Primary Postpartum Hemorrhage
| Site: | EHC | Egyptian Health Council |
| Course: | Obstetric and Gynecology Guidelines |
| Book: | Primary Postpartum Hemorrhage |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 17 December 2025" Download Guideline
- Executive Summary
This guideline offers evidence-based recommendations on prevention and management of postpartum hemorrhage. The recommendations are intended to provide healthcare professionals with practical guidance on risk prevention, treatment interventions and improving health outcomes for cases of postpartum hemorrhage.
List of Recommendations
|
Recommendation |
Strength |
|
1. PREVENTION OF POSTPARTUM HEMORRHAGE |
|
|
1.1 Risk Assessment: |
|
|
1.1.1. Risk factors for PPH may present antenatally or intrapartum; care plans must be modified as and when risk factors arise. |
GPS |
|
1.1.2. Clinicians must be aware of risk factors for PPH and should take these into account when counselling women about place of delivery. |
GPS |
|
1.1.3. Women with known risk factors for PPH should only be delivered in a hospital with a blood bank on site. |
Strong |
|
1.1.4. An individualized risk assessment for postpartum hemorrhage should be documented upon arrival to a labor unit and updated throughout labor and delivery. |
Strong |
|
1.2 Risk Factors |
|
|
1.2.1 Consider the following antenatal risk factors for PPH · Age > 35 · Obesity (BMI >35) · More than 3 prior births · Previous PPH · Previous Cesarean section with placenta previa or PAS · Previous uterine surgery · Presence of uterine fibroid · Multiple pregnancy · Polyhydramnios · Fetal macrosomia · Anemia · Known coagulopathy or other bleeding disorders |
GPS |
|
1.2.2. Consider the following intrapartum risk factors for PPH · Oxytocin use in labor · Prolonged second stage · Prolonged third stage · Retained placenta · Manual removal of placenta · Assisted vaginal birth · Cesarean section whether elective or emergency · Perineal trauma · Uterine rupture · General anaesthesia · Infection · Non-cephalic presentation · Precipitate labor |
GPS |
|
1.3. Antenatal Risk Management |
|
|
1.3.1. Antenatal anemia |
|
|
1.3.1.1. Antenatal anemia should be investigated and treated appropriately as this may reduce the morbidity associated with PPH. |
Strong |
|
1.3.2. Maternal blood disorders |
|
|
1.3.2.1. Involve specialist physician to: · Optimize coagulation profile prior to birth · Advise on birth options (e.g. mode of birth) 1.3.2.2. Seek anesthetic opinion regarding options for analgesia during labor and birth |
Strong |
|
1.3.3. Abnormal placentation |
|
|
1.3.3.1. Determine placental site and if abnormal placental adherence is suspected (PAS): · Refer to a center equipped for placenta accreta management. · Involve multidisciplinary team including urologist, general surgeon, vascular surgeon, hematologist, senior anaesthetist and ICU specialist in preoperative planning. |
Strong |
|
1.4. Intrapartum Risk Management |
|
|
1.4.1. Management of third stage of labor The use of uterotonics for prevention of PPH during the third stage of labor is recommended for all births. |
Strong |
|
1.4.2. Cord clamping: 1.4.2.1. Late cord clamping (performed approximately 1 to 3 minutes after birth) is recommended for all births while initiating simultaneous essential newborn care. 1.4.2.2. Early cord clamping (<1 minute after birth) is not recommended unless the neonate is asphyxiated and needs to be moved immediately for resuscitation. |
Strong |
|
1.4.3. Controlled cord traction: 1.4.3.1 Consider Controlled cord traction (CCT) as part of active/modified active management of third stage as it may. |
Strong |
|
1.4.3.2. Providers employing CCT should only do so after signs of placental separation, and traction should be performed with uterine contraction as these measures reduce the risk of uterine inversion, cord avulsion, and partial detachment of the placenta. |
Strong |
|
1.5. Prophylactic Uterotonics: |
|
|
1.5.1. Oxytocin: 1.5.1.1. In most circumstances, oxytocin is the prophylactic uterotonic of choice. 1.5.1.2 For vaginal birth · If vaginal birth with IV access: Oxytocin 10 IU IV injected slowly over 3–5 minutes is recommended in preference to IM · If vaginal birth without IV access: Oxytocin 10 IU IM · To be transferred ransfer to rational: When compared with IM, IV oxytocin reduces the risk of PPH, need for blood transfusion 60-62 and incidence of retained placenta with no significant difference in side effects (e.g. hypotension and tachycardia) between routes 60,61 1.5.1.3. For CS birth: · Oxytocin 5 IU IV over 1–2 minutes · Monitor for hemodynamic impact · Avoid rapid IV bolus administration 1.5.1.4. If cardiovascular compromise exists (e.g. hypovolemia, shock, cardiac disease), use caution with IV administration. Rational it may result in transient hemodynamic instability 61, 65 |
Strong |
|
1.5.2. Ergometrine: 1.5.2.1 Ergometrine can be given IM or, in life-saving circumstances, as a slow IV injection. 1.5.2.2. Ergometrine should not be used in patients with essential or gestational hypertension, or in patients on HIV protease inhibitors. 1.5.2.3. Though undisputedly extremely effective, potential adverse effects limit ergometrine to a second-line agent. |
Conditional |
|
1.5.3. Carbetocin: 1.5.3.1. Routinely use oxytocin in preference to carbetocin if vaginal birth and cold-chain storage of oxytocin can be guaranteed (e.g. hospital setting). 1.5.3.2. If vaginal birth and cold-chain storage of uterotonics cannot be guaranteed: · Carbetocin is an effective alternative uterotonic · IM is preferred route of administration 1.5.3.2. If CS birth under regional anesthetic: IV carbetocin may be considered as a cost effective uterotonic 1.5.3.3. If CS birth under general anesthetic: Carbetocin is not recommended due to insufficient evidence. 1.5.3.4. If used: used as a single dose only, not for repeated use |
Conditional |
|
1.5.4. Misoprostol: 1.5.4.1. Not recommended if alternative injectable uterotonics are available 1.5.4.2. Use only if no other injectable uterotonic is available (e.g. due to unexpected birth in low resource setting or if storage conditions for uterotonics are inadequate). 1.5.4.3. The dose is 600 micrograms orally or sublingual single dose immediately after birth 1.5.4.4. If in a low resource setting with limited PPH treatment capability, consider use if: · an injectable uterotonic has been administered AND · continued bleeding is anticipated and/or blood loss is estimated to be greater than or equal to 350 mL |
Conditional |
|
1.6. Tranexamic Acid (TXA) For Prophylaxis in High Risk Women |
|
|
1.6.1. Tranexamic acid can be used as a prophylactic agent as an adjunct to uterotonics in patients at high risk for postpartum hemorrhage. 1.6.2. Use TXA within 3 hours of birth of the baby in a fixed dose of 1 g in 10 mL IV over 10 minutes (100 mg/min i.e. 1 ml /minute) |
Strong |
|
1.7. Immediate Postpartum Risk Management |
|
|
1.7.1. Uterine massage: Sustained uterine massage is not recommended as an intervention to prevent PPH in women who have received prophylactic oxytocin. |
GPS |
|
1.7.2. Uterine tonus assessment: Postpartum abdominal uterine tonus assessment for early identification of uterine atony is recommended for all women. |
Strong |
|
1.7.3. Nipple stimulation & breast feeding: Nipple stimulation and/or early breastfeeding may increase uterine activity but has not been shown to reduce bleeding or incidence of PPH. |
GPS |
|
1.7.4. Observation for women with risk factors in the first 2 hours postpartum: · Vital signs: Respiratory rate, pulse rate, and blood pressure, every 15-30 minutes in the first hour and every 30 minutes in the second hour. · Blood Loss every 15-30 minutes by visualizing the labia and perineum and be alert for slow steady trickle. · Temperature every 30 minutes · Uterine tonus assessment · Urine output: after the first 2 hours · After the first 2 hours continue as clinically indicated |
Strong |
|
2. RECOGNITION OF PPH |
|
|
2.1. General Principles In Diagnosis of PPH |
|
|
2.1.1 Early recognition of postpartum hemorrhage (before deterioration in vital signs) is recommended and should be the goal in order to improve outcomes. |
Strong |
|
2.1.2. Clinical signs and symptoms of hypovolemia should be included in the assessment of PPH. However, clinicians should be aware that the signs of hypovolemic shock become less sensitive in pregnancy. |
Strong |
|
2.1.4. Consider initiation of response to PPH when there is excessive bleeding in the first 24 hours post birth, judged clinically, or through estimation of blood loss volume (> 500 ml after VD or 1000ml after CS), or changes in the hemodynamic state. |
Strong |
|
2.2 Visual Assessment of The Amount of Postpartum Blood Loss |
|
|
2.2.1. Visual estimation of blood loss is always subjective and can be imprecise and often leads to underestimation of large volumes or overestimation of small volumes 2.2.2. When conducting visual assessment of blood loss, consider the volume, nature and speed of blood loss. 2.2.3. Simulated scenarios and pictorial guides may improve staff accuracy |
Conditional |
|
2.3. Quantitative Measurement of The Amount of Postpartum Blood Loss |
|
|
2.3.1 Consider that quantitative measurement, provides a more accurate assessment of blood loss when compared with visual estimation. 2.3.2. Consider measurement of blood loss by blood collection drapes for vaginal deliveries,5,10 or the weighing of swabs and weigh blood-soaked items (e.g. linen, pads, swabs, drapes) to quantify volume.5,60 If weighing, 1 gram is equivalent to 1 mL blood loss. |
Conditional |
|
2.4. Diagnosis of PPH & Assessment of Hemodynamic Compromise |
|
|
2.4.1. PPH can be minor (500–1000 ml blood loss) or major (more than 1000 ml blood loss). The condition becomes worse if the patient lost >2000 mL of blood. 2.4.2. Diagnose minor PPH (blood loss 500-1000 mL) if there are: · Normal blood pressure · Mild increase in heart rate (but <100 beats/minute) · Palpitation · Lightheadedness 2.4.3. Diagnose major PPH (blood loss from 1000-2000 mL) if there are: · Drop of blood pressure below the original average of the patient · Tachycardia (≥ 100 beats/minute) · Sweating · Weakness |
GPS |
|
