Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 29 Oct 2025" Download Guideline
- Recommendations
Table 3. Recommendations |
|
| |||
A. History taking |
|
| |||
N | Health question | Source Guideline | Recommendations | Quality of evidence | Strength of Recommendation |
A1 | In infants and children of both sexes, aged from 1 month to 18 years who presents with acute seizure, what is the added value of history taking? And what items should be fulfilled? | Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012)
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012)
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 | The first stage in clinical management is to recognize if abnormal movement or behavior has an epileptic origin
Ask about prior history of epilepsy
AED, AED non-compliance, or known allergies.
Address history of Ingestion of a toxin or drug abuse as possible etiologies of SE. | Moderate
Moderate
Low
Low
| Strong
Strong
Weak (conditional)
Weak (conditional)
|
|
|
|
||||
|
B. Clinical examination |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
In infants and children of both sexes, aged from 1 month to 18 years who presents with suspected acute seizure, what is the added value of vital data recording in diagnosis of etiology, and directing management decisions? |
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
Rule out febrile seizures
Assessment of vital signs: O2 saturation, BP, HR in the immediate (0–2 min) to establish and support baseline vital signs |
Moderate
Very low |
Strong
Weak (conditional)
|
|
B2 |
In infants and children of both sexes, aged from 1 month to 18 years who presents with suspected acute seizures and managed, what is the added value of targeted initial clinical neurologic examination in diagnosis of etiology, and directing management decisions? |
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012)
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
Neurologic exam is urgent (5–10 min) to evaluate for mass lesion, acute intracranial process
Address respiratory monitoring to prevent cardiopulmonary arrest, during anticonvulsant drug treatment in status epilepticus in children. |
Very low
High |
Weak (conditional)
Strong |
|
Table 6. Recommendations |
|
|
|||
|
C. Laboratory Evaluation |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
C1 |
In Infants and children aged 1 month to 18 years of both sexes, who are diagnosed as having acute seizure, regarding the use of laboratory investigations, in diagnostic evaluation, what are the routine laboratory investigations have to be done in all patients? Is serum AED level helpful to improve diagnosis of acute seizures? |
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
I. Laboratory tests should be ordered based on individual clinical circumstances that include suggestive historic or clinical findings such as vomiting, diarrhea, dehydration, or failure to return to baseline alertness.
II. For all patients, order laboratory tests: blood glucose, complete blood count, calcium (total and ionized), magnesium, AED levels. |
Very low
Very low |
Weak (conditional)
Weak (conditional)
|
|
C2 |
In Infants and children aged 1 month to 18 years of both sexes, who are diagnosed as having acute seizure, regarding the use of laboratory investigations, in diagnostic evaluation, is LP analysis helpful to improve diagnosis of acute seizures? Is brain imaging mandatory before it? Are arterial/ venous blood gases, liver function tests, genetic testing, helpful to improve diagnosis of acute seizures?
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012
|
III. Consider based on clinical presentation a) Lumbar puncture (LP). Should be performed in the very young child (<6 months), in the child of any age with persistent (cause unknown) alteration of mental status or failure to return to baseline, or in any child with meningeal signs. If increased intracranial pressure is suspected, the LP should be preceded by an imaging study of the head
b) Comprehensive toxicology panel including toxins that frequently cause seizures (i.e. isoniazid, tricyclic antidepressants, theophylline, cocaine, sympathomimetics, alcohol, organophosphates, and cyclosporine Other laboratory tests: liver function tests, coagulation studies, arterial blood gases, and inborn errors of metabolism |
Very low
Very low |
Weak (conditional)
Weak (conditional)
|
|
Table 7. Recommendations |
|
|
|||
|
D. EEG |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
D1 |
In Infants and children aged 1 month to 18 years of both sexes, who are diagnosed as having acute seizures, regarding the use of EEG in diagnostic evaluation, is standardized EEG helpful to improve diagnosis of acute convulsive seizure? |
Evidence-Based Guidelines for EEG Utilization at the University Teaching Hospital (UTH) – 2011
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012 |
I. In the PICU, An EEG is indicated in all unconscious patients suspected of non-convulsive status epilepticus or subclinical seizures. This includes comatose or obtunded inpatients of unclear etiology especially those in whom seizures preceded the onset of coma. II. EEG is recommended after a first unprovoked seizure as it might offer insights into recurrence risk and/or the need for further neuroimaging.
III. In the treatment of convulsive status epilepticus in children, continuous EEG monitoring is indicated if second therapy fails to stop seizures, and treatment consideration involves the use of anesthetic doses of either thiopental, midazolam, pentobarbital, or propofol
IV. The use of continuous EEG (cEEG) is usually required for the treatment of SE. V. Continuous EEG monitoring should be initiated within 1 h of SE onset if ongoing seizures are suspected VI. The duration of cEEG monitoring should be at least 48 h in comatose patients to evaluate for non-convulsive seizures.
VII. The use of this cEEG is encouraged to be available at tertiary care facilities where the optimum standard of care is provided. |
Very low
Very low
Very low
|
Weak (conditional)
Weak (conditional)
Weak (conditional)
Good practice statement |
|
Table 8. Recommendations |
|
|
|||
|
E. Neuroimaging (CT / MRI Brain) |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
E1 |
In infants and children of both sexes, aged from 1 month to 18 years who presents with acute seizures, whilst the primary assessment and resuscitation are being carried out, Regarding the added value of neuroimaging, what is the role of emergency head CT or MRI in improving diagnosis of acute convulsive seizures? |
Diagnosis and Treatment of the First Epileptic Seizure: Guidelines of the Italian League Against Epilepsy, 2006
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
I. The use of a brain CT scan or MRI in the emergency room is indicated when specific interventions may be needed. II. A CT scan is strictly indicated when a structural lesion is suspected or when the etiology of the seizure cannot be easily identified. Structural lesions include, among others, post-traumatic complications, cerebral hemorrhage, brain edema, and space-occupying lesions, which may be suggested by post-ictal deficits and/or persisting impairment of consciousness. III. In the emergency room, MRI is not indicated, except for selected circumstances, to be evaluated on an individual basis.
IV. The etiology of SE should be diagnosed as soon as possible and occur simultaneously and in parallel with treatment for evaluation for mass lesions, meningitis, encephalitis.
V. All patients with SE should have Head computed tomography (CT) scan
VI. Consider Brain magnetic resonance imaging (MRI)based on clinical presentation |
Moderate
Moderate
|
Strong
Strong
Good practice statement
Good practice statement
|
|
Table 9. Recommendations |
|
|
|||
|
F. Therapies: Initial Therapy: 0 – 20 minutes: 1. Benzodiazepines |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
F1 |
In Infants and children aged 1 month to 18 years of both sexes, who are diagnosed as having acute seizure, what is the role of benzodiazepines as an initial therapy in pre-hospital\hospital setting? |
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
|
Benzodiazepines are the most efficient group of drugs to be initially used in the treatment of acute convulsion after 5 minutes from the start of seizures
I. Pre-hospital Treatment: As early as possible by Trained personal whether medical\Para medical or caretaker.
• Drugs of Choice: -Midazolam (intra-muscular – buccal – nasal) -Diazepam (rectal) Only one dose, not to be repeated
II. Hospital Emergency Room: • History of pre-hospital use of benzodiazepines must be considered as well as timing. • Doses given within one hour before ER presentation must be regarded as an initial dose
O Midazolam • It is the most appropriate type of benzodiazepines to be used. • In the hospital the whole full dose to be given as a single dose better than broken into multiple doses.
O Diazepam IV diazepam could be repeated after 5 minutes convulsion continued
• Alternatively IF VENOUS ACCESS UNAVAILABLE; • Use midazolam: buccal or intranasal. • Use diazepam: rectal • Follow up for signs of respiratory depression as hypoventilation, pallor, decrease oxygen saturation Respiratory support and intubation may be needed |
High
Moderate
Moderate
High
High
Moderate
High |
Strong
Strong
Strong
Strong
Strong
Strong
Strong |
|
Table 10. Recommendations |
|
|
|||
|
G. Therapies: Initial Therapy: 0 – 20 minutes: 2. Non Benzodiazepine Therapy 2.1 Phenytoin and Phenobarbitone: First Phase therapy : 5 - 20 minutes |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
G1 |
In Infants and children aged 1 month to 18 years of both sexes, who are diagnosed as having acute seizure, what is the role of Phenobarbitone and phenytoin, as an initial and second line therapy? |
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012)
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012). Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013. Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
|
Both phenytoin and phenobarbital are recommended
Phenytoin followed by phenobarbital is recommended if benzodiazepines fail
Phenobarbital is recommended in the absence of benzodiazepines
Second phase therapy: 20 - 40 minutes
• Phenytoin and Phenobarbital are recommended
• They are not used sequentially, but alternating. Generally, phenytoin precedes phenobarbital on account of a better safety profile |
High
Moderate
High
Moderate
Moderate |
Strong
Strong
Strong
Strong
Strong
|
|
Table 11. Recommendations |
|
|
|||
|
2.2 Valproate |
|
|
|||
|
N |
Health question |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
H1 |
In infants and children of both sexes, aged from 1 month to 18 years who are diagnosed as having acute seizure, regarding the use of sodium valproate in the treatment, what is the role of sodium valproate, as a third line therapy? |
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: (AES guidelines.
Epilepsy Currents, Vol. 16, No. 1 (January/February) 2016 pp. 48–61:
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· IV Sodium valproate is an option in the second-therapy phase and should begin when the seizure duration reaches 20 minutes.
· The use of IV Sodium valproate is dependent on its availability especially in tertiary care centers where optimum care is provided.
Valproic acid offers a valid alternative to phenytoin and phenobarbital. It should be used with extreme caution, however, particularly in young children, when a possible metabolic etiology has not been ruled out. |
Moderate
Moderate
|
Strong
Good practice statement
Strong |
|
Table 12. Recommendations |
|
|
|||
|
2.3 Pyridoxine |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
I1 |
In infants and children of both sexes, aged from 1 month to 18 years who are diagnosed as having acute seizures and transferred to PICU with convulsive status epilepticus not adequately responding to first, second and third line medications, what is the role of levetiracetam? |
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012)
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
Consider vitamin-dependent diseases (pyridoxine or pyridoxal-phosphate) in the early myoclonic epilepsy group
Young children with epilepsy (under 3 years) who develop SE lasting 30 minutes, especially if myoclonic should receive pyridoxine trial in case they have pyridoxine dependent/responsive seizures.
IV Levetiracetam or oral through a nasogastric tube could be given in the second phase therapy for SE
This drug can be used for continued oral therapy, does not cause significant side effects, and needs no preliminary check of renal function. |
Low
Very low
Very low
|
Good practice statement
Weak (conditional)
Weak (conditional)
Weak (conditional)
|
- Executive Summary
A seizure is a transient occurrence of signs &/or symptoms due to abnormal excessive or synchronous neuronal activity of the brain. Brief seizures are defined as seizures lasting less than 5 minutes, while prolonged seizures last between 5 & 30 minutes. A seizure that is provoked by a transient factor acting on an otherwise normal brain that temporarily lower the seizure threshold is not diagnosed as epilepsy (reactive seizure or acute symptomatic seizure). Epilepsy is a disease of the brain defined by any of the following conditions:
1. At least two unprovoked (or reflex) seizures occurring > 24 hours apart.
2. One unprovoked (or reflex) seizure & a probability of further seizures similar to the general recurrence risk (at least 60%)
3. Diagnosis of an epilepsy syndrome.
Status epilepticus is defined as more than 30 minutes of either, continuous seizure activity or two or more sequential seizures without full recovery of consciousness between them. The 30 - Minute’s definition is based on the duration of convulsive status epilepticus that may lead to permanent neuronal injury by itself. Since the majority of seizures are brief, and once a seizure lasts more than 5 minutes it is likely to be prolonged. Continuous Generalized convulsive SE in adults and children older than 5 years was operationally defined as 5 min or more clinical and/or electrographic seizure activity or (ii) recurrent seizure activity without recovery (returning to baseline) between seizures. Status treatment protocols have used a 5 - minute definition to minimize both the risk of seizures reaching 30 minutes and the adverse outcomes associated with needlessly intervening on brief, self-limited seizures.
This guideline focuses on prevention and management of seizures in children in Egypt and the prevention of their complications.
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1- Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society. American Epilepsy Society Guideline (2016).
2- Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics. International League Against Epilepsy (2015).
3- Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy. ITALIAN LEAGUE (LICE) (2013).
4- Guidelines for the Evaluation and Management of Status Epilepticus Neurocritical Care Society Status Epilepticus Guideline (2012).
5- Evidence-Based Guidelines for EEG Utilization at the University Teaching Hospital (UTH). Neurologic & Psychiatric Society of Zambia (2011).
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following:
A. Diagnosis of Acute epileptic seizures in children.
The guideline covers children beyond the neonatal period (from 1-month to 18-years of age).
This guideline emphasis on diagnosis of children having convulsive (motor) seizures of any type and including serial fits, seizure clustering, and status epilepticus
- Excluding the underlying etiology and co-morbid conditions
B. Management of Acute epileptic seizures in children beyond the neonatal period (from 1-month to 18-years of age)
This section includes recommendations and good practice statements on treatment of acute epileptic seizures among different healthcare professionals in different healthcare settings. Also to provide healthcare professionals with easy-to-apply acute treatment protocol of epileptic seizures in children that will allow immediate and appropriate seizure control and prevention of complications.
C. Prevention of acute seizures attacks & secondary prevention of:
- Injury - Recurrence - Development of status
- Aspiration - vascular insult - Cardiac arrhythmia
- SUEDP
We can summarize the guidelines’ recommendations for management of acute epileptic seizures in children beyond the neonatal period (from 1-month to 18-years of age) in the following:
· We recommend that the first stage in clinical management is to recognize if abnormal movement or behavior has an epileptic origin (strong recommendation).
· We recommend ruling out febrile seizures (strong recommendation).
· We recommend asking about prior history of epilepsy (strong recommendation).
· We suggest asking about AED non-compliance, or known allergies (weak (conditional) recommendation).
· We suggest to address history of Ingestion of a toxin or drug abuse as possible etiologies of SE (weak (conditional) recommendation).
· We suggest assessment of vital signs: O2 saturation, BP, HR in the immediate (0–2 min) to establish and support baseline vital signs (weak (conditional) recommendation).
· We suggest that neurologic exam is urgent (5–10 min) to evaluate for mass lesion, acute intracranial process (weak (conditional) recommendation).
· We recommend to address respiratory monitoring to prevent cardiopulmonary arrest, during anticonvulsant drug treatment in status epilepticus in children (strong recommendation).
· We suggest that laboratory tests should be ordered based on individual clinical circumstances that include suggestive historic or clinical findings such as vomiting, diarrhea, dehydration, or failure to return to baseline alertness (weak (conditional) recommendation).
· We suggest for all patients, order laboratory tests: blood glucose, complete blood count, calcium (total and ionized), magnesium, AED levels (weak (conditional) recommendation).
· We suggest to consider based on clinical presentation, Lumbar puncture (LP) :
- Should be performed in the very young child (<6 months), in the child of any age with persistent (cause unknown) alteration of mental status or failure to return to baseline, or in any child with meningeal signs. If increased intracranial pressure is suspected, the LP should be preceded by an imaging study of the head (weak (conditional) recommendation).
-Comprehensive toxicology panel including toxins that frequently cause seizures (i.e. isoniazid, tricyclic antidepressants, theophylline, cocaine, sympathomimetics, alcohol, organophosphates, and cyclosporine. Other laboratory tests: liver function tests, coagulation studies, arterial blood gases, and inborn errors of metabolism (weak (conditional) recommendation).
· In the PICU we suggest that EEG is indicated in all unconscious patients suspected of non-convulsive status epilepticus or subclinical seizures. This includes comatose or obtunded inpatients of unclear etiology especially those in whom seizures preceded the onset of coma
EEG is recommended after a first unprovoked seizure as it might offer insights into recurrence risk and/or the need for further neuroimaging (weak (conditional) recommendation).
· In the treatment of convulsive status epilepticus in children, we suggest that continuous EEG monitoring is indicated if second therapy fails to stop seizures, and treatment consideration involves the use of anesthetic doses of either thiopental, midazolam, pentobarbital, or propofol (weak (conditional) recommendation).
· We suggest that the use of continuous EEG (cEEG) is usually required for the treatment of SE & that continuous EEG monitoring should be initiated within 1 h of SE onset if ongoing seizures are suspected. The duration of cEEG monitoring should be at least 48 h in comatose patients to evaluate for non-convulsive seizures (weak (conditional) recommendation).
