the Diagnosis and Management of Thyroid Nodules
| الموقع: | EHC | Egyptian Health Council |
| المقرر الدراسي: | جراحه عامه |
| كتاب: | the Diagnosis and Management of Thyroid Nodules |
| طبع بواسطة: | Guest user |
| التاريخ: | الأربعاء، 6 مايو 2026، 3:45 AM |
الوصف
"last update: 8 December 2025" Download Guideline
- Executive summary
These guidelines offer evidence-based recommendations on the targeted levels of clinical practice guidelines and provide healthcare professionals with practice guidance on the surgical management of thyroid nodules and its complications.
· Initial evaluation should include age, personal and family history of MEA, Previous head or neck irradiation, physical evaluation, thyroid function testing, and neck US, (Strong recommendation).
· Neck US, including the thyroid gland and the central and lateral cervical compartments, should be performed in all patients suspected with nodular thyroid disease, (Strong recommendation).
· It is recommended for the US report to describe nodule(s) size, location, US features, and expected risk of malignancy using EU-TIRADS, (Strong recommendation).
· Doppler imaging, elasto-sonography, and CEUS may be recommended as ancillary techniques (Conditional recommendation).
· CEUS may be recommended for defining the size and boundaries of the ablated area after minimally invasive procedures, (Conditional recommendation).
· Thyroid scintigraphy is recommended when serum TSH is subnormal to diagnose functioning nodules and/or multinodularity, avoid FNA and determine eligibility for RAI as an alternative to surgery, (Strong recommendation).
· The use of CT and/or MRI in the study of thyroid nodules should be limited to the assessment of local extension or retrosternal growth of nodular goiter, (Strong recommendation).
· FNA may be recommended after clinical assessment, laboratory evaluation, and US risk stratification, in a shared decision with the patient, (Good practice statement).
· FNA is advised in:
- EU-TIRADS 3: >20 mm, (Conditional recommendation).
· FNA is recommended in:
- EU-TIRADS 4: >15 mm.
- EU-TIRADS 5: >10 mm, (Strong recommendation).
· In high suspicion pattern, we recommend FNA if ≥10 mm, (Strong recommendation).
· In intermediate suspicion pattern, we recommend FNA if ≥10 mm, (Strong recommendation).
· In low suspicion pattern, we advise FNA if ≥15 mm, (Conditional recommendation).
· In very low suspicion pattern, we advise FNA if ≥20 mm (or ultrasound observation), (Conditional recommendation).
· In benign pattern, we recommend against FNA, (Strong recommendation).
· We recommend FNA regardless of lesion size when patients have a history of neck irradiation, a family history of medullary thyroid cancer or MEN2, extracapsular growth, metastatic cervical lymph nodes or Coexistent suspicious clinical findings (e.g., dysphonia), (Strong recommendation).
· Repeat FNA is recommended in case of a first non-diagnostic sample, (except in case of a solitary cyst), a Bethesda class III cytology, discrepancy between US risk score (i.e. high risk) and cytological findings (i.e. benign cytology), and significant nodule growth (an increase ≥20% in at least two nodule diameters with a minimum increase of 2 mm at the time of re-evaluation) of thyroid nodule(s), (Strong recommendation).
· FNA is recommended in suspicious lymph nodes, with thyroglobulin or calcitonin washout dependent on phenotype, (Strong recommendation).
· Core-needle biopsy is not recommended as a first-line tool to assess thyroid nodules after US but could be considered a second line procedure for specific conditions e.g. repeat Bethesda class III cytology and suspicion of poorly differentiated thyroid cancer, thyroid lymphoma, thyroid metastases), (Strong recommendation).
· We recommend correlation of the cytological diagnosis with clinical, ultrasound and laboratory results, (Good practice statement).
· For Bethesda I (non-diagnostic), repeat FNA is advised. If repeat FNA is still non-diagnostic, consider CNB. If still non-diagnostic, consider surgery (Conditional recommendation).
· For Bethesda II, (Benign), we are against further immediate diagnostic studies or treatment, (Strong recommendation).
· For Bethesda III, (Atypia of Undetermined Significance, (AUS)) we recommend repeating FNA and with repeat Bethesda III, consider molecular testing if available or offer surgery, (Conditional recommendation).
· For Bethesda IV, (Follicular Neoplasm), we advise molecular testing or diagnostic lobectomy, (Conditional recommendation).
· For Bethesda V, (Suspicious for malignancy), we recommend molecular testing, lobectomy, or near-total thyroidectomy, (molecular testing may help to decide whether to perform a total thyroidectomy or a thyroid lobectomy), (Strong recommendation).
