Localized rectal Cancer
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Localized rectal Cancer |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
21 Oct 2025" Download Guideline
- Executive Summary
|
Strength of the recommendation |
|
|
l From diagnosis, a dedicated multidisciplinary team (MDT) of expert medical oncologists, radiologists, surgeons, radiation oncologists and pathologists should attend regular meetings to discuss patients |
Good practice statement. |
|
l A full medical history and physical examination, including digital rectal examination (DRE), complete blood count, liver and renal function tests and measurement of serum CEA, should be carried out |
Good practice statement. |
|
l Preoperative colonoscopy to the caecal pole and MRI are recommended to determine tumour level. Tumor height must be defined: low = 0 to <5 from anal verge , mid 5 to <10 cm, upper >10 cm. |
Strong |
|
l ERUS (if available) is recommended for T staging of localized tumors in cases of cT1 versus cT2. |
Conditional |
|
l MRI (rectal protocol) is mandatory as part of the staging work-up to stratify for risk-adapted treatment |
Strong |
|
l MRI reports should include description of tumour infiltration depth, node status, lateral lymph nodes, EMVI status and MRF status
|
Strong |
|
l The recommended high-risk criteria are cT4a or cT4b, involved or threatened mesorectal fascia (MRF+), cN2 (4 uspicious nodes), EMVI + and lateral lymph node enlargement of 7 mm .
|
Strong |
|
l Contrast-enhanced CT of the chest and abdomen is recommended for distant staging (if possible) |
Strong |
|
l Fertility risk discussion is recommended in appropriate patients.
|
Good practice statement. |
|
MANAGEMENT OF LOCALISED DISEASE |
|
|
RT and CRT |
|
|
For lower or middle third tumours when surgery is intended: |
|
|
l Preoperative RT followed by LE cannot generally be recommended in patients with cT2 N0 tumours <4 cm but may be considered for selected patients (e.g. elderly or frail patient at high surgical risk). |
Conditional |
|
l Neoadjuvant SCRT or CRT (not TNT) is recommended for patients with cT2 N+, cT3 N0 or cT3 N1 tumours. |
Strong
|
|
For lower or middle third tumours when watch-and-wait approach is intended: |
|
|
l SCRT or CRT is recommended for patients with cT1-cT2 N0 tumours.
|
Strong
|
|
Total neoadjuvant therapy (TNT). |
|
|
l RT should be offered as long-course CRT (50-50.4 Gy in 25-28 fractions with concomitant capecitabine or infusional 5-FU) or SCRT (25 Gy in five fractions) |
Strong |
|
l Consolidation or induction chemotherapy (CAPOX or FOLFOX) should be administered for 4-6 cycles (i.e. 3-4.5 months) |
Strong
|
|
l If FOLFIRINOX regimen is used, it may be administered in line with the protocol of the PRODIGE 23 trial (indications and doses), see Annex. |
Conditional
|
|
For upper third tumours: |
|
|
l TNT should be offered to patients with cT4 or involved or threatened MRF. |
Strong
|
|
l CRT or SCRT should be considered if TNT is not feasible. |
Strong
|
|
l TNT should be offered to patients with high-risk criteria. |
Strong
|
|
For lower or middle third tumours when watch-and-wait approach is intended: |
|
|
l TNT is recommended for patients with high-risk criteria and patients with cT2 N+ or cT3 any N |
Strong |
|
l Upfront CRT followed by consolidation chemotherapy is recommended to increase the likelihood of cCR. |
Strong
|
|
Neoadjuvant Chemtherapy |
|
|
l When considering neoadjuvant chemotherapy, the inclusion criteria of the PROSPECT study should be used (T2 N+, T3 any N, distance to the CRM ≥3 mm, continence-preserving surgery possible). |
Strong
|
|
l Neoadjuvant chemotherapy should comprise 3 months of CAPOX or FOLFOX . |
Strong
|
|
For upper third tumours: |
|
|
l Neoadjuvant chemotherapy is recommended for patients with cT2 N+ or cT3 any N disease. |
Strong
|
|
l Neoadjuvant chemotherapy is recommended for patients with cT4 any N disease. |
Strong
|
|
For lower or middle third tumours when surgery is intended: |
|
|
l Neoadjuvant chemotherapy is recommended for patients with cT2 N+, cT3 N0 or cT3 N1 disease. |
Strong
|
|
l Salvage RT is recommended in case of intolerance to, or progression on, neoadjuvant chemotherapy. |
Strong
|
|
Strong |
|
|
l In case of a cCR, biopsies are not recommended to determine a watch-and-wait approach, as their value in this setting is unclear. |
Strong |
|
Surgery |
|
|
l PME and TME are the recommended surgical procedures for rectal cancer |
Strong |
|
l Open surgery and minimally invasive approaches are both recommended as they lead to similar oncological results |
Strong
|
|
l A distance of >1 mm from tumour to CRM and other organs is recommended. In case of MRF + or T4b, beyond TME surgery is recommended. |
Strong
|
|
l The distal mesorectal margin should be >5 cm. |
Strong |
|
l A distal resection margin of >1 cm is recommended. |
Strong |
|
l Lateral lymph nodes with a short axis of >7 mm should be resected after neoadjuvant treatment |
Strong |
|
For upper third tumours: |
|
|
l PME and TME are both equally recommended. |
Strong |
|
l LE is recommended as an alternative to PME or TME for low-risk tumours (pT1 without unfavourable pathological features). |
Strong |
|
For lower or middle third tumours when surgery is intended: |
|
|
l TME is the recommended surgical procedure. |
Strong |
|
l LE should be considered as an alternative to TME for low-risk tumours (pT1 without unfavourable pathological features). |
Strong |
|
For lower or middle third tumours when watch-and-wait approach is intended: |
|
|
l Surgery, with the resection method depending on clinical assessment, is recommended for patients who do not achieve a cCR following CRT or TNT |
Strong |
|
l In case of local (endorectal) regrowth after a watch-and-wait procedure, salvage resection should be offered to all patients. |
Strong |
|
Watch and wait approach |
|
|
