Infant and Young Child Feeding in Egypt
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Infant and Young Child Feeding in Egypt |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 29 Oct 2025" Download Guideline
- Executive Summary
Optimal nutrition is critical in the first two years of a child’s life as it improves morbidity and mortality, reduces risk of chronic disease and improves overall development
Complementary feeding is described by the World Health Organization (WHO) as the period from age 6 months onwards where breast milk alone is no longer sufficient to fulfill the nutritional requirements of the infant, therefore requiring complementary feeding to be started
Nutritional deficiencies are common in Egyptian infants, with iron, vitamin D, vitamin A, protein, calories and certain trace elements a key concern
There are currently no detailed guidelines for complementary feeding in Egypt. Consequently, current practice deviates greatly from international guidelines. The Demographic and Health survey also reports that complementary feeding does not follow best practice
➡️ Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- WHO guideline ” Guiding Principles For Complementary Feeding of the Breastfed Child”(2003).
2- ESPGAN position paper “Complementary Feeding: A Position Paper by the European Society for Paediatric Gastroenterology,Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition (2017).
3- American Academy of Pediatrics Committee on Nutrition. Complementary feeding. In: Pediatric Nutrition, 7th ed, Kleinman RE, Greer FR (Eds), American Academy of Pediatrics, Elk Grove Village, IL 2014. p.123.
4- Guideline: Daily iron supplementation in infants and children. Geneva: World Health Organization; 2016.
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
A. Achieve Proper physical & neurodevelopmental outcome of infants.
B. Prevent malnutrition, under nutrition and over nutrition.
C. Prevent micronutrient deficiencies and its comorbidities.
The guideline covers healthy full term infants of both sexes, either breastfed or non-breastfed without co-morbidities.
This guideline emphasis on proper introduction of solid food for healthy full term infants of both sexes, either breastfed or non - breastfed without co-morbidities.
We can summarize the guidelines’ recommendations for Infant and Young Child Feeding in Egypt in the following:
▪️ Exclusive or full breast feeding should be promoted for at least 6 months. (strong recommendation).
▪️ Complementary foods (ie, solid foods and Liquids other than breast milk or infant formula) should not be introduced before 17 weeks of age but should not be delayed beyond 26 weeks of age in formula fed infants. (Weak (conditional) recommendation).
▪️ Complementary food should be adequate in nutrition and as long as iron-rich foods are included in early complementary feeding, foods can be introduced in any order and at a rate that suits the infant, however it is recommended that complementary foods are initiated in following order
Iron fortified cereals
Iron rich vegetables and fruits
Egg yolk
Pureed poultry/meat/fish
yogurt. (strong recommendation).
▪️ Allergenic foods may be introduced when CF is commenced. (Weak (conditional) recommendation).
▪️ Gluten may be introduced into the infant’s diet any time between 6 and 12months of age; consumption of large quantities should be avoided during the first weeks of gluten introduction. Neither breastfeeding nor non breast-feeding during gluten introduction has been shown to reduce the risk of CD. (Weak (conditional) recommendation).
▪️ Start at six months of age with small amounts of food and increase the quantity as the child gets older, while maintaining frequent breastfeeding.
The energy needs from complementary foods for infants with “average” breast milk intake in developing countries are approximately 200 kcal per day at 6-8 months of age, 300 kcal per day at 9-11 months of age, and 550 kcal per day at 12-23 months of age. (Weak (conditional) recommendation).
▪️The appropriate number of feedings depends on the energy density of the local foods and the usual amounts consumed at each feeding. For the average healthy infant, meals of complementary foods should be provided 2-3 times per day at 6-8 months of age and 3-4 times per day at 9-11 and 12-24 months of age, with additional nutritious snack (such as a piece of fruit or bread) offered 1-2 times per day, as desired. (Weak (conditional) recommendation).
▪️ Infants have high nutrient requirements but the capacity to consume small amounts of food. Feed a variety of foods to ensure that nutrient needs are met. Meat, poultry, fish or eggs should be eaten daily, or as often as possible. (Weak (conditional) recommendation).
▪️With the exception of liquid cow milk, which is not recommended before 12 months of age, WHO recommend giving whole cream dairy products as yogurt, cheese & pudding starting after 6m (good source of protein, calcium & DHA). (Weak (conditional) recommendation).
▪️ Honey has a lot of benefits for human, including infants below the age of one year; It has anti-microbial (anti-bacterial, anti-viral, anti- parasitic, anti- fungal); anti-inflammatory; anti-tumor; immune-modulator; prebiotic and probiotic effects.
Honey has been prescribed without prior testing, to thousands of infants below the age of one year for almost 20 years (since 1998) without any single occurrence of infant botulism.
Assuming that honey may rarely contain the spores of Clostridium Botulinum, the recommendation that we should not give honey to infants below the age of one year should also include other sources of C. botulinum, including many other food items such as fruits, vegetables, mushrooms, garlic cloves...etc. Therefore, it is impossible to prevent infant botulism even if we prevent giving honey to infants below the age of one year. (Weak (conditional) recommendation).
▪️ Based on available evidence in Egyptian population the committee suggests honey introduction in small amounts starting 9 month. (Good practice statement).
▪️ Fava beans should be introduced in small amounts with other foods.
Children with family history of G6PD should be screened before introduction. (Weak (conditional) recommendation).
▪️Fruit juice (including 100-percent home made fruit juice) generally should not be offered to infants younger than 12 months. For infants between 6 and 12 months, we suggest consumption of mashed or puréed whole fruit rather than 100-percent fruit juice. (Weak (conditional) recommendation).
▪️Breastfeeding mothers may also need vitamin-mineral supplements or fortified products, both for their own health and to ensure normal concentrations of certain nutrients (particularly vitamins) in their breast milk. (Weak (conditional) recommendation).
▪️ Daily iron supplementation is recommended as a public health intervention in infants and young children aged 6–23 months, living in settings where anaemia is highly prevalent, for preventing iron deficiency and anaemia in dose of 10-12.5 mg elemental iron for 3 consecutive months of year. (Weak (conditional) recommendation).
▪️ All infants should be supplemented with vitamin D (400 IU) since birth. (Weak (conditional) recommendation).
