Management of Pediatric Shock
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Management of Pediatric Shock |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 1 December 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgements
- - Abbreviations
- - Glossary
- - Introduction
- - Methods
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline
- - Updating of the guideline
- - References
- - Annexes
- Executive Summary
Shock is a life-threatening emergency characterized by circulatory failure and impaired tissue
perfusion. In addition to clinical and hemodynamic condition, oxygen utilization and/or cellular
variables have been used to define shock.(1) Hypotension is neither a constant nor an early finding in pediatric shock and prompt recognition requires clinical assessment for tissue hypoperfusion and a high index of suspicion.(2)(3) Involved pathophysiological mechanisms include a combination of reduced intravascular volume, abnormal myocardial function, reduced vascular tone with inappropriate vasodilatation (vasoplegia) and/or circulatory obstruction associated with conditions such as cardiac tamponade, tension pneumothorax or massive pulmonary embolism. Clinical conditions often associated with shock include hypovolemia (eg bleeding and severe dehydration), severe sepsis, cardiogenic shock and anaphylaxis.(4-8)
Severe sepsis and septic shock represent a dysregulated immune response to an invasive infection. (9) Even without shock, children with infections frequently develop fever, tachycardia and vasodilatation as a result of an inflammatory response. Septic shock should be suspected if these manifestations are associated with a change in mental status, and the diagnosis made when tissue perfusion is impaired. Patients with septic shock may present with predominantly low cardiac output, vasoconstriction, delayed capillary refill and cold extremities (cold shock); or with predominantly vasodilatation, wide pulse pressure, warm extremities and increased cardiac output (warm shock).(1)(5) Contrary to the case in adults and some adolescents, most cases of septic shock in infants and children present as cold shock, with low cardiac output associated with hypovolemia (deficient intake and capillary leak) and/or myocardial insufficiency.(2)(5) The clinical distinction between cold and warm shock is not always clear-cut and has sometimes been disputed as a guide to initial inotropic/ vasopressor support. (10)
While cardiogenic etiology of shock may be quite obvious, such as following cardiac surgery and in those with cardiac disease, cardiogenic shock should be suspected in patients with signs such as a gallop rhythm, heart murmur, evidence of circulatory congestion (pulmonary rales, jugular venous distension, hepatomegaly or worsening with volume expansion) or arrhythmia. (4)
Bedside cardiac ultrasonography can be helpful in diagnosis and assessment of myocardial function. (11)
Anaphylaxis is a life-threatening systemic hypersensitivity reaction to triggers such as parenteral medications, insect venoms and food allergens. Prompt intervention is critical and early injection of epinephrine (adrenaline) is essential as it is the only drug shown to reduce mortality and hospitalization. (12-15)
An immediate stepwise approach with ongoing monitoring and clear end-points is necessary for successful management of shock. (1)(7) Initial evaluation and resuscitation should occur irrespective of patient location (emergency department, intensive care unit, general ward), even if it is clear that transfer to a higher level of care will be needed (5)
While most patients with shock benefit from intravascular volume expansion, the required amount and frequency of fluid administration can vary significantly and should depend on assessment of fluid responsiveness. The role of inotropes, vasopressors and vasodilators also varies depending on the prevailing pathophysiology, which can change even in the same patient. (4)(5)(16)(17) There is an increasing role for objective non-invasive hemodynamic assessment using tools such as point-of care ultrasound, electrical impedance cardiometry and
measurement of central venous oxygen saturation (ScvO2) (10)(11)(16)(18) to supplement clinical assessment and enable treatment appropriate to the actual pathophysiological derangements present. Other critical aspects of management include support of other systems and treatment of the underlying cause. (5)
This guideline focuses on management of Pediatric Shock.
➡️Guideline development process and methods
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1. American College of Critical Care Medicine Clinical Practice. Parameters for Hemodynamic Support of Pediatric and Neonatal Septic Shock. Crit Care Med. (2017)
2. Surviving Sepsis Campaign International Guidelines for the Management of Septic Shock and Sepsis-Associated Organ Dysfunction in Children. Pediatr Crit Care (2020)
3. The International Society for Heart and Lung Transplantation, Guidelines for the management of pediatric heart failure. (2014)
4. Guidelines for the Appropriate Use of Bedside General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients.
General Ultrasonography. Crit Care Med. (2015).
Cardiac Ultrasonography. Crit Care Med. (2016)
5. EAACI Food Allergy and Anaphylaxis Guidelines Group. Anaphylaxis: Guidelines from the European Academy of Allergy and Clinical Immunology. (2014)
6. Emergency department diagnosis and treatment of anaphylaxis: a practice parameter. Ann Allergy Asthma Immunol. (2014)
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
➡️Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following four sub-sections:
➡️ Management of Pediatric Shock
This section includes recommendations and good practice statements on
1) promotion of effective and timely management of pediatric patients with shock.
2) improve survival of pediatric patients with shock.
We can summarize the guidelines’ recommendations for management of pediatric shock in the following:
· Shock should be recognized when there are clinical signs of inadequate
tissue perfusion;
including:
Prolonged capillary refill greater than 2 seconds, diminished pulses, mottled
cool extremities (or flash capillary refill, bounding peripheral pulses and
wide pulse pressure), decreased or altered mental status, decreased urine
output.
Hypotension is not necessary; however, its presence is confirmatory (weak (conditional) recommendation).
· Consider hypovolemic shock when there is intravascular fluid loss (eg., hemorrhage, vomiting, diarrhea, osmotic diuresis or capillary leak) (Good practice statemen)t.
· The clinical diagnosis of septic shock is made in children who have clinical signs of inadequate tissue perfusion AND have a suspected infection (weak (conditional) recommendation).
· Consider cardiogenic shock in patients with signs such as a gallop rhythm, heart murmur, evidence of circulatory congestion (pulmonary rales, jugular venous distension, hepatomegaly or worsening with volume expansion) or arrhythmia. Arrhythmias should be appropriately managed (Good practice statement).
· The use for cardiac ultrasonography is recommended to assess the etiology of cardiogenic shock (weak (conditional) recommendation).
· Base the diagnosis of anaphylaxis on the history and physical examination, using scenarios described by the National Institutes of Allergy and Infectious Disease (NIAID) Panel (fig 2, p 28 ), recognizing that there is a broad spectrum of anaphylaxis presentations that require clinical judgment (weak (conditional) recommendation).
· Life-threatening obstructive causes of shock should be identified and treatment initiated for the underlying cause:
Pericardiocentesis for cardiac tamponade, anticoagulation and thormbectomy for pulmonary embolus, chest tube thoracostomy or needle thoracentesis for pneumothorax, or prostaglandin E1 for ductal dependent circulation (Good practice statement).
· The use of cardiac ultrasonography is recommended to recognize/ rule out cardiac Tamponade (Strong recommendation).
· Airway and breathing should be rigorously monitored and maintained. Supplemental oxygen should be given as initial therapy (weak (conditional) recommendation).
· In children with anaphylaxis, prepare for airway management,including intubation if necessary, if there is any suggestion of airway edema (eg, hoarseness or stridor) (weak (conditional) recommendation).
· The decision to intubate and ventilate should be based on clinical assessment of
increased work of breathing, hypoventilation, or impaired mental status. Waiting for confirmatory laboratory tests is discouraged (weak (conditional) recommendation).
· Intubation may be performed for children with fluid-refractory, catecholamine resistant
shock without respiratory failure (Good practice statement).
· If possible, volume loading and peripheral or central inotropic/vasoactive drug support is recommended before and during intubation;
because of relative or absolute hypovolemia, cardiac dysfunction, and the risk of suppressing endogenous stress hormone response with agents that facilitate intubation.
Etomidate is not recommended. Ketamine with atropine pretreatment should be considered the induction combination of choice.
A short-acting neuromuscular blocking agent can facilitate intubation if the provider is confident and skilled (weak (conditional) recommendation).
· Vascular access should be rapidly attained. Intraosseous access should be established if reliable intravenous line cannot be attained in minutes (weak (conditional) recommendation).
· The use of ultrasonography is recommended for central venous access (Strong recommendation).
· Real-time, single operator approach is recommended (Strong recommendation).
Ultrasonography is operator dependent and vascular access should not be delayed in shocked patients (weak (conditional) recommendation).
· A dilute concentration of the initial vasoactive medication (including epinephrine or norepinephrine) may be administered through a peripheral vein or intraosseous line if central venous access is not readily accessible (Good practice statement).
· Patients with hypovolemic shock or distributive shock (including septic & anaphylactic shock) should receive fluid resuscitation:
Amount: 20 mL/Kg per bolus, Type: isotonic crystalloid (eg normal saline) DuraIon: push or rapid infusion over 5-10 min (weak (conditional) recommendation).
· Patients with cardiogenic shock should only receive fluid resuscitation if they are judged to have preload insufficiency (Good practice statement).
· Cardiac ultrasonography evaluation is recommended during such assessment (weak (conditional) recommendation).
· Patients with poor cardiac function may also be volume depleted. Smaller boluses (5-10 mL/kg) should be given more slowly (over 10-20 min) for these patients (Good practice statement).
· Fluid resuscitation should be avoided or discontinued when there is evidence of intravascular volume overload (weak (conditional) recommendation).
· During fluid resuscitation, monitor for the development of increased work of breathing, rales, hypoxemia, cardiac gallop rhythm, hepatomegaly or a diminishing MAP-CVP (weak (conditional) recommendation).
· IniIal volume resuscitaIon requirements may be 0 mL/kg if rales or hepatomegaly are present (weak (conditional) recommendation).
· Fluid boluses may be repeated with the goal of normal perfusion, cardiac output and blood pressure provided there are no signs of fluid overload (weak (conditional) recommendation).
· A total of up to 60 mL/Kg may be needed during the first hour (weak (conditional) recommendation).
· For patients with sepsis In low resource settings with no availability of intensive care: in the absence of hypotension, maintenance fluids should be started without prior bolus fluid administration (Strong recommendation).