l For lower or middle third tumours, a watch-and-wait strategy is recommended in patients with cCR when organ preservation is intended. |
Strong |
|
l Discussion with patients about the importance of adherence to strict follow-up investigations is mandatory. |
Good Practice Statement
|
|
l Follow-up examinations should comprise MRI, endoscopy and DRE every 3 months for the first 2 years and every 6 months thereafter. CT scans of the chest and abdomen should be carried out every 6 months for the first 2 years and annually thereafter. |
Strong |
|
Adjuvant therapy |
|
|
For upper third tumours: |
|
|
Adjuvant Chemotherapy with a fluoropyrimidine and (potentially) oxaliplatin should be offered (according to clinical risk assessment) following PME or TME alone. |
Strong |
|
In patients who did not receive preoperative RT, adjuvant CRT should be offered in case of CRM positivity, pT4b, pN2 with extracapsular spread close to the MRF or poor-quality TME. |
Strong |
|
For lower or middle third tumours after surgery: |
|
|
l Adjuvant therapy with a fluoropyrimidine and (potentially) oxaliplatin should be offered (according to clinical risk assessment) following TME alone. |
Strong |
|
l Adjuvant therapy with a fluoropyrimidine and (potentially) oxaliplatin is recommended after neoadjuvant CRT or SCRT. |
Strong |
|
l In patients who did not receive preoperative RT, adjuvant CRT should be offered in case of CRM positivity, pT4b, pN2 with extracapsular spread close to the MRF or poor-quality TME. |
Strong |
|
For lower or middle third tumours for watch-and-wait approach: |
|
|
l An adjuvant fluoropyrimidine oxaliplatin combination can be offered on a case-by-case basis after RT or fluoropyrimidine-based CRT in patients achieving cCR with initial cN+ disease |
Conditional |
|
l Post-neoadjuvant systemic treatment following TNT (irrespective of surgical or nonsurgical local approach) cannot be generally recommended due to toxicity considerations. This approach should be discussed individually within an MDT. |
Conditional |
|
FOLLOW-UP, LONG-TERM IMPLICATIONS AND SURVIVORSHIP |
|
|
l Proactive surveillance for local recurrence can be considered in patients at high risk of recurrence (e.g. involved CRM). |
Good practice statement.
|
|
l Clinical assessment should be carried out every 3 months for 2 years |
Good practice statement.
|
|
l Serum CEA measurements can be recommended every 3-4 months for the first 3 years. |
Good practice statement.
|
|
l Annual (minimum) CT scan of the chest, abdomen and pelvis can be recommended after the first 2 years for detection of distant metastases. |
Good practice statement.
|
|
l A completion colonoscopy is recommended within the first year (preferably after 6 months) if not carried out at the time of diagnostic work-up (e.g. if an obstruction was present) |
Good practice statement.
|
|
l Medical history and colonoscopy with resection of colonic polyps can be recommended every 5 years up to the age of 75 years |
Good practice statement.
|
|
l Long-term side-effects of treatment should be monitored |
Good practice statement.
|
- Recommendations
DIAGNOSIS, PATHOLOGY AND MOLECULAR BIOLOGY
▪️ From diagnosis, a dedicated multidisciplinary team (MDT) of expert medical oncologists, radiologists, surgeons, radiation oncologists and pathologists should attend regular meetings to discuss patients
Good practice statement.
▪️ A full medical history and physical examination, including digital rectal examination (DRE), complete blood count, liver and renal function tests and measurement of serum CEA, should be carried out
Good practice statement.
▪️ Preoperative colonoscopy to the caecal pole and MRI are recommended to determine tumour level. Tumor height must be defined: low = 0 to <5 from anal verge , mid 5 to <10 cm, upper >10 cm.
Strong recommendation, low grade evidence (5).
▪️ ERUS (if available) is recommended for T staging of localized tumors in cases of cT1 versus cT2.
Conditional recommendation, high grade evidence (6).
▪️ MRI (rectal protocol) is mandatory as part of the staging work-up to stratify for risk-adapted treatment
Strong recommendation, high grade evidence (6)
▪️ MRI reports should include description of tumour infiltration depth, node status, lateral lymph nodes, EMVI status and MRF status
Strong recommendation, low grade evidence (7).
▪️ The recommended high-risk criteria are cT4a or cT4b, involved or threatened mesorectal fascia (MRF+), cN2 (4 uspicious nodes), EMVI + and lateral lymph node enlargement of 7 mm .
Strong recommendation, low grade evidence (8).
▪️ Contrast-enhanced CT of the chest and abdomen is recommended for distant staging.
Strong recommendation, low grade evidence (9).
▪️ Fertility risk discussion is recommended in appropriate patients.
Good practice statememt
MANAGEMENT OF LOCALISED DISEASE
RT and CRT
For lower or middle third tumours when surgery is intended:
▪️ Preoperative RT followed by LE cannot generally be recommended in patients with cT2 N0 tumours <4 cm but may be considered for selected patients (e.g. elderly or frail patient at high surgical risk).
Conditional recommendation, low grade evidence (10).
▪️ Neoadjuvant SCRT or CRT (not TNT) is recommended for patients with cT2 N+, cT3 N0 or cT3 N1 tumours
Strong recommendation, high grade evidence (11,12).
For lower or middle third tumours when watch-and-wait approach is intended:
▪️ SCRT or CRT is recommended for patients with cT1-cT2 N0 tumours
Strong recommendation, high grade evidence (11,12).
Total neoadjuvant therapy (TNT).
▪️ RT should be offered as long-course CRT (50-50.4 Gy in 25-28 fractions with concomitant capecitabine or infusional 5-FU) or SCRT (25 Gy in five fractions)
Strong recommendation, high grade evidence (13-15).
▪️ Consolidation or induction chemotherapy (CAPOX or FOLFOX) should be administered for 4-6 cycles (i.e. 3-4.5 months)
Strong recommendation, high grade evidence (16).