· For Bethesda VI, (Malignant), we recommend lobectomy or near-total thyroidectomy, (Strong recommendation).
• We recommend multidisciplinary workup in case of advanced cancer, (Strong recommendation).
• Molecular testing is recommended in cytologically indeterminate nodules, if available, or offer surgery (Strong recommendation).
• Thyroid hormone treatment is not recommended in euthyroid individuals with nodular thyroid disease, (Strong recommendation).
• Iodine and/or selenium supplementation is not recommended unless individuals are deficient in these micronutrients, (Strong recommendation).
• RAI is recommended as an alternative to surgery and MIT in hyper-functioning solitary thyroid nodules, (Strong recommendation).
• RAI may be recommended as an alternative to surgery in benign normo-functioning multinodular goiter, (Conditional recommendation).
• EA is recommended for pure, or dominantly cystic, thyroid lesions, (Strong recommendation).
• TA is recommended for the treatment of solid benign thyroid nodules that cause local symptoms as an alternative to surgery and for cystic lesions that relapse after EA, (Strong recommendation).
• Benign cytological diagnosis is recommended before MIT. Except for EU-TIRADS 2 nodules, (Strong recommendation).
• After MIT, we recommend follow-up with clinical, biochemical and US assessments after 6 and 12 months and re-evaluating the patient after 3–5 years, (Strong recommendation).
• Surgery is recommended in the following scenarios: Symptomatic nodular thyroid disease, nodules that have been classified as benign at cytology and/or US and become symptomatic over time, calcitonin levels higher than the established cut-offs, responsive calcitonin after stimulation test in RET-mutated gene carriers, nodules with indeterminate cytology (Bethesda class III and IV) that are not suitable for active surveillance and nodules with a Bethesda class V and VI cytology, (Strong recommendation).
- Recommendations
Section 1: Initial evaluation of thyroid nodules:
· Initial evaluation should include age, personal and family history of MEA, Previous head or neck irradiation, physical evaluation, thyroid function testing, and neck US, (Strong recommendation, moderate certainty evidence (7)).
Section 2: Thyroid ultrasound:
· Neck US, including the thyroid gland and the central and lateral cervical compartments, should be performed in all patients suspected with nodular thyroid disease, (Strong recommendation, moderate certainty evidence (8)).
· It is recommended for the US report to describe nodule(s) size, location, US features, and expected risk of malignancy using EU-TIRADS, (Strong recommendation, moderate certainty evidence (8)), (table 3).
· Doppler imaging, elasto-sonography, and CEUS may be recommended as ancillary techniques (Conditional recommendation, very low certainty evidence (8)).
· CEUS may be recommended for defining the size and boundaries of the ablated area after minimally invasive procedures, (Conditional recommendation, low certainty evidence (8)).
Section 3: Non-ultrasound imaging modalities:
· Thyroid scintigraphy is recommended when serum TSH is subnormal to diagnose functioning nodules and/or multinodularity, avoid FNA and determine eligibility for RAI as an alternative to surgery, (Strong recommendation, moderate certainty evidence (3)).
· The use of CT and/or MRI in the study of thyroid nodules should be limited to the assessment of local extension or retrosternal growth of nodular goiter, (Strong recommendation, low certainty evidence (3)).
Section 4: Thyroid biopsy:
· FNA may be recommended after clinical assessment, laboratory evaluation, and US risk stratification, in a shared decision with the patient, (Good practice statement).
· FNA is advised in:
o EU-TIRADS 3: >20 mm, (Conditional recommendation, very low certainty evidence (8)).
· FNA is recommended in:
o EU-TIRADS 4: >15 mm.
o EU-TIRADS 5: >10 mm, (Strong recommendation, very low certainty evidence (8)).
· In high suspicion pattern, we recommend FNA if ≥10 mm, (Strong recommendation, moderate certainty evidence (3)), (Table 4).
· In intermediate suspicion pattern, we recommend FNA if ≥10 mm, (Strong recommendation, low certainty evidence (3)), (Table 4).
· In low suspicion pattern, we advise FNA if ≥15 mm, (Conditional recommendation, low certainty evidence (3)), (Table 4).
· In very low suspicion pattern, we advise FNA if ≥20 mm (or ultrasound observation), (Conditional recommendation, moderate certainty evidence (3)), (Table 4).
· In benign pattern, we recommend against FNA, (Strong recommendation, moderate certainty evidence (3)), (Table 4).
