a second (or even a third) bolus of 20 mL/kg should be administered >10 to 15 minutes and other possible causes of shock should be considered. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Children with severe dehydration requiring IV fluids may receive rapid rehydration with 20mL/kg/h of 0.9% saline solution for 2 to 4 hours. |
Intermediate |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
In IV-rehydrated children, a dextrose-containing solution may be used for maintenance. |
Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
A solution containing not <0.45% saline (at least 77 mmol/L [Na+]) is recommended during the first 24 hours of IV rehydration therapy to prevent hyponatremia. |
Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
After the child starts to urinate and if serum electrolyte values are known, add 20 mmol/L of K+ chloride. |
Very Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Rapid rehydration with 20mL/kg/h for 2 to 4 hours followed by oral rehydration or continuous infusion of dextrose solution is adequate for initial rehydration of most patients requiring hospital assistance. |
Intermediate |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
More rapid IV rehydration may be associated with electrolyte abnormalities and is associated with long time to hospital discharge, and therefore is not recommended. |
Intermediate |
Strong |
|
|
|
|
ESPGHAN/ESPID 2014 |
Isotonic (0.9%) saline solution effectively reduces the risk of hyponatremia and is recommended for initial rehydration in most cases. In the rare but extremely severe cases of shock, Ringer’s lactate solution is recommended. |
Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
Glucose may be added to saline solution once fluid volume has been restored in the subsequent phase of IV rehydration (‘‘maintenance’’). |
Low |
Conditional |
|
|
Management of electrolyte imbalance: |
||||||
|
|
Management Of Hypernatremia: |
ESPGHAN/ESPID 2014 |
Oral or NG rehydration with hypo osmolar ORS is an effective and safe treatment and has fewer adverse effects than IV rehydration. |
Low |
Conditional |
|
|
|
|
ESPGHAN/ESPID 2014 |
If the child is hypernatremia and needs IV rehydration: · Use an isotonic solution (0.9% saline) for fluid deficit replacement and maintenance. · Replace the fluid deficit slowly, typically for 48 hours, with the aim of reducing it to <0.5mmol/L/h. Monitor plasma sodium frequently. |
Low |
Conditional |
|
|
|
Management of Hyponatremia |
WHO 2005 |
ORS solution is safe and effective therapy for nearly all children with hyponatremia. An exception is children with oedema, for whom ORS solution provides too much sodium. |
Good Practice Statement |
|
|
|
|
Management of Hypokalemia |
WHO 2005 |
Hypokalemia is worsened when base (bicarbonate or lactate) is given to treat acidosis without simultaneously providing potassium. |
Good Practice Statement |
|
|
|
|
|
WHO 2005 |
Hypokalaemia can be prevented, and the potassium deficit corrected, by using ORS solution for rehydration therapy and by giving foods rich in potassium during diarrhea and after it has stopped. |
|
|
|
|
Prevention: |
||||||
|
1- Strategies: |
||||||
|
|
|
IDSA 2017 |
Hand hygiene should be performed after using the toilet, changing diapers, before and after preparing food, before eating, after handling garbage or soiled laundry items, and after touching animals or their feces or environments, especially in public settings such as petting zoos. |
Intermediate (moderate) |
Strong |
|
|
|
|
IDSA 2017 |
Infection control measures including use of gloves and gowns, hand hygiene with soap and water, or alcohol-based sanitizers should be followed in the care of people with diarrhea. |
High |
Strong |
|
|
|
|
IDSA 2017 |
The selection of a hand hygiene product should be based upon a known or suspected pathogen and the environment in which the organism may be transmitted. |
low |
Strong |
|
|
|
|
IDSA 2017 |
Appropriate food safety practices are recommended to avoid cross-contamination of other foods or cooking surfaces and utensils during grocery shopping, food preparation, and storage; ensure that foods containing meats and eggs are cooked and maintained at proper temperatures. |
moderate |
Strong |
|
|
|
|
IDSA 2017 |
Healthcare providers should direct educational efforts toward all people with diarrhea, but particularly to people with primary and secondary immune deficiencies, pregnant women, parents of young children, and the elderly as they have increased risk of complications from diarrheal disease. |
low |
Strong |
|
|
|
|
IDSA 2017 |
Ill people with diarrhea should avoid swimming, water-related activities, and sexual contact with other people when symptomatic while adhering to meticulous hand hygiene. |
low |
Strong |
|
|
2- Vaccination |
||||||
|
|
|
IDSA 2017 |
Rotavirus vaccine should be administered to all infants without a known contraindication. |
high |
Strong |
|
|
|
|
IDSA 2017 |