▪️ If FOLFIRINOX regimen is used, it may be administered in line with the protocol of the PRODIGE 23 trial (indications and doses), see Annex.
Conditional recommendation, high grade evidence (17).
For upper third tumours:
▪️ TNT should be offered to patients with cT4 or involved or threatened MRF
Strong recommendation, high grade evidence (18).
▪️ CRT or SCRT should be considered if TNT is not feasible
Strong recommendation, high grade evidence (19).
For lower or middle third tumours when surgery is intended:
▪️ TNT should be offered to patients with high-risk criteria
Strong recommendation, high grade evidence (13).
For lower or middle third tumours when watch-and-wait approach is intended:
▪️ TNT is recommended for patients with high-risk criteria and patients with cT2 N+ or cT3 any N
Strong recommendation, high grade evidence (20).
▪️ Upfront CRT followed by consolidation chemotherapy is recommended to increase the likelihood of cCR.
Strong recommendation, high grade evidence (20).
Neoadjuvant Chemtherapy
▪️ When considering neoadjuvant chemotherapy, the inclusion criteria of the PROSPECT study should be used (T2 N+, T3 any N, distance to the CRM ≥3 mm, continence-preserving surgery possible)
Strong recommendation, high grade evidence (21-23).
▪️ Neoadjuvant chemotherapy should comprise 3 months of CAPOX or FOLFOX
Strong recommendation, high grade evidence (21-23).
For upper third tumours:
▪️ Neoadjuvant chemotherapy is recommended for patients with cT2 N+ or cT3 any N disease
Strong recommendation, high grade evidence (21-23).
▪️ Neoadjuvant chemotherapy is recommended for patients with cT4 any N disease.
Strong recommendation, very low grade evidence (24).
For lower or middle third tumours when surgery is intended:
▪️ Neoadjuvant chemotherapy is recommended for patients with cT2 N+, cT3 N0 or cT3 N1 disease
Strong recommendation, high grade evidence (21-23).
▪️ Salvage RT is recommended in case of intolerance to, or progression on, neoadjuvant chemotherapy
Strong recommendation, high grade evidence (25).
Restaging before surgery or watch-and-wait approach
▪️ Restaging should comprise MRI, endoscopy and DRE
Strong recommendation, high grade evidence (26,27).
▪️ In case of a cCR, biopsies are not recommended to determine a watch-and-wait approach, as their value in this setting is unclear.
Strong recommendation, low grade evidence (28,29).
Surgery
▪️ PME and TME are the recommended surgical procedures for rectal cancer
Strong recommendation, low grade evidence (30).
▪️ Open surgery and minimally invasive approaches are both recommended as they lead to similar oncological results
Strong recommendation, high grade evidence (31).
▪️ A distance of >1 mm from tumour to CRM and other organs is recommended. In case of MRF + or T4b, beyond TME surgery is recommended.
Strong recommendation, low grade evidence (32).
▪️ The distal mesorectal margin should be >5 cm.
Strong recommendation, low grade evidence (32).
▪️ A distal resection margin of >1 cm is recommended.
Strong recommendation, low grade evidence (33,34).
▪️ Lateral lymph nodes with a short axis of >7 mm should be resected after neoadjuvant treatment
Strong recommendation, very low grade evidence (35).
For upper third tumours:
▪️ PME and TME are both equally recommended.
Strong recommendation, low grade evidence (36).
▪️ LE is recommended as an alternative to PME or TME for low-risk tumours (pT1 without unfavourable pathological features).
Strong recommendation, low grade evidence (37).
For lower or middle third tumours when surgery is intended:
▪️ TME is the recommended surgical procedure.
Strong recommendation, low grade evidence (38).
▪️ LE should be considered as an alternative to TME for low-risk tumours (pT1 without unfavourable pathological features).
Strong recommendation, low grade evidence (37).
For lower or middle third tumours when watch-and-wait approach is intended:
▪️ Surgery, with the resection method depending on clinical assessment, is recommended for patients who do not achieve a cCR following CRT or TNT.
Strong recommendation, high grade evidence (39).
▪️ In case of local (endorectal) regrowth after a watch-and-wait procedure, salvage resection should be offered to all patients.
Strong recommendation, low grade evidence (37).
Watch and wait approach
▪️ For lower or middle third tumours, a watch-and-wait strategy is recommended in patients with cCR when organ preservation is intended.
Strong recommendation, high grade evidence (40).
▪️ Discussion with patients about the importance of adherence to strict follow-up investigations is mandatory.
Good Practice Statement
▪️ Follow-up examinations should comprise MRI, endoscopy and DRE every 3 months for the first 2 years and every 6 months thereafter. CT scans of the chest and abdomen should be carried out every 6 months for the first 2 years and annually thereafter
Strong recommendation, high grade evidence (41).
Adjuvant therapy
For upper third tumours:
▪️ Adjuvant Chemotherapy with a fluoropyrimidine and (potentially) oxaliplatin should be offered (according to clinical risk assessment) following PME or TME alone
Strong recommendation, high grade evidence (42,43).
▪️ In patients who did not receive preoperative RT, adjuvant CRT should be offered in case of CRM positivity, pT4b, pN2 with extracapsular spread close to the MRF or poor-quality TME
Strong recommendation, low grade evidence (44).
For lower or middle third tumours after surgery:
▪️ Adjuvant therapy with a fluoropyrimidine and (potentially) oxaliplatin should be offered (according to clinical risk assessment) following TME alone
Strong recommendation, high grade evidence (42,43).
▪️ Adjuvant therapy with a fluoropyrimidine and (potentially) oxaliplatin is recommended after neoadjuvant CRT or SCRT.
Strong recommendation, low grade evidence (45).
▪️ In patients who did not receive preoperative RT, adjuvant CRT should be offered in case of CRM positivity, pT4b, pN2 with extracapsular spread close to the MRF or poor-quality TME.
Strong recommendation, low grade evidence (44).
