Management of Kawasaki Disease and its Cardiac Sequelae
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Management of Kawasaki Disease and its Cardiac Sequelae |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 3 December 2025" Download Guideline
Table of contents
- - Executive Summary
- - Recommendations
- - Acknowledgments
- - Abbreviations
- - Introduction
- - Methods of search
- - Implementation Tools and Considerations
- - Limitations and suggestions for further research needs
- - Monitoring and evaluating the impact of the guideline
- - Updating of the guideline
- - References
- - Annexes
- Executive Summary
|
▪️ The diagnosis of KD should be considered in any child with a febrile illness and evidence of inflammation, particularly if fever persists longer than 4 days (weak) ▪️ There are different diagnostic criteria for KD. The AHA diagnostic criteria should be used to diagnose complete KD (weak) ▪️ Typical or classic KD is diagnosed if a fever is associated with ≥4 diagnostic criteria, with or without CAA, or if fever lasts at least for 4 days with ≥4 diagnostic criteria and eventual demonstration of CAA on echocardiography. (Strong) ▪️ Incomplete KD can be diagnosed when patients present with fever for ≥5 days and lack enough clinical criteria (≤3) to fulfill the diagnosis, with or without CAA. (Strong) ▪️ Atypical KD is diagnosed if fever, not otherwise explained, lasting for ≥ 5 days is associated with classic diagnostic criteria and non-classic manifestations, with or without CAA. (Strong) ▪️ In a patient in whom KD is suspected, but all criteria have not yet been fulfilled, the following clinical signs strengthen the suspicion of KD: Irritability and new erythema and/or induration at the site of previous BCG immunization. (weak) ▪️ MIS-C and KD may share overlapping clinical features, including conjunctival injection, oropharyngeal findings (red and/or cracked lips, strawberry tongue), rash, swollen and/or erythematous hands and feet, and cervical lymphadenopathy (Strong) ▪️ Epidemiologic studies of MIS-C suggest that younger children are more likely to be present with KD-like features, while older children are more likely to develop myocarditis and shock (Strong) ▪️ The following laboratory values should be determined: ESR, CRP, full blood count and liver function (bilirubin, AST/ALT), albumin, serum Na, renal function test, and urinalysis. Ferritin and fibrinogen should be considered if there is a concern for macrophage activation syndrome. Cerebrospinal fluid analysis may be important to rule out infectious meningitis. (Weak) ▪️Laboratory data are nonspecific to KD and can only support diagnosis in patients with suggestive clinical features. (Strong) ▪️ All patients with suspected KD should undergo echocardiography and ECG at baseline, as soon as the diagnosis is suspected. An intermediate echocardiogram, 2 weeks after the first IVIG, should be performed in all patients with KD whose initial echo was normal and in whom disease activity has been arrested (Strong) ▪️ In those with ongoing active inflammation (increasing or persistently elevated CRP and/or persisting signs and symptoms), ECG and echocardiography should be performed at least weekly to monitor the possible development of cardiac sequelae. In those with coronary abnormalities detected on initial echocardiography, echocardiography should then be performed at least twice weekly to monitor progression until there is clinical stabilization. (Strong) ▪️ Persistently febrile non-responders KD patients with CAA, impaired left ventricular function, mild/moderate mitral regurgitation or significant pericardial effusion need a more frequent echocardiogram check-up (at least twice per week). (Strong) ▪️In children with CAA, ECG and echocardiography should be performed 3- to 6-monthly, depending on the severity of the CAA. (weak) ▪️ For coronary artery sequelae, evaluation by Z-score is the standard method, and +2.5 or higher is defined as a long-term significant CAL (sequelae) (Strong ). For those above the age of 5 years, the definition of a giant aneurysm is ≥8mm inside diameter. (weak) ▪️ Cardiovascular CT scan and MR angiography, where available, are important to assess persistent CAA in children with KD and monitor the remodeling of either coronary or systemic arteries in the whole body. (weak)
▪️ IVIG is the standard treatment in KD. It must be administered at dose 2 g/kg of body weight in a single infusion, as soon as the diagnosis is confirmed or strongly suspected, with best response to IVIG documented when given within the 10th day from onset of fever. Administration should be performed over 12 h if the patient’s cardiac function is normal, or in 16–24 h for patients displaying cardiac failure. (Strong) ▪️ All patients diagnosed with KD who are treated with IVIG should be treated with anti-inflammatory dose of aspirin (30-50 mg/kg/day) until fever has settled for 48 h, clinical features are improving, and CRP levels are falling. The dose of aspirin should subsequently be reduced to an antiplatelet dose (3-5 mg/kg once daily). (Strong) ▪️ IVIG may not be administered if fever spontaneously disappears and no CAA are shown, and inflammatory markers (ESR and CRP) are within normal limits. (GPS) ▪️ IVIG should also be administered to children presenting after the 10th day of illness in case of persistent fever or no more fever but aneurysms and ongoing systemic inflammation, as shown by elevation of CRP (Strong) ▪️ In patients without CAA anti-platelet ASA treatment is to be discontinued 8 weeks after KD onset. In children who develop CAA it may be continued until the resolution of CAA lesions or indefinitely in case of its persistence. (Strong) ▪️ High-risk KD patients may receive initial therapy with IVIG + ASA + corticosteroid. (Strong) ▪️ The following laboratory values can be important in assessing risk stratification for IVIG resistance: Low sodium, raised bilirubin, raised Alanine Transferase, Low platelet count, high CRP, Low albumin. (Weak) ▪️ Resistant KD is defined by failure of response to IVIG and is revealed by recrudescent fever reoccurring or persisting 36–48 h after IVIG infusion. (Strong) ▪️ In case of failure (resistant KD), treatment should be implemented with a further infusion of IVIG + low-dose aspirin (3–5 mg/kg/day) and intravenous methylprednisolone or Infliximab (GPS) ▪️Corticosteroid treatment should be given to patients with severe KD (See recommendation C.7). (Strong) ▪️TNF-alpha blockade (e.g. infliximab) should be considered in KD patients with persistent inflammation despite IVIG, aspirin and corticosteroid treatment, after consultation with a specialist unit (Strong) ▪️ The use of Disease Modifying Antirheumatic Drugs (DMARDs) such as cyclosporin, cyclophosphamide and methotrexate, along with anakinra and plasma exchange, cannot be recommended, except on an individual basis after consultation with a specialist unit. (Weak) ▪️ For patients with acute KD and suspected or diagnosed MAS, treatment with IVIG for KD and additional agents to treat MAS is strongly recommended. For children with unexplained MAS, obtaining an echocardiogram with coronary artery measurements is strongly recommended. (Strong) ▪️ KD patients with medium-sized coronary artery aneurysms (Z-score ≥ 5 to 10 or absolute measurement <8 mm) or those with multiple and complex aneurysms may benefit from dual anti-platelet prophylaxis, based on low-dose ASA (at a single dose of 3–5 mg/kg/day) and clopidogrel (at a single dose of 0.2 mg/kg/day in children aged < 24 months and up to 1 mg/kg/day in children aged ≥ 24 months). (Strong) ▪️ Warfarin is used in combination with low-dose aspirin for patients with large CAA , history of MI (myocardial infarction), and thrombosis in the CAA. The dose is adjusted for the international normalized ratio of prothrombin time (PT-INR) target range of 2.0–2.5 (Strong) ▪️ It is imperative to treat KD patients having complex or severe CAA (Z score > 10, diameter >8 mm) with low-dose ASA associated with warfarin (keeping INR targeted at 2.0–3.0) or LMWH (if regular INR checking is difficult). Triple therapy with ASA, warfarin or LMWH and clopidogrel may be considered in KD patients with a relevant risk of thrombosis. (Strong) ▪️ Recombinant tissue plasminogen activator (rtPA) is the first-choice thrombolytic drug in children with KD complicated by coronary artery thrombosis; the glycoprotein IIb/IIIa inhibitor may be used in case of thrombosis with high risk of occlusion. Both therapies require a concomitant association with low-dose ASA and intravenous heparin. (Strong) ▪️ All inactivated vaccines can be safely administered at any time after IVIG in KD patients. Attenuated live virus vaccines (MMR, Varicella, and MMRV vaccines) should be administered 10–12 months after the administration of IVIG to avoid a reduced specific immune response of the vaccine in KD patients; influenza vaccination is recommended in KD patients receiving ASA. (Strong) ▪️ Low-Dose Aspirin is orally administered to patients with persistent CAA (Weak) ▪️ Statins may be used to prevent cardiovascular events in patients with CAL (coronary artery lesion). (Weak) ▪️ACEI (Angiotensin Converting Enzyme inhibitor) or ARB (Angiotensin Receptor Blocker) may be used if coronary artery stenosis is at risk in patients with CAL. Beta-blockers, calcium antagonists, or nitrates can be used to prevent Acute coronary syndrome (ACS) in patients with CAL (Strong) ▪️ Preventing the loss of follow-up (the so-called dropouts) is the most important issue in the management of the adolescent and young adult (AYA) generation. A preset plan of transfer for adult care should be available (GPS) (Weak). Guideline development process and methods After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs): 1- Italian Society of Pediatrics (SIP) CPG on general management of Kawasaki disease (2021) (1). Some recommendations were adopted from earlier version of this CPG (Italian CPG 2018) (2) as it has the same methodology of development. 2- JCS/JSCS 2020 CPG on Diagnosis and Management of Cardiovascular Sequelae in Kawasaki Disease (2020) (3) 3- American College of Rheumatology/Vasculitis Foundation (ACR/VF) CPG for the Management of Kawasaki Disease. (2021) (4). 4- European consensus-based recommendations for the diagnosis and treatment of Kawasaki disease - the SHARE initiative (2019) (5). 5- American College of Rheumatology/Vasculitis Foundation (ACR/VF) CPG for Multisystem Inflammatory Syndrome in Children Associated with SARS-CoV-2 and Hyperinflammation in Pediatric COVID-19: Version 2(2022) (6) 6- Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals from the American Heart Association. Circulation (7), and its updated version “Update on Diagnosis and Management of Kawasaki Disease: A Scientific Statement from the American Heart Association, 2024”. (8) We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development) - Adoption for most of the guideline recommendations. - Development of Good Practice Statements Recommendations and Good Practice Statements (GPS) ➡️This version of the CPG includes recommendations and good practice statements on the following four sub-sections: A. Diagnosis of Kawasaki Disease in Children The guideline covers the recommended criteria for diagnosis of classical cases of KD as well as atypical or incomplete cases. Laboratory investigations needed for initial diagnosis are covered in this section too as well as differentiation between KD and MISC. B. Investigations in Kawasaki disease. This section covers recommendations for laboratory as well as echocardiographic evaluation of patients with KD. Further details are given also on non-echocardiographic imaging using cardiac CT scans and MRI. C. Management of Kawasaki disease and its possible complications This section includes recommendations and good practice statements on management of KD patients, with recommendations on risk stratification, identification of high-risk patients and non-respondents. D. Long term follow-up of patients with Kawasaki disease This section deals with long-term follow-up of patients post convalescence with or without chronic coronary affection. Data are provided also on post-KD activity limitations, vaccinations, and long-term cardiovascular care till successful transition of the child to adult care. ➡️We can summarize the guidelines’ recommendations for KD in the following: • Diagnostic
criteria and KD types (complete, incomplete, atypical). ➡️Guideline Registration PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: (submitted and in process)). Link: http://www.guidelines-registry.org/ |
- Recommendations
|
Table A. Diagnosis of Kawasaki disease |
|||||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A.1: |
When to suspect Kawasaki disease? |
SHARE 2019 CPG
|
The diagnosis of KD should be considered in any child with a febrile exanthematous illness and evidence of inflammation, particularly if it persists longer than 4 days. |
Low |
Weak |
|
A.2: |
What clinical criteria should be used to diagnose Kawasaki disease? |
AHA 2024
|
COMPLETE KD DIAGNOSTIC CRITERIA • Fever for at least 4 d + at least 4/5 principal clinical features at any point during the illness (does not need to be concurrent): • Polymorphous rash • Bulbar conjunctival injection without exudate; bilateral • Oral changes: Erythema and cracking of lips, strawberry tongue, or erythema of oral and pharyngeal mucosa, or all of these o Palmar and plantar erythema : usually accompanied by swelling; resolves with subsequent periungual desquamation in the subacute phase • Cervical adenopathy: usually unilateral, cluster of nodes ≥ 1 .5 cm in diameter • Illness is not explained by known alternative disease process |
Moderate |
Strong |
|
A.3: |
What is the definition of different types of KD (Classic/ Incomplete/ Atypical )? |
SIP 2018 CPG |
Typical or classic KD is diagnosed if a fever is ≥5 days, associated with ≥4 diagnostic criteria, with or without CAA, or if fever lasts at least for 4 days with ≥4 diagnostic criteria and eventual demonstration of CAA on echocardiography. Please refer to A.2
|
Moderate |
Strong |
|
AHA 2024 |
SUSPECTED INCOMPLETE KD DIAGNOSTIC CRITERIA • Prolonged unexplained fever and 2- 3/5 clinical criteria OR infants with unexplained fevers 7 d (day 1 = day of the onset of fever) AND compatible laboratory or echocardiographic findings: o CRP ≥ 3 m g/dL or ESR >40 m m/ h, or both. +3 or more of the following: - Anemia for age - Platelets ≥,450,000 - Album in ≤ 3 g/dL - Elevated ALT - Elevated WBCs ≥15 ,000/mm3 - Urine WBCs ≥10/hpf o Z score of LAD CA or RCA ≥,2.5 Or ≥3 other suggestive features exist, including decreased left ventricular function, mitral regurgitation, pericardial effusion or Z scores in LAD or RCA 2- 2.5 |
Moderate |
Strong |
||
|
SIP 2018 CPG |
Atypical KD is diagnosed if fever, not otherwise explained, lasting for ≥5 days is associated with classic diagnostic criteria and non-classic manifestations, with or without CAA. |
Moderate |
Strong |
||
|
A.4: |
What clinical criteria could increase suspicion of Kawasaki disease in incomplete Cases? |
SHARE 2019 CPG
|
In a patient in whom KD is suspected, but all criteria have not yet been fulfilled, the following clinical signs strengthen the suspicion of KD: Irritability New erythema and/or induration at the site of previous BCG immunization. * * It is now considered a principal criterion in Japanese Society of Cardiology Guidelines. |
Low |
Weak |
|
A.5: |
What are the main differences between KD and MISC-C? |
ARC/VF 2022 CPG |
MIS-C and KD may share overlapping clinical features, including conjunctival infection, oropharyngeal findings (red and/or cracked lips, strawberry tongue), rash, swollen and/or erythematous hands and feet, and cervical lymphadenopathy. |
High |
Strong |
|
Several epidemiologic, clinical, and laboratory features of MIS-C may differ from KD in the following ways: · There is an increased incidence of MIS-C in patients of African, Afro-Caribbean, and Hispanic descent, but a lower incidence in those of East Asian descent. · Patients with MIS-C encompass a broader age range, have more prominent GI and neurologic symptoms, present more frequently in a state of shock, and are more likely to display cardiac dysfunction (ventricular dysfunction and arrhythmias) than children with KD. · At presentation, patients with MIS-C tend to have lower platelet counts, lower absolute lymphocyte counts, and higher CRP levels than patients with KD. · Ventricular dysfunction is more frequently associated with MIS-C whereas KD more frequently manifests with coronary artery aneurysms; however, MIS-C patients without KD features can develop CAA. |
Moderate to high |
Strong |
|||
|
Epidemiologic studies of MIS-C suggest that younger children are more likely to present with KD-like features, while older children are more likely to develop myocarditis and shock |
|||||
|
Table B. Investigations |
|||||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B.1: |
What are the initial laboratory tests for Kawasaki Disease and how can it help in diagnosis? |
SHARE 2019 CPG
|
The following laboratory values should be determined: ESR, CRP, full blood count and liver function (bilirubin, AST/ALT), albumin, serum Na, renal function, and urinalysis. Ferritin and fibrinogen should be considered if there is a concern for macrophage activation syndrome. |
Moderate |
Weak |
|
SHARE 2019 CPG
|
Cerebrospinal fluid analysis may be important to rule out infectious meningitis. |
Low |
Weak |
||
|
B.2: |
Can laboratory data diagnose KD? |
SIP 2018 CPG |
Laboratory data are nonspecific in KD and can only support diagnosis in patients with suggestive clinical features. |
Very low |
Strong |
|
B.3: |
In echocardiography, What protocol an echocardiographer should follow to evaluate cardiac and coronary affection (Timing of imaging, echocardiographic features, coronary imaging)? |
SHARE 2019 CPG |
All patients with suspected KD should undergo echocardiography and ECG at baseline, as soon as the diagnosis is suspected. An intermediate echocardiogram, 2 weeks after the first IVIG, should be performed in all patients with KD whose initial echo was normal and in whom disease activity has been arrested. |
High |
Strong
|
|
SIP 2018 CPG |
Echocardiogram must be performed in all patients with a diagnosis of KD; then after 2, 4, and 8 weeks since KD onset in uncomplicated cases, as CAA can be detected in the subsequent weeks after diagnosis. |
Very low |
Strong |
||
|
SHARE 2019 CPG
|
In those with ongoing active inflammation (increasing or persistently elevated CRP and/or persisting signs and symptoms), ECG and echocardiography should be performed at least weekly to monitor the possible development of cardiac sequelae. In those with coronary abnormalities detected on initial echocardiography, echocardiography should then be performed at least weekly to monitor progression until there is clinical stabilization. |
Moderate |
Strong |
||
|
SIP 2018 CPG |
Persistently febrile non-responders KD patients with CAA, impaired left ventricular function, mild/moderate mitral regurgitation or significant pericardial effusion need a more frequent echocardiogram check-up (at least twice per week). |
Very low |
Strong |
||
|
SHARE 2019 CPG
|
In children with CAA, ECG and echocardiography should be performed 3- to 6-monthly, depending on the severity of the CAA. |
