Indigestion in ruminant
| Site: | EHC | Egyptian Health Council |
| Course: | Large ruminant Medicine and surgery Guidelines |
| Book: | Indigestion in ruminant |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:47 AM |
Description
"last update: 7 December 2025" Download Guideline
Table of contents
- - Acknowledgement
- - Scope
- - Definition of indigestion in ruminants
- - Types of indigestion
- - Simple Indigestion in Cattle
- - Lactic (ruminal) acidosis in ruminants
- - Tympany (Bloat) in Ruminants
- - Abomasal Displacement in Cattle
- - Ruminal parakeratosis
- - Ruminal drinking (reticuloruminal milk accumulation) in young ruminants
- - Secondary indigestion
- - References
- Acknowledgement
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Dr Mohamed Mustafa Lotief.
Head of the Committee: Prof. Dr Ahmed M Byomi
The rapporteur of the Committee: Prof. Dr Mohamed Mohamedy Ghanem.
Scientific Group Members: Prof. Nabil Yassien, Prof. Ashraf Aly Eldesoky Shamaa, Prof. Amany Abbas, Prof. Dalia Mansour, Essam Sobhy Mohamed Elsharkawy, Prof. Gamal A. Sosa., Dr Naglaa Radwan, Dr Hend El Sheikh
Author: Prof. Mohamed Ghanem
- Scope
This guideline targets the veterinarians who are working at large ruminant farms including cattle, buffalo, dairy cows, and fattening calves. The guideline describes the basic diagnosis and management of the diseases causing different types of indigestion and affecting ruminant production. Both clinical and laboratory findings of indigestion will be presented. Differential diagnosis of diseases causing indigestion will be outlined.
- Definition of indigestion in ruminants
it is the functional or mechanical disturbances in the fore-stomach (reticulorumen) that disrupt normal gastrointestinal transit and function, mainly the microbial and enzymatic digestion. It is often triggered by dietary changes, leading to altered ruminal pH, abnormal fermentation, and changes in rumen motility. The net result is the decreased animal production, milk production, decreased body weight gain producing high economic losses.
- Types of indigestion
Indigestion can be classified into
1- Primary indigestion: due to diseases originated from gastrointestinal tract
2- Secondary indigestion: due to systemic diseases
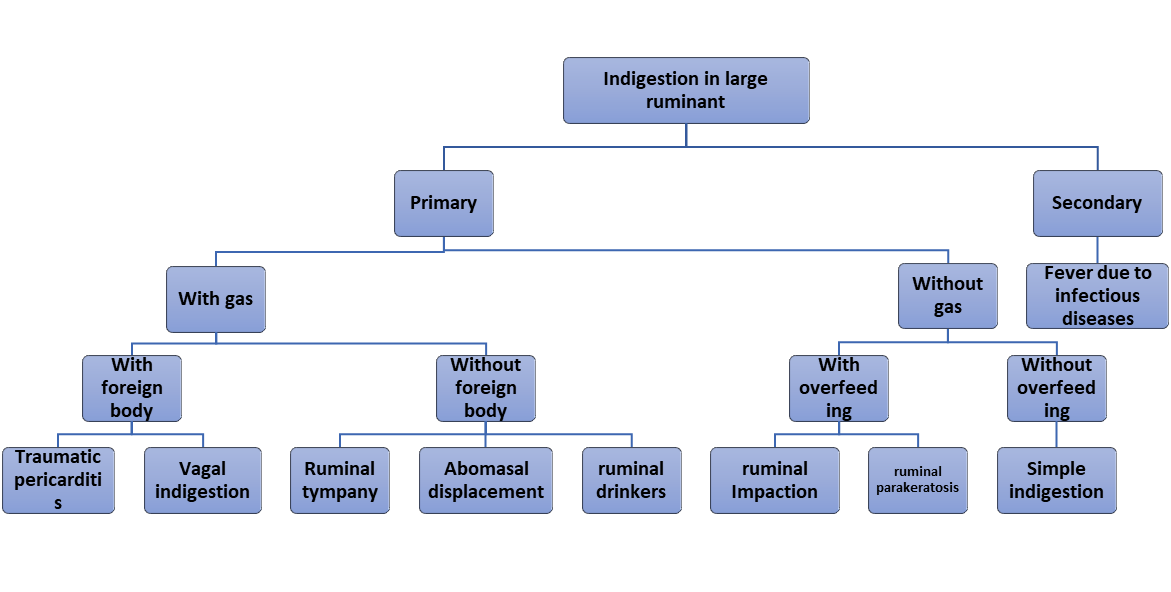
The following diagram summarize the different types of indigestion in ruminant
- Simple Indigestion in Cattle
1. Definition
Simple indigestion in cattle is a mild and transient form of ruminal dysfunction characterized by abnormal fermentation within the rumen. It results in decreased appetite, reduced rumen motility, altered fecal consistency, and mild depression. It typically follows dietary changes, poor-quality feed, or overfeeding of easily fermentable carbohydrates.
2. Etiology
Simple indigestion is usually caused by dietary factors that disrupt normal rumen microbial flora and fermentation patterns. Common etiological factors include:
- Sudden dietary changes, especially to highly fermentable carbohydrates (e.g., grains).
- Spoiled, moldy, or frozen feed.
- Low-fiber or poorly digestible rations.
- Excess intake of silage or concentrates.
- Overfeeding after a fasting period.
- Ingestion of non-nutritive substances (plastic, sand, etc.).
- Abrupt changes in feed composition or feeding schedule.
3. Pathogenesis
The ingestion of inappropriate or excessive feed alters the microbial population within the rumen. This disrupts normal fermentation, leading to:
- Reduced cellulolytic bacterial activity.
- Accumulation of undigested feed.
