the Management of Gastroesophageal Reflux Disease
| Site: | EHC | Egyptian Health Council |
| Course: | General surgery Guidelines |
| Book: | the Management of Gastroesophageal Reflux Disease |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
7 Sep 2025" Download Guideline
- Executive Summary
These guidelines describe the management of gastroesophageal reflux disease (GERD) including diagnosis and treatment.
Changes were made in recent years particularly as related to approaching extra-esophageal symptoms, refractory GERD, and surgical and endoscopic therapies.
We expect that new diagnostic tools and treatments will be developed and those that we have will be further refined. Future research with advanced endoscopic techniques, data on long-term efficacy of surgical intervention, and advances in artificial intelligence and basic science will almost certainly change the way we manage GERD going forward.
Our recommendations are:
· For patients with classic GERD symptoms of heartburn and regurgitation who have no alarm symptoms, we recommend an 8-week trial of empiric proton pump inhibitor (PPI) once daily before a meal, (Strong recommendation).
· We recommend attempting to discontinue the PPIs in patients whose classic GERD symptoms respond to an 8-week empiric trial of PPIs, (Strong recommendation).
· We recommend diagnostic endoscopy, ideally after PPIs are stopped for 2 to 4 weeks, in patients whose classic GERD symptoms do not respond adequately to an 8-week empiric trial of PPIs, or whose symptoms return when PPIs are discontinued, (Strong recommendation).
· In patients who have chest pain without heartburn and who have had adequate evaluation to exclude heart disease, endoscopy and/or reflux monitoring is suggested, (Conditional recommendation).
· We recommend against the use of a barium swallow solely as a diagnostic test for GERD, (Strong recommendation).
· We recommend endoscopy as the first test for evaluation of patients presenting with dysphagia or other alarm symptoms (weight loss, GI bleeding), and for patients with multiple risk factors for Barrett’s esophagus, (Strong recommendation).
· In patients for whom the diagnosis of GERD is suspected but not clear, and endoscopy shows no objective evidence of GERD, we recommend reflux monitoring be performed off therapy to establish the diagnosis, (Strong recommendation).
· We recommend against performing reflux monitoring off therapy solely as a diagnostic test for GERD in patients known to have endoscopic evidence of Los Angeles grade C or D reflux esophagitis, or in patients with long-segment Barrett’s esophagus, (Strong recommendation).
· We recommend against high resolution manometry (HRM) solely as a diagnostic test for GERD, (Strong recommendation).
· We prefer esophageal manometry for excluding rare motility disorders especially prior to surgery. (Conditional recommendation).
· We recommend weight loss in overweight and obese patients for improvement of GERD symptoms, (Strong recommendation).
· We suggest avoiding meals within 2-3 hours of bedtime, (Conditional recommendation).
· We suggest avoidance of tobacco products/smoking in patients with GERD symptoms, (Conditional recommendation).
· We suggest avoidance of "trigger foods" for GERD symptom control, Conditional recommendation).
· We suggest elevating head of bed for night-time GERD symptoms, (Conditional recommendation).
· We recommend treatment with PPI over treatment with H2RA for healing erosive esophagitis, (Strong recommendation).
· We recommend PPI administration 30 to 60 minutes prior to a meal rather than at bedtime for GERD symptom control, (Strong recommendation).
· For GERD patients who do not have erosive esophagitis or Barrett’s esophagus, and whose symptoms have resolved with PPI therapy, an attempt should be made to discontinue PPIs or to switch to on-demand therapy in which PPIs are taken only when symptoms occur and discontinued when they are relieved, (Strong recommendation).
· For GERD patients who require maintenance therapy with PPIs, we suggest that PPIs should be administered in the lowest dose that effectively controls GERD symptoms and maintains healing of reflux esophagitis, (Conditional recommendation).
· We recommend against routine addition of medical therapies in PPI non-responders, (Strong recommendation).
· We recommend maintenance PPI therapy indefinitely or anti-reflux surgery for patients with Los Angeles grade C or D esophagitis, (Strong recommendation).
· We recommend against treatment with a prokinetic agent of any kind for GERD therapy unless there is objective evidence of gastroparesis, (Strong recommendation).
· We recommend against sucralfate for GERD therapy except during pregnancy, (Strong recommendation).