2.4.4. Signs of hemodynamic compromise are a late indicator of PPH and may not be evident until large volumes of blood are lost (e.g. up to 25% of total blood volume or greater than 1500 mL). 2.4.5. Don’t wait for the following to diagnose major PPH as blood loss is already >2000 mL and the condition would be severe enough to be of poorer prognosis: · Systolic blood pressure drops below 70-80 mmHg · Tachycardia (120-140 beats/minute) · Restlessness, confusion and pallor · Oliguria 2.4.6. Conversely, compromise may occur earlier in women with: · Gestational hypertension with proteinuria · Anemia · Dehydration · Small stature · Cardiac disease |
Strong |
|
3. RESPONDING TO AND MANAGEMENT OF PPH |
|
|
3.1. General Principles In Responding To PPH |
|
|
3.1.1. Bleeding after labor is an emergency and responding to it should not be delayed and should start as early as possible and goes hand in hand with the rest of management (Communication with the patient, senior staff, resuscitation investigation, and monitoring). |
GPS |
|
3.1.2. Use an approach that involves maintaining hemodynamic stability while simultaneously identifying and treating the cause of blood loss. |
Strong |
|
3.1.3. The urgency and measures undertaken to resuscitate and arrest hemorrhage need to be tailored to the amount of blood loss (which is usually underestimated), the patient general condition, and the degree of shock. |
Strong |
|
3.1.4 The GDG recommends adoption of the EMOTIVE bundle (Early detection, Massage, Oxytocics, Tranexamic acid, IV fluids, Examination & Escalation) as the standard first-response protocol for all cases of postpartum hemorrhage. Initiate all components within 15 minutes of PPH diagnosis to ensure rapid, standardized, and evidence-based management, with ongoing monitoring and escalation as needed, see Appendix II. |
Strong |
|
3.2. Communication And Multidisciplinary Care: |
|
|
3.2.1. Communication with the patient and her birthing partner is important, and clear information of what is happening should be given from the outset. 3.2.2. Relevant staff with an appropriate level of expertise should be alerted of PPH. 3.2.3. The first-line obstetric and anesthetic staff should be alerted when women present with minor PPH (blood loss 500–1000 ml) without clinical shock 3.2.4. A multidisciplinary team involving senior members of staff should be called to attend to women with major PPH (blood loss of more than 1000 ml) and ongoing bleeding or clinical shock |
GPS |
|
3.3. Responding To & Managing Minor PPH |
|
|
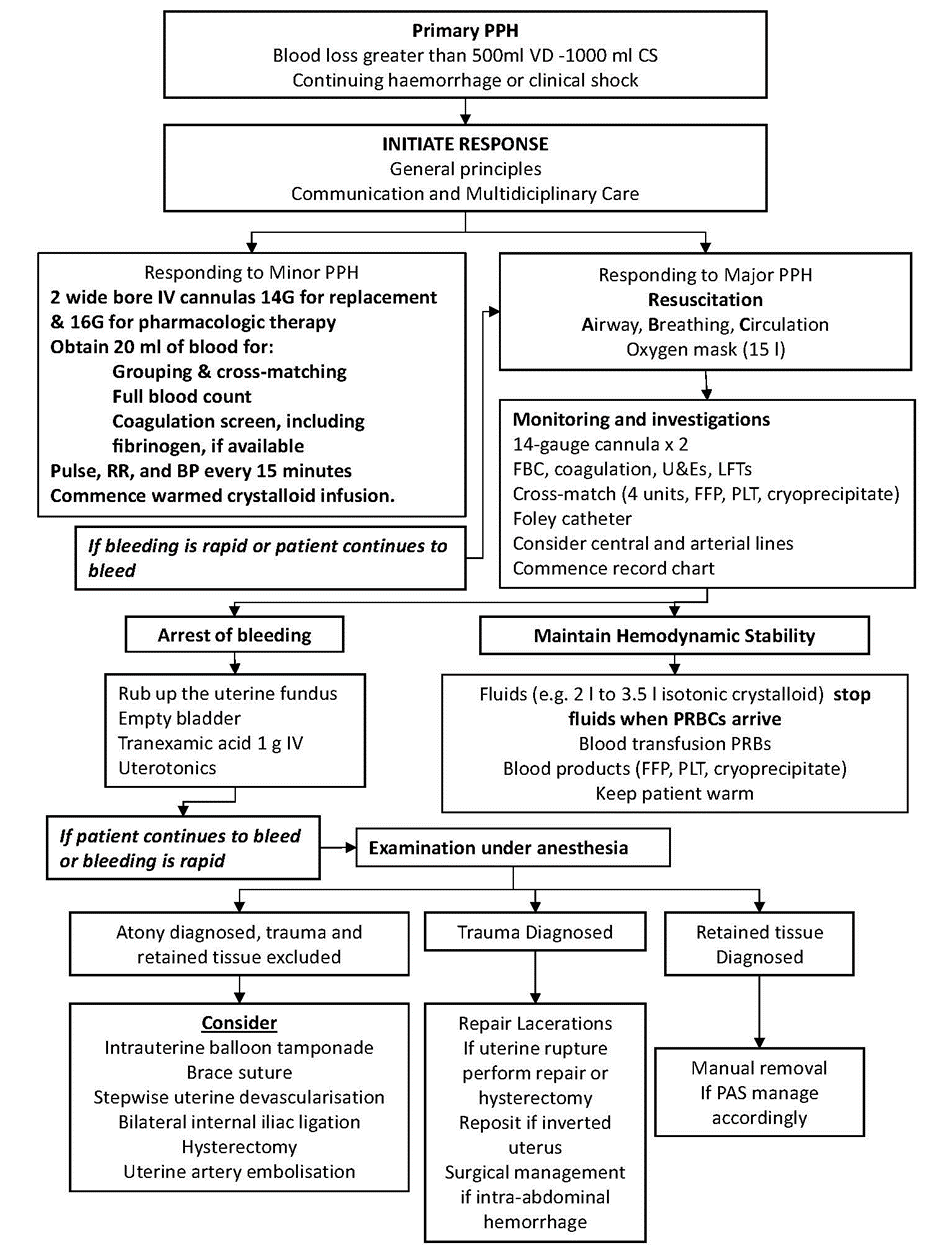
3.3.1. Measures for minor PPH (blood loss 500–1000 ml) without clinical shock: · Establish IV access: 2 wide bore IV cannulas (Orange 14G & Grey 16G) are to be inserted. 14G for fluid and blood replacement & 16G for pharmacologic therapy · Urgent venipuncture and obtain 20 ml of blood for: o Grouping & cross-matching o Full blood count o Coagulation screen, including fibrinogen, if available · Pulse, respiratory rate and blood pressure recording every 15 minutes · Commence warmed crystalloid infusion. · If bleeding is rapid and patient continues to bleed, consider starting the full protocol for major PPH. · In minor PPH, there are no firm criteria for initiating red cell transfusion. The decision to provide blood transfusion should be based on both clinical and hematological assessment. |
GPS |
|
3.4. Identification of The Cause (the 4Ts) |
|
|
3.4.1. If blood loss after delivery seems increased, start the measures of minor PPH and immediately assess for the Tone of the uterus, the presence of Trauma, any retained Tissue or Thrombin activity. · Tone: massage the fundus to see whether the uterus is atonic or well contracted. · Tissue: retained placental fragment. Inspect the placenta for a missing part. Also suspect retained placental fragment or pieces of membranes as a cause if there is atonic fundus unresponsive to uterotonics. · Trauma: if fundus is well contracted, blood clotting is ok and no placental remnants, explore birth canal for improperly repaired birth tract injuries. · Thrombin: The fundus is contracted (may be atonic) and the blood is not clotting. · Other unknown causes: if all of the above is normal, assess for uterine rupture/ inversion, concealed bleeding (e.g. vault hematoma, internal hemorrhage after CS) and non-genital causes (e.g. subcapsular liver rupture). |
GPS |
|
3.5. Responding To & Managing Major PPH |
|
|
3.5.1. Full protocol for major PPH (blood loss greater than 1000 ml) and continuing to bleed or clinical shock: · A and B: assess Airway and Breathing · C – evaluate Circulation · Position the patient flat · Insert two peripheral cannulas (14 gauge) if not inserted before · Immediately draw 20 mL of blood for: o Cross-matching (4 units of blood minimum) o Full blood count o Coagulation screen (PT, INR, aPTT including fibrinogen, if available) o Chemistry profile (Serum Creatinine, SGOT, SGPT, Albumin, LDH, Blood Sugar) · Keep the woman warm using appropriate available measures · Transfuse blood as soon as possible. · Until blood is available, infuse up to 3.5 L of warmed crystalloids as Lactated Ringer’s (1–2 mL for every 1 mL of blood loss)2 · Stop fluid once blood is ready. · The best equipment available should be used to achieve rapid warmed infusion of fluids. · Special blood filters should not be used, as they slow infusions. · If actively bleeding, transfuse early and do not wait unnecessarily for laboratory results and use clinical assessment as the main determinant 3.5.2. Blood transfusion: · 3.5.2.1. If actively bleeding, transfuse early, do not wait unnecessarily for laboratory results. The clinical picture should be the main determinant of the need for blood transfusion and time should not be unnecessarily spent awaiting laboratory results. · 3.5.2.2. Packed RBCs: o Provide emergency blood (Packed RBCs) with immediate issue of group O, rhesus D (RhD)-negative, with a switch to group-specific blood as soon as feasible. o Single Hb/hematocrit estimations may be misleading and can lead to delays in initiating red cell transfusion, serial measurements may be helpful to monitor ongoing progress. · 3.5.2.2. Transfusion of FFP: o Administration of FFP should be guided by coagulation profile test results and whether bleeding is continuing. o But if coagulation profile results are not available and bleeding is continuing, after 4 units of red blood cells, 4 units of FFP should be infused until test results are known. o If coagulation profile results are not available, early FFP should be considered for conditions with a suspected coagulopathy, such as placental abruption or amniotic fluid embolism, or where detection of PPH has been delayed. o If prothrombin time/activated partial thromboplastin time is more than 1.5 times normal and hemorrhage is ongoing, more units of FFP are likely to be needed to correct coagulopathy. · 3.5.2.3. Clinicians should be aware that these blood components must be ordered as soon as a need for them is anticipated, as there will always be a short delay in supply because of the need for thawing. 3.5.3. Cryoprecipitate For Fibrinogen Replacement · A plasma fibrinogen level of greater than 2 g/l should be maintained, if test is available, during ongoing PPH. · Cryoprecipitate should be used for fibrinogen replacement. · Physician should be aware that fibrinogen below 3 g/l and especially below 2 g/l is associated with progression of bleeding, increased RBC and blood component requirements, and the need for invasive procedures. 3.5.4. Transfusion of platelets · During PPH, platelets should be transfused when the platelet count is less than 75 × 109/l (75000 / mm3) based on laboratory monitoring. · The platelets should ideally be group compatible. RhD-negative women should receive RhD-negative platelets. 3.5.5. Use Continued Resuscitation with lab-guided replacement to maintain the following lab parameters: · Hb greater than 8 gm% · Platelet count greater than 50 × 109 /l (50000/mm3) · Prothrombin time (PT) less than 1.5 times normal · Activated partial thromboplastin time (APTT) less than 1.5 times normal · Fibrinogen level greater than 2 g/l. |
GPS |
|
3.5.5. Tranexamic acid (TXA) 3.5.5.1. Tranexamic acid can be used in all patients as an adjunct to uterotonics in the setting of postpartum hemorrhage regardless of whether the bleeding is due to genital tract trauma or other causes. 3.5.5.2. Give TXA as early as possible, ideally within 3 hours of birth, in a fixed dose of 1 g in 10 mL (100 mg/mL) IV at 1 mL per minute (i.e., over 10 minutes). 3.5.5.3. A second dose of 1 g IV may be administered if bleeding continues after 30 minutes of the first dose or for bleeding that stops but restarts within 24 hours of completing the first dose. |
Strong |
|
4. MONITORING AND INVESTIGATIONS IN MAJOR PPH |
|
|
4.1 Immediate venipuncture (20 ml) for: · Cross-matching (4 units of Packed RBCs minimum) · FBC · Coagulation screen (PT, INR, aPTT including fibrinogen, if available) · Chemistry profile (Serum Creatinine, SGOT, SGPT, Albumin, LDH, Blood Sugar) · Repeat as necessary 4.2 Continuous pulse, blood pressure and respiratory rate recording (preferably using oximeter, electrocardiogram and automated blood pressure recording when available) and Monitor temperature every 15 minutes. 4.3. Foley’s catheter to monitor urine output 4.4. Documentation of fluid balance, blood, blood products and procedures. 4.5. Consider transfer to intensive therapy unit once the bleeding is controlled or monitoring at high dependency unit on delivery suite, if appropriate. 4.6. Consider objective scoring system for monitoring as the MEOWS Score (see Appendix II) |
GPS |
|
5. ROLE OF THE ANESTHETIST IN THE MANAGEMENT OF PPH |
|
|
The management of PPH requires a multidisciplinary approach: Allow the anesthetist to play a crucial role in maintaining hemodynamic stability and, in determining and administering the most appropriate method of anesthesia. |
Strong |
|
6. PAIN MANAGEMENT |
|
|
Consider pain relief requirements during initial resuscitation and all subsequent treatments |
GPS |
|
7. PHARMACOLOGICAL AND MECHANICAL STRATEGIES IN MANAGEMENT OF PPH |
|
|
7.1. Clinicians should be prepared to use a combination of pharmacological, mechanical and surgical methods to arrest PPH. These methods should be directed towards the causative factor. |
Strong |
|
7.2. When uterine atony is perceived to be a cause of the bleeding, then a sequence of pharmacological and mechanical measures (in the form of uterine massage and urinary bladder catheterization) should be instituted until the bleeding stops. |
Strong |
|