· When children with presumed hypovolemia have not improved after receiving a total of 60 mL/kg over 30 to 60 minutes, the following should be considered:
-The amount of fluid loss may have been underestimated (eg burn injury)
-There may be significant ongoing fluid loss (eg hemorrhage from blunt abdominal trauma or capillary leak with bowel obstruction)
-Other conditions may be causing or contributing to shock (eg spinal cord injury in a child with multiple trauma, sepsis, myocardial dysfunction, etc) (Good practice statement).
· After the first hour, ongoing fluid replacement should be directed at clinical endpoints including perfusion as well as available tools of hemodynamic monitoring as CO, global end-diastolic volume and PAOP (pulm A occlusion P) (weak (conditional) recommendation).
· Following shock resuscitation, diuretics, peritoneal dialysis or high flux CRRT can be used to remove fluid in paAents who are 10% fluid overloaded and unable to maintain fluid balance with native urine output/ extra-renal losses (weak (conditional) recommendation).
· In children with fluid overload and ventricular dysfunction diuretics (such as furosemide) should be used to return to euvolemic state while monitoring clinical criteria and cardiac output (Good practice statement).
· High-volume hemofiltration (HVHF) is not preferred over standard hemofiltration in children with septic shock or other sepsis-associated organ dysfunction who are treated with renal replacement therapy (weak (conditional) recommendation).
· Crystalloids, rather than 5% albumin, are recommended for the initial resuscitation of children with septic shock (Weak (conditional) recommendation).
· Although controversial, colloid is a reasonable option for patients with hypoalbuminemia (albumin <3g /dL) or hyperchloremic metabolic acidosis who have not improved after initial crystalloid volume expansion (Good practice statement).
· In the acute resuscitation of children with septic shock or other sepsis associated organ dysfunction, it is NOT recommended to use: Starches (Strong recommendation); or Gelatin (Weak (conditional) recommendation).
· Patients with hemorrhagic shock who have not improved should receive blood and require definitive treatment for the cause of hemorrhage (Good practice statement).
· Transfusion of RBCs is not routinely indicated if the blood hemoglobin concentration is greater than or equal to 7 g/dL in hemodynamically stabilized children with septic shock or other sepsis-associated organ dysfunction (weak (conditional) recommendation).
· RBC transfusion may be given to children with Hgb less than 10 g/dL. and poor tissue perfusion despite volume expansion (low CI, low ScvO2) (weak (conditional) recommendation).
· Prophylactic plasma or platelet transfusions are not routinely recommended in nonbleeding children with septic shock or other sepsis associated organ dysfunction solely on the basis of laboratory abnormalities (weak (conditional) recommendation).
· IV immune globulin (IVIG) should not be routinely used in children with septic shock or other sepsis associated organ dysfunction (weak (conditional) recommendation).
· Hypoglycemia must be rapidly diagnosed and promptly treated (weak (conditional) recommendation).
· In paIents with sepsis, a 10% dextrose containing IV solution can be run at maintenance rate to provide age appropriate glucose delivery and to prevent hypoglycemia (weak (conditional) recommendation).
· Blood glucose levels below 180 mg/dL (10 mmol/L) should be targeted (Good practice statement).
· Insulin therapy targeting a blood glucose at or below 140 mg/dL (7.8 mmol/L) is NOT recommended (Strong recommendation).
· Calcium replacement should be directed to normalize ionized calcium concentration (weak (conditional) recommendation).
· Thyroid replacement can be lifesaving in children with thyroid insufficiency and catecholamine-resistant shock (weak (conditional) recommendation).
· The routine use of levothyroxine in children with septic shock and other sepsis associated organ dysfunction in a sick euthyroid state is not recommended (weak (conditional) recommendation).
· The management goals in the first hour should be to maintain/ restore:
*Airway, oxygenation, and ventilation
*Circulation
-normal blood pressure for age (only reliable when pulses palpable)
-normal pulses with no differential between the quality of peripheral & central pulses
-threshold HR
-perfusion: Capillary refill less than or equal to 2 seconds, warm extremities, urine output greater than 1mL/kg/hr, normal mental status
*Normal glucose concentration, normal ionized calcium concentration (weak (conditional) recommendation).
· The following additional goals are applicable beyond the first hour:
-Perfusion pressure (MAP-CVP or MAP-IAP) appropriate for age.
-ScvO2 greater than 70%
-CI greater than 3.3 and less than 6.0L/min/m2
-Normal INR, anion gap, and lactate. (weak (conditional) recommendation).
· On-going resuscitation should be guided by hemodynamic assessment & monitoring including:
- Heart rate, blood pressure, pulse pressure, capillary refill/ skin perfusion analysis and temperature
- Pulse-oximetry and continuous ECG monitoring
- CVP
- Urine output
- Laboratory (Arterial blood gases, ScvO2, lactate, glucose and ionized Ca) (weak (conditional) recommendation).
· Assessment of CI and SVRI using advanced hemodynamic monitoring is recommended when available. Methods include:
- invasive arterial BP monitoring with pulse-contour analysis
- serial ultrasonographic assessment (weak (conditional) recommendation).
· The use of cardiac ultrasonography to assess the efficacy of fluid resuscitation, ventricular function and inotropic support (weak (conditional) recommendation).
- electrical impedance cardiometry (Good practice statement).
· In patients with cardiogenic shock, repeated determination of troponin levels can be used to assess the severity of myocardial involvement as well as the response to treatment (Good practice statement).
· It is reasonable to begin vasoacAve infusions aNer 40–60 mL/kg of fluid resuscitation if the patient continues to have evidence of abnormal perfusion, or sooner if fluid overload develops or other concerns for fluid administration are present (Good practice statement).
· Use of intravenous inotropic agents in the absence of clinical evidence of hypotension, low CO and/or decreased end-organ perfusion is potentially harmful (Strong recommendation).
· In septic shock:
-Central epinephrine can be started for “cold shock” (0.05–0.3 μg/kg/min) or
norepinephrine can be titrated for “warm shock”.
-Central dopamine can be Atrated to a maximum of 10 μg/kg/min.
-Epinephrine or norepinephrine is more likely to be beneficial (weak (conditional) recommendation).
· In cardiogenic shock:
-Milrinone and /or dobutamine can be used as first- line therapy
-It is probably advisable to use milrinone in post- cardiac surgery patients and in
cases with impaired RV function and/or associated pulmonary hypertension (weak (conditional) recommendation).
· Septic shock With Low CI, Normal Blood Pressure, and High SVR:
- Milrinone is considered the first-line inodilator in patients with epinephrine resistant
shock and normal blood pressure.
- Additional volume loading may be necessary to prevent hypotension.
- Norepinephrine can partly reverse hypotension associated with inodilators.
- Nitroprusside or nitroglycerin may be considered as second-line vasodilators.
- Levosimendan and enoximone may have a role with persistently low CO (weak (conditional) recommendation).
· Septic shock With Low CI, Low Blood Pressure, and Low SVR:
-Norepinephrine can be added to/or substituted for epinephrine to increase DBP and SVR.
-Once an adequate blood pressure is achieved, dobutamine, milrinone, enoximone or levosimendan may be added to norepinephrine to improve CI and ScvO2 (weak (conditional) recommendation).
· Septic shock With High CI and Low SVR:
- When titration of norepinephrine and fluid does not resolve hypotension, vasopressin, angiotensin, or terlipressin can be helpful in restoring blood pressure
- These drugs can reduce CO so CO/ScvO2 monitoring is necessary. Low-dose epinephrine or dobutamine may be added to improve CO (weak (conditional) recommendation).
· Cardiogenic shock with low CI refractory to milrinone &/or dobutamine:
-Epinephrine has a role in the face of refractory hypotension and poor end-organ perfusion.
-Levosimendan may be considered in children unresponsive to traditional inotropic therapy (weak (conditional) recommendation).
· Children with refractory shock must be suspected to have unrecognized morbidities; such as:
-Inappropriate source control of infection (remove nidus and use effective antibiotics)
-Pericardial effusion (pericardiocentesis)
-Pneumothorax (thoracentesis)
-Hypoadrenalism (adrenal hormone replacement)
-Hypothyroidism (thyroid hormone replacement)
-Ongoing blood loss (blood replacement/hemostasis)
-Increased IAP (peritoneal catheter or abdominal release)
-Necrotic tissue (nidus removal)
-Excessive immunosuppression (wean immunosuppressants), or immunocompromise (restore immune function; e.g., white cell growth factors/transfusion for neutropenic sepsis) (weak (conditional) recommendation).
· ECMO is an important option to consider in refractory shock when potentially reversible causes are addressed (weak (conditional) recommendation).
· Venovenous ECMO is suggested in children with sepsis-induced PARDS and refractory hypoxia.
Venoarterial ECMO is suggested in children with septic shock refractory to all other treatments (weak (conditional) recommendation).
· IV hydrocortisone may be used if adequate fluid resuscitation and vasopressor therapy are not able to restore hemodynamic stability (weak (conditional) recommendation).
· Ideally after attaining a blood sample for subsequent determination of baseline cortisol concentration (weak (conditional) recommendation).
· In septic shock, broad spectrum antibiotics should be initiated within 60 minutes. After obtaining blood culture if it does not delay antibiotic administration (weak (conditional) recommendation).
· Positioning: patients experiencing anaphylaxis should be positioned supine with elevated lower extremities if they have circulatory instability, sitting up if they have respiratory distress, and in recovery position if unconscious (weak (conditional) recommendation).
· Adrenaline:
Adrenaline must promptly be administered as the first-line treatment for the emergency management of anaphylaxis (weak (conditional) recommendation).
- By intramuscular injection into the mid-outer thigh (Strong recommendation).
- In patients requiring repeat doses of adrenaline, these should be administered at least 5 min apart (weak (conditional) recommendation).
- If the patient is not responding to epinephrine injections, IV infusion of epinephrine should be given in a monitored setting (Strong recommendation).
- Do not routinely administer antihistamines or corticosteroids instead of epinephrine. There is no substitute for epinephrine in the treatment of anaphylaxis (weak (conditional) recommendation).
· Other therapies:
-Trigger of the anaphylaxis episode should be removed (weak (conditional) recommendation).