For lower or middle third tumours for watch-and-wait approach:
▪️ An adjuvant fluoropyrimidine oxaliplatin combination can be offered on a case-by-case basis after RT or fluoropyrimidine-based CRT in patients achieving cCR with initial cN+ disease
Conditional recommendation, high grade evidence (42,43).
▪️ Post-neoadjuvant systemic treatment following TNT (irrespective of surgical or nonsurgical local approach) cannot be generally recommended due to toxicity considerations. This approach should be discussed individually within an MDT
Conditional recommendation, high grade evidence (42,43).
CLINICAL MONITORING, LONG-TERM IMPLICATIONS, AND SURVIVORSHIP
▪️ Clinical examination, pelvic MRI and/or CT can be recommended for detection of locoregional recurrence.
Good practice statement.
▪️ Proactive surveillance for local recurrence can be considered in patients at high risk of recurrence (e.g. involved CRM)
Good practice statement.
▪️ Clinical assessment should be carried out every 3 months for 2 years
Good practice statement.
▪️ Serum CEA measurements can be recommended every 3-4 months for the first 3 years
Good practice statement.
▪️ Annual (minimum) CT scan of the chest, abdomen and pelvis can be recommended after the first 2 years for detection of distant metastases.
Good practice statement.
▪️ A completion colonoscopy is recommended within the first year (preferably after 6 months) if not carried out at the time of diagnostic work-up (e.g. if an obstruction was present)
Good practice statement.
▪️ Medical history and colonoscopy with resection of colonic polyps can be recommended every 5 years up to the age of 75 years
Good practice statement.
▪️ Long-term side-effects of treatment should be monitored
Good practice statement.
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines, for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines: Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Yousri Wasef & Yousri Rostom.
- Chair of the GIT Cancer Scientific Committee: Prof Yousri Rostom
- The rectal cancer Scientific Group Members: (Alphabitical order): Ahmed EL Kassed, Fouad Aboutaleb, Khaled Abdelkarim, Manal EL Mahdy, Omar S. Omar, and Yousri Wasef.
- Abbreviations
5FU fluorouracil
cCR clinical complete remission
CEA carcinoembryonic antigen
CRT chemo-radiotherapy
CT computed tomography
DRE digital rectal examination
EMVI extramural venous invasion
ERUS endorectal ultrasound
LE local excision
MDT multidisciplinary team
MRF mesorectal fascia
MRI magnetic resonance imaging
PME partial mesorectal excision
RT radiotherapy
SCRT Short course radiotherapy
TME total mesorectal excision
TNT total neoadjuvant treatment
- Introduction
Colo-rectal cancer is the 7th most common cancer in the Egyptian population with more than 5900 newly diagnosed cases and more than 3000 deaths in 2022 (4).
- Purpose and scope
These guidelines will help to improve the quality of care for localized rectal cancer patients via providing a uniform standard of care across the country to help in early diagnosis and treatment for localized rectal cancer, with less aggressive treatment options and improved clinical outcomes. These guidelines cover primary diagnosis, staging, treatment and follow-up of localized rectal cancer patients.
- Target audience
Clinicians who are involved in the care and treatment of patients with localized rectal cancer, including medical oncologists, radiation oncologists, clinical oncologists, onco- and gastrointestinal surgeons, radiologists and pathologists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The NCCN, ESMO, NICE guidelines are the main sources used while formulating the national guidelines for rectal cancer (1-3).
- Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
▪️ GRADE working group:https://www.gradeworkinggroup.org
▪️GRADE online training modules:http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
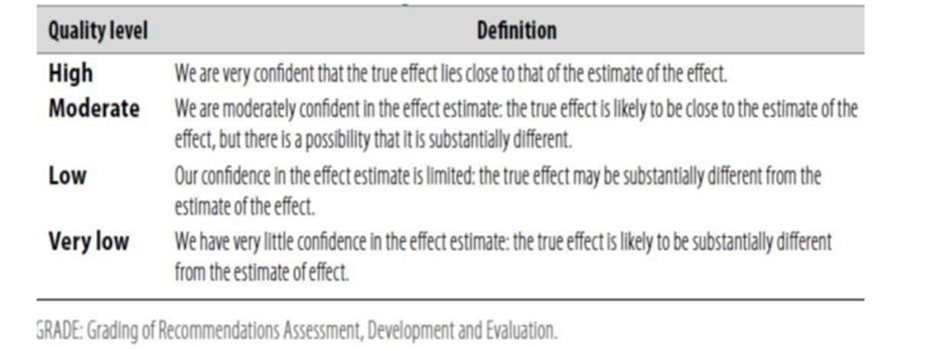
Table 2: Significance of the four levels of evidence

Table 3: Factors that determine how to upgrade or downgrade the quality of evidence.
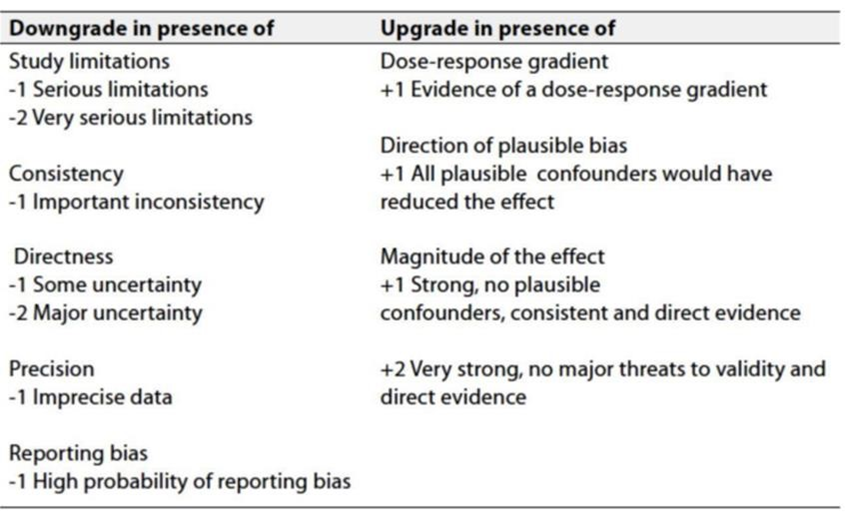
➡️ The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research Gaps
• Evaluation of real world data on the use on new targeted and immune-therapeutic agents in bladder cancer in Egypt.