7.3 Stopping the bleeding should not be delayed and should start as early as possible and goes hand in hand with the rest of management above (Communication and multidisciplinary approach, Resuscitation, Investigation, and Monitoring) |
GPS |
|
7.4. The most common cause of primary PPH is uterine atony. The initial management of PPH should, therefore, involve measures to stimulate myometrial contractions. Thus, mechanical and pharmacological measures should be instituted/administered as needed. 7.5. Palpate the uterine fundus and rub it to stimulate contractions and expel its contents (‘rubbing up the fundus’) 7.6. Ensure that the bladder is empty (Foley catheter & leave in place) 7.7 First line pharmacological therapy for uterine atony (Oxytocin): · Give oxytocin 5 IU IV over 1-2 minutes (even if had the prophylactic dose before). The dose may be repeated, only once, after five minutes but consider the maximum dose for IV oxytocin which is 10 IU IV. · Rapid IV bolus administration may cause hypotension, tachycardia, arrhythmia and myocardial ischemia. · Continue with oxytocin IV infusion 40 IU in 500 mL of either 0.9% sodium chloride (saline) or compound sodium lactate (Ringer’s lactate) at a rate of 5-10 IU/hour via: o Using ordinary IV line about adjust it to deliver 20-40 drop/minute o Infusion pump, if available, equals to 62.5–125 mL per hour o Minimum infusion duration is 2 - 4 hours; use clinical judgement. · Oxytocin infusion may be a safer alternative to a bolus dose of oxytocin in some women (e.g. major cardiovascular disorders) · If the patient had induction of labor (IOL) with oxytocin, you may use the same infusion but at increased rate. · If IV access unavailable or delayed, oxytocin 10 IU IM can be administered · If carbetocin has already been given, consider non-oxytocin uterotonic instead 7.8 Supplemental therapy if no adequate response: give the following: 7.8.1. Ergometrine maleate: o 0.25 - 0.5 mg IV over 1-2 minutes (dilute 250 micrograms in 5 mL 0.9% sodium chloride) that may be repeated every 5 minutes to a maximum of 1 mg o It is contraindicated with retained placenta, severe hypertension, pre-eclampsia, eclampsia severe/persistent sepsis, renal, hepatic, vascular, or cardiac disease 7.8.2. Misoprostol: · Consider misoprostol if alternative uterotonics unavailable or contraindicated (e.g. asthma, hypertension) or if bleeding not effectively controlled with oxytocin. · Give 800 mcg sublingual (rapid onset of action with side effects more likely) · Or 1000 mcg per rectum (slow absorption but prolonged effect) · Consider clinical circumstances when determining optimal route · Side effects: Hyperthermia is a common side effect (>38 C is common) and malignant hyperthermia > 40 C has been reported in 1 - 14% of cases |
GPS |
|
8. SURGICAL TREATMENTS OF PPH |
|
|
8.1. If pharmacological measures fail to control the hemorrhage, surgical interventions should be initiated sooner rather than later and the most experienced, anesthetist and obstetrician should be called to be involved in the management. |
Strong |
|
8.2. Transfer the patient to the operative theatre flat with high-flow oxygen and perform an examination under anesthesia for the exclusion of the presence of remnants (to be evacuated if present) and dealing with any birth tract injuries if found |
Strong |
|
8.3. Bimanual Compression: The use of bimanual uterine compression is recommended as a temporary measure until appropriate care is available for the treatment of PPH due to uterine atony after vaginal delivery |
Strong |
|
8.4. Intrauterine balloon tamponade: · If bimanual compression has been effective consider the use of intrauterine balloon tamponade (e.g. Bakri Balloon) as balloon is an appropriate first-line ‘surgical’ intervention for most women where uterine atony is the only or main cause of hemorrhage after the success of bimanual uterine compression. · After insertion, assess blood loss: · If bleeding continues, balloon tamponade may be ineffective—review aetiology of PPH, check balloon placement and consider other surgical interventions. · If bleeding ceases on insertion, monitor fundal height, uterine cramping and signs of increased blood loss regularly · Balloon is left for 6 hours if bleeding stopped and removed in presence of senior staff lest the return of bleeding. |
Strong |
|
8.5. If bimanual compression is ineffective or balloon tamponade fail: · Consider exploratory laparotomy when less invasive approaches fail to control bleeding. In the setting of a vaginal delivery, it is common to use a midline vertical abdominal incision for laparotomy, to optimize exposure and reduce risk of surgical bleeding. In the setting of cesarean birth, the existing surgical incision may be used. · Conservative surgical interventions (compression sutures and/or arterial ligation) may be attempted as a second line, depending on clinical circumstances and available expertise. · Timing is critical. Surgical interventions should be initiated sooner rather than later. · Weigh benefits of conservative versus aggressive management (hysterectomy) |
Strong |
|
8.6 Hemostatic uterine suture (e.g. B-Lynch suture) · Judiciously apply aortic compression (below the level of the renal arteries) as a temporary measure · Consider B-Lynch compression or other compression suture · It is preferable that a laminated diagram of the brace suture technique, as B-lynch suture, be kept in theatre. |
GPS |
|
8.7. Arterial ligation: Stepwise arterial ligation for uterine devascularization may be considered depending on clinical circumstances and available expertise: · Bilateral uterine artery ligation · Bilateral utero-ovarian artery ligation · If expertise available, bilateral internal iliac artery ligation |
GPS |
|
8.8. Hysterectomy: |
|
|
8.8.1. Resort to hysterectomy early (sooner rather than later) especially if: · Life is threatened (hemodynamic unstable patient), · Bleeding continues after use of conservative treatment options · blood transfusion is limited or not an option. · In cases of placenta accreta or uterine rupture. |
Strong |
|
8.8.2. Subtotal (Supra-Vaginal) hysterectomy is quicker and safer in Major PPH and in packing of the abdomen, use hot packs. |
GPS |
|
8.8.3. Ideally and when feasible, a second experienced clinician should be involved in the decision for hysterectomy. |
GPS |
|
8.8.4. In rare situations in spite of compression, arterial ligation and hysterectomy, bleeding from undefined origin might persist in the pelvis (undefined diffuse venous bleeding or bleeding due to DIC). Tight pelvic packing with radio-opaque packs, done by a senior obstetrician, may be a last resort to be followed by temporarily closing the abdomen, stabilization of the patient condition by multidisciplinary team, then reopening the abdomen again to remove the packs and reassess hemostasis after 24-48 hours. |
Conditional |
|
9. RISK MANAGEMENT |
|
|
9.1. Training and Preparation |
|
|
Every maternity unit should have a multidisciplinary protocol for the management of PPH. |
GPS |
|
All staff involved in maternity care should receive training in the management of obstetric emergencies, including the management of PPH. |
Strong |
|
Training for PPH should be multidisciplinary and multiprofessional including team rehearsals. |
Strong |
|
All cases of PPH involving a blood loss of greater than 1500 ml should be the subject of a formal clinical incident review. |
GPS |
|
9.2. Documentation |
|
|
Accurate documentation of a delivery with PPH is essential. |
GPS |
|
9.3. Consent |
|
|
If treatment is likely to affect woman’s fertility, prioritize gaining informed consent. |
Strong |
|
10. Postnatal Care |
|
|
Upon discharge: Educate about signs, symptoms and self-referral regarding persistent or increasing bleeding, infection and risk of secondary PPH, postnatal depression, and venous thromboembolism |
GPS |
|
Oral iron supplementation, either alone or in combination with folic acid, may be provided to postpartum women for 6–12 weeks after delivery for reducing the risk of anemia. |
Conditional |
|
Offer the woman and family debriefing/clinical disclosure by senior team members, preferably by clinicians who were at the event Offer additional opportunities for discussion/debrief six weeks postpartum |
GPS |
- Recommendations
1.
PREVENTION OF POSTPARTUM
HEMORRHAGE
1.1. Risk Assessment:
- Risk factors for PPH may present antenatally or intrapartum; care plans must be modified as and when risk factors arise. (GPS)
- Clinicians must be aware of risk factors for PPH and should take these into account when counselling women about place of delivery. (GPS)
- Women with known risk factors for PPH should only be delivered in a hospital with a blood bank on site. (Strong, low)5
- An individualized risk assessment for postpartum hemorrhage should be documented upon arrival to a labor unit and updated throughout labor and delivery. (Strong, low)5
1.2 Risk Factors
- Consider the following antenatal risk factors for PPH: (GPS)
· Age > 35
· Obesity (BMI >35)
· More than 3 prior births
· Previous PPH
· Previous Cesarean section with placenta previa or PAS
· Previous uterine surgery
· Presence of uterine fibroid
· Multiple pregnancy
· Polyhydramnios
· Fetal macrosomia
· Anemia
· Known coagulopathy or other bleeding disorders
- Consider the following intrapartum risk factors for PPH: (GPS)
· Oxytocin use in labor
· Prolonged second stage
· Prolonged third stage
· Retained placenta
· Manual removal of placenta
· Assisted vaginal birth
· Cesarean section whether elective or emergency
· Perineal trauma
· Uterine rupture
· General anaesthesia
· Infection
· Non-cephalic presentation
· Precipitate labor
1.3. Antenatal Risk Management
1.3.1. Antenatal anemia
- Antenatal anaemia should be investigated and treated appropriately as this may reduce the morbidity associated with PPH. (strong, low)5
1.3.2. Maternal blood disorders
- Involve specialist physician to Optimise/stabilise coagulation profile prior to birth, advise on birth options (e.g. mode of birth). (strong, very low)11,12
- Seek anaesthetic opinion regarding options for analgesia during labor and birth. (strong, very low)11,12
1.3.3. Abnormal placentation
- Determine placental site and if abnormal placental adherence is suspected (PAS): (Strong, very low)13,14
· Refer to a center equipped for placenta accreta management.
· Involve multidisciplinary team including urologist, general surgeon, vascular surgeon, hematologist, senior anaesthetist and ICU specialist in preoperative planning.