Low |
Weak |
||
|
B.4: |
How can an echocardiographer classify coronary aneurysms? |
JSC\JSCS 2020 CPG |
For coronary artery sequelae, evaluation by Z-score is the standard method, and +2.5 or higher is defined as a long-term significant CAL (sequelae) |
Moderate |
Strong |
|
For those above the age of 5 years, the definition of a giant aneurysm is ≥8mm inside diameter. |
Moderate |
Weak |
|||
|
B.5: |
What are the other non-echocardiographic imaging modalities that should be used to evaluate cardiac affection in KD? |
SIP 2018 CPG |
Cardiovascular CT scan and MR angiography, where available, are important to assess persistent CAA in children with KD and monitor the remodeling of either coronary or systemic arteries in the whole body |
|
Good practice statement |
|
Cardiovascular CT scan, ideally with a DSCT scanner, should be used in patients with KD to: • Confirm CAA (and rule out false positive cases due to coronary artery anomalies or anatomical variants) • Detect middle-distal CAA (not usually seen at routine echocardiograms) • More accurately define the caliber and morphology of CAA and identify coronary artery thrombosis or occlusions • Evaluate other aneurysms, both central and peripheral, in the entire body • Reveal myocardial ischemia or reassess the caliber and morphology of CAAs and better define their treatment. |
Very low |
Weak |
|||
|
SHARE 2019 CPG
|
Cardiovascular MR angiography should be used in patients over 8 years with KD to: - Confirm CAA (and rule out false positive cases due to coronary artery anomalies or anatomical variants). - Identify other aneurysmal dilations, either central or peripheral, in the vascular system. - Assess biventricular global/regional systolic function. - Depict any myocardial scar with contrast enhancement and visualize gross coronary artery anatomy. |
Very low |
Weak |
||
|
Recommendation Table C. Management of Kawasaki disease |
|||||
|
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
|
C.1 |
What is the first line of treatment in the management of KD? |
ARC/VF 2021 CPG |
IVIG is the standard-of-care therapy for the initial treatment of KD. |
High |
High
|
|
SIP 2018 CPG |
IVIG must be administered at dose 2 g/kg of body weight in a single infusion, within the first 7th day of illness, anyway within the 10th day. Administration should be performed over 12 h if the patient’s cardiac function is normal, or in 16–24 h for patients displaying cardiac failure. |
High |
Strong |
||
|
SHARE 2019 CPG
|
All patients diagnosed with KD who are treated with IVIG should be treated with aspirin at a dose of 30-50 mg/kg/day until fever has settled for 48 h, clinical features are improving, and CRP levels are falling. |
Moderate |
Strong |
||
|
SHARE 2019 CPG
|
The dose of aspirin should subsequently be reduced to an antiplatelet dose of 3-5 mg/kg once daily when fever and inflammation have subsided. |
Moderate |
|
||
|
ARC/VF 2021 CPG |
For patients with acute KD, using aspirin is strongly recommended over no aspirin. |
Very low |
Very low |
||
|
C.2 |
When should physicians start treatment? |
SIP 2021 CPG |
IVIG is preferably given within the 10th day, better if within the 7th day of illness, but as soon as possible after diagnosis. |
|
Conditional |
|
SIP 2018 CPG |
IVIG should also be administered to children presenting after the 10th day of illness in case of: – persistent fever – no more fever but aneurysms and ongoing systemic inflammation, as shown by elevation of CRP |
Very low |
Strong |
||
|
SIP 2021 CPG |
IVIG should not be administered if fever spontaneously disappears and no CAA are shown, and if inflammatory markers (ESR and CRP) are within normal limits. |
|
Conditional |
||
|
C.3 |
How long should low dose aspirin be used in patients with acute KD? |
SIP 2018 CPG |
In patients without CAA low-dose ASA is to be discontinued 8 weeks after KD onset. In children who develop CAA low-dose ASA may be continued until the resolution of vascular lesions or indefinitely in case of its persistence. |
Very low |
Strong |
|
C.4 |
How should physicians modify initial treatment in high-risk patients |
SIP 2018 CPG |
High-risk KD patients should receive initial therapy with IVIG + ASA + corticosteroid. |
High |
Strong |
|
C.5 |
What risk factors are suggestive of IVIG resistance? (Laboratory risk assessment of IVIG resistance) |
SHARE 2019 CPG
|
The following laboratory values can be important in assessing risk stratification for IVIG resistance: · Low sodium · Raised bilirubin · Raised Alanine Transferase. · Low platelet count · High CRP · Low albumin. |
Moderate |
Weak |
|
JSC\JSCS 2020 CPG |
Resistant KD is defined by failure in the response to IVIG and is revealed by recrudescent fever reoccurring or persisting 36–48 h after IVIG infusion. The following features are elements of the risk scores for predicting IVIG resistance. (1) Leukocytosis with left shift (2) Thrombocytopenia (3) Hypoalbuminemia (4) Hyponatremia (5) Hyperbilirubinemia (jaundice) (6) Elevation of CRP. (7) Age <1 year |
High |
Strong |
||
|
C.6 |
How should physicians modify treatment in the presence of IVIG resistance? |
SIP 2021 CPG
|
In non-responders, treatment requires additional anti-inflammatory therapy including: second infusion of IVIG, Corticosteroids, anti-TNF alpha, or interleuin-1 antagonist |
|
Conditional |
|
C.7 |
When should corticosteroids be used in pediatric patients with Kawasaki? And what are the proposed regimens? |
SHARE 2019 CPG
|
Corticosteroid treatment should be given to patients with severe KD: (a) Who are IVIG resistant, that is, with ongoing fever and/or persistent inflammation or clinical signs ≥48 h after receiving IVIG as a single dose of 2 g/kg. A second dose of IVIG is at the discretion of the treating physician. (b) Kobayashi score ≥5 (c) With features of MAS. (d) With features of shock (e) Who are under the age of 1 year (f) Who present with coronary and/or peripheral aneurysms |
High
High
Moderate
Low Low Low
|
Strong
Strong
Weak
Weak Weak Weak |
|
SHARE 2019 CPG
|
Proposed regimen: If corticosteroids are indicated, the following regimens would be reasonable: Regimen 1: prednisolone 2mg/kg/D for 5-7 days or until CRP normalizes; wean off over next 2-3 weeks. Regimen 2: methylprednisolone 10-30 mg/kg (up to maximum of 1g/day) once daily for 3 days followed by oral prednisone/prednisolone 2 mg/kg per day until day 7 or until CRP normalizes; then wean over next 2-3 weeks. |
Moderate
|
Strong |
||
|
SHARE 2019 CPG
|
TNF-alpha blockade (e.g. infliximab) should be considered in KD patients with persistent inflammation despite IVIG, aspirin and corticosteroid treatment, after consultation with a specialist unit |
Moderate |
Strong |
||
|
SHARE 2019 CPG
|
The use of Disease Modifying Antirheumatic Drugs (DMARDs) such as ciclosporin, cyclophosphamide and methotrexate, along with anakinra and plasma exchange, cannot be recommended, except on an individual basis after consultation with a specialist unit. |
Moderate |
Weak |
||
|
C.7 |
How can physicians modify treatment in KD patients complicated by Macrophage Activation Syndrome? |
ARC/VF 2021 CPG |
For patients with acute KD and suspected or diagnosed MAS, treatment with IVIG for KD and additional agents to treat MAS is strongly recommended. |
Very low |
|
|
For children with unexplained MAS, obtaining an echocardiogram with coronary artery measurements is strongly recommended. |
|
Strong |
|||
|
Recommendations table D: Management of coronary involvement in children with KD |
|||||
|
D.1 |
How can physicians treat medium sized or multiple aneurysms in KD? |
SIP 2021 CPG
|
KD patients with medium-sized coronary artery aneurysms or those with multiple and complex aneurysms require dual anti-platelet prophylaxis, based on low-dose ASA (at a single dose of 3–5 mg/kg/day) and clopidogrel (at a single dose of 0.2 mg/kg/day in children aged < 24 months and up to 1 mg/kg/day in children aged ≥ 24 months). |
Moderate
|
Strong
|
|
D2 |
How can physicians treat complex or severe CAA (Z score > 10, diameter >8) aneurysms in KD? |
SIP 2021 CPG
|
• It is reasonable to treat KD patients having complex or severe CAA (Z score > 10, diameter >8) with low-dose ASA associated with warfarin (keeping INR targeted at 2.0–3.0) or LMWH (if regular INR checking is difficult). Triple therapy with ASA, warfarin or LMWH and clopidogrel should be considered in KD patients with a relevant risk of thrombosis. • Warfarin is used in combination with low-dose aspirin for patients with large CAA, history of MI, and thrombosis in the CAA. The dose is adjusted for the international normalized ratio of prothrombin time (PT-INR) target range of 2.0–2.5 |
Moderate |
Strong
|
|
D.3 |
How can physicians treat patients with thrombosed coronary aneurysms? |
SIP 2021 CPG
|
Recombinant tissue plasminogen activator (rtPA) is the first-choice thrombolytic drug in children with KD complicated by coronary artery thrombosis; the glycoprotein IIb/IIIa inhibitor abciximab may be used in case of thrombosis with high risk of occlusion. Both therapies require a concomitant association with low-dose ASA and intravenous heparin. |
Moderate |
Strong |
|
Recommendations table (E) Long term management of KD patients |
|||||
|
E1 |
How can physicians manage vaccination in children younger than 5 years of age with KD? |
SIP 2021 CPG
|
All inactivated vaccines can be safely administered at any time after IVIG in KD patients. Attenuated live virus vaccines (MMR, V, and MMRV vaccines) should be administered 10–12 months after the administration of IVIG to avoid a reduced specific immune response in KD patients. influenza vaccination is recommended in KD patients receiving ASA.
|
Moderate |
strong |
|
E2 |
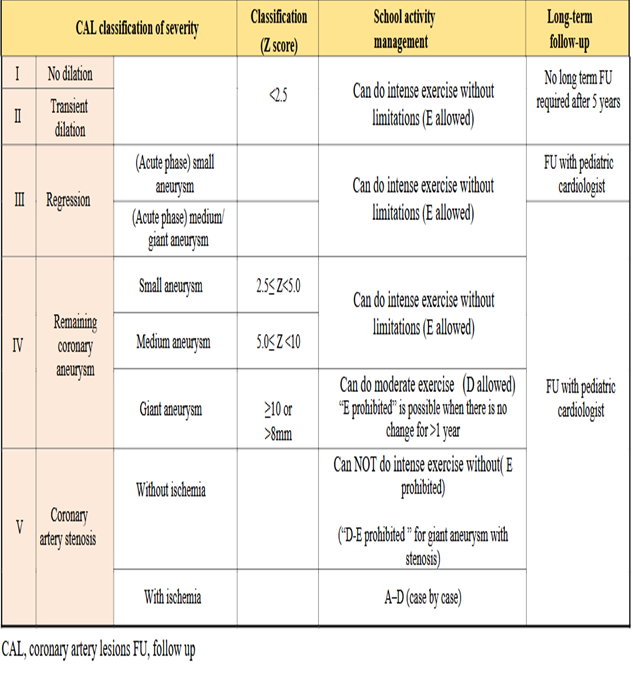
How can school activities be modified in patients post convalescence? |
JSC\JSCS 2020 CPG |
Level of management: A - Requires treatment at home or in hospital, B - Goes to school but must avoid exercise, C - Can do mild exercise, D - Can do moderate exercise, E - Can do intense exercise. Exercise intensity: Mild exercise: Physical activities that do not increase respiratory rate in average children at the same age, Intermediate exercise: Physical activities that increase respiratory rate without causing shortness of breath. Players may talk with others during exercise, and Intense exercise: Physical activities that increase respiratory rate and cause shortness of breath. Express the allowed exercise intensity from “A” to “E”. Only “E” will be noted as “Allowed” or “Prohibited” for school sport club activities and will be referred to as “E-allowed” or “E-prohibited”. |
|
Conditional |
|
E3
|
What is the long-term management of KD patients post convalescence? |
JSC\JSCS 2020 CPG |
Low-Dose Aspirin is orally administered to patients with persistent CAA |
High |
Weak |
|
JSC\JSCS 2020 CPG |
Statins should be used to prevent cardiovascular events in patients with CAL. |
Moderate |
Weak |
||
|
JSC\JSCS 2020 CPG |
ACEI or ARB may be used to prevent coronary artery stenosis in patients with CAL. |
Moderate |
Strong |
||
|
JSC\JSCS 2020 CPG |
Beta-blockers, Calcium antagonists, or nitrates can be used to prevent Acute coronary syndrome (ACS) in patients with CAL |
Moderate |
Strong |
||
|
E4 |
How can patients be safely transferred to adult care? |
JSC\JSCS 2020 CPG |
Preventing the loss of follow-up (the so-called dropouts) is the most important issue in the management of the adolescent and young adult (AYA) generation. |
Moderate |
Low |
- Acknowledgments
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Alyaa Amal Kotby |
Professor of Pediatrics and Pediatric Cardiology Ain Shams University |
Conceptualization, engaging stakeholders and development of the outline and scope of Guidelines. Participating in multiple steps of the CPG adaptation process, Participated in Guidelines selection and appraisal as well as data collection and revision of evidence. Supervising and writing different sections, overseeing the overall structure, style and format.
|
|||
|
Prof. Hala Mounir Agha |
Professor of Pediatrics and Pediatric Cardiology Cairo University |
Participating in multiple steps of the guideline adaptation process, and CPG appraisal |
|||
|
Prof Hanan Ibrahim |
Professor of pediatric Head of pediatric department Ain Shams University |
Revision of the guidelines ensuring its alignment with best practices. Ensured the involvement and collaboration of all relevant stakeholders, both internal and external with the allocation of proper resources for guidelines implementation. |
|||
|
Prof Duaa Raafat |
Professor of Pediatrics and Pediatric Cardiology Assiut University |
Participating in multiple steps of the guideline adaptation process and in CPG selection and appraisal. Shared in writing the background, implementation tools and Arabic instructions to nurses and parents. |
|||
|
Prof Rasha Hassan El-Owaidy |
Assistant Professor of Pediatrics and Pediatric Immunology Ain Shams University |
Participating in multiple steps of the CPG adaptation process, Participated in data collection and revision of evidence as well as in editing and revision of the document |
|||
|
Assistant Prof. Nanies Soliman |
Assistant Professor of Pediatrics and Pediatric Cardiology Ain Shams University |
Participated in multiple steps of the CPG adaptation process, Participated in data collection and revision of evidence as well as in editing and revision of the document. Helped in drafting and revision of the guidelines ensuring consistency in language, tone, and style throughout the document. |
|||
|
Dr. Mahmoud Mohammad Nadder |
Lecturer of Pediatrics , Alexandria University |
Participating in multiple steps of the CPG adaptation process, Participated in data collection and revision of evidence as well as in editing and revision of the document. Shared also in implantation tools creation. |
|||
|
Dr. Mona Saber Mohamed Kamel |
Lecturer of Pediatrics MTI University |
Participating in multiple steps of the guideline adaptation process, Participated in data collection and revision of evidence as well as in editing and revision of the document |
|||
|
Dr. Mohamed Hassan Kamel Garamoon |
Senior Clinical Pharmacist, Children Hospital, Ain Shams University |
Participating in revision of data related to pharmacological agents adopted in the recommendations. |
|||
|
Mrs. Wafaa Fathallah |
Nursing director Children’s Hospital, Ain Shams University |
Provided insights on direct patient care experience ensuring that guidelines are realistic and applicable. Also Acted as a facilitator in the implementation the guidelines within the clinical setting. |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia. 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia. 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Associate Professor of Pediatrics Ain Shams University, Egypt |
Participating in multiple steps of the guideline adaptation process, Participating in Guideline search and retrieval as well as guideline appraisal. |
||||
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process, and revised the whole document. |
|||
|
Reviewers for Clinical Content |
|||||
|
|||||
|
Reviewers |
|||||
|
Professor Nagib Dahdah |
Consultant, Pediatric Cardiology, CHU Ste-Justine. Professor of Pediatrics, Universite de Montreal, Quebec, Canada. |
||||
|
Professor Sanaa A Mahmoud, |
Clinical Professor of Pediatrics, Allergy& Immunology Pikeville university, Pikeville, KY Adjunct Faculty, Lincoln Debusk Memorial University, Harrogate, TN |
||||
|
Professor Mohamed Magdy AbouElkheir |
Professor of Pediatrics &Pediatric Cardiology, Mansoura University Head of Pediatric Department, Horus University, Egypt |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil Member, Board of Trustees, Guidelines International Network (G-I-N)
|
||||
▪️The GDG/ GAG acknowledges EPG for its help in completing this project.
▪️ We acknowledge The European Single Hub and Access point for pediatric Rheumatology in Europe, the Italian Society of Pediatrics, The Japanese Circulation Society\ the Japanese Society of Cardiovascular Surgery, and the American College of Rheumatology/Vasculitis Foundation guidelines (the source original guidelines) for their cooperation in providing the permission for adapting our guidelines.
▪️ We thank Mrs. Safa Rashad for drawing the Kawasaki boy caricature.
▪️ Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
▪️This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions
- Abbreviations
|
- Introduction
➡️Background:
The Egyptian experience in the diagnosis and management of Kawasaki disease (KD) has evolved and intensified over the past years, especially with the emergence of atypical, incomplete cases and with the appearance of Multisystem Inflammatory Syndrome in Children (MISC). Meanwhile, a survey conducted by Kawasaki Disease Arab Initiative (Kawarabi) in 13 Arab countries, showed that the quality of medical services received by children with KD in large cities was rated as excellent in 6/13 or good in 7/13 countries compared to fair in 4/13 or poor in 4/13 countries in rural areas and they concluded that KD patients in mid-size cities and rural areas have limited access to standard healthcare in the Arab world (9). Accordingly, the establishment of Egyptian evidence-based guidelines for the management of KD in children is a necessity with consideration of the available facilities and the local limitations.
➡️What is Kawasaki Disease?
KD is an acute, self-limited febrile illness that predominantly affects young children, especially those under 5 years of age, with a median age of onset of 9 to 11 months. Approximately 25% of cases occur in older children, and it rarely affects adults (10). It is more common in males than females by a ratio of 1.5 to 1 (10). Age and gender play an important role as risk factors for complications, as age less than one year or above 9 years, and male gender have universally been identified to be significant risk factors for developing coronary artery aneurysms (11).
➡️Seasonal peaks: -
Evidence of seasonal variations has been demonstrated with a peak incidence in January through March in the Northern hemisphere, compared to a peak in May through June in the Southern hemisphere (12). This seasonality is suggestive of an environmental agent that may play a role in disease causation in different regions and ethnicities. Because the incidence of Kawasaki disease peaks during winter and spring, an infectious agent as a primary trigger may be suggested (13).
➡️Etiology and pathogenesis:
Despite more than 50 years of study, the exact etiology of Kawasaki disease remains unknown. Immunologic response to an exposure in the respiratory system or gastrointestinal (GI) tract or both in a genetically susceptible child is the most accepted theory. The immunologic cascade leads to systemic inflammation in medium-sized arteries and multiple organs in the acute phase (14).
➡️Diagnostic criteria: -
Recommendations for the diagnosis of KD are shown in table A
KD is categorized into two types: Classic KD(CKD) and Incomplete KD (IKD) (11). Typical KD or CKD have clear diagnostic criteria which are shown in figure (1) a and (1) b. Not all features of KD appear at the same time and watchful waiting might be needed to reach a diagnosis.
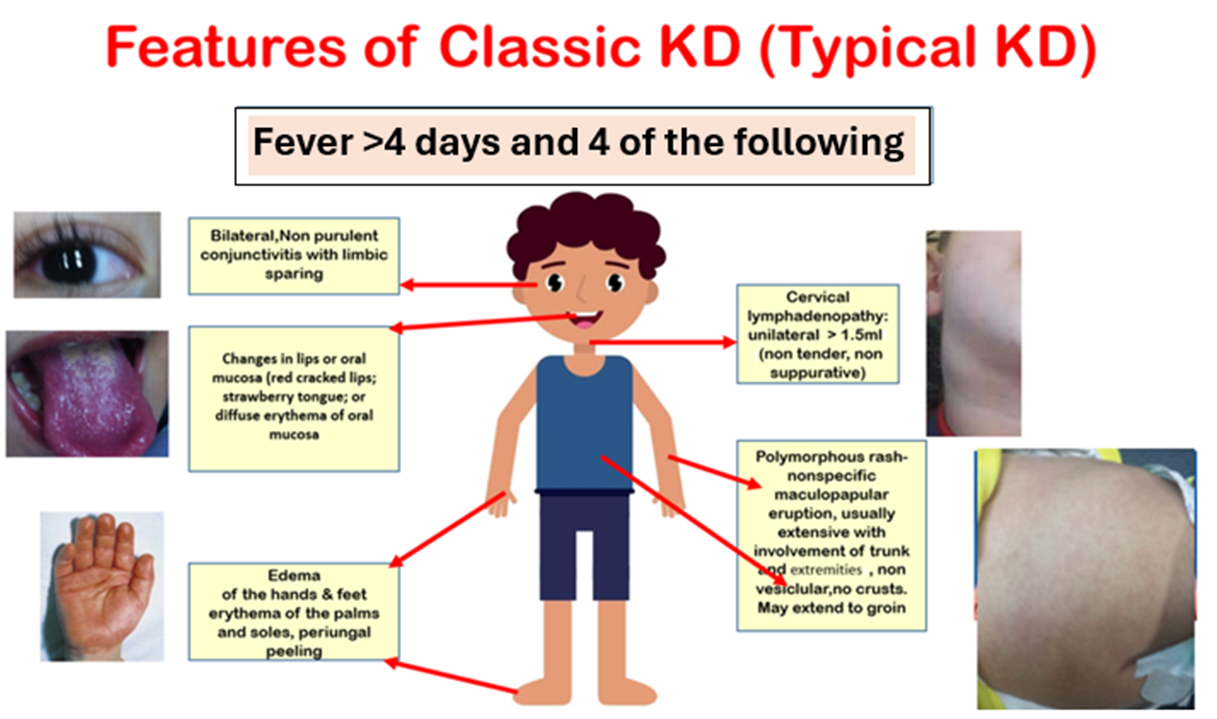
Figure (1a): Features of classic Kawasaki disease (KD).
