Management of Rheumatoid Arthritis
| Site: | EHC | Egyptian Health Council |
| Course: | Rheumatology and clinical immunology Guidelines |
| Book: | Management of Rheumatoid Arthritis |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update:
28 Oct 2025" Download Guideline
- Executive Summary
This guideline offers evidence-based recommendations on the diagnosis and management of Rheumatoid Arthritis (RA). The recommendations are intended to provide healthcare professionals with practical guidance on diagnosis and management of Rheumatoid Arthritis, and improving health outcomes for people living with RA.
➡️Clinical Evaluation:
1. It is advised to suspect that the patient is a case of Rheumatoid Arthritis if he has morning stiffness lasting >1 hour, improving with activity, and small-joint arthritis affecting the hands and feet (Good Practice Statement).
2. You should diagnose a case of RA if the examination of the extremities reveals the presence of synovitis (soft tissue joint swelling), which will typically present as a symmetric polyarthritis affecting the metacarpophalangeal (MCP), metatarsophalangeal (MTP), and proximal interphalangeal (PIP) joints (Good Practice Statement).
➡️ Laboratory Tests:
3. It is advised to do:
· Rheumatoid factor (RF): Present in 70–80% of RA cases (but not specific; seen in other autoimmune diseases and even in healthy elderly persons).
· Anti-Cyclic Citrullinated Peptide (Anti-CCP) Antibodies: highly specific for RA (90–95%), aiding early diagnosis.
· Inflammatory markers: elevated ESR (erythrocyte sedimentation rate) and CRP (C-reactive protein) during activity (Good Practice Statement).
Imaging Studies:
4. Do plain radiographs hands & feet that might demonstrate periarticular osteopenia, joint space narrowing and bone erosions, and their absence does not rule out RA diagnosis (Good Practice Statement).
5. Ultrasonography: may be of help if done by expert person in selected cases & should not be the only tool to rely on for diagnosis and treatment strategies (Good Practice Statement).
6. You can use the ACR/EULAR 2010 classification criteria, to aid the diagnosis (Good Practice Statement).
➡️ Treatment of rheumatoid arthritis:
7. We recommend starting treatment as soon as the diagnosis of RA is made. (SOR: Strong).
8. We recommend applying “Treat-to-target strategy”: treatment should be aimed at reaching a target of sustained remission or low disease activity in every patient, according to ACR/ EULAR provided Boolean- based and index- based remission criteria. (SOR: Strong).
9. We recommend frequent monitoring of the disease activity every 1–3 months during active disease and then every 3–6 months once remission/ low disease activity achieved, using a validated measure. We advise the use of CDAI (Clinical Disease Activity Index) as a tool to measure the activity state (Refer to annexes). Remission ≤ 2.8, low disease activity >2.8-10, moderate disease activity >10-22, high disease activity >22-76. (SOR: Strong).
10. Therapy should be adjusted if there is no improvement by 50% from the baseline measures by 3 months after the start of treatment or the target has not been reached by 6 months (SOR: Strong).
11. MTX should be part of the first treatment strategy. (SOR: Strong).
12. We strongly recommend starting the first line of treatment strategy with conventional disease-modifying antirheumatic drug (csDMARD) monotherapy using methotrexate (15–25 mg/week) s.c. or i.m. injections or oral tablets. Folic acid supplementation 5-10 mg /week should be given 24hrs after MTX administration (in a single or divided doses), not to be taken the day of MTX. (SOR: Strong).
13. We suggest implementing strategies to improve MTX tolerability in patients experiencing side effects, before switching to another DMARD. These include increasing folic acid dose, splitting oral MTX dose over 24 h, or switching between oral and parenteral routes of MTX administration. (SOR: Conditional).
14. We advise treatment with Leflunomide (20 mg/day) or sulfasalazine (3 g/day), when MTX is not tolerated or contraindicated. Consider hydroxychloroquine (200–400 mg) for mild or palindromic disease without poor prognostic factors. (SOR: Conditional).
15. We suggest adding other csDMARD to MTX or using a different combination csDMARD therapy, if no significant improvement at least 50% from the baseline measures has been achieved by 3 months (DMARD combination therapy means double or triple csDMARDs therapy). (SOR: Conditional).
16. You should consider short-term glucocorticoids when initiating or changing csDMARDs (as a bridge therapy); the dose and route of administration may vary, orally at doses up to 5-10 mg/day, or a single intramuscular injection of 120 mg methylprednisolone. Oral steroid therapy should be tapered as rapidly as clinically feasible, within 3-6 months from treatment start provided that the patient doesn't flare. After 6 months, if the patient is still active & doesn't have access to bDMARDs or tsDMARDs, longer term steroids can be used up to 5 mg per day. (SOR: Strong).
17. We advise starting bDMARDs or tsDMARDs after failure of 2 or more csDMARDs (when there is no significant improvement by at least 50% from the baseline measures by 3 months or treatment target is not achieved within 6 months). (SOR: Conditional).
18. The choice of a bDMARD (bio-originator or biosimilar) depends on the availability of the drug, drug cost, associated co-morbidities and patient preference. (SOR: Strong).