▪️Breast fed infants don’t need extra water as breast milk is 80% water. Non-breastfed infant need at least 400-600 mL/d of extra fluids (in addition to the 200-700 mL/d of water that is estimated to come from milk and other foods) in a temperate climate, and 800-1200 mL/d in a hot climate. Plain, clean boiled water should be offered several times per day to ensure that the infant’s thirst is satisfied. (Weak (conditional) recommendation).
Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/- Recommendations
|
Table 3. Recommendations |
|
|
|||
|
A. Time of introduction of solid food |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A1 |
What is the appropriate time to introduce complementary feeding for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2003
ESPAGHN 2017 |
Exclusive or full breast feeding should be promoted for at least 6 months.
Complementary foods (ie, solid foods and Liquids other than breast milk or infant formula) should not be introduced before 17 weeks of age but should not be delayed beyond 26 weeks of age in formula fed infants. |
High
Low |
Strong
Weak (Conditional) |
|
|
|
||||
|
B. Order of introduction of solid food |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
What is the suggested order of introducing complementary foods to healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2003
ESPAGHN 2017
ESPAGHN 2017 |
Complementary food should be adequate in nutrition and as long as long as iron-rich foods are included in early complementary feeding, foods can be introduced in any order and at a rate that suits the infant, however it is recommended that complementary foods are initiated in following order
ü Allergenic foods may be introduced when CF is commenced
ü Gluten may be introduced into infant’s diet any time between 6 and 12months of age; consumption of large quantities should be avoided during the first weeks of gluten introduction. Neither any breastfeeding nor breast-feeding during gluten introduction has been shown to reduce the risk of CD.
|
High
Low
Low |
Strong
Weak (Conditional)
Weak (Conditional) |
|
|
|
||||
|
C. Amount of food to be given |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
C1 |
What is the appropriate amount of complementary food to be served for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2003 |
Start at six months of age with small amounts of food and increase the quantity as the child gets older, while maintaining frequent breastfeeding. The energy needs from complementary foods for infants with “average” breast milk intake in developing countries are approximately 200 kcal per day at 6-8 months of age, 300 kcal per dayat 9-11 months of age, and 550 kcal per day at 12-23 months of age
|
Low |
Weak (Conditional) |
|
Table 6. Recommendations |
|
|
|||
|
D. Number of meals to be given |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
D1 |
How frequent complementary food should be served for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2003 |
The appropriate number of feedings depends on the energy density of the local foods and the usual amounts consumed at each feeding. For the average healthy infant, meals of complementary foods should be provided 2-3 times per day at 6-8 months of age and 3-4 times per day at 9-11 and 12-24 months of age, with additional nutritious snack (such as a piece of fruit or bread) offered 1-2 times per day, as desired |
Low |
Weak (Conditional) |
|
Table 7. Recommendations |
|
|
|||
|
E. Food diversity |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
E1 |
What is the appropriate food diversity in the meals of healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities morbidities |
WHO 2003 |
Infants have high nutrient requirements but the capacity to consume small amounts of food. Feed a variety of foods to ensure that nutrient needs are met. Meat, poultry, fish or eggs should be eaten daily, or as often as possible |
Low |
Weak (Conditional) |
|
Table 8. Recommendations |
|
|
|||
|
F. Dairy products |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
F1 |
Which type of dairy products and when should be served to healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2003 |
With the exception of liquid cow milk, which is not recommended before 12 months of age, WHO recommend giving whole cream dairy products as yogurt, cheese & pudding starting after 6m (good source of protein, calcium & DHA). |
Low
|
Weak (Conditional) |
|
Table 9. Recommendations |
|
|
|||
|
G. Honey introduction |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
G1 |
What is the appropriate time to introduce honey for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
-Medically Graded Honey Supplementation Formula to Preterm Infants as a Prebiotic: A Randomized Controlled Trial. Journal of pediatric gastroenterology and nutrition (2017). (19)
-Effect of honey on febrile neutropenia in children with acute lymphoblastic leukemia: A randomized crossover open-labeled study. Complementary therapies in medicine (2016). (20)
-Effect of honey on gastric emptying of infants with protein energy malnutrition. European journal of clinical investigation (2010). (21)
-Multiplex PCR for detection and genotyping of C. botulinum types A, B, E and F neurotoxin genes in some Egyptian food products. J. Am. Sci, (2011). (22) |
Honey has a lot of benefits for human, including infants below the age of one year; It has anti-microbial (anti-bacterial, anti-viral, anti- parasitic, anti- fungal); anti-inflammatory; anti-tumor; immune-modulator; prebiotic and probiotic effects.
Honey has been prescribed without prior testing, to thousands of infants below the age of one year for almost 20 years (since 1998) without any single occurrence of infant botulism.
Assuming that honey may rarely contain the spores of Clostridium Botulinum, the recommendation that we should not give honey to infants below the age of one year should also include other sources of C. botulinum, including many other food items such as fruits, vegetables, mushrooms, garlic cloves...etc. Therefore, it is impossible to prevent infant botulism even if we prevent giving honey to infants below the age of one year.
Based on available evidence in Egyptian population the committee suggests honey introduction in small amounts starting 9 month |
Very low |
Weak (Conditional)
Good practice statement |
|
Table 10. Recommendations |
|
|
|||
|
H. Fava Beans |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
H1 |
What is the appropriate time to introduce fava beans for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2003+EO |
Fava beans should be introduced in small amounts with other foods Children with family history of G6PD should be screened before introduction |
Low |
Weak (Conditional) |
|
Table 11. Recommendations |
|
|
|||
|
I. Fruit juice |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
I1 |
When is it appropriate to introduce fruit juices and how much for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
ESPAGHN 2017 |
Fruit juice (including 100-percent home made fruit juice) generally should not be offered to infants younger than 12 months. For infants between 6 and 12 months, we suggest consumption of mashed or puréed whole fruit rather than 100-percent fruit juice |
Low |
Weak (Conditional) |
|
|
|
||||
|
J. Supplementation |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
J1 |
What are mandatory supplementations recommended for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2003
WHO 2016
AAP 2014 |
breastfeeding mothers may also need vitamin-mineral supplements or fortified products, both for their own health and to ensure normal concentrations ofcertain nutrients (particularly vitamins) in their breast milk.
