Pharmacological Approach of Type 2 Diabetes Mellitus
| Site: | EHC | Egyptian Health Council |
| Course: | Diabetes and Endocrinology Guidelines |
| Book: | Pharmacological Approach of Type 2 Diabetes Mellitus |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 20 November 2025" Download Guideline
- Executive Summary
This guideline offers evidence-based recommendations on pharmacotherapy for type 2 diabetes in adults. The recommendations are intended to provide healthcare professionals with practical guidance on Pharmacological Approach of Type 2 Diabetes Mellitus and improving health outcomes for people living with Diabetes.
- Recommendations
· Healthy lifestyle behaviors, diabetes self-management education and support, avoidance of therapeutic inertia, and social determinants of health should be considered in the glucose-lowering management of type 2 diabetes.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)[i]’[ii]’[iii]
· The glucose-lowering treatment plan may consider approaches that support weight management goals for adults with type 2 diabetes.
Conditional Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)[iv]’ [v]
· For adults with type 2 diabetes, use pharmacological strategies that provide sufficient effectiveness to achieve and maintain the intended treatment goals.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)[vi],[vii]
· Treatment modification (intensification or de-intensification) for adults not meeting individualized treatment goals should not be delayed.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)9,[viii]
· Medication plan and medication-taking behavior should be reevaluated at regular intervals (e.g., every 3–6 months) and adjusted as needed to incorporate specific factors that impact choice of treatment.
Strong Recommendation
Moderate Quality Evidence (based on observational studies)10,[ix]
· Early combination therapy (oral or injectable) can be considered in adults with type 2 diabetes at treatment initiation to shorten time to attainment of individualized treatment goals.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)9,11
· In adults with type 2 diabetes without cardiovascular and/or kidney disease, pharmacologic agents should address both the individualized glycemic and weight goals.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)11
· In adults with type 2 diabetes who have not achieved their individualized glycemic goals, selection of subsequent glucose-lowering agents should take into consideration the individualized glycemic and weight goals as well as the presence of other metabolic comorbidities and the risk of hypoglycemia.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)[x]
· In adults with type 2 diabetes with established or high risk of atherosclerotic cardiovascular disease, HF and/ or CKD an SGLT2 inhibitor or GLP-1RA is recommended, for glycemic management, improving cardiac and renal outcome and prevention of HF hospitalizations.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)[xi]
· In adults with type 2 diabetes who have CKD (with confirmed estimated glomerular filtration rate [eGFR] of 20–60 mL/min per 1.73 m2 and/or albuminuria), an SGLT2 inhibitor should be used for minimizing progression of CKD, reduction in cardiovascular events, and reduction in hospitalizations for HF; however, the glycemic benefits of SGLT2 inhibitors are reduced at eGFR <45 mL/min per 1.73 m2.
Strong Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)13
· In adults with type 2 diabetes, initiation of insulin should be considered regardless of background glucose-lowering therapy or disease stage if there is evidence of ongoing catabolism (e.g., unexpected weight loss), if symptoms of hyperglycemia are present, or when A1C or blood glucose levels are very high (i.e., A1C >10% or blood glucose ≥300 mg/dL)
Conditional Recommendation (against)
Moderate Quality Evidence (based on observational studies)[xii]
· In adults with type 2 diabetes, glucose-lowering agents may be continued upon initiation of insulin therapy (unless contraindicated or not tolerated) for ongoing glycemic and metabolic benefits (i.e., weight, cardio-metabolic, or kidney benefits).
Conditional Recommendation
High Quality Evidence (based on systematic reviews, randomized controlled trials, and multiple observational studies with consistent effects)14
· To minimize the risk of hypoglycemia and treatment burden when starting insulin therapy in adults with type 2 diabetes, reassess the need for and/or the dose of glucose-lowering agents with higher hypoglycemia risk (i.e., sulfonylureas and meglitinides).
Conditional Recommendation
moderate Quality Evidence (based and multiple observational studies with consistent effects)[xiii]
· Monitor for signs of overbasalization during insulin therapy, such as basal dose exceeding 0.5 units/kg/day, When overbasalization is suspected, a thorough reevaluation could occur promptly to further tailor therapy to the individual’s needs.