· We suggest that the use of this cEEG is encouraged to be available at tertiary care facilities where the optimum standard of care is provided (Good practice statement).
· We recommend that the use of a brain CT scan or MRI in the emergency room is indicated when specific interventions may be needed.
A CT scan is strictly indicated when a structural lesion is suspected or when the etiology of the seizure cannot be easily identified. Structural lesions include, among others, post-traumatic complications, cerebral hemorrhage, brain edema, and space-occupying lesions, which may be suggested by post-ictal deficits and/or persisting impairment of consciousness.
In the emergency room, MRI is not indicated, except for selected circumstances, to be evaluated on an individual basis (strong recommendation).
· We recommend that the etiology of SE should be diagnosed as soon as possible and occur simultaneously and in parallel with treatment for evaluation for mass lesions, meningitis, encephalitis (strong recommendation).
· We suggest that all patients with SE should have Head computed tomography (CT) scan (good practice statement).
· We suggest considering Brain magnetic resonance imaging (MRI)based on clinical presentation (Good practice statement).
· We recommend that Benzodiazepines are the most efficient group of drugs to be initially used in the treatment of acute convulsion after 5 minutes from the start of seizures (strong recommendation).
· Pre-hospital Treatment:
As early as possible by Trained personal whether medical\Para medical or caretaker. We recommend that drugs of Choice are:
Midazolam (intra-muscular – buccal – nasal)
Diazepam (rectal)
(Only one dose, not to be repeated) (strong recommendation).
· Hospital Emergency Room:
We recommend that history of pre-hospital use of benzodiazepines must be considered as well as timing.
Doses given within one hour before ER presentation must be regarded as an initial dose
- Midazolam:
It is the most appropriate type of benzodiazepines to be used.
In the hospital the whole full dose to be given as a single dose better than broken into multiple doses (strong recommendation).
- Diazepam:
IV diazepam could be repeated after 5 minutes convulsion continued (strong recommendation).
- Alternatively:
IF VENOUS ACCESS UNAVAILABLE;
Use midazolam: buccal or intranasal.
Use diazepam: rectal
Follow up for signs of respiratory depression as hypoventilation, pallor, decrease oxygen saturation
Respiratory support and intubation may be needed (strong recommendation).
· - Non Benzodiazepine Therapy: we recommend
- Phenytoin and Phenobarbitone
First Phase therapy: 5 - 20 minutes
- Both phenytoin and phenobarbital are recommended (strong recommendation).
- Phenytoin followed by phenobarbital is recommended if benzodiazepines fail (strong recommendation).
- Phenobarbital is recommended in the absence of benzodiazepines (strong recommendation).
Second phase therapy: 20 - 40 minutes
- Phenytoin and Phenobarbital are recommended (strong recommendation).
- They are not used sequentially but alternating. Generally, phenytoin precedes phenobarbital on account of a better safety profile (strong recommendation).
- Valproate
- IV Sodium valproate is an option in the second-therapy phase and should begin when the seizure duration reaches 20 minutes (strong recommendation).
- The use of IV Sodium valproate is dependent on its availability especially in tertiary care centers where optimum care is provided (Good practice statement).
- Valproic acid offers a valid alternative to phenytoin and phenobarbital. It should be used with extreme caution, however, particularly in young children, when a possible metabolic etiology has not been ruled out (strong recommendation).
- Pyridoxine
- Consider vitamin-dependent diseases (pyridoxine or pyridoxal-phosphate) in the early myoclonic epilepsy group (Good practice statement).
- Young children with epilepsy (under 3 years) who develop SE lasting 30 minutes, especially if myoclonic should receive pyridoxine trial in case they have pyridoxine dependent/responsive seizures (weak (conditional) recommendation).
- Levetiracetam
- IV Levetiracetam or oral through a nasogastric tube could be given in the second phase therapy for SE (weak (conditional) recommendation).
- This drug can be used for continued oral therapy, does not cause significant side effects, and needs no preliminary check of renal function (weak (conditional) recommendation).
Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/
- Acknowledgements
· The GDG/ GAG acknowledge EPG for its help in completing this project.
· We acknowledge the following guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
1. Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society. American Epilepsy Society Guideline (2016)
2. Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics. International League Against Epilepsy (2015)
3. Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy. ITALIAN LEAGUE (LICE) (2013)
4. Guidelines for the Evaluation and Management of Status Epilepticus Neurocritical Care Society Status Epilepticus Guideline (2012)
5. Evidence-Based Guidelines for EEG Utilization at the University Teaching Hospital (UTH). Neurologic & Psychiatric Society of Zambia (2011).
· Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
· This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Abdelsatr Abdallah Elsayeh |
Assistant professor of Pediatrics and Pediatric Neurology Faculty of Medicine for Males Al-Azhar University |
Clinical expert |
|||
|
Ashraf Azmy Al Refaei |
Professor of Child Health and Pediatric Neurology National Research Center |
Clinical expert |
|||
|
Azza Mohamed Elamin Oraby (late professor) |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Cairo University |
Clinical expert |
|||
|
Ebtesam Hussein El Melegy |
Consultant Pediatric Neurologist Former Dean of the Neuromotor Institute Ministry of Health |
Clinical expert |
|||
|
Hammouda Eid El Gazzar |
Fellow and Lecturer in Pediatrics General Organization of Teaching Hospitals Under-secretary of El Beheira Health Directorate |
Clinical expert |
|||
|
Hayam Refaat Tantawi |
Professor of Pediatric Nursing Vice Dean of Community and Environmental Development Faculty of Nursing Ain Shams University |
Clinical expert |
|||
|
Lobna Abdel Gawad Mansour |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Cairo University |
Clinical expert |
|||
|
Marwa Saeed Abd Elmaksod |
Assistant professor of Pediatrics and Pediatric Neurology Faculty of Medicine Alexandria University |
Clinical expert |
|||
|
Moustafa Zakaryia Mohamed |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Cairo University |
Clinical expert |
|||
|
Omnia Fathy El Rashidy
|
Professor of Pediatrics and Pediatric Neurology and Neuro-disability Faculty of Medicine Ain Shams University |
Clinical expert |
|||
|
Sahar Mohamed Ahmed Hassanein |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Ain Shams University |
Clinical expert |
|||
|
Tarek El-Sayed Ismail Omar |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Alexandria University |
Supervisor, Clinical expert |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Professor Hadeer Mahmoud Gamal Eldin Abdelghaffar |
Professor and Head of Pediatrics Department – Consultant Pediatric Neurologist – Faculty of Medicine - Fayoum University |
||||
|
Professor Hoda Yehia Tomoum |
Professor of Pediatrics and Pediatric Neurology – Faculty of Medicine – Ein Ain Shams University |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AED |
Anti-epileptic Drug |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
CNS |
Central Nervous System |
||
|
CPG |
Clinical Practice Guideline |
||
|
CPK |
Creatine Phosphokinase |
||
|
CSE |
Convulsive Status Epilepticus |
||
|
CT |
Computed Tomography |
||
|
DHS |
Demographic and Health Survey |
||
|
ED |
Emergency Department |
||
|
EEG |
Electroencephalogram |
||
|
ER |
Emergency Room |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ERG |
External Review Group |
||
|
FIRES |
Febrile-illness Related Epilepsy Syndrome |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
GTC |
Generalized Tonic-Clonic |
||
|
ILAE |
International League Against Epilepsy |
||
|
IM |
Intra-muscular |
||
|
LP |
Lumbar Puncture |
||
|
MRI |
Magnetic Resonance Imaging |
||
|
NCSE |
Non-convulsive Status Epilepticus |
||
|
NORSE |
New Onset Refractory Status Epilepticus |
||
|
NPCPGC |
National Pediatric Clinical Practice Guideline Committee |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
PICU |
Pediatric Intensive Care Unit |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
||
|
PNES |
Psychogenic Non-epileptic Seizures |
||
|
SE |
Status Epilepticus |
||
|
SUDEP |
Sudden Unexpected Death in Epilepsy |
||
|
WHO |
World Health Organization |
- Glossary
Acceptability
Is the extent to which the users are likely to adopt a recommendation, based on internal qualities such as clarity, comprehensiveness, and logical reasoning and on external factors such as the burden imposed on the process and system of care, patient and providers attitudes and beliefs, and patient’s needs, expectations, and preferences.
Adaptation (of guidelines)
Is the systematic approach to considering the use and/or modification of (a) guidelines(s) produced in one cultural and organizational setting for application in a different context? Adaptation can be used as an alternative to de novo guideline development or for customizing (an) existing guideline(s) to suit the local context.
Admission
Admission, for the purpose of this guideline, refers to a child being registered and entering inpatient care as a patient. This is distinguished from the term “enrolment”, which is used for outpatient care.
Adoption (of a guideline)
Is the acceptance of a guideline as a whole after the assessment of its quality, currency, and content? When health care providers (or other users of recommendations) adopt a guideline, they feel committed to change their practices in accordance with the recommendations of the guideline.
Applicability
Is the extent to which the users are able to put a recommendation into practice, based on internal qualities such as a clearly defined eligible patient population that matches the population to which the intervention is targeted in the local setting and external factors such as the availability of the necessary knowledge, skills, provider time, staff, equipment, and other resources.
Applicability is sometimes taken as a synonym for feasibility:
Feasibility of the acquisition of necessary skills and knowledge
Feasibility of the necessary increase in provider time, staff, equipment, and so on.
Culture
Culture represents the norms and values of a specific group, community, or population.
Diffusion
It is a passive means of transferring knowledge; it is not directed towards a target audience (e.g. publication of articles in medical journals).
Dissemination
It is more active than diffusion in that it targets a specific audience and involves tailoring the information for that audience (e.g. of dissemination strategies include targeted mailings, presentations, and press conferences.
Evidence-based principles
Evidence-Based Medicine (EBM) has been defined as ― the conscientious, explicit, and judicious use of current best evidence in making decisions about the care of individual patients. The practice of EBM means integrating individual clinical expertise with the best available external clinical evidence from systematic research.
Evidence tables
Are summaries of the most salient information from studies identified in the systematic review? The elements of evidence tables are dependent on the types of information in studies related to a particular topic but might include information such as the article reference, the study type (e.g. RCT or Cohort), the number of patients and their characteristics, and the intervention, comparison arms, outcome measures, and effect sizes.
Guideline or Clinical Practice Guideline (CPG)
Systematically developed statements about specific health problems, intended to assist practitioners and patients in making decisions about appropriate health care.
Guideline consistency
Agreement between the evidence and the recommendations, based on the:
Comprehensiveness of the study search and selection process,
Coherence between the results of the studies and their interpretation by the guideline authors, and
Transparency between interpretation and recommendations.
Guideline content
In the ‘ADAPTE Manual and Resource Toolkit for Guideline Adaptation’ document, guideline content refers to the recommendations in the source guidelines.
Guideline currency
A CPG may be considered up to date ―when (no) new information on interventions, outcomes, and performance justifies updating (it).
Guideline quality
By the quality of clinical practice guidelines, we mean the confidence that the potential biases of guideline development addressed adequately and that the recommendations are both internally and externally valid, and are feasible for practice. This process involves taking into account the benefits, harms, and costs of the recommendations, as well as the practical issues attached to them. Therefore, the assessment (of quality) includes judgments about the methods used for developing the guidelines, the content of the final recommendations, and the factors linked to their uptake.
Guideline topic
In the ADAPTE Manual and Resource Toolkit for Guideline Adaptation' document, the topic refers to the theme of the guideline, as described in the guideline title, for a targeted population (disease and patients) and intervention. The purpose, the audience, and the setting intended for the guideline, although not necessarily explicitly stated in the title, are also part of the topic. A guideline on a given topic may contain more than one health question.
Health question or clinical question or key question
Is a precisely described health issue (e.g. clinical, professional practice, or public health) relating to the topic of the guideline? Guideline The guideline may include one or more questions.
Implementation
Implementation includes methods to promote the uptake of research findings into routine healthcare in both clinical and policy contexts and hence to improve the quality and effectiveness of healthcare. It includes the study of influences on healthcare professional and organizational behavior.
Intraclass correlations
Intraclass correlations provide a measurement of the extent to which two or more raters agree when rating the same set of things. It is a reliability index and is typically a ratio of the variance of interest over the sum of the variance of interest plus error.
Recommendation
Any statement that promotes or advocates a particular course of action in clinical care.
Stakeholder
A stakeholder is an individual, group, and/or organization with a stake in your decision to implement a guideline. Stakeholders include individuals or groups who will be directly or indirectly affected by the implementation of a guideline.
Source guideline
In the ADAPTE Manual and Resource Toolkit for Guideline Adaptation' document, source guidelines refer to those guidelines selected to undergo assessments of quality, currency, content, consistency, and acceptability/applicability and upon which an adapted guideline may be based.
- Introduction
DEFINITIONS
A seizure is a transient occurrence of signs &/or symptoms due to abnormal excessive or synchronous neuronal activity of the brain (1). Brief seizures are defined as seizures lasting less than 5 minutes, while prolonged seizures last between 5 & 30 minutes (2). A seizure that is provoked by a transient factor acting on an otherwise normal brain that temporarily lower the seizure threshold is not diagnosed as epilepsy (reactive seizure or acute symptomatic seizure) (3).
The term unprovoked seizure implies to the absence of a temporary or reversible factor lowering the threshold and producing a seizure at that point in time then. The condition of recurrent reflex seizures in response to photic stimuli represents provoked seizures that are defined as epilepsy. Even though the seizures are provoked, the tendency to respond repeatedly to such stimuli with seizures meets the conceptual definition of epilepsy (4).
Febrile seizures are seizure occurring in children between 6 months and 5 years of age in association with a fever greater than 38 C , who do not have evidence of an intracranial cause (e.g. Infection , head trauma , and epilepsy) , another definable cause of seizure (e.g. electrolyte imbalance , hypoglycemia , drug use, or drug withdrawal) or a history of an afebrile seizure (5).
Epilepsy is a disease of the brain defined by any of the following conditions:
1. At least two unprovoked (or reflex) seizures occurring > 24 hours apart.
2. One unprovoked (or reflex) seizure & a probability of further seizures similar to the general recurrence risk (at least 60%)
3. Diagnosis of an epilepsy syndrome (6,7,8&9)
Status epilepticus is defined as more than 30 minutes of either, continuous seizure activity or two or more sequential seizures without full recovery of consciousness between them (2). The 30 - Minute’s definition is based on the duration of convulsive status epilepticus that may lead to permanent neuronal injury by itself (10). Since the majority of seizures are brief, and once a seizure lasts more than 5 minutes it is likely to be prolonged (11). Continuous Generalized convulsive SE in adults and children older than 5 years was operationally defined as 5 min or more clinical and/or electrographic seizure activity or (ii) recurrent seizure activity without recovery (returning to baseline) between seizures. Status treatment protocols have used a 5 - minute definition to minimize both the risk of seizures reaching 30 minutes and the adverse outcomes associated with needlessly intervening on brief, self-limited seizures (12).
This guideline follows this convention & for purposes of treatment, uses the term status epilepticus to represent studies involving both prolonged seizures and traditionally defined status epilepticus.
Status epilepticus presents in several forms:
1. Convulsive status epilepticus consisting of repeated generalized tonic - clinic (GTC) seizures with persistent postictal depression of neurologic function between seizures.
2. Nonconvulsive status epilepticus where seizures produce a continuous or fluctuating epileptic twilight state.
3. Repeated partial seizures manifested as focal motor signs, focal sensory symptoms, or focal impairment of function (e.g. aphasia) not associated with altered awareness (epilepsies partialis continua) (13, 14&15).
DIFFERENTIAL DIAGNOSIS
The diagnosis of epileptic seizures is largely dependent on a good history and witness report, as well as good knowledge of seizure semiology, and of the symptoms of a variety of differential diagnostic conditions. Still, many episodic conditions may be confused with epileptic seizures (16). In the case of status epilepticus, many brief events can be easily excluded from the differential diagnosis. Reflex anoxic seizures and cyanotic breath breath-holding spells are the most common confusing events. They may be associated with brief extensor stiffening and a few seconds of irregular spasms or jerks because of the abrupt loss of cerebral energy supply. Occasionally, an anoxic seizure may provoke a true epileptic seizure. This combination is called an anoxic-epileptic seizure. A positive family history of epilepsy may make some children more prone to anoxic-epileptic seizures. The later may last up to 10 minutes and present as SE (17).