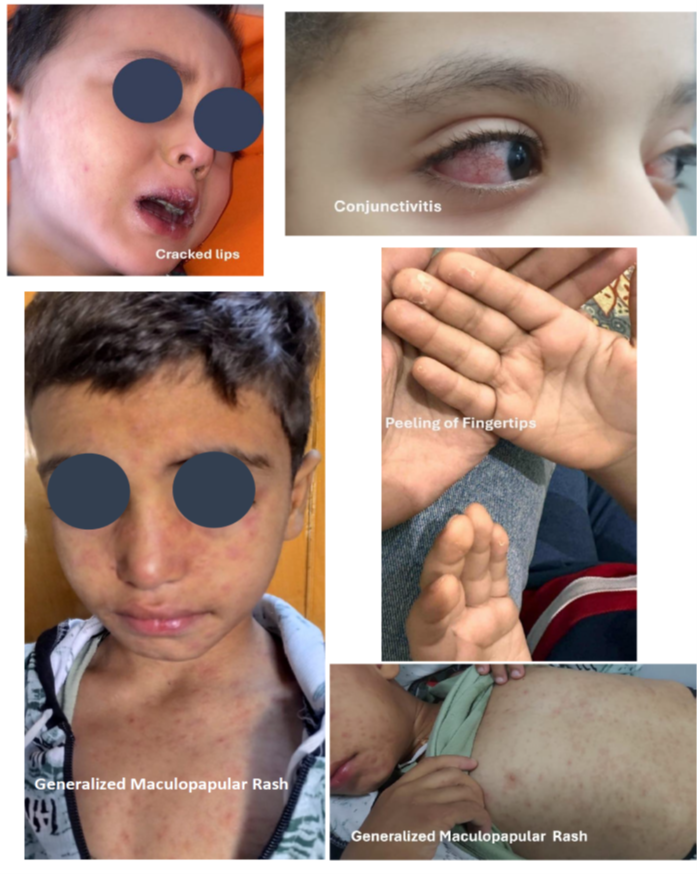
Figure (1b): Features of classic Kawasaki disease (KD).
Additional symptoms can be evident in KD patients but are not encompassed by the principal criteria (fig 1c and fig 1 d), they include arthritis, gastrointestinal involvement, irritability, lethargy, neurological manifestations, cough, and rhinorrhea. Arthritis, primarily affecting the large joints of the lower extremities (such as the knees, hips, and ankles), can be identified in 7.5% to 25% of patients and is typically temporary and non-deforming (15,16). In exceptional instances, abdominal imaging techniques such as radiographs or computed tomography (CT) scans might reveal indications of pseudo-obstruction, a condition that can sometimes manifest before the appearance of cardinal symptoms (17).
In incomplete KD (IKD), children have some symptoms appearing late or not at all, making it somewhat invisible and overlapping with the clinical symptoms of various pediatric infectious or connective tissue diseases. This increases the likelihood of misdiagnosis and underdiagnosis, leading to residual cardiac complications, and even death (18).
The estimated ratio of male to female patients in children with IKD is 1.5: 1.
Among the mucocutaneous manifestations, nail abnormalities can be observed. Orange-brown transverse chromonychia has also been occasionally described. During the convalescent phase, transverse leukonychia and Beau’s lines are the most common (20).
On the other hand, Atypical KD occurs in patients who, along with the usual clinical features of KD, also have a few unusual clinical manifestations, such as pulmonary involvement and renal impairment (21).
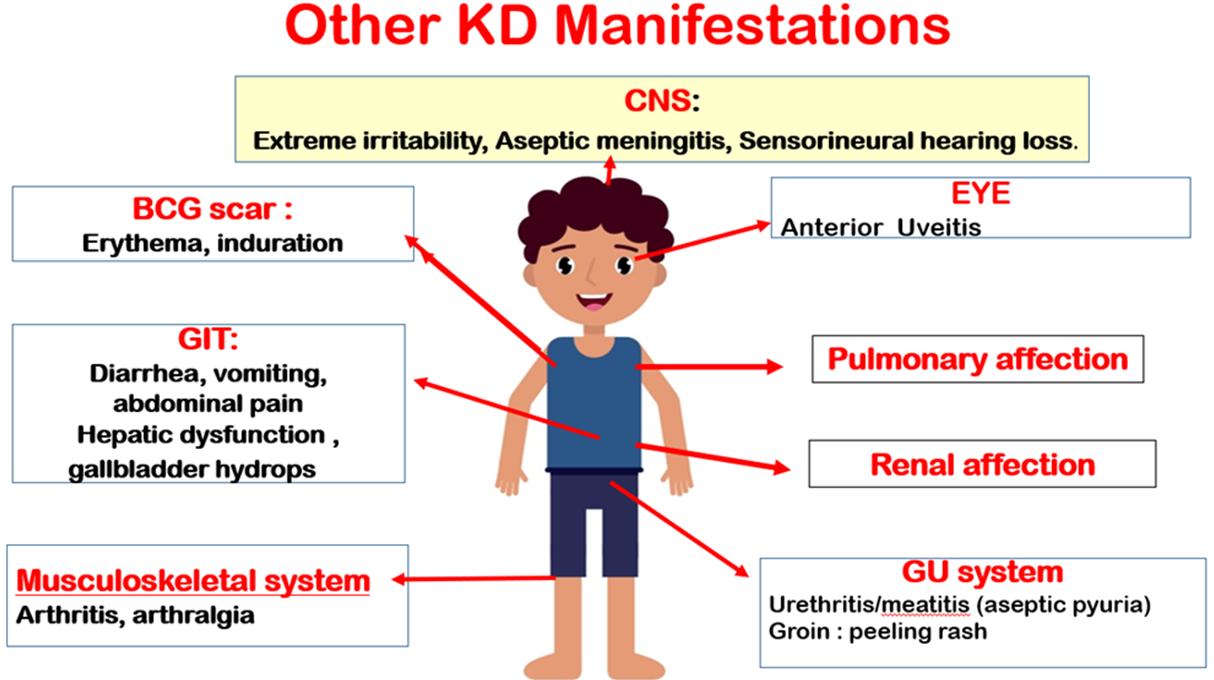
Figure (1)c: Additional clinical features of KD.
CNS: Central Nervous System: Bacillus Calmette Guerin; GIT: Gastrointestinal tract; GU: Genitourinary
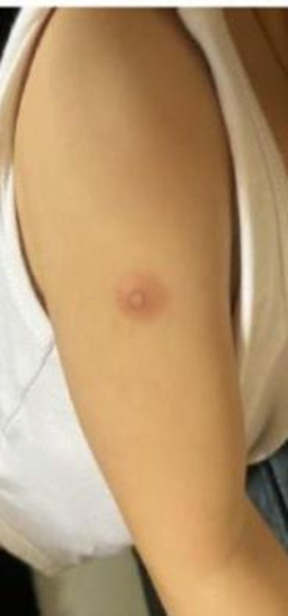
Figure (1)d: Induration of BCG scar in atypical KD patient.
Recommendations for diagnosis of classic, incomplete and atypical Kawasaki disease are shown in Recommendation table A.
➡️Disease Course:
Kawasaki Disease natural clinical course can be divided into three phases:
Acute (1st-2nd Week), Extreme irritability exists, by the end of which CAA can occur
Subacute (3rd-4th week), characterized by defervescence, periungual skin desquamation, thrombocytosis and progress of or appearance of new CAA,
Convalescence phase (5th–8th week), in which all KD signs disappear, inflammatory markers normalize, and transversal nail indentations (Beau’s lines) may appear (22).
➡️Cardiovascular Involvement in Kawasaki Disease:
Coronary artery involvement is the most serious complication in children with KD. Coronary artery abnormalities occur in 25% of untreated patients and 5% of patients treated with intravenous immunoglobulin (IVIG) (7). A study from Egypt reviewed a series of 580 patients ≤ 40 years of age presenting with symptoms of coronary artery ischemia and reported lesions consistent with antecedent KD in 6.7 % of cases (24). Data on exact prevalence of CAA in Egyptian children is not yet determined.
The proximal left anterior descending artery and the proximal right CA are the most frequent locations of CAA, and the posterior descending artery is the least common. (25)
Table 2 Highlights Cardiac affection in KD according to the American Heart ➡️Association (AHA) Guidelines 2017 (7):
|
Positive Echocardiographic findings suggestive of cardiac involvement in acute KD Include: 1. Left anterior descending coronary artery or proximal right coronary artery with a Z-score ≥ 2.5 2. Coronary artery aneurysm formation 3. ≥3 of the following suggestive features may be used to support KD diagnosis if classical clinical features are incomplete: Mitral regurgitation. Pericardial effusion. Decreased left ventricular function. Z-scores in the left anterior descending coronary artery or right coronary artery of 2 to 2.5.
|
Several non-coronary complications have now been identified in this condition, but these are often overlooked. Myocarditis is an integral component of KD and may be more common than coronary artery abnormalities. Myocarditis is universal in almost all patients with KD during the acute phase of disease. Transient left ventricular dysfunction can occur in more than 50% of patients. Pericardial involvement and valvular abnormalities have also been observed in patients with KD (25). Cardiac affection in KD is shown in figure (2).
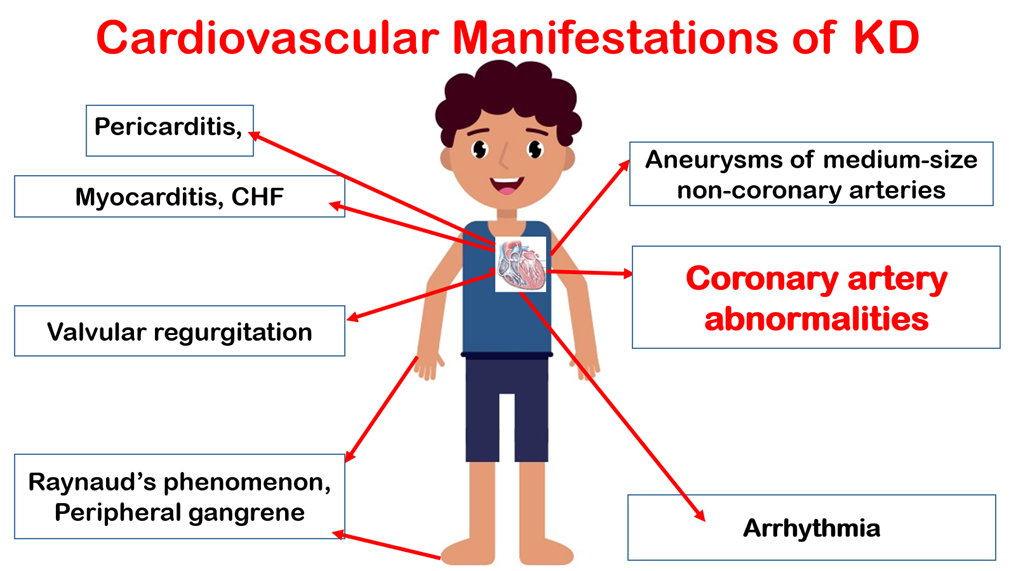
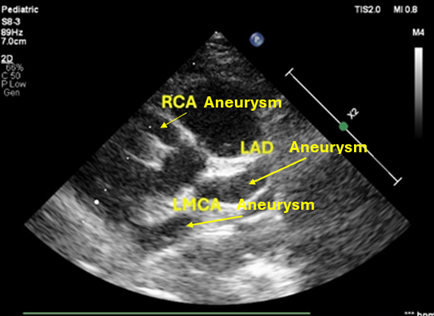
Figure (2): Cardiovascular affection in KD.
CHF: Congestive heart failure, RCA: Right coronary artery, LAD: Left anterior descending, LMCA: Left main coronary artery.
On the other hand, KD shock syndrome (KDSS) is now being recognized and may be difficult to differentiate clinically from toxic shock syndrome (26). In KDSS, KD manifestations are accompanied by multiorgan involvement and reduced organ perfusion due to systolic hypotension. KDSS is a serious condition that can present to the emergency department as an initial feature when typical clinical symptoms of KD have not been detected (27). Patients may have more prominent inflammatory markers and result in shock and hypotension, which requires critical care support at an early stage. Features that enable distinguishing KDSS from the usual KD case presentation include: (1) patients with KDSS are older than the usual patient with KD and have a higher prevalence and severity of GI symptoms; (2) patients with KDSS present with worse biological and inflammatory markers, (3) exhibit a higher resistance rate to IVIG, (4) have a higher rate of CAA, and (5) have greater number of reported cases of ventricular systolic dysfunction and atrioventricular valve regurgitation.
This is different from Toxic Shock syndrome, which should be diagnosed according to the CDC criteria (29)
Kawasaki disease and Multisystem inflammatory syndrome in children: -
Recommendations for differentiation of KD and MISC are shown in Recommendation table A.
MIS-C and KD are considered 2 distinctive diseases triggered by different infectious agents. They may belong to the same umbrella of inflammatory disorders but differ in many aspects of etiology, demography, epidemiology, clinical and laboratory findings, and pathology. Both diseases share many clinical features such as fever, rash, and mucocutaneous involvement, and can affect multiple organ systems. The intensity of the inflammatory response and long-term cardiovascular sequelae diverge between KD and MIS-C. Whereas MIS-C presents a more intense inflammatory syndrome, myocardial dysfunction, and cardiogenic shock. KD vasculitis is associated with pathologic changes in the coronary arteries and long-term cardiovascular sequelae (30,31).
Differential Diagnosis of KD is displayed in Annex Table 4(32-37).
➡️Investigations:
A. Laboratory Investigations
Recommendations on investigations required in KD are provided in Recommendation table B.
Investigations in suspected KD patients are shown in Table2
Table 2: Investigations in suspected KD patients (2)
|
Blood cell count |
|
|
White blood cells |
Increased, especially polymorphonuclear cells. Rare to be decreased. |
|
Red blood cells |
Reduced with normal mean corpuscular volume |
|
Platelets |
Increased till the 2nd or 3rd week, normalized in 4-8 weeks. If decreased suspect Disseminated intravascular coagulopathy |
|
Inflammatory markers |
|
|
Erythrocyte sedimentation rate |
High with slow normalization |
|
C reactive protein* |
High with fast normalization |
|
Liver Function tests |
|
|
Transaminases |
High |
|
Bilirubin |
High |
|
Gamma Glutamyl transferase |
High |
|
Albumin |
Decreased in prolonged severe illness |
|
Other laboratory tests |
|
|
Urine |
>10 white blood cells by High power field |
|
Cerebrospinal fluid |
Aseptic meningitis (mononuclear cells with normal glucose/protein ratio) |
|
Synovial fluid |
Purulent fluid, normal glucose, white blood cells 125000-300000/mm3 |
*Diagnostic positive level used in Egyptian laboratories is > 5mg/L
• CRP: C -reactive protein, ESR: Erythrocyte sedimentation rate, WBCs: White blood cells, HPF: High power field.
N-terminal prohormones of brain natriuretic peptide (NT-proBNP) The utility of NT-proBNP as a biological marker in KD is based on the universal myocardial inflammatory component early in the course of the disease. Patients with KD have higher NT-proBNP at the time of diagnosis than other febrile patients, with a pooled sensitivity of 89%, and a specificity of 72%. Moreover, patients with resistance to intravenous immunoglobulin treatment and CAA were found to have higher levels of NT-proBNP, suggesting a prognostic role. Nevertheless, the non-specificity of NT-proBNP to KD limits its use as a stand-alone test. It is worth mentioning that NT-proBNP proved superior to BNP in the evaluation of KD at the onset of the disease, thanks to the longer half-life of the former providing greater sensitivity and specificity (23)
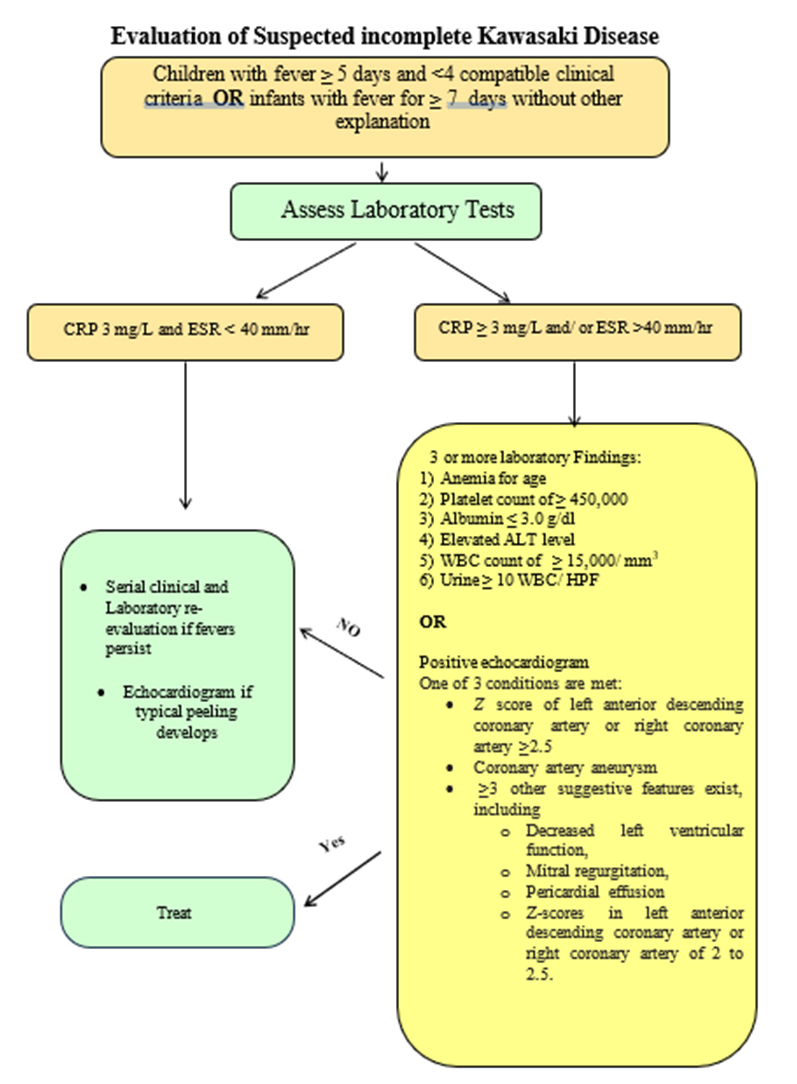
Evaluation of Suspected incomplete KD shown in the following figure
Fig 3: Evaluation of Suspected incomplete Kawasaki Disease (1,36)
B. Echocardiography :
It should be performed in all patients suspected or confirmed to have KD. Recommendations on the protocol of echocardiographic examination in KD are shown in Recommendation table B.