19. When tsDMARDs (JAKi) are considered, risk factors must be taken into account:
· Age > 65 years.
· History of current or past smoking.
· Cardiovascular risk factors: such as diabetes, obesity, and hypertension.
· Risk factors for malignancy.
· Current or previous history of malignancy.
· Risk factors for thromboembolic events: e.g. history of myocardial infarction or heart failure, cancer, inherited blood clotting disorders or a history of blood clots, hormonal contraceptives or hormone replacement therapy, undergoing major surgery or prolonged immobility. (SOR: Strong).
20. If a bDMARD or tsDMARD has failed, treatment with another bDMARD or tsDMARD should be considered. Before switching, reassess adherence, comorbidities, diagnosis, and objective inflammation. Distinguish between:
· Primary treatment failure:
–No clinically relevant improvement ( at least 50% from baseline measures) or treatment target is not achieved within 6 months of adequate therapy.
– →In this case, switch to a drug with a different mechanism of action (e.g., from a TNFi to a non-TNFi such as an IL-6 receptor inhibitor, rituximab, or a JAK inhibitor).
· Secondary treatment failure:
– Loss of efficacy after an initial adequate response.
– Suggests immunogenic or pharmacokinetic failure → switching within the same class can be considered (e.g. from one TNFi to another), especially if immunogenicity or anti-drug antibodies are suspected.
– If the response is not regained, switch to a drug with a different mechanism of action.
Patients who fail multiple b/tsDMARDs despite adequate therapy should be classified as having difficult-to-treat RA, in line with EULAR criteria (SOR: Strong).
21. bDMARDs and tsDMARDs could be combined with a csDMARD usually MTX; in patients who cannot use csDMARDs as comedication, IL- 6i and tsDMARDs may have some advantages compared with other bDMARDs. (SOR: Strong).
22. You should give a trial of tapering the bDMARDs or tsDMARDs after GCs have been discontinued, if the patient has sustained disease remission (i.e. remission for ≥ 6 consecutive months) either by decreasing the dose or increasing the time intervals whenever applicable according to the drug used with continuation of other csDMARDs. (SOR: Strong).
Recommendations in RA patients with comorbidities:
23. In patients who develop double-fold elevation of liver enzymes, reduce MTX & LEF to half dose. If reaches threefold or more, stop MTX and LEF, if persistent over 6 months re-evaluate the virology. (SOR: Strong).
24. In patients with non-alcoholic fatty liver disease (NAFLD), Methotrexate is conditionally recommended over alternative DMARDs for DMARD-naive patients with NAFLD, normal liver enzymes and liver function tests, and no evidence of advanced liver fibrosis who have moderate-to-high disease activity. (SOR: Conditional).
25. Patients with hepatitis B viral infection should receive antiviral treatment before starting bDMARDs with close monitoring as well as follow up with the hepatologists after starting biological therapy. (SOR: Strong).
26. In hypertensive patients, be careful regarding salt and water retention property of Leflunomide. Baseline measurement of blood pressure is recommended with adjustment of blood pressure therapy if required. (SOR: Conditional).
27. In patients with New York Heart Association class 3 or 4 heart failure, non-TNFi bDMARD are recommended. (SOR: Strong).
28. Patients complicated with interstitial lung disease: Interleukin-6 inhibitor (IL-6i) or Rituximab could be considered. (SOR: Conditional).
29. In patients with history of lymphoproliferative disorder, rituximab is preferable over other bDMARDs (SOR: Conditional).
30. MTX, Leflunomide, Rituximab, Tocilizumab and JAKi are contraindicated in pregnancy & lactation. (SOR: Strong).
31. All TNFi, hydroxychloroquine, corticosteroids, and sulfasalazine can be used during pregnancy and lactation. (SOR: Conditional).
- Recommendation
Clinical Evaluation:
1. It is advised to suspect that the patient is a case of Rheumatoid Arthritis if he has morning Stiffness lasting >1 hour, improving with activity, and small-joint arthritis affecting the hands and feet (Good Practice Statement). [3]
2. You should diagnose a case of RA if the examination of the extremities reveals the presence of synovitis (soft tissue joint swelling), which will typically present as a symmetric polyarthritis affecting the metacarpophalangeal (MCP), metatarsophalangeal (MTP), and proximal interphalangeal (PIP) joints (Good Practice Statement). [4]
Laboratory Tests:
3. It is advised to do:
· Rheumatoid factor (RF): Present in 70–80% of RA cases (but not specific; seen in other autoimmune diseases and even in healthy elderly persons).
· Anti-Cyclic Citrullinated Peptide (Anti-CCP) Antibodies: highly specific for RA (90–95%), aiding early diagnosis.
· Inflammatory markers: elevated ESR (erythrocyte sedimentation rate) and CRP (C-reactive protein) during activity (Good Practice Statement). [4-6]
Imaging Studies:
4. Do plain radiographs hands & feet that demonstrate periarticular osteopenia, joint space narrowing and bone erosions, and their absence does not rule out RA diagnosis (Good Practice Statement).