· We suggest on-demand or intermittent PPI therapy for heartburn symptom control in patients with non-erosive reflux disease, (Conditional recommendation).
· We recommend evaluation for non-GERD causes in patients with possible extra-esophageal manifestations before ascribing symptoms to GERD, (Strong recommendation).
· We recommend that patients who have extra-esophageal manifestations of GERD without typical GERD symptoms (e.g. heartburn, regurgitation) undergo reflux testing for evaluation prior to PPI therapy, (Strong recommendation).
· For patients who have both extra-esophageal and typical GERD symptoms we suggest considering a trial of twice-daily PPI therapy for 8 to 12 weeks prior to additional testing, (Conditional recommendation).
· We suggest that upper endoscopy should not be used as the method to establish a diagnosis of GERD-related asthma, chronic cough, or laryngopharyngeal reflux, (Conditional recommendation).
· We recommend optimization of PPI therapy as the first step in management of refractory GERD, (Strong recommendation).
· We suggest esophageal pH monitoring performed off PPIs if the diagnosis of GERD has not been established by a prior pH monitoring study, (Conditional recommendation).
· For patients who have regurgitation as their primary PPI-refractory symptom and who have had abnormal gastroesophageal reflux documented by objective testing, we suggest consideration of anti-reflux surgery, (Conditional recommendation).
· Esophageal high resolution manometry (HRM) is advised as part of the evaluation for refractory GERD in patients with a normal endoscopy and pH monitoring study, and for patients being considered for surgical or endoscopic treatment, (Conditional recommendation).
· We recommend anti-reflux surgery performed by an experienced surgeon as an option for long-term treatment of patients with objective evidence of GERD, especially those who have severe reflux esophagitis (LA grades C or D), large hiatal hernias, and/or persistent, troublesome GERD symptoms, (Strong recommendation).
· We suggest consideration of Roux-en-Y gastric bypass (RYGB) as an option to treat GERD in obese patients who are candidates for this procedure and who are willing to accept its risks and requirements for lifestyle alterations, (Conditional recommendation).
· Since data on the efficacy of radiofrequency energy (Stretta) as an anti-reflux procedure is inconsistent and highly variable, we recommend against its use as an alternative to medical or surgical anti-reflux therapies, (Strong recommendation).
· We suggest the use of an esophageal dilator, (French Bougie 56) to decrease the long-term incidence of dysphagia, (Conditional recommendation).
· We recommend endoscopic mucosal resection in conjunction with antireflux surgery for patients with Barret’s disease especially with low grade dysplasia, (Good practice statement).
· We recommend considering endoscopic deep mucosectomy in conjunction with anti-reflux surgery for patients with high grade dysplasia of Barret’s disease if endosonography excludes malignant invasion as alternative to esophagectomy, (Good practice statement).
- Recommendations
Section 1: Diagnosis of GERD:
· For patients with classic GERD symptoms of heartburn and regurgitation who have no alarm symptoms, we recommend an 8-week trial of empiric proton pump inhibitor (PPI) once daily before a meal, (Strong recommendation, moderate level of evidence, (1)).
· We recommend attempting to discontinue the PPIs in patients whose classic GERD symptoms respond to an 8-week empiric trial of PPIs, (Strong recommendation, low level of evidence, (1)).
· We recommend diagnostic endoscopy, ideally after PPIs are stopped for 2 to 4 weeks, in patients whose classic GERD symptoms do not respond adequately to an 8-week empiric trial of PPIs, or whose symptoms return when PPIs are discontinued, (Strong recommendation, low level of evidence, (1)).
· In patients who have chest pain without heartburn and who have had adequate evaluation to exclude heart disease, endoscopy and/or reflux monitoring is suggested, (Conditional recommendation, low level of evidence, (1)).
· We recommend against the use of a barium swallow solely as a diagnostic test for GERD, (Strong recommendation, low level of evidence, ( 1)).
· We recommend endoscopy as the first test for evaluation of patients presenting with dysphagia or other alarm symptoms (weight loss, GI bleeding), and for patients with multiple risk factors for Barrett’s esophagus, (Strong recommendation, low level of evidence, ( 1)).
· In patients for whom the diagnosis of GERD is suspected but not clear, and endoscopy shows no objective evidence of GERD, we recommend reflux monitoring be performed off therapy to establish the diagnosis, (Strong recommendation, low level of evidence, (1)).