Two typhoid vaccines (oral and injectable) are licensed in the United States but are not recommended routinely. Typhoid vaccination is recommended as an adjunct to hand hygiene and the avoidance of high-risk foods and beverages, for travelers to areas where there is moderate to high risk for exposure to Salmonella enterica subspecies enterica serovar Typhi, people with intimate exposure (eg, household contact) to a documented Salmonella enterica subspecies enterica serovar Typhi chronic carrier, and microbiologists and other laboratory personnel routinely exposed to cultures of Salmonella enterica subspecies enterica serovar Typhi. Booster doses are recommended for people who remain at rik. |
high |
Strong |
|
- Acknowledgements
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge ESPGHAN/ESPID2014, IDSA 2017, and WHO, 2005 guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️ This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Abeer Mohamed Nour ElDin |
Professor of Pediatrics, NRC |
|
|||
|
Prof. Ayman Emil Eskander |
Professor of Pediatrics, Cairo University |
|
|||
|
Dr. Ehab Waheed Abd Elmohsen |
Ass. Lecturer of Pediatrics, AFCM |
|
|||
|
Prof. Manal Sadek Eldefrawy |
Professor of Pediatrics, Benha University |
|
|||
|
Prof. Marwa Talaat Eldeeb |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Prof. Mohamed Saad Eldin Eladawy |
Professor of Pediatrics, AFCM/ Ain Shams University |
|
|||
|
Prof. Mohammed Nabih Almohammady |
Professor of Pediatrics, Cairo University |
|
|||
|
Prof. Mosallam Mohamed Nasser |
Professor of Pediatrics, Alazhar University |
|
|||
|
Dr. Yosra Mohsen Awad |
Lecturer of Pediatrics, Ain Shams University |
|
|||
|
Prof. Tarek Omar |
Professor of Pediatrics, Alexandria University |
|
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Mahmoud Taher Elmogy |
Al-Azhar University |
||||
|
Prof. Nabil Abdel-Aziz Mohsen |
Cairo University |
||||
|
Prof. Hatem Hussein |
Zagazig University |
||||
|
International Peer Reviewers |
|||||
|
Hana Szajewska |
University of Warsaw |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AdV |
Adenovirus |
||
|
AFCM |
Armed Force Collage of Medicine |
||
|
AGGAG |
Acute Gastroenteritis Guidelines Adaptation Group |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
AsV |
Astrovirus |
||
|
CPG |
Clinical Practice guidelines |
||
|
CRP |
C-Reactive Protein |
||
|
̊ C |
Degree Celsius |
||
|
CPG |
Clinical Practice Guideline |
||
|
DHS |
Demographic and Health Survey |
||
|
E. Coli |
Escherichia Coli |
||
|
ED |
Emergency Department |
||
|
EPG |
Egyptian Pediatric Guidelines |
||
|
ESPGHAN |
European Society for Pediatric Gastroenterology, Hepatology, and Nutrition |
||
|
ESPID |
European Society for Pediatric Infectious Diseases |
||
|
ETEC |
Enterotoxigenic Escherichia Coli |
||
|
ERG |
External Review Group |
||
|
GRADE |
Grading of recommendations assessment, development and evaluation |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
IBD |
Inflammatory Bowel Disease |
||
|
ICU |
Intensive care unit |
||
|
IDSA |
Infectious Diseases Society of America |
||
|
IV |
Intravenous |
||
|
K+ |
Potassium |
||
|
LGG |
L rhamnosus GG |
||
|
MOH |
Ministry of Health |
||
|
Na+ |
Sodium |
||
|
NGO |
Non-Governmental Organization |
||
|
NoV |
Norovirus |
||
|
NRC |
National Research Center |
||
|
ORS |
Oral Rehydration Salts Solution |
||
|
PHC |
Primary Health Care |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
PICU |
Pediatric Intensive Care Unit |
||
|
RCTs |
Randomized Control Trials |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
||
|
RV |
Rotavirus |
||
|
STEC |
Shiga Toxin-Producing Escherichia Coli |
||
|
WHO |
World Health Organization |
- Glossary
1. Acceptability
Is the extent to which the users are likely to adopt a recommendation It is based on internal qualities such as clarity, comprehensiveness and logical reasoning and on external factors such as the burden imposed on the process and system of care, patient and providers attitudes and beliefs and patients’ needs expectations and preferences.
2. Adaptation (of guidelines)
It is the systematic approach to considering the use and/or modification of guidelines produced in one cultural and organizational setting for application in different context. Adaptation can be used as an alternative to de novo guidelines development or for customizing existing guidelines to suit the local context.
3. Adoption (of a guidelines)
It is the acceptance of guidelines after the assessment of the quality, currency, and content. When health care providers (or other users of recommendations) use the adopted guidelines, they feel committed to change their practices in accordance with the recommendations of the guidelines.
4. Applicability
It is the extent to which the users can put a recommendation into practice, based on internal qualities such as a clearly defined eligible patient population that matches the population to which the intervention is targeted in the local setting and external factors such as the availability of the necessary knowledge, skills, provider time, staff, equipment, and other resources.
Applicability is sometimes taken as a synonym for feasibility:
• Feasibility of the acquisition of necessary skills and knowledge.