· We recommend FNA regardless of lesion size when patients have a history of neck irradiation, a family history of medullary thyroid cancer or MEN2, extracapsular growth, metastatic cervical lymph nodes or Coexistent suspicious clinical findings (e.g., dysphonia), (Strong recommendation, moderate certainty evidence (7)).
· Repeat FNA is recommended in case of a first non-diagnostic sample, (except in case of a solitary cyst), a Bethesda class III cytology, discrepancy between US risk score (i.e. high risk) and cytological findings (i.e. benign cytology), and significant nodule growth (an increase ≥20% in at least two nodule diameters with a minimum increase of 2 mm at the time of re-evaluation) of thyroid nodule(s), (Strong recommendation, moderate certainty evidence (8)).
· FNA is recommended in suspicious lymph nodes, with thyroglobulin or calcitonin washout dependent on phenotype, (Strong recommendation, moderate certainty evidence (8)).
• Core-needle biopsy is not recommended as a first-line tool to assess thyroid nodules after US but could be considered a second line procedure for specific conditions e.g. repeat Bethesda class III cytology and suspicion of poorly differentiated thyroid cancer, thyroid lymphoma, thyroid metastases), (Strong recommendation, low certainty evidence (8)).
Section 5: Cytopathology-based management, (Based on Bethesda Categories), (Table 5):
· We recommend correlation of the cytological diagnosis with clinical, ultrasound and laboratory results, (Good practice statement).
· For Bethesda I (non-diagnostic), repeat FNA is advised. If repeat FNA is still non-diagnostic, consider CNB. If still non-diagnostic, consider surgery (Conditional recommendation, very low level of evidence, 8).
· For Bethesda II, (Benign), we are against further immediate diagnostic studies or treatment, (Strong recommendation, high certainty evidence (3)).
· For Bethesda III, (Atypia of Undetermined Significance, (AUS)) we recommend repeating FNA and with repeat Bethesda III, consider molecular testing if available or offer surgery, (Conditional recommendation, high certainty evidence (8)).
· For Bethesda IV, (Follicular Neoplasm), we advise molecular testing or diagnostic lobectomy, (Conditional recommendation, low certainty evidence (8)).
· For Bethesda V, (Suspicious for malignancy), we recommend molecular testing, lobectomy, or near-total thyroidectomy, (molecular testing may help to decide whether to perform a total thyroidectomy or a thyroid lobectomy), (Strong recommendation, low certainty evidence (3)).
· For Bethesda VI, (Malignant), we recommend lobectomy or near-total thyroidectomy, (Strong recommendation, moderate certainty evidence (3)).
· We recommend multidisciplinary workup in case of advanced cancer, (Strong recommendation, moderate certainty evidence (8)).
Section 7: Molecular diagnostics of indeterminate thyroid nodule cytology:
· Molecular testing is recommended in cytologically indeterminate nodules, if available, or offer surgery (Strong recommendation, moderate certainty evidence (8)).
Section 8: Non-surgical approaches of thyroid nodules:
· Thyroid hormone treatment is not recommended in euthyroid individuals with nodular thyroid disease, (Strong recommendation, moderate certainty evidence (8)).
· Iodine and/or selenium supplementation is not recommended unless individuals are deficient in these micronutrients, (Strong recommendation, low certainty evidence (8)).
· RAI is recommended as an alternative to surgery and MIT in hyper-functioning solitary thyroid nodules, (Strong recommendation, moderate certainty evidence (8)).
· RAI may be recommended as an alternative to surgery in benign normo-functioning multinodular goiter, (Conditional recommendation, low certainty evidence (8)).
· EA is recommended for pure, or dominantly cystic, thyroid lesions, (Strong recommendation, moderate certainty evidence (8)).
· TA is recommended for the treatment of solid benign thyroid nodules that cause local symptoms as an alternative to surgery and for cystic lesions that relapse after EA, (Strong recommendation, low certainty evidence (8)).
· Benign cytological diagnosis is recommended before MIT. Except for EU-TIRADS 2 nodules, (Strong recommendation, low certainty evidence (8)).
· After MIT, we recommend follow-up with clinical, biochemical and US assessments after 6 and 12 months and re-evaluating the patient after 3–5 years, (Strong recommendation, low certainty evidence (8)).