5. Ultrasonography: may be of help if done by expert person in selected cases & should not be the only tool to rely on for diagnosis and treatment strategies (Good Practice Statement).
6. You can use the ACR/EULAR 2010 classification criteria, to aid the diagnosis (Good Practice Statement). [4, 7, 8]
Treatment of rheumatoid arthritis:
7. We recommend starting treatment as soon as the diagnosis of RA is made. (SOR: Strong; LOE: High). [9]
8. We recommend applying “Treat-to-target strategy”: treatment should be aimed at reaching a target of sustained remission or low disease activity in every patient, according to ACR/ EULAR provided Boolean- based and index- based remission criteria. (SOR: Strong; LOE: High). [10]
9. We recommend frequent monitoring of the disease activity every 1–3 months during active disease and then every 3–6 months once remission/ low disease activity achieved, using a validated measure. We advise the use of CDAI (Clinical Disease Activity Index) as a tool to measure the activity state (Refer to annexes). Remission ≤ 2.8, low disease activity >2.8-10, moderate disease activity >10-22, high disease activity >22-76. (SOR: Strong; LOE: Moderate). [11]
10. Therapy should be adjusted if there is no improvement by 50% from the baseline measures by 3 months after the start of treatment or the target has not been reached by 6 months (SOR: Strong; LOE: Moderate). [12].
11. MTX should be part of the first treatment strategy. (SOR: Strong; LOE: High). [13]
12. We strongly recommend starting the first line of treatment strategy with conventional disease-modifying antirheumatic drug (cDMARD) monotherapy using methotrexate (15–25 mg/week) s.c. or i.m. injections or oral tablets. Folic acid supplementation 5-10 mg /week should be given 24hrs after MTX administration (in a single or divided doses), not to be taken the day of MTX. (SOR: Strong; LOE: High). [14].
13. We suggest implementing strategies to improve MTX tolerability in patients experiencing side effects, before switching to another DMARD. These include increasing folic acid dose, splitting oral MTX dose over 24 h, or switching between oral and parenteral routes of MTX administration. (SOR: Conditional; LOE: very low). [15]
14. We advise treatment with Leflunomide (20 mg/day) or sulfasalazine (3 g/day), when MTX is not tolerated or contraindicated. Consider hydroxychloroquine (200–400 mg) for mild or palindromic disease without poor prognostic factors. (SOR: Conditional; LOE: Moderate). [16, 17]
15. We suggest adding other csDMARD to MTX or using a different combination csDMARD therapy, if no significant improvement at least 50% from the baseline measures has been achieved by 3 months (DMARD combination therapy means double or triple csDMARDs therapy). (SOR: Conditional; LOE: very Low). [18]
16. You should consider short-term glucocorticoids when initiating or changing csDMARDs (as a bridge therapy); the dose and route of administration may vary, orally at doses up to 5-10 mg/day, or a single intramuscular injection of 120 mg methylprednisolone. Oral steroid therapy should be tapered as rapidly as clinically feasible, within 3-6 months from treatment start provided that the patient doesn't flare. After 6 months, if the patient is still active & doesn't have access to bDMARDs or tsDMARDs, longer term steroids can be used up to 5 mg per day. (SOR: Strong; LOE: High). [19, 20]
17. We advise starting bDMARDs or tsDMARDs after failure of 2 or more csDMARDs (when there is no significant improvement by at least 50% from the baseline measures by 3 months or treatment target is not achieved within 6 months). (SOR: Conditional; LOE: very low). [8]
18. The choice of a bDMARD (bio-originator or biosimilar) depends on the availability of the drug, drug cost, associated co-morbidities and patient preference. (SOR: Strong; LOE: very Low). [21]
19. When tsDMARDs (JAKi) are considered, risk factors must be taken into account:
· Age > 65 years.
· History of current or past smoking.
· Cardiovascular risk factors: such as diabetes, obesity, and hypertension.
· Risk factors for malignancy.
· Current or previous history of malignancy.
· Risk factors for thromboembolic events: e.g. history of myocardial infarction or heart failure, cancer, inherited blood clotting disorders or a history of blood clots, hormonal contraceptives or hormone replacement therapy, undergoing major surgery or prolonged immobility. (SOR: Strong; LOE: Moderate). [22]
20. If a bDMARD or tsDMARD has failed, treatment with another bDMARD or tsDMARD should be considered. Before switching, reassess adherence, comorbidities, diagnosis, and objective inflammation. Distinguish between:
· Primary treatment failure:
–No
clinically relevant improvement (at
least 50% from the baseline measures) or treatment target is not achieved
within 6 months of adequate therapy.
– →In this case, switch to a drug with a different
mechanism of action (e.g., from a TNFi to a non-TNFi such as an IL-6 receptor inhibitor, rituximab,
or a JAK inhibitor).
· Secondary treatment failure:
– Loss of efficacy after an initial adequate response.
– Suggests immunogenic or pharmacokinetic failure
→ switching within the same class can be
considered (e.g. from one TNFi to another), especially if immunogenicity or
anti-drug antibodies are suspected.