- Reduced volatile fatty acid (VFA) production.
- Rumen hypomotility or atony.
- mild acidosis or alkalosis, depending on the type of ingested material.
- In grains, acidosis is produced
- In legumes, alkalosis is produced
The net effect is impaired digestion, reduced appetite, and decreased milk production or weight gain.
4. Clinical Signs
Clinical signs are usually mild and nonspecific:
- Anorexia or inappetence
- Reduced rumination and rumen motility
- Mild to moderate drop in milk yield
- Mild abdominal distension (mainly left side)
- Altered feces (dry, scant, or soft and foul-smelling)
- Depression or dullness
- Decreased or absent rumen contractions on auscultation
- Low-pitched, splashing rumen sounds if fluid-filled
Body temperature and heart rate are often within normal ranges or mildly elevated.
5. Diagnosis
a. Clinical Examination
- History of recent feed change or dietary error.
- Observation of typical clinical signs.
b. Rectal Examination
- May reveal firm fecal balls or scant fecal output.
c. Rumen Fluid Analysis
- pH changes: mildly acidic (<6.0) in carbohydrate overload; more alkaline (>7.5) with protein putrefaction (normal 6.8).
- Decreased protozoal activity
- Odor: sour or putrid smell.
d. Laboratory Tests (Optional)
- Mild changes in blood electrolytes or acid-base status.
- Total protein and hematocrit may be mildly increased if dehydration occurs.
6. Differential Diagnosis
- Rumen acidosis
- Rumen alkalosis
- Traumatic reticuloperitonitis
- Vagal indigestion
- Hardware disease
- Displaced abomasum
- Rumen tympany (bloat)
7. Treatment
a. Supportive Care
- Withhold feed for 12–24 hours (rumen rest).
- Provide good-quality hay and fresh water afterward.
b. Rumen Correctives
- Oral administration of rumen buffers (e.g., magnesium oxide, sodium bicarbonate).
- Rumen transfaunation using rumen fluid from a healthy donor to restore microbial flora.
c. Prokinetics or Rumen Stimulants
- commercial rumen stimulants (e.g., ruminotorics like neostigmine, metoclopramide).
d. Laxatives
- Vegetable oils (250–500 ml) to facilitate passage of indigestible material.
e. Fluid Therapy
- Correct dehydration and electrolyte imbalances if necessary.
8. Prognosis
- Excellent if diagnosed early and treated appropriately.
- Return to normal feeding and productivity typically occurs within 1–3 days.
- Complications are rare unless there's a secondary disease or poor management.
9. Prevention
- Gradual changes in diet.
- Avoid feeding spoiled or mouldy feeds.
- Provide consistent feed quality and feeding schedules.
- Ensure balanced rations with adequate fiber.
- Use feed additives (e.g., yeast cultures) to support rumen function.
- Avoid overfeeding concentrates, especially after a period of feed restriction.
- Lactic (ruminal) acidosis in ruminants
Definitions
Lactic acidosis (ruminal acidosis or grain overload) is a metabolic disorder in ruminants caused by excessive accumulation of lactic acid in the rumen and bloodstream following rapid fermentation of highly fermentable carbohydrates. It leads to a sharp drop in ruminal pH (<5.6) and systemic acidosis
Forms
Acute ruminal lactic acidosis (acute acidosis / grain overload): rapid ingestion of large quantities of starches/ sugars → proliferation of lactate-producing bacteria (e.g., Streptococcus bovis, Lactobacillus spp.), marked lactic acid accumulation, rumen pH commonly <5.0, leading to systemic illness and dehydration.
Subacute ruminal acidosis (SARA): recurrent or prolonged periods of moderately depressed rumen pH (commonly defined by many studies as pH <5.6 for extended periods), often insidious with reduced production, intermittent diarrhea, and usually occur at the herd level.
Etiology & risk factors
- Main cause: sudden or excessive intake of highly fermentable carbohydrates (grains, finely processed concentrates, sudden diet changes).
- Rapid introduction or over-feeding of concentrates (poor adaptation).
- Inadequate effective fiber or long particle length in diet.
- Very fine grinding or pelleting of starch sources.
- High stocking density, feeding management
- Lack of feeding frequency or competition at the trough (animals eat large meals).
- Water restriction (can exacerbate effects).
- Species/breed differences and age (young animals sometimes more susceptible).
Pathophysiology (how lactate accumulates and causes disease)
1. Rapid fermentation of soluble carbohydrates → surge in short-chain volatile fatty acids (VFAs) and especially lactic acid produced by lactate-producing bacteria.
2. Rumen pH falls (below ~5.5 for SARA; often <5.0 in acute cases). Low pH selects for lactate producers and suppresses lactate utilizers (e.g., Megasphaera elsdenii, Selenomonas spp.), generating a positive feedback loop.
3. Osmotic and epithelial injury: lactic acid increases ruminal osmolarity → fluid shifts into rumen, dehydration; acidic damage to rumen mucosa (rumenitis) predisposes to translocation of bacteria (liver abscesses, bacteremia).
4. Systemic effects: dehydration, metabolic acidosis, shock; cerebral effects (e.g., polioencephalomalacia) may follow from thiamine deficiency or rumenitis-related sequelae.
Clinical signs & sequelae
Acute (severe) cases
- Sudden anorexia, weakness, depression, ataxia, bloat or rumen distension, rumen stasis (no borborygmi), dehydration, tachycardia, rapid shallow respiration, recumbency, shock, sudden death in severe cases.
- Rumen fluid: very acidic, watery, often full of bacteria and debris; pH often <5.0.
SARA / subclinical forms
- Reduced feed intake and milk fat depression, loose feces or pasty feces, decreased rumination and cud-chewing, decreased milk yield, reproductive inefficiency, hoof problems (laminitis) and increased risk of liver abscesses. SARA may be largely subclinical at the individual level but causes herd-level economic losses.