Daily iron supplementation is recommended as a public health intervention in infants and young children aged 6–23 months, living in settings where anaemia is highly prevalent, for preventing iron deficiency and anaemia in dose of 10-12.5 mg elemental iron for 3 consecutive months of year
All infants should be supplemented with vitamin D (400 IU) since birth |
Low
Low
Low |
Weak (Conditional)
Weak (Conditional)
Weak (Conditional) |
|
Table 13. Recommendations |
|
|
|||
|
K. Water |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
K1 |
When is it appropriate to introduce water and how much for healthy full term infant, both sexes, either breastfed or not breastfed without co-morbidities |
WHO 2005 |
Breast fed infants don’t need extra water as breast milk is 80% water Non-breastfed infan need at least 400-600 mL/d of extra fluids (in addition to the 200-700 mL/d of water that is estimated to come from milk and other foods) in a temperate climate, and 800-1200 mL/d in a hot climate. Plain, clean boiled water should be offered several times per day to ensure that the infant’s thirst is satisfied. |
Low |
Weak (Conditional) |
➡️Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt mos. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
➡️Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
➡️Steps of implementing Infant and Young Child Feeding in Egypt strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
Evidence to Decision Tables: (both ETD and changing strength of recommendation were not done in this guideline)
Guideline Implementation Tools
Educational materials based on this Adapted CPG for national complementaty feeding guidelines are made available in several forms, including
- Booklet with executive summary of guidelines for physicians and primary health care professionals
- Illustrative booklet in arabic directed to mothers with steps of solid food introduction
- Powerpoint presentation for mothers for more illustration of the steps for introduction of complementary feeding.
- Powerpoint presentation for training physicians and primary health care professionals illustrating the steps of introducing complementary feeding and how to explain these steps to the mothers
- Poster with illustrative diagrams for mothers to help them with the steps of solid food introduction to be placed in the primary care facilities, clinics and pediatric hospitals.
Practice Guidelines for Complementary Feeding
Physicians Guide
When To Start?
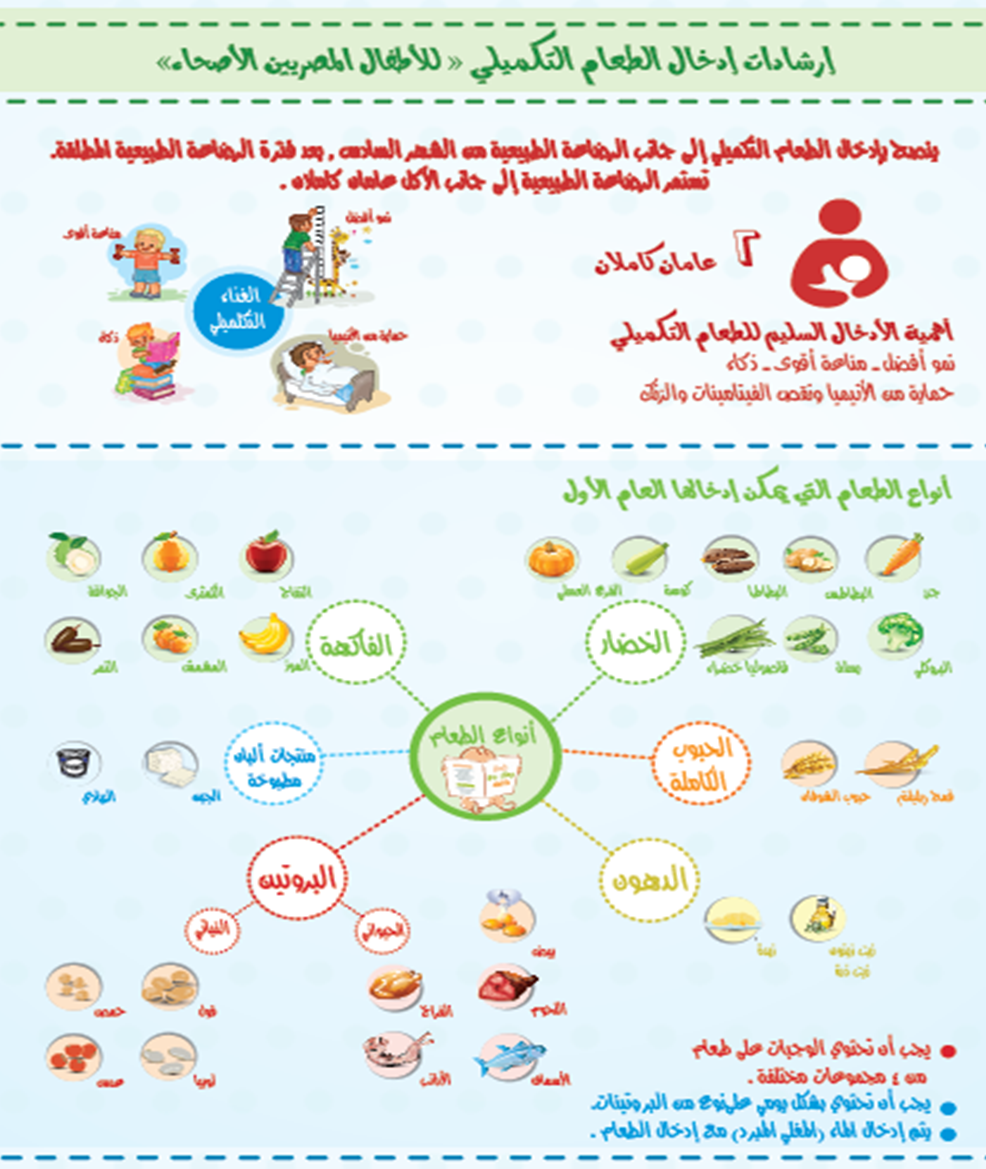
Exclusive or full breast feeding should be promoted for at least 6 months
Order of Introduction
Foods can be introduced in any order and at a rate that suits the infant, however it is recommended that complementary foods are initiated in following order
• Iron fortified cereals
• Iron rich vegetables and fruits
• Egg yolk
• Pureed poultry/meat/fish
• Yogurt.