– If the response is not regained, switch to a drug with a different
mechanism of action.
Patients who fail multiple b/tsDMARDs despite adequate therapy should be classified as having difficult-to-treat RA, in line with EULAR criteria
(SOR: Strong; LOE: Moderate). [23,35,36]
21. bDMARDs and tsDMARDs could be combined with a csDMARD usually MTX; in patients who cannot use csDMARDs as comedication, IL- 6i and tsDMARDs may have some advantages compared with other bDMARDs. (SOR: Strong; LOE: High). [24-26]
22.You should give a trial of tapering the bDMARDs or tsDMARDs after GCs have been discontinued, if the patient has sustained disease remission (i.e. remission for ≥ 6 consecutive months) either by decreasing the dose or increasing the time intervals whenever applicable according to the drug used with continuation of other csDMARDs. (SOR: Strong; LOE: High). [27]
Recommendations in RA patients with comorbidities:
23. In patients who develop double-fold elevation of liver enzymes, reduce MTX & LEF to half dose. If reaches threefold or more, stop MTX and LEF, if persistent over 6 months re-evaluate the virology. (SOR: Strong; LOE: Moderate). [28].
24. In patients with non-alcoholic fatty liver disease (NAFLD), Methotrexate is conditionally recommended over alternative DMARDs for DMARD-naive patients with NAFLD, normal liver enzymes and liver function tests, and no evidence of advanced liver fibrosis who have moderate-to-high disease activity. (SOR: Conditional; LOE: very Low). [29]
25. Patients with hepatitis B viral infection should receive antiviral treatment before starting bDMARDs with close monitoring as well as follow up with the hepatologists after starting biological therapy. (SOR: Strong; LOE: Low). [30]
26. In hypertensive patients, be careful regarding salt and water retention property of Leflunomide. Baseline measurement of blood pressure is recommended with adjustment of blood pressure therapy if required. (SOR: Conditional; LOE: very Low). [31]
27. In patients with New York Heart Association class 3 or 4 heart failure, non-TNFi bDMARD are recommended. (SOR: Strong; LOE: Low). [32]
28. Patients complicated with interstitial lung disease: Interleukin-6 inhibitor (IL-6i) or Rituximab could be considered. (SOR: Conditional; LOE: Low). [33]
29. In patients with history of lymphoproliferative disorder, rituximab is preferable over other bDMARDs (SOR: Conditional; LOE: very Low). [15]
30. MTX, Leflunomide, Rituximab, Tocilizumab and JAKi are contraindicated in pregnancy & lactation. (SOR: Strong; LOE: High). [34]
31. All TNFi, hydroxychloroquine, corticosteroids, and sulfasalazine can be used during pregnancy and lactation. (SOR: Conditional; LOE: Moderate). [34]
- Acknowledgement
We would like to acknowledge the “Rheumatology and Clinical Immunology Scientific Committee” for developing these guidelines.
Chair of the Guideline Development Group (GDG):
Samir Elbadawy, Professor of Rheumatology & Rehabilitation, Cairo University.
Members of the GDG:
1. Samir Ahmed Elbadawy, Professor of Rheumatology, Faculty of Medicine, Cairo University.
2. Mohamed Salah Eldin Abdel Baky Salama, Professor of Internal Medicine and Rheumatology, Ain Shams University.
3. Ali Yousef, Professor and Head of departments Medicine teaching hospitals and institutes.
4. Khaled Tahsin Elhadidi, Professor of Rheumatology, Faculty of Medicine, Cairo University.
5. Amal Mostafa Elganzoury, Professor of Rheumatology and Rehabilitation, Ain Shams University.
6. Mohamed Mahmoud Elwakd, Professor of Rheumatology, Faculty of Medicine, Cairo University.
7. Nevine Mohannad Abbas, Consultant Internal Medicine and Rheumatology, Alexandria University Hospitals, Faculty of Medicine, Alexandria University.
8. Arwa Osama Al-Shaarawy, Lecturer of Rheumatology, Cairo University.
9. Hala Ibrahim Mostafa El Seidy, Lecturer of Rheumatology, Cairo University.
- Abbreviations
ACCP: Anti-cyclic citrullinated peptide.
ACPA: Anti-citrullinated peptide antibody.
ACR: American College of Rheumatology.
bDMARDs: Biologic disease modifying antirheumatic drugs.
bsDMARDs: Biosimilar disease modifying antirheumatic drugs.
CDAI: Clinical disease activity index.
CRP: C-reactive protein.
csDMARDs: Conventional synthetic disease modifying antirheumatic drugs.
DMARDs: Disease modifying antirheumatic drugs.
ESR: erythrocyte sedimentation rate.
EULAR: European League Against Rheumatism.
GCs: Glucocorticoids.
GRADE: Grading of recommendations assessment, development and evaluation.
HbA1c: Glycated hemoglobin A1C.
HCP: Healthcare professionals.
LEF: Leflunomide.
LoA: Level of agreement.
LoE: Level of evidence.