• Feasibility of the necessary increase in provider time, staff, equipment, and so on.
5. Culture
Culture represents the norms and values of a specific group, community or population.
6. Diffusion
It is a passive means of transferring knowledge; it is not directed towards a target audience (e.g. publication of articles in medical journals).
7. Dissemination
It is more active than diffusion in that it targets specific audients and involves tailoring the information for these audients (e.g. dissemination strategies including targeted mailings, presentations and press conferences.
8. Evidence-based principles
Evidence-Based Medicine (EBM) has been defined as the conscientious, explicit and judicious use of the current best evidence in making decisions about the care of individual patients. The practice of EBM means integrating individual clinical expertise with the best available external clinical evidence from systematic researches.
9. Evidence tables
They are summaries of the most salient information from studies identified in the systematic review. The elements of evidence tables are dependent on the types of information in studies related to a particular topic but might include information such as the article reference, the study type (e.g. RCT or Cohort), the number of patients and their characteristics and the intervention, comparison arms, outcome measures and effect sizes.
10. Guidelines or Clinical Practice Guidelines (CPG)
Systematically developed statements about specific health problems, intended to assist practitioners and patients in making decisions about appropriate health care.
11. Guidelines consistency
Agreement between the evidence and the recommendations, based on:
• Comprehensiveness of the study search and selection process.
• Coherence between the results of the studies and their interpretation by the guidelines authors.
• Transparency between interpretation and recommendations.
12. Guidelines content
In the ADAPTE Manual and Resource Toolkit for Guidelines Adaptation document, guidelines content refers to the recommendations in the source guidelines.
13. Guidelines currency
A CPG may be considered up to date when no new information on interventions, outcomes and performance justifies updating it.
14. Guidelines quality
By quality of clinical practice guidelines, we mean the confidence that the potential biases of guidelines development addressed adequately and that the recommendations are both internally and externally valid and are feasible for practice. This process involves taking into account the benefits, harms and costs of the recommendations as well as the practical issues attached to them. Therefore, the assessment of quality includes judgments about the methods used for developing the guidelines, the content of the final recommendations, and the factors linked to their uptake.
15. Guidelines topic
In the ADAPTE Manual and Resource Toolkit for Guidelines Adaptation document, the topic refers to the theme of the guidelines, as described in the guidelines title, for a targeted population (disease and patients) and intervention. The purpose, the audience, and the setting intended for the guidelines, although not necessarily explicitly stated in the title, are also part of the topic. A guideline on a given topic may contain more than one health question.
16. Health question or clinical question or key question
It is a precisely described health issue (e.g. clinical, professional practice or public health) relating to the topic of the guidelines? Guidelines may include one or more questions.
17. Implementation
Implementation includes methods to promote the uptake of research findings into routine healthcare in both clinical and policy contexts and hence to improve the quality and effectiveness of healthcare. It includes the study of influences on healthcare professional and organizational behavior.
18. Intra-class correlations
Intra-class correlations provide a measurement of the extent to which two or more raters agree when rating the same set of things. It is a reliability index and is typically a ratio of the variance of interest over the sum of the variance of interest plus error.
19. Recommendation
Recommendation is any statements that promote or advocate a particular course of action in clinical care.
20. Stakeholder
A stakeholder is an individual, group and/or organization with a stake in your decision to implement a guideline. Stakeholders include individuals or groups who will be directly or indirectly affected by the implementation of a guidelines.
21. Source guidelines
In the ADAPTE Manual and Resource Toolkit for Guidelines Adaptation document, source guidelines refer to those guidelines selected to undergo assessment of quality, currency, content, consistency and acceptability/applicability and upon which an adapted guidelines may be based.
