Cow Milk Protein allergy
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Cow Milk Protein allergy |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
29 Sept 2025" Download Guideline
- Executive Summary
Cow’s milk protein allergy (CMPA) is the most common food allergy in infancy and early childhood, with a variable clinical presentation ranging from mild gastrointestinal symptoms to life-threatening anaphylaxis. The increasing awareness and recognition of food allergies worldwide, along with the significant impact of CMPA on infant growth, family life, and health systems, highlight the need for standardized, evidence-based diagnostic and management protocols.
This Clinical Practice Guideline (CPG) provides adapted, evidence-informed recommendations for the diagnosis and management of CMPA in children, tailored to the Egyptian healthcare context. It draws upon internationally recognized guidelines, specifically the BSACI 2014 and DRACMA 2010 guidelines, and integrates them with local clinical expertise to ensure feasibility, effectiveness, and cultural appropriateness.
The guideline aims to support physicians, especially pediatricians, family doctors, and primary care providers, in making accurate diagnostic decisions and optimizing treatment pathways for children suspected to have CMPA. Ultimately, the goal is to improve patient outcomes, reduce diagnostic errors, and enhance resource utilization across the national healthcare system.
This guideline focuses on prevention
and management of CMPA
. Is intended for use by all healthcare professionals involved in the care of
infants and children with cow’s milk protein allergy (CMPA), including
pediatricians, general practitioners, allergists, dietitians, and other allied
healthcare professionals.
It covers the diagnosis, management, and follow-up of IgE-mediated and non-IgE
mediated CMPA in different healthcare settings, aiming to standardize care,
improve outcomes, and ensure evidence-based practice.
Cow’s milk allergy may be defined as a reproducible adverse reaction of an immunological nature induced by cow’s milk protein. Strong
CMA can be classified into immediate onset and delayed onset according to timing of symptoms and organ involvement Strong
Symptoms of immediate onset hypersensitivity present within minutes to less than 2hours Strong
IgE mediated symptoms affect several target organs, Skin (urticaria,, angioedema),Respiratory (rhinitis/rhino conjunctivitis/asthma/ wheeze, angioedema / stridor), GIT (oral allergy syndrome, vomiting, pain, flatulence and diarrhea) and or CVS ( anaphylactic shock) Strong
Delayed hypersensitivity symptoms to CMP appear more than 2 hours (usually several hours) up to 8 days. Strong
Non-IgE mediated disease varies widely in clinical presentation from eczema exacerbations to life-threatening shock from gastrointestinal fluid loss secondary to inflammation [Food Protein Induced Enterocolitis Syndrome (FPIES)]
- Gastrointestinal symptoms of non-IgE mediated CMA are variable and affect the entire gastrointestinal tract. CMA should be considered in these circumstances where symptoms fail to respond to standard therapy or where other features of allergy are present. Strong
Lactose intolerance can be confused with non-IgE mediated cow’s milk allergy as symptoms overlap. Lactose intolerance should be considered where patients present only with typical gastrointestinal symptoms Strong
A skin prick test (SPT) weal size ≥ 5 mm or ≥ 2 mm in younger infants) is strongly predictive of CMPA.
Negative skin test results are useful for confirming the absence of IgE-mediated reactions, with negative predictive values exceeding 95% Conditional
In settings where oral food challenge is not a requirement (no expertise or not well prepared), the clinical diagnosis of IgE mediated disease is made by a combination of typically presenting symptoms, e.g. urticaria and/or angio-oedema with vomiting and/or wheeze, soon after ingestion of cow’s milk, and evidence of sensitization (presence of specific IgE). Conditional
In patients with low pretest probability of IgE-mediated CMA we suggest using milk-specific IgE measurement with a cut-off value of ‡0.35 IU/L as a triage test to avoid oral food challenge Strong Conditional
The use of milk elimination is an integral step toward the diagnosis of CMA
Conditional
The treatment following the diagnosis of cow’s milk allergy is complete avoidance of cow’s milk and foods containing cow’s milk. Conditional
Diagnostic dietary elimination should be maintained for at least 6 weeks Conditional
In settings where an oral food challenge is a requirement (routinely done) in all patients suspected of IgE mediated CMA, we recommend using oral food challenge with cow’s milk as the only test without measuring a cow’s milk-specific IgE level as a triage or an add-on test to establish a diagnosis. Strong
In practice, OFC is rarely required to make the diagnosis of CMPA . A food challenge may be necessary to confirm the diagnosis in IgE-mediated disease where there is conflict between the history and diagnostic tests.
Food elimination and reintroduction is recommended for the assessment of non-IgE mediated cow’s milk allergy where there is diagnostic uncertainty. Conditional
OFC is not indicated in initial diagnosis of CMPA if:
1- History of anaphylaxis
2- History of Severe delayed reaction (Food Protein Induced Enterocolitis)
3- Generalized immediate allergic reaction with positive specific IgE Conditional
Reintroduction can be performed at home or may need to be supervised in hospital. Conditional
In breast-fed infants breast milk (with elimination of CM from the mother’s diet) is suitable for most infants with cow’s milk allergy.
In children after 2 years of age, a substitute formula may not be necessary.
In infants and children less than 2 years of age, replacement with a substitute formula is mandatory.
The different types of formulas used in CMPA are:
1. Amino acid formula (AAF)
2. Extensively hydrolyzed formula of cow's milk proteins (eHF).
3. Soy formula (SF) more than 6 months of age Conditional
In children with IgE-mediated CMPA at high risk of anaphylactic reactions, we suggest amino acid formula rather than extensively hydrolyzed milk formula. Conditional
In children with IgE-mediated CMPA at low risk of anaphylactic reactions, we suggest extensively hydrolyzed milk formula over amino acid formula. Conditional
In children with IgE-mediated CMA, we suggest extensively hydrolyzed milk formula rather than soy formula.
Conditional
Individuals should be reassessed at 6-12 monthly intervals from 12 months of age to assess for suitability of reintroduction. strong
The reintroduction is achieved by a graded exposure according to the milk ladder with the less allergenic baked milk. Conditional
Reintroduction can be performed at home or may need to be supervised in hospital. Conditional
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- Diagnosis and Rationale for
Action against Cow’s Milk Allergy (DRACMA) Guidelines (2014).