Conditional Recommendation
Moderate Quality Evidence (based on observational studies)14
· Routinely assess all people with diabetes for financial obstacles that could impede their diabetes management. Clinicians, members of the diabetes care team, and social services professionals should work collaboratively, as appropriate and feasible, to support these individuals by implementing strategies to reduce costs, thereby improving their access to evidence-based care.
Conditional Recommendation
Moderate Quality Evidence (based on observational studies)[xiv]
· In adults with diabetes and cost-related barriers, consider use of lower-cost medications for glycemic management within the context of their risks for hypoglycemia, weight gain, cardiovascular and kidney events, and other adverse effects.
Conditional Recommendation
Moderate Quality Evidence (based on observational studies)[xv]
· In adults with type 2 diabetes, metabolic dysfunction–associated steatotic liver disease (MASLD), and overweight or obesity, consider using a GLP-1 RA or a dual glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 RA with potential benefits in metabolic dysfunction–associated steatohepatitis (MASH) for glycemic management and as an adjunctive to healthy interventions for weight loss.
Conditional Recommendation
Moderate Quality Evidence (based on randomized clinical trials)[xvi],[xvii], [xviii], [xix], [xx], [xxi], [xxii],[xxiii].
· In adults with type 2 diabetes and biopsy-proven MASH or those at high risk for liver fibrosis (based on noninvasive tests), pioglitazone, a GLP-1 RA, or a dual GIP and GLP-1 RA is preferred for glycemic management due to potential beneficial effects on MASH.
· Combination therapy with pioglitazone plus a GLP-1 RA can be considered for the treatment of hyperglycemia in adults with type 2 diabetes with biopsy-proven MASH or those at high risk of liver fibrosis (identified with noninvasive tests) due to potential beneficial effects on MASH.
Conditional Recommendation
Moderate Quality Evidence (based on randomized clinical trials)[xxiv],[xxv],- Acknowledgement
We would like to acknowledge the Diabetes and Endocrinology Scientific Committee for developing these guidelines.
Chair of the GDG: Prof. Mohamed Hesham El Hefnawy, National Institute of Diabetes and Endocrinology, Cairo
Members of the Guideline Development Group (GDG):
- Mohamed Abdelhady Mohamed Mashahet, Faculty of Medicine, Fayoum University, Fayoum.
- Athar Reda Ibrahim, National Institute of Diabetes and Endocrinology, Cairo
- Ahmed Mohamed Abdelfattah Hamam, Military Hospitals, Cairo
- Amr Ali Mahfouz, National Institute of Diabetes and Endocrinology, Cairo
- Atef Bassyouni, National Institute of Diabetes and Endocrinology, Cairo
- Elsayed Abdel Fattah Eid, Faculty of Medicine, Delta University for science and technology, Dakahlia
- Fawzy A F Elmessallamy, Faculty of Medicine, Zagazig University, Sharqia
- Mohamed Abdelmoniem Elmikawy, police hospitals, Cairo.
- Randa Salam, Faculty of Medicine, Cairo University, Cairo.
- Yara Muhammad Ahmad Eid, Faculty of Medicine, Ain Shams University, Cairo.
Reviewers of the Clinical Content
- Eman Mahmoud, Faculty of Medicine, Al Azhar University, Cairo
- Ibrahim Elebrashy, Faculty of Medicine, Cairo University, Cairo.
- Kalid El Hadidy, Faculty of Medicine, Beni Suef University, Beni suef.