Routine use of CA Z scores has brought a level of standardization to quantification of CA size. It is important for centers to use the same Z score equation for comparisons over time in patients with KD.
Recommended frequency of echocardiographic examination in KD patients is shown in Fig 4.
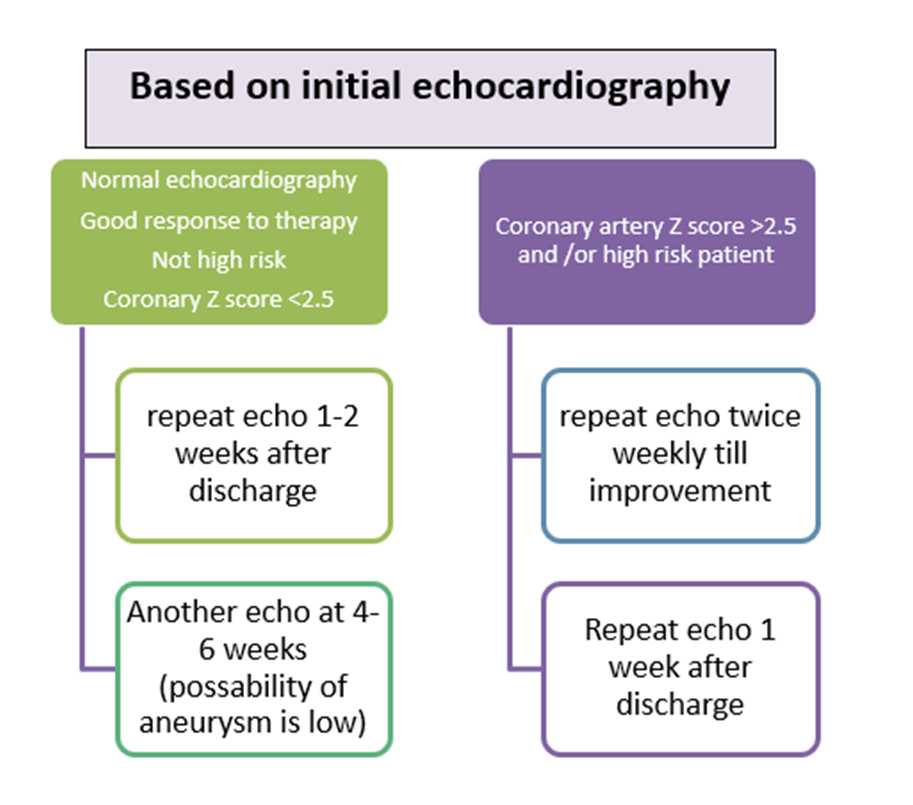
Fig (4): Recommended frequency of echocardiographic examination in KD patients.
Coronary artery measurements should be taken from the inner edge to inner edge of the vessel wall, avoiding the orifices and points of branching which may have normal focal dilatation (23)
The presence of marked perivascular brightness, the absence of the physiological gradual reduction of coronary artery caliber, and mild ectasia, if isolated, are not indicative of KD. These findings could be considered a positive echocardiographic KD sign when are all three simultaneously present (1).
Follow up of KD and CAA: TTE is the primary imaging tool for follow up CAA. Owing to its limited acoustic windows in distal CA, TEE and CT angiography are recommended. Advanced CA imaging using MRI or CTA may be needed for better designations in patients with KD. CTA will delignate aneurysm and stenosis. MRI for Myocardial function analysis and inducible ischemia. Stress echo can be used to follow pts with CCA and ischemia symptoms (36).
➡️KD Treatment: -
At presentation, the low-risk KD should be differentiated from high-risk cases (baseline CAA Z score ≥2.5, infants < 12 months or in high-risk category using the Son risk score, in addition to patients presenting with KD shock syndrome) (8) (Fig 5). Patients with standard risk can be treated with IVIg and aspirin. The general approach for treating low risk KD cases is administering a single high dose (2 g/kg) of IVIG intravenously over 10–24 hours within 10 days of disease onset. This should be accompanied by the oral administration of aspirin (30–50 mg/kg), until the patient is afebrile for 48 to 72 hours. In a classic KD, treatment should be initiated without waiting for echocardiography. (23,5) This can quickly and effectively relieve symptoms and decrease the incidence of CALs and CAAs (37).
To reduce the risk of hemolytic anemia in obese patients, IVIG dosing should be based on lean body mass. (38)
Patients with high-risk KD may benefit from intensification of initial therapy with IVIg plus adjunctive anti-inflammatory therapy to reduce the risk of CAA. These therapies include corticosteroids, tumor necrosis factor α inhibitors (eg, Infliximab & Etanercept), interleukin-1 inhibitors (eg, Anakinra), and cyclosporine (8). Proposed treatment regimen for KD is shown in figure 5.
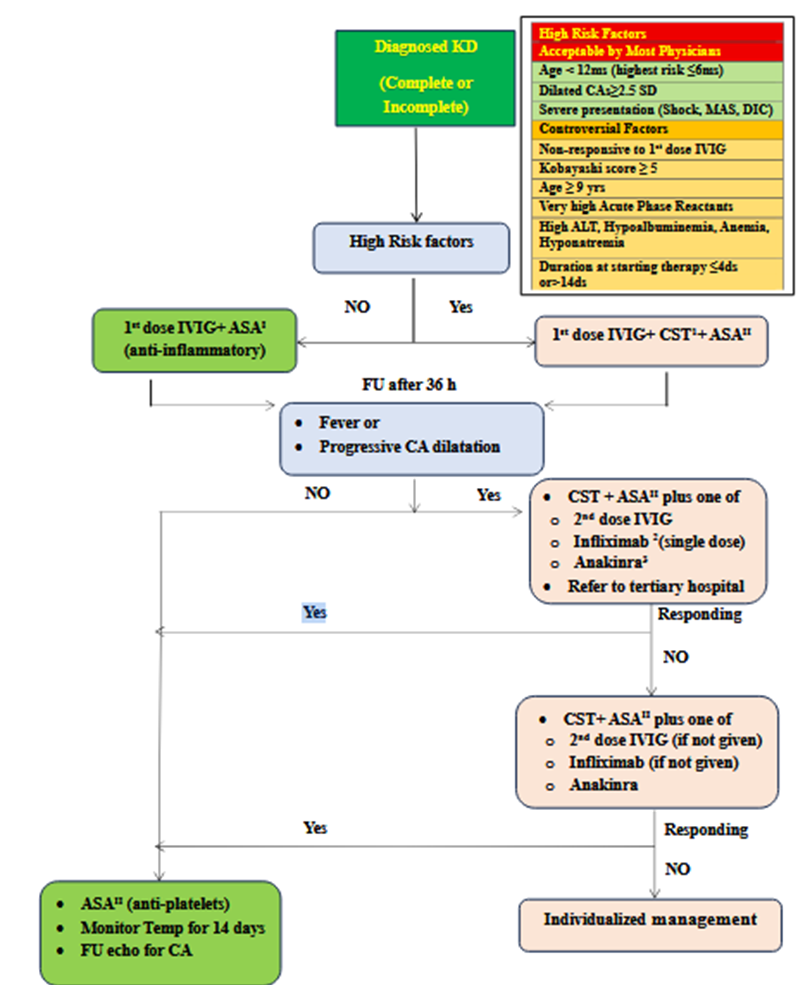
KD: Kawaski Disease, MAS: Macrophage activating Syndrome, DIC: Disseminated Intravascular Coagulation SD: Standard Deviation, ALT: Alanine Transaminase, IVIG: Intravenous Immunoglobulins, ASA: Acetyl Salicylic Acid, CST: Corticosteroid Therapy, CA: Coronary Artery. 1Most practitioners start with standard dose of steroids (2 mg / kg / day IV methylprednisolone) but some practitioners prefer high dose steroids (10-30g / kg / day methylprednisolone for 3-5 days then decrease to standard IV dose ) especially in low income countries in which IVIG treatment could be delayed to 12-24 hours after diagnosis of KD ( or more ) due to shortage of that expensive drug.2 Infliximab is given IV , 10 mg/kg over 2 hours single dose .3 Anakinra ,10 mg/kg/day ,is given IV/SC (preferred IV divided /12 hours).
Figure 5: Proposed algorithm for management of KD(38,39,40).
For corticosteroids therapy as an intensification therapy, the RAISE study suggested the use of IV prednisolone, 2 mg/kg per day divided every 8 h for 5 d (maximum 60 mg/d) while hospitalized; then PO prednisolone 2 mg/kg per d divided every 8 h; slow tapering over 15 d (maximum 30 mg/dose) once CRP normalized. On the other hand, North American studies suggested using Methylprednisolone with the same doses given every 12 hours and to be tapered over 2–4 weeks with the dose cut in half every 5 days. (42,43) Another protocol for non-responders with KD should be managed with a second IVIG cycle and - in case of failure - with 3 pulses of methylprednisolone (30 mg/kg/day), followed by oral prednisone (2 mg/kg/day, then gradually tapered up to the resolution of symptoms and normalization of CRP)(1)
If the child has been ill for more than 14 days and inflammatory symptoms and laboratory changes are no longer present, only low-dose anti- aggregation dose of aspirin (3–5 mg/kg/d) should be administered to prevent thrombosis. Echocardiography should be performed in these patients at the time of presentation and at regular intervals within the next 2 months to follow up on coronary arteries to decide on the duration of aspirin therapy (44). There is increasing evidence that medium- or high-dose aspirin in the acute phase is likely not associated with improved CA outcomes (45,46).
Patients with no coronary dilation should be maintained on LDA (low dose aspirin) for 6-8 weeks with no further interventions. For patients with Z score 5 - <10 and absolute dimension < 8 mm they should be maintained on double antiplatelet ill regression of the aneurysm (23)
Medium-high dose ASA should be replaced in case of concurrent varicella or influenza to avoid the potential development of Reye’s syndrome though low-dose aspirin has not been associated with any documented cases of Reye syndrome and therefore is safe to continue in the event of intercurrent infection. Children may receive dipyridamole (1–5 mg/kg/day divided into 3 doses), ticlopidine (2–7 mg/kg/day divided into 2 doses), or clopidogrel (1 mg/kg/day in a single dose up to a maximum of 75 mg/day in children > 2 years or 0.2 mg/kg of body weight in children < 24 months) in replacement of ASA (47). It is recommended to avoid non-steroidal anti-inflammatory drugs such as ibuprofen in children receiving low-dose aspirin as they can antagonize platelet inhibition sought for with by aspirin (48,49).
|
|
Management recommendations for children with KD are shown in Recommendation table C.
Commonly used medications in treatment are shown in table (3).
|
Table (3): Commonly Used Medications in initial treatment of standard risk KD patients |
|||
|
Drug |
Dose |
Adverse reactions/precautions |
Pharmaceutical form |
|
IVIG |
Single dose ( infusion): 2 g/kg (IV) |
· Possible anaphylactic reaction, especially in children with IgA deficiency (preparation with the least amount of IgA should be selected) · Hemolytic anemia · Aseptic meningitis · Fever, chills, headache, myalgia, nausea, vomiting. Associated with a high infusion rate at beginning of treatment, in 5---15%: During or up to 1---2 days after infusion. · Infusion rate: a test dose is usually done in the first 30-60 min of infusion then the dose is completed. Total Duration 12 hours in patients with Normal Cardiac Function. In Patients with impaired cardiac function usually Total duration is 18-24 hours. |
Vials: 1 g, 2.5 g, 5 g, 10 g
|
|
ASA |
Anti-inflammatory: 30-50 mg/kg /day divided in 3-4 doses (Oral) |
· Gastrointestinal changes and bleeding · Hypoprothrombinemia. · Rhinitis. · Paroxysmal bronchospasm. · Hypersensitivity. · Mild chronic salicylate intoxication, which is characterized by tinnitus and hearing loss. Discontinue treatment if these symptoms appear. Given after meal or with large glass of milk |
Tablets: 75 . 81 . 100, 300, 320 mg Maximum Dose: 4000 mg |
|
|
Anti-platelets: 3-5 mg/kg/day in 1 dose (Oral) |
Tablets: 75. 81. 100 mg Maximum Dose : 150 mg |
|
|
Corticosteroids |
Methylprednisolone: (used in Egypt) · 10-30 mg /kg/day IV for 3-5 days, followed by methylprednisolone, prednisolone or prednisone 2 mg/kg/day IV or Oral with gradual taper based on patient evolution · 2 mg/kg/day IV until fever resolves and CRP levels decrease, with gradual taper based on patient evolution Prednisolone: · 2 mg/kg/day IV until fever resolves and CRP levels decrease, with gradual taper based on patient evolution (not present in Egypt)
|
· High blood pressure. · Fluid retention. · Peptic ulcer. · Infection. · Hirsutism. · Hypokalemia. · Alkalosis. · Weaknesses. · Myopathy with muscle atrophy. · Acne. · Cataract. · Raised intracranial pressure. · Osteoporosis. · Cushing syndrome. · Adrenal suppression. · Glucose intolerance. · Amenorrhoea. · Delayed growth |
Methylprednisolone: Vials: 500, 1000 mg. Maximum Dose: 1000 mg
Prednisolone: Suspension, 1mg/mL, 5 mg/mL Tablets: 5, 20 mg. Maximum Dose : 80 mg
|
IVIG resistance: -
Recommendations for the identification of risk factors of IVIG resistance as well as management are shown in Recommendation table C.
Patients with persistent or recurrent fever ≥36 hours after the completion of the initial IVIg infusion are defined as IVIg resistant (50). Fifteen to 20% of individuals with KD seem to be IVIG resistant and are at greater risk of developing CAA formation (15%) than those who respond to IVIG (5%). Various IVIG resistance (refractory) prediction scores, such as the Kobayashi score (table 4) have been devised for predicting IVIG resistance if total score exceeds 4. Son risk score also can be used to determine risk score. It comprises age <6 months, Asian race, CA Z score >2 on initial echocardiogram, and C-reactive protein >13 mg/dL (each with 1 point assigned, except for 2 points assigned to CA). A risk score ≥3 points is strongly predictive of CAA by 8 weeks after acute illness (23). These scoring systems are thought to contribute to the continuous reduction of CAL occurrence (50). Current evidence supports the use of infliximab as rescue therapy in IVIG- and methylprednisolone-refractory patients with KD. IL-1 blockade with anakinra is highly promising in treating the most dramatically severe multi-refractory patients with KD, with potential benefits also on the cardiovascular complications (51). Table 5 shows the additional anti-inflammatory therapies in high-risk KD patients or those resistant to initial IVIG treatment
|
|
Cut off |
Score |
|
· Na |
≤133 mmol/L |
2 |
|
· AST |
≥100 IU/L |
2 |
|
· Day of starting treatment (or diagnosis) |
Day 4 of illness or earlier |
2 |
|
· Neutrophils |
≥80% |
2 |
|
· CRP |
≥10 mg/dL |
1 |
|
· Platelets |
<300,000 / μL |
1 |
|
· Age (months) |
≤12 months |
1 |
Table (4) Kobayashi Scoring System for prediction of IVIG resistance (50)
AST: Aspartate aminotransferase, CRP: C reactive protein. Kobayashi score ≥5 points has a sensitivity of 76% and a specificity of 80% in Japanese population
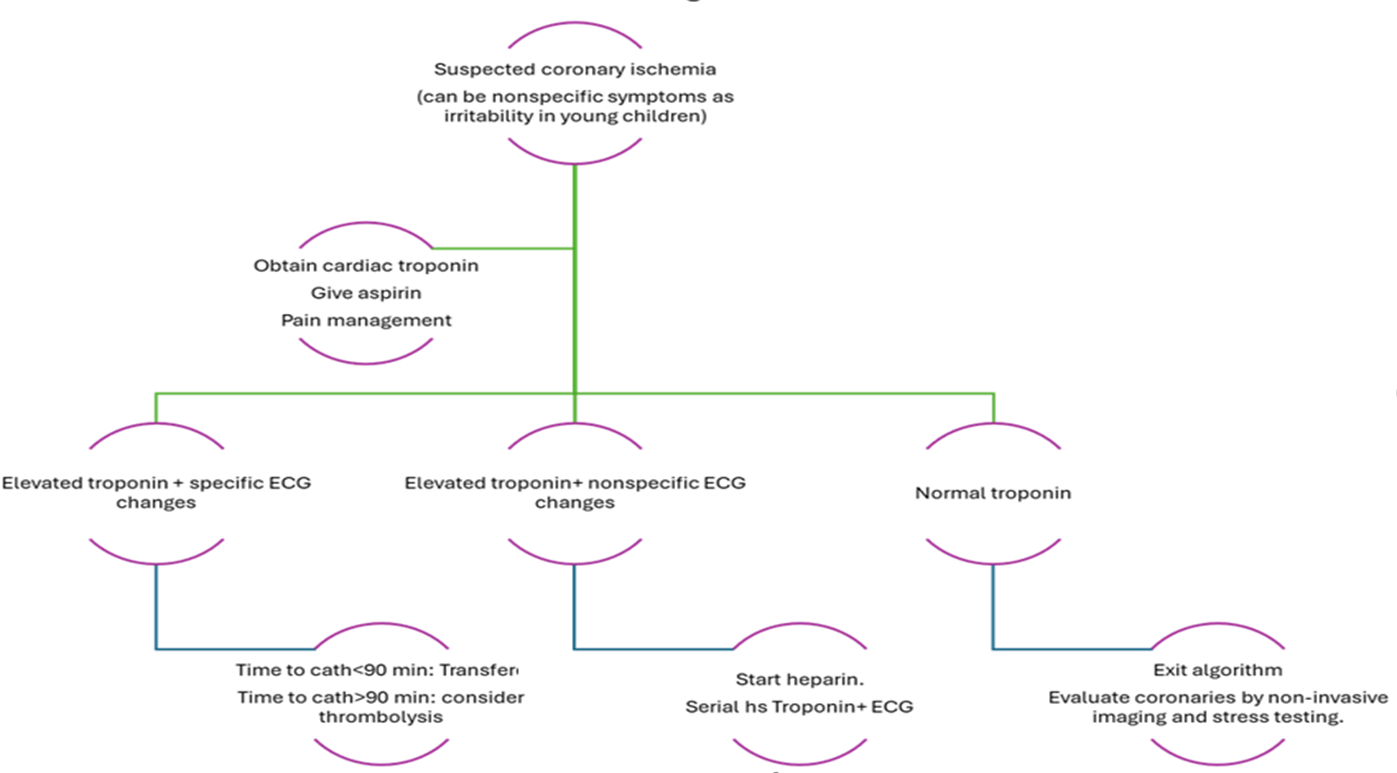
Table (5): Additional anti-inflammatory therapies in high-risk KD patients or those resistant to initial IVIG treatment *(51,52,53)
|
Drug name |
Mechanism of action |
Indication |
Dose |
Precautions |
Pharmaceutical form |
|
Anti-TNF alpha |
|||||
|
|
|||||
|
Infliximab
|
Chimeric murine/human IgG1 monoclonal antibody to TNF-alpha |
in KD patients with persistent inflammation despite IVIG, aspirin and corticosteroid treatment, after consultation with a specialist unit. |
IV, 10 mg/kg given over 2 h |
Exclusion of tuberculosis and infectious hepatitis before use Hold for suspected bacterial infection, fungal infection, varicella, or measles. |
100 mg of lyophilized infliximab in a 20 mL vial for IV use |
|
Etanercept |
Soluble receptor that binds TNFα and TNFβ |
SC, 0.8 mg/kg weekly ×3 doses |
Injection solution vials 25 mg and 50 mg for SC use |
||
|
Anti-IL1 |
|||||
|
Anakinra |
Interleukin-1 receptor antagonist |
In children with a refractory KD, Kawasaki disease shock syndrome, macrophage activation syndrome, persistent fever and laboratory abnormalities, worsening of coronary aneurysms, coronary aneurysms and increased proBNP levels and in patients with features overlapping with MIS-c |
IV/SC, 10 mg/kg per d (IV divided q12 h preferred to SC) while hospitalized; wean once ready for discharge (5 mg/kg per d for 1 d, then stop). |
Exclusion of tuberculosis and infectious hepatitis before use · Serum lipid monitoring after 2–3 months of therapy · Monitor for liver functions baseline and regularly |
Injection solution 100 mg/0.67ml for subcutaneous and intravenous use |
|
Plasma exchange |
|||||
|
|
Mechanical removal of inflammatory cytokines |
a high-risk procedure, should be reserved for extreme cases of refractory KD in whom all reasonable medical therapies have failed, especially if there are complications like severe infections or KD shock syndrome |
Displacing solution set at 5% albumin; 1–1.5× the patient’s circulating plasma volume is exchanged. Usually given for 3 continuous days (upper limit: 6 days) |
|
|
|
Other disease modifying antirheumatic drugs |
|||||
|
Cyclosporine |
Inhibitor of calcineurin – NFAT (nuclear factor of activated T cells) pathway |
May be considered in patients with refractory KD in whom a second IVIG infusion, infliximab, or a course of steroids has failed. In patients with MAS in combination with corticosteroids, IVIG and IL-1 antagonists |
PO, 5 mg/kg per d divided every 12 h; check 2 h level after 3rd dose (goal of 300–600 ng/mL); start to taper (by 10% every 3 d) once patient afebrile, clinically improving, and CRP ≤1.0 mg/dL or 10 d of therapy, whichever is longer |
If using liquid form, use glass dropper; may be mixed with milk, apple juice, or orange juice. Monitor blood pressure. Initial and regular blood counts; AST, ALT, BUN, creatinine, uric acid Reduce dose if creatinine increases by 30% Mg supplementation must be given while on cyclosporine to prevent hypomagnesemia. Should not be administered with statins as both are metabolized by cyt-P450 |
Oral solution (100 mg/ml) Oral capsules 25 mg, 50 mg Solution for intravenous (50 mg/ml) |
|
Cyclophosphamide |
Alkylating agent blocks DNA replication |
Might be used in refractory-KD. Should only be considered in severe refractory cases because of potential adverse reactions |
IV, 10 mg/kg per d in 1 or 2 doses |
Monitor blood counts, AST, ALT, BUN, creatinine. Adjust dosing or discontinue cyclophosphamide if WBC <1500/mm3, platelets <100,000/mm3, or hematuria Administer IV form with Mesna, good hydration and frequent bladder evacuation |
Injection powder solution 1gm/ 2 ml, 200 mg/ml
|
*Plasma exchange and biologic disease modifying anti-rheumatic drugs (DMARDs) like infliximab, anakinra cannot be recommended, except on an individual basis after consultation with a specialist unit.