- Administer additional vasopressors If parenteral epinephrine and fluid resuscitation fail to restore blood pressure (Strong recommendation).
- Administer an inhaled b-agonist if bronchospasm is a component of anaphylaxis (Strong recommendation).
- Administration of antihistamines and corticosteroids should be considered adjunctive therapy (weak (conditional) recommendation).
- Systemic glucocorticosteroids may be used as they may reduce the risk of late phase respiratory symptoms (weak (conditional) recommendation).
- High-dose nebulized glucocorticoids may be beneficial for upper airway obstruction (weak (conditional) recommendation).
· Strongly consider observing patients who have experienced anaphylaxis for at least 4 to 8 hours and observe patients with a history of risk factors for severe anaphylaxis (such as asthma, previous biphasic reactions, or protracted anaphylaxis) for a longer period. Patients who have experienced anaphylaxis should consult an allergist/ immunologist.after discharge (weak (conditional) recommendation).
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/- Recommendations
Table 3. Recommendations |
|
| |||
A. Recognition of shock |
|
| |||
N | Health questions | Source Guideline | Recommendations | Quality of evidence | Strength of Recommendation |
A1 | In pediatric patients with suspected shock/ at risk for shock, when should shock be diagnosed? | ACCM | Shock should be recognized when there are clinical signs of inadequate tissue perfusion; including: Prolonged capillary refill greater than 2 seconds, diminished pulses, mottled cool extremities (or flash capillary refill, bounding peripheral pulses and wide pulse pressure), decreased or altered mental status, decreased urine output. Hypotension is not necessary; however, its presence is confirmatory. | High | Weak (conditional)
|
|
|
|
||||
|
B. Determination of type/ likely etiology |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
In pediatric patients with shock, how could the likely etiology be determined? |
ACCM
US
EDP-Ana; Endorsed by EAACI
US |
· Consider hypovolemic shock when there is intravascular fluid loss (eg., hemorrhage, vomiting, diarrhea, osmotic diuresis or capillary leak).
· The clinical diagnosis of septic shock is made in children who have clinical signs of inadequate tissue perfusion AND have a suspected infection.
· Consider cardiogenic shock in patients with signs such as a gallop rhythm, heart murmur, evidence of circulatory congestion (pulmonary rales, jugular venous distension, hepatomegaly or worsening with volume expansion) or arrhythmia. Arrhythmias should be appropriately managed.
· The use for cardiac ultrasonography is recommended to assess the etiology of cardiogenic shock.
· Base the diagnosis of anaphylaxis on the history and physical examination, using scenarios described by the National Institutes of Allergy and Infectious Disease (NIAID) Panel (fig 2, p 28 ), recognizing that there is a broad spectrum of anaphylaxis presentations that require clinical judgment.
· Life-threatening obstructive causes of shock should be identified and treatment initiated for the underlying cause: Pericardiocentesis for cardiac tamponade, anticoagulation and thormbectomy for pulmonary embolus, chest tube thoracostomy or needle thoracentesis for pneumothorax, or prostaglandin E1 for ductal dependent circulation. · The use of cardiac ultrasonography is recommended to recognize/ rule out cardiac Tamponade. |
High
Moderate
Low
Moderate |
Good practice statement
Weak (conditional)
Good practice statement
weak (conditional)
weak (conditional)
Good practice statement
Strong |
|
Table 5. Respiratory support |
|
|
|||
|
C. What respiratory support is needed for pediatric patients with shock? |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
C1 |
What respiratory support is needed for pediatric patients with shock? |
ACCM
EDP-Ana C
ACCM
SSC
ACCM
|
· Airway and breathing should be rigorously monitored and maintained. Supplemental oxygen should be given as initial therapy.
· In children with anaphylaxis, prepare for airway management,including intubation if necessary, if there is any suggestion of airway edema (eg, hoarseness or stridor).
· The decision to intubate and ventilate should be based on clinical assessment of increased work of breathing, hypoventilation, or impaired mental status. Waiting for confirmatory laboratory tests is discouraged.
· Intubation may be performed for children with fluid-refractory, catecholamine resistant shock without respiratory failure.
· If possible, volume loading and peripheral or central inotropic/vasoactive drug support is recommended before and during intubation; because of relative or absolute hypovolemia, cardiac dysfunction, and the risk of suppressing endogenous stress hormone response with agents that facilitate intubation. Etomidate is not recommended. Ketamine with atropine pretreatment should be considered the induction combination of choice. A short-acting neuromuscular blocking agent can facilitate intubation if the provider is confident and skilled. |
High
Moderate
High
High
|
Weak (conditional)
Weak (conditional)
Weak (conditional)
Good practice Statement
(weak (conditional)
|
|
Table 6. Vascular access |
|
|
|||
|
D. In pediatric patients with shock, how could the likely etiology be determined? |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
D1 |
In pediatric patients with shock, how could the likely etiology be determined? |
ACCM
US
US
US
SSC |
· Vascular access should be rapidly attained. Intraosseous access should be established if reliable intravenous line cannot be attained in minutes. · The use of ultrasonography is recommended for central venous access.
· Real-time, single operator approach is recommended.
· Ultrasonography is operator dependent and vascular access should not be delayed in shocked patient.
· A dilute concentration of the initial vasoactive medication (including epinephrine or norepinephrine) may be administered through a peripheral vein or intraosseous line if central venous access is not readily accessible. |
High
High
High
High
|
Weak (conditional)
Strong
Strong
Weak (conditional)
Good practice statement
|
|
Table 7. Recommendations |
|
|
|||
|
E. Fluid management |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
E1
|
In pediatric patients with shock, when and how should fluid resuscitation be given?
|
ACCM EAACI
US
ACCM SSC
ACCM
ACCM
ACCM
ACCM SSC
SSC
ACCM |
· Patients with hypovolemic shock or distributive shock (including septic & anaphylactic shock) should receive fluid resuscitation: Amount: 20 mL/Kg per bolus, Type: isotonic crystalloid (eg normal saline) DuraIon: push or rapid infusion over 5-10 min
· Patients with cardiogenic shock should only receive fluid resuscitation if they are judged to have preload insufficiency.
· Cardiac ultrasonography evaluation is recommended during such assessment.
· Patients with poor cardiac function may also be volume depleted. Smaller boluses (5-10 mL/kg) should be given more slowly (over 10-20 min) for these patients.
· Fluid resuscitation should be avoided or discontinued when there is evidence of intravascular volume overload.
· During fluid resuscitation, monitor for the development of increased work of breathing, rales, hypoxemia, cardiac gallop rhythm, hepatomegaly or a diminishing MAP-CVP.
· IniIal volume resuscitaIon requirements may be 0 mL/kg if rales or hepatomegaly are present. · Fluid boluses may be repeated with the goal of normal perfusion, cardiac output and blood pressure provided there are no signs of fluid overload.
· A total of up to 60 mL/Kg may be needed during the first hour
· For patients with sepsis In low resource settings with no availability of intensive care: in the absence of hypotension, maintenance fluids should be started without prior bolus fluid administration. Overly aggressive fluid boluses may be harmful in patients with cardiogenic shock, DKA, syndrome of inappropriate antidiuretic hormone secretion, severe malnutrition, or, in resource-limited settings, severe febrile illness in the absence of dehydration, hemorrhage or hypotension.
· When children with presumed hypovolemia have not improved after receiving a total of 60 mL/kg over 30 to 60 minutes, the following should be considered: -The amount of fluid loss may have been underestimated (eg burn injury) -There may be significant ongoing fluid loss (eg hemorrhage from blunt abdominal trauma or capillary leak with bowel obstruction) -Other conditions may be causing or contributing to shock (eg spinal cord injury in a child with multiple trauma, sepsis, myocardial dysfunction, etc). · After the first hour, ongoing fluid replacement should be directed at clinical endpoints including perfusion as well as available tools of hemodynamic monitoring as CO, global end-diastolic volume and PAOP (pulm A occlusion P) In pediatric patients with sepsis, fluid losses and persistent hypovolemia secondary to diffuse capillary leak can continue for days. |
High
Very low
High
Low
High
High
High
Low
High
High |
Weak (conditional)
Weak (conditional)
Good practice statement
Weak (conditional)
Good practice statement
(weak (conditional)
Weak (conditional)
Weak (conditional)
Weak (conditional)
Weak (conditional)
Strong
Good practice statement
Weak (conditional) |
|
E2 |
Following initial resuscitation, what is the role and methods of fluid removal? |
ACCM
SSC |
· Following shock resuscitation, diuretics, peritoneal dialysis or high flux CRRT can be used to remove fluid in paAents who are 10% fluid overloaded and unable to maintain fluid balance with native urine output/ extra-renal losses.
· In children with fluid overload and ventricular dysfunction diuretics (such as furosemide) should be used to return to euvolemic state while monitoring clinical criteria and cardiac output.
· High-volume hemofiltration (HVHF) is not preferred over standard hemofiltration in children with septic shock or other sepsis-associated organ dysfunction who are treated with renal replacement therapy. |
High
Low |
Weak (conditional)
Good practice statement
Weak (conditional)
|
|
Table 8. Recommendations |
|
|
|||
|
F. Colloids and blood products |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
F1 |
What is the role of colloids for volume expansion in shock? |
SSC
SSC |
· Crystalloids, rather than 5% albumin, are recommended for the initial resuscitation of children with septic shoc · Although controversial, colloid is a reasonable option for patients with hypoalbuminemia (albumin <3g /dL) or hyperchloremic metabolic acidosis who have not improved after initial crystalloid volume expansion · In the acute resuscitation of children with septic shock or other sepsis associated organ dysfunction, it is NOT recommended to use: Starches; or Gelatin |
Moderate
Moderate
Low |
Weak (conditional)
Good practice statement
Strong
Weak (conditional) |
|
F2 |
What is the role of blood products in shock? |
SSC
ACCM
SSC SSC |
· Patients with hemorrhagic shock who have not improved should receive blood and require definitive treatment for the cause of hemorrhage,
· Transfusion of RBCs is not routinely indicated if the blood hemoglobin concentration is greater than or equal to 7 g/dL in hemodynamically stabilized children with septic shock or other sepsis-associated organ dysfunction.