MTX: Methotrexate.
NICE: National Institute for Health and Care Excellence.
RA: Rheumatoid Arthritis.
RCT: Randomized controlled trials.
RF: Rheumatoid factor.
SoR: Strength of recommendation.
tsDMARDs: Targeted synthetic disease modifying antirheumatic drugs.
- Glossary and definitions
➡️Poor prognostic factors
· Persistently moderate or high disease activity (after csDMARDs therapy) according to composite measures including joint counts despite csDMARDs therapy.
· High acute phase reactant levels.
· High swollen joint count.
· Presence of rheumatoid factor (RF) and/or anti-citrullinated peptide antibody (ACPA), especially at high levels.
· Presence of early erosions.
· Failure of 2 or more csDMARDs.
➡️Low dose glucocorticoids
· <7.5 mg/day prednisone equivalent.
➡️Short-term
· Up to 3 months.
➡️Tapering
· Reduction of drug dose or increase of the interval between doses.
· May include cessation (tapering to zero), but then only after slow reduction.
➡️Discontinuation, cessation
· Stopping a particular drug.
➡️Disease activity states
· Remission
ACR-EULAR remission definition (Boolean or index-based); sustained remission: ACR-EULAR-defined remission for ≥6 months
· Low disease activity
Low disease activity state according to validated composite disease activity measures that include joint counts, performed by a healthcare professionals (HCP); sustained low disease activity: low disease activity for ≥6 months
· Moderate, high disease activity
Respective disease activity state according to validated composite disease activity measures that include joint counts by a HCP.
➡️DMARD nomenclature
· Conventional synthetic DMARDs: for example, Methotrexate, Leflunomide, Sulfasalazine, Hydroxychloroquine.
· Targeted synthetic DMARDs: for example, Baricitinib, Filgotinib, Tofacitinib, Upadacitinib.
· Biological DMARDs: Biological originator DMARDs including
- TNFi: Adalimumab, Certolizumab, Etanercept, Golimumab, Infliximab.
- IL-6Ri: Sarilumab, Tocilizumab.
- Co-stimulation-i: Abatacept.
- Anti-B-cell (CD20): Rituximab.
· Biosimilar DMARDs: currently for Adalimumab, Etanercept, Infliximab, and Rituximab.
- Introduction
- Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by persistent inflammatory polyarthritis, which can lead to joint destruction and systemic complications. [1]
- Early diagnosis and treatment with disease-modifying antirheumatic drugs (DMARDs) are important to prevent joint damage, disability and systemic complications (e.g., cardiovascular disease). [2]
- Scope and Purpose
· In 2020, the Egyptian health authorities have launched a nationwide Universal Health Insurance System, aiming to ensure that all Egyptians have comprehensive health care for all family members. Setting up guidelines for management plays a vital role in the patients’ management process.
· These guidelines adapt international recommendations to Egypt’s healthcare realities, particularly cost/resource constraints and variable specialist availability.
- Target audience
· The guideline will be of particular interest to health care professionals who treat and manage patients with RA, mainly the rheumatologists.
· The guidelines should also provide a helpful resource for general practitioners, and those in the National Health Service who are in charge of commissioning the care for RA patients.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
· Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence).
· Selecting only the evidence-based guidelines.
· Specific range of dates for publication (using guidelines published or updated in 2018 and later).
· Selecting peer reviewed publications only.
· Selecting guidelines written in English language.
· Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
· Excluding guidelines published without references, the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
The following characteristics of the retrieved guidelines were summarized in:
· Developing organization/authors.
· Date of publication, posting, and release.
· Country/language of publication.
· Date of posting and/or release.
· Dates of the search used by the source guideline developers.
All retrieved guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt: EULAR recommendations for the management of RA with synthetic and biological disease-modifying antirheumatic drugs: 2022 update [23], the 2021 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis [15] & 2018 NICE Guideline for management of Rheumatoid Arthritis [8].
- Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
· GRADE working group:https://www.gradeworkinggroup.org
· GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1: Quality and Significance of the four levels of evidence in GRADE:

Table 2 Factors that determine How to upgrade or downgrade the quality of evidence
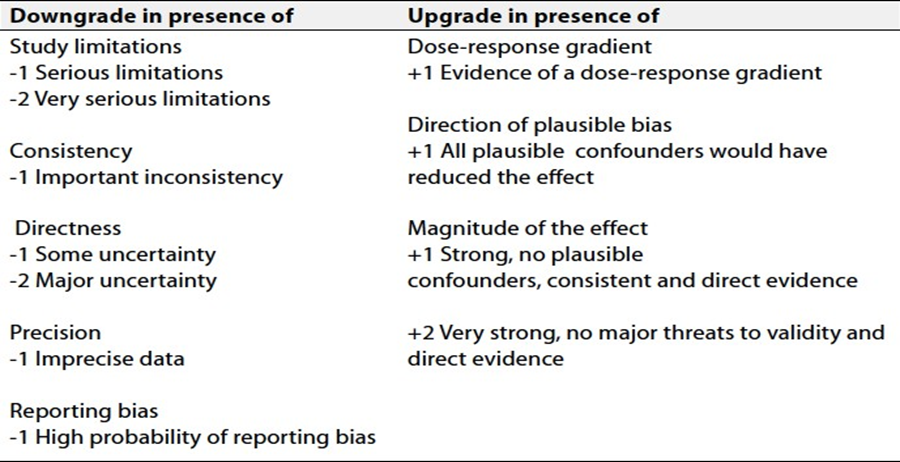
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations:
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations:
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
Good Practice Statements:
Statements based on the opinions of respected authorities and the guideline development group.