2- Milk allergy guideline developed by WAO (2023).
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Adaptation for 2 recommendations according to GRADE criteria to be suitable to our Economic implications (Evidence-to-Decision (EtD) table was done)
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following three sub-sections:
A.Diagnosis of Cow’s Milk Allergy (CMA) The guideline covers the age groups from birth to 2 years. This guideline emphasizes the importance of early and accurate diagnosis of CMA, including clinical suspicion, history taking, and diagnostic tests (such as elimination diets, skin prick tests, specific IgE, and oral food challenges.
B. This section includes recommendations and good practice statements on nutritional management, formula selection, breastfeeding support, pharmacological treatment (if needed), monitoring growth and development, and duration of dietary elimination.
C. This section includes recommendations and good practice statements on Prevention of CMPA through exclusive breastfeeding for the first 6 months of life, and the potential benefit of early introduction of allergenic foods after 4 months of age to reduce the risk of CMA.
We can summarize the guidelines’ recommendations for CMPA in the following:
• Appropriate diagnostic tools for diagnosis of
CMPA.
• Nutritional management.
• Indications for use of different infant formulas.
• Role of breastfeeding and maternal diet.
• Prevention strategies.
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/- Recommendations
Table 3. Recommendations |
|
| |||
A. Management of Cow Milk Allergy |
|
| |||
N | Health questions | Source Guideline | Recommendations | Quality of evidence | Strength of Recommendation |
A1 | When to suspect cow milk allergy (CMA)? | BSACI 2014 | Cow’s milk allergy may be defined as a reproducible adverse reaction of an immunological nature induced by cow’s milk protein. | High | Strong |
| Sub questions |
|
|
|
|
1a | What is the classification of CMA?
| BSACI 2014
| CMA can be classified into immediate onset and delayed onset according to timing of symptoms and organ involvement | High | Strong |
1b | When are symptoms expected in the immediate onset hypersensitivity to CMP or acute onset CMA? | BSACI 2014
| Symptoms of immediate onset hypersensitivity present within minutes to less than 2hours
| Moderate | Strong |
1c | What are the symptoms of the immediate hypersensitivity? | BSACI 2014
| IgE mediated symptoms affect several target organs Skin (urticaria, angioedema) Respiratory (rhinitis/rhino conjunctivitis/asthma/ wheeze, angioedema / stridor) GIT (oral allergy syndrome , vomiting, pain, flatulence and diarrhea) And or CVS ( anaphylactic shock) | Moderate | Strong |
1d | When are symptoms expected in delayed hypersensitivity to CMP? | BSACI 2014
| Delayed hypersensitivity symptoms to CMP appear more than 2 hours (usually several hours) up to 8 days. | Moderate | Strong |
1e | What are the symptoms of delayed hypersensitivity to CMP? | BSACI 2014
| Non-IgE mediated disease varies widely in clinical presentation from eczema exacerbations to life-threatening shock from gastrointestinal fluid loss secondary to inflammation [Food Protein Induced Enterocolitis Syndrome (FPIES)] - Gastrointestinal symptoms of non-IgE mediated CMA are variable and affect the entire gastrointestinal tract. CMA should be considered in these circumstances where symptoms fail to respond to standard therapy or where other features of allergy are present. | Moderate | Strong |
Table 4. Recommendations |
|
| |||
B. Management of Cow Milk Allergy |
|
| |||
N | Health questions | Source Guideline | Recommendations | Quality of evidence | Strength of Recommendation |
2 | When should lactose intolerance be considered? | BSACI 2014 | Lactose intolerance can be confused with non-IgE mediated cow’s milk allergy as symptoms overlap. Lactose intolerance should be considered where patients present only with typical gastrointestinal symptoms. | Moderate | Strong |
3 | What is the value of skin prick test in the diagnosis of suspected IgE-mediated CMPA?
| BSACI 2014
BSACI 2014
| A skin prick test (SPT) weal size ≥ 5 mm or ≥ 2 mm in younger infants) is strongly predictive of CMPA.
Negative skin test results are useful for confirming the absence of IgE-mediated reactions, with negative predictive values exceeding 95% |
Low
Low | Conditional
Conditional |
4 | Should in vitro specific IgE determination be used for the diagnosis of suspected IgE-mediated CMPA? | BSACI 2014
DRACMA 2022 | In settings where oral food challenge is not a requirement (no expertise or not well prepared), the clinical diagnosis of IgE mediated disease is made by a combination of typically presenting symptoms, e.g. urticaria and/or angio-oedema with vomiting and/or wheeze, soon after ingestion of cow’s milk, and evidence of sensitization (presence of specific IgE).
In patients with low pretest probability of IgE-mediated CMA we suggest using milk-specific IgE measurement with a cut-off value of ‡0.35 IU/L as a triage test to avoid oral food challenge | Moderate
Low | Strong
Conditional |
5 | What is the role of diagnostic elimination diet after suspicion of CMPA? | BSACI 2014 | The use of milk elimination is an integral step toward the diagnosis of CMA | Very low | Conditional
|
| Sub questions |
|
|
|
|
5a | What to eliminate? | BSACI 2014
| The treatment following the diagnosis of cow’s milk allergy is complete avoidance of cow’s milk and foods containing cow’s milk | Very low | Conditional
|
5b | For how long? | BSACI 2014
| Diagnostic dietary elimination should be maintained for at least 6 weeks | Low
| Conditional
|
6 | What is the role of oral food challenge (OFC) test in CMPA diagnosis? |
|
|
|
|
| Sub questions |
|
|
|
|
6a | When to do a diagnostic OFC? | DRACMA 2022
BSACI 2014
BSACI 2014
BSACI 2014
| In settings where an oral food challenge is a requirement (routinely done) in all patients suspected of IgE mediated CMA, we recommend using oral food challenge with cow’s milk as the only test without measuring a cow’s milk-specific IgE level as a triage or an add-on test to establish a diagnosis
In practice, OFC is rarely required to make the diagnosis of CMPA
A food challenge may be necessary to confirm the diagnosis in IgE-mediated disease where there is conflict between the history and diagnostic tests.