- Introduction
Acute Gastroenteritis (AGE) is a common pediatric illness .In the Middle East region, AGE persists as the second major cause of pediatric mortality and morbidity following acute lower respiratory tract infections3,4Acute gastroenteritis (AGE) in children is considered one of the most common causes of visits to health centers, one of the most frequent reason of hospitalization and the third leading cause of death related to infectious diseases worldwide5,6.The incidence of diarrhea ranges from 0.5 to 2 episodes per child per year in children <3 years in Europe8. On average, children below 3 years of age in developing countries experience 3 episodes of diarrhea each year7.Rotavirus (RV)is the leading cause of AGE in infants and young children and the major contributor to hospitalization for diarrhea in countries that have no RV vaccines in their national immunization schedules9,10,11. With the continuing decline in cases of RV-associated AGE, since the implementation of routine childhood vaccination against RV, norovirus (NoV) infection has become the most common cause of medically treated AGE 12,13.In 2012, a study conducted in two locations in Egypt over 2112 children to determine the causes of acute diarrhea in children younger than 5-years seeking treatment. Bacteria were identified as a sole pathogen in 20%, RV in 14% and Cryptosporidium in 5% of the cases. Adenovirus (AdV), astrovirus (AsV), NoV and G. lamblia were detected as the sole pathogen in 2%, 3%, 9% and 7% of the cases, respectively. E. histolytica was never detected as the sole pathogen14.The most common enteric infections in the 0-12 months age group were NoV, RV, enterotoxigenic E.coli (ETEC), AsV, Campylobacter spp. and AdV. When identified, Shigella was more commonly identified during the second year after birth (13-24 months). The rates of infection for Cryptosporidium spp. and G. lamblia were similar among children of both age groups14.High fever (>40 ̊C), overt fecal blood, abdominal pain, and central nervous system involvement each suggests a bacterial pathogen. Vomiting and respiratory symptoms are associated with a viral etiology7.Many diarrheal deaths are caused by dehydration8. It would be helpful to have a common tool to evaluate dehydration. The Clinical Dehydration Scale (CDS) is easy to use in the assessment of dehydration7.Dehydration from AGE of any etiology and at any age, except when it is severe, can be safely and effectively treated in over 90% of cases by Oral Rehydration Salts (ORS) solution8.For prevention of recurrent AGE, exclusive breastfeeding until age six months, and continued breastfeeding with complementary foods until two years of age is considered an important aspect in prevention7.Contact and standard precautions including (hand hygiene, personal protective equipment, soiled patient-care equipment, environmental control including textiles, laundry and adequate patient placement)8.
- Purpose and Scope
This guideline focuses on prevention and management of acute gastroenteritis in infants and young children. The objectives of these Clinical Practice Guidelines are to provide evidence-based guidance on the diagnosis, management and prevention of Acute Gastroenteritis (AGE) specifically adjusted to the customs of the Egyptian community and to integrate it with the already existing health care system through the following:
1- Standardization of clinical practice of acute gastroenteritis in Egypt.
2- Reduction of morbidity and mortality from acute gastroenteritis.
These Clinical Practice Guidelines intended to be used in infants and young children less than 5 years of age presenting with acute gastroenteritis without co-morbidities in all healthcare facilities
Patient Population (P):
· Both genders
· Age: 2months - 5 years
· Disease: Acute gastroenteritis
· Co-morbidities: No
Intervention (I):
· Diagnosis
· Management
· Prevention
Professionals (P):
· Pediatricians, Family Physicians, Emergency Physicians, Primary Health Care Practitioners.
· Nurses.
· Parents and Care-givers.
Outcomes (O):
· Primary outcome: standardization of clinical practice of acute gastroenteritis in Egypt.
· Secondary outcome: reduction of morbidity and mortality from acute gastroenteritis.
Healthcare Settings (H):
· Primary, secondary and tertiary healthcare facilities.
· Governmental: University, MOH, Ministry of Defense, Ministry of Interior.
· Non-governmental: Private, NGO.
- Methods
Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: acute, gastroenteritis, infants, young children, ORS
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1.CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 2 guidelines:
ESPGHAN/ESPID guidelines 2014
2- IDSA guidelines 2017
3-WHO guidelines 2005:
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of gastroenterology.
The main functions of the clinical panel were adolopment of ESPGHAN/ESPID guidelines 2014, IDSA guidelines 2017, WHO guidelines 2005:
Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinician’s subgroup, generation of the EtD frameworks whenever applicable.
External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as eminent international reviewers
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of
the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing gastroenteritis diagnosis, treatment, and prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for management of acute malnutrition
3. Arabic Educational materials for nurses and mothers
Implementation
To improve acute gastroenteritis care and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels, and integrated into clinical practice.
Dissemination involves educating health care providers to improve their awareness, knowledge, and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders, and should take into account local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps in implementing AGE diagnosis, management and prevention strategy into a health system:
1- Develop a multidisciplinary working group.
2- Assess the current status of acute gastroenteritis care delivery, care gaps and current needs.
3- Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4- Identify barriers to, and facilitators of implementation.
5- Select an implementation framework and its component strategies.
6- Develop a step-by step implementation plan:
• Select the target populations and evaluate the outcome.
• Identify the local resources to support the implementation.
• Set timelines.
• Distribute the tasks to the members.
• Evaluate the outcomes.
7- Continuously review the progress and results to determine if the strategy requires modification.
Clinical Practical Guidelines Implementation strategies will focus on:
1. Practitioners
Dissemination and implementation of CPG through the following strategies:
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional–patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. Patients
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behavior.
· Mass media campaigns.
3. Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. Stakeholders
Plans have been made to contact with all the health sectors in Egypt including:
Ministry of Health, Health Insurance Organizations, University Hospitals, Ministry of Interior, Ministry of Defense, Curative Care Organizations, Non-Governmental Organizations, Private sectors and Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative procedures/policies.
· Formularies: Drug safety programs, electronic medication administration records.
Other activities to assist the implementation of the guideline’s recommendations include:
A. International initiative: Dissemination of the presented adapted CPG internationally via sending the final draft to the Guidelines International Network (G.I.N) and the ADAPTE Working Group and contact the CPG developers.