- Research needs
1. Is the risk of glucocorticoids (GCs) different if a specific cumulative dose has been used within a relatively short period of time, such as up to 3 or 6 months, or chronically over a number of years?
2. Does the concomitant use of GCs at very low doses (1–3 mg prednisone equivalent) increase therapeutic success without producing unacceptable side effects?
3. What are the effectiveness and safety profiles of (repeated) intramuscular glucocorticoids, for example, methylprednisolone 120 mg or triamcinolone 80 mg 1–4 times yearly?
4. How safe and efficacious is the use of a JAKi after another JAKi has failed?
5. Can we identify new biomarkers to stratify patients and to predict therapeutic responses or lack of response?
6. How good is patient adherence to a bDMARD or tsDMARD and can non-adherence explain secondary loss of efficacy?
7. Is Leflunomide equivalent to MTX as first line csDMARD therapy?
8. Does the risk stratification for bDMARD/tsDMARD initiation based on presence of good or bad prognostic factors as recommended by EULAR translate into improved outcomes for both prognosis groups?
9. What is the optimal treatment approach to refractory RA?
10. What is the optimal (therapeutic) approach to arthralgia suspicious for progression to RA?
- Monitoring and evaluating the impact of the guidelines
· RF measurement.
· ACCP measurement.
· X-ray hands.
· Assessment of effectiveness of Treatment: using CDAI Score.
- Updating of the guidelines
These guidelines will be updated whenever there is new evidence.
Adapted from: EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update [23], EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update [27], the 2021 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis [15] & 2018 NICE Guideline for management of Rheumatoid Arthritis [8].
- References
[1] Smolen JS, Aletaha D, McInnes IB. Rheumatoid arthritis. Lancet. 2016 Oct 22;388(10055):2023-2038. doi: 10.1016/S0140-6736(16)30173-8. Epub 2016 May 3. Erratum in: Lancet. 2016 Oct 22;388(10055):1984. doi: 10.1016/S0140-6736(16)30794-2. PMID: 27156434.
[2] van der Linden MP, le Cessie S, Raza K, et al. Long-term impact of delay in assessment of patients with early arthritis. Arthritis Rheum. 2010 Dec;62(12):3537-46. doi: 10.1002/art.27692. PMID: 20722031.
[3] Orange DE, Blachere NE, DiCarlo EF,et al. Rheumatoid Arthritis Morning Stiffness Is Associated With Synovial Fibrin and Neutrophils. Arthritis Rheumatol. 2020 Apr;72(4):557-564. doi: 10.1002/art.41141. Epub 2020 Feb 12. PMID: 31612614; PMCID: PMC7113097.
[4] Aletaha D, Neogi T, Silman AJ, et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010 Sep;62(9):2569-81. doi: 10.1002/art.27584. PMID: 20872595.
[5] Sokolova MV, Schett G, Steffen U. Autoantibodies in Rheumatoid Arthritis: Historical Background and Novel Findings. Clin Rev Allergy Immunol. 2022 Oct;63(2):138-151. doi: 10.1007/s12016-021-08890-1. Epub 2021 Sep 8. PMID: 34495490; PMCID: PMC9464122.
[6] Niewold TB, Harrison MJ, Paget SA. Anti-CCP antibody testing as a diagnostic and prognostic tool in rheumatoid arthritis. QJM. 2007 Apr;100(4):193-201. doi: 10.1093/qjmed/hcm015. PMID: 17434910.
[7] Mohammed RH, Bhutta BS. Hand and Wrist Rheumatoid Arthritis. [Updated 2023 Aug 17]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK560890/
[8] Rheumatoid arthritis in adults: diagnosis and management. London: National Institute for Health and Care Excellence (NICE); 2018 Jul 11. PMID: 30102507.
[9] Nell VP, Machold KP, Eberl G, Stamm TA, Uffmann M, Smolen JS. Benefit of very early referral and very early therapy with disease-modifying anti-rheumatic drugs in patients with early rheumatoid arthritis. Rheumatology (Oxford). 2004 Jul;43(7):906-14. doi: 10.1093/rheumatology/keh199. Epub 2004 Apr 27. PMID: 15113999.
[10] Felson DT, Smolen JS, Wells G, et al. American College of Rheumatology/European League against rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Ann Rheum Dis 2011;70:404–13.doi:10.1136/ard.2011.149765
[11] Aletaha D , Alasti F , Smolen JS . Optimisation of a treat-to-target approach in rheumatoid arthritis: strategies for the 3-month time point. Ann Rheum Dis 2016;75:1479–85.doi:10.1136/annrheumdis-2015-208324
[12] Aletaha D, Funovits J, Keystone EC, et al. Disease activity early in the course of treatment predicts response to therapy after one year in rheumatoid arthritis patients. Arthritis Rheum 2007;56:3226–35.