- Mohamed Halawa, Faculty of Medicine, Ain Shams University, Cairo
- Nabil El-Kafrawy, Faculty of Medicine, Menoufia University, Menoufia
- Yehia Ghanem, Faculty of Medicine, Alexandria University, Alexandria
Reviewers of the Adaptation Methodology
- Athar Reda Ibrahim, National Institute of Diabetes and Endocrinology, Cairo, Egypt
- Mai Mohammed Salama, National Hepatology and Tropical Medicine Research Institute, Cairo
- Abbreviations
BMI
Body Mass Index
CKD
Chronic kidney disease
CVD
Cardiovascular disease
DPP
Diabetes Prevention Program
DPP – 4i
Dipeptidyl peptidase - 4 inhibitors
GIP
Glucose dependent insulinotropic polypeptide
GLP-1Ra
Glucagon like peptide – 1 receptor agonists
GRADE
Grading of Recommendations Assessment, Development and Evaluation
HbA1c
Glycated hemoglobin A1c
HF
Heart failure
MASH
Metabolic dysfunction-associated steatohepatitis
RCT
Randomized controlled trial
SGLT2i
Sodium glucose co transporter 2 inhibitors
- Glossary
Cardiovascular diseases (CVDs)
A group of disorders of the heart and blood vessels that include coronary heart disease and cerebrovascular disease and peripheral arterial diseases
Dipeptidyl peptidase - 4 inhibitors (DPP - 4 inhibitors or gliptins)
Oral antidiabetic drugs used in treating type 2 diabetes. They suppress the degradation of incretins by blocking the action of the enzyme dipeptidyl - peptidase 4. This stimulates insulin secretion and suppresses glucagon release.
HbA1c
Haemoglobin glycated by non-enzymatic attachment of glucose to haemoglobin. The concentration of HbA1c is the most commonly used measure of chronic glycaemia in clinical trials and diabetes management. It is considered to reflect the integrated mean glucose level over the previous 8–12 weeks.
Metformin
A biguanide oral anti diabetic agent used in treating type 2 diabetes. It decreases glucose production by the liver and increases the insulin sensitivity of body tissues.
Type 1 diabetes
Diabetes caused by immune destruction of pancreatic beta-cells, resulting in lack of insulin production by the pancreas and need for insulin injections for survival.
Type 2 diabetes
Diabetes characterized by various degrees of disorders of insulin action in the body and insulin secretion by the pancreas. Insulin injections are not needed for survival, but might be needed for controlling blood glucose levels.
SGLT2
Sodium glucose co transporters -2 a group of drugs used in T2DM treatment as they inhibit glucose reabsorption from the renal tubules and increase glucose execration.
- Introduction
A comprehensive, multifaceted, and person-centered approach is recommended for managing type 2 diabetes and its lifelong consequences. Treatment should be tailored to individual glycemic and weight goals, risk of hypoglycemia, and history or risk factors for cardiovascular, kidney, liver, and other diabetes-related complications. Additionally, treatment options must consider medication tolerability, side effects, complexity of the regimen, and the individual's ability to adhere to it given their personal circumstances. Access, cost, and availability of medications are also crucial factors. Prioritizing lifestyle changes and healthy habits alongside pharmaceutical therapy is essential for optimal health outcomes.[i]’[ii]