· RBC transfusion may be given to children with Hgb less than 10 g/dL. and poor tissue perfusion despite volume expansion (low CI, low ScvO2).
·Prophylactic plasma or platelet transfusions are not routinely recommended in nonbleeding children with septic shock or other sepsis associated organ dysfunction solely on the basis of laboratory abnormalities. · IV immune globulin (IVIG) should not be routinely used in children with septic shock or other sepsis associated organ dysfunction. N.B. Although therapies may not be routinely recommended, select patients may benefit |
Low
High
Very low
Low |
Good practice statement
Weak (conditional)
Weak (conditional)
Weak (conditional)
Weak (conditional)
|
|
|
|
||||
|
G. Metabolic abnormalities |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
G1 |
What is the target blood glucose in patients with shock? |
ACCM
ACCM
SSC
SSC |
· Hypoglycemia must be rapidly diagnosed and promptly treated.
· In paIents with sepsis, a 10% dextrose containing IV solution can be run at maintenance rate to provide age appropriate glucose delivery and to prevent hypoglycemia.
· Blood glucose levels below 180 mg/dL (10 mmol/L) should be targeted.
· Insulin therapy targeting a blood glucose at or below 140 mg/dL (7.8 mmol/L) is NOT recommended |
High
High
Moderate |
Weak (conditional)
Weak (conditional)
Good practice statement
Strong
|
|
G2 |
What is the target calcium concentration in patients with shock? |
ACCM |
· Calcium replacement should be directed to normalize ionized calcium concentration |
High |
Weak (conditional)
|
|
G3 |
What is the role of thyroid replacement in patients with shock? |
ACCM
SSC |
· Thyroid replacement can be lifesaving in children with thyroid insufficiency and catecholamine-resistant shock. · The routine use of levothyroxine in children with septic shock and other sepsis associated organ dysfunction in a sick euthyroid state is not recommended. |
High
Low |
Weak (conditional)
Weak (conditional)
|
|
Table 10. Recommendations |
|
|
|||
|
H. TherapeuAc end-points and hemodynamic assessment/ monitoring |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
H1 |
What are the end-points/ resuscitation goals denoting successful resuscitation & stabilization during the first hour of management of shock (primarily in the ED)? |
ACCM
|
· The management goals in the first hour should be to maintain/ restore: *Airway, oxygenation, and ventilation *Circulation -normal blood pressure for age (only reliable when pulses palpable) -normal pulses with no differential between the quality of peripheral & central pulses -threshold HR -perfusion: Capillary refill less than or equal to 2 seconds, warm extremities, urine output greater than 1mL/kg/hr, normal mental status *Normal glucose concentration, normal ionized calcium concentration. |
High
|
Weak (conditional)
|
|
H2 |
What additional end-points/ therapeutic goals are applicable after the first hour of management of shock (primarily in the PICU/ HDU)? |
ACCM |
· The following additional goals are applicable beyond the first hour: -Perfusion pressure (MAP-CVP or MAP-IAP) appropriate for age. -ScvO2 greater than 70% -CI greater than 3.3 and less than 6.0L/min/m2 -Normal INR, anion gap, and lactate. |
High |
Weak (conditional)
|
|
H3 |
What are the methods of hemodynamic assessment/ monitoring needed to guide the management of shock? |
ACCM
SSC
US |
· On-going resuscitation should be guided by hemodynamic assessment & monitoring including: - Heart rate, blood pressure, pulse pressure, capillary refill/ skin perfusion analysis and temperature - Pulse-oximetry and continuous ECG monitoring - CVP - Urine output - Laboratory (Arterial blood gases, ScvO2, lactate, glucose and ionized Ca. · Assessment of CI and SVRI using advanced hemodynamic monitoring is recommended when available. Methods include: - invasive arterial BP monitoring with pulse-contour analysis - serial ultrasonographic assessment.
· The use of cardiac ultrasonography to assess the
efficacy of fluid resuscitation, ventricular
function and inotropic support. - electrical impedance cardiometry. · In patients with cardiogenic shock, repeated determination of troponin levels can be used to assess the severity of myocardial involvement as well as the response to treatment. |
High
Low
High |
Weak (conditional)
Weak (conditional)
Weak (conditional)
Good practice statement
Good practice statement |
|
Table 11. Recommendations |
|
|
|||
|
I. Inotropes, vasopressors and vasodilators |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
I1 |
When should inotropic/ vasopressor support be initiated? |
SSC
ISHLT |
· It is reasonable to begin vasoacAve infusions aNer 40–60 mL/kg of fluid resuscitation if the patient continues to have evidence of abnormal perfusion, or sooner if fluid overload develops or other concerns for fluid administration are present.
· Use of intravenous inotropic agents in the absence of clinical evidence of hypotension, low CO and/or decreased end-organ perfusion is potentially harmful. |
Moderate |
Good practice statement
Strong
|
|
I2 |
Which inotropes/ vasopressors should initially be used? |
ACCM
ISHLT |
· In septic shock: -Central epinephrine can be started for “cold shock” (0.05–0.3 μg/kg/min) or norepinephrine can be titrated for “warm shock”. -Central dopamine can be Atrated to a maximum of 10 μg/kg/min. -Epinephrine or norepinephrine is more likely to be beneficial. N.B. In children, “cold shock” is more common. · In cardiogenic shock: -Milrinone and /or dobutamine can be used as first- line therapy -It is probably advisable to use milrinone in post- cardiac surgery patients and in cases with impaired RV function and/or associated pulmonary hypertension. |
High
Low |
Weak (conditional)
Weak (conditional)
|
|
I3 |
How should inotropic/ vasoactive support be modified in patients with shock not responding to first-line agents? |
ACCM
ACCM
ACCM
ISHLT |
When a patient requires the use of inotropes/ vasopressors, frequent evaluation of BP, CO, SVR and peripheral perfusion is needed to guide further combination of drugs and/or fluids. · Septic shock With Low CI, Normal Blood Pressure, and High SVR: - Milrinone is considered the first-line inodilator in patients with epinephrine resistant shock and normal blood pressure. - Additional volume loading may be necessary to prevent hypotension. - Norepinephrine can partly reverse hypotension associated with inodilators. - Nitroprusside or nitroglycerin may be considered as second-line vasodilators. - Levosimendan and enoximone may have a role with persistently low CO.
· Septic shock With Low CI, Low Blood Pressure, and Low SVR: -Norepinephrine can be added to/or substituted for epinephrine to increase DBP and SVR. -Once an adequate blood pressure is achieved, dobutamine, milrinone, enoximone or levosimendan may be added to norepinephrine to improve CI and ScvO2 .
· Septic shock With High CI and Low SVR: - When titration of norepinephrine and fluid does not resolve hypotension, vasopressin, angiotensin, or terlipressin can be helpful in restoring blood pressure - These drugs can reduce CO so CO/ScvO2 monitoring is necessary. Low-dose epinephrine or dobutamine may be added to improve CO. · Cardiogenic shock with low CI refractory to milrinone &/or dobutamine: -Epinephrine has a role in the face of refractory hypotension and poor end-organ perfusion. -Levosimendan may be considered in children unresponsive to traditional inotropic therapy. |
High
High
High
Low |
Weak (conditional)
Weak (conditional)
Weak (conditional)
Weak (conditional)
|
|
Table 12. Recommendations |
|
|
|||
|
J. Refractory shock |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
J1 |
What is the management of pediatric shock refractory to fluids and pharmacological support? |
ACCM
ACCM
SSC |
· Children with refractory shock must be suspected to have unrecognized morbidities; such as: -Inappropriate source control of infection (remove nidus and use effective antibiotics) -Pericardial effusion (pericardiocentesis) -Pneumothorax (thoracentesis) -Hypoadrenalism (adrenal hormone replacement) -Hypothyroidism (thyroid hormone replacement) -Ongoing blood loss (blood replacement/hemostasis) -Increased IAP (peritoneal catheter or abdominal release) -Necrotic tissue (nidus removal) -Excessive immunosuppression (wean immunosuppressants), or immunocompromise (restore immune function; e.g., white cell growth factors/transfusion for neutropenic sepsis).
· ECMO is an important option to consider in refractory shock when potentially reversible causes are addressed.
· Venovenous ECMO is suggested in children with sepsis-induced PARDS and refractory hypoxia. Venoarterial ECMO is suggested in children with septic shock refractory to all other treatments. |
Moderate
High
Very low
|
Weak (conditional)
Weak (conditional)
Weak (conditional)
|
|
Table 13. Recommendations |
|
|
|||
|
K. Coticosteroids and antibiotics |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
K1 |
What is the role of corticosteroids and antibiotics in septic shock? |
SSC
ACCM
ACCM |
· IV hydrocortisone may be used if adequate fluid resuscitation and vasopressor therapy are not able to restore hemodynamic stability.
· Ideally after attaining a blood sample for subsequent determination of baseline cortisol concentration.
· In septic shock, broad spectrum antibiotics should be initiated within 60 minutes After obtaining blood culture if it does not delay antibiotic administration |
Low
High
High
|
Weak (conditional)
Weak (conditional)
Weak (conditional)
|
|
Table 14. Recommendations |
|
|
|||
|
L. Specific management of anaphylactic shock |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
L1 |
What is the specific management of anaphylactic shock? |
EAACI
EAACI
EDP-Ana
EAACI
EDP-Ana
EAACI EDP-Ana |
· Positioning: patients experiencing anaphylaxis should be positioned supine with elevated lower extremities if they have circulatory instability, sitting up if they have respiratory distress, and in recovery position if unconscious.
· Adrenaline: Adrenaline must promptly be administered as the first-line treatment for the emergency management of anaphylaxis.
- By intramuscular injection into the mid-outer thigh.
- In patients requiring repeat doses of adrenaline, these should be administered at least 5 min apart.
- If the patient is not responding to epinephrine injections, IV infusion of epinephrine should be given in a monitored setting.