1.4. Intrapartum Risk Management
1.4.1. Management of third stage of labor
- The use of uterotonics for prevention of PPH during the third stage of labor is recommended for all births. (strong, high)10,15
1.4.2. Cord clamping:
1.4.2.1. Late cord clamping (performed approximately 1 to 3 minutes after birth) is recommended for all births while initiating simultaneous essential newborn care. (strong, moderate)16
1.4.2.2. Early cord clamping (<1 minute after birth) is not recommended unless the neonate is asphyxiated and needs to be moved immediately for resuscitation. (strong, moderate)16
1.4.3. Controlled cord traction:
- Consider Controlled cord traction (CCT) as part of active/modified active management of third stage. (strong, low)17
- Providers employing CCT should only do so after signs of placental separation, and traction should be performed with uterine contraction as these measures reduce the risk of uterine inversion, cord avulsion, and partial detachment of the placenta. (strong, moderate)10,17
1.5. Prophylactic Uterotonics:
1.5.1. Oxytocin:
- The following are recommendations for Oxytocin use in the prevention of PPH: (Strong, High-quality evidence)5,9,10,11,18,19,20,23,24.25
o In most circumstances, oxytocin is the prophylactic uterotonic of choice.9,18
o For vaginal birth with IV access: Oxytocin 10 IU IV injected slowly over 3–5 minutes is recommended in preference to IM.10,11,19
o For vaginal birth without IV access: Oxytocin 10 IU IM is recommended.5,20
Rational: When compared with IM, IV oxytocin reduces the risk of PPH, need for blood transfusion,19,21,22 and incidence of retained placenta with no significant difference in side effects (e.g. hypotension and tachycardia) between routes.19,20,
- For CS birth Oxytocin 5 IU IV over 1–2 minutes, avoid rapid IV bolus administration and monitor for hemodynamic impact.5,23,24
- If cardiovascular compromise exists (e.g. hypovolemia, shock, cardiac disease), use caution with IV administration.19,25
Rational: Oxytocin may result in transient hemodynamic instability.19,25
1.5.2. Ergometrine
- The following are recommendations for Ergometrine use in the prevention of PPH:5,20
o Ergometrine can be given IM or, in life-saving circumstances, as a slow IV injection. (Conditional, low)5
o Ergometrine should not be used in patients with essential or gestational hypertension, or in patients on HIV protease inhibitors. (Strong, moderate)5
o Though undisputedly extremely effective, potential adverse effects limit ergometrine to a second-line agent.5,20
Rationale
The GDG is aware of the 2025 WHO position not to recommend the prophylactic use of fixed-dose combination of oxytocin and ergometrine because of concerns about safety. However, if the health worker and the woman regard the additional benefits of a combination of oxytocin and ergometrine (over either of these agents alone) is important in improving overall maternal outcomes, the use of ergometrine could be considered.
1.5.3. Carbetocin
- The following are recommendations for Carbetocin use in the prevention of PPH: (Conditional, moderate)18,20,26,27.28,29
o Routinely use oxytocin in preference to carbetocin if vaginal birth and cold-chain storage of oxytocin can be guaranteed (e.g. hospital setting).18,26
o If vaginal birth and cold-chain storage of uterotonics cannot be guaranteed Carbetocin (100 µg, intramuscularly/intravenously) is an effective alternative uterotonic.10,27
o IM is preferred route of Carbetocin administration.27
o If CS birth under regional anesthetic: IV carbetocin may be considered as a cost effective uterotonic.20,28,29
o If CS birth under general anesthetic: Carbetocin is not recommended due to insufficient evidence.27
o Use Carbetocin as a single dose only, not for repeated use.27
1.5.4. Misoprostol
- The following are recommendations for Misoprostol use in the prevention of PPH: (Conditional, moderate)9,10,29,30,31.32,33
o Misoprostol is Not recommended as a prophylactic uterotonic if alternative injectable uterotonics are available.29,30
o Use Misoprostol only if no other injectable uterotonic is available (e.g. due to unexpected birth in low resource setting or if storage conditions for uterotonics are inadequate).9,30,31,32
o Misoprostol dose is 600 micrograms orally or sublingual single dose immediately after birth.9,10,30,32
o If in a low resource setting with limited PPH treatment capability, consider Misoprostol use if an injectable uterotonic has been administered AND continued bleeding is anticipated and/or blood loss is estimated to be greater than or equal to 350 mL9,33
1.6. Tranexamic Acid (TXA) For Prophylaxis in High-Risk Women
- Tranexamic acid can be used as a prophylactic agent as an adjunct to uterotonics in patients at high risk for postpartum hemorrhage. (strong, high)34
- Use TXA within 3 hours of birth of the baby in a fixed dose of 1 g in 10 mL IV over 10 minutes (100 mg/min i.e. 1 ml /minute) (strong, high)34,35,36
Rationale:
GDG is aware of the WHO recommendation in 2025; “Tranexamic acid is not recommended for the prevention of postpartum hemorrhage at vaginal birth”.
The GDG considered that since there is no clear evidence of increased risk of maternal or newborn harms, or serious life-threatening adverse events with TXA, and considering the risk of thromboembolic events that may increase with the use of TXA, the GDG agreed that the use of TXA for prevention of PPH is recommended but limited to women at high risk for PPH where in this population the benefit for reducing postpartum blood loss at childbirth outweighs the risk of TXA.
1.7. Immediate Postpartum Risk Management
1.7.1. Uterine massage:
- Sustained uterine massage is not recommended as an intervention to prevent PPH in women who have received prophylactic oxytocin. (Strong, low-quality evidence)10,37
1.7.2. Uterine tonus assessment:
- Postpartum abdominal uterine tonus assessment for early identification of uterine atony is recommended for all women. (Strong, low-quality evidence)10,38
1.7.3. Nipple stimulation & breast feeding:
- Nipple stimulation and/or early breastfeeding may increase uterine activity but has not been shown to reduce bleeding or incidence of PPH. (Strong, low-quality evidence)39
1.7.4. Observation for women with risk factors:
- The following should be observed for women with PPH risk factors in the first 2 hours postpartum: (Strong, moderate-quality evidence)7
· Vital signs: Respiratory rate, pulse rate, and blood pressure, every 15-30 minutes in the first hour and every 30 minutes in the second hour.
· Blood Loss every 15-30 minutes by visualizing the labia and perineum and be alert for slow steady trickle.
· Temperature every 30 minutes
· Uterine tonus assessment
· Urine output: after the first 2 hours
· After the first 2 hours continue observations as clinically indicated7
2.
RECOGNITION OF PPH
2.1. General Principles in Diagnosis of PPH
- Early recognition of postpartum hemorrhage (before deterioration in vital signs) is recommended and should be the goal in order to improve outcomes. (Strong, high)6,10
- Clinical signs and symptoms of hypovolemia should be included in the assessment of PPH. However, clinicians should be aware that the signs of hypovolemic shock become less sensitive in pregnancy. (strong, moderate)5,6,18,40,41
- Consider initiation of response to PPH when there is excessive bleeding in the first 24 hours post birth, judged clinically, or through estimation of blood loss volume (> 500 ml after VD or 1000ml after CS), or changes in the hemodynamic state. (Strong, moderate)5,7
2.2. Assessment of postpartum blood loss: Identification of the severity of Hemorrhage
2.2.1. Visual Assessment of The Amount of Postpartum Blood Loss
- Care givers should consider that visual estimation of blood loss is always subjective, can be imprecise and often leads to Underestimation of large volumes Overestimation of small volumes. (conditional, moderate)43,44,45
- When conducting visual assessment of blood loss, consider Volume, Nature and speed of blood loss. (conditional, low)6,7
- Simulated scenarios and pictorial guides are recommended as this may improve staff accuracy. (conditional, moderate)45,46
2.2.2 Quantitative Measurement of The Amount of Postpartum Blood Loss
- Consider that quantitative measurement, provides a more accurate assessment of blood loss when compared with visual estimation. (conditional, high)5-10,45,46
- Consider measurement of blood loss by blood collection drapes for vaginal deliveries, or the weighing of swabs and weigh blood-soaked items (e.g. linen, pads, swabs, drapes) to quantify volume. If weighing, 1 gram is equivalent to 1 mL blood loss. (conditional, moderate)5,10,28,46
2.3. Diagnosis of PPH & Assessment of Hemodynamic Compromise
- Diagnose minor PPH (blood loss 500-1000 mL) if there are: (GPS)
· Normal blood pressure
· Mild increase in heart rate (but <100 beats/minute)
· Palpitation
· Lightheadedness
- Diagnose major PPH (blood loss from 1000-2000 mL) if there are: (GPS)
· Drop of blood pressure below the original average of the patient
· Tachycardia (≥ 100 beats/minute)
· Sweating
· Weakness
- Be aware that signs of hemodynamic compromise are a late indicator of PPH and may not be evident until large volumes of blood are lost (e.g. up to 25% of total blood volume or greater than 1500 mL). (Strong, moderate)40
- Don’t wait for the following to diagnose major PPH as blood loss is already >2000 mL and the condition would be severe enough to be of poorer prognosis: (Strong, moderate)48
· Systolic blood pressure drops below 70-80 mmHg
· Tachycardia (120-140 beats/minute)
· Restlessness, confusion and pallor
· Oliguria
- Consider that compromise may occur earlier in women with: (Strong, moderate)7
· Gestational hypertension with proteinuria
· Anemia
· Dehydration
· Small stature
· Cardiac disease
3.
RESPONDING TO AND
MANAGEMENT OF PPH
3.1. General Principles in Responding To PPH
3.1.2. Use an approach that involves maintaining hemodynamic stability while simultaneously identifying and treating the cause of blood loss. (strong, low)6
3.1.3. The urgency and measures undertaken to resuscitate and arrest hemorrhage need to be tailored to the amount of blood loss (which is usually underestimated), the patient general condition, and the degree of shock. (strong, low)5,47
3.1.4. The GDG recommends adoption of the EMOTIVE bundle (Early detection, Massage, Oxytocics, Tranexamic acid, IV fluids, Examination & Escalation) as the standard first-response protocol for all cases of postpartum hemorrhage. Initiate all components within 15 minutes of PPH diagnosis to ensure rapid, standardized, and evidence-based management, with ongoing monitoring and escalation as needed, see Appendix I. (Strong, low)10
3.2. Communication And Multidisciplinary Care:
3.2.1. Communication with the patient and her birthing partner is important, and clear information of what is happening should be given from the outset. (GPS)
3.2.2. Relevant staff with an appropriate level of expertise should be alerted of PPH. (GPS)
3.2.3. The first-line obstetric and anesthetic staff should be alerted when women present with minor PPH (blood loss 500–1000 ml) without clinical shock. (GPS)
3.2.4. A multidisciplinary team involving senior members of staff should be called to attend to women with major PPH (blood loss of more than 1000 ml) and ongoing bleeding or clinical shock. (GPS)
3.3. Responding To & Managing Minor PPH
- Perform the following actions promptly in response to minor PPH: (GPS)
· Establish IV access: 2 wide bore IV cannulas (Orange 14G & Grey 16G) are to be inserted. 14G for fluid and blood replacement & 16G for pharmacologic therapy
· Urgent venipuncture and obtain 20 ml of blood for:
o Grouping & cross-matching
o Full blood count
o Coagulation screen, including fibrinogen, if available
· Pulse, respiratory rate and blood pressure recording every 15 minutes
· Commence warmed crystalloid infusion.
· If bleeding is rapid and patient continues to bleed, consider starting the full protocol for major PPH.
· In minor PPH, there are no firm criteria for initiating red cell transfusion. The decision to provide blood transfusion should be based on both clinical and hematological assessment.