- Scope and Purpose
The objective of this chapter is to provide guidance on pharmacotherapy for type 2 diabetes in adults to be used by policy-makers, health care professionals and primary health care providers to offer better treatment strategies for patients with type 2 diabetes.
- Target Audience
This guideline targets; healthcare professionals, policy makers, national diabetes programme managers, as well as non-governmental organizations (NGOs) and other stakeholders to afford the most appropriate tools for individuals with diabetes.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
1. Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2015 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in:
• Developing organisation/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt the American Diabetes Association – Standards of Care in Diabetes – 2024, 2025. 1,2
Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of
Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
• GRADE working group: https://www.gradeworkinggroup.org/
• GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1 Quality and Significance of the four levels of evidence in GRADE:
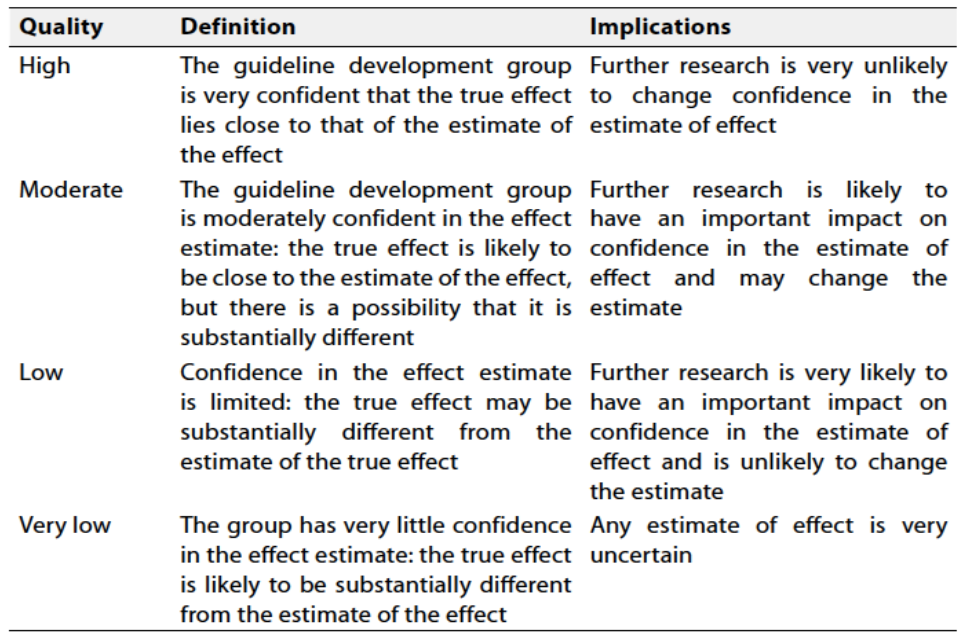
Table 2 Factors that determine How to upgrade or downgrade the quality of evidence
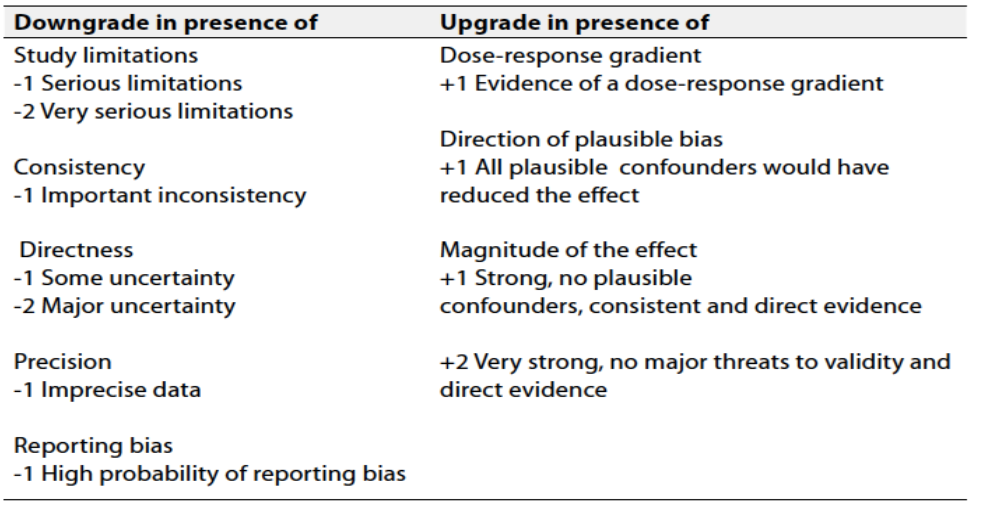
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation- Research needs
During the review of evidence and the development of recommendations, several research gaps were identified regarding Egyptian population. Addressing these will help inform revision of these guidelines.
1. Long-Term Effectiveness: Conduct studies to evaluate the long-term effectiveness of oral anti diabetic medications.
2. Tailoring Interventions: Investigate the effectiveness of personalized or tailored pharmacological therapies. Explore how individual characteristics, cultural factors, socioeconomic status, and health literacy influence program outcomes and identify strategies for optimizing prescription customization.
3. Cost-Effectiveness Analysis: Conduct cost-effectiveness analyses of diabetes prevention programs to assess the economic impact of these interventions. Evaluate the balance between costs, health outcomes, and potential healthcare savings to inform policy decisions and resource allocation.
- Monitoring and evaluating the impact of the guideline
There are potential indicators that can be used to evaluate the success of the pharmacological therapies in treating type 2 diabetic patients
1. Diabetes control rate (Hb A1c below 7 mg %) Rate Reduction: Calculate the percentage reduction in HbA1c using the following formula:
Percentage Reduction = ((Baseline HbA1C - Post-Implementation HbA1C) / Baseline HbA1c) 100
2. The same can be done to evaluate the effect different anti diabetic medications on hypoglycaemia , body weight , renal and cardiovascular outcome
3. Reporting the side effects of different pharmacological therapies
4. Program Engagement and Participation: Evaluate the level of engagement and participation of the target audience, including attendance rates for educational sessions, participation, and utilization of support resources. Higher engagement indicates increased program reach and potential effectiveness.