What about Allergic Food
- Allergenic foods may be introduced when Complementary Feeding is commenced any time after 4 months (17 weeks).
- Gluten may be introduced into infant’s diet any time between 4 and 12months of age; consumption of large quantities should be avoided during the first weeks of gluten introduction
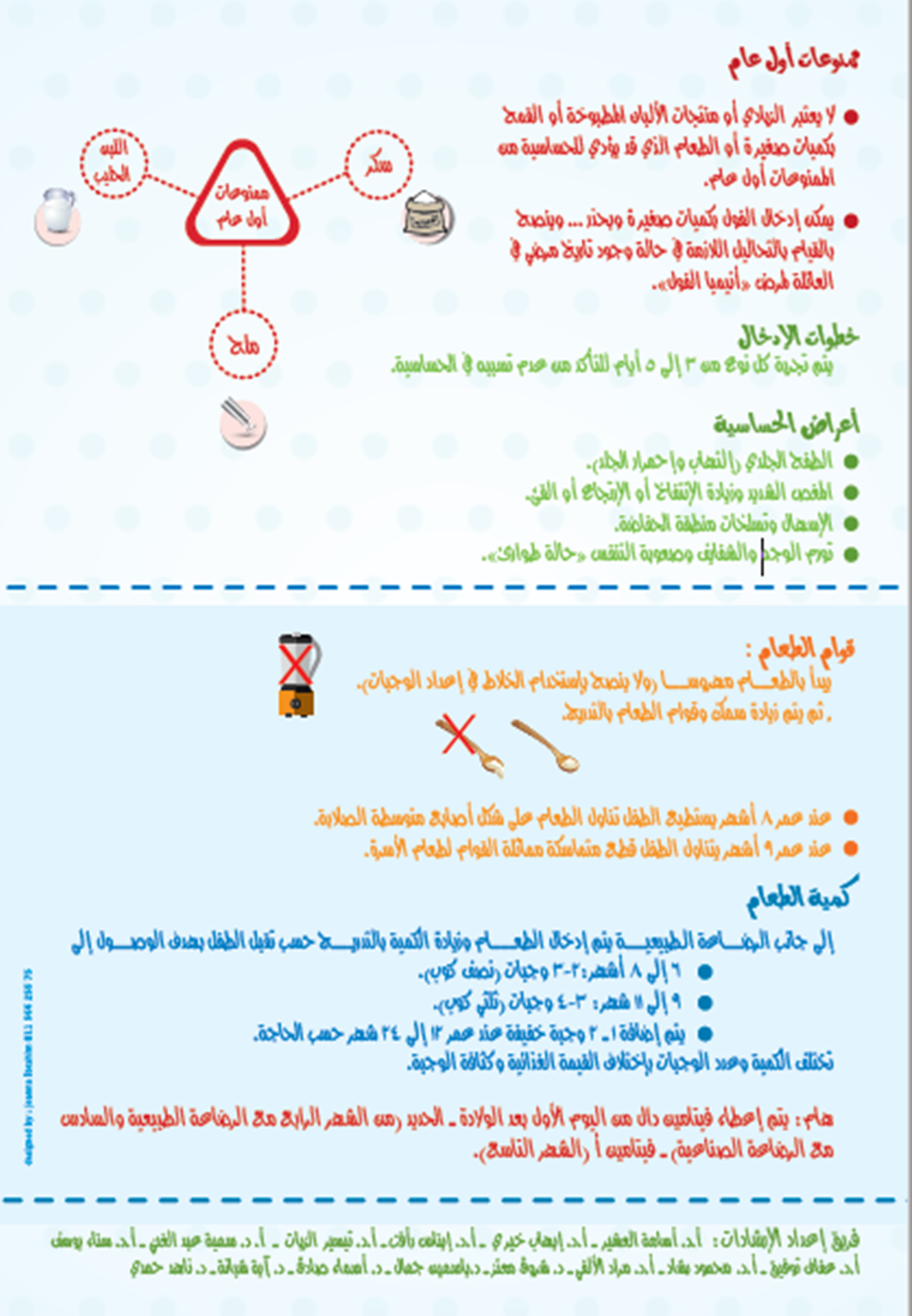
Food Amount, and consistency
Start at six months of age with small amounts of food and increase the quantity as the child gets older, while maintaining frequent breastfeeding.
The appropriate number of feedings also depends on the energy density of the local foods and the usual amounts consumed at each feeding.
Also In practice, caregivers will not be measuring the energy content of foods to be offered. Thus, the amount of food to be offered should be based on the principles of responsive feeding
Practicing responsive feeding, applying the principles of psycho-social care.
a) feed infants directly and assist older children when they feed themselves, being sensitive to their hunger and satiety cues.
b) feed slowly and patiently, and encourage children to eat, but do not force them.
c) if children refuse much food, experiment with different food combinations, tastes, textures and methods of encouragement.
d) minimize distractions during meals if the child loses interest easily.
e) Remember that feeding times are periods of learning and love - talk to children during feeding, with eye-to-eye contact.
Food consistency and variety should be increased with the child’s
age.
Practical guidance on the quality, frequency and amount of food to offer children 6-23 months of age besides milk (either breast milk or its substitutes )
|
Age In months |
Energy needed per day in addition to breastmilk |
Texture |
Frequency |
Amount of food an average child will usually eat at each meal |
|
6-8 |
200 kcal/day |
Start by thick porridge,well mashed food Continue with mashed family foods By eight months most infants can also eat “finger foods” (snacks that can be eaten by children alone).
|
-2-3 meals per day Depending on the child’s appetite,1-2 snacks may be offered |
Start with 2-3 tabblespoonfuls per feed,increasing gradually to of a 250 ml cup |
|
9-11 |
300 kcal/day |
Finely chopped or mashed foods, and fods that baby can pick up |
-3-4 meals per day Depending on the child’s appetite,1-2 snacks may be offered |
to of a 250 ml cup/bowel |
|
12-23 |
550kcal/day |
Family foods, chopped or mashed if necessary |
-3-4 meals per day Depending on the child’s appetite,1-2 snacks may be offered |
to of a 250 ml cup/bowel |
Avoid foods in a form that may cause choking, such as whole nuts, whole grapes or raw carrots, whole or in pieces
Food diversity
Infants have high nutrient requirements but the capacity to consume small amounts of food. Feed a variety of foods to ensure that nutrient needs are met.