- Do not routinely administer antihistamines or corticosteroids instead of epinephrine. There is no substitute for epinephrine in the treatment of anaphylaxis.
· Other therapies: -Trigger of the anaphylaxis episode should be remove.
- Administer additional vasopressors If parenteral epinephrine and fluid resuscitation fail to restore blood pressure.
- Administer an inhaled b-agonist if bronchospasm is a component of anaphylaxis.
- Administration of antihistamines and corticosteroids should be considered adjunctive therap.
- Systemic glucocorticosteroids may be used as they may reduce the risk of late phase respiratory symptoms.
- High-dose nebulized glucocorticoids may be beneficial for upper airway obstruction.
· Strongly consider observing patients who have experienced anaphylaxis for at least 4 to 8 hours and observe patients with a history of risk factors for severe anaphylaxis (such as asthma, previous biphasic reactions, or protracted anaphylaxis) for a longer period. Patients who have experienced anaphylaxis should consult an allergist/ immunologist.after discharge. |
Very low
Very low
Moderate
Very low
Moderate
Moderate
Very low
Moderate
Moderate
Moderate
Very low
Very low
Moderate |
Weak (conditional)
Weak (conditional)
Strong
Weak (conditional)
Strong
Weak (conditional)
Weak (conditional)
Strong
Strong
Weak (conditional)
Weak (conditional)
Weak (conditional)
Weak (conditional)
|
➡️Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Professor of Pediatrics and Pediatric Intensive Care, kasr Alainy, Cairo University |
Clinical expert |
||||
|
Shereen Abdel Monem Mohamed |
Assistant Professor of Pediatrics and Pediatric Intensive Care, kasr Alainy, Cairo University |
Clinical expert |
|||
|
Tarek Abd El Latef Abd El Aziz |
Lecturer of Pediatrics and Pediatric Intensive Care, Zagazig University |
Clinical expert |
|||
|
Dalia A. Abdelrahman |
Professor of pediatric critical care medicine, Zagazig university |
Clinical expert |
|||
|
Marwa Nabil Saad |
Assistant lecturer of pediatric critical care, Cairo University |
Clinical expert |
|||
|
Hafez Bazaraa |
Professor of Pediatrics Head of Pediatric Critical Care Unit, Cairo University |
Clinical expert |
|||
|
Noha El Anwar |
Lecturer of pediatric critical care, Cairo University |
Clinical expert |
|||
|
Mervat Gamal Eldin Mansour |
Professor of Pediatrics and Pediatric Critical Care, Ain Shams university |
Clinical expert |
|||
|
Azza Ahmed Eltayeb |
Professor of pediatrics and Pediatric intensive care, Assiut University |
Clinical expert |
|||
|
Effat Hussein Assar |
Assistant professor of pediatrics. Head of PICU, Benha university hospital
|
Clinical expert |
|||
|
Khaled Talaat |
Professor of Pediatrics Head of Pediatric Critical Care Unit Tanta University |
Clinical expert |
|||
|
Baher Matta Hanna |
Professor of Pediatrics & Pediatric Cardiology Cairo University & AFCM |
Clinical expert |
|||
|
Ahmed rezk ahmed |
Associate professor of pediatrics and PICU Ain shams university |
Clinical expert |
|||
|
Mohamed Mahmood Ahmed Romih |
professor of pediatrics and PICU, Zagazig university (ZU) |
Clinical expert |
|||
|
Sally Ahmed Farid El-Sahrigy |
professor of Pediatrics and Pediatric Cardiology Former Head Of Pediatrics Department Medical Research and Clinical Studies Institute National Research Center NRC |
Clinical expert |
|||
|
Hanaa Abdel Rady |
Associate professor of pediatrics and PICU ain shams university |
Clinical expert |
|||
|
Tarek Abdel Lateef |
Associate professor of pediatrics and PICU ain shams university |
Clinical expert |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
|
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Alyaa A. Kotby |
Professor of Pediatrics Former head of the Pediatric Department & Pediatric Cardiology Division Children Hospital, Ain Shams University |
||||
|
Prof. Khaled Talaat Muhammad |
Professor of Pediatrics Head of Tanta University Hospital PICU Faculty of Medicine, Tanta University |
||||
|
Prof. Mohammed Attia El-Bayoumi |
Professor of Pediatrics and Pediatric Critical Care Faculty of Medicine, Mansoura University |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
▪️ The GDG/ GAG acknowledge EPG for its help in completing this project.
▪️ We acknowledge …………………guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️ This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
||
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
||
|
BP |
Blood pressure |
||
|
CI |
Cardiac index |
||
|
CO |
Cardiac output |
||
|
CPG |
Clinical Practice Guideline |
||
|
CRRT |
Continuous renal replacement therapy |
||
|
CVP |
Central venous pressure |
||
|
DBP |
Diastolic blood pressure |
||
|
DHS |
Demographic and Health Survey |
||
|
ECG |
Electrocardiography |
||
|
ECMO |
Extra-corporeal membrane oxygenation |
||
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
||
|
EPG CPG |
EPG Clinical Practice Guideline |
||
|
ERG |
External Review Group |
||
|
GAG |
Guideline Adaptation Group |
||
|
GDG |
Guideline Development Group |
||
|
GPS |
Good Practice Statement |
||
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
||
|
HDU |
High dependency unit |
||
|
HR |
Heart rate |
||
|
HVHF |
High volume hemofiltration |
||
|
IAP |
Intra-abdominal pressure |
||
|
INR |
International normalized ratio |
||
|
IVIG |
Intravenous immune globulin |
||
|
MAP |
Mean arterial blood pressure |
||
|
NIAID |
National Institute of allergy and infection |
||
|
PAOP |
Pulmonary artery occlusion pressure |
||
|
PH |
Pulmonary hypertension |
||
|
PICO |
population, intervention, comparison, and outcomes |
||
|
Patient population, intervention, professionals, outcomes, and healthcare context |
||
|
PICU |
Pediatric intensive care unit |
||
|
PP |
Pulse pressure |
||
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
||
|
RV |
Right ventricle/ right ventricular |
||
|
ScvO2 |
Central venous oxygen saturation |
||
|
SI |
Stroke index |
||
|
SpO2 |
Arterial oxygen saturation by pulse oximeter |
||
|
SV |
Stroke volume |
||
|
SVR |
Systemic vascular resistance |
||
|
SVRI |
Systemic vascular resistance index |
||
|
US |
Ultrasonography/ ultrasonographic |
- Glossary
➡️Acceptability
Is the extent to which the users are likely to adopt a recommendation, based on internal qualities such as clarity, comprehensiveness, and logical reasoning and on external factors such as the burden imposed on the process and system of care, patient and providers attitudes and beliefs, and patients needs, expectations, and preferences.
➡️Adaptatioon (of guidelines)
Is the systematic approach to considering the use and/or modification of (a) guidelines(s) produced in one cultural and organizational setting for application in different context?
Adaptation can be used as an alternative to de novo guideline development or for customizing (an) existing guideline(s) to suit the local context.
➡️Admission
Admission, for the purpose of this guideline, refers to a child being registered and entering inpatient care as a patient. This is distinguished from the term “enrolment”, which is used for outpatient care.
➡️Adoption (of a guideline)
Is the acceptance of a guideline as a whole after the assessment of its quality, currency, and content. When health care providers (or other users of recommendations) adopt a guideline, they feel committed to change their practices in accordance with the recommendations of the guideline.
➡️Applicability
Is the extent to which the users are able to put a recommendation into practice, based on internal qualities such as a clearly defined eligible patient population that matches the population to which the intervention is targeted in the local setting and external factors such as the availability of the necessary knowledge, skills, provider time, staff, equipment, and other
resources.
➡️Applicability is sometimes taken as a synonym for feasibility:
- Feasibility of the acquisition of necessary skills and knowledge
- Feasibility of the necessary increase in provider time, staff, equipment, and so on.
➡️Culture
Culture represents the norms and values of a specific group, community, or population.
➡️Diffusion
Is a passive means of transferring knowledge; it is not directed towards a target audience (e.g. publication of articles in medical journals).
➡️Dissemination
Is more active than diffusion in that it targets a specific audience and involve tailoring the information for that audience (e.g. of dissemination strategies include targeted mailings, presentations, and press conferences.
➡️Evidence-based principles
Evidence-Based Medicine (EBM) has been defined as ― the conscienIous, explicit, and judicious use of current best evidence in making decisions about the care of individual patients. The practice of EBM means integrating individual clinical expertise with the best available external clinical evidence from systematic research.
➡️Evidence tables
Are summaries of the most salient information from studies identified in the systematic review. The elements of evidence tables are dependent on the types of information in studies related to a particular topic but might include information such as the article reference, the study type (e.g. RCT or Cohort), the number of patients and their characteristics, and the intervention, comparison arms, outcome measures, and effect sizes.
➡️Guideline or Clinical Practice Guideline (CPG)
Systematically developed statements about specific health problems, intended to assist practitioners and patients in making decisions about appropriate health care.
➡️Guideline consistency
Agreement between the evidence and the recommendations, based on the:
- Comprehensiveness of the study search and selection process,
- Coherence between the results of the studies and their interpretation by the guideline authors, and
- Transparency between interpretation and recommendations.
➡️Guideline content
In the ‘ADAPTE Manual and Resource Toolkit for Guideline Adaptation’ document, guideline content refers to the recommendations in the source guidelines.
➡️Guideline currency
A CPG may be considered up to date ―when (no) new informaIon on intervenIons, outcomes, and performance justifies updating (it).
➡️Guideline quality
By quality of clinical practice guidelines, we mean the confidence that the potential biases of guideline development addressed adequately and that the recommendations are both internally and externally valid, and are feasible for practice. This process involves taking into account the benefits, harms and costs of the recommendations, as well as the practical issues attached to them. Therefore, the assessment (of quality) includes judgments about the methods used for developing the guidelines, the content of the final recommendations, and the factors linked to their uptake.
➡️Guideline topic
In the ADAPTE Manual and Resource Toolkit for Guideline Adaptation' document, the topic
refers to the theme of the guideline, as described in the guideline title, for a targeted population (disease and patients) and intervention. The purpose, the audience, and the setting intended for the guideline, although not necessarily explicitly stated in the title, are also part of the topic. A guideline on a given topic may contain more than one health question.