Section 9: Surgical approaches of thyroid nodules:
· Surgery is recommended in the following scenarios: Symptomatic nodular thyroid disease, nodules that have been classified as benign at cytology and/or US and become symptomatic over time, calcitonin levels higher than the established cut-offs, responsive calcitonin after stimulation test in RET-mutated gene carriers, nodules with indeterminate cytology (Bethesda class III and IV) that are not suitable for active surveillance and nodules with a Bethesda class V and VI cytology, (Strong recommendation, moderate certainty evidence (8)).
- Acknowledgement
We would like to acknowledge the Guideline Development Group, (GDG) committee for developing these guidelines.
Chair of GDG: Mostafa Abdel-Hamed Soliman, Professor of Surgery, Cairo University.
Moderator of GDG: Mohamed Ali Mohamed Nada, Professor of Surgery, Ain Shams University.
Members of GDG (Alphabetically):
Abdel-Wahab Mohamed Ezzat, Professor of Surgery, Ain Shams University.
Ahmed Abdel-Raouf Elgeidie, Professor of Digestive Surgery, Mansoura University.
Alaa Abdallah, Professor of Surgery, Ain Shams University.
Atef Abdel-Ghani Salem, Professor of Surgery, Benha University.
Hesham Abdel-Raouf El-Akkad, Professor of Surgery, Ain Shams University.
Ibrahim El-Zayat, Head of Surgery Department, Aswan University.
Khaled Abdallah El-Fiky, Professor of Surgery, Ain Shams University.
Khaled Amer, Professor of Surgery, Military Medical Academy.
Khaled Safwat, Professor of Surgical Oncology and Endoscopy, Zagazig University.
Mohamed Ibrahim Abdel-Hamed Al-Said, Professor of Surgery, Zagazig University.
Tarek Ibrahim, Professor of Surgery, National Liver Institute, Menofia University.- Abbreviations
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation II |
|
AUS |
Atypia of Undetermined Significance. |
|
CEUS |
Contrast-Enhanced Ultrasound. |
|
CNB |
Core-Needle Biopsy. |
|
CT |
Computed Tomography. |
|
EA |
Ethanol Ablation. |
|
FNA |
Fine Needle Aspiration. |
|
FNAB |
Fine Needle Aspiration Biopsy. |
|
GDG |
Guideline Development Group. |
|
GRADE |
Grade of Recommendations, Assessment, Development and Evaluation. |
|
MEA |
Multiple Endocrine Anaplasia. |
|
MEN2 |
Multiple Endocrine Neoplasia 2 |
|
MIT |
Minimally Invasive Techniques. |
|
MRI |
Magnetic Resonance Imaging |
|
PRO |
Patient-Reported Outcome. |
|
RAI |
Radioactive Iodine. |
|
ROM |
Risk of Malignancy |
|
RSS |
Risk Stratification System. |
|
TA |
Thermal Ablation. |
|
TBSRTC |
Bethesda System for Reporting Thyroid Cytopathology. |
|
EU-TIRADS |
European Thyroid Imaging and Reporting Data System. |
|
US |
Ultrasound. |
|
TSH |
Thyroid-Stimulating Hormone. |
- Introduction
Thyroid nodules are a common clinical problem. Epidemiologic studies have shown that the prevalence of palpable thyroid nodules to be approximately 5% in women and 1% in men living in iodine-sufficient parts of the world (1). In contrast, high-resolution ultrasound (US) can detect thyroid nodules in 19%–68% of randomly selected individuals, with higher frequencies in women and the elderly (2). Thyroid cancer occurs in 4%–15% of cases depending on age, sex, radiation exposure history, family history, and other factors (3-4).
Ultrasonography (US) is the primary imaging modality for evaluating thyroid nodules. The US risk stratification system (RSS) and the Thyroid Imaging Reporting and Data System (TIRADS) can help in estimating malignancy risk and guide fine-needle aspiration biopsy decisions (5).
Once FNAB has been obtained, the results can be classified according to the Bethesda System for Reporting Thyroid Cytopathology (TBSRTC). The third edition of TBSRTC, was published in 2023. Assigned single name for each of the 6 diagnostic categories: (i) nondiagnostic; (ii) benign; (iii) atypia of undetermined significance; (iv) follicular neoplasm; (v) suspicious for malignancy; and (vi) malignant, also it offers an average ROM for each category (6).
- Scope and Purpose
The scope of these guidelines is to establish a national guideline for patients with thyroid nodules as regard the need for surgery, timing, and type of surgery to achieve a best outcome as much as we can.
The main purposes of these guidelines are to minimize malpractice and poor surgical decision, to improve the quality of medical care and surgical service, to provide the best clinical practice to
our patients, and finally to reduce the cost of medical service.