The daily diet should include:
Ø Vitamin A-rich foods: (e.g. dark coloured fruits and vegetables; red palm oil; vitamin A-fortified oil or foods);
Ø vitamin C-rich foods: (e.g. many fruits, vegetables and potatoes) consumed with meals to enhance iron absorption.
Ø Vitamin B rich foods: (e.g. liver, egg, dairy products, green leafy vegetables, soybeans), vitamin B6 (e.g. meat, poultry, fish, banana, green leafy vegetables, potato and other tubers, peanuts) and folate (e.g. legumes, green leafy vegetables, orange juice
Meat, poultry, fish or eggs should be eaten daily, or as often as possible
If milk and other animal-source foods are not eaten in adequate amounts, both
grains and legumes should be consumed daily, if possible, within the same meal, to
ensure adequate protein quality.
Dairy products
With the exception of liquid cow milk, which is not recommended before 12 months of age,
WHO recommend giving whole cream dairy products like yogurt, cheese & pudding starting from 6m (good source of protein, calcium & DHA).
Skimmed(non-fat) milk is not recommended as a major food source for children under two
Honey
Honey has a lot of benefits for humans, including infants below the age of one year; It has anti-microbial (anti-bacterial, anti-viral, anti- parasitic, anti- fungal); anti-inflammatory; anti-tumor; immune modulator; prebiotic and probiotic effects.
Based on available evidence in Egyptian population the committee suggests honey introduction in small amounts starting 9 months.
Fava Beans
Fava beans should be introduced in small amounts with other foods
Children with family history of G6PD should be screened before introduction
Fruit juice
Fruit juice (including 100-percent homemade fruit juice) generally should not be offered to infants younger than 12 months. For infants between 6 and 12 months, we suggest consumption of mashed or puréed whole fruit rather than 100-percent fruit juice
Supplementations
Breastfeeding mothers may also need vitamin-mineral supplements
or fortified products, both for their own health and to ensure normal concentrations of certain nutrients (particularly vitamins) in their breast milk.
Daily iron supplementation is recommended as a public health intervention in infants and young children aged 6–23 months, living in settings where anemia is highly prevalent, for preventing iron deficiency and anemia in dose of 10-12.5 mg elemental iron for 3 consecutive months of year
All infants should be supplemented with vitamin D (400 IU) since birth
Water
Breast fed infants don’t need extra water as breast milk is 80% water
Non-breastfed infants need at least 400-600 mL/d of extra fluids (in addition to the 200-700 mL/d of water that is estimated to come from milk and other foods) in a temperate climate, and 800-1200 mL/d in a hot climate.
Plain, clean boiled water should be offered several times per day to ensure that the infant’s thirst is satisfied.
The Five Keys to Safer Food include
1) keep hands, food preparation surfaces and equipment clean,
2) separate raw meat, poultry and seafood from other foods and use separate utensils and cutting boards for their preparation,
3) cook foods thoroughly, especially meat, poultry, eggs and seafood,
4) keep food at safe temperatures
5) use safe water and raw materials
- Acknowledgements
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge World Health organization infant and Young Child guidelines (2003) (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️The authors appreciate the authors to WHO guideline” Guiding Principles For Complementary Feeding of the Breastfed Child” and the authors of the ES PGAN position paper “Complementary Feeding: A Position Paper by the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition” from which this guideline is adapted.
▪️Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) <Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Sanaa Youssef Shaaban |
Professor of Pediatrics, Ain shams University |
Supervisor, Clinical expert |
|||
|
Prof. Mourad Alfy Ramzy Tadros |
Professor of Pediatrics, Armed forced faculty of medicine. |
Supervisor, Clinical expert |
|||
|
Prof. Hanna Aboulghar |
Professor of Pediatrics, Cairo University |
Clinical expert |
|||
|
Prof. Ehab Khairy El khashab |
Professor of Pediatrics, Ain Shams University |
Clinical expert |
|||
|
Prof. Mahmoud Rashad |
Professor of Pediatrics, Al-Azhar University |
Clinical expert |
|||
|
Prof. Somaya Mohamed Abd El -Ghany |
Professor of Pediatrics, Al-Azhar University Faculty of Medicine for Girls |
Clinical expert |
|||
|
Prof. Tayseer Mohamed El Zayat |
Professor of Pediatrics, Al-Azhar University Faculty of Medicine for Girls |
Clinical expert |
|||
|
Prof. Osama M Al-Asheer |
Professor of Pediatrics, Assuit University |
Clinical expert |
|||
|
Prof. Enas Raafat |
Professor of child health, National Research Center |
Clinical expert |
|||
|
Prof. Afaf Abdel Fattah Tawfik |
Previous dean of National Nutrition Institute |
Clinical expert |
|||
|
Prof. Yasmin Gamal El Gendy |
Professor of Pediatrics and Clinical Nutrition, AinShams University, AFCM |
Coordinator, editor, Clinical expert |
|||
|
Dr. Shrouk MoatazAbdallah |
Lecturer of Pediatrics Cairo University |
Clinical expert |
|||
|
Dr. Asmaa Sadek A. Sadek |
Pediatric & neonatology consultant |
Clinical expert |
|||
|
Dr. Ayah Shabana |
Lecturer of Pediatrics, Suez University |
Clinical expert |
|||
|
Dr. Nahed Mohamed hamdy |
Pediatric & neonatology consultant |
Clinical expert |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Dr. Abla AL Alfy |
Regional Advisor for Middle East and Africa- Royal College of Paediatrics and Child Health (RCPCH) Head of Egyptian Members Assoc- RCPCH Co-Chair –Scientific Council-Military Paediatric Board IBCLC |
||||
|
Dr. Ahmed El Saeed Younis |
Head of Egyptian Paediatric Society |
||||
|
Dr. Aliaa Hafiz |
Head of nutrition unit, world food program IBCLC |
||||
|
Dr. Mona Hafez El Naka |
Ministry of Health IBCLC |
||||
|
Dr. Nahla Roshdy |
Ministry of Health IBCLC |
||||
|
External Reviewer(s) for methodology |
|||||
|
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
|||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
- Abbreviations
|
AAP |
American Academy of Pediatrics |
||
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AFCM |
Armed Forces College of Medicine |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
ASD |
Alternative Service Delivery |
||
|
BF |
Breast Feeding |
||
|
CD |
Coeliac Disease |
||
|
CF |
Complementary Feeding |
||
|
CPG |
Clinical Practice Guideline |
||
|
DHA |
Docosahexaenoic Acid |
||
|
DHS |
Demographic and Health Survey |
||
|
EBM |
Evidence Based Medicine |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ERG |
External Review Group |
||
|
ESPGHAN |
the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition |
||
|
G6PD |
Glucose-6-Phosphate Dehydrogenase Deficiency |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
OPC |
Outpatient Clinic |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
RCT |
Randomized Controlled Trial |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
||
|
UNICEF |
United Nations International Children's Emergency Fund |
||
|
WHO |
World Health Organization |
- Glossary
Acceptability
Is the extent to which the users are likely to adopt a recommendation, based on internal qualities such as clarity, comprehensiveness, and logical reasoning and on external factors such as the burden imposed on the process and system of care, patient and providers attitudes and beliefs, and patients needs, expectations, and preferences.