3.4. Identification of The Cause (the 4Ts)
3.4.1. If blood loss after delivery seems increased, start the measures of minor PPH and immediately assess for the Tone of the uterus, the presence of Trauma, any retained Tissue or Thrombin activity. (GPS)
· Tone: massage the fundus to see whether the uterus is atonic or well contracted.
· Tissue: retained placental fragment. Inspect the placenta for a missing part. Also suspect retained placental fragment or pieces of membranes as a cause if there is atonic fundus unresponsive to uterotonics.
· Trauma: if fundus is well contracted, blood clotting is ok and no placental remnants, explore birth canal for improperly repaired birth tract injuries.
· Thrombin: The fundus is contracted (may be atonic) and the blood is not clotting.
Other unknown causes: if all of the above is normal, assess for uterine rupture/ inversion, concealed bleeding (e.g. vault hematoma, internal hemorrhage after CS) and non-genital causes (e.g. subcapsular liver rupture).
3.5. Responding To & Managing Major PPH
3.5.1. Full protocol for major PPH (blood loss greater than 1000 ml) and continuing to bleed or clinical shock:
- Perform the following actions promptly in response to major PPH: (GPS)
· A and B: assess Airway and Breathing
· C – evaluate Circulation
· Position the patient flat
· Insert two peripheral cannulas (14 gauge) if not inserted before
· Immediately draw 20 mL of blood for:
o Cross-matching (4 units of blood minimum)
o Full blood count
o Coagulation screen (PT, INR, aPTT including fibrinogen, if available)
o Chemistry profile (Serum Creatinine, SGOT, SGPT, Albumin, LDH, Blood Sugar)
· Keep the woman warm using appropriate available measures
· Transfuse blood as soon as possible.
· Until blood is available, infuse up to 3.5 L of warmed crystalloids as Lactated Ringer’s (1–2 mL for every 1 mL of blood loss)2
· Stop fluid once blood is ready.
· The best equipment available should be used to achieve rapid warmed infusion of fluids.
· Special blood filters should not be used, as they slow infusions.
· If actively bleeding, transfuse early and do not wait unnecessarily for laboratory results and use clinical assessment as the main determinant
Rationale:
o Stopping crystalloid infusion as soon as blood is ready and avoiding excessive crystalloid use (transfusing no more than 3.5 Liters) is associated with better coagulation and metabolic response as the excessive use of crystalloids leads to dilutional coagulopathy (exacerbates bleeding), decreases O2 carrying capacity (impairs tissue perfusion), metabolic acidosis and hypothermia.
o This occurs secondary to the decrease in hemoglobin concentration, hematocrit value, platelets concentration, fibrinogen level, prolongation of aPTT and PTT, and the reduction of the buffering capacity of plasma bicarbonate in response to excessive crystalloids transfusion.
o Additionally, aggressive fluid resuscitation can rapidly raise arterial pressure before haemostasis is achieved, potentially disrupting early clot formation, dislodging fragile thrombi, and accelerating ongoing haemorrhage.
3.5.2. Blood transfusion
· 3.5.2.1. If actively bleeding, transfuse early, do not wait unnecessarily for laboratory results5,69 The clinical picture should be the main determinant of the need for blood transfusion and time should not be unnecessarily spent awaiting laboratory results. (strong, moderate)5,47,49-51
· 3.5.2.2. Packed RBCs:
o Provide emergency blood (Packed RBCs) with immediate issue of group O, rhesus D (RhD)-negative, with a switch to group-specific blood as soon as feasible. (conditional, low)49-51
o Single Hb/hematocrit estimations may be misleading and can lead to delays in initiating red cell transfusion, serial measurements may be helpful to monitor ongoing progress. (conditional, low)5,47
Rational: Packed RBCs- (to replace red cell loss) should be transfused as soon as available, for restoration of both blood volume and oxygen-carrying capacity.
· 3.5.2.2. Transfusion of FFP:
o Administration of FFP should be guided by coagulation profile test results and whether bleeding is continuing. (GPS)
o If coagulation profile results are not available and bleeding is continuing, after 4 units of red blood cells, 4 units of FFP should be infused until test results are known. (GPS)
o If coagulation profile results are not available, early FFP should be considered for conditions with a suspected coagulopathy, such as placental abruption or amniotic fluid embolism, or where detection of PPH has been delayed. (GPS)
o If prothrombin time/activated partial thromboplastin time is more than 1.5 times normal and hemorrhage is ongoing, more units of FFP are likely to be needed to correct coagulopathy. (GPS)
· 3.5.2.3. Clinicians should be aware that these blood components must be ordered as soon as a need for them is anticipated, as there will always be a short delay in supply because of the need for thawing. (GPS)
3.5.3. Cryoprecipitate For Fibrinogen Replacement
· A plasma fibrinogen level of greater than 2 g/l should be maintained, if test is available, during ongoing PPH. (GPS)
· Cryoprecipitate should be used for fibrinogen replacement.
· Physician should be aware that fibrinogen below 3 g/l and especially below 2 g/l is associated with progression of bleeding, increased RBC and blood component requirements, and the need for invasive procedures. (GPS)
3.5.4. Transfusion of platelets
· During PPH, platelets should be transfused when the platelet count is less than 75 × 109/l (75000 / mm3) based on laboratory monitoring. (GPS)
· The platelets should ideally be group compatible. RhD-negative women should receive RhD-negative platelets. (strong, low)5, 53
3.5.5. Use Continued Resuscitation with lab-guided replacement to maintain the following lab parameters: (conditional, high)5,54,55
· Hb greater than 8 gm%
· Platelet count greater than 50 × 109 /l (50000/mm3)
· Prothrombin time (PT) less than 1.5 times normal
· Activated partial thromboplastin time (APTT) less than 1.5 times normal
· Fibrinogen level greater than 2 g/l.
3.5.5. Tranexamic acid (TXA)
- Tranexamic acid can be used in all patients as an adjunct to uterotonics in the setting of postpartum hemorrhage regardless of whether the bleeding is due to genital tract trauma or other causes. (Strong, high)5-10
- Give TXA as early as possible, ideally within 3 hours of birth, in a fixed dose of 1 g in 10 mL (100 mg/mL) IV at 1 mL per minute (i.e., over 10 minutes). (Strong, high) 5-10
- A second dose of 1 g IV may be administered if bleeding continues after 30 minutes of the first dose or for bleeding that stops but restarts within 24 hours of completing the first dose. (Strong, high) 5-10
4. MONITORING AND INVESTIGATIONS IN MAJOR PPH
- The following should be considered for monitoring and guide investigations in major PPH (GPS)
4.1 Immediate venipuncture (20 ml) for:
· Cross-matching (4 units of Packed RBCs minimum)
· FBC
· Coagulation screen (PT, INR, aPTT including fibrinogen, if available)
· Chemistry profile (Serum Creatinine, SGOT, SGPT, Albumin, LDH, Blood Sugar)
· Repeat as necessary
4.2 Continuous pulse, blood pressure and respiratory rate recording (preferably using oximeter, electrocardiogram and automated blood pressure recording when available) and Monitor temperature every 15 minutes.
4.3. Foley’s catheter to monitor urine output
4.4. Documentation of fluid balance, blood, blood products and procedures.
4.5. Consider transfer to intensive therapy unit once the bleeding is controlled or monitoring at high dependency unit on delivery suite, if appropriate.
4.6. Consider objective scoring system for monitoring as the MEOWS Score (see Appendix II)
5. ROLE OF THE ANESTHETIST IN THE MANAGEMENT OF PPH
- The management of PPH requires a multidisciplinary approach: Allow the anesthetist to play a crucial role in maintaining hemodynamic stability and, in determining and administering the most appropriate method of anesthesia. (Strong, low)5
6. PAIN MANAGEMENT
- Consider pain relief requirements during initial resuscitation and all subsequent treatments. (GPS)
7. PHARMACOLOGICAL AND MECHANICAL STRATEGIES IN MANAGEMENT OF PPH IN PPH
7.1. Clinicians should be prepared to use a combination of pharmacological, mechanical and surgical methods to arrest PPH. These methods should be directed towards the causative factor. (Strong, low)5,10
7.2. When uterine atony is perceived to be a cause of the bleeding, then a sequence of pharmacological and mechanical measures (in the form of uterine massage and urinary bladder catheterization) should be instituted until the bleeding stops. (Strong, low)10,56
7.3 Stopping the bleeding should not be delayed and should start as early as possible and goes hand in hand with the rest of management above (Communication and multidisciplinary approach, Resuscitation, Investigation, and Monitoring) (GPS)
7.4. The most common cause of primary PPH is uterine atony. The initial management of PPH should, therefore, involve measures to stimulate myometrial contractions. Thus, mechanical and pharmacological measures should be instituted/administered as needed. (GPS)
7.5. Palpate the uterine fundus and rub it to stimulate contractions and expel its contents (‘rubbing up the fundus’) (GPS)
7.6. Ensure that the bladder is empty (Foley catheter & leave in place) (GPS)
7.7. First line pharmacological therapy for uterine atony (Oxytocin): (GPS)
· Give oxytocin 5 IU IV over 1-2 minutes (even if had the prophylactic dose before). The dose may be repeated, only once, after five minutes but consider the maximum dose for IV oxytocin which is 10 IU IV.
· Rapid IV bolus administration may cause hypotension, tachycardia, arrhythmia and myocardial ischemia.
· Continue with oxytocin IV infusion 40 IU in 500 mL of either 0.9% sodium chloride (saline) or compound sodium lactate (Ringer’s lactate) at a rate of 5-10 IU/hour via:
o Using ordinary IV line about adjust it to deliver 20-40 drop/minute
o Infusion pump, if available, equals to 62.5–125 mL per hour
o Minimum infusion duration is 2 - 4 hours; use clinical judgement.
· Oxytocin infusion may be a safer alternative to a bolus dose of oxytocin in some women (e.g. major cardiovascular disorders)
· If the patient had induction of labor (IOL) with oxytocin, you may use the same infusion but at increased rate.
· If IV access unavailable or delayed, oxytocin 10 IU IM can be administered
· If carbetocin has already been given, consider non-oxytocin uterotonic instead
7.8. Supplemental therapy if no adequate response, give the following: (GPS)
7.8.1. Ergometrine maleate:
o 0.25 - 0.5 mg IV over 1-2 minutes (dilute 250 micrograms in 5 mL 0.9% sodium chloride) that may be repeated every 5 minutes to a maximum of 1 mg
o It is contraindicated with retained placenta, severe hypertension, pre-eclampsia, eclampsia severe/persistent sepsis, renal, hepatic, vascular, or cardiac disease
7.8.2. Misoprostol:
- Consider misoprostol if alternative uterotonics unavailable or contraindicated (e.g. asthma, hypertension) or if bleeding not effectively controlled with oxytocin.
- Give 800 mcg sublingual (rapid onset of action with side effects more likely)
- Or 1000 mcg per rectum (slow absorption but prolonged effect)
- Consider clinical circumstances when determining optimal route
- Side effects: Hyperthermia is a common side effect (>38 C is common) and malignant hyperthermia > 40 C has been reported in 1 - 14% of cases
8. SURGICAL TREATMENTS OF PPH
- If pharmacological measures fail to control the hemorrhage, surgical interventions should be initiated sooner rather than later and the most experienced, anesthetist and obstetrician should be called to be involved in the management. (Strong, low)7,9
8.1. Examination of the genital tract under anesthesia:
- Transfer the patient to the operative theatre flat with high-flow oxygen and perform an examination under anesthesia for the exclusion of the presence of remnants (to be evacuated if present) and dealing with any birth tract injuries if found. (Strong, low)7,9
- Bimanual Compression: The use of bimanual uterine compression is recommended as a temporary measure until appropriate care is available for the treatment of PPH due to uterine atony after vaginal delivery (Strong, low)10
8.2. Intrauterine balloon tamponade:
- The following is recommended when considering the use of intrauterine balloon tamponade: (Strong, low)5-10
· If bimanual compression has been effective consider the use of intrauterine balloon tamponade (e.g. Bakri Balloon) as balloon is an appropriate first-line ‘surgical’ intervention for most women where uterine atony is the only or main cause of hemorrhage after the success of bimanual uterine compression.