Economic impact of lactic acidosis
· SARA is especially insidious at the herd level: reduced milk fat, decreased feed efficiency, increased culling, hoof problems, and greater veterinary costs.
· Acute cases carry higher mortality and treatment costs. Prevention and good feeding practice are typically far more cost-effective than treating outbreaks.
Diagnosis
History & herd context: sudden access to grain, recent diet change, feeding pattern.
Physical exam: dehydration, rumen atony and stasis
Rumen fluid analysis (most direct): rumenocentesis or rumen fluid
collected by stomach tube to assess pH (lower than 505), odour, colour,
motility of protozoa. Acidic pH (<5.0–5.5) is diagnostic when correlated
with history.
Laboratory tests: CBC (hemoconcentration, leukocytosis if secondary
infection), serum electrolytes and blood gas analysis (metabolic acidosis).
Herd-level diagnostic approaches for SARA: rumen pH profiling across
high-risk cows, rumen bolus pH telemetry in research/advanced systems,
Treatment (clinical management)
Immediate (acute) treatment goals
1. Restore circulation and correct dehydration / acid–base disturbances
o Prompt IV fluids (crystalloids) to correct dehydration and improve perfusion.
o Use blood gas/electrolytes to guide therapy.
2. Reduce and neutralize rumen acidity
o Oral/ruminal administration of alkaline buffers (e.g., magnesium hydroxide or sodium bicarbonate in warmed water) administered directly into the rumen to raise pH.
o Reported ruminal magnesium hydroxide dose: e.g., 500 g per 450 kg animal diluted and administered into rumen solutions
3. Rumen evacuation / lavage if large grain overload and severe: rumenotomy may be indicated in severe cases to mechanically remove grain and acids.
4. Transfaunation / microbial restoration: restore rumen microflora using rumen fluid from a healthy donor (transfaunation) or using commercially available inoculants/rumen-adapted probiotics.
5. Antimicrobials / anti-inflammatories: use selectively if there is systemic infection or to control secondary infections; NSAIDs can help control inflammation and endotoxemia. Avoid misuse of antibiotics that further disrupt rumen flora.
6. Supportive care: thiamine (if neurologic signs), rumenotonic agents
7. Treat sequelae (laminitis, liver abscesses)
Prevention & herd control
- Gradual adaptation to high-concentrate rations (stepwise increases over 2–3 weeks).
- Maintain adequate effective fiber (long-stem forage) to stimulate chewing and saliva production (natural buffer).
- Feeding management: frequent feeding, multiple smaller meals, ensure access to feed so animals do not eat large ‘binge’ meals; reduce competition at the bunk.
- Physical form of the diet: avoid excessively fine grinding; include particle size that promotes cud chewing.
- Dietary buffers: inclusion of sodium bicarbonate or other buffers at recommended levels for herds at risk. Studies support strategic buffering to reduce ruminal pH excursions.
- Feed additives: ionophores (where permitted) can reduce risk by modifying fermentation patterns; careful use of yeasts, direct-fed microbials and lactate-utilizing probiotics may reduce incidence — evidence varies and should be used as part of a broader plan.
- Monitoring: routine rumen pH monitoring in high-risk groups, track milk-fat depression and other production indicators; consider rumen pH boluses in research or intensive herds.
Complications
- Complications: laminitis, liver abscesses, rumenitis with bacterial translocation, peritonitis (if rumen wall damaged), polioencephalomalacia, chronic production losses and reproductive problems.
Prognosis:
Depends on severity and promptness of treatment.
· Mild cases (SARA) can be reversed with management changes but cause production losses.
· Severe acute acidosis can be fatal or lead to long-term sequelae despite treatment.
- Tympany (Bloat) in Ruminants
1. Definition
Tympany, commonly referred to as bloat, is a condition in ruminants characterized by excessive accumulation of gas in the rumen and/or reticulum. It results in distension of the left paralumbar fossa and can lead to respiratory distress, decreased cardiac output, and death if untreated.
2. Classification
Tympany is classified into two main types:
A. Primary (Frothy) Bloat
- Caused by ingestion of legumes (e.g., alfalfa, clover), which produce a stable foam that traps fermentation gases in the rumen.
- Foam prevents gas from being eructated (belched).
- Common in pasture-fed cattle.
B. Secondary (Free-Gas) Bloat
- Results from physical or functional obstruction of the esophagus or failure of normal eructation.
- Causes include:
- Esophageal obstruction (e.g., foreign body, tumors)
- Vagal indigestion
- Hypocalcemia
- Lateral recumbency (as during anesthesia)
- Tetanus or pharyngeal paralysis
3. Epidemiology
- Affects cattle more commonly than sheep or goats due to grazing behavior and rumen capacity.
- Young animals and high-producing dairy cows are at higher risk.
- Frothy bloat is more common during spring and early summer with lush (juicy) pasture growth.
4. Etiology and Risk Factors
|
Type |
Cause |
Risk Factors |
|
Primary |
Ingestion of legumes, grains |
High-protein pastures, finely ground feed, rapid dietary change |
|
Secondary |
Esophageal or ruminal dysfunction |
Obstruction, hypocalcemia, TRP, vagus nerve injury, tetanus |
5. Pathophysiology
- Rumen fermentation naturally produces gas (CO₂, CH₄, N₂).
- Normally, gases are eliminated by eructation.
- When eructation is impaired:
- Intraluminal pressure rises → compression of diaphragm → dyspnea.
- Venous return to heart decreases → hypotension, shock.
- Death occurs from asphyxiation or cardiovascular collapse.
In frothy bloat, gas is trapped in a stable foam; in free-gas bloat, gas is not trapped but cannot escape.