• Cost effective analysis of new therapeutic agents in Egypt.
• Define the molecular biologic profiles of our Egyptian patients
- Update of the guideline
• This guideline will be updated whenever there is new evidence.
- References
1. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1453.
2. https://www.esmo.org/guidelines/guidelines-by-topic/esmo-clinical-practice-guideline
3. https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/nice-guidelines
4. https://gco.iarc.who.int/media/globocan/factsheets/populations/818-egypt-fact-sheet.pdf
5. Jacobs L, Meek DB, van Heukelom J, Bollen TL, Siersema PD, Smits AB, Tromp E, Los M, Weusten
BL, van Lelyveld N.: Comparison of MRI and colonoscopy in determining tumor height in rectal
cancer.United European Gastroenterol J. 2018 Feb;6(1):131-137.
6. Lahaye MJ, Engelen SM, Nelemans PJ, et al. Imaging for predicting the risk factors–the
circumferential resection margin and nodal dis- ease–of local recurrence in rectal cancer: a meta-analysis. Semin Ultrasound CT MR. 2005;26(4):259-268.
7. Di Fabio F, Allievi N, Lord A, et al. MRI-predicted extramural vascular invasion and tumour deposit are main predictors of disease-free survival in patients undergoing surgical resection for rectal cancer. BJS Open. 2024;8(1):zrad139.
8. Conroy T, Bosset JF, Etienne PL, et al. Neoadjuvant chemotherapy with FOLFIRINOX and preoperative chemoradiotherapy for patients with locally advanced rectal cancer (UNICANCER-PRODIGE 23): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22(5):702-715.
9. Van Cutsem E, Cervantes A, Adam R, et al. ESMO consensus guide- lines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016;27(8):1386-1422.
10. Bujko k., Richter p., Smith F.M. et al., Preoperative radiotherapy and local excision of rectal cancer with immediate radical re-operation for poor responders: A prospective multicentre study January 2013, Radiotherapy and Oncology 106(2).
11. Bujko K, Nowacki MP, Nasierowska-Guttmejer A, et al. Long-term results of a randomized trial comparing preoperative short-course radiotherapy with preoperative conventionally fractionated chemo- radiation for rectal cancer. Br J Surg. 2006;93(10):1215-1223.
12. Ngan SY, Burmeister B, Fisher RJ, et al. Randomized trial of short- course radiotherapy versus long-course chemoradiation comparing rates of local recurrence in patients with T3 rectal cancer: Trans- Tasman Radiation Oncology Group trial 01.04. J Clin Oncol. 2012;30(31):3827-3833.
13. Lutz MP, Zalcberg JR, Glynne-Jones R, et al. Second St. Gallen European Organisation for Research and Treatment of Cancer Gastrointestinal Cancer Conference: consensus recommendations on controversial issues in the primary treatment of rectal cancer. Eur J Cancer. 2016;63:11-24.
14. Dijkstra EA, Nilsson PJ, Hospers GAP, et al. Locoregional failure during and after short-course radiotherapy followed by chemotherapy and surgery compared with long-course chemoradiotherapy and surgery:a 5-year follow-up of the RAPIDO trial, Ann Surg. 2023;278(4).
15. Zhang J, Chi P, Shi L, et al. Neoadjuvant modified infusional fluoro- uracil, leucovorin, and oxaliplatin with or without radiation versus fluorouracil plus radiation for locally advanced rectal cancer: updated results of the FOWARC study after a median follow-up of 10 years. J Clin Oncol. 2025;43(6):633-640.
16. Glimelius B, Khan T, Adolfsson K, et al. Total neoadjuvant treatment using short-course radiotherapy and four CAPOX cycles in locally advanced rectal cancer with high-risk criteria for recurrence: a Swedish nationwide cohort study (LARCT-US). EClinicalMedicine. 2024;75:102771.
17. Conroy T, Castan F, Etienne PL, et al. Total neoadjuvant therapy with mFOLFIRINOX versus preoperative chemoradiotherapy in patients with locally advanced rectal cancer: long-term results of the UNICANCER-PRODIGE 23 trial. Ann Oncol. 2024;35(10):873- 881.
18. Boublikova L., Novakova A., Simsa J.,Lohynska R., Total neoadjuvant therapy in rectal cancer: the evidence and expectations, Crit Rev Oncol Hematol. 2023 Dec:192:104196.
19. van der Valk MJM, Marijnen CAM, van Etten B, et al. Compliance and tolerability of short-course radiotherapy followed by preoperative chemotherapy and surgery for high-risk rectal cancer e results of the international randomized RAPIDO-trial. Radiother Oncol. 2020;147:75-83.
20. Verheij FS, Omer DM, Williams H, et al. Long-term results of organ preservation in patients with rectal adenocarcinoma treated with total neoadjuvant therapy: the randomized phase II OPRA trial. J Clin Oncol. 2024;42(5):500-506.
21. Schrag D, Shi Q, Weiser MR, et al. Preoperative treatment of locally advanced rectal cancer. N Engl J Med. 2023;389(4):322-334.
22. Ding PR, Wang XZ, Li YF, et al. LBA26 Neoadjuvant chemotherapy with CAPOX versus chemoradiation for locally advanced rectal cancer with uninvolved mesorectal fascia (CONVERT): final results of a phase III trial. Ann Oncol. 2023;34(suppl 2):S1267-S1268.