Differential Diagnosis of status epilepticus should include movement disorders and non-epileptic seizures (18) like psychogenic non-epileptic seizures. Sandifer syndrome can be considered a prototype of dystonic posturing that is usually evaluated as intractable seizures. It is a paroxysmal movement disorder characterized by dystonic movements of the head, neck, and trunk in association with gastroesophageal reflux disease (19&20).
However, dystonic postures can be Epileptic. A dystonic posture of a limb is a feature seen in focal seizures. It is infrequently seen in the very young but becomes more common in the school-age child. Dystonic postures of the hand are usually contralateral to the seizure focus. They may often be coupled with ipsilateral (to the seizure focus) limb automatisms. A unilateral dystonic posture suggests a focal seizure (21).
Both symmetric and asymmetric tonic postures, are seen with focal seizures. In the very young, it is possible that these tonic postures ar these tonic postures that may be generated in the deeper brainstem or subcortical structures and are not direct manifestations of the ictal discharges. In accordance with Following this, some asymmetric tonic postures can be reversed by passive turning of the head during a seizure, in a fashion similar to the tonic neck reflex elicited in the newborn (21).
Decerebrate and decorticate postures are not likely to be mistaken for a seizure, those posturing typically occur in response to noxious stimuli (22).
Other differential diagnoses of events that have duration’s compatible with status epileptics include self-stimulatory behaviors as seen in autistic children (23) and stereotyped behaviors associated with established neurologic diseases as cerebral palsy (22).
Psychogenic non-epileptic seizures (PNES) are time-limited disturbances of consciousness and motor-sensory control, not accompanied by ictal activity on electroencephalogram (EEG) and are best conceptualized as atypical neurophysiological responses to emotional distress, physiological stressors, and danger (24).
In cases where status epilepticus occurs in a newly diagnosed child or it is the first presentation of a seizure disorder, the following diagnoses should be considered; CNS vasculitis whether primary or secondary, anti NMDA encephalitis and other autoimmune illnesses (25), as well as febrile infection‐related epilepsy syndrome (FIRES) and new‐onset refractory status epilepticus (NORSE) (26). It is important to identify these disorders as early as possible to apply specific therapeutic agents.
EPIDEMIOLOGY OF ACUTE SEIZURES
Although the epidemiology of epilepsy has been well studied, less has been identified about the epidemiology of acute seizures. Studies suggest that around 4–10% of children have an experience of seizure before 16 years of age where 1/5th of total children with unprovoked seizures may develop epilepsy later. Each year, about 150,000 children and adolescents will come to medical attention for evaluation of a newly occurring seizure disorder of some type. Between 2% and 4% of all children in Europe and the United States experience at least one convulsion associated with a febrile illness before the age of 5 years (27).
EPIDEMIOLOGY OF SEIZURES IN THE EMERGENCY DEPARTMENT (ED):
Seizures are one of the common causes for hospital admissions in children with significant mortality and morbidity especially in younger children, it is estimated to be 2% of visits to children’s ED hospitals, (28) and 3.4% of admission to children’s tertiary care hospital (29). The high occurrence among small children, in comparison to older ones, is a result of the febrile seizure, accounting for 28% of all pediatric ED seizures (30).
GENDER:
Males account for a greater proportion of seizures in the ED setting, with an odds ratio of 1.4 for the association between male gender and seizure among ED visits (31). On a population level (rather than in the ED setting), acute symptomatic seizures occur in males versus females in a ratio of 1.85 to 1, with a lifetime risk of 5.0% in males and 2.7% in females (32). By contrast, epilepsy is only slightly more common in males. (33) The increased incidence of acute symptomatic seizures in males, in previously mentioned studies, is not due to trauma, as the male predominance persists across all etiologic categories. (31)
ETIOLOGY:
Most seizures managed in the ED are secondary to an underlying disease process, that is, they are either acute symptomatic seizures or pediatric febrile seizures. (28) Among children, the febrile seizure is the most common, accounting for almost a third of pediatric ED seizures. (34) Febrile seizures in children are classified as simple versus complex. Simple febrile seizures are the most common and have an excellent prognosis, while 20% of first febrile seizures are complex. The peak incidence of a first febrile convulsion occurs in the second year of life. The risk of recurrence is high, and ranges from about 12% in children whose first febrile seizure occurs in infancy to about 50% in those whose first febrile seizure occurs later. The subsequent risk of epilepsy is the same as for children who have never had a febrile seizure. Complex febrile seizures, on the other hand, do indicate an increased risk for epilepsy, and do not indicate an increased risk for future occurrence of simple febrile seizures. (36)
Between 0.5% and 1% of children and adolescents experience a seizure associated with acute metabolic causes; hyponatremia contributed to 70% of metabolic causes especially in children younger than 6 months and those in the neonatal period. (37) The contribution of “breakthrough seizures” among chronic epileptics to the burden of seizure care in the ED setting is unknown. The aforementioned study suggested that only 6.8% of seizures in the ED were due to epilepsy. (38) However, a smaller study in a different setting found that 46% of seizure visits to an urban ED were by known epileptics. (35)
SEMIOLOGY:
In the majority of studies, Generalized tonic tonic-clonic seizures were the commonest seizure type and were seen in; 63.3% in one study. (39) 82.1% by Chaudhary et al, (29) and even up to (98.0%) by Mwipopo1 et al. (40)
STATUS EPILEPTICUS:
In the ED-based series, when status epilepticus was mentioned, it represented 6% to 7% of seizures. (41) In 1995, DeLorenzo and colleagues estimated 195,000 status events in 152,000 patients per year in the United States, with 42,000 deaths. The highest rate was in infants. Fewer than half of the cases were managed by neurologists, and the majority (58%) occurred in patients with no prior history of epilepsy. In children, mortality was estimated with 3% in, while risk factors were: infection (52%), remote insult (39%), low antiepileptic drug levels (21%), stroke, metabolic, hypoxia, idiopathic, and trauma. (42)
EPIDEMIOLOGY OF EPILEPSY WITH SPECIAL REFERENCE TO EGYPT:
Epilepsy affects 50 million people worldwide, with an estimated 2–3 million living in the United States, 6 million in Europe, and at least 40 million in the developing world. Epilepsy poses a significant burden on the quality of life of affected individuals and their families. Nearly 80% of the people with epilepsy are found in developing regions. Seventy percent of people who develop epilepsy do so in the first two decades of life. Seizures and epilepsy affect infants and children more than any other age group. (43, 44)
Studies (with population size more than 1000) conducted in the Mediterranean region have shown that the prevalence of epilepsy ranges from as low as 4.04 per 1000 population to those as high as 12 or more per 1000 population. Furthermore, higher rates are reported in young age groups and in rural populations (45) In a comprehensive review of the epidemiology of epilepsy in the Arab World (22 countries), and epidemiological literature search was conducted utilizing Google and PubMed, Google provided 21 prevalence, four CAMP and nine incidence estimates while PubMed provided 10 such estimates none of which has been identified by Google. No epidemiological data about epilepsy were found in 10/22 countries. (46)
Regarding Egypt, the epidemiological profile of epilepsy in New Valley Governorate in Egypt (2013) was conducted to estimate its magnitude of treatment gap and revealed that life time prevalence of epilepsy in Al Kharga district is 6.76/1000, with the highest peak during early childhood. The annual incidence rate is 43.14/100, 00047. Another study was conducted to estimate the prevalence of different types of epilepsy in the region of the Assiut Governorate/Egypt (2013). The study revealed that the prevalence rate of active epilepsy was 9.3/1000 and the incidence rate was 1.5/1000. (75%) of them had idiopathic epilepsy. (CPR 9.5/1000) that high prevalence rate of epilepsy in Egypt is comparable with that in other Arabic and European countries (48).
Another study was conducted to evaluate the prevalence of epilepsy in primary school children in El-Minia City, Egypt (2009). Lifetime prevalence was 7.2/1000 in conventional schools and 133.3/1000 in schools for subnormal children. Male: Female ratio was 2:1. Prevalence was significantly higher among the lower socioeconomic class. In a door to door survey, a study conducted to determine prevalence and clinical patterns of epilepsy among children and adolescents in Upper Egypt. It revealed that lifetime prevalence was 9.7/1000 with a higher prevalence among children < 12 years than adolescents. The age-specific prevalence was highest among early childhood (12.01/1000) and lowest in adolescence. More than half of the target population have has idiopathic epilepsy. The most frequent causes of symptomatic/structural epilepsy were perinatal complications particularly in infancy, followed by CNS infection in early childhood and post-traumatic epilepsy in adolescence. Partial seizures were more frequent in infancy while generalized seizures were commoner in childhood and adolescence (49).
Another study for epilepsy prevalence in Al-Maniel Island using a door to door survey revealed a lifetime point prevalence of 6.9/1000 inhabitants and active epilepsy prevalence of 5.1/1000 inhabitants. The age distribution showed bimodal peaks one in adolescents and another in the elderly with equal sex ratio. Focal seizures were the most frequent (58.3%) and the treatment gap was (66.7%) (50). Studying stigma about epilepsy, a review was published in 2016 stated that people with epilepsy devalued and stigmatized. There are many factors that Many factors affect these stigmas including: false false-negative historical background of epilepsy, psychological aspects, knowledge, and attitudes towards people with epilepsy, uncontrolled epilepsy, and the effect of anticonvulsant drugs. (51) Studying the health-related quality of life in epilepsy revealed that patients with epilepsy are at a high risk of developing the impaired quality of life with lower mean scores in all domains especially those with generalized, frequent fits, those on polytherapy and those with younger age of onset of seizures and a longer duration of illness (52).
CLASSIFICATION OF SEIZURES & EPILEPSY
Figure (1): The basic ILAE 2017 operational classification of seizure types (54)
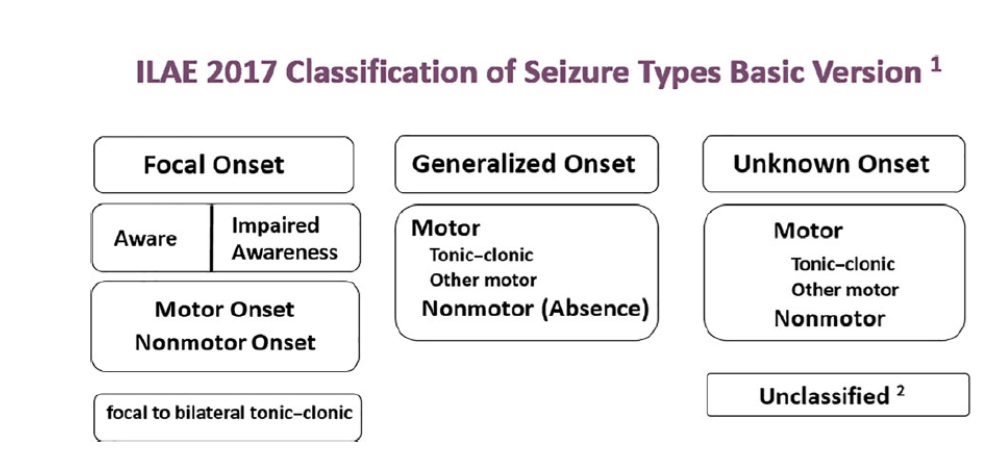
Figure (2): The expanded version of ILAE 2017 operational classification of seizure types (54)
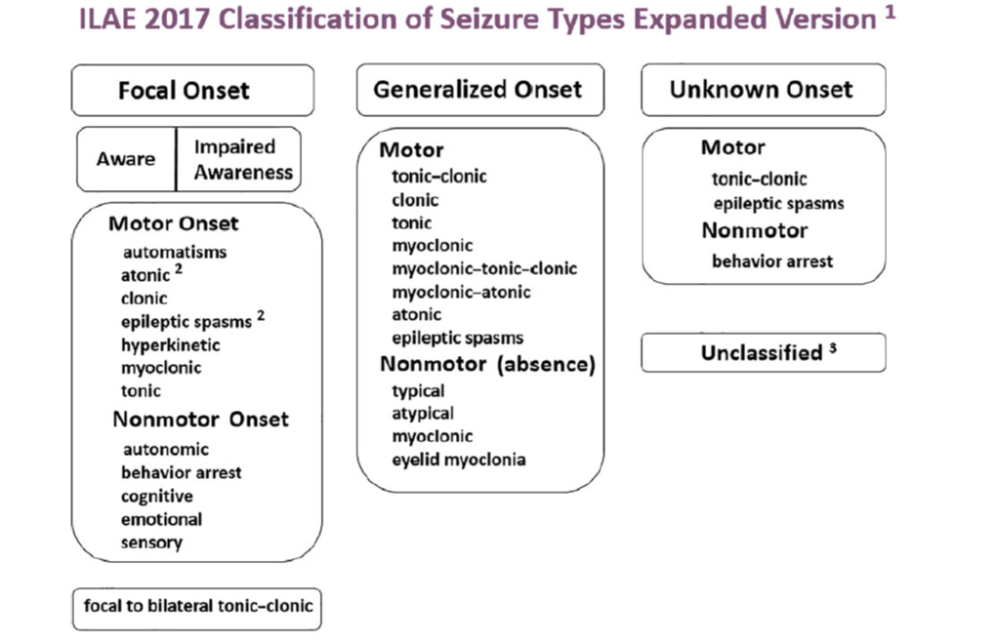
Seizure and epilepsy classification systems have been used in clinical practice and research since the 1970s(53). Over the years, multiple revisions have been implemented, the most recent of which is the 2017 International League Against Epilepsy (ILAE) operational epilepsy classification system. (54) This system aims to better define seizures and epilepsies by classifying them using key clinical features, EEG findings, imaging, and genetics. Seizure classification starts with whether the initial manifestations of the seizure are focal or generalized. Focal seizures originate within a neuronal network limited to one hemisphere that may be discretely localized or more widely distributed, whereas generalized seizures originate at some point within the brain and rapidly engage bilateral distributed networks. (55) If the onset of the seizure is missed or is unclear, the seizure is of unknown onset. Focal seizures are classified according to the patient’s level of awareness and the first most prominent motor or non-motor features of the seizure. (56) These early prominent features are important to consider when localizing the seizure onset or of the epileptogenic zone. The final feature used in the classification of focal seizures is whether the focal seizure evolves to a bilateral tonic-clonic seizure. A focal aware seizure replaces the previously termed simple partial seizure, and a focal impaired awareness seizure replaces the term complex partial seizure. Focal motor seizures can be more specifically defined. Motor-onset manifestations include automatisms, epileptic spasms, and atonic, clonic, hyperkinetic, myoclonic, or tonic seizures. Focal seizures with non-motor symptoms as the first prominent feature include autonomic, behavior arrest, cognitive, emotional, or sensory seizures. (57) Focal seizures can be further classified as to whether they evolve to a bilateral tonic-clonic seizure. This spread is typically clearly seen on EEG. (56) Generalized Seizures; similar to focal seizures, are classified according to a motor or non-motor manifestations. Broadly, motor seizures are either tonic-clonic or other motor seizures. Non-motor generalized seizures primarily refer to absence seizures. Motor onset more specifically includes tonic-clonic, clonic, tonic, myoclonic, myoclonic-tonic-clonic, myoclonic-atonic, atonic, or epileptic spasms. Non-motor or absence seizures include typical, atypical, myoclonic, or eyelid myoclonia. (58) Unknown Seizures are seizures of unknown onset and can be classified by a motor (tonic-clonic, epileptic spasms) or nonmotor (behavior arrest) presentations. If information is inadequate or if the seizure cannot be categorized, then the seizure is considered unclassified. (57)
The second level of classification is the epilepsy type. The epilepsy type is predominantly determined clinically; characteristic EEG findings provide supportive evidence. Similar to seizure classification, the epilepsies are classified as generalized or focal. (59) The new classification system additionally recognizes two new categories: combined generalized and focal epilepsy and unknown epilepsy. Patients with generalized epilepsy have one or more of the generalized seizure types, and their EEGs typically display generalized spike-wave activity. For individuals who have generalized seizure types and a normal EEG, other data are needed to determine whether the epilepsy is generalized. Having myoclonic jerks or a pertinent family history supports the diagnosis of a generalized epilepsy type. (60) Clinically, patients with one or more focal seizure types have focal epilepsy. These epilepsies can be either unifocal or multifocal. Although not always seen, focal EEG findings such as focal slowing or epileptiform discharges support the diagnosis of focal epilepsy. Concordant focal MRI findings are also supportive. Designation The designation of combined generalized and focal epilepsy is for patients with both focal and generalized seizures. EEG may reveal both focal and generalized electrographic findings. (56) Examples of combined generalized and focal epilepsy include Dravet syndrome and Lennox-Gastaut syndrome. When the patient has epilepsy as defined by the ILAE but it remains undetermined whether the patient has focal or generalized epilepsy, the classification of unknown epilepsy type is used. (61)
The epilepsy syndrome is a new addition to the current classification system and is defined as “a cluster of features incorporating seizure types, EEG, and imaging features that tend to occur together.” Factors that contribute to epilepsy syndrome include the age of onset, remission, triggers, diurnal variation, intellectual and psychiatric dysfunction, EEG findings, imaging studies, family history, and genetics. (56) The etiology of seizures and epilepsies is emphasized in the new classification system. Six etiologic categories (structural, genetic, infectious, metabolic, immune, unknown) have been defined. When multiple potential etiologies are present, priority should be given to the etiology with more relevant management issues. (62)
Status epilepticus (SE) is classified by four axes: (1) semiology; (2) etiology; (3) electroencephalography (EEG) correlates; and (4) age. Axis 1 (semiology) lists different forms of SE divided into those with prominent motor systems, those without prominent motor systems, and currently indeterminate conditions (such as acute confusional states with epileptiform EEG patterns). Those forms with prominent motor symptoms and impairment of consciousness may be summarized as convulsive SE as opposed to the non-convulsive forms of SE (NCSE). Axis 2 (etiology) is divided into subcategories of known and unknown causes. The etiologic classification of SE includes 1) acute symptomatic, 2) remote symptomatic, 3) remote symptomatic with an acute precipitant, 4) progressive encephalopathy, 5) febrile, and 6) cryptogenic (idiopathic). Axis 3 (EEG correlates) adopts the latest recommendations by consensus panels to use the following descriptors for the EEG: name of the pattern, morphology, location, time-related features, modulation, and effect of the intervention. Finally, axis 4 divides age groups into neonatal, infancy, childhood, adolescent and adulthood, and elderly. (63)
COMPLICATIONS OF ACUTE CONVULSIONS
Complication A complication is was defined as an event that requires additional attention, time, and effort by the treating physician and nursing staff. The majority of the generalized tonic tonic-clonic seizures (GTCS) are benign but some lead to complications such as cardiac arrhythmias, fractures/falls, rhabdomyolysis, and increase creatinin phosphokinase enzyme (CPK), aspiration pneumonia, acute kidney injury and disturbed level of consciousness or acute confusional state. While briefer, focal, and myoclonic seizures have less few complications. (64) The first stage of convulsive status epilepticus (CSE) is characterized by a high level of catecholamines in the blood, which leads to hyper -adrenergic stimulation and extreme stress on the body systems. It is triggered by intense and prolonged brain electrical activity and ending by blood vessels damage of many organs. (65)
All complications encountered could be summarized in the following table:
|
Classification |
Complications |
|
Systemic: |
|
|
Cardiac: |
Hypertension, tachycardia (reversing after 30 minutes), arrhythmias, and cardiac arrest. |
|
Pulmonary: |
Apnea, respiratory failure, hypoxia, neurogenic pulmonary edema, and aspiration pneumonia. |
|
Autonomic: |
Fever and hyperpyrexia, sweating, hypersecretion (including tracheobronchial), and vomiting. |
|
Metabolic: |
Hyperkalemia, hyperglycemia then hypoglycemia, volume depletion, metabolic acidosis, venous stasis, and possible thrombosis/ hemorrhage. |
|
Endocrine: |
Increased prolactin and cortisol |
|
Gastrointestinal complications: |
Stress ulcer, gastrointestinal hemorrhage, diarrhea, and paralytic ileus. |
|
Others: |
Leukocytosis, cerebrospinal fluid pleocytosis, vertebral and other fractures, physical injury, rhabdomyolysis, renal failure and disseminated intravascular coagulation |
|
Cerebral: |
Neuronal damage ، cerebral edema, raised intracranial pressure،, cortical vein thrombosis, and cerebral hemorrhage. |
|
Neurologic sequelae: |
Increased seizure frequency, recurrent status epilepticus, and decreased cognitive function. |
Adapted from: Drislane FW. Status epilepticus. In: Schachter SC, Schomer DL. Eds. The comprehensive evaluation and treatment of epilepsy. San Diego, CA: Academic Press: 1997. P. 149-172. With permission from Elsevier (www.elsevier.com)(3). (66)
Another source of complications is the medications used to control seizures as Benzodiazepines, which may cause respiratory and cardiovascular depression as well as immunologic dysfunction. Other drugs used in the treatment of SE include anesthetic and non-anesthetic drugs are also accompanied by various complications such as hypersensitivity, breathing difficulty, hemodynamic alterations, and renal injury. The distributive shock caused by these drugs can cause further deterioration of the neurological status by inducing or enhancing neuronal necrosis.