- Target Audience
The principle targeted audiences are the practicing surgeons, radiologists and cytopathology specialists.
- Methods
A comprehensive search for guidelines was undertaken to identify the relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2015 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in:
• Developing organization/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer, 2016; American college of endocrinology, and association medici endocrinologi medical guidelines for clinical practice for the diagnosis and management of thyroid nodules, 2016 and European Thyroid Association Clinical Practice Guidelines for thyroid nodule management, 2023.
- Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
· GRADE working group: https://www.gradeworkinggroup.org
· GRADE online training modules: http://cebgrade.mcmaster.ca/
Specifically, the quality of evidence was graded as ‘High’, ‘Moderate’, ‘Low’ or ‘Very low’, (table 1& 2).
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
these are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research Needs
1. Evaluation of MITs as out-patient procedures in management of nodular goiter.
2. FNA versus CNB in assessment of thyroid nodules.
3. Molecular testing in cytologically indeterminate thyroid nodules.
- Clinical Indicators of Monitoring
1. TSH, T3, T4 testing.
2. US of the neck and TIRADS class.
3. FNA or CNB.
4. Bethesda classification.
5. Documentation of the indication of surgery in thyroid disease.
6. Documentation of the type of operation.
- Updating of the Guideline
The GDG committee for guidelines development is responsible for the continuous evaluation of evidence available about thyroid disease. The present guidelines will be updated in case of significant changes based on new evidence.
- Annexes
Table 1. Quality and Significance of the four levels of evidence in GRADE
|
Quality |
Current definition |
Previous definition |
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect |
Further research is very unlikely to change our confidence in the estimate of effect |
|
Moderate |
We are moderately confident in the effect estimate: The true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different |
Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate |
|
Low |
Our confidence in the effect estimate is limited: The true effect may be substantially different from the estimate of the effect |
Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate |
|
Very low |
We have very little confidence in the effect estimate: The true effect is likely to be substantially different from the estimate of effect |
Any estimate of effect is very uncertain |
Table 2. Factors that determine How to upgrade or downgrade the quality of evidence
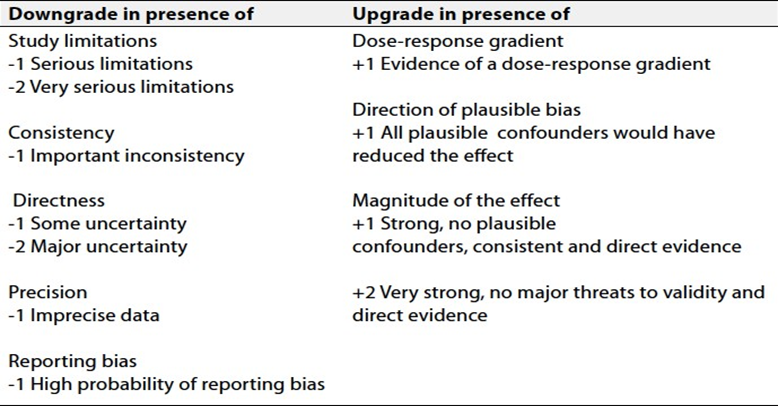
Table 3: EU-TIRADS categories with corresponding malignancy risks and indication of FNAC(8)
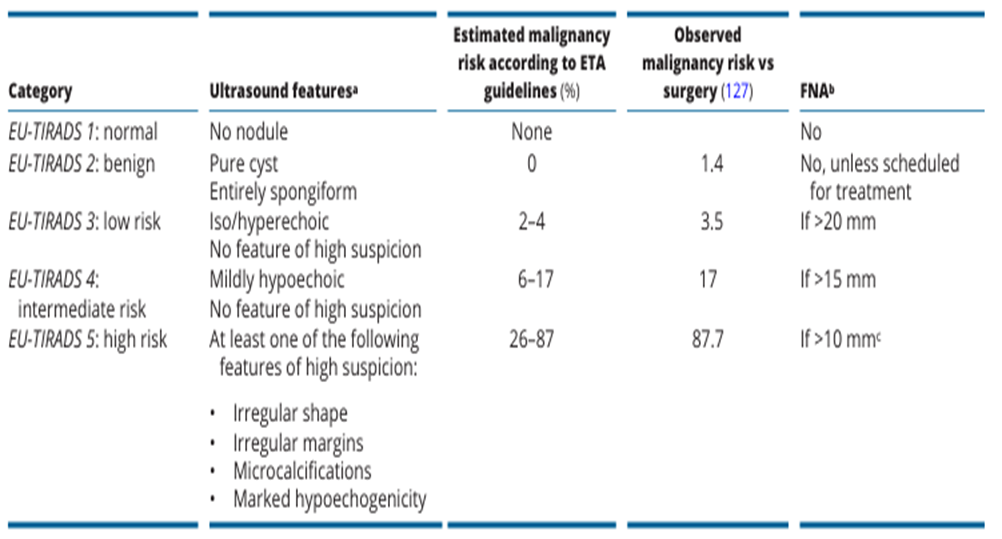
Table 4: Sonographic Patterns, Estimated Risk of Malignancy, and Fine-Needle Aspiration Guidance for Thyroid Nodules (3).