· We recommend against performing reflux monitoring off therapy solely as a diagnostic test for GERD in patients known to have endoscopic evidence of Los Angeles grade C or D reflux esophagitis, or in patients with long-segment Barrett’s esophagus, (Strong recommendation, low level of evidence, (1)).
· We recommend against high resolution manometry (HRM) solely as a diagnostic test for GERD, (Strong recommendation, low level of evidence, (1)).
· We prefer esophageal manometry for excluding rare motility disorders especially prior to surgery. (Conditional recommendation, low level of evidence, (1)).
Section 2: Medical Management of GERD:
· We recommend weight loss in overweight and obese patients for improvement of GERD symptoms, (Strong recommendation, moderate level of evidence, (1)).
· We suggest avoiding meals within 2-3 hours of bedtime, (Conditional recommendation, low level of evidence, (1)).
· We suggest avoidance of tobacco products/smoking in patients with GERD symptoms, (Conditional recommendation, low level of evidence, (1)).
· We suggest avoidance of "trigger foods" for GERD symptom control, Conditional recommendation, low level of evidence, (1)).
· We suggest elevating head of bed for night-time GERD symptoms, (Conditional recommendation, low level of evidence, (1)).
· We recommend treatment with PPI over treatment with H2RA for healing erosive esophagitis, (Strong recommendation, high level of evidence, (1)).
· We recommend PPI administration 30 to 60 minutes prior to a meal rather than at bedtime for GERD symptom control, (Strong recommendation, moderate level of evidence, (1)).
· For GERD patients who do not have erosive esophagitis or Barrett’s esophagus, and whose symptoms have resolved with PPI therapy, an attempt should be made to discontinue PPIs or to switch to on-demand therapy in which PPIs are taken only when symptoms occur and discontinued when they are relieved, (Strong recommendation, low level of evidence, (1)).
· For GERD patients who require maintenance therapy with PPIs, we suggest that PPIs should be administered in the lowest dose that effectively controls GERD symptoms and maintains healing of reflux esophagitis, (Conditional recommendation, low level of evidence (1)).
· We recommend against routine addition of medical therapies in PPI non-responders, (Strong recommendation, moderate level of evidence, (1)).
· We recommend maintenance PPI therapy indefinitely or anti-reflux surgery for patients with Los Angeles grade C or D esophagitis, (Strong recommendation, moderate level of evidence, (1)).
· We recommend against treatment with a prokinetic agent of any kind for GERD therapy unless there is objective evidence of gastroparesis, (Strong recommendation, low level of evidence, (1)).
· We recommend against sucralfate for GERD therapy except during pregnancy, (Strong recommendation, low level of evidence, (1)).
· We suggest on-demand or intermittent PPI therapy for heartburn symptom control in patients with non-erosive reflux disease, (Conditional recommendation, low level of evidence, (1)).
Section 3: Extra-esophageal GERD Symptoms:
· We recommend evaluation for non-GERD causes in patients with possible extra-esophageal manifestations before ascribing symptoms to GERD, (Strong recommendation, moderate level of evidence, (1)).
· We recommend that patients who have extra-esophageal manifestations of GERD without typical GERD symptoms (e.g. heartburn, regurgitation) undergo reflux testing for evaluation prior to PPI therapy, (Strong recommendation, moderate level of evidence, (1)).
· For patients who have both extra-esophageal and typical GERD symptoms we suggest considering a trial of twice-daily PPI therapy for 8 to 12 weeks prior to additional testing, (Conditional recommendation, low level of evidence, (1)).
· We suggest that upper endoscopy should not be used as the method to establish a diagnosis of GERD-related asthma, chronic cough, or laryngopharyngeal reflux, (Conditional recommendation, low level of evidence, (1)).
Section 4: Refractory GERD:
· We recommend optimization of PPI therapy as the first step in management of refractory GERD, (Strong recommendation, moderate level of evidence, (1)).
· We suggest esophageal pH monitoring performed off PPIs if the diagnosis of GERD has not been established by a prior pH monitoring study, (Conditional recommendation, low level of evidence, (1)).
· For patients who have regurgitation as their primary PPI-refractory symptom and who have had abnormal gastroesophageal reflux documented by objective testing, we suggest consideration of anti-reflux surgery, (Conditional recommendation, low level of evidence, (1)).