Adaptation (of guidelines)
Is the systematic approach to considering the use and/or modification of (a) guidelines(s) produced in one cultural and organizational setting for application in different context? Adaptation can be used as an alternative to de novo guideline development or for customizing (an) existing guideline(s) to suit the local context.
Admission
Admission, for the purpose of this guideline, refers to a child being registered and entering inpatient care as a patient. This is distinguished from the term “enrolment”, which is used for outpatient care.
Adoption (of a guideline)
Is the acceptance of a guideline as a whole after the assessment of its quality, currency, and content. When health care providers (or other users of recommendations) adopt a guideline, they feel committed to change their practices in accordance with the recommendations of the guideline.
Applicability
Is the extent to which the users are able to put a recommendation into practice, based on internal qualities such as a clearly defined eligible patient population that matches the population to which the intervention is targeted in the local setting and external factors such as the availability of the necessary knowledge, skills, provider time, staff, equipment, and other resources.
Applicability is sometimes taken as a synonym for feasibility:
- Feasibility of the acquisition of necessary skills and knowledge
- Feasibility of the necessary increase in provider time, staff, equipment, and so on.
Culture
Culture represents the norms and values of a specific group, community, or population.
Diffusion
Is a passive means of transferring knowledge; it is not directed towards a target audience (e.g. publication of articles in medical journals).
Dissemination
Is more active than diffusion in that it targets a specific audience and involve tailoring the information for that audience (e.g. of dissemination strategies include targeted mailings, presentations, and press conferences.
Evidence-based principles
Evidence-Based Medicine (EBM) has been defined as ― the conscientious, explicit, and judicious use of current best evidence in making decisions about the care of individual patients. The practice of EBM means integrating individual clinical expertise with the best available external clinical evidence from systematic research.
Evidence tables
Are summaries of the most salient information from studies identified in the systematic review. The elements of evidence tables are dependent on the types of information in studies related to a particular topic but might include information such as the article reference, the study type (e.g. RCT or Cohort), the number of patients and their characteristics, and the intervention, comparison arms, outcome measures, and effect sizes.
Guideline or Clinical Practice Guideline (CPG)
Systematically developed statements about specific health problems, intended to assist practitioners and patients in making decisions about appropriate health care.
Guideline consistency
Agreement between the evidence and the recommendations, based on the:
- Comprehensiveness of the study search and selection process,
- Coherence between the results of the studies and their interpretation by the guideline authors, and
- Transparency between interpretation and recommendations.
Guideline content
In the ‘ADAPTE Manual and Resource Toolkit for Guideline Adaptation’ document, guideline content refers to the recommendations in the source guidelines.
Guideline currency
A CPG may be considered up to date ―when (no) new information on interventions, outcomes, and performance justifies updating (it).
Guideline quality
By quality of clinical practice guidelines, we mean the confidence that the potential biases of guideline development addressed adequately and that the recommendations are both internally and externally valid, and are feasible for practice. This process involves taking into account the benefits, harms and costs of the recommendations, as well as the practical issues attached to them. Therefore, the assessment (of quality) includes judgments about the methods used for developing the guidelines, the content of the final recommendations, and the factors linked to their uptake.
Guideline topic
In theADAPTE Manual and Resource Toolkit for Guideline Adaptation' document, the topic refers to the theme of the guideline, as described in the guideline title, for a targeted population (disease and patients) and intervention. The purpose, the audience, and the setting intended for the guideline, although not necessarily explicitly stated in the title, are also part of the topic. A guideline on a given topic may contain more than one health question.
Health question or clinical question or key question
Is a precisely described health issue (e.g. clinical, professional practice or public health) relating to the topic of the guideline? Guideline may include one or more questions.
Implementation
Implementation includes methods to promote the uptake of research findings into routine healthcare in both clinical and policy contexts and hence to improve the quality and effectiveness of healthcare. It includes the study of influences on healthcare professional and organizational behavior.
Intra-class correlations
Intra-class correlations provide a measurement of the extent to which two or more raters agree when rating the same set of things. It is a reliability index and is typically a ratio of the variance of interest over the sum of the variance of interest plus error.
Recommendation
Any statement that promote or advocate a particular course of action in clinical care.
Stakeholder
A stakeholder is an individual, group and/or organization with a stake in your decision to implement a guideline. Stakeholders include individuals or groups who will be directly or indirectly affected by the implementation of a guideline.
Source guideline
In the ADAPTE Manual and Resource Toolkit for Guideline Adaptation' document, source guideline refer to those guidelines selected to undergo assessments of quality, currency, content, consistency, and acceptability/applicability and upon which an adapted guideline may be based.