Complications may also arise from the long duration of severe illness and immobilization following the onset of SE and prolonged stay in the intensive care unit may lead to various respiratory and infectious ones. These may include nosocomial pneumonia, ventilator-associated pneumonia, pulmonary embolism, atelectasis or collapse of the lung alveoli, adult respiratory distress syndrome, and neurogenic pulmonary edema. Clostridial colitis and other infectious complications could also be enhanced by the slowed gastrointestinal motility. (65)
Sudden Unexplained Death in Epilepsy (SUDEP)
SUDEP is defined as a death that occurred suddenly, unexpectedly and of non-traumatic and non-drawing, witnessed or unwitnessed, with or without the evidence of a seizure, excluding status epilepticus, and without a toxicological or anatomical cause of death in post-mortem examination and will be responsible for about 7.5 to 17% of all deaths in epilepsy. Risk factors possible associated with SUDEP are refractoriness of the epilepsy, the occurrence of generalized tonic-clonic seizures, antiepileptic drugs polytherapy, early age of epilepsy onset, long term seizure disorder (more than 15 years), cardiovascular and pulmonary abnormalities and genetic factors. (67)
LABORATORY INVESTIGATIONS
All patients with acute seizure presentation should be investigated with routine blood tests to exclude infection or metabolic disturbance. (68) When evaluating a first seizure occurring in children, the American Academy of Neurology has recommended that laboratory screening tests should be ordered based on individual clinical circumstances. (69)
Acute and/or severe electrolyte imbalances can manifest with rapidly progressive neurologic symptoms or seizures, which may be the sole presenting symptom. (70)
Obtaining intravenous access is the next step after establishing the airway and circulation. (71) Blood should be measured for glucose, serum electrolyte; calcium, magnesium, sodium, potassium, blood urea nitrogen. Antiepileptic drug levels, as well as a toxic drug screen and complete blood cell count, are also required. Lumbar puncture was recommended when fever occurred with SE, especially in young children, unless a contraindication to LP was present. (72)
TREATMENT OF ACUTE SEIZURE
Given the urgency of terminating seizures, the initial diagnostic assessment and medical treatment should occur simultaneously. Though treatment options and diagnostics may be dictated by available medications and capacity, it is possible for virtually any healthcare setting to develop a relevant and feasible local guideline for seizure management. Clear specifications on when to refer to a higher level of care should be part of the care plan. (73&74)
PREHOSPITAL SEIZURE MANAGEMENT
In the prehospital setting or in a health care facility that is not equipped to administer intravenous medications, the WHO Guidelines Approved by the Guidelines Review Committee 2016 recommended that 0.5 mg/kg (maximum dose of 10 mg) of liquid diazepam can be given rectally. (74)
ACUTE MANAGEMENT
The urgent priority is terminating seizures, the WHO Review Committee 2016(75) recommended that the initial diagnostic assessment and emergency medical treatment should occur simultaneously. A brief history and rapid clinical examination should be done to find underlying etiology (e.g., neuro-infection, poisoning, established epilepsy) and to confirm that the event is a seizure. (73)
World Health Organization (WHO) updated their its emergency triage assessment and treatment (ETAT) guidelines. The guidelines are used to identify children with the life-threatening conditions seen most frequently in developing countries. Thus, children presenting with the following signs should be treated as emergencies: obstructed or absent breathing, severe respiratory distress, central cyanosis, signs of shock (cold extremities with capillary refill time > 3 s and weak and fast pulse), coma (or seriously reduced level of consciousness), seizures or, in a child with diarrhea, any two signs of severe dehydration signs: lethargy or unconsciousness, sunken eyes, very slow return of skin after pinching. Immediate treatment is begun when any emergency sign is identified (by assessment of Airway, Breathing and Circulation, ABC) until the end of the algorithm. Children who require prompt, full assessment, and rapid treatment are then checked for priority signs. Children without emergency or priority signs are deemed “non-urgent” on completion of triage. (74&76)
ABC with high concentration oxygen. Check glucose and establish an IV line. First First-line with IV is lorazepam. If lorazepam not available then diazepam. If no IV, then buccal midazolam. Second Second-line includes phenobarbital or phenytoin. Refractory status with midazolam or thiopental. (77)
The guideline, which reviewed all available adult and pediatric evidence, provides a treatment algorithm that comprises three phases of treatment. It also offers evidence-based answers to the effectiveness, safety, and tolerability questions regarding the treatment of convulsive status epilepticus.
• Stabilization phase (0-5 minutes of seizure activity), includes standard initial first aid for seizures and initial assessments and monitoring.
• Initial therapy phase (5-20 minutes of seizure activity) when it is clear the seizure requires medical intervention, a benzodiazepine (specifically IM midazolam, IV lorazepam, or IV diazepam) is recommended as the initial therapy of choice, given its demonstrated efficacy, safety, and tolerability. A benzodiazepine is the initial therapy of choice (Level A):
Choose one of the following 3 equivalent first first-line options with dosing and frequency:
1. Intramuscular midazolam (10 mg for > 40 kg, 5 mg for 13-40 kg, single dose, Level A) OR
2. Intravenous lorazepam (0.1 mg/kg/dose, max: 4 mg/dose, may repeat dose once, Level A) OR
3. Intravenous diazepam (0.15-0.2 mg/kg/dose, max: 10 mg/dose, may repeat dose once, Level A)
If none of the 3 options above are available, choose one of the following:
1. Intravenous phenobarbital (15 mg/kg/dose, single single-dose, Level A) OR
2. Rectal diazepam (0.2-0.5 mg/kg, max: 20 mg/dose, single dose, Level B) OR
3. Intranasal midazolam (Level B), buccal midazolam (Level B)
• Second therapy phase (20-40 minutes of seizure activity) when a response (or lack of response) to the initial therapy should be apparent. Reasonable options include fosphenytoin, valproic acid, and levetiracetam. There is no clear evidence that any one of these options is better than the others. Because of adverse events, IV phenobarbital is a reasonable second-therapy alternative if none of the three recommended therapies are available.
Choose one of the following second second-line options and give as a single dose:
1. Intravenous fosphenytoin (20 mg PE/kg, max: 1500 mg PE/dose, single single-dose, Level U) OR
2. Intravenous valproic acid (40 mg/kg, max: 3000 mg/dose, single dose, Level B) OR
3. Intravenous levetiracetam (60 mg/kg, max: 4500 mg/dose, single dose, Level U)
If none of the options above are available, choose one of the following (if not given already)
Intravenous phenobarbital (15 mg/kg, single dose, Level B)
• Children younger than 2 years presenting with seizures lasting more than 30 minutes, especially if myoclonic or if they are known to have pyridoxine dependent / /responsive seizures, should receive pyridoxine trial of 50-100 mg IM. It is to be noted that the IV forms are not available in Egypt, and using the IM route also avoids possible side effects previously mentioned with the IV route.
Third therapy phase (40+minutes of seizure activity). There is no clear evidence to guide therapy in this phase. The guideline found strong evidence that initial second therapy is often less effective than initial therapy, and the third therapy is substantially less effective than initial therapy. Thus, if second therapy fails to stop the seizures, treatment considerations should include repeating second-line therapy or anesthetic doses of either thiopental, midazolam, pentobarbital, or propofol (all with continuous EEG monitoring). (78)
PROGNOSIS
Seizure recurrences
- For febrile seizures, epilepsy occurs more frequently than in the general population. In a normal child with a simple febrile seizure, the risk is approximately 1 to 2 percent, only slightly above that of the general population. (79) For children with complex febrile seizures, an abnormal developmental history, or a family history of epilepsy, the risk is closer to 5 to 10 percent. (80)
- Children who have an acute symptomatic seizure (e.g., acute infection, acute head injury), have a low risk of seizure recurrence compared with other children with a first seizure and seizure recurrence is likely only if the underlying etiology recurs (Examples include seizures associated with febrile illnesses, metabolic derangements such as hyponatremia, and concussion). (81)
- The child who is a neurologically normal neurologically normal child with, has no history of a prior neurologic illness who, and has an unprovoked seizure with no evident acute cause has an approximately 25 percent risk of having another seizure in the next year and a 45 percent risk over the next three years. Clinical factors associated with an increased risk of recurrent seizures include: 1) Prior neurologic insult, 2) Significant brain MRI findings, 3) Abnormal EEG. (82-85)
Morbidity and Mortality
- The prognosis for children with febrile seizures is favorable. While early reports had suggested that febrile seizures were associated with an increased risk of sudden death, the results from a large population-based study indicate that the small excess in mortality among children with febrile seizures is restricted to those with complex febrile seizures. (86) Furthermore, the increased risk in those patients is explained by preexisting neurologic abnormalities and subsequent epilepsy. (87)
- Neurologic sequelae, including new neurologic deficits, intellectual impairment, and behavioral disorders, are rare following febrile seizures. (88)
- Children with status epilepticus (SE) have an overall mortality of approximately 0% to 3%. Considering only children admitted to pediatric intensive care units, the mortality is approximately 5% to 8%. Further, children surviving SE are at risk of lifelong sequelae, including cognitive and neurodevelopmental impairments, new-onset epilepsy, and recurrent SE. The occurrence of seizures and the recurrence of SE are influenced by the underlying etiology, with structural or metabolic lesions having the highest risk. (89)
- Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of seizures in children in Egypt.
It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to provide standardization of the acute management of seizures in children in Egypt and the prevention of their complications. This version of the guideline includes recommendations and good practice statements for seizures in children in Egypt.
- Methods
Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: convulsions, seizure, convulsions beyond neonatal period.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2011 and later or the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria the GDG/ GAG recommended using 5 guidelines:
1- Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society. American Epilepsy Society Guideline (2016).
2- Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics. International League Against Epilepsy (2015).
3- Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy. ITALIAN LEAGUE (LICE) (2013).
4- Guidelines for the Evaluation and Management of Status Epilepticus Neurocritical Care Society Status Epilepticus Guideline (2012).
5- Evidence-Based Guidelines for EEG Utilization at the University Teaching Hospital (UTH). Neurologic & Psychiatric Society of Zambia (2011).
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of pediatrics’ neurology.
The main functions of the clinical panel were adolopment of Emergency Management (Diagnosis and Treatment) of Seizures in Children Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 5 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 2 clinical national experts who have interest and expertise in pediatric neurology
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [91]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations (both ETD and changing strength of recommendation were not done in this guideline).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include awell-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendation. both ETD and changing strength of recommendation were not done in this guideline).
Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
➡️Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period have been made available in several forms including:
Table (13): CPG implementation strategies
|
Focus of Strategy |
Strategies |
|
Practitioners |
· Educational meetings: Conferences, lectures, workshops or traineeships, grand rounds, seminars, and symposia. |
|
· Educational materials: Printed or electronic information. |
|
|
· Web-based education: Computer-based educational activities. |
|
|
· Educational outreach/academic detailing: A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s). |
|
|
· Audit and feedback: Any summary of the clinical provision of health care over a specified period; may include recommendations for clinical action. The information is obtained from medical records, databases, or observations of patients. The summary may be targeted at the individual practitioner or the organization. |
|
|
· Reminders: The provision of information verbally, on paper or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care. |
|
|
· Local opinion leaders: Providers nominated by their colleagues as “educationally influential.” In general, such individuals are identified by their peer colleagues, are trained as change agents, and operate within their communities to teach and enable change. |
|
|
· Patient-mediated interventions: Interventions directed at patients (e.g., mass media campaigns, reminders, education materials) to optimize professional-patient interactions. |
|
|
· Practice tools: Tools designed to facilitate behavioral/practice changes, e.g., flow charts. |
|
|
Patients |
· Patient education materials: Printed/electronic information aimed at the patient, consumer, family, caregivers, etc. |
|
· Mass media campaigns |
|
|
· Reminders: The provision of information verbally, on paper, or electronically to remind a patient/consumer to perform a particular health-related behavior. |
|
|
· Decision-support tools: Aids designed to facilitate shared decisions by patients and their physicians |
|
|
Organizations and regulatory bodies |
· Changes to health care teams: Changing tasks or responsibilities of health professionals or compositions of health professional groups. |
|
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc. |
|
|
· Audit and feedback: Any summary of a clinical provision of health care over a specified period; may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. The summary may be targeted at the individual practitioner or the organization. |
|
|
· Administrative procedures/policies |
|
|
· Formularies: Drug safety programs, electronic medication administration records. |
|
|
· Financial incentives or penalties: The use of remuneration for the performance of certain functions or actions, e.g., screening procedures in primary care. |
|
|
· Mandated practices |
➡️Example of Dissemination and Implementation Proposed Resources
Educational materials based on this Adapted CPG for acute management of seizures in children beyond the neonatal period shall be made available in several forms, including a pocket guide for health care professionals and for parents/caregivers.
proposed Implementation tools:-
• Patient Resources (Information) (Arabic).