Food elimination and reintroduction is recommended for the assessment of non-IgE mediated cow’s milk allergy where there is diagnostic uncertainty. | High
Very low
Very low
Low | Strong
Conditional
Conditional
Conditional
|
6b | When not to do a diagnostic OFC? | Panel Consensus
| OFC is not indicated in initial diagnosis of CMPA if: 1- History of anaphylaxis 2- History of Severe delayed reaction (Food Protein Induced Enterocolitis) 3- Generalized immediate allergic reaction with positive specific IgE |
Very low
|
Conditional
|
6c | Where to do OFC diagnostic test? | BSACI 2014
| Reintroduction can be performed at home or may need to be supervised in hospital. | Very low
| Conditional
|
|
|
|
|
| |
Table 4. Recommendations |
|
| |||
B. Management of Cow Milk Allergy |
|
| |||
N | Health questions | Source Guideline | Recommendations | Quality of evidence | Strength of Recommendation |
7 | What are the milk substitutes? | In breast-fed infants breast milk (with elimination of CM from the mother’s diet) is suitable for most infants with cow’s milk allergy. In children after 2 years of age, a substitute formula may not be necessary. In infants and children less than 2 years of age, replacement with a substitute formula is mandatory. The different types of formulas used in CMPA are: 1. Amino acid formula (AAF) 2. Extensively hydrolyzed formula of cow's milk proteins (eHF). 3. Soy formula (SF) more than 6 months of age | Very low | Conditional | |
7a | When it is preferred to use AAF over eHf in IgE-mediated CMPA? | DRACMA 2022 | In children with IgE-mediated CMPA at high risk of anaphylactic reactions, we suggest amino acid formula rather than extensively hydrolyzed milk formula. | Very low
| Conditional
|
7b | When it is preferred to use eHF over AAF in IgE-mediated CMPA | DRACMA 2022 | In children with IgE-mediated CMPA at low risk of anaphylactic reactions, we suggest extensively hydrolyzed milk formula over amino acid formula. | Very low | Conditional
|
7c | Which is preferred in IgE-mediated CMPA eHF or SF? | DRACMA 2022 | In children with IgE-mediated CMA, we suggest extensively hydrolyzed milk formula rather than soy formula. | Very low | Conditional
|
| 8-When and how to test for tolerance? Population: children with CMPA |
|
|
|
|
| Sub questions |
|
|
|
|
8a | when to test for tolerance ? | BSACI 2014
| Individuals should be reassessed at 6-12 monthly intervals from 12 months of age to assess for suitability of reintroduction. | Moderate
| Strong
|
8b | How to do it? | BSACI 2014
| The reintroduction is achieved by a graded exposure according to the milk ladder with the less allergenic baked milk) | Very low | Conditional
|
8c | Where to do it? | BSACI 2014
| Reintroduction can be performed at home or may need to be supervised in hospital. | Very low | Conditional
|
Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing cow milk protein allergy (CMPA) diagnosis, treatment, and prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for management of acute malnutrition
3. Arabic Educational materials for nurses and mothers
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Afaf Korraa |
Professor of Pediatrics, Al-Azhar University |
|
|||
|
Prof. Ahmed Hamdy |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Prof. Mohamed Genina |
Professor of Pediatrics, Cairo University |
|
|||
|
Prof. Sanaa Youssef |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Prof. Shereen Reda |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Prof. Somyah Abdel Ghani |
Professor of Pediatrics, Al-Azhar University |
|
|||
|
Tayseer Zayed .Prof |
Professor of Pediatrics, Al-Azhar University |
|
|||
|
Tarek Hamed .Prof |
Professor of Pediatrics, Zagazig University |
|
|||
|
Prof. Suzan Samir |
Professor of Pediatrics, Suez Canal University |
|
|||
|
Prof. Zeinab Awad El-Sayed |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Dr. Baher Hanaa |
Associate professor of Pediatrics, Cairo University |
|
|||
|
Ahmed Hendawy .Dr |
Lecturer of Pediatrics, Al-Azhar University |
|
|||
|
Dr. Nesrine Radwan(Coordinator) |
Lecturer of Pediatrics, Ain Shams University/ AFCM |
|
|||
|
Dr. Sarah Tarek (Coordinator) |
Lecturer of Pediatrics, Cairo University |
|
|||
|
Dr. Hend Fayez (Coordinator) |
Assistant Lecturer of Pediatrics, AFCM |
|
|||
|
Dr. Tarek Omar |
Professor of Pediatrics, Alexandria University |
|
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Dr. Nahla Gamal ElDin Abdel Hakim |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Sami Bahna |
Professor of pediatrics & medicine. Chief of Allergy &Immunology Louisiana State University Health Sciences Center. |
||||
|
Prof. Yvan Vandepllas |
Head of Department of Pediatrics, University Hospital Brussels (UZ Brussel, KidZ Health Castle), and Chair of Pediatrics, Vrije Universiteit Brussel (VUB), Brussels, Belgium. |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge BSACI (British Society for Allergy and Clinical Immunology) 2014 and DRACMA (Diagnosis and Rationale for Action against Cow’s Milk Allergy) 2022 guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
BSACI |
British Society for Allergy and Clinical Immunology |
||
|
CMA |
Cow Milk Protein Allergy |
||
|
CMAGAG |
Cow Milk Protein Allergy Guideline Adaptation Group |
||
|
CPG |
Clinical Practice Guideline |
||
|
DHS |
Demographic and Health Survey |
||
|
DRACMA |
Diagnosis and Rationale for Action against Cow's Milk Allergy |
||
|
eHF |
Extensively Hydrolyzed Formulae |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ERG |
External Review Group |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
PHC |
Primary Health Care |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, Professional, Outcomes, and healthcare context . |
||
|
RCTs |
Randomized Control Trials |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
- Introduction
Cow's milk allergy may be defined as a reproducible adverse reaction to one or more milk proteins (usually caseins or whey β-lactoglobulin).4
Cow's milk allergy is classified by the underlying immune mechanism, timing of presentation and organ system involvement.
➡️Epidemiology
The perception of milk allergy is far more frequent than confirmed CMA.