6. Clinical Signs
|
Early Signs |
Advanced Signs |
|
|
Abdominal distension (left flank) |
Severe respiratory distress |
|
|
Discomfort, kicking at abdomen |
Open-mouth breathing |
|
|
Decreased appetite, milk yield |
Cyanosis, collapse |
|
|
Ruminal tympany on percussion |
Death (within hours if untreated) |
|
In sheep and goats, signs are similar but may progress more rapidly in lambs/kids.
7. Diagnosis
A. Clinical Diagnosis
- History of sudden onset after pasture change.
- Left-sided abdominal distension ("Papple" shape in advanced cases).
- Ping sounds and fluid/gas on auscultation.
B. Diagnostic Procedures
- Passing a stomach tube:
- If gas escapes freely → free-gas bloat
- If no gas escapes → likely frothy bloat
- Rumen fluid examination:
- Frothy content in primary bloat.
- pH may be reduced in grain-associated bloat.
8. Treatment
A. Emergency Decompression
- Stomach tube: Immediate relief in free-gas bloat.
- If unsuccessful: Trocarization of left paralumbar fossa using trocar and cannula.
B. Antifoaming Agents (Frothy Bloat)
- Poloxalene, mineral oil, vegetable oils (sunflower, soybean): Break down stable foam.
- Administer orally via stomach tube.
C. Other Supportive Treatments
- Calcium borogluconate (if hypocalcemia suspected).
- Broad-spectrum antibiotics (if secondary infection or TRP involved).
- NSAIDs or corticosteroids in severe inflammation.
9. Prevention
A. Pasture Management
- Avoid grazing hungry animals on lush legume pastures.
- Mix grasses with legumes to reduce risk.
- Feed dry hay before pasture turnout.
B. Feed Management
- Avoid finely ground grains.
- Introduce dietary changes gradually.
- Use ionophores (monensin, lasalocid) in feed: reduce foam-producing bacteria.
10. Complications
- Aspiration pneumonia: If animal regurgitates or is in lateral recumbency.
- Shock and death: Rapid onset in severe cases.
- Chronic rumenitis: From prolonged pressure and impaired motility.
11. Prognosis
- Good if treated early, especially in free-gas bloat.
- Poor in cases of chronic obstruction or when treatment is delayed.
- Recurrent cases suggest underlying anatomical or neurological issue.
13. Summary Table
|
Type |
Cause |
Diagnosis |
Treatment |
Prevention |
|
Frothy |
Foam-forming legumes or grains |
No relief via stomach tube |
Antifoaming agents, trocar |
Poloxalene, hay before pasture |
|
Free-Gas |
Obstruction, TRP, vagal issues |
Relief with tube |
Tube, treat cause |
Avoid foreign bodies, prevent TRP |
- Abomasal Displacement in Cattle
1. Definition
Abomasal displacement is a common gastrointestinal disorder in high-producing dairy cattle, particularly during the early postpartum period. It involves the shifting of the abomasum from its normal position on the ventral abdominal floor to an abnormal location either on the left (LDA) or right side (RDA) of the abdomen. Severe cases, particularly right-sided displacement with volvulus (RAV), can be life-threatening and require emergency intervention. Abomasal displacement is a multifactorial condition in dairy cattle, especially around parturition. Early diagnosis, appropriate treatment, and effective preventive strategies are essential to minimize economic losses and improve animal welfare. Integration of nutrition, herd management, and monitoring technologies plays a crucial role in reducing the incidence.
2. Types of Displacement
1. Left Displaced Abomasum (LDA)
- Most common (80–90% of cases)
- Abomasum shifts between the rumen and the left abdominal wall.
2. Right Displaced Abomasum (RDA)
- Less common, but more dangerous
- Abomasum moves to the right side between the liver and the body wall.
3. Right Abomasal Volvulus (RAV)
- A complication of RDA
- Twisting of the abomasum leading to ischemia, necrosis, and shock
3. Epidemiology
- Predominantly affects high-yielding dairy cows.
- Typically occurs within the first 6 weeks post-partum.
- Risk increases in Holstein-Friesians.
- Incidence varies from 0.5% to 5% in herds but may be higher in poorly managed systems.
4. Etiology and Risk Factors
- Negative energy balance in early lactation
- Hypocalcemia
- Ketosis
- High-concentrate, low-fiber diets
- Sudden dietary changes
- Reduced feed intake (anorexia)
- Concurrent diseases:
- Metritis
- Mastitis
- Retained placenta
- Fatty liver syndrome
- Overconditioning or fat cows at calving
- Genetic predisposition (certain breeds are more susceptible)