• Pocket Guide/Quick Reference Summary (English)
• Power Point Slide Presentation.
• Clinical Algorithm
• Drug Table
1. Pediatric Emergency Triage:
Pocketbook of hospital care for children: Second edition.
Guidelines for the management of common childhood illnesses http://apps.who.int/iris/bitstream/10665/204463/1/9789241510219_eng.pdf?ua=1
Pediatric emergency triage, assessment, and treatment: care of critically-ill children Updated guideline
http://apps.who.int/iris/bitstream/10665/204463/1/9789241510219_eng.pdf?ua=1

2.Quick Summary Guide
|
Guideline Source |
Recommendation |
Level of Evidence |
|
History taking |
||
|
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015 |
1. The first stage in clinical management is to recognize if abnormal movement or behavior has an epileptic origin |
Level B |
|
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015 |
2. Rule out febrile seizures |
Level B |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
3. Ask about prior history of epilepsy |
Level B |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
4. AED given, AED non-compliance, or known allergies. |
Level C |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
5. Address history of Ingestion of a toxin or drug abuse as possible etiologies of SE. |
Level C
|
|
Clinical Examination |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
1. Assessment of vital signs: O2 saturation, BP, HR in the immediate (0–2 min) to establish and support baseline vital signs |
Level U |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
2. Neurologic exam is urgent (5–10 min) to evaluate for mass lesion, acute intracranial process |
Level U |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
3.Address respiratory monitoring to prevent cardiopulmonary arrest, during anticonvulsant drug treatment in status epilepticus in children. |
Level A |
Laboratory Evaluation |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012 |
1. Laboratory tests should be ordered based on individual clinical circumstances that include suggestive historic or clinical findings such as vomiting, diarrhea, dehydration, or failure to return to baseline alertness. |
Level U |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
2. For all patients, order laboratory tests: blood glucose, complete blood count, calcium (total and ionized), magnesium, AED levels. |
Level U |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012
|
3. Consider based on clinical presentation c) Lumbar puncture (LP). Should be performed in the very young child (<6 months), in the child of any age with persistent (cause unknown) alteration of mental status or failure to return to baseline, or in any child with meningeal signs. If increased intracranial pressure is suspected, the LP should be preceded by an imaging study of the head d) Comprehensive toxicology panel including toxins that frequently cause seizures (i.e. isoniazid, tricyclic antidepressants, theophylline, cocaine, sympathomimetics, alcohol, organophosphates, and cyclosporine e) Other laboratory tests: liver function tests, coagulation studies, arterial blood gases, and inborn errors of metabolism |
Level U
Level U |
|
EEG |
||
|
Evidence-Based Guidelines for EEG Utilization at the University Teaching Hospital (UTH) – 2011 |
V. In the PICU, An EEG is indicated in all unconscious patients suspected of non-convulsive status epilepticus or subclinical seizures. This includes comatose or obtunded inpatients of unclear etiology especially those in whom seizures preceded the onset of coma. VI. EEG is recommended after a first unprovoked seizure as it might offer insights into recurrence risk and/or the need for further neuroimaging. |
Level U |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
VII. In the treatment of convulsive status epilepticus in children, continuous EEG monitoring is indicated if second therapy fails to stop seizures, and treatment consideration involves the use of anesthetic doses of either thiopental, midazolam, pentobarbital, or propofol. |
Level U |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012 |
VIII. The use of continuous EEG (cEEG) is usually required for the treatment of SE. IX. Continuous EEG monitoring should be initiated within 1 h of SE onset if ongoing seizures are suspected X. The duration of cEEG monitoring should be at least 48 h in comatose patients to evaluate for non-convulsive seizures. XI. The use of this cEEG is encouraged to be available at tertiary care facilities where the optimum standard of care is provided. |
Level U
Guideline Adaptation Group and External Reviewer |
|
Neuroimaging (CT / MRI Brain) |
||
|
Diagnosis and Treatment of the First Epileptic Seizure: Guidelines of the Italian League Against Epilepsy, 2006 |
i. The use of a brain CT scan or MRI in the emergency room is indicated when specific interventions may be needed. ii. A CT scan is strictly indicated when a structural lesion is suspected or when the etiology of the seizure cannot be easily identified. Structural lesions include, among others, post-traumatic complications, cerebral hemorrhage, brain edema, and space-occupying lesions, which may be suggested by post-ictal deficits and/or persisting impairment of consciousness. iii. In the emergency room, MRI is not indicated, except for much selected circumstances, to be evaluated on an individual basis. |
Level 2 |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
· The etiology of SE should be diagnosed as soon as possible and occur simultaneously and in parallel with treatment for evaluation for mass lesions, meningitis, encephalitis. · All patients with SE should have Head computed tomography (CT) scan · Consider Brain magnetic resonance imaging (MRI)based on clinical presentation |
strong recommendation
Expert opinion Expert opinion |
Therapies |
||
|
Initial Therapy: 0 – 20 minutes |
||
|
1. Benzodiazepines |
||
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
|
· Benzodiazepines are the most efficient group of drugs to be initially used in the treatment of acute convulsion after 5 minutes from the start of seizures
|
Level A
|
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
1. Pre-hospital Treatment: - As early as possible by Trained personal whether medical\Para medical or caretaker. |
Level 1B Level 2B |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
- Drugs of Choice: - Midazolam (intra-muscular – buccal – nasal) - Diazepam (rectal) - Only one dose Not to be repeated |
Level1B
Level1B Level1B Level B
|
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
|
2. Hospital Emergency Room: · History of pre-hospital use of benzodiazepines must be considered as well as timing. · Doses given within one hour prior to ER presentation must be regarded as an initial dose - Midazolam It is the most appropriate type of benzodiazepines to be used. In the hospital the whole full dose to be given as a single dose better than broken into multiple doses. - Diazepam IV diazepam could be repeated after 5 minutes convulsion continued · Alternatively IF VENOUS ACCESS UNAVAILABLE. - Use midazolam: buccal or intranasal. - Use diazepam: rectal · Follow up for signs of respiratory depression as hypoventilation, pallor decreased oxygen saturation. · Respiratory support and intubation may be needed |
Level A
Level A
Level B
Level A
|
|
2. Non Benzodiazepine Therapy 2.1 Phenytoin and Phenobarbitone |
||
|
First Phase therapy: 5 - 20 minutes |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
· Both phenytoin and phenobarbital are recommended |
Level A
|
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· Phenytoin followed by phenobarbital is recommended if benzodiazepines fail |
Level 2B |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
· Phenobarbital is recommended in the absence of benzodiazepines |
Level. A |
|
Second phase therapy: 20 - 40 minutes |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
- Phenytoin and Phenobarbital is recommended
- They are not used sequentially but alternating. Generally, phenytoin precedes phenobarbital on account of better safety profile |
Class II a Class II b |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· Level B |
|
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
Level B |
|
|
2.2 Valproate |
||
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: (AES guidelines: Epilepsy Currents, Vol. 16, No. 1 (January/February) 2016 pp. 48–61: |
|
Level B
Recommendations of Guideline Adaptation Group and External Reviewers |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· Valproic acid offers a valid alternative to phenytoin and phenobarbital. It should be used with extreme caution, however, particularly in young children, when a possible metabolic etiology has not been ruled out. |
Level 1B |
|
2.3 Pyridoxine |
||
|
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
· Consider vitamin-dependent diseases (pyridoxine or pyridoxal-phosphate) in the early myoclonic epilepsy group
· Young children with epilepsy (under 3 years) who develop SE lasting 30 minutes, especially if myoclonic should receive pyridoxine trial in case they have pyridoxine dependent/responsive seizures. |
Expert opinion class 4
Level C
|
2.4 Levetiracetam |
||
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· IV Levetiracetam or oral through a nasogastric tube could be given in the second phase therapy for SE
· This drug can be used for continued oral therapy, does not cause significant side effects, and needs no preliminary check of renal function; |
Level U
Level 4 |
3. Overall Seizure Care Pathway
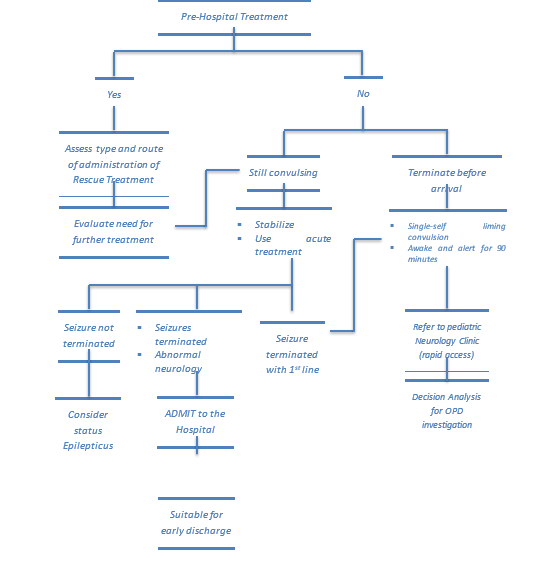
4. Detailed National Seizure Care Pathway

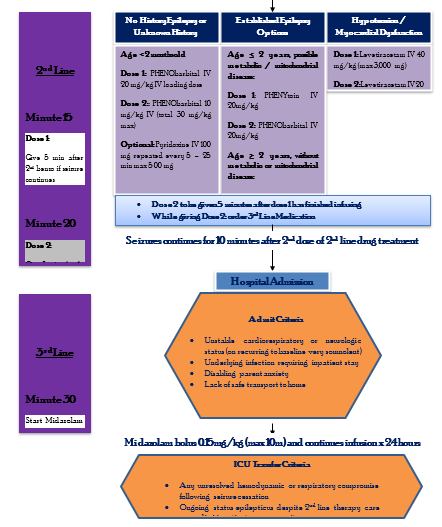
|
Assess Risk of Meningitis or Intracranial Infection |
|
Acute Seizure Management: Diagnostic Tests |
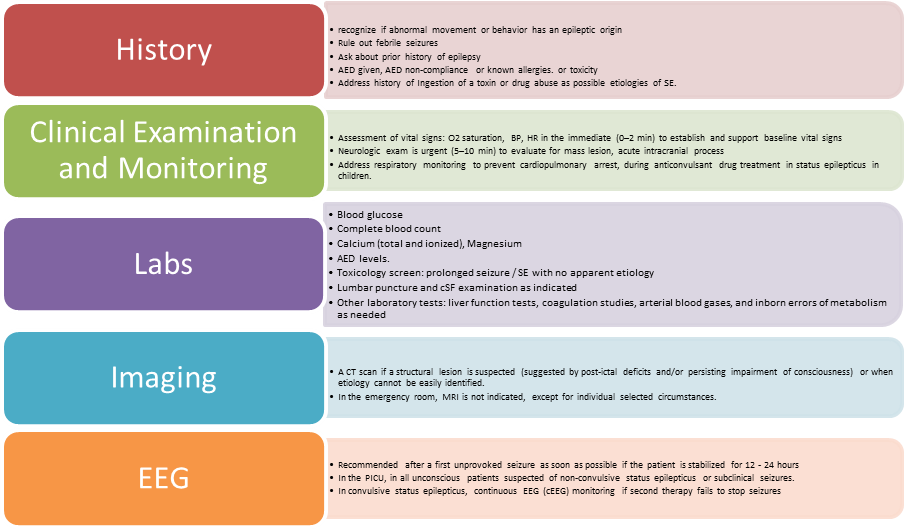
Lumbar puncture (LP) should be performed in:
1. The very young child (<6 months),
2. The child of any age with persistent (cause unknown) alteration of mental status or failure to return to baseline,
3. Any child with meningeal signs.
If increased intracranial pressure is suspected, the LP should be preceded by an imaging study of the head
|
Assess Risk of Meningitis or Intracranial Infection |
||
|
History · >3 days duration of illness · Seen by the previous doctor in previous 24 hours · Drowsiness or vomiting at home · Infants 6 – 12 months old deficient in Hib or pneumococcal vaccines or immunization status cannot be determined · Pretreated with antibiotics |
Physical Signs · Petechiae · Questionable nuchal rigidity · Drowsiness · Convulsing on examination · Weakness or neurological deficit on examination · Signs of infection of head or neck with potential for intracranial extension (e.g. mastoiditis, sinusitis..etc) · Bulging anterior fontanelle |
Complex Features · Focal seizures · Seizure duration > 15 minutes · Multiple seizures in 24 hours
Meningitis Less Likely · Prior febrile seizures · Pre-existing neurological findings |
|
Rationale for Use of Antiepileptic Drugs |
|
Benzodiazepines (1st Line: 2 doses) |

|
2nd Line Medications |
· Phenytoin followed by phenobarbital is recommended if benzodiazepines fail (Level 2B, Grade B).
· Phenobarbital is recommended if no benzodiazepines available (Level A).
· Phenytoin and phenobarbital are not used sequentially but alternating. Generally, phenytoin precedes phenobarbital based on better safety profile (Level B)
· IV valproate is used as an alternative based on its availability especially in tertiary care centers (Expert Opinion)
· IV valproate is used with extreme caution especially in young children when a metabolic etiology has been ruled out (Level 1B, Grade A)
· IV levetiracetam or through a nasogastric tube is an alternative (Level U). It can be used for continued oral therapy, does not cause significant side effects, and needs no preliminary check for renal function (Level 4, Grade C).
|
Consider 2nd Line IV bolus with drug patient is already on Dose 2 to be given 5 minutes after dose 1 has finished infusing |
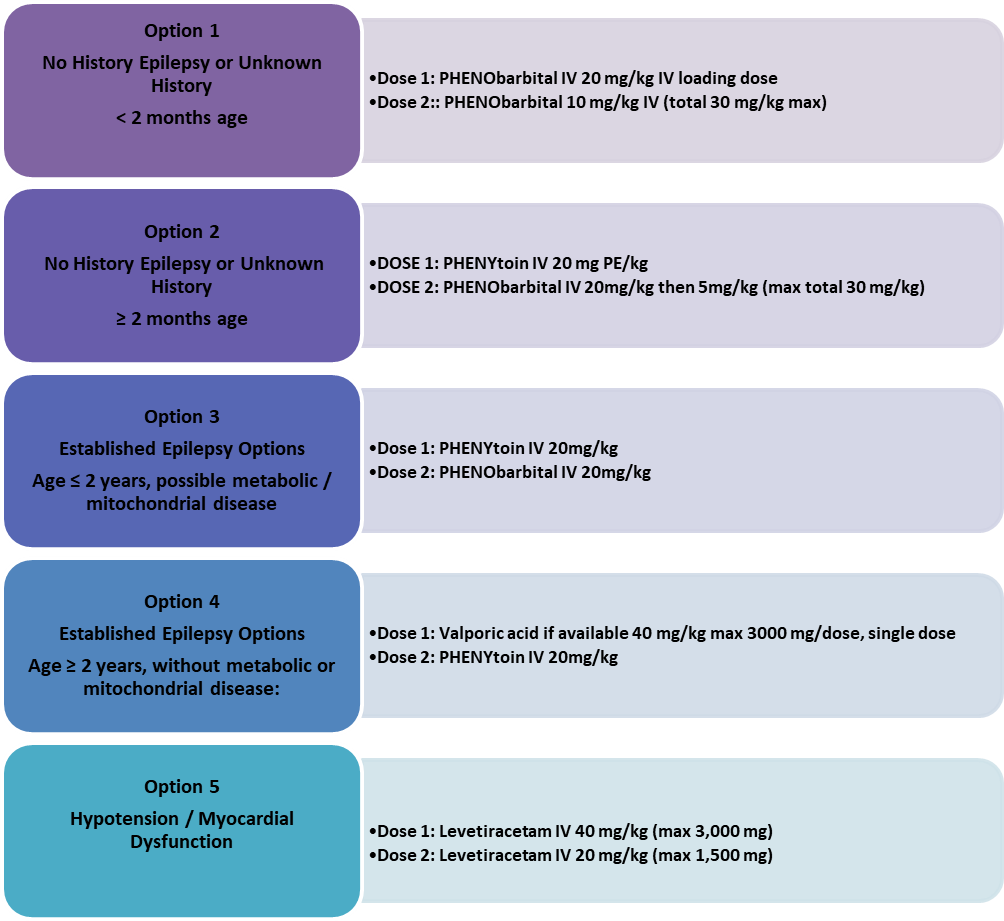
|
Pre-hospital Treatment |
- As early as possible (Level 1B – Grade A and Level 2 – Grade B)
- Better by trained personnel (medical/paramedical) or by a caregiver.