*Non-biologic DMARDs such as cyclosporin, cyclophosphamide and methotrexate might be used in refractory-KD. They should only be considered in severe refractory cases because of potential adverse reactions, after consultation with a specialist unit.
IgG: immunoglobulin G, TNF: Tumor necrosis factor, SC: Subcutaneous, AST: Aspartate aminotransferase, ALT: Alanine aminotransferase, BUN: Blood urea nitrogen, MAS: Macrophage activation syndrome.
KD and Macrophage activation syndrome (MAS):
The recommendation of the identification and management of MAS in patients with KD is shown in Recommendation table C. Formal diagnostic criteria for MAS in the setting of KD have not been developed. However, drawing on experience with other secondary Hemophagocytic syndrome presentations, MAS may be suspected in KD patients with persistent fever, splenomegaly, elevated ferritin levels, and thrombocytopenia. Inadequate treatment of either KD or MAS could result in severe consequences. These include large coronary aneurysms or coronary artery stenosis, leading to death via cardiac infarct or coronary rupture in KD, or death due to multiorgan dysfunction in MAS. Thus, to ensure appropriate therapy, each disease entity should be considered separately with appropriate targeted therapy. KD should be treated with IVIG as the first-line therapy, and MAS should also be treated with appropriate agents for targeting cytokine storms or underlying triggers. Anakinra and glucocorticoids are preferred for treatment in these patients over a primary HLH-directed treatment protocol with cytotoxic agents. (51)
Thromboprophylaxis
KD patients with small or medium-size aneurysms receive antiplatelet therapy, following the belief that thrombus development is initiated by platelet activation and adhesion. In large aneurysms, however, humoral clotting factors are involved secondary to flow stasis; therefore, anticoagulant therapy is deemed necessary. Both Warfarin and LMWH have been shown to have similar efficiency in preventing thrombosis in KD patients with large aneurysms. They both have similar safety profiles with no substantial differences in the cumulative incidence of major bleeding complications (52).
Frequent blood work is required for routine monitoring of warfarin to ensure maintenance within a therapeutic window, and dosing can be challenging due to warfarin’s possible interactions with food. Certain foods that are rich in vitamin K may affect the therapeutic effects of warfarin, while other foods, such as grapefruit juice, interact with the metabolism of warfarin via the cytochrome P450 pathway (53). Furthermore, warfarin has many known drug interactions. Antimicrobials such as trimethoprim-sulfamethoxazole and erythromycin have been known to enhance the effects of warfarin, whereas rifampicin has been reported to reduce its effects. In addition, genetic polymorphisms have been found to be associated with warfarin metabolism, which further complicates the issues with dosing (54).
LMWH has added advantages over warfarin because of its somewhat less intensive monitoring requirements and faster achievement of monitoring levels within the therapeutic window, although potential negative effects on bone health associated with long-term treatment and discomfort associated with the injections make it less than ideal as a solution. It is a particularly useful option for younger patients in whom frequent blood work is less feasible (55)
Warfarin is advantageous compared with LMWH because of its ability to counter the anticoagulation effect promptly by means of vitamin K administration. The anti–factor Xa activity of LMWH is only partially reversible with protamine.
In the last decade, direct-acting oral anticoagulants, namely, direct thrombin inhibitors and anti–factor Xa agents, and novel antiplatelet agents have been introduced in adults. (29) These agents have been known to have certain advantages over classic anticoagulants, such as minor or absent food and drug interactions. In addition, they require limited or no monitoring and are delivered via the oral route, which may be a better option for patients who are averse to daily injections. Although their nonreversible nature has long been a limitation of direct oral anticoagulants from a safety point of view, antidote therapies for those agents are emerging which will allow reversal in case of severe bleeding events. However, these agents have not been adequately studied in children. Once there is more safety and efficacy data, regulatory approval, and availability of pediatric preparations, direct-acting oral anticoagulants will likely replace warfarin, not only for KD patients, but also for many other pediatric patients (56).
Recommendations table D shows the Management of coronary involvement in children with KD.
Coronary thrombosis and Myocardial Infarction in relation to KD:
KD patients with CAA are at highest risk of myocardial infarction in the first 2-3 months after illness. The presentation of these patients is different than adults. Patients with giant CAA have a lifelong risk of ischemia (58,59,3). The algorithm for management is shown in figure 6 .
Figure (6): Management of possible coronary thrombosis in children with KD
Long-term management (after convalescence) of KD patients with coronary lesions
Recommendations table E shows the long-term management of KD
Medical therapy for myocardial protection such as beta- blockers (carvedilol, metoprolol, or bisoprolol) decrease the risk of myocardial infarction and death by reducing myocardial oxygen demand. Angiotensin converting enzyme (ACE) inhibitors or Angiotensin receptor blockers (ARBs) also protect against myocardial infarction and death. Statins in addition to their cholesterol-lowering action have other pleiotropic effects in inflammation, endothelial dysfunction, oxidative stress, platelet aggregation, coagulation, and fibrinolysis, which make them useful in the management of KD. Huang, et al. reported a beneficial effect of short-term (3 months) statin treatment (simvastatin, 10 mg/day as a single dose at bedtime) in KD patients complicated with CAL. Chronic vascular inflammation is also significantly improved, as well as endothelial dysfunction, with no adverse effects. (61) However, long-term and randomized control trials are needed before further conclusions can be made.
The use of an ACE inhibitor in combination with a beta-blocker during the acute phase of Kawasaki disease (KD) in normotensive infants has been proposed in some observational studies; however, this approach has not been formally recommended by current guidelines. While there is a theoretical rationale based on the role of angiotensin II in promoting endothelial proliferation and vascular remodeling, which may contribute to coronary artery aneurysm (CAA) progression, the clinical evidence supporting such an intervention in the acute phase of KD is limited and of low certainty. Therefore, the panel judged this strategy to be speculative and not supported by sufficient high-quality data to inform a clinical recommendation. Further research is needed to assess the potential benefits and risks of this approach.
Following up of patients depends largely on the degree of coronary affection:
Patients with no coronary dilation should be maintained on LDA (low dose aspirin) for 6 weeks with no further interventions. If dilatation only was present at the acute stage, it should be reassessed at regular intervals till 1 year post convalescence.
For patients with small coronary artery aneurysms (Z score 2.5–4.9), regular follow-up with echocardiography is recommended. Aspirin may continue until normalization of coronary dimensions. The routine use of stress testing or coronary CT angiography (CTA) in this group is not currently supported by strong evidence and may be reserved for selected cases based on clinical risk factors or symptoms. Further studies are needed to determine the optimal imaging interval in this subgroup.
Patients with Medium and Large Coronary Aneurysms (Z score ≥5):
For medium aneurysms (Z 5–<10, diameter <8 mm): Dual antiplatelet therapy (e.g., aspirin + clopidogrel) should be considered until aneurysm regression with baseline ischemia assessment (stress echo or coronary CTA) at ~1-year post-illness to be repeated every 2–5 years, based on symptoms or clinical risk. Beta-blockers and stains may be considered for possible endothelial protection and ischemia risk reduction (62).
For Large aneurysms (Z ≥10 or diameter ≥8 mm): should consider combined antiplatelet and anticoagulation therapy in addition to beta blockers. Contact or high-impact sports should be restricted due to bleeding and ischemic risk. In addition to regular advanced imaging and close specialist follow-up (63).
Transition to adult care:
Patients with KD and coronary artery lesions, either persistent or those that have been remodeled and have decreased to a normal internal luminal dimension, should have long‐term follow‐up, with their care transitioned to an adult cardiologist once they reach adulthood, generally between the ages of 18 and 21 years.
The transition from pediatric to adult care for these patients needs to be a process involving a deliberate and coordinated series to ensure uninterrupted care. Transition should involve 6 cores:
1.The policy: preparing an office transition guide for families, including discussion of the practice's approach to an adult model of care around privacy and consent.
2. Tracking: ensuring that patient is offered a transition readiness/self‐care skill assessment conducted periodically.
3. Transition readiness. The pediatric clinician should have a standardized way of assessing the youth's self‐care skills.
4. Planning should include creating a transition plan jointly developed with youth and families that includes preparing decision-making support, assembling a medical summary and emergency care plan shared with youth and families, identifying an adult cardiologist and primary care clinician, and preparing transfer information for and communication with the new clinician.
5. Transfer of care, which requires engagement during this vulnerable transition time of both the adult clinician and the pediatric subspecialist to help the young adult schedule a follow‐up appointment with the adult health care clinician within 3 to 6 months of leaving the pediatric clinician.
6. The sixth core element is transition completion, during which the pediatric clinician confirms that the young adult has been seen by the adult clinician and facilitates obtaining patient feedback about the HCT process.
The 6 core elements is a process that evolves over several years and includes working with the adolescent's developmental stages to achieve maturity, education, and self‐management. More details on transition can be checked on the scientific statement from AHA 2021 (64).
- Methods of search
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for the search are Kawasaki disease, Cardiovascular sequelae of Kawasaki disease, Coronary affection in Kawasaki disease in children, Coronary artery aneurysms, coronary artery lesions.
Inclusion/exclusion criteria followed in the search and retrieval of guidelines were adapted:
• Selecting only evidence-based guidelines (guideline must include a report on the methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published within the last 5 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in the English language
• Excluding guidelines written by a single author
➡️The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 5 guidelines:
1. Revised recommendations of the Italian Society of Pediatrics about the general management of Kawasaki disease (2021) based on the Guidelines of the Italian Society of Pediatrics on Management of Kawasaki disease 2018.
2. American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Kawasaki Disease. (2021).
3. European consensus-based recommendations for the diagnosis and treatment of Kawasaki disease - the SHARE initiative (2019)
4. JCS/JSCS 2020 Guideline on Diagnosis and Management of Cardiovascular Sequelae in Kawasaki Disease (2020)
5. American College of Rheumatology Clinical Guidance for Multisystem Inflammatory Syndrome in Children Associated with SARS-CoV-2 and Hyperinflammation in Pediatric COVID-19: Version 2(2022)
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
➡️Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups: the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills, and diverse perspectives in the field of Pediatric Cardiology, as well as rheumatology and immunology. and clinical pharmacy.
The main functions of the clinical panel were adolopment of Kawasaki disease Guidelines, determining the scope of the guideline and guidelines, reviewing the evidence, and formulating evidence-informed recommendations in case of changing the strength of recommendations.
➡️Guideline Methodologists Subgroup
Guidelines methodologists with expertise in guidelines development, adaptation, GRADE, and translation of evidence into recommendations participated in the adaptation process. They provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinician's subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprised 3 clinical international and national experts who have interest and expertise in Kawasaki disease. They were identified by the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and the objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interest form before the peer review process.
Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [61]. Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 5. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 6. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is necessary about actual healthcare practice
2- Have large net positive consequences (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. The cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low-resource settings.
➡️Steps of implementing Kawasaki disease diagnosis, treatment, and prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps, and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment, and prevention, and adapt them to the local context or environment.
4. Identify barriers to and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
➡️Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for atypical kawaski , and differentiation of KD from other diseases.
3. Arabic Educational materials for nurses and mothers
- Limitations and suggestions for further research needs
➡️Future research recommendations for the management of KD and its cardiac sequalae in children in the Egyptian context could include:
· Large RCTs on the use of low dose IVIG versus high dose or IVIG replacement by steroids.
· Study of the long term microvascular coronary affection in children with history of Kawasaki disease.
· The benefits of cardio-protective medications post KD treatment.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for KD in children.
➡️Challenges
· Difficulty of reaching diagnosis of KD especially in incomplete or atypical forms.
· Lack of sufficient data on cardiac sequalae in children with KD.
Strengthen the evidence base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline
The following are three performance measures or indicators for implementing this adapted CPG for
KD management in children:
1. Adherence to KD Guidelines
· Numerator: Number of children with KD who received treatment as per guideline recommendations.
· Denominator: Total number of children diagnosed with KD
· Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
· Numerator: Total number of hospitals stay days for children with KD.
· Denominator: Total number of children admitted.
· Data Source: Hospital admission and discharge records.
3. Rate of Readmission
· Numerator: Number of children readmitted with symptoms of KD within a certain period (e.g., 30 days) after discharge.
· Denominator: Total number of children initially admitted with KD
· Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG KD GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (Checkups) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1- Marchesi A, Rigante D, Cimaz R, Ravelli A, Tarissi de Jacobis I, Rimini A, Cardinale F, Cattalini M, De Zorzi A, Dellepiane RM, Salice P, Secinaro A, Taddio A, Palma P, El Hachem M, Cortis E, Maggio MC, Corsello G, Villani A. Revised recommendations of the Italian Society of Pediatrics about the general management of Kawasaki disease. Ital J Pediatr. 2021 Jan 25;47(1):16. doi: 10.1186/s13052-021-00962-4. PMID: 33494789; PMCID: PMC7830049.
2- Marchesi A, Tarissi de Jacobis I, Rigante D, Rimini A, Malorni W, Corsello G, Bossi G, Buonuomo S, Cardinale F, Cortis E, De Benedetti F, De Zorzi A, Duse M, Del Principe D, Dellepiane RM, D'Isanto L, El Hachem M, Esposito S, Falcini F, Giordano U, Maggio MC, Mannarino S, Marseglia G, Martino S, Marucci G, Massaro R, Pescosolido C, Pietraforte D, Pietrogrande MC, Salice P, Secinaro A, Straface E, Villani A. Kawasaki disease: guidelines of the Italian Society of Pediatrics, part I - definition, epidemiology, etiopathogenesis, clinical expression and management of the acute phase. Ital J Pediatr. 2018 Aug 30;44(1):102. doi: 10.1186/s13052-018-0536-3. PMID: 30157897; PMCID: PMC6116535.
3- Fukazawa R, Kobayashi J, Ayusawa M, Hamada H, Miura M, Mitani Y, Tsuda E, Nakajima H, Matsuura H, Ikeda K, Nishigaki K, Suzuki H, Takahashi K, Suda K, Kamiyama H, Onouchi Y, Kobayashi T, Yokoi H, Sakamoto K, Ochi M, Kitamura S, Hamaoka K, Senzaki H, Kimura T; Japanese Circulation Society Joint Working Group. JCS/JSCS 2020 Guideline on Diagnosis and Management of Cardiovascular Sequelae in Kawasaki Disease. Circ J. 2020 Jul 22;84(8):1348-1407. doi: 10.1253/circj.CJ-19-1094. Epub 2020 Jul 8. PMID: 32641591.
4- Gorelik M, Chung SA, Ardalan K, Binstadt BA, Friedman K, Hayward K, Imundo LF, Lapidus SK, Kim S, Son MB, Sule S, Tremoulet AH, Van Mater H, Yildirim-Toruner C, Langford CA, Maz M, Abril A, Guyatt G, Archer AM, Conn DL, Full KA, Grayson PC, Ibarra MF, Merkel PA, Rhee RL, Seo P, Stone JH, Sundel RP, Vitobaldi OI, Warner A, Byram K, Dua AB, Husainat N, James KE, Kalot M, Lin YC, Springer JM, Turgunbaev M, Villa-Forte A, Turner AS, Mustafa RA. 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Kawasaki Disease. Arthritis Rheumatol. 2022 Apr;74(4):586-596. doi: 10.1002/art.42041. Epub 2022 Mar 7. PMID: 35257501.
5- Graeff N, Groot N, Ozen S, Eleftheriou D, Avcin T, Bader-Meunier B, Dolezalova P, Feldman BM, Kone-Paut I, Lahdenne P, McCann L, Pilkington C, Ravelli A, van Royen-Kerkhof A, Uziel Y, Vastert B, Wulffraat N, Kamphuis S, Brogan P, Beresford MW. European consensus-based recommendations for the diagnosis and treatment of Kawasaki disease - the SHARE initiative. Rheumatology (Oxford). 2019 Apr 1;58(4):672-682. doi: 10.1093/rheumatology/key344. PMID: 30535127.