- Introduction
Complementary feeding (CF), as defined by the World Health Organization (WHO) in 2002, is ‘‘the process starting when breast milk alone is no longer sufficient to meet the nutritional requirements of infants’’ so that ‘‘other foods and liquids are needed, along with breast milk’’ (1).
Adequate nutrition during infancy and early childhood is fundamental to the development of each child’s full human potential. It is well recognized that the period from birth to two years of age is a “critical window” for the promotion of optimal growth, health and behavioral development.
Longitudinal studies have consistently shown that this is the peak age for growth faltering, deficiencies of certain micronutrients, and common childhood illnesses such as diarrhea. After a child reaches 2 years of age, it is very difficult to reverse stunting that has occurred earlier (1).
In contrast to the large literature on breast and formula feeding, less attention has been paid to the CF period, especially to the type of foods given, or whether this period of significant dietary change influences later health, development, or behavior.
The more limited scientific evidence base is reflected in considerable variation in CF recommendations and practices between and within countries. (2).
Child growth, development and wellbeing are determined by the feeding practices and nutritional status of the child. Infant feeding practices in most of Egyptian surveys showed that the majority of infants are breastfed. Exclusive breast-feeding is common but not universal in early infancy. median duration of breastfeeding was 17 months Surveys showed that breast-feeding continued for the majority of children beyond the first year of life. The percentage of children aged 6-9 months who received both breast milk and solid food is higher in urban areas. In rural areas, mothers are more likely to initiate and continue breast-feeding than mothers in urban areas (3,4).
Less than one-quarter of children aged 6-23 months were being fed according to minimum Infant and Young Child Feeding standards for diet diversity and meal frequency. One in five Egyptian children under age 5 was stunted, 8 percent were wasted (thin for their height), and 6 percent were underweight (thin for their age). Fifteen percent of children were overweight (heavy for their age). Slightly more than one-quarter of children age 6-59 months were anemic (5)
Regarding micronutrient deficiencies, anaemia is considered the most prevalent. The groups most affected are preschool children and their mothers. The World Health Organization have estimated that 29.9% of preschool aged children in Egypt suffer from anemia (WHO 2014). Studies suggest the problem may be more widespread; a 2012 study of 300 infants at Ain Shams University Children’s Hospital identified an anaemia prevalence of 66%, of which 43% was iron deficiency anaemia. (6).
Similarly, vitamin A deficiency is also a challenge in Egypt with recent data from the WHO indicating that 11.9% of preschool children are vitamin A deficient. Vitamin D status is also a concern, with a recent study indicating that 60% and 32.6% of Egyptian neonates were vitamin D deficient and insufficient respectively (8) this was associated with low maternal vitamin D status and a consequence of low fish consumption and limited skin exposure. Cross-sectional studies suggest that poor vitamin D status of Egyptian infants persists into childhood and adolescence (7,8)
These health problems in Egyptian infants are multifactorial and may be due to lack of nutritional guidelines especially for introduction of complementary feeding, socioeconomic factors, cultural barriers and peer pressures.
There are currently no detailed guidelines for complementary feeding in Egypt. Consequently, current practice deviates greatly from international guidelines
The purpose of guideline is to review current recommendations and practice; summarize evidence for nutritional aspects and short-and long-term health effects of the timing and composition of CF; provide advice to health care providers for proper CF considering different aspects of CF with respect to developmental readiness, nutritional adequacy, and health effects; content, with respect to nutritional requirements and health effects; method of feeding; and specific dietary practices.
- Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the Introduction of complementary feeding in Egyptian Children.
It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to achieve Proper physical & neurodevelopmental outcome of infants, prevent malnutrition, under nutrition and over nutrition and prevent micronutrient deficiencies and its comorbidities.
This version of the guideline includes recommendations and good practice statements for proper introduction of solid food for healthy full term infants of both sexes, either breastfed or non - breastfed without co-morbidities.
- Methods
➡️ Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Keywords used for search are: Infant feeding, weaning, complementary feeding.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1.CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria the GDG/ GAG recommended using 4 guidelines:
1- WHO guideline ” Guiding Principles For Complementary Feeding of the Breastfed Child”(2003).
2- ESPGAN position paper “Complementary Feeding: A Position Paper by the European Society for Paediatric Gastroenterology,Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition (2017).
3- American Academy of Pediatrics Committee on Nutrition. Complementary feeding. In: Pediatric Nutrition, 7th ed, Kleinman RE, Greer FR (Eds), American Academy of Pediatrics, Elk Grove Village, IL 2014. p.123.
4- Guideline: Daily iron supplementation in infants and children. Geneva: World Health Organization; 2016.
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
➡️Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of Infant and Young Child Feeding
The main functions of the clinical panel were adolopment of Introduction of complementary feeding in Egyptian Children Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 3 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 5 clinical national experts who have interest and expertise in as well as eminent international reviewers.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that include the following definitions [12]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations. (both ETD and changing strength of recommendation were not done in this guideline).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5.Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include awell-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- research needs
Future research recommendations for Infant and Young Child Feeding in the Egyptian context could include:
· Iron supplementation (timing & dose) among infants whom receive breastfeeding & formula feeding
· Family education about importance of introducing all kind of macro & micronutrients to the infants.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for Infant and Young Child Feeding.
➡️Challenges:
Family education about alternative types of high biological value protein.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Updating of the guideline
The EPG Infant and Young Child Feeding GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. WHO (World Health Organization). 2003. Complementary Feeding. Report of the Global Consultation. Geneva, 10–13 December 2001. Summary of Guiding Principles. http://www.who.int/nutrition/ publications/Complementary_Feeding.pdf. Accessed april 11,2018..
2. Fewtrell, Mary; Bronsky, Jiri; Campoy, Cristina; Domellöf, Magnus; Embleton, Nicholas; Fidler Mis, Nataša; Hojsak, Iva; Hulst, Jessie M; Indrio, Flavia; Lapillonne, Alexandre; Molgaard, Christian. Complementary Feeding: A Position Paper by the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition. Journal of Pediatric Gastroenterology and Nutrition. 64(4):653, April 2017.