- Drugs of Choice:
- Midazolam:
- Intra-muscular:
|
13 – 40 kg: |
5 mg |
|
>40 kg: |
10 mg |
- Buccal: 0.5 mg/kg (max 10 mg)
- Diazepam:
- Per-rectal:
|
2 – 5 years: |
0.5 mg/kg |
|
6 – 11 years: |
0.3 mg/kg |
|
>12 years: |
0.2 mg/kg |
|
Buccal Midazolam
|
It can be given by the “Syringe Method” where the IV preparation 5 mg/ml trickled from a syringe into which the midazolam has been drawn up using insulin syringe to deliver 1 ml i.e. 5 mg midazolam.
|
Buccal Midazolam |
Buccal Midazolam – Syringe Method:
|
Step 1: Open the ampoule of midazolam |
|
|
Step 2: Insert the syringe into the ampoule and hold the ampoule and syringe upright with the ampoule above the syringe. Withdraw the required dose as prescribed by your doctor into a 1mL syringe. Push out any big air bubbles and check the amount of midazolam in the syringe. Remove the ampoule from the syringe. |
|
|
Step 3: Lay your child on their side (recovery position). Open their lips and slowly trickle the midazolam from the syringe into the inside of the lower cheek The calculated dose is applied to the buccal cavity between the gum and the cheek. Massage the cheek gently after administration. There is no need to open your child's jaw. The solution does not need to be swallowed. |
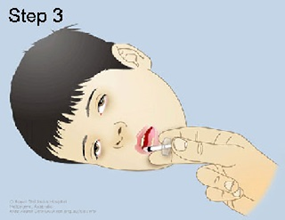
|
|
Step 4: Watch your child's breathing and seizure activity while they remain lying on their side in the first aid position. |
|
|
Step 5: Write down the time that the seizure started, when the midazolam was given and when the seizure stopped. |
|
It can be given by the “Syringe Method” where the IV preparation 5 mg/ml trickled from a syringe into which the diazepam has been drawn up using insulin syringe to deliver 1 ml i.e. 5 mg diazepam.

Rectal Diazepam – Syringe Method:
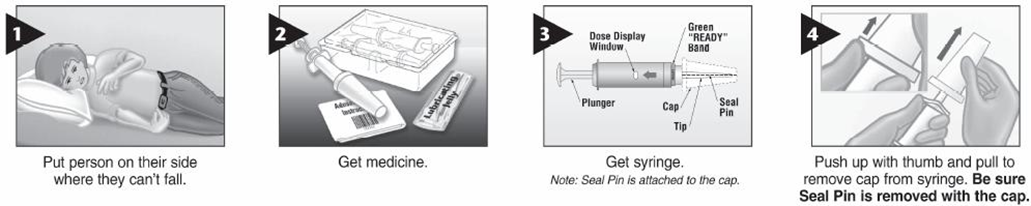
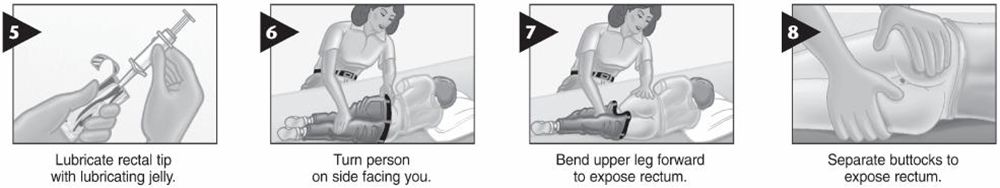
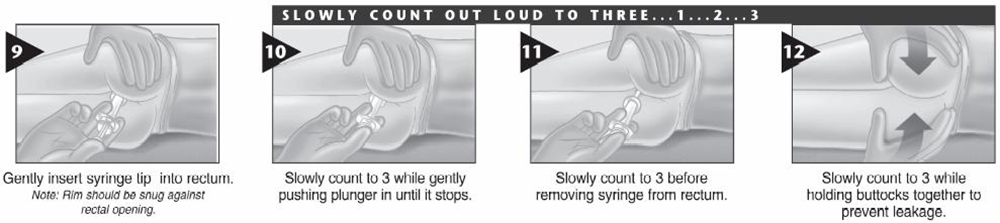
5. Emergency Seizure Management Algorithm
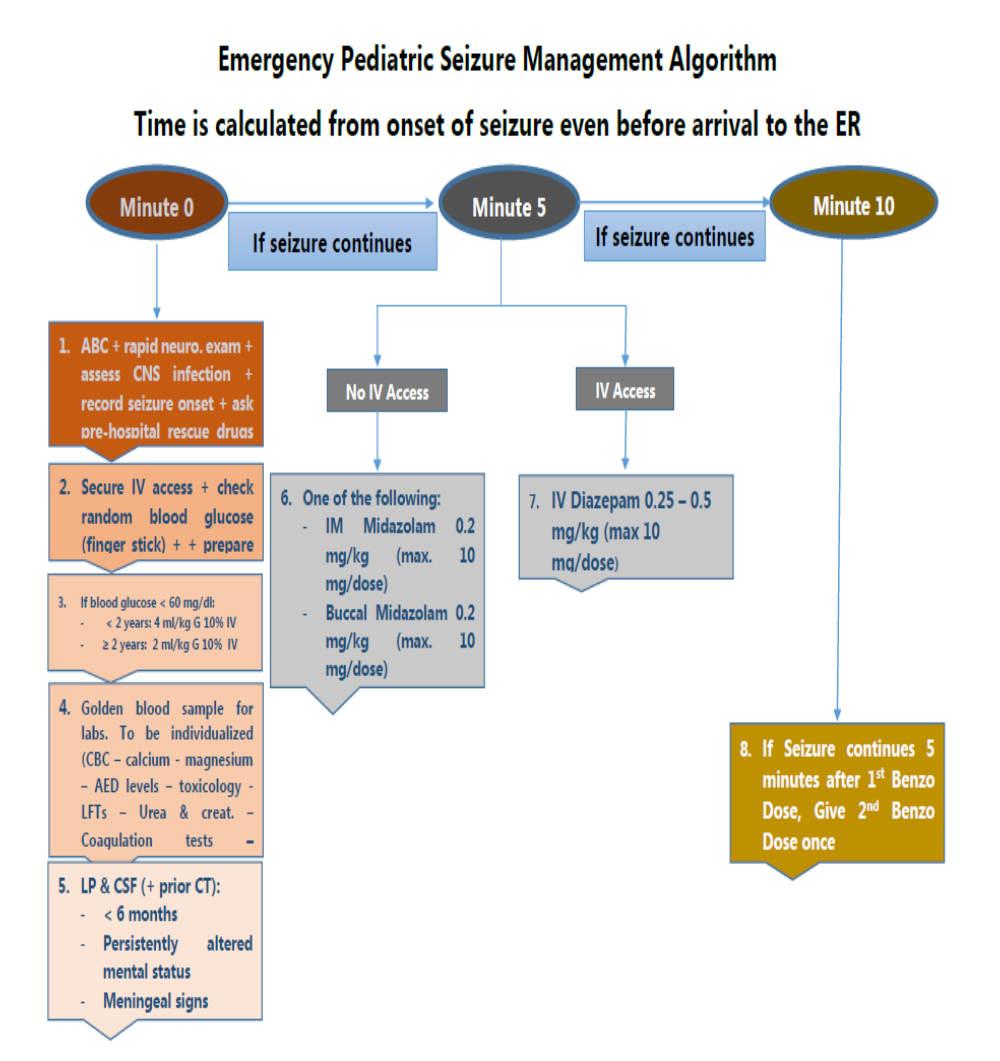
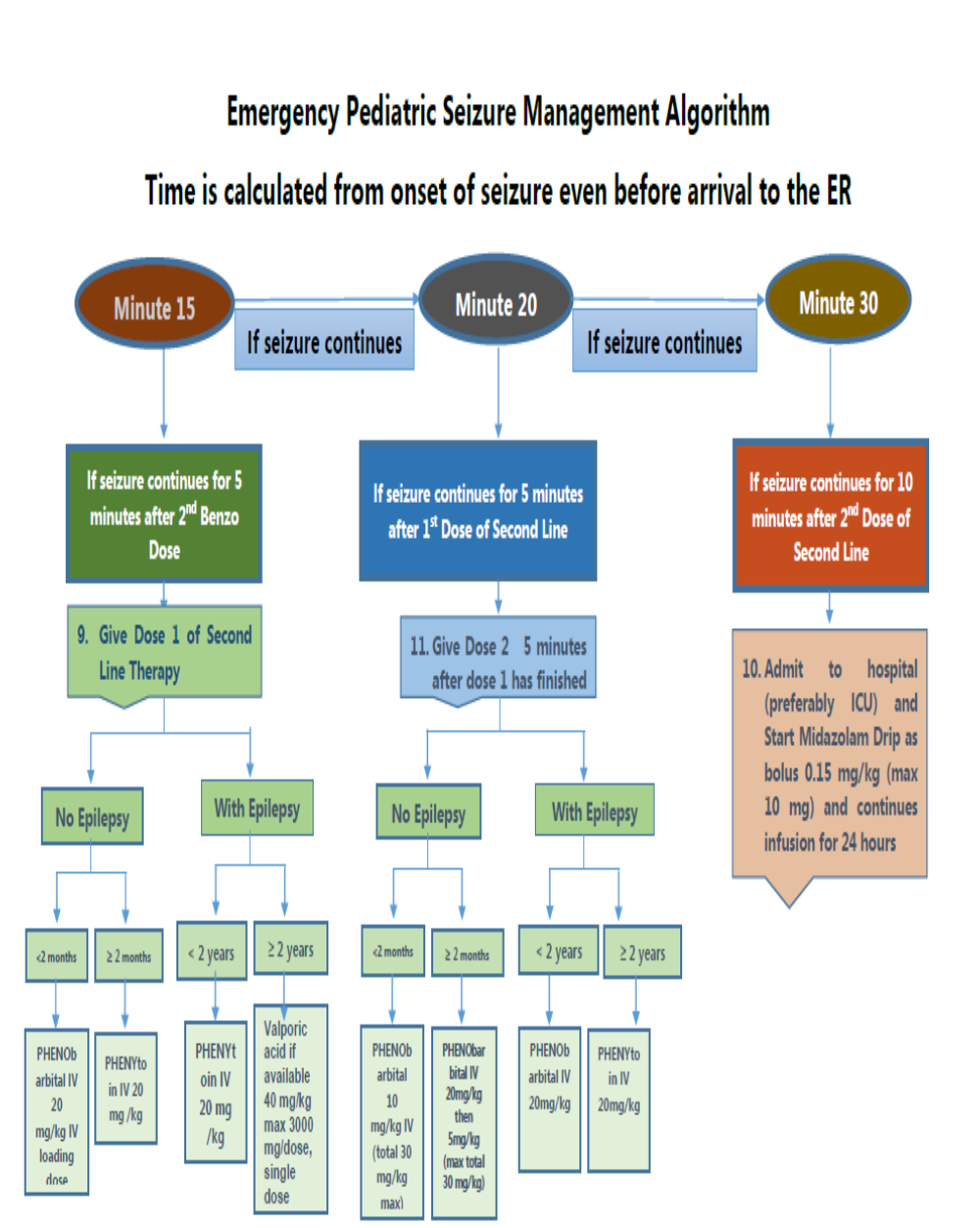
6. Drug Table
7. Table: Intermittent Drug Dosing in Emergency Seizure and Status Epilept
Management
|
Drug |
Initial Dosing |
Administration Rates and Alternative Drug Dosing Recommendations |
Serious Adverse Effects |
Considerations |
|
Diazepam |
IV 0.25 – 0.5 mg/kg max 10 mg/dose, may repeat in 5 min |
IV: Up to 5 mg/min (IVP), maximum 10 mg/dose
Pre-Hospital Treatment: - 2–5 years, 0.5 mg/kg (rectal); - 6–11 years, 0.3 mg/kg (PR); - greater than 12 years, 0.2 mg/kg (rectal) - maximum 20 mg/dose |
Hypotension Respiratory depression signs (hypo-ventilation, pallor, Decrease oxygen saturation. Respiratory support and intubation may be needed). |
- diazepam could be repeated once after 5 minutes convulsion continued - History of pre-hospital use of benzodiazepines must be considered as well as timing. - doses given within one hour before ER presentation must be regarded as an initial dose - Rapid redistribution (short duration), the active metabolite, - IV contains propylene glycol |
|
Midazolam |
0.2 mg/kg IM up to maximum of 10 mg |
10 mg IM (>40 kg); 5 mg IM (13–40 kg);
0.2 mg/kg max 10 mg/dose, (buccal)
|
Hypotension Respiratory depression signs (hypo-ventilation, pallor, Decrease oxygen saturation. Respiratory support and intubation may be needed). |
- Midazolam is the most appropriate type of benzodiazepines to be used - The whole full dose to be given as a single dose better than broken into multiple doses) - The active metabolite, renal elimination, rapid redistribution (short duration) |
|
Phenytoin |
Initial loading dose :20 mg/kg IV, may give an additional 5–10 mg/kg, maximum 1500 mg/dose 10 minutes after the initial loading dose
Maintenance dose: 4-8 mg/kg/day IV divided twice daily |
up to 1 mg/kg/min (if<50kg: give over 20 min, if >50 kg give at rate 50 mg/ min
|
Arrhythmias Hypotension Purple glove syndrome |
- It is preferred to phenobarbital as it has fewer side effects in terms of cardiovascular depression, sedation, and respiratory depression than phenobarbital given after benzodiazepines. - It should be infused using independent venous access in a large-caliber vessel to reduce the risk of phlebitis. It is recommended to monitor heart rate and blood pressure( cardiac arrhythmias, hypotension). Purple glove syndrome, Stevens-Johnson - Phenytoin is contraindicated in patients with Grade II atrioventricular block or severe hypotension - Only compatible in saline in a concentration not exceeding 10 mg/ml - IV contains propylene glycol - If already on phenytoin, halve the loading dose |
|
Phenobarbitone |
20 mg/kg IV, may give an additional 5–10 mg/kg
|
50–100 mg/min IV, may give additional dose 10 min after loading infusion
Must not be faster than 1 mg/ kg/ min (at least 20 min), Must be diluted with water to a concentration, not exceeding10 mg/ ml
(If <30 kg infuse over 20 min) , if > 30 kg infuse at 30 mg / min |
Hypotension Respiratory depression |
- IV contains propylene glycol - If already on phenobarbitone, halve the above loading dose of that drug. - Monitoring of heart rate, electrocardiography, and blood pressure for risk of respiratory depression and hypotension. It should be administered with a resuscitator and/ or trained medical personnel to support advanced ventilation - Respiratory depression is the most clinically significant treatment-emergent adverse event with anticonvulsant drugs in status epilepticus |
|
Valproate |
20–40 mg/kg IV, may give an additional 20 mg/kg |
1.5–3 mg/kg/min Maximum 3000 mg/dose. Single-dose |
Hyperammonemia Pancreatitis Thrombocytopenia Hepatotoxicity |
- Use with caution in patients with traumatic head injury; maybe a preferred agent in patients with glioblastoma multiforme |
|
Pyridoxine |
Loading: administering 100 mg of pyridoxine intravenously If clinical response is not demonstrated, the dose should be repeated every 5 – 25 min up to a maximum of 500 mg.
|
while monitoring the EEG, oxygen saturation, and vital signs In individuals with pyridoxine-dependent epilepsy, clinical seizures generally cease over several minutes. A corresponding change should be observed in the EEG; in some circumstances, the change may be delayed by several hours. |
In some individuals with pyridoxine-dependent epilepsy, significant neurologic, and cardiorespiratory depression follows this trial, making close systemic monitoring essential.
|
|
|
Levetiracetam |
20–60 mg/kg IV twice daily
|
bolus of 13–70 mg/kg – 60 mg/kg, maximum 4500 mg/dose, single-dose, (maximum dose 4 g), typical starting dose 30 mg/kg in 15-min intravenous infusion (from 5 to 60 min) for a total of 100 ml (but at high concentration and low volume: 50 mg/ml) Keppra Injection is for intravenous use only and should be diluted in 100 mL of a compatible diluent before administration. If a smaller volume is required (e.g. pediatric patients), the amount of diluent should be calculated to not exceed a maximum levetiracetam concentration of 15 mg per mL of diluted solution. Consideration Diluents: Sodium chloride (0.9%) injection, USP Lactated Ringer's injection Dextrose 5% injection, USP 2–5 mg/kg/min IV |
The following adverse reactions may be observed: - Behavioral Abnormalities and Psychotic Symptoms - Somnolence and Fatigue - Anaphylaxis and Angioedema - Serious Dermatological Reactions - Coordination Difficulties - Hematologic Abnormalities - Increase in Blood Pressure |
- Minimal drug interactions - Not hepatically metabolized - Levetiracetam in the oral formulation can be administered through a nasogastric tube. - In cases where it was effective, seizures stopped in 25– 30 min after intravenous injection and in 1.5 days when given through a nasogastric tube. - The following calculation should be used to determine the appropriate daily dose of Keppra Injection for pediatric patients - Total daily dose (mL/day) = Daily dose (mg/kg/day) × patient weight (kg)/ 100 mg/mL |
- research needs
Future research recommendations for the Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period in the Egyptian context could include:
· Further studies on pharmacological & surgical role in convulsions in children.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for seizures in children.