· After insertion, assess blood loss:
o If bleeding continues, balloon tamponade may be ineffective—review aetiology of PPH, check balloon placement and consider other surgical interventions.
o If bleeding ceases on insertion, monitor fundal height, uterine cramping and signs of increased blood loss regularly
· Balloon is left for 6 hours if bleeding stopped and removed in presence of senior staff lest the return of bleeding.
- If bimanual compression is ineffective or balloon tamponade fail:
· Consider exploratory laparotomy when less invasive approaches fail to control bleeding. In the setting of a vaginal delivery, it is common to use a midline vertical abdominal incision for laparotomy, to optimize exposure and reduce risk of surgical bleeding. In the setting of cesarean birth, the existing surgical incision may be used. (Strong, low)6
· Conservative surgical interventions (compression sutures and/or arterial ligation) may be attempted as a second line, depending on clinical circumstances and available expertise. (strong, low)5-10
· Timing is critical. Surgical interventions should be initiated sooner rather than later. (Strong, low) 5-10
· Weigh benefits of conservative versus aggressive management (hysterectomy) (Strong, low) 5-10
8.3. Hemostatic uterine suture (e.g. B-Lynch suture)
- Judiciously apply aortic compression (below the level of the renal arteries) as a temporary measure. (GPS)
- Consider B-Lynch compression or other compression suture. (GPS)
- It is preferable that a laminated diagram of the brace suture technique, as B-lynch suture, be kept in theatre. (GPS)
8.4. Arterial ligation:
- Stepwise arterial ligation for uterine devascularization may be considered depending on clinical circumstances and available expertise (GPS)
· Bilateral uterine artery ligation
· Bilateral utero-ovarian artery ligation
· If expertise available, bilateral internal iliac artery ligation
8.5. Hysterectomy:
8.8.1. Resort to hysterectomy early (sooner rather than later) especially if: (Strong, low)5-10
o Life is threatened
o Hemodynamic unstable patient,
o Bleeding continues after use of conservative treatment options
o blood transfusion is limited or not an option,
o In cases of placenta accreta or uterine rupture.
8.8.2. Subtotal (Supra-Vaginal) hysterectomy is quicker and safer in Major PPH and in packing of the abdomen, use hot packs (GPS)
8.8.3. Ideally and when feasible, a second experienced clinician should be involved in the decision for hysterectomy (GPS)
8.8.4. In rare situations in spite of compression, arterial ligation and hysterectomy, bleeding from undefined origin might persist in the pelvis (undefined diffuse venous bleeding or bleeding due to DIC). Tight pelvic packing with radio-opaque packs, done by a senior obstetrician, may be a last resort to be followed by temporarily closing the abdomen, stabilization of the patient condition by multidisciplinary team, then reopening the abdomen again to remove the packs and reassess hemostasis after 24-48 hours. (Conditional, low)9
9. RISK MANAGEMENT
9.1. Training and preparation:
- Every maternity unit should have a multidisciplinary protocol for the management of PPH. (GPS)
- All staff involved in maternity care should receive training in the management of obstetric emergencies, including the management of PPH. (strong, very-low)5
- Training for PPH should be multiprofessional and include team rehearsals. (strong, very-low)5
- All cases of PPH involving a blood loss of greater than 1500 ml should be the subject of a formal clinical incident review. (GPS)
9.2. Documentation:
- Accurate documentation of a delivery with PPH is essential. (GPS)
9.3. Consent:
- If treatment is likely to affect woman’s fertility, prioritize gaining informed consent. (Strong, moderate)5-10
10. POSTNATAL CARE
- Offer the woman and family debriefing/clinical disclosure by senior team member(s), preferably by clinicians who were at the event. (GPS)
- Upon discharge Educate about signs, symptoms and self-referral regarding persistent or increasing bleeding, infection and risk of secondary PPH, postnatal depression, venous thromboembolism (VTE) (GPS)
- Oral iron supplementation, either alone or in combination with folic acid, may be provided to postpartum women for 6–12 weeks after delivery for reducing the risk of anaemia. (conditional, low)10
- Offer additional opportunities for discussion/debrief six weeks postpartum. (GPS)
- Acknowledgements
We would like to acknowledge Egyptian Health Council and Obstetrics & Gynecology Guidelines Development Committee.
Chair of the GDG:
Abdelhamid Mohamed Attia, MD, FRCOG, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Cairo University
Rapporteur of the GDG:
Alaa Eldin Hamed ElFeky, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Ain Shams University
Members of the GDG:
- Aboubakr Mohamed ElNashar, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Banha University
- Ahmed Ezz El-din Mahran, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Minia University
- Ahmed Fawzy Galal, MD, FRCOG, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Alexandria University
- Ahmed Sekotory Mahmoud, MD, FRCOG, FACOG, Consultant of Obstetrics and Gynecology, Private sector
- Amr Abdel Aziz Nadim, MD, Dean, Faculty of Medicine, 6 October University
- Amr Ahmed AboAlyazid, MD, EFOG, Senior Consultant of Obstetrics and Gynecology, Police Medical Services.
- Amr Essam Abdel Rahman, MD, Senior Consultant of Obstetrics and Gynecology, Ain-Shams University
- Diaa Monier Ajlan, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Tanta University
- Ihab Hassan Abdelfataah, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Galala University
- Magdy Ibrahim Mostafa, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Cairo University
- Mervat Ali Elsersy, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Alexandria University
- Mohamed Morad, MD, FRCOG, Consultant of Obstetrics and Gynecology, AlGalaa Teaching Hospital, MOHP
- Mohamed Mahmoud Fathalla, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine, Assiut University
- Osama Omar Amer, MD, FRCOG, Major General Dr. of Obstetrics and Gynecology, Armed Forces Hospitals
- Taiseer Maarouf Afifi, MD, Prof. of Obstetrics and Gynecology, Faculty of Medicine for girls, AlAzhar University
- Wafaa Benjamin Basta, FRCOG, Obstetrics and Gynecology, Mataria Teaching Hospital, MOHP
- Abbreviations
- GDG: Guidelines Development Group
- GPS: Good Practice Statement.
- GRADE: Grading of Recommendations Assessment, Development and Evaluation
- PPH: In this document PPH refers to Primary Postpartum hemorrhage
- EUA: Examination under anesthesia
- PAS: Placenta Accreta Spectrum
- UBT: Uterine Balloon Tamponade
- UAE: Uterine artery embolization
- Glossary
- Primary postpartum haemorrhage is identified when excessive bleeding in the first 24 hours post birth occurs.
- Active Management of Third Stage of Labor (AMTSL): A set of interventions (uterotonic use, controlled cord traction, uterine massage) to prevent PPH.
- Uterotonic Agents: Medications (e.g., oxytocin, misoprostol, ergometrine, carbetocin) that stimulate uterine contractions.
- Tranexamic Acid (TXA): An antifibrinolytic drug used to reduce bleeding in PPH.
- Uterine Atony: Failure of the uterus to contract adequately after birth, the leading cause of PPH.
- Placenta Accreta Spectrum (PAS): Abnormal adherence of the placenta to the uterine wall, increasing PPH risk.
- Balloon Tamponade: Insertion of a balloon device into the uterus to apply pressure and control bleeding.
- B-Lynch Suture: A compression suture technique applied to the uterus to control severe bleeding.
- Hemodynamic Instability: Clinical state of compromised circulation (hypotension, tachycardia) due to blood loss.
- Shock Index (SI): Ratio of heart rate to systolic blood pressure; useful for early detection of shock.
- FIGO (International Federation of Gynecology and Obstetrics): Professional body setting international guidelines on PPH management.
- WHO (World Health Organization): Provides global recommendations for PPH prevention and treatment.
- Introduction
Postpartum hemorrhage (PPH) is defined as blood loss exceeding 500 ml following vaginal birth and severe PPH is characterized as a loss of more than 1000 ml, while in cases of caesarean birth PPH is defined as blood loss exceeding 1000 ml.1 The incidence of PPH differs widely according to the definition and criteria used, methods of measuring postpartum blood loss, and the population being studied, with the highest incidence reported in low- and lower-middle-income countries.2
Each year, worldwide, about 14 million people of women have postpartum hemorrhage, with about 70 000 of them ending in death.1 In the immediate postpartum period, complications of postpartum hemorrhage include hypovolemic shock from massive blood loss, disseminated intravascular coagulopathy, acute renal failure, hepatic failure, and complications of blood transfusion, including transfusion-related acute lung injury, acute respiratory distress syndrome, transfusion-associated circulatory over- load, and death. Late complications e.g. pituitary necrosis and pan- hypopituitarism as well as infertility may also occur.3
In Egypt, according to the last demographic and health survey, postpartum hemorrhage contributed to 19% of causes of maternal mortality.4
Late recognition and inadequate response are major contributors to the morbidity and mortality in PPH. This is why guidelines for management of postpartum hemorrhage emphasize early recognition and prompt response.
- Scope and Purpose
The objectives of this guideline are:
- To provide guidance for the proper prevention and management of primary postpartum hemorrhage.
- To optimize outcomes for patients who are at risk of or developed primary postpartum hemorrhage.
- Target Audience
- This guideline targets; healthcare professionals working as Obstetricians & Gynecologists, Anesthetists, ICU physicians, Nurses, policy makers, hospital managers, and other stakeholders to apply the best practice and afford the most appropriate tools for women or developed primary postpartum hemorrhage.
- Methodology
A comprehensive search for guidelines was done to identify the most relevant ones to consider for adaptation. The inclusion/exclusion criteria that were followed in the search and retrieval of guidelines to be adapted are:
We select guidelines only if they are:
- Evidence-based guidelines
- National and/or international guidelines
- Guidelines published from 2015 to 2025
- Peer reviewed publications
- Guidelines written in English language
We Exclude guidelines that are:
- Written by a single author not on behalf of an organization as guideline to be valid and comprehensive ideally requires multidisciplinary input.
- Published without references as the panel needs to know whether a thorough literature review was conducted and whether the current evidence was used in the preparation of the recommendations.
The following characteristics of the retrieved guidelines were summarized in a table:
- Developing organisation/authors
- Date of publication, posting, and release
- Country/language of publication
- Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained).
Guidelines used in the adaptation process: 5-10
1. Royal College of Obstetricians and Gynaecologists. Prevention and Management of Postpartum Haemorrhage. Green-top Guideline No. 52. London: RCOG; 2016. https://www.rcog.org.uk/guidance/browse-all-guidance/green-top-guidelines/prevention-and-management-of-postpartum-haemorrhage-green-top-guideline-no-52/
2. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 183: Postpartum Hemorrhage. Obstet Gynecol. 2017;130(4):e168-e186.