5. Pathogenesis
1. Decreased rumen fill and reduced motility allow the abomasum to shift.
2. Accumulation of gas in the abomasum leads to floatation.
3. In LDA, the abomasum traps gas and displaces between the rumen and left body wall.
4. In RDA/RAV, it shifts to the right side, and in RAV, torsion obstructs blood flow.
5. Displacement disrupts digesta flow, causing hypochloremia, metabolic alkalosis, and dehydration.
6. Clinical Signs
LDA:
- Gradual decrease in milk yield
- Anorexia (especially grain refusal)
- Mild to moderate dehydration
- "Ping" on auscultation/percussion on the left side (9th–13th rib area)
- Reduced fecal output (scanty pasty feces)
- Dark fecal matter
- Normal or slightly elevated temperature
RDA:
- Similar signs to LDA but on the right side
- More severe systemic signs (dehydration, electrolyte imbalance)
- Risk of progression to volvulus
RAV:
- Acute onset, rapid deterioration
- Severe dehydration and weakness
- Tachycardia (HR > 100 bpm)
- Cool extremities, shock
- Abdominal distension
- No feces or very dark, tarry stool
7. Diagnosis
- Physical Examination:
- Abdominal auscultation + percussion → high-pitched “ping”
- Rectal exam: may detect gas-filled structure
- Laboratory Tests:
- Hypochloremia
- Hypokalemia
- Metabolic alkalosis
- Ketosis
- Ultrasound: Confirms position and displacement
- Laparotomy: Definitive diagnosis and treatment
8. Treatment
LDA and RDA (without volvulus):
- Surgical Correction (gold standard):
- Right paramedian abomasopexy
- Right flank omentopexy
- Left flank abomasopexy (less common)
- Rolling (casting method):
- Only temporary; high recurrence
- Not recommended as sole treatment
- Supportive Therapy:
- IV fluids
- Electrolytes (K+, Cl–, Ca++)
- NSAIDs (e.g., flunixin)
- Oral calcium and propylene glycol (especially in hypocalcemic cows)
RAV (Volvulus):
- Emergency surgery required
- Delay leads to necrosis and death
- Prognosis depends on early recognition and prompt correction
- Post-op care: antibiotics, anti-inflammatories, fluids, calcium, and monitoring
9. Prognosis
- LDA: Good to excellent with timely surgical correction
- RDA: Fair to good if volvulus does not develop
- RAV: Guarded to poor; depends on duration and degree of torsion
10. Prevention
- Optimize transition cow management
- Feed adequate fiber in pre- and post-partum diets
- Minimize stress and disease around calving
- Maintain consistent feed intake and avoid sudden changes
- Prevent hypocalcemia and ketosis:
- Use of oral calcium
- Monitor NEFA and BHBA levels
- Good herd monitoring and early detection of off-feed cows
11. Economic Impact
- Significant losses due to:
- Decreased milk yield
- Treatment and veterinary costs
- Premature culling or death
- Ruminal parakeratosis
Definition
Ruminal parakeratosis is a nutrition-related lesion of the rumen epithelium characterized by excessive keratin accumulation (hyperkeratosis/parakeratosis), thickened/hardened and often clumped papillae, reduced absorptive function and variable sloughing/erosion of the epithelium. It’s most commonly seen when ruminants receive high-concentrate/low-effective-fiber rations or experience prolonged ruminal pH depression (acidosis).
hardening of the rumen's papillae (small, finger-like projections) in ruminants like cattle. This keratinization can hinder VFA absorption, affecting feed efficiency and weight gain. It can also be associated with zinc deficiencies or a diet low in vitamin A.
Pathogenesis (how it happens)
· Excess rapidly fermentable carbohydrate → increased VFA (esp. butyrate) production and repeated/ prolonged ruminal pH depression.
· The altered chemical environment changes epithelial cell proliferation and differentiation: keratin layers accumulate (parakeratosis), cell adhesion is compromised, and papillae become short, thick, clumped or even slough. This impairs short-chain fatty acid (SCFA) absorption and the epithelial barrier.
Risk factors
· Abrupt transition to high-grain rations (feedlot finishing, early concentrate starters in calves).
·Low effective fiber or poor particle size (insufficient rumination).
· Heat-treated or pelleted forages (can predispose), and prolonged ruminal milk accumulation in bottle-fed calves.
· Zinc or vitamin A deficiency
Clinical signs
· The diseases is often subclinical
· Reduced feed efficiency, lower weight gain or milk production.
· In more severe cases: anorexia, rumen dysfunction (hypomotility), paste-like rumen contents, secondary rumenitis or ulcers, and increased risk of systemic effects from translocated bacteria/toxins.
· Gross and histologic lesions (what you’ll see) Gross: papillae thickened, clumped, occasionally short/atrophic; surface may be rough, scaly or show erosions/ulcers. (See macroscopic image in the carousel.)
· Histology: retained nucleated keratin layers (parakeratosis), hyperplasia of keratinocytes, variable spongiosis/vacuolar change, inflammatory cell infiltration and sloughing of the stratum corneum. (See histology images in the carousel.)
Diagnosis
· History of diet (high concentrates, abrupt change).
· Clinical exam (rumen dysfunction, production loss).
· Rumen fluid analysis: low pH supports SARA/acidotic states but pH alone doesn’t prove parakeratosis.
· Rumenoscopy or rumenotomy: direct visualization of papillae changes.
· Biopsy/histopathology of rumen papillae is definitive (shows parakeratosis/hyperkeratosis).
Treatment & control
· Dietary correction is mainstay: increase physically effective fiber, reduce rapidly fermentable carbohydrates, and gradually adapt animals when shifting rations. Restore rumen function and avoid abrupt changes. Merck Veterinary Manual +1
· Buffers/alkalinizing agents (e.g., oral bicarbonate) may be used to correct acute pH drops, but long-term control relies on ration formulation and management. Merck Veterinary Manual
· Feed additives: ionophores (where permitted) and rumen modifiers can reduce SARA risk in some systems — use according to regulations and veterinary guidance. Frontiers
· Severe erosions/ulcers: treat as rumenitis (antibiotics if secondary infection suspected, anti-inflammatories) under veterinary guidance.
· Prognosis Variable. With diet correction, ruminal epithelium can recover but full structural/functional recovery may take weeks to months; chronic thickening or scarring can lead to lasting reductions in absorptive efficiency. Early identification and correction improve outcomes.
· Practical management checklist (for herd use) Evaluate TMR particle size and effective fiber (shredded long hay, straw buffers).
· Review feeding schedule and ensure gradual transitions (step-up grain adaptation).
· Monitor rumen health: rumen pH spot checks, fecal consistency, milk fat depressions or drop in gain.
· Consider ration reformulation with a ruminant nutritionist; use ionophores only if appropriate.