· Esophageal high resolution manometry (HRM) is advised as part of the evaluation for refractory GERD in patients with a normal endoscopy and pH monitoring study, and for patients being considered for surgical or endoscopic treatment, (Conditional recommendation, low level of evidence, (2)).
Section 5: Surgical and Endoscopic Options for GERD:
· We recommend anti-reflux surgery performed by an experienced surgeon as an option for long-term treatment of patients with objective evidence of GERD, especially those who have severe reflux esophagitis (LA grades C or D), large hiatal hernias, and/or persistent, troublesome GERD symptoms, (Strong recommendation, moderate level of evidence, (1)).
· We suggest consideration of Roux-en-Y gastric bypass (RYGB) as an option to treat GERD in obese patients who are candidates for this procedure and who are willing to accept its risks and requirements for lifestyle alterations, (Conditional recommendation, low level of evidence, (1)).
· Since data on the efficacy of radiofrequency energy (Stretta) as an anti-reflux procedure is inconsistent and highly variable, we recommend against its use as an alternative to medical or surgical anti-reflux therapies, (Strong recommendation, low level of evidence, (1)).
· We suggest the use of an esophageal dilator, (French Bougie 56) to decrease the long-term incidence of dysphagia, (Conditional recommendation, high level of evidence, (1)).
Section 6: Management of Barrett’s oesophagus:
· We recommend endoscopic mucosal resection in conjunction with antireflux surgery for patients with Barret’s disease especially with low grade dysplasia, (Good practice statement).
· We recommend considering endoscopic deep mucosectomy in conjunction with anti-reflux surgery for patients with high grade dysplasia of Barret’s disease if endosonography excludes malignant invasion as alternative to esophagectomy, (Good practice statement).
- Acknowledgement
We would like to acknowledge the Guideline General Surgery, (GGS) committee for developing this guideline.
Chair of GGS: Mostafa Abdel-Hamed Soliman, Professor of Surgery, Cairo University.
Moderator of GGS: Mohamed Ali Mohamed Nada, Professor of Surgery, Ain-Shams University.
Members of GGS (Alphabetically):
Abel-Motey Hussein Aly, Professor of Surgery, Cairo University.
Abdel-Wahab Mohamed Ezzat, Professor of Surgery, Ain -Shams University.
Ahmed Abdel-Raouf Elgeidie, Professor of Digestive Surgery, Mansoura University.
Alaa Abdallah, Professor of Surgery, Ain -Shams University.
Atef Abdel-Ghani Salem, Professor of Surgery, Benha University.
Hesham Abdel-Raouf El-Akkad, Professor of Surgery, Ain-Shams University.
Ibrahim El-Zayat, Head of Surgery Department, Aswan University.
Khaled Abdallah El-Fiky, Professor of Surgery, Ain-Shams University.
Khaled Amer, Professor of Surgery, Military Medical Academy.
Khaled Safwat, Professor of Surgical Oncology and Endoscopy, Zagazig University.
Mohamed Ibrahim Abdel-Hamed Al-Said, Professor of Surgery, Zagazig University.
Tarek Ibrahim, Professor of Surgery, National Liver Institute, Menofia University.
- Abbreviations
|
AGREEII |
Appraisal of Guidelines for Research and Evaluation II |
|
GERD |
Gastroesophageal Reflux Disease |
|
GGS |
Guidelines General Surgery (Committee) |
|
GRADE |
Grading of Recommendations, Assessment, Development and Evaluation |
|
HRM |
High Resolution Manometry |
|
H2RA |
Histamine2 Receptor Antagonist |
|
LA |
Los Angeles |
|
RYGB |
Roux-en-Y Gastric Bypass |
|
PPI |
Proton Pump Inhibitor |
- Introduction
Gastroesophageal reflux disease (GERD) continues to be among the most common diseases seen by gastroenterologists, surgeons, and primary care physicians. Clinically important advances in surgical and endoscopic therapy of GERD have emerged. Our understanding of the varied presentations of GERD, enhancements in diagnostic testing, and approach to patient management have evolved. During this time, scrutiny of proton pump inhibitors (PPI) has increased considerably. While PPIs remain the medical treatment of choice for GERD, multiple publications have raised questions about adverse events, raising doubts about the safety of long-term use and increasing concern about over-prescribing of PPIs. In this document, we provide updated, evidence-based recommendations and practical guidance for the evaluation and management of GERD, including pharmacologic, lifestyle, surgical, and endoscopic management. The management of functional heartburn and other functional upper GI symptoms is beyond the scope of this guideline. The GRADE system was used to evaluate the evidence and the strength of recommendations, (1&2).