➡️Challenges
· Availability of anti-convulsions drugs in pediatrics’ emergency rooms.
· Family education about anti-convulsions.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline
The following are three performance measures or indicators for implementing this adapted CPG for Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period:
1. Adherence to Emergency Management (Diagnosis and Treatment) of Seizures in Children Guidelines
· Numerator: Number of children with Seizures who received treatment as per guideline recommendations.
· Denominator: Total number of children diagnosed with Seizures.
· Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
· Numerator: Total number of hospital stay days for children with Seizures.
· Denominator: Total number of children admitted with Seizures.
· Data Source: Hospital admission and discharge records.
3. Rate of Readmission
· Numerator: Number of children readmitted with symptoms of Seizures within a certain period (e.g., 30 days) after discharge.
· Denominator: Total number of children initially admitted with Seizures.
· Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
➡️Updating of the guideline
The EPG Egyptian Childhood Seizure and Epilepsy Working Group has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Fisher R S, van Emde Boas W, Blume W, et al. Epileptic seizures and epilepsy: Definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE) . Epilepsia 2005 ; 46 : 470 - 472
2. Glauser T A. Designing practical evidence - based treatment plans for children with prolonged seizures and status epilepticus. J Child Neurology 2007 ; 22 (suppl) 385 - 465
3. Beghi E, Carpio A, Forsgren L, et al. Recommendation for a definition of acute symptomatic seizure. Epilepsia 2010 ; 51 : 671 - 675
4. Harding G. The reflex epilepsia with emphasis on photosensitive epilepsy. Suppl Clin Neurophysiol 2004 ; 57: 433 - 438
5. Leung A K, Robson W L. Febrile seizures J Pediatr Health Care. 2007 ; 21 (4) : 250 - 255
6. Wilden J A, Cohen-Dadol A. Evaluation of first nonfebrile seizures , Am Fam Physician 2012 ;86 : 334 - 340
7. Stronik H, Brouwer O F, Arts W F, et al. The first unprovoked, untreated seizure in childhood: a hospital based study of the accuracy of the diagnosis, rate of recurrence, and long term outcome after recurrence. Dutch study of epilepsy in childhood. J Neurol Neurosurg Psychiatry 1998 ; 64 : 595 - 600
8. Berg A T , Berkovic S F , Brodie M J , et al (2010): Revised terminology and concepts for organization of seizures and epilepsies : Report of the ILAE Commission on Classification and Terminology , 2010; 51: 676 - 685
9. Fisher RS, Acevedo C, Arzimanoglou et al, (2004) A practical clinical definition of epilepsy. Epilepsia ; 55 (4) : 475 - 482 , 2014
10. Treatment of convulsive status epilepticus: Recommendation of the Epilepsy Foundation of America’s Working Group on status Epilepticus JAMA 1993 ; 270 : 854 - 859
11. Shinnar S, Berg A T, Moshe S L, Shinnar R. How long do new - onset seizures in childhood last? Ann Neurol 2001 ; 49 : 659 - 664
12. De Lorenzo R J , Hauser W A , Towne A R , Boggs J G ,Pellock J M , Penberthy L , Garnett L , Fortner C A , Ko D . A prospective, population - based epidemiological study of status epilepticus in Richmond, Virginia. Neurology 1996 ; 46 : 1029 - 1035
13. De Lorenzo R J , Hauser W A , Towne A R , Boggs J G , Pellock J M , Penberthy L , Garnett L , Fortner C A , Ko D . A prospective, population - based epidemiologic study of status epilepticus in Richmond, Virginia. Neurology 2002 ; 58 : 1070 - 1076
14. Wu Y W, Shewk D W, Garcia P A, Zhao S, Johnston S C. Incidence and mortality of generalized convulsive status epilepticus in California Neurology 2002; 58 : 1070 - 1076.
15. Hauser W A. Status epilepticus: Epidemiologic considerations. Neurology 1990 ; 40 : 9 - 13
16. Malmgren K, Stödberg T, Amandusson Å, Strandberg M, Diagnosing epileptic seizures and epilepsy Lakartidningen. 2018 May 21; 115. pii: E47D.
17. Horrocks IA1, Nechay A, Stephenson JB, Zuberi SM. Anoxic-epileptic seizures: observational study of epileptic seizures induced by syncopes. Arch Dis Child. 2005 Dec; 90(12):1283-7. Epub 2005 Sep 13
18. Texas children’s hospital, evidence-based outcomes center, Initial Management of Status Epilepticus Evidence-Based Guideline, July 2018
19. Kabakus, N. Abdullah K., Sandifer Syndrome: A continuing problem of misdiagnosis Pediatrics international, 22 November 2006 https://doi.org/10.1111/j.1442-200X.2006.02280.x
20. Nanvaakkara C. S., & Paton J. Y Sandifer Syndrome an Overlooked Diagnosis? Developmental Medicine & Child Neurology (DMCN) First published: December 1985 https://doi.org/10.1111/j.1469-8749.1985.tb03808.x
21. Nordli D. Jr. In Swaiman K., Ashwal S., Ferriero D., & Schor N., Focal and Multifocal Seizures, In Swaimans Pediatric Neurology 2012 p.753
22. Benbadis SR, Chen S, Melo M. What's shaking in the ICU? The differential diagnosis of seizures in the intensive care setting. Epilepsia 2010; 51(11):2338-40. PMID 20738384
23. Camfield P., and Camfield C., Pediatric Epilepsy: An Overview. In Swaimans Pediatric Neurology 2012 p707
24. Kozlowska K., Chudleigh C., Cruz C., Lim M., McClure G., Savage B., Shah U., Cook A., Scher S., Carrive P., and Gill P. Psychogenic non-epileptic seizures in children and adolescents: Part II – explanations to families, treatment, and group outcomes, Clin Child Psychol Psychiatry. 2018 Jan; 23(1): 160–176.
25. Morton L. and Pellock J., Status Epilepticus in. In Swaimans Pediatric Neurology 2012 p803
26. Culleton S., Marios S., Kaliakatsos M., Pujar S., D'Arco F., The spectrum of neuroimaging findings in febrile infection‐related epilepsy syndrome (FIRES): A literature review First published: 10 March 2019 https://doi.org/10.1111/epi.14684
27. McAbee GN, Wark JE. A practical approach to uncomplicated seizures in children. American family physician. 2000;62(5):1109-16.
28. Martindale JL, Goldstein JN, Pallin DJ. Emergency department seizure epidemiology. Emergency medicine clinics of North America. 2011;29(1):15-27.
29. Chaudhary N, Gupta MM, Shrestha S. Clinicodemographic Profile of Children with Seizures in a Tertiary Care Hospital: A Cross-Sectional Observational Study. 2017;2017:1524548.
30. Pallin DJ, Goldstein JN, Moussally JS, et al. Seizure visits in US emergency departments: epidemiology and potential disparities in care. Int J Emerg Med 2008;1(2):97–105.
31. Annegers JF, Hauser WA, Lee JR, et al. Incidence of acute symptomatic seizures in Rochester, Minnesota, 1935–1984. Epilepsia 1995;36(4):327–33.
32. Kotsopoulos IA, van Merode T, Kessels FG, et al. Systematic review and meta-analysis of incidence studies of epilepsy and unprovoked seizures. Epilepsia 2002;43(11):1402–9.
33. Sharma S, Riviello JJ, Harper MB, et al. The role of emergent neuroimaging in children with new-onset afebrile seizures. Pediatrics 2003; 111(1):1–5.
34. Ong S, Talan DA, Moran GJ, et al. Neurocysticercosis in radiographically imaged seizure patients in U.S. emergency departments. Emerg Infect Dis 2002; 8(6): 608–13.
35. Verity CM, Golding J. Risk of epilepsy after febrile convulsions: a national cohort study. BMJ 1991; 303(6814):1373–6.
36. Berg AT. Febrile seizures and epilepsy: the contributions of epidemiology. Paediatr Perinat Epidemiol 1992;6(2):145–52
37. Farrar HC, Chande VT, Fitzpatrick DF, et al. Hyponatremia as the cause of seizures in infants: a retrospective analysis of incidence, severity, and clinical predictors. Ann Emerg Med 1995; 26(1):42–8.
38. Krumholz A, Grufferman S, Orr ST, et al. Seizures and seizure care in an emergency department. Epilepsia 1989; 30(2):175–81.
39. Sastry CPVR, Reddy RM. Study of prevalence and clinical spectrum of seizures in children in a teaching hospital in rural Telangana, India. Int J Contemp Pediatr 2018; 5:862-6.
40. Mwipopo EE, Akhatar S, Fan P, Zhao D. Profile and clinical characterization of seizures in hospitalized children
41. Huff JS, Morris DL, Kothari RU, et al. Emergency department management of patients with seizures: a multicenter study. Acad Emerg Med 2001; 8(6):622–8.
42. DeLorenzo RJ, Hauser WA, Towne AR, et al. A prospective, population-based epidemiologic study of status epilepticus in Richmond, Virginia. Neurology 1996; 46(4):1029–35.
Reference epidemiology with special reference to Egypt:
43. Reference epidemiology with special reference to Egypt. The global burden of disease: 2004 update. Geneva, World Health Organization, 2004(http://www.who.int/healthinfo/global_burden_disease/2004_report_update/en/index.html, accessed 22 January 2010).
44. Attia-Romdhane N, Mrabet A, Ben Hamida M. Prevalence of epilepsy in Kelibia, Tunisia. G, 1993, 34(6):1028–1032.
45. The work of WHO in the Eastern 1. Mediterranean Region. Annual report of the Regional Director, 1 January – 31 December 2008. Cairo, WHO Regional Office for the Eastern Mediterranean, 2007.
46. Devender Bhalla., Elham Lotfalinezhad, Utsav Timalsina, Saloni Kapoor, Kailash Suresh Kumar, Abdallah Abdelrahman, Brenda Giagante, Manjari Tripathi, Kavita Srivastava, Irmansyah Irmansyah. A comprehensive review of epilepsy in the Arab world. Seizure 2016 ; 34: 54-59.
47. El-Tallawy HN1, Farghaly WM, Shehata GA, Abdel-Hakeem NM, Rageh TA, Abo-Elftoh NA, Hegazy A, Badry R. Epidemiology of epilepsy in New Valley Governorate, Al Kharga District, Egypt. Epilepsy Res. 2013;104(1-2):167-74
48. Khedr EM, Shawky OA, Ahmed MA, Elfetoh NA, Al Attar G, Ali AM, Kandil MR, Farweez H. A community based epidemiological study of epilepsy in Assiut Governorate/Egypt. Epilepsy Res. 2013; 103(2-3):294-302
49. Wafaa M.Farghaly, Mohamed A. And Elhamed, Inas M. Hassan, Wael T. Soliman, Mohamed A. Yehia, Nermeen A. Hamdy. Prevalence of Childhood and Adolescence Epilepsy in Upper Egypt (Desert Areas). The Egypt J Neurol, Psychiatr Neurosurg 2018, 54(34): 1 – 7.
50. Hashem S., Al-Kattan M, Ibrahim SY, Shalaby NM, Shamloul RM, Farrag M. Epilepsy Prevalence in Al-Manial Island, Egypt: A door to door survey. Epilepsy Res 2015; 117: 133 – 7.
51. Ghaydaa A. Shehata. A review of epilepsy stigma in Egypt. Acta Psychopathologica 2016; 2(2): 13: 1 – 7
52. Zeinab M. Monir, Inas R. EL-Alameey, Eman Eltahlawy. Health related quality of life of children with epilepsy in Egypt. J Arab Soc Med Res 2013; 8:53–66.
References: classification :
53. Panayiotopoulos C.P. The Epilepsies: Seizures, Syndromes and Management. Oxfordshire (UK): Bladon Medical Publishing; 2005.
54. Fisher R.S. The New Classification of Seizures by the International League Against Epilepsy. Article in Current Neurology and Neuroscience Reports 17(6); June 2017.
55. Nickels K. How Have We Come This Far? Epilepsy Classification through the Ages. Epilepsy Curr. 17(4): 229–231, 2017.
56. Pack A.M. Epilepsy Overview and Revised Classification of Seizures and Epilepsies. Continuum (Minneap Minn); 25(2):306-321, 2019.
57. Fisher R.S., Cross J.H., D’Souza C., French J.A., Haut S.R., Higurashi N., Hirsch E., Jansen F.E., Lagae L., Moshe S.L., Peltola J., Perez E.R., Scheffer I.E., Schulze-Bonhage A., Somerville E., Sperling M., Yacubian E.M., and Zuberi S.M.on behalf of the ILAE Commission for Classification and Terminology. Instruction manual for the ILAE 2017 operational classification of seizure types. Epilepsia, 58(4):531–542, 2017.
58. Stafstrom CE. And Carmant L. Seizures and Epilepsy: An Overview for Neuroscientists. Cold Spring Harb Perspect Med.; Jun; 5(6): a022426 (2015).
59. Scheffer I.E., Berkovic S., Capovilla G., Connolly M.B.,et al. ILAE Classification of the Epilepsies Position Paper of the ILAE Commission for Classification and Terminology. Epilepsies. 58(4): 512–521, 2017.
60. Brodie M.J., Zuberi S.M., Scheffer I.E.,Fisher R.S. The 2017 ILAE classification of seizure types and the epilepsies: what do people with epilepsy and their caregivers need to know? Epileptic Disord; 20 (2): 77-87, 2018.
61. Chang R.S., Leung C.Y.W., Ho C.C.A. And Yung A. Classifications of seizures and epilepsies, where are we? – A brief historical review and update. Journal of the Formosan Medical Association; Vol 116, Issue 10, 736-741, 2017.
62. Berg A.T., Berkovic S.F., Brodie M.J., Buchhalter J., Cross J.H., van Emde Boas W., Engel J., French J., Glauser T.A., Mathern G.W., et al. Revised terminology and concepts for organization of seizures and epilepsies: Report of the ILAE Commission on Classification and Terminology; Epilepsia. 2010; 51:676–685, 2009.
63. Trinka E., Cock H., Hesdorffer D., Rossetti A. O., Scheffer I. E., Shinnar S., Shorvon S., Lowenstein D. H. A definition and classification of status epilepticus--Report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia; 56(10):1515-23, 2015.
REFRENCES complications :
64. Laboratory markers of cardiac and metabolic complications after generalized tonic-clonic seizures. Robert D. Nass,Sina Meiling , René P. Andrié, Christian E. Elger1 and Rainer Surges.BMC Neurology (2017) 17:187 DOI 10.1186/s12883-017-0965-4
65. StatusEpilepticus risk factors and complications. By Dr. Liji Thomas, MD. Reviewed by Afsaneh Khetrapal, BSc Saved from URL: https://www.news-medical.net/health/Status-Epilepticus-Risk-Factors-and-Complications.aspx
66. The comprehensive evaluation and treatment of epilepsy. Byrislane FW. Status epilepticus. In:Schachter SC, Schomer DL. EdsSan Diego, CA: Academic Press: 1997. P. 149-172. With permission from Elsevier (www.elsevier.com).