23. Zhang J, Chi P, Shi L, et al. Neoadjuvant modified infusional fluoro- uracil, leucovorin, and oxaliplatin with or without radiation versus fluorouracil plus radiation for locally advanced rectal cancer: updated results of the FOWARC study after a median follow-up of 10 years. J Clin Oncol. 2025;43(6):633-640
24. Zi-Tong Zhang, Wei-Wei Xiao, Li-Ren Li , et al., Neoadjuvant chemoradiotherapy versus neoadjuvant chemotherapy for initially unresectable locally advanced colon cancer: short-term outcomes of an open-label, single-centre, randomised, controlled, phase 3 trial, eClinicalMedicine, . 2024 Sep 21;76:102836. doi: 10.1016/j.eclinm.2024.102836
25. Mari Miyata, Takayuki Ohguri, Katsuya Yahara, Shinsaku Yamaguchi, Hajime Imada, Yukunori Korogi, Salvage radiotherapy for second oligo-recurrence in patients with breast cancer, J Radiat Res
2017 Nov 22;59(1):58–66. doi: 10.1093/jrr/rrx066
26. Lambregts DMJ, Bogveradze N, Blomqvist LK, et al. Current contro- versies in TNM for the radiological staging of rectal cancer and how to deal with them: results of a global online survey and multidisciplinary expert consensus. Eur Radiol. 2022;32(7):4991-5003.
27. Shihab OC, How P, West N, et al. Can a novel MRI staging system for low rectal cancer aid surgical planning? Dis Colon Rectum. 2011;54(10): 1260-1264.
28. Custers PA, Geubels BM, Beets GL, et al. Defining near-complete response following (chemo)radiotherapy for rectal cancer: systematic review. Br J Surg. 2022;110(1):43-49.
29. panelKerKan Tan, Charles B. Tsang, Staging of Rectal Cancer—Technique and Interpretation of Evaluating Rectal Adenocarcinoma, uT1-4, N Disease: 2D and 3D Evaluation, Seminars in Colon and Rectal Surgery Volume 21, Issue 4, December 2010, Pages 197-204.
30. Volkan Özben, Nuri Okkabaz, Turkish Colorectal Cancer Database Study Group, Partial Versus Total Mesorectal Excision for the Surgical Treatment of Mid-Rectal Cancer: An Assessment from the Turkish Society of Colon and Rectal Surgery’s Colorectal Cancer Database, Turk J Colorectal Dis 2024;34:41-49
31. Pimentel-Nunes P, Libânio D, Bastiaansen BAJ, et al. Endoscopic submucosal dissection for superficial gastrointestinal lesions: Euro- pean Society of Gastrointestinal Endoscopy (ESGE) Guideline - update 2022. Endoscopy. 2022;54(6):591-622.
32. Burton S, Brown G, Daniels IR, et al. MRI directed multidisciplinary team preoperative treatment strategy: the way to eliminate positive circumferential margins? Br J Cancer. 2006;94(3):351-357.
33. Rutkowski A, Bujko K, Nowacki MP, et al. Distal bowel surgical margin shorter than 1 cm after preoperative radiation for rectal cancer: is it safe? Ann Surg Oncol. 2008;15(11):3124-3131. 81.
34. Fitzgerald TL, Brinkley J, Zervos EE. Pushing the envelope beyond a centimeter in rectal cancer: oncologic implications of close, but negative margins. J Am Coll Surg. 2011;213(5):589-595.
35. Chen Z, Sasaki K, Murono K, et al. Oncologic status of obturator lymph node metastases in locally advanced low rectal cancer: a Japanese multi-institutional study of 3487 patients. Ann Surg Oncol. 2022;29:4210-4219.
36. Quirke P, Steele R, Monson J, et al. Effect of the plane of surgery achieved on local recurrence in patients with operable rectal cancer: a prospective study using data from the MRC CR07 and NCIC-CTG CO16 randomised clinical trial. Lancet. 2009;373(9666):821-828.
38. Elisa Cassinotti, Giorgio Palazzini, Massimiliano Della Porta, Ilaria Grosso, Luigi Bon, Transanal total mesorectal excision (TaTME): tips and tricks of a new surgical technique.,Ann Laparosc Endosc Surg 2017;2:1-8.
39. Dajana Cuicchi, Giovanni Castagna, Stefano Cardelli, Cristina Larotonda, Benedetta Petrello, Gilberto Poggioli,Restaging rectal cancer following neoadjuvant chemoradiotherapy,
World J Gastrointest Oncol. 2023 May 15;15(5):700–712. doi: 10.4251/wjgo.v15.i5.700
40. J. Wyatt y, S.G. Powell y, S. Ahmed et al., Watch and Wait in Rectal Cancer After a Complete Response to Chemoradiotherapy Is It Safe and Are We Doing Enough?,Clinical Oncology 35 (2023) 117e123.
41. Shiru L Liu, Winson Y Cheung, Role of surveillance imaging and endoscopy in colorectal cancer follow-up: Quality over quantity?, World J Gastroenterol 2019 January 7; 25(1): 59-68.
42. 7. Petersen SH, Harling H, Kirkeby LT, et al. Postoperative adjuvant chemotherapy in rectal cancer operated for cure. Cochrane Database Syst Rev. 2012;2012(3):Cd004078. 98.
43. Bosset JF, Calais G, Mineur L, et al. Fluorouracil-based adjuvant chemotherapy after preoperative chemoradiotherapy in rectal cancer: long-term results of the EORTC 22921 randomised study. Lancet Oncol. 2014;15(2):184-190.
44. Jin‑hu Chen, Qing Ye and Feng Huang, Determining the survival benefit of postoperative radiotherapy in patients with pT1-3N1M0 rectal cancer undergoing total mesorectal excision: a retrospective analysis,BMC Gastroenterology (2023) 23:83.
45. Margaret Lee, Peter Gibbs, Rachel Wong, Multidisciplinary Management of Locally Advanced Rectal Cancer—An Evolving Landscape?, Clinical Colorectal Cancer14,4, 2015: 251-261.
- Annexes
Annex 1.