5. Cost-effectiveness: Assess the cost-effectiveness of the different treatment strategies
6. Patient Satisfaction and Feedback: Collect feedback from participants through surveys or interviews to gauge their satisfaction with the program, perceived benefits, and suggestions for improvement. Positive patient satisfaction indicates a well-received and impactful program.
- Update of the guideline;
These guidelines will be updated whenever there is new evidence.
- References
[1] ElSayed, N. A., McCoy, R. G., Aleppo, G., Bajaj, M., Balapattabi, K., Beverly, E. A., ... & Pekas, E. J. (2025). 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2025. Diabetes Care, 48.
[1] ElSayed, N. A., Aleppo, G., Bannuru, R. R., Bruemmer, D., Collins, B. S., Ekhlaspour, L., ... & American Diabetes Association Professional Practice Committee. (2024). 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2024. Diabetes Care, 47.
[1] Lambrinou E, Hansen TB, Beulens JW. Lifestyle factors, self-management and patient empowerment in diabetes care. Eur J Prev Cardiol. 2019 Dec;26(2_suppl):55-63.
[1] Staimez LR, Weber MB, Gregg EW. The role of lifestyle change for prevention of cardiovascular disease in diabetes. Curr Atheroscler Rep. 2014 Dec;16(12):460.
[1] Powers MA, Bardsley JK, Cypress M, Funnell MM, Harms D, Hess-Fischl A, et al.. Diabetes self-management education and support in adults with type 2 diabetes: a consensus report of the American Diabetes Association, the Association of Diabetes Care & Education Specialists, the Academy of Nutrition and Dietetics, the American Academy of Family Physicians, the American Academy of PAs, the American Association of Nurse Practitioners, and the American Pharmacists Association. Diabetes Care. (2020) 43:1636–49 10.2337/dci20-0023.
[1] Wang X, Qin LQ, Arafa A, Eshak ES, Hu Y, Dong JY. Smoking Cessation, Weight Gain, Cardiovascular Risk, and All-Cause Mortality: A Meta-analysis. Nicotine Tob Res. 2021 Nov 05;23(12):1987-1994.
[1] Riobó Serván P. [Diet recomendations in diabetes and obesity]. Nutr Hosp. 2018 Jun 12;35(Spec No4):109-115.
[1] Satpathy SV, Datta S, Upreti B. Utilization study of antidiabetic agents in a teaching hospital of Sikkim and adherence to current standard treatment guidelines. J Pharm Bioallied Sci. 2016 Jul-Sep;8(3):2238. doi: 10.4103/0975-7406.175975.
[1] Das P, Das BP, Rauniar GP, Roy RK, Sharma SK. Drug utilization pattern and effectiveness analysis in diabetes mellitus at a tertiary care centre in eastern Nepal. Indian J Physiol Pharmacol. 2011 Jul-Sep;55(3):272–80.
[1] Xu Y, Yang Z, Lin H, Shen P, Wang H, Zhan S, et al. Long-term patterns of antidiabetic medications use in patients with type 2 diabetes. Med Sci Monit. 2018 Dec 2;24:8707–15. doi: 10.12659/MSM.913603.
[1] Agarwal AA, Jadhav PR, Deshmukh YA. Prescribing pattern and efficacy of anti-diabetic drugs in maintaining optimal glycemic levels in diabetic patients. J Basic Clin Pharm. 2014 Jun;5(3):79–83. doi: 10.4103/0976-0105.139731.
[1] ADA “Standards of Care in Diabetes” 2024 Diabetes Care 2024;47(Suppl. 1):S158–S178 | https://doi.org/10.2337/dc24-S009
[1] Sowmya Swamy Sahibzadi Mahrukh Noo and Roy O. Mathew Cardiovascular Disease in Diabetes and Chronic Kidney Disease J. Clin. Med. 2023, 12(22), 6984; https://doi.org/10.3390/jcm12226984
[1] Ji Chun, Jodi Strong, and Scott Urquhart Insulin Initiation and Titration in Patients With Type 2 Diabetes Diabetes Spectr. 2019 May; 32(2): 104–111
[1] Gangji AS, Cukierman T, Gerstein HC, Goldsmith CH, Clase CM. A systematic review and meta-analysis of hypoglycemia and cardiovascular events: a comparison of glyburide with other secretagogues and with insulin. Diabetes Care. 2007;30:389–394.