3. Queensland Clinical Guidelines. Primary postpartum haemorrhage (PPH) [Internet]. Brisbane (AU): Queensland Health; 2024 Aug [amended 2024 Nov; cited 2025 Oct 15]. 46 p. Report No.: MN24.1-V12-R29. Available from: https://www.health.qld.gov.au/qcg
4. Mundle S, Bracken H, Khedikar V, Easterling T, Winikoff B, Zorkun N, et al. SOGC Clinical Practice Guideline: Postpartum Hemorrhage and Hemorrhagic Shock. J Obstet Gynaecol Can. 2022 Dec;44(12):1273-1307.
5. Escobar MF, Nassar AH, Theron G, Barnea ER, Nicholson W, Ramasauskaite D, et al. FIGO recommendations on the management of postpartum hemorrhage 2022. Int J Gynaecol Obstet. 2022 Mar;157 Suppl 1(Suppl 1):3-50.
6. WHO recommendations Consolidated guidelines for the prevention, diagnosis and treatment of postpartum haemorrhage. Geneva: World Health Organization; 2025. Licence: CC BY-NC-SA 3.0 IGO.
Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
- GRADE working group: http://www.gradeworkinggroup.org
- GRADE online training modules: http://cebgrade.mcmaster.ca/
- GRADE profile software: http://ims.cochrane.org/revman/gradepro
Table 1: Quality and Significance of the four levels of evidence in GRADE:
|
Quality |
Definition |
Implications |
|
High |
The guideline development group is very confident that the true effect lies close to that of the estimate of the effect |
Further research is very unlikely to change confidence in the estimate of effect |
|
Moderate |
The guideline development group is moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different |
Further research is likely to have an important impact on confidence in the estimate of effect and may change the estimate |
|
Low |
Confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the true effect |
Further research is very likely to have an important impact on confidence in the estimate of effect and is unlikely to change the estimate |
|
Very low |
The group has very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of the effect |
Any estimate of effect is very uncertain |
Table 2; Factors that determine How to upgrade or downgrade the quality of evidence
|
Downgrade in presence of |
Upgrade in presence of |
|
Study limitations -1 Serious limitations -2 Very serious limitations |
Dose-response gradient + 1 Evidence of a dose-response gradient |
|
Consistency -1 Important inconsistency |
Direction of plausible bias + 1 All plausible confounders would have reduced the effect |
|
Directness -1 Some uncertainty -2 Major uncertainty |
Magnitude of the effect + 1 Strong, no plausible confounders, consistent and direct evidence + 2 Very strong, no major threats to validity and direct evidence |
|
Precision -1 Imprecise data |
|
|
Reporting bias -1 High probability of reporting bias |
|
The strength of recommendations
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations: The GDG found that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted.
Conditional recommendations: This means that the GDG found that there is:
- Greater uncertainty about the strength of evidence, or
- The recommendation may account for a greater variety in patient values and preferences, or
- The resource use makes the intervention suitable for some, but not for other locations.
Conditional recommendations are still the best available evidence to date and it can be adopted if it meets the conditions mentioned with it.
Good Practice Statement: Statements based on opinion of respected authorities, e.g. the RCOG, ACOG, and the guidelines development group.
- Implementation considerations
Several barriers may hinder the effective implementation and scale-up of the recommendations in this guideline. These factors may be related to the behaviours of patients (or families), the behavior of healthcare professionals, the organization of care, health service delivery or financial arrangements.
Obstacles to effective implementation include:
- Patient engagement
- Collaboration; person centered, team-based collaboration between clinician, dietitian, pharmacist and others involved in care delivery
- Behavior changes: information, guidance and support delivered easily and consistently can help assess sustained behavioral changes.
- Research needs
Epidemiology of PPH in Egypt
- Accurate national and regional incidence and mortality data.
- Risk factor profiles among Egyptian women, including the role of anaemia, high parity, and caesarean section rates.
Early recognition, Health System Capacity & Implementation
- Validation study for early obstetric warning score (EOWS) trigger chart, considering feasibility as an objective maternity evaluation and monitoring tool compared to use of vital data subjectively.
- Study to evaluate MEOWS optimum trigger values and optimum response to abnormal score.
- Effectiveness of active management of the third stage of labour (AMTSL) across different levels of healthcare (primary, secondary, tertiary).
- Barriers to timely administration of uterotonics, tranexamic acid, and blood products in Egyptian hospitals.
- Evaluation of referral pathways and emergency obstetric transport systems in rural vs. urban settings.
Pharmacological & Non-Pharmacological Interventions
- Comparative effectiveness of oxytocin, misoprostol, and carbetocin in Egyptian populations, considering cost-effectiveness and storage challenges.
- Feasibility and outcomes of balloon tamponade, B-Lynch sutures, and interventional radiology in resource-variable settings.
Blood Management and Transfusion Practices
- Outcomes of implementing Massive Transfusion Protocols (MTP) in Egyptian obstetric units.
- Strategies to optimize blood availability, cross-matching practices, and patient blood management in maternity care.
Maternal Health Determinants
- Impact of nutritional anemia, chronic diseases, and social determinants (access, literacy, socioeconomic status) on PPH outcomes.
- Cultural and behavioral factors influencing care-seeking and acceptance of interventions.
Training and Simulation-Based Research
- Effectiveness of structured training programs, PPH emergency drills, and multidisciplinary team simulations in reducing delays and errors in real practice.
Cost-Effectiveness and Health Economics
- Economic evaluation of different uterotonics, surgical techniques, and emergency interventions in Egyptian hospitals.
- Budget impact of scaling up WHO-recommended interventions at national level.
Clinical Quality Standards for Monitoring
|
- Delivery: Investigations & Cross Match |
|
|
QS.1 |
All women with one or more risk factors for PPH should have an IV line with 16 G canula, and blood sample sent for CBC, blood group, and cross match of 2 units of blood. |
|
QM.1 |
Numerator: number of women with risk factors admitted to labor who have an IV access (16G) and a sample of blood sent for CBC, blood group, and cross match of 2 units of blood. Denominator: total number of women with risk factors admitted to labor |
|
- Delivery: Prophylaxis at Cesarean Section |
|
|
QS.2 |
All women with risk factors for PPH should be given IV Tranexamic acid at Cesarean Section (CS). |
|
QM.2 |
Numerator: number of women with risk factors for PPH given IV Tranexamic acid at CS Denominator: number of women with risk factors for PPH who had CS |
|
- Delivery: Management of the 3rd Stage of Labor |
|
|
QS.3 |
All women after delivery of the baby are given oxytocin, or misoprostol if oxytocin is not available, within one minute maximally of delivery of the baby. |
|
QM.3 |
Numerator: number of patients who received oxytocin (or misoprostol) within 1 minute of delivery of the baby. Denominator: total number of deliveries. |
|
- Management of the Fourth Stage of Labor |
|
|
QS.4 |
Vital signs and bleeding should be monitored and recorded every 15 mins in all women for the first two hours after birth. |
|
QM.4 |
Numerator: number of women monitored for vital signs and bleeding in the first 2 hours after birth and data recorded in their file. Denominator: total number of women who gave birth. |
|
- PPH Management: Senior Staff Notification & Call |
|
|
QS.5 |
When a woman presents with minor PPH, the required senior staff are notified and called as per protocol. |
|
QM.5 |
Numerator: number of senior staff notified and called with minor PPH. Denominator: total number of women with minor PPH. |
|
- PPH Mortality |
|
|
QS.6 |
The majority of PPH mortalities are avoidable with proper preventive strategies and timely management by a multidisciplinary senior staff when needed. |
|
QM.6 |
Numerator: number of women who died of PPH. Denominator: total number of women who developed PPH. |
- Updating of the guidelines
This guideline will be updated whenever there is new evidence.
- References
|
Say L, Chou D, Gemmill A, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health 2014;2(6):e323-e333. |
|
Borovac-Pinheiro A, Pacagnella RC, Cecatti JG, et al. Postpartum hemorrhage: new insights for definition and diagnosis. Am J Obstet Gynecol 2018;219:162-8. |
|
Committee on Practice Bulletins-Obstetrics. Practice bulletin no. 183: postpartum hemorrhage. Obstet Gynecol 2017;130(4):e168-e186 |
|
Elzanaty and Way. Egypt Demographic and Health Survey. 2014. |
|
Royal College of Obstetricians and Gynaecologists. Prevention and Management of Postpartum Haemorrhage. Green-top Guideline No. 52. London: RCOG; 2016. https://www.rcog.org.uk/guidance/browse-all-guidance/green-top-guidelines/prevention-and-management-of-postpartum-haemorrhage-green-top-guideline-no-52/ |
|
American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 183: Postpartum Hemorrhage. Obstet Gynecol. 2017;130(4):e168-e186. |
|
Queensland Clinical Guidelines. Primary postpartum haemorrhage (PPH): Queensland Health; 2024. Available from: https://www.health.qld.gov.au/qcg |
|
Mundle S, Bracken H, Khedikar V, Easterling T, Winikoff B, Zorkun N, et al. SOGC Clinical Practice Guideline: Postpartum Hemorrhage and Hemorrhagic Shock. J Obstet Gynaecol Can. 2022 Dec;44(12):1273-1307. |
|
Escobar MF, Nassar AH, Theron G, Barnea ER, Nicholson W, Ramasauskaite D, et al. FIGO recommendations on the management of postpartum hemorrhage 2022. Int J Gynaecol Obstet. 2022 Mar;157 Suppl 1(Suppl 1):3-50. |
|
WHO recommendations Consolidated guidelines for the prevention, diagnosis and treatment of postpartum haemorrhage. Geneva: World Health Organization; 2025. Licence: CC BY-NC-SA 3.0 IGO |
|
Muñoz M, Stensballe J, Ducloy-Bouthors AS, Bonnet MP, De Robertis E, Fornet I, et al. Patient blood management in obstetrics: prevention and treatment of postpartum Hemorrhage. A NATA consensus statement. Blood Transfus 2019;17(2):112-36. doi:10.2450/2019.0245-18 |
|
National Blood Authority (NBA). Patient blood management guidelines: module 5 obstetrics and maternity. Canberra Australia: NBA; 2015 [cited 2024 February 26]. Available from: https://blood.gov.au |
|