Symptoms suggestive of cow's milk allergy based on self-reports vary widely, and only about one in three children presenting with symptoms is confirmed to be cow's milk allergic using strict, well defined elimination and open challenge criteria. 5 With these criteria, cow's milk allergy is shown to affect between 1.8% and 7.5% of infants in the first year of life. This may still be an overestimate.6 Clinicians should therefore anticipate that between 2–3% of children have cow's milk allergy.7
Patients with CMA develop gastrointestinal symptoms in 32 to 60% of cases, skin symptoms in 5 to 90%, and anaphylaxis in 0.8 to 9% of cases. This frequency of anaphylaxis is the main concern pointed out in many CMA studies.8
In Egypt, there are no population-based surveys on prevalence of cow milk allergy in adults or children.9
➡️Etiology and pathogenesis of cow’s milk allergy
Cow’s milk allergy (CMA) results from a defect in the immune tolerance toward cow’s milk proteins. The immune reactions involved can be immunoglobulin (Ig) E-mediated, cell-mediated or both (Table 1).10,11
The major milk proteins incriminated in CMA are whey proteins (ß-lactoglobulin) and caseins. 12,13
➡️IgE-Mediated CMA (Immediate hypersensitivity reaction)
The mechanism of IgE-mediated allergy is more understood than non- IgE-mediated reactions and is relatively easily suspected because of rapid onset of symptoms (Table 1). It occurs in 2 steps, the first is the sensitization step that starts when the immune system is abnormally programmed to produce IgE antibodies to milk proteins. These antibodies when binding to the surface of mast cells and basophils, prime them with an allergen-specific trigger. The second step is the activation of the primed mast cells upon re-exposure to milk proteins and subsequently trigger the rapid release of powerful inflammatory mediators. 4,10
➡️Non-IgE-mediated (Delayed hypersensitivity reaction)
In non-IgE-mediated reactions usually there is delayed onset of symptoms occurring within 1 hour to several days following cow’s milk intake. Usually the symptoms are confined to the gut ranging from allergic proctatitis to severe shock-like reactions with metabolic acidosis that is characteristic for the food protein – induced enterocolitis syndrome (Table 1).14-16
➡️Risk Factors
There are several risk factors for development of CMA, such as; artificial feeding, prematurity, antibiotic use, over intake or deficiency of vitamin D and family history.
➡️Diagnosis of CMA
The diagnosis of CMA should be undertaken in a stepwise approach, starting with history taking, clinical examination, and tests for specific IgE and dietary cow milk elimination/challenge.
➡️Clinical assessment
History taking and physical examination can establish the likelihood of the diagnosis, suggest whether an IgE or non-IgE mechanism is involved, can provide a guide to appropriately select and interpret further investigations and identify the potential food triggers.
A thorough medical history should ascertain the following information:
- The food suspected of provoking the reaction and the quantity ingested,
- The form of food (raw, cooked or baked),
- The length of time between ingestion and the development of symptoms,
- Whether ingesting the suspected food produced similar symptoms on other occasions,
- How long ago the patient experienced the last reaction to the food.
➡️Clinical examination: In conjunction with the medical history, clinical examination helps determine the useful diagnostic test or tests.17 Atopic asthma, allergic rhinitis, or atopic dermatitis might indicate an increased risk of IgE-mediated food allergy. Failure to thrive or dermatitis herpetiformis, might indicate other non–IgE-mediated, autoimmune, or nonimmunologic disease. The clinical evaluation should include examination of nutritional status and growth.18
Table 1 Main characteristics of IgE-mediated and non-IgE-mediated allergy
|
Characteristic |
IgE-mediated |
Non-IgE-mediated |
|
Time from exposure to reaction |
Minutes to 2 h |
Several hours to days |
|
Severity |
Mild to anaphylaxis |
Mild to moderate. May cause failure to thrive and edema |
|
Duration |
May persist beyond 1 year of age |
Usually resolved by 1 year of age |
|
Diagnosis |
Specific serum IgE, skin prick tests Oral challenge |
Elimination Oral challenge |
Adapted from: Lifschitz C, Szajewska H;201519
➡️Clinical spectrum of CMA:
From a clinical and diagnostic standpoint, it is most useful to subdivide food hypersensitivity disorders according to the predominant target organ and immune mechanisms (Table 2, 3 and 4).
Table 2. Clinical spectrum of food allergy:
|
|
IgE-mediated min to 2 h |
Non-IgE or cell-mediated 2 h to several days |
Combined IgE and cell-mediated |
|
Skin |
Urticaria/ angioedema |
Allergic contact dermatitis |
Atopic dermatitis |
|
Respiratory |
Bronchial hyperreactivity
Rhino-conjunctivitis |
Heiner's syndrome
Extrinsic allergic alveolitis |
Bronchial asthma |
|
GIT |
Oral allergy syndrome (food pollen syndrome)
Gastrointestinal anaphylaxis |
Food protein induced enterocolitis Food protein induced allergic proctocolitis |
Allergic eosinophilic esophagitis
Eosinophilic gastroenteritis |
|
Generalized |
Anaphylaxis, and food associated, exercise-induced anaphylaxis |
||