- Scope and Purpose
The scope of this guideline is to set recommendations for the diagnose and treatment of GERD. The main purpose of these guidelines is to minimize malpractice and poor surgical decision, to improve the quality of medical care and surgical service, to provide the Good Surgical Practice to our patients, and finally to be cost effective.
- Target Audience
The principle targeted candidates are the practicing surgeons, however endoscopists and radiologists and all specialists involved in the treatment of GERD are also included.
- Methods
A comprehensive search for guidelines was undertaken to identify the relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2022 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in:
• Developing organization/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The GGS decided to adapt the ACG Clinical Guideline: Guidelines for the Diagnosis and Management of Gastroesophageal Reflux Disease, 2022 and AGA Clinical Practice Update on the Personalized Approach to the Evaluation and Management of GERD: Expert Review, 2022 .
Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
· GRADE working group: https://www.gradeworkinggroup.org/
· GRADE online training modules: http://cebgrade.mcmaster.ca/
Specifically, the quality of evidence was graded as ‘High’, ‘Moderate’, ‘Low’ or ‘Very low’, (table 1& 2).
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
these are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research Needs
1. Comparison between adding to, not to add a drainage procedure to Nissen fundoplication for severe GERD associated with delayed gastric emptying.
2. Comparing the regression of Barrett’s epithelium after fundoplication between patients who had prior endoscopic mucosectomy to those who did not.
3. Comparing deep endoscopic mucosectomy to esophagectomy for patients with high grade esophageal mucosal dysplasia after exclusion of invasive cancer by endosonography.
4. Comparing the sensitivity of diagnosing Barrett’s metaplasia by endoscopic biopsies taken during presence of esophagitis to biopsies taken after 3 weeks of high dose PPI treatment.
- Clinical Indicators for Monitoring
1. Upper endoscopy.
2. PH manometry study.
3. Oesophageal biopsy.
- Updating of the Guideline
The GGS committee for guidelines development is responsible for the continuous evaluation of evidence available about GERD. The present guidelines will be updated in case of significant changes based on new evidence.
- Annexes
Table 1: Quality and Significance of the Four Levels of Evidence in GRADE

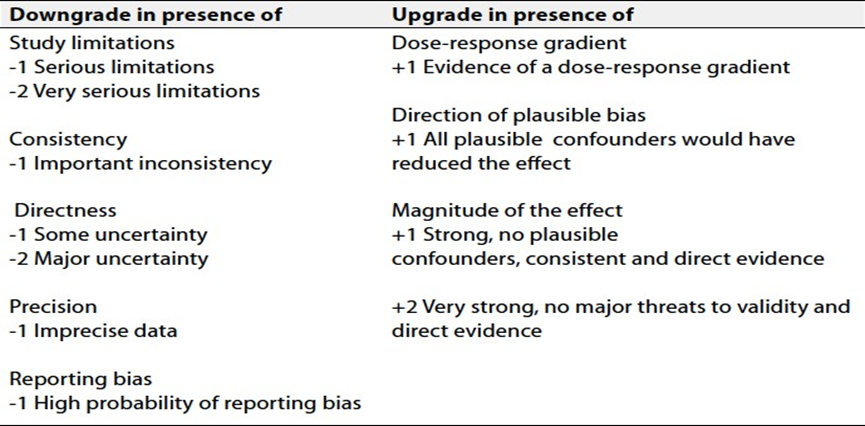
Table 2: Factors that Determine How to Upgrade or Downgrade the Quality of Evidence
- References
1. ACG Clinical Guideline: Guidelines for the Diagnosis and Management of Gastroesophageal Reflux Disease. Am J Gastroenterol. 2022 January 01; 117(1): 27–56. doi:10.14309/ajg.0000000000001538.
2. AGA Clinical Practice Update on the Personalized Approach to the Evaluation and Management of GERD: Expert Review. Clin Gastroenterol Hepatol. 2022 May ; 20(5): 984–994.e1. doi:10.1016/j.cgh.2022.01.025.