➡️Health question or clinical question or key question
Is a precisely described health issue (e.g. clinical, professional practice or public health) relating to the topic of the guideline? Guideline may include one or more questions.
➡️Implementation
Implementation includes methods to promote the uptake of research findings into routine healthcare in both clinical and policy contexts and hence to improve the quality and effectiveness of healthcare. It includes the study of influences on healthcare professional and organizational behavior.
➡️Intra-class correlations
Intra-class correlations provide a measurement of the extent to which two or more raters agree
when rating the same set of things. It is a reliability index and is typically a ratio of the variance
of interest over the sum of the variance of interest plus error.
➡️Recommendation
Any statement that promote or advocate a particular course of action in clinical care.
➡️Stakeholder
A stakeholder is an individual, group and/or organization with a stake in your decision to implement a guideline. Stakeholders include individuals or groups who will be directly or indirectly affected by the implementation of a guideline.
➡️Source guideline
In the ADAPTE Manual and Resource Toolkit for Guideline Adaptation' document, source guideline refer to those guidelines selected to undergo assessments of quality, currency, content, consistency, and acceptability/applicability and upon which an adapted guideline may
be based.
- Introduction
Shock is a life-threatening emergency characterized by circulatory failure and impaired tissue
perfusion. In addition to clinical and hemodynamic condition, oxygen utilization and/or cellular
variables have been used to define shock.(1) Hypotension is neither a constant nor an early finding in pediatric shock and prompt recognition requires clinical assessment for tissue hypoperfusion and a high index of suspicion.(2)(3) Involved pathophysiological mechanisms include a combination of reduced intravascular volume, abnormal myocardial function, reduced vascular tone with inappropriate vasodilatation (vasoplegia) and/or circulatory obstruction associated with conditions such as cardiac tamponade, tension pneumothorax or massive pulmonary embolism. Clinical conditions often associated with shock include hypovolemia (eg bleeding and severe dehydration), severe sepsis, cardiogenic shock and anaphylaxis.(4-8)
Severe sepsis and septic shock represent a dysregulated immune response to an invasive infection. (9) Even without shock, children with infections frequently develop fever, tachycardia and vasodilatation as a result of an inflammatory response. Septic shock should be suspected if these manifestations are associated with a change in mental status, and the diagnosis made when tissue perfusion is impaired. Patients with septic shock may present with predominantly low cardiac output, vasoconstriction, delayed capillary refill and cold extremities (cold shock); or with predominantly vasodilatation, wide pulse pressure, warm extremities and increased cardiac output (warm shock).(1)(5) Contrary to the case in adults and some adolescents, most cases of septic shock in infants and children present as cold shock, with low cardiac output associated with hypovolemia (deficient intake and capillary leak) and/or myocardial insufficiency.(2)(5) The clinical distinction between cold and warm shock is not always clear-cut and has sometimes been disputed as a guide to initial inotropic/ vasopressor support. (10)
While cardiogenic etiology of shock may be quite obvious, such as following cardiac surgery and in those with cardiac disease, cardiogenic shock should be suspected in patients with signs such as a gallop rhythm, heart murmur, evidence of circulatory congestion (pulmonary rales, jugular venous distension, hepatomegaly or worsening with volume expansion) or arrhythmia. (4)
Bedside cardiac ultrasonography can be helpful in diagnosis and assessment of myocardial function. (11)
Anaphylaxis is a life-threatening systemic hypersensitivity reaction to triggers such as parenteral medications, insect venoms and food allergens. Prompt intervention is critical and early injection of epinephrine (adrenaline) is essential as it is the only drug shown to reduce mortality and hospitalization. (12-15)
An immediate stepwise approach with ongoing monitoring and clear end-points is necessary for successful management of shock. (1)(7) Initial evaluation and resuscitation should occur irrespective of patient location (emergency department, intensive care unit, general ward), even if it is clear that transfer to a higher level of care will be needed (5)
While most patients with shock benefit from intravascular volume expansion, the required amount and frequency of fluid administration can vary significantly and should depend on assessment of fluid responsiveness. The role of inotropes, vasopressors and vasodilators also varies depending on the prevailing pathophysiology, which can change even in the same patient. (4)(5)(16)(17) There is an increasing role for objective non-invasive hemodynamic assessment using tools such as point-of care ultrasound, electrical impedance cardiometry and
measurement of central venous oxygen saturation (ScvO2) (10)(11)(16)(18) to supplement clinical assessment and enable treatment appropriate to the actual pathophysiological derangements present. Other critical aspects of management include support of other systems and treatment of the underlying cause. (5)
Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the management of Pediatric shock. It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to
1) To promote effective and timely management of pediatric patients with shock.
2) To improve survival of pediatric patients with shock.
This version of the guideline includes recommendations and good practice statements for Management of Pediatric shock.
- Methods
Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: pediatric, emergency, shock.
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in the last 10 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% in the AGREE II Domain 3 (rigor of development) was retained).
Six guidelines were considered eligible for the AGREE II appraisal instrument which were:
1. American College of Critical Care Medicine Clinical Practice (ACCM). Parameters for Hemodynamic Support of Pediatric and Neonatal Septic Shock. Crit Care Med. (2017)
2. Surviving Sepsis Campaign (SSC) International Guidelines for the Management of Septic Shock and Sepsis-Associated Organ Dysfunction in Children. Pediatr Crit Care (2020)
3. The International Society for Heart and Lung Transplantation (ISHLT), Guidelines for the management of pediatric heart failure. (2014)
4. Guidelines for the Appropriate Use of Bedside General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients.
General Ultrasonography. Crit Care Med. (2015).
Cardiac Ultrasonography. Crit Care Med. (2016)
5. EAACI Food Allergy and Anaphylaxis Guidelines Group. Anaphylaxis: Guidelines from the European Academy of Allergy and Clinical Immunology. (2014)
6. Emergency department diagnosis and treatment of anaphylaxis (EDP-Ana): a practice parameter. Ann Allergy Asthma Immunol. (2014)
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements.
➡️Contributors to the guideline development process:
➡️Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of emergency
The main functions of the clinical panel were adolopment of management of pediatric shock guideline, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as eminent international reviewers in the management of shock.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [25]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations (both ETD and changing strength of recommendation were not done in this guideline).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7.Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include awell-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing pediatric shock management strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for management of acute malnutrition
3. Arabic Educational materials for nurses and mothers
Data from Pediatric Advanced Life Support Provider Manual, American Hear t Association
Cardiac index, Stroke index and Systemic vascular resistance index
|
|
|
Target range |
|
Cardiac index (CI) |
Cardiac output/ m2 BSA |
3.5-5.5 L/min/m2 |
|
Stroke index (SI) |
Stroke volume/ m2 BSA |
30-60 mL/m2 |
|
Systemic vascular resistance (SVRI) |
Systemic vascular resistance |
Systemic vascular resistance |
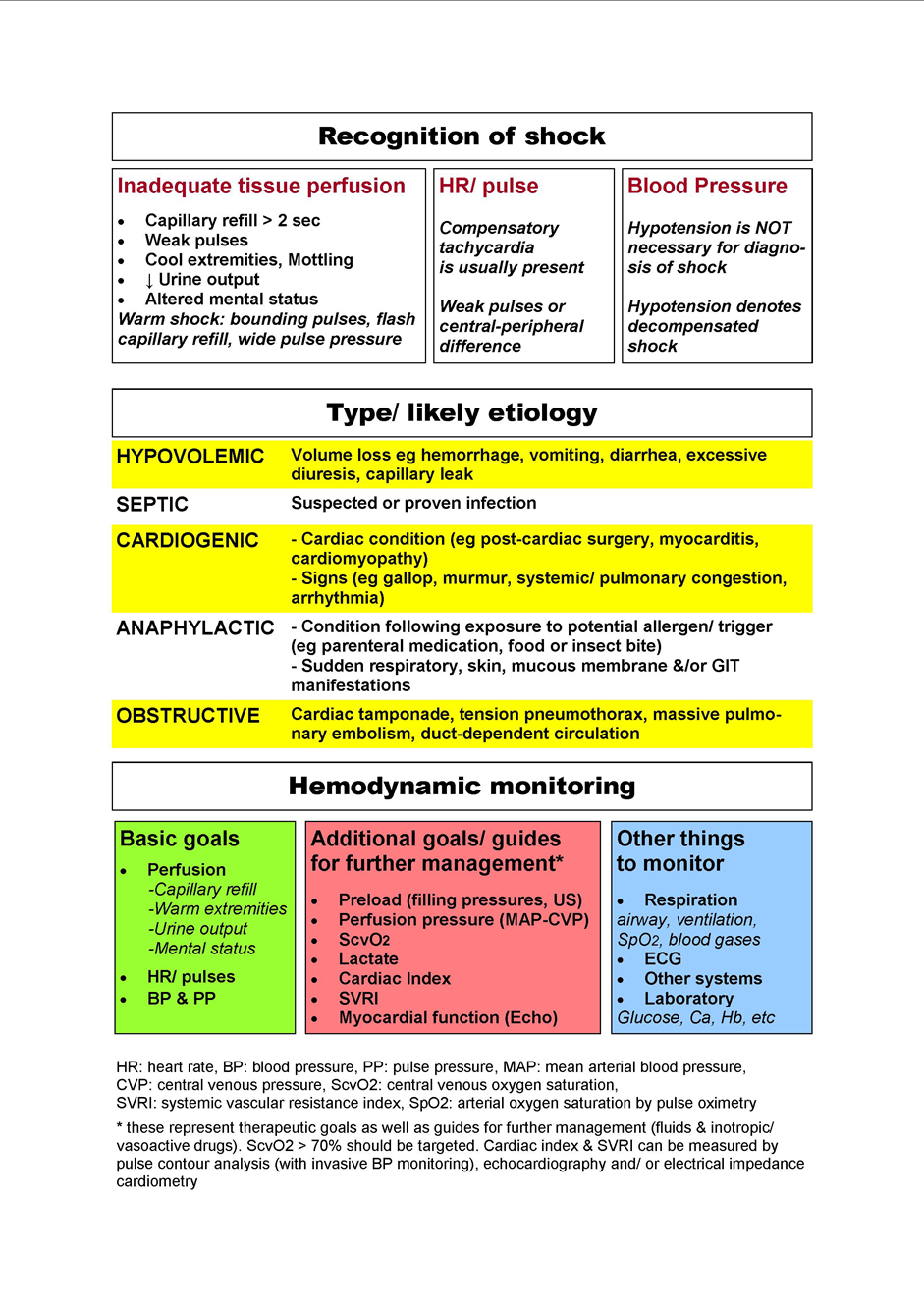
Chart for assessment
and monitoring in shock
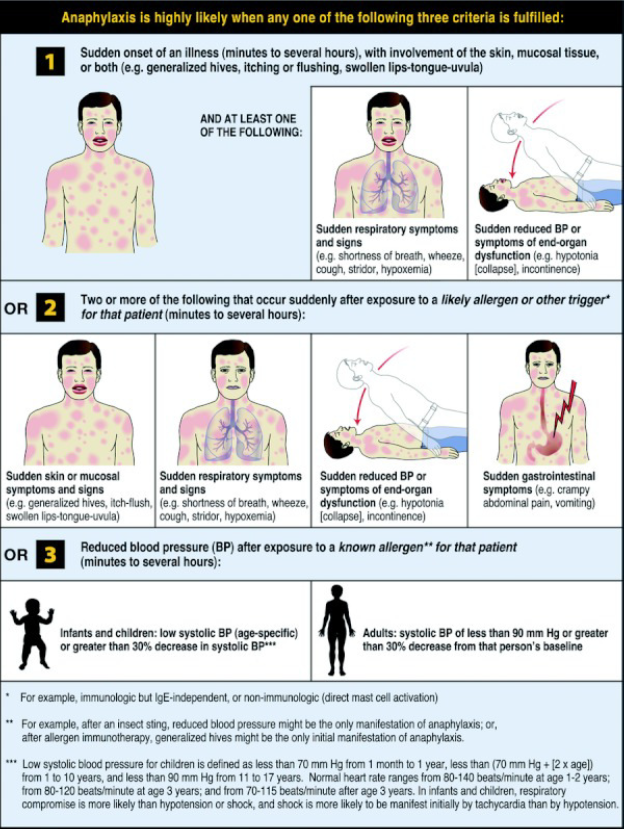
Anaphylaxis scenarios
scenarios described by the National Institutes of Allergy and Infectious Disease (NIAID) Panel (19)
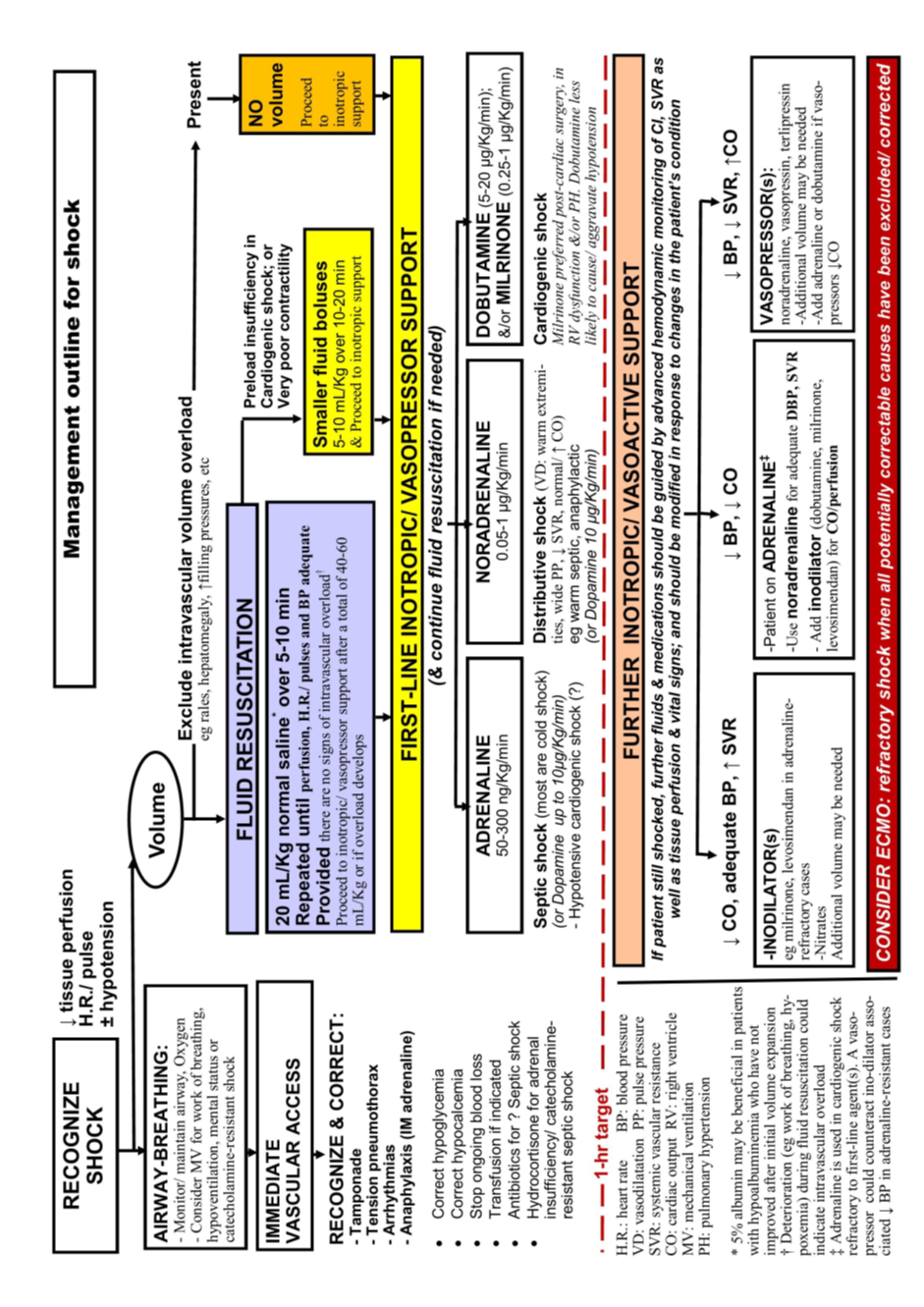
Doses of inotropes, vasopressors and vasodilators
|
Agent |
Main action |
Dose |
Remarks |
|
Adrenaline |
Inotrope, dose dependent |
0.05-1μg/Kg/min |
Dose >0.3μg/Kg/min has significant vasopressor effect; addition of nor adrenaline may be preferred then |
|
Adrenaline (anaphylaxis) |
|
0.01mg/Kg IM (max. 0.5mg) |
With autoinjector: 0.15mg from 7.5Kg 0.3mg from 25-30Kg |
|
Noradrenaline |
Vasopressor |
0.05-2μg/Kg/min |
Typical up to 0.5 μg/Kg/min |
|
Dopamine |
Inotrope, dosedependent |
5-10μg/Kg/min |
Doses from 10μg/Kg/min noradrenaline preferred then |
|
Dobutamine |
Ino-dilator |
5-20μg/Kg/min |
|
|
Milrinone |
Ino-dilator |
0.25-1μg/Kg/min |
Loading dose (50 μg/Kg/5-10min) usually omitted to avoid hypotension |
|
Levosimendan |
Ino-dilator |
0.1-0.2μg/Kg/min |
|
|
Nitroglycerin |
Vasodilator |
0.5-20μg/Kg/min |
Typical up to 5μg/Kg/min |
|
Vasopressin |
Vasopressin |
0.05-2mU/Kg/min |
Typical up to 0.5mU/kg/min |
|
Terlipressin |
Vasopressor |
Loading 20μg/Kg, then 4-20 μg/Kg/hr |
|
- Limitations and suggestions for further research needs
➡️Future research recommendations for the management of shock in children in the Egyptian context could include:
· Conduct national and regional studies to determine the epidemiology, incidence, and mortality of different types of pediatric shock in Egypt.
· Identify Egypt-specific risk factors contributing to shock severity and outcomes, including malnutrition, anemia, and delayed hospital presentation.
· Investigate the optimal choice and dosing of inotropes and vasopressors in resource-limited settings, and their outcomes in both secondary and tertiary hospitals.
· Investigate shock outcomes in special pediatric populations, such as children with congenital heart disease, and immunocompromised children.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for shock in children.
➡️Challenges
· Insufficient national epidemiological data on the burden, patterns, and outcomes of pediatric shock in Egypt.
· Lack of standardized, evidence-based fluid resuscitation practices adapted to local comorbidities and resource constraints.
· Ongoing gaps in provider training related to early detection and timely management of pediatric shock.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline
The following are three performance measures or indicators for implementing this adapted CPG for management of shock in children:
1. Adherence to management of shock Guidelines
·Numerator: Number of children with shock who received treatment as per guideline recommendations.
· Denominator: Total number of children diagnosed with shock.
· Data Source: Hospital or clinic patient records.
2.Duration of Hospital Stay
· Numerator: Total number of hospital stay days for children with shock.
· Denominator: Total number of children admitted with shock.
· Data Source: Hospital admission and discharge records.
3. Rate of Readmission
· Numerator: Number of children readmitted with symptoms of shock within a certain period (e.g., 30 days) after discharge.
· Denominator: Total number of children initially admitted with shock.
· Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG emergency GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Davis A, Carcillo J, Aneja R, et al. American College of Critical Care Medicine Clinical Practice Parameters for Hemodynamic Support of Pediatric and Neonatal Septic Shock. Crit Care Med. 2017;45(6): 1061-1093.
2. Aneja R, Carcillo J. Differences between adult and pediatric septic shock. Minerva anestesiologica. 2011;77:986–92.
3. RecogniIon of shock. In: Pediatric Advanced Life Support Provider Manual, Chameides L, Samson RA, Schexnayder SM, Hazinski MF (Eds), American Heart AssociaIon, 2011.