Table 3: Gastrointestinal food hypersensitivities
|
Disorder |
Mechanism |
Symptoms |
Diagnosis |
|
Pollen‐food allergy syndrome (oral allergy syndrome) |
IgE-mediated |
Mild pruritus, tingling and/or angioedema of the lips, palate, tongue or oropharynx; occasional sensation of tightness in the throat and rarely systemic symptoms |
Clinical history and positive SPT to relevant food proteins (prick‐to‐prick method); ± oral challenge (positive with fresh food, negative with cooked food) |
|
Gastrointestinal anaphylaxis |
IgE-mediated |
Rapid onset of nausea, abdominal pain, cramps, vomiting, and/or diarrhea; other target organ responses, i.e. skin, respiratory tract, often involved |
Clinical history and positive SPTs or sIgE; ± oral challenge |
|
Allergic eosinophilic oesophagitis |
IgE and/or cell mediated |
Gastro-oesophageal reflux or excessive spitting up or emesis, dysphagia, intermittent abdominal pain, irritability, sleep disturbance, failure to respond to conventional reflux medications |
Clinical history; SPTs; endoscopy and biopsy; elimination diet and challenge |
|
Allergic eosinophilic gastroenteritis |
IgE and/or cell mediated |
Recurrent abdominal pain, irritability, early satiety, intermittent vomiting, failure to thrive and/or weight loss |
Clinical history; SPTs; endoscopy and biopsy; elimination diet and challenge |
|
Food protein induced proctocolitis |
Cell mediated |
Gross or occult blood in stool; typically thriving; usually presents in first few months of life |
SPTs negative; elimination of food protein results in clearing of most bleeding within 72 h; ± endoscopy and biopsy; challenge induces bleeding within 72 h |
|
Food protein induced enterocolitis |
Cell mediated |
Protracted vomiting and diarrhea (± bloody) not infrequently with dehydration; abdominal distention, failure to thrive; vomiting typically delayed 1–3 h post feeding |
SPTs negative; elimination of food protein results in clearing of symptoms within 24–72 h; challenge induces recurrent vomiting within 1–2 h, ∼15% develop hypotension |
|
Food protein induced enteropathy, e.g. coeliac disease (gluten sensitive enteropathy) |
Cell mediated |
Diarrhea or steatorrhea, abdominal distention and flatulence, weight loss or failure to thrive, ± nausea and vomiting, oral ulcers |
Endoscopy and biopsy IgA; elimination diet with resolution of symptoms and food challenge; coeliac disease: IgA anti-gliadin and anti-transglutaminase antibodies |
sIgE, specific immunoglobulin E; SPT, skin prick test. Quoted from Sampson, 200520
Table 4. Respiratory food hypersensitivities:
|
Disorder |
Mechanism |
Symptoms |
Diagnosis |
|
Allergic rhino-conjunctivitis |
IgE mediated |
Periocular pruritus, tearing, and conjunctival erythema, nasal congestion, rhinorrhea, sneezing |
Clinical history, SPTs, elimination diet, food challenge |
|
Asthma |
IgE and cell mediated |
Cough, dyspnea, wheezing |
Clinical history, SPTs, elimination diet, food challenge |
|
Heiner's syndrome (food induced pulmonary hemosiderosis) |
Unknown |
Recurrent pneumonia, pulmonary infiltrates, iron deficiency anemia, failure to thrive |
Clinical history, peripheral eosinophilia, milk precipitins (if due to milk), ± lung biopsy, elimination diet |
- IgE, immunoglobulin E; SPT, skin prick test. Adapted from Sampson 200520
Prognosis: Several studies found that 15% of children with IgE-mediated CMA remained allergic after 8.6 years while all children with non-IgE-mediated CMA outgrew their allergy by 5 years.6
The Cow's Milk-related Symptom Score (CoMiSS) is a clinical resource for primary healthcare providers which aims to increase awareness of CMPA symptoms.
Co-Miss score for clinical evaluation of CMA:
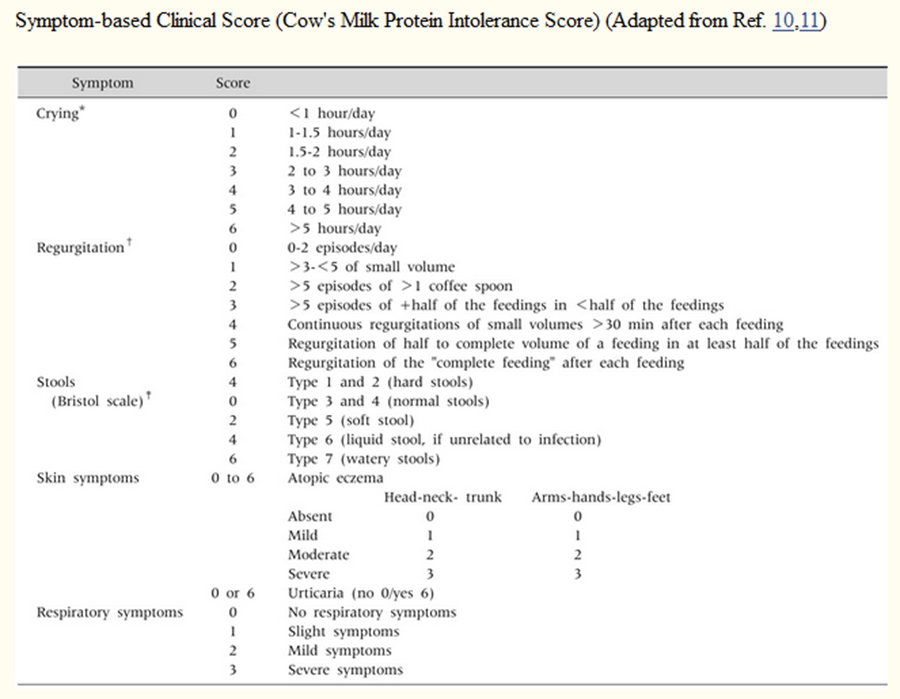
A score of 12 or more indicates that symptoms are cow milk related, potentially CMA. Adapted from DRACMA 2010 and Kattan et al 2016.4,21
➡️Specific IgE-based tests:
Detection of specific IgE in IgE mediated reactions can be performed through either skin prick test (SPT) or serum specific IgE(sIgE) measurement. All IgE based tests should be interpreted in the context of the history. This is because a number of sensitized patients will not react to ingestion of cow milk.22
Serum CMP specific IgE measurement can be especially helpful in patients with severe systemic manifestations for fear of anaphylaxis, those with extensive eczema, or those who could not stop antihistamines where skin testing is not feasible. The sensitivity is greater than 90% for skin testing and 70%–90% for serum food-specific IgE measurement. The specificity of both tests is less than 50%.22,23
In the presence of a good clinical history, cut-off levels for SPT wheal size of ≥ 3 mm larger than the negative control or sIgE ≥ 0.35 kU/L is strongly supporting diagnosis of CMA.24
Results of skin prick tests and specific IgE with ≥ 95% positive predictive values
|
Test |
Infants ≤ 2 years |
> 2 years |
|
Skin prick test (wheal diameter in mm) |
6 |
8 |
|
Specific IgE levels (U/mL) |
5 |
15 |
Quoted from Du Toit et al., 200925.
➡️The basophil activation test (BAT):
The BAT is a flow cytometry-based assay of the expression of activation markers on the surface of basophils following stimulation with allergen.26 It is still not available for practical use in Egypt.