3. EDHS (Egypt Demographic and Health Survey), 2014
4. EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific opinion on the appropriate age for introduction of complementary feeding of infants. EFSA J 2009; 7:1423.
5. World Health Organization (2014) Global Targets 2025. To Improve Maternal, Infant and Young Child Nutrition [Online]. Available at: www.who.int/nutrition/topics/nutrition_globaltargets2025/en/
6. Justine A. Kavle, Valerie L. Flax, Ali Abdelmegeid, Farouk Salah, Seham Hafez, Magda Ramzy, Doaa Hamed, Gulsen Saleh, and Rae Galloway. Factors associated with early growth in Egyptian infants: implications for addressing the dual burden of malnutrition. Matern Child Nutr. 2016 Jan; 12(1): 139–151.
7. Elalfy, M. S., A. M. Hamdy, S. S. Maksoud and R. I. Megeed (2012). "Pattern of Milk Feeding and Family Size as Risk Factors for Iron Deficiency Anemia among Poor Egyptian Infants 6 to 24 Months Old." Nutrition research 32(2): 93-99.
8. El Rifai, N. M., G. A. Abdel Moety, H. M. Gaafar and D. A. Hamed (2013). "Vitamin D Deficiency in Egyptian Mothers and Their Neonates and Possible Related Factors." The journal of maternal-fetal & neonatal medicine : the official journal of the European Association of Perinatal Medicine, the Federation of Asia and Oceania Perinatal Societies, the International Society of Perinatal Obstetricians.
9. Amr, N., A. Hamid, M. Sheta and H. Elsedfy (2012). "Vitamin D Status in Healthy Egyptian Adolescent Girls." Georgian medical news(210): 65-71.
10. Mansour, M. M. and K. M. Alhadidi (2012). "Vitamin D Deficiency in Children Living in Jeddah, Saudi Arabia." Indian journal of endocrinology and metabolism 16(2): 263-269.
11. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
12. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
13. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
14. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
15. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
16. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
17. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
18. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
19. Aly, H., Said, R.N., Wali, I.E., Elwakkad, A., Soliman, Y., Awad, A.R., Shawky, M.A., Alam, M.S.A. and Mohamed, M.A., 2017. Medically Graded Honey Supplementation Formula to Preterm Infants as a Prebiotic: A Randomized Controlled Trial. Journal of pediatric gastroenterology and nutrition, 64(6), pp.966-970.
20. Abdulrhman, M.A., Hamed, A.A., Mohamed, S.A. and Hassanen, N.A.A., 2016. Effect of honey on febrile neutropenia in children with acute lymphoblastic leukemia: A randomized crossover open-labeled study. Complementary therapies in medicine, 25, pp.98-103.
21. Shaaban, S.Y., Abdulrhman, M.A., Nassar, M.F. and Fathy, R.A., 2010. Effect of honey on gastric emptying of infants with protein energy malnutrition. European journal of clinical investigation, 40(5), pp.383-387.
22. Ahmed, S.H., Badary, M.S., Mohamed, W.A. and Elkhawaga, A.A., 2011. Multiplex PCR for detection and genotyping of C. botulinum types A, B, E and F neurotoxin genes in some Egyptian food products. J. Am. Sci, 7, pp.176-190.
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Sanaa Youssef Shaaban |
Professor of Pediatrics, Ain shams University |
None |
Not Applicable |
|
Prof. Mourad Alfy Ramzy Tadros |
Professor of Pediatrics, Armed forced faculty of medicine. |
None |
Not Applicable |
|
Prof. Hanna Aboulghar |
Professor of Pediatrics, Cairo University |
None |
Not Applicable |
|
Prof. Ehab Khairy El khashab |
Professor of Pediatrics, Ain Shams University |
None |
Not Applicable |
|
Prof. Mahmoud Rashad |
Professor of Pediatrics, Al-Azhar University |
None |
Not Applicable |
|
Prof. Somaya Mohamed Abd El -Ghany |
Professor of Pediatrics, Al-Azhar University Faculty of Medicine for Girls |
None |
Not Applicable |
|
Prof. Tayseer Mohamed El Zayat |
Professor of Pediatrics, Al-Azhar University Faculty of Medicine for Girls |
None |
Not Applicable |
|
Prof. Osama M Al-Asheer |
Professor of Pediatrics, Assuit University |
None |
Not Applicable |
|
Prof. Enas Raafat |
Professor of child health, National Research Center |
None |
Not Applicable |
|
Prof. Afaf Abdel Fattah Tawfik |
Previous dean of National Nutrition Institute |
None |
Not Applicable |
|
Prof. Yasmin Gamal El Gendy |
Professor of Pediatrics and Clinical Nutrition, AinShams University, AFCM |
None |
Not Applicable |
|
Dr. Shrouk MoatazAbdallah |
Lecturer of Pediatrics Cairo University |
None |
Not Applicable |
|
Dr. Asmaa Sadek A. Sadek |
Pediatric & neonatology consultant |
None |
Not Applicable |
|
Dr. Ayah Shabana |
Lecturer of Pediatrics, Suez University |
None |
Not Applicable |
|
Dr. Nahed Mohamed hamdy |
Pediatric & neonatology consultant |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Dr.Abla AL Alfy |
Regional Advisor for Middle East and Africa- Royal College of Paediatrics and Child Health (RCPCH) Head of Egyptian Members Assoc- RCPCH Co-Chair –Scientific Council-Military Paediatric Board IBCLC |
None |
Not Applicable |
|
Dr. Ahmed El Saeed Younis |
Head of Egyptian Paediatric Society |
None |
Not Applicable |
|
Dr. Aliaa Hafiz |
Head of nutrition unit ,world food program IBCLC |
None |
Not Applicable |
|
Dr. Mona Hafez El Naka- |
Ministry of Health IBCLC |
None |
Not Applicable |
|
Dr. Nahla Roshdy |
Ministry of Health IBCLC |
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
None |
Not Applicable |
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for Infant and Young Child Feeding in the Egyptian context. " on PubMed are: infant feeding, weaning, complementary feeding”.
Annex Table 3. Annex Nurses and Parents Educational Guide in Arabic


Appendix
Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|