|
Sonographic pattern |
US features |
ROM % |
FNA size cutoff |
|
High suspicion |
Solid hypoechoic nodule or solid hypoechoic component of a partially cystic nodule with one or more of the following features: irregular margins (infiltrative, micro lobulated), microcalcifications, taller than wide shape, rim calcifications with small extrusive soft tissue component, evidence of ETE |
>70–90 |
Recommend FNA at ≥1 cm |
|
Intermediate suspicion |
Hypoechoic solid nodule with smooth margins without microcalcifications, ETE, or taller than wide shape |
10–20 |
Recommend FNA at ≥1 cm |
|
Low suspicion |
Isoechoic or hyperechoic solid nodule, or partially cystic nodule with eccentric solid areas, without microcalcification, irregular margin or ETE, or taller than wide shape. |
5–10 |
Recommend FNA at ≥1.5 cm |
|
Very low suspicion |
Spongiform or partially cystic nodules without any of the sonographic features described in low, intermediate, or high suspicion patterns |
<3 |
Consider FNA
at ≥2 cm a reasonable option |
|
Benign |
Purely cystic nodules (no solid component) |
<1 |
No biopsy |
Table 5: The Bethesda system for reporting thyroid cytopathology, ROM with clinical management recommendations (6).
|
Diagnostic category |
ROM average% (range) |
Usual management |
|
Nondiagnostic |
13 (5–20) |
Repeat with ultrasound guidance |
|
Benign |
4 (2–7) |
Clinical and sonographic follow-up |
|
Atypia of Undetermined Significance |
22 (12–30) |
Repeat FNA, molecular testing, diagnostic lobectomy, or surveillance |
|
Follicular neoplasm |
30 (23–34) |
Molecular testing, diagnostic lobectomy |
|
Suspicious for Malignancy |
74 (67–83) |
Molecular testing, lobectomy, or near-total thyroidectomy |
|
Malignant |
97 (97–100) |
Lobectomy or near-total thyroidectomy |
- References
1. Tunbridge WM, Evered DC, Hall R, Appleton D, Brewis M, Clark F, Evans JG, Young E, Bird T, Smith PA. The spectrum of thyroid disease in a community: the Whickham survey. Clin Endocrinol (Oxf) 1977; 7:481–493.
2. Guth S, Theune U, Aberle J, et al. Very high prevalence of thyroid nodules de tected by high frequency (13 MHz) ultrasound examination. Eur J Clin Invest 2009;39(8):699–706.
3. Haugen BR, Alexander EK, Bible KC, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 2016;26(1):1–133.
4. Hegedus L, Clinical practice. The thyroid nodule. N Engl J Med 2004; 351:1764–1771.
5. Lee JY, Baek JH, Ha EJ, Sung JY, Shin JH, Kim JH, et al. 2020 Imaging guidelines for thyroid nodules and differentiated thyroid cancer: Korean Society of Thyroid Radiology. Korean J Radiol 2021; 22:840-860.
6. Riddhi AP, and Keval P: The 2023 Bethesda System for Reporting Thyroid Cytopathology Journal of Clinical and Scientific Research 2024;13 (4) 297-307
7. Gharib H, Papini E, Garber JR, Duick DS, Harrell RM, Hegedüs L, Paschke R, Valcavi R, Vitti P, AACE/ACE/AME Task Force on Thyroid Nodules, et al. American college of endocrinology, and association medici endocrinologi medical guidelines for clinical practice for the diagnosis and management of thyroid nodules–2016 update. Endocr Pract. 2016;22: 622–639.
8. Durante C, Hegedüs L, Czarniecka A, Paschke R, Russ G, Schmitt F, et al.2023 European Thyroid Association Clinical Practice Guidelines for thyroid nodule management. Eur. Thyroid. J. 2023;12(5) e230067.