6- Henderson LA, Canna SW, Friedman KG, Gorelik M, Lapidus SK, Bassiri H, Behrens EM, Ferris A, Kernan KF, Schulert GS, Seo P, Son MBF, Tremoulet AH, Yeung RSM, Mudano AS, Turner AS, Karp DR, Mehta JJ. American College of Rheumatology Clinical Guidance for Multisystem Inflammatory Syndrome in Children Associated With SARS-CoV-2 and Hyperinflammation in Pediatric COVID-19: Version 2. Arthritis Rheumatol. 2021 Apr;73(4):e13-e29. doi: 10.1002/art.41616. Epub 2021 Feb 15. PMID: 33277976; PMCID: PMC8559788.
7- McCrindle BW, Rowley AH, Newburger JW, Burns JC, Bolger AF, Gewitz M, Baker AL, Jackson MA, Takahashi M, Shah PB, Kobayashi T, Wu MH, Saji TT, Pahl E; American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young; Council on Cardiovascular and Stroke Nursing; Council on Cardiovascular Surgery and Anesthesia; and Council on Epidemiology and Prevention. Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals From the American Heart Association. Circulation. 2017 Apr 25;135(17):e927-e999. doi: 10.1161/CIR.0000000000000484. Epub 2017 Mar 29. Erratum in: Circulation. 2019 Jul 30;140(5):e181-e184. doi: 10.1161/CIR.0000000000000703. PMID: 28356445.
8- Jone PN, Tremoulet A, Choueiter N, Dominguez SR, Harahsheh AS, Mitani Y, Zimmerman M, Lin MT, Friedman KG; American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young; Council on Cardiovascular and Stroke Nursing; Council on Cardiovascular Radiology and Intervention; and Council on Clinical Cardiology. Update on Diagnosis and Management of Kawasaki Disease: A Scientific Statement From the American Heart Association. Circulation. 2024 Dec 3;150(23):e481-e500. doi: 10.1161/CIR.0000000000001295. Epub 2024 Nov 13. PMID: 39534969.
9- Alzyoud R, El-Kholy N, Arab Y, Choueiter N, Harahsheh AS, Aselan AS, Kotby A, Bouaziz A, Salih AF, Abushhaiwia A, Alahmadi F, Agha HM, Elmarsafawy HM, Alrabte H, Al-Saloos H, Boudiaf H, Hijazi I, Bouayed K, Al Senaidi KS, Boughammoura L, Jalal M, Ladj MS, Abu-Shukair ME, ElGanzoury MM, Hammadouche N, Elsamman N, Mouawad P, Boukari R, Benalikhoudja N, Jdour S, Abu Al-Saoud SY, Touri SN, Kammoun T, Fitouri Z, Dahdah N. Access to Care and Therapy for Kawasaki Disease in the Arab Countries: A Kawasaki Disease Arab Initiative (Kawarabi) Multicenter Survey. Pediatr Cardiol. 2023 Aug;44(6):1277-1284. doi: 10.1007/s00246-023-03166-1. Epub 2023 May 1. PMID: 37126143.
10- Holman RC, Belay ED, Christensen KY, Folkema AM, Steiner CA, Schonberger LB. Hospitalizations for Kawasaki syndrome among children in the United States, 1997-2007. Pediatr Infect Dis J. 2010 Jun;29(6):483-8. doi: 10.1097/INF.0b013e3181cf8705. PMID: 20104198.
11- Jindal AK, Pilania RK, Guleria S, Vignesh P, Suri D, Gupta A, Singhal M, Rawat A, Singh S. Kawasaki Disease in Children Older Than 10 Years: A Clinical Experience From Northwest India. Front Pediatr. 2020 Feb 14;8:24. doi: 10.3389/fped.2020.00024. PMID: 32117831; PMCID: PMC7034337.
12- Burns JC, Herzog L, Fabri O, Tremoulet AH, Rodó X, Uehara R, Burgner D, Bainto E, Pierce D, Tyree M, Cayan D; Kawasaki Disease Global Climate Consortium. Seasonality of Kawasaki disease: a global perspective. PLoS One. 2013 Sep 18;8(9):e74529. doi: 10.1371/journal.pone.0074529. PMID: 24058585; PMCID: PMC3776809.
13- Aggarwal R, Pilania RK, Sharma S, Kumar A, Dhaliwal M, Rawat A, Singh S. Kawasaki disease and the environment: an enigmatic interplay. Front Immunol. 2023 Dec 18;14:1259094. doi: 10.3389/fimmu.2023.1259094. PMID: 38164136; PMCID: PMC10757963.
14- Burns JC. The etiologies of Kawasaki disease. J Clin Invest. 2024 Mar 1;134(5):e176938. doi: 10.1172/JCI176938. PMID: 38426498; PMCID: PMC10904046
15- Kuo HC. Diagnosis, Progress, and Treatment Update of Kawasaki Disease. Int J Mol Sci. 2023 Sep 11;24(18):13948. doi: 10.3390/ijms241813948. PMID: 37762250; PMCID: PMC10530964
16- Gong GW, McCrindle BW, Ching JC, Yeung RS. Arthritis presenting during the acute phase of Kawasaki disease. J Pediatr. 2006 Jun;148(6):800-5. doi: 10.1016/j.jpeds.2006.01.039. PMID: 16769390.
17- Colomba C, La Placa S, Saporito L, Corsello G, Ciccia F, Medaglia A, Romanin B, Serra N, Di Carlo P, Cascio A. Intestinal Involvement in Kawasaki Disease. J Pediatr. 2018 Nov;202:186-193. doi: 10.1016/j.jpeds.2018.06.034. Epub 2018 Jul 17. PMID: 30029859
18- Agha HM, Hamza HS. Incomplete Kawasaki disease in Egypt. Glob Cardiol Sci Pract. 2017 Oct 31;2017(3):e201724. doi: 10.21542/gcsp.2017.24. PMID: 29564345; PMCID: PMC5856965.
19- Jiao F, Pan Y, Du Z, Deng F, Yang X, Wang H, Shen J, Xiang W, Mu Z, Gao C, Bai J. Guideline for the diagnosis and treatment of incomplete Kawasaki disease in children in China. BMC Pediatr. 2024 Jul 26;24(1):477. doi: 10.1186/s12887-024-04961-2. PMID: 39060924; PMCID: PMC11282762.
20- Mitsuishi T, Miyata K, Ando A, Sano K, Takanashi JI, Hamada H: Characteristic nail lesions in Kawasaki disease: case series and literature review. J Dermatol. 2022, 49:232-8. 10.1111/1346-8138.16276
21- Liu YC, Hou CP, Kuo CM, Liang CD, Kuo HC. Atypical kawasaki disease: literature review and clinical nursing. Hu Li Za Zhi. 2010 Dec;57(6):104-10. Chinese. PMID: 21140351
22- Falcini F. Kawasaki disease. Curr Opin Rheumatol. 2006 Jan;18(1):33-8. doi: 10.1097/01.bor.0000197998.50450.f6. PMID: 16344617.
23- Dionne A, Dahdah N. A Decade of NT-proBNP in Acute Kawasaki Disease, from Physiological Response to Clinical Relevance. Children (Basel). 2018 Oct 12;5(10):141. doi: 10.3390/children5100141. PMID: 30322059; PMCID: PMC6210997.
24- Rizk SR, El Said G, Daniels LB, Burns JC, El Said H, Sorour KA, Gharib S, Gordon JB. Acute myocardial ischemia in adults secondary to missed Kawasaki disease in childhood. Am J Cardiol. 2015 Feb 15;115(4):423-7. doi: 10.1016/j.amjcard.2014.11.024. Epub 2014 Nov 29. PMID: 25555655; PMCID: PMC4697961.
25- Dionne A, Dahdah N. A Decade of NT-proBNP in Acute Kawasaki Disease, from Physiological Response to Clinical Relevance. Children. 2018; 5(10):141. https://doi.org/10.3390/children5100141
26- Wang W, Wang H, Wang H, Cheng J. Kawasaki disease with shock as the primary manifestation: How to distinguish from toxic shock syndrome?: A case report and literature review. Medicine (Baltimore). 2024 Aug 2;103(31):e39199. doi: 10.1097/MD.0000000000039199. PMID: 39093792; PMCID: PMC11296402.
27- Chen PS, Chi H, Huang FY, Peng CC, Chen MR, Chiu NC. Clinical manifestations of Kawasaki disease shock syndrome: a case-control study. J Microbiol Immunol Infect. 2015 Feb;48(1):43-50. doi: 10.1016/j.jmii.2013.06.005. Epub 2013 Aug 6. PMID: 23927822.
28- Lamrani L, Manlhiot C, Elias MD, Choueiter NF, Dionne A, Harahsheh AS, Portman MA, McCrindle BW, Dahdah N. Kawasaki Disease Shock Syndrome vs Classical Kawasaki Disease: A Meta-analysis and Comparison With SARS-CoV-2 Multisystem Inflammatory Syndrome. Can J Cardiol. 2021 Oct;37(10):1619-1628. doi: 10.1016/j.cjca.2021.05.014. Epub 2021 Jun 6. PMID: 34090979; PMCID: PMC8180353.
29- CDC U.S. Centers of disease control and prevention Clinical Guidance for Streptococcal Toxic Shock Syndrome May, 2024
30- Panupattanapong S, Brooks EB. New spectrum of COVID-19 manifestations in children: Kawasaki-like syndrome and hyperinflammatory response. Cleve Clin J Med. 2020 Dec 31. doi: 10.3949/ccjm.87a.ccc039. Epub ahead of print. PMID: 32493734
31- Noval Rivas M, Arditi M. Kawasaki Disease and Multisystem Inflammatory Syndrome in Children: Common Inflammatory Pathways of Two Distinct Diseases. Rheum Dis Clin North Am. 2023 Aug;49(3):647-659. doi: 10.1016/j.rdc.2023.03.002. Epub 2023 Mar 17. PMID: 37331738; PMCID: PMC10020039.
32- Lim KYY, Chua MC, Tan NWH, Chandran S. Reactivation of BCG inoculation site in a child with febrile exanthema of 3 days duration: an early indicator of incomplete Kawasaki disease. BMJ Case Rep. 2020 Dec 17;13(12):e239648. doi: 10.1136/bcr-2020-239648. PMID: 33334773; PMCID: PMC7747535
33- Iio K, Fukushima N, Akamine K, Uda K, Hataya H, Miura M. Acute Rheumatic Fever and Kawasaki Disease Occurring in a Single Patient. Front Pediatr. 2020 Sep 3;8:562. doi: 10.3389/fped.2020.00562. PMID: 33014943; PMCID: PMC7494961.
34- Gewitz MH, Baltimore RS, Tani LY, Sable CA, Shulman ST, Carapetis J, Remenyi B, Taubert KA, Bolger AF, Beerman L, Mayosi BM, Beaton A, Pandian NG, Kaplan EL; American Heart Association Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease of the Council on Cardiovascular Disease in the Young. Revision of the Jones Criteria for the diagnosis of acute rheumatic fever in the era of Doppler echocardiography: a scientific statement from the American Heart Association. Circulation. 2015 May 19;131(20):1806-18. doi: 10.1161/CIR.0000000000000205. Epub 2015 Apr 23. Erratum in: Circulation. 2020 Jul 28;142(4):e65. doi: 10.1161/CIR.0000000000000889. PMID: 25908771.
35- Cox JR and Sallis RE. Recognition of Kawasaki Disease. The Permanente Journal volume 13, No 1Published Online: March 1, 2009 https://doi.org/10.7812/TPP/08-042
36- Phuong, L. K., Bonetto, C., Buttery, J., Pernus, Y. B., Chandler, R., Goldenthal, K. L., Kucuku, M., Monaco, G., Pahud, B., Shulman, S. T., Top, K. A., Ulloa-Gutierrez, R., Varricchio, F., de Ferranti, S., Newburger, J. W., Dahdah, N., Singh, S., Bonhoeffer, J., & Burgner, D. (2016). Kawasaki disease and immunisation: Standardised case definition & guidelines for data collection, analysis. Vaccine, 34(51), 6582-6596. https://doi.org/10.1016/j.vaccine.2016.09.025
37- Zhang QY, Xu BW, Du JB. Similarities and differences between multiple inflammatory syndrome in children associated with COVID-19 and Kawasaki disease: clinical presentations, diagnosis, and treatment. World J Pediatr. 2021 Aug;17(4):335-340. doi: 10.1007/s12519-021-00435-y. Epub 2021 May 20. PMID: 34013488; PMCID: PMC8134825.
38- Salgado AP, Ashouri N, Berry EK, Sun X, Jain S, Burns JC, Tremoulet AH. High Risk of Coronary Artery Aneurysms in Infants Younger than 6 Months of Age with Kawasaki Disease. J Pediatr. 2017 Jun;185:112-116.e1. doi: 10.1016/j.jpeds.2017.03.025. Epub 2017 Apr 10. PMID: 28408126; PMCID: PMC5529235.
39- Van Anh KY, Shah S, Tremoulet AH. Hemolysis From Intravenous Immunoglobulin in Obese Patients With Kawasaki Disease. Front Pediatr. 2020 Apr 3;8:146. doi: 10.3389/fped.2020.00146. PMID: 32318529; PMCID: PMC7146618
40- Shaanxi Provincial Diagnosis and Treatment Center of Kawasaki Disease/Children's Hospital of Shaanxi Provincial People's Hospital; Beijing Children's Hospital, Capital Medical University; Shanghai Children's Medical Center; Children's Hospital of Shanghai Jiao Tong University; Xianyang Children's Hospital of Shaanxi Province; Suzhou Children's Hospital, Suzhou University; Children's Hospital of Chongqing Medical University; Expert Committee of Advanced Training for Pediatrician, China Maternal and Children's Health Association; Editorial Board of Chinese Journal of Contemporary Pediatrics. Pediatric expert consensus on the application of glucocorticoids in Kawasaki disease. Zhongguo Dang Dai Er Ke Za Zhi. 2022 Mar 15;24(3):225-231. English, Chinese. doi: 10.7499/j.issn.1008-8830.2112033. PMID: 35351250; PMCID: PMC8974659.
41- Nadig PL, Joshi V, Pilania RK, Kumrah R, Kabeerdoss J, Sharma S, Suri D, Rawat A, Singh S. Intravenous Immunoglobulin in Kawasaki Disease-Evolution and Pathogenic Mechanisms. Diagnostics (Basel). 2023 Jul 11;13(14):2338. doi: 10.3390/diagnostics13142338. PMID: 37510082; PMCID: PMC10378342.
42- Rigante D, Andreozzi L, Fastiggi M, Bracci B, Natale MF, Esposito S. Critical Overview of the Risk Scoring Systems to Predict Non-Responsiveness to Intravenous Immunoglobulin in Kawasaki Syndrome. Int J Mol Sci. 2016 Feb 24;17(3):278. doi: 10.3390/ijms17030278. PMID: 26927060; PMCID: PMC4813142
43- Sleeper LA, Minich LL, McCrindle BM, Li JS, Mason W, Colan SD, Atz AM, Printz BF, Baker A, Vetter VL, Newburger JW; Pediatric Heart Network Investigators. Evaluation of Kawasaki disease risk-scoring systems for intravenous immunoglobulin resistance. J Pediatr. 2011 May;158(5):831-835.e3. doi: 10.1016/j.jpeds.2010.10.031. Epub 2010 Dec 18. PMID: 21168857; PMCID: PMC3075321.
44- obayashi T, Saji T, Otani T, Takeuchi K, Nakamura T, Arakawa H, Kato T, Hara T, Hamaoka K, Ogawa S, Miura M, Nomura Y, Fuse S, Ichida F, Seki M, Fukazawa R, Ogawa C, Furuno K, Tokunaga H, Takatsuki S, Hara S, Morikawa A; RAISE study group investigators. Efficacy of immunoglobulin plus prednisolone for prevention of coronary artery abnormalities in severe Kawasaki disease (RAISE study): a randomised, open-label, blinded-endpoints trial. Lancet. 2012 Apr 28;379(9826):1613-20. doi: 10.1016/S0140-6736(11)61930-2. Epub 2012 Mar 8. PMID: 22405251.
45- Catella-Lawson F, Reilly MP, Kapoor SC et al. Cyclooxygenase inhibitors and the antiplatelet effects of aspirin. N Engl J Med 2001;345:1809_17.
46- Moreno E, Garcia SD, Bainto E, Salgado AP, Parish A, Rosellini BD, Ulloa-Gutierrez R, Garrido-Garcia LM, Dueñas L, Estripeaut D, Luciani K, Rodríguez-Quiroz FJ, Del Aguila O, Camacho-Moreno G, Gómez V, Viviani T, Alvarez-Olmos MI, de Souza Marques HH, Faugier-Fuentes E, Saltigeral-Simental P, López-Medina E, Miño-León G, Beltrán S, Martínez-Medina L, Pirez MC, Cofré F, Tremoulet AH; The REKAMLATINA-2 Study Group Investigators. Presentation and Outcomes of Kawasaki Disease in Latin American Infants Younger Than 6 Months of Age: A Multinational Multicenter Study of the REKAMLATINA Network. Front Pediatr. 2020 Jul 16;8:384. doi: 10.3389/fped.2020.00384. PMID: 32766186; PMCID: PMC7378383.
47- Cameron SA, Carr M, Pahl E, DeMarais N, Shulman ST, Rowley AH. Coronary artery aneurysms are more severe in infants than in older children with Kawasaki disease. Arch Dis Child. 2019 May;104(5):451-455. doi: 10.1136/archdischild-2018-314967. Epub 2018 Nov 9. PMID: 30413485.
48- Fitzgerald DA. Aspirin and Reye syndrome. Paediatr Drugs. 2007;9(3):205-6. doi: 10.2165/00148581-200709030-00009. PMID: 17523701.
49- Suzuki T, Michihata N, Hashimoto Y, Yoshikawa T, Saito K, Matsui H, Fushimi K, Yasunaga H. Association between aspirin dose and outcomes in patients with acute Kawasaki disease: a nationwide retrospective cohort study in Japan. Eur J Pediatr. 2024 Jan;183(1):415-424. doi: 10.1007/s00431-023-05302-8. Epub 2023 Nov 2. PMID: 37917176.
50- Kim GB, Yu JJ, Yoon KL, Jeong SI, Song YH, Han JW, Hong YM, Joo CU. Medium- or Higher-Dose Acetylsalicylic Acid for Acute Kawasaki Disease and Patient Outcomes. J Pediatr. 2017 May;184:125-129.e1. doi: 10.1016/j.jpeds.2016.12.019. Epub 2016 Dec 30. PMID: 28043685.
51- Kobayashi T, Inoue Y, Tamura K, Morikawa A, Kobayashi T. External validation of a scoring system to predict resistance to intravenous immunoglobulin. J Pediatr. 2007 Apr;150(4):e37; author reply e38. doi: 10.1016/j.jpeds.2006.12.036. PMID: 17382098.
52- Manlhiot C, Newburger JW, Low T, Dahdah N, Mackie AS, Raghuveer G, Giglia TM, Dallaire F, Mathew M, Runeckles K, Pahl E, Harahsheh AS, Norozi K, de Ferranti SD, Friedman K, Yetman AT, Kutty S, Mondal T, McCrindle BW; International Kawasaki Disease Registry. Low-Molecular-Weight Heparin vs Warfarin for Thromboprophylaxis in Children With Coronary Artery Aneurysms After Kawasaki Disease: A Pragmatic Registry Trial. Can J Cardiol. 2020 Oct;36(10):1598-1607. doi: 10.1016/j.cjca.2020.01.016. Epub 2020 Jul 1. PMID: 32621885.