The Royal Australian and New Zealand College of Obstetricians and Gynaecologists. Placenta accreta spectrum (PAS) C-Obs 20. [Internet]. 2023. [cited 2024 March 28]. Available from: https://ranzcog.edu.au. 5. Australian College of Midwives. Aims and scope [Internet]. 2024 [cited 2024 March 03]. Available from: https://www.womenandbirth.org |
|
Stewart MJ, Richmond D, Mooney S, Esler S, Churilov L, Israelsohn N, et al. Diagnostic utility of MRI features of placental adhesion disorder for abnormal placentation and massive postpartum hemorrhage. AJR Am J Roentgenol 2021;217(2):378-88. doi:10.2214/AJR.19.22661. |
|
Queensland Clinical Guidelines. Induction of labour. Guideline No. MN22.22-V9-R27. [Internet]. Queensland Health. 2022. [Cited 2025 March 19]. Available from: https://www.health.qld.gov.au/qcg |
|
Chaudhary P, Priyadarshi M, Singh P, Chaurasia S, Chaturvedi J, Basu S. Effects of delayed cord clamping at different time intervals in late preterm and term neonates: a randomized controlled trial. European Journal of Pediatrics 2023;182(8):3701-11. doi:10.1007/s00431-023-05053-6. |
|
Hofmeyr GJ, Mshweshwe NT, Gulmezoglu AM. Controlled cord traction for the third stage of labour. Cochrane Database of Systematic Reviews. [Internet]. 2015, [Cited 2025 February 15]. Issue 1. Art No.: CD008020. DOI:10.1002/14651858.CD008020.pub2. |
|
The Royal Australian and New Zealand College of Obstetricians and Gynaecologists. Management of postpartum haemorrhage. [Internet]. 2021. [cited 2024 January 31]. Available from: https://ranzcog.edu.au |
|
Wu Y, Wang H, Wu QY, Liang XL, Wang J. A meta-analysis of the effects of intramuscular and intravenous injection of oxytocin on the third stage of labor. Arch Gynecol Obstet 2020;301(3):643-53. doi:10.1007/s00404-020-05467-9 |
|
National Institute for Health and Clinical Excellence (NICE). Intrapartum care. Clinical Guideline 235. 2023. [Internet]. [cited 2024 January 29]. Available from: https://www.nice.org.uk |
|
Oladapo OT, Okusanya BO, Abalos E, Gallos ID, Papadopoulou A. Intravenous versus intramuscular prophylactic oxytocin for the third stage of labour. Cochrane Database of Systematic Reviews. [Internet]. 2020, [cited 2024 February 12]. Issue 11. Art No.: CD009332. DOI:10.1002/14651858.CD009332 |
|
Zhou YH, Xie Y, Luo YZ, Liu XW, Zhou J, Liu Q. Intramuscular versus intravenous oxytocin for the third stage of labor after vaginal delivery to prevent postpartum hemorrhage: a meta-analysis of randomized controlled trials. Eur J Obstet Gynecol Reprod Biol 2020;250:265-71. doi:10.1016/j.ejogrb.2020.04.007 |
|
Tyagi A, Deep S, Salhotra R, Malhotra R, Singla A. Minimum effective dose of oxytocin bolus during the caesarean section for patients at high vs low risk of uterine atony: A non-randomized, dual-arm, dose-finding prospective trial. Indian Journal of Anaesthesia 2023;67(8):690-6. doi:10.4103/ija.ija_760_22 |
|
Baliuliene V, Vitartaite M, Rimaitis K. Prophylactic dose of oxytocin for uterine atony during caesarean delivery: A systematic review. International Journal of Environmental Research and Public Health 2021;18(9):5029. doi:10.3390/ijerph18095029 |
|
Lee S, Cauldwell M, Wendler R. Cardiac effects of drugs used for induction of labour and prevention and treatment of postpartum haemorrhage. International Journal of Cardiology Congenital Heart Disease 2021;5:100208. doi:10.1016/j.ijcchd.2021.100208 |
|
World Health Organization. WHO recommendations: uterotonics for the prevention of postpartum haemorrhage. 2018. [cited 2024 January 29]. Available from: https://who.int |
|
Australian Medicines Handbook. Carbetocin. [Internet]. Adelaide: Australian Medicines Handbook Pty Ltd; January 2024 [cited 2024 February 21] |
|
Robinson d, Basso M, Chan C, Duckitt K, Lett R. Guideline No. 431: postpartum hemorrhage and hemorrhagic shock. J Obstet Gynaecol Can 2022;44(12):1293-310.e1. doi:10.1016/j.jogc.2022.10.002 |
|
National Institute for Health and Care Excellence (NICE). Evidence reviews for uterotonics for the prevention of postpartum haemorrhage: Intrapartum care: Evidence review [M] [Internet]. 2023 [cited 2024 February 15]. Available from: www.nice.org.uk |
|
Australian Medicines Handbook. Misoprostol (obstetrics). [Internet]. Adelaide: Australian Medicines Handbook Pty Ltd; January 2024 |
|
Drew T, Carvalho JCA. Pharmacologic prevention and treatment of postpartum hemorrhage. Current Anesthesiology Reports 2021;11(1):37-47. doi:10.1007/s40140-021-00444-7. |
|
World Health Organization. WHO recommendations for the prevention and treatment of postpartum haemorrhage. 2012. |
|
International Federation of Gynaecology and Obstetrics. Misoprostol - only: recommended regimens 2017 [Internet]. 2017 |
|
Collaborators WT. Effect of early tranexamic acid administration on mortality, hysterectomy, and other morbidities in women with post-partum haemorrhage (WOMAN): an international, randomised, double-blind, placebo-controlled trial. Lancet 2017;389(10084):2105-16. doi:10.1016/S0140-6736(17)30638-4. |
|
National Blood Authority (NBA). Patient blood management guideline for adults with critical bleeding [Internet]. 2023 [cited 2024 January 29]. Available from: https://www.blood.gov.au/clinical-guidance/patient-blood-management |
|
WHO recommendation on tranexamic acid for the treatment of postpartum Hemorrhage. Geneva: World Health Organization; 2017 (https://iris.who.int/handle/10665/259374). |
|
Hofmeyr GJ, Abdel-Aleem H, Abdel-Aleem MA. Uterine massage for preventing postpartum haemorrhage. Cochrane Database Syst Rev 2013;(7):CD006431 |
|
Queensland Clinical Guidelines. Normal birth. Guideline No. MN22.25-V5-R27. [Internet]. Queensland Health. 2022. [cited 2024 March 13]. Available from: https://www.health.qld.gov.au/qcg. |
|
Abedi P, Jahanfar S, Namvar F, Lee J. Breastfeeding or nipple stimulation for reducing postpartum haemorrhage in the third stage of labour. Cochrane Database of Systematic Reviews. [Internet]. 2016, [cited 2024 February 7]. Issue 1. Art No.: CD010845. DOI:10.1002/14651858.CD010845.pub2. |
|
Rath WH. Postpartum hemorrhage – update on problems of definitions and diagnosis. Acta Obstet Gynecol Scand 2011;90:421–8 |
|
Nathan HL, El Ayadi A, Hezelgrave NL, Seed P, Butrick E, Miller S, et al. Shock index: an effective predictor of outcome in postpartum haemorrhage? BJOG 2015;122:268–75. |
|
Sentilhes L, Merlot B, Madar H, Sztark F, Brun S, Deneux-Tharaux C. Postpartum Hemorrhage: prevention and treatment. Expert Rev Hematol 2016;9(11):1043-61. doi:10.1080/17474086.2016.1245135. |
|
Ayala M, Nookala V, Fogel J, Fatehi M. Visual estimation of blood loss versus quantitative blood loss for maternal outcomes related to obstetrical hemorrhage. Baylor University Medical Center Proceedings 2023;36(3):341-5. doi:10.1080/08998280.2023.2187248 |
|
Wiklund I, Fernández SA, Jonsson M. Midwives' ability during third stage of childbirth to estimate postpartum Hemorrhage. Eur J Obstet Gynecol Reprod Biol X 2022;15:100158. doi:10.1016/j.eurox.2022.100158. |
|
Ruiz MT, Azevedo NF, Resende CV, Rodrigues WF, Meneguci J, Contim D, et al. Quantification of blood loss for the diagnosis of postpartum hemorrhage: a systematic review and meta-analysis. Rev Bras Enferm 2023;76(6):e20230070. doi:10.1590/0034-7167-2023-0070. |
|
American College of Obstetricians and Gynecologists (ACOG). Quantitative blood loss in obstetric hemorrhage: committee opinion. No. 794. Obstetrics and Gynecology 2019;134(6):e150-e6. doi:10.1097/aog.0000000000003564 |
|
American College of Surgeons Committee on Trauma. Advanced Trauma Life Support (ATLS) Student Course Manual. 10th ed. Chicago: American College of Surgeons; 2018 |
|
Bonnar, J. Bailliere's Clinical Obstetrics and Gynaecology Vol. 14, No. 1, pp 1–18, 2000 |
|
Hunt BJ, Allard S, Keeling D, Norfolk D, Stanworth SJ, Pendry K; British Committee for Standards in Haematology. A practical guideline for the haematological management of major haemorrhage. Br J Haematol 2015;170:788–803 |
|
Spahn DR, Bouillon B, Cerny V, Duranteau J, Filipescu D, Hunt BJ, et al. The European guideline on management of major bleeding and coagulopathy following trauma: fifth edition. Crit Care. 2019;23(1):98. |
|
National Blood Authority (Australia). Patient Blood Management Guidelines: Module 1 - Critical Bleeding/Massive Transfusion. [Internet]Canberra: National Blood Authority; 2011. Available from: https://www.blood.gov.au/patient-blood-management-guideline-adults-critical-bleeding |
|
Muñoz M, Stensballe J, Ducloy-Bouthors AS, Bonnet MP, De Robertis E, Fornet I, et al. Patient blood management in obstetrics: prevention and treatment of postpartum haemorrhage. A NATA consensus statement. Blood Transfus 2019;17(2):112-36. doi:10.2450/2019.0245-18. |
|
United Kingdom Blood Services; Norfolk D, editor. Handbook of Transfusion Medicine. 5th ed. Norwich: TSO; 2013. |
|
Association of Anaesthetists of Great Britain and Ireland, Thomas D, Wee M, Clyburn P, Walker I, Brohi K, et al. Blood transfusion and the anaesthetist: management of massive haemorrhage. Anaesthesia 2010;65:1153–61. |
|
Hunt BJ, Allard S, Keeling D, Norfolk D, Stanworth SJ, Pendry K; British Committee for Standards in Haematology. A practical guideline for the haematological management of major haemorrhage. Br J Haematol 2015;170:788–803. |
|
WHO recommendations on the assessment of postpartum blood loss and treatment bundles for postpartum haemorrhage. Geneva: World Health Organization; 2023. |
- Appendix
The EMOTIVE FIRST BUNDLE:Top of Form
Early detection, Massage the uterus, Oxytocic drugs, Tranexamic acid, IV fluids,
èExamination & Escalation

Appendix II
Components of the Modified Early Obstetric Warning Score (MEOWS) (Joint Trust Guideline for the use of the Modified Early Obstetric Warning Score (MEOWS) in Detecting the Seriously Ill and Deteriorating Woman; 25/02/2022, Internet download from https://www.nnuh.nhs.uk/publication/download/modified-early-obstetric-warning-score-meows-mid33-ao13-v7/),
|
MEOW Sore |
3 |
2 |
1 |
0 |
1 |
2 |
3 |
|
1.Temp oC |
< 35 |
35-35.9 |
36-37.4 |
37.5 -37.9 |
38-38.9 |
≥ 39 |
|
|
2.Sys BP |
< 70 |
70-79 |
80-89 |
90-139 |
140-149 |
150-159 |
≥ 160 |
|
3.Dias BP |
≤ 49 |
50-89 |
90-99 |
100-109 |
≥ 110 |
||
|
4.Pulse |
<40 |
40-49 |
50-99 |
100-109 |
110-129 |
≥ 130 |
|
|
5.R R |
≤ 10 |
11-19 |
20-24 |
25-29 |
≥ 30 |
||
|
6.AVPU |
Alert |
Respond to Voice |
Respond to Pain |
Unresponsive |
|||
|
7.UOP ml/hr |
< 10 |
< 30 |
> 30 |
||||
|
8. ± O2 Sat* |
≤ 90% |
91-94% |
≥ 95% |
||||
|
*Any woman who triggers a MEOWS of ≥ 4, or 3 in any one parameter should have their oxygen saturations recorded with each full set of observations. |
|||||||
Green Pathway - expedited management, Score 1-3
Amber Pathway - urgent management, Score 4 or 5 or score 3 in a single item
Red Pathway - emergency management, Score ≥ 6
There is no substitute for wise physician judgment
Appendix III:
A flow chart of the different steps for the management of PPH