B. Gantt Chart has been designed by the Epidemiologist supervised by Prof. Dr. Tarek Omer to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
The Implementation tools:
Examples of Dissemination and Implementation Proposed Resources
Educational materials based on this Adapted CPG for treatment of acute gastroenteritis in children have been made available in several forms including:
Table (4) Pediatric Dehydration Scale (PDS) for Children16.
|
Characteristics |
0 |
1 |
2 |
|
General appearance |
Normal |
Thirsty, restless or lethargic but irritable when touched |
Drowsy, limp, cold or sweaty ±comatose |
|
Eyes |
Normal |
Slightly sunken |
Extremely sunken |
|
Mucous membranes (tongue) |
Moist |
Sticky |
Dry |
|
Skin Turgor |
Goes back immediately |
Delayed (<2sec) |
Very delayed (>2sec) |
A score of 0 represents no dehydration; a score of 1 to 4, some dehydration; and a score of 5 to 8 moderate/severe dehydration.
Table (5) Indications for Hospitalization7.
|
Indications for Hospitalization: |
|
· Shock |
|
· Severe dehydration (>9% of body weight) |
|
· Neurological abnormalities (lethargy, seizures, etc) |
|
· Intractable or bilious vomiting |
|
· Failure of oral rehydration |
|
· Suspected surgical condition |
|
· Conditions for a safe follow-up and home management are not met · Complicated AGE ( electrolyte imbalance, DIC) |
Table (6) Indications for Discharge7.
|
Indications for Discharge: |
|
· Sufficient rehydration is achieved as indicated by weight gain and/or clinical status |
|
· IV fluids are no longer required |
|
· Oral intake equals or exceeds losses |
|
· Medical follow-up is available via telephone or office visit |
Table (7) Indications for IV Rehydration7.
|
Indications for IV Rehydration: |
|
· Shock |
|
· Dehydration with altered level of consciousness or severe acidosis |