· If individual animals show signs, perform rumen fluid analysis and consider rumenoscopy/biopsy.
PM lesions
Macroscopic rumen lesions / thickened papillae — from a slaughterhouse study.
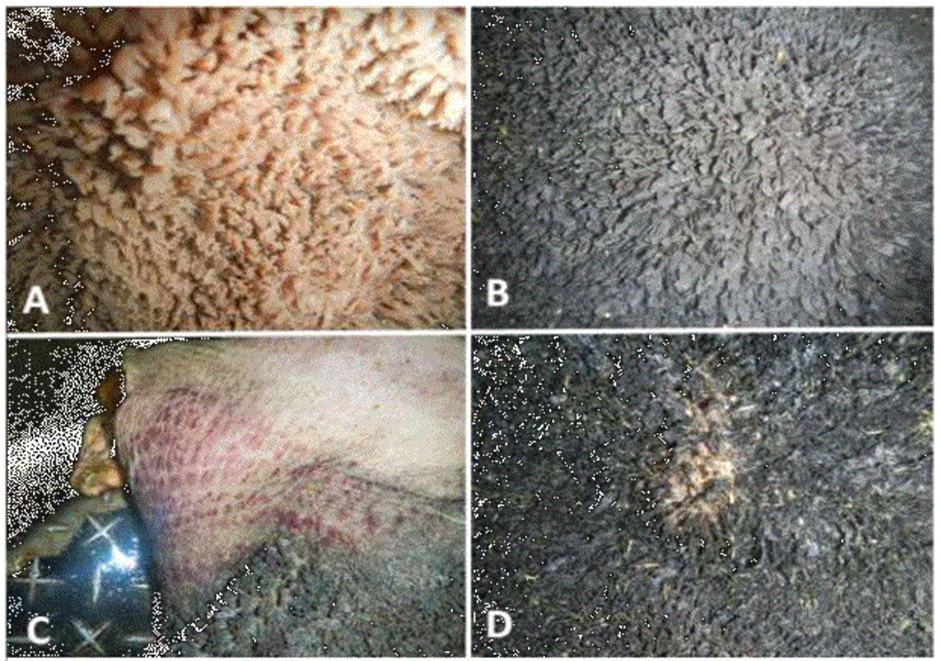
Pm lesion of ruminal parakeratosis. A/ normal rumen mucosa. B/ thickening and atrophy of the rumen papillae. C/ ruminal hemorrhages. D/E erosions, ulcers and ruminal scars (LUNA-MENDEZ et al., 2020)
- Ruminal drinking (reticuloruminal milk accumulation) in young ruminants
Definition
Ruminal drinking (reticuloruminal milk accumulation) is a disorder of neonatal/pre-weaned calves in which milk fails to bypass the forestomach due to failure/insufficiency of the esophageal/reticular groove reflex and is deposited directly into the rumen/reticulum instead of the abomasum. This leads to ruminal fermentation, producing lactic acid and other volatile fatty acids that lower the rumen's pH, resulting in ruminal acidosis, inflammation, poor growth, diarrhea, and even death.
Epidemiology & importance
- Most commonly seen in artificially-fed dairy calves, but reported in lambs, kids, and other young ruminants.
- Can present as an acute, life-threatening problem (bloat, toxemia) or as chronic underperformance (poor growth, recurrent scours, hair loss).
- It is an important cause of mortality and reduced productivity in calf-rearing systems.
Predisposing factors / risk situations
- Failure of the esophageal groove reflex secondary to illness (neonatal diarrhea, systemic disease, weakness).
- Management factors: bucket feeding (open bucket), inappropriate teat design (wide holes, worn teats), feeding at wrong height, feeding very cold milk, irregular feeding schedules, over-feeding (exceed abomasal capacity), poor milk replacer quality or wrong concentration, force-feeding or tube feeding without care. Stress and pain also predispose.
Pathogenesis
1. Milk enters rumen instead of abomasum → substrate for ruminal microbes.
2. Microbial fermentation of milk produces lactic acid and VFA → ruminal pH falls (ruminal acidosis).
3. Acid injury to ruminal epithelium → rumenitis, ulceration; altered motility and impaired gastric development.
4. Clinical consequences: recurrent bloat (gas accumulation), diarrhoea (malabsorption plus systemic effects), dehydration, d-lactic encephalopathy in severe cases, secondary bacterial/fungal infections, and chronic ruminal scarring that impairs future function.
Clinical signs
- Acute form: abdominal distension (left flank/bulging), discomfort, dyspnea if severe bloat, weakness, inappetence, depression, sometimes sudden death.
- Subacute/chronic form: poor weight gain, rough hair coat, intermittent scours (often greyish), recurrent ruminal bloat after milk feeding, decreased suck reflex, poor body condition, patchy alopecia and reduced coordination. On auscultation you may hear fluid splashing. Palpation/percussion shows rumen distension.
Clinicopathologic/diagnostic tests
- History & observation: bucket feeding, irregular feeding, diarrhoea, weak neonate.
- Rumen fluid analysis (when obtainable): low pH (<5.5–6 depending on stage), increased lactic acid, abnormal smell/appearance consistent with fermented milk.
- Blood analysis: metabolic acidosis (in severe cases), electrolyte derangements, dehydration.
- Response to therapy and necropsy findings (ulcerative rumenitis, parakeratosis, mucosal necrosis) support diagnosis.
- Imaging rarely required; in severe bloat, emergency decompression is clinical.
Differential diagnoses
- Primary bloat,
- choke/obstruction,
- neonatal septicemia,
- parasitic or viral enteritis causing secondary dysfunction of the groove,
- congenital defects of swallowing,
- pneumonia (if respiratory signs predominant).