67. Febrile seizures associated with risk of SUDEP in patients with chronic epilepsy? Terra VC, Machado HR, Sakamoto AC, Arida RM, Scorza FA. Arq Neuropsiquiatr. 2011; 69 (2B): 384-6.
References for lab investigations :
68. Elizabeth G, and Howard J: Seizures and epilepsy in the acute medical setting: presentation and management, Clinical Medicine 2018 Vol 18, No 5: 409–13ع^
69. Hirtz D, Ashwal S, Berg A, Bettis D, Camfield C, Camfield P, et al. Practice parameter: evaluating a first non-febrile seizure in children: report of the quality standards subcommittee of the American Academy of Neurology, The Child Neurology Society, and The American Epilepsy Society. Neurology 2000; 55:616–623.
70. Raffaele N, Francesco B, and Eugen T; Acute Symptomatic Seizures Caused by Electrolyte Disturbances. J Clin Neurol. 2016 Jan; 12(1):21-33.
71. Joseph I, Sirven M, & Elizabeth W. Management of status epilepticus, Am Fam Physician. 2003 Aug 1; 68(3):469-476.
72. J. J. Riviello, Jr, S. Ashwal, D. Hirtz, et al: Diagnostic assessment of the child with status epilepticus (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society, Neurology 2006;67;1542-1550
References management :
73. Ciccone, O, M Mathews, and GL Birbeck. 2017. Management of acute seizures in children: A review with special consideration of care in resource-limited settings. African Journal of Emergency Medicine 7:s3-s9.
74. WHO Guidelines Approved by the Guidelines Review Committee. 2016. Guideline: Updates on Paediatric Emergency Triage, Assessment and Treatment: Care of Critically-Ill Children. In Guideline: Updates on Paediatric Emergency Triage, Assessment and Treatment: Care of Critically-Ill Children. eneva.
75. WHO Review Committee. 2016. mhGAP Intervention Guide for Mental, Neurological and Substance Use Disorders in Non-Specialized Health Settings: Mental Health Gap Action Programme (mhGAP): Version 2.0. In mhGAP Intervention Guide for Mental, Neurological and Substance Use Disorders in Non-Specialized Health Settings: Mental Health Gap Action Programme (mhGAP): Version 2.0, edited by W. G. A. b. t. G. R. Committee. Geneva.
76. Hategeka, C., L. Mwai, and L. Tuyisenge. 2017. Implementing the Emergency Triage, Assessment and Treatment plus admission care (ETAT+) clinical practice guidelines to improve quality of hospital care in Rwandan district hospitals: healthcare workers' perspectives on relevance and challenges. BMC Health Serv Res 17 (1):256.
77. Bashiri, F. A., M. H. Hamad, Y. S. Amer, M. M. Abouelkheir, S. Mohamed, A. Y. Kentab, M. A. Salih, M. N. Al Nasser, A. A. Al-Eyadhy, M. A. Al Othman, T. Al-Ahmadi, S. M. Iqbal, A. M. Somily, H. A. Wahabi, K. J. Hundallah, A. H. Alwadei, R. S. Albaradie, W. A. Al-Twaijri, M. M. Jan, F. Al-Otaibi, A. M. Alnemri, and L. A. Al-Ansary. 2017. Management of convulsive status epilepticus in children: an adapted clinical practice guideline for pediatricians in Saudi Arabia. Neurosciences (Riyadh) 22 (2):146-155.
78. Glauser, T., S. Shinnar, D. Gloss, B. Alldredge, R. Arya, J. Bainbridge, M. Bare, T. Bleck, W. E. Dodson, L. Garrity, A. Jagoda, D. Lowenstein, J. Pellock, J. Riviello, E. Sloan, and D. M. Treiman. 2016. Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society. Epilepsy Curr 16 (1):48-61.
References Prognosis :
79. Wo SB, Lee JH, Lee YJ, et al. Risk for developing epilepsy and epileptiform discharges on EEG in patients with febrile seizures. Brain Dev 2013; 35:307.
80. Pavlidou E, Panteliadis C. Prognostic factors for subsequent epilepsy in children with febrile seizures. Epilepsia 2013; 54:2101.
81. Martin ET, Kerin T, Christakis DA, et al. Redefining outcome of first seizures by acute illness. Pediatrics 2010; 126:e1477.
82. Shinnar S, Berg AT, Moshé SL, et al. Risk of seizure recurrence following a first unprovoked seizure in childhood: a prospective study. Pediatrics 1990; 85:1076.
83. Shinnar S, O'Dell C, Berg AT. Mortality following a first unprovoked seizure in children: a prospective study. Neurology 2005; 64:880.
84. Garcia Pierce J, Aronoff S, Del Vecchio M. Systematic Review and Meta-analysis of Seizure Recurrence after a First Unprovoked Seizure in 815 neurologically and Developmentally Normal Children. J Child Neurol 2017; 32:1035.
85. Arthur TM, deGrauw TJ, Johnson CS, et al. Seizure recurrence risk following a first seizure in neurologically normal children. Epilepsia 2008; 49:1950.
86. Vestergaard M, Pedersen MG, Ostergaard JR, et al. Death in children with febrile seizures: a population-based cohort study. Lancet 2008; 372:457.
87. Holm IA, Poduri A, Crandall L, et al. Inheritance of febrile seizures in sudden unexplained death in toddlers. Pediatr Neurol 2012; 46:235
88. Leaffer EB, Hinton VJ, Hesdorffer DC. Longitudinal assessment of skill development in children with first febrile seizure. Epilepsy Behav 2013; 28:83.
89. Raspall-Chaure, M., Chin, R.F., Neville, B.G., et al., 2006. Outcome of paediatric convulsive status epilepticus: a systematic review. Lancet Neurol. 5, 769–779.
90. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
91. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
92. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
93. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
94. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
95. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
96. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
97. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024).
مراجع جزء رسائل الأمهات
98. Web MD. Epilepsy. Retrieved on the 23rd of July, 2019, From: https://www.webmd.com/epilepsy/default.htm
99. Jeanne Morisson. Epilespsy. Retrieved on the 23rd of July, 2019, From: https://www.healthline.com/health/epilepsy#epilepsy-and-behavior
100. Neil Lava. Types of Seizures and Their Symptoms. Retrieved on the 23rd of July, 2019, From: https://www.webmd.com/epilepsy/types-of-seizures-their-symptoms#1
101. Epilepsy.com. What is Epilepsy?. Retrieved on the 23rd of July, 2019, From: https://www.epilepsy.com/learn/about-epilepsy-basics/what-epilepsy
102. https://gigglemagazine.com/children-and-febrile-seizures/
103. https://www.epilepsy.ie/content/seizure-first-aid
104. https://www.cdc.gov/epilepsy/groups/parents.htm
105. http://www.epilepsy.org.au/
106. Video for GTC https://www.youtube.com/watch?time_continue=4&v=WP4NZcfadIk&feature=emb_logo.
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Abdelsatr Abdallah Elsayeh |
Assistant professor of Pediatrics and Pediatric Neurology Faculty of Medicine for Males Al-Azhar University |
None |
Not Applicable |
|
Ashraf Azmy Al Refaei |
Professor of Child Health and Pediatric Neurology National Research Center |
None |
Not Applicable |
|
Azza Mohamed Elamin Oraby (late professor) |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Cairo University |
None |
Not Applicable |
|
Ebtesam Hussein El Melegy |
Consultant Pediatric Neurologist Former Dean of the Neuromotor Institute Ministry of Health |
None |
Not Applicable |
|
Hammouda Eid El Gazzar |
Fellow and Lecturer in Pediatrics General Organization of Teaching Hospitals Under-secretary of El Beheira Health Directorate |
None |
Not Applicable |
|
Hayam Refaat Tantawi |
Professor of Pediatric Nursing Vice Dean of Community and Environmental Development Faculty of Nursing Ain Shams University |
None |
Not Applicable |
|
Lobna Abdel Gawad Mansour |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Cairo University |
None |
Not Applicable |
|
Marwa Saeed Abd Elmaksod |
Assistant professor of Pediatrics and Pediatric Neurology Faculty of Medicine Alexandria University |
None |
Not Applicable |
|
Moustafa Zakaryia Mohamed |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Cairo University |
None |
Not Applicable |
|
Omnia Fathy El Rashidy
|
Professor of Pediatrics and Pediatric Neurology and Neuro-disability Faculty of Medicine Ain Shams University |
None |
Not Applicable |
|
Sahar Mohamed Ahmed Hassanein |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Ain Shams University |
None |
Not Applicable |
|
Tarek El-Sayed Ismail Omar |
Professor of Pediatrics and Pediatric Neurology Faculty of Medicine Alexandria University |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Professor Hadeer Mahmoud Gamal Eldin Abdelghaffar |
Professor and Head of Pediatrics Department – Consultant Pediatric Neurologist – Faculty of Medicine - Fayoum University |
None |
Not Applicable |
|
Professor Hoda Yehia Tomoum |
Professor of Pediatrics and Pediatric Neurology – Faculty of Medicine – Ein Ain Shams University |
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
None |
Not Applicable |
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period in the Egyptian " on PubMed are: convulsions, seizures, pediatric convulsions.

Annex Table. Annex Nurses and Parents Educational Guide in Arabic
ارشادات هامة للتعامل مع النوبات الصرعية الكبرى
مقدمة
الصرع هو مرض مزمن يظهر على شكل تشنجات دماغية متكررة أو اختلاجات دماغية. مرض الصرع ليس إعاقة عقلية ولا مرضاً نفسياً وهو غير معدي، و قد يصاحب بعض أمراض الدماغ الأخرى وهو مرض يصيب الدماغ وينتج عنه نشاط كهربائي غير طبيعي يؤدي إلى نوبات والى سلوكيات وأحاسيس غير معتادة وأحياناً ما يتسبب في فقدان الوعي. وتترتب على الصرع آثار عصبية و ادراكية ونفسية واجتماعية
قد تختلف أعراض نوبة الصرع حسب نوع النوبة، ووقت حدوث هذه الأعراض، حيث تسبق بعض الأعراض النوبة وتكون بمثابة أعراض تحذيرية، بينما تتزامن الأعراض الأخرى مع حدوث النوبة أو تليها.
فيما يلي أهم أعراض نوبات التشنج أو الصرع العامة generalized seizures
· يسبقها أحياناً حالة الهالة التي قد تشمل هلاوس سمعية، أو شم روائح خيالية، أو ضيق بالبطن، أو شعور بالضغط داخل الدماغ.
· نوبات التشنج الكبرى العامة تبدأ بفقدان مفاجئ للوعي، وتيبس الجسم، وقد يصبح الجسم منتصباً مثل السيخ المعدني،
· أو قد ينحني كالقوس إلى الخلف وتحدث بعد ذلك سلسلة من حركات الارتجاف في الذراعين والساقين والجذع، مع الصر على الأسنان.
· التبول أو التبرز اللاإرادي عند البعض.
· عادة ما تستمر نوبة التشنج بالكامل إلى ما لا يزيد عن دقيقتين.
بعد نوبة الصرع، قد يشعر الشخص بالتشويش والإرهاق لعدة ساعات وقد يصاب بصداع وتنتابه رغبة في النوم. ولا يتذكر الشخص أنه أصيب بنوبة صرع، لكنه قد يتذكر نوبة الهالة التي سبقتها.
· يسبقها أحياناً حالة الهالة التي قد تشمل هلاوس سمعية، أو شم روائح خيالية، أو ضيق بالبطن، أو شعور بالضغط داخل الدماغ.
· نوبات التشنج الكبرى العامة تبدأ بفقدان مفاجئ للوعي، وتيبس الجسم، وقد يصبح الجسم منتصباً مثل السيخ المعدني،
· أو قد ينحني كالقوس إلى الخلف وتحدث بعد ذلك سلسلة من حركات الارتجاف في الذراعين والساقين والجذع، مع الصر على الأسنان.
· التبول أو التبرز اللاإرادي عند البعض.
· عادة ما تستمر نوبة التشنج بالكامل إلى ما لا يزيد عن دقيقتين.
بعد نوبة الصرع، قد يشعر الشخص بالتشويش والإرهاق لعدة ساعات وقد يصاب بصداع وتنتابه رغبة في النوم. ولا يتذكر الشخص أنه أصيب بنوبة صرع، لكنه قد يتذكر نوبة الهالة التي سبقتها.
· إذا حدثت نوبات صرعية كبرى لمدة أطول من خمس دقائق أو تكررت مرات عديدة متعاقبة دون أن يستيقظ الشخص فيما بينها، قد تهدد حياة المريض، ويجب بدء الاسعافات الأولية و نقله فورا إلى أقرب مشفى.
1. ما الذي يجب عمله أثناء إصابة الطفل بالنوبة؟
قد تكون لحظات فقدان الوعي أثناء النوبة قصيرة جدا وبالتالي هناك القليل الذي يمكن عمله للطفل أثناءها ومع هذا فإن مشاهدة حالة الصرع تجربة قاسية لكل شخص يتواجد حول الطفل إلا الطفل نفسه الذي يكون فاقدا للوعي تماما لما يدور حوله.
من المهم معرفة الطريقة الصحيحة للتعامل مع مريض الصرع أثناء النوبة، وذلك لتجنب حدوث أي مضاعفات أو حوادث ناجمة عن التعامل الخاطئ.
وفيما يلي نقدم لك بعض الإرشادات البسيطة حول ما يجب وما لا يجب عمله أثناء إصابة الطفل بالنوبة:
الإسعافات الأولية لنوبة الصرع:
- ساعد المصاب على الاستلقاء على الأرض. ضع الطفل في رقدته على جانبه، واجعل الرأس مائلا قليلا إلى الخلف للسماح للعاب بالخروج ولتمكينه من التنفس. وضع الافاقة
https://dmgpzc86o16rj.cloudfront.net/wp-content/uploads/2018/10/Screen-Shot-2018-10-01-at-11.27.45-AM.png
https://www.cdc.gov/epilepsy/images/groups/seizure-first-aid.jpg
- إرخاء الملابس الضيقة حول الرقبة.
- امنع الطفل من إيذاء نفسه – حاول أن تبعد أي شيء صلب أو حاد أو ساخن من الممكن أن يؤذي المصاب.
- تجنب وضع أي شيء في فم المصاب. لاتحاول إعطاءه أي دواء أثناء النوبة ولا تحاول إيقاظه منها.
- عندما يبدأ في استعادة وعيه ضعه على أحد جانبيه مع التأكد أن المصاب لا يعاني أي صعوبة في التنفس
- بعد انتهاء النوبة إذا لم يكن الشخص بمنزله وما زال يبدو مترنحا أو ضعيف فمن الأفضل مصاحبته إلى المنزل
- إذا استمرت النوبة لأكثر من خمس دقائق يجب عليك الاستعانة بمساعدة طبيبه في الحال.
- تذكر دائما أن الطفل بعد النوبة يكون متعبا أو خائفا، حاول أن تهدئ من روعه قدر استطاعتك .
- تذكر أن تسجيلك لحالة الطفل أثناء النوبة ومدة النوبة نفسها مفيد للطبيب المعالج .
- لا تدع المريض إلا بعد انقضاء النوبة فالمريض يكون لا يزال مضطرباً ومرتبكاً لعدة دقائق.
- ثمة دلائل تؤكد أن الذي حدث معه نوبة صرع:
غالباً المريض لا يتذكر ما حدث.
قام بقضم لسانه.
حدث له تبول.
1. كيفية إعطاء دواء الميدازولام بالفم أو الأنف Midazolam 5mg /ml ampoule
• يوضع الطفل في وضع الإفاقة.
• يسحب كمية الدواءالمحسوبة بواسطة الطبيب مسبقا في ارشاداته في سرنجة إنسولين وينزع السن. يمكن إستخدام أمبولات الميدازولام المتوفرة للحقن الوريدي أو العضلي حيث أنه غير متوفر في مصر امبولات مخصوصة.
• تدخل السرنجة بين الاسنان والخد السفلي ليمتص الدواء من الغشاء المخاطي للثة وليس عن طريق البلع.
• يتم الضغط على مبسم السرنجة برفق حتى الانتهاء من اعطاء الدواء.
• يترك الطفل في وضع الافاقة


Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|