➡️Atopy patch test:
There is insufficient evidence for the routine use of atopy patch test for the evaluation of cow's milk allergy and does not have an additional value in predicting outcomes of OFC.17
Total serum IgE measurements have negligible -if any- role in the diagnosis of food allergy .22
➡️The elimination diet in the work-up of CMA:
Milk elimination is integral to the diagnosis of CMA especially in cases with negative specific IgE result. A definite improvement in symptoms without the need for medication supports the diagnosis. The duration of elimination ranges from 3 to 5 days in children with immediate clinical reactions (eg, angioedema, vomiting, exacerbation of eczema within 2 hours) to 1 to 2 weeks in children with delayed clinical reactions (eg, exacerbation of eczema, rectal bleeding, ). In patients with gastrointestinal reactions (eg, chronic diarrhea, growth faltering), it may take 2 to 4 weeks on a CMP-free diet to judge the response. Care should be undertaken to avoid accidental ingestion and contact with CM and or inhalation of milk vapor (strict elimination).5,27 Goat’s and sheep’s milk should be strictly avoided because of high cross reactivity with CMP.28 Maternal elimination diet is used in case of exclusively breast-fed infants.
➡️Oral Food Challenge (OFC):
The clinician should consider OFC test to confirm the diagnosis of CMA unless there is clinical history consistent with anaphylaxis.16 In patients who demonstrate significant improvement on the elimination diet, the diagnosis of CMA should be confirmed by a standardized oral challenge test. OFC is also indicated to demonstrate tolerance and monitor immunotherapy.27
In the first year of life, a challenge test should be performed with an infant formula based on cow’s milk. Fresh pasteurized cow’s milk can be used above 12 months of age. A false positive challenge due to primary lactose intolerance, may be avoided by using lactose-free CMP-containing formula or milk.27
The starting dose during an oral milk challenge should be lower than a dose that can induce a reaction and then be increased stepwise to 100mL (eg, in children with a delayed reaction, stepwise doses of 1, 3.0, 10.0, 30.0, and 100mL may be given at 30-minute intervals). If severe reactions are expected, then the challenge should begin with minimal volumes (eg, stepwise dosing of 0.1, 0.3, 1.0, 3.0, 10.0, 30.0, and 100mL given at 30-minute intervals). There should be documentation of any signs and symptoms and the amount of milk that provokes symptoms. If no reaction occurs, then the milk should be continued at home every day with at least 200 mL/day for at least 2 weeks. The parents should be contacted by telephone to document any late reactions.27
➡️Differential diagnosis of CMA:
So, the accurate diagnosis depends on high index of suspicion and exclusion of the extremely common functional GI disorders which affects approximately half of infants (40-60%) all over the world.28
For infants presenting with vomiting and posseting: In functional infant regurgitation. the vomiting tends to be effortless and does not upset the infant, and pain is not usually prominent. However, this is not the case in cow’s milk allergic infants with vomiting who are often miserable, rather irritable babies who suffer frequent back-arching and screaming episodes. Feed refusal and aversion to lumps are also prominent features.5
For infants presenting with infant colic: Colic is often confused with simple fussiness, and adding unsortable or inconsolable crying as an additional criterion will help identify ‘true’ colic from simple fussiness [Middle East Consensus Statement on the Diagnosis and Management of Functional Gastrointestinal Disorders in <12 Months Old Infants.28 Observational studies have suggested cow’s milk allergy as a contributing factor in some infants demonstrating extreme colic.5
➡️For infants presenting with diarrhea:
Lactose intolerance can be confused with non-IgE mediated cow’s milk allergy as symptoms overlap. Lactose intolerance should be considered where patients present only with typical gastrointestinal symptoms including abdominal discomfort, bloating, flatulence, and explosive non-bloody watery diarrhea.5
A common cause of diarrhea among infants and toddlers is functional diarrhea (Toddler's diarrhea) which is characterized by daily painless recurrent passage of 4 or more large unformed stools for more than 4 weeks in a well growing child (6 to 60 months age).29
For well thriving infants presenting with bloody stools (suggestive of allergic proctosigmoiditis), exclude presence of anal fissure and gut infections. But if the bleeding is significant or associated with severe anemia, growth failure or systemic manifestations, the differential diagnosis should include primary immune deficiency, inflammatory bowel disease, antibiotic associated diarrhea and other gut conditions.
For children presenting with dysphagia: This symptom always warrants endoscopy to exclude presence of anatomical esophageal abnormalities (stricture, web) and get biopsies for diagnosis of eosinophilic esophagitis.5
Management of CMA:
Principles of management:
Key principles in the management of CMA:
▪️ The key principle in the management of CMA, regardless of the clinical type, is dietary elimination of CMP.
▪️ A substitute formula may not be necessary in infants who are breastfed and children above the age of 2 years.
▪️ Replacement of cow’s milk with a substitute formula is recommended for children below the age of 2 years and non-breastfed children.
➡️Elimination and avoidance of CMP
The management of CMA comprises the avoidance of cow’s milk and cow’s milk products and dietary substitution with an allergenically and nutritionally suitable milk alternative.4 Avoidance of CMP is not limited to exposure via the oral route.30-32 Avoidance of other bovine proteins should be evaluated on a case-by-case basis; while practically all children allergic to beef are allergic to milk,33the opposite is not true.34 A periodical re-evaluation of cow’s milk tolerance every 6-12 months with an open OFC (until tolerance develops) is recommended. Different types of formulas are available to replace cow’s milk in managing CMA.35 The choice of substitute formula should take into account the patient’s preferences, dietary requirements and individual circumstances, as well as cost and availability of the formula.36
Strategies for the management of CMA
➡️Exclusively breastfed children:
Mothers should be encouraged to continue breastfeeding and usually do not require dietary dairy restrictions unless the infant has symptoms whilst being breastfed. The incidence of CMA in exclusively breastfed children is very low; approximately 0.5% of breastfed children have CMA.37 As with all cases of CMA, avoidance of CMP is essential. It takes an average of 2–4 weeks for symptoms to improve or disappear. If symptoms do not improve after 2–4 weeks, an alternative diagnosis should be considered. If symptoms improve or disappear during maternal elimination diet, an open OFC can be performed 6 months later.38
➡️Formula-fed children:
Avoidance of CMP from the diet is essential. eHF is recommended as a substitute to cow’s milk formula in mild-to-moderate cases. AAF is recommended for infants presenting with anaphylaxis, allergic eosinophilic oesophagitis or if the symptoms in mild-to-moderate cases do not improve on eHF after 2–4 weeks.39
➡️Cross reactivity:
Studies on cross reactivity using oral challenges showed that 9.7% of 62 children with CMA reacted to beef. Well-cooked beef is less likely to cause symptoms for those with CMA. There is extensive cross-reactivity among sheep’s, cow’s, and goat’s milk but not with camel’s milk.