53- Sadowski C, Reinert JP. The efficacy and safety of direct oral anticoagulants in the treatment of the acute phase of heparin-induced thrombocytopenia: A systematic review. Am J Health Syst Pharm. 2024 Sep 23;81(19):e584-e593. doi: 10.1093/ajhp/zxae109. PMID: 38651828
54- . Takahashi H, Wilkinson GR, Nutescu EA, Morita T, Ritchie MD, Scordo MG, Pengo V, Barban M, Padrini R, Ieiri I, Otsubo K, Kashima T, Kimura S, Kijima S, Echizen H. Different contributions of polymorphisms in VKORC1 and CYP2C9 to intra- and inter-population differences in maintenance dose of warfarin in Japanese, Caucasians and African-Americans. Pharmacogenet Genomics. 2006 Feb;16(2):101-10. doi: 10.1097/01.fpc.0000184955.08453.a8. PMID: 16424822.
55- van Veen JJ, Maclean RM, Hampton KK, Laidlaw S, Kitchen S, Toth P, Makris M. Protamine reversal of low molecular weight heparin: clinically effective? Blood Coagul Fibrinolysis. 2011 Oct;22(7):565-70. doi: 10.1097/MBC.0b013e3283494b3c. PMID: 21959588
56- Mekaj YH, Mekaj AY, Duci SB, Miftari EI. New oral anticoagulants: their advantages and disadvantages compared with vitamin K antagonists in the prevention and treatment of patients with thromboembolic events. Ther Clin Risk Manag. 2015 Jun 24;11:967-77. doi: 10.2147/TCRM.S84210. PMID: 26150723; PMCID: PMC4485791.
57- Oné-Paut I, Tellier S, Belot A, Brochard K, Guitton C, Marie I, Meinzer U, Cherqaoui B, Galeotti C, Boukhedouni N, Agostini H, Arditi M, Lambert V, Piedvache C. Phase II Open Label Study of Anakinra in Intravenous Immunoglobulin-Resistant Kawasaki Disease. Arthritis Rheumatol. 2021 Jan;73(1):151-161. doi: 10.1002/art.41481. Epub 2020 Nov 17. PMID: 32779863.
58- Buda P, Friedman-Gruszczyńska J, Książyk J. Anti-inflammatory Treatment of Kawasaki Disease: Comparison of Current Guidelines and Perspectives. Front Med (Lausanne). 2021 Nov 30;8:738850. doi: 10.3389/fmed.2021.738850. PMID: 34917629; PMCID: PMC8669475.
59- Scherler L, Haas NA, Tengler A, Pattathu J, Mandilaras G, Jakob A. Acute phase of Kawasaki disease: a review of national guideline recommendations. Eur J Pediatr. 2022 Jul;181(7):2563-2573. doi: 10.1007/s00431-022-04458-z. Epub 2022 Apr 11. PMID: 35403975; PMCID: PMC8995165..
60- Burns JC, El-Said H, Tremoulet AH, Friedman K, Gordon JB, Newburger JW. Management of Myocardial Infarction in Children with Giant Coronary Artery Aneurysms after Kawasaki Disease. J Pediatr. 2020 Jun;221:230-234. doi: 10.1016/j.jpeds.2020.02.033. Epub 2020 Apr 17. PMID: 32312552.
61- Zeng YY, Zhang M, Ko S, Chen F. An Update on Cardiovascular Risk Factors After Kawasaki Disease. Front Cardiovasc Med. 2021 Apr 16;8:671198. doi: 10.3389/fcvm.2021.671198. PMID: 33937365; PMCID: PMC8086797
62- Brogan P, Burns JC, Cornish J, Diwakar V, Eleftheriou D, Gordon JB, Gray HH, Johnson TW, Levin M, Malik I, MacCarthy P, McCormack R, Miller O, Tulloh RMR; Kawasaki Disease Writing Group, on behalf of the Royal College of Paediatrics and Child Health, and the British Cardiovascular Society. Lifetime cardiovascular management of patients with previous Kawasaki disease. Heart. 2020 Mar;106(6):411-420. doi: 10.1136/heartjnl-2019-315925. Epub 2019 Dec 16. PMID: 31843876; PMCID: PMC7057818.
63- Huang SM, Weng KP, Chang JS, Lee WY, Huang SH, Hsieh KS. Effects of statin therapy in children complicated with coronary arterial abnormality late after Kawasaki disease: a pilot study. Circ J. 2008 Oct;72(10):1583-7. doi: 10.1253/circj.cj-08-0121. Epub 2008 Sep 1. PMID: 18758088.
64- Dahdah N, Kung SC, Friedman KG, Marelli A, Gordon JB, Belay ED, Baker AL, Kazi DS, White PH, Tremoulet AH; American Heart Association Rheumatic Fever, Endocarditis, Kawasaki Disease Committee of the Council on Lifelong Congenital Heart Disease and Heart Health in the Young, and the Council on Arteriosclerosis, Thrombosis and Vascular Biology. Falling Through the Cracks: The Current Gap in the Health Care Transition of Patients With Kawasaki Disease: A Scientific Statement From the American Heart Association. J Am Heart Assoc. 2021 Oct 19;10(20):e023310. doi: 10.1161/JAHA.121.023310. Epub 2021 Oct 11. PMID: 34632822; PMCID: PMC8751858.
65- Hill KD, Frush DP, Han BK, Abbott BG, Armstrong AK, DeKemp RA, Glatz AC, Greenberg SB, Herbert AS, Justino H, Mah D, Mahesh M, Rigsby CK, Slesnick TC, Strauss KJ, Trattner S, Viswanathan MN, Einstein AJ; Image Gently Alliance. Radiation Safety in Children With Congenital and Acquired Heart Disease: A Scientific Position Statement on Multimodality Dose Optimization From the Image Gently Alliance. JACC Cardiovasc Imaging. 2017 Jul;10(7):797-818. doi: 10.1016/j.jcmg.2017.04.003. Epub 2017 May 18. PMID: 28514670; PMCID: PMC5542588
66- Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
67- Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
68- Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
69- Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
70- Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
71- Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
72- Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
73- Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
- Annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the Diagnosis and management of KD and its cardiac sequalae in children " on PubMed are Kawasaki disease, Coronary aneurysm, IVIG, Cardiac sequalae.
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof Alyaa Kotby |
Professor Of Pediatrics and Pediatric Cardiology- Ain Shams University- Egypt |
None |
Not Applicable |
|
Prof Hala Agha |
Professor Of Pediatrics and Pediatric Cardiology- Cairo University- Egypt |
None |
Not Applicable |
|
Prof Duaa Raafat |
Professor Of Pediatrics and Pediatric Cardiology- Assuit University- Egypt |
None |
Not Applicable |
|
Prof Rasha Hussin |
Professor Of Pediatrics and Pediatric Immunology- Ain Shams University- Egypt |
None |
Not Applicable |
|
A. Prof Nanies Soliman |
Assistant Professor Of Pediatrics and Pediatric Cardiology- Ain Shams University- Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics and Pediatric Cardiology- MTI University- Egypt |
None |
Not Applicable |
|
Dr Mahmoud Nadder |
Assistant lecturer of Pediatrics |
None |
Not applicable |
|
|
|
|
|
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanis Soliman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Dr. Nagib Dahdah |
Associate professor of pediatrics and pediatric Cardiology, Montréal university. |
None |
Not Applicable |
|
Dr. Sanaa Mahmoud |
Clinical Professor of Pediatrics, Allergy& Immunology Pikeville university, Pikeville, KY Adjunct Faculty, Lincoln Debusk Memorial University, Harrogate, TN |
None |
Not Applicable |
|
Dr. Mahmoud Magdy Abu el Kheir |
Professor of Pediatrics and Pediatric Cardiology, Mansoura University. Head of Pediatric Departement, Faculty of Medcine, Horus University. |
None |
Not Applicable |
Annex Table 2. Results of the AGREE II assessment of the Five source guidelines
|
Guidelines |
Domain 1 Scope& purpose |
Domain 2
Stakehold. involve |
Domain 3 Rigor of develop. |
Domain 4 Clarity of present. |
Domain 5 Applicab. |
Domain 6
Editorial independ. |
Overall |
Can be used? |
|
1. Revised recommendations of the Italian Society of Pediatrics about the general management of Kawasaki disease. |
91% |
72% |
94% |
98% |
81% |
100% |
89% |
Yes:3 |
|
2. 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of Kawasaki Disease. |
81% |
83% |
87% |
96% |
79% |
94% |
87% |
Yes:2 Yes with modification. |
|
3. European consensus-based recommendations for the diagnosis and treatment of Kawasaki disease - the SHARE initiative. |
83% |
72% |
87% |
96% |
74% |
89% |
83% |
Yes:3 |
|
4. JCS/JSCS 2020 Guideline on Diagnosis and Management of Cardiovascular Sequelae in Kawasaki Disease. |
89% |
69% |
66% |
94% |
69% |
86% |
78% |
Yes: 3 |
|
5. American College of Rheumatology Clinical Guidance for Multisystem Inflammatory Syndrome in Children Associated With SARS-CoV-2 and Hyperinflammation in Pediatric COVID-19: Version 2. |
91% |
69% |
89% |
98% |
82% |
67% |
89% |
Yes: 3 |
Annex 3. Nurses and Parents Educational Guide in Arabic
الملخص التعليمى الخاص بالتمريض و أهالى الأطفال المصابين بداء كاوازاكى
أهداف و ارشادات التمريض:-
قد تشمل الأهداف والنتائج المتوقعة ما يلي:
Ø أن يحافظ الطفل على درجة حرارته الطبيعية.
Ø أن يكون الغشاء المخاطي لفم الطفل خاليًا من الجفاف والتهيج.
Ø أن يشعر الأهل بقلق أقل.
Ø أن يشعر الطفل بألم أقل.
Ø أن يبدأ شفاء الطفل من الطفح الجلدى.
Ø أن يقوم الطفل بالاعتماد على نفسه فى الأنشطة الحياتية أو أقصى ما يكون في حدود المرض.
كيفية الوصول الى تنفيذ الأهداف:-
١. إلمساعدة على التحكم فى الالتهابات وارتفاع الحرارة
المرضى الذين يعانون من مرض كاواساكي، وهي حالة تسبب التهاب الأوعية الدموية في جميع أنحاء الجسم، قد يعانون من ارتفاع درجة الحرارة كجزء من أعراضهم. يجب خفض حمى المريض ومراقبة علاماته الحيوية لمنع المضاعفات المرتبطة بارتفاع الحرارة. علاوة على ذلك، يمكن أن يسبب مرض كاواساكي التهابًا في الفم والحلق والشفتين، مما يؤدي إلى احمرار وتورم وألم في الغشاء المخاطي للفم. يمكن أن يؤدي ذلك إلى صعوبة في البلع والتحدث وتناول الطعام، الأمر الذي يتطلب رعاية تمريضية دقيقة لمنع المزيد من المضاعفات وتعزيز الشفاء.
v مراقبة درجة الحرارة كل ٤ ساعات؛ كل ساعتين إذا كانت مرتفعة.
v يبدأ مرض كاواساكي في البداية بحمى شديدة (٣٩ درجة إلى ٤٠ درجة) لمدة 5 أيام أو أكثر.
v تقييم التغيرات في الشفاه وتجويف الفم.
v تشمل التغيرات النموذجية في الغشاء المخاطي احمرار الفم واللسان الذى بشبه الفراولة والشفاه الحمراء الجافة والمتشققة.
v توفير حمامات الاسفنج الفاترة لدرجات الحرارة التى تزيد عن ٣٨ درجة وذلك يساعد على فقدان الحرارة من خلال التوصيل والتبخر.
v توفير فترات راحة كافية. فالراحة في الفراش تقلل من متطلبات التمثيل الغذائي واستهلاك الأكسجين.
v تشجيع تناول كمية كافية من السوائل لأن الجفاف أو التعرق يزيد من الحمى.
v إعطا ء الطفل المريض الدواء كما هو محدد.
v •أسبرين. وهو دواء مضاد للالتهابات يعطى لتقليل الالتهاب.
• الغلوبولين المناعي الوريدى يتم إعطاؤه بجرعة واحدة لعلاج الالتهاب وتقليله وبالتالي تقليل مدة الحمى.
v تقديم الأطعمة الناعمة وغير المهيجة .
v يتطلب الطعام اللين مضغًا أقل ويقلل التهيجً أ للغشاء المخاطي للفم.
v توفير السوائل الباردة للمحافظة على ترطيب الفم وتقليل الام الفم بوضع المراهم المهدئة على الشفاه.
v الحفاظ على الشفاه رطبة باستخدام زبدة كاكاو مرطبة لتجنب الألم.
v استخدام فرشاة ذات شعيرات ناعمة أثناء العناية بالفم حيث ان الفرشاة ذات الشعيرا ت الناعمة تحد من تهيج الغشاء المخاطي.
v توفير رعاية منتظمة للفم باستخدام غسول الفم الخالي من الكحول الذى يحد من تراكم البكتيريا التي يمكن أن تسبب العدوى.
٢- كيفية المساعدة على التحكم فى الألم وتخفيفه:-
v مرض كاواساكي يسبب التهابًا في جدران الأوعية الدموية في جميع أنحاء الجسم. يمكن أن يؤدي هذا الالتهاب إلى آلام في أجزاء مختلفة من الجسم، بما في ذلك المفاصل والبطن والصدر. ويعد تخفيف الألم عنصرًا مهمًا في خطط الرعاية التمريضية للمرضى الذين يعانون من مرض كاواساكي للمساعدة في تخفيف انزعاجهم وتحسين نوعية حياتهم.
v تقييم مستوى الألم من خلال الملاحظة (التعبيرات اللفظية عن الألم، تكشيرة الوجه)، وذلك باستخدام تقييم مقياس الألم، ومن خلال الحصول على معلومات الألم ذات الصلة من الوالدين.
v الحفاظ على غرفة الطفل هادئة وإبقاء الاضاءة خافتة للاقلال من انزعاج العين الناجم عن التهاب الملتحمة.
v الشرح للوالدين سبب انزعاج/تهيج الطفل يعزز التفاهم والتعاون.
v الشرح للوالدين أن التهيج قد يستمر لمدة تصل إلى شهرين؛و أن تقشر الجلد في اليدين والقدمين أمر طبيعي وغير مؤلم.
v الشرح للوالدين أن آلام المفاصل قد تستمر لعدة أسابيع
v تعليم الوالدين تمارين المدى الحركى لمفصل الركبة والتمارين السلبية في حمام دافئ تساعد على زيادة المرونة
v ضع قطعة قماش باردة على الجلد، وغسولًا، وملابس ناعمة وفضفاضة على الطفل يخفف من حكة الجلد، وبالتالي يعزز الراحة.
v التعامل مع الطفل بلطف وتجنب الحركات غير الضروريةل ان الحركة تسبب عدم الراحة.
٣. الحفاظ على سلامة الجلد و الاهتمام بالأغشية المخاطية:-
قد يصاب المرضى المصابون بمرض كاواساكي بطفح جلدي وتقشير ومظاهر جلدية أخرى بسبب الالتهاب الناجم عن المرض. وهذا يمكن أن يؤدي إلى ضعف سلامة الجلد، مما يجعل المرضى أكثر عرضة للإصابة بالتهابات الجلد والمضاعفات الأخرى. تعد العناية بالبشرة والمراقبة المناسبة من المكونات الأساسية لخطط الرعاية التمريضية للمرضى الذين يعانون من مرض كاواساكي لتعزيز الشفاء ومنع المزيد من تلف الجلد.
v تقييم الجلد من حيث الملمس والتورم واللون والرطوبة وأى شكل غير صحى.
v تشتمل السمات الجلدية الكلاسيكية لمرض كاواساكي على احمرار وتورم وطفح جلدي متعدد الأشكال وتقشير يؤثر على جلد الأطراف .
v ألبسي الطفل ملابس خفيفة. الملابس الثقيلة قد تؤدي إلى تهيج الطفح الجلدي
v عدم التشجيع على استخدام الصابون لانه يجعل الجلد جافًا .
v قم بإزالة أغطية السرير الرطبة والمتجعدة.
v ضع كمادة باردة ورطبة على مناطق الحكة الجلدية لتوفير الراحة وتقليل الحكة.
v تشجيع تناول كمية كافية من السوائل للحفاظ على الترطيب وتقليل ألم الفم.
v التشجيع على تناول الأطعمة الغنية بالبروتين مثل البيض والفاصوليا والدجاج.
لان البروتين ضروري لصحة وصيانة الجلد
٤. تعزيز القدرة على الحركة البدنية:-
مرض كاواساكي يمكن أن يؤدي إلى تمدد يالأوعية الدموية في الشرايين التاجية. وقد يسبب مرض كاواساكي فى ضعف العضلات وآلام المفاصل والتورم، مما قد يؤدي إلى محدودية الحركة، ويتطلب ذلك رعاية تمريضية دقيقة لمنع المضاعفات وتعزيز التعافي.
v تقييم مستوى طاقة الطفل وقدرته على أداء أنشطة الحياة اليومية.
v تؤثر الحركة المقيدة الناتجة عن آلام المفاصل على القدرة على أداء أنشطة الحياة اليومية بشكل فعال.
v توفير الدعم المشترك باستخدام الوسائد.
v يمكن استخدام الوسائد لتثبيت المفصل وتقليل خطر الإصابة بقرح الضغط.
v تزويد الطفل بالوقت الكافي لإنجاز الأنشطة المتعلقة بالتنقل وتشجيعه على الراحة بينهما.
v تعظيم التزام الطفل ومشاركته في النشاط؛ يعزز الحفاظ على الطاقة وتقليل التعب.
v المساعدة في تمارين المدى الحركى لمفصل الركبة السلبية كما هو مسموح به.
v يحافظ على وظيفة المفاصل وقوة العضلات والقدرة على التحمل بشكل عام ويحسنها.
v تقديم تعليمات حول استخدام الأجهزة المساعدة المناسبة أثناء التمشي.
v التشجيع على تناول الأطعمة مثل السلمون والتونة والحبوب الكاملة والجزر.
v تساعد هذه المواد الغذائية على تقوية الحركة وتعظيم إنتاج الطاقة
٥-التحكم فى القلق :-
-قد يعاني المرضى المصابون بمرض كاواساكي من القلق بسبب عدم اليقين المحيط بالمرض، واحتمال حدوث مضاعفات طويلة الأمد، والحاجة إلى تدخلات طبية متكررة. بالإضافة إلى ذلك، قد يكون الأطفال المصابون بمرض كاواساكي عرضة بشكل خاص للقلق وذلك لطبيعة المرض نفسه.
v تثقيف الوالدين حول المعلومات المتعلقة بالمرض وعلاماته وأعراضه وتشخيصه وطرق التعامل معه.
v الشرح بأن سبب المرض غيرمعروف يساعد في تخفيف أي شعور بالذنب لدى الوالدين فيما يتعلق باكتساب المرض.
v إبلاغ الوالدين بالتعامل بلطف مع الطفل حسب الحاجة.
v -توفير المعلومات التي يحتاجها الآباء لتوفير الراحة
v تجنب اللمس غير الضروري للمريض.
v تشجيع الوالدين على التعبير عن مشاعرهم بحرية. طمأنة الوالدين بأن بعض القلق طبيعى عندما يكون طفلهما مريضاً.
v يساعد التشجيع والطمأنينة الوالدين على التعرف على عواطفهم واستعادة السيطرة عليها.
v الشرح للوالدين أن الطفل قد يعاني من حمى متكررة في المنزل ونعلمهم كيفية قياس درجة حرارة الطفل ومتى يجب إخطار الطبيب .
v التأكد من حصول الطفل على رعاية فعالة في المنزل يقلل من القلق المرتبط بعدم اليقين.
v الشرح للوالدين أن التهيج هو أحد أعراض مرض كاواساكي وأنه ليس خطاهم ؛ وتشجيعهم على أخذ قسط من الراحة بينما تعتني الممرضة بالطفل.
v تقديم الدعم والإغاثة للآباء والأمهات في أوقات الأحداث العصيبة.