[1] Obakiro SB, Kiyimb K, Napyo A, Knyike AM, Mayoka WJ, Nnassozi AG, et al. Appropriateness and affordability of prescriptions to diabetic patients attending a tertiary hospital in Eastern Uganda: A retrospective cross-sectional study. PLos One. 2021 Jan;16(1):e0245036. doi: 10.1371/journal.pone.0245036.
[1] Acharya KG, Shah KN, Solanki ND, Rana DA. Evaluation of antidiabetic prescriptions, cost and adherence to treatment guidelines: A prospective, cross-sectional study at a tertiary care teaching hospital. J Basic Clin Pharm. 2013 Sep;4(4):82–7. doi: 10.4103/0976-0105.121653.
[1] Belfort R, Harrison SA, Brown K, et al. A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis. N Engl J Med 2006;355:2297–2307
[1] Cusi K, Orsak B, Bril F, Xuet al. Liraglutide or insulin glargine treatments improves hepatic fat in obese patients with type 2 diabetes and nonalcoholic fatty liver disease in twenty-six weeks: a randomized placebo-controlled trial. Diabetes Res Clin Pract 2020;170:108487
[1] Flint A, Andersen G, Hockings P, et al. Randomised clinical trial: semaglutide versus placebo reduced liver steatosis but not liver stiffness in subjects with non-alcoholic fatty liver disease assessed by magnetic resonance imaging. Aliment Pharmacol Ther 2021;54:1150–1161
[1] Bizino MB, Jazet IM, de Heer P, et al. Placebo-controlled randomised trial with liraglutide on magnetic resonance endpoints in individuals with type 2 diabetes: a pre-specified secondary study on ectopic fat accumulation. Diabetologia 2020;63:65–74
[1] Newsome PN, Buchholtz K, Cusi K, et al.; NN9931-4296 Investigators. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med 2021;384:1113–1124
[1] Loomba R, Hartman ML, Lawitz EJ, et al.; SYNERGY-NASH Investigators. Tirzepatide for metabolic dysfunction-associated steatohepatitis with liver fibrosis. N Engl J Med 2024;391:299–310
[1] Gastaldelli A, Cusi K, Fernández Landó L, Bray R, Brouwers B, Rodríguez Á. Effect of tirzepatide versus insulin degludec on liver fat content and abdominal adipose tissue in people with type 2 diabetes (SURPASS-3 MRI): a substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial. Lancet Diabetes Endocrinol 2022;10:393–406
[1] Armstrong MJ, Gaunt P, Aithal GP, et al.; LEAN Trial Team. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet 2016;387:679–690
[1] Sathyanarayana P, Jogi M, Muthupillai R, Krishnamurthy R, Samson SL, Bajaj M. Effects of combined exenatide and pioglitazone therapy on hepatic fat content in type 2 diabetes. Obesity (Silver Spring) 2011;19:2310–2315
[1] Abdul-Ghani M, Migahid O, Megahed A, DeFronzo RA, Al-Ozairi E, Jayyousi A. Combination therapy with pioglitazone/exenatide improves beta-cell function and produces superior glycaemic control compared with basal/bolus insulin in poorly controlled type 2 diabetes: a 3-year follow-up of the Qatar study. Diabetes Obes Metab 2020;22:2287–2294
[1] Lavynenko O, Abdul-Ghani M, Alatrach M, et al. Combination therapy with pioglitazone/exenatide/metformin reduces the prevalence of hepatic fibrosis and steatosis: the efficacy and durability of initial combination therapy for type 2 diabetes (EDICT). Diabetes Obes Metab 2022;24:899–907.
- Annexes
Drugs used in treatment of T2 DM
Early initiation of pharmacologic therapy is associated with improved glycemic control and reduced long-term complications in type 2 diabetes.