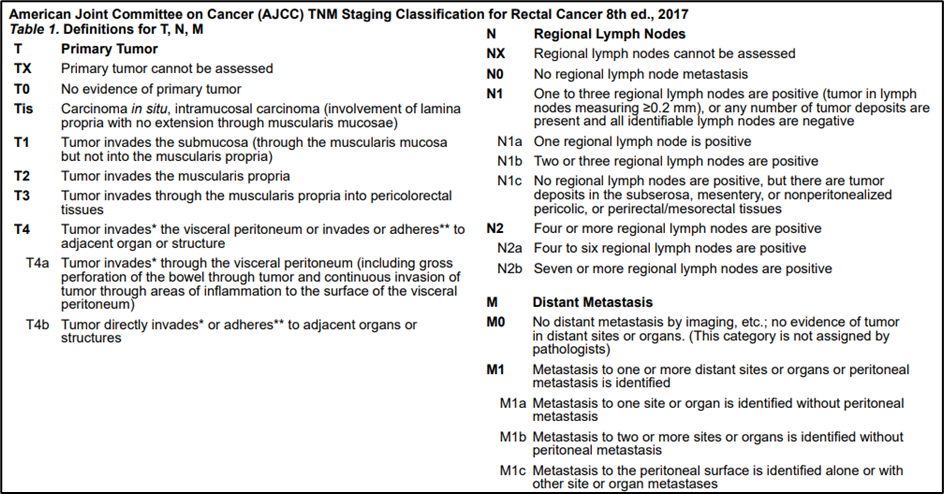
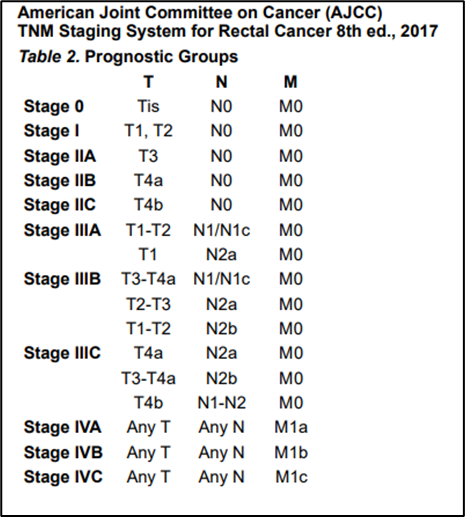
Annex 2.
REGIMENS (PERIOPERATIVE THERAPY)
Perioperative Chemotherapy:
• mFOLFOX 6
Oxaliplatin 85 mg/m2 IV, day 1, leucovorin 400 mg/m2 IV day 1, 5-FU 400 mg/m2
IV bolus on day 1, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over
46–48 hours) continuous infusion. Repeat every 2 weeks to a total of 6 months perioperative therapy.
• CAPEOX
Oxaliplatin 130 mg/m2 IV day 1. Capecitabine 1000 mg/m2 PO twice daily
for 14 days every 3 weeks. Repeat every 3 weeks to a total of 6 months perioperative therapy.
• FOLFIRINOX
Oxaliplatin 85 mg/m² IV on day 1, leucovorin 400 mg/m² IV over 2 hours on
day 1, irinotecan 180 mg/m² IV over 30–90 minutes on day 1, 5-FU 400 mg/m²
IV push day 1, 5-FU 1200 mg/m²/day x 2 days (total 2400 mg/m² over 46 hours)
continuous infusion. Repeat every 2 weeks.
• Modified FOLFIRINOX
Oxaliplatin 85 mg/m² IV on day 1, leucovorin 400 mg/m² IV over 2 hours on day
1, irinotecan 150 mg/m² IV over 30–90 minutes on day 1, 5-FU 1200 mg/m²/day
x 2 days (total 2400 mg/m² over 46 hours) continuous infusion. Repeat every 2
weeks.
Dosing Schedules for Concurrent Chemotherapy/RT:
• RT + continuous infusion 5-FU
5-FU 225 mg/m2 IV over 24 hours daily on days 1–5 or days 1–7 for 5 weeks with RT
• RT + capecitabine
Capecitabine 825 mg/m2 PO BID, Monday–Friday, on days of radiation
treatment only, throughout the duration of RT (typically 28–30 treatment
days)
• RT + 5-FU/leucovorin
5-FU 400 mg/m2 IV bolus + leucovorin 20 mg/m2 IV bolus for 4 days during weeks 1 and 5 of RT
SYSTEMIC THERAPY FOR ADVANCED OR METASTATIC DISEASE
➡️ mFOLFOX 6
Oxaliplatin 85 mg/m2 IV day 1z Leucovorin 400 mg/m2 IV day 1aa
5-FU 400 mg/m2 IV bolus on day 1, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) IV continuous infusion
Repeat every 2 weeks
➡️ mFOLFOX 7
Oxaliplatin 85 mg/m2 IV day 1z Leucovorin 400 mg/m2 IV day 1aa
5-FU 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) IV continuous infusion
Repeat every 2 weeks
➡️ FOLFOX + bevacizumab
Bevacizumab mg/kg IV, day 1 Repeat every 2 weeks
➡️ FOLFOX + panitumumab (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes, day 1 ---Repeat every 2 weeks
➡️ FOLFOX + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 IV over 2 hours first infusion, followed by 250 mg/m2 IV over 60 minutes weekly or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks (preferred for every 2 weeks)
➡️ CAPEOX8
Oxaliplatin 130 mg/m2 IV day 1z
Capecitabine 1000cc mg/m2 twice daily PO for 14 days
Repeat every 3 weeks
➡️ CAPEOX + bevacizumab
Oxaliplatin 130 mg/m2 IV day 1z
Capecitabine 1000cc mg/m2 PO twice daily for 14 days
Bevacizumab 7.5 mg/kg IV day 1 Repeat every 3 weeks
➡️CAPEOX + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 IV over 2 hours first infusion, followed by 250 mg/m2 IV over 60 minutes weekly or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks (preferred for every 2 weeks)
➡️CAPEOX + panitumumab9-11 (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes, day 1 Repeat every 2 weeks
➡️FOLFIRI
Irinotecan 180 mg/m2 IV over 30–90 minutes, day 1
Leucovorinaa 400 mg/m2 IV infusion to match duration of irinotecan infusion,
day 1
5-FU 400 mg/m2 IV bolus day 1, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion
Repeat every 2 weeks
➡️ FOLFIRI + bevacizumab
Bevacizumab 5 mg/kg IV, day 1 Repeat every 2 weeks
➡️ FOLFIRI + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 IV over 2 hours first infusion, followed by 250 mg/m2 IV over 60 minutes weekly or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks (preferred
for every 2 weeks)
➡️ FOLFIRI + panitumumab (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes, day 1 Repeat every 2 weeks
➡️ FOLFIRINOX20
Oxaliplatin 85 mg/m² IV on day 1,z leucovorin 400 mg/m² IV over 2 hours on day 1, irinotecan 165–180 mg/m² IV over 30–90 minutes on day 1, 5-FU 400 mg/m² IV push day 1, 5-FU 1200 mg/m²/day x 2 days (total 2400 mg/m² over 46 hours) continuous infusion.