٦-مراقبة العلامات الحيوية للطفل عن كثب أثناء إعطاء الجلوبيولين المناعي الوريدى :-
v غاما جلوبيولين هو أحد منتجات الدم ويتطلب المراقبة الدقيقة وهذا يطمئن الوالدين بأن طفلهم يتلقى الرعاية المناسبة
v قم بإنهاء التسريب فى حالة حدوثه والإبلاغ فورًا عن ردود الفعل غير المرغوب فيها مثل الحمى والقشعريرة وضيق الصدر وضيق التنفس والغثيان / القيء.
v تعليم الوالدين كيفية التعامل مع الأسبرين و الإبلاغ عن أي علامات سمية (طنين الأذن، والصداع، والدوخة، والارتباك). اشرح أن الأسبرين قد يسبب كدمات
v يجب إيقاف الأسبرين وإخطار الطبيب إذا تعرض الطفل للجديرى المائى أو الأنفلونزا (خطر الإصابة بمتلازمة راي).
٧- مساعدة أولياء الأمور في تحديد مواعيد الإحالة والمتابعة للطفل.
المساعدة تساعد على تقليل القلق.
ارشادات الآباء:
ما هو مرض كاواساكي؟
مرض كاواساكي هو التهاب فى الاوردة يؤدى لارتفاع حاد فى درجات الحرارة وغالبًا ما يصيب الأطفال الذين تقل أعمارهم عن ٥ سنوات ولكنه وارد الحدوث فى الأطفال الاكبر سنا
و هو نتيجة التهاب جهاز الأوعية الدموية مجهول السبب، وعادة ما ينتهى المرض من تلقاء نفسه وعلى الرغم من أن التنبؤ بتمام الشفاء جيد مع العلاج، إلا ان حوالى٢٥% من الذين لا يعالجون يصابوا بمضاعفات قلبية مثل التهاب الشرايين التاجية و تمدد الأوعية الدموية .
الاسباب:-
حتى الان لم يتم اكتشاف السبب المؤدى لمرض كواساكى ولكن يعتقد انه التهاب فيروسى أو بكتبرى سابق مصحوب بتفاعل مناعى يؤدى الى التهابات بالأوعية الدموية
اعراض المرض:-
ينقسم المرض الى ثلاث مراحل:-
المرحلة الحاده (الأسبوع الأول و الثانى):-
Ø تبدا بحمى شديدة قد تصل درجة حرارة جسم طفلك إلى 40 درجة مئوية.
و الحرارة لا تستجيب للمضادات الحيوية أو مخفضات الجرارة مثل الإيبوبروفين أو الباراسيتامول.
Ø عادة ما تستمر درجة الحرارة المرتفعة لمدة ٥ أيام على الأقل، ولكنها يمكن أن تستمر لمدة اسابيع اذا لم يعطى العلاج المناسب لمرض كاواساكي.
§ وارتفاع الحرارة يكون مصحوبا باربعة من الاعراض التالية:
v طفح جلدى على الجسم والوجه والاذرع والسيقان وله أشكال مختلفة
v التهاب بالحلق واللسان والشفايف
v التهاب العينبن واحمرارهما
v وتورم اليدين و القدمين
v تضخم العقد الليمفاوية بالرقبة
· الشفاه والفم والحلق واللسان:-
Ø قد تكون شفاه طفلك حمراء أو جافة أو متشققة. وقد تنتفخ أيضًا وتتقشر أو تنزف.
Ø قد يكون أيضًا الجزء الداخلي من فم طفلك وحلقه ملتهبًا.
Ø قد يكون السان أحمر اللون، منتفخًا ويُعرف هذا باسم "لسان الفراولة".
· اليدين والقدمين
Ø قد تتورم يدي طفلك وأقدامه، وقد يصبح الجلد على يديه وأقدامه أحمر أو جامدا
Ø قد يشعر طفلك بأن يديه وأقدامه مؤلمة عند لمسها أو ، ,وقد يجد صعوبة في المشي أو الزحف أثناء استمرار
· تورم الغدد الليمفاوية وهى كتل منتفخة فى رقبة طفلك عادة على جانب واحد
ويعتبر التهيج هو أحد أعراض مرض كاواساكي وقد تكون هناك اعراض اخرى مثل مشاكل في القلب ,آلام البطن ,القيء,إسهال,بول يحتوي على صديد,الشعور بالنعاس ونقص الطاقة ,صداع ,آلام المفاصل وتورم المفاصل واصفرار الجلد وبياض العينين (اليرقان)
قد لا يعاني الأطفال الذين تقل أعمارهم عن سنة واحدة من العديد من الأعراض الرئيسية مقارنة بالأطفال الأكبر سنًا. والتشخيص قد يكون صعبا فى ذلك السن.
المرحلة تحت الحادة(الأسبوع الثالث والرابع):-
تصبح أعراض طفلك أقل حدة.
من المفترض أن تهدأ درجة الحرارة المرتفعة، لكن قد يظل طفلك عصبيًا.
قد تشمل الأعراض تقشر جلد اليدين ثم القدمين، وأحيانًا أيضًا على راحتي اليدين أو باطن القدمين ويبدا التقشير فى اليدين ثم القدمين و يكون الطفل أكثر عرضة للاصابة بتمدد الأوعية الدموية التاجية وتزيد الصفائح الدموية.
مرحلة النقاهة (من خمسة الى ثمانى اسابيع)
حيث تختفى الأعراض بالتدريح وتبدا نتائج التحاليل فى التحسن.
لكن قد يظل طفلك يعاني من نقص الطاقة ويتعب بسهولة خلال هذا الوقت.
التشخيص:-
لايوجد اختبار محدد لتشخيص مرض كاواساكي، ولكن هناك بعض العلامات الاكلينيكية الرئيسية التي تشير إلى احتمال إصابة الطفل بهذه الحالة و بعض التحاليل التى يجب اجرائها .
الرجاء اخبار طبيبك المعالج بأى عرض يظهر ويستمر أو يختفى
التشخيص المعملى:
يمكن إجراء العديد من الاختبارات للمساعدة في دعم تشخيص مرض كاواساكى. قد لا تكون الاختبارات حاسمة بشكل فردي ، ولكن عند دمجها مع بعض الأعراض الرئيسية لمرض كاواساكي، فإنها يمكن أن تساعد في تأكيد التشخيص وقد يحتاج الطفل إلى إجراء اختبارات لاستبعاد الامراض الأخرى التي قد تسبب أعراض مشايهة
فحوصات القلب:-
عادةً ما تؤثر مضاعفات مرض كاواساكي على القلب. وهذا يعني أن طفلك قد يحتاج إلى بعض الاختبارات للتأكد من أن قلبه يعمل بشكل طبيعي مثل:
١- مخطط كهربية القلب (ECG)
٢- مخطط صدى القلب والتي يمكن أن تؤكد ما إذا كانت هناك أي مشاكل في بنية القلب أو وظيفته
العلاج:
يتم علاج مرض كاواساكي في المستشفى لأنه يمكن أن يسبب مضاعفات خطيرة. يجب أن يبدأ العلاج في أقرب وقت ممكن.
قد يستغرق طفلك وقتًا أطول للتعافي و يزيد خطر الإصابة بتمدد الشراين التاجية إذا لم يتم العلاج على الفور.
العلاجان الرئيسيان لمرض كاواساكي هما:
v الجلوبيولين المناعي الوريديIVIG.
o أظهرت الأبحاث أن IVIG يمكن أن يقلل من الحمى وخطر الإصابة بمشاكل في القلب.
o بعد إعطاء طفلك IVIG، يجب أن تتحسن أعراضه خلال ٣٦ ساعة.
إذا لم تتحسن درجة حرارتهم المرتفعة بعد ٣٦ ساعة، فقد يتم إعطاؤهم جرعة ثانية من IVIG و علاجات اخرى.
v الأسبرين
o يتم استخدامه لعلاج مرض كاواساكي لأنه يمكن تخفيف الألم والانزعاج. يمكن أن يساعد في تقليل درجة الحرارة المرتفعة
عند تناول جرعات عالية، يعتبر الأسبرين مضادًا للالتهابات (يخفف التورم). عند تناول جرعات منخفضة، يعتبر الأسبرين مضادًا للصفيحات (يمنع تكون جلطات الدم)
جرعة الأسبرين الموصوفة لطفلك والمدة التي يحتاجها لتناولها تعتمد على الأعراض التي يعاني منها.
o يتم إعطاء جرعة عالية من الأسبرين حتى تنخفض درجة الحرارة.
o ويمكن بعد ذلك وصف جرعة منخفضة من الأسبرين لهم لمدة 6 أسابيع.وذلك لتقليل جلطات الدم .
الكورتيكوستيرويدات
يوصى بها إذا لم تكن IVIG فعالة، أو إذا وجد أن طفلك معرض لخطر كبير للإصابة بمشاكل في القلب.
بعد العلاج
عندما يخرج طفلك من المستشفى، يجب أن تحصل على النصائح حول كيفية العناية به في المنزل.
قد يشمل ذلك التأكد من أنهم مرتاحون قدر الإمكان وأنهم يشربون الكثير من السوائل.
تأكد من أن طفلك يستمر في تناول أي دواء موصوف له وابحث عن أي آثار جانبية.
سيتم تحديد موعد متابعة لطفلك وستستمر مراقبة قلبه.
بمجرد التأكد من فحص القلب بالموجات فوق الصوتية (مخطط صدى القلب) أن طفلك لا يعاني من أي مشاكل في القلب، يمكنه عادةً التوقف عن تناول الأسبرين.
قد يستغرق التعافي الكامل حوالي ٦ أسابيع، ولكنه قد يستغرق وقتًا أطول عند بعض الأطفال.
قد تكون هناك حاجة إلى متابعة العلاج إذا أصيب طفلك بمزيد من المضاعفات.
الآثار الجانبية للأسبرين
عادةً لا يُعطى الأسبرين للأطفال دون سن ١٦ عامًا لأنه يمكن أن يسبب آثارًا جانبية، بما في ذلك متلازمة راي. متلازمة راي نادرة وتشمل أعراضها القيء المستمر ونقص الطاقة.
احصل على المساعدة الطبية فورًا إذا كان طفلك يعاني من أي من هذه الأعراض.
المضاعفات: اصابة الشرايين التاجية للقلب
مع العلاج الفوري، يتعافي معظم الأطفال
وترتبط مضاعفات مرض كاواساكي بشكل أساسي بالقلب.
تحدث نتيجة لالتهاب وتمدد الأوعية الدموية يالقلب وقد يؤثر هذا أحيانًا على الأوعية الدموية خارج القلب
.وقد يسبب هذا فى نوبة قلبية - حيث يموت جزء من عضلة القلب بسبب حرمانها من الأكسجين
ومن الممكن أيضًا أن تتأثر الشرايين الأخرى المتوسطة الحجم.
وقدتشفى بعض هذه المضاعفات من تلقاء نفسها مع مرور الوقت. لكن قد يتعرض بعض الأطفال لمزيد من المضاعفات التي تتطلب متابعة وعلاج يمنع التجلط فى تمددات الأوعية الدموية.
تعد المضاعفات المرتبطة بالقلب المرتبطة بمرض كاواساكي خطيرة وقد تكون قاتلة في 2 إلى 3٪ من الحالات التي لا يتم علاجها.
من المعروف أن الأطفال الذين تقل أعمارهم عن سنة واحدة أكثر عرضة لخطر الإصابة بمضاعفات خطيرة بالقلب.
علاج المضاعفات
تشمل العلاجات الممكنة ما يلي:
استخدام الأدوية المضادة للتخثر والأدوية المضادة للصفيحات
– الأدوية التي توقف تخثر الدم، والتي قد تمنع إصابة طفلك بنوبة قلبية إذا كان يعانى من تمدد شرايين القلب.
وقد يحتاج الطفل لرأب الأوعية التاجية – إجراء لتوسيع الشرايين التاجية المسدودة أو الضيقة لتحسين تدفق الدم إلى القلب؛ وفي بعض الأحيان يتم إدخال دعامة في الشريان المسدود لإبقائه مفتوحًا
نادرا ما يحتاج الطفل لعملية جراحية لتحويل الدم بالشرايين الضيقة أو المسدودة، لتحسين تدفق الدم وإمداد الأكسجين إلى القلب
قد يعاني الأطفال الذين يعانون من مضاعفات خطيرة من تلف دائم في عضلات القلب أو الصمامات.
هؤلاء الاطفال سيكون لديهم مواعيد متابعة منتظمة مع أستشارى قلب الاطفال حتى يمكن مراقبة حالتهم عن كثب.
وبعد سن ثمانى عشر سنة يتم تحويل المريض الى رعاية طبيب قلب الكبار متخصص فى مرض كواساكى .
Annex Table 4: Differential diagnosis:
|
Point of comparison |
Kawasaki disease, |
Scarlet fever (Scarlatina) |
Measles |
|
Etiology |
Unknown? viral |
Group A Strptococcus bacterial throat infection |
Morbilivirus |
|
Fever |
+ve |
+ve, usually >10days |
+ ve, Prodromal symptoms |
|
Rash and its distribution |
Erythematous, maculopapullar and polymorphous on trunk, face, hands and feet
|
Red, fine, "sandpaper-like" rash is usually found on the neck, forehead, cheeks, and chest, and then may spread to the arms and back, it begins 1-2 days after fever. It fades after 3-4 days. Circumoral pallor |
Maculopapular that may be confluent usually on the face and upper neck, spreads over 3 days, eventually to the hands and feet, lasts 5–6 days before fading.
|
|
Desquamation |
Starts periungal , glove & stocking peeling |
Desquamative “bran like” rash begins 1 week after onset
|
Dry desquamation |
|
Mucosal involvement
|
strawberry tongue, fissured or erythematous, dry fissured lips |
Pharyngitis |
Koplik’s spot
|
|
Conjunctivitis |
non purulent |
normal eyes
|
Conjunctivitis |
|
Lymphadenopathy |
unilateral cervical lymphadenpathy
|
Painful neck lymphadenopathy |
+/- |
|
Extremities |
Erythema, edema, induration and painful hands and feet |
Linear petchia in anticubital and axillary folds (Lines of Pastia)
|
-- |
Table (4)a DD of Kawasaki disease, scarlet fever and Measles.
|
Point pf comparison |
Kawasaki Disease |
Systemic onset juvenile idiopathic arthritis |
Rheumatic Fever |
Steven Johnson Syndrome |
|
Age |
Usually, <5years |
2-5 years |
>5 years |
Any age |
|
Fever |
persistent fever
|
Prolonged daily fever or twice daily for at least 2 weeks |
One of the minor criteria
|
Prolonged |
|
Jones criteria |
- |
- |
Major criteria Arthritis, carditis, Chorea erythema marginatum subcutaneous nodules Minor Criteria Arthralgia, fever, ↑ESR, ↑CRP, ↑PR interval |
- |
|
Arthritis Arthralgia |
+/- |
arthritis is a must
|
Migratory, fleeting polyarthritis, big joints |
arthralgia
|
|
Eye changes |
non exudative conjunctivitis Limbal sparing
|
- |
- |
exudative conjunctivitis, keratitis
|
|
Oral mucosa |
Diffuse erythema, strawberry tongue |
normal |
+ve throat swab for group A β hemolytic strept |
Erythema, ulceration, pseudo membrane formation |
|
Rash |
Erythematous, maculopapular and polymorphous on trunk, face, hands and feet
|
transient salmon pink rash
|
Erythema Marginatum |
erythema, ulcerations, pseudo membrane formation, target lesions |
|
Lymphadenopathy |
At least 1 cervical lymph node >1.5cm
|
Generalized lymphadenopathy
|
- |
- |
|
Investigations |
↑ESR ,↑CRP anemia Elevated liver enzymes thrombocytosis after day 7 |
↑ESR, ↑CRP anemia
|
↑ASOT ↑ESR, ↑CRP Prolonged PR interval
|
associated herpes virus infection
|
|
Echocardiography
|
mainly coronary involvement |
pericarditis
|
mainly endocarditis valvular involvement
|
- |
Table (4)b DD Kawasaki Disease with autoimmune: Systemic onset juvenile idiopathic arthritis, Rheumatic Fever and Steven Johnson Syndrome.
|
Point of comparison |
Kawasaki disease (KD) |
Kawasaki disease shock syndrome |
Multisystem inflammatory Syndrome in Children |
Toxic shock syndrome |
|
age |
usually < 5 years
|
6 months to 10 years common in girls
|
6-10years
|
All age groups acute in nature toxin induced renal injury is common |
|
fever |
Persistent unresponsive to antipyretic |
Persistent unresponsive to antipyretic |
present |
Usually less than 10 days But hypothermia can occur in patients with shock |
|
Rash |
Erythematous, maculopapullar and polymorphous on trunk, face, hands and feet |
Maculopapular erythematous and polymorphous on trunk, face, hands and feet
|
Hetero -genous |
Erythroderma Macular rash which may desquamate
|
|
Cardiac affection |
coronary involvement if untreated with IVIG 25% Treated 5-8%
uncommon myocarditis with ventricular dysfunction rarely pericarditis and valvulitis |
Common coronary artery affection (frequency unknown)
Pericarditis common |
increased incidence of myocarditis and myocardial involvement
|
Shock +/- myocarditis |
|
GIT |
+/- hepatitis |
+/- diarrhea
|
increased incidence of GIT affection |
- |
|
other |
Irritable arthritis desquamation of perineal area, Inflammation of BCG site Uncommon respiratory symptoms
|
Relatively rare Full criteria for Kawasaki disease and toxic shock syndrome including shock diarrhea CNS and muscle involvement Possibly IV IG is nonresponsive than KD |
shock Macrophage activation syndrome more commonly seen |
coagulopathy mental status changes myositis renal dysfunction hypotension and shock
|
|
Investigations |
Thrombocytosis is characteristic rarely lymphopenia and increased ferritin and D Dimer |
As KD |
Increased ferritin, thrombocytopenia leucopenia lymphopenia
|
leukocytosis or leucopenia isolation of bacteria on culture thrombocytopenia |
|
Medications |
IVIg, steroids and biologic drugs are second line therapy
|
Needs Support and aggressive treatment Repeat dose of IVIG Methyl prednisolone Biologic Drugs |
inotropes vasopressors mechanical ventilation ECMO, IVIg steroids Biologic drugs
|
antibiotics and management of shock |
Table (4)c Critical cases commonly mistaken for Kawasaki disease
Appendix Table 5. The RIGHT-Ad@pt checklist |
|||||
|
|
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|