|
· Worsening of dehydration or lack of improvement despite oral or enteral rehydration therapy |
|
· Persistent vomiting despite appropriate fluid administration orally or via an NG tube |
|
· Severe abdominal distension and ileus |
NG= Nasogastric
Table (8) Holliday–Segar Method To Calculate Maintenance Fluid17.
|
Child’s weight |
Baseline daily fluid requirement |
|
1–10 kg |
100 ml/kg |
|
10–20 kg |
1000 ml + 50 ml/kg for each kg >10 kg |
|
>20 kg |
1500 ml + 20 ml/kg for each kg >20 kg |
kg=Kilogram, ml=Milliliter
Table (9) Signs of Shock 18.
|
Signs of compensated shock: |
|
· Tachycardia |
|
· Cool extremities |
|
· Prolonged capillary refill (despite warm ambient temperature) · Weak peripheral pulses compared with central pulses · Normal blood pressure |
|
Signs of decompensated shock: |
|
In addition to the above, these signs include: |
|
· Depressed mental status · Decreased urine output · Metabolic acidosis · Tachypnea |
|
· Weak central pulses |
|
* Signs of decompensated shock include the signs listed above plus hypotension. In the absence of blood pressure measurement, decompensated shock is indicated by the non-detectable distal pulses with weak central pulses in an infant or child with other signs and symptoms consistent with inadequate tissue oxygen delivery. |
Figure (2) Management of a Case of AGE
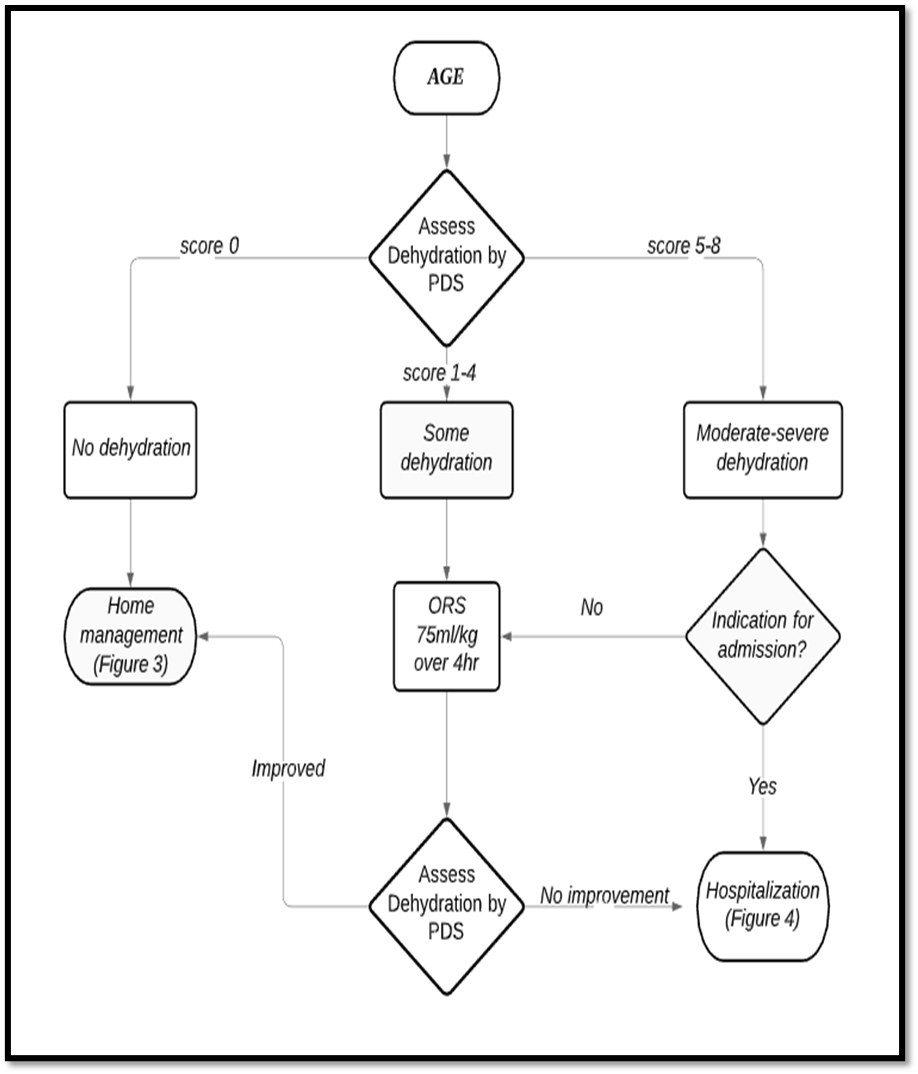
AGE= Acute gastroenteritis, PDS= Pediatric dehydration scale, ORS= Oral rehydration salts solution.
Figure (3) Home Management of a Case of AGE

AGE= Acute gastroenteritis, ORS= Oral rehydration salts solution.
Figure (4) Hospital Management of a Case of AGE
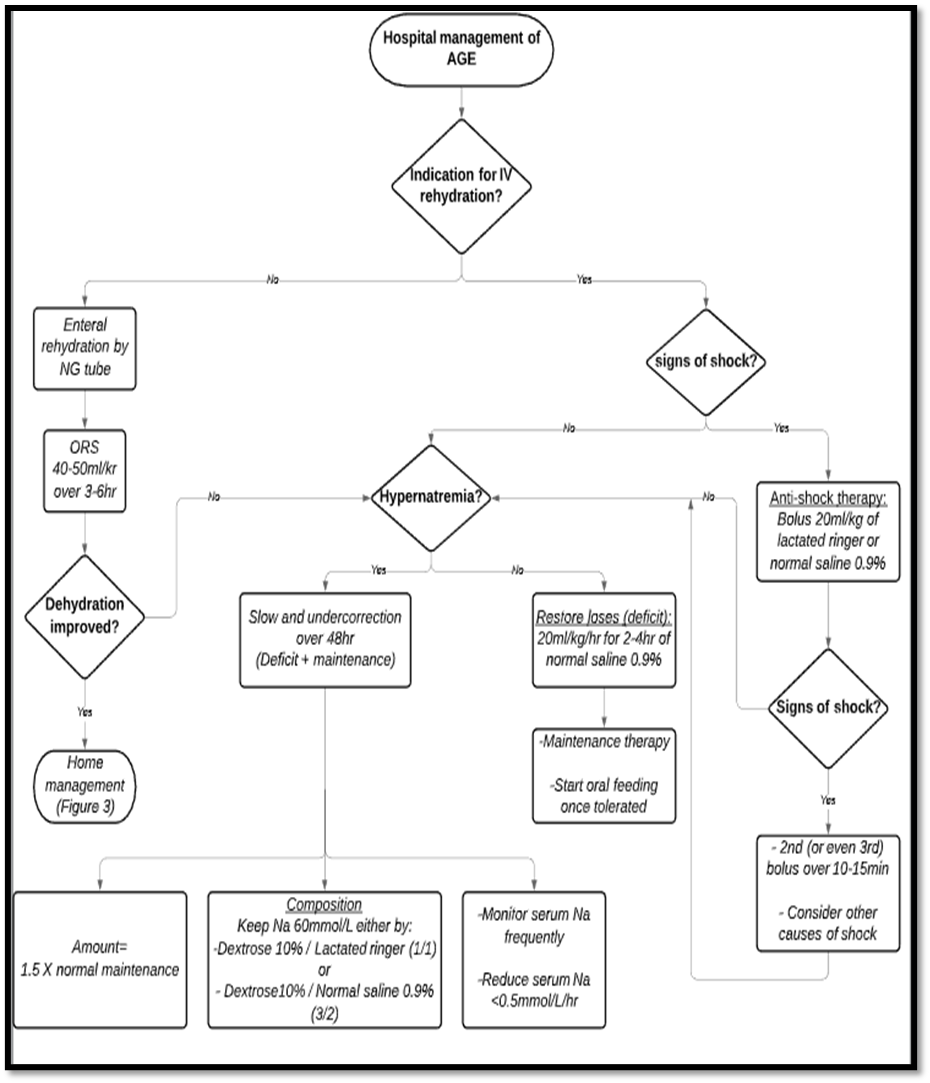
AGE= Acute gastroenteritis, IV= Intravenous, Na= Sodium, NG= Nasogastric, ORS= Oral rehydration salts solution.