[13] Pincus T, Yazici Y, Sokka T, et al. Methotrexate as the “anchor drug” for the treatment of early rheumatoid arthritis. Clin Exp Rheumatol 2003;21(Suppl 31):S178–85.
[14] Visser K, van der Heijde D. Optimal dosage and route of administration of methotrexate in rheumatoid arthritis: a systematic review of the literature. Ann Rheum Dis 2009;68:1094–9.
[15] Fraenkel L, Bathon JM, England BR, et al. 2021 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis. Arthritis Care Res (Hoboken). 2021 Jul;73(7):924-939. doi: 10.1002/acr.24596. Epub 2021 Jun 8. PMID: 34101387; PMCID: PMC9273041.
[16] Smolen JS, Kalden JR, Scott DL, et al. Efficacy and safety of leflunomide compared with placebo and sulphasalazine in active rheumatoid arthritis: a double-blind, randomised, multicentre trial. Lancet 1999;353:259–66.
[17] Katz SJ, Russell AS. Re-evaluation of antimalarials in treating rheumatic diseases: re-appreciation and insights into new mechanisms of action. Curr Opin Rheumatol 2011;23:278–81.
[18] O'Dell JR, Mikuls TR, Taylor TH, et al; CSP 551 RACAT Investigators. Therapies for active rheumatoid arthritis after methotrexate failure. N Engl J Med. 2013 Jul 25;369(4):307-18. doi: 10.1056/NEJMoa1303006. Epub 2013 Jun 11. PMID: 23755969.
[19] Svensson B, Boonen A, Albertsson K, et al. Low-dose prednisolone in addition to the initial disease-modifying antirheumatic drug in patients with early active rheumatoid arthritis reduces joint destruction and increases the remission rate: a two-year randomized trial. Arthritis Rheum 2005;52:3360–70.
[20] da Silva JA, Jacobs JW, Kirwan JR, et al. Safety of low dose glucocorticoid treatment in rheumatoid arthritis: published evidence and prospective trial data. Ann Rheum Dis 2006;65:285–93.
[21] Holloway K, van Dijk L. The world medicines situation 2011. rational use of medicines. Geneva: World Health Organzization, 2011.doi:WHO/EMP/MIE/2011.2.2
[22] European Medicines Agency. Xeljanz (tofacitinib): increased risk of major adverse cardiovascular events and malignancies with use of tofacitinib relative to TNF-alpha inhibitors, 2021.
[23] Smolen JS, Landewé RBM, Bergstra SA, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update. Ann Rheum Dis. 2023 Jan;82(1):3-18. doi: 10.1136/ard-2022-223356. Epub 2022 Nov 10. Erratum in: Ann Rheum Dis. 2023 Mar;82(3):e76. doi: 10.1136/ard-2022-223356corr1. PMID: 36357155.
[24] Burmester G-R, Kivitz AJ, Kupper H, et al. Efficacy and safety of ascending methotrexate dose in combination with adalimumab: the randomised concerto trial. Ann Rheum Dis 2015;74:1037–44.doi:10.1136/annrheumdis-2013-204769
[25] Gabay C, Emery P, van VR, et al. Tocilizumab monotherapy versus adalimumab monotherapy for treatment of rheumatoid arthritis (ADACTA): a randomised, double-blind, controlled phase 4 trial. Lancet 2013;381:1541–50.
[26] Fleischmann R, Takeuchi T, Schlichting D, et al. Baricitinib, methotrexate, or baricitinib plus methotrexate in patients with early rheumatoid arthritis who had received limited or no treatment with disease-modifying anti-rheumatic drugs (DMARDs): phase 3 trial results [abstract]. Arhritis Rheum 2015;67 (Suppl 10).
[27] Smolen JS, Landewé RBM, Bijlsma JWJ, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. Ann Rheum Dis 2020;79:685–99.doi:10.1136/annrheumdis-2019-216655pmid: http://www.ncbi.nlm.nih.gov/pubmed/31969328
[28] Gossec L, Bijlsma JW, Bombardier C, et al. Dissemination and evaluation of the 3E initiative recommendations for use of methotrexate in rheumatic disorders: results of a study among 2233 rheumatologists. Ann Rheum Dis. 2011 Feb;70(2):388-9. doi: 10.1136/ard.2010.128652. Epub 2010 May 14. PMID: 20472594.
[29] American Association for the Study of Liver Diseases. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Clin Liver Dis (Hoboken) 2018; 11: 81
[30] Terrault NA, Lok ASF, McMahon BJ, Chang KM, Hwang JP, Jonas MM, et al. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology 2018; 67: 1560–99.