Treatment
Emergency (severe bloat/respiratory compromise):
1. Immediate decompression: pass orogastric tube (if gas/fluid can be released) or trocar/cannula left flank in life-threatening free-gas bloat
2. Oxygen and supportive care if dyspneic.
Medical & supportive therapy:
1. Stop milk feeding temporarily. Provide appropriate oral electrolytes if calf able to suck; otherwise IV fluids. Correction of systemic dehydration and metabolic acidosis is often required — isotonic crystalloids plus bicarbonate when indicated (calculate base deficit clinically).
2. Rumen lavage (orogastric lavage) in selected cases to remove fermenting milk; some clinicians perform rumen lavage with warm isotonic fluid in severe, accessible cases.
3. Oral antacids/rumen buffers are used by some clinicians (evidence variable).
4. Antibiotics when secondary bacterial infection or aspiration pneumonia suspected — choose based on local protocols. Topical antifungal/systemic antifungals if secondary fungal overgrowth documented.
5. Nutritional support and nursing care; gradually reintroduce milk correctly.
6. Severe, chronic lesions with persistent failure may require euthanasia if prognosis poor.
Prognosis
- Acute severe cases with marked acidosis/bloat may die despite therapy.
- Early identified, mild–moderate rumen drinking with appropriate supportive therapy and management correction has fair to good prognosis.
- Chronic rumenitis with scarring leads to lifelong poor performance and guarded prognosis.
- Prognosis worsens if underlying systemic disease (severe diarrhea, septicemia) is present.
Prevention & control
- Feed milk from teats/nipples (not open buckets); ensure teat hole size controls drinking speed. Teat should be at nose height.
- Feed milk at physiological temperature (~38–40 °C). Avoid very cold milk.
- Avoid over-feeding; divide daily allowance into appropriate feedings to avoid overflow of abomasal capacity.
- Avoid force-feeding or inappropriate tube feeding techniques that bypass normal triggers for groove closure. If tube-feeding, use correct technique and volume limits.
- Correct and treat primary diseases rapidly (diarrhea, sepsis) so weak neonates will suck and trigger the groove reflex normally.
- Use quality milk replacer at correct concentration; avoid spoiled/waste milk unless pasteurized and of known quality.
- Consistent feeding schedule and reduced stress. Training calves to nurse properly on teat feeders rather than buckets reduces incidence.
Some practical points to be considered at the farm
- Switch bucket-fed calves to teat feeders where possible.
- Check teat hole size / replace worn teats.
- Feed at body temperature and at consistent times.
- Monitor for scours/weak calves and treat early.
- If bloat observed after milk, suspect rumen drinking — act quickly to decompress and seek veterinary care.
- Secondary indigestion
Secondary indigestion due to infectious diseases in cattle occurs when an underlying infectious condition disrupts normal rumen function. In such cases, the infectious disease doesn’t directly target the rumen, but its systemic effects (like fever, toxemia, or inflammation) cause rumen stasis or hypomotility, leading to indigestion.
Common Infectious Diseases Causing Secondary Indigestion in Cattle:
|
Infectious Disease |
Mechanism Leading to Secondary Indigestion |
Associated Signs |
|
Traumatic reticuloperitonitis (Hardware disease) |
Bacterial infection of peritoneum causes inflammation, pain, and rumen stasis |
Fever, grunt on pressure, arched back |
|
Peritonitis (bacterial origin) |
Inflammation of the abdominal cavity disrupts ruminal motility |
Tense abdomen, reduced rumen contractions |
|
Liver abscesses (e.g., Fusobacterium necrophorum) |
Toxins and pain suppress appetite and rumen activity |
Weight loss, reduced milk yield, fever |
|
Septic metritis (uterine infection post-partum) |
Systemic infection leads to toxemia and ruminal hypomotility |
Foul uterine discharge, fever, dullness |
|
Severe mastitis (e.g., coliform mastitis) |
Endotoxemia depresses rumen motility |
Swollen udder, hot quarters, systemic signs |
|
Pneumonia (e.g., Mannheimia haemolytica, Pasteurella multocida) |
Generalized infection leads to poor appetite and decreased rumen activity |
Coughing, fever, nasal discharge |
|
Enterotoxemia (Clostridium perfringens) |
Toxins affect nervous system and GI motility |
Sudden death, bloating, diarrhea |
|
Johne’s Disease (Mycobacterium avium subsp. paratuberculosis) |
Chronic infection leads to gradual rumen dysfunction |
Chronic diarrhea, weight loss |
|
Salmonellosis |
Systemic effects and enteritis reduce digestion and rumen activity |
Fever, diarrhea, dehydration |
Pathophysiology:
- Fever and toxemia → depress appetite and microbial fermentation
- Inflammation or pain → reduces vagal tone, causing rumen stasis
- Endotoxins → affect smooth muscle motility and microbial flora
- Anorexia → leads to poor nutrient input and worsens rumen function
Clinical Signs of Secondary Indigestion:
- Fever
- Decreased appetite or anorexia
- Reduced rumination
- Dullness or depression
- Mild to moderate rumen distension
- Decreased or absent rumen contractions
- Dry, firm feces or diarrhea (depending on disease)
- Signs of primary disease (e.g., pain, fever, signs of peritonitis)
Diagnosis:
- Identification of the primary infectious disease
- Rumen fluid analysis (alkaline pH, reduced protozoa)
- Clinical examination and history
- Blood tests (elevated WBCs, inflammatory markers)
Treatment:
1. Address the infectious cause:
- Appropriate antibiotics or anti-infectives
- Anti-inflammatory and supportive therapy
2. Restore rumen function:
- Rumenotorics (e.g., neostigmine, metoclopramide)
- Transfaunation (healthy rumen fluid)
- Vitamin B complex, fluids, and electrolytes
Prognosis:
- Depends on the severity of the primary disease and the duration of rumen stasis.