4. Waltzman M. IniIal management of shock in children. Uptodate. 2016. www.uptodate.com.
5. MarIn K, Weiss SL. IniIal resuscitaIon and management of pediatric septic shock. Minerva Pediatr. 2015;67(2):141-158.
6. Hartman ME, L-ZW, Angus DC, Watson RS. Trends in the epidemiology of pediatric severe
sepsis. Pediatric Critical Care Medicine. 2013;14:686–93.
7. Brissaud O, Botte A, Cambonie G, et al. Experts’ recommendations for the management of
cardiogenic shock in children. Ann. Intensive Care. 2016; 6(1):14-29.
8. Simons F, Motohiro Ebisawa M, Sanchez-Borges M, et al. 2015 update of the evidence base: World Allergy Organization anaphylaxis guidelines. World Allergy Organ J. 2015; 8(1):32-47.
9. Hotchkiss RS, Karl IE. The pathophysiology and treatment of sepsis. The New England journal of medicine. 2003;348:138–50.
10. Weiss S, Peters M, Alhazzani W, et al. Surviving Sepsis Campaign International Guidelines for the Management of Septic Shock and Sepsis-Associated Organ Dysfunction in Children. Pediatr Crit Care Med. 2020;21(2):e52-106.
11. Levitov A, Frankel H, Blaivas M, et al. Guidelines for the Appropriate Use of Bedside General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients—Part II: Cardiac Ultrasonography. Crit Care Med.2016;44(6):1206-1227.
12. Muraro A, Roberts G, Worm M, et al., EAACI Food Allergy and Anaphylaxis Guidelines Group. Anaphylaxis: Guidelines from the European Academy of Allergy and Clinical Immunology. Allergy. 2014; 69(8):1026-1045.
13. Campbell RL, Li JT, Nicklas RA, Sadosty AT; members of the joint task force; pracIce parameter workgroup. Emergency department diagnosis and treatment of anaphylaxis: a practice parameter. Ann Allergy Asthma Immunol. 2014;113(6):599-608.
14. Fleming JT, Clark S, Camargo CA, Rudders SA. Early treatment of food induced anaphylaxis with epinephrine is associated with a lower risk of hospitalizaIon. J Allergy Clin Immunol Pract. 2015;3:57–62.
15. Xu YS, Kastner M, Harada L, Xu A, Salter J, Waserman S. Anaphylaxis-related deaths in Ontario: a retrospecIve review of cases from 1986 to 2011.Allergy Asthma Clin Immunol. 2014;10:38.
16. Fathi E, Narchi H, Chedid F. Noninvasive hemodynamic monitoring of septic shock in children. World J Methodol. 2018;8(1):1-8.
17. Boluyt N, Bollen CW, Bos AP, Kok JH, Offringa M. Fluid resuscitation in neonatal and pediatric hypovolemic shock: a Dutch Pediatric Society evidence-based clinical practice guideline. Intensive Care Med. 2006;32(7):995.
18. Frankel H, Kirkpatrick A, Elbarbary M, et al. Guidelines for the Appropriate Use of Bedside General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients-Part I: General Ultrasonography. Crit Care Med. 2015;43(11):2479-2502.
19. Simons FE, Ardusso LR, Bilò MB, El-Gamal YM, Ledford DK, Ring J, et al. World allergy organization guidelines for the assessment and management of anaphylaxis. [World Allergy Organ J. 2011;4(2):13-37.
20. University of Iowa, Stead family children hospital, PICU handbook https://uichildrens.org/health-library/continuous-infusions-picu-chart
21. Simons FER, Gu X, Silver NA, Simons KJ. EpiPen Jr versus EpiPen in young children weighing 15 to 30 kg at risk for anaphylaxis. J Allergy Clin Immunol. 2002;109:171–175.
22. Rodríguez-Núñez A, Oulego-Erroz I, Gil-Antón J, et al. Continuous terlipressin infusion as rescue treatment in a case series of children with refractory septic shock. Ann Pharmacother.
2310;44(10):1545-1553.
24. Kirk R, Dipchand A, Rosenthal D, et al. The International Society for Heart and Lung Transplantation Guidelines for the management of pediatric heart failure. J Heart Lung Transplant. 2014;33(9):888-909
25. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0.
26. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
27. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
28. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press).
29. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479.
30. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449.
31. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
32. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024).
References of materials used
in creating this EBCPG
1. ADAPTE Resource Toolkit versions 2.0 (2009) Available
from: www.g-i-n.net/documentstore/adapte-resource-toolkit-guideline-adaptation-version-2
(Version 2.0 downloaded free without registration).
2. Yasser Sami Amer YS, Elzalabany MM, Omar TEI, Ibrahim
AG and Dowidar NL.The ‘Adapted ADAPTE’:an approach to
improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria
Center for Evidence-Based Clinical Practice Guidelines. Journal of Evaluation
in Clinical PracIce 2015; 21: 1095 – 1106.
3. AGREE (II) Instrument available from the
www.agreecollaboration.org/instrument/.21
The following are source
guideline(s)
(used to produce the final single
adapted CPG):
ACCM: (1)
Davis A, Carcillo J, Aneja R, et al.
American College of Critical Care Medicine Clinical Practice Parameters for
Hemodynamic Support of Pediatric and Neonatal Septic Shock. Crit Care Med. 2017;45(6):
1061-1093
SSC: (10)
Weiss S, Peters M, Alhazzani W, et al. Surviving Sepsis Campaign International Guidelines for the Management of Septic Shock and Sepsis-Associated Organ Dysfunction in Children. Pediatr Crit Care Med. 2020;21(2):e52-106.
ISHLT: (23)
Kirk R, Dipchand A, Rosenthal D, et al. The International Society for Heart and Lung Transplantation Guidelines for the management of pediatric heart failure. J Heart Lung Transplant. 2014;33(9):888-909.
US: (18)(11).
Frankel H, Kirkpatrick A, Elbarbary M, et al. Guidelines for the Appropriate Use of Bedside
General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients-Part I: General
Ultrasonography. Crit Care Med. 2015;43(11):2479-2502.
Levitov A, Frankel H, Blaivas M, et al. Guidelines for the Appropriate Use of Bedside General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients—Part II: Cardiac Ultrasonography. Crit Care Med.2016;44(6):1206-1227.
EAACI: (12)
Muraro A, Roberts G, Worm M, et al., EAACI Food Allergy and Anaphylaxis Guidelines Group. Anaphylaxis: Guidelines from the European Academy of Allergy and Clinical Immunology. Allergy. 2014; 69(8):1026-1045.
EDP-Ana: (13)
Campbell RL, Li JT, Nicklas RA, Sadosty AT; members of the joint task force; practice parameter workgroup. Emergency department diagnosis and treatment of anaphylaxis: a practice parameter. Ann Allergy Asthma Immunol. 2014;113(6):599-608.
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Hanaa Ibrahim Rady |
Professor of Pediatrics and Pediatric Intensive Care, kasr Alainy, Cairo University |
None |
Not Applicable |
|
Shereen Abdel Monem Mohamed |
Assistant Professor of Pediatrics and Pediatric Intensive Care, kasr Alainy, Cairo University |
None |
Not Applicable |
|
Tarek Abd El Latef Abd El Aziz |
Lecturer of Pediatrics and Pediatric Intensive Care, Zagazig University |
None |
Not Applicable |
|
Dalia A. Abdelrahman |
Professor of pediatric critical care medicine, Zagazig university |
None |
Not Applicable |
|
Marwa Nabil Saad |
Assistant lecturer of pediatric critical care, Cairo University |
None |
Not Applicable |
|
Hafez Bazaraa |
Professor of Pediatrics Head of Pediatric Critical Care Unit, Cairo University |
None |
Not Applicable |
|
Noha El Anwar |
Lecturer of pediatric critical care, Cairo University |
None |
Not Applicable |
|
Mervat Gamal Eldin Mansour |
Professor of Pediatrics and Pediatric Critical Care, Ain Shams university |
None |
Not Applicable |
|
Azza Ahmed Eltayeb |
Professor of pediatrics and Pediatric intensive care, Assiut University |
None |
Not Applicable |
|
Effat Hussein Assar |
Assistant professor of pediatrics. Head of PICU, Benha university hospital
|
None |
Not Applicable |
|
Khaled Talaat |
Professor of Pediatrics Head of Pediatric Critical Care Unit Tanta University |
None |
Not Applicable |
|
Baher Matta Hanna |
Professor of Pediatrics & Pediatric Cardiology Cairo University & AFCM |
None |
Not Applicable |
|
Ahmed rezk ahmed |
Associate professor of pediatrics and PICU Ain shams university |
None |
Not Applicable |
|
Mohamed Mahmood Ahmed Romih |
professor of pediatrics and PICU, Zagazig university (ZU) |
None |
Not Applicable |
|
Sally Ahmed Farid El-Sahrigy |
professor of Pediatrics and Pediatric Cardiology Former Head Of Pediatrics Department Medical Research and Clinical Studies Institute National Research Center NRC |
None |
Not Applicable |
|
Hanaa Abdel Rady |
Associate professor of pediatrics and PICU ain shams university |
None |
Not Applicable |
|
Tarek Abdel Lateef |
Associate professor of pediatrics and PICU ain shams university |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanis Sulieman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not Applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Prof. Alyaa A. Kotby |
Professor of Pediatrics Former head of the Pediatric Department & Pediatric Cardiology Division Children Hospital, Ain Shams University |
None |
Not Applicable |
|
Prof. Khaled Talaat Muhammad |
Professor of Pediatrics Head of Tanta University Hospital PICU Faculty of Medicine, Tanta University |
None |
Not Applicable |
|
Prof. Mohammed Attia El-Bayoumi |
Professor of Pediatrics and Pediatric Critical Care Faculty of Medicine, Mansoura University |
None |
Not Applicable |
➡️ Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the prevention and management of Shock in pediatrcis" on PubMed are: pediatrics, emergency, shock.
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|