علاج النزلات المعوية فى المنزل
1. محلول معالجة الجفاف:
▪️طريقة الاعداد:
يتم اضافة محتويات الكيس على 200 سم
ماء فى كوب نظيف-
يتم اعطاء المحلول للاطفال باستخدام
ملعقة صغيرة نظيفة (للاطفال اقل من عامين) او عن- طريق اخذ رشفات مباشرة من الكوب.
الاطفال اقل من 6 اشهر يمكن اعطائهم
المحلول عن طريق حقنة منزوعة السن ويتم تقطير المحلول فى فم الطفل (لا يتم اعطاء
المحلول فى زجاجات الرضاعة).
▪️الجرعة المناسبة:
- يتم اعطاء الطفل 5-10 مل (1-2 ملعقة صغيرة) لكل كيلوجرام من وزن الطفل بعد كل حركة اسهال.
· طريقة اعطاء محلول معالجة الجفاف:
- يتم اعطاء ملعقة صغيرة او رشفة من الكوب للطفل كل 1-2 دقيقة.
- اذا حدث قئ يتم الانتظار لمدة 10 دقائق ثم نعيد اعطاء المحلول للطفل ببطء أكثر (ملعقة صغيرة او رشفة من الكوب للطفل كل 2-3 دقيقة).
2. تغذية الطفل:
· لا يجب ايقاف التغذية للطفل سواء كانت رضاعة طبيعية او صناعية او غذاء اعتيادى.
· يجب الإكثار من إعطاء السوائل للطفل قدر المستطاع إلى حين توقف الإسهال.
· السوائل التى لا يجب إعطائها للطفل هى:
- المشروبات الغازية
- العصائر التجارية المعلبة
- المشروبات المحلاه بالسكر
- المشروبات التى تحتوى على مادة الكافيين
3. الزنك:
يتم اعطاء شراب الزنك للأطفال الذين يعانون من النزلات المعوية لمدة 14 يوما وتكون الجرعة:
- 10 مجم يوميا للأطفال أقل من 6 أشهر.
- 20 مجم يوميا للأطفال أكبر من 6 أشهر.
4. البروبيوتيك.
▪️ يجب زيارة الطبيب فى الحالات الآتية:
- القئ المستمر
- قيام الطفل بتمرير العديد من البراز المائي
- ارتفاع درجة الحرارة
- الأكل أو الشرب القليل
- العطش الشديد
- وجود دم بالبراز
- عدم تحسن الطفل خلال 3 أيام.
Gantt Chart for the Second Wave: Appendix
- research needs
Future research recommendations for the management of acute gastroenteritis in infants and young children in the Egyptian context could include:
· Relation of gastroenteritis and malnutrition
· Effect of different types of ORS and outcome.
· Epidemiological study of gastroenteritis in Egypt
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for acute gastroenteritis in infants and young children.
Challenges
· High prevalence of gastroenteritis in poor social class
· Malnutrition should be managed in combination in context of malnutrition and dehydration
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for Acute Gastroenteritis in infants and young children:
1. Adherence to Acute Gastroenteritis Guidelines
· Numerator: Number of children with acute gastroenteritis who received treatment as per guideline recommendations.
· Denominator: Total number of children diagnosed with acute gastroenteritis
· Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
· Numerator: Total number of hospitals stay days for children with acute gastroenteritis
· Denominator: Total number of children admitted with acute gastroenteritis
· Data Source: Hospital admission and discharge records.
3. Rate of Readmission
· Numerator: Number of children readmitted with symptoms of acute gastroenteritis within a certain period (e.g., 30 days) after discharge.
· Denominator: Total number of children initially admitted with acute gastroenteritis
· Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG Acute Gastroenteritis GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. ADAPTE Resource Toolkit versions 2.0 (2009): Available from:
www.g-i-n.net/document-store/adapte-resource-toolkit-guidelines-adaptation-version-2.
2. AGREE (II) Instrument available from: www.agreecollaboration.org/instrument/ (downloaded free).976– 987
3. Harb H. The Statistical Bulletin of the Ministry of Public Health (MOPH) 2017. In: Statistical Bulletins of the MoPH. Beirut: Ministry of Public Health (MoPH), World Health Organization (WHO); 2017.