- Early detection and treatment improve outcomes.
- References
· Allen MS. Relationship between fermentation acid production in the rumen and the requirement for physically effective fiber. Journal of Dairy Science. 1997;80(7):1447–1462.
· Andrews, A. H., Blowey, R. W., Boyd, H., Eddy, R. G. Bovine Medicine.
· Aschenbach JR, Penner GB, Stumpff F, Gabel G. Ruminant nutrition symposium: Role of fermentation acid absorption in the regulation of ruminal pH. Journal of Animal Science. 2011;89(4):1092–1107.
· Bevans DW, Beauchemin KA, Schwartzkopf-Genswein KS, McKinnon JJ, McAllister TA. Effect of rapid or gradual grain adaptation on subacute acidosis and feed intake by feedlot cattle. Journal of Animal Science. 2005;83(5):1116–1132.
· Constable, P. D., Hinchcliff, K. W., Done, S. H., & Grünberg, W. (2017). Veterinary Medicine: A textbook of the diseases of cattle, horses, sheep, pigs, and goats (11th ed.). Elsevier.
· Duffield TF, Plaizier JC, Fairfield A, et al. A meta-analysis of the impact of monensin in lactating dairy cattle. Journal of Dairy Science. 2008;91(4):1334–1346.
· Enemark JMD. The monitoring, prevention and treatment of sub-acute ruminal acidosis (SARA): A review. Veterinary Journal. 2008;176(1):32–43.
· Fenta M, Li F, Wang H, Jin G, Jin L. Probiotics in ruminants and their potential roles in prevention and treatment of ruminal acidosis. Frontiers in Veterinary Science. 2023;10:1282031.
· Geishauser, T. et al. (2000). Diagnosis of left displaced abomasum in dairy cows. Journal of Dairy Science.
· Gerardy-Schahn, R., Wooding, F. B. P., & Leipold, H. W. (1989). Dysfunction of the esophageal groove (“ruminal drinking”) in calves: Clinical and pathological findings. Journal of Veterinary Medicine Series A, 36(1), 1–14. https://doi.org/10.1111/j.1439-0442.1989.tb00669.x
· Goff, J. P., and Horst, R. L. (1997). Physiological changes at parturition and their relation to metabolic disorders. Compendium on Continuing Education for the Practicing Veterinarian.
· Golder HM. Ruminal acidosis—A review of ruminal pH, rumen microbiome, and microbial activities. Frontiers in Veterinary Science. 2024;11:1384699.
· Kaba, J., Nowicki, M., Nowicki, S., & Anusz, K. (2018). Esophageal groove dysfunction: A cause of ruminal bloat in newborn calves. BMC Veterinary Research, 14(1), 63. https://doi.org/10.1186/s12917-018-1374-3
· Khafipour E, Krause DO, Plaizier JC. A grain-based subacute ruminal acidosis challenge causes translocation of lipopolysaccharide and triggers inflammation. Journal of Dairy Science. 2009;92(3):1060–1070.
· LUNA-MENDEZ, Alejandra et al. Ruminal lesions in cattle slaughtered in slaughterhouses. Abanico vet [online]. 2020, vol.10, e105. Epub 02-Mar-2021. ISSN 2448-6132. https://doi.org/10.21929/abavet2020.9.
· Merck Veterinary Manual. (2023). Reticuloruminal milk accumulation (“ruminal drinking”) in calves. Merck Sharp & Dohme Corp. Retrieved from https://www.merckvetmanual.com
· Merck Veterinary Manual. Grain Overload (Acute Ruminal Acidosis) in Ruminants. In: The Merck Veterinary Manual. Merck & Co., Inc.; Updated 2024.
· Nagaraja TG, Lechtenberg KF. Acidosis in feedlot cattle. Veterinary Clinics of North America: Food Animal Practice. 2007;23(2):333–350.
· Nocek JE. Bovine acidosis: Implications on laminitis. Journal of Dairy Science. 1997;80(5):1005–1028.
· Oetzel GR. Monitoring and testing dairy herds for metabolic disease. Veterinary Clinics of North America: Food Animal Practice. 2004;20(3):651–674.
· Orós J., Pathological Changes of the Rumen in Small Ruminants Associated with Indigestible Foreign Objects. MDPI; histology figures and discussion of hyperkeratosis/parakeratosis changes.
· Owens FN, Secrist DS, Hill WJ, Gill DR. Acidosis in cattle: A review. Journal of Animal Science. 1998;76(1):275–286.
· Plaizier JC, Krause DO, Gozho GN, McBride BW. Subacute ruminal acidosis in dairy cows: The physiological causes, incidence, and consequences. The Veterinary Journal. 2008;176(1):21–31.
· Smith, B.P. Large Animal Internal Medicine, 6th Edition.
· Smith, G. W. (2009). Treatment of calf diarrhea: Oral fluid therapy. Veterinary Clinics of North America: Food Animal Practice, 25(1), 55–72. https://doi.org/10.1016/j.cvfa.2008.10.007
· Steele MA, et al. Bovine rumen epithelium undergoes rapid structural adaptations... Am J Physiol Regul Integr Comp Physiol. 2010. (review of epithelial adaptations, parakeratosis and SCFA handling). Physiology Journals
· Steele, M.A., AlZahal, O., Hook, S.E. et al. Ruminal acidosis and the rapid onset of ruminal parakeratosis in a mature dairy cow: a case report. Acta Vet Scand 51, 39 (2009). https://doi.org/10.1186/1751-0147-51-39
· Zebeli Q, Metzler-Zebeli BU. Interactions between rumen digestion and health in dairy cows fed high-grain diets. Animal Feed Science and Technology. 2012;173(1–2):22–33.