Drug classes used for the treatment of type 2 diabetes include the following:
· Biguanides
· Sulfonylureas
· Meglitinide derivatives
· Alpha-glucosidase inhibitors
· Thiazolidinediones (TZDs)
· Glucagonlike peptide–1 (GLP-1) agonists
· GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonists
· Dipeptidyl peptidase IV (DPP-4) inhibitors
· Selective sodium-glucose transporter–2 (SGLT-2) inhibitors
Biguanides
Metformin is the only biguanide in clinical use and has proved effective and safe. Lactic acidosis during metformin use is very rare and is associated with concurrent comorbidity. Metformin lowers basal and postprandial plasma glucose levels. Metformin works by decreasing hepatic gluconeogenesis. decreases intestinal absorption of glucose and improves insulin sensitivity by increasing peripheral glucose uptake and utilization.
Sulfonylureas
Sulfonylureas (eg, glyburide, glipizide, glimepiride) are insulin secretagogues that stimulate insulin release from pancreatic beta cells and probably have the greatest efficacy for glycemic lowering of any of the oral agents. However, that effect is only short-term and quickly dissipates. Sulfonylureas are indicated for use as adjuncts to diet and exercise in adult patients with type 2 diabetes mellitus. They are generally well-tolerated, with hypoglycemia the most common side effect.
Meglitinide derivatives
Meglitinides (eg, repaglinide, nateglinide) are much shorter-acting insulin secretagogues than the sulfonylureas are, with preprandial dosing potentially achieving more physiologic insulin release and less risk for hypoglycemia.
Meglitinides may be used in patients who have allergy to sulfonylurea medications.
They have a similar risk for inducing weight gain as sulfonylureas do but possibly carry less risk for hypoglycemia.
Alpha-glucosidase inhibitors
These agents delay sugar absorption and help to prevent postprandial glucose surges. Alpha-glucosidase inhibitors prolong the absorption of carbohydrates, but their induction of flatulence greatly limits their use. They should be titrated slowly to reduce gastrointestinal (GI) intolerance.
Thiazolidinediones
TZDs (eg, pioglitazone the only TZD currently available) act as insulin sensitizers; thus, they require the presence of insulin to work. They must be taken for 12-16 weeks to achieve maximal effect..They are the only antidiabetic agents that have been shown to slow the progression of diabetes (particularly in early disease).pioglitazone was found to reduce the progression to frank diabetes by 72% in patients with IGT . However, the drug was associated with significant edema and weight gain. TZDs generally decrease triglyceride levels and increase HDL cholesterol levels. They increase LDL cholesterol, but this increase may involve large, buoyant LDL, which may be less atherogenic.
Glucagon like peptide–1 agonist
GLP-1 agonists (ie, exenatide, liraglutide, albiglutide, dulaglutide and semagutide) mimic the endogenous incretin GLP-1; they stimulate glucose-dependent insulin release, reduce glucagon, and slow gastric emptying so improving glycemic control with significant weight loss without hypoglycemia
Dipeptidyl peptidase IV inhibitors
DPP-4 inhibitors (eg, vildagliptin, sitagliptin, saxagliptin, linagliptin and allogliptin) are a class of drugs that prolong the action of incretin hormones. DPP-4 degrades numerous biologically active peptides, including the endogenous incretins GLP-1 and glucose-dependent insulinotropic polypeptide (GIP). DPP-4 inhibitors can be used as a monotherapy or in combination with metformin or a TZD. They are given once daily and are weight neutral.
Selective sodium-glucose transporter-2 inhibitors
SGLT-2 inhibition (Canagliflozin, dapagliflozin, empagliflozin and others ) lowers the renal glucose threshold (ie, the plasma glucose concentration that exceeds the maximum glucose reabsorption capacity of the kidney). Lowering the renal glucose threshold results in increased urinary glucose excretion.
Received FDA approval for the treatment of diabetic kidney disease (DKD) and, in patients with type 2 diabetes and DKD, reduction in the risk of hospitalization for heart failure. prevention of cardiovascular disease–related death in adults with type 2 diabetes who also have cardiovascular disease
Insulin
See insulin in type 1 DM treatment
Indications of insulin in type 2 diabetes
1- Very high blood glucose with catabolic state
2- During pregnancy
3- Peri operative
4- In hospitalized patients
5- In critically ill patients
6- Failure of oral medication to keep glycemic control
7- In patient with liver or kidney disease