4. World Health Organization. Global Health Observatory data repository 2018. Geneva: World Health Organization; 2018.
5. Akan H, Izbirak G, Gürol Y, et al. Rotavirus and adenovirus frequency among patients with acute gastroenteritis and their relationship to clinical parameters: A retrospective study in Turkey. Asia Pac Fam Med 2009;8:8.
6. Tate JE, Burton AH, Boschi-Pinto C, et al. Global,regional, and national estimates of rotavirus mortalityin children <5 years of age, 2000–2013. ClinInfect Dis 2016;62:96-105.
7. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition/ European Society for Pediatric Infectious Diseases Evidence-Based Guidelines for the Management of Acute Gastroenteritis in Children in Europe: Update 2014 available from: http://www.espghan.org/fileadmin/user_upload/guidelines_pdf/Guidelines_2404/European_Society_for_Pediatric_Gastroenterology_.26.pdf
8. World Health Organization. The treatment of diarrhoea, A manual for physicians and other senior health workers. Available from: https://www.who.int/maternal_child_adolescent/documents/9241593180/en
9. Friesema IH, de Boer RF, Duizer E, et al. Etiology of acute gastroenteritis in children requiring hospitalization in the Netherlands. Eur J Clin MicrobiolInfect Dis 2012;31:405-15.
10. Gimenez-Sanchez F, Delgado-Rubio A, Martinon-Torres F, et al. Rotascore: Research Group Multicenter prospective study analysing the role of rotavirus on acute gastroenteritis in Spain. ActaPaediatr 2010;99:738-42.
11. Aydin H, Aktaş O. Rotavirus genotypes in children with gastroenteritis in Erzurum: first detection of G12P[6] and G12P[8] genotypes in Turkey.Gastroenterology Review 2017; 12:122-7.
12. Hungerford D, Smith K, Tucker A, et al. Populationeffectiveness of the pentavalent and monovalentrotavirus vaccines: a systematic review andmeta-analysis of observational studies. BMC InfectDis 2017;17:569.
13. Payne DC, Vinjé J, Szilagyi PG, et al. Norovirusand medically attended gastroenteritis in U.S. children.N Engl J Med 2013;368:1121–30.
14. El-Mohammady H, Mansour A, Shaheen HI, et al. Increase in the detection rate of viral and parasitic enteric pathogens among Egyptian children with acute diarrhea. J Infect Dev Ctries 2012; 6(11):774-781.
15. Infectious Diseases Society of America ClinicalPractice Guidelines for the Diagnosis and Management of Infectious Diarrhea, 2017. Available from: http://www.saei.org/documentos/biblioteca/biblioteca-capitulo-358-capitulos-50409.pdf
16. Goldman RD, Friedman JN, Parkin PC. Validation of the clinical dehydration scale for children with acute gastroenteritis. Pediatrics. 2008 Sep;122(3):545-549.
17. Holliday MA, Segar WE. The maintenance need for water in parenteral fluid therapy. Pediatrics. 1957;19:823-832.
18. Atkins DL, Berg MD, Berg RA, et al. 2005 American Heart Association (AHA) guidelines for cardiopulmonary resuscitation (CPR) and emergency cardiovascular care (ECC) of pediatrics and neonatal patients: Pediatric advanced life support. Pediatrics, 117(5). http://doi.org/10.1542/peds.2006-0346.
Methodology References:
12. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
13. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
14. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
15. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
16. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
17. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
18. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
19. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Abeer Mohamed Nour ElDin |
Professor of Pediatrics, NRC |
|
|||
|
Prof. Ayman Emil Eskander |
Professor of Pediatrics, Cairo University |
|
|||
|
Dr. Ehab Waheed Abd Elmohsen |
Ass. Lecturer of Pediatrics, AFCM |
|
|||
|
Prof. Manal Sadek Eldefrawy |
Professor of Pediatrics, Benha University |
|
|||
|
Prof. Marwa Talaat Eldeeb |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Prof. Mohamed Saad Eldin Eladawy |
Professor of Pediatrics, AFCM/ Ain Shams University |
|
|||
|
Prof. Mohammed Nabih Almohammady |
Professor of Pediatrics, Cairo University |
|
|||
|
Prof. Mosallam Mohamed Nasser |
Professor of Pediatrics, Alazhar University |
|
|||
|
Dr. Yosra Mohsen Awad |
Lecturer of Pediatrics, Ain Shams University |
|
|||
|
Prof. Tarek Omar |
Professor of Pediatrics, Alexandria University |
|
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
12. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 13. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 14. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 15. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Mahmoud Taher Elmogy |
Al-Azhar University |
||||
|
Prof. Nabil Abdel-Aziz Mohsen |
Cairo University |
||||
|
Prof. Hatem Hussein |
Zagazig University |
||||
|
International Peer Reviewers |
|||||
|
|
|
||||
|
|
|
||||
|
|
|
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the prevention and management of acute gastroenteritis in infants and young children" on PubMed are: gastroenteritits, acute, infants, children
Annex Table 2. Annex Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|