Repeat every 2 weeks
➡️ Modified FOLFIRINOX
Oxaliplatin 85 mg/m² IV on day 1,z leucovorin 400 mg/m² IV over 2 hours on day 1, irinotecan 150 mg/m² IV over 30–90 minutes on day 1, 5-FU 1200 mg/m²/day x 2 days (total 2400 mg/m² over 46 hours) continuous infusion. Repeat every 2 weeks
➡️ FOLFIRINOX or mFOLFIRINOX + bevacizumab
Bevacizumab 5 mg/kg IV, day 1
Repeat every 2 weeks
➡️ IROX
Oxaliplatin 85 mg/m2 IV
followed by irinotecan 200 mg/m2 over 30–90 minutes every 3 weeks
➡️ IROX + bevacizumab
Bevacizumab 7.5 mg/kg IV on day 1 Repeat every 3 weeks
➡️ Bolus or infusional 5-FU/leucovorin Roswell Park regimen
Leucovorin 500 mg/m2 IV over 2 hours, days 1, 8, 15, 22, 29, and 36
5-FU 500 mg/m2 IV bolus 1 hour after start of leucovorin,
days 1, 8, 15, 22, 29, and 36
Repeat every 8 weeks
➡️ Simplified biweekly infusional 5-FU/leucovorin (sLV5FU2)
Leucovorinaa 400 mg/m2 IV over 2 hours on day 1,
followed by 5-FU bolus 400 mg/m2 followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion Repeat every 2 weeks
Weekly Leucovorin 20 mg/m2 IV over 2 hours on day 1, 5-FU 500 mg/ m2 IV bolus injection 1 hour after the start of leucovorin. Repeat weekly27
or
5-FU 2600 mg/m2 by 24-hour infusion plus leucovorin 500 mg/m2 Repeat every week
➡️ Bolus or infusional 5-FU + bevacizumab
Bevacizumab 5 mg/kg IV on day 1
Repeat every 2 weeks
➡️ Capecitabine
Capecitabine 850–1250 mg/m2 PO twice daily for 14 days Repeat every 3 weeks
➡️ Capecitabine + bevacizumab
Bevacizumab 7.5 mg/kg IV, day 1 Repeat every 3 weeks
➡️ Irinotecan
Irinotecan 125 mg/m2 IV over 30–90 minutes, days 1 and 8 Repeat every 3 weeks
or Irinotecan 180 mg/m2 IV over 30–90 minutes, day 1 Repeat every 2 weeks
or Irinotecan 300–350 mg/m2 IV over 30–90 minutes, day 1 Repeat every 3 weeks
➡️ Irinotecan + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 first infusion, followed by 250 mg/m2 IV weekly32
or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks16
(preferred for every 2 weeks)
➡️ Irinotecan + panitumumab17,33 (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes every 2 weeks
➡️ Irinotecan + bevacizumab34,bb Irinotecan 180 mg/m2 IV, day 1
Bevacizumab 5 mg/kg IV, day 1 Repeat every 2 weeks
or
Irinotecan 300–350 mg/m2 IV, day 1
Bevacizumab 7.5 mg/kg IV, day 1 Repeat every 3 weeks
Cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 first infusion, followed by 250 mg/m2 IV weekly32
or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks16
(preferred for every 2 weeks)
Panitumumab35 (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes every 2 weeks
➡️ Trastuzumab + pertuzumab
(HER2-amplified and RAS and BRAF WT)
Trastuzumab 8 mg/kg IV loading dose on day 1 of cycle 1, followed by 6 mg/kg IV every 21 days
Pertuzumab 840 mg IV loading dose on day 1 of cycle 1, followed by 420 mg IV every 21 days
➡️ Trastuzumab + lapatinib
(HER2-amplified and RAS and BRAF WT)
Trastuzumab 4 mg/kg IV loading dose on day 1 of cycle 1, followed by 2 mg/kg IV weekly
Lapatinib 1000 mg PO daily
Annex 3. PRODIGE 23 TRIAL PROTOCOL
(Indications and doses)
Eligible patients:
- adults aged 18–75 years,
- newly diagnosed,
- biopsy-proven,
- staged cT3 or cT4 M0
- WHO performance status of 0–1.
Doses, and procedures
- oxaliplatin 85 mg/m2, irinotecan 180 mg/m2, leucovorin 400 mg/m2, and fluorouracil 2400 mg/m2 intravenously every 14 days for 6 cycles),
- chemoradiotherapy (50 Gy during 5 weeks and 800 mg/m2 concurrent oral capecitabine twice daily for 5 days per week),
- total mesorectal excision,
- adjuvant chemotherapy (3 months of modified FOLFOX6 [intravenous oxaliplatin 85 mg/m2 and leucovorin 400 mg/m2, followed by intravenous 400 mg/m2 fluorouracil bolus and then continuous infusion at a dose of 2400 mg/m2 over 46 h every 14 days for six cycles] or capecitabine [1250 mg/m2 orally twice daily on days 1–14 every 21 days]).