Special considerations:
Immunotherapy for CMA:
While there may seem to be potentially large benefits of oral immunotherapy in the management of CMA, frequent and serious adverse events have also been associated with its use. Oral immunotherapy is a promising but still experimental method to treat children with cow’s milk allergy.39 Until further research is done, immunotherapy is not recommended in the treatment of CMPA.
Prevention of cow’s milk protein allergy
Family history is the most important determinant of allergic risk in infancy.
P[ZAE1] revention in high-risk infants:
• Exclusive breastfeeding for 4–6 months. Some recent studies report that early exposure to CMP might promote tolerance.
• When breastfeeding is not possible, consider a hydrolyzed formula. There is no conclusive evidence to support the use of formulas with reduced allergenicity for preventive purposes in healthy infants without a family history of allergic disease.
• Avoidance of CMP during pregnancy is not necessary
- Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of the prevention, diagnosis and management of cow’s milk allergy (CMA).
It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to improve the quality of care for infants and children with CMA, reduce misdiagnosis and delayed diagnosis, and provide consistent messages for healthcare professionals and families. Across all levels of care.
This version of the guideline includes recommendations and good practice statements for:-
•Management of infants and children with suspected or confirmed IgE and non-IgE-mediated CMA across different care settings. including primary care, secondary care, and tertiary hospitals
• Prevention of CMA through infant feeding choices and nutritional support in high-risk infants.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are:
CMPA, CMA, IgE-mediated, non-IgE-mediated, diagnosis, management, oral food challenge, elimination diet, infant formula, pediatric allergy.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2013 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 2 guidelines:
1- BSACI guideline on the management of cow milk protein allergy (2014).
2- DRACMA guideline (Diagnosis and Rationale for Action against Cow's Milk Allergy) (2022).
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Adaptation for 2 recommendations according to GRADE criteria to be suitable to our Economic implications
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of cow milk protein allergy (CMPA) in children.
The main functions of the clinical panel were adolopment of cow milk protein allergy (CMPA) in children. Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinician’s subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in in cow milk protein allergy (CMPA) in children, as well as eminent international reviewers.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of
the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations.
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
➡️GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include awell-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- research needs
Future research recommendations for the management of CMPA in children in the Egyptian context could include:
- Conducting large-scale epidemiological studies to determine the true prevalence and burden of CMPA among Egyptian children.
-Evaluating the effectiveness, safety, and cost-effectiveness of different nutritional interventions (extensively hydrolyzed formulas, amino acid–based formulas, and soy-based alternatives) in local healthcare settings.
- Studying genetic, environmental, and cultural dietary factors that may influence the presentation and severity of CMPA in Egyptian children.
- Developing and testing culturally adapted educational tools for parents, caregivers, and healthcare providers.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for CMPA in children.
➡️Challenges
· Limited availability and high cost of specialized hypoallergenic formulas in low-resource settings.
· Low awareness and variable diagnostic practices among primary healthcare providers.
· Sociocultural factors that may delay seeking medical care or lead to reliance on unproven remedies.
· Lack of structured referral systems between primary care and tertiary allergy/immunology centers.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline
The following are three performance measures or indicators for implementing this adapted CPG for Cow Milk Protein Allergy (CMPA) in children:
1. Adherence to CMPA Guidelines
· Numerator: Number of children with CMPA who received treatment as per guideline recommendations.
· Denominator: Total number of children diagnosed with CMPA.
· Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
· Numerator: Total number of hospitals stay days for children with CMPA.
· Denominator: Total number of children admitted with CMPA.
· Data Source: Hospital admission and discharge records.
3. Rate of Readmission
· Numerator: Number of children readmitted with symptoms of CMPA within a certain period (e.g., 30 days) after discharge.
· Denominator: Total number of children initially admitted with CMPA.
· Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG Gastroenterology, Nutrition and allergy GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
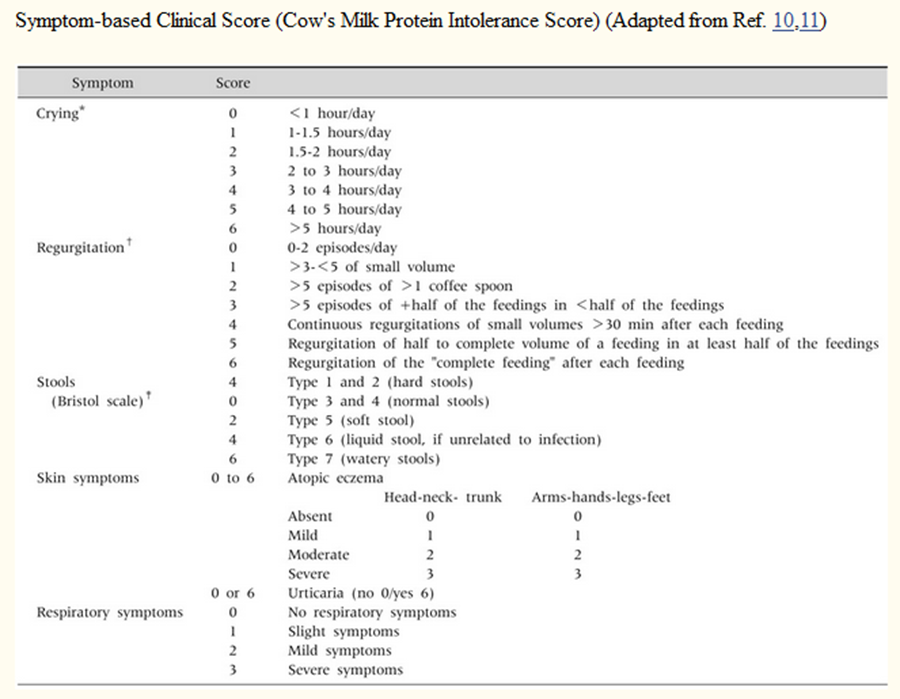
A score of 12 or more indicates that symptoms are cow milk related, potentially CMA. Adapted from DRACMA 2010 and Kattan et al 2016.4,21
- References
1. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
2. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
3. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
4. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
5. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
6. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
7. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
8. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Afaf Korraa |
Professor of Pediatrics,Al-Azhar University |
|
|||
|
Prof.Ahmed Hamdy |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Prof. Mohamed Genina |
Professor of Pediatrics,Cairo University |
|
|||
|
Prof. Sanaa Youssef |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Prof. Shereen Reda |
Professor of Pediatrics,Ain Shams University |
|
|||
|
Prof. Somyah Abdel Ghani |
Professor of Pediatrics, Al-Azhar University |
|
|||
|
Tayseer Zayed .Prof |
Professor of Pediatrics, Al-Azhar University |
|
|||
|
Tarek Hamed .Prof |
Professor of Pediatrics, Zagazig University |
|
|||
|
Prof. Suzan Samir |
Professor of Pediatrics,Suez Canal University |
|
|||
|
Prof. Zeinab Awad El-Sayed |
Professor of Pediatrics, Ain Shams University |
|
|||
|
Dr. Baher Hanaa |
Associate professor of Pediatrics, Cairo University |
|
|||
|
Ahmed Hendawy .Dr |
Lecturer of Pediatrics, Al-Azhar University |
|
|||
|
Dr. Nesrine Radwan(Coordinator) |
Lecturer of Pediatrics, Ain Shams University/ AFCM |
|
|||
|
Dr. Sarah Tarek (Coordinator) |
Lecturer of Pediatrics, Cairo University |
|
|||
|
Dr. Hend Fayez (Coordinator) |
Assistant Lecturer of Pediatrics, AFCM |
|
|||
|
Dr. Tarek Omar |
Professor of Pediatrics, Alexandria University |
|
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Dr. Nahla Gamal ElDin Abdel Hakim |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
|
|
||||
|
Prof. Sami Bahna |
Professor of pediatrics & medicine. Chief of Allergy &Immunology Louisiana State University Health Sciences Center. |
||||
|
Prof. Yvan Vandepllas |
Diensthoofd/Head of Departement. |
||||
|
|
|
||||
|
|
|
||||
|
International Peer Reviewers |
|||||
|
|
|
||||
|
|
|
||||
|
|
|
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the prevention and management of CMPA. " on PubMed are:
CMPA, CMA, IgE-mediated, non-IgE-mediated, diagnosis, management, oral food challenge, elimination diet, infant formula, pediatric allergy.
Annex Table 2. Results of the AGREE II assessment of the source guidelines for BCASI Guidelines for the diagnosis ana Management of Cow’s Milk Allergy
|
AGREE II/ CPGs |
BCASI Guidelines for the diagnosis ana Management of Cow’s Milk Allergy
|
World Allergy Organization and Rationale for Action against Cows Milk Allergy Guidelines |
|
Domain 1 (Scope) |
14 |
17 |
|
Domain 2 (Stakeholder) |
14 |
19 |
|
Domain 3 (Rigour) |
43 |
45 |
|
Domain 4 (Clarity) |
19 |
18 |
|
Domain 5 (Applicability) |
12 |
13 |
|
Domain 6 (Independence) |
11 |
9 |
|
Overall assessment . |
% |
6 |
|
Recommend for use (Overall assessment .) |
YES .,Yes with modifications |
YES .,Yes with modifications |
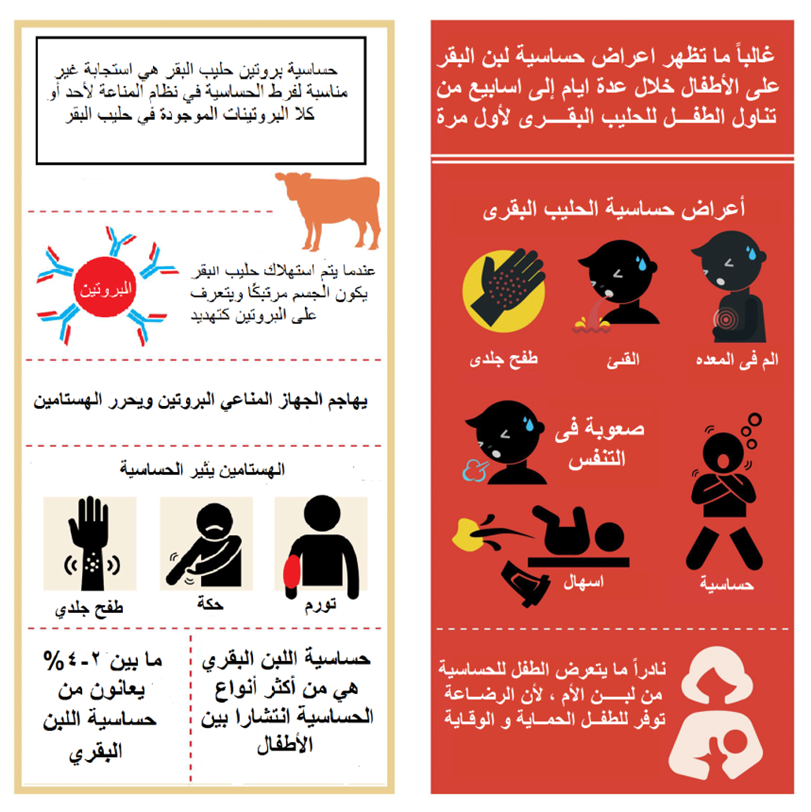
Annex Table 3. Annex Nurses and Parents Educational Guide in Arabic
Implementation Tools:
Arabic Posters: By AFCM Students
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Inclusion/exclusion selection criteria for
retrieved source CPGs checklist
|
Item |
BSAIC |
DRACMA |
ESPGHAN |
EACCI |
|
Method of development |
Evidence based/consensus |
Evidence based |
Evidence based |
consensus |
|
Authors |
Society |
Experts |
Society |
experts |
|
Country (National/International) |
National |
International |
International |
International |
|
Years of Publications |
5 years |
Published 2010 and updated in 2016 < 5years |
> 5 years |
< 5 years |
|
Language |
English |
English |
English |
English |
|
Status (Original/adapted) |
original |
original |
original |
adapted |
|
Decision |
included |
included |
excluded |
excluded |