[31] Baker JF, Sauer B, Teng CC,et al. Initiation of Disease-Modifying Therapies in Rheumatoid Arthritis Is Associated With Changes in Blood Pressure. J Clin Rheumatol. 2018 Jun;24(4):203-209. doi: 10.1097/RHU.0000000000000736. PMID: 29664818; PMCID: PMC7461421.
[32] Chung ES, Packer M, Lo KH, Fasanmade AA, Willerson JT. Randomized, double-blind, placebo-controlled, pilot trial of infliximab, a chimeric monoclonal antibody to tumor necrosis factor-alpha, in patients with moderate-to-severe heart failure: results of the anti-TNF Therapy Against Congestive Heart Failure (ATTACH) trial. Circulation 2003; 107: 3133–40.
[33] Narváez J, Díaz Del Campo Fontecha P, Brito García N,et al. SER-SEPAR recommendations for the management of rheumatoid arthritis-related interstitial lung disease. Part 2: Treatment. Reumatol Clin (Engl Ed). 2022 Nov;18(9):501-512. doi:10.1016/j.reumae.2022.03.004. Epub 2022 Sep 3. PMID: 36064885.
[34] Russell MD, Dey M, Flint J,et al.; BSR Standards, Audit and Guidelines Working Group. Executive Summary: British Society for Rheumatology guideline on prescribing drugs in pregnancy and breastfeeding: immunomodulatory anti-rheumatic drugs and corticosteroids. Rheumatology (Oxford). 2023 Apr 3;62(4):1370-1387. doi:10.1093/rheumatology/keac558. PMID: 36318965; PMCID: PMC10070067.
[35] Vallejo-Yagüe E, Keystone EC, Kandhasamy S, Micheroli R, Finckh A, Burden AM. Primary and secondary non-response: in need of operational definitions in observational studies. Ann Rheum Dis. 2021 Aug;80(8):961-964. doi: 10.1136/annrheumdis-2021-220202. Epub 2021 Apr 21. PMID: 33883161; PMCID: PMC8292559.
[36] Nagy G, Roodenrijs NMT, Welsing PM, et al. EULAR definition of difficult-to-treat rheumatoid arthritis. Ann Rheum Dis. 2021 Jan;80(1):31-35. doi: 10.1136/annrheumdis-2020-217344. Epub 2020 Oct 1. PMID: 33004335; PMCID: PMC7788062.
- Annexes
Annex 1:
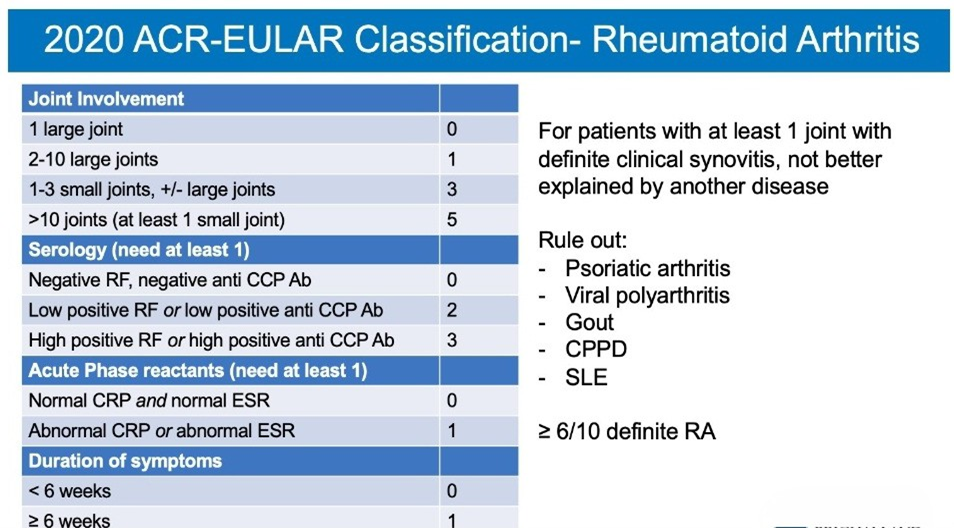
Annex 2:
Clinical Disease Activity Index (CDAI) for Rheumatoid Arthritis
When to Use
● All patients with rheumatoid arthritis.
● Re-assessing patients after starting a new medication for RA
Pearls and pitfalls
● Assessment of disease activity in rheumatoid arthritis (RA) should be viewed as a dynamic, on-going process; many experts recommend assessing the Clinical Disease Activity Index (CDAI) at every patient visit.
● Early treatment is important for optimal RA outcomes, and the CDAI can help give a gestalt view of a patient’s disease severity and activity
Why Use
● The CDAI provides an excellent overview of a patient's disease severity (it correlates closely with the SDAI which requires laboratory data) and the DAS-28 (which is a much longer, 28-joint assessment).
● CDAI can also help guide therapy based on severity of disease
FORMULA
CDAI = Tender Joint Count + Swollen Joint Count + Patient Global Activity + Provider Global Activity
Score interpretation:
CDAI Score Range Disease Severity
≤ 2.8 Remission
> 2.8 - 10.0 Low
> 10.0 - 22.0 Moderate
> 22.0 High